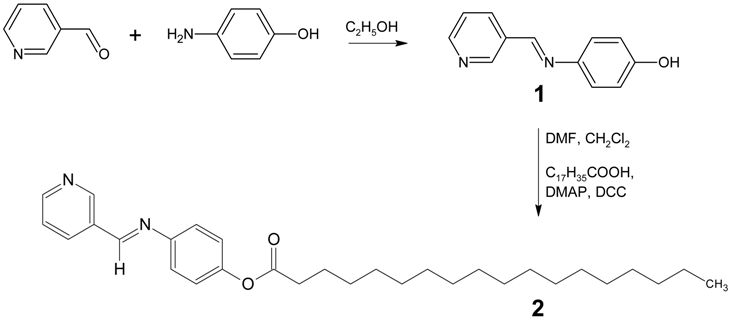
4-[(Pyridin-3-ylmethylene)amino]phenyloctadecanoate
Abstract
:
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgements
References and Notes
- Hadjoudis, E.; Vittorakis, M.; Moustakali-Mavridis, I. Tetrahedron 1987, 43, 1345–1360. [CrossRef]
- Hadjoudis, E.; Rontoyianni, A.; Ambroziak, K.; Dziembowska, T.; Mavridis, I.M. J. Photochem. Photobiol. A: Chem. 2004, 162, 521–530. [CrossRef]
- Oshima, A.; Momotake, A.; Arai, T. J. Photochem. Photobiol. A: Chem. 2004, 162, 473–479.
- Suzuki, T.; Arai, T. Chem. Lett. 2001, 124–125.
- Ha, S.T.; Ong, L.K.; Win, Y.F.; Koh, T.M.; Yeap, G.Y. Molbank 2008, (3), M582.
- Ha, S.T.; Ong, L.K.; Win, Y.F.; Koh, T.M.; Yeap, G.Y. Molbank 2009, (1), M584.
- Ha, S.T.; Ong, L.K.; Win, Y.F.; Koh, T.M.; Yeap, G.Y. Molbank 2009, (1), M585.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ha, S.-T.; Ong, L.-K.; Win, Y.-F.; Koh, T.-M.; Yeap, G.-Y. 4-[(Pyridin-3-ylmethylene)amino]phenyloctadecanoate. Molbank 2009, 2009, M591. https://doi.org/10.3390/M591
Ha S-T, Ong L-K, Win Y-F, Koh T-M, Yeap G-Y. 4-[(Pyridin-3-ylmethylene)amino]phenyloctadecanoate. Molbank. 2009; 2009(1):M591. https://doi.org/10.3390/M591
Chicago/Turabian StyleHa, Sie-Tiong, Lay-Khoon Ong, Yip-Foo Win, Teck-Ming Koh, and Guan-Yeow Yeap. 2009. "4-[(Pyridin-3-ylmethylene)amino]phenyloctadecanoate" Molbank 2009, no. 1: M591. https://doi.org/10.3390/M591
APA StyleHa, S.-T., Ong, L.-K., Win, Y.-F., Koh, T.-M., & Yeap, G.-Y. (2009). 4-[(Pyridin-3-ylmethylene)amino]phenyloctadecanoate. Molbank, 2009(1), M591. https://doi.org/10.3390/M591



