Unveiling Anti-Diabetic Potential of Baicalin and Baicalein from Baikal Skullcap: LC–MS, In Silico, and In Vitro Studies
Abstract
1. Introduction
2. Results
2.1. Identification and Screening of Effective Active Small Molecules in Baikal Skullcap
2.2. Identification of T2DM-Related Targets and Active Compounds in Baikal Skullcap
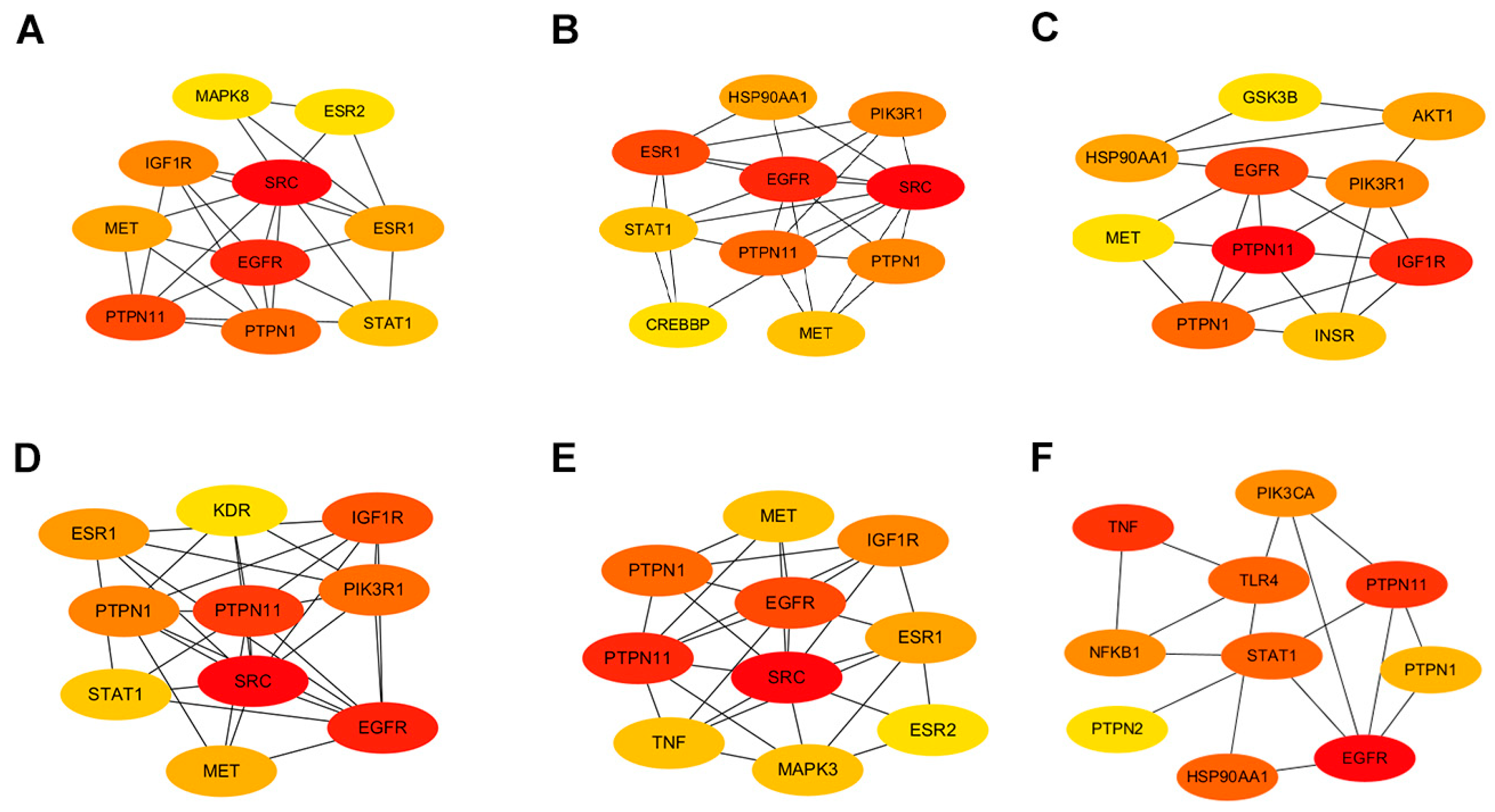
2.3. Stratification of Key Targets in Baikal Skullcap’s Anti-T2DM Mechanisms
2.4. Functional Enrichment of Baikal Skullcap’s Intersection with T2DM
2.5. Quantum Chemical Analysis of Baikal Skullcap Constituents
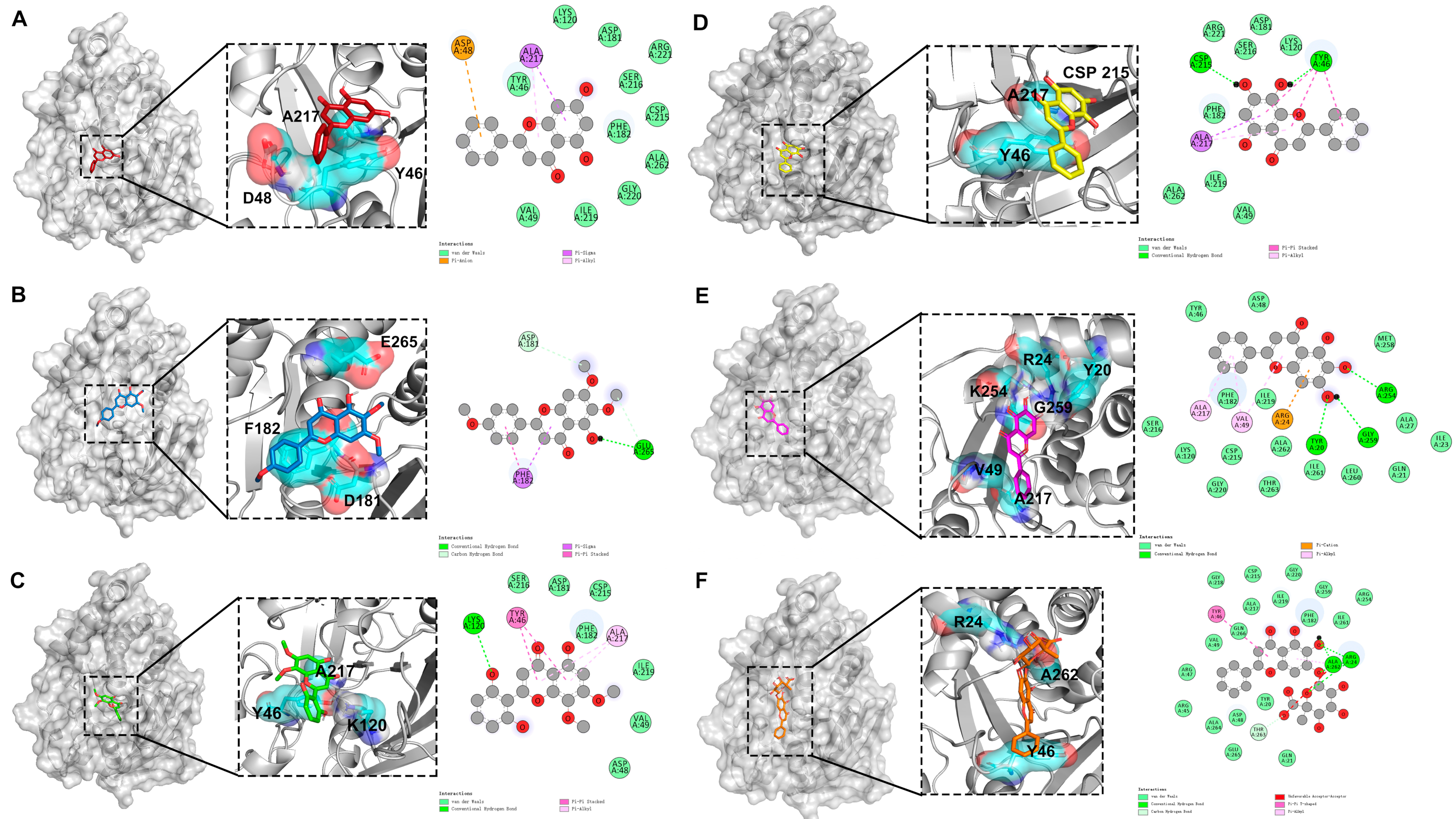
2.6. Molecular Docking Studies
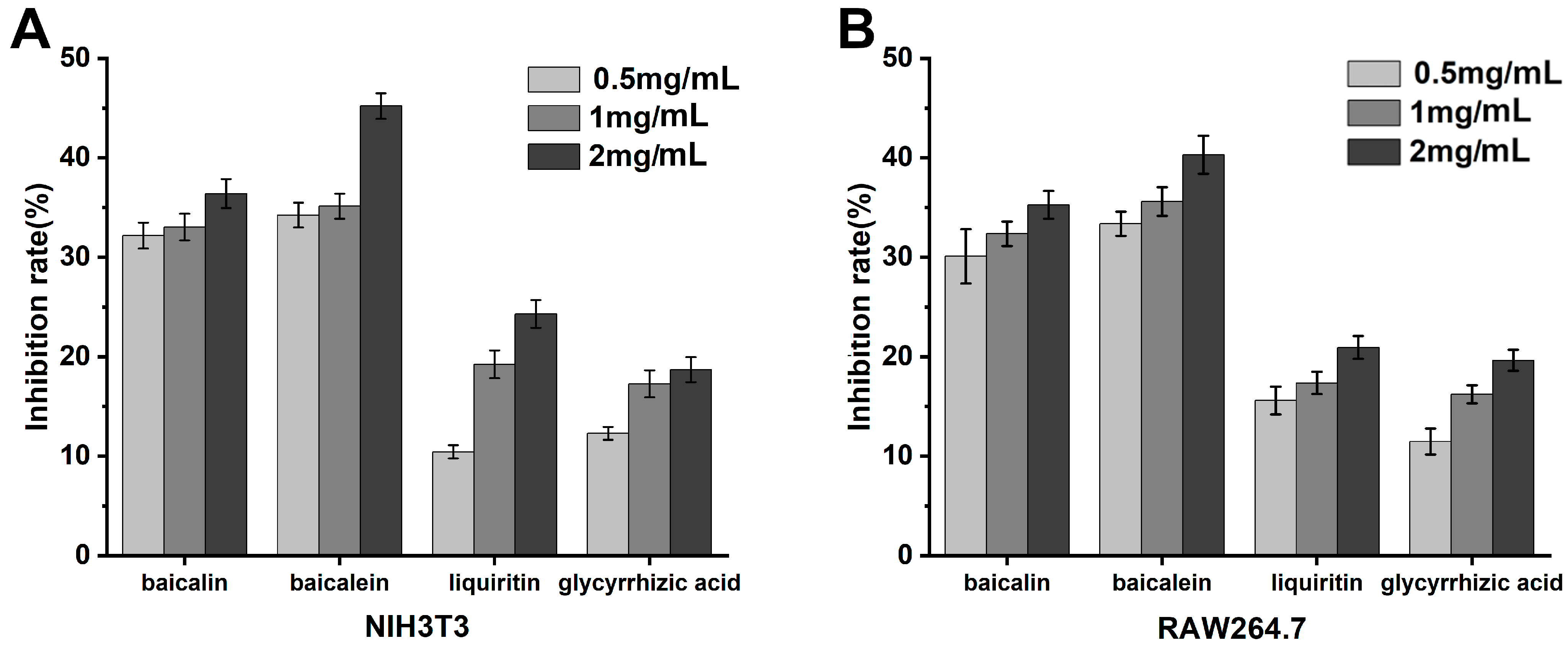
2.7. In Vitro Cellular Anti-Inflammatory Assays
3. Discussion
4. Materials and Methods
4.1. LC–MS Experiments
4.2. In Silico Experiments
4.2.1. Predictive Analysis of T2DM Potential Targets and Baikal Skullcap Component Interactions
4.2.2. Precise Construction of the Protein–Protein Interaction Network
4.2.3. Comprehensive Enrichment Analysis of GO and KEGG Pathways
4.2.4. Quantum Chemical Calculations and Molecular Docking of Active Components in Baikal Skullcap
4.3. In Vitro Cellular Anti-Inflammatory Assays
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef]
- Liu, G.; Li, Y.; Pan, A.; Hu, Y.; Chen, S.; Qian, F.; Rimm, E.B.; Manson, J.E.; Stampfer, M.J.; Giatsidis, G.; et al. Adherence to a Healthy Lifestyle in Association with Microvascular Complications Among Adults with Type 2 Diabetes. JAMA Netw. Open 2023, 6, e2252239. [Google Scholar] [CrossRef]
- Lv, C.; Sun, Y.; Zhang, Z.Y.; Aboelela, Z.; Qiu, X.; Meng, Z.-X. β-cell dynamics in type 2 diabetes and in dietary and exercise interventions. J. Mol. Cell Biol. 2022, 14, mjac046. [Google Scholar] [CrossRef] [PubMed]
- Marx, N.; Davies, M.J.; Grant, P.J.; Mathieu, C.; Petrie, J.R.; Cosentino, F.; Buse, J.B. Guideline recommendations and the positioning of newer drugs in type 2 diabetes care. Lancet Diabetes Endocrinol. 2021, 9, 46–52, reprinted in Lancet Diabetes Endocrinol. 2021, 9, e1. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.-H.; Lee, J.-H.; Kim, J.-W.; Cho, J.H.; Choi, Y.-H.; Ko, S.-H.; Zimmet, P.; Son, H.-Y. Epidemic obesity and type 2 diabetes in Asia. Lancet 2006, 368, 1681–1688. [Google Scholar] [CrossRef]
- Li, X.-Q.; Jia, S.-S.; Yuan, K.; Jin, S.-H. Hypoglycemic Effect of the N-Butanol Fraction of Torreya grandis Leaves on Type 2 Diabetes Mellitus in Rats through the Amelioration of Oxidative Stress and Enhancement of β-Cell Function. BioMed Res. Int. 2022, 2022, 5648896. [Google Scholar] [CrossRef]
- Gu, J.; Wu, Y.; Huang, W.; Fan, X.; Chen, X.; Zhou, B.; Lin, Z.; Feng, X. Effect of vitamin D on oxidative stress and serum inflammatory factors in the patients with type 2 diabetes. J. Clin. Lab. Anal. 2022, 36, e24430. [Google Scholar] [CrossRef]
- García-Aguilar, A.; Guillén, C. Targeting pancreatic beta cell death in type 2 diabetes by polyphenols. Front. Endocrinol. 2022, 13, 1052317. [Google Scholar] [CrossRef] [PubMed]
- Volpe, C.M.O.; Abreu, L.F.M.; Gomes, P.S.; Gonzaga, R.M.; Veloso, C.A.; Nogueira-Machado, J.A. The production of nitric oxide, IL-6, and TNF-alpha in palmitate-stimulated PBMNCs is enhanced through hyperglycemia in diabetes. Oxid. Med. Cell. Longev. 2014, 2014, 479587. [Google Scholar] [CrossRef]
- Shah, A.B.; Baiseitova, A.; Lee, G.; Kim, J.H.; Park, K.H. Analogues of Dihydroflavonol and Flavone as Protein Tyrosine Phosphatase 1B Inhibitors from the Leaves of Artocarpus elasticus. ACS Omega 2024, 9, 9053–9062. [Google Scholar] [CrossRef]
- Shah, A.B.; Yoon, S.; Kim, J.H.; Zhumanova, K.; Ban, Y.J.; Lee, K.W.; Park, K.H. Effectiveness of cyclohexyl functionality in ugonins from Helminthostachys zeylanica to PTP1B and α-glucosidase inhibitions. Int. J. Biol. Macromol. 2020, 165 (Pt B), 1822–1831. [Google Scholar] [CrossRef]
- Zhao, Q.; Chen, X.-Y.; Martin, C. Scutellaria baicalensis, the golden herb from the garden of Chinese medicinal plants. Sci. Bull. 2016, 61, 1391–1398. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.D.; Deng, Q.-G.; Chen, S.; Xiong, S.; Koop, D.; Tsukamoto, H. Hepatoprotective mechanisms of Yan-gan-wan. Hepatol. Res. 2005, 32, 202–212. [Google Scholar] [CrossRef] [PubMed]
- Scheck, A.C.; Perry, K.; Hank, N.C.; Clark, W.D. Anticancer activity of extracts derived from the mature roots of Scutellaria baicalensis on human malignant brain tumor cells. BMC Complement. Altern. Med. 2006, 6, 27. [Google Scholar] [CrossRef] [PubMed]
- Shan, B.; Cai, Y.-Z.; Brooks, J.D.; Corke, H. The in vitro antibacterial activity of dietary spice and medicinal herb extracts. Int. J. Food Microbiol. 2007, 117, 112–119. [Google Scholar] [CrossRef]
- Chen, J.; Fan, S.; Guo, J.; Yang, J.; Pan, L.; Xia, Y. Discovery of anticancer function of Febrifugine: Inhibition of cell proliferation, induction of apoptosis and suppression steroid synthesis in bladder cancer cells. Toxicol. Appl. Pharmacol. 2024, 484, 116878. [Google Scholar] [CrossRef]
- Tu, Y. Artemisinin—A Gift from Traditional Chinese Medicine to the World (Nobel Lecture). Angew. Chem. Int. Ed. Engl. 2016, 55, 10210–10226. [Google Scholar] [CrossRef]
- Thiruvengadam, M.; Khalil, A.A.; Rauf, A.; Alhumaydhi, F.A.; Aljohani, A.S.M.; Javed, M.S.; Khan, M.A.; Khan, I.A.; El-Esawi, M.A.; Bawazeer, S.; et al. Recent Developments and Anticancer Therapeutics of Paclitaxel: An Update. Curr. Pharm. Des. 2022, 28, 3363–3373. [Google Scholar] [CrossRef]
- Dempsey, D.A.; Vlot, A.C.; Wildermuth, M.C.; Klessig, D.F. Salicylic Acid biosynthesis and metabolism. Arab. Book 2011, 9, e0156. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.; Zheng, G.; Wang, C.; Chen, Z.; Mao, T.; Gao, J.; Yan, Y.; Chen, X.; Ji, X.; Yu, J.; et al. HIT 2.0: An enhanced platform for Herbal Ingredients’ Targets. Nucleic Acids Res. 2022, 50, D1238–D1243. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Cui, H.; Liu, K.; He, Q.; Fu, X.; Li, W.; Han, W. Exploring the anti-gout potential of sunflower receptacles alkaloids: A computational and pharmacological analysis. Comput. Biol. Med. 2024, 172, 108252. [Google Scholar] [CrossRef] [PubMed]
- Kao, T.-C.; Wu, C.-H.; Yen, G.-C. Bioactivity and potential health benefits of licorice. J. Agric. Food Chem. 2014, 62, 542–553. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liu, F.; Li, W.; Zhang, H.; Wang, W.; Liu, M.; Zhang, D.; Zhang, H. Study on the Composition and Mechanism of Santiao Decoction in Treating Insomnia Based on UPLC and Network Pharmacology and Molecular Docking Technology. Comb. Chem. High Throughput Screen. 2023. published online ahead of print. [Google Scholar] [CrossRef]
- Wang, M.; Yang, X.; Gao, Y.; Han, W. Computer-Aided Screening and Revealing Action Mechanism of Green Tea Polyphenols Intervention in Alzheimer’s Disease. Foods 2023, 12, 635. [Google Scholar] [CrossRef] [PubMed]
- Piñero, J.; Ramírez-Anguita, J.M.; Saüch-Pitarch, J.; Ronzano, F.; Centeno, E.; Sanz, F.; I Furlong, L. The DisGeNET knowledge platform for disease genomics: 2019 update. Nucleic Acids Res. 2020, 48, D845–D855. [Google Scholar] [CrossRef] [PubMed]
- Stelzer, G.; Rosen, N.; Plaschkes, I.; Zimmerman, S.; Twik, M.; Fishilevich, S.; Stein, T.I.; Nudel, R.; Lieder, I.; Mazor, Y.; et al. The GeneCards Suite: From Gene Data Mining to Disease Genome Sequence Analyses. Curr. Protoc. Bioinform. 2016, 54, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Whirl-Carrillo, M.; Huddart, R.; Gong, L.; Sangkuhl, K.; Thorn, C.F.; Whaley, R.; Klein, T.E. An Evidence-Based Framework for Evaluating Pharmacogenomics Knowledge for Personalized Medicine. Clin. Pharmacol. Ther. 2021, 110, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Bateman, A.; Martin, M.J.; Orchard, S.; Magrane, M.; Agivetova, R.; Ahmad, S.; Alpi, E.; Bowler-Barnett, E.H.; Britto, R.; Bursteinas, B.; et al. UniProt: The universal protein knowledgebase in 2021. Nucleic Acids Res. 2021, 49, D480–D489. [Google Scholar] [CrossRef]
- Keiser, M.J.; Roth, B.L.; Armbruster, B.N.; Ernsberger, P.; Irwin, J.J.; Shoichet, B.K. Relating protein pharmacology by ligand chemistry. Nat. Biotechnol. 2007, 25, 197–206. [Google Scholar] [CrossRef]
- Nickel, J.; Gohlke, B.-O.; Erehman, J.; Banerjee, P.; Rong, W.W.; Goede, A.; Dunkel, M.; Preissner, R. SuperPred: Update on drug classification and target prediction. Nucleic Acids Res. 2014, 42, W26–W31. [Google Scholar] [CrossRef]
- Gfeller, D.; Michielin, O.; Zoete, V. Shaping the interaction landscape of bioactive molecules. Bioinformatics 2013, 29, 3073–3079. [Google Scholar] [CrossRef] [PubMed]
- Szklarczyk, D.; Gable, A.L.; Lyon, D.; Junge, A.; Wyder, S.; Huerta-Cepas, J.; Simonovic, M.; Doncheva, N.T.; Morris, J.H.; Bork, P.; et al. STRING v11: Protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019, 47, D607–D613. [Google Scholar] [CrossRef] [PubMed]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef] [PubMed]
- Ashburner, M.; Ball, C.A.; Blake, J.A.; Botstein, D.; Butler, H.; Cherry, J.M.; Davis, A.P.; Dolinski, K.; Dwight, S.S.; Eppig, J.T.; et al. Gene ontology: Tool for the unification of biology. Nat. Genet. 2000, 25, 25–29. [Google Scholar] [CrossRef] [PubMed]
- The Gene Ontology Consortium. The Gene Ontology resource: Enriching a GOld mine. Nucleic Acids Res. 2021, 49, D325–D334. [Google Scholar] [CrossRef]
- Kanehisa, M.; Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef]
- Hosseini, S.; Ketabi, S.; Hasheminasab, G. QSAR study of antituberculosis activity of oxadiazole derivatives using DFT calculations. J. Recept. Signal Transduct. Res. 2022, 42, 503–511. [Google Scholar] [CrossRef]
- Lu, T.; Chen, F. Multiwfn: A multifunctional wavefunction analyzer. J. Comput. Chem. 2012, 33, 580–592. [Google Scholar] [CrossRef]
- Seeliger, D.; de Groot, B.L. Ligand docking and binding site analysis with PyMOL and Autodock/Vina. J. Comput. Aided. Mol. Des. 2010, 24, 417–422. [Google Scholar] [CrossRef]
- Berman, H.M.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.N.; Weissig, H.; Shindyalov, I.N.; Bourne, P.E. The Protein Data Bank. Nucleic Acids Res. 2000, 28, 235–242. [Google Scholar] [CrossRef]
- Reddy, M.S.; Ghadiyaram, C.; Panigrahi, S.K.; Krishnamurthy, N.R.; Hosahalli, S.; Chandrasekharappa, A.P.; Manna, D.; Badiger, S.E.; Dubey, P.K.; Mangamoori, L.N. X-ray structure of PTP1B in complex with a new PTP1B inhibitor. Protein Pept. Lett. 2014, 21, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Alotaibi, B.; El-Masry, T.A.; Elekhnawy, E.; A Mokhtar, F.; El-Seadawy, H.M.; A Negm, W. Studying the effects of secondary metabolites isolated from Cycas thouarsii R.Br. leaves on MDA-MB-231 breast cancer cells. Artif. Cells Nanomed. Biotechnol. 2024, 52, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Donadio, J.L.; Prado, S.B.R.D.; Soares, C.G.; Tamarossi, R.I.; Heidor, R.; Moreno, F.S.; Fabi, J.P. Ripe papaya pectins inhibit the proliferation of colon cancer spheroids and the formation of chemically induced aberrant crypts in rats colons. Carbohydr. Polym. 2024, 331, 121878. [Google Scholar] [CrossRef] [PubMed]



| Molecule Name | MW | OB (%) | DL |
|---|---|---|---|
| chrysin | 254.25 | 22.61 | 0.18 |
| baicalein | 270.25 | 33.52 | 0.21 |
| Salvigenin | 328.34 | 49.07 | 0.33 |
| 5,2′,6′-Trihydroxy-7,8-dimethoxyflavone | 330.31 | 45.05 | 0.33 |
| Norwogonin | 270.25 | 39.4 | 0.21 |
| Baicalin | 460.42 | 29.53 | 0.77 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, W.; Cui, H.; Liu, K.; Yang, X.; Xing, S.; Li, W. Unveiling Anti-Diabetic Potential of Baicalin and Baicalein from Baikal Skullcap: LC–MS, In Silico, and In Vitro Studies. Int. J. Mol. Sci. 2024, 25, 3654. https://doi.org/10.3390/ijms25073654
Zhao W, Cui H, Liu K, Yang X, Xing S, Li W. Unveiling Anti-Diabetic Potential of Baicalin and Baicalein from Baikal Skullcap: LC–MS, In Silico, and In Vitro Studies. International Journal of Molecular Sciences. 2024; 25(7):3654. https://doi.org/10.3390/ijms25073654
Chicago/Turabian StyleZhao, Wencheng, Huizi Cui, Kaifeng Liu, Xiaotang Yang, Shu Xing, and Wannan Li. 2024. "Unveiling Anti-Diabetic Potential of Baicalin and Baicalein from Baikal Skullcap: LC–MS, In Silico, and In Vitro Studies" International Journal of Molecular Sciences 25, no. 7: 3654. https://doi.org/10.3390/ijms25073654
APA StyleZhao, W., Cui, H., Liu, K., Yang, X., Xing, S., & Li, W. (2024). Unveiling Anti-Diabetic Potential of Baicalin and Baicalein from Baikal Skullcap: LC–MS, In Silico, and In Vitro Studies. International Journal of Molecular Sciences, 25(7), 3654. https://doi.org/10.3390/ijms25073654






