The Use of Hematopoietic Stem Cells for Heart Failure: A Systematic Review
Abstract
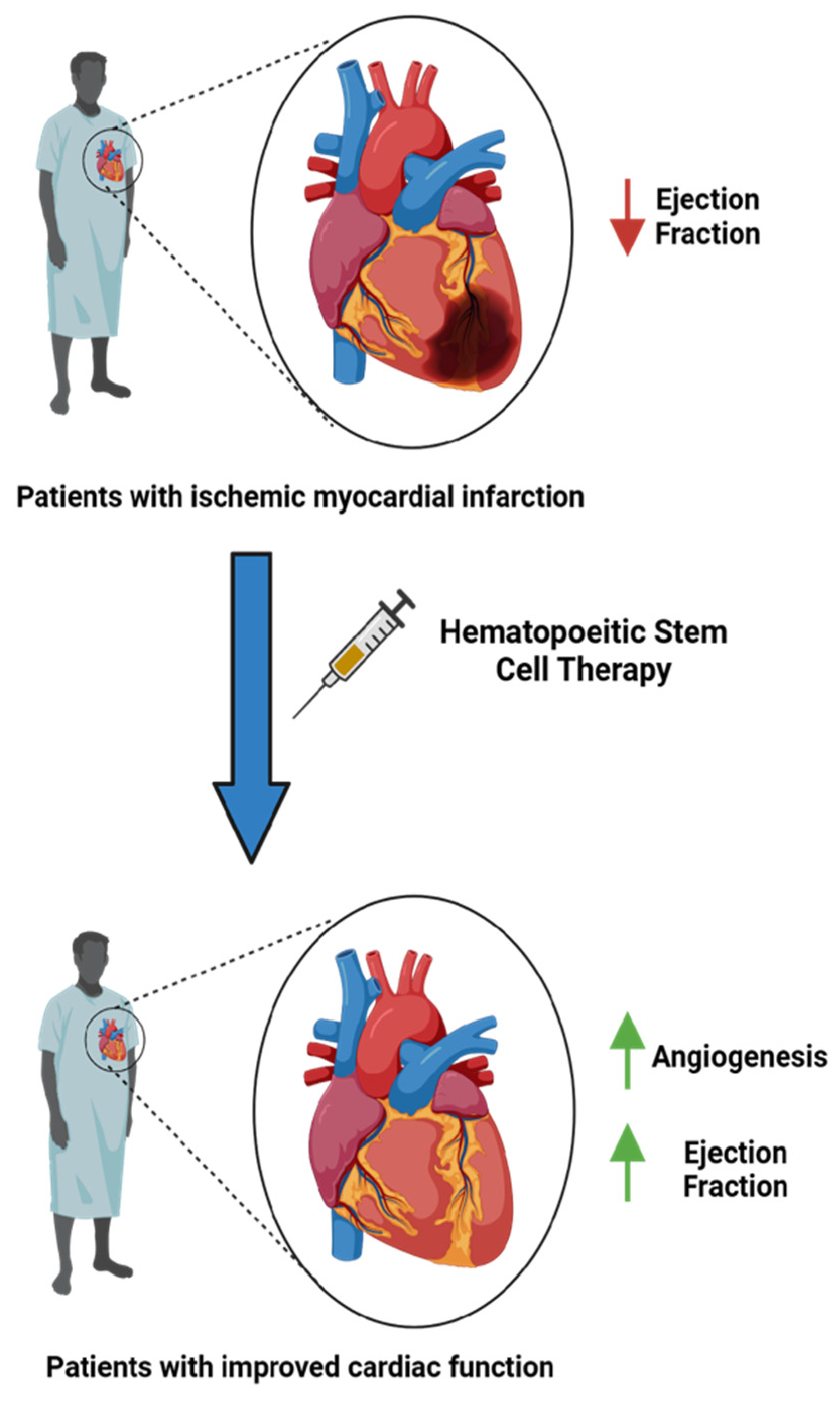
1. Introduction
2. Materials and Methods
2.1. Search Strategy
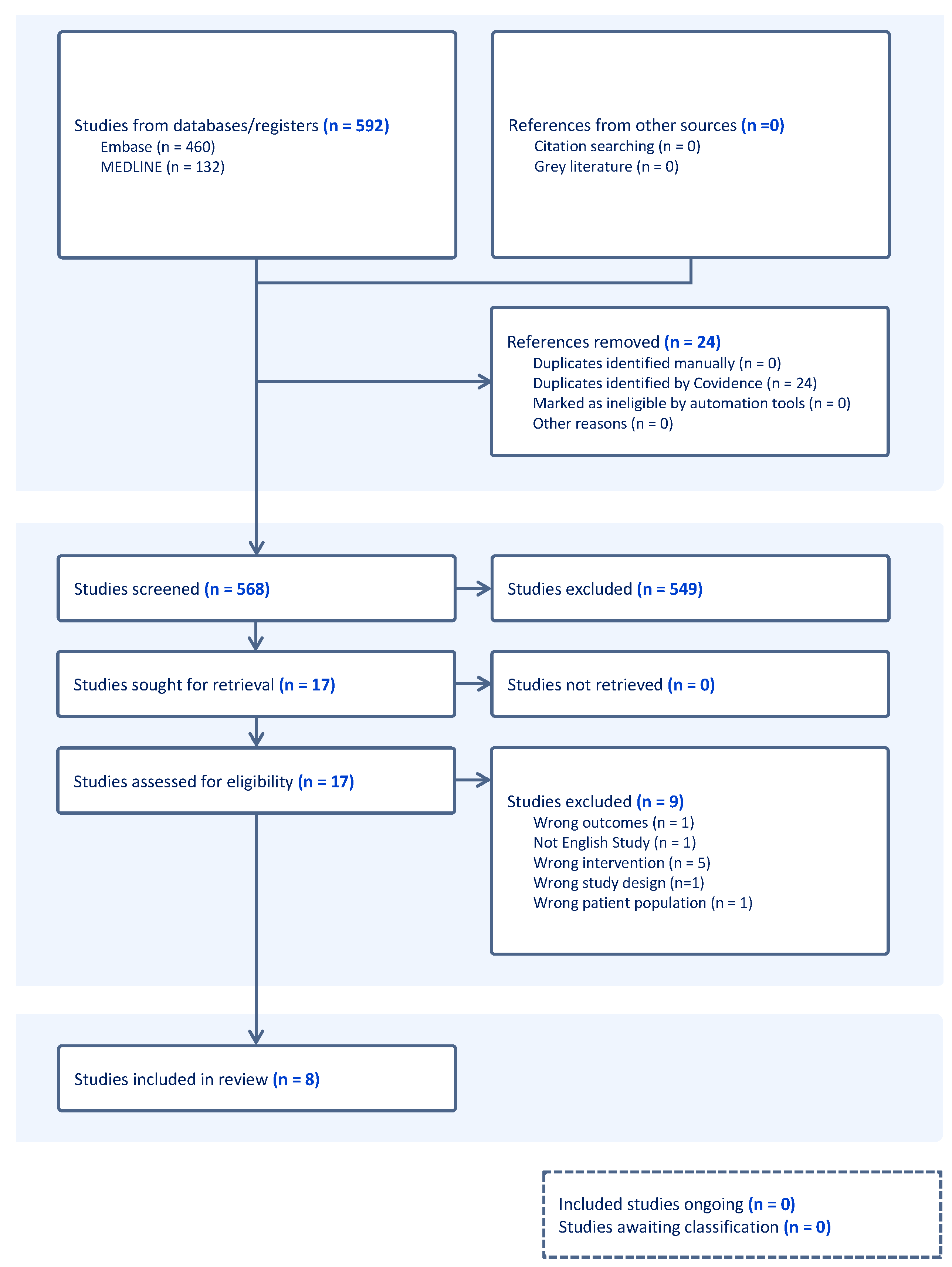
2.2. Study Selection
3. Results
3.1. Study Design
3.2. Impact of BMSC on LVEF and Myocardial Perfusion
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| HSC | Hematopoietic Stem Cells |
| MSC | Mesenchymal Stem Cells |
| NYHA | New York Heart Association |
| VEGF | Vascular Endothelial Growth Factor |
| BNP | B-type Natriuretic Peptide |
| EF | Ejection Fraction |
| BMSC | Bone Marrow Stem Cell |
| AMI | Acute Myocardial Infarction |
| BMC | Bone Marrow-derived progenitor Cells |
| BMMNC | Bone Marrow Mononuclear Cell |
| PBSC | Peripheral Blood Stem Cells |
| G-CSF | Granulocyte Colony-Stimulating Factor |
| LVEF | Left Ventricular Ejection Fraction |
| RCT | Randomized Controlled Trials |
| CD33 | Cluster of Differentiation 33 |
| CD34 | Cluster of Differentiation 33 |
| T2DM | Type 2 Diabetes Mellitus |
| CABG | Coronary Artery Bypass Grafting |
| HF | Heart Failure |
| AMI | Acute Myocardial Infarction |
| BMC | Bone Marrow-derived progenitor Cells |
| BMMNC | Bone Marrow Mononuclear Cell |
| BMSC | Bone Marrow Stem Cell |
| BNP | B-type Natriuretic Peptide |
| CABG | Coronary Artery Bypass Grafting |
| CD33 | Cluster of Differentiation 33 |
| CD34 | Cluster of Differentiation 33 |
| EF | Ejection Fraction |
| G-CSF | Granulocyte Colony-Stimulating Factor |
| HF | Heart Failure |
| HSC | Hematopoietic Stem Cells |
| LVEF | Left Ventricular Ejection Fraction |
| MSC | Mesenchymal Stem Cells |
| NYHA | New York Heart Association |
| PBSC | Peripheral Blood Stem Cells |
| RCT | Randomized Controlled Trials |
| T2DM | Type 2 Diabetes Mellitus |
| VEGF | Vascular Endothelial Growth Factor |
References
- Malik, A.; Brito, D.; Vaqar, S.; Chhabra, L. Congestive Heart Failure. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Bozkurt, B.; Ahmad, T.; Alexander, K.M.; Baker, W.L.; Bosak, K.; Breathett, K.; Fonarow, G.C.; Heidenreich, P.; Ho, J.E.; Hsich, E.; et al. Heart Failure Epidemiology and Outcomes Statistics: A Report of the Heart Failure Society of America. J. Card. Fail. 2023, 29, 1412–1451. [Google Scholar] [CrossRef] [PubMed]
- Heidenreich, P.A.; Bozkurt, B.; Aguilar, D.; Allen, L.A.; Byun, J.J.; Colvin, M.M.; Deswal, A.; Drazner, M.H.; Dunlay, S.M.; Evers, L.R.; et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2022, 145, e895–e1032. [Google Scholar] [CrossRef]
- Kikuchi, K.; Poss, K.D. Cardiac Regenerative Capacity and Mechanisms. Annu. Rev. Cell Dev. Biol. 2012, 28, 719–741. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Bian, W.; Zhou, Y.; Zhang, J. Cardiac Fibroblasts and Myocardial Regeneration. Front. Bioeng. Biotechnol. 2021, 9, 599928. [Google Scholar] [CrossRef] [PubMed]
- Lindeman, A.; Pepine, C.J.; March, K.L. Cardiac Stem Cell Therapy among Clinics of Uncertain Regulatory Status (COURS): Under-Regulated, under-Observed, Incompletely Understood. J. Transl. Med. 2020, 18, 285. [Google Scholar] [CrossRef]
- Hoang, D.M.; Pham, P.T.; Bach, T.Q.; Ngo, A.T.L.; Nguyen, Q.T.; Phan, T.T.K.; Nguyen, G.H.; Le, P.T.T.; Hoang, V.T.; Forsyth, N.R.; et al. Stem Cell-Based Therapy for Human Diseases. Signal Transduct. Target. Ther. 2022, 7, 272. [Google Scholar] [CrossRef] [PubMed]
- Bolli, R.; Solankhi, M.; Tang, X.-L.; Kahlon, A. Cell Therapy in Patients with Heart Failure: A Comprehensive Review and Emerging Concepts. Cardiovasc. Res. 2021, 118, 951–976. [Google Scholar] [CrossRef] [PubMed]
- du Pré, B.C.; Doevendans, P.A.; van Laake, L.W. Stem Cells for Cardiac Repair: An Introduction. J. Geriatr. Cardiol. 2013, 10, 186–197. [Google Scholar] [CrossRef] [PubMed]
- Martin-Rendon, E.; Brunskill, S.J.; Hyde, C.J.; Stanworth, S.J.; Mathur, A.; Watt, S.M. Autologous Bone Marrow Stem Cells to Treat Acute Myocardial Infarction: A Systematic Review. Eur. Heart J. 2008, 29, 1807–1818. [Google Scholar] [CrossRef]
- Strauer, B.-E.; Steinhoff, G. 10 Years of Intracoronary and Intramyocardial Bone Marrow Stem Cell Therapy of the Heart: From the Methodological Origin to Clinical Practice. J. Am. Coll. Cardiol. 2011, 58, 1095–1104. [Google Scholar] [CrossRef]
- Gupta, S.; Sharma, A.; Sharma, A.; Verma, R.S. Mesenchymal Stem Cells for Cardiac Regeneration: From Differentiation to Cell Delivery. Stem Cell Rev. Rep. 2021, 17, 1666–1694. [Google Scholar] [CrossRef] [PubMed]
- Manginas, A.; Goussetis, E.; Koutelou, M.; Karatasakis, G.; Peristeri, I.; Theodorakos, A.; Leontiadis, E.; Plessas, N.; Theodosaki, M.; Graphakos, S.; et al. Pilot Study to Evaluate the Safety and Feasibility of Intracoronary CD133+ and CD133− CD34+ Cell Therapy in Patients with Nonviable Anterior Myocardial Infarction. Catheter. Cardiovasc. Interv. 2007, 69, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Assmus, B.; Rolf, A.; Erbs, S.; Elsässer, A.; Haberbosch, W.; Hambrecht, R.; Tillmanns, H.; Yu, J.; Corti, R.; Mathey, D.G.; et al. Clinical Outcome 2 Years after Intracoronary Administration of Bone Marrow-Derived Progenitor Cells in Acute Myocardial Infarction. Circ. Heart Fail. 2010, 3, 89–96. [Google Scholar] [CrossRef] [PubMed]
- De Rosa, S.; Seeger, F.H.; Honold, J.; Fischer-Rasokat, U.; Lehmann, R.; Fichtlscherer, S.; Schächinger, V.; Dimmeler, S.; Zeiher, A.M.; Assmus, B. Procedural Safety and Predictors of Acute Outcome of Intracoronary Administration of Progenitor Cells in 775 Consecutive Procedures Performed for Acute Myocardial Infarction or Chronic Heart Failure. Circ. Cardiovasc. Interv. 2013, 6, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Mozid, A.M.; Holstensson, M.; Choudhury, T.; Ben-Haim, S.; Allie, R.; Martin, J.; Sinusas, A.J.; Hutton, B.F.; Mathur, A. Clinical Feasibility Study to Detect Angiogenesis Following Bone Marrow Stem Cell Transplantation in Chronic Ischaemic Heart Failure. Nucl. Med. Commun. 2014, 35, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Choudry, F.A.; Yeo, C.; Mozid, A.; Martin, J.F.; Mathur, A. Increases in Plasma Tβ4 after Intracardiac Cell Therapy in Chronic Ischemic Heart Failure Is Associated with Symptomatic Improvement. Regen. Med. 2015, 10, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Duan, F.; Qi, Z.; Liu, S.; Lu, X.; Wang, H.; Gao, Y.; Wang, J. Effectiveness of Bone Marrow Mononuclear Cells Delivered through a Graft Vessel for Patients with Previous Myocardial Infarction and Chronic Heart Failure: An Echocardiographic Study of Left Ventricular Remodeling. Med. Ultrason. 2015, 17, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Bocchi, E.A.; Bacal, F.; Guimarães, G.; Mendroni, A.; Mocelin, A.; Filho, A.E.; da Cruz, F.d.D.; Resende, M.C.; Chamone, D. Granulocyte-Colony Stimulating Factor or Granulocyte-Colony Stimulating Factor Associated to Stem Cell Intracoronary Infusion Effects in Non Ischemic Refractory Heart Failure. Int. J. Cardiol. 2010, 138, 94–97. [Google Scholar] [CrossRef]
- Gu, X.; Xie, Y.; Gu, J.; Sun, L.; He, S.; Xu, R.; Duan, J.; Zhao, J.; Hang, F.; Xu, H.; et al. Repeated Intracoronary Infusion of Peripheral Blood Stem Cells with G-CSF in Patients with Refractory Ischemic Heart Failure—A Pilot Study. Circ. J. 2011, 75, 955–963. [Google Scholar] [CrossRef][Green Version]
- Fan, M.; Huang, Y.; Chen, Z.; Xia, Y.; Chen, A.; Lu, D.; Wu, Y.; Zhang, N.; Qian, J. Efficacy of Mesenchymal Stem Cell Therapy in Systolic Heart Failure: A Systematic Review and Meta-Analysis. Stem Cell Res. Ther. 2019, 10, 150. [Google Scholar] [CrossRef]
- Attar, A.; Bahmanzadegan Jahromi, F.; Kavousi, S.; Monabati, A.; Kazemi, A. Mesenchymal Stem Cell Transplantation after Acute Myocardial Infarction: A Meta-Analysis of Clinical Trials. Stem Cell Res. Ther. 2021, 12, 600. [Google Scholar] [CrossRef]
- Gnecchi, M.; He, H.; Noiseux, N.; Liang, O.D.; Zhang, L.; Morello, F.; Mu, H.; Melo, L.G.; Pratt, R.E.; Ingwall, J.S.; et al. Evidence Supporting Paracrine Hypothesis for Akt-Modified Mesenchymal Stem Cell-Mediated Cardiac Protection and Functional Improvement. FASEB J. 2006, 20, 661–669. [Google Scholar] [CrossRef]
- Zhou, B.O.; Ding, L.; Morrison, S.J. Hematopoietic Stem and Progenitor Cells Regulate the Regeneration of Their Niche by Secreting Angiopoietin-1. eLife 2015, 4, e05521. [Google Scholar] [CrossRef]
- Perin, E.C.; Silva, G.V.; Assad, J.A.R.; Vela, D.; Buja, L.M.; Sousa, A.L.S.; Litovsky, S.; Lin, J.; Vaughn, W.K.; Coulter, S.; et al. Comparison of Intracoronary and Transendocardial Delivery of Allogeneic Mesenchymal Cells in a Canine Model of Acute Myocardial Infarction. J. Mol. Cell. Cardiol. 2008, 44, 486–495. [Google Scholar] [CrossRef]
- Fukushima, S.; Varela-Carver, A.; Coppen, S.R.; Yamahara, K.; Felkin, L.E.; Lee, J.; Barton, P.J.R.; Terracciano, C.M.N.; Yacoub, M.H.; Suzuki, K. Direct Intramyocardial But Not Intracoronary Injection of Bone Marrow Cells Induces Ventricular Arrhythmias in a Rat Chronic Ischemic Heart Failure Model. Circulation 2007, 115, 2254–2261. [Google Scholar] [CrossRef] [PubMed]
- AbuSamra, D.B.; Aleisa, F.A.; Al-Amoodi, A.S.; Jalal Ahmed, H.M.; Chin, C.J.; Abuelela, A.F.; Bergam, P.; Sougrat, R.; Merzaban, J.S. Not Just a Marker: CD34 on Human Hematopoietic Stem/Progenitor Cells Dominates Vascular Selectin Binding along with CD44. Blood Adv. 2017, 1, 2799–2816. [Google Scholar] [CrossRef] [PubMed]
- Rennert, R.C.; Sorkin, M.; Januszyk, M.; Duscher, D.; Kosaraju, R.; Chung, M.T.; Lennon, J.; Radiya-Dixit, A.; Raghvendra, S.; Maan, Z.N.; et al. Diabetes Impairs the Angiogenic Potential of Adipose-Derived Stem Cells by Selectively Depleting Cellular Subpopulations. Stem Cell Res. Ther. 2014, 5, 79. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Han, J.W.; Lee, J.Y.; Choi, Y.J.; Sohn, Y.-D.; Song, M.; Yoon, Y. Diabetic Mesenchymal Stem Cells Are Ineffective for Improving Limb Ischemia Due to Their Impaired Angiogenic Capability. Cell Transplant. 2015, 24, 1571–1584. [Google Scholar] [CrossRef]
- Siggins, R.W.; Hossain, F.; Rehman, T.; Melvan, J.N.; Zhang, P.; Welsh, D.A. Cigarette Smoke Alters the Hematopoietic Stem Cell Niche. Med. Sci. 2014, 2, 37–50. [Google Scholar] [CrossRef]
- Kowalówka, A.R.; Wojakowski, W.; Bachowski, R.; Deja, M. Stem Cells Mobilization by Cardiopulmonary Bypass after Coronary Artery Bypass Grafting. Adv. Interv. Cardiol. 2022, 18, 450–458. [Google Scholar] [CrossRef]
- Cui, X.; Zhao, X.; Liang, Y. Sex Differences in Normal and Malignant Hematopoiesis. Blood Sci. 2022, 4, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.; Gandhi, D.; Srivastava, S.; Shah, K.J.; Mansukhani, R. Heart Failure: A Class Review of Pharmacotherapy. Pharm. Ther. 2017, 42, 464–472. [Google Scholar]
- Williams, S.; Green, R.; Morrison, A.; Watson, D.; Buchanan, S. The Psychosocial Aspects of Donating Blood Stem Cells: The Sibling Donor Perspective. J. Clin. Apher. 2003, 18, 1–9. [Google Scholar] [CrossRef] [PubMed]
- MacLeod, K.D.; Whitsett, S.F.; Mash, E.J.; Pelletier, W. Pediatric Sibling Donors of Successful and Unsuccessful Hematopoietic Stem Cell Transplants (HSCT): A Qualitative Study of Their Psychosocial Experience. J. Pediatr. Psychol. 2003, 28, 223–230. [Google Scholar] [CrossRef]
- Pillay, B.; Lee, S.J.; Katona, L.; De Bono, S.; Warren, N.; Fletcher, J.; Burney, S. The Psychosocial Impact of Haematopoietic SCT on Sibling Donors. Bone Marrow Transplant. 2012, 47, 1361–1365. [Google Scholar] [CrossRef]
| Parameter | Description |
|---|---|
| Population (P) | Adult patients (age 18+) with heart failure |
| Intervention (I) | Hematopoietic stem cell therapy |
| Comparison (C) | Hematopoietic stem cell therapy or placebo or pre-therapy (baseline) measures or post-therapy measures |
| Outcome (O) | Peak VO2, New York Heart Association (NYHA) symptom class, major adverse cardiovascular events, ejection fraction, duration of follow-up appointments |
| Study | Country | Study Design | Group | Sample Size | Follow-Up | Age * | % Male | Etiology | Ejection Fraction * |
|---|---|---|---|---|---|---|---|---|---|
| [13] | Greece | Prospective cohort study | Total | 24 | NR | Ischemic HF | |||
| Treatment | 12 | 50.1 ± 8.5 | 91.66 | 27.2 ± 6.8 | |||||
| Control | 12 | 64.8 ± 10.8 | 91.66 | 33.9 ± 69.1 | |||||
| [14] | Germany | RCT | Total | 204 | 24 months | 56 ± 11 | 82 | Ischemic HF | |
| Treatment | 103 | 57 ± 11 | NR | 45.4 ± 9.4 | |||||
| Control | 101 | 55 ± 11 | NR | 48.7 ± 10.4 | |||||
| [15] | Germany | Retrospective cohort study | Total | 775 | 1 month | ||||
| AMI | 126 | 54 ± 11 | 93.65 | AMI | 49 ± 10 | ||||
| Chronic HF | 562 | 62 ± 11 | 88.26 | Ischemic HF | 37 ± 11 | ||||
| Chronic HF | 87 | 57 ± 14 | 75.86 | Non-ischemic HF | 31 ± 11 | ||||
| [16] | UK | RCT | Total | 9 | 12 months | Ischemic HF | |||
| Treatment | 5 | 59 ± 11 | 80 | 30.5 ± 11.9 | |||||
| Control | 4 | 58 ± 4 | 100 | 27.8 ± 10.1 | |||||
| [17] | UK | RCT | Total | 27 | NR | Ischemic HF | |||
| Intramyocardial BMSC group | 8 | 67.1 ± 10.5 | 100 | 32.0 ± 9.1 | |||||
| Intracoronary serum group | 8 | 59.3 ± 12.2 | 100 | 32.4 ± 8.3 | |||||
| BMSC Group | 5 | 62.0 ± 8.9 | 100 | 30.1 ± 3.4 | |||||
| Serum Group | 6 | 63.8 ± 6.6 | 100 | 27.6 ± 10.9 | |||||
| [18] | China | RCT | Total | 42 | 12 months | Ischemic HF | |||
| CABG | 18 | 56.56 ± 9.09 | 96 | NR | |||||
| CABG + BMMNC | 24 | 57.88 ± 8.52 | 94.4 | NR | |||||
| [19] | Brazil | RCT | Total | 22 | <1190 days | NR | NR | NR | 31.4 ± 10 |
| G-CSF (control) | 14 | NR | NR | NR | 29 ± 6.9 | ||||
| BMSC | 8 | NR | NR | NR | 35.8 ± 11.8 |
| Study | MACE |
|---|---|
| [13] | HF deterioration: 25%; stent re-stenosis 8.22%; |
| [14] | Cardiac death—Placebo/control: 5%; Treatment: 3%. Myocardial infarction—Placebo/control: 7%; Treatment: 0%. Revascularization—Placebo/control: 37%; Treatment: 25%. Documented ventricular arrhythmia—Placebo/control: 5%; Treatment: 6%. Stroke—Placebo/control: 2%; Treatment: 1%. |
| [15] | ICM: Native vessel-related complications: 4/455 dissection (nonflow-limitating), 1/455 main vessel occlusion, 1/455 side vessel occlusion, 1/455 thrombus formation/embolization, 2/455 arrhythmmia. Arterial graft-related complications: dissection (nonflow-limitating) 3/47. Venous graft-related complications: 1/60 arrhythmia DCM: 1/87 arrhythmiam 1/87 stroke, 6/87 repeat myocardial infarction, 4/87 deaths AMI: Procedural Complications Related to the Sole cell Administration Procedure: 1/126 dissection, 1/126 side branch occlusion, 2/126 thrombus formation. |
| [16] | NR |
| [17] | NR |
| [18] | CABG: 1/18 died |
| [19] | NR |
| Study | Group | HTN % | Hyperlipidemia % | Diabetes % | Smoking % | CAD % |
|---|---|---|---|---|---|---|
| [14] | Total | NR | NR | NR | NR | NR |
| Treatment group | 54 | 52 | 12 | NR | NR | |
| Control group | 60 | 59 | 21 | NR | NR | |
| [15] | AMI | NR | 51 | 26 | 100 | |
| ICM | NR | 82 | 32 | 73 | 100 | |
| DCM | NR | 53 | 18 | 68 | 100 | |
| [16] | Total | NR | NR | NR | NR | NR |
| Patients receiving intracoronary infusion of BMSC | NR | NR | 40 | NR | NR | |
| Patients receiving intracoronary infusion of serum only | NR | NR | 0 | NR | NR | |
| [17] | Total | NR | NR | NR | NR | NR |
| Intramyocardial BMSC group | NR | NR | 37.5 | NR | NR | |
| Intracoronary serum group | NR | NR | 25 | NR | NR | |
| Intracoronary BMSC group | NR | NR | 20 | |||
| Intracoronary serum group | NR | NR | 33.3 | NR | NR | |
| [18] | Total | NR | NR | NR | NR | NR |
| CABG | 11.1 | NR | NR | NR | NR | |
| CABG + BMMNC | 16.7 | NR | NR | NR | NR |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seth, J.; Sharma, S.; Leong, C.J.; Vaibhav, V.; Nelson, P.; Shokravi, A.; Luo, Y.; Shirvani, D.; Laksman, Z. The Use of Hematopoietic Stem Cells for Heart Failure: A Systematic Review. Int. J. Mol. Sci. 2024, 25, 6634. https://doi.org/10.3390/ijms25126634
Seth J, Sharma S, Leong CJ, Vaibhav V, Nelson P, Shokravi A, Luo Y, Shirvani D, Laksman Z. The Use of Hematopoietic Stem Cells for Heart Failure: A Systematic Review. International Journal of Molecular Sciences. 2024; 25(12):6634. https://doi.org/10.3390/ijms25126634
Chicago/Turabian StyleSeth, Jayant, Sohat Sharma, Cameron J. Leong, Venkat Vaibhav, Pierce Nelson, Arveen Shokravi, Yuchen Luo, Daniel Shirvani, and Zachary Laksman. 2024. "The Use of Hematopoietic Stem Cells for Heart Failure: A Systematic Review" International Journal of Molecular Sciences 25, no. 12: 6634. https://doi.org/10.3390/ijms25126634
APA StyleSeth, J., Sharma, S., Leong, C. J., Vaibhav, V., Nelson, P., Shokravi, A., Luo, Y., Shirvani, D., & Laksman, Z. (2024). The Use of Hematopoietic Stem Cells for Heart Failure: A Systematic Review. International Journal of Molecular Sciences, 25(12), 6634. https://doi.org/10.3390/ijms25126634






