Ab Initio Studies on the Preferred Site of Protonation in Cytisine in the Gas Phase and Water
Abstract
:Introduction
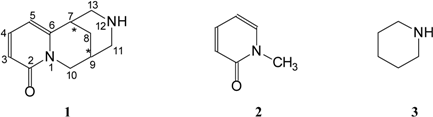
Computational Details
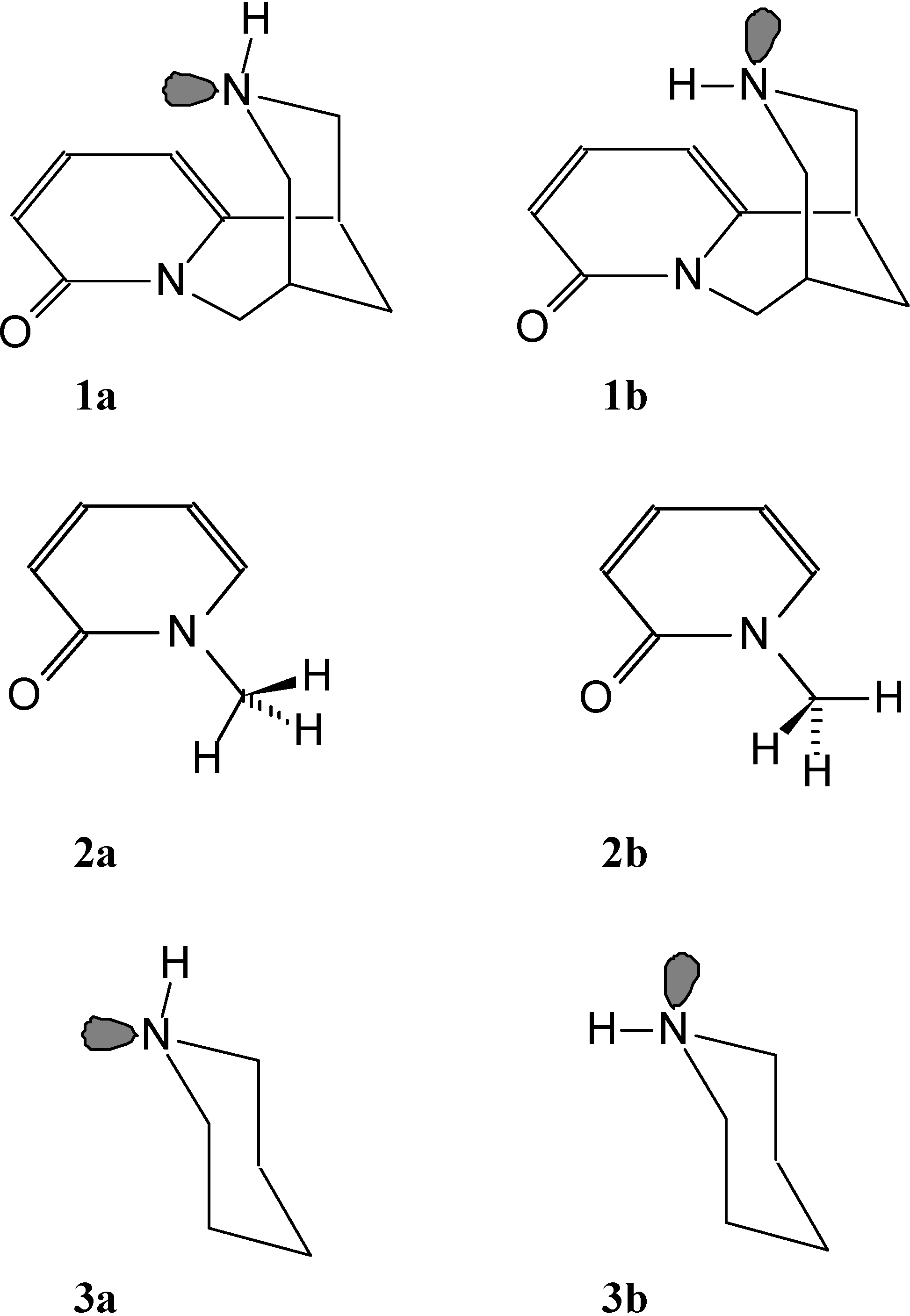
Results and Discussion
1. Preferred Structures for Isolated and Solvated Neutral Molecules
| Compound | Method | Phase | ΔE | ΔG | Preferred Conformation |
|---|---|---|---|---|---|
| 1 | HF/6-31G*b | gas | -0.365 | -0.315 | 1a |
| HF/6-311G**c | gas | -0.435 | -0.58 | 1a | |
| MP2/6-31G*b | gas | -0.005 | 1a | ||
| MP2/6-311G**c | gas | -0.11 | 1a | ||
| DFT(B3LYP)/6-31G*b | gas | 0.29 | 1b | ||
| DFT(B3LYP)/6-311G**c | gas | 0.08 | 1b | ||
| PCMb | water | -0.66 | 1a | ||
| PCMc | water | -0.78 | 1a | ||
| 2 | HF/6-31G*b | gas | 0.63 | 0.10 | 2b |
| HF/6-311G**c | gas | 0.58 | 1.30 | 2b | |
| MP2/6-31G*b | gas | 0.79 | 2b | ||
| MP2/6-311G**c | gas | 0.85 | 2b | ||
| DFT(B3LYP)/6-31G*b | gas | 0.55 | 2b | ||
| DFT(B3LYP)/6-311G**c | gas | 0.41 | 2b | ||
| PCMb | water | 1.00 | 2b | ||
| PCMc | water | 0.80 | 2b | ||
| 3 | HF/6-31G* b | gas | -0.82 | -0.66 | 3a |
| HF/6-311G**c | gas | -0.88 | -0.71 | 3a | |
| MP2/6-31G*b | gas | -0.49 | 3a | ||
| MP2/6-311G**c | gas | -0.53 | 3a | ||
| DFT(B3LYP)/6-31G*b | gas | -0.30 | 3a | ||
| DFT(B3LYP)/6-311G**c | gas | -0.55 | 3a | ||
| PCMb | water | -0.33 | 3a | ||
| PCMc | water | -0.42 | 3a |
2. Geometrical Parameters

3. Possible Basic Sites
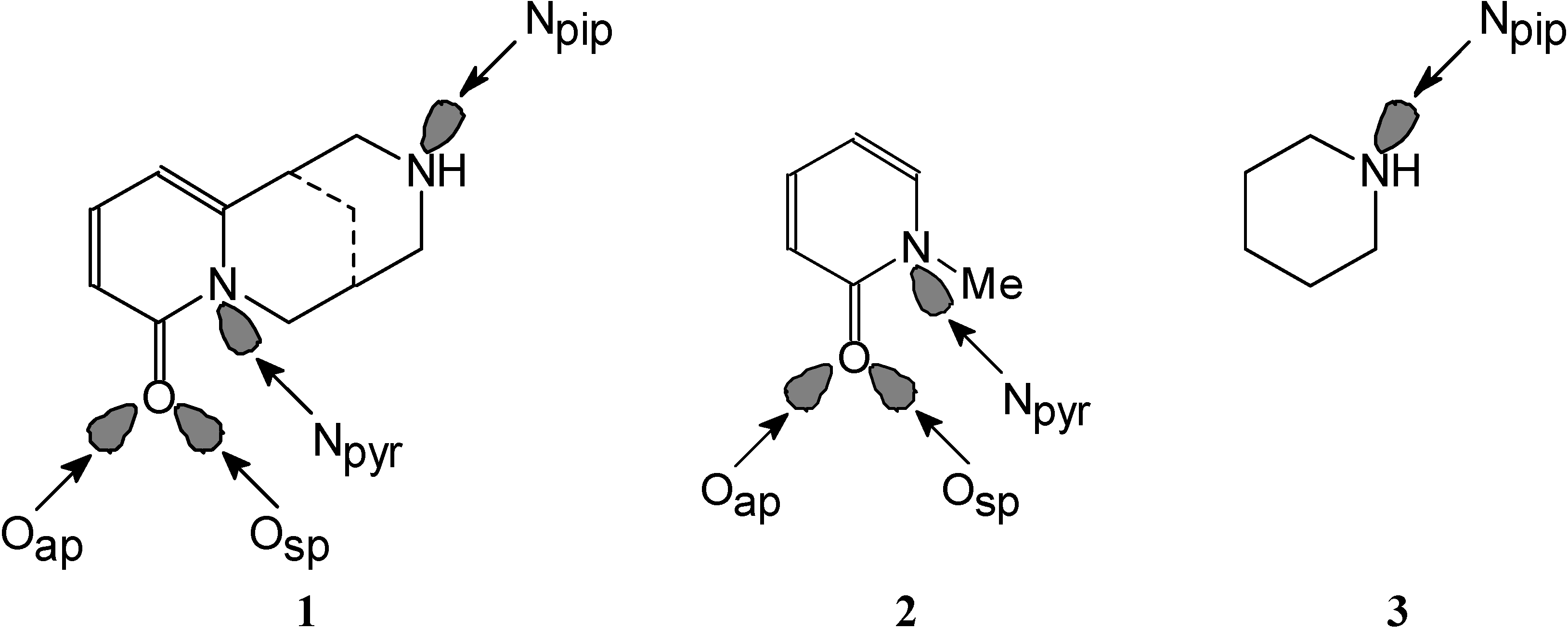
4. Favoured Site of Protonation in the Gas Phase
| Isomer | Method | -Eprot/gas | ||||
|---|---|---|---|---|---|---|
| Osp | Oap | Npyr | Npip | |||
| 1a | HF/6-31G*c | 239.5 | 244.7 | 203.4 | 236.3 | 8.4 |
| HF/6-311G**d | 242.0 | 247.2 | 204.7 | 237.4 | 9.8 | |
| MP2/6-31G*c | 235.2 | 239.9 | 199.3 | 233.7 | 6.2 | |
| MP2/6-311G**d | 238.1 | 242.4 | 201.1 | 233.6 | 8.8 | |
| DFT(B3LYP)/6-31G*c | 237.5 | 241.9 | 201.4 | 234.4 | 7.5 | |
| DFT(B3LYP)/6-311G**d | 237.5 | 241.8 | 200.5 | 233.2 | 8.6 | |
| 1b | HF/6-31G*c | 233.9 | 239.4 | 197.9 | 236.7 | 2.7 |
| HF/6-311G**d | 236.8 | 242.2 | 199.6 | 237.9 | 4.3 | |
| MP2/6-31G*c | 229.2 | 234.2 | 193.4 | 233.7 | 0.5 | |
| MP2/6-311G**d | 232.5 | 237.1 | 195.8 | 233.7 | 3.4 | |
| DFT(B3LYP)/6-31G*c | 232.7 | 237.4 | 196.3 | 234.1 | 3.3 | |
| DFT(B3LYP)/6-311G**d | 233.0 | 237.6 | 195.7 | 233.1 | 4.5 | |
| Isomer | Method | -Eprot/gas | |||
|---|---|---|---|---|---|
| Osp(2) | Oap(2) | Npip(3) | |||
| a | HF/6-31G*c | 230.4 | 234.7 | 241.4 | 6.7 |
| HF/6-311G**d | 232.7 | 237.0 | 242.7 | 5.7 | |
| MP2/6-31G*c | 225.8 | 229.8 | 238.2 | 8.4 | |
| MP2/6-311G**d | 229.0 | 232.9 | 238.2 | 5.3 | |
| DFT(B3LYP)/6-31G*c | 228.0 | 231.7 | 239.3 | 7.6 | |
| DFT(B3LYP)/6-311G**d | 227.7 | 231.5 | 238.1 | 6.6 | |
| b | HF/6-31G*c | 229.8 | 235.0 | 242.2 | 7.2 |
| HF/6-311G**d | 232.1 | 237.4 | 243.5 | 6.1 | |
| MP2/6-31G*c | 225.0 | 230.2 | 238.7 | 8.5 | |
| MP2/6-311G**d | 228.1 | 233.1 | 238.8 | 5.7 | |
| DFT(B3LYP)/6-31G*c | 227.4 | 232.1 | 239.6 | 7.5 | |
| DFT(B3LYP)/6-311G**d | 227.3 | 232.0 | 238.6 | 6.6 | |
| Method | PA | ΔPAb | GB | |||
|---|---|---|---|---|---|---|
| Oap(2b) | Npip(3a) | Oap(2b) | Npip(3a) | |||
| Experiment | 221.3 | 228.0 | 6.7 | 213.9 | 220.1 | 6.2 |
| HF/6-31G*c | 224.8 | 229.6 | 4.8 | 217.0 | 222.0 | 5.0 |
| HF/6-311G**d | 227.0 | 230.9 | 3.9 | 218.9 | 223.3 | 4.4 |
| MP2/6-31G*c | 220.0 | 226.5 | 6.5 | 212.2 | 218.9 | 6.7 |
| MP2/6-311G**d | 222.7 | 226.4 | 3.7 | 214.6 | 218.8 | 4.2 |
| DFT(B3LYP)/6-31G*c | 221.9 | 227.5 | 5.6 | 214.1 | 219.9 | 5.8 |
| DFT(B3LYP)/6-311G**d | 221.6 | 226.3 | 4.7 | 213.5 | 218.7 | 5.2 |
5. Basic Site Preference in Water
| Site | -Eprot/aq | |||||
|---|---|---|---|---|---|---|
| 1a | 1b | 2a | 2b | 3a | 3b | |
| Oap | 28.0 | 27.0 | 26.2 | 26.2 | ||
| Osp | 24.1 | 22.8 | 23.7 | 22.7 | ||
| Npip | 37.8 | 38.5 | 44.9 | 45.2 | ||
Acknowledgments
References
- a)Caldin, E.; Gold, V. (Eds.) Proton-Transfer Reactions; Chapman & Hall: London, 1975.b)Reinchardt, C. Solvents and Solvent Effects in Organic Chemistry; Wiley-VCH: Weinheim, 2002. [Google Scholar]
- Charton, M. J. Org. Chem. 1965, 30, 3346.Hernández-Laguna, A.; Abboud, J.-L. M.; Notario, R.; Homan, H.; Smeyers, Y. G. J. Am. Chem. Soc. 1993, 115, 1450.Notario, R.; Abboud, J.-L. M.; Cativiela, C.; Garcia, J. Y.; Herreros, M.; Homan, H.; Mayoral, J. A.; Salvatella, L. J. Am. Chem. Soc. 1998, 120, 13224.Collado, J. A.; Tuñón, I.; Silla, E.; Ramírez, F. J. J. Phys. Chem. A 2000, 104, 2120.Toyama, A.; Ono, K.; Hashimoto, S.; Takeuchi, H. J. Phys. Chem. A 2002, 106, 3403.
- Gawinecki, R.; Raczyńska, E. D.; Rasała, D.; Styrcz, S. Tetrahedron 1997, 53, 17211.Raczyńska, E. D.; Darowska, M.; Rudka, T.; Górnicka, E. Cur. Top. Anal. Chem. 2002, 3, 125.Raczyńska, E. D.; Darowska, M.; Dąbkowska, I.; Decouzon, M.; Gal, J.-F.; Maria, P.-C.; Dubin-Poliart, C. J. Org. Chem. 2004, 69, 4023.
- a)Cordell, G. A. An Introduction to Alkaloids: a Biogenic Approach; Wiley: New York, 1981. [Google Scholar]Michael, J. P. Nat. Prod. Rep. 2001, 18, 520.
- Chandler, C. J.; Stolerman, I. P. Psychopharmacology (Berlin) 1997, 129, 257.Zhang, J.; Steinbach, J. H. Brain Res. 2003, 959, 98.
- Imming, P.; Klaperski, P.; Stubbs, M. T.; Seitz, G.; Gundisch, D. Eur. J. Med. Chem. 2001, 36, 375.Houlihan, L. M.; Slater, Y.; Guerra, D. L.; Peng, J. H.; Kuo, Y. P.; Lukas, R. J.; Cassels, B. K.; Bermudez, I. J. Neurochem. 2001, 78, 1029.Nicolotti, O.; Canu Boido, C.; Sparatore, F.; Carotti, A. Farmaco 2002, 57, 469.Fredrickson, P.; Boules, M.; Yerbury, S.; Richelson, E. Eur. J. Pharmacol. 2003, 458, 111.
- Colquhoun, L. M.; Patrick, J. W. Adv. Pharmacol. 1997, 39, 191.Gotti, C.; Fornasari, D.; Clementi, F. Prog. Neurobiol. 1997, 53, 199.Wenger, B. W.; Bryant, D. L.; Boyd, R. T.; McKay, D. B. J. Pharmacol. Exp. Ther. 1997, 281, 905.Changeux, J. P.; Bertrand, D.; Corringer, P. J.; Dehaene, S.; Edelstein, S.; Lena, C.; Le Novere, N.; Marubio, L.; Picciotto, M.; Zoli, M. Brain Res. Rev. 1998, 26, 198.Leonard, S.; Bertrand, D. Nicotine Tob. Res. 2001, 3, 203.
- Sihver, W.; Gillberg, P. G.; Svensson, A. L.; Nordberg, A. Neuroscience 1999, 94, 685.Court, J.; Spurden, D.; Lloyd, S.; McKeith, I.; Ballard, C.; Cairns, N.; Kerwin, R.; Perry, R.; Perry, E. J. Neurochem. 1999, 73, 1590.Durany, N.; Zochling, R.; Boissl, K. W.; Paulus, W.; Ransmayr, G.; Tatschner, T.; Danielczyk, W.; Jellinger, K.; Deckert, J.; Riederer, P. Neurosci. Lett. 2000, 287, 109.Tizabi, Y.; Rezvanil, A. H.; Russell, L. T.; Tyler, K. Y.; Overstreet, D. H. Pharmacol. Biochem. Behav. 2000, 66, 73.Lloyd, G. K.; Williams, M. J Pharmacol. Exp. Ther. 2000, 292, 4617.Tizabi, Y.; Getachew, B.; Davila-Garcia, M.; Taylor, R. E. Alcohol Alcohol 2001, 36, 318.Marutle, A.; Zhang, X.; Court, J.; Piggott, M.; Johnson, M.; Perry, R.; Perry, E.; Nordberg, A. J. Chem. Neuroanat. 2001, 22, 115.
- Mascagni, P.; Christodoulou, M.; Gibbons, W. A.; Asres, K.; Phillipson, J. D.; Niccolai, N.; Mangani, S. J. Chem. Soc., Perkin Trans. 2 1987, 1159.Freer, A. A.; Robins, D. J.; Sheldrake, G. N. Acta Crystallogr. Sect. C: Cryst. Struct. Commun. 1987, C43, 1119.Barlow, R. B.; Johnson, O. Br. J. Pharmacol. 1989, 98, 799.
- Górnicka, E.; Rode, J. E.; Raczyńska, E. D.; Dasiewicz, B.; Dobrowolski, J. Cz. Vibr. Spectrosc. 2004, 36, 105.
- Liu, Z.; Yang, L.; Jia, Z.; Chen, J. Magn. Reson. Chem. 1992, 30, 511.Wysocka, W.; Przybył, A.; Brukwicki, T. J. Monatsh. Chem. 1994, 125, 1267.Wysocka, W.; Brukwicki, T. J. Mol. Struct. 1996, 385, 23.Brukwicki, T.; Wysocka, W. J. Mol. Struct. 1999, 474, 215.
- Górnicka, E.; Raczyńska, E. D. Talanta 2002, 57, 609.Rode, J. E.; Raczyńska, E. D.; Górnicka, E.; Dobrowolski, J. Cz. J. Mol. Struct. 2005, (in press).
- Górnicka, E.; Makowski, M.; Darowska, M.; Raczyńska, E. D. Pol. J. Chem. 2001, 75, 1483.b)Darowska, M.; Makowski, M.; Górnicka, E.; Raczyńska, E. D. The Ninth International Conference on Correlation Analysis in Chemistry; Borówno, 2001. [Google Scholar]
- Barlow, R. B.; McLeod, L. J. Br. J. Pharmacol. 1969, 35, 161.
- a)Hehre, W. J.; Radom, L.; Schleyer, P.v.R.; Pople, J. A. Ab Initio Molecular Orbital Theory; Wiley: New York, 1986; p. 785. [Google Scholar]b)Parr, R. G.; Yang, W. Density Functional Theory of Atoms and Molecules; Oxford University Press: New York, 1989. [Google Scholar]c)Density Functionals Methods in Chemistry; Labanowski, J. K.; Andzelm, J. W. (Eds.) Springer-Verlag: New York, 1991.Möller, C.; Plesset, M. S. Phys. Rev. 1934, 46, 618.Pople, J. A.; Brinkley, J. S.; Seeger, R. Int. Quantum. Chem. Symp. 1976, 10, 1.Becke, A. D. J. Chem. Phys. 1993, 98, 5648.Lee, C.; Yang, W.; Parr, R.G. Phys. Rev. 1988, 37, 785.
- Miertu, S.; Scrocco, E.; Tomasi, J. Chem. Phys. 1981, 55, 117.Szafran, M.; Karelson, M. M.; Katritzky, A. R.; Koput, J.; Zerner, M. C. J. Comput. Chem. 1993, 1, 371.Cammi, R.; Tomasi, J. J. Comput. Chem. 1995, 16, 1449.Pomelli, C. S.; Tomasi, J. Theor. Chem. Acc. 1997, 96, 39.
- Raczyńska, E. D.; Mishima, M.; Mustanir. Bull. Chem. Soc. Jpn. 1998, 71, 2175.Makowski, M.; Raczyńska, E. D.; Chmurzyński, L. J. Phys. Chem. A 2001, 105, 869.Raczyńska, E. D.; Darowska, M.; Cyrański, M. K.; Makowski, M.; Rudka, T.; Gal, J.-F.; Maria, P.-M. J. Phys. Org. Chem. 2003, 16, 783.
- Raczyńska, E. D.; Woniak, K.; Dolecka, E.; Darowska, M. J. Phys. Org. Chem. 2002, 15, 706. [CrossRef]
- Smith, B. J.; Radom, L. J. Am. Chem. Soc. 1993, 115, 4885. [CrossRef]
- Amekraz, B.; Tortajada, J.; Morizur, J.-P.; González, A. I.; Mó, O.; Yáñez, M.; Leito, I.; Maria, P.-C.; Gal, J.-F. New J. Chem. 1996, 20, 1011.Notario, R.; Castaño, O.; Herreros, M.; Abboud, J. L. M. J. Mol. Struct. (Theochem) 1996, 371, 21.
- Maksi, Z. B.; Kovaevi, B. J. Org. Chem. 2000, 65, 3303.Koppel, I. A.; Schwesinger, R.; Breuer, T.; Burk, P.; Herodes, K.; Koppel, I.; Leito, I.; Mishima, M. J. Phys. Chem. A 2001, 105, 9575.
- Howard, S. T.; Platts, J. A. J. Org. Chem. 1998, 63, 3568. [CrossRef]
- Schmidt, M. W.; Baldridge, K. K.; Boatz, J. A.; Elbert, S. T.; Gordon, M. S.; Jensen, J. J.; Koseki, S.; Matsunaga, N.; Nguyen, K. A.; Su, S.; Windus, T. L.; Dupuis, M.; Montgomery, J. A. J. Comput.Chem. 1993, 14, 1347. [CrossRef]
- Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Montgomery, J. A., Jr.; Vreven, T.; Kudin, K. N.; Burant, J. C.; Millam, J. M.; Iyengar, S. S.; Tomasi, J.; Barone, V.; Mennucci, B.; Cossi, M.; Scalmani, G.; Rega, N.; Petersson, G. A.; Nakatsuji, H.; Hada, M.; Ehara, K.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Klene, M.; Li, X.; Knox, J. E.; Hratchian, H. P.; Cross, J. B.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C.; Ochterski, J. W.; Ayala, P. Y.; Morokuma, K.; Voth, G. A.; Salvador, P.; Dannenberg, J. J.; Zakrzewski, V. G.; Dapprich, S.; Daniels, A. D.; Strain, M. C.; Farkas, O.; Malick, D. K.; Rabuck, A. D.; Raghavachari, K.; Foresman, J. B.; Ortiz, J. V.; Cui, Q.; Baboul, A. G.; Clifford, S.; Cioslowski, J.; Stefanov, B. B.; Liu, G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Martin, R.L.; Fox, D. J.; Keith, T.; Al-Laham, M. A.; Peng, C. Y.; Nanayakkara, A.; Challacombe, M.; Gill, P. M. W.; Johnson, B. G.; Chen, W.; Wong, M. W.; Gonzalez, C.; Pople, J. A. Gaussian; Gaussian, Inc.: Pittsburgh, PA, 2003. [Google Scholar]
- a)Kier, L.B. Molecular Orbital Theory in Drug Research; Academic Press: New York, 1971; Chapter 8. [Google Scholar]Sheridan, R.P.; Nilakantan, R.; Dixon, J. S.; Venkataraghavan, R. J. Med. Chem. 1986, 29, 899.Barlow, R. B.; Howard, J. A. K.; Johnson, O. Acta Crystallogr. 1986, C42, 853.Barlow, R. B.; Johnson, O. Br. J. Pharmacol. 1989, 98, 799.
- Sihver, W.; Gillberg, P. G.; Nordberg, A. Neuroscience 1998, 85, 1121.Galvez-Ruano, E.; Iriepa-Canalda, I.; Morreale, A.; Lipkowitz, K. B. J. Comput. Aided Mol. Des. 1999, 13, 57.Brejc, K.; van Dijk, W. J.; Klaassen, R. V.; Schuurmans, M.; van Der Oost, J.; Smit, A. B.; Sixma, T. K. Nature 2001, 411, 269.Grutter, T.; Changeux, J. P. Trends Biochem. Sci. 2001, 26, 459.Morreale, A.; Maseras, F.; Iriepa, I.; Galvez, E. J. Mol. Graph. Model. 2002, 21, 111.Schapira, M.; Abagyan, R.; Totrov, M. BMC Struct. Biol. 2002, 2, 1.
- Krygowski, T. M. J. Chem. Inf. Comput. Sci. 1993, 33, 70.Krygowski, T. M.; Cyrański, M. K. Chem Rev. 2001, 101, 1385.
- Takeshima, T.; Fukumoto, R.; Egawa, T.; Konaka, S. J. Phys. Chem. A 2002, 106, 8734. [CrossRef]
- Hunter, E. P. L.; Lias, S. G. J. Phys. Chem. Ref. Data 1998, 27, 413. [CrossRef]
- Graton, J.; Bethelot, M.; Gal, J.-F.; Girard, S.; Laurence, C.; Lebreton, J.; Le Questel, J.-Y.; Maria, P.-C.; Naus, P. J. Am. Chem. Soc. 2002, 124, 10552. [CrossRef]
- Albert, A.; Serjeant, E. P. The Determination of Ionization Constants; Chapman & Hall: London, 1984. [Google Scholar]
© 2005 by MDPI (http://www.mdpi.org).
Share and Cite
Raczyńska, E.D.; Makowski, M.; Górnicka, E.; Darowska, M. Ab Initio Studies on the Preferred Site of Protonation in Cytisine in the Gas Phase and Water. Int. J. Mol. Sci. 2005, 6, 143-156. https://doi.org/10.3390/i6010143
Raczyńska ED, Makowski M, Górnicka E, Darowska M. Ab Initio Studies on the Preferred Site of Protonation in Cytisine in the Gas Phase and Water. International Journal of Molecular Sciences. 2005; 6(1):143-156. https://doi.org/10.3390/i6010143
Chicago/Turabian StyleRaczyńska, Ewa D., Mariusz Makowski, Elżbieta Górnicka, and Małgorzata Darowska. 2005. "Ab Initio Studies on the Preferred Site of Protonation in Cytisine in the Gas Phase and Water" International Journal of Molecular Sciences 6, no. 1: 143-156. https://doi.org/10.3390/i6010143
APA StyleRaczyńska, E. D., Makowski, M., Górnicka, E., & Darowska, M. (2005). Ab Initio Studies on the Preferred Site of Protonation in Cytisine in the Gas Phase and Water. International Journal of Molecular Sciences, 6(1), 143-156. https://doi.org/10.3390/i6010143




