Probing the Skin–Brain Axis: New Vistas Using Mouse Models
Abstract
1. Introduction–Skin Disease and Mental Health
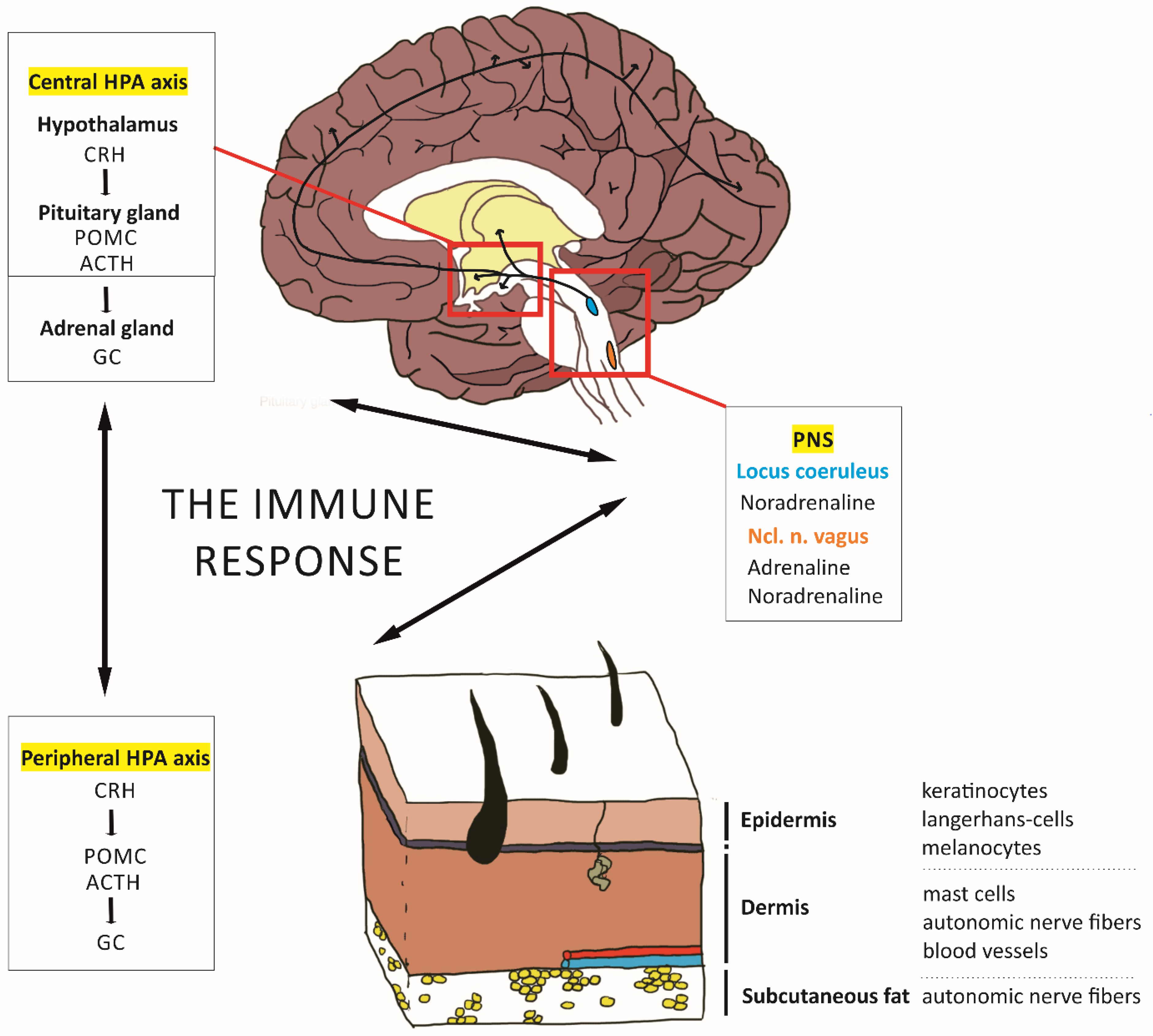
2. Modeling the Skin–Brain Axis
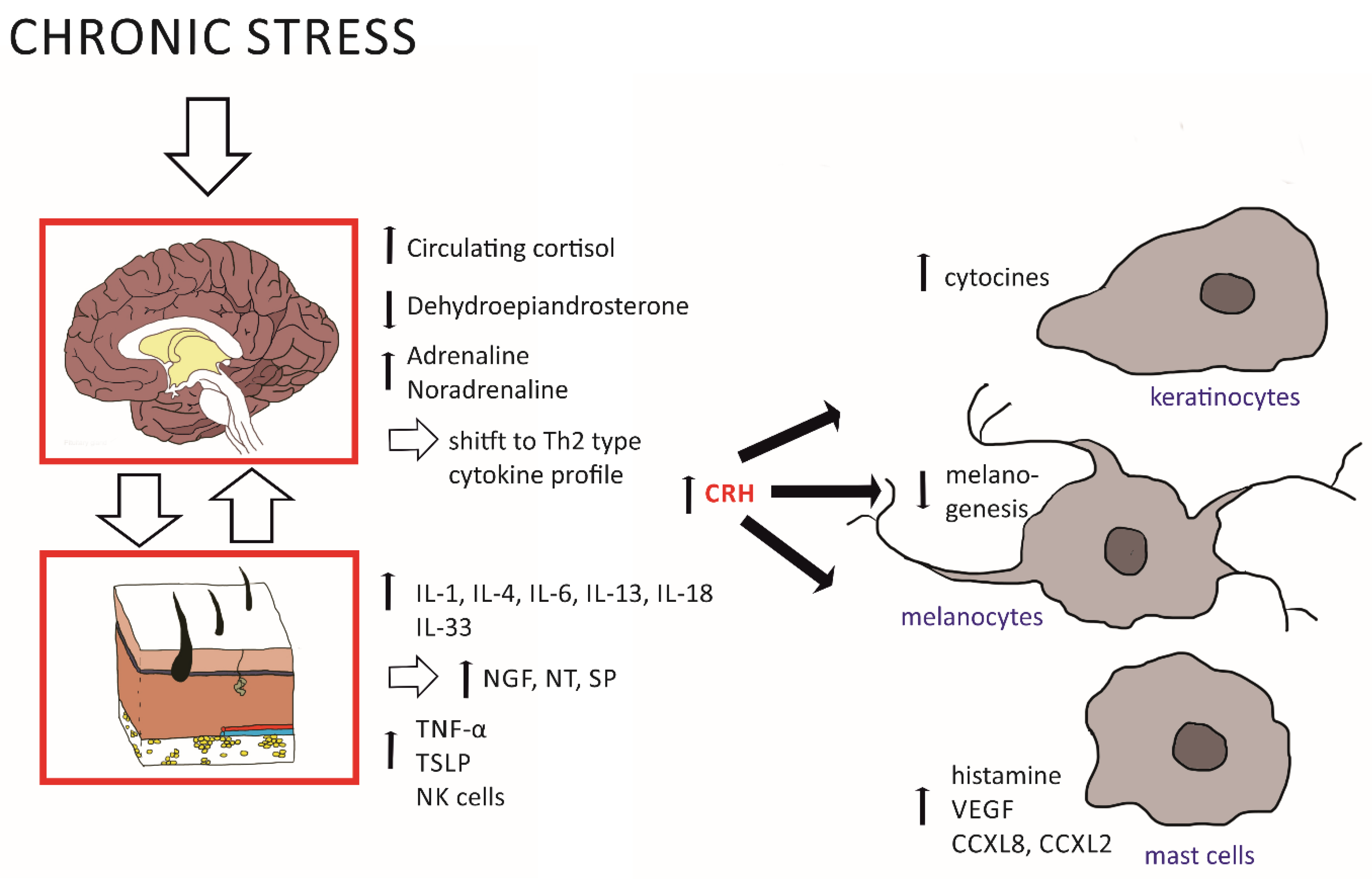
2.1. Effects of the HPA Axis on Skin Inflammation
2.2. Effects of Peripheral Inflammation on Brain Plasticity and Stress-Related Behavioral Domains
| Active Substance | Site of Action | Function | Reference |
|---|---|---|---|
| IL-4 | Astrocytes, neurons | ↓ POMC expression; induces differentiation of CD4- into Th2 cells; macrophage activation | [2,32] |
| IL-13 | Blood | Macrophage activation | [32] |
| TNF-α | Astrocytes, neurons | Astrocytic Ca2+ levels ↓ Glutamatergic exocytotic vesicles at the synapse p38–MAPK, ERK, JNK * pathways ↓ Spine size ↓ Glutamate receptors of the AMPA subtype | [2,30] |
| TSLP ***** | Blood, skin | ↑ Lymphoid cell response; CD4- T-cell polarization into Th2 cells | [33] |
| IL-1β | Neurons | Production of NGF **; BDNF *** release activation of the tropomyosin receptor kinase B (TrkB)–ERK pathway High level: ↓ LTP, spatial and working memory | [30,34,35] |
| IL-6 | Neurons blood | ERK1/2 pathway High levels: ↓ LTD, ↓ memory, ↓ functional brain connectivity mPFC–striatum ↓ Functional brain connectivity mPFC–amygdala | [30] |
| IL-18 | Blood, skin | ↑ Th2 cytokines | [33] |
| IL-33 | Blood/blood vessels | ↑ Vascular endothelial growth factor (VEGF) release | [24] |
| NT **** | Blood/skin | ↑ Histamine release from mast cells | [24] |
| CRH | Skin, blood, neurons, | ↑ Activates mast cells ↑ HPA axis: POMC/ACTH/glucocorticoid levels | [24,25,36] |
| SP | Blood/skin | ↑ Activates mast cells, ↑ Histamine, cytokines, NGF | [24] |
| VEGF | Blood/skin | Maturation of dendritic cells | [37] |
2.3. Influences of a Neglected Regulatory System: The Microbiome
3. Tools for Probing the Skin–Brain Axis
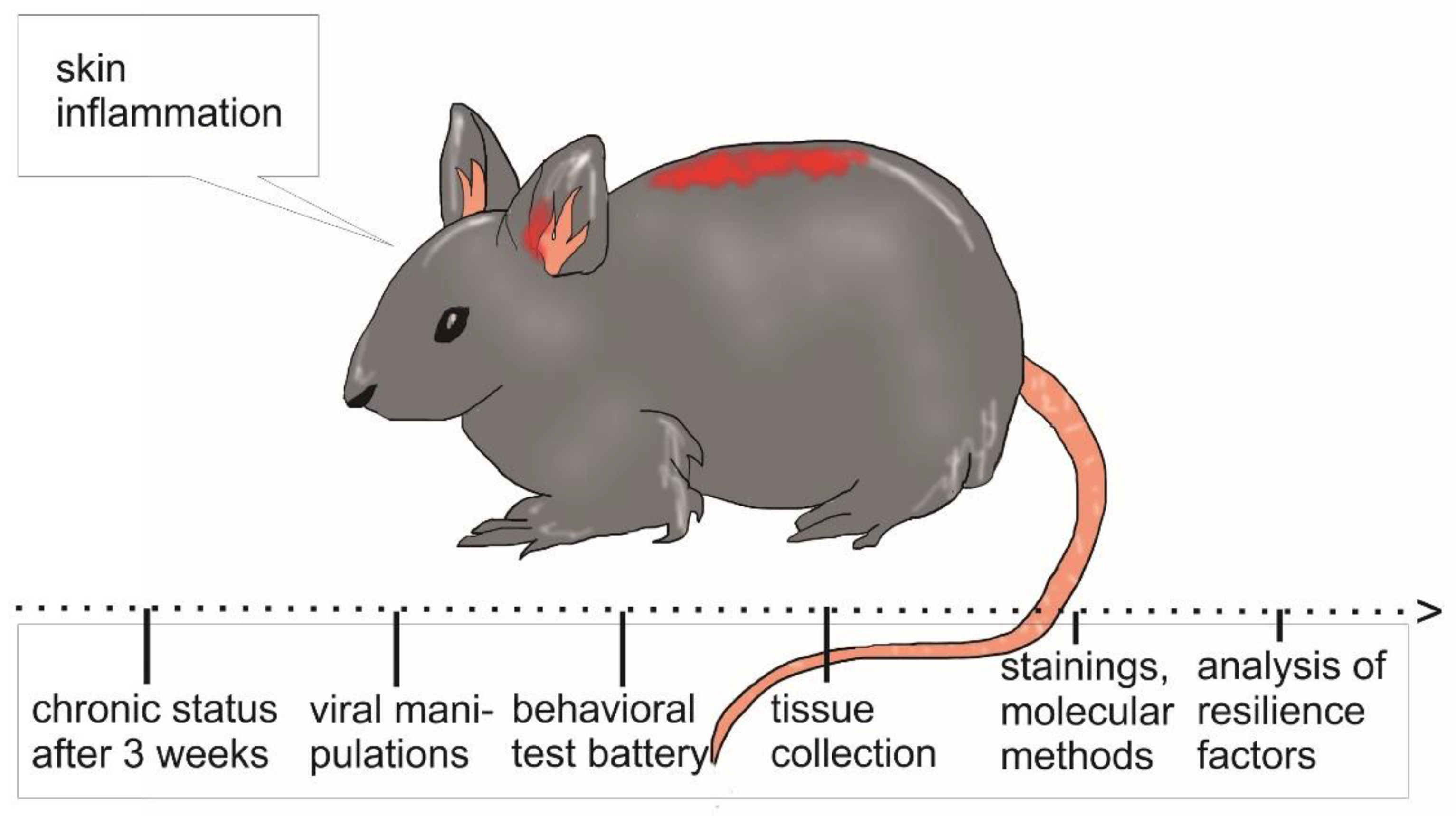
3.1. Mouse Models for Chronic Skin Inflammation
3.2. Translational Testing of Neuropsychopathologies in Mice
3.2.1. Basic Characterization of the Neurological Status
3.2.2. Anxiety Testing
3.2.3. Depression
3.2.4. Addiction
3.2.5. Learning and Memory
4. Interventional Approaches and Translational Relevance for Probing the Skin–Brain Axis
4.1. Interventional Approaches
4.2. Translational Outlook
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| ACTH | adrenocorticotropic hormone |
| AI | artificial intelligence |
| AD | atopic dermatitis |
| ADHD | attention deficit hyperactivity disorder |
| BLMH | bleomycin hydrolase |
| BAM | border-associated macrophage |
| BDNF | brain-derived neurotrophic factor |
| JNK | c-Jun N-terminal kinase |
| CNS | central nervous system |
| OVA | chicken-egg albumin-ovalbumin |
| CRH | corticotropin-releasing hormone |
| DMTS | delayed matching to sample |
| DREADDS | Designer Receptors Exclusively Activated by Designer Drugs |
| DRG | dorsal root ganglion |
| EPM | elevated plus maze |
| ERK | extracellular signal-regulated kinase |
| FGF | fibroblast growth factor |
| 5CSRTT | five-choice serial-reaction time task |
| 4BC test | four-bottle choice test |
| GC | glucocorticoid hormone |
| HPA axis | hypothalamic-pituitary-adrenal axis |
| IMQ | imiquimod |
| IEG | immediate early gene |
| IL | interleukin |
| LDP | long-term depression |
| LTP | long-term potentiation |
| MBT | marble burying test |
| mPFC | medial prefrontal cortex |
| MGB axis | microbiota-gut-brain axis |
| MAPK | mitogen-activated protein kinases |
| NGF | nerve growth factor |
| NT | neurotensin |
| ORT | object recognition test |
| OCD | obsessive-compulsive disorder |
| OF | open field |
| OXA | oxazolone |
| PNS | peripheral nervous system |
| PET | positron emission tomography |
| PFC | prefrontal cortex |
| POMC | pro-opiomelanocortin |
| STAT3 | signal transducers and activators of transcription 3 |
| Scd1 | stearoyl-CoA desaturase-1 |
| SP | substance P |
| TSLP | thymic stromal lymphopoietin |
| TARC | thymus and activation-regulated chemokine |
| TLR | toll-like receptor |
| TGF | transforming growth factor |
| TrkB | tropomyosin receptor kinase B |
| TNF | tumor necrosis factor |
| 2BC test | two-bottle choice test |
| Th17 | type 17 T helper |
| Th2 | type 2 T helper |
| VEGF | vascular endothelial growth factor |
References
- Gilhar, A.; Reich, K.; Keren, A.; Kabashima, K.; Steinhoff, M.; Paus, R. Mouse models of atopic dermatitis: A critical reappraisal. Exp. Dermatol. 2021, 30, 319–336. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.-K.; Zhong, L.; Santiago, J.L. Association between stress and the HPA axis in the atopic dermatitis. Int. J. Mol. Sci. 2017, 18, 2131. [Google Scholar] [CrossRef] [PubMed]
- Nutten, S. Atopic dermatitis: Global epidemiology and risk factors. Ann. Nutr. Metab. 2015, 66, 8–16. [Google Scholar] [CrossRef]
- Yang, L.; Fu, J.; Zhou, Y. Research progress in atopic march. Front. Immunol. 2020, 11, 1907. [Google Scholar] [CrossRef]
- Hagenström, K.; Sauer, K.; Mohr, N.; Dettmann, M.; Glaeske, G.; Petersen, J.; Garbe, C.; Steimle, T.; Augustin, M. Prevalence and medications of atopic dermatitis in Germany: Claims data analysis. Clin. Epidemol. 2021, 13, 593–602. [Google Scholar] [CrossRef] [PubMed]
- Parisi, R.; Symmons, D.P.; Griffiths, C.E.; Ashcroft, D.M. Global epidemiology of psoriasis: A systematic review of incidence and prevalence. J. Investig. Dermatol. 2013, 133, 377–385. [Google Scholar] [CrossRef]
- Capon, F. The genetic basis of psoriasis. Int. J. Mol. Sci. 2017, 18, 2526. [Google Scholar] [CrossRef]
- Takeshita, J.; Grewal, S.; Langam, S.M.; Mehta, N.N.; Ogdie, A.; Van Voorhees, A.S.; Gelfand, J.M. Psoriasis and comorbid diseases part I. Epidemiology. J. Am. Acad. Dermatol. 2017, 76, 377–390. [Google Scholar] [CrossRef]
- Jafferany, M.; Ferreira, B.R.; Abdelmaksoud, A.; Mkhoyan, R. Management of psychocutaneous disorders: A practical approach for dermatologists. Dermatol. Ther. 2020, 33, e13969. [Google Scholar] [CrossRef]
- Miller, A.H.; Raison, C.L. The role of inflammation in depression: From evolutionary imperative to modern treatment target. Nat. Rev. Immunol. 2016, 16, 22–34. [Google Scholar] [CrossRef]
- Peters, E.M.J.; Michenko, A.; Kupfer, J.; Kummer, W.; Wiegand, S.; Niemeier, V.; Potekaev, N.N.; Lvov, A.; Gieler, U. Mental stress in atopic dermatitis–neuronal plasticity and the cholinergic system are affected in atopic dermatitis and in response to acute experimental mental stress in a randomized controlled pilot study. PLoS ONE 2014, 9, e113552. [Google Scholar] [CrossRef] [PubMed]
- Rousset, L.; Halioua, B. Stress and psoriasis. Int. J. Dermatol. 2018, 57, 1165–1172. [Google Scholar] [CrossRef] [PubMed]
- Kantor, R.; Kim, A.; Thyssen, J.P.; Silverberg, J.I. Association of atopic dermatitis with smoking: A systematic review and meta-analysis. J. Am. Acad. Dermatol. 2016, 75, 1119–1125.e1. [Google Scholar] [CrossRef]
- Rønnstad, A.T.M.; Halling-Overgaard, A.-S.; Hamann, C.R.; Skov, L.; Egeberg, A.; Thyssen, J.P. Association of atopic dermatitis with depression, anxiety, and suicidal ideation in children and adults: A systemic review and meta-analysis. J. Am. Acad. Dermatol. 2018, 79, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Yousaf, M.; Ayasse, M.; Ahmed, A.; Gwillim, E.C.; Janmohamed, S.R.; Yousaf, A.; Patel, K.R.; Thyssen, J.P.; Silverberg, J.I. Association between atopic dermatitis and hypertension: A systematic review and meta-analysis. Br. J. Dermatol. 2021, 186, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Leistner, C.; Menke, A. Hypothalamic-pituitary-adrenal axis and stress. Handb. Clin. Neurol. 2020, 175, 55–64. [Google Scholar]
- Haapakoski, R.; Ebmeier, K.P.; Alenius, H.; Kivimäki, M. Innate and adaptive immunity in the development of depression: An update on current knowledge and technological advances. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 66, 63–72. [Google Scholar] [CrossRef]
- Patel, N.; Nadkarni, A.; Cardwell, L.A.; Vera, N.; Frey, C.; Patel, N.; Feldman, S.R. Psoriasis, depression, and inflammatory overlap: A review. Am. J. Clin. Dermatol. 2017, 18, 613–620. [Google Scholar] [CrossRef]
- Wang, X.; Li, Y.; Wu, L.; Xiao, S.; Ji, Y.; Tan, Y.; Jiang, C.; Zhang, G. Dysregulation of the gut-brain-skin axis and key overlapping inflammatory and immune mechanisms of psoriasis and depression. Biomed. Pharmacother. 2021, 137, 111065. [Google Scholar] [CrossRef]
- Hashizume, H.; Takigawa, M. Anxiety in allergy and atopic dermatitis. Curr. Opin. Allergy Clin. Immunol. 2006, 6, 335–339. [Google Scholar] [CrossRef]
- Pavlovsky, L.; Friedman, A. Pathogenesis of stress-associated skin disorders: Exploring the brain-skin axis. Curr. Probl. Dermatol. 2017, 35, 136–145. [Google Scholar]
- Ohtsu, H.; Seike, M. Histamine and histamine receptors in allergic dermatitis. Handb. Exp. Pharmacol. 2017, 241, 222–245. [Google Scholar]
- Tausk, F.A.; Nousari, H. Stress and the skin. Arch. Dermatol. 2001, 137, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Stewart, J.M.; Taracanova, A.; Conti, P.; Zouboulis, C.C. Neuroendocrinology of the skin. Rev. Endocr. Metab. Discord. 2016, 17, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Zbytek, B.; Nikolakis, G.; Manna, P.R.; Skobowiat, C.; Zmijewski, M.; Li, W.; Janjetovic, Z.; Postlethwaite, A.; Zouboulis, C.C.; et al. Steroidogenesis in the skin: Implications for local immune functions. J. Steroid Biochem. Mol. Biol. 2013, 137, 107–123. [Google Scholar] [CrossRef]
- Alexopoulos, A.; Chrousos, G.P. Stress-related skin disorders. Rev. Endocr. Metab. Disord. 2016, 17, 295–304. [Google Scholar] [CrossRef]
- Dong, X.; Dong, X. Peripheral and central mechanisms of itch. Neuron 2018, 98, 482–494. [Google Scholar] [CrossRef]
- Shang, H.; Cao, X.-L.; Wan, Y.-J.; Meng, J.; Guo, L.-H. IL-4 gene polymorphism may contribute to an increased risk of atopic dermatitis in children. Dis. Markers 2016, 2016, 1021942. [Google Scholar] [CrossRef]
- Huang, E.; Ong, P.Y. Severe atopic dermatitis in children. Curr. Allergy Asthma Rep. 2018, 18, 35. [Google Scholar] [CrossRef]
- Bourgognon, J.-M.; Cavanagh, J. The role of cytokines in modulating learning and memory and brain plasticity. Brain Neurosci. Adv. 2020, 18, 2398212820979802. [Google Scholar] [CrossRef]
- Sanders, K.M.; Akiyama, T. The vicious cycle of itch and anxiety. Neurosci. Biobehav. Rev. 2018, 87, 17–26. [Google Scholar] [CrossRef]
- Juntilla, I. Turning the cytokine responses: An update on interleukin (IL)-4 and IL-13 receptor complexes. Front. Immunol. 2018, 9, 888. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lyga, J. Brain-skin connection: Stress, inflammation and skin aging. Inflamm. Allergy Drug Targets 2014, 13, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Yirmiya, R.; Goshen, I. Immune modulation of learning, memory, neural plasticity and neurogenesis. Brain Behav. Immun. 2011, 25, 181–213. [Google Scholar] [CrossRef] [PubMed]
- Michopoulos, V.; Powers, A.; Gillespie, C.F.; Ressler, K.J.; Jovanovic, T. Inflammation in Fear- and Anxiety-Based Disorders: PTSD, GAD, and Beyond. Neuropsychopharmacology 2017, 42, 254–270. [Google Scholar] [CrossRef] [PubMed]
- Elenkov, I.J.; Chrousos, G.P. Stress system-organization, physiology and immunoregulation. Neuroimmunomodulation 2006, 13, 257–267. [Google Scholar] [CrossRef]
- Li, Y.-L.; Zhao, H.; Ren, X.-B. Relationship of VEGF/VEGFR with immune and cancer cells: Staggering or forward? Cancer Biol. Med. 2016, 13, 206–214. [Google Scholar] [CrossRef]
- Sandhu, K.V.; Sherwin, E.; Schellekens, H.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Feeding the microbiota-gut-brain axis: Diet, microbiome, and neuropsychiatry. Transl. Res. 2017, 179, 223–244. [Google Scholar] [CrossRef]
- Cryan, J.F.; O’Riordan, K.J.; Cowan, C.S.M.; Sandhu, K.V.; Bastiaanssen, T.F.S.; Boehme, M.; Codagone, M.G.; Cussotto, S.; Fulling, C.; Golubeva, A.V.; et al. The microbiota-gut-brain axis. Physiol. Rev. 2019, 99, 1877–2013. [Google Scholar] [CrossRef]
- Tremblay, A.; Lingrand, L.; Maillard, M.; Feuz, B.; Tompkins, T.A. The effects of psychobiotics on the microbiota-gut-brain axis in early-life stress and neuropsychiatric disorders. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2021, 105, 110142. [Google Scholar] [CrossRef]
- Dumitrescu, L.; Popescu-Olaru, J.; Cozma, L.; Tulba, D.; Hinescu, M.E.; Ceafalan, L.C.; Gherghiceanu, M.; Popescu, B.O. Oxidative stress and the microbiota–gut–brain axis. Oxid. Med. Cell. Longev. 2018, 2018, 2406594. [Google Scholar]
- García-Cabrerizo, R.; Carbia, C.; O’Riordan, K.J.; Schellekens, H.; Cryan, J.F. Microbiota-gut-brain axis as a regulator of reward processes. J. Neurochem. 2020, 157, 1495–1524. [Google Scholar] [CrossRef] [PubMed]
- Pasparakis, M.; Haase, I.; Nestle, F.O. Mechanisms regulating skin immunity and inflammation. Nat. Rev. Immunol. 2014, 14, 289–301. [Google Scholar] [CrossRef]
- Bocheńska, K.; Smolińska, E.; Moskot, M.; Jakóbkiewicz-Banecka, J.; Gabig-Cimińska, M. Models in the research process of psoriasis. Int. J. Mol. Sci. 2017, 18, 2514. [Google Scholar] [CrossRef]
- Baurecht, H.; Rühlemann, M.C.; Rodríguez, E.; Thielking, F.; Harder, I.; Erkens, A.-S.; Stölzl, D.; Ellinghaus, E.; Hotze, M.; Lieb, W.; et al. Epidermal lipid composition, barrier integrity, and eczematous inflammation are associated with skin microbiome configuration. J. Allergy Clin. Immunol. 2018, 141, 1668–1676. [Google Scholar] [CrossRef] [PubMed]
- Gudjonsson, J.; Johnston, A.; Dyson, M.; Valdimarsson, H.; Elder, J.T. Mouse Models of Psoriasis. J. Investig. Dermatol. 2007, 127, 1292–1308. [Google Scholar] [CrossRef]
- Kim, D.; Kobayashi, T.; Nagao, K. Research techniques made simple: Mouse models of atopic dermatitis. J. Investig. Dermatol. 2019, 139, 984–990. [Google Scholar] [CrossRef]
- Smith, L.; Gatault, S.; Casals-Diaz, L.; Kelly, P.; Camerer, E.; Métais, C.; Knaus, U.G.; Eissner, G.; Steinhoff, M. House dust mite-treated PAR2 over-expressor mouse: A novel model of atopic dermatitis. Exp. Dermatol. 2019, 28, 1298–1308. [Google Scholar] [CrossRef]
- Kitagaki, H.; Ono, N.; Hayakawa, K.; Kitazawa, T.; Watanabe, K.; Shiohara, T. Repeated elicitation of contact hypersensitivity induces a shift in cutaneous cytokine milieu from a T helper cell type 1 to a T helper cell type 2 profile. Immunology 1997, 159, 2484–2491. [Google Scholar]
- Spergel, J.M.; Mizoguchi, E.; Brewer, J.P.; Martin, T.R.; Bhan, A.K.; Geha, R.S. Epicutaneous sensitization with protein antigen induces localized allergic dermatitis and hyperresponsiveness to methacholine after single exposure to aerosolized antigen in mice. J. Clin. Investig. 1998, 101, 1614–1622. [Google Scholar] [CrossRef]
- Jin, H.; Oyoshi, M.K.; Le, Y.; Bianchi, T.; Koduru, S.; Mathias, C.B.; Kumar, L.; Le Bras, S.; Young, D.; Collins, M.; et al. IL-21R is essential for epicutaneous sensitization and allergic skin inflammation in humans and mice. J. Clin. Investig. 2009, 119, 47–60. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.S.; Siracusa, M.C.; Saenz, S.A.; Noti, M.; Monticelli, L.A.; Sonnenberg, G.F.; Hepworth, M.R.; Van Voorhees, A.S.; Comeau, M.R.; Artis, D. TSLP elicits IL-33-independent innate lymphoid cell responses to promote skin inflammation. Sci. Transl. Med. 2013, 30, 170ra16. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Du, J.; Zhu, J.; Yang, X.; Zhou, B. Thymic stromal lymphopoietin signaling in CD4(+) T cells is required for TH2 memory. J. Allergy Clin. Immunol. 2015, 135, 781–791.e3. [Google Scholar] [CrossRef]
- Chen, J.; Niu, X.; Gao, Y.; Ma, L.; Gao, X.; Chen, H.; Qi, R. IL-18 knockout alleviates atopic dermatitis-like skin lesions induced by MC903 in a mouse model. Int. J. Mol. Med. 2020, 46, 880–888. [Google Scholar] [CrossRef] [PubMed]
- Moosburger-Martinz, V.; Schmuth, M.; Dubrac, S. A Mouse Model for Atopic Dermatitis Using Topical Application of Vitamin D3 or of Its Analog MC903. Methods Mol. Biol. 2017, 1559, 91–106. [Google Scholar]
- Naidoo, K.; Jagot, F.; Elsen, L.V.D.; Pellefigues, C.; Jones, A.; Luo, H.; Johnston, K.; Painter, G.; Roediger, B.; Lee, J.; et al. Eosinophils determine dermal thickening and water loss in an MC903 model of atopic dermatitis. J. Investig. Dermatol. 2018, 138, 2606–2616. [Google Scholar] [CrossRef]
- Schwartz, C.; Moran, T.; Saunders, S.P.; Kaszlikowska, A.; Floudas, A.; Bom, J.; Nunez, G.; Iwakura, Y.; O’Neill, L.; Irvine, A.D.; et al. Spontaneous atopic dermatitis in mice with a defective skin barrier is independent of ILC2 and mediated by IL-1β. Allergy 2019, 74, 1920–1933. [Google Scholar] [CrossRef]
- Thyssen, J.P.; Jakasa, I.; Riethmüller, C.; Schön, M.P.; Braun, A.; Haftek, M.; Fallon, P.G.; Wróblewski, J.; Jakubowski, H.; Eckhart, L.; et al. Filaggrin expression and processing deficiencies impair corneocyte surface texture and stiffness in mice. J. Investig. Dermatol. 2020, 140, 615–623. [Google Scholar] [CrossRef]
- Horváth, S.; Komlódi, R.; Perkecz, A.; Pintér, E.; Gyulai, R.; Kemény, A. Methodological refinement of Aldara-induced psoriasisiform dermatitis model in mice. Sci. Rep. 2019, 9, 3685. [Google Scholar] [CrossRef]
- Sundberg, J.P. Handbook of Mouse Mutations with Skin and Hair Abnormalities; CRC Press: Boca Raton, FL, USA, 1994. [Google Scholar]
- HogenEsch, H.; Gijbels, M.J.; Offerman, E.; Van Hooft, J.; Van Bekkum, D.W.; Zurcher, C. A spontaneous mutation characterized by chronic proliferative dermatitis in C57BL mice. Am. J. Pathol. 1993, 143, 972–982. [Google Scholar]
- Zheng, Y.; Eilertsen, K.J.; Ge, L.; Zhang, L.; Sundberg, J.P.; Prouty, S.M.; Stenn, K.S.; Parimoo, S. Scd1 is expressed in sebaceous glands and is disrupted in the asebia mouse. Nat. Genet. 1999, 23, 268–270. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, H.; Conklin, D.; Xu, W.; Grossmann, A.; Brender, T.; Carollo, S.; Eagan, M.; Foster, D.; Haldeman, B.A.; Hammond, A.; et al. Interleukin 20: Discovery, receptor identification, and role in epidermal function. Cell 2001, 104, 9–19. [Google Scholar] [CrossRef]
- Rico, L.; Del Rio, M.; Bravo, A.; Ramirez, A.; Jorcano, J.L.; Page, A.; Larcher, F. Targeted overexpression of leptin to keratinocytes in transgenic mice results in lack of skin phenotype but induction of early leptin resistance. Endocrinology 2005, 146, 4167–4176. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lowe, N.J.; Breeding, J.; Kean, C.; Cohn, M.L. Psoriasiform dermatosis in a rhesus monkey. J. Investig. Dermatol. 1981, 76, 141–143. [Google Scholar] [CrossRef] [PubMed]
- Zanolli, M.D.; Jayo, M.J.; Jayo, J.M.; Blaine, D.; Hall, J.; Jorizzo, J.L. Evaluation of psoriatic plaques that spontaneously developed in a cynomolgus monkey (Macaca fascicularis). Acta. Derm. Venereol. Suppl. (Stockh.) 1989, 146, 58. [Google Scholar] [PubMed]
- Sundberg, J.P.; King, L.E., Jr. Mouse mutations as animal models and biomedical tools for dermatological research. J. Investig. Dermatol. 1996, 106, 368–376. [Google Scholar] [CrossRef]
- Raychaudhuri, S.P.; Sanyal, M.; Weltman, H.; Kundu-Raychaudhuri, S. K252a, a high-affinity nerve growth factor receptor blocker, improves psoriasis: An in vivo study using the severe combined immunodeficient mouse-human skin model. J. Investig. Dermatol. 2004, 122, 812–819. [Google Scholar] [CrossRef]
- Wang, Y.; Li, P.; Zhang, L.; Fu, J.; Di, T.; Li, N.; Meng, Y.; Guo, J.; Zhao, J. Stress aggravates and prolongs imiquimod-induced psoriasis-like epidermal hyperplasia and IL-1β/IL-23p40 production. J. Leukoc. Biol. 2020, 108, 267–281. [Google Scholar] [CrossRef]
- Pu, J.; Wang, R.; Zhang, G.; Wang, J. FGF-7 facilitates the process of psoriasis by inducing TNF-α expression in HaCaT cells. Acta Biochim. Biophys. Sin. 2019, 51, 1056–1063. [Google Scholar] [CrossRef]
- Ju, N.; Shimamura, M.; Hayashi, H.; Ikeda, Y.; Yoshida, S.; Nakamura, A.; Morishita, R.; Rakugi, H.; Nakagami, H. Preventative effects of the partial RANKL peptide MHP1-AcN in a mouse model of imiquimod-induced psoriasis. Sci. Rep. 2019, 9, 15434. [Google Scholar] [CrossRef]
- Rogers, D.C.; Peters, J.; Martin, J.E.; Ball, S.; Nicholson, S.J.; Witherden, A.S.; Hafezparast, M.; Latcham, J.; Robinson, T.L.; Quilter, C.A.; et al. SHIRPA, a protocol for behavioral assessment: Validation for longitudinal study of neurological dysfunction in mice. Neurosci. Lett. 2001, 306, 89–92. [Google Scholar] [CrossRef]
- Robinson, L.; Riedel, G. Comparison of automated home-cage monitoring systems: Emphasis on feeding behaviour, activity and spatial learning following pharmacological interventions. J. Neurosci. Methods 2014, 234, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Deacon, R.M.J. Measuring motor coordination in mice. J. Vis. Exp. 2013, 75, e2609. [Google Scholar] [CrossRef] [PubMed]
- Luong, T.N.; Carlisle, H.J.; Southwell, A.; Patterson, P.H. Assessment of motor balance and coordination in mice using the balance beam. J. Vis. Exp. 2011, 10, 2376. [Google Scholar] [CrossRef]
- Sturman, O.; Germain, P.-L.; Bohacek, J. Exploratory rearing: A context- and stress-sensitive behavior recorded in the open-field test. Stress 2018, 21, 443–552. [Google Scholar] [CrossRef]
- Kraeuter, A.-K.; Guest, P.C.; Sarnyai, Z. The Elevated Plus Maze Test for Measuring Anxiety-Like Behavior in Rodents. Methods Mol. Biol. 2019, 1916, 69–74. [Google Scholar]
- Kulesskaya, N.; Voikar, V. Assessment of mouse anxiety-like behavior in the light–dark box and open-field arena: Role of equipment and procedure. Physiol. Behav. 2014, 133, 30–38. [Google Scholar] [CrossRef]
- Himanshu; Dharmila; Sarkar, D.; Nutan. A review of behavioral tests to evaluate different types of anxiety and anti-anxiety effects. Clin. Psychopharmacol. Neuroci. 2020, 18, 341–351. [Google Scholar] [CrossRef]
- Liu, M.-Y.; Yin, C.-Y.; Zhu, L.-J.; Zhu, X.-H.; Xu, C.; Luo, C.-X.; Chen, H.; Zhu, D.-Y.; Zhou, Q.-G. Sucrose preference test for measurement of stress-induced anhedonia in mice. Nat. Protoc. 2018, 13, 1686–1698. [Google Scholar] [CrossRef]
- Kaidanovich-Beilin, O.; Lipina, T.; Vukobradovic, I.; Roder, J.; Woodgett, J.R. Assessment of social interaction behaviors. J. Vis. Exp. 2011, 48, e2473. [Google Scholar] [CrossRef]
- Dixit, P.V.; Sahu, R.; Mishra, D.K. Marble-burying behavior test as a murine model of compulsive-like behavior. J. Pharmacol. Toxicol. Methods 2020, 102, 106676. [Google Scholar] [CrossRef] [PubMed]
- Eisenhardt, M.; Leixner, S.; Spanagel, R.; Bilbao, A. Quantification of alcohol drinking patterns in mice. Addict. Biol. 2015, 20, 1001–1011. [Google Scholar] [CrossRef] [PubMed]
- Vogel-Ciernia, A.; Wood, M.A. Examining Object Location and Object Recognition Memory in Mice. Curr. Protoc. Neurosci. 2014, 69, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Denninger, J.K.; Smith, B.M.; Kirby, E.D. Novel Object Recognition and Object Location Behavioral Testing in Mice on a Budget. J. Vis. Exp. 2018, 141, e58593. [Google Scholar] [CrossRef]
- Othman, M.Z.; Hassan, Z.; Has, A.T.C. Morris water maze: A versatile and pertinent tool for assessing spatial learning and memory. Exp. Anim. 2022, 21-0120. [Google Scholar] [CrossRef]
- Kay, C.; Harper, D.N.; Hunt, M. The effects of binge MDMA on acquisition and reversal learning in a radial-arm maze task. Neurobiol. Learn. Mem. 2011, 95, 473–483. [Google Scholar] [CrossRef]
- Leggio, G.M.; Torrisi, S.A.; Mastrogiacomo, R.; Mauro, D.; Chisari, M.; Devroye, C.; Scheggia, D.; Nigro, M.; Geraci, F.; Pintori, N.; et al. The epistatic interaction between the dopamine D3 receptor and dysbindin-1 modulates higher-order cognitive functions in mice and humans. Mol. Psychiatry 2019, 26, 1272–1285. [Google Scholar] [CrossRef]
- Birtalan, E.; Bánhidi, A.; Sanders, J.I.; Balázsfi, D.; Hangya, B. Efficient training of mice on the 5-choice serial reaction time task in an automated rodent training system. Sci. Rep. 2020, 10, 22362. [Google Scholar] [CrossRef]
- Kim, W.B.; Cho, J.-H. Encoding of contextual fear memory in hippocampal–amygdala circuit. Nat. Commun. 2020, 11, 1382. [Google Scholar] [CrossRef]
- LeDoux, J.E.; Moscarello, J.; Sears, R.; Campese, V. The birth, death and resurrection of avoidance: A reconceptualization of a troubled paradigm. Mol. Psychiatry 2017, 22, 24–36. [Google Scholar] [CrossRef]
- Shiotsuki, H.; Yoshimi, K.; Shimo, Y.; Funayama, M.; Takamatsu, Y.; Ikeda, K.; Takahashi, R.; Kitazawa, S.; Hattori, N. A rotarod test for evaluation of motor skill learning. J. Neurosci. Methods 2010, 189, 180–185. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association, APA Diagnostic and Statistical Manual of Mental Disorders (DSM-5); American Psychiatric Publishing: Arlington, TX, USA, 2013.
- Jirkof, P. Burrowing and nest building behavior as indicators of well-being in mice. J. Neurosci. Methods 2014, 234, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Molendjiek, M.L.; de Kloet, E.R. Coping with the forced swim stressor: Current state-of-the-art. Behav. Brain Res. 2019, 364, 2813–2831. [Google Scholar]
- Molendjiek, M.L.; de Kloet, E.R. Forced swim stressor: Trends in usage and mechanistic consideration. Eur. J. Neurosci. 2021, 55, 1–19. [Google Scholar]
- Mayfield, J.; Arends, M.; Harris, R.; Blednov, Y. Genes and alcohol consumption: Study with mutant mice. Int. Rev. Neurobiol. 2016, 126, 293–355. [Google Scholar] [CrossRef]
- Pitts, M.W. Barnes maze procedure for spatial learning and memory in mice. Bio. Protoc. 2018, 8, e2744. [Google Scholar] [CrossRef]
- Stopford, C.L.; Thompson, J.C.; Neary, D.; Richardson, A.M.; Snowden, J.S. Working memory, attention, and executive function in Alzheimer’s disease and frontotemporal dementia. Cortex 2012, 48, 429–446. [Google Scholar] [CrossRef]
- Lind, J.; Enquist, M.; Ghirlanda, S. Animal memory: A review of delayed matching-to-sample data. Behav. Processes 2015, 117, 52–58. [Google Scholar] [CrossRef]
- Josselyn, S.A.; Köhler, S.; Frankenland, P.W. Finding the engram. Nat. Rev. Neurosci. 2015, 16, 521–534. [Google Scholar] [CrossRef]
- McLellan, M.A.; Rosenthal, N.A.; Pinto, A.R. Cre-loxP-mediated recombination: General principles and experimental considerations. Curr. Protoc. Mouse Biol. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Poth, K.M.; Texakalidis, P.; Boulis, N.M. Chemogenetics: Beyond lesions and electrodes. Neurosurgery 2021, 89, 185–195. [Google Scholar] [CrossRef]
- Rost, B.R.; Schneider-Warme, F.; Schmitz, D.; Hegemann, P. Optogenetic tools for subcellular application in neuroscience. Neuron 2017, 96, 572–603. [Google Scholar] [CrossRef]
- Gallo, F.T.; Katche, C.; Morici, J.F.; Mediana, J.H.; Weisstaub, N. Immediate early genes, memory and psychiatric disorders: Focus on c-Fos, Egr1 and Arc. Front. Behav. Neurosci. 2018, 12, 79. [Google Scholar] [CrossRef] [PubMed]
- Franceschini, A.; Costantini, I.; Pavone, F.S.; Silvestri, L. Dissecting neuronal activation on a brain-wide scale with immediate early genes. Front. Neurosci. 2020, 14, 569517. [Google Scholar] [CrossRef] [PubMed]
- Ali, F.; Kwan, A.C. Interpreting in vivo calcium signals from neuronal cell bodies, axons, and dendrites: A review. Neurophotonics 2020, 7, 011402. [Google Scholar] [CrossRef]
- Wall, N.R.; Wickersham, I.R.; Cetin, A.; De La Parra, M.; Callaway, E.M. Monosynaptic circuit tracing in vivo through Cre-dependent targeting and complementation of modified rabies virus. Proc. Natl. Acad. Sci. USA 2010, 107, 21848–21853. [Google Scholar] [CrossRef] [PubMed]
- Lanciego, J.L.; Wouterlood, F.G. Neuroanatomical tract-tracing techniques that did go viral. Brain Struct. Funct. 2020, 225, 1193–1224. [Google Scholar] [CrossRef]
- Navabpour, S.; Kwapis, J.L.; Jarome, T.J. A neuroscientist’s guide to transgenic mice and other genetic tools. Neurosci. Biobehav. Rev. 2020, 108, 703–748. [Google Scholar] [CrossRef]
- Dando, S.J.; Kazanis, R.; Chinnery, H.R.; McMenamin, P.G. Regional and functional heterogeneity of antigen presenting cells in the mouse brain and meninges. Glia 2019, 67, 935–949. [Google Scholar] [CrossRef]
- Eme-Scolan, E.; Dando, S.J. Tools and approaches for studying microglia in vivo. Front. Immunol. 2020, 11, 583647. [Google Scholar] [CrossRef]
- Plemel, J.R.; Stratton, J.A.; Michaels, N.J.; Rawji, K.S.; Zhang, E.; Sinha, S.; Baaklini, C.S.; Dong, Y.; Ho, M.; Thorburn, K.; et al. Microglia response following acute demyelination is heterogeneous and limits infiltrating macrophage dispersion. Sci. Adv. 2020, 6, eaay6324. [Google Scholar] [CrossRef] [PubMed]
- Parkhurst, C.N.; Yang, G.; Ninan, I.; Savas, J.N.; Yates, J.R., III; Lafaille, J.J.; Hempstead, B.L.; Littman, D.R.; Gan, W.-B. Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell 2013, 155, 1596–1609. [Google Scholar] [CrossRef] [PubMed]
- Salter, M.W.; Stevens, B. Microglia emerge as central players in brain disease. Nature Med. 2017, 23, 1018–1027. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Jin, Y.; Shapiro, T.M.; Hinduja, A.; Baas, P.W.; Tom, V.J. Chronic neuronal activation increases dynamic microtubules to enhance functional axon regeneration after dorsal root crush injury. Nat. Commun. 2020, 11, 6131. [Google Scholar] [CrossRef] [PubMed]
- Krames, E.S. The Role of the Dorsal Root Ganglion in the Development of Neuropathic Pain. Pain Med. 2014, 15, 1669–1685. [Google Scholar] [CrossRef] [PubMed]
- Daigle, T.L.; Madisen, L.; Hage, T.A.; Valley, M.T.; Knoblich, U.; Larsen, R.S.; Takeno, M.M.; Huang, L.; Gu, H.; Larsen, R.; et al. A suite of transgenic driver and reporter mouse lines with enhanced brain-cell-type targeting and functionality. Cell 2018, 174, 465–480.e422. [Google Scholar] [CrossRef]
- Breschi, A.; Gingeras, T.R.; Guigó, R. Comparative transcriptomics in human and mouse. Nat. Rev. Genet. 2017, 18, 425–440. [Google Scholar] [CrossRef]
- Hodge, R.D.; Bakken, T.E.; Miller, J.A.; Smith, K.A.; Barkan, E.R.; Graybuck, L.T.; Close, J.L.; Long, B.; Johansen, N.; Penn, O.; et al. Conserved cell types with divergent features in human versus mouse cortex. Nature 2019, 573, 61–68. [Google Scholar] [CrossRef]
- Khanna, R.; Burrows, S.R. Human immunology: A case for the ascent of non-furry immunology. Immunol. Cell Biol. 2011, 89, 330–331. [Google Scholar] [CrossRef]
- Perlman, R.L. Mouse models of human disease. Evol. Med. Public Health 2016, 2016, 170–176. [Google Scholar]
- Nakajima, M.; Schmitt, L.I. Understanding the circuit basis of cognitive functions using mouse models. Neurosci. Res. 2020, 152, 44–58. [Google Scholar] [CrossRef] [PubMed]
- Raza, S.A.; Albrecht, A.; Caliskan, G.; Müller, B.; Demiray, Y.E.; Ludewig, S.; Meis, S.; Faber, N.; Hartig, R.; Schraven, B.; et al. HIPP neurons in the dentate gyrus mediate the cholinergic modulation of background context memory salience. Nat. Commun. 2017, 8, 189. [Google Scholar] [CrossRef] [PubMed]
- Regev-Tsur, S.; Demiray, Y.E.; Tripathi, K.; Stork, O.; Richter-Levin, G.; Albrecht, A. Region-specific involvement of interneuron subpopulaitons in trauma-related pathology and resilience. Neurobiol. Dis. 2020, 143, 104974. [Google Scholar] [CrossRef] [PubMed]
| (A) AD | Description | Advantages/Caveats | Reference |
|---|---|---|---|
| Oxazolone (OXA) application | Destroyed integrity of skin barrier Th2 immune response | (+) rapid, low cost (−) model for allergic contact dermatitis | [1,47,48] |
| Chicken-egg albumin-ovalbumin (OVA) application | Triggers Th2 immune response | (+) chronic AD-like skin lesions (−) variable OVA allergen composition (−) not sufficient in certain mouse strains (e.g., C57BL/6) | [1,49,50,51] |
| Calcipotriol (MC903) application | Activation of ILC2 type-2 immune response with eosinophilia, skin swelling, inflammation | (+) model for type 2 immune response initiation (+) to study TSLP and neutrophils in scratching behavior | [52,53,54,55,56] |
| Flg ft/ft or Flg -/- “Flaky tail mice” | Filaggrin deficient mice | (+) spontaneous dermatitis (+) to study skin microbiome | [57,58] |
| Blmh -/- mice | Bleomycin hydrolase (BLMH) deficiency impairs filaggrin processing | (+) decreased levels of natural moisturizing factors (+) decreased levels of BLMH in AD | [58] |
| Interleukin overexpression | Overexpression of IL-4, IL-5, IL-13, IL-18, IL-31, TSLP | (+) exploration of specific pathways | [47] |
| Imiquimod application | Acute skin inflammation | (+) erythema and scales as in human disorder (+) used to study stress–skin symptom correlation | [59] |
| Ttc7 fsn/Ttc7 fsn | Spontaneous mutation in tertratricopeptide repeat domain 7 | (+) progressive papulosquamous as in human disease | [60] |
| cpdm/cpdm | Spontaneous proliferative dermatitis mutation mouse | (+) red and scaling skin as in human disease | [61] |
| Scd1 ab/Scd1 ab | Asebia mouse, defective stearoyl-CoA desaturase-1 (Scd1) gene | (+) leads to hypoplastic sebaceous glands | [62] |
| Interleukin signaling | Overexpression/knock out of IL-6, IL-20, STAT3 pathway | (+) hyperproliferation of keratinocytes and altered differentiation via STAT3 pathway | [46,63,64] |
| Transgenic mice for aberrant T-cell function | Via TGF ****** regulating T cell development | (+) altered keratinocyte regulation | [46] |
| Assay | Read-Outs | Associated Psychiatric Feature | Reference |
|---|---|---|---|
| SHIRPA test | Movement, posture, reflexes | Basic neurological characterization | [72] |
| PhenoTyper | Activity | Circadian rhythm, basal activity | [73] |
| Rotarod | Motor learning | Neurological motor and coordination deficits | [74] |
| Beam walking | Motor coordination | Neurological motor and coordination deficits | [75] |
| Open field | Time and distance covered | Locomotory activity, anxiety | [76] |
| Elevated plus maze | Time, distance, and entries in open and closed arms | Locomotory activity, anxiety | [77] |
| Light–dark box | Transitions between compartments, time spent in compartments | Anxiety | [78] |
| Marble burying test | Numbers of marbles covered by bedding | Anxiety, compulsive behavior, repetitive behavior | [79] |
| Sucrose preference test | Consumption of plain water vs. water with sweetener | Anhedonia, depression | [80] |
| Social interaction test | Time contacting a social interaction partner restricted in a tube | Social preference, social anhedonia, social memory | [81] |
| Nest building | Complexity scores of nests built from tissues | Reduced wellbeing, depression, compulsive and repetitive behavior | [82] |
| Two-bottle choice test | Consumption of plain water vs. ethanol | Addiction | [83] |
| Object recognition | Time spent with familiar vs. novel objects or object locations | Recognition and spatial memory | [84,85] |
| Water maze/Barnes maze | Latency to reach an escape platform/hole, time spent at the escape platform/hole | Spatial memory | [86] |
| Radial arm maze | Latency to reach a reward arm | Spatial memory, working memory | [87] |
| Delayed matching to sample test | According to a learning rule correctly entering a specific arm on a T-maze after a delay | Working memory | [88] |
| 5CSRTT | Correct choices for retaining a reward associated with sequences of stimuli | Working memory, attention, impulsivity | [89] |
| Fear conditioning | Freezing to a conditioned stimulus or context | Aversive memory | [90,91] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weiglein, A.; Gaffal, E.; Albrecht, A. Probing the Skin–Brain Axis: New Vistas Using Mouse Models. Int. J. Mol. Sci. 2022, 23, 7484. https://doi.org/10.3390/ijms23137484
Weiglein A, Gaffal E, Albrecht A. Probing the Skin–Brain Axis: New Vistas Using Mouse Models. International Journal of Molecular Sciences. 2022; 23(13):7484. https://doi.org/10.3390/ijms23137484
Chicago/Turabian StyleWeiglein, Aliće, Evelyn Gaffal, and Anne Albrecht. 2022. "Probing the Skin–Brain Axis: New Vistas Using Mouse Models" International Journal of Molecular Sciences 23, no. 13: 7484. https://doi.org/10.3390/ijms23137484
APA StyleWeiglein, A., Gaffal, E., & Albrecht, A. (2022). Probing the Skin–Brain Axis: New Vistas Using Mouse Models. International Journal of Molecular Sciences, 23(13), 7484. https://doi.org/10.3390/ijms23137484







