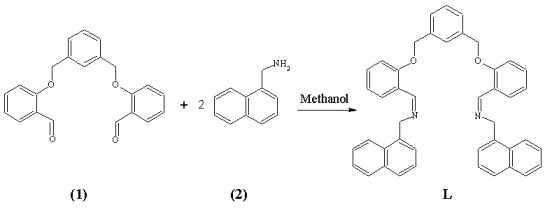
N,N'-{1,3-Phenylenebis[methyleneoxy-2,1-phenylene(Z)methylylidene]}bis[1-(1-naphthyl)methanamine]
Abstract
:Introduction
Experimental
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgements
References
- Lodeiro, C.; Capelo, J.L.; Mejuto, J.C.; Oliveira, E.; Santos, H.M.; Pedras, B.; Nuñez, C. Light and colour as analytical detection tools: A journey into the periodic table using polyamines to bio-inspired systems as chemosensors. Chem. Soc. Rev. 2010, 39, 2948–2976. [Google Scholar]
- Lodeiro, C.; Pina, F. Luminescent and chromogenic molecular probes based on polyamines and related compounds. Coordin. Chem. Rev. 2009, 253, 1353–1385. [Google Scholar]
- Bencini, A.; Lippolis, V. 1,10-phenanthroline: A versatile building block for the construction of ligands for various purposes. Coordin. Chem. Rev. 2010, 254, 2096–2180. [Google Scholar]
- Mewis, R.E.; Archibald, S.J. Biomedical applications of macrocyclic ligand complexes. Coordin. Chem. Rev. 2010, 254, 1686–1712. [Google Scholar]
- Pazos, E.; Vázquez, O.; Mascareñas, J.L.; Vázquez, E.M. Peptide-based fluorescent biosensors. Chem. Soc. Rev. 2009, 38, 3348–3359. [Google Scholar]
- Pedras, B.; Santos, H.M.; Fernades, L.; Covelo, B.; Tamayo, A.; Bertolo, E.; Capelo, J.L.; Avilés, T.; Lodeiro, C. Sensing metal ions with twoazomethine-thiophene pincer ligands (NSN): Fluorescence and MALDI-TOF-MS applications. Inorg. Chem. Commun. 2007, 10, 925–929. [Google Scholar]
- Liu, W.; Xu, L.; Sheng, R.; Wang, P.; Li, H.; Wu, S. A Water Soluble “Switching On” fluorescent Chemosenor of Selectivity to Cd2+. Org. Lett. 2007, 9, 3829–3832. [Google Scholar]
- Yson, M.M. Synthesis and characterisation of a N2O2 containing compound with potential application as a sensor for Cu(II) and Zu (II). BSc Thesis, Canterbury Christ Church University, 2007. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
George, L.A.; Bertolo, E.; Lodeiro, C. N,N'-{1,3-Phenylenebis[methyleneoxy-2,1-phenylene(Z)methylylidene]}bis[1-(1-naphthyl)methanamine]. Molbank 2010, 2010, M702. https://doi.org/10.3390/M702
George LA, Bertolo E, Lodeiro C. N,N'-{1,3-Phenylenebis[methyleneoxy-2,1-phenylene(Z)methylylidene]}bis[1-(1-naphthyl)methanamine]. Molbank. 2010; 2010(4):M702. https://doi.org/10.3390/M702
Chicago/Turabian StyleGeorge, Lucy A., Emilia Bertolo, and Carlos Lodeiro. 2010. "N,N'-{1,3-Phenylenebis[methyleneoxy-2,1-phenylene(Z)methylylidene]}bis[1-(1-naphthyl)methanamine]" Molbank 2010, no. 4: M702. https://doi.org/10.3390/M702
APA StyleGeorge, L. A., Bertolo, E., & Lodeiro, C. (2010). N,N'-{1,3-Phenylenebis[methyleneoxy-2,1-phenylene(Z)methylylidene]}bis[1-(1-naphthyl)methanamine]. Molbank, 2010(4), M702. https://doi.org/10.3390/M702