Reduction of Obesity and Insulin Resistance through Dual Targeting of VAT and BAT by a Novel Combination of Metabolic Cofactors
Abstract
1. Introduction
2. Results
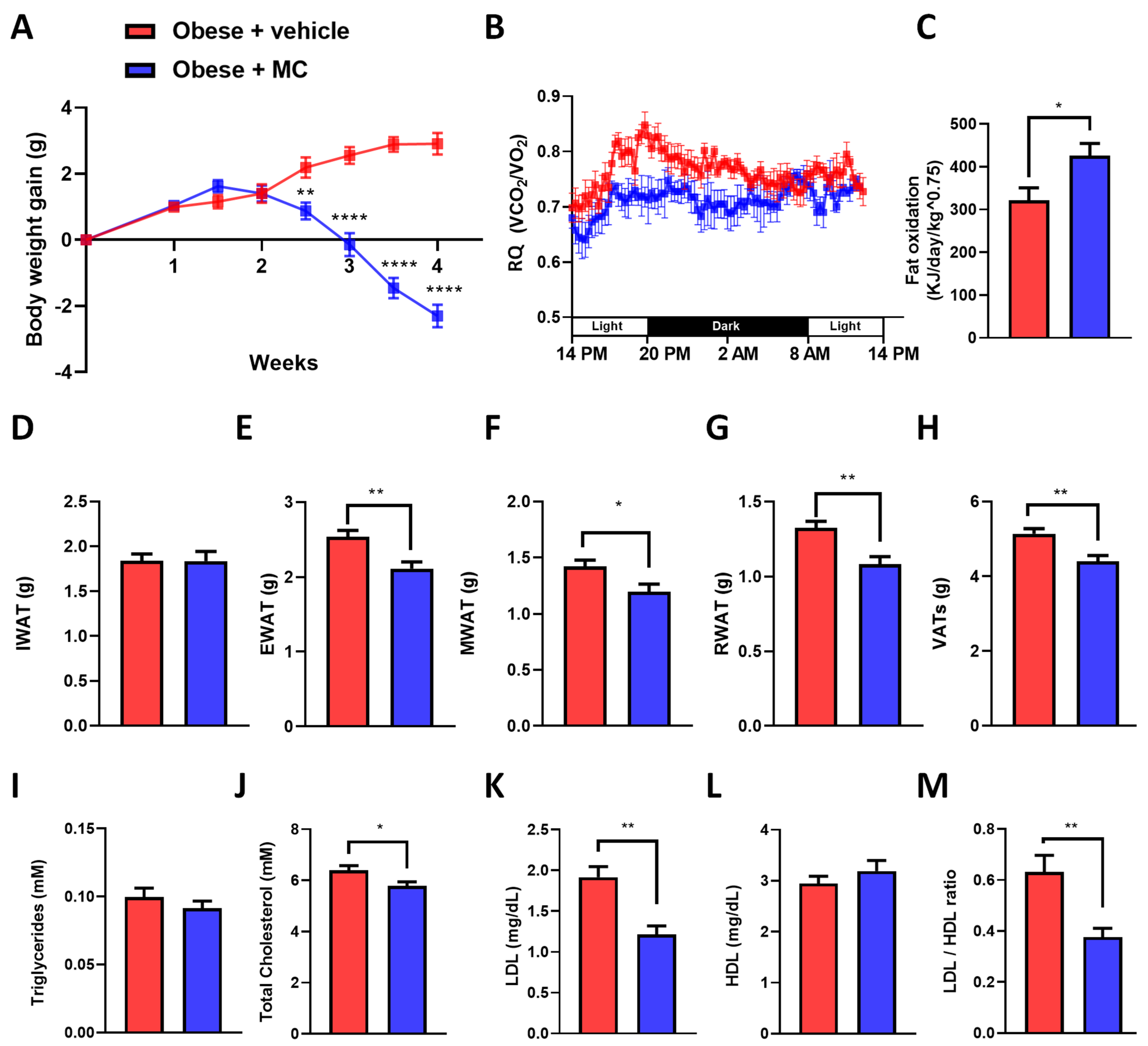
2.1. MC Supplementation Alleviates Obese Diet-Induced Obesity
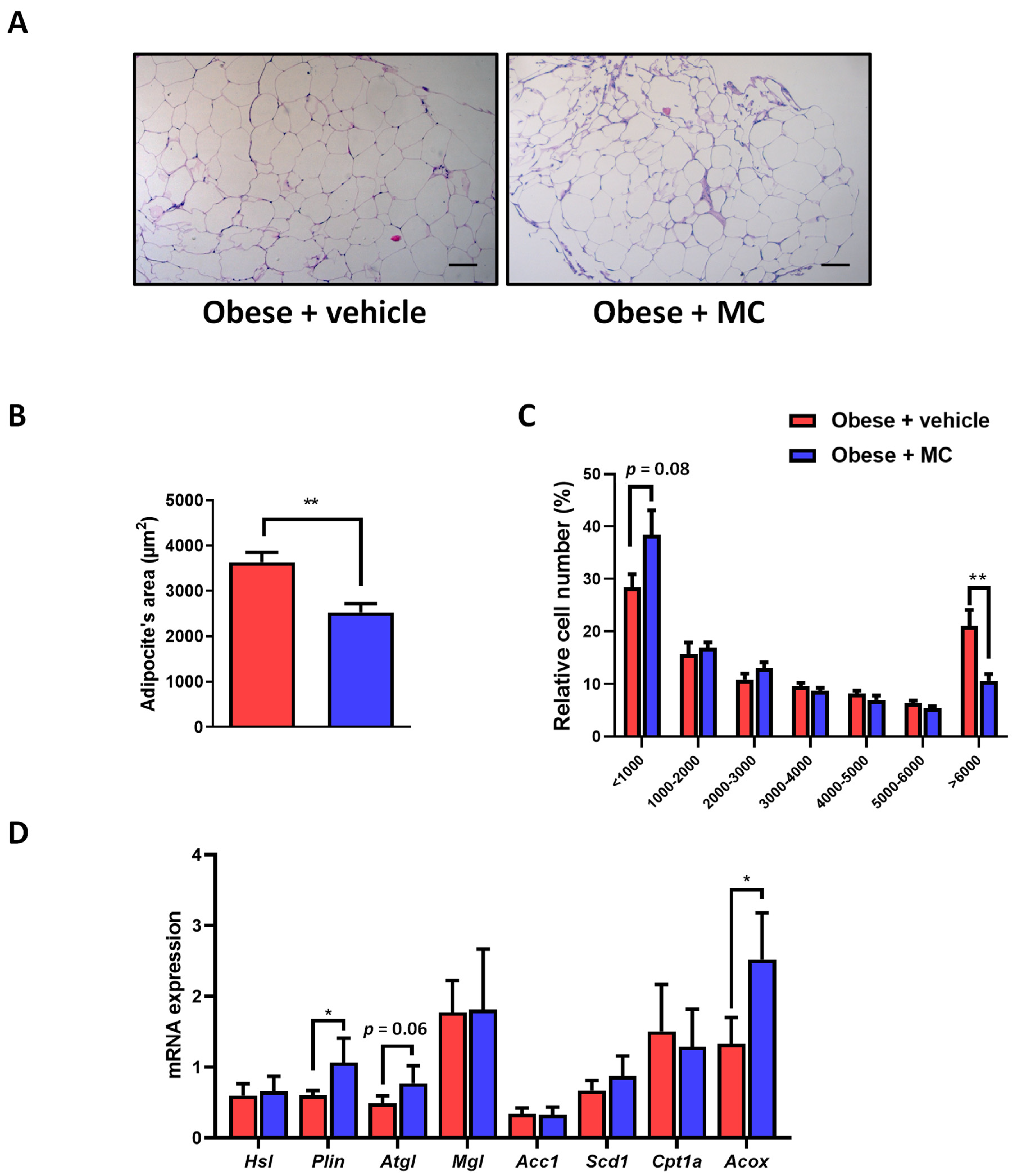
2.2. MC Supplementation Improves Adiposity by Decreasing Adipocyte Hypertrophy in Epididymal Adipose Tissue, with Beneficial Effects on Lipolysis and Fatty Acid Oxidation
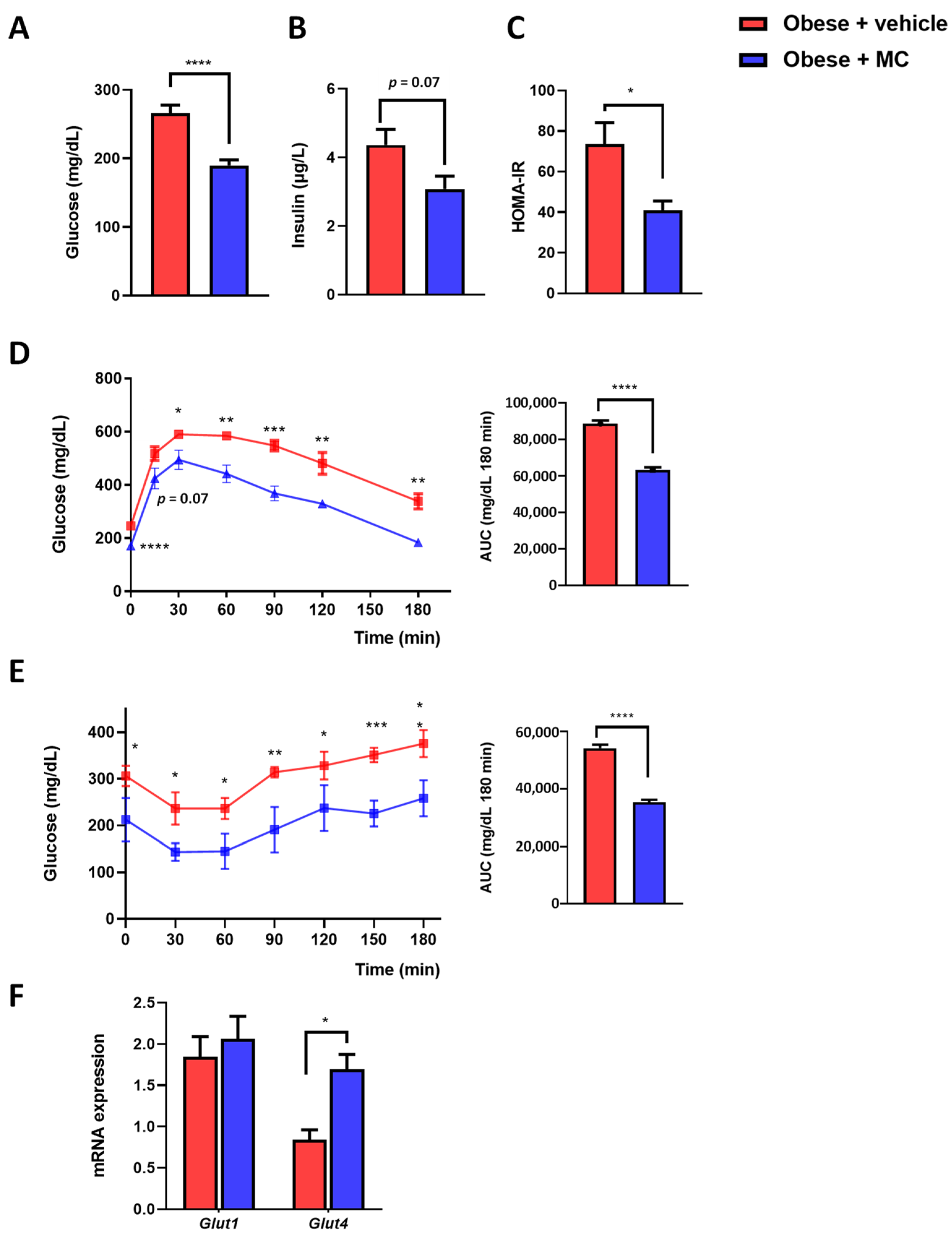
2.3. MC Supplementation Reduced Insulin Resistance Associated with Obesity
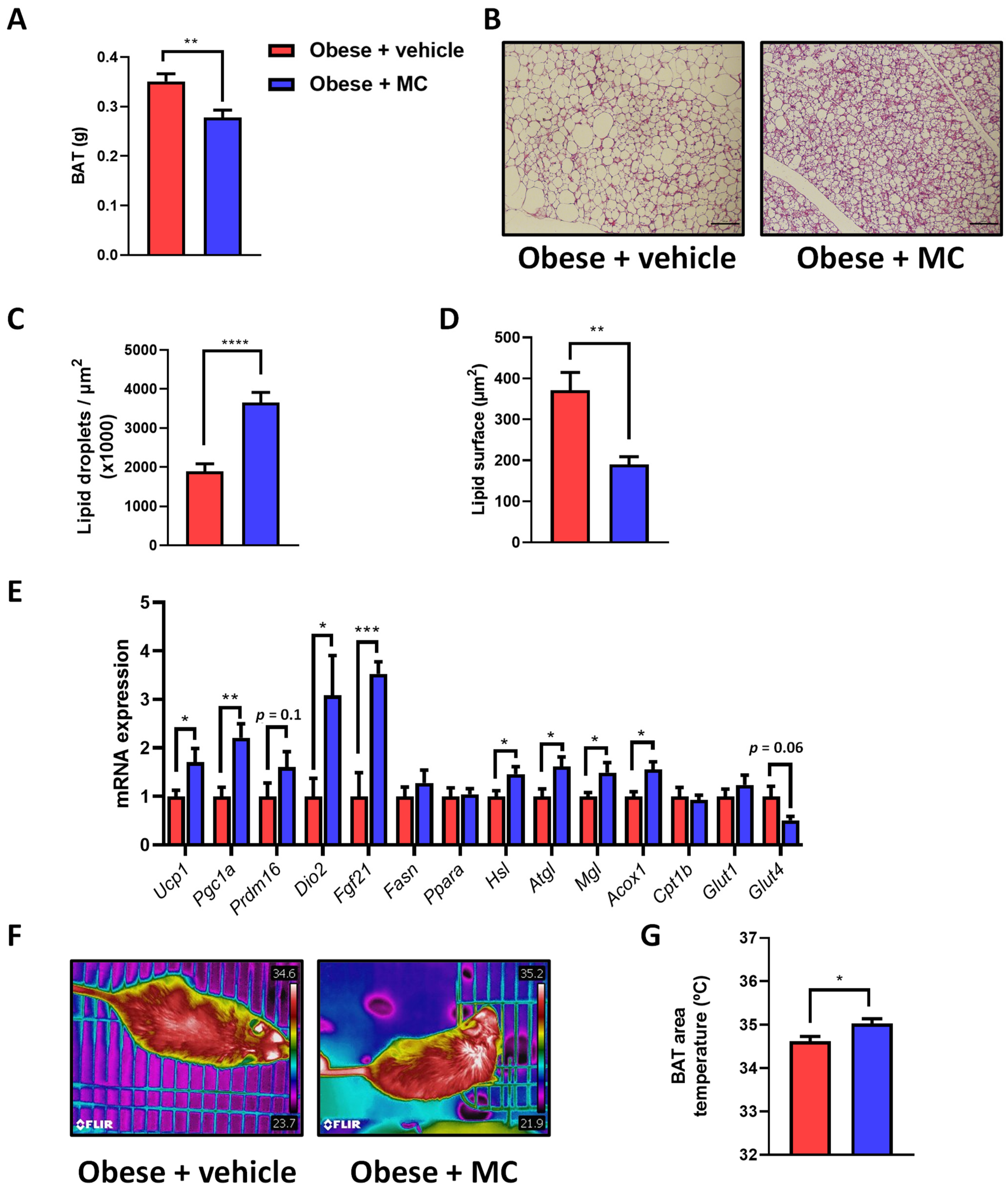
2.4. MC Supplementation Reduces Fat Accumulation in Brown Adipose Tissue by Activation of Lipolysis, Lipid Oxidation and Thermogenesis
3. Discussion
4. Materials and Methods
4.1. Animal Model
4.2. Serum and Blood Analysis
4.3. Indirect Calorimetry
4.4. Histological Analysis
4.5. mRNA Extraction and Quantitative Polymerase Chain Reaction
4.6. BAT Temperature Measurements
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- WHO. New WHO Report: Deaths from Noncommunicable Diseases on the Rise, with Developing World Hit Hardest. Cent. Eur. J. Public Health 2011, 19, 114–120. [Google Scholar]
- Duval, C.; Thissen, U.; Keshtkar, S.; Accart, B.; Stienstra, R.; Boekschoten, M.V.; Roskams, T.; Kersten, S.; Müller, M. Adipose Tissue Dysfunction Signals Progression of Hepatic Steatosis towards Nonalcoholic Steatohepatitis in C57Bl/6 Mice. Diabetes 2010, 59, 3181–3191. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, S. Functional Foods against Metabolic Syndrome (Obesity, Diabetes, Hypertension and Dyslipidemia) and Cardiovasular Disease. Trends Food Sci. Technol. 2014, 35, 114–128. [Google Scholar] [CrossRef]
- Petta, S.; Amato, M.C.; di Marco, V.; Cammà, C.; Pizzolanti, G.; Barcellona, M.R.; Cabibi, D.; Galluzzo, A.; Sinagra, D.; Giordano, C.; et al. Visceral Adiposity Index Is Associated with Significant Fibrosis in Patients with Non-Alcoholic Fatty Liver Disease. Aliment. Pharmacol. Ther. 2012, 35, 238–247. [Google Scholar] [CrossRef]
- Azzu, V.; Vacca, M.; Virtue, S.; Allison, M.; Vidal-Puig, A. Adipose Tissue-Liver Cross Talk in the Control of Whole-Body Metabolism: Implications in Nonalcoholic Fatty Liver Disease. Gastroenterology 2020, 158, 1899–1912. [Google Scholar] [CrossRef]
- Rinella, M.E. Nonalcoholic Fatty Liver Disease a Systematic Review. JAMA J. Am. Med. Assoc. 2015, 313, 2263–2273. [Google Scholar] [CrossRef]
- Basak Engin, A.; Atilla, E. (Eds.) Obesity and Lipotoxicity; Springer: Cham, Switzerland, 2017; Volume 960. [Google Scholar]
- Mardinoglu, A.; Bjornson, E.; Zhang, C.; Klevstig, M.; Söderlund, S.; Ståhlman, M.; Adiels, M.; Hakkarainen, A.; Lundbom, N.; Kilicarslan, M.; et al. Personal Model-Assisted Identification of NAD + and Glutathione Metabolism as Intervention Target in NAFLD. Mol. Syst. Biol. 2017, 13, 916. [Google Scholar] [CrossRef]
- Shi, W.; Hegeman, M.A.; Doncheva, A.; Bekkenkamp-Grovenstein, M.; de Boer, V.C.J.; Keijer, J. High Dose of Dietary Nicotinamide Riboside Induces Glucose Intolerance and White Adipose Tissue Dysfunction in Mice Fed a Mildly Obesogenic Diet. Nutrients 2019, 11, 2439. [Google Scholar] [CrossRef]
- Charron, M.J.; Williams, L.; Seki, Y.; Du, X.Q.; Chaurasia, B.; Saghatelian, A.; Summers, S.A.; Katz, E.B.; Vuguin, P.M.; Reznik, S.E. Antioxidant Effects of N-Acetylcysteine Prevent Programmed Metabolic Disease in Mice. Diabetes 2020, 69, 1650–1661. [Google Scholar] [CrossRef]
- Shi, W.; Hegeman, M.A.; van Dartel, D.A.M.; Tang, J.; Suarez, M.; Swarts, H.; van der Hee, B.; Arola, L.; Keijer, J. Effects of a Wide Range of Dietary Nicotinamide Riboside (NR) Concentrations on Metabolic Flexibility and White Adipose Tissue (WAT) of Mice Fed a Mildly Obesogenic Diet. Mol. Nutr. Food Res. 2017, 61, 1600878. [Google Scholar] [CrossRef]
- Wang, Z.; Yao, T.; Pini, M.; Zhou, Z.; Fantuzzi, G.; Song, Z.; Betaine, S.Z. Betaine Improved Adipose Tissue Function in Mice Fed a High-Fat Diet: A Mechanism for Hepatoprotective Effect of Betaine in Nonalcoholic Fatty Liver Disease. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 298, 634–642. [Google Scholar] [CrossRef] [PubMed]
- Ranjbar Kohan, N.; Tabandeh, M.R.; Nazifi, S.; Soleimani, Z. L-Carnitine Improves Metabolic Disorders and Regulates Apelin and Apelin Receptor Genes Expression in Adipose Tissue in Diabetic Rats. Physiol. Rep. 2020, 8, e14641. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, M.; Gargante, M.P.; Russo, C.; Antic, T.; Vacante, M.; Malaguarnera, M.; Avitabile, T.; Li Volti, G.; Galvano, F. L-Carnitine Supplementation to Diet: A New Tool in Treatment of Nonalcoholic Steatohepatitisa Randomized and Controlled Clinical Trial. Am. J. Gastroenterol. 2010, 105, 1338–1345. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Chen, J.; Chen, J.; Wu, W.; Wang, X.; Wang, Y. The Beneficial Effects of Betaine on Dysfunctional Adipose Tissue and N6-Methyladenosine MRNA Methylation Requires the AMP-Activated Protein Kinase A1 Subunit. J. Nutr. Biochem. 2015, 26, 1678–1684. [Google Scholar] [CrossRef] [PubMed]
- Suárez, M.; Boqué, N.; del Bas, J.M.; Mayneris-Perxachs, J.; Arola, L.; Caimari, A. Mediterranean Diet and Multi-Ingredient-Based Interventions for the Management of Non-Alcoholic Fatty Liver Disease. Nutrients 2017, 9, 1052. [Google Scholar] [CrossRef] [PubMed]
- Khodayar, M.J.; Kalantari, H.; Khorsandi, L.; Rashno, M.; Zeidooni, L. Betaine Protects Mice against Acetaminophen Hepatotoxicity Possibly via Mitochondrial Complex II and Glutathione Availability. Biomed. Pharmacother. 2018, 103, 1436–1445. [Google Scholar] [CrossRef]
- Xia, Y.; Li, Q.; Zhong, W.; Dong, J.; Wang, Z.; Wang, C. L-Carnitine Ameliorated Fatty Liver in High-Calorie Diet/STZ-Induced Type 2 Diabetic Mice by Improving Mitochondrial Function. Diabetol. Metab. Syndr. 2011, 3, 31. [Google Scholar] [CrossRef]
- Cantó, C.; Houtkooper, R.H.; Pirinen, E.; Youn, D.Y.; Oosterveer, M.H.; Cen, Y.; Fernandez-Marcos, P.J.; Yamamoto, H.; Andreux, P.A.; Cettour-Rose, P.; et al. The NAD+ Precursor Nicotinamide Riboside Enhances Oxidative Metabolism and Protects against High-Fat Diet-Induced Obesity. Cell Metab. 2012, 15, 838–847. [Google Scholar] [CrossRef]
- Dou, X.; Xia, Y.; Chen, J.; Qian, Y.; Li, S.; Zhang, X.; Song, Z. Rectification of Impaired Adipose Tissue Methylation Status and Lipolytic Response Contributes to Hepatoprotective Effect of Betaine in a Mouse Model of Alcoholic Liver Disease. Br. J. Pharmacol. 2014, 171, 4073–4086. [Google Scholar] [CrossRef]
- Salic, K.; Gart, E.; Seidel, F.; Verschuren, L.; Caspers, M.; van Duyvenvoorde, W.; Wong, K.E.; Keijer, J.; Bobeldijk-Pastorova, I.; Wielinga, P.Y.; et al. Combined Treatment with L-Carnitine and Nicotinamide Riboside Improves Hepatic Metabolism and Attenuates Obesity and Liver Steatosis. Int. J. Mol. Sci. 2019, 20, 84359. [Google Scholar] [CrossRef]
- Marin, V.; Rosso, N.; Dal Ben, M.; Raseni, A.; Boschelle, M.; Degrassi, C.; Nemeckova, I.; Nachtigal, P.; Avellini, C.; Tiribelli, C.; et al. An Animal Model for the Juvenile Non-Alcoholic Fatty Liver Disease and Non-Alcoholic Steatohepatitis. PLoS ONE 2016, 11, e0158817. [Google Scholar] [CrossRef] [PubMed]
- Sanches, S.C.L.; Ramalho, L.N.Z.; Augusto, M.J.; da Silva, D.M.; Ramalho, F.S. Nonalcoholic Steatohepatitis: A Search for Factual Animal Models. BioMed Res. Int. 2015, 2015, 574832. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Indias, I.; Tinahones, F.J. Impaired Adipose Tissue Expandability and Lipogenic Capacities as Ones of the Main Causes of Metabolic Disorders. J. Diabetes Res. 2015, 2015, 970375. [Google Scholar] [CrossRef] [PubMed]
- Houjeghani, S.; Kheirouri, S.; Faraji, E.; Jafarabadi, M.A. L-Carnosine Supplementation Attenuated Fasting Glucose, Triglycerides, Advanced Glycation End Products, and Tumor Necrosis Factor–α Levels in Patients with Type 2 Diabetes: A Double-Blind Placebo-Controlled Randomized Clinical Trial. Nutr. Res. 2018, 49, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Townsend, K.L.; Tseng, Y.H. Brown Fat Fuel Utilization and Thermogenesis. Trends Endocrinol. Metab. 2014, 25, 168–177. [Google Scholar] [CrossRef]
- Yu, J.; Shen, J.; Sun, T.T.; Zhang, X.; Wong, N. Obesity, Insulin Resistance, NASH and Hepatocellular Carcinoma. Semin. Cancer Biol. 2013, 23, 483–491. [Google Scholar] [CrossRef]
- Friedman, S.L.; Neuschwander-Tetri, B.A.; Rinella, M.; Sanyal, A.J. Mechanisms of NAFLD Development and Therapeutic Strategies. Nat. Med. 2018, 24, 908–922. [Google Scholar] [CrossRef]
- Huang, X.; Liu, G.; Guo, J.; Su, Z. The PI3K/AKT Pathway in Obesity and Type 2 Diabetes. Int. J. Biol. Sci. 2018, 14, 1483–1496. [Google Scholar] [CrossRef]
- Quesada-Vázquez, S.; Colom-Pellicer, M.; Navarro-Masip, È.; Aragonès, G.; del Bas, J.M.; Caimari, A.; Escoté, X. Supplementation with a Specific Combination of Metabolic Cofactors Ameliorates Non-Alcoholic Fatty Liver Disease and, Hepatic Fibrosis, and Insulin Resistance in Mice. Nutrients 2021, 13, 3532. [Google Scholar] [CrossRef]
- Jang, A.; Kim, D.; Sung, K.S.; Jung, S.; Kim, H.J.; Jo, C. The Effect of Dietary α-Lipoic Acid, Betaine, l-Carnitine, and Swimming on the Obesity of Mice Induced by a High-Fat Diet. Food Funct. 2014, 5, 1966–1974. [Google Scholar] [CrossRef]
- Mun, E.G.; Soh, J.R.; Cha, Y.S. L-Carnitine Reduces Obesity Caused by High-Fat Diet in C57BL_6J Mice. Food Sci. Biotechnol. 2007, 16, 228–233. [Google Scholar]
- Shen, F.C.; Weng, S.W.; Tsao, C.F.; Lin, H.Y.; Chang, C.S.; Lin, C.Y.; Lian, W.S.; Chuang, J.H.; Lin, T.K.; Liou, C.W.; et al. Early Intervention of N-Acetylcysteine Better Improves Insulin Resistance in Diet-Induced Obesity Mice. Free Radic. Res. 2018, 52, 1296–1310. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Gao, M.; Liu, D. N-Acetylcysteine Protects Mice from High Fat Diet-Induced Metabolic Disorders. Pharm. Res. 2016, 33, 2033–2042. [Google Scholar] [CrossRef] [PubMed]
- Zhuhua, Z.; Zhiquan, W.; Zhen, Y.; Yixin, N.; Weiwei, Z.; Xiaoyong, L.; Yueming, L.; Hongmei, Z.; Li, Q.; Qing, S. A Novel Mice Model of Metabolic Syndrome: The High-Fat-High-Fructose Diet-Fed ICR Mice. Exp. Anim. 2015, 64, 435–442. [Google Scholar] [CrossRef]
- Luo, Y. The Role of Sugar-Sweetened Water in the Progression of Nonalcoholic Fatty Liver Disease. Ph.D. Thesis, Auburn University, Auburn, AL, USA, 2016. [Google Scholar]
- Donohoe, C.L.; Doyle, S.L.; Reynolds, J.V. Visceral Adiposity, Insulin Resistance and Cancer Risk. Diabetol. Metab. Syndr. 2011, 3, 12. [Google Scholar] [CrossRef] [PubMed]
- Ejaz, A.; Martinez-Guino, L.; Goldfine, A.B.; Ribas-Aulinas, F.; de Nigris, V.; Ribó, S.; Gonzalez-Franquesa, A.; Garcia-Roves, P.M.; Li, E.; Dreyfuss, J.M.; et al. Dietary Betaine Supplementation Increases Fgf21 Levels to Improve Glucose Homeostasis and Reduce Hepatic Lipid Accumulation in Mice. Diabetes 2016, 65, 902–912. [Google Scholar] [CrossRef]
- Bruls, Y.M.; de Ligt, M.; Lindeboom, L.; Phielix, E.; Havekes, B.; Schaart, G.; Kornips, E.; Wildberger, J.E.; Hesselink, M.K.; Muoio, D.; et al. Carnitine Supplementation Improves Metabolic Flexibility and Skeletal Muscle Acetylcarnitine Formation in Volunteers with Impaired Glucose Tolerance: A Randomised Controlled Trial. EBioMedicine 2019, 49, 318–330. [Google Scholar] [CrossRef]
- Klaus, S.; Pültz, S.; Thöne-Reineke, C.; Wolfram, S. Epigallocatechin Gallate Attenuates Diet-Induced Obesity in Mice by Decreasing Energy Absorption and Increasing Fat Oxidation. Int. J. Obes. 2005, 29, 615–623. [Google Scholar] [CrossRef]
- Singh, A.; Zapata, R.C.; Pezeshki, A.; Chelikani, P.K. Dietary Lactalbumin and Lactoferrin Interact with Inulin to Modulate Energy Balance in Obese Rats. Obesity 2017, 25, 1050–1060. [Google Scholar] [CrossRef]
- Crescenti, A.; del Bas, J.M.; Arola-Arnal, A.; Oms-Oliu, G.; Arola, L.; Caimari, A. Grape Seed Procyanidins Administered at Physiological Doses to Rats during Pregnancy and Lactation Promote Lipid Oxidation and Up-Regulate AMPK in the Muscle of Male Offspring in Adulthood. J. Nutr. Biochem. 2015, 26, 912–920. [Google Scholar] [CrossRef]
- Wanders, R.J.A.; Vreken, P.; Ferdinandusse, S.; Jansen, G.A.; Waterham, H.R.; van Roermund, C.W.T.; van Grunsven, E.G. Peroxisomal Fatty Acid α- and β-Oxidation in Humans: Enzymology, Peroxisomal Metabolite Transporters and Peroxisomal Diseases. Biochem. Soc. Trans. 2001, 29, 250. [Google Scholar] [CrossRef] [PubMed]
- Fiamoncini, J.; Turner, N.; Hirabara, S.M.; Salgado, T.M.L.; Marçal, A.C.; Leslie, S.; da Silva, S.M.A.; Deschamps, F.C.; Luz, J.; Cooney, G.J.; et al. Enhanced Peroxisomal β-Oxidation Is Associated with Prevention of Obesity and Glucose Intolerance by Fish Oil-Enriched Diets. Obesity 2013, 21, 1200–1207. [Google Scholar] [CrossRef] [PubMed]
- Wanders, R.J.A.; Waterham, H.R. Biochemistry of Mammalian Peroxisomes Revisited. Annu. Rev. Biochem. 2006, 75, 295–332. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Tan, F.; Mu, J.; Yi, R.; Zhou, X.; Zhao, X. Anti-Obesity Effects of Lactobacillus Fermentum CQPC05 Isolated from Sichuan Pickle in High-Fat Diet-Induced Obese Mice through PPAR-α Signaling Pathway. Microorganisms 2019, 7, 194. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Lara, K.A.; Rodríguez-Millán, E.; Sebastián, D.; Blanco-Soto, R.; Camacho, M.; Nan, M.N.; Diarte-Añazco, E.M.G.; Mato, E.; Lope-Piedrafita, S.; Roglans, N.; et al. Nicotinamide Protects Against Diet-Induced Body Weight Gain, Increases Energy Expenditure, and Induces White Adipose Tissue Beiging. Mol. Nutr. Food Res. 2021, 65, 2100111. [Google Scholar] [CrossRef] [PubMed]
- Morigny, P.; Houssier, M.; Mouisel, E.; Langin, D. Adipocyte Lipolysis and Insulin Resistance. Biochimie 2016, 125, 259–266. [Google Scholar] [CrossRef]
- Gao, X.; Sun, G.; Randell, E.; Tian, Y.; Zhou, H. Systematic Investigation of the Relationships between Trimethylamine N-Oxide and L-Carnitine with Obesity in Both and Rodents. Food Funct. 2020, 11, 7707–7716. [Google Scholar] [CrossRef]
- Du, J.; Shen, L.; Tan, Z.; Zhang, P.; Zhao, X.; Xu, Y.; Gan, M.; Yang, Q.; Ma, J.; Jiang, A.; et al. Betaine Supplementation Enhances Lipid Metabolism and Improves Insulin Resistance in Mice Fed a High-Fat Diet. Nutrients 2018, 10, 131. [Google Scholar] [CrossRef]
- Ozaki, K.; Sano, T.; Tsuji, N.; Matsuura, T.; Narama, I. Carnitine Is Necessary to Maintain the Phenotype and Function of Brown Adipose Tissue. Lab. Investig. 2011, 91, 704–710. [Google Scholar] [CrossRef]
- Crisol, B.M.; Veiga, C.B.; Lenhare, L.; Braga, R.R.; Silva, V.R.R.; da Silva, A.S.R.; Cintra, D.E.; Moura, L.P.; Pauli, J.R.; Ropelle, E.R. Nicotinamide Riboside Induces a Thermogenic Response in Lean Mice. Life Sci. 2018, 211, 1–7. [Google Scholar] [CrossRef]
- Huang, B.-W.; Chiang, M.-T.; Yao, H.-T.; Chiang, W. The Effect of High-Fat and High-Fructose Diets on Glucose Tolerance and Plasma Lipid and Leptin Levels in Rats. Diabetes Obes. Metab. 2004, 6, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.S.; Koves, T.R.; Pettway, Y.D.; Draper, J.A.; Slentz, D.H.; Grimsrud, P.A.; Ilkayeva, O.R.; Muoio, D.M. Nicotinamide Riboside Supplementation Confers Marginal Metabolic Benefits in Obese Mice without Remodeling the Muscle Acetyl-Proteome. iScience 2022, 25, 103635. [Google Scholar] [CrossRef] [PubMed]
- Zayed, E.A.; AinShoka, A.A.; el Shazly, K.A.; Abd El Latif, H.A. Improvement of Insulin Resistance via Increase of GLUT4 and PPARγ in Metabolic Syndrome-Induced Rats Treated with Omega-3 Fatty Acid or l-Carnitine. J. Biochem. Mol. Toxicol. 2018, 32, e22218. [Google Scholar] [CrossRef] [PubMed]
- Koivisto, U.M.; Martinez-Valdez, H.; Bilan, P.J.; Burdett, E.; Ramlal, T.; Klip, A. GLUT-1 and GLUT-4 in Muscle Cells. J. Biol. Chem. 1991, 266, 2615–2621. [Google Scholar] [CrossRef]
- Zhao, G.; He, F.; Wu, C.; Li, P.; Li, N.; Deng, J.; Zhu, G.; Ren, W.; Peng, Y. Betaine in Inflammation: Mechanistic Aspects and Applications. Front. Immunol. 2018, 9, 1070. [Google Scholar] [CrossRef]
- Nejabati, H.R.; Samadi, N.; Shahnazi, V.; Mihanfar, A.; Fattahi, A.; Latifi, Z.; Bahrami-asl, Z.; Roshangar, L.; Nouri, M. Nicotinamide and Its Metabolite N1-Methylnicotinamide Alleviate Endocrine and Metabolic Abnormalities in Adipose and Ovarian Tissues in Rat Model of Polycystic Ovary Syndrome. Chem. Biol. Interact. 2020, 324, 109093. [Google Scholar] [CrossRef]
- Quesada-Vázquez, S.; Bone, C.; Saha, S.; Triguero, I.; Colom-Pellicer, M.; Aragonès, G.; Hildebrand, F.; del Bas, J.M.; Caimari, A.; Beraza, N.; et al. Microbiota Dysbiosis and Gut Barrier Dysfunction Associated with Non-Alcoholic Fatty Liver Disease Are Modulated by a Specific Metabolic Cofactors’ Combination. Int. J. Mol. Sci. 2022, 23, 13675. [Google Scholar] [CrossRef]
- Yang, H.; Mayneris-Perxachs, J.; Boqué, N.; del Bas, J.M.; Arola, L.; Yuan, M.; Türkez, H.; Uhlén, M.; Borén, J.; Zhang, C.; et al. Combined Metabolic Activators Decrease Liver Steatosis by Activating Mitochondrial Metabolism in Hamsters Fed with a High-Fat Diet. Biomedicines 2021, 9, 1440. [Google Scholar] [CrossRef]
- Reagan-Shaw, S.; Nihal, M.; Ahmad, N. Dose Translation from Animal to Human Studies Revisited. FASEB J. 2008, 22, 659–661. [Google Scholar] [CrossRef]
- Carraro, F.; Stuart, C.A.; Hartl, W.H.; Rosenblatt, J.; Wolfe, R.R.; Robert Wolfee, A.R. Effect of Exercise and Recovery on Muscle Protein Synthesis in Human Subjects. Am. J. Physiol.-Endocrinol. Metab. 1990, 259, E470–E476. [Google Scholar] [CrossRef]
- Bircher, S.; Knechtle, B. Relationship between Fat Oxidation and Lactate Threshold in Athletes and Obese Women and Men. J. Sports Sci. Med. 2004, 3, 174–181. [Google Scholar] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Sun, W.; Qian, J.; Tang, Y. Fasting Exacerbates Hepatic Growth Differentiation Factor 15 to Promote Fatty Acid β-Oxidation and Ketogenesis via Activating XBP1 Signaling in Liver. Redox Biol. 2018, 16, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Geurts, L.; Everard, A.; van Hul, M.; Essaghir, A.; Duparc, T.; Matamoros, S.; Plovier, H.; Castel, J.; Denis, R.G.P.; Bergiers, M.; et al. Adipose Tissue NAPE-PLD Controls Fat Mass Development by Altering the Browning Process and Gut Microbiota. Nat. Commun. 2015, 6, 6495. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Xu, M.; Peng, Y.; Naren, Q.; Xu, Y.; Wang, X.; Yang, G.; Shi, X.; Li, X. Triptolide Enhances Lipolysis of Adipocytes by Enhancing ATGL Transcription via Upregulation of P53. Phytother. Res. 2020, 34, 3298–3310. [Google Scholar] [CrossRef]
- Kimura, R.; Takahashi, N.; Lin, S.; Goto, T.; Murota, K.; Nakata, R.; Inoue, H.; Kawada, T. DHA Attenuates Postprandial Hyperlipidemia via Activating PPARα in Intestinal Epithelial Cells. J. Lipid Res. 2013, 54, 3258–3268. [Google Scholar] [CrossRef]
- Ma, Y.; Gao, M.; Sun, H.; Liu, D. Interleukin-6 Gene Transfer Reverses Body Weight Gain and Fatty Liver in Obese Mice. Biochim. Biophys. Acta Mol. Basis Dis. 2015, 1852, 1001–1011. [Google Scholar] [CrossRef]
- Li, Y.; Wong, K.; Giles, A.; Jiang, J.; Lee, J.W.; Adams, A.C.; Kharitonenkov, A.; Yang, Q.; Gao, B.; Guarente, L.; et al. Hepatic SIRT1 Attenuates Hepatic Steatosis and Controls Energy Balance in Mice by Inducing Fibroblast Growth Factor 21. Gastroenterology 2014, 146, 539–549.e7. [Google Scholar] [CrossRef]
- Zhou, J.; Febbraio, M.; Wada, T.; Zhai, Y.; Kuruba, R.; He, J.; Lee, J.H.; Khadem, S.; Ren, S.; Li, S.; et al. Hepatic Fatty Acid Transporter Cd36 Is a Common Target of LXR, PXR, and PPARγ in Promoting Steatosis. Gastroenterology 2008, 134, 556–567. [Google Scholar] [CrossRef]
- Vinaik, R.; Barayan, D.; Auger, C.; Abdullahi, A.; Jeschke, M.G. Regulation of Glycolysis and the Warburg Effect in Wound Healing. JCI Insight 2020, 5, e138949. [Google Scholar] [CrossRef]
- Atkinson, B.J.; Griesel, B.A.; King, C.D.; Josey, M.A.; Olson, A.L. Moderate Glut4 Overexpression Improves Insulin Sensitivity and Fasting Triglyceridemia in High-Fat Diet-Fed Transgenic Mice. Diabetes 2013, 62, 2249–2258. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Liu, M.; Jin, Z.; Yaqoob, S.; Zheng, M.; Cai, D.; Liu, J.; Guo, S. Ginsenoside Rg2 Inhibits Adipogenesis in 3T3-L1 Preadipocytes and Suppresses Obesity in High-Fat-Diet-Induced Obese Mice through the AMPK Pathway. Food Funct. 2019, 10, 3603–3614. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, N.; Gill, J.F.; Brink, M.; Handschin, C. Moderate Modulation of Cardiac PGC-1α Expression Partially Affects Age-Associated Transcriptional Remodeling of the Heart. Front. Physiol. 2018, 9, 242. [Google Scholar] [CrossRef] [PubMed]
- Montagner, A.; Polizzi, A.; Fouché, E.; Ducheix, S.; Lippi, Y.; Lasserre, F.; Barquissau, V.; Régnier, M.; Lukowicz, C.; Benhamed, F.; et al. Liver PPARα Is Crucial for Whole-Body Fatty Acid Homeostasis and Is Protective against NAFLD. Gut 2016, 65, 1202–1214. [Google Scholar] [CrossRef]
- Shen, Y.; Zhou, H.; Jin, W.; Lee, H.J. Acute Exercise Regulates Adipogenic Gene Expression in White Adipose Tissue. Biol. Sport 2016, 33, 381–391. [Google Scholar] [CrossRef]
- Choi, H.; Kim, C.S.; Yu, R. Quercetin Upregulates Uncoupling Protein 1 in White/Brown Adipose Tissues through Sympathetic Stimulation. J. Obes. Metab. Syndr. 2018, 27, 102–109. [Google Scholar] [CrossRef]
- Martínez-Sánchez, N.; Seoane-Collazo, P.; Contreras, C.; Varela, L.; Villarroya, J.; Rial-Pensado, E.; Buqué, X.; Aurrekoetxea, I.; Delgado, T.C.; Vázquez-Martínez, R.; et al. Hypothalamic AMPK-ER Stress-JNK1 Axis Mediates the Central Actions of Thyroid Hormones on Energy Balance. Cell Metab. 2017, 26, 212–229.e12. [Google Scholar] [CrossRef]
| Primers | Forward | Reverse | Reference |
|---|---|---|---|
| Acc1 | GATGAACCATCTCCGTTGGC | CCCAATTATGAATCGGGAGTGC | [65] |
| Acox1 | CTATGGGATCAGCCAGAAAG | AGTCAAAGGCATCCACCAAAG | [66] |
| Atgl | CAACGCCACTCACATCTACGG | GGACACCTCAATAATGTTGGCAC | [67] |
| Cpt1α | CTCAGTGGGAGCGACTCTTCA | GGCCTCTGTGGTACACGACAA | [68] |
| Cpt1b | CGAGGATTCTCTGGAACTGC | GGCCTCTGTGGTACACGACAA | [69] |
| Dio2 | AGAGTGGAGGCGCATGCT | GGCATCTAGGAGGAAGCTGTTC | [70] |
| Fasn | GCTGCGGAAACTTCAGGAAAT | AGAGACGTGTCACTCCTGGACTT | [71] |
| Glut1 | TCAACACGGCCTTCACTG | CACGATGCTCAGATAGGACATC | [72] |
| Glut4 | AAAAGTGCCTGAAACCAGAG | TCACCTCCTGCTCTAAAAGG | [73] |
| Hsl | TCCTGGAACTAAGTGGACGCAAG | CAGACACACTCCTGCGCATAGAC | [74] |
| Mgl | CGGAACAAGTCGGAGGTTGA | TGTCCTGACTCCGGGATGAT | [67] |
| Pgc1a | AGCCGTGACCACTGACAACGAG | GCTGCATGGTTCTGAGTGCTAAG | [75] |
| Plin1 | GTCAATGAACAAGGGCCCAAC | CACAGGCAGCTGCAGAACTCTC | [74] |
| Pparα | CCCTGTTTGTGGCTGCTATAATTT | GGGAAGAGGAAGGTGTCATCTG | [76] |
| Prdm16 | CAGCACGGTGAAGCCATTC | GCGTGCATCCGCTTGTG | [77] |
| Scd1 | AGATCTCCAGTTCTTACACGACCAC | GACGGATGTCTTCTTCCAGGTG | [65] |
| Ucp1 | ACTGCCACACCTCCAGTCATT | CTTTGCCTCACTCAGGATTGG | [78] |
| 36b4 | AGTCCCTGCCCTTTGTACACA | CGATCCGAGGGCCTCACTA | [30] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quesada-Vázquez, S.; Antolín, A.; Colom-Pellicer, M.; Aragonès, G.; Herrero, L.; Del Bas, J.M.; Caimari, A.; Escoté, X. Reduction of Obesity and Insulin Resistance through Dual Targeting of VAT and BAT by a Novel Combination of Metabolic Cofactors. Int. J. Mol. Sci. 2022, 23, 14923. https://doi.org/10.3390/ijms232314923
Quesada-Vázquez S, Antolín A, Colom-Pellicer M, Aragonès G, Herrero L, Del Bas JM, Caimari A, Escoté X. Reduction of Obesity and Insulin Resistance through Dual Targeting of VAT and BAT by a Novel Combination of Metabolic Cofactors. International Journal of Molecular Sciences. 2022; 23(23):14923. https://doi.org/10.3390/ijms232314923
Chicago/Turabian StyleQuesada-Vázquez, Sergio, Anna Antolín, Marina Colom-Pellicer, Gerard Aragonès, Laura Herrero, Josep Maria Del Bas, Antoni Caimari, and Xavier Escoté. 2022. "Reduction of Obesity and Insulin Resistance through Dual Targeting of VAT and BAT by a Novel Combination of Metabolic Cofactors" International Journal of Molecular Sciences 23, no. 23: 14923. https://doi.org/10.3390/ijms232314923
APA StyleQuesada-Vázquez, S., Antolín, A., Colom-Pellicer, M., Aragonès, G., Herrero, L., Del Bas, J. M., Caimari, A., & Escoté, X. (2022). Reduction of Obesity and Insulin Resistance through Dual Targeting of VAT and BAT by a Novel Combination of Metabolic Cofactors. International Journal of Molecular Sciences, 23(23), 14923. https://doi.org/10.3390/ijms232314923










