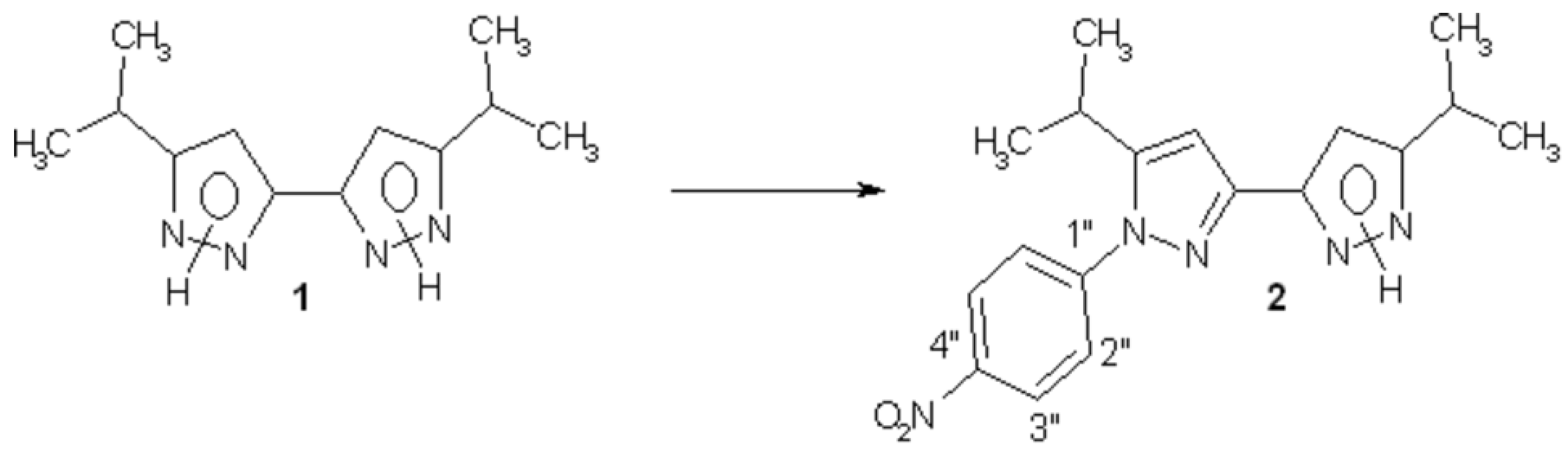
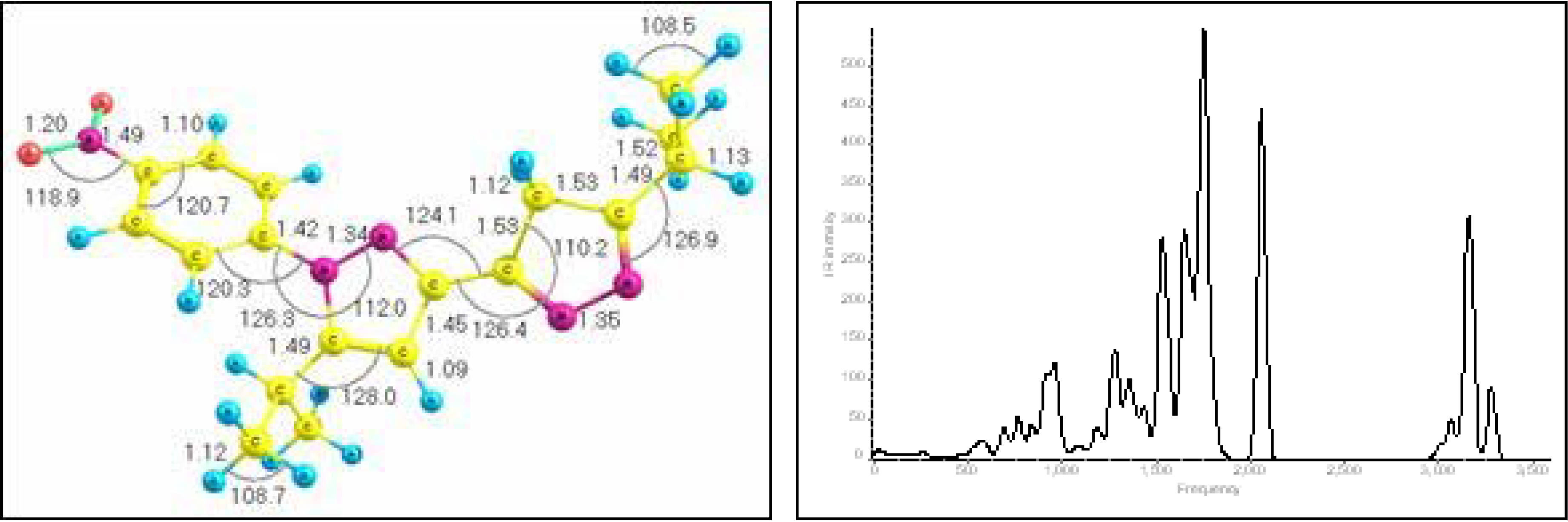
New 1-(4-nitrophenyl)-5,5’-diisopropyl-3,3’-bipyrazole
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References
- Bouabdallah, I.; Ramdani, A.; Zidane, I.; Touzani, R.; Eddike, D.; Radi, S.; Haidoux, A. J. Mar. Chim. Heterocycl. 2004, 3, 1, 39.
- Bouabdallah, I.; Ramdani, A.; Zidane, I.; Eddike, D.; Tillard, M.; Belin, C. Acta Cryst. 2005, E61, 4243.
- Frisch, M.J.; et al. GAUSSIAN 03, Revision A.1. Gaussian, Inc.: Pittsburgh PA, 2003. [Google Scholar]
- Foresman, J.B.; Frisch, Æ. Exploring Chemistry with Electronic Structure Methods, 2nd edition; Gaussian, INC.: Pittsburgh, PA, 1996. [Google Scholar]
- Jalbout, A.F.; Nazari, F.; Turker, L. J. Mol. Struct. (THEOCHEM) 2004, 1, 627. Jalbout, A.F.; Adamowicz, L. Adv. Quant. Chem.(Book Series, Ed. J. Sabin). 2006. xxx-xxx (Reviews).
| 100 K | 298.15 K | 1000 K | 1200 K | 1500 K | 2000 K | |
| Cp | 172.80 | 367.03 | 846.73 | 907.32 | 969.69 | 1030.02 |
| S | 457.64 | 732.22 | 1462.85 | 1622.88 | 1832.55 | 2120.72 |
| ΔH | 11.09 | 64.28 | 522.32 | 698.05 | 980.29 | 1481.85 |
© 2006 MDPI. All rights reserved.
Share and Cite
Bouabdallah, I.; Zidane, I.; Touzani, R.; Ramdani, A.; Jalbout, A.F.; Trzaskowski, B. New 1-(4-nitrophenyl)-5,5’-diisopropyl-3,3’-bipyrazole. Molbank 2006, 2006, M490. https://doi.org/10.3390/M490
Bouabdallah I, Zidane I, Touzani R, Ramdani A, Jalbout AF, Trzaskowski B. New 1-(4-nitrophenyl)-5,5’-diisopropyl-3,3’-bipyrazole. Molbank. 2006; 2006(5):M490. https://doi.org/10.3390/M490
Chicago/Turabian StyleBouabdallah, Ibrahim, Ismail Zidane, Rachid Touzani, Abdelkrim Ramdani, Abraham F. Jalbout, and B. Trzaskowski. 2006. "New 1-(4-nitrophenyl)-5,5’-diisopropyl-3,3’-bipyrazole" Molbank 2006, no. 5: M490. https://doi.org/10.3390/M490
APA StyleBouabdallah, I., Zidane, I., Touzani, R., Ramdani, A., Jalbout, A. F., & Trzaskowski, B. (2006). New 1-(4-nitrophenyl)-5,5’-diisopropyl-3,3’-bipyrazole. Molbank, 2006(5), M490. https://doi.org/10.3390/M490




