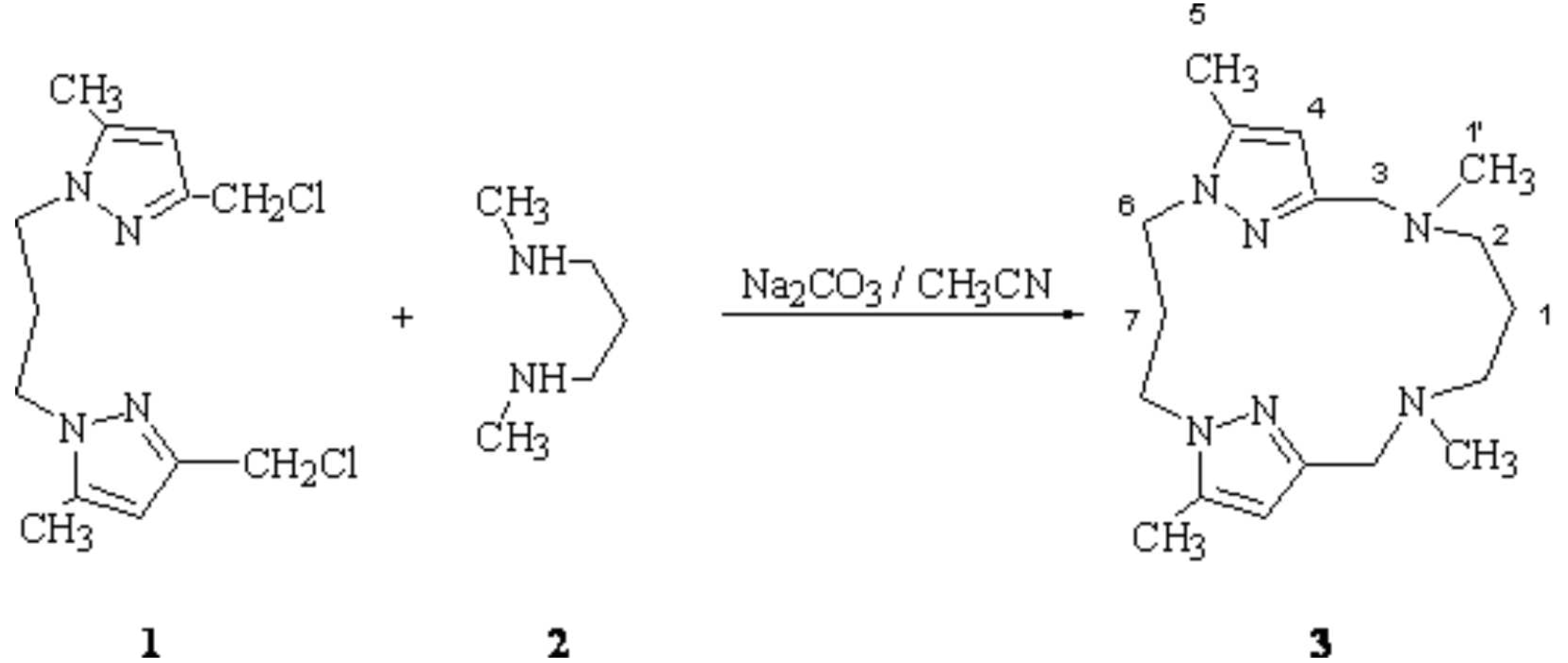
6,10,14,18-Tetramethyl-1,5,10,14,19,20-hexaaza-tricyclo[14.2.1.15,8]eicosa-6,8(20),16(19),17-tetraene
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References
- Tarrago, G.; El Kadiri, S.; Marzin, C.; Coquelet, C. New .J. Chem. 1991, 15, 677.
- Bienvenue, E.; Choua, S.; Lobo-Recio, M-HAS.; Marzin, C.; Pacheco, P.; Seta, P.; Tarrago, G. J. Inorg. Biochem. 1995, 57, 157.
- Sample Availability: Available from MDPI.
© 2005 MDPI. All rights reserved.
Share and Cite
Touzani, R.; Ramdani, A.; El Kadiri, S. 6,10,14,18-Tetramethyl-1,5,10,14,19,20-hexaaza-tricyclo[14.2.1.15,8]eicosa-6,8(20),16(19),17-tetraene. Molbank 2005, 2005, M440. https://doi.org/10.3390/M440
Touzani R, Ramdani A, El Kadiri S. 6,10,14,18-Tetramethyl-1,5,10,14,19,20-hexaaza-tricyclo[14.2.1.15,8]eicosa-6,8(20),16(19),17-tetraene. Molbank. 2005; 2005(4):M440. https://doi.org/10.3390/M440
Chicago/Turabian StyleTouzani, Rachid, Abdelkrim Ramdani, and Sghir El Kadiri. 2005. "6,10,14,18-Tetramethyl-1,5,10,14,19,20-hexaaza-tricyclo[14.2.1.15,8]eicosa-6,8(20),16(19),17-tetraene" Molbank 2005, no. 4: M440. https://doi.org/10.3390/M440
APA StyleTouzani, R., Ramdani, A., & El Kadiri, S. (2005). 6,10,14,18-Tetramethyl-1,5,10,14,19,20-hexaaza-tricyclo[14.2.1.15,8]eicosa-6,8(20),16(19),17-tetraene. Molbank, 2005(4), M440. https://doi.org/10.3390/M440



