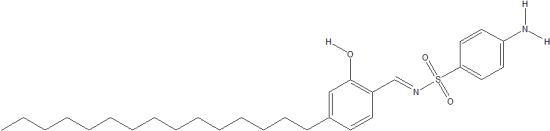
4-Amino-N-(2-hydroxy-4-pentadecylbenzylidene)benzenesulfonamide
Abstract
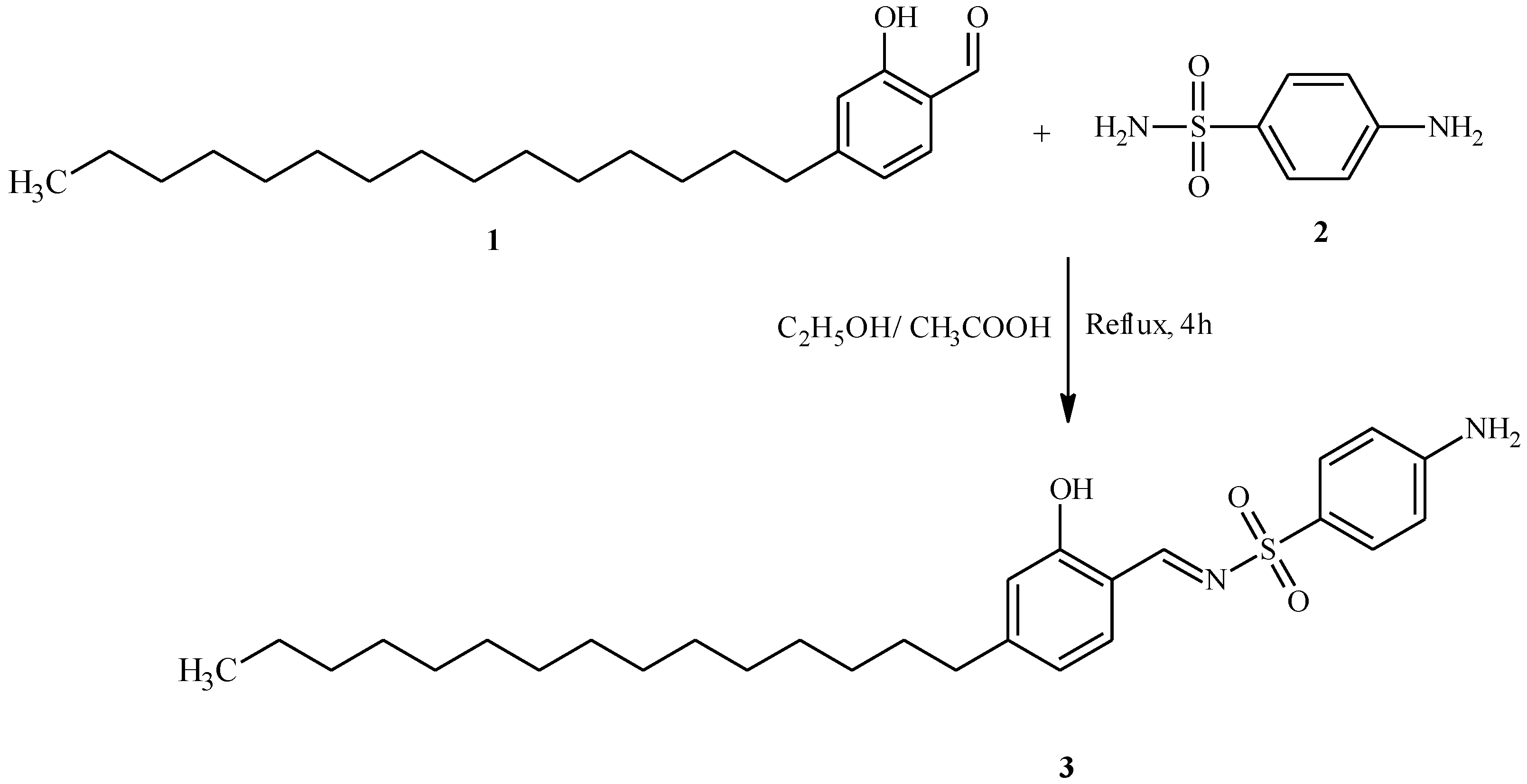
:Experimental
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgments
References
- Hadjoudis, E.; Vittorakis, M.; Moustakali-Mavridis, I. Photochromism and thermochromism of schiff bases in the solid state and in rigid glasses. Tetrahedron 1987, 43, 1345–1360. [Google Scholar] [CrossRef]
- Hadjoudis, E.; Rontoyianni, A.; Ambroziak, K.; Dziembowska, T.; Mavridis, I.M. Photochromism and thermochromism of solid trans-N,N′-bis(salicylidene)-1,2- cyclohexanediamines and trans-N,N′-bis-(2-hydroxynaphylidene)-1,2-cyclohexanediamine. J. Photochem. Photobiol. A Chem. 2004, 162, 521–530. [Google Scholar] [CrossRef]
- Oshima, A.; Momotake, A.; Arai, T. Photochromism, thermochromism, and solvatochromism of naphthalene-based analogues of salicylideneaniline in solution. J. Photochem. Photobiol. A Chem. 2004, 162, 473–479. [Google Scholar] [CrossRef]
- Yeap, G.Y.; Ha, S.T.; Lim, P.L.; Boey, P.L.; Ito, M.M.; Sanehisa, S.; Youhei, Y. Synthesis, physical and mesomorphic properties of Schiff’s base esters containing ortho-, meta and para-substituents in benzylidene-4’-alkanoyloxyanilines. Liq. Cryst. 2006, 33, 205–211. [Google Scholar] [CrossRef]
- Ha, S.T.; Ong, L.K.; Wong, J.P.W.; Yeap, G.Y.; Lin, H.C.; Ong, S.T.; Koh, T.M. Mesogenic Schiff′s base ether with dimethylamino end group. Phase Transit. 2009, 82, 387–397. [Google Scholar] [CrossRef]
- Ha, S.T.; Ong, L.K.; Ong, S.T.; Yeap, G.Y.; Wong, J.P.W.; Koh, T.M.; Lin, H.C. Synthesis and mesomorphic properties of new Schiff base esters with different alkyl chains. Chin. Chem. Lett. 2009, 20, 767–770. [Google Scholar] [CrossRef]
- Ha, S.T.; Ong, L.K.; Yasodha, S.; Yeap, G.Y.; Boey, P.L.; Lin, H.C. New mesogenic Schiff base esters with polar chloro substituent: synthesis, thermotropic properties and X-ray diffraction studies. Am. J. Appl. Sci. 2010, 7, 214–220. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Naganagowda, G.; Petsom, A. 4-Amino-N-(2-hydroxy-4-pentadecylbenzylidene)benzenesulfonamide. Molbank 2011, 2011, M739. https://doi.org/10.3390/M739
Naganagowda G, Petsom A. 4-Amino-N-(2-hydroxy-4-pentadecylbenzylidene)benzenesulfonamide. Molbank. 2011; 2011(4):M739. https://doi.org/10.3390/M739
Chicago/Turabian StyleNaganagowda, Gadada, and Amorn Petsom. 2011. "4-Amino-N-(2-hydroxy-4-pentadecylbenzylidene)benzenesulfonamide" Molbank 2011, no. 4: M739. https://doi.org/10.3390/M739
APA StyleNaganagowda, G., & Petsom, A. (2011). 4-Amino-N-(2-hydroxy-4-pentadecylbenzylidene)benzenesulfonamide. Molbank, 2011(4), M739. https://doi.org/10.3390/M739