Current Insights in Prolactin Signaling and Ovulatory Function
Abstract
1. Introduction
2. Prolactin (PRL)
2.1. Structure
2.2. Prolactin Receptor (PRLR)
2.2.1. PRLR Structure
2.2.2. PRLR Signaling
3. Ovulation
| Class/Stage of the Follicle | Expression of Selected Genes | ||
|---|---|---|---|
| Granulosa Cells | Oocyte | Theca Cells | |
| Primordial | 3βHSD, ALK3, BMPRII, Erβ, KITLG, StAR, WTI | ALK3, ALK6, BMP6, BMPRII, C-kit, Erβ, GDF9, GJA4, TGFBR3 | – |
| Primary | βB-activin, ActRIIB, ALK6, AMH, AMHRII, FSH-R, GJA1, IGFR1 | BMP15, FIGα | – |
| Small preantral | ALK5, FSRP, FST, TGFBR3 | – | ActRIIB, ALK3, ALK5, ALK6?, BMPRII, FSRP, IGFR1, TGF-β1, TGF-β2, TGFBR3, TGFBR11 |
| Large preantral | AR, ERα, InhA | – | 3βHSD, ARErβ, CYP17A1, IGF2, LHR, PR, SF1, StAR |
3.1. Hormonal Regulation of Ovulation
The General Characteristics of the Main Endocrine Hormones Involved in the Menstrual Cycle and Ovulation (See also Table 2 for the Schematic Structure)
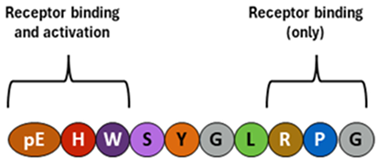 Gonadotropin-releasing hormone (GnRH) | 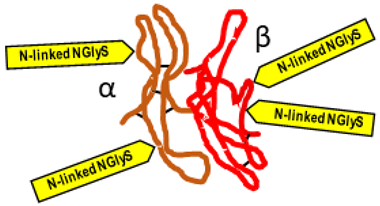 Follicle-stimulating hormone (FSH) | 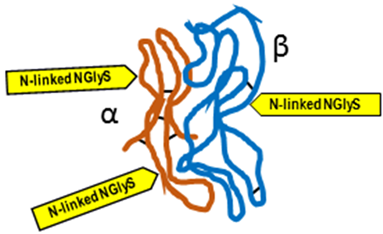 Luteinizing hormone (LH) |
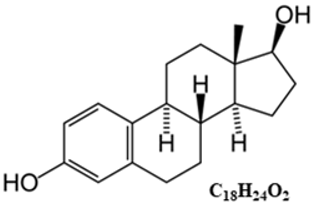 Estradiol (E2) | 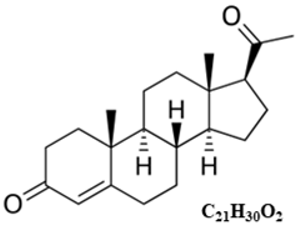 Progesterone (P4) |  Inhibins |
3.2. Mechanism of Follicle Rupture during Ovulation
3.3. Ovulatory Disorders
3.3.1. PRL and Ovulatory Disorders
PRL and the Release of Gonadotropins
PRL–Kisspeptin Interaction
4. Concluding Remarks
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 17-OHP | 17-hydroxyprogesterone |
| 3β-HSD | 3β-hydroxysteroid dehydrogenase |
| 5-HT | serotonin (5-hydroxytryptamine) |
| aa | amino acid count |
| ACTH | adrenocorticotropic hormone |
| AKT | protein kinase B |
| APC | antigen-presenting cells |
| ARC | arcuate nucleus (caudal region of the hypothalamus) |
| BAT | brown adipose tissue |
| BSs, BS1, BS2 | binding sites, binding site 1, binding site 2, respectively |
| Box-1, Box-2 | the proline-rich and hydrophobic regions in the intracellular domain of cytokine receptor 1 and 2, respectively |
| cAMP | cyclic adenosine monophosphate |
| C-C | carbon-carbon bond |
| CNS | central nervous system |
| D1, D2 | the two fibronectin type III domains of the prolactin receptor |
| DA | dopamine |
| DYN | dynorphin |
| E1, E2, E3, E4 | estrone, estradiol, estriol, and estetrol, respectively |
| ECR | extracellular region of receptor |
| EPOR | erythropoietin receptor |
| ER-α | estrogen receptor-α |
| ERK1/2 | extracellular signal-regulated kinase ½ |
| FAK | focal adhesion kinase |
| FIGO | International Federation of Gynecology and Obstetrics |
| FSH | follicle-stimulating hormone |
| FSHR | follicle-stimulating hormone receptor |
| GABA | gamma-aminobutyric acid |
| GC | granulosa cells |
| GDM | gestational diabetes mellitus |
| GDP, GTP | guanosine diphosphate and guanosine triphosphate, respectively |
| GH | growth hormone |
| GnRH | gonadotropin-releasing hormone (gonadoliberin) |
| GPER | G protein-coupled estrogen-receptor |
| GRB2 | growth factor receptor-bound protein 2 |
| hCG | human chorionic gonadotropin |
| HETE | hydroxyeicosatetraenoic acid |
| hGLC | human granulosa cells |
| HPG axis | hypothalamic–pituitary–gonadal axis |
| hPL | human placental lactogen (also called human chorionic somatotropin—hCS) |
| HPO | hypothalamic–pituitary–ovarian axis |
| HRT | hormonal replacement therapy |
| ICR | intracellular (cytoplasmic) region of receptor |
| icv | intracerebroventricular |
| IL-2R | interleukin-2 receptor |
| InhA | inhibin A (also marked as αβA) |
| InhB | inhibin B (also marked as αβB) |
| IRS | insulin receptor substrate |
| JAK2 | Janus kinase 2 |
| KISS-1R | kisspeptin receptor (also known as GPR54) |
| KNDγ neurons | kisspeptin/neurokinin B/dynorphin neurons |
| KP | kisspeptin |
| LH | luteinizing hormone |
| LTH | lactotrophs (lactotropic cells) |
| MAOIs | monoamine oxidase inhibitors |
| MAPK | mitogen-activated protein kinase |
| MBH | mediobasal region of the hypothalamus |
| ME | median eminence of the hypothalamus |
| mTOR | mammalian target of rapamycin (serine-threonine protein kinase) |
| NFPAs | non-functioning pituitary adenomas |
| NKB | neurokinin B |
| N-linked NGlyS | N-linked glycosylation sites in human proteins |
| NPFFR1 | neuropeptide FF receptor 1 |
| P4 | progesterone |
| PCOS | polycystic ovary syndrome |
| pE | pyroglutamate (pyroglutamic acid) |
| PI3 | phosphoinositide 3-kinase |
| PIH | pregnancy-induced hypertension |
| Pit-1 | transcription factor, a member of the POU (Pit-Oct-Unc) homeodomain protein family |
| POA | preoptic area (rostral region of the hypothalamus) |
| PRFs | prolactin-releasing factors |
| PRL | prolactin |
| PRLBP | prolactin binding protein |
| PRLR | prolactin receptor (a member of the class I cytokine receptor family) |
| PRLR-LF | long form of prolactin receptor |
| pSTAT5 | phosphorylated signal transducer and activator of transcription 5 |
| Ras/Raf | Ras/Raf kinases |
| SER | smooth endoplasmic reticulum |
| SHP2 | Src homology 2 (SH2) domain |
| SMARCA3 | SWI/SNF-related matrix-associated actin-dependent regulator of chromatin subfamily A, member 3 |
| SOS | son of sevenless, refers to a set of genes encoding guanine nucleotide exchange factors that act on the Ras subfamily of small GTPases |
| SRC | Src family kinases |
| SSRIs | selective serotonin reuptake inhibitors |
| STAT5 | signal transducer and activator of transcription 5 |
| STATs | signal transducer and activator of transcription proteins |
| TCA | tricyclic antidepressants |
| TGF-β | transforming growth factor-beta |
| TIDA neurons | tuberoinfundibular dopamine neurons |
| TMR/TMD | transmembrane region/transmembrane domain of receptor |
| TpoR | thrombopoietin receptor (also known as MPL) |
| TRH | thyrotropin-releasing hormone |
| TSH | thyroid-stimulating hormone |
| VIP | vasoactive intestinal peptide |
| VMAT2 | vesicular monoamine transporter type-2 |
| WHO | World Health Organization |
| WSXWS | a conserved amino acid sequence (WS motif) in prolactin receptor |
References
- Stricker, P.; Grüter, F. Action du lobe antérieur de l’hypophyse sur la montée laiteuse. CR Soc Biol. 1928, 99, 1978–1980. [Google Scholar]
- Flückiger, E.W.; del Pozo, E.; von Werder, K. Prolactin. Physiology, Pharmacology and Clinical Findings; Monographs on Endocrinology 23; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1982. [Google Scholar] [CrossRef]
- Riddle, O.; Braucher, P.F. Control of the special secretion of the crop gland in pigeons by an anterior pituitary hormone. Am. J. Physiol. 1931, 97, 617–625. [Google Scholar] [CrossRef]
- Riddle, O.; Bates, R.W.; Dykshorn, S.W. The preparation, identification and assay of prolactin–a hormone of the anterior pituitary. Am. J. Physiol. 1933, 105, 191–216. [Google Scholar] [CrossRef]
- Freeman, M.E.; Kanyicska, B.; Lerant, A.; Nagy, G. Prolactin: Structure, function, and regulation of secretion. Physiol. Rev. 2000, 80, 1523–1631. [Google Scholar] [CrossRef]
- Bernard, V.; Young, J.; Binart, N. Prolactin—A pleiotropic factor in health and disease. Nat. Rev. Endocrinol. 2019, 15, 356–365. [Google Scholar] [CrossRef]
- Al-Chalabi, M.; Bass, A.N.; Alsalman, I. Physiology, Prolactin. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK507829/ (accessed on 3 January 2024).
- Grattan, D.R. 60 years of neuroendocrinology: The hypothalamo-prolactin axis. J. Endocrinol. 2015, 226, T101–T122. [Google Scholar] [CrossRef]
- Atıcı, Ö.K.; Govindrajan, N.; Lopetegui-González, I.; Shemanko, C.S. Prolactin: A hormone with diverse functions from mammary gland development to cancer metastasis. Semin. Cell Dev. Biol. 2021, 114, 159–170. [Google Scholar] [CrossRef]
- Bachelot, A.; Binart, N. Reproductive role of prolactin. Reproduction 2007, 133, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Bouilly, J.; Sonigo, C.; Auffret, J.; Gibori, G.; Binart, N. Prolactin signaling mechanisms in ovary. Mol. Cell Endocrinol. 2012, 356, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Donato, J., Jr.; Frazão, R. Interactions between prolactin and kisspeptin to control reproduction. Arch. Endocrinol. Metab. 2016, 60, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Basini, G.; Baioni, L.; Bussolati, S.; Grolli, S.; Grasselli, F. Prolactin is a potential physiological modulator of swine ovarian follicle function. Regul. Pept. 2014, 189, 22–30. [Google Scholar] [CrossRef]
- Holesh, J.E.; Bass, A.N.; Lord, M. Physiology, Ovulation. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Laron, Z. The growth hormone-prolactin relationship: A neglected issue. Pediatr. Endocrinol. Rev. 2011, 9, 546–548. [Google Scholar]
- Ben-Jonathan, N.; Hugo, E. Prolactin (PRL) in adipose tissue: Regulation and functions. In Recent Advances in Prolactin Research; Advances in Experimental Medicine and Biology; Springer: Cham, Switzerland, 2015; Volume 846, pp. 1–35. [Google Scholar] [CrossRef]
- Brandebourg, T.; Hugo, E.; Ben-Jonathan, N. Adipocyte prolactin: Regulation of release and putative functions. Diabetes Obes. Metab. 2007, 9, 464–476. [Google Scholar] [CrossRef] [PubMed]
- Macotela, Y.; Triebel, J.; Clapp, C. Time for a New Perspective on Prolactin in Metabolism. Trends Endocrinol. Metab. 2020, 31, 276–286. [Google Scholar] [CrossRef]
- Pirchio, R.; Graziadio, C.; Colao, A.; Pivonello, R.; Auriemma, R.S. Metabolic effects of prolactin. Front. Endocrinol. 2022, 13, 1015520. [Google Scholar] [CrossRef] [PubMed]
- Breves, J.P.; Popp, E.E.; Rothenberg, E.F.; Rosenstein, C.W.; Maffett, K.M.; Guertin, R.R. Osmoregulatory actions of prolactin in the gastrointestinal tract of fishes. Gen. Comp. Endocrinol. 2020, 298, 113589. [Google Scholar] [CrossRef] [PubMed]
- Foitzik, K.; Krause, K.; Conrad, F.; Nakamura, M.; Funk, W.; Paus, R. Human scalp hair follicles are both a target and a source of prolactin, which serves as an autocrine and/or paracrine promoter of apoptosis-driven hair follicle regression. Am. J. Pathol. 2006, 168, 748–756. [Google Scholar] [CrossRef] [PubMed]
- Langan, E.A.; Ramot, Y.; Hanning, A.; Poeggeler, B.; Bíró, T.; Gaspar, E.; Funk, W.; Griffiths, C.E.; Paus, R. Thyrotropin-releasing hormone and oestrogen differentially regulate prolactin and prolactin receptor expression in female human skin and hair follicles in vitro. Br. J. Dermatol. 2010, 162, 1127–1131. [Google Scholar] [CrossRef] [PubMed]
- Torner, L. Actions of Prolactin in the Brain: From Physiological Adaptations to Stress and Neurogenesis to Psychopathology. Front. Endocrinol. 2016, 7, 25. [Google Scholar] [CrossRef]
- Borba, V.V.; Zandman-Goddard, G.; Shoenfeld, Y. Prolactin and Autoimmunity. Front. Immunol. 2018, 9, 73. [Google Scholar] [CrossRef]
- Rasmi, Y.; Jalali, L.; Khalid, S.; Shokati, A.; Tyagi, P.; Ozturk, A.; Nasimfar, A. The effects of prolactin on the immune system, its relationship with the severity of COVID-19, and its potential immunomodulatory therapeutic effect. Cytokine 2023, 169, 156253. [Google Scholar] [CrossRef]
- Flores-Espinosa, P.; Méndez, I.; Irles, C.; Olmos-Ortiz, A.; Helguera-Repetto, C.; Mancilla-Herrera, I.; Ortuño-Sahagún, D.; Goffin, V.; Zaga-Clavellina, V. Immunomodulatory role of decidual prolactin on the human fetal membranes and placenta. Front. Immunol. 2023, 14, 1212736. [Google Scholar] [CrossRef]
- Goffin, V. Prolactin receptor targeting in breast and prostate cancers: New insights into an old challenge. Pharmacol. Ther. 2017, 179, 111–126. [Google Scholar] [CrossRef]
- Standing, D.; Dandawate, P.; Anant, S. Prolactin receptor signaling: A novel target for cancer treatment—Exploring anti-PRLR signaling strategies. Front. Endocrinol. 2023, 13, 1112987. [Google Scholar] [CrossRef] [PubMed]
- Ben-Jonathan, N.; Liby, K.; McFarland, M.; Zinger, M. Prolactin as an autocrine/paracrine growth factor in human cancer. Trends Endocrinol. Metab. 2002, 13, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.; Stanford, P.M.; Oakes, S.R.; Ormandy, C.J. Prolactin and the prolactin receptor: New targets of an old hormone. Ann. Med. 2004, 36, 414–425. [Google Scholar] [CrossRef] [PubMed]
- Brooks, C.L. Molecular mechanisms of prolactin and its receptor. Endocr. Rev. 2012, 33, 504–525. [Google Scholar] [CrossRef] [PubMed]
- Gorvin, C.M. The prolactin receptor: Diverse and emerging roles in pathophysiology. J. Clin. Transl. Endocrinol. 2015, 2, 85–91. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Marano, R.J.; Ben-Jonathan, N. Minireview: Extrapituitary prolactin: An update on the distribution, regulation, and functions. Mol. Endocrinol. 2014, 28, 622–633. [Google Scholar] [CrossRef] [PubMed]
- Featherstone, K.; White, M.R.; Davis, J.R. The prolactin gene: A paradigm of tissue-specific gene regulation with complex temporal transcription dynamics. J. Neuroendocrinol. 2012, 24, 977–990. [Google Scholar] [CrossRef] [PubMed]
- Perks, C.M.; Newcomb, P.V.; Grohmann, M.; Wright, R.J.; Mason, H.D.; Holly, J.M. Prolactin acts as a potent survival factor against C2-ceramide-induced apoptosis in human granulosa cells. Hum. Reprod. 2003, 18, 2672–2677. [Google Scholar] [CrossRef]
- Schwärzler, P.; Untergasser, G.; Hermann, M.; Dirnhofer, S.; Abendstein, B.; Berger, P. Prolactin gene expression and prolactin protein in premenopausal and postmenopausal human ovaries. Fertil. Steril. 1997, 68, 696–701. [Google Scholar] [CrossRef]
- Vlahos, N.P.; Bugg, E.M.; Shamblott, M.J.; Phelps, J.Y.; Gearhart, J.D.; Zacur, H.A. Prolactin receptor gene expression and immunolocalization of the prolactin receptor in human luteinized granulosa cells. Mol. Hum. Reprod. 2001, 7, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Porter, M.B.; Brumsted, J.R.; Sites, C.K. Effect of prolactin on follicle-stimulating hormone receptor binding and progesterone production in cultured porcine granulosa cells. Fertil. Steril. 2000, 73, 99–105. [Google Scholar] [CrossRef]
- Ben-Jonathan, N.; LaPensee, C.R.; LaPensee, E.W. What can we learn from rodents about prolactin in humans? Endocr. Rev. 2008, 29, 1–41. [Google Scholar] [CrossRef] [PubMed]
- Munro, M.G.; Balen, A.H.; Cho, S.; Critchley, H.O.D.; Díaz, I.; Ferriani, R.; Henry, L.; Mocanu, E.; van der Spuy, Z.M.; FIGO Committee on Menstrual Disorders and Related Health Impacts; et al. The FIGO Ovulatory Disorders Classification System. Hum. Reprod. 2022, 37, 2446–2464. [Google Scholar] [CrossRef] [PubMed]
- Borght, M.V.; Wyns, C. Fertility and infertility: Definition and epidemiology. Clin. Biochem. 2018, 62, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, A.; Mangal, N.S. Hyperprolactinemia. J. Hum. Reprod. Sci. 2013, 6, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Shibli-Rahhal, A.; Schlechte, J. Hyperprolactinemia and infertility. Endocrinol. Metab. Clin. N. Am. 2011, 40, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Auriemma, R.S.; Del Vecchio, G.; Scairati, R.; Pirchio, R.; Liccardi, A.; Verde, N.; de Angelis, C.; Menafra, D.; Pivonello, C.; Conforti, A.; et al. The Interplay between Prolactin and Reproductive System: Focus on Uterine Pathophysiology. Front. Endocrinol. 2020, 11, 594370. [Google Scholar] [CrossRef]
- Štelcl, M.; Vrublovský, P.; Machač, Š. Prolactin and alteration of fertility. Ceska Gynekol. 2018, 83, 232–235. [Google Scholar] [PubMed]
- Iancu, M.E.; Albu, A.I.; Albu, D.N. Prolactin Relationship with Fertility and In Vitro Fertilization Outcomes-A Review of the Literature. Pharmaceuticals 2023, 16, 122. [Google Scholar] [CrossRef] [PubMed]
- Keeler, C.; Dannies, P.S.; Hodsdon, M.E. The tertiary structure and backbone dynamics of human prolactin. J. Mol. Biol. 2003, 328, 1105–1121. [Google Scholar] [CrossRef] [PubMed]
- Sinha, Y.N. Structural variants of prolactin: Occurrence and physiological significance. Endocr. Rev. 1995, 16, 354–369. [Google Scholar] [CrossRef]
- Horseman, N.D.; Yu-Lee, L.Y. Transcriptional regulation by the helix bundle peptide hormones: Growth hormone, prolactin, and hematopoietic cytokines. Endocr. Rev. 1994, 15, 627–649. [Google Scholar] [CrossRef] [PubMed]
- Corbacho, A.; De La Escalera, G.M.; Clapp, C. Roles of prolactin and related members of the prolactin/growth hormone/placental lactogen family in angiogenesis. J. Endocrinol. 2002, 173, 219–238. [Google Scholar] [CrossRef] [PubMed]
- Owerbach, D.; Rutter, W.J.; Cooke, N.E.; Martial, J.A.; Shows, T.B. The prolactin gene is located on chromosome 6 in humans. Science 1981, 212, 815–816. [Google Scholar] [CrossRef] [PubMed]
- McNamara, A.V.; Awais, R.; Momiji, H.; Dunham, L.; Featherstone, K.; Harper, C.V.; A Adamson, A.; Semprini, S.; A Jones, N.; Spiller, D.G.; et al. Transcription Factor Pit-1 Affects Transcriptional Timing in the Dual-Promoter Human Prolactin Gene. Endocrinology 2021, 162, bqaa249. [Google Scholar] [CrossRef]
- Trott, J.F.; Hovey, R.C.; Koduri, S.; Vonderhaar, B.K. Alternative splicing to exon 11 of human prolactin receptor gene results in multiple isoforms including a secreted prolactin-binding protein. J. Mol. Endocrinol. 2003, 30, 31–47. [Google Scholar] [CrossRef]
- Kasum, M.; Orešković, S.; Čehić, E.; Šunj, M.; Lila, A.; Ejubović, E. Laboratory and clinical significance of macroprolactinemia in women with hyperprolactinemia. Taiwan J. Obstet. Gynecol. 2017, 56, 719–724. [Google Scholar] [CrossRef]
- Vilar, L.; Vilar, C.F.; Lyra, R.; Freitas, M.D.C. Pitfalls in the Diagnostic Evaluation of Hyperprolactinemia. Neuroendocrinology 2019, 109, 7–19. [Google Scholar] [CrossRef]
- Thirunavakkarasu, K.; Dutta, P.; Sridhar, S.; Dhaliwal, L.; Prashad, G.R.; Gainder, S.; Sachdeva, N.; Bhansali, A. Macroprolactinemia in hyperprolactinemic infertile women. Endocrine 2013, 44, 750–755. [Google Scholar] [CrossRef]
- Koniares, K.; Benadiva, C.; Engmann, L.; Nulsen, J.; Grow, D. Macroprolactinemia: A mini-review and update on clinical practice. F&S Rep. 2023, 4, 245–250. [Google Scholar] [CrossRef]
- Kasum, M.; Pavičić-Baldani, D.; Stanić, P.; Orešković, S.; Sarić, J.M.; Blajić, J.; Juras, J. Importance of macroprolactinemia in hyperprolactinemia. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 183, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Bugge, K.; Papaleo, E.; Haxholm, G.W.; Hopper, J.T.; Robinson, C.V.; Olsen, J.G.; Lindorff-Larsen, K.; Kragelund, B.B. A combined computational and structural model of the full-length human prolactin receptor. Nat. Commun. 2016, 7, 11578. [Google Scholar] [CrossRef]
- Araya-Secchi, R.; Bugge, K.; Seiffert, P.; Petry, A.; Haxholm, G.W.; Lindorff-Larsen, K.; Pedersen, S.F.; Arleth, L.; Kragelund, B.B. The prolactin receptor scaffolds Janus kinase 2 via co-structure formation with phosphoinositide-4,5-bisphosphate. eLife 2023, 12, e84645. [Google Scholar] [CrossRef] [PubMed]
- A Lee, S.; A Haiman, C.; Burtt, N.P.; Pooler, L.C.; Cheng, I.; Kolonel, L.N.; Pike, M.C.; Altshuler, D.; Hirschhorn, J.N.; E Henderson, B.; et al. A comprehensive analysis of common genetic variation in prolactin (PRL) and PRL receptor (PRLR) genes in relation to plasma prolactin levels and breast cancer risk: The multiethnic cohort. BMC Med. Genet. 2007, 8, 72. [Google Scholar] [CrossRef]
- Gorvin, C.M.; Newey, P.J.; Thakker, R.V. Identification of prolactin receptor variants with diverse effects on receptor signalling. J. Mol. Endocrinol. 2023, 70, e220164. [Google Scholar] [CrossRef]
- Dagil, R.; Knudsen, M.J.; Olsen, J.G.; O’Shea, C.; Franzmann, M.; Goffin, V.; Teilum, K.; Breinholt, J.; Kragelund, B.B. The WSXWS motif in cytokine receptors is a molecular switch involved in receptor activation: Insight from structures of the prolactin receptor. Structure 2012, 20, 270–282. [Google Scholar] [CrossRef]
- Bole-Feysot, C.; Goffin, V.; Edery, M.; Binart, N.; Kelly, P.A. Prolactin (PRL) and its receptor: Actions, signal transduction pathways and phenotypes observed in PRL receptor knockout mice. Endocr. Rev. 1998, 19, 225–268. [Google Scholar] [CrossRef]
- Vara, J.F.; Carretero, M.V.; Gerónimo, H.; Ballmer-Hofer, K.; Martín-Pérez, J. Stimulation of c-Src by prolactin is independent of Jak2. Biochem. J. 2000, 345 Pt 1, 17–24. [Google Scholar] [CrossRef]
- Kavarthapu, R.; Dufau, M.L. Prolactin receptor gene transcriptional control, regulatory modalities relevant to breast cancer resistance and invasiveness. Front. Endocrinol. 2022, 13, 949396. [Google Scholar] [CrossRef] [PubMed]
- Haxholm, G.W.; Nikolajsen, L.F.; Olsen, J.G.; Fredsted, J.; Larsen, F.H.; Goffin, V.; Pedersen, S.F.; Brooks, A.J.; Waters, M.J.; Kragelund, B.B. Intrinsically disordered cytoplasmic domains of two cytokine receptors mediate conserved interactions with membranes. Biochem. J. 2015, 468, 495–506. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.Z.; Meng, J.; Dufau, M.L. Isolation and characterization of two novel forms of the human prolactin receptor generated by alternative splicing of a newly identified exon 11. J. Biol. Chem. 2001, 276, 41086–41094. [Google Scholar] [CrossRef] [PubMed]
- Tsai-Morris, C.H.; Dufau, M.L. PRLR (prolactin receptor). In Atlas Genet Cytogenet Oncol Haematol; Institute for Biomedical Research of Salamanca: Salamanca, Spain, 2011; Available online: http://atlasgeneticsoncology.org/gene/42891/ (accessed on 26 December 2023).
- Abramicheva, P.A.; Smirnova, O.V. Prolactin Receptor Isoforms as the Basis of Tissue-Specific Action of Prolactin in the Norm and Pathology. Biochemistry 2019, 84, 329–345. [Google Scholar] [CrossRef]
- Berthon, P.; Kelly, P.A.; Djiane, J. Water-soluble prolactin receptors from porcine mammary gland. Proc. Soc. Exp. Biol. Med. 1987, 184, 300–306. [Google Scholar] [CrossRef]
- Postel-Vinay, M.C.; Belair, L.; Kayser, C.; Kelly, P.A.; Djiane, J. Identification of prolactin and growth hormone binding proteins in rabbit milk. Proc. Natl. Acad. Sci. USA 1991, 88, 6687–6690. [Google Scholar] [CrossRef]
- Ezoe, K.; Miki, T.; Ohata, K.; Fujiwara, N.; Yabuuchi, A.; Kobayashi, T.; Kato, K. Prolactin receptor expression and its role in trophoblast outgrowth in human embryos. Reprod. Biomed. Online 2021, 42, 699–707. [Google Scholar] [CrossRef]
- Utama, F.E.; Tran, T.H.; Ryder, A.; LeBaron, M.J.; Parlow, A.F.; Rui, H. Insensitivity of human prolactin receptors to nonhuman prolactins: Relevance for experimental modeling of prolactin receptor-expressing human cells. Endocrinology 2009, 150, 1782–1790. [Google Scholar] [CrossRef]
- Pezet, A.; Buteau, H.; Kelly, P.A.; Edery, M. The last proline of Box 1 is essential for association with JAK2 and functional activation of the prolactin receptor. Mol. Cell Endocrinol. 1997, 129, 199–208. [Google Scholar] [CrossRef]
- Brockman, J.L.; Schuler, L.A. Prolactin signals via Stat5 and Oct-1 to the proximal cyclin D1 promoter. Mol. Cell Endocrinol. 2005, 239, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Aksamitiene, E.; Achanta, S.; Kolch, W.; Kholodenko, B.N.; Hoek, J.B.; Kiyatkin, A. Prolactin-stimulated activation of ERK1/2 mitogen-activated protein kinases is controlled by PI3-kinase/Rac/PAK signaling pathway in breast cancer cells. Cell Signal. 2011, 23, 1794–1805. [Google Scholar] [CrossRef] [PubMed]
- Derwich, A.; Sykutera, M.; Bromińska, B.; Rubiś, B.; Ruchała, M.; Sawicka-Gutaj, N. The Role of Activation of PI3K/AKT/mTOR and RAF/MEK/ERK Pathways in Aggressive Pituitary Adenomas-New Potential Therapeutic Approach-A Systematic Review. Int. J. Mol. Sci. 2023, 24, 10952. [Google Scholar] [CrossRef] [PubMed]
- Bishop, J.D.; Nien, W.L.; Dauphinee, S.M.; Too, C.K. Prolactin activates mammalian target-of-rapamycin through phosphatidylinositol 3-kinase and stimulates phosphorylation of p70S6K and 4E-binding protein-1 in lymphoma cells. J. Endocrinol. 2006, 190, 307–312. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hewetson, A.; Moore, S.L.; Chilton, B.S. Prolactin signals through RUSH/SMARCA3 in the absence of a physical association with Stat5a. Biol. Reprod. 2004, 71, 1907–1912. [Google Scholar] [CrossRef] [PubMed]
- Hannan, F.M.; Elajnaf, T.; Vandenberg, L.N.; Kennedy, S.H.; Thakker, R.V. Hormonal regulation of mammary gland development and lactation. Nat. Rev. Endocrinol. 2023, 19, 46–61. [Google Scholar] [CrossRef] [PubMed]
- Auriemma, R.S.; Pirchio, R.; Pivonello, C.; Garifalos, F.; Colao, A.; Pivonello, R. Approach to the Patient with Prolactinoma. J. Clin. Endocrinol. Metab. 2023, 108, 2400–2423. [Google Scholar] [CrossRef]
- Moriwaki, M.; Welt, C.K. PRL Mutation Causing Alactogenesis: Insights into Prolactin Structure and Function Relationships. J. Clin. Endocrinol. Metab. 2021, 106, e3021–e3026. [Google Scholar] [CrossRef]
- Naylor, M.J.; Lockefeer, J.A.; Horseman, N.D.; Ormandy, C.J. Prolactin regulates mammary epithelial cell proliferation via autocrine/paracrine mechanism. Endocrine 2003, 20, 111–114. [Google Scholar] [CrossRef]
- Gabrielson, M.; Ubhayasekera, K.; Ek, B.; Andersson Franko, M.; Eriksson, M.; Czene, K.; Bergquist, J.; Hall, P. Inclusion of Plasma Prolactin Levels in Current Risk Prediction Models of Premenopausal and Postmenopausal Breast Cancer. JNCI Cancer Spectr. 2018, 2, pky055. [Google Scholar] [CrossRef]
- Sackmann-Sala, L.; Guidotti, J.E.; Goffin, V. Minireview: Prolactin regulation of adult stem cells. Mol. Endocrinol. 2015, 29, 667–681. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Membrillo, M.; Siqueiros-Márquez, L.; Núñez, F.F.; Díaz-Lezama, N.; Adán-Castro, E.; Ramírez-Hernández, G.; Adán, N.; Macotela, Y.; de la Escalera, G.M.; Clapp, C.; et al. Prolactin stimulates the vascularisation of the retina in newborn mice under hyperoxia conditions. J. Neuroendocrinol. 2020, 32, e12858. [Google Scholar] [CrossRef] [PubMed]
- Yousefvand, S.; Hadjzadeh, M.A.; Vafaee, F.; Dolatshad, H. The protective effects of prolactin on brain injury. Life Sci. 2020, 263, 118547. [Google Scholar] [CrossRef] [PubMed]
- Molina-Salinas, G.; Rivero-Segura, N.A.; Cabrera-Reyes, E.A.; Rodríguez-Chávez, V.; Langley, E.; Cerbon, M. Decoding signaling pathways involved in prolactin-induced neuroprotection: A review. Front. Neuroendocrinol. 2021, 61, 100913. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Chavez, V.; Moran, J.; Molina-Salinas, G.; Zepeda Ruiz, W.A.; Rodriguez, M.C.; Picazo, O.; Cerbon, M. Participation of Glutamatergic Ionotropic Receptors in Excitotoxicity: The Neuroprotective Role of Prolactin. Neuroscience 2021, 461, 180–193. [Google Scholar] [CrossRef] [PubMed]
- Clevenger, C.V.; Rui, H. Breast Cancer and Prolactin—New Mechanisms and Models. Endocrinology 2022, 163, bqac122. [Google Scholar] [CrossRef]
- Ramírez-de-Arellano, A.; Villegas-Pineda, J.C.; Hernández-Silva, C.D.; Pereira-Suárez, A.L. The Relevant Participation of Prolactin in the Genesis and Progression of Gynecological Cancers. Front. Endocrinol. 2021, 12, 747810. [Google Scholar] [CrossRef]
- Baeyens, L.; Hindi, S.; Sorenson, R.L.; German, M.S. β-Cell adaptation in pregnancy. Diabetes Obes. Metab. 2016, 18 (Suppl. S1), 63–70. [Google Scholar] [CrossRef]
- Al-Nami, M.S.; Al-Kuraishy, H.M.; Al-Gareeb, A.I.; Al-Mamoori, F. Metabolic profile and prolactin serum levels in men with type 2 diabetes mellitus: Old-new rubric. Int. J. Crit. Illn. Inj. Sci. 2019, 9, 120–126. [Google Scholar] [CrossRef]
- Zhu, C.; Wen, X.; You, H.; Lu, L.; Du, L.; Qian, C. Improved Insulin Secretion Response and Beta-cell Function Correlated with Increased Prolactin Levels after Laparoscopic Sleeve Gastrectomy in Morbidly Obese Patients with Acanthosis Nigricans. Obes. Surg. 2023, 33, 2405–2419. [Google Scholar] [CrossRef]
- Retnakaran, R.; Ye, C.; Kramer, C.K.; Connelly, P.W.; Hanley, A.J.; Sermer, M.; Zinman, B. Maternal Serum Prolactin and Prediction of Postpartum β-Cell Function and Risk of Prediabetes/Diabetes. Diabetes Care. 2016, 39, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Rassie, K.; Giri, R.; Joham, A.E.; Mousa, A.; Teede, H. Prolactin in relation to gestational diabetes and metabolic risk in pregnancy and postpartum: A systematic review and meta-analysis. Front. Endocrinol. 2022, 13, 1069625. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Lu, J.; Xu, Y.; Li, M.; Sun, J.; Zhang, J.; Xu, B.; Xu, M.; Chen, Y.; Bi, Y.; et al. Circulating prolactin associates with diabetes and impaired glucose regulation: A population-based study. Diabetes Care. 2013, 36, 1974–1980. [Google Scholar] [CrossRef]
- Yang, H.; Lin, J.; Li, H.; Liu, Z.; Chen, X.; Chen, Q. Prolactin Is Associated with Insulin Resistance and Beta-Cell Dysfunction in Infertile Women with Polycystic Ovary Syndrome. Front. Endocrinol. 2021, 12, 571229. [Google Scholar] [CrossRef] [PubMed]
- Rasheed, H.A.; Al-Kuraishy, H.M.; Al-Gareeb, A.I.; Hussien, N.R.; Al-Nami, M.S. Effects of diabetic pharmacotherapy on prolactin hormone in patients with type 2 diabetes mellitus: Bane or Boon. J. Adv. Pharm. Technol. Res. 2019, 10, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Shao, S.; Yao, Z.; Lu, J.; Song, Y.; He, Z.; Yu, C.; Zhou, X.; Zhao, L.; Zhao, J.; Gao, L. Ablation of prolactin receptor increases hepatic triglyceride accumulation. Biochem. Biophys. Res. Commun. 2018, 498, 693–699. [Google Scholar] [CrossRef]
- de Winne, C.; Pascual, F.L.; Lopez-Vicchi, F.; Etcheverry-Boneo, L.; Mendez-Garcia, L.F.; Ornstein, A.M.; Lacau-Mengido, I.M.; Sorianello, E.; Becu-Villalobos, D. Neuroendocrine control of brown adipocyte function by prolactin and growth hormone. J. Neuroendocrinol. 2023, e13248. [Google Scholar] [CrossRef]
- Ghoreshi, Z.A.; Akbari, H.; Sharif-Zak, M.; Arefinia, N.; Abbasi-Jorjandi, M.; Asadikaram, G. Recent findings on hyperprolactinemia and its pathological implications: A literature review. J. Investig. Med. 2022, 70, 1443–1451. [Google Scholar] [CrossRef]
- Corona, G.; Rastrelli, G.; Comeglio, P.; Guaraldi, F.; Mazzatenta, D.; Sforza, A.; Vignozzi, L.; Maggi, M. The metabolic role of prolactin: Systematic review, meta-analysis and preclinical considerations. Expert. Rev. Endocrinol. Metab. 2022, 17, 533–545. [Google Scholar] [CrossRef]
- Xu, P.; Zhu, Y.; Ji, X.; Ma, H.; Zhang, P.; Bi, Y. Lower serum PRL is associated with the development of non-alcoholic fatty liver disease: A retrospective cohort study. BMC Gastroenterol. 2022, 22, 523. [Google Scholar] [CrossRef]
- Deachapunya, C.; Poonyachoti, S.; Krishnamra, N. Regulation of electrolyte transport across cultured endometrial epithelial cells by prolactin. J. Endocrinol. 2008, 197, 575–582. [Google Scholar] [CrossRef]
- Radojkovic, D.; Pesic, M.; Radojkovic, M.; Dimic, D.; Nikolic, M.V.; Stoimenov, T.J.; Radenkovic, S.; Golubovic, M.V.; Petkovic, T.R.; Antic, S. Expression of prolactin receptors in the duodenum, kidneys and skeletal system during physiological and sulpiride-induced hyperprolactinaemia. Endocrine 2018, 62, 681–691. [Google Scholar] [CrossRef]
- Ibarra, F.; Crambert, S.; Eklöf, A.C.; Lundquist, A.; Hansell, P.; Holtbäck, U. Prolactin, a natriuretic hormone, interacting with the renal dopamine system. Kidney Int. 2005, 68, 1700–1707. [Google Scholar] [CrossRef]
- Crambert, S.; Sjöberg, A.; Eklöf, A.C.; Ibarra, F.; Holtbäck, U. Prolactin and dopamine 1-like receptor interaction in renal proximal tubular cells. Am. J. Physiol. Ren. Physiol. 2010, 299, F49–F54. [Google Scholar] [CrossRef] [PubMed]
- Georgescu, T.; Swart, J.M.; Grattan, D.R.; Brown, R.S.E. The Prolactin Family of Hormones as Regulators of Maternal Mood and Behavior. Front. Glob. Womens Health 2021, 2, 767467. [Google Scholar] [CrossRef] [PubMed]
- Faron-Górecka, A.; Latocha, K.; Pabian, P.; Kolasa, M.; Sobczyk-Krupiarz, I.; Dziedzicka-Wasylewska, M. The Involvement of Prolactin in Stress-Related Disorders. Int. J. Environ. Res. Public Health 2023, 20, 3257. [Google Scholar] [CrossRef] [PubMed]
- Reavley, A.; Fisher, A.D.; Owen, D.; Creed, F.H.; Davis, J.R. Psychological distress in patients with hyperprolactinaemia. Clin. Endocrinol. 1997, 47, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Elgellaie, A.; Larkin, T.; Kaelle, J.; Mills, J.; Thomas, S. Plasma prolactin is higher in major depressive disorder and females, and associated with anxiety, hostility, somatization, psychotic symptoms and heart rate. Compr. Psychoneuroendocrinol. 2021, 6, 100049. [Google Scholar] [CrossRef] [PubMed]
- Telfer, E.E.; Grosbois, J.; Odey, Y.L.; Rosario, R.; Anderson, R.A. Making a good egg: Human oocyte health, aging, and in vitro development. Physiol. Rev. 2023, 103, 2623–2677. [Google Scholar] [CrossRef] [PubMed]
- Marques, P.; Skorupskaite, K.; Rozario, K.S.; Anderson, R.A.; George, J.T. Physiology of GnRH and Gonadotropin Secretion. In Endotext [Internet]; Feingold, K.R., Anawalt, B., Blackman, M.R., Boyce, A., Chrousos, G., Corpas, E., de Herder, W.W., Dhatariya, K., Dungan, K., Hofland, J., et al., Eds.; Updated 5 January 2022; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. Available online: https://www.ncbi.nlm.nih.gov/books/NBK279070/ (accessed on 15 December 2023).
- Marshall, J.C.; Dalkin, A.C.; Haisenleder, D.J.; Paul, S.J.; Ortolano, G.A.; Kelch, R.P. Gonadotropin-releasing hormone pulses: Regulators of gonadotropin synthesis and ovulatory cycles. Recent. Prog. Horm. Res. 1991, 47, 155–187. [Google Scholar] [CrossRef] [PubMed]
- Orlowski, M.; Sarao, M.S. Physiology, Follicle Stimulating Hormone. In StatPearls [Internet]; Updated 1 May 2023; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK535442/ (accessed on 15 December 2023).
- Reed, B.G.; Carr, B.R. The Normal Menstrual Cycle and the Control of Ovulation. In Endotext [Internet]; Feingold, K.R., Anawalt, B., Blackman, M.R., Boyce, A., Chrousos, G., Corpas, E., de Herder, W.W., Dhatariya, K., Dungan, K., Hofland, J., et al., Eds.; Updated 5 August 2018; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. Available online: https://www.ncbi.nlm.nih.gov/books/NBK279054/ (accessed on 22 December 2023).
- Nedresky, D.; Singh, G. Physiology, Luteinizing Hormone. In StatPearls [Internet]; Updated 26 September 2022; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK539692/ (accessed on 19 December 2023).
- Cable, J.K.; Grider, M.H. Physiology, Progesterone. In StatPearls [Internet]; Updated 1 May 2023; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK558960/ (accessed on 19 December 2023).
- Findlay, J.K.; Drummond, A.E.; Dyson, M.; Baillie, A.J.; Robertson, D.M.; Ethier, J.F. Production and actions of inhibin and activin during folliculogenesis in the rat. Mol. Cell Endocrinol. 2001, 180, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Vale, W.; Wiater, E.; Gray, P.; Harrison, C.; Bilezikjian, L.; Choe, S. Activins and inhibins and their signaling. Ann. N. Y. Acad. Sci. 2004, 1038, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Roseff, S.J.; Bangah, M.L.; Kettel, L.M.; Vale, W.; Rivier, J.; Burger, H.G.; Yen, S.S. Dynamic changes in circulating inhibin levels during the luteal-follicular transition of the human menstrual cycle. J. Clin. Endocrinol. Metab. 1989, 69, 1033–1039. [Google Scholar] [CrossRef] [PubMed]
- Welt, C.K.; Schneyer, A.L. Differential regulation of inhibin B and inhibin a by follicle-stimulating hormone and local growth factors in human granulosa cells from small antral follicles. J. Clin. Endocrinol. Metab. 2001, 86, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Block, E. A quantitative morphological investigation of the follicular system in newborn female infants. Acta Anat. 1953, 17, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Forabosco, A.; Sforza, C.; De Pol, A.; Vizzotto, L.; Marzona, L.; Ferrario, V.F. Morphometric study of the human neonatal ovary. Anat. Rec. 1991, 231, 201–208. [Google Scholar] [CrossRef] [PubMed]
- McGee, E.A.; Hsueh, A.J. Initial and cyclic recruitment of ovarian follicles. Endocr. Rev. 2000, 21, 200–214. [Google Scholar] [CrossRef] [PubMed]
- DiVall, S.A.; Radovick, S. Endocrinology of female puberty. Curr. Opin. Endocrinol. Diabetes Obes. 2009, 16, 1–4. [Google Scholar] [CrossRef]
- Mikhael, S.; Punjala-Patel, A.; Gavrilova-Jordan, L. Hypothalamic-Pituitary-Ovarian Axis Disorders Impacting Female Fertility. Biomedicines 2019, 7, 5. [Google Scholar] [CrossRef]
- Messinis, I.E.; Messini, C.I.; Dafopoulos, K. Novel aspects of the endocrinology of the menstrual cycle. Reprod. Biomed. Online 2014, 28, 714–722. [Google Scholar] [CrossRef]
- Welt, C.K. Regulation and function of inhibins in the normal menstrual cycle. Semin. Reprod. Med. 2004, 22, 187–193. [Google Scholar] [CrossRef]
- Buffet, N.C.; Bouchard, P. The neuroendocrine regulation of the human ovarian cycle. Chronobiol. Int. 2001, 18, 893–919. [Google Scholar]
- Fischer-Holzhausen, S.; Röblitz, S. Hormonal regulation of ovarian follicle growth in humans: Model-based exploration of cycle variability and parameter sensitivities. J. Theor. Biol. 2022, 547, 111150. [Google Scholar] [CrossRef]
- Scaramuzzi, R.J.; Baird, D.T.; Campbell, B.K.; Driancourt, M.-A.; Dupont, J.; Fortune, J.E.; Gilchrist, R.B.; Martin, G.B.; McNatty, K.P.; McNeilly, A.S.; et al. Regulation of folliculogenesis and the determination of ovulation rate in ruminants. Reprod. Fertil. Dev. 2011, 23, 444–467. [Google Scholar] [CrossRef]
- Lundy, T.; Smith, P.; O’Connell, A.; Hudson, N.L.; McNatty, K.P. Populations of granulosa cells in small follicles of the sheep ovary. J. Reprod. Fertil. 1999, 115, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Dunlop, C.E.; Anderson, R.A. The regulation and assessment of follicular growth. Scand. J. Clin. Lab. Investig. Suppl. 2014, 244, 13–17. [Google Scholar] [CrossRef]
- Gershon, E.; Dekel, N. Newly Identified Regulators of Ovarian Folliculogenesis and Ovulation. Int. J. Mol. Sci. 2020, 21, 4565. [Google Scholar] [CrossRef] [PubMed]
- Evans, A.C.; Fortune, J.E. Selection of the dominant follicle in cattle occurs in the absence of differences in the expression of messenger ribonucleic acid for gonadotropin receptors. Endocrinology 1997, 138, 2963–2971. [Google Scholar] [CrossRef] [PubMed][Green Version]
- de Andrade, L.G.; Portela, V.M.; Dos Santos, E.C.; Aires, K.d.V.; Ferreira, R.; Missio, D.; da Silva, Z.; Koch, J.; Antoniazzi, A.Q.; Gonçalves, P.B.D.; et al. FSH Regulates YAP-TEAD Transcriptional Activity in Bovine Granulosa Cells to Allow the Future Dominant Follicle to Exert Its Augmented Estrogenic Capacity. Int. J. Mol. Sci. 2022, 23, 14160. [Google Scholar] [CrossRef] [PubMed]
- Casarini, L.; Paradiso, E.; Lazzaretti, C.; D’Alessandro, S.; Roy, N.; Mascolo, E.; Zaręba, K.; García-Gasca, A.; Simoni, M. Regulation of antral follicular growth by an interplay between gonadotropins and their receptors. J. Assist. Reprod. Genet. 2022, 39, 893–904. [Google Scholar] [CrossRef] [PubMed]
- Rosen, M.P.; Zamah, A.M.; Shen, S.; Dobson, A.T.; McCulloch, C.E.; Rinaudo, P.F.; Lamb, J.D.; Cedars, M.I. The effect of follicular fluid hormones on oocyte recovery after ovarian stimulation: FSH level predicts oocyte recovery. Reprod. Biol. Endocrinol. 2009, 7, 35. [Google Scholar] [CrossRef]
- Chauvin, S.; Cohen-Tannoudji, J.; Guigon, C.J. Estradiol Signaling at the Heart of Folliculogenesis: Its Potential Deregulation in Human Ovarian Pathologies. Int. J. Mol. Sci. 2022, 23, 512. [Google Scholar] [CrossRef]
- Stringer, J.M.; Alesi, L.R.; Winship, A.L.; Hutt, K.J. Beyond apoptosis: Evidence of other regulated cell death pathways in the ovary throughout development and life. Hum. Reprod. Update 2023, 29, 434–456. [Google Scholar] [CrossRef] [PubMed]
- Shelling, A.N.; Nasef, N.A. The Role of Lifestyle and Dietary Factors in the Development of Premature Ovarian Insufficiency. Antioxidants 2023, 12, 1601. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.C.; Wang, Y.Y.; Sun, X.F.; Cheng, S.F.; Li, L.; Zhao, Y.; Shen, W.; Chen, H. The role of autophagy during murine primordial follicle assembly. Aging 2018, 10, 197–211. [Google Scholar] [CrossRef] [PubMed]
- Schuh-Huerta, S.M.; Johnson, N.A.; Rosen, M.P.; Sternfeld, B.; Cedars, M.I.; Pera, R.A.R. Genetic markers of ovarian follicle number and menopause in women of multiple ethnicities. Hum. Genet. 2012, 131, 1709–1724. [Google Scholar] [CrossRef] [PubMed]
- Irving-Rodgers, H.F.; van Wezel, I.L.; Mussard, M.L.; Kinder, J.E.; Rodgers, R.J. Atresia revisited: Two basic patterns of atresia of bovine antral follicles. Reproduction 2001, 122, 761–775. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Peng, X.; Mei, S. Autophagy in Ovarian Follicular Development and Atresia. Int. J. Biol. Sci. 2019, 15, 726–737. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Shi, X.; Shi, Y.; Wang, Z. The Signaling Pathways Involved in Ovarian Follicle Development. Front. Physiol. 2021, 12, 730196. [Google Scholar] [CrossRef]
- Gellersen, B.; Brosens, J.J. Cyclic decidualization of the human endometrium in reproductive health and failure. Endocr. Rev. 2014, 35, 851–905. [Google Scholar] [CrossRef]
- Baerwald, A.; Pierson, R. Ovarian follicular waves during the menstrual cycle: Physiologic insights into novel approaches for ovarian stimulation. Fertil. Steril. 2020, 114, 443–457. [Google Scholar] [CrossRef]
- Bulletti, C.; Bulletti, F.M.; Sciorio, R.; Guido, M. Progesterone: The Key Factor of the Beginning of Life. Int. J. Mol. Sci. 2022, 23, 14138. [Google Scholar] [CrossRef]
- Klusmann, H.; Schulze, L.; Engel, S.; Bücklein, E.; Daehn, D.; Lozza-Fiacco, S.; Geiling, A.; Meyer, C.; Andersen, E.; Knaevelsrud, C.; et al. HPA axis activity across the menstrual cycle—A systematic review and meta-analysis of longitudinal studies. Front. Neuroendocr. 2022, 66, 100998. [Google Scholar] [CrossRef] [PubMed]
- Javed, Z.; Qamar, U.; Sathyapalan, T. The role of kisspeptin signalling in the hypothalamic-pituitary-gonadal axis—Current perspective. Endokrynol. Pol. 2015, 66, 534–547. [Google Scholar] [CrossRef]
- Skorupskaite, K.; George, J.T.; Anderson, R.A. The kisspeptin-GnRH pathway in human reproductive health and disease. Hum. Reprod. Update 2014, 20, 485–500. [Google Scholar] [CrossRef]
- Stevenson, H.; Bartram, S.; Charalambides, M.M.; Murthy, S.; Petitt, T.; Pradeep, A.; Vineall, O.; Abaraonye, I.; Lancaster, A.; Koysombat, K.; et al. Kisspeptin-neuron control of LH pulsatility and ovulation. Front. Endocrinol. 2022, 13, 951938. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, A.S. Neuroendocrine mechanisms underlying estrogen positive feedback and the LH surge. Front. Neurosci. 2022, 16, 953252. [Google Scholar] [CrossRef] [PubMed]
- Tsukamura, H. Kobayashi Award 2019: The neuroendocrine regulation of the mammalian reproduction. Gen. Comp. Endocrinol. 2022, 315, 113755. [Google Scholar] [CrossRef]
- Namwanje, M.; Brown, C.W. Activins and Inhibins: Roles in Development, Physiology, and Disease. Cold Spring Harb. Perspect. Biol. 2016, 8, a021881. [Google Scholar] [CrossRef]
- Perakakis, N.; Upadhyay, J.; Ghaly, W.; Chen, J.; Chrysafi, P.; Anastasilakis, A.D.; Mantzoros, C.S. Regulation of the activins-follistatins-inhibins axis by energy status: Impact on reproductive function. Metabolism 2018, 85, 240–249. [Google Scholar] [CrossRef]
- Tumurgan, Z.; Kanasaki, H.; Tumurbaatar, T.; Oride, A.; Okada, H.; Hara, T.; Kyo, S. Role of activin, follistatin, and inhibin in the regulation of Kiss-1 gene expression in hypothalamic cell models. Biol. Reprod. 2019, 101, 405–415. [Google Scholar] [CrossRef]
- Walker, M.H.; Tobler, K.J. Female Infertility. In StatPearls [Internet]; Updated 19 December 2022; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK556033/ (accessed on 29 January 2024).
- Agents Stimulating Gonadal Function in the Human. Report of a WHO scientific group. World Health Organ. Tech. Rep. Ser. 1973, 514, 1–30.
- Urman, B.; Yakin, K. Ovulatory disorders and infertility. J. Reprod. Med. 2006, 51, 267–282. [Google Scholar] [PubMed]
- Provan, D. (Ed.) Oxford Handbook of Clinical and Laboratory Investigation, 4th ed.; Oxford University Press: New York, NY, USA, 2018. [Google Scholar]
- Sikaris, K.A. Enhancing the Clinical Value of Medical Laboratory Testing. Clin. Biochem. Rev. 2017, 38, 107–114. [Google Scholar] [PubMed]
- Sperduti, S.; Limoncella, S.; Lazzaretti, C.; Paradiso, E.; Riccetti, L.; Turchi, S.; Ferrigno, I.; Bertacchini, J.; Palumbo, C.; Potì, F.; et al. GnRH Antagonists Produce Differential Modulation of the Signaling Pathways Mediated by GnRH Receptors. Int. J. Mol. Sci. 2019, 20, 5548. [Google Scholar] [CrossRef] [PubMed]
- Choi, D. Evolutionary Viewpoint on GnRH (gonadotropin-releasing hormone) in Chordata—Amino Acid and Nucleic Acid Sequences. Dev. Reprod. 2018, 22, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Ferris, H.A.; Shupnik, M.A. Mechanisms for pulsatile regulation of the gonadotropin subunit genes by GNRH1. Biol. Reprod. 2006, 74, 993–998. [Google Scholar] [CrossRef] [PubMed]
- Nippoldt, T.B.; Reame, N.E.; Kelch, R.P.; Marshall, J.C. The roles of estradiol and progesterone in decreasing luteinizing hormone pulse frequency in the luteal phase of the menstrual cycle. J. Clin. Endocrinol. Metab. 1989, 69, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Romano, G.J.; Krust, A.; Pfaff, D.W. Expression and estrogen regulation of progesterone receptor mRNA in neurons of the mediobasal hypothalamus: An in situ hybridization study. Mol. Endocrinol. 1989, 3, 1295–1300, Erratum in Mol. Endocrinol. 1989, 3, 1860. [Google Scholar] [CrossRef]
- Soules, M.R.; Steiner, R.A.; Clifton, D.K.; Cohen, N.L.; Aksel, S.; Bremner, W.J. Progesterone modulation of pulsatile luteinizing hormone secretion in normal women. J. Clin. Endocrinol. Metab. 1984, 58, 378–383. [Google Scholar] [CrossRef]
- Berga, S.L.; Yen, S.S. Opioidergic regulation of LH pulsatility in women with polycystic ovary syndrome. Clin. Endocrinol. 1989, 30, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Neal-Perry, G.; Lebesgue, D.; Lederman, M.; Shu, J.; Zeevalk, G.D.; Etgen, A.M. The excitatory peptide kisspeptin restores the luteinizing hormone surge and modulates amino acid neurotransmission in the medial preoptic area of middle-aged rats. Endocrinology 2009, 150, 3699–3708. [Google Scholar] [CrossRef] [PubMed]
- Araki, S.; Takanashi, N.; Chikazawa, K.; Motoyama, M.; Akabori, A.; Konuma, S.; Tamada, T. Reevaluation of immunoreactive gonadotropin-releasing hormone (GnRH) levels in general circulation in women: Changes in levels and episodic patterns before, during and after gonadotropin surges. Endocrinol. Jpn. 1986, 33, 457–468. [Google Scholar] [CrossRef] [PubMed]
- Andersen, C.Y.; Ezcurra, D. Human steroidogenesis: Implications for controlled ovarian stimulation with exogenous gonadotropins. Reprod. Biol. Endocrinol. 2014, 12, 128. [Google Scholar] [CrossRef] [PubMed]
- Dias, J.A.; Ulloa-Aguirre, A. New Human Follitropin Preparations: How Glycan Structural Differences May Affect Biochemical and Biological Function and Clinical Effect. Front. Endocrinol. 2021, 12, 636038. [Google Scholar] [CrossRef] [PubMed]
- Bousfield, G.R.; Butnev, V.Y.; White, W.K.; Hall, A.S.; Harvey, D.J. Comparison of Follicle-Stimulating Hormone Glycosylation Microheterogenity by Quantitative Negative Mode Nano-Electrospray Mass Spectrometry of Peptide-N Glycanase-Released Oligosaccharides. J. Glycom. Lipidom. 2015, 5, 129. [Google Scholar] [CrossRef]
- Fink, G. Gonadotropin secretion and its control. In The Physiology of Reproduction; Knobil, E., Neill, J.D., Ewing, L.L., Greenwald, G.S., Markert, L., Pfaff, D.W., Eds.; Raven: New York, NY, USA, 1988; pp. 1349–1377. [Google Scholar]
- Oduwole, O.O.; Huhtaniemi, I.T.; Misrahi, M. The Roles of Luteinizing Hormone, Follicle-Stimulating Hormone and Testosterone in Spermatogenesis and Folliculogenesis Revisited. Int. J. Mol. Sci. 2021, 22, 12735. [Google Scholar] [CrossRef]
- Amsterdam, A.; Rotmensch, S. Structure-function relationships during granulosa cell differentiation. Endocr. Rev. 1987, 8, 309–337. [Google Scholar] [CrossRef]
- Larson, S.B.; McPherson, A. The crystal structure of the β subunit of luteinizing hormone and a model for the intact hormone. Curr. Res. Struct. Biol. 2019, 1, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Filicori, M. The role of luteinizing hormone in folliculogenesis and ovulation induction. Fertil. Steril. 1999, 71, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Sait, S.F. Luteinizing hormone and its dilemma in ovulation induction. J. Hum. Reprod. Sci. 2011, 4, 2–7. [Google Scholar] [CrossRef]
- Duffy, D.M.; Ko, C.; Jo, M.; Brannstrom, M.; Curry, T.E. Ovulation: Parallels with Inflammatory Processes. Endocr. Rev. 2019, 40, 369–416. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Sciences Policy; Committee on the Clinical Utility of Treating Patients with Compounded Bioidentical Hormone Replacement Therapy; Jackson, L.M.; Parker, R.M.; Mattison, D.R. (Eds.) The Clinical Utility of Compounded Bioidentical Hormone Therapy: A Review of Safety, Effectiveness, and Use; 4, Reproductive Steroid Hormones: Synthesis, Structure, and Biochemistry; National Academies Press: Washington, DC, USA, 2020. Available online: https://www.ncbi.nlm.nih.gov/books/NBK562873/ (accessed on 10 December 2023).
- Xu, X.L.; Huang, Z.Y.; Yu, K.; Li, J.; Fu, X.W.; Deng, S.L. Estrogen Biosynthesis and Signal Transduction in Ovarian Disease. Front. Endocrinol. 2022, 13, 827032. [Google Scholar] [CrossRef] [PubMed]
- Daniel, J.M.; Lindsey, S.H.; Mostany, R.; Schrader, L.A.; Zsombok, A. Cardiometabolic health, menopausal estrogen therapy and the brain: How effects of estrogens diverge in healthy and unhealthy preclinical models of aging. Front. Neuroendocrinol. 2023, 70, 101068. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, B.; Byemerwa, J.; Krebs, T.; Lim, F.; Chang, C.Y.; McDonnell, D.P. Estrogen Receptor Signaling in the Immune System. Endocr. Rev. 2023, 44, 117–141. [Google Scholar] [CrossRef]
- Kim, H.J.J.; Dickie, S.A.; Laprairie, R.B. Estradiol-dependent hypocretinergic/orexinergic behaviors throughout the estrous cycle. Psychopharmacology 2023, 240, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Bühler, M.; Stolz, A. Estrogens-Origin of Centrosome Defects in Human Cancer? Cells 2022, 11, 432. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, K.J.; Arao, Y.; Korach, K.S. Estrogen hormone physiology: Reproductive findings from estrogen receptor mutant mice. Reprod. Biol. 2014, 14, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Christenson, L.K.; Devoto, L. Cholesterol transport and steroidogenesis by the corpus luteum. Reprod. Biol. Endocrinol. 2003, 1, 90. [Google Scholar] [CrossRef]
- Ng, S.W.; Norwitz, G.A.; Pavlicev, M.; Tilburgs, T.; Simón, C.; Norwitz, E.R. Endometrial Decidualization: The Primary Driver of Pregnancy Health. Int. J. Mol. Sci. 2020, 21, 4092. [Google Scholar] [CrossRef]
- Tuckey, R.C. Progesterone synthesis by the human placenta. Placenta 2005, 26, 273–281. [Google Scholar] [CrossRef]
- Robertson, D.M.; Cahir, N.; Findlay, J.K.; Burger, H.G.; Groome, N. The biological and immunological characterization of inhibin A and B forms in human follicular fluid and plasma. J. Clin. Endocrinol. Metab. 1997, 82, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Walton, K.L.; Makanji, Y.; Wilce, M.C.; Chan, K.L.; Robertson, D.M.; Harrison, C.A. A common biosynthetic pathway governs the dimerization and secretion of inhibin and related transforming growth factor beta (TGFbeta) ligands. J. Biol. Chem. 2009, 284, 9311–9320. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Shao, L.E.; Lemas, V.; Yu, A.L.; Vaughan, J.; Rivier, J.; Vale, W. Importance of FSH-releasing protein and inhibin in erythrodifferentiation. Nature 1987, 330, 765–767. [Google Scholar] [CrossRef] [PubMed]
- Vassalli, A.; Matzuk, M.M.; Gardner, H.A.; Lee, K.F.; Jaenisch, R. Activin/inhibin beta B subunit gene disruption leads to defects in eyelid development and female reproduction. Genes. Dev. 1994, 8, 414–427. [Google Scholar] [CrossRef]
- Matzuk, M.M. Functional analysis of mammalian members of the transforming growth factor-beta superfamily. Trends Endocrinol. Metab. 1995, 6, 120–127. [Google Scholar] [CrossRef]
- Morikawa, M.; Derynck, R.; Miyazono, K. TGF-β and the TGF-β Family: Context-Dependent Roles in Cell and Tissue Physiology. Cold Spring Harb. Perspect. Biol. 2016, 8, a021873. [Google Scholar] [CrossRef]
- Wang, N.F.; Bogstad, J.W.; Petersen, M.R.; Pinborg, A.; Andersen, C.Y.; Løssl, K. Androgen and inhibin B levels during ovarian stimulation before and after 8 weeks of low-dose hCG priming in women with low ovarian reserve. Hum. Reprod. 2023, 38, 1807–1815. [Google Scholar] [CrossRef]
- Sehested, A.; Juul, A.A.; Andersson, A.M.; Petersen, J.H.; Jensen, T.K.; Müller, J.; Skakkebaek, N.E. Serum inhibin A and inhibin B in healthy prepubertal, pubertal, and adolescent girls and adult women: Relation to age, stage of puberty, menstrual cycle, follicle-stimulating hormone, luteinizing hormone, and estradiol levels. J. Clin. Endocrinol. Metab. 2000, 85, 1634–1640. [Google Scholar] [CrossRef]
- Hillier, S.G. Paracrine support of ovarian stimulation. Mol. Hum. Reprod. 2009, 15, 843–850. [Google Scholar] [CrossRef]
- Canipari, R.; Cellini, V.; Cecconi, S. The ovary feels fine when paracrine and autocrine networks cooperate with gonadotropins in the regulation of folliculogenesis. Curr. Pharm. Des. 2012, 18, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Padmanabhan, V.; Cardoso, R.C. Neuroendocrine, autocrine, and paracrine control of follicle-stimulating hormone secretion. Mol. Cell Endocrinol. 2020, 500, 110632. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Ohnishi, J. Molecular mechanism of follicle rupture during ovulation. Zoolog Sci. 1995, 12, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Canipari, R.; Strickland, S. Studies on the hormonal regulation of plasminogen activator production in the rat ovary. Endocrinology 1986, 118, 1652–1659. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Leung, A. Gonadotropins regulate plasminogen activator production by rat granulosa cells. Endocrinology 1983, 112, 1201–1207. [Google Scholar] [CrossRef]
- Lumsden, M.A.; Kelly, R.W.; Templeton, A.A.; Van Look, P.F.; Swanston, I.A.; Baird, D.T. Changes in the concentration of prostaglandins in preovulatory human follicles after administration of hCG. J. Reprod. Fertil. 1986, 77, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Espey, L.L.; Tanaka, N.; Adams, R.F.; Okamura, H. Ovarian hydroxyeicosatetraenoic acids compared with prostanoids and steroids during ovulation in rats. Am. J. Physiol. 1991, 260 Pt 1, E163–E169. [Google Scholar] [CrossRef]
- Borba, V.V.; Zandman-Goddard, G.; Shoenfeld, Y. Prolactin and autoimmunity: The hormone as an inflammatory cytokine. Best. Pract. Res. Clin. Endocrinol. Metab. 2019, 33, 101324. [Google Scholar] [CrossRef]
- Hu, S.; Duggavathi, R.; Zadworny, D. Regulatory Mechanisms Underlying the Expression of Prolactin Receptor in Chicken Granulosa Cells. PLoS ONE 2017, 12, e0170409. [Google Scholar] [CrossRef]
- Yang, R.; Duan, C.; Zhang, S.; Liu, Y.; Zhang, Y. Prolactin Regulates Ovine Ovarian Granulosa Cell Apoptosis by Affecting the Expression of MAPK12 Gene. Int. J. Mol. Sci. 2023, 24, 10269. [Google Scholar] [CrossRef]
- Legro, R.S. The new International Federation of Gynecology and Obstetrics (FIGO) classification of ovulatory disorders: Getting from here to there. Fertil. Steril. 2023, 119, 560–561. [Google Scholar] [CrossRef] [PubMed]
- Munro, M.G.; Balen, A.H.; Cho, S.; Critchley, H.O.D.; Díaz, I.; Ferriani, R.; Henry, L.; Mocanu, E.; van der Spuy, Z.M.; FIGO Committee on Menstrual Disorders and Related Health Impacts; et al. The FIGO ovulatory disorders classification system. Int. J. Gynaecol. Obstet. 2022, 159, 1–20. [Google Scholar] [CrossRef]
- Azziz, R. Introduction: Determinants of polycystic ovary syndrome. Fertil. Steril. 2016, 106, 4–5. [Google Scholar] [CrossRef] [PubMed]
- Deswal, R.; Narwal, V.; Dang, A.; Pundir, C.S. The Prevalence of Polycystic Ovary Syndrome: A Brief Systematic Review. J. Hum. Reprod. Sci. 2020, 13, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wu, Q.; Hao, Y.; Jiao, M.; Wang, X.; Jiang, S.; Han, L. Measuring the global disease burden of polycystic ovary syndrome in 194 countries: Global Burden of Disease Study 2017. Hum. Reprod. 2021, 36, 1108–1119. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, S.; Conigliaro, R.L. Polycystic Ovarian Syndrome. Med. Clin. N. Am. 2023, 107, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Smet, M.E.; McLennan, A. Rotterdam criteria, the end. Australas. J. Ultrasound Med. 2018, 21, 59–60. [Google Scholar] [CrossRef] [PubMed]
- Szukiewicz, D.; Trojanowski, S.; Kociszewska, A.; Szewczyk, G. Modulation of the Inflammatory Response in Polycystic Ovary Syndrome (PCOS)-Searching for Epigenetic Factors. Int. J. Mol. Sci. 2022, 23, 14663. [Google Scholar] [CrossRef]
- Harada, M. Pathophysiology of polycystic ovary syndrome revisited: Current understanding and perspectives regarding future research. Reprod. Med. Biol. 2022, 21, e12487. [Google Scholar] [CrossRef]
- Davoudi, Z.; Araghi, F.; Vahedi, M.; Mokhtari, N.; Gheisari, M. Prolactin Level in Polycystic Ovary Syndrome (PCOS): An approach to the diagnosis and management. Acta Biomed. 2021, 92, e2021291. [Google Scholar] [CrossRef]
- Kim, S.I.; Yoon, J.H.; Park, D.C.; Yang, S.H.; Kim, Y.I. What is the optimal prolactin cutoff for predicting the presence of a pituitary adenoma in patients with polycystic ovary syndrome? Int. J. Med. Sci. 2023, 20, 463–467. [Google Scholar] [CrossRef]
- Liu, Y.X.; Peng, X.R.; Chen, Y.J.; Carrico, W.; Ny, T. Prolactin delays gonadotrophin-induced ovulation and down-regulates expression of plasminogen-activator system in ovary. Hum. Reprod. 1997, 12, 2748–2755. [Google Scholar] [CrossRef]
- Chang, S.; Copperman, A.B. New insights into human prolactin pathophysiology: Genomics and beyond. Curr. Opin. Obstet. Gynecol. 2019, 31, 207–211. [Google Scholar] [CrossRef]
- Thapa, S.; Bhusal, K. Hyperprolactinemia. In StatPearls [Internet]; Updated 24 July 2023; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK537331/ (accessed on 29 January 2024).
- Stojkovic, M.; Radmanovic, B.; Jovanovic, M.; Janjic, V.; Muric, N.; Ristic, D.I. Risperidone Induced Hyperprolactinemia: From Basic to Clinical Studies. Front. Psychiatry 2022, 13, 874705. [Google Scholar] [CrossRef]
- Malik, A.A.; Aziz, F.; Beshyah, S.A.; Aldahmani, K.M. Aetiologies of Hyperprolactinaemia: A retrospective analysis from a tertiary healthcare centre. Sultan Qaboos Univ. Med. J. 2019, 19, e129–e134. [Google Scholar] [CrossRef]
- Aliberti, L.; Gagliardi, I.; Dorizzi, R.M.; Pizzicotti, S.; Bondanelli, M.; Zatelli, M.C.; Ambrosio, M.R. Hypeprolactinemia: Still an insidious diagnosis. Endocrine 2021, 72, 928–931. [Google Scholar] [CrossRef]
- Grattan, D.R.; Steyn, F.J.; Kokay, I.C.; Anderson, G.M.; Bunn, S.J. Pregnancy-induced adaptation in the neuroendocrine control of prolactin secretion. J. Neuroendocrinol. 2008, 20, 497–507. [Google Scholar] [CrossRef] [PubMed]
- Kirk, S.E.; Xie, T.Y.; Steyn, F.J.; Grattan, D.R.; Bunn, S.J. Restraint stress increases prolactin-mediated phosphorylation of signal transducer and activator of transcription 5 in the hypothalamus and adrenal cortex in the male mouse. J. Neuroendocrinol. 2017, 29. [Google Scholar] [CrossRef] [PubMed]
- Hackney, A.C.; Saeidi, A. The thyroid axis, prolactin, and exercise in humans. Curr. Opin. Endocr. Metab. Res. 2019, 9, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Mogavero, M.P.; Cosentino, F.I.I.; Lanuzza, B.; Tripodi, M.; Lanza, G.; Aricò, D.; DelRosso, L.M.; Pizza, F.; Plazzi, G.; Ferri, R. Increased Serum Prolactin and Excessive Daytime Sleepiness: An Attempt of Proof-of-Concept Study. Brain Sci. 2021, 11, 1574. [Google Scholar] [CrossRef] [PubMed]
- Kapcala, L.P.; Lakshmanan, M.C. Thoracic stimulation and prolactin secretion. J. Endocrinol. Investig. 1989, 12, 815–821. [Google Scholar] [CrossRef]
- Nteeba, J.; Kubota, K.; Wang, W.; Zhu, H.; Vivian, J.; Dai, G.; Soares, M. Pancreatic prolactin receptor signaling regulates maternal glucose homeostasis. J. Endocrinol. 2019, 241, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Krüger, T.H.; Hartmann, U.; Schedlowski, M. Prolactinergic and dopaminergic mechanisms underlying sexual arousal and orgasm in humans. World J. Urol. 2005, 23, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Samperi, I.; Lithgow, K.; Karavitaki, N. Hyperprolactinaemia. J. Clin. Med. 2019, 8, 2203. [Google Scholar] [CrossRef] [PubMed]
- Shukla, P.; Bulsara, K.R.; Luthra, P. Pituitary Hyperplasia in Severe Primary Hypothyroidism: A Case Report and Review of the Literature. Case Rep. Endocrinol. 2019, 2019, 2012546. [Google Scholar] [CrossRef] [PubMed]
- Dourado, M.; Cavalcanti, F.; Vilar, L.; Cantilino, A. Relationship between Prolactin, Chronic Kidney Disease, and Cardiovascular Risk. Int. J. Endocrinol. 2020, 2020, 9524839. [Google Scholar] [CrossRef] [PubMed]
- Adachi, N.; Lei, B.; Deshpande, G.; Seyfried, F.J.; Shimizu, I.; Nagaro, T.; Arai, T. Uraemia suppresses central dopaminergic metabolism and impairs motor activity in rats. Intensive Care Med. 2001, 27, 1655–1660. [Google Scholar] [CrossRef] [PubMed]
- Jha, S.K.; Kannan, S. Serum prolactin in patients with liver disease in comparison with healthy adults: A preliminary cross-sectional study. Int. J. Appl. Basic. Med. Res. 2016, 6, 8–10. [Google Scholar] [CrossRef]
- Pekic, S.; Miljic, D.; Popovic, V. Hypopituitarism Following Cranial Radiotherapy. In Endotext [Internet]; Feingold, K.R., Anawalt, B., Blackman, M.R., Boyce, A., Chrousos, G., Corpas, E., de Herder, W.W., Dhatariya, K., Dungan, K., Hofland, J., et al., Eds.; Updated 9 August 2021; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. Available online: https://www.ncbi.nlm.nih.gov/books/NBK532082/ (accessed on 15 November 2023).
- Niculescu, D.A.; Gheorghiu, M.L.; Poiana, C. Radiotherapy in aggressive or dopamine agonists resistant prolactinomas; is it still worthwhile? Eur. J. Endocrinol. 2023, 188, R88–R97. [Google Scholar] [CrossRef]
- Rutberg, L.; Fridén, B.; Karlsson, A.K. Amenorrhoea in newly spinal cord injured women: An effect of hyperprolactinaemia? Spinal Cord. 2008, 46, 189–191. [Google Scholar] [CrossRef][Green Version]
- Arinola, E.; Adeleye, J.O.; Lawal, O.A.; Oluwakemi, B.A. Hyperprolactinemia following traumatic myelopathy. Afr. J. Endocrinol. Metab. 2022, 12, 93–95. [Google Scholar] [CrossRef]
- Hermann, A.L.; Fell, G.L.; Kemény, L.V.; Fung, C.Y.; Held, K.D.; Biggs, P.J.; Rivera, P.D.; Bilbo, S.D.; Igras, V.; Willers, H.; et al. β-Endorphin mediates radiation therapy fatigue. Sci. Adv. 2022, 8, eabn6025. [Google Scholar] [CrossRef]
- Panahi, Y.; Fathi, E.; Shafiian, M.A. The link between seizures and prolactin: A study on the effects of anticonvulsant medications on hyperprolactinemia in rats. Epilepsy Res. 2023, 196, 107206. [Google Scholar] [CrossRef] [PubMed]
- Bahceci, M.; Tuzcu, A.; Bahceci, S.; Tuzcu, S. Is hyperprolactinemia associated with insulin resistance in non-obese patients with polycystic ovary syndrome? J. Endocrinol. Investig. 2003, 26, 655–659. [Google Scholar] [CrossRef] [PubMed]
- Le Bescont, A.; Vitte, A.-L.; Debernardi, A.; Curtet, S.; Buchou, T.; Vayr, J.; de Reyniès, A.; Ito, A.; Guardiola, P.; Brambilla, C.; et al. Receptor-Independent Ectopic Activity of Prolactin Predicts Aggressive Lung Tumors and Indicates HDACi-Based Therapeutic Strategies. Antioxid. Redox Signal. 2015, 23, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Letson, G.W.; Moore, D.C. Galactorrhea secondary to chest wall surgery in an adolescent. J. Adolesc. Health Care 1984, 5, 277–278. [Google Scholar] [CrossRef]
- Bushe, C.; Shaw, M.; Peveler, R.C. A review of the association between antipsychotic use and hyperprolactinaemia. J. Psychopharmacol. 2008, 22 (Suppl. S2), 46–55. [Google Scholar] [CrossRef]
- Junqueira, D.R.; Bennett, D.; Huh, S.Y.; Casañas IComabella, C. Clinical Presentations of Drug-Induced Hyperprolactinaemia: A Literature Review. Pharm. Med. 2023, 37, 153–166. [Google Scholar] [CrossRef]
- Kim, S.; Jeong, J.H.; Um, Y.H.; Kim, T.W.; Seo, H.J.; Hong, S.C. Prolactin Level Changes according to Atypical Antipsychotics Use: A Study Based on Clinical Data Warehouse. Clin. Psychopharmacol. Neurosci. 2023, 21, 769–777. [Google Scholar] [CrossRef]
- Bhattacharjee, S.; Biswas, R.; Mandal, N. Selective serotonin reuptake inhibitor-induced galactorrhea with hyperprolactinemia. Indian. J. Psychiatry 2021, 63, 613–616. [Google Scholar] [CrossRef]
- Ashbury, J.E.; Lévesque, L.E.; Beck, P.A.; Aronson, K.J. Selective Serotonin Reuptake Inhibitor (SSRI) Antidepressants, Prolactin and Breast Cancer. Front. Oncol. 2012, 2, 177. [Google Scholar] [CrossRef] [PubMed]
- Torre, D.L.; Falorni, A. Pharmacological causes of hyperprolactinemia. Ther. Clin. Risk Manag. 2007, 3, 929–951. [Google Scholar]
- Rodier, C.; Courbière, B.; Fernandes, S.; Vermalle, M.; Florence, B.; Resseguier, N.; Brue, T.; Cuny, T. Metoclopramide Test in Hyperprolactinemic Women with Polycystic Ovarian Syndrome: Old Wine into New Bottles? Front. Endocrinol. 2022, 13, 832361. [Google Scholar] [CrossRef] [PubMed]
- Kertesz, E.; Somoza, G.M.; D’Eramo, J.; Libertun, C. Further evidence for endogenous hypothalamic serotonergic neurons involved in the cimetidine-induced prolactin release in the rat. Brain Res. 1987, 413, 10–14. [Google Scholar] [CrossRef]
- Meltzer, H.Y.; Maes, M.; Lee, M.A. The cimetidine-induced increase in prolactin secretion in schizophrenia: Effect of clozapine. Psychopharmacology 1993, 112 (Suppl. S1), S95–S104. [Google Scholar] [CrossRef]
- Gupta, M.; Al Khalili, Y. Methyldopa. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Kelley, S.R.; Kamal, T.J.; Molitch, M.E. Mechanism of verapamil calcium channel blockade-induced hyperprolactinemia. Am. J. Physiol. 1996, 270 Pt 1, E96–E100. [Google Scholar] [CrossRef] [PubMed]
- Veselinović, T.; Schorn, H.; Vernaleken, I.B.; Schiffl, K.; Klomp, M.; Gründer, G. Impact of different antidopaminergic mechanisms on the dopaminergic control of prolactin secretion. J. Clin. Psychopharmacol. 2011, 31, 214–220. [Google Scholar] [CrossRef]
- Ren, N.; Carratala-Ros, C.; Ecevitoglu, A.; Rotolo, R.A.; Edelstein, G.A.; Presby, R.E.; Stevenson, I.H.; Chrobak, J.J.; Salamone, J.D. Effects of the dopamine depleting agent tetrabenazine on detailed temporal parameters of effort-related choice responding. J. Exp. Anal. Behav. 2022, 117, 331–345. [Google Scholar] [CrossRef]
- Enjalbert, A.; Ruberg, M.; Arancibia, S.; Priam, M.; Kordon, C. Endogenous opiates block dopamine inhibition of prolactin secretion in vitro. Nature 1979, 280, 595–597. [Google Scholar] [CrossRef]
- Arbogast, L.A.; Voogt, J.L. Endogenous opioid peptides contribute to suckling-induced prolactin release by suppressing tyrosine hydroxylase activity and messenger ribonucleic acid levels in tuberoinfundibular dopaminergic neurons. Endocrinology 1998, 139, 2857–2862. [Google Scholar] [CrossRef]
- Chourpiliadi, C.; Paparodis, R. Physiology, Pituitary Issues During Pregnancy. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Perez, P.A.; Petiti, J.P.; Picech, F.; Guido, C.B.; dVSosa, L.; Grondona, E.; Mukdsi, J.H.; De Paul, A.L.; Torres, A.I.; Gutierrez, S. Estrogen receptor β regulates the tumoral suppressor PTEN to modulate pituitary cell growth. J. Cell Physiol. 2018, 233, 1402–1413. [Google Scholar] [CrossRef]
- Lyons, D.J.; Broberger, C. TIDAL WAVES: Network mechanisms in the neuroendocrine control of prolactin release. Front. Neuroendocrinol. 2014, 35, 420–438. [Google Scholar] [CrossRef]
- Pérez, C.T.; Ferraris, J.; van Lunteren, J.A.; Hellysaz, A.; Iglesias, M.J.; Broberger, C. Adaptive Resetting of Tuberoinfundibular Dopamine (TIDA) Network Activity during Lactation in Mice. J. Neurosci. 2020, 40, 3203–3216. [Google Scholar] [CrossRef]
- Lyons, D.J.; Ammari, R.; Hellysaz, A.; Broberger, C. Serotonin and Antidepressant SSRIs Inhibit Rat Neuroendocrine Dopamine Neurons: Parallel Actions in the Lactotrophic Axis. J. Neurosci. 2016, 36, 7392–7406. [Google Scholar] [CrossRef] [PubMed]
- Bernard, V.; Lamothe, S.; Beau, I.; Guillou, A.; Martin, A.; Le Tissier, P.; Grattan, D.; Young, J.; Binart, N. Autocrine actions of prolactin contribute to the regulation of lactotroph function in vivo. FASEB J. 2018, 32, 4791–4797. [Google Scholar] [CrossRef] [PubMed]
- Yatavelli, R.K.R.; Bhusal, K. Prolactinoma. In StatPearls [Internet]; Updated 31 July 2023; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK459347 (accessed on 15 November 2023).
- Lyu, L.; Yin, S.; Hu, Y.; Chen, C.; Jiang, Y.; Yu, Y.; Ma, W.; Wang, Z.; Jiang, S.; Zhou, P. Hyperprolactinemia in clinical non-functional pituitary macroadenomas: A STROBE-compliant study. Medicine 2020, 99, e22673. [Google Scholar] [CrossRef] [PubMed]
- Bergsneider, M.; Mirsadraei, L.; Yong, W.H.; Salamon, N.; Linetsky, M.; Wang, M.B.; McArthur, D.L.; Heaney, A.P. The pituitary stalk effect: Is it a passing phenomenon? J. Neurooncol. 2014, 117, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Schury, M.P.; Adigun, R. Sheehan Syndrome. In StatPearls [Internet]; Updated 4 September 2023; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK459166/ (accessed on 5 December 2023).
- Montejo, Á.L.; Arango, C.; Bernardo, M.; Carrasco, J.L.; Crespo-Facorro, B.; Cruz, J.J.; Del Pino-Montes, J.; García-Escudero, M.A.; Garcia-Rizo, C.; González-Pinto, A.; et al. Multidisciplinary consensus on the therapeutic recommendations for iatrogenic hyperprolactinemia secondary to antipsychotics. Front. Neuroendocrinol. 2017, 45, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Cano, A.M.G.; Rosillo, M.; Lozano, A.G.; Mendiguchía, L.J.; Pinedo, M.M.; Torres, A.R.; Araujo-Castro, M. Pharmacological hyperprolactinemia: A retrospective analysis of 501 hyperprolactinemia cases in primary care setting. Naunyn Schmiedebergs Arch. Pharmacol. 2023. [Google Scholar] [CrossRef]
- Montejo, Á.L.; Arango, C.; Bernardo, M.; Carrasco, J.L.; Crespo-Facorro, B.; Cruz, J.J.; del Pino, J.; Escudero, M.A.G.; Rizo, C.G.; González-Pinto, A.; et al. Spanish consensus on the risks and detection of antipsychotic drug-related hyperprolactinaemia. Rev. Psiquiatr. Salud Ment. 2016, 9, 158–173. [Google Scholar] [CrossRef] [PubMed]
- Wiciński, M.; Malinowski, B.; Puk, O.; Socha, M.; Słupski, M. Methyldopa as an inductor of postpartum depression and maternal blues: A review. Biomed. Pharmacother. 2020, 127, 110196. [Google Scholar] [CrossRef]
- Petersenn, S.; Fleseriu, M.; Casanueva, F.F.; Giustina, A.; Biermasz, N.; Biller, B.M.K.; Bronstein, M.; Chanson, P.; Fukuoka, H.; Gadelha, M.; et al. Diagnosis and management of prolactin-secreting pituitary adenomas: A Pituitary Society international Consensus Statement. Nat. Rev. Endocrinol. 2023, 19, 722–740, Erratum in Nat. Rev. Endocrinol. 2024, 20, 62. [Google Scholar] [CrossRef]
- Chanson, P.; Borson-Chazot, F.; Chabre, O.; Estour, B. Drug treatment of hyperprolactinemia. Ann. Endocrinol. 2007, 68, 113–117. [Google Scholar] [CrossRef]
- Concepción-Zavaleta, M.J.; Coronado-Arroyo, J.C.; Quiroz-Aldave, J.E.; Durand-Vásquez, M.D.C.; Ildefonso-Najarro, S.P.; Rafael-Robles, L.D.P.; Concepción-Urteaga, L.A.; Gamarra-Osorio, E.R.; Suárez-Rojas, J.; Paz-Ibarra, J. Endocrine factors associated with infertility in women: An updated review. Expert. Rev. Endocrinol. Metab. 2023, 18, 399–417. [Google Scholar] [CrossRef]
- Chutpiboonwat, P.; Yenpinyosuk, K.; Sridama, V.; Kunjan, S.; Klaimukh, K.; Snabboon, T. Macroprolactinemia in patients with hyperprolactinemia: An experience from a single tertiary center. Pan Afr. Med. J. 2020, 36, 8. [Google Scholar] [CrossRef] [PubMed]
- Richa, V.; Rahul, G.; Sarika, A. Macroprolactin; a frequent cause of misdiagnosed hyperprolactinemia in clinical practice. J. Reprod. Infertil. 2010, 11, 161–167. [Google Scholar] [PubMed]
- Soh, N.A.A.C.; Yaacob, N.M.; Omar, J.; Jelani, A.M.; Shafii, N.; Ismail, T.S.T.; Azman, W.N.W.; Ghazali, A.K. Global Prevalence of Macroprolactinemia among Patients with Hyperprolactinemia: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2020, 17, 8199. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, H.; Yang, W.; Jin, W.; Yu, W.; Wang, W.; Zhang, K.; Song, G. A New Method of Using Polyethylene Glycol (PEG) Precipitation of Macroprolactin to Detect Genuine Hyperprolactinemia. J. Clin. Lab. Anal. 2016, 30, 1169–1174. [Google Scholar] [CrossRef] [PubMed]
- Shimatsu, A.; Hattori, N. Macroprolactinemia: Diagnostic, clinical, and pathogenic significance. Clin. Dev. Immunol. 2012, 2012, 167132. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, Y.; Nakamura, Y.; Yamada, H.; Ando, M.; Ubukata, Y.; Oda, T.; Suzuki, M. Possible contribution of prolactin in the process of ovulation and oocyte maturation. Horm. Res. 1991, 35 (Suppl. S1), 22–32. [Google Scholar] [CrossRef]
- Stocco, C. The long and short of the prolactin receptor: The corpus luteum needs them both! Biol. Reprod. 2012, 86, 85. [Google Scholar] [CrossRef]
- Ezoe, K.; Fujiwara, N.; Miki, T.; Kato, K. Post-warming culture of human vitrified blastocysts with prolactin improves trophoblast outgrowth. Reprod. Biol. Endocrinol. 2023, 21, 6. [Google Scholar] [CrossRef]
- Alila, H.W.; Rogo, K.O.; Gombe, S. Effects of prolactin on steroidogenesis by human luteal cells in culture. Fertil. Steril. 1987, 47, 947–955. [Google Scholar] [CrossRef]
- Grattan, D.R.; Jasoni, C.L.; Liu, X.; Anderson, G.M.; Herbison, A.E. Prolactin regulation of gonadotropin-releasing hormone neurons to suppress luteinizing hormone secretion in mice. Endocrinology 2007, 148, 4344–4351. [Google Scholar] [CrossRef]
- Brown, R.S.; Piet, R.; Herbison, A.E.; Grattan, D.R. Differential actions of prolactin on electrical activity and intracellular signal transduction in hypothalamic neurons. Endocrinology 2012, 153, 2375–2384. [Google Scholar] [CrossRef] [PubMed]
- Cortasa, S.A.; Schmidt, A.R.; Proietto, S.; Corso, M.C.; Inserra, P.I.F.; Giorgio, N.P.D.; Lux-Lantos, V.; Vitullo, A.D.; Halperin, J.; Dorfman, V.B. Hypothalamic GnRH expression and pulsatility depends on a balance of prolactin receptors in the plains vizcacha, Lagostomus maximus. J. Comp. Neurol. 2023, 531, 720–742. [Google Scholar] [CrossRef]
- Xie, Q.; Kang, Y.; Zhang, C.; Xie, Y.; Wang, C.; Liu, J.; Yu, C.; Zhao, H.; Huang, D. The Role of Kisspeptin in the Control of the Hypothalamic-Pituitary-Gonadal Axis and Reproduction. Front. Endocrinol. 2022, 13, 925206. [Google Scholar] [CrossRef] [PubMed]
- Iwasa, T.; Matsuzaki, T.; Yano, K.; Mayila, Y.; Irahara, M. The roles of kisspeptin and gonadotropin inhibitory hormone in stress-induced reproductive disorders. Endocr. J. 2018, 65, 133–140. [Google Scholar] [CrossRef]
- Nejad, S.Z.; Tehrani, F.R.; Zadeh-Vakili, A. The Role of Kisspeptin in Female Reproduction. Int. J. Endocrinol. Metab. 2017, 15, e44337. [Google Scholar] [CrossRef]
- Franssen, D.; Tena-Sempere, M. The kisspeptin receptor: A key G-protein-coupled receptor in the control of the reproductive axis. Best. Pract. Res. Clin. Endocrinol. Metab. 2018, 32, 107–123. [Google Scholar] [CrossRef]
- de Tassigny, X.D.; Colledge, W.H. The role of kisspeptin signaling in reproduction. Physiology 2010, 25, 207–217, Erratum in Physiology 2010, 25, 378. [Google Scholar] [CrossRef][Green Version]
- Goodman, R.L.; Hileman, S.M.; Nestor, C.C.; Porter, K.L.; Connors, J.M.; Hardy, S.L.; Millar, R.P.; Cernea, M.; Coolen, L.M.; Lehman, M.N. Kisspeptin, neurokinin B, and dynorphin act in the arcuate nucleus to control activity of the GnRH pulse generator in ewes. Endocrinology 2013, 154, 4259–4269. [Google Scholar] [CrossRef] [PubMed]
- Moore, A.M.; Coolen, L.M.; Lehman, M.N. Kisspeptin/Neurokinin B/Dynorphin (KNDy) cells as integrators of diverse internal and external cues: Evidence from viral-based monosynaptic tract-tracing in mice. Sci. Rep. 2019, 9, 14768. [Google Scholar] [CrossRef] [PubMed]
- Merkley, C.M.; Porter, K.L.; Coolen, L.M.; Hileman, S.M.; Billings, H.J.; Drews, S.; Goodman, R.L.; Lehman, M.N. KNDy (kisspeptin/neurokinin B/dynorphin) neurons are activated during both pulsatile and surge secretion of LH in the ewe. Endocrinology 2012, 153, 5406–5414. [Google Scholar] [CrossRef] [PubMed]
- Mittelman-Smith, M.A.; Krajewski-Hall, S.J.; McMullen, N.T.; Rance, N.E. Neurokinin 3 Receptor-Expressing Neurons in the Median Preoptic Nucleus Modulate Heat-Dissipation Effectors in the Female Rat. Endocrinology 2015, 156, 2552–2562. [Google Scholar] [CrossRef] [PubMed]
- Duittoz, A.; Cayla, X.; Fleurot, R.; Lehnert, J.; Khadra, A. Gonadotrophin-releasing hormone and kisspeptin: It takes two to tango. J. Neuroendocrinol. 2021, 33, e13037. [Google Scholar] [CrossRef] [PubMed]
- Constantin, S. Physiology of the gonadotrophin-releasing hormone (GnRH) neurone: Studies from embryonic GnRH neurones. J. Neuroendocrinol. 2011, 23, 542–553. [Google Scholar] [CrossRef] [PubMed]
- de Tassigny, X.D.; Fagg, L.A.; Carlton, M.B.L.; Colledge, W.H. Kisspeptin can stimulate gonadotropin-releasing hormone (GnRH) release by a direct action at GnRH nerve terminals. Endocrinology 2008, 149, 3926–3932. [Google Scholar] [CrossRef] [PubMed]
- Tng, E.L. Kisspeptin signalling and its roles in humans. Singap. Med. J. 2015, 56, 649–656. [Google Scholar] [CrossRef]
- Seminara, S.B.; Messager, S.; Chatzidaki, E.E.; Thresher, R.R.; Acierno, J.S.; Shagoury, J.K.; Bo-Abbas, Y.; Kuohung, W.; Schwinof, K.M.; Hendrick, A.G.; et al. The GPR54 gene as a regulator of puberty. N. Engl. J. Med. 2003, 349, 1614–1627. [Google Scholar] [CrossRef]
- de Roux, N.; Genin, E.; Carel, J.C.; Matsuda, F.; Chaussain, J.L.; Milgrom, E. Hypogonadotropic hypogonadism due to loss of function of the KiSS1-derived peptide receptor GPR54. Proc. Natl. Acad. Sci. USA 2003, 100, 10972–10976. [Google Scholar] [CrossRef]
- Funes, S.; Hedrick, J.A.; Vassileva, G.; Markowitz, L.; Abbondanzo, S.; Golovko, A.; Yang, S.; Monsma, F.J.; Gustafson, E.L. The KiSS-1 receptor GPR54 is essential for the development of the murine reproductive system. Biochem. Biophys. Res. Commun. 2003, 312, 1357–1363. [Google Scholar] [CrossRef]
- Teles, M.G.; Bianco, S.D.; Brito, V.N.; Trarbach, E.B.; Kuohung, W.; Xu, S.; Seminara, S.B.; Mendonca, B.B.; Kaiser, U.B.; Latronico, A.C. A GPR54-activating mutation in a patient with central precocious puberty. N. Engl. J. Med. 2008, 358, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, M.; Jaillardon, L. Pathogenesis of the crosstalk between reproductive function and stress in animals-part 1: Hypothalamo-pituitary-adrenal axis, sympatho-adrenomedullary system and kisspeptin. Reprod. Domest. Anim. 2023, 58 (Suppl. S2), 176–183. [Google Scholar] [CrossRef]
- Brown, R.S.; Herbison, A.E.; Grattan, D.R. Prolactin regulation of kisspeptin neurones in the mouse brain and its role in the lactation-induced suppression of kisspeptin expression. J. Neuroendocrinol. 2014, 26, 898–908. [Google Scholar] [CrossRef] [PubMed]
- Araujo-Lopes, R.; Crampton, J.R.; Aquino, N.S.; Miranda, R.M.; Kokay, I.C.; Reis, A.M.; Franci, C.R.; Grattan, D.R.; Szawka, R.E. Prolactin regulates kisspeptin neurons in the arcuate nucleus to suppress LH secretion in female rats. Endocrinology 2014, 155, 1010–1020. [Google Scholar] [CrossRef]
- Kokay, I.C.; Petersen, S.L.; Grattan, D.R. Identification of prolactin-sensitive GABA and kisspeptin neurons in regions of the rat hypothalamus involved in the control of fertility. Endocrinology 2011, 152, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Rao, A.; Pereira, A.; Clarke, I.J.; Smith, J.T. Kisspeptin cells in the ovine arcuate nucleus express prolactin receptor but not melatonin receptor. J. Neuroendocrinol. 2011, 23, 871–882. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, P.; Ladyman, S.R.; McFadden, S.; Larsen, C.; Aung, Z.K.; Brown, R.S.E.; Bunn, S.J.; Grattan, D.R. Prolactin receptor-mediated activation of pSTAT5 in the pregnant mouse brain. J. Neuroendocrinol. 2020, 32, e12901. [Google Scholar] [CrossRef]
- Calik-Ksepka, A.; Stradczuk, M.; Czarnecka, K.; Grymowicz, M.; Smolarczyk, R. Lactational Amenorrhea: Neuroendocrine Pathways Controlling Fertility and Bone Turnover. Int. J. Mol. Sci. 2022, 23, 1633. [Google Scholar] [CrossRef]
- Grattan, D.R.; Szawka, R.E. Kisspeptin and Prolactin. Semin. Reprod. Med. 2019, 37, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Hara, T.; Kanasaki, H.; Tumurbaatar, T.; Oride, A.; Okada, H.; Kyo, S. Role of kisspeptin and Kiss1R in the regulation of prolactin gene expression in rat somatolactotroph GH3 cells. Endocrine 2019, 63, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Ammari, R.; Broberger, C. Pre- and post-synaptic modulation by GABAB receptors of rat neuroendocrine dopamine neurones. J. Neuroendocrinol. 2020, 32, e12881. [Google Scholar] [CrossRef] [PubMed]
- Aquino, N.S.S.; Kokay, I.C.; Perez, C.T.; Ladyman, S.R.; Henriques, P.C.; Silva, J.F.; Broberger, C.; Grattan, D.R.; Szawka, R.E. Kisspeptin Stimulation of Prolactin Secretion Requires Kiss1 Receptor but Not in Tuberoinfundibular Dopaminergic Neurons. Endocrinology 2019, 160, 522–533. [Google Scholar] [CrossRef]
- Ribeiro, A.B.; Leite, C.M.; Kalil, B.; Franci, C.R.; Anselmo-Franci, J.A.; Szawka, R.E. Kisspeptin regulates tuberoinfundibular dopaminergic neurones and prolactin secretion in an oestradiol-dependent manner in male and female rats. J. Neuroendocrinol. 2015, 27, 88–99. [Google Scholar] [CrossRef] [PubMed]
- Hoskova, K.; Kayton Bryant, N.; Chen, M.E.; Nachtigall, L.B.; Lippincott, M.F.; Balasubramanian, R.; Seminara, S.B. Kisspeptin Overcomes GnRH Neuronal Suppression Secondary to Hyperprolactinemia in Humans. J. Clin. Endocrinol. Metab. 2022, 107, e3515–e3525, Erratum in J. Clin. Endocrinol. Metab. 2022, 107, e3543. [Google Scholar] [CrossRef]
- Rezaei-Tazangi, F.; Kooshesh, L.; Tayyebiazar, A.; Taghizabet, N.; Tavakoli, A.; Hassanpour, A.; Aliakbari, F.; Kharazinejad, E.; Sharifi, A.M. Effects of kisspeptin on the maturation of human ovarian primordial follicles in vitro. Zygote 2023, 32, 66–70. [Google Scholar] [CrossRef]
- Sonigo, C.; Bouilly, J.; Carré, N.; Tolle, V.; Caraty, A.; Tello, J.; Simony-Conesa, F.J.; Millar, R.; Young, J.; Binart, N. Hyperprolactinemia-induced ovarian acyclicity is reversed by kisspeptin administration. J. Clin. Investig. 2012, 122, 3791–3795. [Google Scholar] [CrossRef]
- Uenoyama, Y.; Pheng, V.; Tsukamura, H.; Maeda, K.I. The roles of kisspeptin revisited: Inside and outside the hypothalamus. J. Reprod. Dev. 2016, 62, 537–545. [Google Scholar] [CrossRef]
- Yang, F.; Zhao, S.; Wang, P.; Xiang, W. Hypothalamic neuroendocrine integration of reproduction and metabolism in mammals. J. Endocrinol. 2023, 258, e230079. [Google Scholar] [CrossRef] [PubMed]
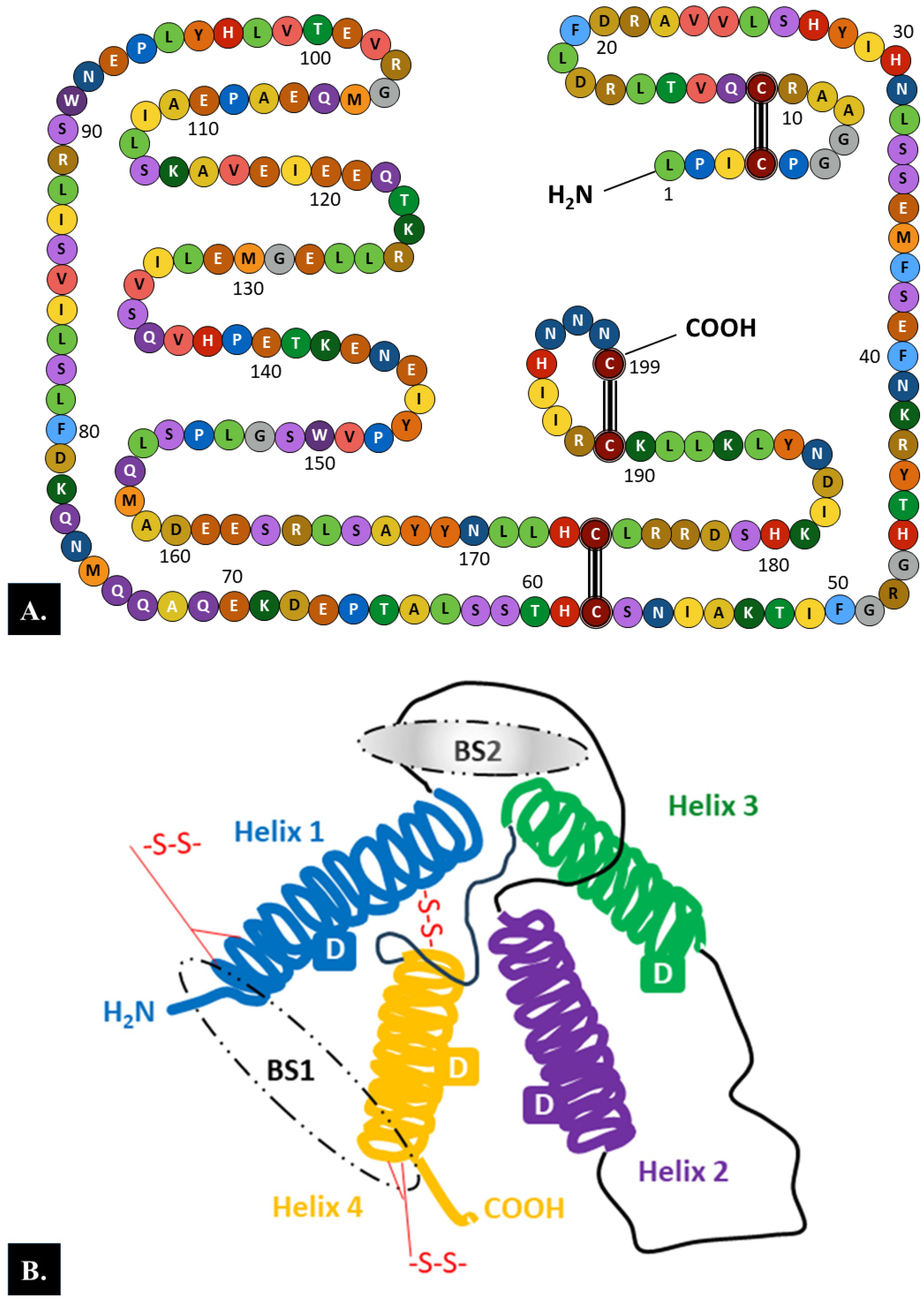
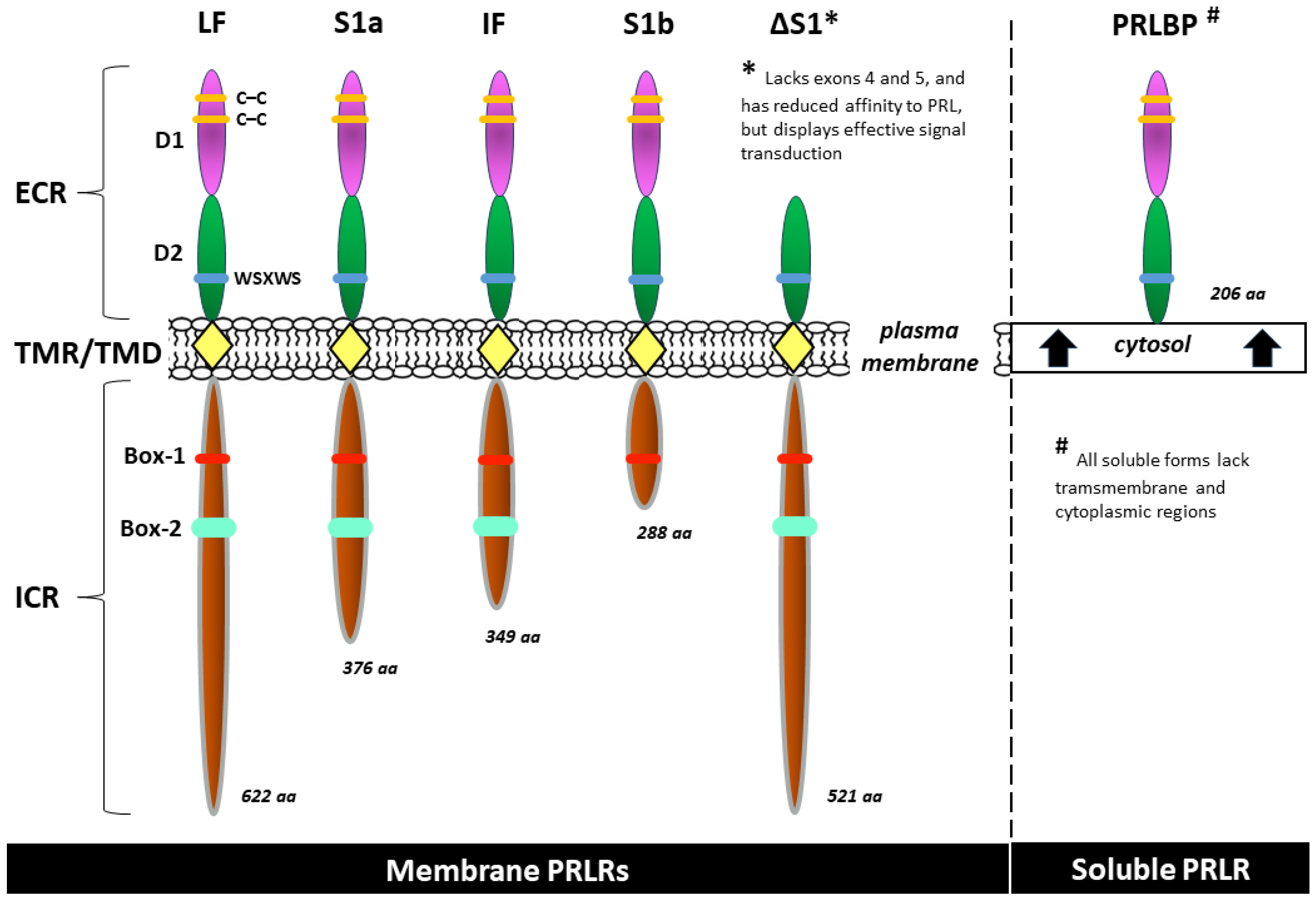
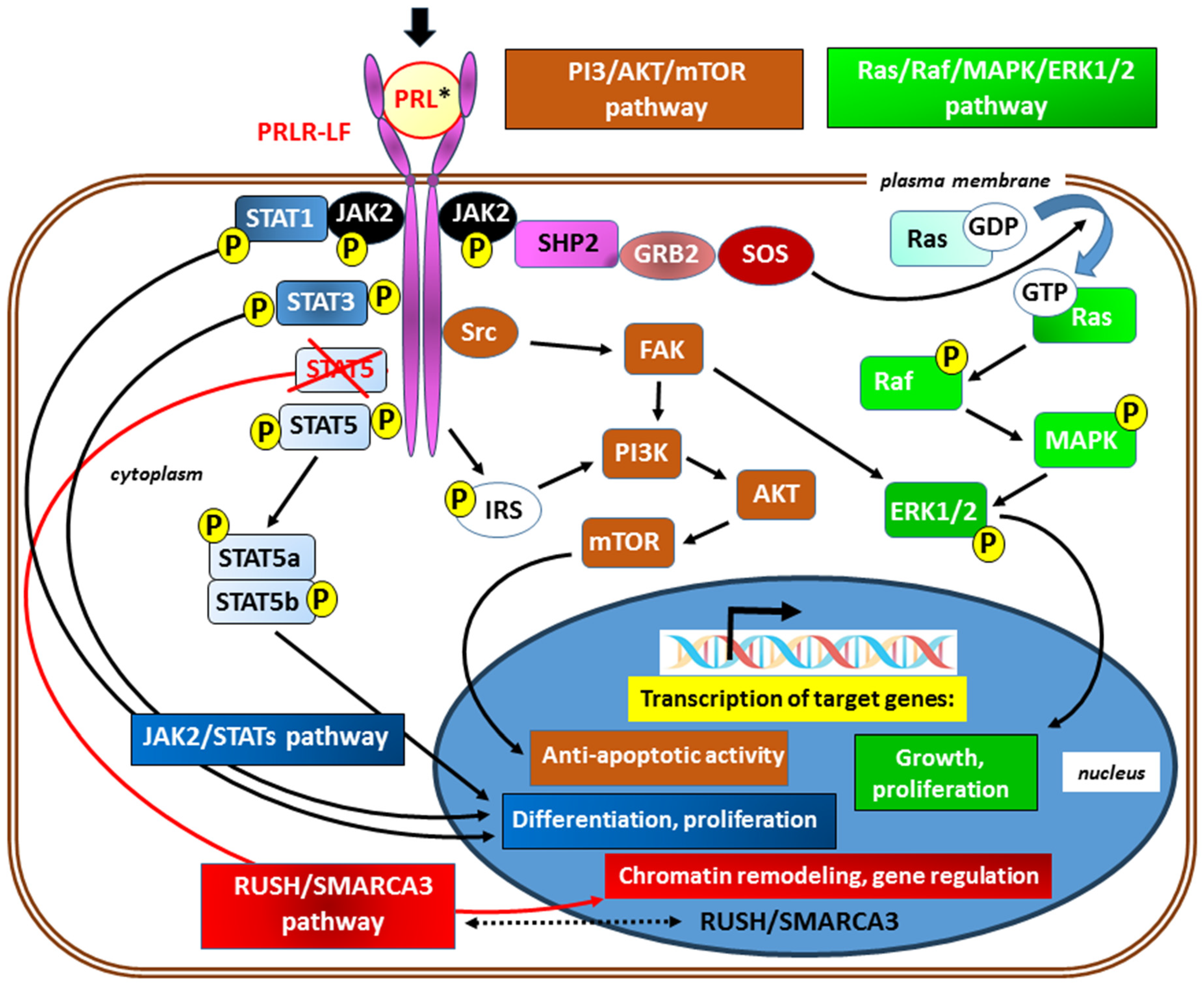

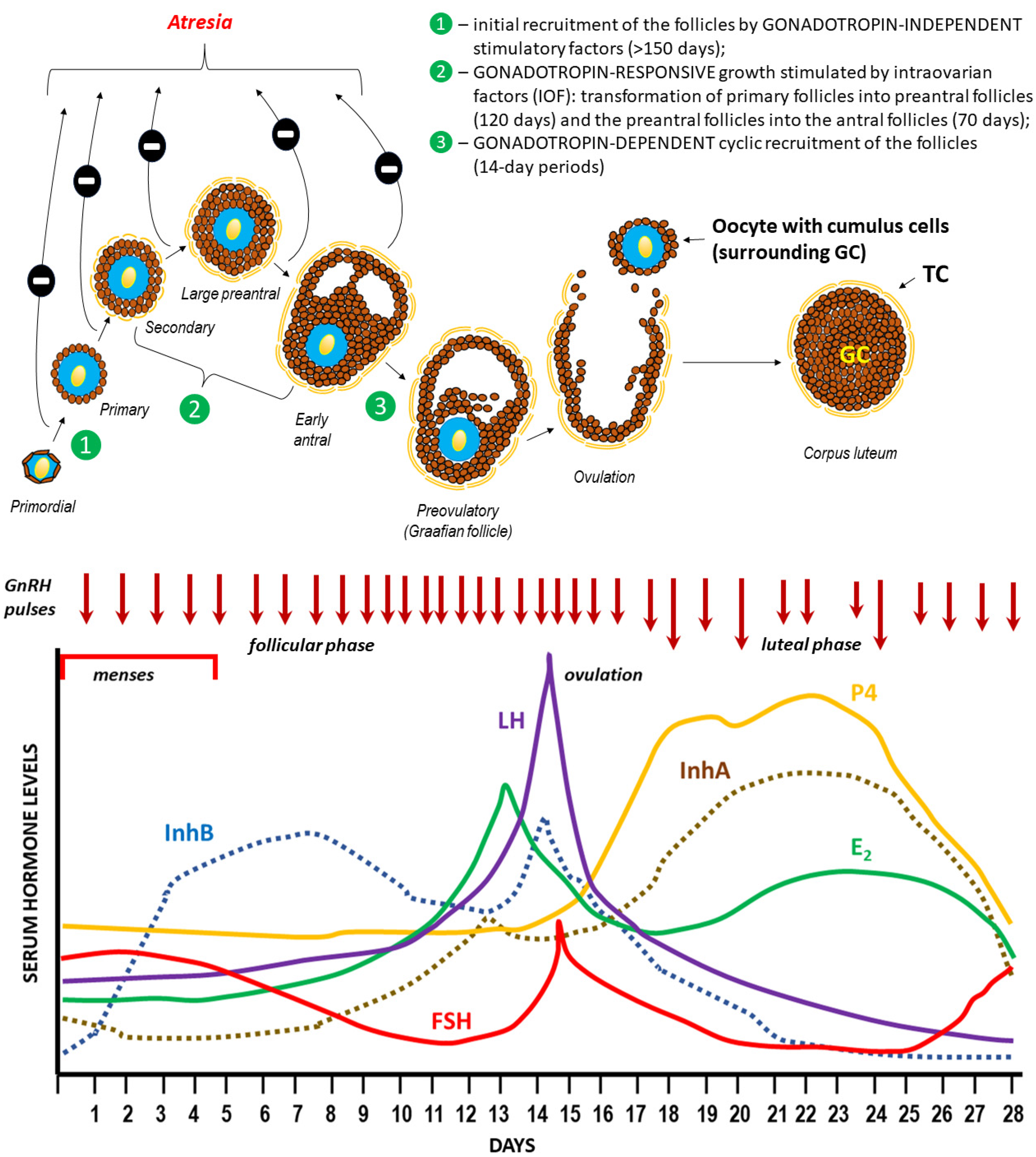
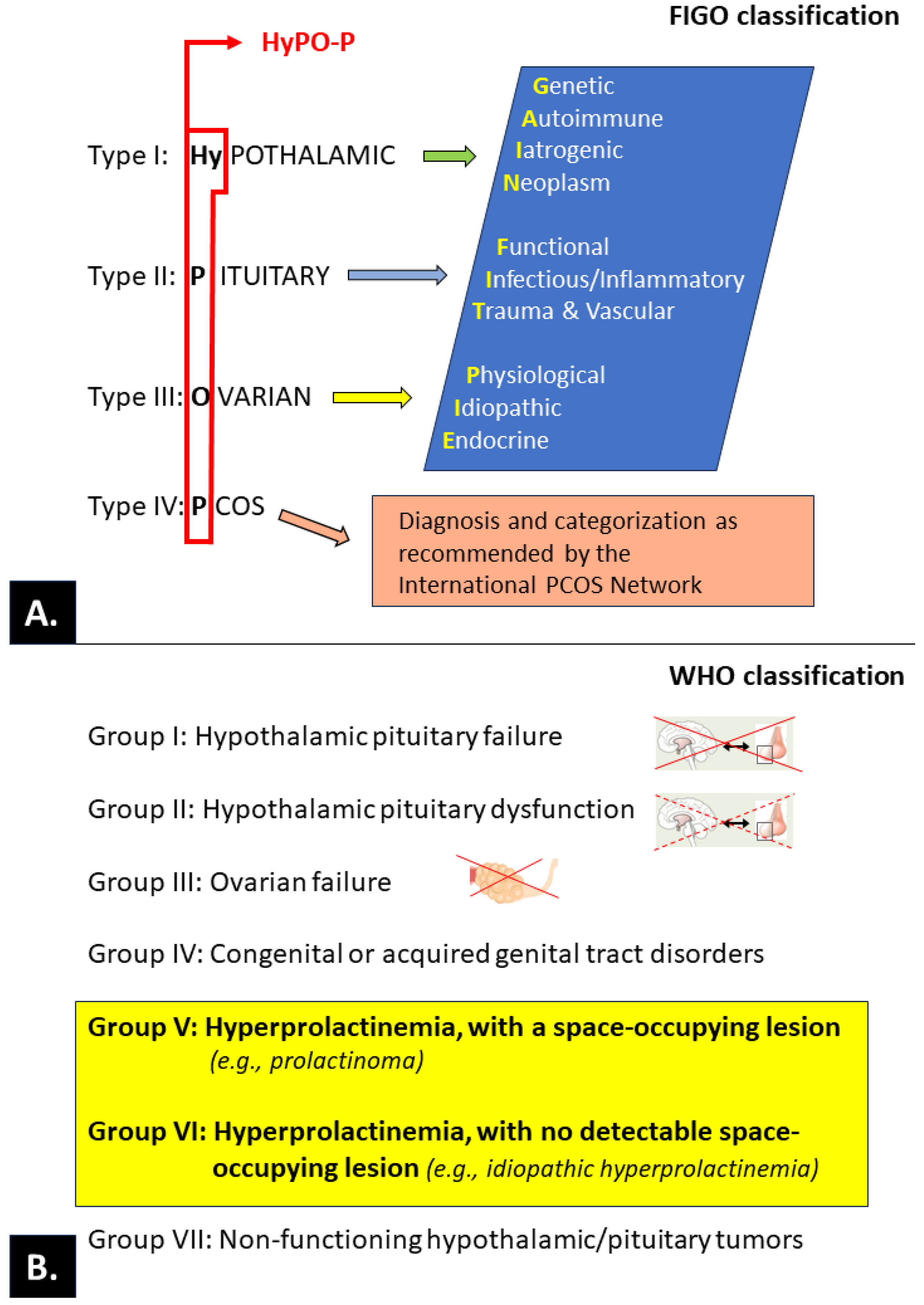

| Category of Hyperprolactinemia | Examples, Incl. Medications | Possible Etiology (Direct Cause, If Known) |
|---|---|---|
| PHYSIOLOGICAL | Pregnancy and postpartum Breast feeding (nipple stimulation, suckling) Nursing | Reduced dopamine secretion from tuberoinfundibular dopamine (TIDA) neurons of the arcuate nucleus in the hypothalamus and disappearance of the negative-feedback mechanism between PRL and TIDA activity, resulting in unresponsiveness of TIDA to increased PRL concentration [232]. |
| Stress | Complex neuroendocrine response to physical and emotional distress reduces PRL signaling in TIDA neurons and thus potentially a decline in their inhibitory dopamine-dependent influence on PRL secretion [233]. | |
| Exercise, especially high intensity | Hypothalamic thyrotropin-releasing hormone (TRH) release caused by physical exercise stimulates both release of thyroid-stimulating hormone (TSH) and thus the production of thyroid hormones as well as the release of PRL from lactotrophic cells [234]. | |
| Sleep | Unspecified connection with the sleep-wake cycle [235]. | |
| Chest wall stimulation | Neurogenic. It is hypothesized that even indirect stimulation of the intercostal nerves may result in induction of a stimulatory reflex conducted by intercostal nerves with subsequent stimulation of hypothalamic centers controlling lactation [236]. | |
| Food ingestion | Probably because PRL signaling is implicated in the regulation of glucose homeostatic adaptations through its impact in pancreatic islet cell physiology and glucose metabolism [237]. | |
| Sexual intercourse | PRL increases following orgasm are involved in a feedback loop that serves to decrease arousal through inhibitory central dopaminergic and probably peripheral processes [238]. | |
| PATHOLOGICAL | Pituitary disease:
| Differential diagnosis is broad (including primary tumors, metastases, and lympho-proliferative diseases) and multifaceted. Excessive production of PRL by hypertrophic or neoplastic pituitary tissue and/or disinhibition of lactotrophs due to mechanical interruption of the portal transport of dopamine from the hypothalamus to the anterior pituitary gland (stalk effect) [82,239]. |
| Primary hypothyroidism | Increased levels of TRH can cause to rise PRL levels by stimulation of TRH receptors on lactotrophic cells [240]. | |
| Chronic renal insufficiency | Reduction in metabolic clearance of PRL and direct stimulation of PRL secretion from lactotrophs due to reduced availability of dopamine in the brain in the uremic state [241,242]. | |
| Severe liver failure (inc. cirrhosis) | Decompensated liver function leads to an alteration in the type of amino acids entering the central nervous system with an increase in the synthesis of false neurotransmitters such as octopamine and phenylethanolamine. These false neurotransmitters may inhibit the dopamine release contributing to hyperprolactinemia [243]. | |
| Neuraxis irradiation (radiation therapy) | Hyperprolactinemia may develop in 20–50% of cases after high dose (>50 Gy) cranial radiotherapy as a result of hypothalamic damage and reduced inhibitory dopamine activity [244]. Elevated PRL levels may decline and normalize during follow-up due to radiation-induced reduction of the pituitary lactotroph cells [245]. | |
| Spinal cord lesions | Secondary hyperprolactinemia due to elevated endorphins and opioid phenotypes in the central nervous system (CNS) following spinal cord injury-related shock as well as production of PRL-releasing factors [246,247,248]. | |
| Seizures | Activation of certain pathways in the brain that regulate PRL secretion or the activation of the hypothalamic–pituitary–adrenal (HPA) axis, which can subsequently increase PRL levels in the blood [249]. | |
| Polycystic ovary syndrome (PCOS) | Probably secondary to increased E2 levels and/or insulin-resistance in PCOS patients [224,250]. | |
| Ectopic secretion of PRL (bronchogenic carcinoma, hypernephroma) | Excess PRL from tumor tissue not subject to negative feedback regulation by dopamine [251]. | |
| Chest wall trauma (including surgery, herpes zoster) | A damage or sectioning of the intercostal nerves may result in reflex stimulation of hypothalamic centers controlling lactation through the same neural pathways involved in puerperal lactation [252]. | |
| Idiopathic | Unknown cause or no clear cause | |
| PHARMACOLOGICAL (iatrogenic) | Typical antipsychotics: haloperidol, phenothiazines, thioridazine, clomipramine, fluphenazine, pimozide, prochlorperazine PRL-raising atypical antipsychotics: risperidone, olanzapine, molindone, paliperidone | Antipsychotic-associated dopamine D2 receptor antagonism. Blockade of D2 by typical antipsychotics and risperidone can cause hyperprolactinemia [253,254]. Atypical antipsychotics other than risperidone are less likely to cause sustained hyperprolactinemia; asymptomatic and transient hyperprolactinemia is more common, because of their lower affinity for D2 receptors [255]. |
| Antidepressant agents: selective serotonin reuptake inhibitors—SSRIs (citalopram, escitalopram, fluoxetine, fluvoxamine, milnacipran, paroxetine, sertraline, venlafaxine); tricyclic antidepressants—TCAs (amitriptyline, amoxapine, clomipramine, desipramine); monoamine oxidase inhibitors—MAOIs (clorgyline, pargyline) | Inhibition of the tuberoinfundibular dopaminergic (D2) pathway through stimulation of gamma aminobutyric acid (GABA)ergic neurons and release of PRL-regulating factors, such as vasoactive intestinal peptide (VIP) or oxytocin [256,257]. May cause indirect modulation of PRL release by increasing serotonin (5-HT) [258]. | |
| Gastrointestinal drugs: metoclopramide, domperidone, prochlorperazine, metiamide, cimetidine (intravenous) | Dopamine D2 receptor antagonism [259]. Cimetidine, a histamine H2 receptor antagonist acting in the hypothalamus inhibits dopamine secretion, as well as increases 5-HT release within dopamine-GABA-serotoninergic system [258,260,261]. | |
| Antihypertensive agents: - methyldopa, - verapamil, - reserpine, tetrabenazine | Alpha-methyldopa causes hyperprolactinemia by inhibiting the enzyme l-aromatic amino acid decarboxylase (which is responsible for converting L-dopa to dopamine) and by acting as a false neurotransmitter to decrease dopamine synthesis [262]. Verapamil, a phenylalkylamine calcium channel blocker, blocks hypothalamic (tuberoinfundibular) production of dopamine [263]. Reserpine and tetrabenazine produce a reversible depletion of dopamine by inhibition of the vesicular monoamine transporter type-2 (VMAT2) that blocks dopamine storage in synaptic vesicles of neurons [264,265]. | |
| Opiates: codeine, morphine | Disinhibition of lactotrophs by the inhibitory effect of TIDA neurons (decreased dopaminergic activity due to decrease in the turnover and release of hypothalamic dopamine) [266,267]. | |
| Hormone preparations: antiandrogens, combined oral contraceptives, estrogens | Estrogen-stimulated lactotroph hyperplasia [268,269]. | |
| ANALYTICAL (assay artefacts)—misdiagnosis | Macroprolactin | Macroprolactinemia represents a state of hyperprolactinemia characterized by the predominance of macroprolactin (also known as big–big PRL), a macromolecule with limited bioavailability and bioactivity, and it is mainly suspected in asymptomatic individuals or those without the typical hyperprolactinemia-related symptoms [54,57,231]. |
| Heterophilic antibodies (endogenous proteins that bind animal antigens) | Heterophilic antibodies interact poorly and nonspecifically with the assay antibodies. Depending on the type of such antibody and the immunoassay format, heterophilic antibodies can lead to both falsely high and low analyte concentrations according to the site of interference [231]. | |
| Prozone phenomenon (hook effect), i.e., falsely normal or mildly elevated PRL while the true PRL concentration is many fold higher than the upper limit | This phenomenon occurs when extremely high PRL concentration (i.e., observed in large pituitary macroadenomas (≥3 cm)) saturates both the capture and the labeled antibody during immunoassay, preventing the formation of the “sandwich” and causing false-negative results [231]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szukiewicz, D. Current Insights in Prolactin Signaling and Ovulatory Function. Int. J. Mol. Sci. 2024, 25, 1976. https://doi.org/10.3390/ijms25041976
Szukiewicz D. Current Insights in Prolactin Signaling and Ovulatory Function. International Journal of Molecular Sciences. 2024; 25(4):1976. https://doi.org/10.3390/ijms25041976
Chicago/Turabian StyleSzukiewicz, Dariusz. 2024. "Current Insights in Prolactin Signaling and Ovulatory Function" International Journal of Molecular Sciences 25, no. 4: 1976. https://doi.org/10.3390/ijms25041976
APA StyleSzukiewicz, D. (2024). Current Insights in Prolactin Signaling and Ovulatory Function. International Journal of Molecular Sciences, 25(4), 1976. https://doi.org/10.3390/ijms25041976






