The Origin and Biomedical Relevance of Cannabigerol
Abstract
:1. Introduction
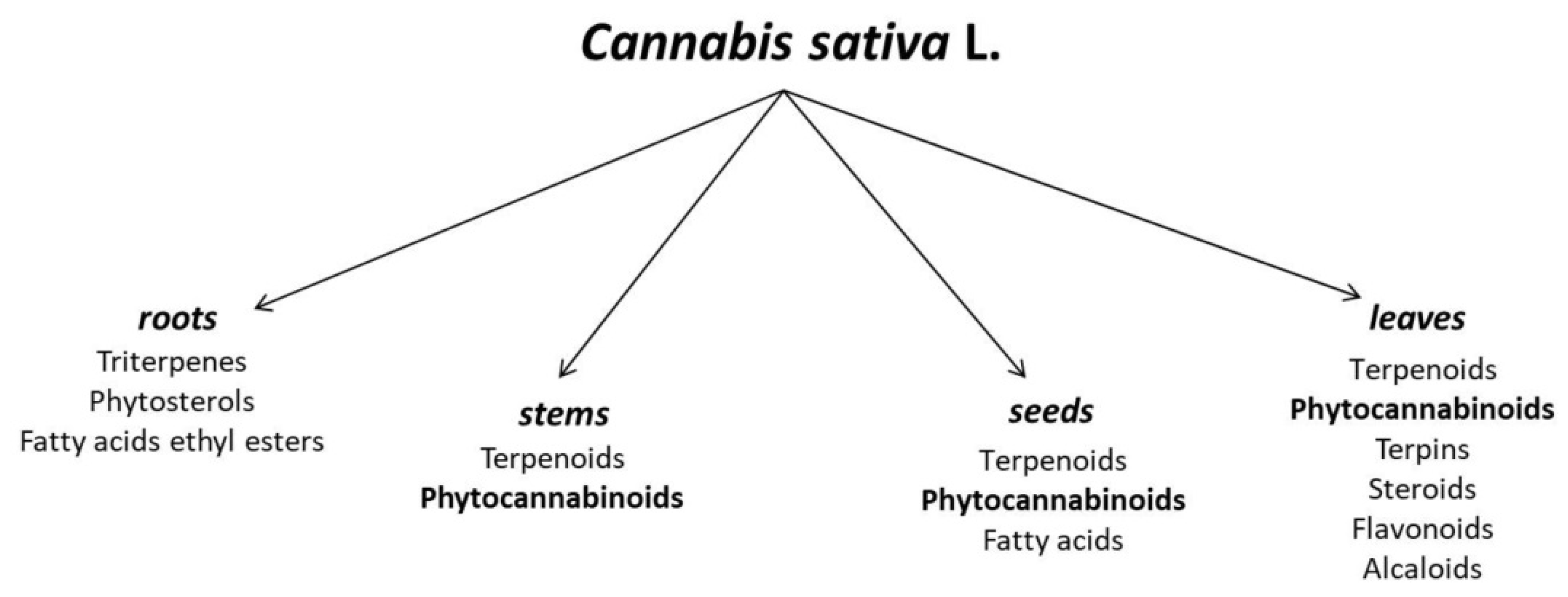
2. Cannabis sativa L. as a Source of Cannabigerol
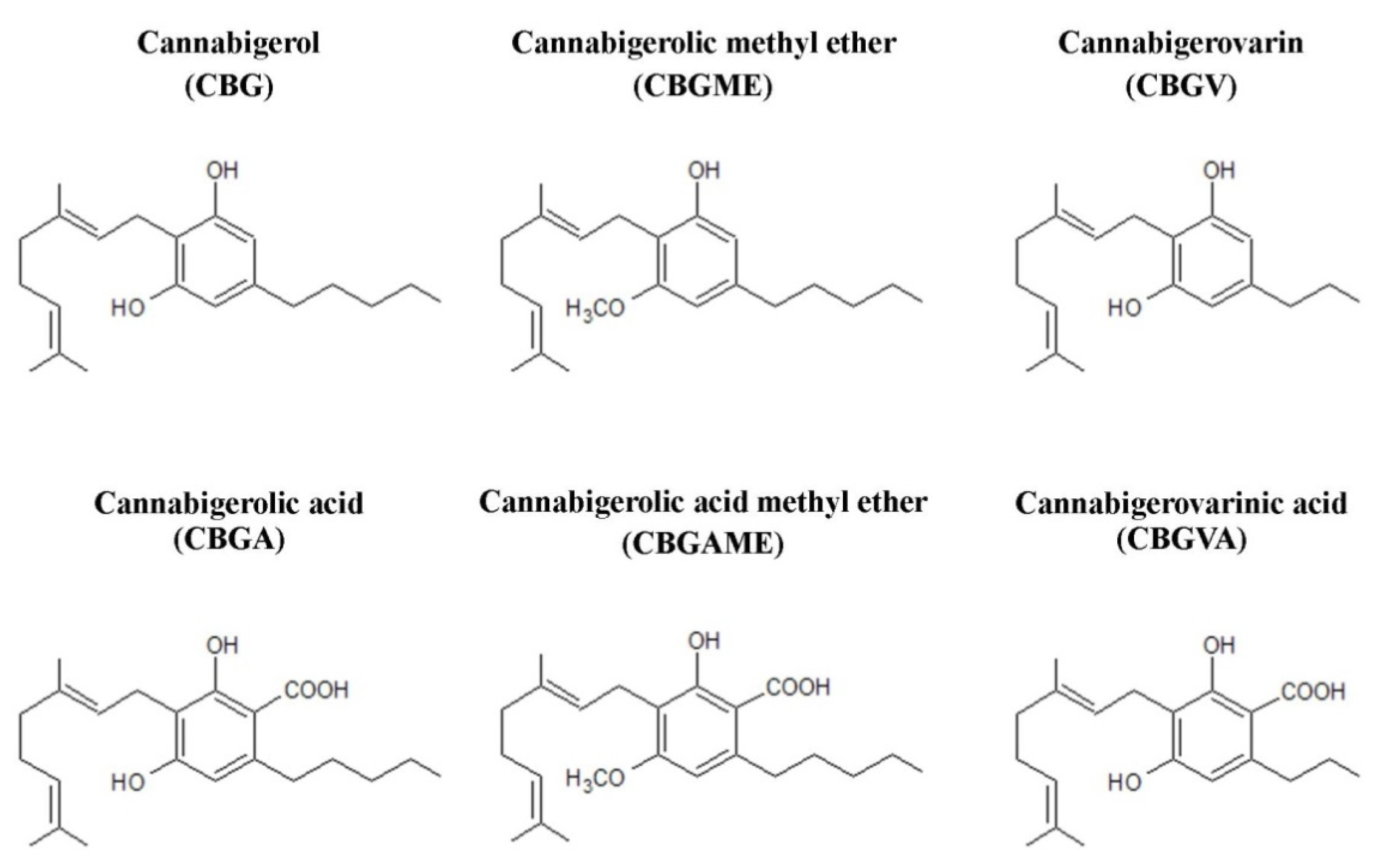
3. Structure of Compounds from the Cannabigerol-Type Group
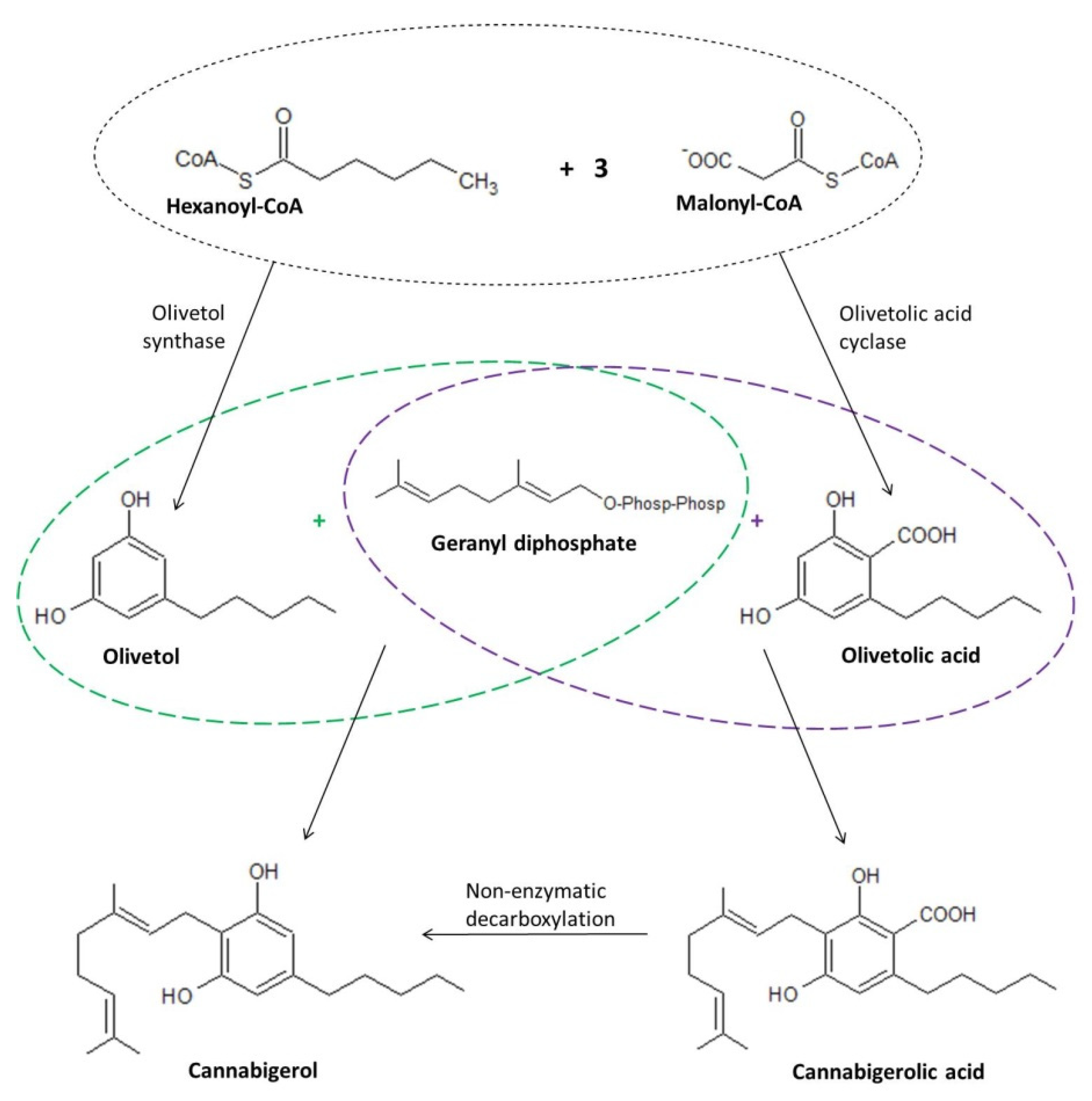
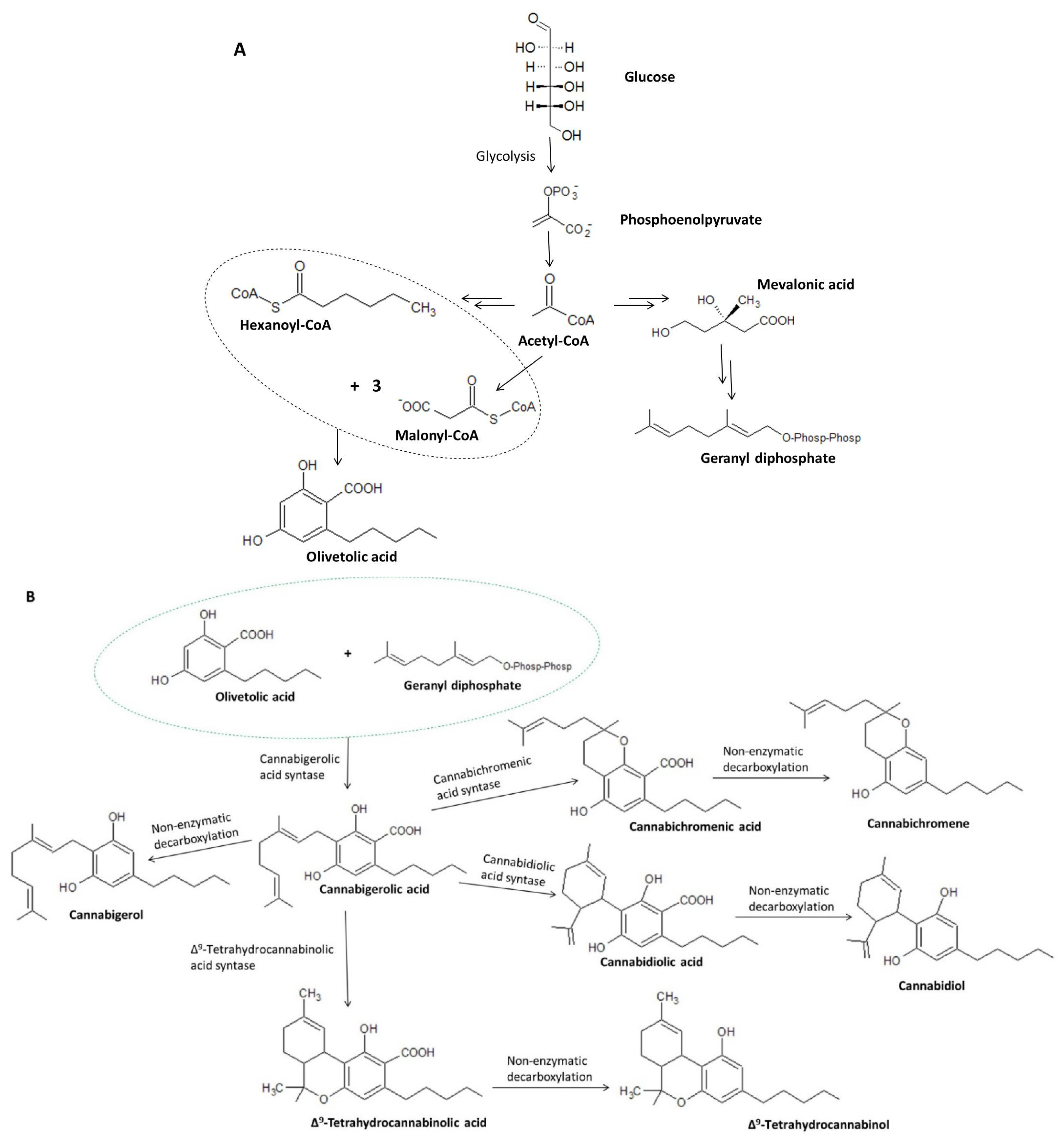
4. Biosynthesis of Cannabigerol
5. Biological Activity of Cannabigerol
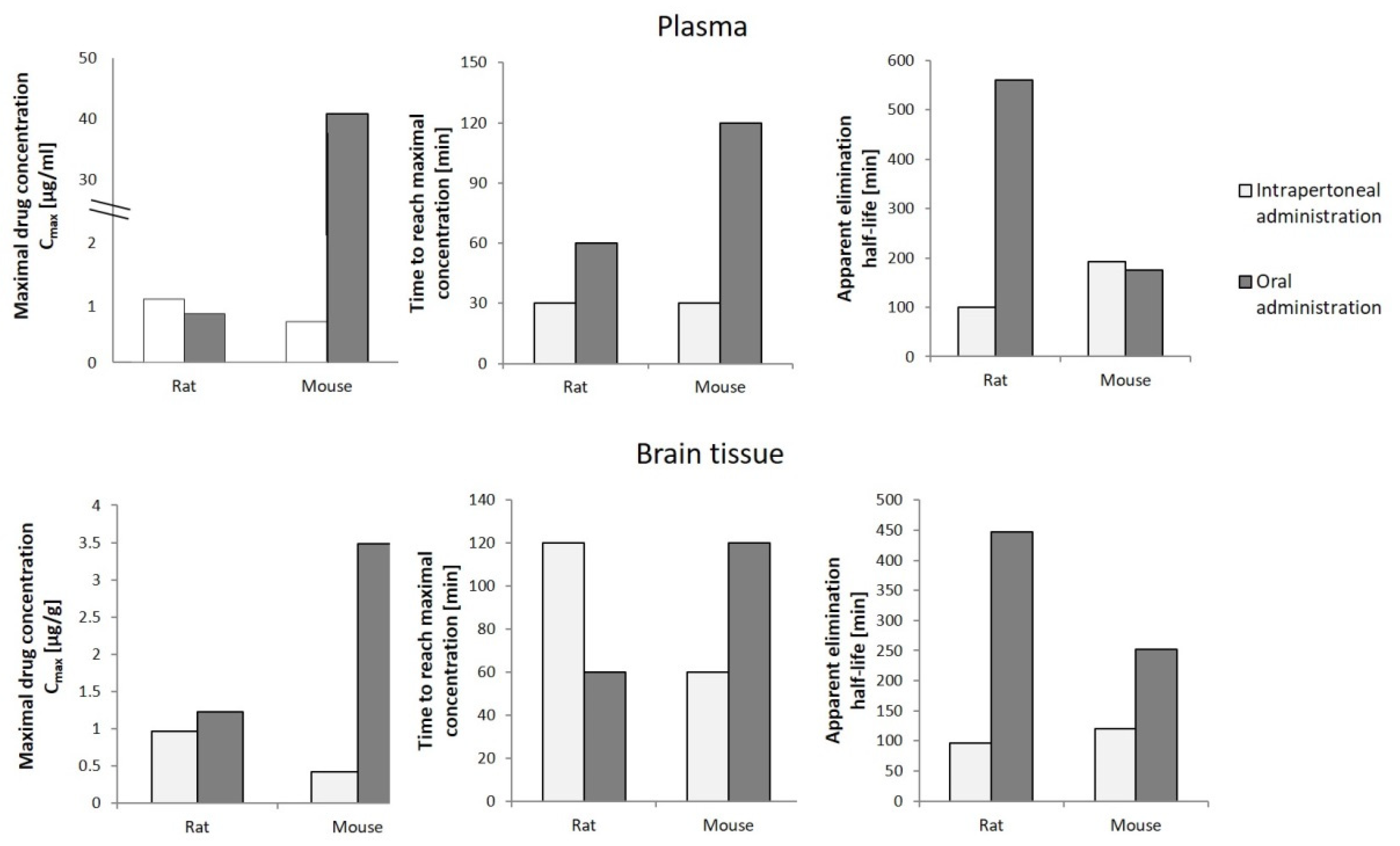
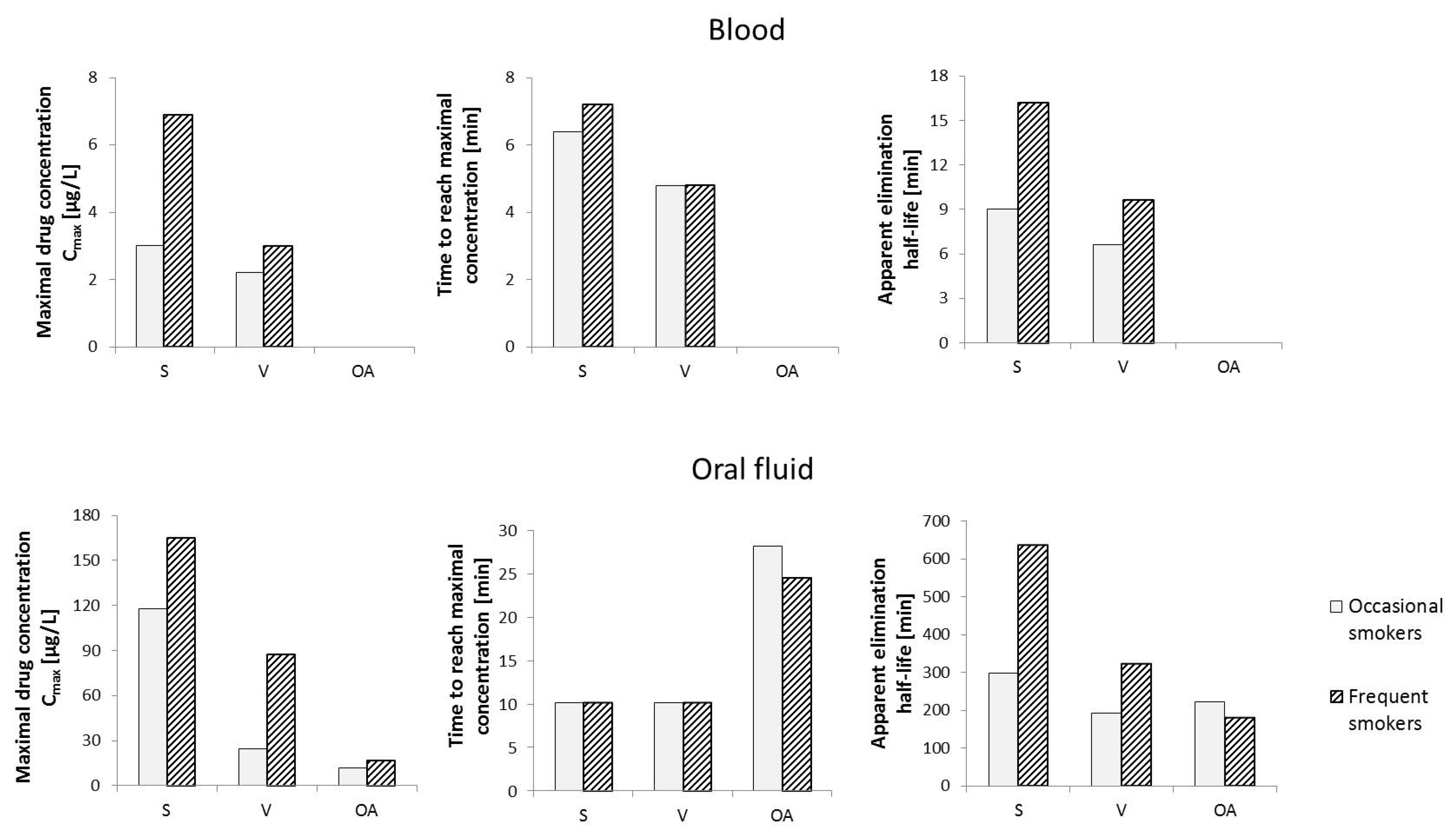
6. Pharmacokinetics of Cannabigerol
7. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Karimi, A.; Majlesi, M.; Rafieian-Kopaei, M. Herbal versus Synthetic Drugs; Beliefs and Facts. J. Nephropharmacology 2015, 4, 27–30. [Google Scholar]
- Manandhar, S.; Luitel, S.; Dahal, R.K. In Vitro Antimicrobial Activity of Some Medicinal Plants against Human Pathogenic Bacteria. J. Trop. Med. 2019, 2019, 1895340. [Google Scholar] [CrossRef] [Green Version]
- Arsene, M.M.J.; Jorelle, A.B.J.; Sarra, S.; Viktorovna, P.I.; Davares, D.; Ingrid, N.K.C.; Steve, A.A.F.; Andreevna, S.L.; Vyacheslavovna, Y.N.; Carime, B.Z. Short Review on the Potential Alternatives to Antibiotics in the Era of Antibiotic Resistance. J. Appl. Pharm. Sci. 2022, 12, 29–40. [Google Scholar] [CrossRef]
- Keshari, A.K.; Srivastava, A.; Upadhayaya, M.; Srivastava, R. Antioxidants and Free Radicals Scavenging Activity of Medicinal Plants. J. Pharmacogn. Phytochem. 2018, 7, 1499–1504. [Google Scholar]
- Ribeiro, V.P.; Arruda, C.; Abd El-Salam, M.; Bastos, J.K. Brazilian Medicinal Plants with Corroborated Anti-Inflammatory Activities: A Review. Pharm. Biol. 2018, 56, 253–268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Majolo, F.; de Oliveira Becker Delwing, L.K.; Marmitt, D.J.; Bustamante-Filho, I.C.; Goettert, M.I. Medicinal Plants and Bioactive Natural Compounds for Cancer Treatment: Important Advances for Drug Discovery. Phytochem. Lett. 2019, 31, 196–207. [Google Scholar] [CrossRef]
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis Sativa: A Comprehensive Ethnopharmacological Review of a Medicinal Plant with a Long History. J. Ethnopharmacol. 2018, 227, 300–315. [Google Scholar] [CrossRef]
- Hartsel, J.A.; Eades, J.; Hickory, B.; Makriyannis, A. Chapter 53—Cannabis Sativa and Hemp. In Nutraceuticals; Gupta, R.C., Ed.; Academic Press: Boston, MA, USA, 2016; pp. 735–754. ISBN 978-0-12-802147-7. [Google Scholar]
- Stasiłowicz, A.; Tomala, A.; Podolak, I.; Cielecka-Piontek, J. Cannabis sativa L. as a Natural Drug Meeting the Criteria of a Multitarget Approach to Treatment. Int. J. Mol. Sci. 2021, 22, 778. [Google Scholar] [CrossRef]
- García-Valverde, M.T.; de Medina, V.S.; Codesido, V.; Hidalgo-García, J.; Ferreiro-Vera, C. Exploring the Mysteries of Cannabis through Gas Chromatography. In Recent Advances in Gas Chromatography; Mutelet, F., Ed.; IntechOpen: London, UK, 2020; ISBN 978-1-83962-603-6. [Google Scholar]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and Anti-Inflammatory Properties of Cannabidiol. Antioxidants 2020, 9, 21. [Google Scholar] [CrossRef] [Green Version]
- Deiana, S. Chapter 99—Potential Medical Uses of Cannabigerol: A Brief Overview. In Handbook of Cannabis and Related Pathologies; Preedy, V.R., Ed.; Academic Press: San Diego, CA, USA, 2017; pp. 958–967. ISBN 978-0-12-800756-3. [Google Scholar]
- Hill, A.J.; Williams, C.M.; Whalley, B.J.; Stephens, G.J. Phytocannabinoids as Novel Therapeutic Agents in CNS Disorders. Pharmacol. Ther. 2012, 133, 79–97. [Google Scholar] [CrossRef] [Green Version]
- Bernstein, N.; Gorelick, J.; Koch, S. Interplay between Chemistry and Morphology in Medical Cannabis (Cannabis sativa L.). Ind. Crops Prod. 2019, 129, 185–194. [Google Scholar] [CrossRef]
- Schilling, S.; Melzer, R.; McCabe, P.F. Cannabis Sativa. Curr. Biol. CB 2020, 30, R8–R9. [Google Scholar] [CrossRef] [PubMed]
- Brenneisen, R. Chemistry and Analysis of Phytocannabinoids and Other Cannabis Constituents. In Marijuana and the Cannabinoids; ElSohly, M.A., Ed.; Forensic Science and Medicine; Humana Press: Totowa, NJ, USA, 2007; pp. 17–49. ISBN 978-1-59259-947-9. [Google Scholar]
- Kornpointner, C.; Sainz Martinez, A.; Marinovic, S.; Haselmair-Gosch, C.; Jamnik, P.; Schröder, K.; Löfke, C.; Halbwirth, H. Chemical Composition and Antioxidant Potential of Cannabis sativa L. Roots. Ind. Crops Prod. 2021, 165, 113422. [Google Scholar] [CrossRef]
- Audu, B.S.; Ofojekwu, P.C.; Ujah, A.; Ajima, M.N.O. Phytochemical, Proximate Composition, Amino Acid Profile and Characterization of Marijuana (Cannabis sativa L.). J. Phytopharm. 2014, 3, 35–43. [Google Scholar] [CrossRef]
- Nigro, E.; Formato, M.; Crescente, G.; Daniele, A. Cancer Initiation, Progression and Resistance: Are Phytocannabinoids from Cannabis sativa L. Promising Compounds? Molecules 2021, 26, 2668. [Google Scholar] [CrossRef]
- Tagen, M.; Klumpers, L.E. Review of Delta-8-Tetrahydrocannabinol (Δ8-THC): Comparative Pharmacology with Δ9-THC. Br. J. Pharmacol. 2022, 179, 3915–3933. [Google Scholar] [CrossRef]
- Filipiuc, L.E.; Ababei, D.C.; Alexa-Stratulat, T.; Pricope, C.V.; Bild, V.; Stefancescu, R.; Stanciu, G.D.; Tamba, B.-I. Major Phytocannabinoids and Their Related Compounds: Should We Only Search for Drugs That Act on Cannabinoid Receptors? Pharmaceutics 2021, 13, 1823. [Google Scholar] [CrossRef]
- Lu, D.; Potter, D.E. Chapter 58—Cannabinoids and the Cannabinoid Receptors: An Overview. In Handbook of Cannabis and Related Pathologies; Preedy, V.R., Ed.; Academic Press: San Diego, CA, USA, 2017; pp. 553–563. ISBN 978-0-12-800756-3. [Google Scholar]
- Prandi, C.; Blangetti, M.; Namdar, D.; Koltai, H. Structure-Activity Relationship of Cannabis Derived Compounds for the Treatment of Neuronal Activity-Related Diseases. Mol. Basel Switz. 2018, 23, E1526. [Google Scholar] [CrossRef] [Green Version]
- Morales, P.; Hurst, D.P.; Reggio, P.H. Molecular Targets of the Phytocannabinoids: A Complex Picture. Prog. Chem. Org. Nat. Prod. 2017, 103, 103–131. [Google Scholar] [CrossRef] [Green Version]
- Andre, C.M.; Hausman, J.-F.; Guerriero, G. Cannabis Sativa: The Plant of the Thousand and One Molecules. Front. Plant Sci. 2016, 7, 19. [Google Scholar] [CrossRef] [Green Version]
- ElSohly, M.A.; Gul, W. Constituents of Cannabis Sativa. In Handbook of Cannabis; Oxford University Press: Oxford, UK, 2014; ISBN 978-0-19-966268-5. [Google Scholar]
- Turner, C.E.; Elsohly, M.A.; Boeren, E.G. Constituents of Cannabis sativa L. XVII. A Review of the Natural Constituents. J. Nat. Prod. 1980, 43, 169–234. [Google Scholar] [CrossRef] [PubMed]
- Andriotis, E.G.; Chachlioutaki, K.; Monou, P.K.; Bouropoulos, N.; Tzetzis, D.; Barmpalexis, P.; Chang, M.-W.; Ahmad, Z.; Fatouros, D.G. Development of Water-Soluble Electrospun Fibers for the Oral Delivery of Cannabinoids. AAPS PharmSciTech 2021, 22, 23. [Google Scholar] [CrossRef] [PubMed]
- Perrotin-Brunel, H.; Kroon, M.C.; van Roosmalen, M.J.E.; van Spronsen, J.; Peters, C.J.; Witkamp, G.J. Solubility of Non-Psychoactive Cannabinoids in Supercritical Carbon Dioxide and Comparison with Psychoactive Cannabinoids. J. Supercrit. Fluids 2010, 55, 603–608. [Google Scholar] [CrossRef]
- Degenhardt, F.; Stehle, F.; Kayser, O. Chapter 2—The Biosynthesis of Cannabinoids. In Handbook of Cannabis and Related Pathologies; Preedy, V.R., Ed.; Academic Press: San Diego, CA, USA, 2017; pp. 13–23. ISBN 978-0-12-800756-3. [Google Scholar]
- Taura, F.; Tanaka, S.; Taguchi, C.; Fukamizu, T.; Tanaka, H.; Shoyama, Y.; Morimoto, S. Characterization of Olivetol Synthase, a Polyketide Synthase Putatively Involved in Cannabinoid Biosynthetic Pathway. FEBS Lett. 2009, 583, 2061–2066. [Google Scholar] [CrossRef] [Green Version]
- Gagne, S.J.; Stout, J.M.; Liu, E.; Boubakir, Z.; Clark, S.M.; Page, J.E. Identification of Olivetolic Acid Cyclase from Cannabis Sativa Reveals a Unique Catalytic Route to Plant Polyketides. Proc. Natl. Acad. Sci. USA 2012, 109, 12811–12816. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schachtsiek, J.; Warzecha, H.; Kayser, O.; Stehle, F. Current Perspectives on Biotechnological Cannabinoid Production in Plants. Planta Med. 2018, 84, 214–220. [Google Scholar] [CrossRef] [Green Version]
- Booth, J.K.; Page, J.E.; Bohlmann, J. Terpene Synthases from Cannabis Sativa. PLoS ONE 2017, 12, e0173911. [Google Scholar] [CrossRef] [Green Version]
- Karlic, H.; Varga, F. Mevalonate Pathway. In Encyclopedia of Cancer, 3rd ed.; Boffetta, P., Hainaut, P., Eds.; Academic Press: Oxford, UK, 2019; pp. 445–457. ISBN 978-0-12-812485-7. [Google Scholar]
- Nachnani, R.; Raup-Konsavage, W.M.; Vrana, K.E. The Pharmacological Case for Cannabigerol. J. Pharmacol. Exp. Ther. 2021, 376, 204–212. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, P.; Cai, S.; Haughn, G.; Page, J.E. Three Novel Transcription Factors Involved in Cannabinoid Biosynthesis in Cannabis sativa L. Plant Mol. Biol. 2021, 106, 49–65. [Google Scholar] [CrossRef]
- Morales, P.; Reggio, P.H.; Jagerovic, N. An Overview on Medicinal Chemistry of Synthetic and Natural Derivatives of Cannabidiol. Front. Pharmacol. 2017, 8, 422. [Google Scholar] [CrossRef] [Green Version]
- de Meijer, E.P.M.; Hammond, K.M. The Inheritance of Chemical Phenotype in Cannabis sativa L. (II): Cannabigerol Predominant Plants. Euphytica 2005, 145, 189–198. [Google Scholar] [CrossRef]
- Cabrera, C.L.R.; Keir-Rudman, S.; Horniman, N.; Clarkson, N.; Page, C. The Anti-Inflammatory Effects of Cannabidiol and Cannabigerol Alone, and in Combination. Pulm. Pharmacol. Ther. 2021, 69, 102047. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, F.; Fasolino, I.; Romano, B.; Capasso, R.; Maiello, F.; Coppola, D.; Orlando, P.; Battista, G.; Pagano, E.; Di Marzo, V.; et al. Beneficial Effect of the Non-Psychotropic Plant Cannabinoid Cannabigerol on Experimental Inflammatory Bowel Disease. Biochem. Pharmacol. 2013, 85, 1306–1316. [Google Scholar] [CrossRef] [PubMed]
- Gugliandolo, A.; Pollastro, F.; Grassi, G.; Bramanti, P.; Mazzon, E. In Vitro Model of Neuroinflammation: Efficacy of Cannabigerol, a Non-Psychoactive Cannabinoid. Int. J. Mol. Sci. 2018, 19, 1992. [Google Scholar] [CrossRef] [Green Version]
- Giacoppo, S.; Gugliandolo, A.; Trubiani, O.; Pollastro, F.; Grassi, G.; Bramanti, P.; Mazzon, E. Cannabinoid CB2 Receptors Are Involved in the Protection of RAW264.7 Macrophages Against the Oxidative Stress: An in Vitro Study. Eur. J. Histochem. 2017, 61, 2749. [Google Scholar] [CrossRef] [Green Version]
- Di Marzo, V.; Piscitelli, F. The Endocannabinoid System and Its Modulation by Phytocannabinoids. Neurotherapeutics 2015, 12, 692–698. [Google Scholar] [CrossRef]
- Jacobson, M.R.; Watts, J.J.; Boileau, I.; Tong, J.; Mizrahi, R. A Systematic Review of Phytocannabinoid Exposure on the Endocannabinoid System: Implications for Psychosis. Eur. Neuropsychopharmacol. 2019, 29, 330–348. [Google Scholar] [CrossRef]
- Zagzoog, A.; Mohamed, K.A.; Kim, H.J.J.; Kim, E.D.; Frank, C.S.; Black, T.; Jadhav, P.D.; Holbrook, L.A.; Laprairie, R.B. In Vitro and in Vivo Pharmacological Activity of Minor Cannabinoids Isolated from Cannabis Sativa. Sci. Rep. 2020, 10, 20405. [Google Scholar] [CrossRef]
- Starkus, J.; Jansen, C.; Shimoda, L.M.N.; Stokes, A.J.; Small-Howard, A.L.; Turner, H. Diverse TRPV1 Responses to Cannabinoids. Channels Austin Tex 2019, 13, 172–191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Almeida, D.L.; Devi, L.A. Diversity of Molecular Targets and Signaling Pathways for CBD. Pharmacol. Res. Perspect. 2020, 8, e00682. [Google Scholar] [CrossRef]
- De Petrocellis, L.; Ligresti, A.; Moriello, A.S.; Allarà, M.; Bisogno, T.; Petrosino, S.; Stott, C.G.; Di Marzo, V. Effects of Cannabinoids and Cannabinoid-Enriched Cannabis Extracts on TRP Channels and Endocannabinoid Metabolic Enzymes. Br. J. Pharmacol. 2011, 163, 1479–1494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramer, R.; Heinemann, K.; Merkord, J.; Rohde, H.; Salamon, A.; Linnebacher, M.; Hinz, B. COX-2 and PPAR-γ Confer Cannabidiol-Induced Apoptosis of Human Lung Cancer Cells. Mol. Cancer Ther. 2013, 12, 69–82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruhaak, L.R.; Felth, J.; Karlsson, P.C.; Rafter, J.J.; Verpoorte, R.; Bohlin, L. Evaluation of the Cyclooxygenase Inhibiting Effects of Six Major Cannabinoids Isolated from Cannabis Sativa. Biol. Pharm. Bull. 2011, 34, 774–778. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cascio, M.G.; Gauson, L.A.; Stevenson, L.A.; Ross, R.A.; Pertwee, R.G. Evidence That the Plant Cannabinoid Cannabigerol Is a Highly Potent Alpha2-Adrenoceptor Agonist and Moderately Potent 5HT1A Receptor Antagonist. Br. J. Pharmacol. 2010, 159, 129–141. [Google Scholar] [CrossRef] [Green Version]
- Zagožen, M.; Čerenak, A.; Kreft, S. Cannabigerol and Cannabichromene in Cannabis sativa L. Acta Pharm. 2021, 71, 355–364. [Google Scholar] [CrossRef]
- Carberry, J.J. Composition of Olivetol and Method of Use to Reduce or Inhibit the Efects of Tetrahydrocannabinol in the Human Body. U.S. Patent No. 9,918,947, 20 March 2018. [Google Scholar]
- Borrelli, F.; Pagano, E.; Romano, B.; Panzera, S.; Maiello, F.; Coppola, D.; De Petrocellis, L.; Buono, L.; Orlando, P.; Izzo, A.A. Colon Carcinogenesis Is Inhibited by the TRPM8 Antagonist Cannabigerol, a Cannabis-Derived Non-Psychotropic Cannabinoid. Carcinogenesis 2014, 35, 2787–2797. [Google Scholar] [CrossRef] [Green Version]
- Echeverry, C.; Prunell, G.; Narbondo, C.; de Medina, V.S.; Nadal, X.; Reyes-Parada, M.; Scorza, C. A Comparative In Vitro Study of the Neuroprotective Effect Induced by Cannabidiol, Cannabigerol, and Their Respective Acid Forms: Relevance of the 5-HT1A Receptors. Neurotox. Res. 2021, 39, 335–348. [Google Scholar] [CrossRef]
- García, C.; Gómez-Cañas, M.; Burgaz, S.; Palomares, B.; Gómez-Gálvez, Y.; Palomo-Garo, C.; Campo, S.; Ferrer-Hernández, J.; Pavicic, C.; Navarrete, C.; et al. Benefits of VCE-003.2, a Cannabigerol Quinone Derivative, against Inflammation-Driven Neuronal Deterioration in Experimental Parkinson’s Disease: Possible Involvement of Different Binding Sites at the PPARγ Receptor. J. Neuroinflamm. 2018, 15, 19. [Google Scholar] [CrossRef] [Green Version]
- Navarro, G.; Varani, K.; Reyes Resina, I.; Sánchez de Medina, V.; Rivas-Santisteban, R.; Sánchez-Carnerero Callado, C.; Vincenzi, F.; Casano, S.; Ferreiro-Vera, C.; Canela Campos, E.I.; et al. Cannabigerol Action at Cannabinoid CB1 and CB2 Receptors and at CB1-CB2 Heteroreceptor Complexes. Front. Pharmacol. 2018, 9, 632. [Google Scholar] [CrossRef]
- Walsh, K.B.; McKinney, A.E.; Holmes, A.E. Minor Cannabinoids: Biosynthesis, Molecular Pharmacology and Potential Therapeutic Uses. Front. Pharmacol. 2021, 12, 777804. [Google Scholar] [CrossRef]
- Dawidowicz, A.L.; Olszowy-Tomczyk, M.; Typek, R. CBG, CBD, Δ9-THC, CBN, CBGA, CBDA and Δ9-THCA as Antioxidant Agents and Their Intervention Abilities in Antioxidant Action. Fitoterapia 2021, 152, 104915. [Google Scholar] [CrossRef] [PubMed]
- di Giacomo, V.; Chiavaroli, A.; Recinella, L.; Orlando, G.; Cataldi, A.; Rapino, M.; Di Valerio, V.; Ronci, M.; Leone, S.; Brunetti, L.; et al. Antioxidant and Neuroprotective Effects Induced by Cannabidiol and Cannabigerol in Rat CTX-TNA2 Astrocytes and Isolated Cortexes. Int. J. Mol. Sci. 2020, 21, 3575. [Google Scholar] [CrossRef]
- D’Aniello, E.; Fellous, T.; Iannotti, F.A.; Gentile, A.; Allarà, M.; Balestrieri, F.; Gray, R.; Amodeo, P.; Vitale, R.M.; Di Marzo, V. Identification and Characterization of Phytocannabinoids as Novel Dual PPARα/γ Agonists by a Computational and in Vitro Experimental Approach. Biochim. Biophys. Acta BBA-Gen. Subj. 2019, 1863, 586–597. [Google Scholar] [CrossRef] [PubMed]
- Stone, N.L.; Murphy, A.J.; England, T.J.; O’Sullivan, S.E. A Systematic Review of Minor Phytocannabinoids with Promising Neuroprotective Potential. Br. J. Pharmacol. 2020, 177, 4330–4352. [Google Scholar] [CrossRef]
- Díaz-Alonso, J.; Paraíso-Luna, J.; Navarrete, C.; del Río, C.; Cantarero, I.; Palomares, B.; Aguareles, J.; Fernández-Ruiz, J.; Bellido, M.L.; Pollastro, F.; et al. VCE-003.2, a Novel Cannabigerol Derivative, Enhances Neuronal Progenitor Cell Survival and Alleviates Symptomatology in Murine Models of Huntington’s Disease. Sci. Rep. 2016, 6, 29789. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Meo, C.; Tortolani, D.; Standoli, S.; Angelucci, C.B.; Fanti, F.; Leuti, A.; Sergi, M.; Kadhim, S.; Hsu, E.; Rapino, C.; et al. Effects of Rare Phytocannabinoids on the Endocannabinoid System of Human Keratinocytes. Int. J. Mol. Sci. 2022, 23, 5430. [Google Scholar] [CrossRef]
- Rodríguez-Cueto, C.; Santos-García, I.; García-Toscano, L.; Espejo-Porras, F.; Bellido, M.L.; Fernández-Ruiz, J.; Muñoz, E.; de Lago, E. Neuroprotective Effects of the Cannabigerol Quinone Derivative VCE-003.2 in SOD1G93A Transgenic Mice, an Experimental Model of Amyotrophic Lateral Sclerosis. Biochem. Pharmacol. 2018, 157, 217–226. [Google Scholar] [CrossRef]
- Carrillo-Salinas, F.J.; Navarrete, C.; Mecha, M.; Feliú, A.; Collado, J.A.; Cantarero, I.; Bellido, M.L.; Muñoz, E.; Guaza, C. A Cannabigerol Derivative Suppresses Immune Responses and Protects Mice from Experimental Autoimmune Encephalomyelitis. PLoS ONE 2014, 9, e94733. [Google Scholar] [CrossRef]
- Burgaz, S.; García, C.; Gómez-Cañas, M.; Navarrete, C.; García-Martín, A.; Rolland, A.; del Río, C.; Casarejos, M.J.; Muñoz, E.; Gonzalo-Consuegra, C.; et al. Neuroprotection with the Cannabigerol Quinone Derivative VCE-003.2 and Its Analogs CBGA-Q and CBGA-Q-Salt in Parkinson’s Disease Using 6-Hydroxydopamine-Lesioned Mice. Mol. Cell. Neurosci. 2021, 110, 103583. [Google Scholar] [CrossRef]
- Muñoz, E.; Pollastro, F.; Taglialatela-Scafati, O.; Appendino, G. Chapter 69—Peroxisome Proliferator Activated Receptors and Cannabinoids. In Handbook of Cannabis and Related Pathologies; Preedy, V.R., Ed.; Academic Press: San Diego, CA, USA, 2017; pp. 671–679. ISBN 978-0-12-800756-3. [Google Scholar]
- Granja, A.G.; Carrillo-Salinas, F.; Pagani, A.; Gómez-Cañas, M.; Negri, R.; Navarrete, C.; Mecha, M.; Mestre, L.; Fiebich, B.L.; Cantarero, I.; et al. A Cannabigerol Quinone Alleviates Neuroinflammation in a Chronic Model of Multiple Sclerosis. J. Neuroimmune Pharmacol. 2012, 7, 1002–1016. [Google Scholar] [CrossRef]
- Ligresti, A.; Moriello, A.S.; Starowicz, K.; Matias, I.; Pisanti, S.; De Petrocellis, L.; Laezza, C.; Portella, G.; Bifulco, M.; Di Marzo, V. Antitumor Activity of Plant Cannabinoids with Emphasis on the Effect of Cannabidiol on Human Breast Carcinoma. J. Pharmacol. Exp. Ther. 2006, 318, 1375–1387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deiana, S.; Watanabe, A.; Yamasaki, Y.; Amada, N.; Arthur, M.; Fleming, S.; Woodcock, H.; Dorward, P.; Pigliacampo, B.; Close, S.; et al. Plasma and Brain Pharmacokinetic Profile of Cannabidiol (CBD), Cannabidivarine (CBDV), Δ9-Tetrahydrocannabivarin (THCV) and Cannabigerol (CBG) in Rats and Mice Following Oral and Intraperitoneal Administration and CBD Action on Obsessive-Compulsive Behaviour. Psychopharmacology 2012, 219, 859–873. [Google Scholar] [CrossRef] [PubMed]
- World Drug Report 2021. Available online: https://www.unodc.org/unodc/en/data-and-analysis/wdr2021.html (accessed on 27 June 2022).
- Newmeyer, M.N.; Swortwood, M.J.; Barnes, A.J.; Abulseoud, O.A.; Scheidweiler, K.B.; Huestis, M.A. Free and Glucuronide Whole Blood Cannabinoids’ Pharmacokinetics after Controlled Smoked, Vaporized, and Oral Cannabis Administration in Frequent and Occasional Cannabis Users: Identification of Recent Cannabis Intake. Clin. Chem. 2016, 62, 1579–1592. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swortwood, M.J.; Newmeyer, M.N.; Andersson, M.; Abulseoud, O.A.; Scheidweiler, K.B.; Huestis, M.A. Cannabinoid Disposition in Oral Fluid after Controlled Smoked, Vaporized, and Oral Cannabis Administration. Drug Test. Anal. 2017, 9, 905–915. [Google Scholar] [CrossRef]
- Klawitter, J.; Sempio, C.; Mörlein, S.; De Bloois, E.; Klepacki, J.; Henthorn, T.; Leehey, M.A.; Hoffenberg, E.J.; Knupp, K.; Wang, G.S.; et al. An Atmospheric Pressure Chemical Ionization MS/MS Assay Using Online Extraction for the Analysis of 11 Cannabinoids and Metabolites in Human Plasma and Urine. Ther. Drug Monit. 2017, 39, 556–564. [Google Scholar] [CrossRef]
- Krämer, M.; Schäper, M.; Dücker, K.; Philipsen, A.; Losacker, M.; Dreimüller, N.; Engelmann, J.; Madea, B.; Hess, C. Detectability of Cannabinoids in the Serum Samples of Cannabis Users: Indicators of Recent Cannabis Use? A Follow-up Study. Drug Test. Anal. 2021, 13, 1614–1626. [Google Scholar] [CrossRef]
- Andersson, M.; Scheidweiler, K.B.; Sempio, C.; Barnes, A.J.; Huestis, M.A. Simultaneous Quantification of 11 Cannabinoids and Metabolites in Human Urine by Liquid Chromatography Tandem Mass Spectrometry Using WAX-S Tips. Anal. Bioanal. Chem. 2016, 408, 6461–6471. [Google Scholar] [CrossRef] [Green Version]
- Hidvégi, E.; Somogyi, G.P. Detection of Cannabigerol and Its Presumptive Metabolite in Human Urine after Cannabis Consumption. Pharm.-Int. J. Pharm. Sci. 2010, 65, 408–411. [Google Scholar] [CrossRef]
- Lah, T.; Novak, M.; Almidon, M.; Marinelli, O.; Baškovič, B.; Majc, B.; Mlinar, M.; Bošnjak, R.; Breznik, B.; Zomer, R.; et al. Cannabigerol Is a Potential Therapeutic Agent in a Novel Combined Therapy for Glioblastoma. Cells 2021, 10, 340. [Google Scholar] [CrossRef]
| CBG | CBGA | VCE-003 | VCE-003.2 | |
|---|---|---|---|---|
| Activity Area | ||||
| Receptors | Agonist: CB1, CB2, PPARγ, TRPA1, TRPV1, TRPV2, TRPV3, TRPV4, α2-adenoceptors | Agonist: CB1, CB2, PPARγ, TRPA1, TRPV1, TRPV2, TRPV3, TRPV4 | Agonist: CB1, CB2, PPARγ | Agonist: CB1, CB2, PPARγ |
| Antagonist: TRPM8, 5-HT1A | Antagonist: TRPM8 | |||
| Endocannabinoid system enzymes | Inhibition: FAAH, DAGLα, MAGL | Inhibition: FAAH, MAGL, DAGLα | Inhibition: MAGL | Inhibition: FAH, MAGL |
| Redox status and inflammation | Downregulation: TNFα, NFκB, IL-1ß, IL-6, INF-γ, PGE2 | Downregulation: TNFα, NFκB, IL-1ß, IL-6, INF-γ, PGE2 | Downregulation: TNFα, IL-1ß, IL-2, IL-6, IL-17, INF-γ, PGE2 | Downregulation: TNFα, NFκB, IL-1ß, IL-6, PGE2, Caspase 3 |
| Inhibition: iNOS, COX-1, COX-2, PLA2 | Inhibition: iNOS, COX-1, COX-2, PLA2 | Inhibition: iNOS, | Inhibition: iNOS, COX-2 | |
| Upregulation: Catalase, SOD-1 | Upregulation: Nrf2 | |||
| Biological activity | ||||
| antioxidant anti-inflammatory antibacterial neuromodulatory neuroprotective | antioxidant antibacterial neuromodulatory neuroprotective | anti-inflammatory neuromodulatory neuroprotective | anti-inflammatory neuromodulatory neuroprotective | |
| References | ||||
| [21,57,58,59,60,61,62,63,64] | [21,49,58,59,62,65] | [57,64,66,67,68,69,70] | [57,63,64,66,68] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jastrząb, A.; Jarocka-Karpowicz, I.; Skrzydlewska, E. The Origin and Biomedical Relevance of Cannabigerol. Int. J. Mol. Sci. 2022, 23, 7929. https://doi.org/10.3390/ijms23147929
Jastrząb A, Jarocka-Karpowicz I, Skrzydlewska E. The Origin and Biomedical Relevance of Cannabigerol. International Journal of Molecular Sciences. 2022; 23(14):7929. https://doi.org/10.3390/ijms23147929
Chicago/Turabian StyleJastrząb, Anna, Iwona Jarocka-Karpowicz, and Elżbieta Skrzydlewska. 2022. "The Origin and Biomedical Relevance of Cannabigerol" International Journal of Molecular Sciences 23, no. 14: 7929. https://doi.org/10.3390/ijms23147929
APA StyleJastrząb, A., Jarocka-Karpowicz, I., & Skrzydlewska, E. (2022). The Origin and Biomedical Relevance of Cannabigerol. International Journal of Molecular Sciences, 23(14), 7929. https://doi.org/10.3390/ijms23147929