TFIP11, CCNL1 and EWSR1 Protein-protein Interactions, and Their Nuclear Localization
Abstract
:1. Introduction
2. Experimental Section
2.1. Northern blot analysis
2.2. Plasmid constructs
2.3. Cell culture and transient transfection
2.4. Immunofluorescence assay and confocal imaging
2.5. Immunoprecipitation assay and western blot analysis
3. Results
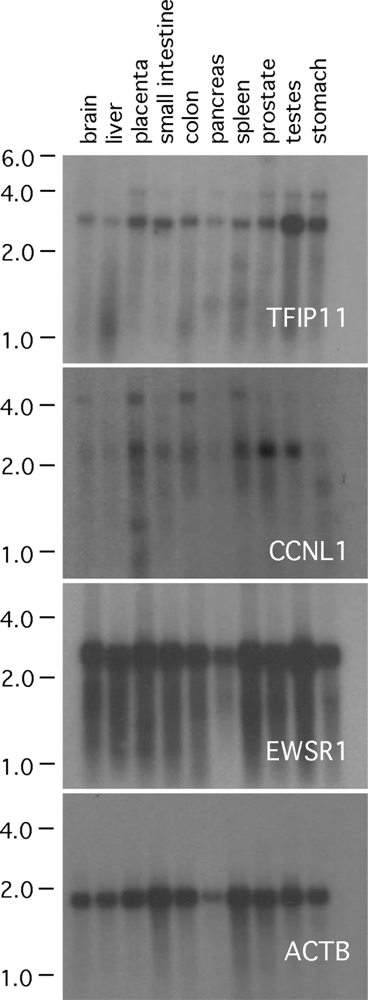
3.1. TFIP11, CCNL1 and EWSR1 Northern blot analysis
3.2. Subcellular localization of GFP-tagged CCNL1 and RFP-tagged EWSR1
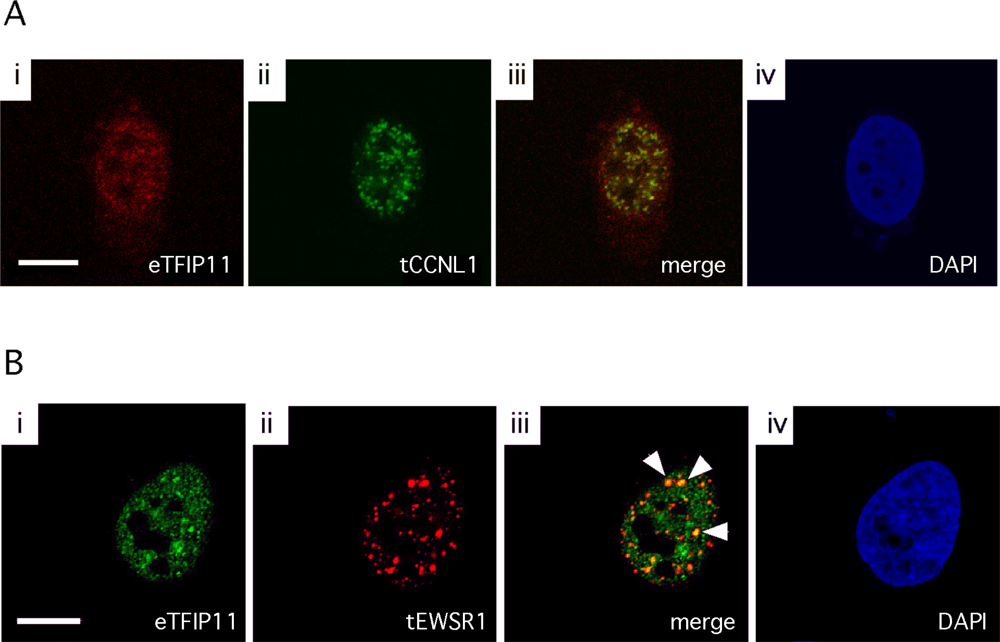
3.3. Colocalization of TFIP11 and CCNL1, and TFIP11 and EWSR1
3.4. EWSR1 interacts with TFIP11 in HEK293 cells
4. Discussion
Acknowledgments
References
- Zhou, Z; Licklider, LJ; Gygi, SP; Reed, R. Comprehensive proteomic analysis of the human spliceosome. Nature 2002, 419, 182–185. [Google Scholar]
- Jurica, MS; Moore, MJ. Pre-mRNA splicing: Awash in a sea of proteins. Mol.Cell 2003, 12, 5–14. [Google Scholar]
- Makarov, EM; Makarova, OV; Urlaub, H; Gentzel, M; Will, CL; Wilm, M; Luhrmann, R. Small nuclear ribonucleoprotein remodeling during catalytic activation of the spliceosome. Science 2002, 298, 2205–2208. [Google Scholar]
- Rappsilber, J; Ryder, U; Lamond, AI; Mann, M. Large-scale proteomic analysis of the human spliceosome. Genome Res 2002, 12, 1231–1245. [Google Scholar]
- Wen, X; Lei, YP; Zhou, YL; Okamoto, CT; Snead, ML; Paine, ML. Structural organization and cellular localization of tuftelin-interacting protein 11 (TFIP11). Cell. Mol. Life Sci 2005, 62, 1038–1046. [Google Scholar]
- Boon, KL; Auchynnikava, T; Edwalds-Gilbert, G; Barrass, JD; Droop, AP; Dez, C; Beggs, JD. Yeast ntr1/spp382 mediates prp43 function in postspliceosomes. Mol. Cell. Biol 2006, 26, 6016–6023. [Google Scholar]
- Tsai, RT; Fu, RH; Yeh, FL; Tseng, CK; Lin, YC; Huang, YH; Cheng, SC. Spliceosome disassembly catalyzed by Prp43 and its associated components Ntr1 and Ntr2. Genes Dev 2005, 19, 2991–3003. [Google Scholar]
- Tsai, RT; Tseng, CK; Lee, PJ; Chen, HC; Fu, RH; Chang, KJ; Yeh, FL; Cheng, SC. Dynamic interactions of Ntr1-Ntr2 with Prp43 and with U5 govern the recruitment of Prp43 to mediate spliceosome disassembly. Mol. Cell. Biol 2007, 27, 8027–8037. [Google Scholar]
- Tanaka, N; Aronova, A; Schwer, B. Ntr1 activates the Prp43 helicase to trigger release of lariat-intron from the spliceosome. Genes Dev 2007, 21, 2312–2325. [Google Scholar]
- Wang, HJ; Tannukit, S; Shapiro, JL; Snead, ML; Paine, ML. Using the yeast two-hybrid assay to discover protein partners for the leucine-rich amelogenin peptide (LRAP) and for tuftelin-interacting protein 11 (TFIP11). Eur. J. Oral Sci 2006, 114(Suppl. 1), 276–279. [Google Scholar]
- Chen, HH; Wang, YC; Fann, MJ. Identification and characterization of the CDK12/cyclin L1 complex involved in alternative splicing regulation. Mol. Cell. Biol 2006, 26, 2736–2745. [Google Scholar]
- Chen, HH; Wong, YH; Geneviere, AM; Fann, MJ. CDK13/CDC2L5 interacts with L-type cyclins and regulates alternative splicing. Biochem. Biophys. Res. Commun 2007, 354, 735–40. [Google Scholar]
- Law, WJ; Cann, KL; Hicks, GG. TLS, EWS and TAF15: A model for transcriptional integration of gene expression. Brief. Func. Gen. & Prot 2006, 5, 8–14. [Google Scholar]
- Dickinson, LA; Edgar, AJ; Ehley, J; Gottesfeld, JM. Cyclin L is an RS domain protein involved in pre-mRNA splicing. J. Biol. Chem 2002, 277, 25465–25473. [Google Scholar]
- Graveley, BR. Sorting out the complexity of SR protein functions. RNA 2000, 6, 1197–1211. [Google Scholar]
- Arvand, A; Denny, CT. Biology of EWS/ETS fusions in Ewing’s family tumors. Oncogene 2001, 20, 5747–5754. [Google Scholar]
- Bertolotti, A; Melot, T; Acker, J; Vigneron, M; Delattre, O; Tora, L. EWS, but not EWS-FLI-1, is associated with both TFIID and RNA polymerase II: Interactions between two members of the TET family, EWS and hTAFII68, and subunits of TFIID and RNA polymerase II complexes. Mol. Cell. Biol 1998, 18, 1489–1497. [Google Scholar]
- Zhang, D; Paley, AJ; Childs, G. The transcriptional repressor ZFM1 interacts with and modulates the ability of EWS to activate transcription. J. Biol. Chem 1998, 273, 18086–18091. [Google Scholar]
- Knoop, LL; Baker, SJ. The splicing factor U1C represses EWS/FLI-mediated transactivation. J. Biol. Chem 2000, 275, 24865–24871. [Google Scholar]
- Sambrook, J; Russell, DW. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Laboratory Press: New York, 2001. [Google Scholar]
- Loyer, P; Trembley, JH; Grenet, JA; Busson, A; Corlu, A; Zhao, W; Kocak, M; Kidd, VJ; Lahti, JM. Characterization of cyclin L1 and L2 interactions with CDK11 and splicing factors: Influence of cyclin L isoforms on splice site selection. J. Biol. Chem. 2008, in press. [Google Scholar]
- Herrmann, A; Fleischer, K; Czajkowska, H; Muller-Newen, G; Becker, W. Characterization of cyclin L1 as an immobile component of the splicing factor compartment. FASEB J 2007, 21, 3142–3152. [Google Scholar]
- Jurica, MS; Licklider, LJ; Gygi, SR; Grigorieff, N; Moore, MJ. Purification and characterization of native spliceosomes suitable for three-dimensional structural analysis. RNA 2002, 8, 426–439. [Google Scholar]
- Hartmuth, K; Urlaub, H; Vornlocher, HP; Will, CL; Gentzel, M; Wilm, M; Luhrmann, R. Protein composition of human prespliceosomes isolated by a tobramycin affinity-selection method. Proc. Natl. Acad. Sci. USA 2002, 99, 16719–16724. [Google Scholar]
- Lamond, AI; Spector, DL. Nuclear speckles: a model for nuclear organelles. Nat. Rev. Mol. Cell. Biol 2003, 4, 605–612. [Google Scholar]
- Young, PJ; Francis, JW; Lince, D; Coon, K; Androphy, EJ; Lorson, CL. The Ewing’s sarcoma protein interacts with the Tudor domain of the survival motor neuron protein. Mol. Brain Res 2003, 119, 37–49. [Google Scholar]
- Knoop, LL; Baker, SJ. EWS/FLI alters 5’-splice site selection. J. Biol. Chem 2001, 276, 22317–22322. [Google Scholar]


© 2008 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/). This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tannukit, S.; Wen, X.; Wang, H.; Paine, M.L. TFIP11, CCNL1 and EWSR1 Protein-protein Interactions, and Their Nuclear Localization. Int. J. Mol. Sci. 2008, 9, 1504-1514. https://doi.org/10.3390/ijms9081504
Tannukit S, Wen X, Wang H, Paine ML. TFIP11, CCNL1 and EWSR1 Protein-protein Interactions, and Their Nuclear Localization. International Journal of Molecular Sciences. 2008; 9(8):1504-1514. https://doi.org/10.3390/ijms9081504
Chicago/Turabian StyleTannukit, Sissada, Xin Wen, HongJun Wang, and Michael L. Paine. 2008. "TFIP11, CCNL1 and EWSR1 Protein-protein Interactions, and Their Nuclear Localization" International Journal of Molecular Sciences 9, no. 8: 1504-1514. https://doi.org/10.3390/ijms9081504
APA StyleTannukit, S., Wen, X., Wang, H., & Paine, M. L. (2008). TFIP11, CCNL1 and EWSR1 Protein-protein Interactions, and Their Nuclear Localization. International Journal of Molecular Sciences, 9(8), 1504-1514. https://doi.org/10.3390/ijms9081504



