Multi-Omics Techniques for Analysis Antifungal Mechanisms of Lipopeptides Produced by Bacillus velezensis GS-1 against Magnaporthe oryzae In Vitro
Abstract
:1. Introduction
2. Results
2.1. Screening of Antagonistic Activity against Plant Pathogenic Fungi
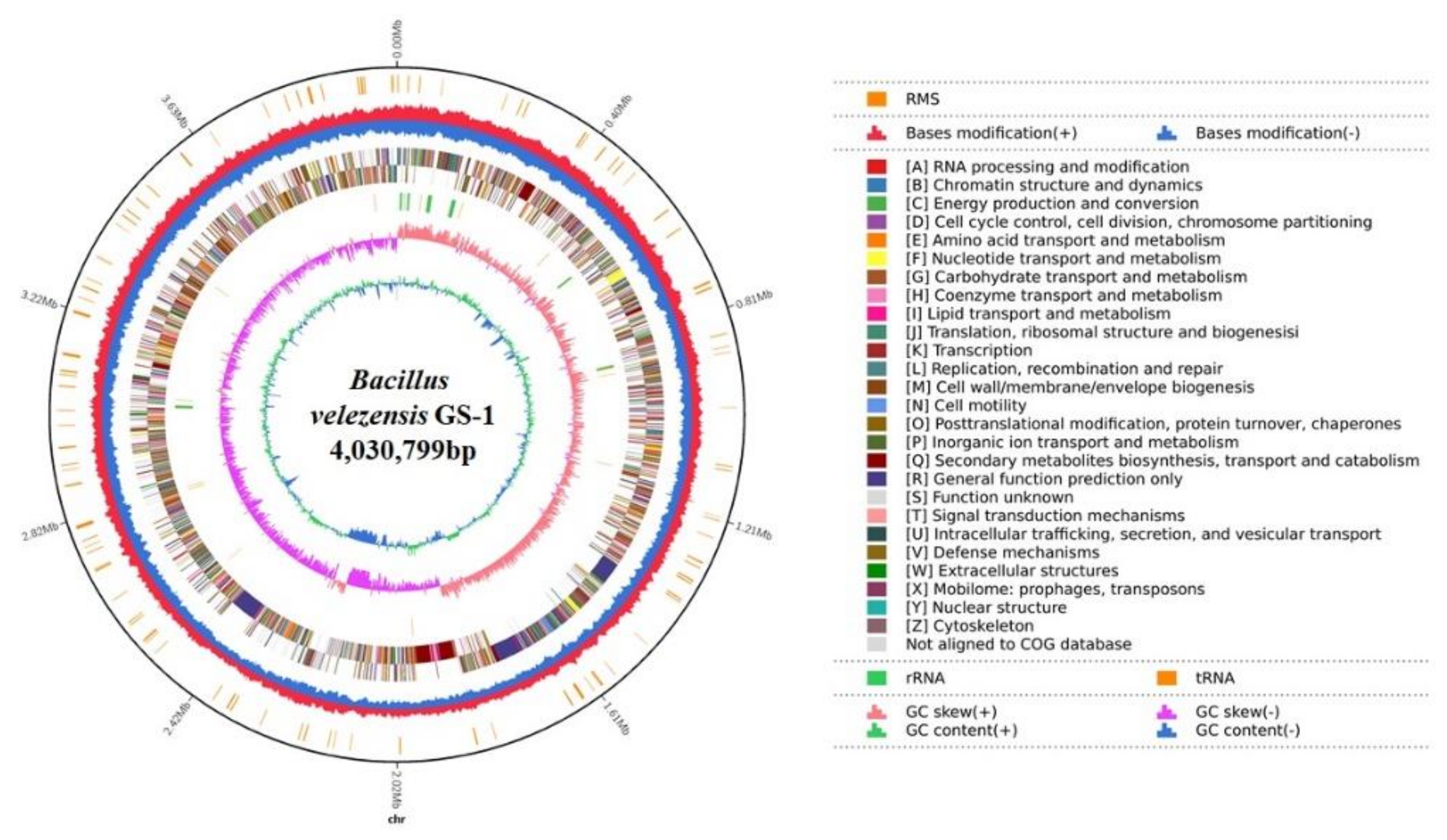
2.2. Genomic Features of B. velezensis GS-1
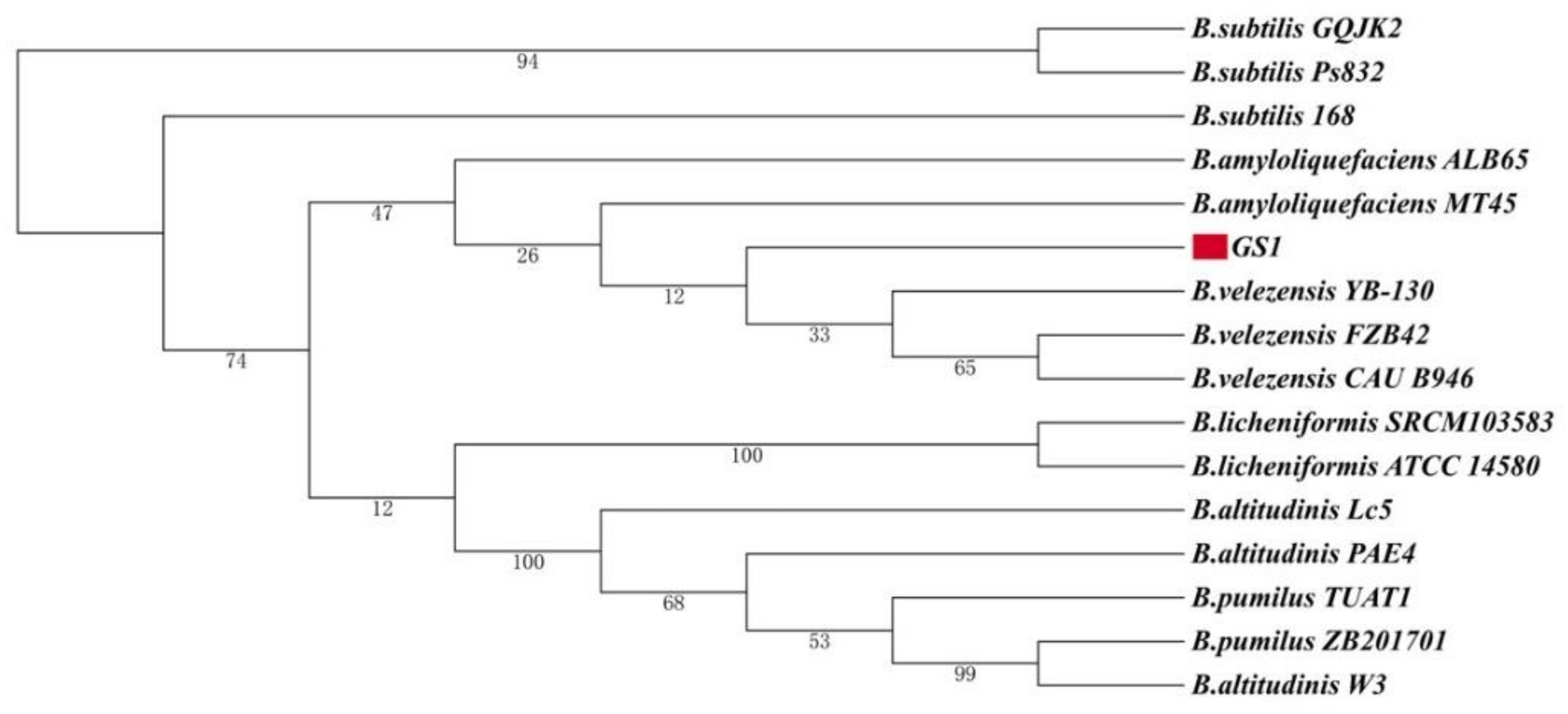
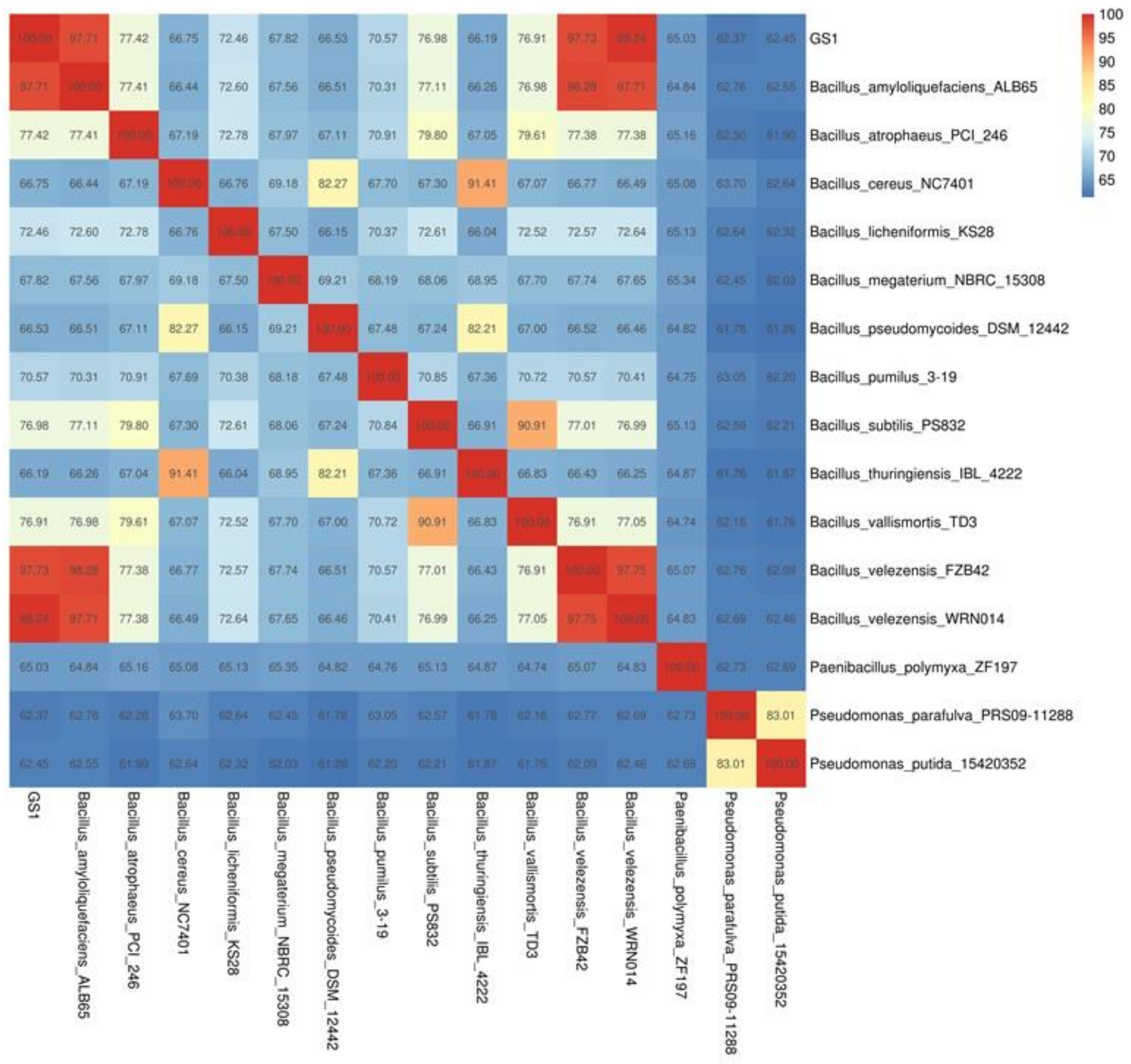
2.3. Phylogeny of B. velezensis GS-1
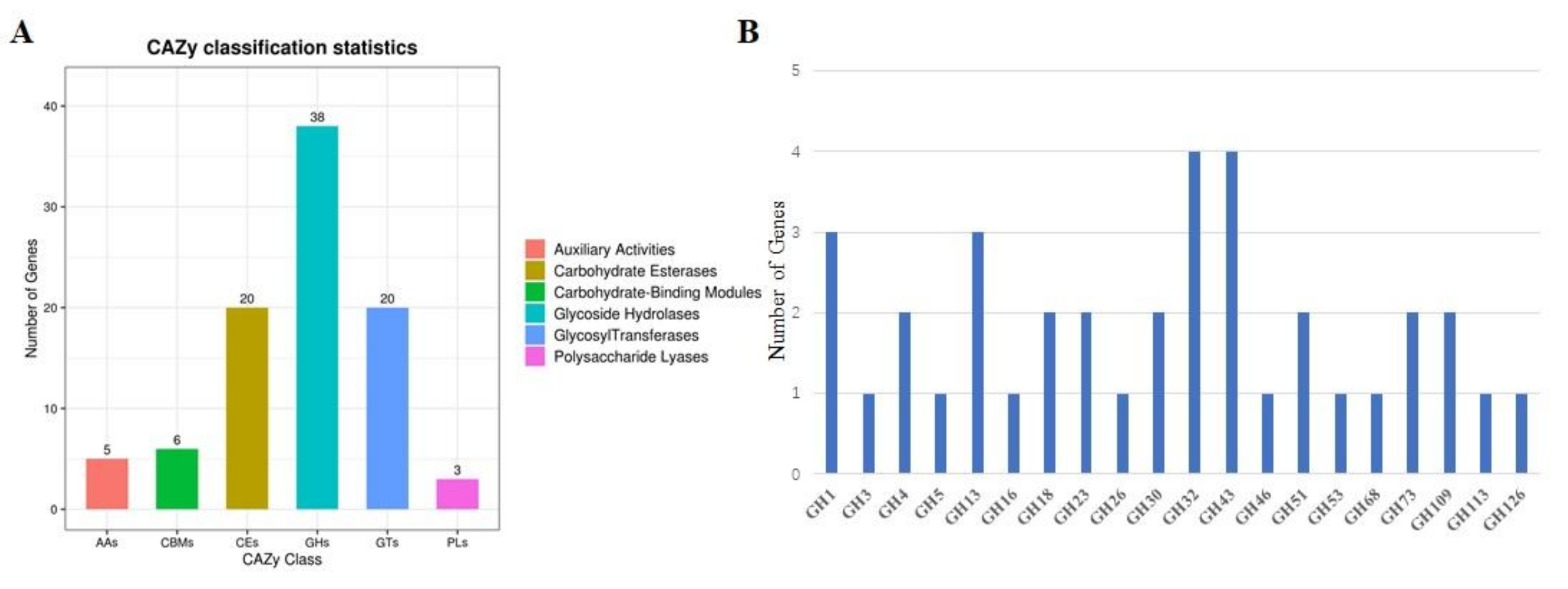
2.4. Analysis of CAZyme Genes in B. velezensis GS-1 Genome
2.5. Secondary Metabolic Related Genes of GS-1
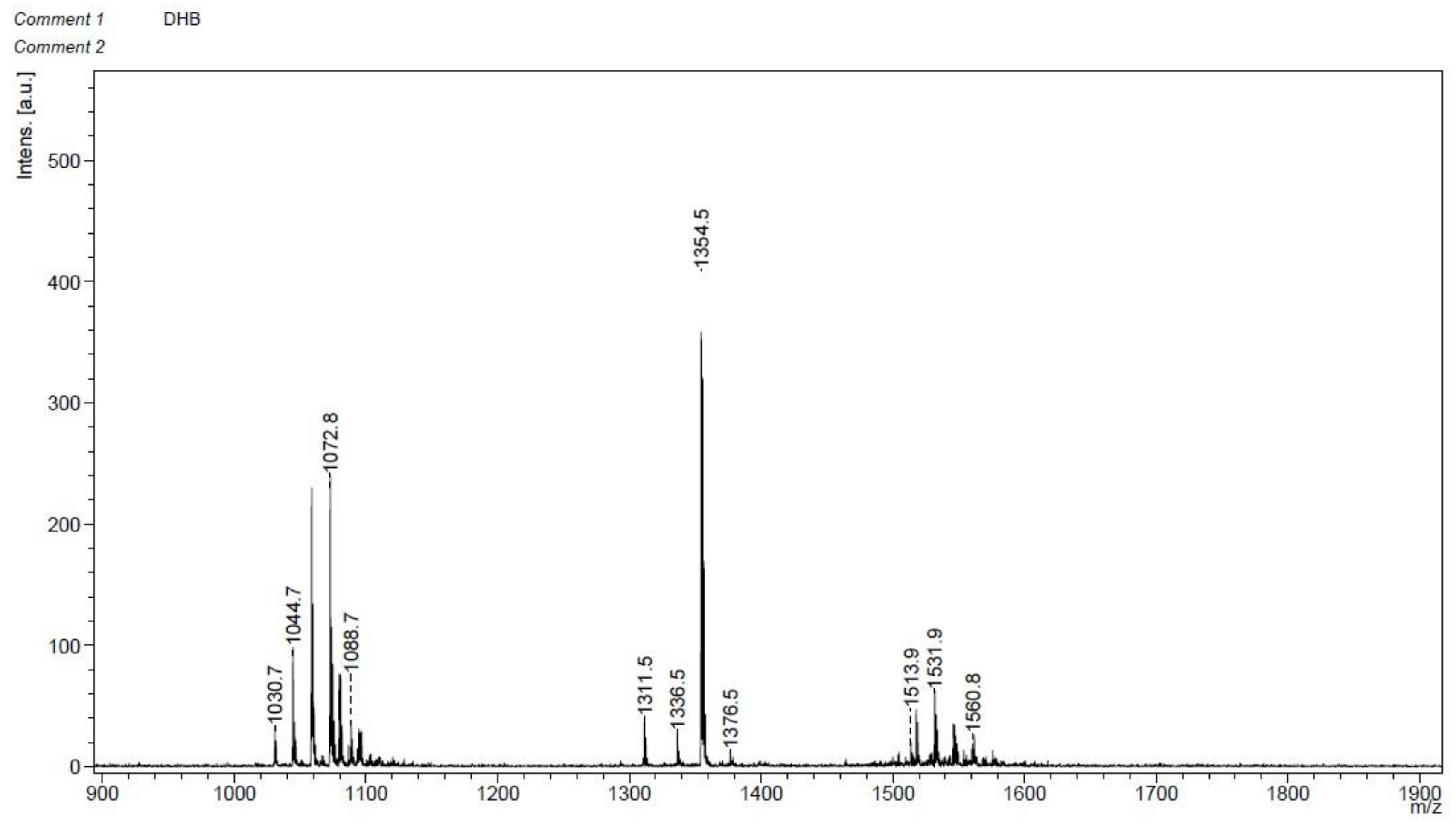
2.6. Crude Lipopeptide Extracts from B. velezensis GS-1 Inhibited M. oryzae
2.7. Transcriptomics Analysis of the Effect of Lipopeptide Extracts on M. oryzae
2.8. Metabolomics Analysis of the Effect of Lipopeptide Extracts on M. oryzae
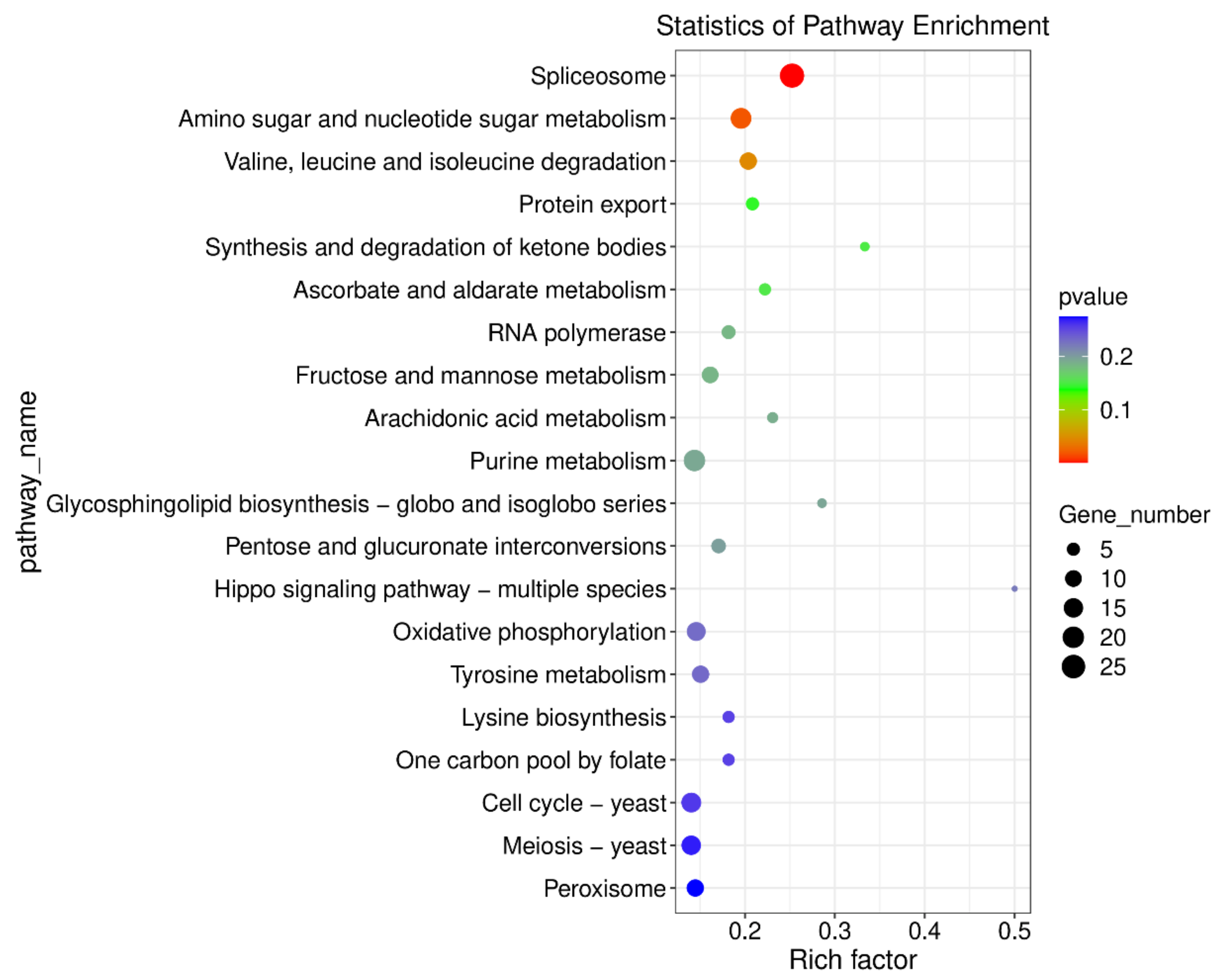
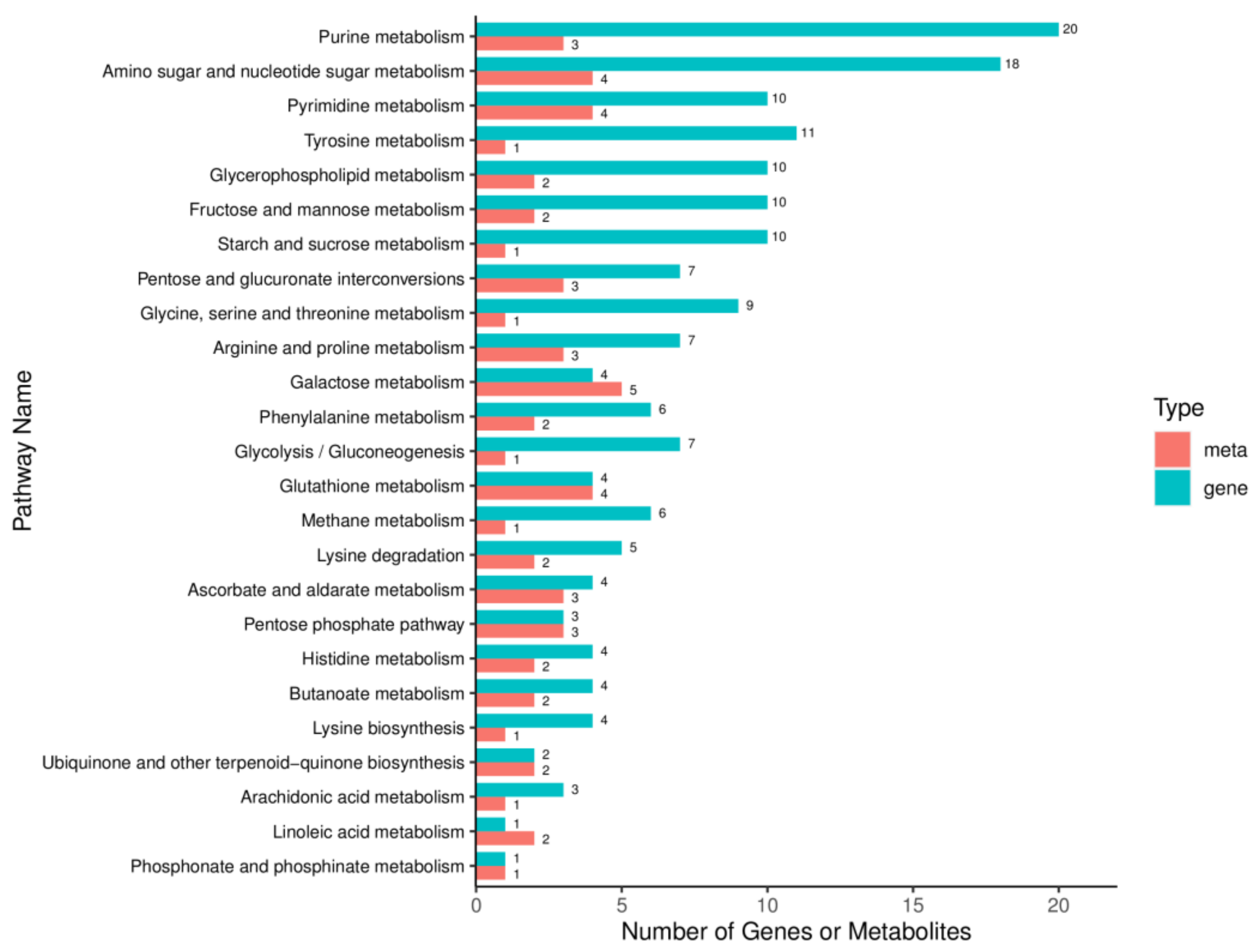
2.9. Integrated Analysis of Transcriptome and Metabolome in M. oryzae
3. Discussion
4. Materials and Methods
4.1. Strains
4.2. In Vitro Antagonistic Activity of B. velezensis GS-1
4.3. DNA Extraction, Genome Sequencing and Assembly
4.4. Genome Annotation of GS-1
4.5. Identification of GS-1
4.6. Analysis of CAZymes and Secondary Metabolic Genes
4.7. Preparation of Crude Lipopeptide Extracts from B. velezensis GS-1
4.8. RNA Extraction and Transcriptomics Analysis
4.9. Verification of RNA-Seq
4.10. Metabolome Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dagdas, Y.F.; Yoshino, K.; Dagdas, G.; Ryder, L.S.; Bielska, E.; Steinberg, G.; Talbot, N.J. Septin-mediated plant cell invasion by the rice blast fungus, Magnaporthe Oryzae. Science 2012, 336, 1590–1595. [Google Scholar] [CrossRef]
- Shan, H.; Zhao, M.; Chen, D.; Cheng, J.; Li, J.; Feng, Z.; Ma, Z.; An, D. Biocontrol of rice blast by the phenaminomethylacetic acid producer of Bacillus methylotrophicus strain BC79. Crop Prot. 2013, 44, 29–37. [Google Scholar] [CrossRef]
- Spence, C.; Alff, E.; Johnson, C.; Ramos, C.; Donofrio, N.; Sundaresan, V. Natural rice rhizospheric microbes suppress rice blast infections. BMC Plant Biol. 2014, 14, 130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, J.; Soytong, K.; Kanokmedhakul, S. Antifungal activity of Chaetomium elatum against Pyricularia oryzae causing rice blast. Int. J. Agric. Technol. 2016, 12, 1437–1447. [Google Scholar]
- Subhalakshmi, T.; Indira, D.S. Blast of rice in Manipur and its biocontrol by Pseudomonas fluorescens and Trichoderma sp. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 1619–1634. [Google Scholar]
- Vurukonda, S.S.K.P.; Giovanardi, D.; Stefani, E. Plant growth promoting and biocontrol activity of Streptomyces spp. as endophytes. Int. J. Mol. Sci. 2018, 19, 952. [Google Scholar] [CrossRef] [Green Version]
- Pang, Y.; Yang, J.; Chen, X.; Jia, Y.; Li, T.; Jin, J.; Liu, H.; Jiang, L.; Hao, Y.; Zhang, H.; et al. An antifungal chitosanase from Bacillus subtilis SH21. Molecules 2021, 26, 1863. [Google Scholar] [CrossRef]
- Jia, Y.; Huang, J.; Qi, L.; Zhang, X.; Liu, J.; Guan, H.; Wang, C.; Tang, G.; Dou, X.; Lu, M. Bacillus subtilis strain BS06 protects soybean roots from Fusarium oxysporum infection. FEMS Microbiol. Lett. 2021, 368, fnab102. [Google Scholar] [CrossRef]
- Kim, Y.; Choi, Y.; Yu, Y.; Ha, N.; Song, J. Plant growth-promoting activity of beta-propeller protein YxaL secreted from Bacillus velezensis strain GH1-13. PLoS ONE 2019, 25, e0207968. [Google Scholar] [CrossRef] [Green Version]
- Azizoglu, U. Bacillus thuringiensis as a Biofertilizer and Biostimulator: A Mini-Review of the Little-Known Plant Growth-Promoting Properties of Bt. Curr. Microbiol. 2019, 76, 1379–1385. [Google Scholar] [CrossRef]
- Chen, L.; Shi, H.; Heng, J.; Wang, D.; Bian, K. Antimicrobial, plant growth-promoting and genomic properties of the peanut endophyte Bacillus velezensis LDO2. Microbiol. Res. 2019, 218, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Qiu, Y.; Yang, S.; Wang, Y.; Wang, K.; Jiang, L.; Wang, H. Antagonistic activity of Bacillus velezensis SDTB038 against Phytophthora infestans in Potato. Plant Dis. 2021, 105, 1738–1747. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.H.; Won, S.J.; Maung, C.E.H.; Choi, J.H.; Choi, S.I.; Ajuna, H.B.; Ahn, Y.S. Bacillus velezensis CE 100 inhibits root rot diseases (Phytophthora spp.) and promotes growth of Japanese Cypress (Chamaecyparis obtusa Endlicher) Seedlings. Microorganisms 2021, 9, 821. [Google Scholar] [CrossRef] [PubMed]
- Pan, D.; Mionetto, A.; Tiscornia, S.; Bettucci, L. Endophytic bacteria from wheat grain as biocontrol agents of Fusarium graminearum and deoxynivalenol production in wheat. Mycotoxin Res. 2015, 31, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Sha, Y.X.; Zeng, Q.C.; Sui, S.T. Screening and application of Bacillus strains isolated from nonrhizospheric rice soil for the biocontrol of rice blast. Plant Pathol. J. 2020, 36, 231–243. [Google Scholar] [CrossRef]
- Zhou, H.; Ren, Z.; Zu, X.; Yu, X.; Zhu, H.; Li, X.; Zhong, J.; Liu, E. Efficacy of plant growth-promoting bacteria Bacillus cereus YN917 for biocontrol of rice blast. Front. Microbiol. 2021, 12, 684888. [Google Scholar] [CrossRef]
- Zhang, L.; Sun, C. Fengycins, cyclic lipopeptides from marine Bacillus subtilis strains, kill the plant-pathogenic fungus Magnaporthe grisea by inducing reactive oxygen species production and chromatin condensation. Appl. Environ. Microbiol. 2018, 84, e00445-18. [Google Scholar] [CrossRef] [Green Version]
- Jin, P.F.; Wang, H.N.; Tan, Z.; Xuan, Z.; Dahar, G.Y.; Li, Q.; Miao, W.; Liu, W. Antifungal mechanism of bacillomycin D from Bacillus velezensis HN-2 against Colletotrichum gloeosporioides Penz. Pestic. Biochem. Physiol. 2020, 163, 102–107. [Google Scholar] [CrossRef]
- Luo, W.; Liu, L.; Qi, G.; Yang, F.; Shi, X.; Zhao, X. Embedding Bacillus velezensis NH-1 in Microcapsules for Biocontrol of Cucumber Fusarium Wilt. Appl. Environ. Microbiol. 2019, 85, e03128-18. [Google Scholar] [CrossRef] [Green Version]
- Wei, Y.; Li, L.; Hu, W.; Ju, H.; Zhang, M.; Qin, Q.; Zhang, S.; Li, G. Suppression of rice blast by bacterial strains isolated from cultivated soda saline-sodic soils. Int. J. Environ. Res. Public Health 2020, 17, 5248. [Google Scholar] [CrossRef]
- Prasanna, S.; Prasannakumar, M.K.; Mahesh, H.B.; Babu, G.V.; Kirnaymayee, P.; Puneeth, M.E.; Narayan, K.; Pramesh, D. Diversity and biopotential of Bacillus velezensis strains A6 and P42 against rice blast and bacterial blight of pomegranate. Arch. Microbiol. 2021, 203, 4189–4199. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Koumoutsi, A.; Scholz, R.; Eisenreich, A.; Schneider, K.; Heinemeyer, I.; Morgenstern, B.; Voss, B.; Hess, W.R.; Reva, O.; et al. Comparative analysis of the complete genome sequence of the plant growth-promoting bacterium Bacillus amyloliquefaciens FZB42. Nat. Biotechnol. 2007, 25, 1007–1014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rabbee, M.F.; Ali, M.S.; Choi, J.; Hwang, B.S.; Jeong, S.C.; Baek, K.H. Bacillus velezensis: A valuable member of bioactive molecules within plant microbiomes. Molecules 2019, 24, 1046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, W.; Zhang, L.; Goodwin, P.; Xia, M.; Zhang, J.; Wang, Q.; Liang, J.; Sun, R.; Wu, C.; Yang, L. Isolation, identification and complete genome assembly of an endophytic Bacillus velezensis YB-130, potential biocontrol agent against Fusarium graminearum. Front. Microbiol. 2020, 11, 598285. [Google Scholar] [CrossRef]
- Scholz, R.; Molohon, K.J.; Nachtigall, J.; Vater, J.; Markley, A.L.; Süssmuth, R.D. Plantazolicin, a novel microcin B17/streptolysin S-like natural product from Bacillus amyloliquefaciens FZB42. J. Bacteriol. 2011, 193, 215–224. [Google Scholar] [CrossRef] [Green Version]
- Muhammad, F.R.; Baek, K.H. Antimicrobial Activities of lipopeptides and polyketides of Bacillus velezensis for agricultural applications. Molecules 2020, 25, 4973. [Google Scholar] [CrossRef]
- Liu, Y.; Teng, K.; Wang, T.; Dong, E.; Zhang, M.; Tao, Y.; Zhong, J. Antimicrobial Bacillus velezensis HC6: Production of three kinds of lipopeptides and biocontrol potential in maize. J. Appl. Microbiol. 2020, 128, 242–254. [Google Scholar] [CrossRef] [Green Version]
- Yan, Y.; Xu, W.; Chen, W.; Hu, Y.; Wang, Z. Complete genome sequence of Bacillus velezensis YYC, a bacterium isolated from the tomato rhizosphere. Arch Microbiol. 2021, 204, 44. [Google Scholar] [CrossRef]
- Deng, Q.; Wang, R.; Sun, D.; Sun, L.; Wang, Y.; Pu, Y.; Fang, Z.; Xu, D.; Liu, Y.; Ye, R.; et al. Complete genome of Bacillus velezensis CMT-6 and comparative genome analysis reveals lipopeptide diversity. Biochem. Genet. 2020, 58, 1–15. [Google Scholar] [CrossRef]
- Olanrewaju, O.S.; Ayilara, M.S.; Ayangbenro, A.S.; Babalola, O.O. Genome mining of three plant growth-promoting Bacillus Species from maize rhizosphere. Appl. Biochem. Biotechnol. 2021, 193, 3949–3969. [Google Scholar] [CrossRef]
- Sun, H.; Xiang, X.; Li, Q.; Lin, H.; Wang, X.; Sun, J.; Luo, L.; Zheng, A. Comparative genome analysis of Bacillus thuringiensis strain HD521 and HS18-1. Sci. Rep. 2021, 11, 16590. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.; Bruno, V.M.; Rasko, D.A.; Cuomo, C.A.; Muñoz, J.F.; Livny, J.; Shetty, A.C.; Mahurkar, A.; Dunning, H.J.C. Best practices on the differential expression analysis of multi-species RNA-seq. Genome Biol. 2021, 22, 121. [Google Scholar] [CrossRef] [PubMed]
- Shafi, J.; Tian, H.; Ji, M. Bacillus species as versatile weapons for plant pathogens: A review. Biotechnol. Biotechnol. Eq. 2017, 31, 446–459. [Google Scholar] [CrossRef] [Green Version]
- Wang, G.; Meng, J.; Tian, T.; Xiao, X.; Zhang, B.; Xiao, Y. Endophytic Bacillus velezensis strain B-36 is a potential biocontrol agent against lotus rot caused by Fusarium oxysporum. J. Appl. Microbiol. 2019, 128, 1153–1162. [Google Scholar] [CrossRef] [Green Version]
- Alvarez, F.; Castro, M.; Principe, A.; Borioli, G.; Fischer, S.; Mori, G.; Jofré, E. The plant-associated Bacillus amyloliquefaciens strains MEP218 and ARP23 capable of producing the cyclic lipopeptides iturin or surfactin and fengycin are effective in biocontrol of sclerotinia stem rot disease. J. Appl. Microbiol. 2012, 112, 159–174. [Google Scholar] [CrossRef]
- Xu, Y.; Chen, M.; Zhang, Y.; Wang, M.; Wang, Y.; Huang, Q.; Wang, X.; Wang, G. The phosphotransferase system gene ptsI in the endophytic bacterium Bacillus cereus is required for biofilm formation, colonization, and biocontrol against wheat sharp eyespot. FEMS Microbiol. Lett. 2014, 354, 142–152. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Yong, D.; Zhang, Y.; Shi, X.; Li, B.; Li, G.; Liang, W.; Wang, C. Streptomyces rochei A-1 induces resistance and defense-related responses against Botryosphaeria dothidea in apple fruit during storage. Postharvest Biol. Tech. 2016, 115, 30–37. [Google Scholar] [CrossRef]
- Hanif, A.; Zhang, F.; Li, P.; Li, C.; Xu, Y.; Zubair, M.; Zhang, M.; Jia, D.; Zhao, X.; Liang, J.; et al. Fengycin produced by Bacillus amyloliquefaciens FZB42 inhibits Fusarium graminearum growth and mycotoxins biosynthesis. Toxins 2019, 11, 295. [Google Scholar] [CrossRef] [Green Version]
- Yang, L.; Qu, X.; Xue, B.; Goodwin, P.H.; Lu, S.; Wang, J.; Wei, D.; Wu, C. Isolation and identification of Bacillus subtilis strain YB-05 and its antifungal substances showing antagonism against Gaeumannomyces graminis var. tritici. Biol. Control 2014, 85, 52–58. [Google Scholar] [CrossRef]
- Medeot, D.B.; Fernandez, M.; Morales, G.M.; Jofre, E. Fengycins from Bacillus amyloliquefaciens MEP2 18 exhibit antibacterial activity by producing alterations on the cell surface of the pathogens Xanthomonas axonopodis pv. vesicatoria and Pseudomonas aeruginosa PA01. Front. Microbiol. 2020, 10, 3107. [Google Scholar] [CrossRef]
- Wu, L.; Wu, H.; Chen, L.; Yu, X.; Borriss, R.; Gao, X. Difficidin and bacilysin from Bacillus amyloliquefaciens FZB42 have antibacterial activity against Xanthomonas oryzae rice pathogens. Sci. Rep. 2015, 5, 12975. [Google Scholar] [CrossRef] [PubMed]
- Arguelles, A.; Ongena, M.; Halimi, B.; Lara, Y.; Brans, A.; Joris, B.; Fickers, P. Bacillus amyloliquefaciens GA1 as a source of potent antibiotics and other secondary metabolites for biocontrol of plant pathogens. Microb. Cell Fact. 2009, 8, 63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ortiz, A.; Sansinenea, E. Macrolactin antibiotics: Amazing natural products. Med. Chem. 2020, 20, 584–600. [Google Scholar] [CrossRef] [PubMed]
- Palazzini, J.M.; Dunlap, C.A.; Bowman, M.J.; Chulze, S.N. Bacillus velezensis RC 218 as a biocontrol agent to reduce Fusarium head blight and deoxynivalenol accumulation: Genome sequencing and secondary metabolite cluster profiles. Microbiol. Res. 2016, 192, 30–36. [Google Scholar] [CrossRef]
- Shen, D.; Wang, J.; Dong, Y.; Zhang, M.; Tang, Z.; Xia, Q.; Nyawira, K.T.; Jing, M.; Dou, D.; Xia, A. The glycoside hydrolase 18 family chitinases are associated with development and virulence in the mosquito pathogen Pythium guiyangense. Fungal Genet. Biol. 2020, 135, 103290. [Google Scholar] [CrossRef]
- Xia, Y.; Ma, Z.; Qiu, M.; Guo, B.; Zhang, Q.; Jiang, H.; Zhang, B.; Lin, Y.; Xuan, M.; Sun, L.; et al. N-glycosylation shields Phytophthora sojae apoplastic effector PsXEG1 from a specific host aspartic protease. Proc. Natl. Acad. Sci. USA 2020, 117, 27685–27693. [Google Scholar] [CrossRef]
- García, N.; González, M.A.; González, C.; Brito, N. Simultaneous silencing of xylanase genes in Botrytis cinerea. Front. Plant Sci. 2017, 22, 2174. [Google Scholar] [CrossRef]
- Kim, T.; Lee, S.H.; Oh, Y.T.; Jeon, J. A histone deacetylase, MoHDA1 regulates asexual development and virulence in the rice blast fungus. Plant Pathol. J. 2020, 36, 314–322. [Google Scholar] [CrossRef]
- Ma, H.; Li, L.; Gai, Y.; Zhang, X.; Chen, Y.; Zhuo, X.; Cao, Y.; Jiao, C.; Gmitter, F.G.; Li, H. Histone acetyltransferases and deacetylases are required for virulence, conidiation, DNA damage repair, and multiple stresses resistance of Alternaria alternate. Front. Microbiol. 2021, 12, 783633. [Google Scholar] [CrossRef]
- Kim, I.Y.; Choi, B.; Park, W.R.; Kim, Y.J.; Kim, B.E.; Mun, S.; Choic, H.S.; Kim, D.K. Nuclear receptor HR96 up-regulates cytochrome P450 for insecticide detoxification in Tribolium castaneum. Pest Manag. Sci. 2022, 78, 230–239. [Google Scholar] [CrossRef]
- Wang, J.; Ohno, H.; Ide, Y.; Ichinose, H.; Mori, T.; Kawagishi, H.; Hirai, H. Identification of the cytochrome P450 involved in the degradation of neonicotinoid insecticide acetamiprid in Phanerochaete chrysosporium. J. Hazard. Mater. 2019, 371, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Shan, T.; Liu, Y.; Shi, X.; Gao, X. Identification of a novel cytochrome P450 CYP3356A1 linked with insecticide detoxification in Bradysia odoriphaga. Pest Manag. Sci. 2019, 75, 1006–1013. [Google Scholar] [CrossRef] [PubMed]
- Green, R.; Sang, H.; Im, J.; Jung, G. Chlorothalonil biotransformation by cytochrome P450 monooxygenases in Sclerotinia homoeocarpa. FEMS Microbiol. Lett. 2018, 365, fny214. [Google Scholar] [CrossRef]
- Wang, Z.; Jiang, S.; Mota-Sanchez, D.; Wang, W.; Li, X.; Gao, Y.; Lu, X.; Yang, X. Cytochrome P450-Mediated λ-Cyhalothrin-Resistance in a Field Strain of Helicoverpa armigera from Northeast China. J. Agric. Food Chem. 2019, 67, 3546–3553. [Google Scholar] [CrossRef] [PubMed]
- Qian, H.; Du, J.; Chi, M.; Sun, X.; Liang, W.; Huang, J.; Li, B. The Y137H mutation in the cytochrome P450 FgCYP51B protein confers reduced sensitivity to tebuconazole in Fusarium graminearum. Pest Manag. Sci. 2018, 74, 1472–1477. [Google Scholar] [CrossRef]
- Vela-Corcía, D.; Srivastava, D.A.; Dafa-Berger, A.; Rotem, N.; Barda, O.; Levy, M. MFS transporter from Botrytis cinerea provides tolerance to glucosinolate-breakdown products and is required for pathogenicity. Nat. Commun. 2019, 10, 2886. [Google Scholar] [CrossRef] [PubMed] [Green Version]




| Gene Clusters | Types | Genome Locations | Most Similar Known Clusters | Similarity |
|---|---|---|---|---|
| Cluster 1 | NRPS | 304,866–369,675 | surfactin | 82% |
| Cluster 2 | thiopeptide, LAP | 585,385–614,269 | kijanimicin | 4% |
| Cluster 3 | LAP | 698,756–720,938 | plantazolicin | 91% |
| Cluster 4 | PKS-like | 930,568–971,812 | butirosin A/butirosin B | 7% |
| Cluster 5 | terpene | 1,056,704–1,074,038 | ||
| Cluster 6 | transAT-PKS | 1,372,752–1,460,872 | macrolactin H | 100% |
| Cluster 7 | transAT-PKS, T3PKS, transAT-PKS-like, NRPS | 1,680,080–1,789,798 | bacillaene | 100% |
| Cluster 8 | NRPS, transAT-PKS, betalactone | 1,847,452–1,983,301 | fengycin | 100% |
| Cluster 9 | terpene | 2,006,887–2,028,770 | ||
| Cluster 10 | T3PKS | 2,097,450–2,138,550 | ||
| Cluster 11 | transAT-PKS-like, transAT-PKS | 2,391,006–2,497,179 | difficidin | 100% |
| Cluster 12 | NRPS, bacteriocin | 3,126,907–3,178,699 | bacillibactin | 100% |
| Cluster 13 | other | 3,691,456–3,732,874 | bacilysin | 100% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Zhao, M.; Chen, W.; Yu, H.; Jia, W.; Pan, H.; Zhang, X. Multi-Omics Techniques for Analysis Antifungal Mechanisms of Lipopeptides Produced by Bacillus velezensis GS-1 against Magnaporthe oryzae In Vitro. Int. J. Mol. Sci. 2022, 23, 3762. https://doi.org/10.3390/ijms23073762
Zhang Y, Zhao M, Chen W, Yu H, Jia W, Pan H, Zhang X. Multi-Omics Techniques for Analysis Antifungal Mechanisms of Lipopeptides Produced by Bacillus velezensis GS-1 against Magnaporthe oryzae In Vitro. International Journal of Molecular Sciences. 2022; 23(7):3762. https://doi.org/10.3390/ijms23073762
Chicago/Turabian StyleZhang, Yanhua, Meixi Zhao, Wei Chen, Huilin Yu, Wantong Jia, Hongyu Pan, and Xianghui Zhang. 2022. "Multi-Omics Techniques for Analysis Antifungal Mechanisms of Lipopeptides Produced by Bacillus velezensis GS-1 against Magnaporthe oryzae In Vitro" International Journal of Molecular Sciences 23, no. 7: 3762. https://doi.org/10.3390/ijms23073762
APA StyleZhang, Y., Zhao, M., Chen, W., Yu, H., Jia, W., Pan, H., & Zhang, X. (2022). Multi-Omics Techniques for Analysis Antifungal Mechanisms of Lipopeptides Produced by Bacillus velezensis GS-1 against Magnaporthe oryzae In Vitro. International Journal of Molecular Sciences, 23(7), 3762. https://doi.org/10.3390/ijms23073762





