Extracellular Vesicles in Atrial Fibrillation—State of the Art
Abstract
1. Introduction
2. Extracellular Vesicles in Patients with Atrial Fibrillation
2.1. Preclinical Studies
2.2. EVs in Atrial Fibrillation Patients Treated with Anticoagulants and Other Drugs
2.3. EVs Containing Nucleic Acids in Atrial Fibrillation Patients
2.4. EVs in Atrial Fibrillation Patients Undergoing Ablation or Other Invasive Procedures
2.5. Other Research Studies Concerning EVs in Atrial Fibrillation Patients
3. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomström-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.A.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur. Heart J. 2021, 42, 373–498. [Google Scholar] [CrossRef] [PubMed]
- Thrall, G.; Lane, D.; Carroll, D.; Lip, G.Y. Quality of life in patients with atrial fibrillation: A systematic review. Am. J. Med. 2006, 119, 448.e1–448.e19. [Google Scholar] [CrossRef] [PubMed]
- Odutayo, A.; Wong, C.X.; Hsiao, A.J.; Hopewell, S.; Altman, D.G.; Emdin, C.A. Atrial fibrillation and risks of cardiovascular disease, renal disease, and death: Systematic review and meta-analysis. BMJ 2016, 354, i4482. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das, S.R.; et al. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation 2019, 139, e56–e528. [Google Scholar] [CrossRef]
- Deng, H.; Bai, Y.; Shantsila, A.; Fauchier, L.; Potpara, T.S.; Lip, G.Y.H. Clinical scores for outcomes of rhythm control or arrhythmia progression in patients with atrial fibrillation: A systematic review. Clin. Res. Cardiol. 2017, 106, 813–823. [Google Scholar] [CrossRef]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef]
- Yáñez-Mó, M.; Siljander, P.R.; Andreu, Z.; Zavec, A.B.; Borràs, F.E.; Buzas, E.I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J.; et al. Biological properties of extracellular vesicles and their physiological functions. J. Extracell. Vesicles 2015, 4, 27066. [Google Scholar] [CrossRef]
- Mathieu, M.; Martin-Jaular, L.; Lavieu, G.; Théry, C. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat. Cell Biol. 2019, 21, 9–17. [Google Scholar] [CrossRef]
- Shah, R.; Patel, T.; Freedman, J.E. Circulating Extracellular Vesicles in Human Disease. N. Engl. J. Med. 2018, 379, 958–966. [Google Scholar] [CrossRef]
- Thulin, Å.; Christersson, C.; Alfredsson, J.; Siegbahn, A. Circulating cell-derived microparticles as biomarkers in cardiovascular disease. Biomark Med. 2016, 10, 1009–1022. [Google Scholar] [CrossRef]
- Voukalis, C.; Shantsila, E.; Lip, G.Y.H. Microparticles and cardiovascular diseases. Ann. Med. 2019, 51, 193–223. [Google Scholar] [CrossRef]
- Martins-Marques, T.; Girão, H. The good, the bad and the ugly: The impact of extracellular vesicles on the cardiovascular system. J. Physiol. 2022. [Google Scholar] [CrossRef]
- Jesel, L.; Abbas, M.; Toti, F.; Cohen, A.; Arentz, T.; Morel, O. Microparticles in atrial fibrillation: A link between cell activation or apoptosis, tissue remodelling and thrombogenicity. Int. J. Cardiol. 2013, 168, 660–669. [Google Scholar] [CrossRef]
- Chen, C.; Chen, Q.; Cheng, K.; Zou, T.; Pang, Y.; Ling, Y.; Xu, Y.; Zhu, W. Exosomes and Exosomal Non-coding RNAs Are Novel Promises for the Mechanism-Based Diagnosis and Treatments of Atrial Fibrillation. Front. Cardiovasc. Med. 2021, 8, 782451. [Google Scholar] [CrossRef]
- Huang, S.; Deng, Y.; Xu, J.; Liu, J.; Liu, L.; Fan, C. The Role of Exosomes and Their Cargos in the Mechanism, Diagnosis, and Treatment of Atrial Fibrillation. Front. Cardiovasc. Med. 2021, 8, 712828. [Google Scholar] [CrossRef]
- Xiang, K.; Akram, M.; Elbossaty, W.F.; Yang, J.; Fan, C. Exosomes in atrial fibrillation: Therapeutic potential and role as clinical biomarkers. Heart Fail. Rev. 2021, 27, 1211–1221. [Google Scholar] [CrossRef]
- Berezin, A.E.; Berezin, A.A. Extracellular Vesicles and Thrombogenicity in Atrial Fibrillation. Int. J. Mol. Sci. 2022, 23, 1774. [Google Scholar] [CrossRef]
- Li, S.; Gao, Y.; Liu, Y.; Li, J.; Yang, X.; Hu, R.; Liu, J.; Zhang, Y.; Zuo, K.; Li, K.; et al. Myofibroblast-Derived Exosomes Contribute to Development of a Susceptible Substrate for Atrial Fibrillation. Cardiology 2020, 145, 324–332. [Google Scholar] [CrossRef]
- Cao, F.; Li, Z.; Ding, W.; Yan, L.; Zhao, Q. Angiotensin II-Treated Cardiac Myocytes Regulate M1 Macrophage Polarization via Transferring Exosomal PVT1. J. Immunol. Res. 2021, 2021, 1994328. [Google Scholar] [CrossRef]
- Yao, Y.; He, S.; Wang, Y.; Cao, Z.; Liu, D.; Fu, Y.; Chen, H.; Wang, X.; Zhao, Q. Blockade of Exosome Release Suppresses Atrial Fibrillation by Alleviating Atrial Fibrosis in Canines With Prolonged Atrial Pacing. Front. Cardiovasc. Med. 2021, 8, 699175. [Google Scholar] [CrossRef]
- Yan, B.; Liu, T.; Yao, C.; Liu, X.; Du, Q.; Pan, L. LncRNA XIST shuttled by adipose tissue-derived mesenchymal stem cell-derived extracellular vesicles suppresses myocardial pyroptosis in atrial fibrillation by disrupting miR-214-3p-mediated Arl2 inhibition. Lab. Investig. 2021, 101, 1427–1438. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Fan, Y.; Wu, L.; Zhang, C.; Chu, M.; Wang, Y.; Zhuang, W. Exosomes from Bone Marrow Mesenchymal Stem Cells with Overexpressed Nrf2 Inhibit Cardiac Fibrosis in Rats with Atrial Fibrillation. Cardiovasc. Ther. 2022, 2022, 2687807. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Man, Y.; Chen, Z. microRNA-148a in Exosomes Derived from Bone Marrow Mesenchymal Stem Cells Alleviates Cardiomyocyte Apoptosis in Atrial Fibrillation by Inhibiting SMOC2. Mol. Biotechnol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Chirinos, J.A.; Castrellon, A.; Zambrano, J.P.; Jimenez, J.J.; Jy, W.; Horstman, L.L.; Willens, H.J.; Castellanos, A.; Myerburg, R.J.; Ahn, Y.S. Digoxin use is associated with increased platelet and endothelial cell activation in patients with nonvalvular atrial fibrillation. Heart Rhythm. 2005, 2, 525–529. [Google Scholar] [CrossRef]
- Lau, Y.C.; Xiong, Q.; Blann, A.D.; Lip, G.Y. Relationship between renal function and circulating microparticles, soluble P-selectin and E-selectin levels in atrial fibrillation. J. Thromb. Thrombolysis 2017, 43, 18–23. [Google Scholar] [CrossRef]
- Lenart-Migdalska, A.; Drabik, L.; Kaźnica-Wiatr, M.; Tomkiewicz-Pająk, L.; Podolec, P.; Olszowska, M. Flow Cytometric Assessment of Endothelial and Platelet Microparticles in Patients With Atrial Fibrillation Treated With Dabigatran. Clin. Appl. Thromb. Hemost. 2020, 26, 1076029620972467. [Google Scholar] [CrossRef]
- Lenart-Migdalska, A.; Drabik, L.; Kaźnica-Wiatr, M.; Tomkiewicz-Pająk, L.; Podolec, P.; Olszowska, M. Increased Levels of Platelets and Endothelial-Derived Microparticles in Patients With Non-Valvular Atrial Fibrillation During Rivaroxaban Therapy. Clin. Appl. Thromb. Hemost. 2021, 27, 10760296211019465. [Google Scholar] [CrossRef]
- Weiss, L.; Keaney, J.; Szklanna, P.B.; Prendiville, T.; Uhrig, W.; Wynne, K.; Kelliher, S.; Ewins, K.; Comer, S.P.; Egan, K.; et al. Nonvalvular atrial fibrillation patients anticoagulated with rivaroxaban compared with warfarin exhibit reduced circulating extracellular vesicles with attenuated pro-inflammatory protein signatures. J. Thromb. Haemost. 2021, 19, 2583–2595. [Google Scholar] [CrossRef]
- Duarte, R.C.F.; Rios, D.R.A.; Figueiredo, E.L.; Caiaffa, J.R.S.; Silveira, F.R.; Lanna, R.; Alves, L.C.V.; Martins, G.L.; Reis, H.J.; Reis, E.A.; et al. Thrombin Generation and other hemostatic parameters in patients with atrial fibrillation in use of warfarin or rivaroxaban. J. Thromb. Thrombolysis 2021, 51, 47–57. [Google Scholar] [CrossRef]
- Wang, S.; Min, J.; Yu, Y.; Yin, L.; Wang, Q.; Shen, H.; Yang, J.; Zhang, P.; Xiao, J.; Wang, Z. Differentially expressed miRNAs in circulating exosomes between atrial fibrillation and sinus rhythm. J. Thorac. Dis. 2019, 11, 4337–4348. [Google Scholar] [CrossRef]
- Mun, D.; Kim, H.; Kang, J.Y.; Park, H.; Park, H.; Lee, S.H.; Yun, N.; Joung, B. Expression of miRNAs in circulating exosomes derived from patients with persistent atrial fibrillation. FASEB J. 2019, 33, 5979–5989. [Google Scholar] [CrossRef]
- Soltész, B.; Urbancsek, R.; Pös, O.; Hajas, O.; Forgács, I.N.; Szilágyi, E.; Nagy-Baló, E.; Szemes, T.; Csanádi, Z.; Nagy, B. Quantification of peripheral whole blood, cell-free plasma and exosome encapsulated mitochondrial DNA copy numbers in patients with atrial fibrillation. J. Biotechnol. 2019, 299, 66–71. [Google Scholar] [CrossRef]
- Wei, Z.; Bing, Z.; Shaohuan, Q.; Yanran, W.; Shuo, S.; Bi, T.; Feiyu, Z.; Heng, Z.; Qin, G.; Pinfang, K. Expression of miRNAs in plasma exosomes derived from patients with atrial fibrillation. Clin. Cardiol. 2020, 43, 1450–1459. [Google Scholar] [CrossRef]
- Liu, L.; Chen, Y.; Shu, J.; Tang, C.E.; Jiang, Y.; Luo, F. Identification of microRNAs enriched in exosomes in human pericardial fluid of patients with atrial fibrillation based on bioinformatic analysis. J. Thorac. Dis. 2020, 12, 5617–5627. [Google Scholar] [CrossRef]
- Wang, S.; Li, L.; Hu, X.; Liu, T.; Jiang, W.; Wu, R.; Ren, Y.; Wang, M. Effects of Atrial Fibrillation-Derived Exosome Delivery of miR-107 to Human Umbilical Vein Endothelial Cells. DNA Cell Biol. 2021, 40, 568–579. [Google Scholar] [CrossRef]
- Liu, L.; Luo, F.; Lei, K. Exosomes Containing LINC00636 Inhibit MAPK1 through the miR-450a-2-3p Overexpression in Human Pericardial Fluid and Improve Cardiac Fibrosis in Patients with Atrial Fibrillation. Mediat. Inflamm. 2021, 2021, 9960241. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, X.; Li, H.; Li, Y.; Cheng, D.; Tang, Y.; Sang, H. Serum extracellular vesicles containing MIAT induces atrial fibrosis, inflammation and oxidative stress to promote atrial remodeling and atrial fibrillation via blockade of miR-485-5p-mediated CXCL10 inhibition. Clin. Transl. Med. 2021, 11, e482. [Google Scholar] [CrossRef]
- Siwaponanan, P.; Kaewkumdee, P.; Phromawan, W.; Udompunturak, S.; Chomanee, N.; Udol, K.; Pattanapanyasat, K.; Krittayaphong, R. Increased expression of six-large extracellular vesicle-derived miRNAs signature for nonvalvular atrial fibrillation. J. Transl. Med. 2022, 20, 4. [Google Scholar] [CrossRef]
- Zhu, P.; Li, H.; Zhang, A.; Li, Z.; Zhang, Y.; Ren, M.; Zhang, Y.; Hou, Y. MicroRNAs sequencing of plasma exosomes derived from patients with atrial fibrillation: miR-124-3p promotes cardiac fibroblast activation and proliferation by regulating AXIN1. J. Physiol. Biochem. 2022, 78, 85–98. [Google Scholar] [CrossRef]
- Mun, D.; Kim, H.; Kang, J.Y.; Yun, N.; Youn, Y.N.; Joung, B. Small extracellular vesicles derived from patients with persistent atrial fibrillation exacerbate arrhythmogenesis via miR-30a-5p. Clin. Sci. 2022, 136, 621–637. [Google Scholar] [CrossRef]
- Herrera Siklódy, C.; Arentz, T.; Minners, J.; Jesel, L.; Stratz, C.; Valina, C.M.; Weber, R.; Kalusche, D.; Toti, F.; Morel, O.; et al. Cellular damage, platelet activation, and inflammatory response after pulmonary vein isolation: A randomized study comparing radiofrequency ablation with cryoablation. Heart Rhythm. 2012, 9, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Jesel, L.; Arentz, T.; Herrera-Siklody, C.; Trenk, D.; Zobairi, F.; Abbas, M.; Weber, R.; Minners, J.; Toti, F.; Morel, O. Do atrial differences in endothelial damage, leukocyte and platelet activation, or tissue factor activity contribute to chamber-specific thrombogenic status in patients with atrial fibrillation? J. Cardiovasc. Electrophysiol. 2014, 25, 266–270. [Google Scholar] [CrossRef] [PubMed]
- Liles, J.; Liles, J.; Wanderling, C.; Syed, M.; Hoppensteadt, D.; Fareed, J. Increased Level of Thrombotic Biomarkers in Patients with Atrial Fibrillation Despite Traditional and New Anticoagulant Therapy. Clin. Appl. Thromb. Hemost. 2016, 22, 743–748. [Google Scholar] [CrossRef] [PubMed]
- Pourtau, L.; Sellal, J.M.; Lacroix, R.; Poncelet, P.; Bernus, O.; Clofent-Sanchez, G.; Hocini, M.; Haïssaguerre, M.; Dignat-George, F.; Sacher, F.; et al. Platelet function and microparticle levels in atrial fibrillation: Changes during the acute episode. Int. J. Cardiol. 2017, 243, 216–222. [Google Scholar] [CrossRef]
- Meng, H.; Kou, J.; Ma, R.; Ding, W.; Kou, Y.; Cao, M.; Dong, Z.; Bi, Y.; Thatte, H.S.; Shi, J. Prognostic implications and procoagulant activity of phosphatidylserine exposure of blood cells and microparticles in patients with atrial fibrillation treated with pulmonary vein isolation. Mol. Med. Rep. 2017, 16, 8579–8588. [Google Scholar] [CrossRef]
- Mørk, M.; Andreasen, J.J.; Rasmussen, L.H.; Lip, G.Y.H.; Pedersen, S.; Bæk, R.; Jørgensen, M.M.; Kristensen, S.R. Elevated blood plasma levels of tissue factor-bearing extracellular vesicles in patients with atrial fibrillation. Thromb. Res. 2019, 173, 141–150. [Google Scholar] [CrossRef]
- Amabile, N.; Bagdadi, I.; Armero, S.; Elhadad, S.; Sebag, F.; Landolff, Q.; Saby, L.; Mechulan, A.; Boulanger, C.M.; Caussin, C. Impact of left atrial appendage closure on circulating microvesicles levels: The MICROPLUG study. Int. J. Cardiol. 2020, 307, 24–30. [Google Scholar] [CrossRef]
- Shaihov-Teper, O.; Ram, E.; Ballan, N.; Brzezinski, R.Y.; Naftali-Shani, N.; Masoud, R.; Ziv, T.; Lewis, N.; Schary, Y.; Levin-Kotler, L.P.; et al. Extracellular Vesicles From Epicardial Fat Facilitate Atrial Fibrillation. Circulation 2021, 143, 2475–2493. [Google Scholar] [CrossRef]
- Choudhury, A.; Chung, I.; Blann, A.D.; Lip, G.Y.H. Elevated platelet microparticle levels in nonvalvular atrial fibrillation: Relationship to p-selectin and antithrombotic therapy. Chest 2007, 131, 809–815. [Google Scholar] [CrossRef]
- Ederhy, S.; Di Angelantonio, E.; Mallat, Z.; Hugel, B.; Janower, S.; Meuleman, C.; Boccara, F.; Freyssinet, J.M.; Tedgui, A.; Cohen, A. Levels of circulating procoagulant microparticles in nonvalvular atrial fibrillation. Am. J. Cardiol. 2007, 100, 989–994. [Google Scholar] [CrossRef]
- Azzam, H.; Zagloul, M. Elevated platelet microparticle levels in valvular atrial fibrillation. Hematology 2009, 14, 357–360. [Google Scholar] [CrossRef]
- Wang, H.; Yan, H.M.; Tang, M.X.; Wang, Z.H.; Zhong, M.; Zhang, Y.; Deng, J.T.; Zhang, W. Increased serum levels of microvesicles in nonvalvular atrial fibrillation determinated by ELISA using a specific monoclonal antibody AD-1. Clin. Chim. Acta 2010, 411, 1700–1704. [Google Scholar] [CrossRef]
- Hayashi, M.; Takeshita, K.; Inden, Y.; Ishii, H.; Cheng, X.W.; Yamamoto, K.; Murohara, T. Platelet activation and induction of tissue factor in acute and chronic atrial fibrillation: Involvement of mononuclear cell-platelet interaction. Thromb. Res. 2011, 128, e113–e118. [Google Scholar] [CrossRef]
- Idriss, N.K.; Blann, A.D.; Sayed, D.M.; Gaber, M.A.; Hassen, H.A.; Kishk, Y.T. Circulating Endothelial Cells and Platelet Microparticles in Mitral Valve Disease With and Without Atrial Fibrillation. Angiology 2015, 66, 631–637. [Google Scholar] [CrossRef]
- Wang, L.; Bi, Y.; Yu, M.; Li, T.; Tong, D.; Yang, X.; Zhang, C.; Guo, L.; Wang, C.; Kou, Y.; et al. Phosphatidylserine-exposing blood cells and microparticles induce procoagulant activity in non-valvular atrial fibrillation. Int. J. Cardiol. 2018, 258, 138–143. [Google Scholar] [CrossRef]
- Siwaponanan, P.; Keawvichit, R.; Udompunturak, S.; Hunnangkul, S.; Reesukumal, K.; Sukapirom, K.; Pattanapanyasat, K.; Krittayaphong, R. Altered profile of circulating microparticles in nonvalvular atrial fibrillation. Clin. Cardiol. 2019, 42, 425–431. [Google Scholar] [CrossRef]
- Thulin, Å.; Lindbäck, J.; Granger, C.B.; Wallentin, L.; Lind, L.; Siegbahn, A. Extracellular vesicles in atrial fibrillation and stroke. Thromb. Res. 2020, 193, 180–189. [Google Scholar] [CrossRef]
- Wang, H.; Song, N.P.; Li, J.P.; Wang, Z.H.; Ti, Y.; Li, Y.H.; Zhang, W.; Zhong, M. The microvesicle/CD36 complex triggers a prothrombotic phenotype in patients with non-valvular atrial fibrillation. J. Cell. Mol. Med. 2020, 24, 7331–7340. [Google Scholar] [CrossRef]
- Voukalis, C.; Lip, G.Y.H.; Shantsila, E. Effects of antithrombotic drugs on the prothrombotic state in patients with atrial fibrillation: The west Birmingham atrial fibrillation project. Thromb. Res. 2021, 200, 149–155. [Google Scholar] [CrossRef]
- Ni, H.; Pan, W.; Jin, Q.; Xie, Y.; Zhang, N.; Chen, K.; Lin, T.; Lin, C.; Xie, Y.; Wu, J.; et al. Label-free proteomic analysis of serum exosomes from paroxysmal atrial fibrillation patients. Clin. Proteom. 2021, 18, 1. [Google Scholar] [CrossRef]
- Zietzer, A.; Al-Kassou, B.; Jamme, P.; Rolfes, V.; Steffen, E.; Bulic, M.; Hosen, M.R.; Goody, P.R.; Tiyerili, V.; Zimmer, S.; et al. Large extracellular vesicles in the left atrial appendage in patients with atrial fibrillation-the missing link? Clin. Res. Cardiol. 2022, 111, 34–49. [Google Scholar] [CrossRef]
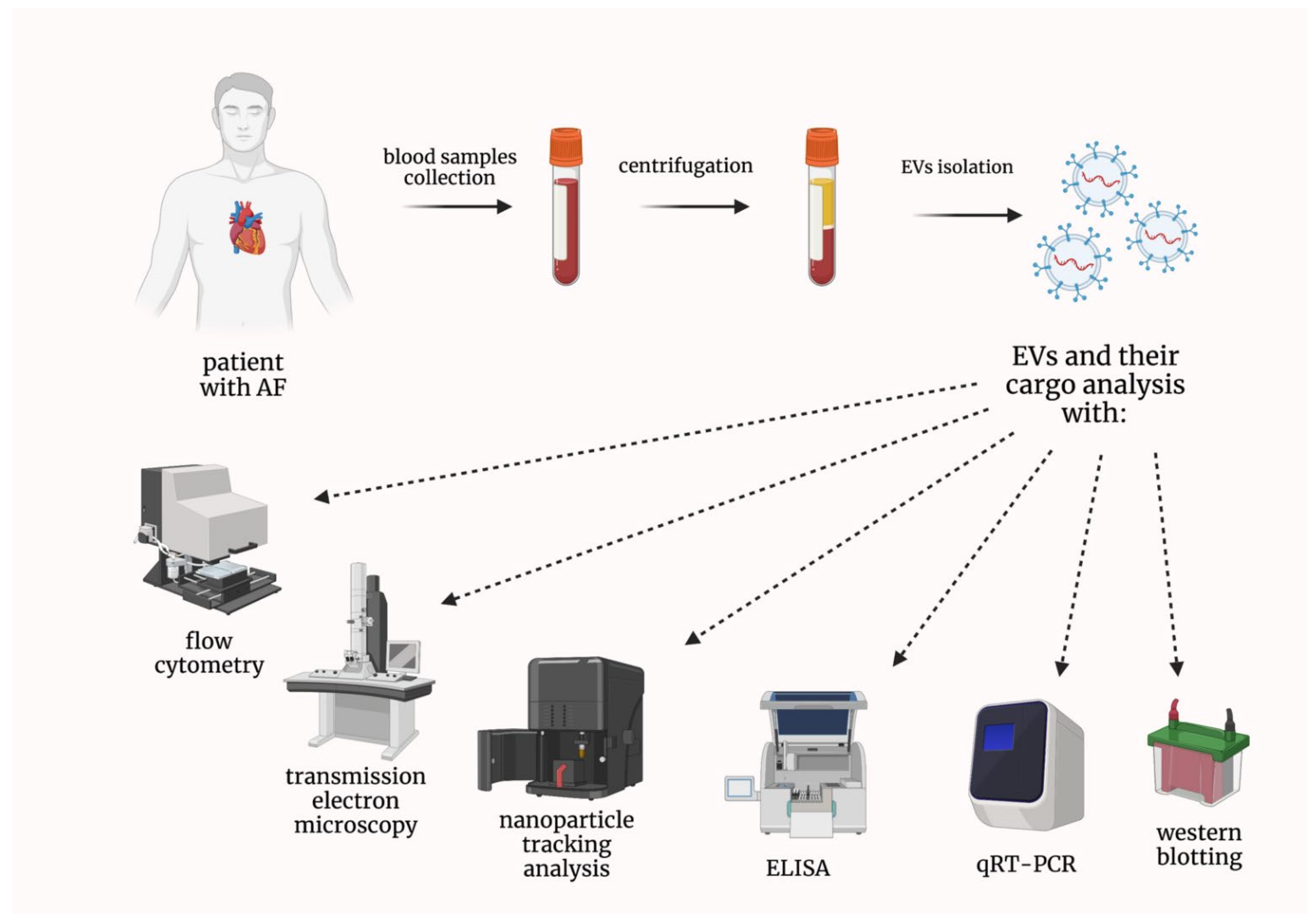
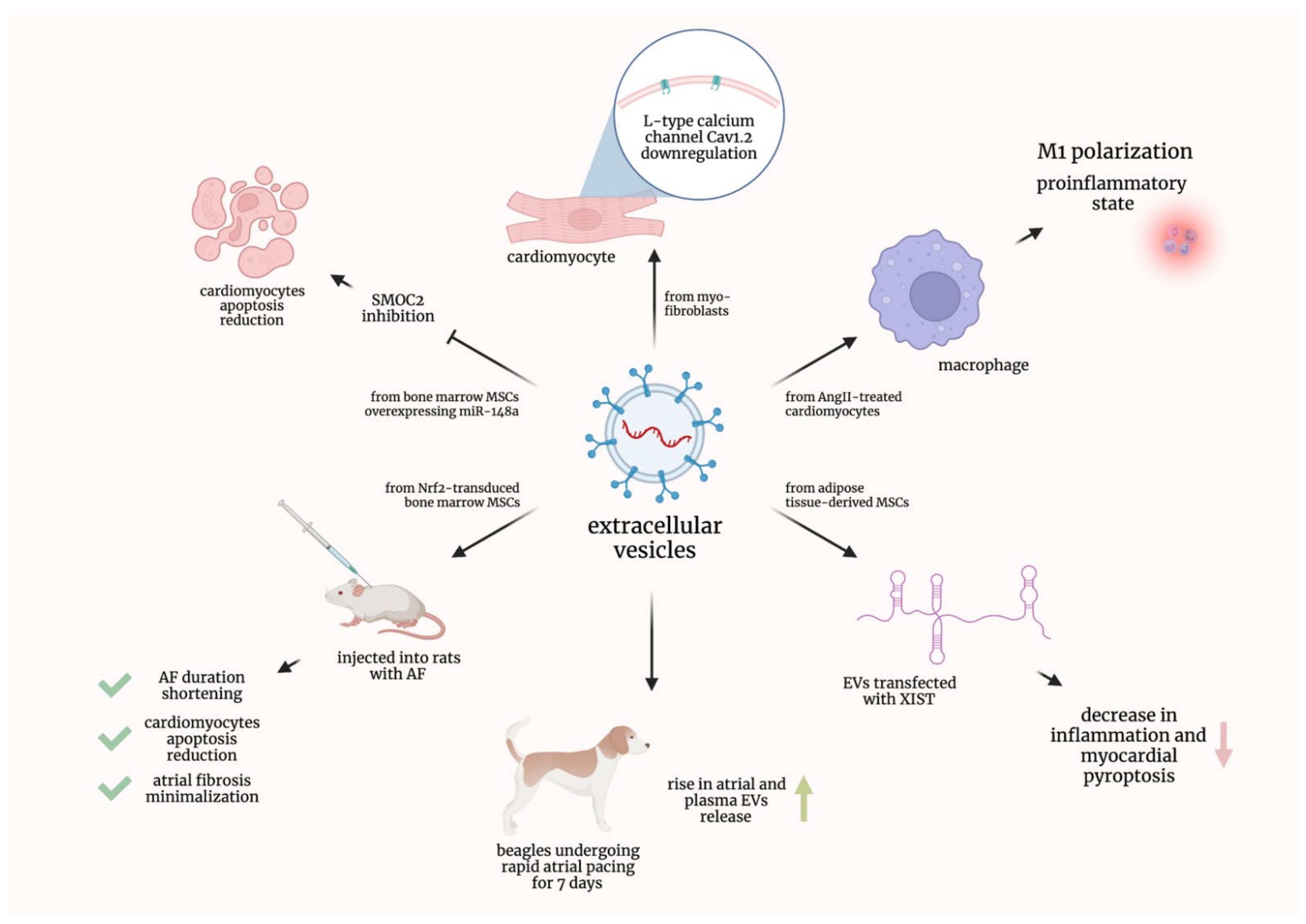
| Ref. | Population | EVs’ Origin | Comparison | Outcomes | Method |
|---|---|---|---|---|---|
| [24] | 30 NV AF pts | platelets endothelial cells | pts taking digoxin vs. pts not taking digoxin | platelet EVs not significantly altered ↑ endothelial EVs in AF pts taking digoxin | EVs measured in PPP with fluorescent mAbs |
| [25] | 160 NV AF pts treated with warfarin | platelets mixed from endothelial cells and platelets (EPEVs) | correlations with different variables | EPEVs correlated negatively with eGFR no correlations btw platelet-derived EVs and eGFR | EVs measured in PFP by FC |
| [26] | 39 NV AF pts treated with dabigatran | platelets endothelial cells | correlations with dabigatran intake | ↑ platelet EVs after taking dabigatran change in platelet EVs correlated negatively with dabigatran concentration endothelial EVs unaltered by dabigatran | EVs measured by FC |
| [27] | 34 NV AF pts treated with rivaroxaban | platelets endothelial cells | correlations with rivaroxaban intake | ↑ platelet and endothelial EVs after rivaroxaban administration | EVs measured in PFP by FC |
| [28] | 8 NV AF pts treated with rivaroxaban | circulating EVs | 15 NV AF control pts treated with warfarin | EVs of pts treated with rivaroxaban: ↓ proinflammatory proteins and complement factors ↑ negative regulators of inflammatory pathways | EVs examined by NTA, FC, and proteomics |
| [29] | 78 NV AF pts (39 pts on rivaroxaban + 39 pts on warfarin) | platelets endothelial cells | 42 control subjects in SR | ↑ platelet-derived EVs in AF pts no differences in endothelial-derived EVs | EVs measured in PPP by FC |
| Ref. | Population | EV Content | Comparison | Outcomes | Method |
|---|---|---|---|---|---|
| [30] | 40 NV persistent AF pts | miRs | 20 controls in SR | ↑miR-483-5p in AF pts ↓ miR-142-5p, miR-223-3p, miR-223-5p in AF pts miR-483-5p, miR-142-5p, miR-223-3p related with AF (univariate analysis) miR-483-5p independently correlated with AF (multivariate analysis) | EVs examined by TEM and NTA miRs levels by qRT-PCR |
| [31] | 40 persistent AF pts | miRs | 20 controls with SVT | ↑ miR-107, miR-320d, miR-103a-3p, miR-486-5p, and let-7b-5p in AF pts | EVs examined by TEM and NTA miRs levels by qRT-PCR |
| [32] | 60 AF pts | mtDNA | 72 healthy controls | no differences in EV-encapsulated mtDNA copy numbers between AF pts and controls | EVs from cell-free plasma mtDNA copy number by qRT-PCR |
| [33] | 20 AF pts | miRs | 20 pts in SR | ↑ miR-92b-3p, miR-1306-5p, and miR-let-7b-3p in AF pts | plasma-derived EVs miRs levels by qRT-PCR |
| [34] | 60 adult pts with CHD and persistent AF | miRs | 60 adult pts with CHD in SR | ↑ miR-3126-5p in AF pts ↓ miR-382-3p and miR-450a-2-3p in AF pts | EVs from pericardial fluid miRs levels by qRT-PCR |
| [35] | AF pts | miRs | healthy controls | ↑ miR-107 in AF pts | miRs levels by qRT-PCR |
| [36] | 12 AF pts | RNAs | 12 non-AF pts | ↓ LINC00636 and miR-450a-2-3p in AF pts | EVs from pericardial fluid RNAs levels by qRT-PCR |
| [37] | 20 AF pts | DNA | 20 healthy individuals | ↑ MIAT expression in EVs in AF pts | EVs examined by TEM, NTA, and Western blots qRT-PCR |
| [38] | 30 AF pts | miRs | 30 control subjects in SR | ↑ miR-106b-3p, miR-590-5p, miR-339-3p, miR-378-3p, miR-328-3p, and miR-532-3p in AF pts | EVs examined by FC, TEM, NTA, and Western blots miRs levels by qRT-PCR |
| [39] | 40 AF pts | miRs | 40 pts in SR | ↑ miR-124-3p, miR-378d, miR-2110, and miR-3180-3p in AF pts ↓ miR-223-5p, miR-574-3p, miR-125a-3p, and miR-1299 in AF pts | EVs examined by TEM, NTA, and Western blots miRs levels by qRT-PCR |
| Ref. | Population | EVs Origin | Comparison | Outcomes | Methods |
|---|---|---|---|---|---|
| [41] | 60 AF pts undergoing ablation | procoagulant EVs from leukocytes, endothelial cells, platelets | cryoablation: 30 AF pts RF ablation: 30 AF pts | both groups: release of procoagulant EVs (platelet and leukocyte, but not endothelial) no significant differences btw groups | EVs measured in PPP PS content measured by FPA |
| [42] | 22 AF pts undergoing ablation | procoagulant EVs from leukocytes, endothelial cells, platelets | comparison of EV levels btw right and left atria | ↑ endothelial EVs in the right atrium no differences in total, platelet, nor leukocyte EVs | EVs measured in PPP collected in right and left atria PS content measured by FPA |
| [43] | 30 NV AF pts prior to ablation | TF-EVs | Healthy controls NV AF pts divided into groups according to ACT | ↑ TF-EVs in AF pts compared with healthy controls ↑ TF-EVs in AF pts on ACT compared with AF pts not on ACT no differences btw AF pts on warfarin and AF pts on apixaban/rivaroxaban | TF-EVs measured in plasma by ELISA |
| [44] | 37 AF pts undergoing ablation (21 paroxysmal + 16 persistent) | fibrinolytic and TF-dependent PCA of EVs | 11 healthy controls 10 AF pts in SR subjected to induced AF | ↓ fibrinolytic EVs in paroxysmal AF pts compared with controls ↓ procoagulant EVs in both paroxysmal and persistent AF pts compared with controls ↓ PCA after 20 min of induced AF (unaltered fibrinolytic activity) | measurements in PFP fluorogenic assay of factor Xa chromogenic test of plasmin |
| [45] | 56 AF pts undergoing PVI ablation | platelet, leukocyte, endothelial-cell, erythrocyte, TF-EVs | 40 healthy controls | endothelial EVs > 355/μL as independent predictor of ERAF ↑ EVs of all types except erythrocyte EVs in AF pts compared with controls | EVs measured in PFP by FC |
| [46] | 13 NV AF pts undergoing elective AVS or CABG | AnnV-positive EVs TF-EVs | 12 patients in SR undergoing elective AVS or CABG | ↑ total EVs in AF patients ↑ TF-EVs in AF patients | plasma EVs measured using protein microarray platform |
| [47] | 25 NV AF pts treated with LAA occlusion | platelet, endothelial-cell, leukocyte, erythrocyte, AnnV-positive EVs | 25 control patients who underwent coronary angiography | ↑ AnnV-positive, platelet, erythrocyte, and leukocyte EVs after LAA occlusion ↑ AnnV-positive in controls after angiography | EVs measured in PFP by FC |
| [48] | 30 AF pts undergoing elective heart surgery | epicardial-fat-derived EVs | 32 pts in SR undergoing elective heart surgery | ↑ EVs (with proinflammatory and profibrotic cytokines and profibrotic miR) in cultured explants from AF pts | EVs analyzed by FC, NTA, ELISA of organ cultures |
| Ref. | Population | EVs Origin | Comparison | Outcomes | Methods |
|---|---|---|---|---|---|
| [49] | 70 NV AF pts (35 paroxysmal + 35 permanent) | platelets | 46 pts in SR + 33 healthy people in SR | ↑ EVs in AF pts and SR pts compared with healthy SR | EVs measured in PPP by FC |
| [50] | 45 NV AF pts (permanent or persistent) | platelet, endothelial-cell, AnnV-positive EVs | 45 control subjects with CV RFs + 45 control subjects without CV RFs | ↑ AnnV-positive EVs in AF pts ↑ platelet and endothelial EVs in both AF pts and control subjects with CV RFs compared with control subjects without CV RFs | EVs measured in PPP wells coated with antibodies |
| [51] | 20 valvular AF pts with chronic rheumatic MS | platelets | 10 healthy volunteers in SR | ↑ platelet EVs in AF pts correlation btw the severity of MS and EV levels | EVs measured by FC |
| [52] | 53 NV AF pts (37 persistent + 16 paroxysmal) | EVs captured with mAbs | 37 normal controls | ↑ EVs in persistent AF pts compared with normal controls and paroxysmal AF pts | EVs measured in serum amount of EVs by LAP activity |
| [53] | 28 NV AF pts | EVs expressing P-selectin | 13 control subjects | ↑ EVs in paroxysmal AF pts after induction of AF no differences in EVs level btw chronic AF pts and control subjects | EVs measured in PRP by FC |
| [54] | 21 AF MVD pts | EVs with positive binding to anti CD41-a | 20 healthy controls 24 MVD pts in SR | ↑ EVs in both MVD pts groups compared with healthy controls (no differences btw MVD pts groups) | EVs measured in PPP by FC as percentage of total platelet count |
| [55] | 78 NV AF pts (18 with left atrial thrombi and 60 without) | PS-positive EVs from platelets, leukocytes, erythrocytes, endothelial cells | 36 matched controls | ↑ EVs in both groups of NV AF pts compared with controls ↑ EVs in pts with left atrial thrombi compared with pts without thrombi PCA inhibition by blockade of exposed PS on EVs with lactadherin positive correlation btw thrombus diameter and amount of EVs | EVs measured by FC PCA evaluated with clotting time, factor Xa, thrombin, and fibrin formation |
| [56] | 66 NV AF pts (31 permanent + 28 paroxysmal + 7 persistent) | platelet, leukocyte, endothelial-cell, erythrocyte, and PS-positive EVs, TF-EVs | 33 healthy individuals | ↑ total EVs and EVs from platelets and endothelial cells in AF pts compared with controls | EVs measured in centrifuged serum by FC |
| [57] | 836 AF pts (of which 280 pts with ischemic stroke or systemic embolism) | platelet, leukocyte, erythrocyte, endothelial-cell, PS-positive EVs | 1007 randomly selected 70 yo control individuals | ↑ EVs (of all origins except endothelial derived) in AF pts compared with control individuals similar EV levels among all AF pts (stroke cases vs. others) | EVs measured by FC and SP-PLA |
| [58] | 210 NV AF pts (paroxysmal or persistent) | platelets AnnV-positive | 35 healthy controls pts divided in “low to moderate risk” or “high risk” of stroke | ↑ AnnV-positive and platelet EVs in pts at “high risk” EVs from NV AF bound to platelet CD36 and activated platelets | EVs measured in PFP by ELISA |
| [59] | 47 AF pts | platelets AnnV-positive | 39 IHD pts in SR | ↑ AnnV-positive EVs Similar levels of platelet-derived EVs | EVs measured in PPP by FC |
| [60] | 15 paroxysmal AF pts | CD9- and CD63-positive EVs | 15 healthy donors | altered expression of proteins involved in anticoagulation, complement system, and protein folding | EVs examined with TEM and Western blots quantitative proteomic analysis |
| [61] | 48 AF pts (27 permanent AF + 21 non-permanent AF) | large EVs from platelets | 10 pts with no AF | ↑ platelet-derived EVs in AF pts ↑ platelet-derived EVs in permanent AF compared with non-permanent AF | PFP from LAA EVs examined by FC, TEM, NTA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Procyk, G.; Bilicki, D.; Balsam, P.; Lodziński, P.; Grabowski, M.; Gąsecka, A. Extracellular Vesicles in Atrial Fibrillation—State of the Art. Int. J. Mol. Sci. 2022, 23, 7591. https://doi.org/10.3390/ijms23147591
Procyk G, Bilicki D, Balsam P, Lodziński P, Grabowski M, Gąsecka A. Extracellular Vesicles in Atrial Fibrillation—State of the Art. International Journal of Molecular Sciences. 2022; 23(14):7591. https://doi.org/10.3390/ijms23147591
Chicago/Turabian StyleProcyk, Grzegorz, Dominik Bilicki, Paweł Balsam, Piotr Lodziński, Marcin Grabowski, and Aleksandra Gąsecka. 2022. "Extracellular Vesicles in Atrial Fibrillation—State of the Art" International Journal of Molecular Sciences 23, no. 14: 7591. https://doi.org/10.3390/ijms23147591
APA StyleProcyk, G., Bilicki, D., Balsam, P., Lodziński, P., Grabowski, M., & Gąsecka, A. (2022). Extracellular Vesicles in Atrial Fibrillation—State of the Art. International Journal of Molecular Sciences, 23(14), 7591. https://doi.org/10.3390/ijms23147591








