Functions of Phytochrome Interacting Factors (PIFs) in Adapting Plants to Biotic and Abiotic Stresses
Abstract
1. Introduction
2. PIFs Are Involved in Regulating Drought Stress Tolerance in Plants
3. Key Roles of PIFs in Regulating Low-Temperature Responses in Plants
4. Role of PIFs in High-Temperature Stress
5. Role of PIFs in Salt Stress
6. Role of PIFs in Biotic Stress
7. Summary and Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Silva, C.A., Jr.; D’Amico-Damião, V.; Carvalho, R.F. Phytochrome type B family: The abiotic stress responses signaller in plants. Ann. Appl. Biol. 2020, 178, 135–148. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lee, J.-H.; Park, C.-M. A Multifaceted Action of Phytochrome B in Plant Environmental Adaptation. Front. Plant Sci. 2021, 12, 659712. [Google Scholar] [CrossRef]
- Voitsekhovskaja, O.V. Phytochromes and Other (Photo)Receptors of Information in Plants. Russ. J. Plant Physiol. 2019, 66, 351–364. [Google Scholar] [CrossRef]
- Zheng, C.C.; Potter, D.; O’Neill, S.D. Phytochrome gene expression and phylogenetic analysis in the short-day plant Pharbitis nil (Convolvulaceae): Differential regulation by light and an endogenous clock. Am. J. Bot. 2009, 96, 1319–1336. [Google Scholar] [CrossRef]
- Sineshchekov, V.A. Two Distinct Molecular Types of Phytochrome A in Plants: Evidence of Existence and Implications for Functioning. Int. J. Mol. Sci. 2023, 24, 8139. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.-M.; Kim, S.-H.; Han, Y.-J.; Kim, J.-I. Regulation of Plant Photoresponses by Protein Kinase Activity of Phytochrome A. Int. J. Mol. Sci. 2023, 24, 2110. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Mroginski, M.A.; Lang, C.; Kopycki, J.; Gärtner, W.; Matysik, J.; Hughes, J. 3D Structures of Plant Phytochrome A as Pr and Pfr From Solid-State NMR: Implications for Molecular Function. Front. Plant Sci. 2018, 9, 498. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Burgie, E.S.; Gannam, Z.T.K.; Li, H.; Vierstra, R.D. Plant phytochrome B is an asymmetric dimer with unique signalling potential. Nature 2022, 604, 127–133. [Google Scholar] [CrossRef]
- Klose, C.; Nagy, F.; Schäfer, E. Thermal Reversion of Plant Phytochromes. Mol. Plant 2019, 13, 386–397. [Google Scholar] [CrossRef]
- Quail, P.H. Phytochrome-interacting factors. Semin. Cell Dev. Biol. 2000, 11, 457–466. [Google Scholar] [CrossRef]
- Leivar, P.; Quail, P.H. PIFs: Pivotal components in a cellular signaling hub. Trends Plant Sci. 2011, 16, 19–28. [Google Scholar] [CrossRef]
- Nakamura, Y.; Kato, T.; Yamashino, T.; Murakami, M.; Mizuno, T. Characterization of a Set of Phytochrome-Interacting Factor-Like bHLH Proteins in Oryza sativa. Biosci. Biotechnol. Biochem. 2007, 71, 1183–1191. [Google Scholar] [CrossRef]
- Rosado, D.; Gramegna, G.; Cruz, A.; Lira, B.S.; Freschi, L.; de Setta, N.; Rossi, M. Phytochrome Interacting Factors (PIFs) in Solanum lycopersicum: Diversity, Evolutionary History and Expression Profiling during Different Developmental Processes. PLoS ONE 2016, 11, e0165929. [Google Scholar] [CrossRef]
- Gao, Y.; Ren, X.; Qian, J.; Li, Q.; Tao, H.; Chen, J. The phytochrome-interacting family of transcription factors in maize (Zea mays L.): Identification, evolution, and expression analysis. Acta Physiol. Plant. 2019, 41, 8. [Google Scholar] [CrossRef]
- Sharma, A.; Samtani, H.; Sahu, K.; Sharma, A.K.; Khurana, J.P.; Khurana, P. Functions of Phytochrome-Interacting Factors (PIFs) in the regulation of plant growth and development: A comprehensive review. Int. J. Biol. Macromol. 2023, 244, 125234. [Google Scholar] [CrossRef]
- Zhuang, H.; Guo, Z.; Wang, J.; Chen, T. Genome-wide identification and comprehensive analysis of the phytochrome-interacting factor (PIF) gene family in wheat. PLoS ONE 2024, 19, e0296269. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.-Y.; Jia, M.-Z.; Wang, S.-N.; Han, S.; Jiang, J. Identification and characterization of cotton PHYTOCHROME-INTERACTING FACTORS in temperature-dependent flowering. J. Exp. Bot. 2023, 74, 3765–3780. [Google Scholar] [CrossRef] [PubMed]
- Saud, S.; Shi, Z.; Xiong, L.; Danish, S.; Datta, R.; Ahmad, I.; Fahad, S.; Banout, J. Recognizing the Basics of Phytochrome-Interacting Factors in Plants for Abiotic Stress Tolerance. Plant Stress 2021, 3, 100050. [Google Scholar] [CrossRef]
- Zheng, P.-F.; Wang, X.; Yang, Y.-Y.; You, C.-X.; Zhang, Z.-L.; Hao, Y.-J. Identification of Phytochrome-Interacting Factor Family Members and Functional Analysis of MdPIF4 in Malus domestica. Int. J. Mol. Sci. 2020, 21, 7350. [Google Scholar] [CrossRef] [PubMed]
- Huq, E.; Al-Sady, B.; Hudson, M.; Kim, C.; Apel, K.; Quail, P.H. PHYTOCHROME-INTERACTING FACTOR 1 Is a Critical bHLH Regulator of Chlorophyll Biosynthesis. Science 2004, 305, 1937–1941. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, X.-W.; Liu, X.; Zheng, P.-F.; Su, L.; Wang, G.-L.; Wang, X.-F.; Li, Y.-Y.; You, C.-X.; An, J.-P. Phytochrome interacting factor MdPIF7 modulates anthocyanin biosynthesis and hypocotyl growth in apple. Plant Physiol. 2022, 188, 2342–2363. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, M.; Wang, Y.; Zheng, X.; Zhang, H.; Zhang, L.; Tan, B.; Ye, X.; Wang, W.; Li, J.; et al. PpPIF8, a DELLA2-interacting protein, regulates peach shoot elongation possibly through auxin signaling. Plant Sci. 2022, 323, 111409. [Google Scholar] [CrossRef]
- Fernández-Milmanda, G.L.; Ballaré, C.L. Shade Avoidance: Expanding the Color and Hormone Palette. Trends Plant Sci. 2021, 26, 509–523. [Google Scholar] [CrossRef]
- Zhang, Y.; Pfeiffer, A.; Tepperman, J.M.; Dalton-Roesler, J.; Leivar, P.; Grandio, E.G.; Quail, P.H. Central clock components modulate plant shade avoidance by directly repressing transcriptional activation activity of PIF proteins. Proc. Natl. Acad. Sci. USA 2020, 117, 3261–3269. [Google Scholar] [CrossRef]
- Rehman, S.; Ahmad, Z.; Ramakrishnan, M.; Kalendar, R.; Zhuge, Q. Regulation of plant epigenetic memory in response to cold and heat stress: Towards climate resilient agriculture. Funct. Integr. Genom. 2023, 23, 298. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, N.; Ahmad, S.; Alghamdi, S.S.; Rehman, H.M.; Javed, M.A.; Tabassum, J.; Shao, G. CRISPR-Cas System, a Possible “Savior” of Rice Threatened by Climate Change: An Updated Review. Rice 2023, 16, 39. [Google Scholar] [CrossRef] [PubMed]
- Paik, I.; Kathare, P.K.; Kim, J.-I.; Huq, E. Expanding Roles of PIFs in Signal Integration from Multiple Processes. Mol. Plant 2017, 10, 1035–1046. [Google Scholar] [CrossRef]
- Bandurska, H. Drought Stress Responses: Coping Strategy and Resistance. Plants 2022, 11, 922. [Google Scholar] [CrossRef] [PubMed]
- Soma, F.; Takahashi, F.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Cellular Phosphorylation Signaling and Gene Expression in Drought Stress Responses: ABA-Dependent and ABA-Independent Regulatory Systems. Plants 2021, 10, 756. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Lu, M.; Wang, Y.; Wang, Y.; Liu, Z.; Chen, S. Response Mechanism of Plants to Drought Stress. Horticulturae 2021, 7, 50. [Google Scholar] [CrossRef]
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S.M.A. Plant drought stress: Effects, mechanisms and management. Agron. Sustain. Dev. 2009, 29, 185–212. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, W.; Liu, H.; Liu, P.; Li, Z. Genome-wide analysis of the soybean DREB gene family: Identification, genomic organization and expression profiles in response to drought stress. Plant Breed. 2020, 139, 1158–1167. [Google Scholar] [CrossRef]
- Yang, S.U.; Kim, H.; Kim, R.J.; Kim, J.; Suh, M.C. AP2/DREB Transcription Factor RAP2.4 Activates Cuticular Wax Biosynthesis in Arabidopsis Leaves Under Drought. Front. Plant Sci. 2020, 11, 895. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Song, X.; Chen, R.; Zhang, B.; Li, C.; Liang, Y.; Qiu, L.; Fan, Y.; Zhou, Z.; Zhou, H.; et al. Genome-Wide Analysis of the DREB Subfamily in Saccharum spontaneum Reveals Their Functional Divergence During Cold and Drought Stresses. Front. Genet. 2020, 10, 1326. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Ma, Y.; Wisniewski, M.; Cheng, Z.-M. Meta-analysis of the effect of overexpression of CBF/DREB family genes on drought stress response. Environ. Exp. Bot. 2017, 142, 1–14. [Google Scholar] [CrossRef]
- Wang, B.; Li, L.; Liu, M.; Peng, D.; Wei, A.; Hou, B.; Lei, Y.; Li, X. TaFDL2-1A confers drought stress tolerance by promoting ABA biosynthesis, ABA responses, and ROS scavenging in transgenic wheat. Plant J. 2022, 112, 722–737. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.-Y.; Du, Y.-T.; Fu, J.-D.; Yu, T.-F.; Wang, C.-T.; Chen, M.; Chen, J.; Ma, Y.-Z.; Xu, Z.-S. Wheat CBL-interacting protein kinase 23 positively regulates drought stress and ABA responses. BMC Plant Biol. 2018, 18, 93. [Google Scholar] [CrossRef]
- Mukherjee, A.; Dwivedi, S.; Bhagavatula, L.; Datta, S. Integration of light and ABA signaling pathways to combat drought stress in plants. Plant Cell Rep. 2023, 42, 829–841. [Google Scholar] [CrossRef]
- Gu, L.; Chen, P.; Yu, S. The cytochrome P450 gene GhCYP94C1 is involved in drought stress in upland cotton (Gossypium hirsutum L.). Czech J. Genet. Plant Breed. 2023, 59, 189–195. [Google Scholar] [CrossRef]
- Cordeiro, A.M.; Figueiredo, D.D.; Tepperman, J.; Borba, A.R.; Lourenço, T.; Abreu, I.A.; Ouwerkerk, P.B.; Quail, P.H.; Oliveira, M.M.; Saibo, N.J. Rice phytochrome-interacting factor protein OsPIF14 represses OsDREB1B gene expression through an extended N-box and interacts preferentially with the active form of phytochrome B. Biochim. Biophys. Acta (BBA)—Gene Regul. Mech. 2015, 1859, 393–404. [Google Scholar] [CrossRef]
- Kudo, M.; Kidokoro, S.; Yoshida, T.; Mizoi, J.; Todaka, D.; Fernie, A.R.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Double overexpression of DREB and PIF transcription factors improves drought stress tolerance and cell elongation in transgenic plants. Plant Biotechnol. J. 2016, 15, 458–471. [Google Scholar] [CrossRef] [PubMed]
- Todaka, D.; Nakashima, K.; Maruyama, K.; Kidokoro, S.; Osakabe, Y.; Ito, Y.; Matsukura, S.; Fujita, Y.; Yoshiwara, K.; Ohme-Takagi, M.; et al. Rice phytochrome-interacting factor-like protein OsPIL1 functions as a key regulator of internode elongation and induces a morphological response to drought stress. Proc. Natl. Acad. Sci. USA 2012, 109, 15947–15952. [Google Scholar] [CrossRef]
- Gao, Y.; Jiang, W.; Dai, Y.; Xiao, N.; Zhang, C.; Li, H.; Lu, Y.; Wu, M.; Tao, X.; Deng, D.; et al. A maize phytochrome-interacting factor 3 improves drought and salt stress tolerance in rice. Plant Mol. Biol. 2015, 87, 413–428. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Wu, M.; Zhang, M.; Jiang, W.; Ren, X.; Liang, E.; Zhang, D.; Zhang, C.; Xiao, N.; Li, Y.; et al. A maize phytochrome-interacting factors protein ZmPIF1 enhances drought tolerance by inducing stomatal closure and improves grain yield in Oryza sativa. Plant Biotechnol. J. 2018, 16, 1375–1387. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Wu, M.; Zhang, M.; Jiang, W.; Liang, E.; Zhang, D.; Zhang, C.; Xiao, N.; Chen, J. Roles of a maize phytochrome-interacting factors protein ZmPIF3 in regulation of drought stress responses by controlling stomatal closure in transgenic rice without yield penalty. 2018, 97, 311–323. Plant Mol. Biol. 2018, 97, 311–323. [Google Scholar] [CrossRef]
- Wang, X.-R.; Wang, Y.-H.; Jia, M.; Zhang, R.-R.; Liu, H.; Xu, Z.-S.; Xiong, A.-S. The phytochrome-interacting factor DcPIF3 of carrot plays a positive role in drought stress by increasing endogenous ABA level in Arabidopsis. Plant Sci. 2022, 322, 111367. [Google Scholar] [CrossRef]
- Qiu, J.-R.; Xiang, X.-Y.; Wang, J.-T.; Xu, W.-X.; Chen, J.; Xiao, Y.; Jiang, C.-Z.; Huang, Z. MfPIF1 of Resurrection Plant Myrothamnus flabellifolia Plays a Positive Regulatory Role in Responding to Drought and Salinity Stresses in Arabidopsis. Int. J. Mol. Sci. 2020, 21, 3011. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, Y.; Pan, X.; Li, B.; Yang, Q.; Yang, C.; Zhang, J.; Wu, F.; Yang, A.; Li, Y. PIF1, a phytochrome-interacting factor negatively regulates drought tolerance and carotenoids biosynthesis in tobacco. Int. J. Biol. Macromol. 2023, 247, 125693. [Google Scholar] [CrossRef]
- Wang, F.; Chen, X.; Dong, S.; Jiang, X.; Wang, L.; Yu, J.; Zhou, Y. Crosstalk of PIF4 and DELLA modulates CBF transcript and hormone homeostasis in cold response in tomato. Plant Biotechnol. J. 2019, 18, 1041–1055. [Google Scholar] [CrossRef]
- He, Y.; Li, Y.; Cui, L.; Xie, L.; Zheng, C.; Zhou, G.; Zhou, J.; Xie, X. Phytochrome B Negatively Affects Cold Tolerance by Regulating OsDREB1 Gene Expression through Phytochrome Interacting Factor-Like Protein OsPIL16 in Rice. Front. Plant Sci. 2016, 7, 1963. [Google Scholar] [CrossRef]
- Lee, C.-M.; Thomashow, M.F. Photoperiodic regulation of the C-repeat binding factor (CBF) cold acclimation pathway and freezing tolerance in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2012, 109, 15054–15059. [Google Scholar] [CrossRef]
- Jiang, B.; Shi, Y.; Peng, Y.; Jia, Y.; Yan, Y.; Dong, X.; Li, H.; Dong, J.; Li, J.; Gong, Z.; et al. Cold-Induced CBF–PIF3 Interaction Enhances Freezing Tolerance by Stabilizing the phyB Thermosensor in Arabidopsis. Mol. Plant 2020, 13, 894–906. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Shi, Y.; Zhang, X.; Xin, X.; Qi, L.; Guo, H.; Li, J.; Yang, S. PIF3 is a negative regulator of the CBF pathway and freezing tolerance in Arabidopsis. Proc. Natl. Acad. Sci. USA 2017, 114, E6695–E6702. [Google Scholar] [CrossRef]
- Pan, C.; Yang, D.; Zhao, X.; Liu, Y.; Li, M.; Ye, L.; Ali, M.; Yu, F.; Lamin-Samu, A.T.; Fei, Z.; et al. PIF4 negatively modulates cold tolerance in tomato anthers via temperature-dependent regulation of tapetal cell death. Plant Cell 2021, 33, 2320–2339. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Zhao, T.; Yin, Z.; Liu, J.; Cheng, Y.; Xu, J. The phytochrome-interacting transcription factor CsPIF8 contributes to cold tolerance in citrus by regulating superoxide dismutase expression. Plant Sci. 2020, 298, 110584. [Google Scholar] [CrossRef]
- Zheng, P.-F.; Yang, Y.-Y.; Zhang, S.; You, C.-X.; Zhang, Z.-L.; Hao, Y.-J. Identification and functional characterization of MdPIF3 in response to cold and drought stress in Malus domestica. Plant Cell Tissue Organ Cult. 2020, 144, 435–447. [Google Scholar] [CrossRef]
- Yang, J.; Qu, X.; Ji, L.; Li, G.; Wang, C.; Wang, C.; Zhang, Y.; Zheng, L.; Li, W.; Zheng, X. PIF4 Promotes Expression of HSFA2 to Enhance Basal Thermotolerance in Arabidopsis. Int. J. Mol. Sci. 2022, 23, 6017. [Google Scholar] [CrossRef]
- Kim, C.; Kim, S.J.; Jeong, J.; Park, E.; Oh, E.; Park, Y.-I.; Lim, P.O.; Choi, G. High Ambient Temperature Accelerates Leaf Senescence via PHYTOCHROME-INTERACTING FACTOR 4 and 5 in Arabidopsis. Mol. Cells 2020, 43, 645–661. [Google Scholar] [CrossRef] [PubMed]
- Koini, M.A.; Alvey, L.; Allen, T.; Tilley, C.A.; Harberd, N.P.; Whitelam, G.C.; Franklin, K.A. High Temperature-Mediated Adaptations in Plant Architecture Require the bHLH Transcription Factor PIF4. Curr. Biol. 2009, 19, 408–413. [Google Scholar] [CrossRef]
- Li, N.; Bo, C.; Zhang, Y.; Wang, L. PHYTOCHROME INTERACTING FACTORS PIF4 and PIF5 promote heat stress induced leaf senescence in Arabidopsis. J. Exp. Bot. 2021, 72, 4577–4589. [Google Scholar] [CrossRef]
- Sun, J.; Qi, L.; Li, Y.; Chu, J.; Li, C. PIF4–Mediated Activation of YUCCA8 Expression Integrates Temperature into the Auxin Pathway in Regulating Arabidopsis Hypocotyl Growth. PLoS Genet. 2012, 8, e1002594. [Google Scholar] [CrossRef]
- Franklin, K.A.; Lee, S.H.; Patel, D.; Kumar, S.V.; Spartz, A.K.; Gu, C.; Ye, S.; Yu, P.; Breen, G.; Cohen, J.D.; et al. PHYTOCHROME-INTERACTING FACTOR 4 (PIF4) regulates auxin biosynthesis at high temperature. Proc. Natl. Acad. Sci. USA 2011, 108, 20231–20235. [Google Scholar] [CrossRef]
- Lau, O.S.; Song, Z.; Zhou, Z.; Davies, K.A.; Chang, J.; Yang, X.; Wang, S.; Lucyshyn, D.; Tay, I.H.Z.; Wigge, P.A.; et al. Direct Control of SPEECHLESS by PIF4 in the High-Temperature Response of Stomatal Development. Curr. Biol. 2018, 28, 1273–1280.e3. [Google Scholar] [CrossRef]
- Mishra, D. Take it easy in the heat: Transcription factors PIF4 and TCP4 interplay to slow leaf growth. Plant Physiol. 2022, 190, 2074–2076. [Google Scholar] [CrossRef] [PubMed]
- Saini, K.; Dwivedi, A.; Ranjan, A. High temperature restricts cell division and leaf size by coordination of PIF4 and TCP4 transcription factors. Plant Physiol. 2022, 190, 2380–2397. [Google Scholar] [CrossRef] [PubMed]
- Sakuraba, Y.; Bülbül, S.; Piao, W.; Choi, G.; Paek, N. Arabidopsis EARLY FLOWERING3 increases salt tolerance by suppressing salt stress response pathways. Plant J. 2017, 92, 1106–1120. [Google Scholar] [CrossRef] [PubMed]
- Balazadeh, S.; Wu, A.; Mueller-Roeber, B. Salt-triggered expression of the ANAC092-dependent senescence regulon in Arabidopsis thaliana. Plant Signal. Behav. 2010, 5, 733–735. [Google Scholar] [CrossRef] [PubMed]
- Balazadeh, S.; Siddiqui, H.; Allu, A.D.; Matallana-Ramirez, L.P.; Caldana, C.; Mehrnia, M.; Zanor, M.-I.; Köhler, B.; Mueller-Roeber, B. A gene regulatory network controlled by the NAC transcription factor ANAC092/AtNAC2/ORE1 during salt-promoted senescence. Plant J. 2010, 62, 250–264. [Google Scholar] [CrossRef]
- Mo, W.; Tang, W.; Du, Y.; Jing, Y.; Bu, Q.; Lin, R. PHYTOCHROME-INTERACTING FACTOR-LIKE14 and SLENDER RICE1 Interaction Controls Seedling Growth under Salt Stress. Plant Physiol. 2020, 184, 506–517. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Han, R.; Yang, Y.; Liu, X.; Li, H.; Zhao, X.; Li, J.; Fu, H.; Huo, Y.; Sun, L.; et al. Phytochromes enhance SOS2-mediated PIF1 and PIF3 phosphorylation and degradation to promote Arabidopsis salt tolerance. Plant Cell 2023, 35, 2997–3020. [Google Scholar] [CrossRef]
- Han, R.; Ma, L.; Lv, Y.; Qi, L.; Peng, J.; Li, H.; Zhou, Y.; Song, P.; Duan, J.; Li, J.; et al. SALT OVERLY SENSITIVE2 stabilizes phytochrome-interacting factors PIF4 and PIF5 to promote Arabidopsis shade avoidance. Plant Cell 2023, 35, 2972–2996. [Google Scholar] [CrossRef]
- Yang, Y.; Guang, Y.; Wang, F.; Chen, Y.; Yang, W.; Xiao, X.; Luo, S.; Zhou, Y. Characterization of Phytochrome-Interacting Factor Genes in Pepper and Functional Analysis of CaPIF8 in Cold and Salt Stress. Front. Plant Sci. 2021, 12, 746517. [Google Scholar] [CrossRef]
- Huang, Z.; Tang, R.; Yi, X.; Xu, W.; Zhu, P.; Jiang, C.-Z. Overexpressing PhytochromeInteractingFactor 8 of Myrothamnus flabellifolia Enhanced Drought and Salt Tolerance in Arabidopsis. Int. J. Mol. Sci. 2022, 23, 8155. [Google Scholar] [CrossRef] [PubMed]
- Xiang, S.; Wu, S.; Zhang, H.; Mou, M.; Chen, Y.; Li, D.; Wang, H.; Chen, L.; Yu, D. The PIFs Redundantly Control Plant Defense Response against Botrytis cinerea in Arabidopsis. Plants 2020, 9, 1246. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, O.; Piqueras, R.; Sánchez-Serrano, J.J.; Solano, R. ETHYLENE RESPONSE FACTOR1 Integrates Signals from Ethylene and Jasmonate Pathways in Plant Defense. Plant Cell 2003, 15, 165–178. [Google Scholar] [CrossRef]
- Huang, L.; Zhang, J.; Lin, Z.; Yu, P.; Lu, M.; Li, N. The AP2/ERF transcription factor ORA59 regulates ethylene-induced phytoalexin synthesis through modulation of an acyltransferase gene expression. J. Cell. Physiol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Milmanda, G.L.; Crocco, C.D.; Reichelt, M.; Mazza, C.A.; Köllner, T.G.; Zhang, T.; Cargnel, M.D.; Lichy, M.Z.; Fiorucci, A.-S.; Fankhauser, C.; et al. A light-dependent molecular link between competition cues and defence responses in plants. Nat. Plants 2020, 6, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wu, X.; Xing, Q.; Zhao, Y.; Yu, B.; Ma, Y.; Wang, F.; Qi, H. PIF8-WRKY42-mediated salicylic acid synthesis modulates red light induced powdery mildew resistance in oriental melon. Plant Cell Environ. 2023, 46, 1726–1742. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, D.; Zhao, P.; Sun, Y.; Fang, R.-X.; Ye, J. Near-infrared light and PIF4 promote plant antiviral defense by enhancing RNA interference. Plant Commun. 2024, 5, 100644. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Katsura, K.; Maruyama, K.; Taji, T.; Kobayashi, M.; Seki, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Functional Analysis of Rice DREB1/CBF-type Transcription Factors Involved in Cold-responsive Gene Expression in Transgenic Rice. Plant Cell Physiol. 2006, 47, 141–153. [Google Scholar] [CrossRef]
- Li, J.; Sima, W.; Ouyang, B.; Wang, T.; Ziaf, K.; Luo, Z.; Liu, L.; Li, H.; Chen, M.; Huang, Y.; et al. Tomato SlDREB gene restricts leaf expansion and internode elongation by downregulating key genes for gibberellin biosynthesis. J. Exp. Bot. 2012, 63, 6407–6420. [Google Scholar] [CrossRef]
- Li, X.; Zhang, J.; Li, M.; Zhou, B.; Zhang, Q.; Wei, Q. Influence of six dwarfing interstocks on the ‘Fuji’ apple under drought stress. Indian J. Hortic. 2017, 74, 346–350. [Google Scholar] [CrossRef]
- Li, F.; Chen, G.; Xie, Q.; Zhou, S.; Hu, Z. Down-regulation of SlGT-26 gene confers dwarf plants and enhances drought and salt stress resistance in tomato. Plant Physiol. Biochem. 2023, 203, 108053. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.-J.; Cho, K.-C.; Hwang, O.-J.; Choi, Y.-S.; Shin, A.-Y.; Hwang, I.; Kim, J.-I. Overexpression of an Arabidopsis β-glucosidase gene enhances drought resistance with dwarf phenotype in creeping bentgrass. Plant Cell Rep. 2012, 31, 1677–1686. [Google Scholar] [CrossRef] [PubMed]
- Skirycz, A.; De Bodt, S.; Obata, T.; De Clercq, I.; Claeys, H.; De Rycke, R.; Andriankaja, M.; Van Aken, O.; Van Breusegem, F.; Fernie, A.R.; et al. Developmental Stage Specificity and the Role of Mitochondrial Metabolism in the Response of Arabidopsis Leaves to Prolonged Mild Osmotic Stress. Plant Physiol. 2009, 152, 226–244. [Google Scholar] [CrossRef] [PubMed]
- Peres, A.; Churchman, M.L.; Hariharan, S.; Himanen, K.; Verkest, A.; Vandepoele, K.; Magyar, Z.; Hatzfeld, Y.; Van Der Schueren, E.; Beemster, G.T.S.; et al. Novel Plant-specific Cyclin-dependent Kinase Inhibitors Induced by Biotic and Abiotic Stresses. J. Biol. Chem. 2007, 282, 25588–25596. [Google Scholar] [CrossRef] [PubMed]
- Aguirrezabal, L.; Bouchier-Combaud, S.; Radziejwoski, A.; Dauzat, M.; Cookson, S.J.; Granier, C. Plasticity to soil water deficit in Arabidopsis thaliana: Dissection of leaf development into underlying growth dynamic and cellular variables reveals invisible phenotypes. Plant Cell Environ. 2006, 29, 2216–2227. [Google Scholar] [CrossRef]
- Ding, Y.; Shi, Y.; Yang, S. Advances and challenges in uncovering cold tolerance regulatory mechanisms in plants. New Phytol. 2019, 222, 1690–1704. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Liu, D.; Chong, K. Cold signaling in plants: Insights into mechanisms and regulation. J. Integr. Plant Biol. 2018, 60, 745–756. [Google Scholar] [CrossRef]
- Wang, D.-Z.; Jin, Y.-N.; Ding, X.-H.; Wang, W.-J.; Zhai, S.-S.; Bai, L.-P.; Guo, Z.-F. Gene regulation and signal transduction in the ICE–CBF–COR signaling pathway during cold stress in plants. Biochemistry 2017, 82, 1103–1117. [Google Scholar] [CrossRef]
- Hwarari, D.; Guan, Y.; Ahmad, B.; Movahedi, A.; Min, T.; Hao, Z.; Lu, Y.; Chen, J.; Yang, L. ICE-CBF-COR Signaling Cascade and Its Regulation in Plants Responding to Cold Stress. Int. J. Mol. Sci. 2022, 23, 1549. [Google Scholar] [CrossRef]
- Mei, C.; Yang, J.; Mei, Q.; Jia, D.; Yan, P.; Feng, B.; Mamat, A.; Gong, X.; Guan, Q.; Mao, K.; et al. MdNAC104 positively regulates apple cold tolerance via CBF-dependent and CBF-independent pathways. Plant Biotechnol. J. 2023, 21, 2057–2073. [Google Scholar] [CrossRef] [PubMed]
- An, J.-P.; Yao, J.-F.; You, C.-X.; Wang, X.-F.; Hao, Y.-J. MdHY5 positively regulates cold tolerance via CBF-dependent and CBF-independent pathways in apple. J. Plant Physiol. 2017, 218, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Lin, Y.; Chen, C.; Pervaiz, T.; Wang, X.; Luo, H.; Fang, J.; Shangguan, L. Whole genome identification of CBF gene families and expression analysis in Vitis vinifera L. Czech J. Genet. Plant Breed. 2023, 59, 119–132. [Google Scholar] [CrossRef]
- Yahia, N.; Wani, S.H.; Kumar, V. CBF-Dependent and CBF-Independent Transcriptional Regulation of Cold Stress Responses in Plants. In Cold Tolerance in Plants: Physiological, Molecular and Genetic Perspectives; Wani, S.H., Herath, V., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 89–102. [Google Scholar]
- Shi, Y.; Ding, Y.; Yang, S. Molecular Regulation of CBF Signaling in Cold Acclimation. Trends Plant Sci. 2018, 23, 623–637. [Google Scholar] [CrossRef] [PubMed]
- Li, D.-D.; Xue, J.-S.; Zhu, J.; Yang, Z.-N. Gene Regulatory Network for Tapetum Development in Arabidopsis thaliana. Front. Plant Sci. 2017, 8, 1559. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Ouyang, S.; Zhao, Y.; Tie, L.; Shao, C.; Duan, H. Plant responses to high temperature and drought: A bibliometrics analysis. Front. Plant Sci. 2022, 13, 1052660. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Wu, S.; Li, C.; Quan, W.; Wang, A. Response Mechanisms of Woody Plants to High-Temperature Stress. Plants 2023, 12, 3643. [Google Scholar] [CrossRef]
- Grover, A.; Mittal, D.; Negi, M.; Lavania, D. Generating high temperature tolerant transgenic plants: Achievements and challenges. Plant Sci. 2013, 205–206, 38–47. [Google Scholar] [CrossRef]
- Lämke, J.; Brzezinka, K.; Bäurle, I. HSFA2 orchestrates transcriptional dynamics after heat stress in Arabidopsis thaliana. Transcription 2016, 7, 111–114. [Google Scholar] [CrossRef]
- Liu, X.; Liu, X.; Chen, H.; Chen, H.; Li, S.; Li, S.; Lecourieux, D.; Lecourieux, D.; Duan, W.; Duan, W.; et al. Natural variations of HSFA2 enhance thermotolerance in grapevine. Hortic. Res. 2022, 10, uhac250. [Google Scholar] [CrossRef]
- Hwang, G.; Park, J.; Kim, S.; Park, J.; Seo, D.; Oh, E. Overexpression of BBX18 Promotes Thermomorphogenesis Through the PRR5-PIF4 Pathway. Front. Plant Sci. 2021, 12, 782352. [Google Scholar] [CrossRef]
- Xu, Y.; Zhu, Z. PIF4 and PIF4-Interacting Proteins: At the Nexus of Plant Light, Temperature and Hormone Signal Integrations. Int. J. Mol. Sci. 2021, 22, 10304. [Google Scholar] [CrossRef]
- Lee, S.; Wang, W.; Huq, E. Spatial regulation of thermomorphogenesis by HY5 and PIF4 in Arabidopsis. Nat. Commun. 2021, 12, 3656. [Google Scholar] [CrossRef]
- Lee, S.; Paik, I.; Huq, E. SPAs promote thermomorphogenesis via regulating the phyB-PIF4 module in Arabidopsis. Development 2020, 147, dev189233. [Google Scholar] [CrossRef]
- Ma, D.; Li, X.; Guo, Y.; Chu, J.; Fang, S.; Yan, C.; Noel, J.P.; Liu, H. Cryptochrome 1 interacts with PIF4 to regulate high temperature-mediated hypocotyl elongation in response to blue light. Proc. Natl. Acad. Sci. USA 2015, 113, 224–229. [Google Scholar] [CrossRef]
- Zhai, H.; Xiong, L.; Li, H.; Lyu, X.; Yang, G.; Zhao, T.; Liu, J.; Liu, B. Cryptochrome 1 Inhibits Shoot Branching by Repressing the Self-Activated Transciption Loop of PIF4 in Arabidopsis. Plant Commun. 2020, 1, 100042. [Google Scholar] [CrossRef] [PubMed]
- Duek, P.D.; Elmer, M.V.; van Oosten, V.R.; Fankhauser, C. The Degradation of HFR1, a Putative bHLH Class Transcription Factor Involved in Light Signaling, Is Regulated by Phosphorylation and Requires COP1. Curr. Biol. 2004, 14, 2296–2301. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Jiang, F.; Zhou, R.; Lv, H.; Wen, J.; Cui, S.; Wu, Z. NADPH-H2O2 shows different functions in regulating thermotolerance under different high temperatures in Solanum pimpinellifolium L. Sci. Hortic. 2019, 261, 108997. [Google Scholar] [CrossRef]
- Bawa, G.; Feng, L.; Chen, G.; Chen, H.; Hu, Y.; Pu, T.; Cheng, Y.; Shi, J.; Xiao, T.; Zhou, W.; et al. Gibberellins and auxin regulate soybean hypocotyl elongation under low light and high-temperature interaction. Physiol. Plant. 2020, 170, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Wang, X.; Wang, X.; Mao, T. PHYTOCHROME INTERACTING FACTOR 4 regulates microtubule organization to mediate high temperature-induced hypocotyl elongation in Arabidopsis. Plant Cell 2023, 35, 2044–2061. [Google Scholar] [CrossRef]
- Tang, Y.; Lu, S.; Fang, C.; Liu, H.; Dong, L.; Li, H.; Su, T.; Li, S.; Wang, L.; Cheng, Q.; et al. Diverse flowering responses subjecting to ambient high temperature in soybean under short-day conditions. Plant Biotechnol. J. 2023, 21, 782–791. [Google Scholar] [CrossRef]
- Qadir, M.; Quillérou, E.; Nangia, V.; Murtaza, G.; Singh, M.; Thomas, R.; Drechsel, P.; Noble, A. Economics of salt-induced land degradation and restoration. Nat. Resour. Forum 2014, 38, 282–295. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Fujita, M. Plant Responses and Tolerance to Salt Stress: Physiological and Molecular Interventions. Int. J. Mol. Sci. 2022, 23, 4810. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zhang, Q.; Liu, M.; Zhou, H.; Ma, C.; Wang, P. Regulation of Plant Responses to Salt Stress. Int. J. Mol. Sci. 2021, 22, 4609. [Google Scholar] [CrossRef]
- Liu, Z.; Guo, C.; Wu, R.; Wang, J.; Zhou, Y.; Yu, X.; Zhang, Y.; Zhao, Z.; Liu, H.; Sun, S.; et al. Identification of the Regulators of Epidermis Development under Drought- and Salt-Stressed Conditions by Single-Cell RNA-Seq. Int. J. Mol. Sci. 2022, 23, 2759. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Shen, L.; Han, X.; He, G.; Fan, W.; Li, Y.; Yang, S.; Zhang, Z.; Yang, Y.; Jin, W.; et al. Phosphatidic acid–regulated SOS2 controls sodium and potassium homeostasis in Arabidopsis under salt stress. EMBO J. 2023, 42, e112401. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Li, J.; Wang, Y.; Liang, X.; Zhang, M.; Lu, M.; Guo, Y.; Qin, F.; Jiang, C. The classical SOS pathway confers natural variation of salt tolerance in maize. New Phytol. 2022, 236, 479–494. [Google Scholar] [CrossRef]
- Zhu, J.-K. Salt and drought stress signal transduction in plants. Annu. Rev. Plant Biol. 2002, 53, 247–273. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, Z.; Iqbal, M.S.; Hashem, A.; Abd_Allah, E.F.; Ansari, M.I. Plant Defense Responses to Biotic Stress and Its Interplay With Fluctuating Dark/Light Conditions. Front. Plant Sci. 2021, 12, 631810. [Google Scholar] [CrossRef] [PubMed]
- Saijo, Y.; Loo, E.P. Plant immunity in signal integration between biotic and abiotic stress responses. New Phytol. 2020, 225, 87–104. [Google Scholar] [CrossRef] [PubMed]
- Gidda, S.K.; Miersch, O.; Levitin, A.; Schmidt, J.; Wasternack, C.; Varin, L. Biochemical and Molecular Characterization of a Hydroxyjasmonate Sulfotransferase from Arabidopsis thaliana. J. Biol. Chem. 2003, 278, 17895–17900. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.-L.; Yao, J.; Mei, C.-S.; Tong, X.-H.; Zeng, L.-J.; Li, Q.; Xiao, L.-T.; Sun, T.-P.; Li, J.; Deng, X.-W.; et al. Plant hormone jasmonate prioritizes defense over growth by interfering with gibberellin signaling cascade. Proc. Natl. Acad. Sci. USA 2012, 109, E1192–E1200. [Google Scholar] [CrossRef] [PubMed]
- Yamashino, T.; Kitayama, M.; Mizuno, T. Transcription of ST2A Encoding A Sulfotransferase Family Protein That Is Involved in Jasmonic Acid Metabolism Is Controlled According to the Circadian Clock- and PIF4/PIF5-Mediated External Coincidence Mechanism in Arabidopsis thaliana. Biosci. Biotechnol. Biochem. 2013, 77, 2454–2460. [Google Scholar] [CrossRef] [PubMed]
- Alamholo, M.; Tarinejad, A. Molecular mechanism of drought stress tolerance in barley (Hordeum vulgare L.) via a combined analysis of the transcriptome data. Czech J. Genet. Plant Breed. 2023, 59, 76–94. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, X.; Wang, W.; Cai, G.; Bi, G.; Chen, S.; Sun, C.; Zhou, J. PIF3 is phosphorylated by MAPK to modulate plant immunity. New Phytol. 2023, 240, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Ballaré, C.L. Light Regulation of Plant Defense. Annu. Rev. Plant Biol. 2014, 65, 335–363. [Google Scholar] [CrossRef]
- Ranade, S.S.; Seipel, G.; Gorzsás, A.; García-Gil, M.R. Enhanced lignin synthesis and ecotypic variation in defense-related gene expression in response to shade in Norway spruce. Plant Cell Environ. 2022, 45, 2671–2681. [Google Scholar] [CrossRef]
- Fiorucci, A.-S.; Michaud, O.; Schmid-Siegert, E.; Trevisan, M.; Petrolati, L.A.; Ince, Y.; Fankhauser, C. Shade suppresses wound-induced leaf repositioning through a mechanism involving PHYTOCHROME KINASE SUBSTRATE (PKS) genes. PLoS Genet. 2022, 18, e1010213. [Google Scholar] [CrossRef]
- Sng, B.J.R.; Van Vu, K.; Choi, I.K.Y.; Chin, H.J.; Jang, I.-C. LONG HYPOCOTYL IN FAR-RED 1 mediates a trade-off between growth and defence under shade in Arabidopsis. J. Exp. Bot. 2023, 74, 3560–3578. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; Pierik, R.; Van Wees, S.C.M. Different shades of JAZ during plant growth and defense. New Phytol. 2014, 204, 261–264. [Google Scholar] [CrossRef]
- Ballaré, C.L.; Austin, A.T. Recalculating growth and defense strategies under competition: Key roles of photoreceptors and jasmonates. J. Exp. Bot. 2019, 70, 3425–3434. [Google Scholar] [CrossRef]
- Li, K.; Yu, R.; Fan, L.-M.; Wei, N.; Chen, H.; Deng, X.W. DELLA-mediated PIF degradation contributes to coordination of light and gibberellin signalling in Arabidopsis. Nat. Commun. 2016, 7, 11868. [Google Scholar] [CrossRef] [PubMed]
- Leone, M.; Keller, M.M.; Cerrudo, I.; Ballaré, C.L. To grow or defend? Low red: Far-red ratios reduce jasmonate sensitivity in Arabidopsis seedlings by promoting DELLA degradation and increasing JAZ10 stability. New Phytol. 2014, 204, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Davière, J.-M.; de Lucas, M.; Prat, S. Transcriptional factor interaction: A central step in DELLA function. Curr. Opin. Genet. Dev. 2008, 18, 295–303. [Google Scholar] [CrossRef]
- De Lucas, M.; Davière, J.-M.; Rodríguez-Falcón, M.; Pontin, M.; Iglesias-Pedraz, J.M.; Lorrain, S.; Fankhauser, C.; Blazquez, M.A.; Titarenko, E.; Prat, S. A molecular framework for light and gibberellin control of cell elongation. Nature 2008, 451, 480–484. [Google Scholar] [CrossRef]
- Cerrudo, I.; Keller, M.M.; Cargnel, M.D.; Demkura, P.V.; de Wit, M.; Patitucci, M.S.; Pierik, R.; Pieterse, C.M.; Ballaré, C.L. Low Red/Far-Red Ratios Reduce Arabidopsis Resistance to Botrytis cinerea and Jasmonate Responses via a COI1-JAZ10-Dependent, Salicylic Acid-Independent Mechanism. Plant Physiol. 2012, 158, 2042–2052. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Qian, W.; Hua, J. Temperature Modulates Plant Defense Responses through NB-LRR Proteins. PLoS Pathog. 2010, 6, e1000844. [Google Scholar] [CrossRef]
- Pham, T.A.; Hwang, S.-Y. High temperatures reduce nutrients and defense compounds against generalist Spodoptera litura F. in Rorippa dubia. Arthropod-Plant Interact. 2020, 14, 333–344. [Google Scholar] [CrossRef]
- Zhang, Y.; Goritschnig, S.; Dong, X.; Li, X. A Gain-of-Function Mutation in a Plant Disease Resistance Gene Leads to Constitutive Activation of Downstream Signal Transduction Pathways in suppressor of npr1-1, constitutive 1. Plant Cell 2003, 15, 2636–2646. [Google Scholar] [CrossRef]
- Gangappa, S.N.; Berriri, S.; Kumar, S.V. PIF4 Coordinates Thermosensory Growth and Immunity in Arabidopsis. Curr. Biol. 2016, 27, 243–249. [Google Scholar] [CrossRef]
- Edgerton, M.D. Increasing Crop Productivity to Meet Global Needs for Feed, Food, and Fuel. Plant Physiol. 2009, 149, 7–13. [Google Scholar] [CrossRef]
- Guo, T.; Lin, H.-X. Creating future crops: A revolution for sustainable agriculture. J. Genet. Genom. 2021, 48, 97–101. [Google Scholar] [CrossRef]
- Tian, Z.; Wang, J.; Li, J.; Han, B. Designing future crops: Challenges and strategies for sustainable agriculture. Plant J. 2020, 105, 1165–1178. [Google Scholar] [CrossRef] [PubMed]
- Lesk, C.; Rowhani, P.; Ramankutty, N. Influence of extreme weather disasters on global crop production. Nature 2016, 529, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Han, P.; Wang, C.; Li, F.; Li, M.; Nie, J.; Xu, M.; Feng, H.; Xu, L.; Jiang, C.; Guan, Q.; et al. Valsa mali PR1-like protein modulates an apple valine-glutamine protein to suppress JA signaling-mediated immunity. Plant Physiol. 2024. [Google Scholar] [CrossRef] [PubMed]
- Manasse, R.S. Ecological Risks of Transgenic Plants: Effects of Spatial Dispersion on Gene Flow. Ecol. Appl. 1992, 2, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Leivar, P.; Monte, E. PIFs: Systems Integrators in Plant Development. Plant Cell 2014, 26, 56–78. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Oh, E. PIF4 Integrates Multiple Environmental and Hormonal Signals for Plant Growth Regulation in Arabidopsis. Mol. Cells 2016, 39, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Hwang, G.; Zhu, J.-Y.; Lee, Y.K.; Kim, S.; Nguyen, T.T.; Kim, J.; Oh, E. PIF4 Promotes Expression of LNG1 and LNG2 to Induce Thermomorphogenic Growth in Arabidopsis. Front. Plant Sci. 2017, 8, 1320. [Google Scholar] [CrossRef]
- Foreman, J.; Johansson, H.; Hornitschek, P.; Josse, E.; Fankhauser, C.; Halliday, K.J. Light receptor action is critical for maintaining plant biomass at warm ambient temperatures. Plant J. 2010, 65, 441–452. [Google Scholar] [CrossRef]
- Jiang, Y.; Yang, C.; Huang, S.; Xie, F.; Xu, Y.; Liu, C.; Li, L. The ELF3-PIF7 Interaction Mediates the Circadian Gating of the Shade Response in Arabidopsis. iScience 2019, 22, 288–298. [Google Scholar] [CrossRef]
- Ashraf, M.F.; Hou, D.; Hussain, Q.; Imran, M.; Pei, J.; Ali, M.; Shehzad, A.; Anwar, M.; Noman, A.; Waseem, M.; et al. Entailing the Next-Generation Sequencing and Metabolome for Sustainable Agriculture by Improving Plant Tolerance. Int. J. Mol. Sci. 2022, 23, 651. [Google Scholar] [CrossRef] [PubMed]
- Depuydt, T.; Vandepoele, K. Multi-omics network-based functional annotation of unknown Arabidopsis genes. Plant J. 2021, 108, 1193–1212. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Y.; Doudna, J.A. CRISPR technology: A decade of genome editing is only the beginning. Science 2023, 379, eadd8643. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Guo, W.; Le, L.; Yu, J.; Wu, Y.; Li, D.; Wang, Y.; Wang, H.; Lu, X.; Qiao, H.; et al. Integration of high-throughput phenotyping, GWAS, and predictive models reveals the genetic architecture of plant height in maize. Mol. Plant 2023, 16, 354–373. [Google Scholar] [CrossRef]
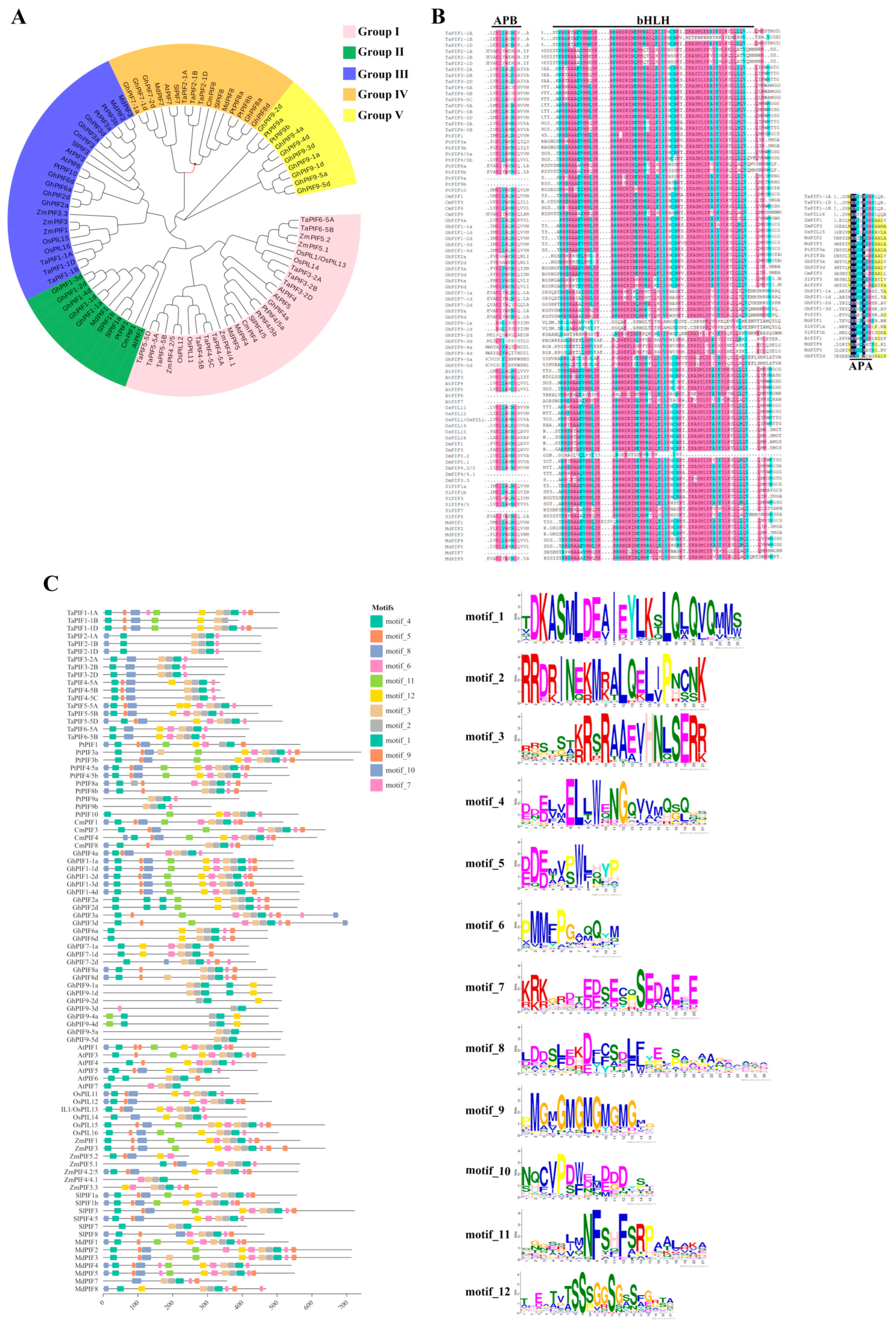
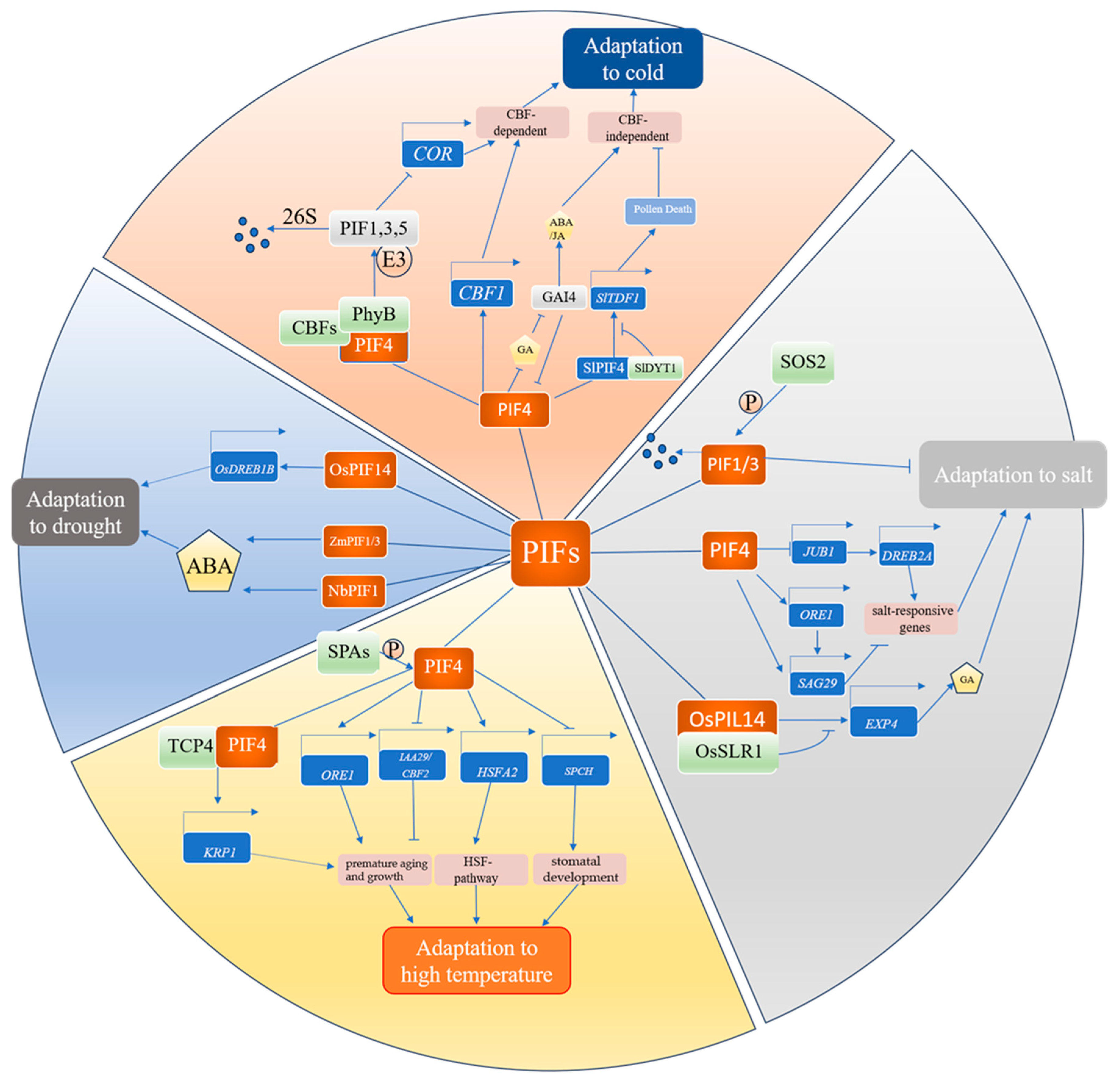
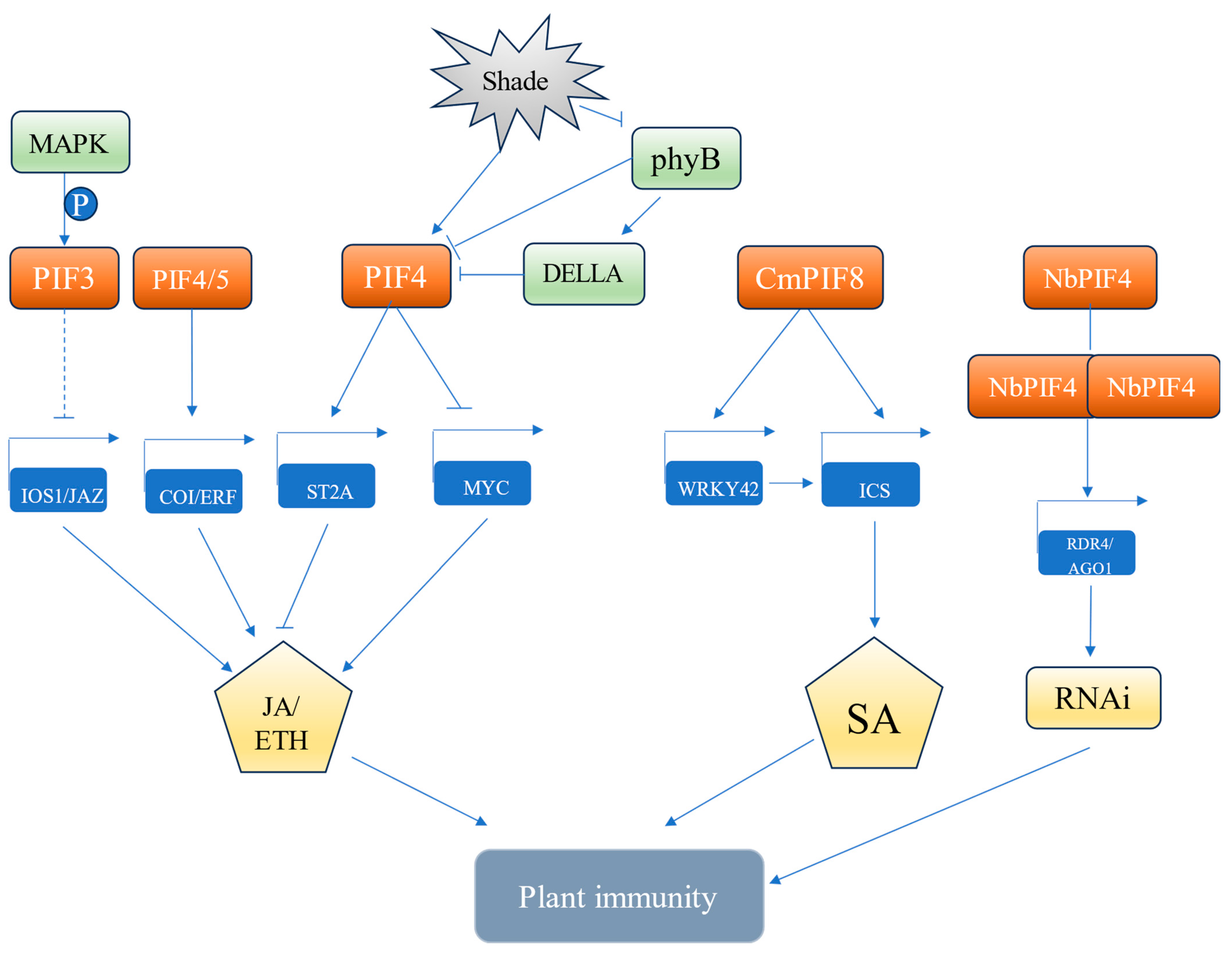
| Stress Conditions | PIF TFs | Associated Responses | References |
|---|---|---|---|
| Drought | OsPIF14, OsPIL1, | DREB-mediated pathway | [40,41,42] |
| ZmPIF1, ZmPIF3, DcPIF3, MfPIF1, NbPIF1/NtPIF1 | ABA signaling pathways | [43,44,45,46,47,48] | |
| Low temperature | SlPIF4, OsPIL16, AtPIF1, AtPIF3, AtPIF4, AtPIF5, AtPIF7 | CBF-dependent regulatory pathways | [49,50,51,52,53] |
| SlPIF4, CsPIF8, MdPIF3 | CBF-independent regulatory pathways | [49,54,55,56] | |
| High temperature | AtPIF4 | HSF-mediated heat stress response pathway | [57] |
| AtPIF4, AtPIF5 | Premature aging and growth caused by high temperature | [58,59,60,61,62] | |
| AtPIF4 | Stomatal development | [63,64,65] | |
| Salt | AtPIF4, OsPIL14 | Transcriptional regulation of salt-responsive genes | [66,67,68,69] |
| AtPIF1/3, AtPIF4/5 | SOS pathway | [70,71] | |
| MfPIF1/8, CaPIF8 | GA signaling pathways | [47,72,73] | |
| Biotic stress | AtPIF3, AtPIF4, AtPIF5 | JA/ETH | [74,75,76,77] |
| CmPIF8 | SA | [78] | |
| NbPIF4 | RNAi | [79] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Z.-Y.; Ma, N.; Zhang, F.-J.; Li, L.-Z.; Li, H.-J.; Wang, X.-F.; Zhang, Z.; You, C.-X. Functions of Phytochrome Interacting Factors (PIFs) in Adapting Plants to Biotic and Abiotic Stresses. Int. J. Mol. Sci. 2024, 25, 2198. https://doi.org/10.3390/ijms25042198
Li Z-Y, Ma N, Zhang F-J, Li L-Z, Li H-J, Wang X-F, Zhang Z, You C-X. Functions of Phytochrome Interacting Factors (PIFs) in Adapting Plants to Biotic and Abiotic Stresses. International Journal of Molecular Sciences. 2024; 25(4):2198. https://doi.org/10.3390/ijms25042198
Chicago/Turabian StyleLi, Zhao-Yang, Ning Ma, Fu-Jun Zhang, Lian-Zhen Li, Hao-Jian Li, Xiao-Fei Wang, Zhenlu Zhang, and Chun-Xiang You. 2024. "Functions of Phytochrome Interacting Factors (PIFs) in Adapting Plants to Biotic and Abiotic Stresses" International Journal of Molecular Sciences 25, no. 4: 2198. https://doi.org/10.3390/ijms25042198
APA StyleLi, Z.-Y., Ma, N., Zhang, F.-J., Li, L.-Z., Li, H.-J., Wang, X.-F., Zhang, Z., & You, C.-X. (2024). Functions of Phytochrome Interacting Factors (PIFs) in Adapting Plants to Biotic and Abiotic Stresses. International Journal of Molecular Sciences, 25(4), 2198. https://doi.org/10.3390/ijms25042198






