FKBP52 in Neuronal Signaling and Neurodegenerative Diseases: A Microtubule Story
Abstract
1. Introduction
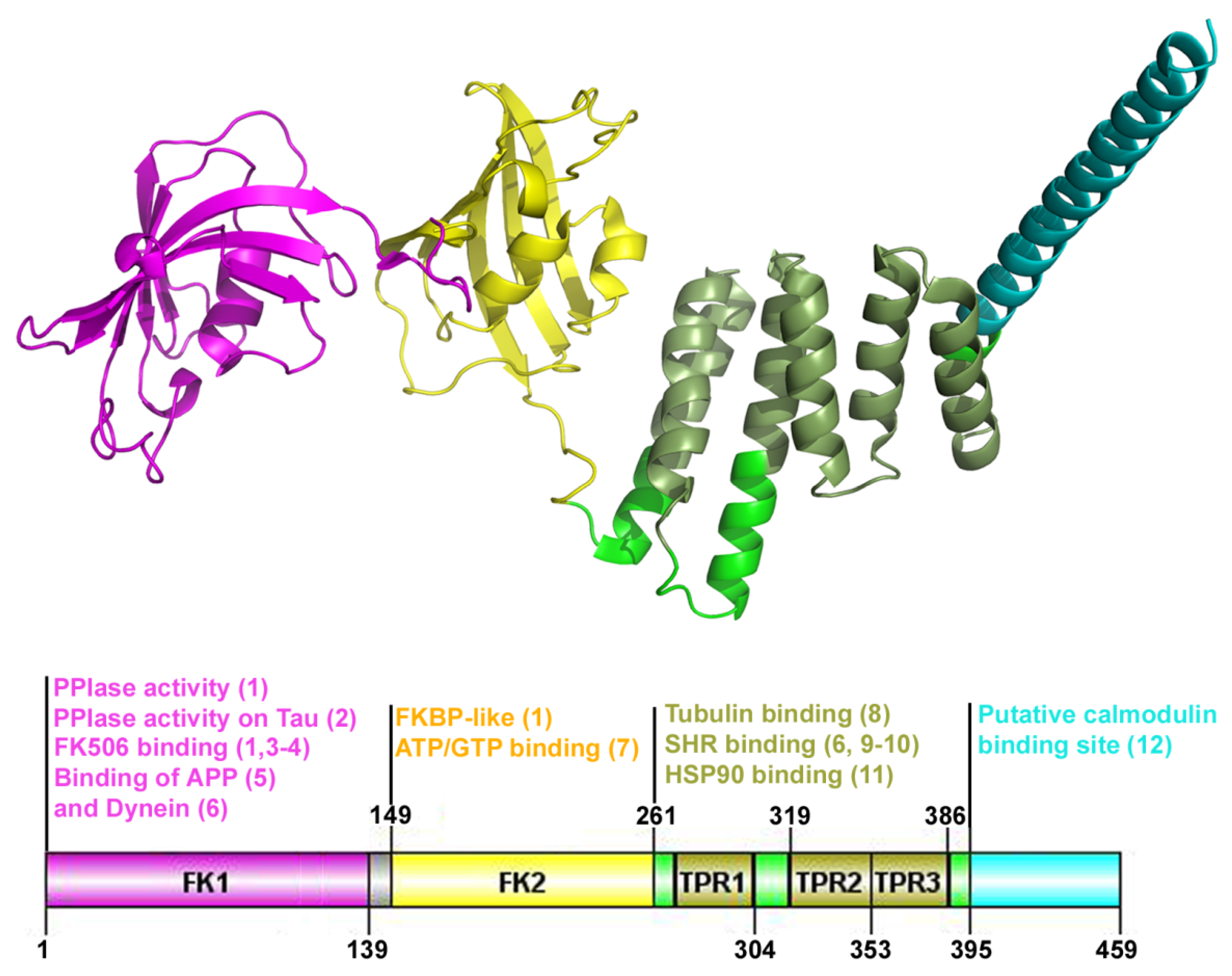
2. FKBP52, a Member of FKBP Family Proteins
3. FKBP52 in Microtubule Dynamics
4. FKBP52 in Microtubule-Dependent Trafficking
5. FKBP52 in Microtubule-Associated Protein Aggregation and Clearance
6. Involvement of FKBP52 in Neurodegenerative Diseases
6.1. FKBP52 and Microtubule Dynamics
6.2. FKBP52 in Protein Aggregation and Clearance
6.3. FKBP52 in Stress and Inflammation
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AD | Alzheimer disease |
| ALP | autophagy lysosomal pathway |
| APP | amyloid precursor protein |
| Atox1 | antioxidant protein 1 |
| Aβ | amyloid beta |
| ER | endoplasmic reticulum |
| FK1 | FK506 binding domain 1 |
| FKBPs | FK506-binding proteins |
| FTLD-Tau | frontotemporal lobar degeneration linked to a point-mutation of Tau gene |
| GR | glucocorticoid receptor |
| Hsp90 | heat shock protein of 90 kDa |
| hTERT | human telomerase reverse transcriptase |
| IRF4 | interferon regulating factor |
| MT | microtubule |
| Nf-kappaB | nuclear factor K-light chain enhancer of activated lymphocyte B cells |
| PD | Parkinson disease |
| PPIase | peptidyl-prolyl cis trans isomerase |
| SHR | steroid hormone receptor |
| Tau | tubulin associated unit |
| TPR | tetratricopeptide repeat |
| α-syn | alpha-synuclein |
References
- Schiene, C.; Fischer, G. Enzymes that catalyse the restructuring of proteins. Curr. Opin. Struct. Biol. 2000, 10, 40–45. [Google Scholar] [CrossRef]
- Wedemeyer, W.J.; Welker, E.; Scheraga, H.A. Proline cis-trans isomerization and protein folding. Biochemistry 2002, 41, 14637–14644. [Google Scholar] [CrossRef] [PubMed]
- Chambraud, B.; Rouviere-Fourmy, N.; Radanyi, C.; Hsiao, K.; Peattie, D.A.; Livingston, D.J.; Baulieu, E.E. Overexpression of p59-HBI (FKBP90), full length and domains, and characterization of PPlase activity. Biochem. Biophys. Res. Commun. 1993, 196, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, S.L. Chemistry and biology of the immunophilins and their immunosuppressive ligands. Science 1991, 251, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Harding, M.W.; Galat, A.; Uehling, D.E.; Schreiber, S.L. A receptor for the immunosuppressant FK506 is a cis-trans peptidyl-prolyl isomerase. Nature 1989, 341, 758–760. [Google Scholar] [CrossRef] [PubMed]
- Steiner, J.P.; Dawson, T.M.; Fotuhi, M.; Glatt, C.E.; Snowman, A.M.; Cohen, N.; Snyder, S.H. High brain densities of the immunophilin FKBP colocalized with calcineurin. Nature 1992, 358, 584–587. [Google Scholar] [CrossRef] [PubMed]
- Snyder, S.H.; Sabatini, D.M. Immunophilins and the nervous system. Nat. Med. 1995, 1, 32–37. [Google Scholar] [CrossRef]
- Storer, C.L.; Dickey, C.A.; Galigniana, M.D.; Rein, T.; Cox, M.B. FKBP51 and FKBP52 in signaling and disease. Trends Endocrinol. Metab. TEM 2011, 22, 481–490. [Google Scholar] [CrossRef]
- Guy, N.C.; Garcia, Y.A.; Sivils, J.C.; Galigniana, M.D.; Cox, M.B. Functions of the Hsp90-binding FKBP immunophilins. Sub-Cell. Biochem. 2015, 78, 35–68. [Google Scholar] [CrossRef]
- Hong, C.; Li, T.; Zhang, F.; Wu, X.; Chen, X.; Cui, X.; Zhang, G.; Cui, Y. Elevated FKBP52 expression indicates a poor outcome in patients with breast cancer. Oncol. Lett. 2017, 14, 5379–5385. [Google Scholar] [CrossRef][Green Version]
- Maeda, K.; Habara, M.; Kawaguchi, M.; Matsumoto, H.; Hanaki, S.; Masaki, T.; Sato, Y.; Matsuyama, H.; Kunieda, K.; Nakagawa, H.; et al. FKBP51 and FKBP52 regulate androgen receptor dimerization and proliferation in prostate cancer cells. Mol. Oncol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Chambraud, B.; Sardin, E.; Giustiniani, J.; Dounane, O.; Schumacher, M.; Goedert, M.; Baulieu, E.E. A role for FKBP52 in Tau protein function. Proc. Natl. Acad. Sci. USA 2010, 107, 2658–2663. [Google Scholar] [CrossRef] [PubMed]
- Gerard, M.; Deleersnijder, A.; Daniels, V.; Schreurs, S.; Munck, S.; Reumers, V.; Pottel, H.; Engelborghs, Y.; Van den Haute, C.; Taymans, J.M.; et al. Inhibition of FK506 binding proteins reduces alpha-synuclein aggregation and Parkinson’s disease-like pathology. J. Neurosci. Off. J. Soc. Neurosci. 2010, 30, 2454–2463. [Google Scholar] [CrossRef]
- Fusco, D.; Vargiolu, M.; Vidone, M.; Mariani, E.; Pennisi, L.F.; Bonora, E.; Capellari, S.; Dirnberger, D.; Baumeister, R.; Martinelli, P.; et al. The RET51/FKBP52 complex and its involvement in Parkinson disease. Hum. Mol. Genet. 2010, 19, 2804–2816. [Google Scholar] [CrossRef]
- Siekierka, J.J.; Hung, S.H.; Poe, M.; Lin, C.S.; Sigal, N.H. A cytosolic binding protein for the immunosuppressant FK506 has peptidyl-prolyl isomerase activity but is distinct from cyclophilin. Nature 1989, 341, 755–757. [Google Scholar] [CrossRef] [PubMed]
- Avramut, M.; Achim, C.L. Immunophilins and their ligands: Insights into survival and growth of human neurons. Physiol. Behav. 2002, 77, 463–468. [Google Scholar] [CrossRef]
- Nigam, S.K.; Jin, Y.J.; Jin, M.J.; Bush, K.T.; Bierer, B.E.; Burakoff, S.J. Localization of the FK506-binding protein, FKBP 13, to the lumen of the endoplasmic reticulum. Biochem. J. 1993, 294, 511–515. [Google Scholar] [CrossRef]
- Tremmel, D.; Tropschug, M. Neurospora crassa FKBP22 is a novel er chaperone and functionally cooperates with BiP. J. Mol. Biol. 2007, 369, 55–68. [Google Scholar] [CrossRef]
- Patterson, C.E.; Schaub, T.; Coleman, E.J.; Davis, E.C. Developmental regulation of FKBP65. An er-localized extracellular matrix binding-protein. Mol. Biol. Cell 2000, 11, 3925–3935. [Google Scholar] [CrossRef]
- Jin, Y.J.; Albers, M.W.; Lane, W.S.; Bierer, B.E.; Schreiber, S.L.; Burakoff, S.J. Molecular cloning of a membrane-associated human FK506- and rapamycin-binding protein, FKBP-13. Proc. Natl. Acad. Sci. USA 1991, 88, 6677–6681. [Google Scholar] [CrossRef]
- Rulten, S.L.; Kinloch, R.A.; Tateossian, H.; Robinson, C.; Gettins, L.; Kay, J.E. The human FK506-binding proteins: Characterization of human FKBP19. Mamm. Genome Off. J. Int. Mamm. Genome Soc. 2006, 17, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Baumann, M.; Giunta, C.; Krabichler, B.; Ruschendorf, F.; Zoppi, N.; Colombi, M.; Bittner, R.E.; Quijano-Roy, S.; Muntoni, F.; Cirak, S.; et al. Mutations in FKBP14 cause a variant of Ehlers-Danlos syndrome with progressive kyphoscoliosis, myopathy, and hearing loss. Am. J. Hum. Genet. 2012, 90, 201–216. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Yabe, D.; Kanazawa, N.; Tashiro, K.; Sasayama, S.; Honjo, T. Molecular cloning, characterization, and chromosomal localization of fkbp23, a novel FK506-binding protein with Ca2+-binding ability. Genomics 1998, 54, 89–98. [Google Scholar] [CrossRef]
- Riviere, S.; Menez, A.; Galat, A. On the localization of FKBP25 in T-lymphocytes. FEBS Lett. 1993, 315, 247–251. [Google Scholar] [CrossRef]
- Galigniana, M.D.; Echeverria, P.C.; Erlejman, A.G.; Piwien-Pilipuk, G. Role of molecular chaperones and TPR-domain proteins in the cytoplasmic transport of steroid receptors and their passage through the nuclear pore. Nucleus 2010, 1, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Helander, S.; Montecchio, M.; Lemak, A.; Fares, C.; Almlof, J.; Yi, Y.; Yee, A.; Arrowsmith, C.; DhePaganon, S.; Sunnerhagen, M. Basic tilted helix bundle - a new protein fold in human FKBP25/FKBP3 and HectD1. Biochem. Biophys. Res. Commun. 2014, 447, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, O.; Nakamura, F.; Yamashita, N.; Tomita, Y.; Suto, F.; Okada, T.; Iwamatsu, A.; Kondo, E.; Fujisawa, H.; Takei, K.; et al. FKBP133: A novel mouse FK506-binding protein homolog alters growth cone morphology. Biochem. Biophys. Res. Commun. 2006, 346, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Gallo, L.I.; Lagadari, M.; Piwien-Pilipuk, G.; Galigniana, M.D. The 90-kda heat-shock protein (Hsp90)-binding immunophilin fkbp51 is a mitochondrial protein that translocates to the nucleus to protect cells against oxidative stress. J. Biol. Chem. 2011, 286, 30152–30160. [Google Scholar] [CrossRef]
- Meduri, G.; Guillemeau, K.; Dounane, O.; Sazdovitch, V.; Duyckaerts, C.; Chambraud, B.; Baulieu, E.E.; Giustiniani, J. Caspase-cleaved tau-d(421) is colocalized with the immunophilin FKBP52 in the autophagy-endolysosomal system of Alzheimer’s disease neurons. Neurobiol. Aging 2016, 46, 124–137. [Google Scholar] [CrossRef]
- Perrot-Applanat, M.; Cibert, C.; Geraud, G.; Renoir, J.M.; Baulieu, E.E. The 59 kda FK506-binding protein, a 90 kda heat shock protein binding immunophilin (FKBP59-HBI), is associated with the nucleus, the cytoskeleton and mitotic apparatus. J. Cell Sci. 1995, 108, 2037–2051. [Google Scholar] [CrossRef]
- Chambraud, B.; Belabes, H.; Fontaine-Lenoir, V.; Fellous, A.; Baulieu, E.E. The immunophilin FKBP52 specifically binds to tubulin and prevents microtubule formation. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2007, 21, 2787–2797. [Google Scholar] [CrossRef] [PubMed]
- Dilworth, D.; Gudavicius, G.; Xu, X.; Boyce, A.K.J.; O’Sullivan, C.; Serpa, J.J.; Bilenky, M.; Petrochenko, E.V.; Borchers, C.H.; Hirst, M.; et al. The prolyl isomerase FKBP25 regulates microtubule polymerization impacting cell cycle progression and genomic stability. Nucleic Acids Res. 2018, 46, 2459–2478. [Google Scholar] [CrossRef] [PubMed]
- Ikura, T.; Ito, N. Peptidyl-prolyl isomerase activity of FK506 binding protein 12 prevents tau peptide from aggregating. Protein Eng. Des. Sel. PEDS 2013, 26, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.L.; Liu, T.Y.; Kung, F.L. FKBP12 regulates the localization and processing of amyloid precursor protein in human cell lines. J. Biosci. 2014, 39, 85–95. [Google Scholar] [CrossRef]
- Gant, J.C.; Blalock, E.M.; Chen, K.C.; Kadish, I.; Porter, N.M.; Norris, C.M.; Thibault, O.; Landfield, P.W. FK506-binding protein 1b/12.6: A key to aging-related hippocampal Ca2+ dysregulation? Eur. J. Pharmacol. 2014, 739, 74–82. [Google Scholar] [CrossRef][Green Version]
- Ochocka, A.M.; Kampanis, P.; Nicol, S.; Allende-Vega, N.; Cox, M.; Marcar, L.; Milne, D.; Fuller-Pace, F.; Meek, D. FKBP25, a novel regulator of the p53 pathway, induces the degradation of MDM2 and activation of p53. FEBS Lett. 2009, 583, 621–626. [Google Scholar] [CrossRef]
- Bailus, B.J.; Scheeler, S.M.; Simons, J.; Sanchez, M.A.; Tshilenge, K.T.; Creus-Muncunill, J.; Naphade, S.; Lopez-Ramirez, A.; Zhang, N.; Lakshika Madushani, K.; et al. Modulating FKBP5/FKBP51 and autophagy lowers HTT (huntingtin) levels. Autophagy 2021, 17, 4119–4140. [Google Scholar] [CrossRef]
- Jarczowski, F.; Fischer, G.; Edlich, F. FKBP36 forms complexes with clathrin and Hsp72 in spermatocytes. Biochemistry 2008, 47, 6946–6952. [Google Scholar] [CrossRef]
- Francke, U. Williams-Beuren syndrome: Genes and mechanisms. Hum. Mol. Genet. 1999, 8, 1947–1954. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, Y.; Li, H.; Zhang, W.; Wu, D.; Mi, H. The mouse FKBP23 binds to bip in er and the binding of c-terminal domain is interrelated with Ca2+ concentration. FEBS Lett. 2004, 559, 57–60. [Google Scholar] [CrossRef]
- Bai, X.; Ma, D.; Liu, A.; Shen, X.; Wang, Q.J.; Liu, Y.; Jiang, Y. Rheb activates mTOR by antagonizing its endogenous inhibitor, FKBP38. Science 2007, 318, 977–980. [Google Scholar] [CrossRef]
- Brown, C.A.; Schmidt, C.; Poulter, M.; Hummerich, H.; Klohn, P.C.; Jat, P.; Mead, S.; Collinge, J.; Lloyd, S.E. In vitro screen of prion disease susceptibility genes using the scrapie cell assay. Hum. Mol. Genet. 2014, 23, 5102–5108. [Google Scholar] [CrossRef] [PubMed]
- Sezen, S.F.; Lagoda, G.; Burnett, A.L. Role of immunophilins in recovery of erectile function after cavernous nerve injury. J. Sex. Med. 2009, 6 (Suppl 3), 340–346. [Google Scholar] [CrossRef]
- Alanay, Y.; Avaygan, H.; Camacho, N.; Utine, G.E.; Boduroglu, K.; Aktas, D.; Alikasifoglu, M.; Tuncbilek, E.; Orhan, D.; Bakar, F.T.; et al. Mutations in the gene encoding the RER protein FKBP65 cause autosomal-recessive osteogenesis imperfecta. Am. J. Hum. Genet. 2010, 86, 551–559. [Google Scholar] [CrossRef] [PubMed]
- van de Hoef, D.L.; Hughes, J.; Livne-Bar, I.; Garza, D.; Konsolaki, M.; Boulianne, G.L. Identifying genes that interact with Drosophila presenilin and amyloid precursor protein. Genesis 2009, 47, 246–260. [Google Scholar] [CrossRef]
- Jarczowski, F.; Jahreis, G.; Erdmann, F.; Schierhorn, A.; Fischer, G.; Edlich, F. FKBP36 is an inherent multifunctional glyceraldehyde-3-phosphate dehydrogenase inhibitor. J. Biol. Chem. 2009, 284, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Shirane, M.; Nakayama, K.I. Inherent calcineurin inhibitor FKBP38 targets Bcl-2 to mitochondria and inhibits apoptosis. Nat. Cell Biol. 2003, 5, 28–37. [Google Scholar] [CrossRef]
- Hahle, A.; Merz, S.; Meyners, C.; Hausch, F. The many faces of FKBP51. Biomolecules 2019, 9, 35. [Google Scholar] [CrossRef]
- Linnert, M.; Lin, Y.J.; Manns, A.; Haupt, K.; Paschke, A.K.; Fischer, G.; Weiwad, M.; Lucke, C. The FKBP-type domain of the human aryl hydrocarbon receptor-interacting protein reveals an unusual Hsp90 interaction. Biochemistry 2013, 52, 2097–2107. [Google Scholar] [CrossRef]
- McKeen, H.D.; McAlpine, K.; Valentine, A.; Quinn, D.J.; McClelland, K.; Byrne, C.; O’Rourke, M.; Young, S.; Scott, C.J.; McCarthy, H.O.; et al. A novel FKBP-like binding protein interacts with the glucocorticoid receptor and regulates steroid receptor signaling. Endocrinology 2008, 149, 5724–5734. [Google Scholar] [CrossRef]
- Gerard, M.; Debyser, Z.; Desender, L.; Baert, J.; Brandt, I.; Baekelandt, V.; Engelborghs, Y. FK506 binding protein 12 differentially accelerates fibril formation of wild type alpha-synuclein and its clinical mutants A30P or A53T. J. Neurochem. 2008, 106, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Deleersnijder, A.; Van Rompuy, A.S.; Desender, L.; Pottel, H.; Buee, L.; Debyser, Z.; Baekelandt, V.; Gerard, M. Comparative analysis of different peptidyl-prolyl isomerases reveals FK506-binding protein 12 as the most potent enhancer of alpha-synuclein aggregation. J. Biol. Chem. 2011, 286, 26687–26701. [Google Scholar] [CrossRef] [PubMed]
- Sugata, H.; Matsuo, K.; Nakagawa, T.; Takahashi, M.; Mukai, H.; Ono, Y.; Maeda, K.; Akiyama, H.; Kawamata, T. A peptidyl-prolyl isomerase, FKBP12, accumulates in alzheimer neurofibrillary tangles. Neurosci. Lett 2009, 459, 96–99. [Google Scholar] [CrossRef] [PubMed]
- Giustiniani, J.; Chambraud, B.; Sardin, E.; Dounane, O.; Guillemeau, K.; Nakatani, H.; Paquet, D.; Kamah, A.; Landrieu, I.; Lippens, G.; et al. Immunophilin FKBP52 induces Tau-P301L filamentous assembly in vitro and modulates its activity in a model of tauopathy. Proc. Natl. Acad. Sci. USA 2014, 111, 4584–4589. [Google Scholar] [CrossRef] [PubMed]
- Giustiniani, J.; Guillemeau, K.; Dounane, O.; Sardin, E.; Huvent, I.; Schmitt, A.; Hamdane, M.; Buee, L.; Landrieu, I.; Lippens, G.; et al. The FK506-binding protein FKBP52 in vitro induces aggregation of truncated tau forms with prion-like behavior. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2015, 29, 3171–3181. [Google Scholar] [CrossRef]
- Kamah, A.; Cantrelle, F.X.; Huvent, I.; Giustiniani, J.; Guillemeau, K.; Byrne, C.; Jacquot, Y.; Landrieu, I.; Baulieu, E.E.; Smet, C.; et al. Isomerization and oligomerization of truncated and mutated tau forms by FKBP52 are independent processes. J. Mol. Biol. 2016, 428, 1080–1090. [Google Scholar] [CrossRef] [PubMed]
- Tatro, E.T.; Everall, I.P.; Kaul, M.; Achim, C.L. Modulation of glucocorticoid receptor nuclear translocation in neurons by immunophilins FKBP51 and FKBP52: Implications for major depressive disorder. Brain Res. 2009, 1286, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.Y.; Her, J.; Oh, S.Y.; Chung, I.K. Hsp90-binding immunophilin FKBP52 modulates telomerase activity by promoting the cytoplasmic retrotransport of htert. Biochem. J. 2016, 473, 3517–3532. [Google Scholar] [CrossRef]
- Erlejman, A.G.; De Leo, S.A.; Mazaira, G.I.; Molinari, A.M.; Camisay, M.F.; Fontana, V.; Cox, M.B.; Piwien-Pilipuk, G.; Galigniana, M.D. Nf-kappab transcriptional activity is modulated by FK506-binding proteins FKBP51 and FKBP52: A role for peptidyl-prolyl isomerase activity. J. Biol. Chem. 2014, 289, 26263–26276. [Google Scholar] [CrossRef]
- Mamane, Y.; Sharma, S.; Petropoulos, L.; Lin, R.; Hiscott, J. Posttranslational regulation of IRF-4 activity by the immunophilin FKBP52. Immunity 2000, 12, 129–140. [Google Scholar] [CrossRef]
- Chambraud, B.; Daguinot, C.; Guillemeau, K.; Genet, M.; Dounane, O.; Meduri, G.; Pous, C.; Baulieu, E.E.; Giustiniani, J. Decrease of neuronal FKBP4/FKBP52 modulates perinuclear lysosomal positioning and mapt/tau behavior during mapt/tau-induced proteotoxic stress. Autophagy 2021, 17, 3491–3510. [Google Scholar] [CrossRef]
- Blair, L.J.; Baker, J.D.; Sabbagh, J.J.; Dickey, C.A. The emerging role of peptidyl-prolyl isomerase chaperones in tau oligomerization, amyloid processing, and Alzheimer’s disease. J. Neurochem. 2015, 133, 1–13. [Google Scholar] [CrossRef]
- Jinwal, U.K.; Koren, J., 3rd; Borysov, S.I.; Schmid, A.B.; Abisambra, J.F.; Blair, L.J.; Johnson, A.G.; Jones, J.R.; Shults, C.L.; O’Leary, J.C., 3rd; et al. The Hsp90 cochaperone, FKBP51, increases tau stability and polymerizes microtubules. J. Neurosci. Off. J. Soc. Neurosci. 2010, 30, 591–599. [Google Scholar] [CrossRef]
- Lagadari, M.; Zgajnar, N.R.; Gallo, L.I.; Galigniana, M.D. Hsp90-binding immunophilin FKBP51 forms complexes with htert enhancing telomerase activity. Mol. Oncol. 2016, 10, 1086–1098. [Google Scholar] [CrossRef]
- Gassen, N.C.; Hartmann, J.; Schmidt, M.V.; Rein, T. FKBP5/FKBP51 enhances autophagy to synergize with antidepressant action. Autophagy 2015, 11, 578–580. [Google Scholar] [CrossRef] [PubMed]
- Bhujabal, Z.; Birgisdottir, A.B.; Sjottem, E.; Brenne, H.B.; Overvatn, A.; Habisov, S.; Kirkin, V.; Lamark, T.; Johansen, T. FKBP8 recruits LC3A to mediate parkin-independent mitophagy. EMBO Rep. 2017, 18, 947–961. [Google Scholar] [CrossRef]
- Viklund, I.M.; Aspenstrom, P.; Meas-Yedid, V.; Zhang, B.; Kopec, J.; Agren, D.; Schneider, G.; D'Amato, M.; Olivo-Marin, J.C.; Sansonetti, P.; et al. WAFL, a new protein involved in regulation of early endocytic transport at the intersection of actin and microtubule dynamics. Exp. Cell Res. 2009, 315, 1040–1052. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Li, P.; Liu, Y.; Lou, Z.; Ding, Y.; Shu, C.; Ye, S.; Bartlam, M.; Shen, B.; Rao, Z. 3d structure of human FK506-binding protein 52: Implications for the assembly of the glucocorticoid receptor/Hsp90/immunophilin heterocomplex. Proc. Natl. Acad. Sci. USA 2004, 101, 8348–8353. [Google Scholar] [CrossRef] [PubMed]
- Massol, N.; Lebeau, M.C.; Renoir, J.M.; Faber, L.E.; Baulieu, E.E. Rabbit FKBP59-heat shock protein binding immunophillin (HBI) is a calmodulin binding protein. Biochem. Biophys. Res. Commun. 1992, 187, 1330–1335. [Google Scholar] [CrossRef]
- Le Bihan, S.; Renoir, J.M.; Radanyi, C.; Chambraud, B.; Joulin, V.; Catelli, M.G.; Baulieu, E.E. The mammalian heat shock protein binding immunophilin (p59/HBI) is an ATP and GTP binding protein. Biochem. Biophys. Res. Commun. 1993, 195, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.; Weikl, T.; Bugl, H.; Buchner, J. Chaperone function of Hsp90-associated proteins. Science 1996, 274, 1715–1717. [Google Scholar] [CrossRef] [PubMed]
- Radanyi, C.; Chambraud, B.; Baulieu, E.E. The ability of the immunophilin FKBP59-HBI to interact with the 90-kda heat shock protein is encoded by its tetratricopeptide repeat domain. Proc. Natl. Acad. Sci. USA 1994, 91, 11197–11201. [Google Scholar] [CrossRef] [PubMed]
- Kelliher, M.T.; Saunders, H.A.; Wildonger, J. Microtubule control of functional architecture in neurons. Curr. Opin. Neurobiol. 2019, 57, 39–45. [Google Scholar] [CrossRef]
- Janke, C.; Bulinski, J.C. Post-translational regulation of the microtubule cytoskeleton: Mechanisms and functions. Nat. Rev. Mol. Cell Biol. 2011, 12, 773–786. [Google Scholar] [CrossRef] [PubMed]
- Tai, P.K.; Maeda, Y.; Nakao, K.; Wakim, N.G.; Duhring, J.L.; Faber, L.E. A 59-kilodalton protein associated with progestin, estrogen, androgen, and glucocorticoid receptors. Biochemistry 1986, 25, 5269–5275. [Google Scholar] [CrossRef]
- Renoir, J.M.; Radanyi, C.; Jung-Testas, I.; Faber, L.E.; Baulieu, E.E. The nonactivated progesterone receptor is a nuclear heterooligomer. J. Biol. Chem. 1990, 265, 14402–14406. [Google Scholar] [CrossRef]
- Silverstein, A.M.; Galigniana, M.D.; Kanelakis, K.C.; Radanyi, C.; Renoir, J.M.; Pratt, W.B. Different regions of the immunophilin FKBP52 determine its association with the glucocorticoid receptor, Hsp90, and cytoplasmic dynein. J. Biol. Chem. 1999, 274, 36980–36986. [Google Scholar] [CrossRef]
- Sinars, C.R.; Cheung-Flynn, J.; Rimerman, R.A.; Scammell, J.G.; Smith, D.F.; Clardy, J. Structure of the large FK506-binding protein FKBP51, an Hsp90-binding protein and a component of steroid receptor complexes. Proc. Natl. Acad. Sci. USA 2003, 100, 868–873. [Google Scholar] [CrossRef]
- Mustafi, S.M.; LeMaster, D.M.; Hernandez, G. Differential conformational dynamics in the closely homologous FK506-binding domains of FKBP51 and FKBP52. Biochem. J. 2014, 461, 115–123. [Google Scholar] [CrossRef]
- Wochnik, G.M.; Ruegg, J.; Abel, G.A.; Schmidt, U.; Holsboer, F.; Rein, T. FK506-binding proteins 51 and 52 differentially regulate dynein interaction and nuclear translocation of the glucocorticoid receptor in mammalian cells. J. Biol. Chem. 2005, 280, 4609–4616. [Google Scholar] [CrossRef]
- Quinta, H.R.; Galigniana, M.D. The neuroregenerative mechanism mediated by the hsp90-binding immunophilin FKBP52 resembles the early steps of neuronal differentiation. Br. J. Pharmacol. 2012, 166, 637–649. [Google Scholar] [CrossRef]
- Shim, S.; Yuan, J.P.; Kim, J.Y.; Zeng, W.; Huang, G.; Milshteyn, A.; Kern, D.; Muallem, S.; Ming, G.L.; Worley, P.F. Peptidyl-prolyl isomerase FKBP52 controls chemotropic guidance of neuronal growth cones via regulation of TRPC1 channel opening. Neuron 2009, 64, 471–483. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, E.T.; Salmon, E.D.; Erickson, H.P. How calcium causes microtubule depolymerization. Cell Motil. Cytoskelet. 1997, 36, 125–135. [Google Scholar] [CrossRef]
- Bandleon, S.; Strunz, P.P.; Pickel, S.; Tiapko, O.; Cellini, A.; Miranda-Laferte, E.; Eder-Negrin, P. FKBP52 regulates TRPC3-dependent Ca2+ signals and the hypertrophic growth of cardiomyocyte cultures. J. Cell Sci. 2019, 132. [Google Scholar] [CrossRef] [PubMed]
- Gkika, D.; Topala, C.N.; Hoenderop, J.G.; Bindels, R.J. The immunophilin FKBP52 inhibits the activity of the epithelial Ca2+ channel TRPV5. Am. J. Physiol. Ren. Physiol. 2006, 290, F1253–F1259. [Google Scholar] [CrossRef] [PubMed]
- Galigniana, M.D.; Radanyi, C.; Renoir, J.M.; Housley, P.R.; Pratt, W.B. Evidence that the peptidylprolyl isomerase domain of the Hsp90-binding immunophilin FKBP52 is involved in both dynein interaction and glucocorticoid receptor movement to the nucleus. J. Biol. Chem. 2001, 276, 14884–14889. [Google Scholar] [CrossRef] [PubMed]
- Gallo, L.I.; Ghini, A.A.; Piwien Pilipuk, G.; Galigniana, M.D. Differential recruitment of tetratricorpeptide repeat domain immunophilins to the mineralocorticoid receptor influences both heat-shock protein 90-dependent retrotransport and hormone-dependent transcriptional activity. Biochemistry 2007, 46, 14044–14057. [Google Scholar] [CrossRef]
- Garabedian, M.J.; Harris, C.A.; Jeanneteau, F. Glucocorticoid receptor action in metabolic and neuronal function. F1000Research 2017, 6, 1208. [Google Scholar] [CrossRef]
- Allan, R.K.; Ratajczak, T. Versatile tpr domains accommodate different modes of target protein recognition and function. Cell Stress Chaperones 2011, 16, 353–367. [Google Scholar] [CrossRef]
- Ratajczak, T.; Cluning, C.; Ward, B.K. Steroid receptor-associated immunophilins: A gateway to steroid signalling. The Clin. Biochem. Rev. 2015, 36, 31–52. [Google Scholar]
- Zgajnar, N.R.; De Leo, S.A.; Lotufo, C.M.; Erlejman, A.G.; Piwien-Pilipuk, G.; Galigniana, M.D. Biological actions of the Hsp90-binding immunophilins FKBP51 and FKBP52. Biomolecules 2019, 9, 52. [Google Scholar] [CrossRef] [PubMed]
- Tak, P.P.; Firestein, G.S. NF-kappaB: A key role in inflammatory diseases. J. Clin. Investig. 2001, 107, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-kappaB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [PubMed]
- Annett, S.; Moore, G.; Robson, T. FK506 binding proteins and inflammation related signalling pathways; basic biology, current status and future prospects for pharmacological intervention. Pharmacol. Ther. 2020, 215, 107623. [Google Scholar] [CrossRef]
- Mikenberg, I.; Widera, D.; Kaus, A.; Kaltschmidt, B.; Kaltschmidt, C. Transcription factor nf-kappab is transported to the nucleus via cytoplasmic dynein/dynactin motor complex in hippocampal neurons. PloS ONE 2007, 2, e589. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Li, Z.Z.; Jiang, D.S.; Lu, Y.Y.; Liu, Y.; Gao, L.; Zhang, S.M.; Lei, H.; Zhu, L.H.; Zhang, X.D.; et al. IRF4 is a novel mediator for neuronal survival in ischaemic stroke. Cell Death Differ. 2014, 21, 888–903. [Google Scholar] [CrossRef]
- Sanokawa-Akakura, R.; Dai, H.; Akakura, S.; Weinstein, D.; Fajardo, J.E.; Lang, S.E.; Wadsworth, S.; Siekierka, J.; Birge, R.B. A novel role for the immunophilin FKBP52 in copper transport. J. Biol. Chem. 2004, 279, 27845–27848. [Google Scholar] [CrossRef]
- Lim, C.M.; Cater, M.A.; Mercer, J.F.; La Fontaine, S. Copper-dependent interaction of dynactin subunit p62 with the n terminus of ATP7B but not ATP7A. J. Biol. Chem. 2006, 281, 14006–14014. [Google Scholar] [CrossRef]
- Chen, G.F.; Sudhahar, V.; Youn, S.W.; Das, A.; Cho, J.; Kamiya, T.; Urao, N.; McKinney, R.D.; Surenkhuu, B.; Hamakubo, T.; et al. Copper transport protein antioxidant-1 promotes inflammatory neovascularization via chaperone and transcription factor function. Sci. Rep. 2015, 5, 14780. [Google Scholar] [CrossRef]
- Giannakakou, P.; Sackett, D.L.; Ward, Y.; Webster, K.R.; Blagosklonny, M.V.; Fojo, T. P53 is associated with cellular microtubules and is transported to the nucleus by dynein. Nat. Cell Biol. 2000, 2, 709–717. [Google Scholar] [CrossRef]
- Galigniana, M.D.; Harrell, J.M.; O’Hagen, H.M.; Ljungman, M.; Pratt, W.B. Hsp90-binding immunophilins link p53 to dynein during p53 transport to the nucleus. J. Biol. Chem. 2004, 279, 22483–22489. [Google Scholar] [CrossRef] [PubMed]
- Harada, A.; Takei, Y.; Kanai, Y.; Tanaka, Y.; Nonaka, S.; Hirokawa, N. Golgi vesiculation and lysosome dispersion in cells lacking cytoplasmic dynein. J. Cell Biol. 1998, 141, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Weingarten, M.D.; Lockwood, A.H.; Hwo, S.Y.; Kirschner, M.W. A protein factor essential for microtubule assembly. Proc. Natl. Acad. Sci. USA 1975, 72, 1858–1862. [Google Scholar] [CrossRef]
- Kadavath, H.; Hofele, R.V.; Biernat, J.; Kumar, S.; Tepper, K.; Urlaub, H.; Mandelkow, E.; Zweckstetter, M. Tau stabilizes microtubules by binding at the interface between tubulin heterodimers. Proc. Natl. Acad. Sci. USA 2015, 112, 7501–7506. [Google Scholar] [CrossRef] [PubMed]
- Alim, M.A.; Ma, Q.L.; Takeda, K.; Aizawa, T.; Matsubara, M.; Nakamura, M.; Asada, A.; Saito, T.; Kaji, H.; Yoshii, M.; et al. Demonstration of a role for alpha-synuclein as a functional microtubule-associated protein. J. Alzheimer’s Dis. JAD 2004, 6, 435–442; discussion 443–449. [Google Scholar] [CrossRef] [PubMed]
- Cartelli, D.; Aliverti, A.; Barbiroli, A.; Santambrogio, C.; Ragg, E.M.; Casagrande, F.V.; Cantele, F.; Beltramone, S.; Marangon, J.; De Gregorio, C.; et al. Alpha-synuclein is a novel microtubule dynamase. Sci Rep 2016, 6, 33289. [Google Scholar] [CrossRef] [PubMed]
- Stamer, K.; Vogel, R.; Thies, E.; Mandelkow, E.; Mandelkow, E.M. Tau blocks traffic of organelles, neurofilaments, and app vesicles in neurons and enhances oxidative stress. J. Cell Biol. 2002, 156, 1051–1063. [Google Scholar] [CrossRef]
- Eisbach, S.E.; Outeiro, T.F. Alpha-synuclein and intracellular trafficking: Impact on the spreading of parkinson’s disease pathology. J. Mol. Med. 2013, 91, 693–703. [Google Scholar] [CrossRef]
- Carnwath, T.; Mohammed, R.; Tsiang, D. The direct and indirect effects of alpha-synuclein on microtubule stability in the pathogenesis of Parkinson’s disease. Neuropsychiatr. Dis. Treat. 2018, 14, 1685–1695. [Google Scholar] [CrossRef]
- Grundke-Iqbal, I.; Iqbal, K.; Tung, Y.C.; Quinlan, M.; Wisniewski, H.M.; Binder, L.I. Abnormal phosphorylation of the microtubule-associated protein tau (tau) in alzheimer cytoskeletal pathology. Proc. Natl. Acad. Sci. USA 1986, 83, 4913–4917. [Google Scholar] [CrossRef]
- Gotz, J.; Ittner, L.M.; Fandrich, M.; Schonrock, N. Is tau aggregation toxic or protective: A sensible question in the absence of sensitive methods? J. Alzheimer’s Dis. JAD 2008, 14, 423–429. [Google Scholar] [CrossRef]
- Kopito, R.R. Ron r. Kopito: Unfolding the secrets of protein aggregation. Trends Cell Biol. 2016, 26, 559–560. [Google Scholar] [CrossRef] [PubMed]
- Fassler, J.S.; Skuodas, S.; Weeks, D.L.; Phillips, B.T. Protein aggregation and disaggregation in cells and development. . J. Mol. Biol. 2021, 433, 167215. [Google Scholar] [CrossRef] [PubMed]
- Blair, L.J.; Nordhues, B.A.; Hill, S.E.; Scaglione, K.M.; O’Leary, J.C., 3rd; Fontaine, S.N.; Breydo, L.; Zhang, B.; Li, P.; Wang, L.; et al. Accelerated neurodegeneration through chaperone-mediated oligomerization of tau. J. Clin. Investig. 2013, 123, 4158–4169. [Google Scholar] [CrossRef] [PubMed]
- Weickert, S.; Wawrzyniuk, M.; John, L.H.; Rudiger, S.G.D.; Drescher, M. The mechanism of Hsp90-induced oligomerizaton of tau. Sci. Adv. 2020, 6, eaax6999. [Google Scholar] [CrossRef]
- Karagoz, G.E.; Duarte, A.M.; Akoury, E.; Ippel, H.; Biernat, J.; Moran Luengo, T.; Radli, M.; Didenko, T.; Nordhues, B.A.; Veprintsev, D.B.; et al. Hsp90-tau complex reveals molecular basis for specificity in chaperone action. Cell 2014, 156, 963–974. [Google Scholar] [CrossRef]
- Liberek, K.; Lewandowska, A.; Zietkiewicz, S. Chaperones in control of protein disaggregation. EMBO J. 2008, 27, 328–335. [Google Scholar] [CrossRef]
- Mok, S.A.; Condello, C.; Freilich, R.; Gillies, A.; Arhar, T.; Oroz, J.; Kadavath, H.; Julien, O.; Assimon, V.A.; Rauch, J.N.; et al. Mapping interactions with the chaperone network reveals factors that protect against tau aggregation. Nat. Struct. Mol. Biol. 2018, 25, 384–393. [Google Scholar] [CrossRef]
- Salminen, A.; Ojala, J.; Kaarniranta, K.; Hiltunen, M.; Soininen, H. Hsp90 regulates tau pathology through co-chaperone complexes in Alzheimer’s disease. Prog. Neurobiol. 2011, 93, 99–110. [Google Scholar] [CrossRef]
- Daturpalli, S.; Waudby, C.A.; Meehan, S.; Jackson, S.E. Hsp90 inhibits alpha-synuclein aggregation by interacting with soluble oligomers. J. Mol. Biol. 2013, 425, 4614–4628. [Google Scholar] [CrossRef]
- Jia, C.; Ma, X.; Liu, Z.; Gu, J.; Zhang, X.; Li, D.; Zhang, S. Different heat shock proteins bind alpha-synuclein with distinct mechanisms and synergistically prevent its amyloid aggregation. Front. Neurosci. 2019, 13, 1124. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Martinez-Vicente, M.; Kruger, U.; Kaushik, S.; Wong, E.; Mandelkow, E.M.; Cuervo, A.M.; Mandelkow, E. Tau fragmentation, aggregation and clearance: The dual role of lysosomal processing. Hum. Mol. Genet. 2009, 18, 4153–4170. [Google Scholar] [CrossRef] [PubMed]
- Mak, S.K.; McCormack, A.L.; Manning-Bog, A.B.; Cuervo, A.M.; Di Monte, D.A. Lysosomal degradation of alpha-synuclein in vivo. J. Biol. Chem. 2010, 285, 13621–13629. [Google Scholar] [CrossRef]
- Mackeh, R.; Perdiz, D.; Lorin, S.; Codogno, P.; Pous, C. Autophagy and microtubules - new story, old players. J. Cell Sci. 2013, 126, 1071–1080. [Google Scholar] [CrossRef] [PubMed]
- Matteoni, R.; Kreis, T.E. Translocation and clustering of endosomes and lysosomes depends on microtubules. J. Cell Biol. 1987, 105, 1253–1265. [Google Scholar] [CrossRef] [PubMed]
- Korolchuk, V.I.; Rubinsztein, D.C. Regulation of autophagy by lysosomal positioning. Autophagy 2011, 7, 927–928. [Google Scholar] [CrossRef]
- Sferra, A.; Nicita, F.; Bertini, E. Microtubule dysfunction: A common feature of neurodegenerative diseases. Int. J. Mol. Sci. 2020, 21, 7354. [Google Scholar] [CrossRef]
- Oliveira, L.M.A.; Gasser, T.; Edwards, R.; Zweckstetter, M.; Melki, R.; Stefanis, L.; Lashuel, H.A.; Sulzer, D.; Vekrellis, K.; Halliday, G.M.; et al. Alpha-synuclein research: Defining strategic moves in the battle against Parkinson’s disease. NPJ Parkinson’s Dis. 2021, 7, 65. [Google Scholar] [CrossRef]
- Lee, V.M.; Goedert, M.; Trojanowski, J.Q. Neurodegenerative tauopathies. Annu. Rev. Neurosci. 2001, 24, 1121–1159. [Google Scholar] [CrossRef]
- Goedert, M.; Spillantini, M.G. A century of Alzheimer’s disease. Science 2006, 314, 777–781. [Google Scholar] [CrossRef]
- Pellegrini, L.; Wetzel, A.; Granno, S.; Heaton, G.; Harvey, K. Back to the tubule: Microtubule dynamics in Parkinson’s disease. Cell. Mol. Life Sci. CMLS 2017, 74, 409–434. [Google Scholar] [CrossRef]
- Cartelli, D.; Cappelletti, G. Microtubule destabilization paves the way to Parkinson’s disease. Mol. Neurobiol. 2017, 54, 6762–6774. [Google Scholar] [CrossRef] [PubMed]
- Alonso, A.C.; Grundke-Iqbal, I.; Iqbal, K. Alzheimer’s disease hyperphosphorylated tau sequesters normal tau into tangles of filaments and disassembles microtubules. Nat. Med. 1996, 2, 783–787. [Google Scholar] [CrossRef] [PubMed]
- Alonso, A.D.; Grundke-Iqbal, I.; Barra, H.S.; Iqbal, K. Abnormal phosphorylation of tau and the mechanism of Alzheimer neurofibrillary degeneration: Sequestration of microtubule-associated proteins 1 and 2 and the disassembly of microtubules by the abnormal tau. Proc. Natl. Acad. Sci. USA 1997, 94, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Brandt, R.; Bakota, L. Microtubule dynamics and the neurodegenerative triad of Alzheimer’s disease: The hidden connection. J. Neurochem. 2017, 143, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.A.; Higgins, G.A. Alzheimer’s disease: The amyloid cascade hypothesis. Science 1992, 256, 184–185. [Google Scholar] [CrossRef] [PubMed]
- Duyckaerts, C.; Delatour, B.; Potier, M.C. Classification and basic pathology of Alzheimer disease. Acta Neuropathol. 2009, 118, 5–36. [Google Scholar] [CrossRef]
- King, M.E.; Kan, H.M.; Baas, P.W.; Erisir, A.; Glabe, C.G.; Bloom, G.S. Tau-dependent microtubule disassembly initiated by prefibrillar beta-amyloid. J. Cell Biol. 2006, 175, 541–546. [Google Scholar] [CrossRef]
- Giustiniani, J.; Sineus, M.; Sardin, E.; Dounane, O.; Panchal, M.; Sazdovitch, V.; Duyckaerts, C.; Chambraud, B.; Baulieu, E.E. Decrease of the immunophilin FKBP52 accumulation in human brains of Alzheimer’s disease and FTDP-17. J. Alzheimer’s Dis. JAD 2012, 29, 471–483. [Google Scholar] [CrossRef]
- Herrmann, L.; Ebert, T.; Rosen, H.; Novak, B.; Philipsen, A.; Touma, C.; Schreckenbach, M.; Gassen, N.C.; Rein, T.; Schmidt, U. Analysis of the cerebellar molecular stress response led to first evidence of a role for fkbp51 in brain fkbp52 expression in mice and humans. Neurobiol. Stress 2021, 15, 100401. [Google Scholar] [CrossRef]
- Goedert, M.; Crowther, R.A.; Spillantini, M.G. Tau mutations cause frontotemporal dementias. Neuron 1998, 21, 955–958. [Google Scholar] [CrossRef]
- Hasegawa, M.; Smith, M.J.; Goedert, M. Tau proteins with ftdp-17 mutations have a reduced ability to promote microtubule assembly. FEBS Lett. 1998, 437, 207–210. [Google Scholar] [CrossRef]
- Paquet, D.; Bhat, R.; Sydow, A.; Mandelkow, E.M.; Berg, S.; Hellberg, S.; Falting, J.; Distel, M.; Koster, R.W.; Schmid, B.; et al. A zebrafish model of tauopathy allows in vivo imaging of neuronal cell death and drug evaluation. J. Clin. Investig. 2009, 119, 1382–1395. [Google Scholar] [CrossRef] [PubMed]
- Frost, B.; Jacks, R.L.; Diamond, M.I. Propagation of tau misfolding from the outside to the inside of a cell. . J. Biol. Chem. 2009, 284, 12845–12852. [Google Scholar] [CrossRef]
- Clavaguera, F.; Bolmont, T.; Crowther, R.A.; Abramowski, D.; Frank, S.; Probst, A.; Fraser, G.; Stalder, A.K.; Beibel, M.; Staufenbiel, M.; et al. Transmission and spreading of tauopathy in transgenic mouse brain. Nat. Cell Biol. 2009, 11, 909–913. [Google Scholar] [CrossRef]
- Guo, J.L.; Lee, V.M. Seeding of normal tau by pathological Tau conformers drives pathogenesis of Alzheimer-like tangles. J. Biol. Chem. 2011, 286, 15317–15331. [Google Scholar] [CrossRef]
- Criado-Marrero, M.; Gebru, N.T.; Blazier, D.M.; Gould, L.A.; Baker, J.D.; Beaulieu-Abdelahad, D.; Blair, L.J. Hsp90 co-chaperones, FKBP52 and Aha1, promote tau pathogenesis in aged wild-type mice. Acta Neuropathol. Commun. 2021, 9, 65. [Google Scholar] [CrossRef]
- Criado-Marrero, M.; Gebru, N.T.; Gould, L.A.; Blazier, D.M.; Vidal-Aguiar, Y.; Smith, T.M.; Abdelmaboud, S.S.; Shelton, L.B.; Wang, X.; Dahrendorff, J.; et al. FKBP52 overexpression accelerates hippocampal-dependent memory impairments in a tau transgenic mouse model. NPJ Aging Mech. Dis. 2021, 7, 9. [Google Scholar] [CrossRef]
- Sanokawa-Akakura, R.; Cao, W.; Allan, K.; Patel, K.; Ganesh, A.; Heiman, G.; Burke, R.; Kemp, F.W.; Bogden, J.D.; Camakaris, J.; et al. Control of Alzheimer’s amyloid beta toxicity by the high molecular weight immunophilin FKBP52 and copper homeostasis in Drosophila. PLoS ONE 2010, 5, e8626. [Google Scholar] [CrossRef]
- Green, K.N.; Billings, L.M.; Roozendaal, B.; McGaugh, J.L.; LaFerla, F.M. Glucocorticoids increase amyloid-beta and tau pathology in a mouse model of Alzheimer’s disease. J. Neurosci. Off. J. Soc. Neurosci. 2006, 26, 9047–9056. [Google Scholar] [CrossRef]
- Park, H.J.; Lee, S.; Jung, J.W.; Kim, B.C.; Ryu, J.H.; Kim, D.H. Glucocorticoid- and long-term stress-induced aberrant synaptic plasticity are mediated by activation of the glucocorticoid receptor. Arch. Pharmacal Res. 2015, 38, 1204–1212. [Google Scholar] [CrossRef] [PubMed]
- Lopes, S.; Vaz-Silva, J.; Pinto, V.; Dalla, C.; Kokras, N.; Bedenk, B.; Mack, N.; Czisch, M.; Almeida, O.F.; Sousa, N.; et al. Tau protein is essential for stress-induced brain pathology. Proc. Natl. Acad. Sci. USA 2016, 113, E3755–E3763. [Google Scholar] [CrossRef] [PubMed]
- Baschant, U.; Tuckermann, J. The role of the glucocorticoid receptor in inflammation and immunity. J. Steroid Biochem. Mol. Biol. 2010, 120, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, H.; Barger, S.; Barnum, S.; Bradt, B.; Bauer, J.; Cole, G.M.; Cooper, N.R.; Eikelenboom, P.; Emmerling, M.; Fiebich, B.L.; et al. Inflammation and Alzheimer’s disease. Neurobiol. Aging. 2000, 21, 383–421. [Google Scholar] [CrossRef]
- Aktas, O.; Ullrich, O.; Infante-Duarte, C.; Nitsch, R.; Zipp, F. Neuronal damage in brain inflammation. Arch. Neurol. 2007, 64, 185–189. [Google Scholar] [CrossRef]
- Kinney, J.W.; Bemiller, S.M.; Murtishaw, A.S.; Leisgang, A.M.; Salazar, A.M.; Lamb, B.T. Inflammation as a central mechanism in Alzheimer’s disease. Alzheimer’s Dement. 2018, 4, 575–590. [Google Scholar] [CrossRef]
- Roos, P.M.; Vesterberg, O.; Nordberg, M. Metals in motor neuron diseases. Exp. Biol. Med. 2006, 231, 1481–1487. [Google Scholar] [CrossRef]
- Desai, V.; Kaler, S.G. Role of copper in human neurological disorders. Am. J. Clin. Nutr. 2008, 88, 855S–858S. [Google Scholar] [CrossRef]
| Gene | FKBPs | FK506 Binding/PPIase Activity | Cellular Location | Cellular Function | CNS Expression | CNS Disease Association |
|---|---|---|---|---|---|---|
| FKBP1a | FKBP12 | Yes/Yes | Cytoplasm | - Regulates Tau aggregation [33] - Regulates APP processing [34] - Increases α-Syn aggregation [13,51,52] - Regulates ryanodine receptors (RyRs) [35] | -Detected in all brain regions and spinal cord -Expressed in Neurons and dystrophic neurites [53] | - Tauopathies (AD) - α-Synucleinopathies (PD) |
| FKBP1b | FKBP12.6 | Yes/Yes | Cytoplasm | - Regulates ryanodine receptors (RyRs) [35] | Detected in all brain regions and spinal cord | AD [35] |
| FKBP2 | FKBP13 | Yes/Yes | Endoplasmic reticulum | -ER Chaperone [20] | Detected in all brain regions and spinal cord | Unknown |
| FKBP3 | FKBP25 | Yes/Yes | Nucleus and cytoplasm | - Regulates MT dynamics and is involved in Nucleus-Cytoplasm shuttling [32] - Regulates p53 signaling [36] | - Detected in all brain regions and spinal cord -Expressed in neurons | Unknown |
| FKBP4 | FKBP52 | Yes/Yes | Cytoplasm, nucleus, MT network, endo-lysosomal system | - Regulates Tau aggregation [54,55,56] - Increases α-Syn aggregation [13,52] - Regulates MT dynamics [12,31] - Regulates MT-dependent trafficking (i.e., SHR, Nf-kappaB, IRF4, hTERT) [57,58,59,60] - Involved in ALP function [61] | - Detected in all brain regions and spinal cord - Highly expressed in neurons | - Tauopathies (AD) - α-Synucleinopathies (PD) |
| FKBP5 | FKBP51 | Yes/Yes | Cytoplasm, nucleus, MT network, mitochondria | - Regulates Tau aggregation [62] - Regulates MT dynamics [63] - Regulates MT-dependent trafficking (i.e., SHR, Nf-kappaB, hTERT) [57,59,64] - Involved in ALP function [65] | - Detected in all brain regions and spinal cord - Highly expressed in neurons | - Tauopathies (AD) - α-Synucleinopathies (PD) - Huntington disease [37] - Stress-related and psychiatric diseases [8] |
| FKBP6 | FKBP36 | No/Yes | Cytoplasm and nucleus | - Regulates GAPDH signalling [46] - Involved in spermatogenesis [38] | Undefined | - Williams-Beuren syndrome [39] |
| FKBP7 | FKBP23 | Undefined/Yes | Endoplasmic reticulum | - Calcium binding ability [23] - Regulates interaction with BiP [40] | - Detected in all brain regions and spinal cord | Unknown |
| FKBP8 | FKBP38 | No/Yes | Mitochondria | - Involved in mitophagy [66] - Inhibits apoptosis [47] - Endogenous inhibitor of mTOR [41] | - Detected in all brain regions and spinal cord -Expressed in neurons | Unknown |
| FKBP9 | FKBP60 | Yes/Yes | Endoplasmic reticulum | - Possible role in prion propagation or clearance [42] | Detected in all brain regions and spinal cord | Unknown |
| FKBP10 | FKBP65 | Yes/Yes | Endoplasmic reticulum | - Possible neuroprotective effect linked to regulation of protein folding [43] - Participates in type I procollagen folding [44] | Detected in all brain regions and spinal cord | Unknown |
| FKBP11 | FKBP19 | Yes/Yes | Cytoplasm, Endoplasmic reticulum | -Protein folding and secretion [21] | Detected in all brain regions and spinal cord | Unknown |
| FKBP14 | FKBP22 | Yes/Yes | Cytoplasm, Endoplasmic reticulum | - Involved in protein folding, trafficking and in collagen synthesis [22] - Regulates Presenilin and Notch signalling [45] | Detected in all brain regions and spinal cord | Unknown |
| FKBP15 | FKBP133 | No/No | Cytoplasm, nucleus and endosome | - Possible role in cytoskeletal organization of neuronal growth cones [67] - Involved in early endosomes transport [67] | Detected in all brain regions and spinal cord | Unknown |
| AIP | FKBP37 | No/No | Cytoplasm | - Interaction with Hsp90 [49] -Regulates aromatic hydrocarbon receptor signalling [49] | Detected in all brain regions and spinal cord | - Pituitary adenoma |
| FKBPL | FKBP-L | No/No | Cytoplasm | - Regulates MT-dependent trafficking of GR [50] | Detected in all brain regions and spinal cord | Unknown |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chambraud, B.; Byrne, C.; Meduri, G.; Baulieu, E.E.; Giustiniani, J. FKBP52 in Neuronal Signaling and Neurodegenerative Diseases: A Microtubule Story. Int. J. Mol. Sci. 2022, 23, 1738. https://doi.org/10.3390/ijms23031738
Chambraud B, Byrne C, Meduri G, Baulieu EE, Giustiniani J. FKBP52 in Neuronal Signaling and Neurodegenerative Diseases: A Microtubule Story. International Journal of Molecular Sciences. 2022; 23(3):1738. https://doi.org/10.3390/ijms23031738
Chicago/Turabian StyleChambraud, Béatrice, Cillian Byrne, Geri Meduri, Etienne Emile Baulieu, and Julien Giustiniani. 2022. "FKBP52 in Neuronal Signaling and Neurodegenerative Diseases: A Microtubule Story" International Journal of Molecular Sciences 23, no. 3: 1738. https://doi.org/10.3390/ijms23031738
APA StyleChambraud, B., Byrne, C., Meduri, G., Baulieu, E. E., & Giustiniani, J. (2022). FKBP52 in Neuronal Signaling and Neurodegenerative Diseases: A Microtubule Story. International Journal of Molecular Sciences, 23(3), 1738. https://doi.org/10.3390/ijms23031738






