The Upregulation of Regenerative Activity for Extracellular Vesicles with Melatonin Modulation in Chemically Defined Media
Abstract
1. Introduction
2. Results and Discussion
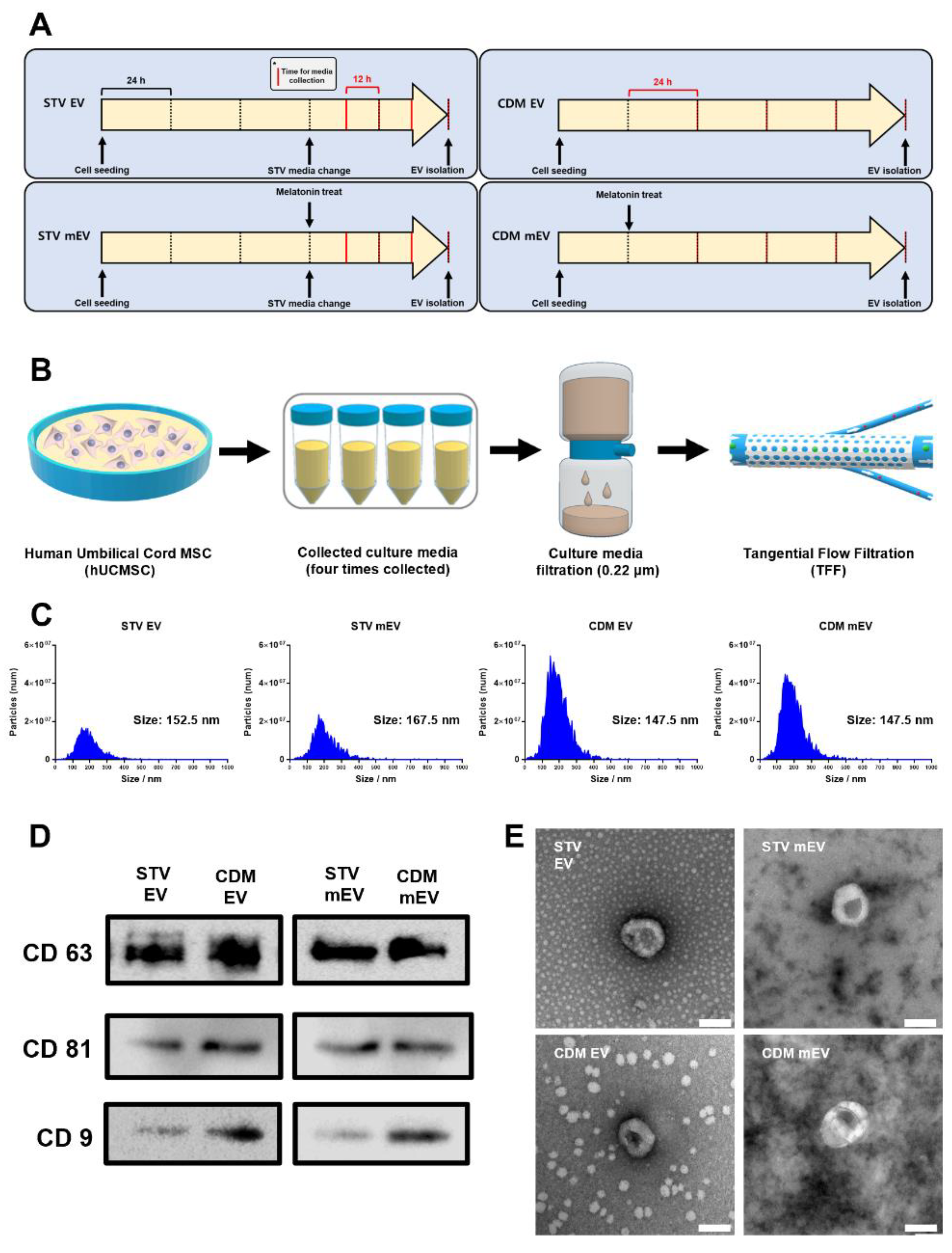
2.1. Characterization of hUCMSC-Derived EVs Depending on Culture Conditions
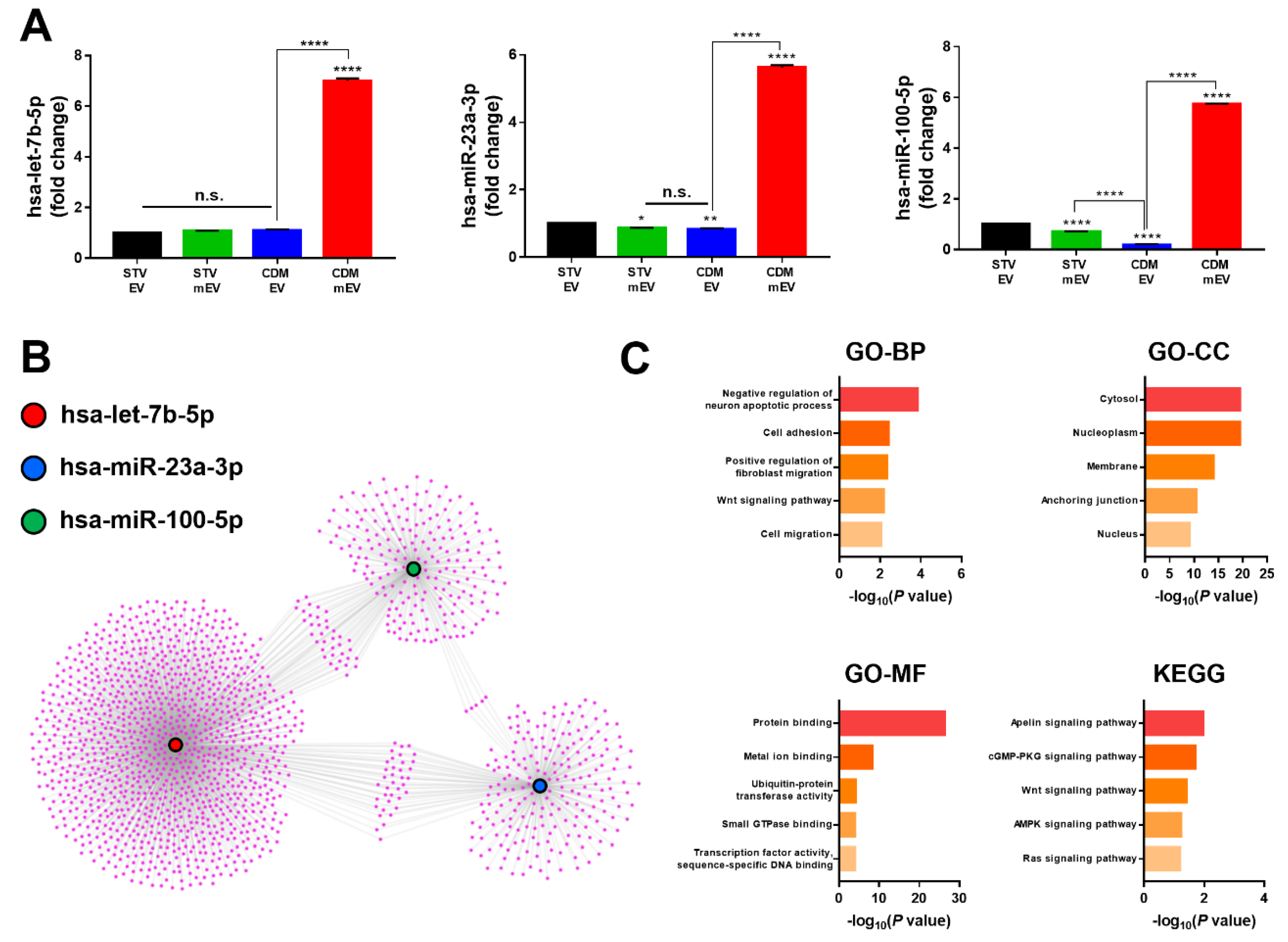
2.2. The Expectations for the Functionality of EVs through Comparative Analysis on Internal miRNA of EVs
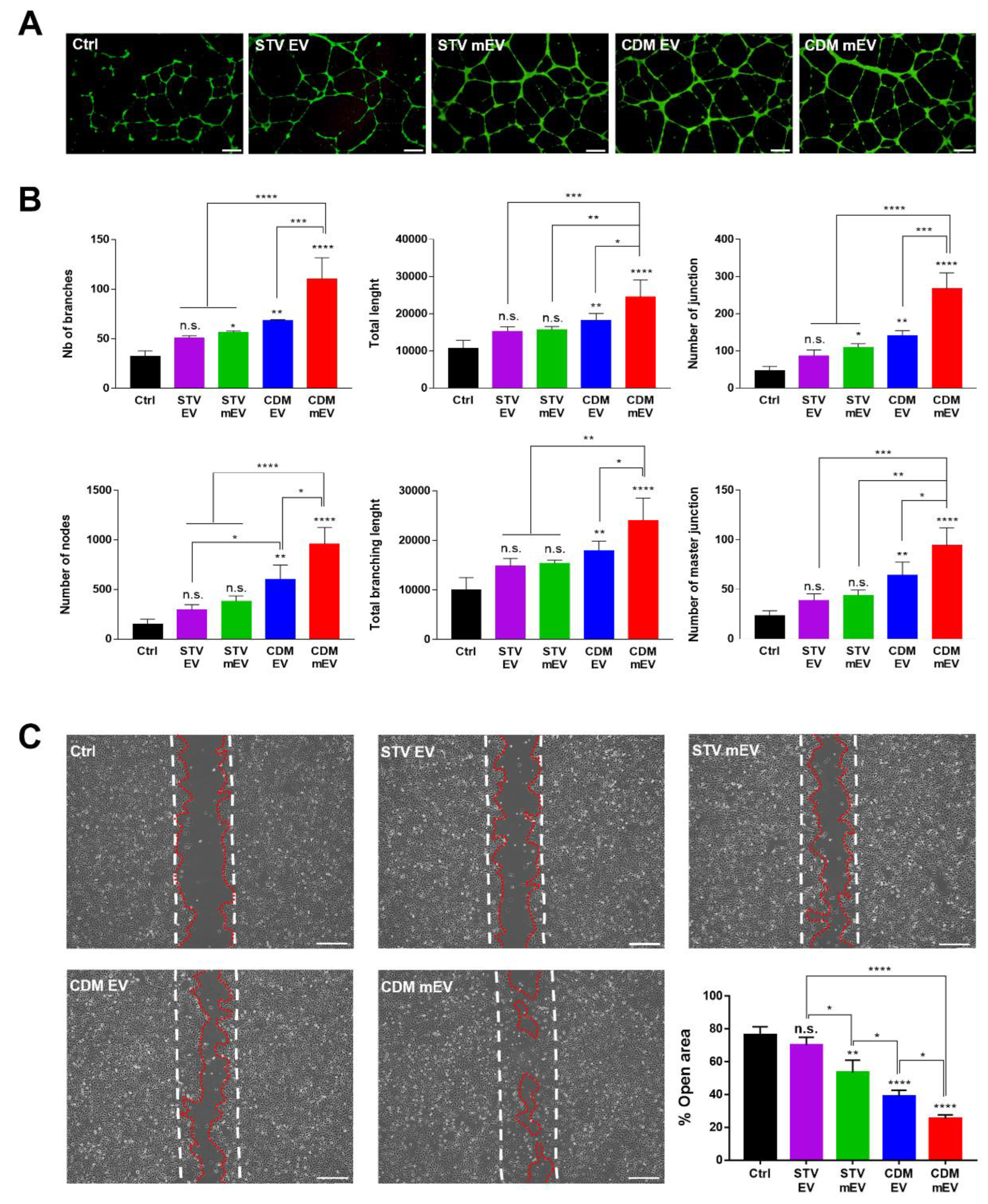
2.3. Angiogenesis and Wound-Healing Effects of EVs
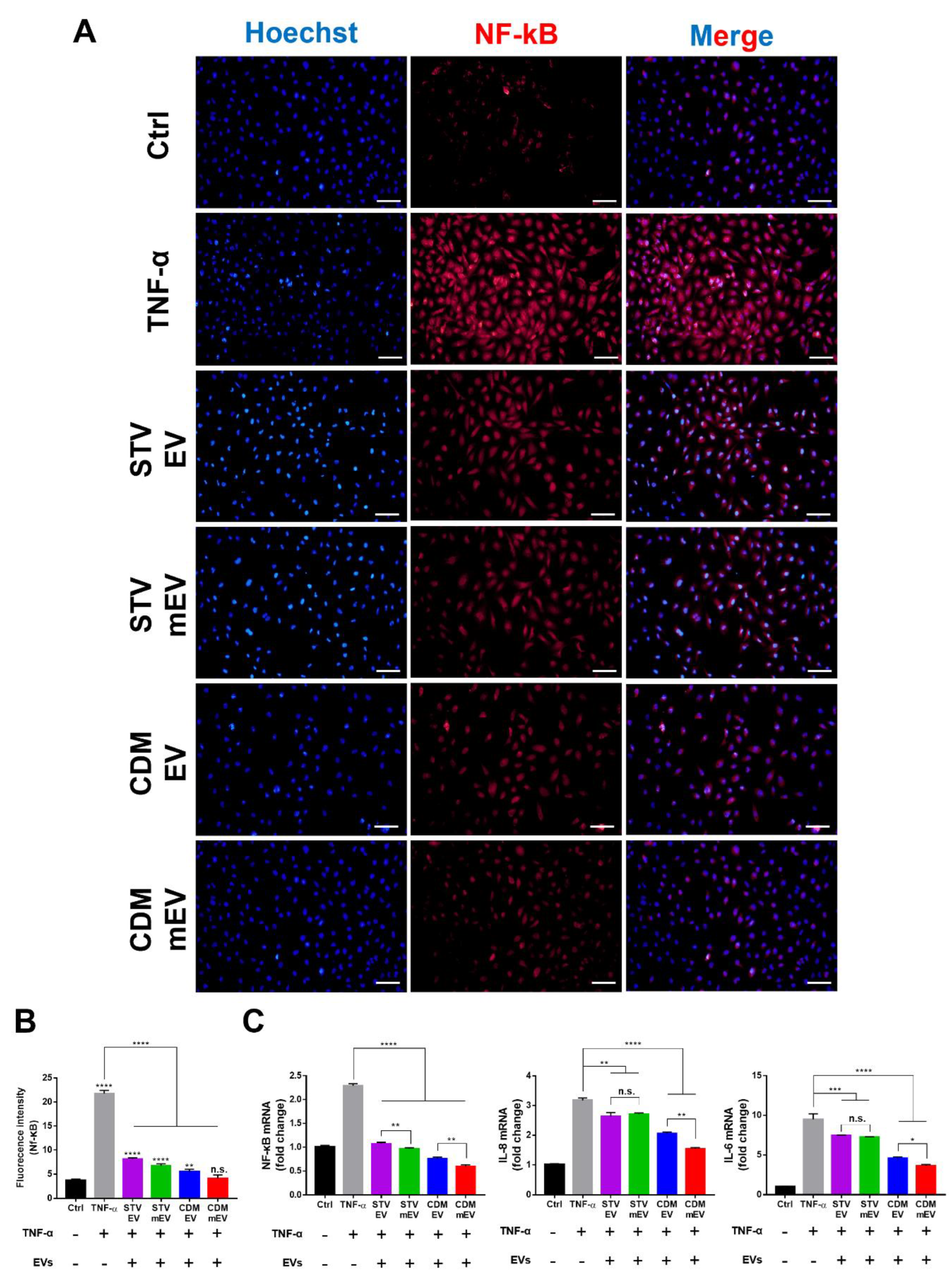
2.4. Anti-Inflammation Effects of EVs
2.5. Anti-Oxidative and Anti-Apoptosis Effects of EVs
3. Materials and Methods
3.1. Cell Culture
3.2. Isolation and Characterization of Extracellular Vesicles (EVs)
3.3. Western Blot Analysis
3.4. Quantification of the miRNA in EVs
3.5. Bioinformatics Analysis of the Internal miRNA in EVs
3.6. Tube Formation Assay
3.7. Cell Migration Assay
3.8. Immunocytochemistry (ICC)
3.9. RT-qPCR for Pro-Inflammatory Factors
3.10. Cellular ROS Assay with 2′,7′-Dichlorofluorescin Diacetate (DCF-DA)
3.11. Apoptosis Assay
3.12. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mazini, L.; Rochette, L.; Malka, G. Exosomes Contribution in COVID-19 Patients’ Treatment. J. Transl. Med. 2021, 19, 234. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Yeo, R.W.Y.; Lai, R.C.; Sim, E.W.K.; Chin, K.C.; Lim, S.K. Mesenchymal Stromal Cell Exosome–Enhanced Regulatory T-Cell Production through an Antigen-Presenting Cell–Mediated Pathway. Cytotherapy 2018, 20, 687–696. [Google Scholar] [CrossRef]
- Munoz-Perez, E.; Gonzalez-Pujana, A.; Igartua, M.; Santos-Vizcaino, E.; Hernandez, R.M. Mesenchymal Stromal Cell Secretome for the Treatment of Immune-Mediated Inflammatory Diseases: Latest Trends in Isolation, Content Optimization and Delivery Avenues. Pharmaceutics 2021, 13, 1802. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, V.; Sengupta, S.; Lazo, A.; Woods, P.; Nolan, A.; Bremer, N. Exosomes Derived from Bone Marrow Mesenchymal Stem Cells as Treatment for Severe COVID-19. Stem Cells Dev. 2020, 29, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Fujii, S.; Miura, Y.; Fujishiro, A.; Shindo, T.; Shimazu, Y.; Hirai, H.; Tahara, H.; Takaori-Kondo, A.; Ichinohe, T.; Maekawa, T. Graft-Versus-Host Disease Amelioration by Human Bone Marrow Mesenchymal Stromal/Stem Cell-Derived Extracellular Vesicles Is Associated with Peripheral Preservation of Naive T Cell Populations. Stem Cells 2018, 36, 434–445. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yu, D.; Liu, Z.; Zhou, F.; Dai, J.; Wu, B.; Zhou, J.; Heng, B.C.; Zou, X.H.; Ouyang, H.; et al. Exosomes from Embryonic Mesenchymal Stem Cells Alleviate Osteoarthritis through Balancing Synthesis and Degradation of Cartilage Extracellular Matrix. Stem Cell Res. Ther. 2017, 8, 189. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhu, Q.; Cheng, L.; Wang, Y.; Li, M.; Yang, Q.; Hu, L.; Lou, D.; Li, J.; Dong, X.; et al. Exosome Detection via the Ultrafast-Isolation System: EXODUS. Nat. Methods 2021, 18, 212–218. [Google Scholar] [CrossRef]
- Kim, J.Y.; Rhim, W.K.; Yoo, Y.I.; Kim, D.S.; Ko, K.W.; Heo, Y.; Park, C.G.; Han, D.K. Defined MSC Exosome with High Yield and Purity to Improve Regenerative Activity. J. Tissue Eng. 2021, 12, 20417314211008626. [Google Scholar] [CrossRef]
- Kim, J.Y.; Rhim, W.K.; Seo, H.J.; Lee, Y.J.; Park, C.G.; Han, D.K. Comparative Analysis of MSC-Derived Exosomes Depending on Cell Culture Media for Regenerative Bioactivity. Tissue Eng. Regen. Med. 2021, 18, 355–367. [Google Scholar] [CrossRef]
- Haraszti, R.A.; Miller, R.; Stoppato, M.; Sere, Y.Y.; Coles, A.; Didiot, M.C.; Wollacott, R.; Sapp, E.; Dubuke, M.L.; Li, X.; et al. Exosomes Produced from 3D Cultures of MSCs by Tangential Flow Filtration Show Higher Yield and Improved Activity. Mol. Ther. 2018, 26, 2838–2847. [Google Scholar] [CrossRef]
- Gonzalez-King, H.; García, N.A.; Ontoria-Oviedo, I.; Ciria, M.; Montero, J.A.; Sepúlveda, P. Hypoxia Inducible Factor-1α Potentiates Jagged 1-Mediated Angiogenesis by Mesenchymal Stem Cell-Derived Exosomes. Stem Cells 2017, 35, 1747–1759. [Google Scholar] [CrossRef] [PubMed]
- Sung, D.K.; Sung, S.I.; Ahn, S.Y.; Chang, Y.S.; Park, W.S. Thrombin Preconditioning Boosts Biogenesis of Extracellular Vesicles from Mesenchymal Stem Cells and Enriches Their Cargo Contents via Protease-Activated Receptor-Mediated Signaling Pathways. Int. J. Mol. Sci. 2019, 20, 2899. [Google Scholar] [CrossRef] [PubMed]
- Noronha, N.D.C.; Mizukami, A.; Caliári-Oliveira, C.; Cominal, J.G.; Rocha, J.L.M.; Covas, D.T.; Swiech, K.; Malmegrim, K.C.R. Priming Approaches to Improve the Efficacy of Mesenchymal Stromal Cell-Based Therapies. Stem Cell Res. Ther. 2019, 10, 131. [Google Scholar] [CrossRef] [PubMed]
- Tocharus, C.; Puriboriboon, Y.; Junmanee, T.; Tocharus, J.; Ekthuwapranee, K.; Govitrapong, P. Melatonin Enhances Adult Rat Hippocampal Progenitor Cell Proliferation via ERK Signaling Pathway through Melatonin Receptor. Neuroscience 2014, 275, 314. [Google Scholar] [CrossRef]
- Chen, K.; Tong, C.; Yang, J.; Cong, P.; Liu, Y.; Shi, X.; Liu, X.; Zhang, J.; Zou, R.; Xiao, K.; et al. Injectable Melatonin-Loaded Carboxymethyl Chitosan (CMCS)-Based Hydrogel Accelerates Wound Healing by Reducing Inflammation and Promoting Angiogenesis and Collagen Deposition. J. Mater. Sci. Technol. 2021, 63, 236–245. [Google Scholar] [CrossRef]
- Gómez-Florit, M.; Ramis, J.M.; Monjo, M. Anti-Fibrotic and Anti-Inflammatory Properties of Melatonin on Human Gingival Fibroblasts in Vitro. Biochem. Pharmacol. 2013, 86, 1784–1790. [Google Scholar] [CrossRef]
- Yea, J.H.; Yoon, Y.M.; Lee, J.H.; Yun, C.W.; Lee, S.H. Exosomes Isolated from Melatonin-Stimulated Mesenchymal Stem Cells Improve Kidney Function by Regulating Inflammation and Fibrosis in a Chronic Kidney Disease Mouse Model. J. Tissue Eng. 2021, 12, 20417314211059624. [Google Scholar] [CrossRef]
- Han, Y.S.; Yoon, Y.M.; Go, G.; Lee, J.H.; Lee, S.H. Melatonin Protects Human Renal Proximal Tubule Epithelial Cells against High Glucose-Mediated Fibrosis via the Cellular Prion Protein-Tgf-β-Smad Signaling Axis. Int. J. Med. Sci. 2020, 17, 1235–1245. [Google Scholar] [CrossRef]
- Marino, A.; Battaglini, M.; Desii, A.; Lavarello, C.; Genchi, G.; Petretto, A.; Ciofani, G. Liposomes Loaded with Polyphenol-Rich Grape Pomace Extracts Protect from Neurodegeneration in a Rotenone-Based in Vitro Model of Parkinson’s Disease. Biomater. Sci. 2021, 9, 8171–8188. [Google Scholar] [CrossRef]
- Takhtfooladi, H.; Takhtfooladi, M.; Moayer, F.; Mobarakeh, S. Melatonin Attenuates Lung Injury in a Hind Limb Ischemia-Reperfusion Rat Model. Rev. Port. Pneumol. 2015, 21, 30–35. [Google Scholar] [CrossRef]
- Zhou, L.; Zhao, D.; An, H.; Zhang, H.; Jiang, C.; Yang, B. Melatonin Prevents Lung Injury Induced by Hepatic Ischemia-Reperfusion through Anti-Inflammatory and Anti-Apoptosis Effects. Int. Immunopharmacol. 2015, 29, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Nickkholgh, A.; Yi, X.; Bruns, H.; Gross, M.L.; Hoffmann, K.; Mohr, E.; Zorn, M.; Büchler, M.W.; Schemmer, P. Melatonin Protects Kidney Grafts from Ischemia/Reperfusion Injury through Inhibition of NF-KB and Apoptosis after Experimental Kidney Transplantation. J. Pineal Res. 2009, 46, 365–372. [Google Scholar] [CrossRef]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal Information for Studies of Extracellular Vesicles 2018 (MISEV2018): A Position Statement of the International Society for Extracellular Vesicles and Update of the MISEV2014 Guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef]
- Rikkert, L.G.; Nieuwland, R.; Terstappen, L.W.M.M.; Coumans, F.A.W. Quality of Extracellular Vesicle Images by Transmission Electron Microscopy Is Operator and Protocol Dependent. J. Extracell. Vesicles 2019, 8, 1555419. [Google Scholar] [CrossRef]
- Park, S.-Y.; Kim, D.-S.; Kim, H.-M.; Lee, J.-K.; Hwang, D.-Y.; Kim, T.-H.; You, S.; Han, D.K. Human Mesenchymal Stem Cell-Derived Extracellular Vesicles Promote Neural Differentiation of Neural Progenitor Cells. Int. J. Mol. Sci. 2022, 23, 7047. [Google Scholar] [CrossRef] [PubMed]
- Aday, S.; Hazan-Halevy, I.; Chamorro-Jorganes, A.; Anwar, M.; Goldsmith, M.; Beazley-Long, N.; Sahoo, S.; Dogra, N.; Sweaad, W.; Catapano, F.; et al. Bioinspired Artificial Exosomes Based on Lipid Nanoparticles Carrying Let-7b-5p Promote Angiogenesis in Vitro and in Vivo. Mol. Ther. 2021, 29, 2239–2252. [Google Scholar] [CrossRef]
- Hu, H.; Dong, L.; Bu, Z.; Shen, Y.; Luo, J.; Zhang, H.; Zhao, S.; Lv, F.; Liu, Z. MiR-23a-3p-Abundant Small Extracellular Vesicles Released from Gelma/Nanoclay Hydrogel for Cartilage Regeneration. J. Extracell. Vesicles 2020, 9, 1778883. [Google Scholar] [CrossRef]
- Liu, Z.; Yang, Y.; Ju, J.; Zhang, G.; Zhang, P.; Ji, P.; Jin, Q.; Cao, G.; Zuo, R.; Wang, H.; et al. MiR-100-5p Promotes Epidermal Stem Cell Proliferation through Targeting MTMR3 to Activate PIP3/AKT and ERK Signaling Pathways. Stem Cells Int. 2022, 2022, 1474273. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Li, Q.; Tong, K.; Zhu, J.; Wang, H.; Chen, B.; Chen, L. BMSC-Derived Exosomes Promote Tendon-Bone Healing after Anterior Cruciate Ligament Reconstruction by Regulating M1/M2 Macrophage Polarization in Rats. Stem Cell Res. Ther. 2022, 13, 295. [Google Scholar] [CrossRef]
- Wu, J.; Kuang, L.; Chen, C.; Yang, J.; Zeng, W.N.; Li, T.; Chen, H.; Huang, S.; Fu, Z.; Li, J.; et al. MiR-100-5p-Abundant Exosomes Derived from Infrapatellar Fat Pad MSCs Protect Articular Cartilage and Ameliorate Gait Abnormalities via Inhibition of MTOR in Osteoarthritis. Biomaterials 2019, 206, 87–100. [Google Scholar] [CrossRef]
- Moparthi, L.; Koch, S. Wnt Signaling in Intestinal Inflammation. Differentiation 2019, 108, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Reis, M.; Liebner, S. Wnt Signaling in the Vasculature. Exp. Cell Res. 2013, 319, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Helker, C.S.M.; Eberlein, J.; Wilhelm, K.; Sugino, T.; Malchow, J.; Schuermann, A.; Baumeister, S.; Kwon, H.B.; Maischein, H.M.; Potente, M.; et al. Apelin Signaling Drives Vascular Endothelial Cells towards a Pro-Angiogenic State. eLife 2020, 9, e55589. [Google Scholar] [CrossRef]
- Schuman, E.M.; Madison, D.V. Nitric Oxide and Angiogenesis. Am. J. Physiol. 1994, 272, 31–35. [Google Scholar]
- Nagata, D.; Mogi, M.; Walsh, K. AMP-Activated Protein Kinase (AMPK) Signaling in Endothelial Cells Is Essential for Angiogenesis in Response to Hypoxic Stress. J. Biol. Chem. 2003, 278, 31000–31006. [Google Scholar] [CrossRef] [PubMed]
- Meadows, K.N.; Bryant, P.; Pumiglia, K. Vascular Endothelial Growth Factor Induction of the Angiogenic Phenotype Requires Ras Activation. J. Biol. Chem. 2001, 276, 49289–49298. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; He, L.; Chen, L. Apelin/APJ System: A Promising Therapy Target for Hypertension. Mol. Biol. Rep. 2014, 41, 6691–6703. [Google Scholar] [CrossRef]
- Zeng, X.J.; Zhang, L.K.; Wang, H.X.; Lu, L.Q.; Ma, L.Q.; Tang, C.S. Apelin Protects Heart against Ischemia/Reperfusion Injury in Rat. Peptides 2009, 30, 1144–1152. [Google Scholar] [CrossRef]
- Clevers, H.; Loh, K.M.; Nusse, R. An Integral Program for Tissue Renewal and Regeneration: Wnt Signaling and Stem Cell Control. Science 2014, 346, 1248012. [Google Scholar] [CrossRef]
- Zhou, F.; Wang, M.; Ju, J.; Wang, Y.; Liu, Z.; Zhao, X.; Yan, Y.; Yan, S.; Luo, X.; Fang, Y. Schizandrin a Protects against Cerebral Ischemia-Reperfusion Injury by Suppressing Inflammation and Oxidative Stress and Regulating the AMPK/Nrf2 Pathway Regulation. Am. J. Transl. Res. 2019, 11, 199–209. [Google Scholar] [PubMed]
- Xie, L.; Overbeek, P.A.; Reneker, L.W. Ras Signaling Is Essential for Lens Cell Proliferation and Lens Growth during Development. Dev. Biol. 2006, 298, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Molina, J.R.; Adjei, A.A. The Ras/Raf/MAPK Pathway. J. Thorac. Oncol. 2006, 1, 7–9. [Google Scholar] [CrossRef] [PubMed]
- Nour, S.; Imani, R.; Chaudhry, G.R.; Sharifi, A.M. Skin Wound Healing Assisted by Angiogenic Targeted Tissue Engineering: A Comprehensive Review of Bioengineered Approaches. J. Biomed. Mater. Res.—Part A 2021, 109, 453–478. [Google Scholar] [CrossRef] [PubMed]
- DiPietro, L.A. Angiogenesis and Wound Repair: When Enough Is Enough. J. Leukoc. Biol. 2016, 100, 979–984. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.Y.; Park, H.H.; Park, W.; Kim, H.; Jang, J.G.; Hong, K.S.; Lee, J.Y.; Seo, H.S.; Na, D.H.; Kim, T.H.; et al. Long-Acting Nanoparticulate DNase-1 for Effective Suppression of SARS-CoV-2-Mediated Neutrophil Activities and Cytokine Storm. Biomaterials 2021, 267, 120389. [Google Scholar] [CrossRef]
- Ko, K.-W.; Park, S.-Y.; Lee, E.H.; Yoo, Y.-I.; Kim, D.-S.; Kim, J.Y.; Kwon, T.G.; Han, D.K. Integrated Bioactive Scaffold with Polydeoxyribonucleotide and Stem-Cell-Derived Extracellular Vesicles for Kidney Regeneration. ACS Nano 2021, 15, 7575–7585. [Google Scholar] [CrossRef]
- Yang, G.H.; Lee, Y.B.; Kang, D.; Choi, E.; Nam, Y.; Lee, K.H.; You, H.J.; Kang, H.J.; An, S.H.; Jeon, H. Overcome the Barriers of the Skin: Exosome Therapy. Biomater. Res. 2021, 25, 22. [Google Scholar] [CrossRef]
- Kim, S.; Kim, Y.; Hyun, Y.S.; Choi, H.; Kim, S.Y.; Kim, T.G. Exosomes from Human Cord Blood Plasma Accelerate Cutaneous Wound Healing by Promoting Fibroblast Function, Angiogenesis, and M2 Macrophage Differentiation. Biomater. Sci. 2021, 9, 3028–3039. [Google Scholar] [CrossRef]
- Xiong, H.; Wang, S.; Sun, Z.; Li, J.; Zhang, H.; Liu, W.; Ruan, J.; Chen, S.; Gao, C.; Fan, C. The ROS-responsive Scavenger with Intrinsic Antioxidant Capability and Enhanced Immunomodulatory Effects for Cartilage Protection and Osteoarthritis Remission. Appl. Mater. Today 2022, 26, 101366. [Google Scholar] [CrossRef]
- Hong, Y.; Sun, Y.; Rong, X.; Li, D.; Lu, Y.; Ji, Y. Exosomes from Adipose-Derived Stem Cells Attenuate UVB-Induced Apoptosis, ROS, and the Ca2+ Level in HLEC Cells. Exp. Cell Res. 2020, 396, 112321. [Google Scholar] [CrossRef]
- Xu, X.; Lai, Y.; Hua, Z.C. Apoptosis and Apoptotic Body: Disease Message and Therapeutic Target Potentials. Biosci. Rep. 2019, 39, BSR20180992. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Romero, M.; Ratajczak, P.; Lebœuf, C.; Belhadj, S.; Peffault De Latour, R.; Zhao, W.L.; Socié, G.; Janin, A. Increased Apoptosis Is Linked to Severe Acute GVHD in Patients with Fanconi Anemia. Bone Marrow Transplant. 2013, 48, 849–853. [Google Scholar] [CrossRef] [PubMed]
- Simon, H.U.; Haj-Yehia, A.; Levi-Schaffer, F. Role of Reactive Oxygen Species (ROS) in Apoptosis Induction. Apoptosis 2000, 5, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.H.; Kim, Y.J.; Lee, D.Y. Hepatic and Renal Cellular Cytotoxic Effects of Heparin-Coated Superparamagnetic Iron Oxide Nanoparticles. Biomater. Res. 2021, 25, 36. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Jiang, W.; Tan, Y.; Zou, S.; Zhang, H.; Mao, F.; Gong, A.; Qian, H.; Xu, W. HucMSC Exosome-Derived GPX1 Is Required for the Recovery of Hepatic Oxidant Injury. Mol. Ther. 2017, 25, 465–479. [Google Scholar] [CrossRef]
- Zhao, X.M.; Hao, H.S.; Du, W.H.; Zhao, S.J.; Wang, H.Y.; Wang, N.; Wang, D.; Liu, Y.; Qin, T.; Zhu, H. Bin Melatonin Inhibits Apoptosis and Improves the Developmental Potential of Vitrified Bovine Oocytes. J. Pineal Res. 2016, 60, 132–141. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.Y.; Rhim, W.-K.; Woo, J.; Cha, S.-G.; Park, C.G.; Han, D.K. The Upregulation of Regenerative Activity for Extracellular Vesicles with Melatonin Modulation in Chemically Defined Media. Int. J. Mol. Sci. 2022, 23, 15089. https://doi.org/10.3390/ijms232315089
Kim JY, Rhim W-K, Woo J, Cha S-G, Park CG, Han DK. The Upregulation of Regenerative Activity for Extracellular Vesicles with Melatonin Modulation in Chemically Defined Media. International Journal of Molecular Sciences. 2022; 23(23):15089. https://doi.org/10.3390/ijms232315089
Chicago/Turabian StyleKim, Jun Yong, Won-Kyu Rhim, Jiwon Woo, Seung-Gyu Cha, Chun Gwon Park, and Dong Keun Han. 2022. "The Upregulation of Regenerative Activity for Extracellular Vesicles with Melatonin Modulation in Chemically Defined Media" International Journal of Molecular Sciences 23, no. 23: 15089. https://doi.org/10.3390/ijms232315089
APA StyleKim, J. Y., Rhim, W.-K., Woo, J., Cha, S.-G., Park, C. G., & Han, D. K. (2022). The Upregulation of Regenerative Activity for Extracellular Vesicles with Melatonin Modulation in Chemically Defined Media. International Journal of Molecular Sciences, 23(23), 15089. https://doi.org/10.3390/ijms232315089






