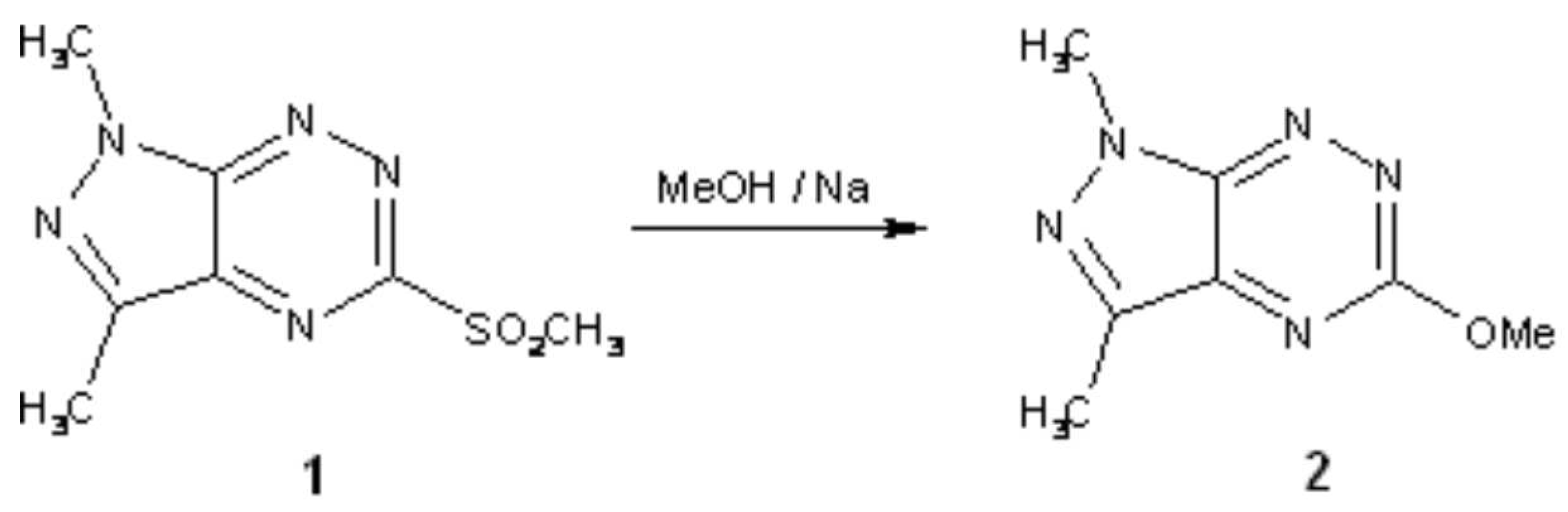
5-Methoxy-1,3-dimethyl-1H-pyrazolo[4,3-e][1,2,4]triazine
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References
- Smirnov, V.V.; Kiprianowa, E.A.; Garagulya, A.D.; Esipov, S. E.; Dovjenko, S. A. FEMS Microbiology Lett. 1997, 153, 357.
- Hirata, K.; Nakagami, H.; Takashina, J.; Mahmud, T.; Kobayashi, M.; In, Y.; Ishida, T.; Miyamoto, K. Heterocycles 1996, 43, 1513.
- Sample Availability: Available from MDPI.
© 2005 MDPI. All rights reserved.
Share and Cite
Mojzych, M.; Branowska, D. 5-Methoxy-1,3-dimethyl-1H-pyrazolo[4,3-e][1,2,4]triazine. Molbank 2005, 2005, M431. https://doi.org/10.3390/M431
Mojzych M, Branowska D. 5-Methoxy-1,3-dimethyl-1H-pyrazolo[4,3-e][1,2,4]triazine. Molbank. 2005; 2005(4):M431. https://doi.org/10.3390/M431
Chicago/Turabian StyleMojzych, Mariusz, and Danuta Branowska. 2005. "5-Methoxy-1,3-dimethyl-1H-pyrazolo[4,3-e][1,2,4]triazine" Molbank 2005, no. 4: M431. https://doi.org/10.3390/M431
APA StyleMojzych, M., & Branowska, D. (2005). 5-Methoxy-1,3-dimethyl-1H-pyrazolo[4,3-e][1,2,4]triazine. Molbank, 2005(4), M431. https://doi.org/10.3390/M431



