PD-1/PD-L1 Inhibitors and Chemotherapy Synergy: Impact on Drug Resistance and PD-L1 Expression in Breast Cancer-Immune Cell Co-Cultures
Abstract
1. Introduction
2. Results
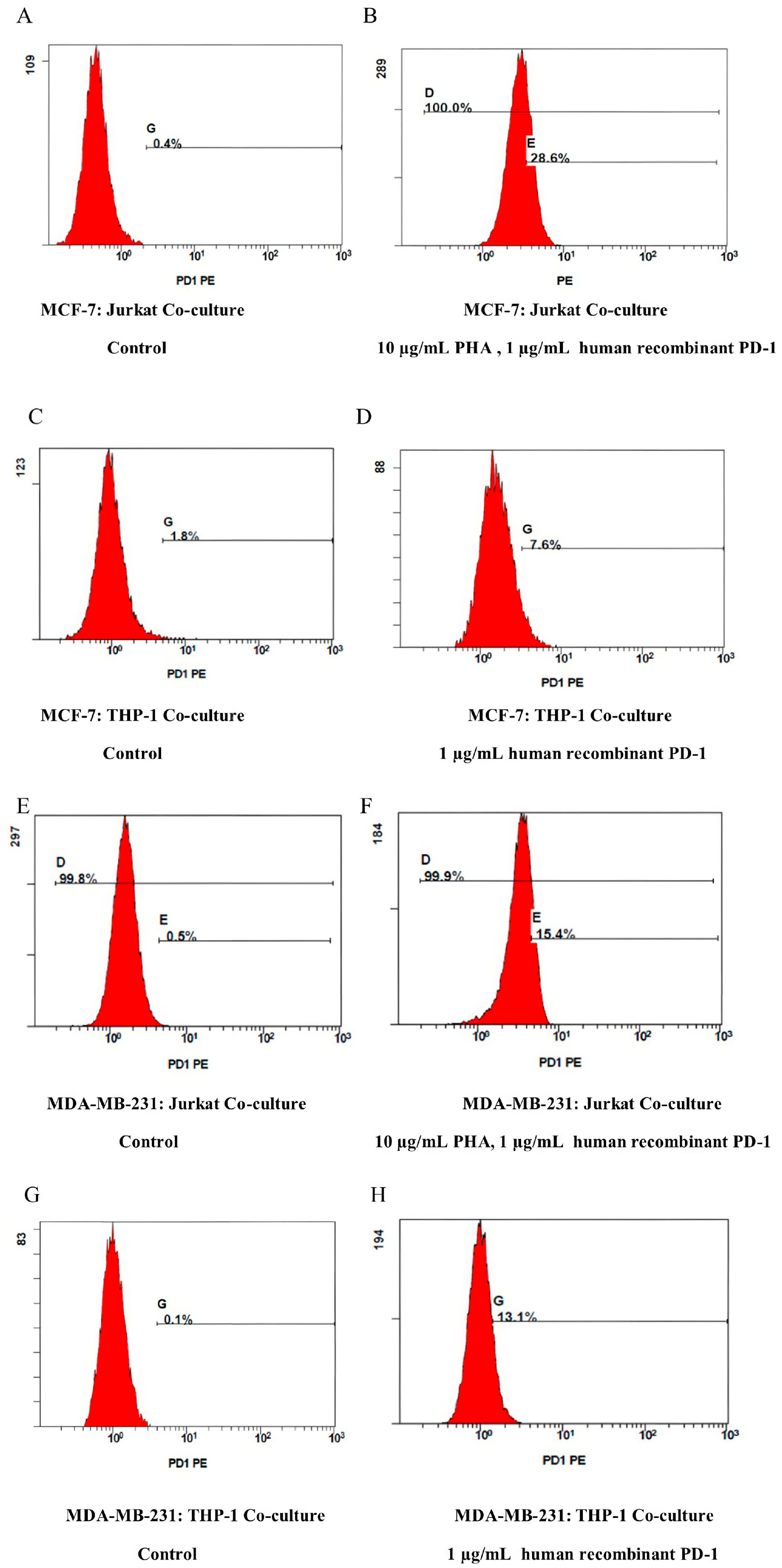
2.1. Co-Culture Model Optimization
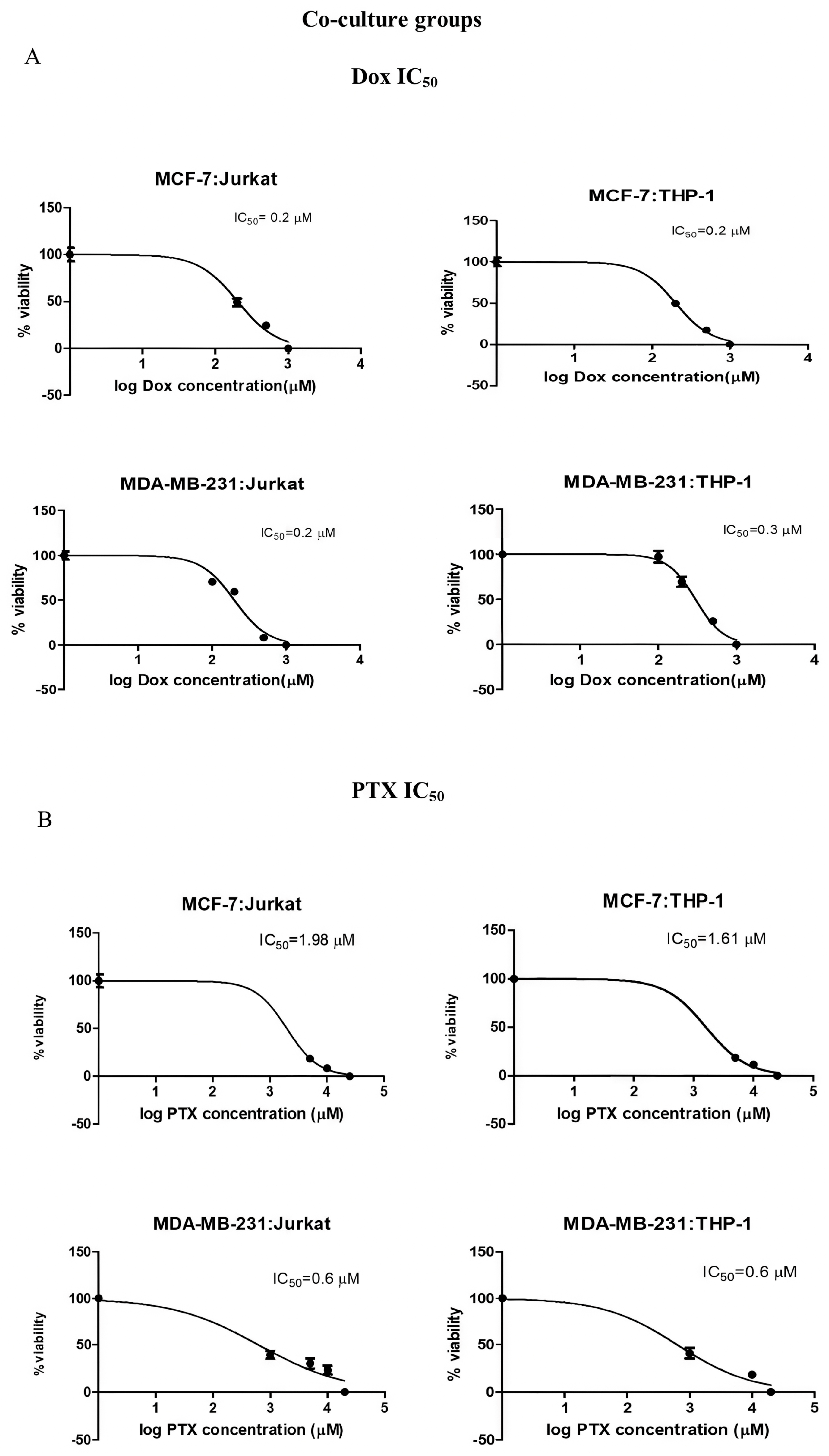
2.2. Dox and PTX Treatments in the Breast Cancer Co-Culture Models
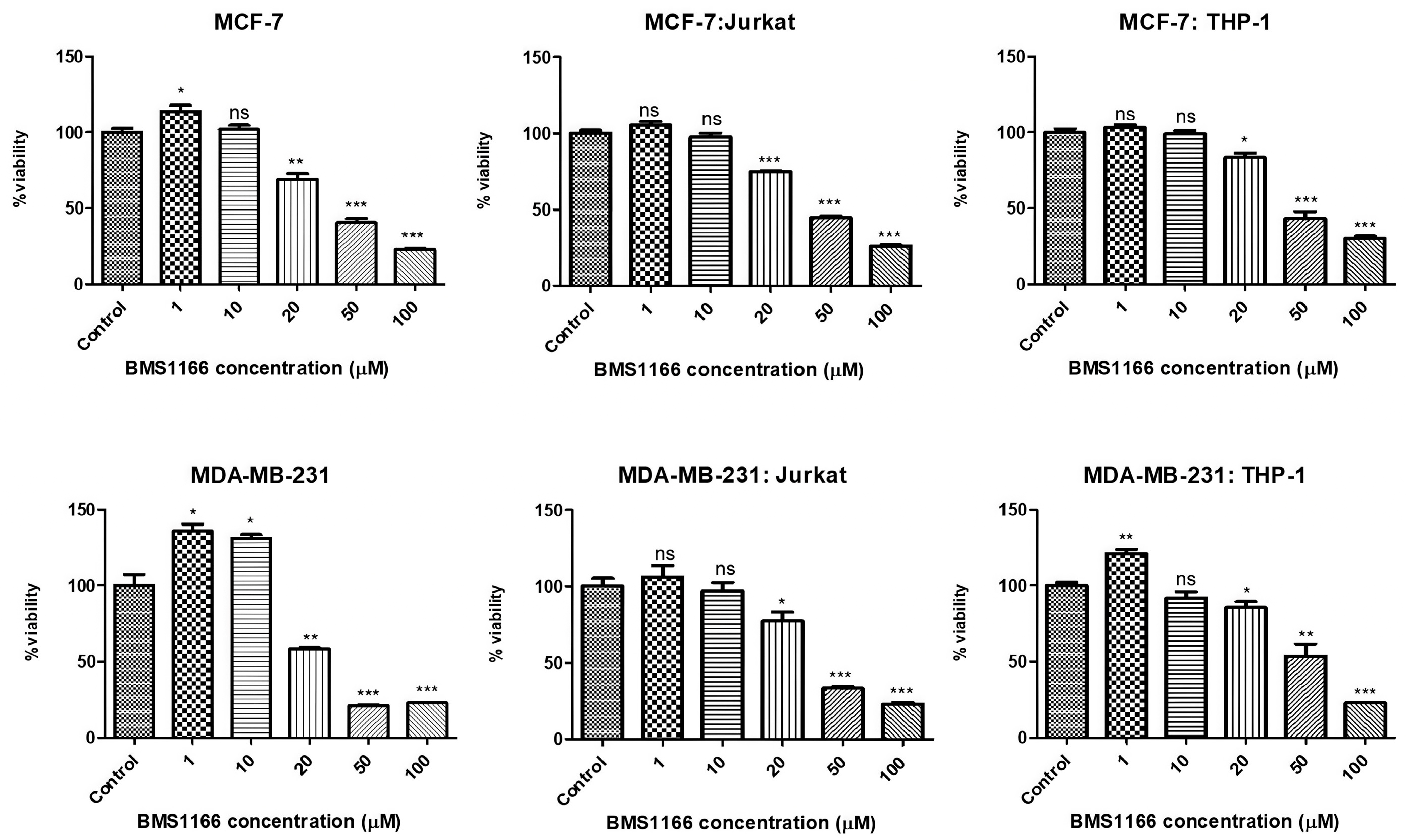
2.3. BMS-1166 and Its Effect on Breast Cancer Cell Viability
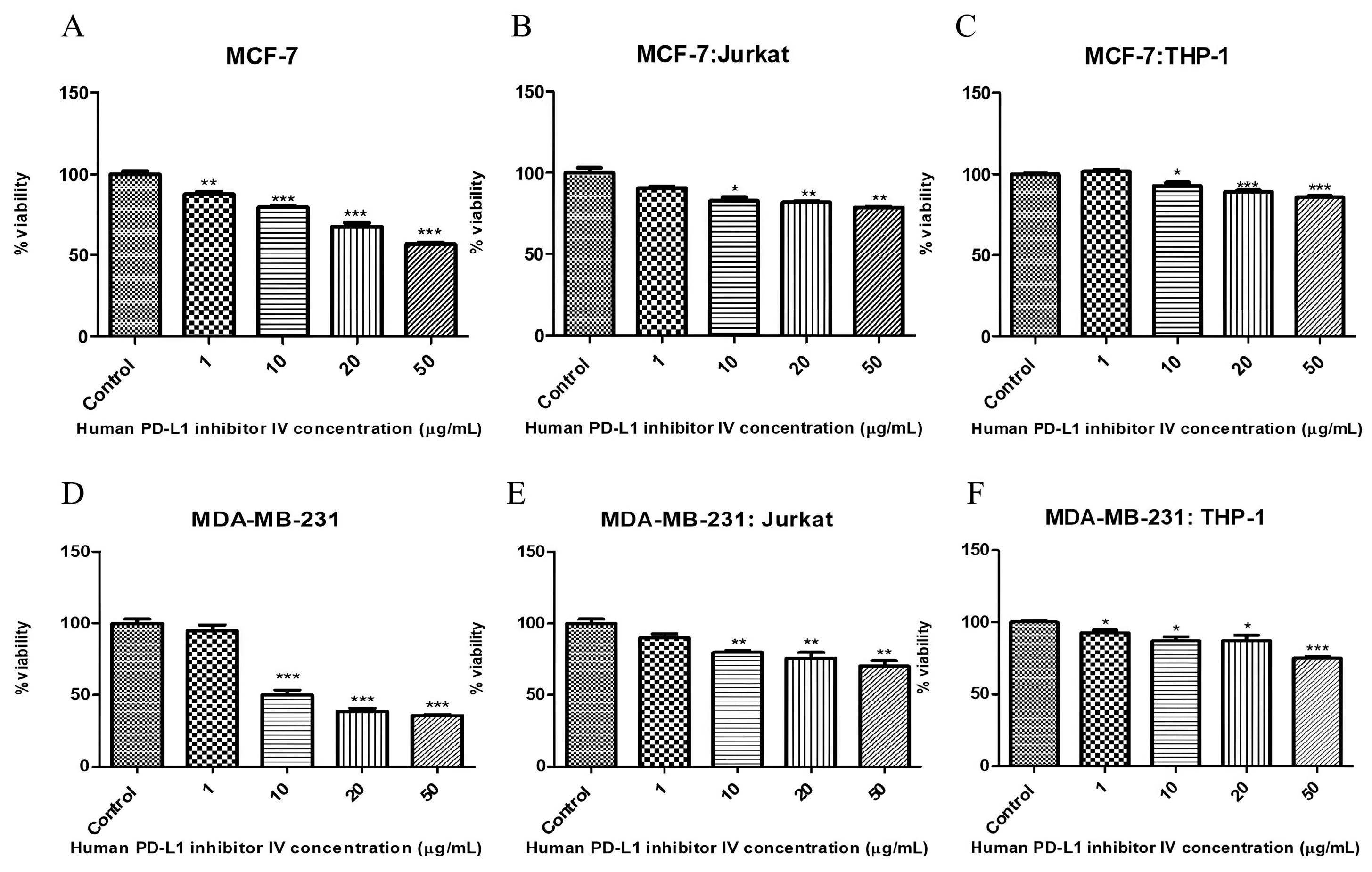
2.4. Human PD-L1 Inhibitor IV and Its Effect on Breast Cancer Cell Viability
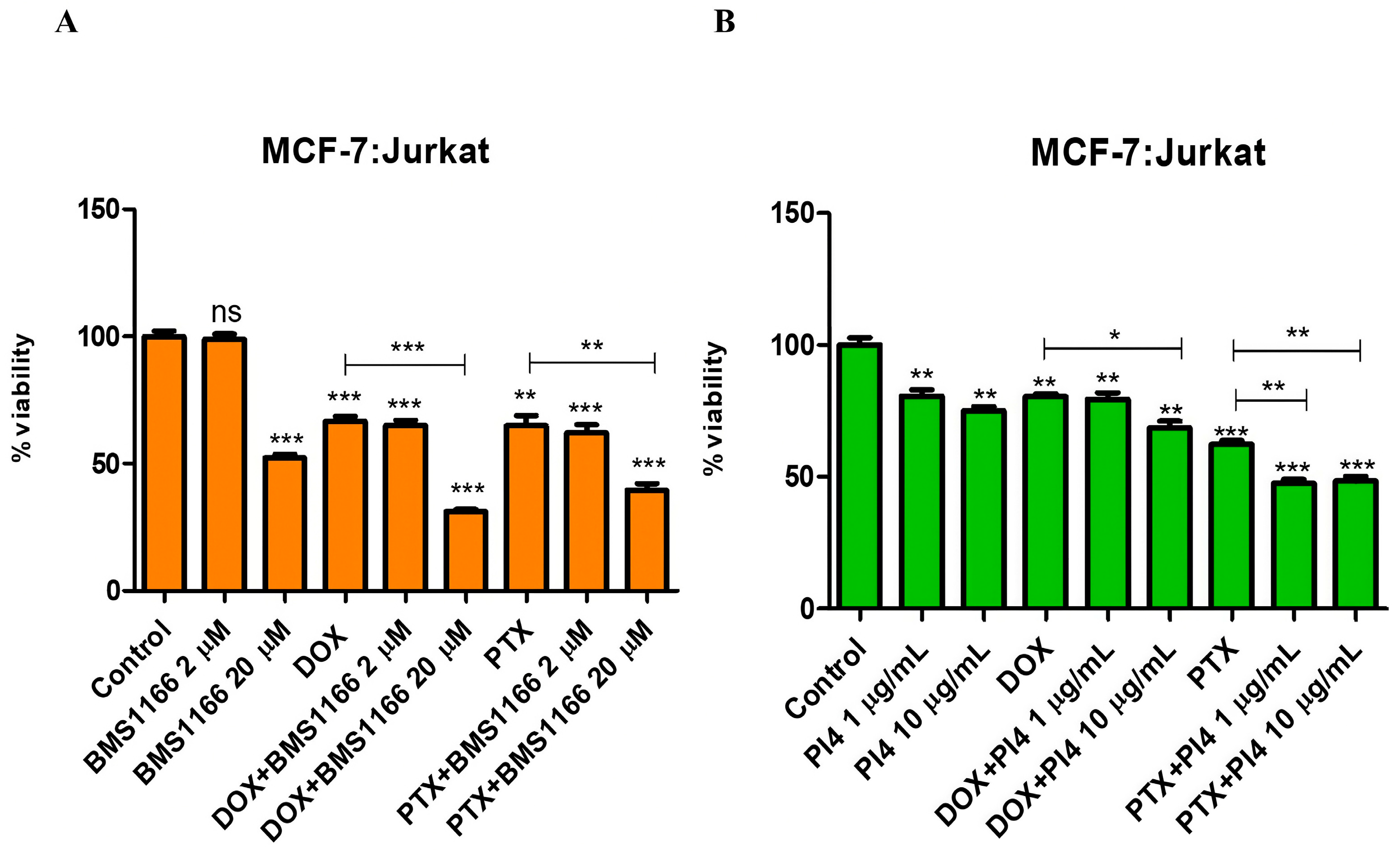
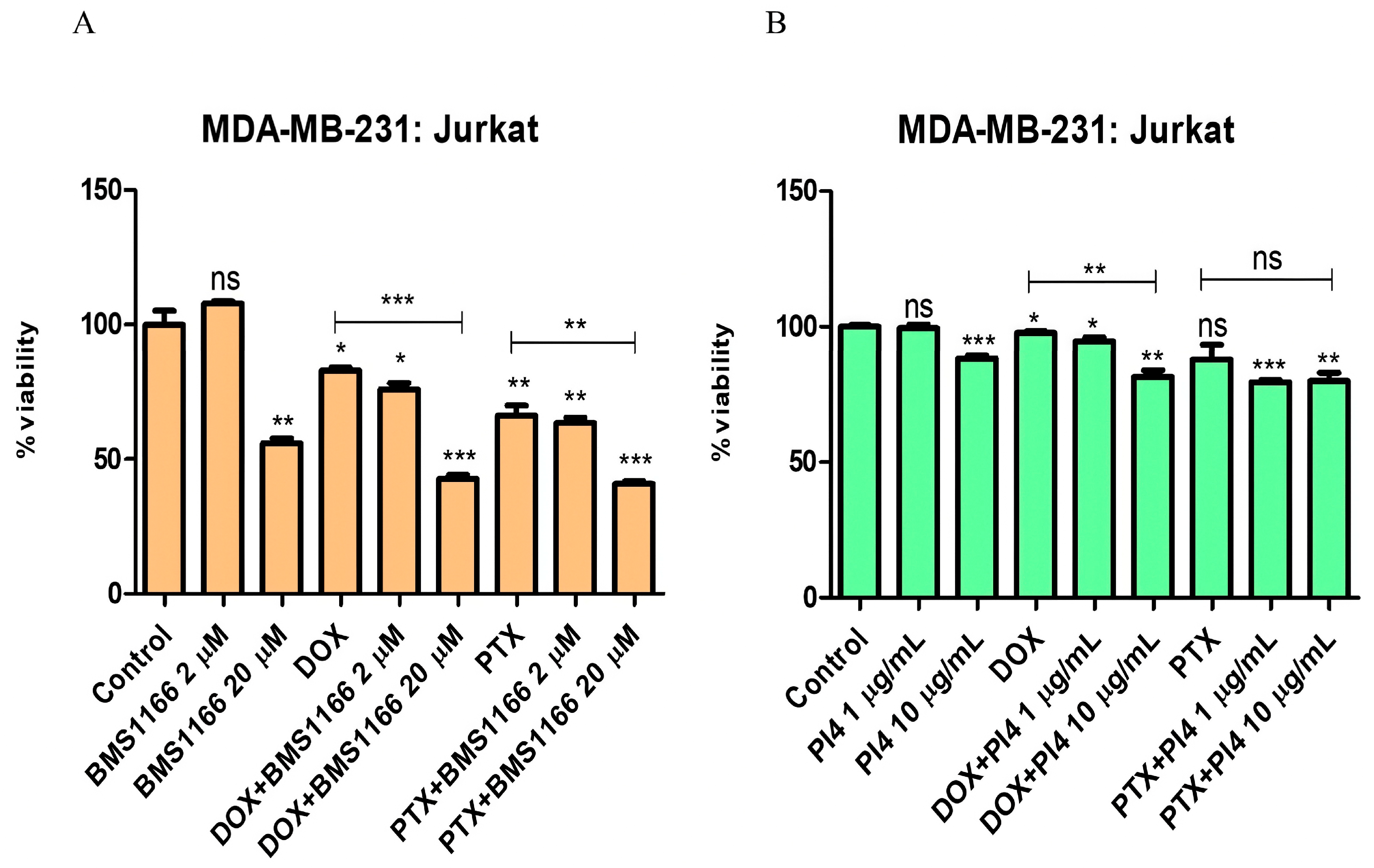
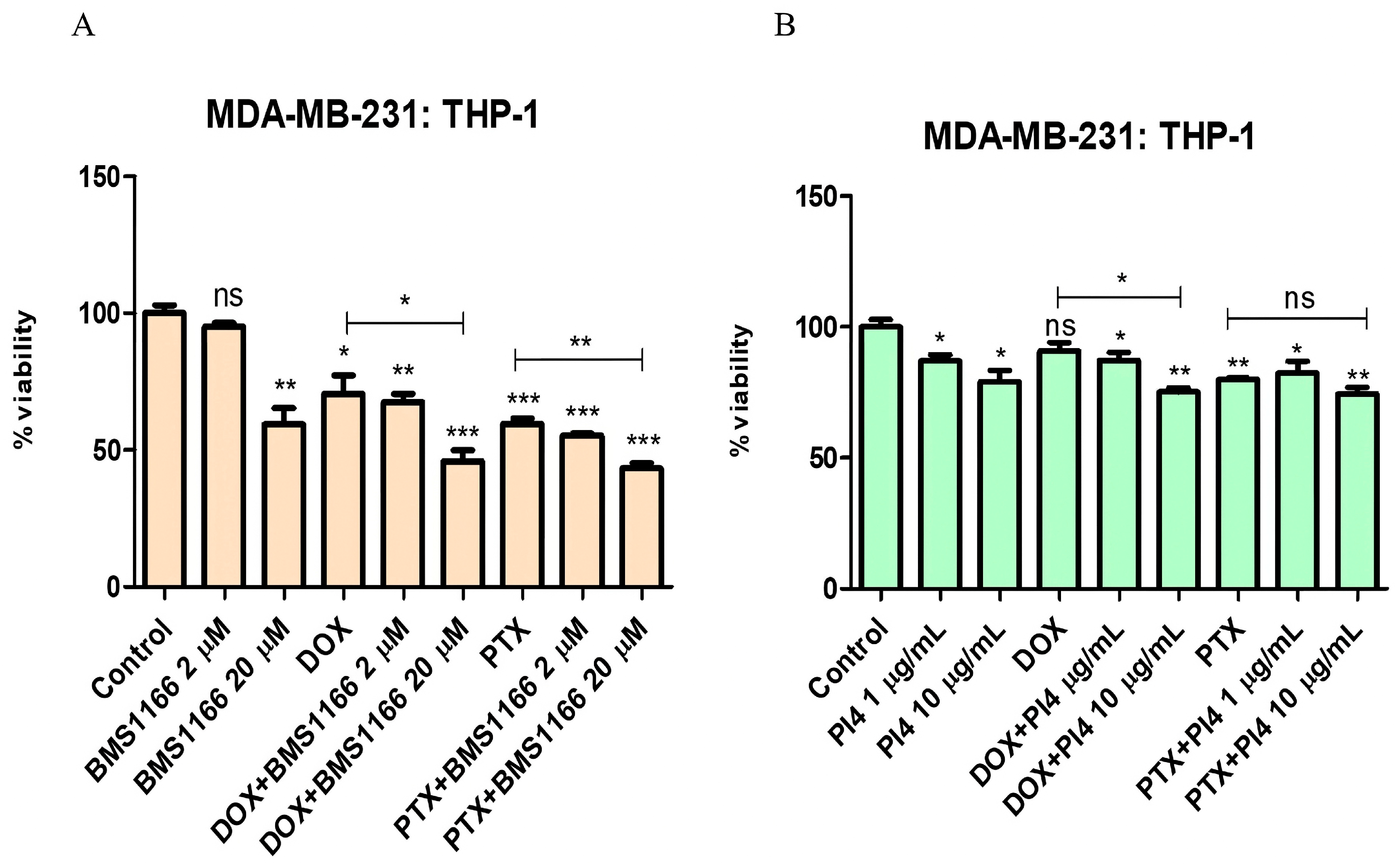
2.5. Cell Viability Analyses of the Combined Use of PD-1/PD-L1 Inhibitors and Chemotherapeutic Agents
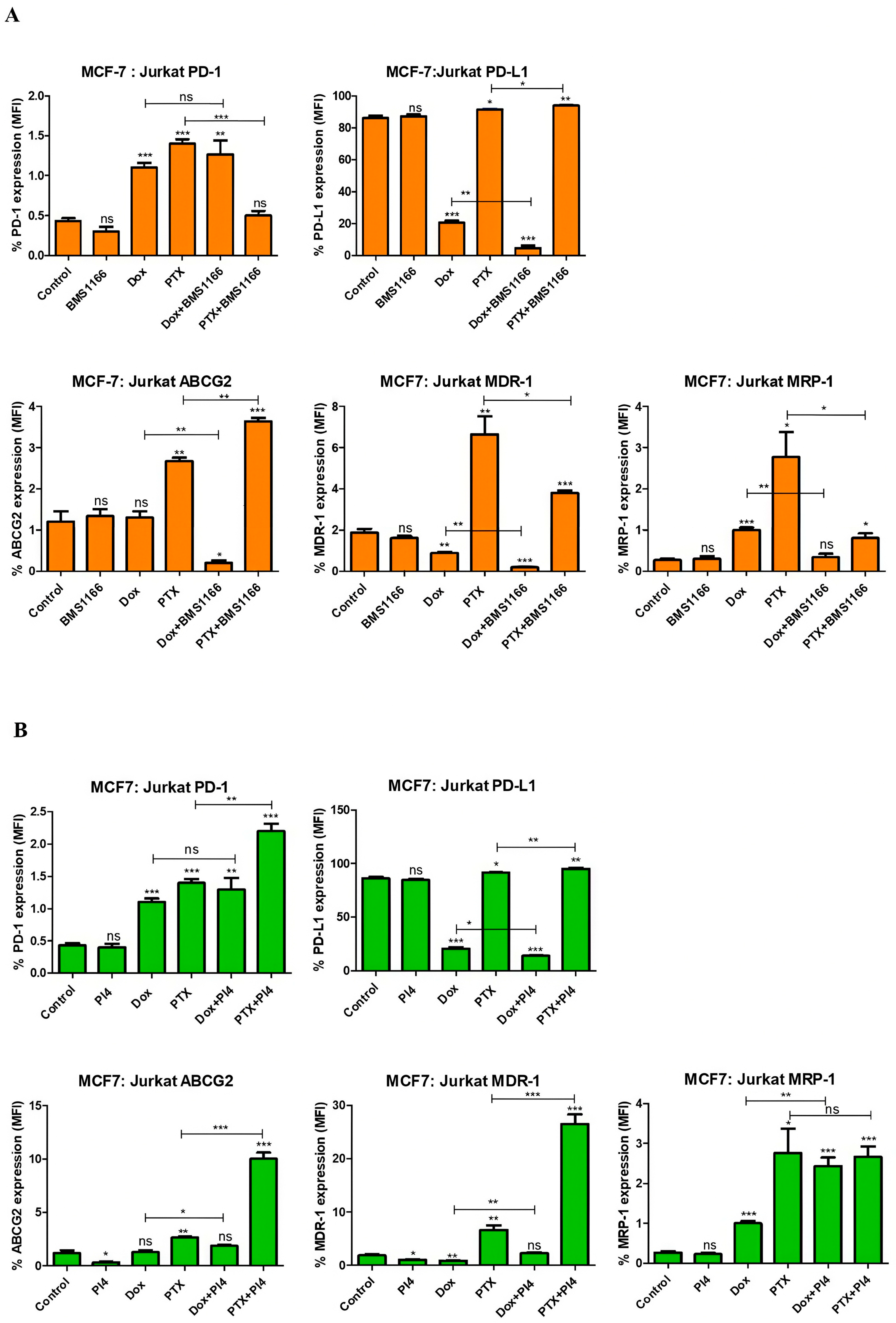
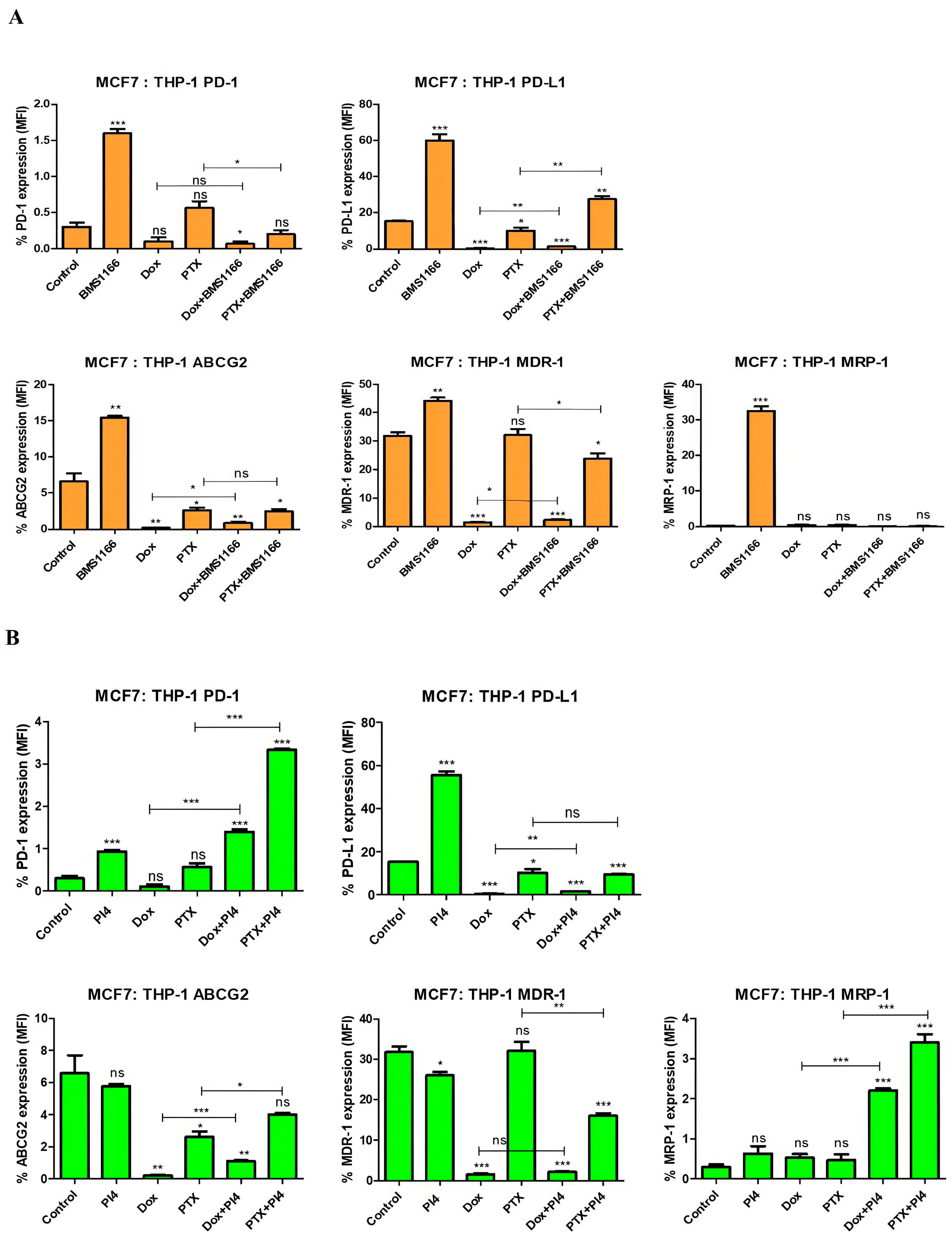
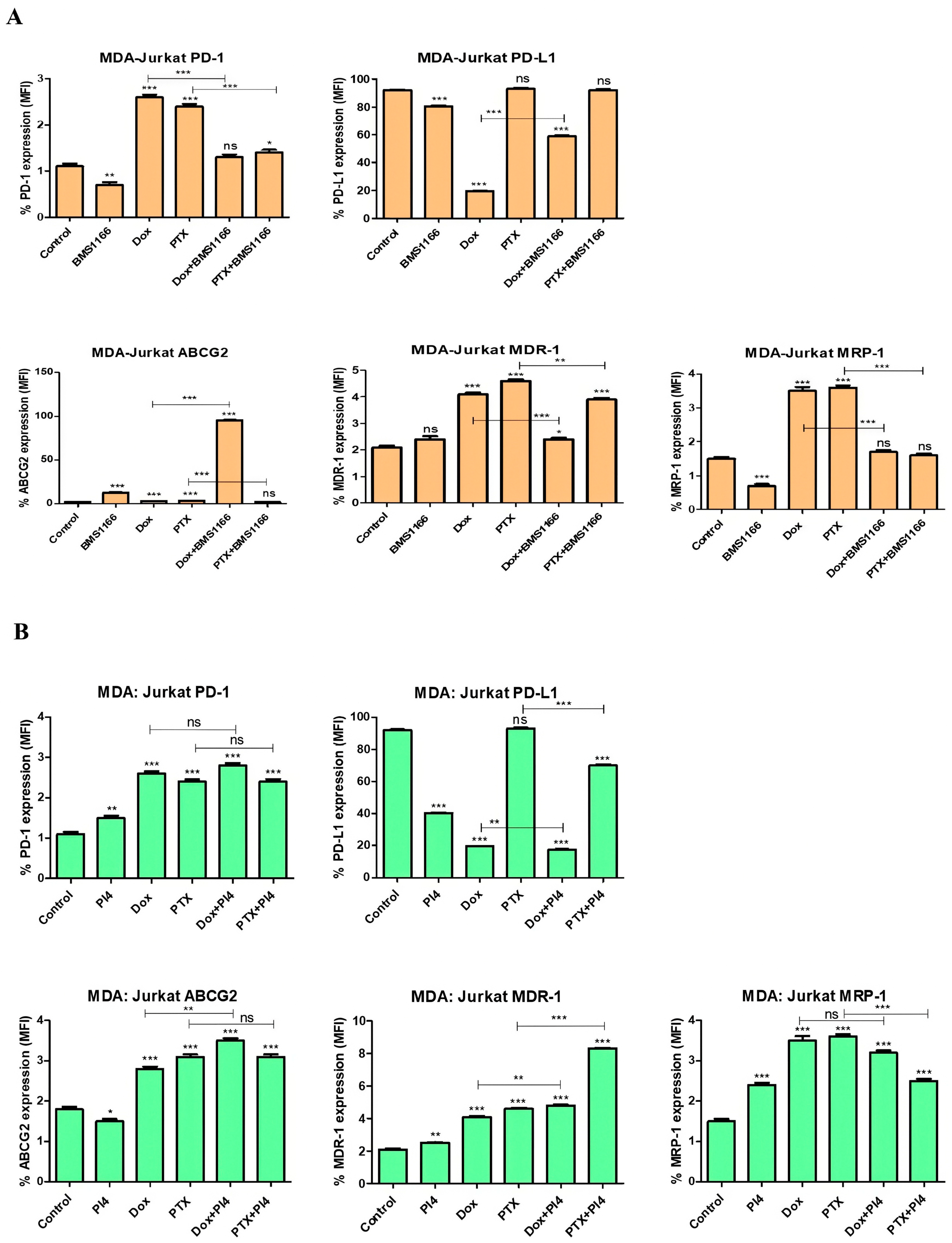
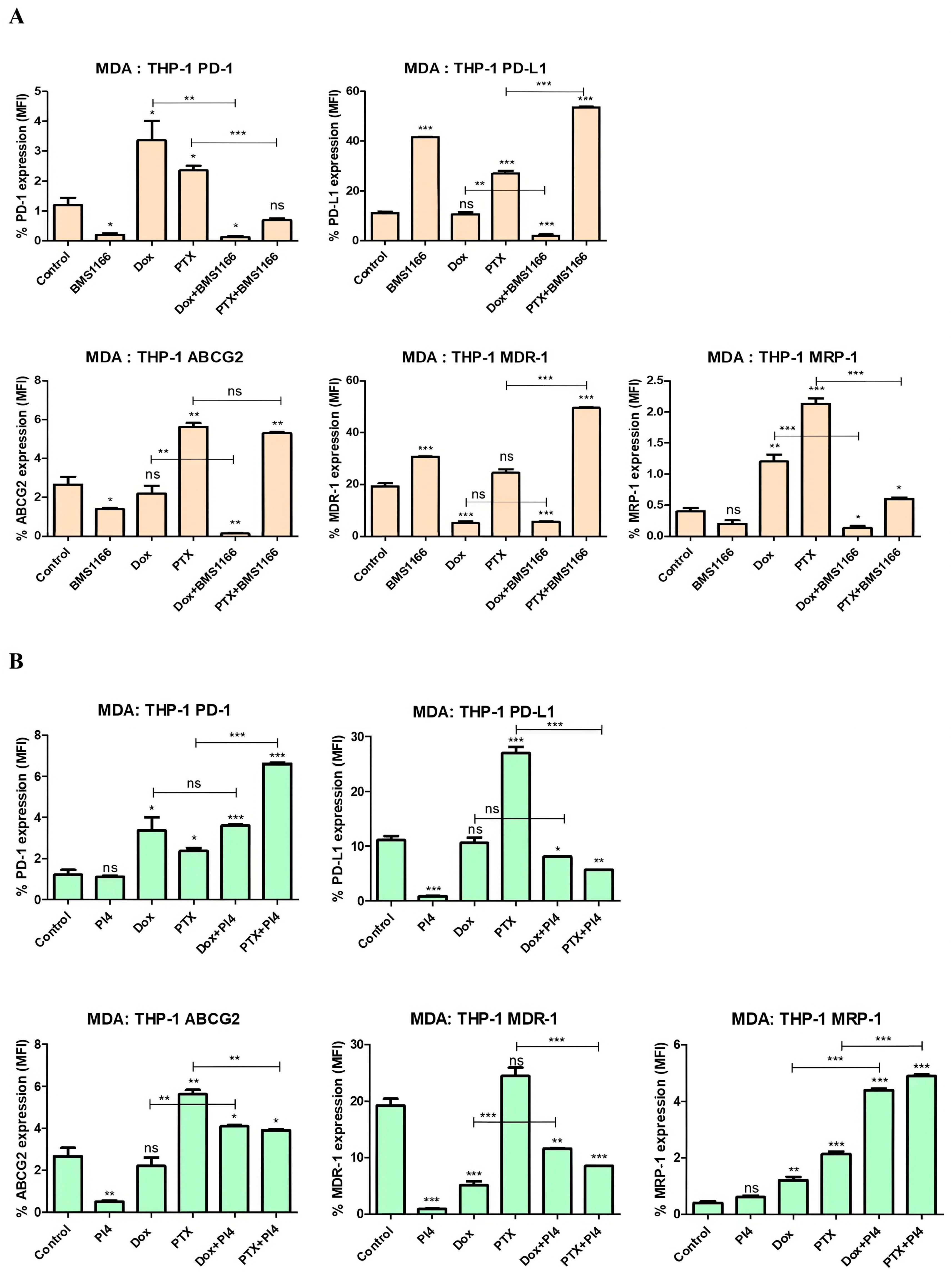
2.6. Drug Resistance and PD-1/PD-L1 Connection in Breast Cancer
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. In Vitro Co-Culture Model
4.3. Cell Viability Assay
4.4. Flow Cytometry
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2024, 74, 229–263. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Kong, D.; Liu, J.; Zhan, L.; Luo, L.; Zheng, W.; Zheng, Q.; Chen, C.; Sun, S. Breast cancer heterogeneity and its implication in personalized precision therapy. Exp. Hematol. Oncol. 2023, 12, 3. [Google Scholar] [CrossRef] [PubMed]
- Roskoski, R., Jr. Targeted and cytotoxic inhibitors used in the treatment of breast cancer. Pharmacol. Res. 2024, 210, 107534. [Google Scholar] [CrossRef] [PubMed]
- Smyth, M.J.; Ngiow, S.F.; Ribas, A.; Tang, M.W.L. Combination cancer immunotherapies tailored to the tumour microenvironment. Nat. Rev. Clin. Oncol. 2016, 13, 143–158. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.Y.W.; Tanaka, Y.; Iwasaki, M.; Gittis, A.G.; Su, H.P.; Mikami, B.; Okazaki, T.; Honjo, T.; Minato, N.; Garboczi, D.N. The PD-1/PD-L1 complex resembles the antigen-binding Fv domains of antibodies and T cell receptors. Proc. Natl. Acad. Sci. USA 2008, 105, 3011–3016. [Google Scholar] [CrossRef] [PubMed]
- Nallasamy, P.; Chava, S.; Verma, S.S.; Mishra, S.; Gorantla, S.; Coulter, D.W.; Byrareddy, S.N.; Batra, S.K.; Gupta, S.C.; Challagundla, K.B. PD-L1, inflammation, non-coding RNAs, and neuroblastoma: Immuno-oncology perspective. Semin. Cancer Biol. 2018, 52 Pt 2, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Gatalica, Z.; Snyder, C.; Maney, T.; Ghazalpour, A.; Holterman, D.A.; Xiao, N.; Overberg, P.; Rose, I.; Basu, G.D.; Vranic, S.; et al. Programmed cell death 1 (PD-1) and its ligand (PD-L1) in common cancers and their correlation with molecular cancer type. Cancer Epidemiol. Biomark. Prev. 2014, 23, 2965–2970. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wetherilt, C.S.; Krishnamurti, U.; Yang, J.; Ma, Y.; Styblo, T.M.; Meisel, J.L.; Peng, L.; Siddiqui, M.T.; Cohen, C.; et al. Stromal PD-L1 expression is associated with better disease-free survival in triple-negative breast cancer. Am. J. Clin. Pathol. 2016, 146, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Mittendorf, E.A.; Philips, A.V.; Meric-Bernstam, F.; Qiao, N.; Wu, Y.; Harrington, S.; Su, X.; Wang, Y.; Gonzalez-Angulo, A.M.; Akcakanat, A.; et al. PD-L1 expression in triple-negative breast cancer. Cancer Immunol. Res. 2014, 2, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Ghebeh, H.; Mohammed, S.; Al-Omair, A.; Qattant, A.; Lehe, C.; Al-Quadaihi, G.; Elkum, N.; Alshabanah, M.; Bin Amer, S.; Tulbah, A.; et al. The B7-H1 (PD-L1) T lymphocyte-inhibitory molecule is expressed in breast cancer patients with infiltrating ductal carcinoma: Correlation with important high-risk prognostic factors. Neoplasia 2006, 8, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Bartelt, R.R.; Cruz-Orcutt, N.; Collins, M.; Houtman, J.C. Comparison of T cell receptor-induced proximal signaling and downstream functions in immortalized and primary T cells. PLoS ONE 2009, 4, e5430. [Google Scholar] [CrossRef] [PubMed]
- Auwerx, J. The human leukemia cell line, THP-1: A multifacetted model for the study of monocyte-macrophage differentiation. Experientia 1991, 47, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Ribas, A.; Wolchok, J.D. Cancer immunotherapy using checkpoint blockade. Science 2018, 359, 1350–1355. [Google Scholar] [CrossRef] [PubMed]
- Guidotti, G.; Brambilla, L.; Rossi, D. Cell-penetrating peptides: From basic research to clinics. Trends Pharmacol. Sci. 2017, 38, 406–424. [Google Scholar] [CrossRef] [PubMed]
- Marqus, S.; Pirogova, E.; Piva, T.J. Evaluation of the use of therapeutic peptides for cancer treatment. J. Biomed. Sci. 2017, 24, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Zhan, M.M.; Hu, X.Q.; Liu, X.X.; Ruan, B.F.; Xu, J.; Liao, C. From monoclonal antibodies to small molecules: The development of inhibitors targeting the PD-1/PD-L1 pathway. Drug Discov. Today 2016, 21, 1027–1036. [Google Scholar] [CrossRef] [PubMed]
- Ai, L.; Chen, J.; Yan, H.; He, Q.; Luo, P.; Xu, Z.; Yang, X. Research Status and Outlook of PD-1/PD-L1 Inhibitors for Cancer Therapy. Drug Des. Dev. Ther. 2020, 14, 3625–3649. [Google Scholar] [CrossRef] [PubMed]
- Bailly, C.; Vergoten, G. Flurbiprofen as a biphenyl scaffold for the design of small molecules binding to PD-L1 protein dimer. Biochem. Pharmacol. 2020, 178, 114042. [Google Scholar] [CrossRef] [PubMed]
- Guzik, K.; Tomala, M.; Muszak, D.; Konieczny, M.; Hec, A.; Błaszkiewicz, U.; Pustuła, M.; Butera, R.; Dömling, A.; Holak, T.A. Development of the Inhibitors that Target the PD-1/PD-L1 Interaction-A Brief Look at Progress on Small Molecules, Peptides and Macrocycles. Molecules 2019, 24, 2071. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Quan, L.; Lyu, J.; He, Z.; Wang, X.; Meng, J.; Zhao, Z.; Zhu, L.; Liu, X.; Li, H. Discovery of peptide inhibitors targeting human programmed death 1 (PD-1) receptor. Oncotarget 2016, 7, 64967. [Google Scholar] [CrossRef] [PubMed]
- Majidi, M.; Safaee, S.; Amini, M.; Baghbanzadeh, A.; Hajiasgharzadeh, K.; Hashemzadeh, S.; Sandoghchian Shotorbani, S.; Mokhtarzadeh, A.; Baradaran, B. The effects of chemotherapeutic drugs on PD-L1 gene expression in breast cancer cell lines. Med. Oncol. 2021, 38, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Samanta, D.; Park, Y.; Ni, X.; Li, H.; Zahnow, C.A.; Gabrielson, E.; Pan, F.; Semenza, G.L. Chemotherapy induces enrichment of CD47+/CD73+/PDL1+ immune evasive triple- negative breast cancer cells. Proc. Natl. Acad. Sci. USA 2018, 115, E1239–E1248. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Chen, S.; Yuan, W.; Wang, H.; Chen, K.; Li, D.; Li, D. PD-1/PD-L1 interaction up-regulates MDR1/P-gp expression in breast cancer cells via PI3K/AKT and MAPK/ERK pathways. Oncotarget 2017, 8, 99901. [Google Scholar] [CrossRef] [PubMed]
- Taylor, N.M.; Manolaridis, I.; Jackson, S.M.; Kowal, J.; Stahlberg, H.; Locher, K.P. Structure of the human multidrug transporter ABCG2. Nature 2017, 546, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Sharom, F.J. The P-glycoprotein multidrug transporter. Essays Biochem. 2011, 50, 161–178. [Google Scholar] [CrossRef] [PubMed]
- Gillet, J.P.; Gottesman, M. Advances in the molecular detection of ABC transporters involved in multidrug resistance in cancer. Curr. Pharm. Biotechnol. 2011, 12, 686–692. [Google Scholar] [CrossRef] [PubMed]
- Gote, V.; Nookala, A.R.; Bolla, P.K. Drug resistance in metastatic breast cancer: Tumor targeted nanomedicine to the rescue. Int. J. Mol. Sci. 2021, 22, 4673. [Google Scholar] [CrossRef] [PubMed]
- Black, M.; Barsoum, I.B.; Truesdell, P.; Cotechini, T.; Macdonald-Goodfellow, S.K.; Petroff, M.; Siemens, D.; Koti, M.; Craig, A.W.B.; Graham, C.H. Activation of the PD-1/PD-L1 immune checkpoint confers tumor cell chemoresistance associated with increased metastasis. Oncotarget 2016, 7, 10557. [Google Scholar] [CrossRef] [PubMed]
- Nandi, D.; Sharma, D. Integrating immunotherapy with conventional treatment regime for breast cancer patients-an amalgamation of armamentarium. Front. Immunol. 2024, 15, 1477980. [Google Scholar] [CrossRef] [PubMed]
- Dijkstra, K.K.; Cattaneo, C.M.; Weeber, F.; Chalabi, M.; van de Haar, J.; Fanchi, L.F.; Slagter, M.; van der Velden, D.L.; Kaing, S.; Kelderman, S.; et al. Generation of tumor-reactive T cells by co-culture of peripheral blood lymphocytes and tumor organoids. Cell 2018, 174, 1586–1598. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Fan, X.; Deng, H.; Brezski, R.J.; Rycyzyn, M.; Jordan, R.E.; Strohl, W.R.; Zou, Q.; Zhang, N.; An, Z. Trastuzumab triggers phagocytic killing of high HER2 cancer cells in vitro and in vivo by interaction with Fcγ receptors on macrophages. J. Immunol. 2015, 194, 4379–4386. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Li, B.; Li, L.; Wang, G.; Li, Y.; Fu, R.; Ming, Y.; Ni, Y.; Wang, J.; Ye, G.; et al. The immunostimulative effect and mechanisms of a novel mouse anti-human PD-1 monoclonal antibody on Jurkat lymphocytic cells cocultured with hepatoma cells. OncoTargets Ther. 2020, 13, 12225. [Google Scholar] [CrossRef] [PubMed]
- Naba, N.M.; Tolay, N.; Erman, M.B.; Yazgan, A.S. Doxorubicin inhibits miR-140 expression and upregulates PD-L1 expression in HCT116 cells, opposite to its effects on MDA-MB-231 cells. Turk. J. Biol. 2020, 44, 15–23. [Google Scholar] [PubMed]
- Ghebeh, H.; Lehe, C.; Barhoush, E.; Al-Romaih, K.; Tulbah, A.; Al-Alwan, M.; Hendrayani, S.F.; Manogaran, P.; Alaiya, A.; Al-Tweigeri, T.; et al. Doxorubicin downregulates cell surface B7-H1 expression and upregulates its nuclear expression in breast cancer cells: Role of B7-H1 as an anti-apoptotic molecule. Breast Cancer Res. 2010, 12, R48. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Su, D.M.; Liang, M.; Fu, J. Chemopreventive agents induce programmed death-1-ligand 1 (PD-L1) surface expression in breast cancer cells and promote PD-L1-mediated T cell apoptosis. Mol. Immunol. 2008, 45, 1470–1476. [Google Scholar] [CrossRef] [PubMed]
- Pelekanou, V.; Carvajal-Hausdorf, D.E.; Altan, M.; Wasserman, B.; Carjaval-Hausdorf, D.E.; Wimberly, H.; Brown, J.; Lannin, D.; Pusztai, L.; Rimm, D.L. Effect of neoadjuvant chemotherapy on tumor-infiltrating lymphocytes and PD-L1 expression in breast cancer and its clinical significance. Breast Cancer Res. 2017, 19, 91. [Google Scholar] [CrossRef] [PubMed]
- Theodoulou, F.L.; Kerr, I.D. ABC transporter research: Going strong 40 years on. Biochem. Soc. Trans. 2015, 43, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Schinkel, A.H.; Jonker, J.W. Mammalian drug efflux transporters of the ATP binding cassette (ABC) family: An overview. Adv. Drug Deliv. Rev. 2012, 64, 138–153. [Google Scholar] [CrossRef]
- Martins-Gomes, C.; Silva, A.M. Natural products as a tool to modulate the activity and expression of multidrug resistance proteins of intestinal barrier. J. Xenobiotics 2023, 13, 172–192. [Google Scholar] [CrossRef] [PubMed]
- Konieczny, M.; Musielak, B.; Kocik, J.; Skalniak, L.; Sala, D.; Czub, M.; Magiera- Mularz, K.; Rodriguez, I.; Myrcha, M.; Steg, M.; et al. Di-bromo-based small-molecule inhibitors of the PD-1/PD-L1 immune checkpoint. J. Med. Chem. 2020, 63, 11271–11285. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xu, W.; Li, L.; Zhang, Z.; Lu, M.; Xia, X. Dual PI3K/mTOR Inhibitor BEZ235 combined with BMS-1166 Promoting Apoptosis in Colorectal Cancer. Int. J. Med. Sci. 2024, 21, 1814. [Google Scholar] [CrossRef] [PubMed]
- Patysheva, M.; Frolova, A.; Larionova, I.; Afanas’ev, S.; Tarasova, A.; Cherdyntseva, N.; Kzhyshkowska, J. Monocyte programming by cancer therapy. Front. Immunol. 2022, 13, 994319. [Google Scholar] [CrossRef] [PubMed]
- Antony, G.R.; Augustine, P.; Parambil, S.T.; Littleflower, A.B.; Kattoor, J.; Krishna, K.M.J.; Subhadradevi, L. Immunohistochemical expression of PD-L1 and MDR1 in breast tumors: Association with clinico-pathological parameters and treatment outcome. Clin. Exp. Med. 2023, 23, 859–869. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; To, K.K.W.; Chen, Z.S.; Fu, L. ABC transporters affects tumor immune microenvironment to regulate cancer immunotherapy and multidrug resistance. Drug Resist. Updates 2023, 66, 100905. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef] [PubMed]


| Cells/Co-Culture Groups | DOX | PTX |
|---|---|---|
| MCF-7 | 0.15 µM | 0.85 µM |
| MDA-MB-231 | 0.22 µM | 0.1 µM |
| Jurkat | 0.09 µM | 0.9 µM |
| THP-1 | 0.11 µM | 0.82 µM |
| MCF-7:Jurkat | 0.2 µM | 1.98 µM |
| MCF-7:THP-1 | 0.2 µM | 1.61 µM |
| MDA-MB-231:Jurkat | 0.2 µM | 0.6 µM |
| MDA-MB-231:THP-1 | 0.3 µM | 0.6 µM |
| Cells/Co-Culture Groups | BMS-1166 | PI4 |
|---|---|---|
| MCF-7 | 21.7 µM | >50 µg/mL |
| MCF-7:Jurkat | 28.7 µM | >50 µg/mL |
| MCF-7:THP-1 | 30 µM | >50 µg/mL |
| MDA-MB-231 | 17.6 µM | 10 µg/mL |
| MDA-MB-231:Jurkat | 24.7 µM | >50 µg/mL |
| MDA-MB-231:THP-1 | 24.5 µM | >50 µg/mL |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Özen Eroğlu, G.; Erol Bozkurt, A.; Yaylım, İ.; Kuruca, D.S. PD-1/PD-L1 Inhibitors and Chemotherapy Synergy: Impact on Drug Resistance and PD-L1 Expression in Breast Cancer-Immune Cell Co-Cultures. Int. J. Mol. Sci. 2025, 26, 6876. https://doi.org/10.3390/ijms26146876
Özen Eroğlu G, Erol Bozkurt A, Yaylım İ, Kuruca DS. PD-1/PD-L1 Inhibitors and Chemotherapy Synergy: Impact on Drug Resistance and PD-L1 Expression in Breast Cancer-Immune Cell Co-Cultures. International Journal of Molecular Sciences. 2025; 26(14):6876. https://doi.org/10.3390/ijms26146876
Chicago/Turabian StyleÖzen Eroğlu, Güneş, Ayşe Erol Bozkurt, İlhan Yaylım, and Dürdane Serap Kuruca. 2025. "PD-1/PD-L1 Inhibitors and Chemotherapy Synergy: Impact on Drug Resistance and PD-L1 Expression in Breast Cancer-Immune Cell Co-Cultures" International Journal of Molecular Sciences 26, no. 14: 6876. https://doi.org/10.3390/ijms26146876
APA StyleÖzen Eroğlu, G., Erol Bozkurt, A., Yaylım, İ., & Kuruca, D. S. (2025). PD-1/PD-L1 Inhibitors and Chemotherapy Synergy: Impact on Drug Resistance and PD-L1 Expression in Breast Cancer-Immune Cell Co-Cultures. International Journal of Molecular Sciences, 26(14), 6876. https://doi.org/10.3390/ijms26146876







