Neuronal-Immune Cell Units in Allergic Inflammation in the Nose
Abstract
:1. Introduction
2. Combined Neuroimmune System
3. Neuro Molecules of the Neuroimmune Network
4. Functional Structure of the Muciliary Barrier in the Nasal Cavity
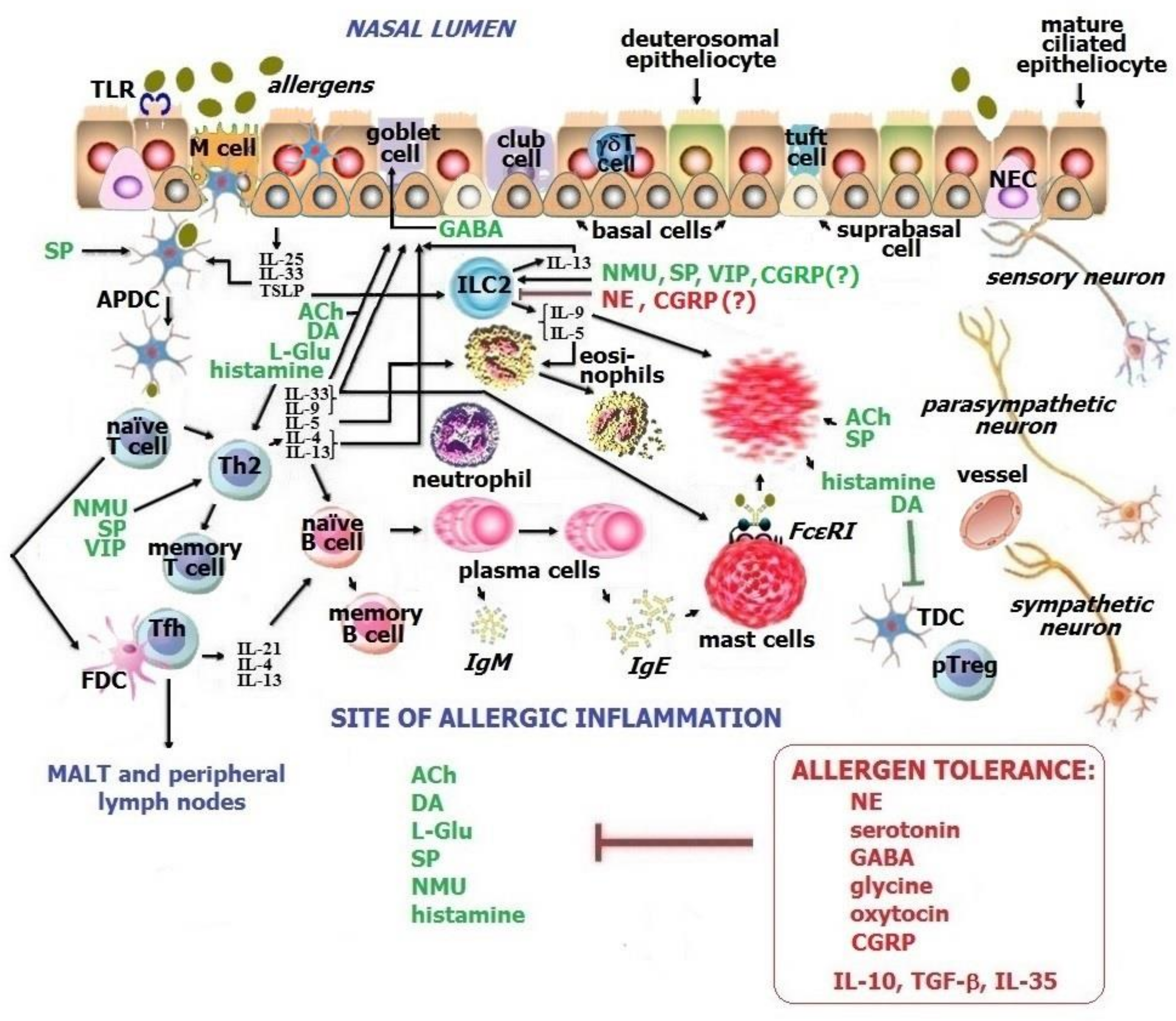
5. Allergic Inflammation at the Sites of Nasal Mucosa
6. Neuronal-Immune Cell Interplays in the Upper Respiratory Tract
6.1. Neuronal-Lymphocyte Unit
6.2. Neuronal-Dendritic Cellunit
6.3. Neuronal-Macrophage Unit
6.4. Neuronal-ILC2Unit
6.5. Neuronal-Mast Cell Unit
6.6. Neuronal-Eosinophil Unit
6.7. Neuronal-Neutrophil Unit
6.8. Neuronal-NEC Unit
7. Discussion
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| scRNA-seq | single-cell RNA-sequencing |
| CNS | central nervous system |
| NEC | neuroendocrine cell |
| HPA | hypothalamic-pituitary-adrenal axis |
| L-Glu | L-glutamate |
| DA | dopamine |
| ACh | acetylcholine |
| a7nAchR | nicotinic acetylcholine receptor |
| MAchR | muscarinic acetylcholine receptor |
| mGluR | metabotropic L-glutamate receptor |
| iGluR | ionotropic L-glutamate receptor |
| AVR | arginine-vasopressin receptor |
| SP | substance P |
| NKR | substance P receptor |
| NMU | neuromedin U |
| NMUR | neuromedin U receptor |
| NE | norepinephrine |
| AR | adrenoreceptor |
| GABA | γ Aminobutiric acid |
| GlyR | glycine receptor |
| OXTR | oxytocin receptor |
| CGRP | calcitonin-gene-related peptide |
| CLR | calcitonin-gene-related peptide receptor |
| ATP | adenosine triphosphate |
| NGF | nerve growth factor |
| Trk | nerve growth factor receptor |
| BDNF | brain-derived neurotrophic factor |
| GPCR | G-protein-coupled receptor |
| TSLP | thymic stromal lymphopoietin |
| ILC2 | group 2 innate lymphoid cell |
| Th | helper T cell |
| APDC | allergen-presenting dendritic cell |
| TLR | Toll-like receptors |
| DC | dendritic cell |
| TDC | tolerogenic dendritic cell |
| pTreg | peripheral regulatory T cell |
References
- Coyle, P.K. Introduction to neuroimmunology. In Clinical Neuroimmunology; Rizvi, S.A., Cahil, J.F., Coyle, P.K., Eds.; Humana Press: New York, NY, USA, 2019; pp. 3–15. [Google Scholar] [CrossRef]
- Hodo, T.W.; de Aquino, M.T.P.; Shimamoto, A.; Shanker, A. Critical neurotransmitters in the neuroimmune network. Front. Immunol. 2020, 11, 1869. [Google Scholar] [CrossRef] [PubMed]
- Foster, S.L.; Seehus, C.R.; Woolf, C.J.; Talbot, S. Sense and immunity: Context-dependent neuro-immune interplay. Front. Immunol. 2017, 8, 1463. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-G.; Pavon, L.; Tu, H. Novel aspects of neurotransmitters. In Frontiers in Cell and Developmental Biology and Frontiers in Pharmacology; Zhang, Z.-G., Pavón, L., Tu, H., Eds.; Frontiers Media SA: Lausanne, Switzerland, 2021; pp. 4–5. [Google Scholar] [CrossRef]
- Godinho-Silva, C.; Cardoso, F.; Veiga-Fernandes, H. Neuro-immune cell units: A new paradigm in physiology. Annu. Rev. Immunol. 2019, 37, 19–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jean, E.E.; Good, O.; Herbert, D.-B.R. Neuroimmune regulatory networks of the airway mucosa in allergic inflammatory disease. J. Leuk. Biol. 2021, 111, 209–221. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, R.J.; Lloyd, C.M. Regulation of immune responses by the airway epithelial cell landscape. Nat. Rev. Immunol. 2021, 21, 347–362. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.D.; Wypych, T.P. Cellular and functional heterogeneity of the airway epithelium. Mucosal Immunol. 2021, 14, 978–990. [Google Scholar] [CrossRef] [PubMed]
- Ruiz García, S.; Deprez, M.; Lebrigand, K.; Cavard, A.; Paquet, A.; Arguel, M.J.; Magnone, V.; Truchi, M.; Caballero, I.; Leroy, S.; et al. Novel dynamics of human mucociliary differentiation revealed by single-cell RNA sequencing of nasal epithelial cultures. Development 2019, 146, dev177428. [Google Scholar] [CrossRef] [Green Version]
- Ruysseveldt, E.; Martens, K.; Steelant, B. Airway basal cells, protectors of epithelial walls in health and respiratory diseases. Front. Allergy 2021, 2, 787128. [Google Scholar] [CrossRef]
- Travaglini, K.J.; Nabhan, A.N.; Penland, L.; Sinha, R.; Gillich, A.; Sit, R.V.; Chang, S.; Conley, S.D.; Mori, Y.; Seita, J.; et al. A molecular cell atlas of the human lung from single cell RNA sequencing. Nature 2020, 587, 619–625. [Google Scholar] [CrossRef]
- Croese, T.; Castellani, G.; Schwartz, M. Immune cell compartmentalization for brain surveillance and protection. Nat. Immunol. 2021, 22, 1083–1092. [Google Scholar] [CrossRef]
- Chen, C.-S.; Barnoud, C.; Scheiermann, C. Peripheral neurotransmitters in the immune system. Curr. Opin. Physiol. 2021, 19, 73–79. [Google Scholar] [CrossRef]
- Korin, B.; Dubovik, T.; Rolls, A. Mass cytometry analysis of immune cells in the brain. Nat. Protoc. 2018, 13, 377–391. [Google Scholar] [CrossRef]
- Jain, A.; Hakim, S.; Woolf, C.J. Unraveling the plastic peripheral neuroimmune interactome. J. Immunol. 2020, 204, 257–263. [Google Scholar] [CrossRef]
- Selye, H. Stress and the general adaptation syndrome. Br. Med. J. 1950, 1, 1383–1392. [Google Scholar] [CrossRef] [Green Version]
- Chesné, J.; Cardoso, V.; Veiga-Fernandes, H. Neuro-immune regulation of mucosal physiology. Mucosal Immunol. 2019, 12, 10–20. [Google Scholar] [CrossRef] [Green Version]
- Voisin, T.; Bouvier, A.; Chiu, I.V. Neuro-immune interactions in allergic diseases: Novel targets for therapeutics. Int. Immunol. 2017, 29, 247–261. [Google Scholar] [CrossRef] [Green Version]
- Drokhlyansky, E.; Smillie, C.S.; Van Wittenberghe, N.; Ericsson, M.; Griffin, G.K.; Eraslan, G.; Dionne, D.; Cuoco, M.S.; Goder-Reiser, M.N.; Sharova, T.; et al. The human and mouse enteric nervous system at single-cell resolution. Cell 2020, 182, 1606–1622.e23. [Google Scholar] [CrossRef]
- Ivanova, D.; Cousin, M.A. Synaptic vesicle recycling and the endolysosomal system: A reappraisal of form and function. Front. Synaptic Neurosci. 2022, 14, 826098. [Google Scholar] [CrossRef]
- Carlton, S.M. Nociceptive primary afferents: They have a mind of their own. J. Physiol. 2014, 592, 3403–3411. [Google Scholar] [CrossRef]
- Kabata, H.; Artis, D. Neuro-immune crosstalk and allergic inflammation. J. Clin. Investig. 2019, 129, 1475–1482. [Google Scholar] [CrossRef] [Green Version]
- Noguchi, M.; Furukawa, K.T.; Morimoto, M. Pulmonary neuroendocrine cells: Physiology, tissue homeostasis and disease. Dis. Model. Mech. 2020, 13, dmm046920. [Google Scholar] [CrossRef] [PubMed]
- Delpech, J.-C.; Herron, S.; Botros, M.B.; Ikezu, T. Neuroimmune crosstalk through extracellular vesicles in health and disease. Trends Neurosci. 2019, 42, 361–372. [Google Scholar] [CrossRef] [PubMed]
- Wisniewski, J.; Agrawal, R.; Woodfolk, J.A. Mechanisms of tolerance induction in allergic disease: Integrating current and emerging concepts. Clin. Exp. Allergy 2013, 43, 164–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klimov, V.V. From Basic to Clinical Immunology; Springer Nature: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
- Kerage, D.; Sloan, E.K.; Mattarollo, S.R.; McCombe, P.A. Interaction of neurotransmitters and neurochemicals with lymphocytes. J.Neuroimmunol. 2019, 332, 99–111. [Google Scholar] [CrossRef] [Green Version]
- Samuels, A. Dose dependent toxicity of glutamic acid: A review. Int. J. Food Prop. 2020, 23, 412–419. [Google Scholar] [CrossRef] [Green Version]
- Weng, M.; Xie, X.; Liu, C.; Lim, K.-L.; Zhang, C.-W.; Li, L. The sources of reactive oxygen species and its possible role in the pathogenesis of Parkinson’s disease. Parkinson’s Dis. 2018, 2018, 9163040. [Google Scholar] [CrossRef] [Green Version]
- Larenas-Linnemann, D.E.; Ortega-Martell, J.A.; Brandón-Vijil, M.V.; Rodríguez-Péréz, N.; Luna-Pech, J.A.; Estrada-Cardona, A.; Arias-Cruz, A.; Del Rio-Navarro, B.E.; Navarrete Rodríguez, E.M.; Pozo-Beltrán, C.F.; et al. Coronavirus disease 2019, allergic diseases, and allergen immunotherapy: Possible favorable mechanisms of interaction. Allergy Asthma Proc. 2021, 42, 187–197. [Google Scholar] [CrossRef]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Skaper, S.D. Nerve growth factor: A neuroimmune crosstalk mediator for all seasons. Immunology 2017, 151, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Cuevas, J. Neurotransmitters and Their Life Cycle. Available online: http://www.sciencedirect.com/science/article/pii/B9780128012383113182 (accessed on 31 May 2022).
- Bosmans, G.; Bassi, G.S.; Florens, M.; Gonzalez-Dominguez, E.; Matteoli, G.; Boeckxstaens, G.E. Cholinergic modulation of type 2 immune responses. Front. Immunol. 2017, 8, 1873. [Google Scholar] [CrossRef] [Green Version]
- Cox, M.A.; Bassi, C.; Saunders, M.E.; Nechanitzky, R.; Morgado-Palacin, I.; Zheng, C.; Mak, T.W. Beyond neurotransmission: Acetylcholine in immunity and inflammation. J. Int. Med. 2020, 287, 120–133. [Google Scholar] [CrossRef]
- Gori, S.; Vermeulen, M.; Remes-Lenicov, F.; Jancic, C.; Scordo, W.; Caballos, A.; Towstyka, N.; Bestach, Y.; Belli, C.; Sabbione, F.; et al. Acetylcholine polarizes dendritic cells toward a Th2-promoting profile. Allergy 2017, 72, 221–231. [Google Scholar] [CrossRef]
- Saunders, C.J.; Christensen, M.; Finger, T.E.; Tizzano, M. Cholinergic neurotransmission links solitary chemosensory cells to nasal inflammation. Proc. Natl. Acad. Sci. USA 2014, 111, 6075–6080. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Cohen, J.A.; Wallrapp, A.; Trieu, K.G.; Barrios, J.; Shao, F.; Krishnamoorthy, N.; Kuchroo, V.K.; Jones, M.R.; Fine, A.; et al. Age-related dopaminergic innervation augments T helper 2-type allergic inflammation in the postnatal lung. Immunity 2019, 51, 1102–1118.e1107. [Google Scholar] [CrossRef]
- Matt, S.M.; Gaskill, P.J. Where is Dopamine and how do immune cells see it? Dopamine-mediated immune cell function in health and disease. J. NeuroimmPharmacol. 2020, 15, 114–164. [Google Scholar] [CrossRef]
- Alim, M.A.; Grujic, M.; Ackerman, P.W.; Kristiansson, P.; Eliasson, P.; Peterson, M.; Pejler, G. Glutamate triggers the expression of functional ionotropic and metabotropic glutamate receptors in mast cells. Cell. Mol. Immunol. 2020, 18, 2383–2392. [Google Scholar] [CrossRef] [Green Version]
- Contreras Healey, D.C.; Cephus, J.Y.; Barone, S.M.; Chowdhury, N.U.; Dahunsi, D.O.; Madden, M.Z.; Ye, X.; Yu, X.; Olszewski, K.; Young, K.; et al. Targeting in vivo metabolic vulnerabilities of Th2 and Th17 cells reduces airway inflammation. J. Immunol. 2021, 206, 1127–1139. [Google Scholar] [CrossRef]
- Thangam, E.B.; Jemima, E.A.; Singh, H.; Baig, M.S.; Khan, M.; Mathias, C.B.; Church, M.K.; Saluja, R. The role of histamine and histamine receptors in mast cell-mediated allergy and inflammation: The hunt for new therapeutic targets. Front. Immunol. 2018, 9, 1873. [Google Scholar] [CrossRef] [Green Version]
- Yamauchi, K.; Ogasawara, M. The Role of histamine in the pathophysiology of asthma and the clinical efficacy of antihistamines in asthma therapy. Int. J. Mol. Sci. 2019, 20, 1733. [Google Scholar] [CrossRef] [Green Version]
- Dale, H.H.; Laidlaw, P.P. Histamine shock. J. Physiol. 2019, 52, 355–390. [Google Scholar] [CrossRef]
- Saeki, M.; Nishimura, T.; Kaminuma, O.; Ohtsu, H.; Mori, A.; Hiroi, T. Crosstalk between histamine and T cells in allergic diseases. Curr. Immunol. Rev. 2016, 12, 10–13. [Google Scholar] [CrossRef]
- Scammell, T.E.; Jackson, A.C.; Franks, N.P.; Wisden, W.; Dauvilliers, Y. Histamine: Neural circuits and new medications. SleepJ 2019, 42, 1–8. [Google Scholar] [CrossRef]
- Wiedermann, F.J.; Watzinger, K.; Stichlberger, M.; Joannidis, M.; Kaehler, C.; Lederer, W. Effects of arginine vasopressin on migration and respiratory burst activity in human leukocytes. Open Med. 2018, 13, 122–129. [Google Scholar] [CrossRef]
- Al-kuraishy, H.M.; Al-Gareeb, A.I.; Qusti, S.; Alshammari, E.M.; Atanu, F.O.; Batiha, G.E.-S. Arginine vasopressin and pathophysiology of COVID-19: An innovative perspective. Biomed. Pharmacother. 2021, 143, 112193. [Google Scholar] [CrossRef]
- Berczi, I.; Stephano, A. Vasopressin, the acute phase response and healing. In Insights to Neuroimmune Biology, 2nd ed.; Berczi, I., Ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 185–199. [Google Scholar] [CrossRef]
- Marseglia, L.; D’Angelo, G.; Manti, S.; Salpietro, C.; Arrigo, T.; Barberi, I.; Reiter, R.J.; Gitto, E. Melatonin and atopy: Role in atopic dermatitis and asthma. Int. J. Mol. Sci. 2014, 15, 13482–13493. [Google Scholar] [CrossRef] [Green Version]
- Luo, J.; Zhang, Z.; Sun, H.; Song, J.; Chen, X.; Huang, J.; Lin, X.; Zhou, R. Effect of melatonin on T/B cell activation and immune regulation in pinealectomy mice. Life Sci. 2020, 242, 117191. [Google Scholar] [CrossRef]
- Guan, R.; Malkani, R.G. Melatonin, sleep, and allergy. In Allergy and Sleep; Fishbein, A., Sheldon, S., Eds.; Springer: Cham, Switzerland, 2019; pp. 367–384. [Google Scholar] [CrossRef]
- Stauch, B.; Johansson, L.C.; Cherezov, V. Structural insights into melatonin receptors. FEBS J. 2020, 287, 1496–1510. [Google Scholar] [CrossRef] [Green Version]
- Perner, C.; Flayer, C.H.; Zhu, X.; Aderhold, P.A.; Dewan, Z.N.A.; Voisin, T.; Camire, R.B.; Chow, O.A.; Chiu, I.M.; Sokol, C.L. Substance P release by sensory neurons triggers dendritic cell migration and initiates the type-2 immune response to allergens. Immunity 2020, 53, 1063–1077.e7. [Google Scholar] [CrossRef]
- Mashaghi, A.; Marmalidou, A.; Tehrani, M.; Grace, P.T.; Pothoulakis, C.; Dana, R. Neuropeptide substance P and the immune response. Cell Mol. Life Sci. 2016, 73, 4249–4264. [Google Scholar] [CrossRef] [Green Version]
- Larsson, O.; Tengroth, L.; Xu, Y.; Uddman, R.; Georen, S.K.; Cardell, L.-O. Substance P represents a novel first-line defense mechanism in the nose. J. Allergy Clin. Immunol. 2017, 141, 128–136.e3. [Google Scholar] [CrossRef] [Green Version]
- Larsson, O.J.; Georen, S.K.; Cardell, L.-O. Rapid activation of brainstem nuclei following TLR stimulation of the nasal mucosa. Acta Neurobiol. Exp. 2020, 80, 353–357. [Google Scholar] [CrossRef]
- Klose, C.S.N.; Veiga-Fernandes, H. Neuroimmune interactions in peripheral tissues. Eur. J. Immunol. 2021, 51, 1602–1614. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Dong, F.; Zhuang, Y.; Wang, Y.; Ma, W. Effect of neuromedin U on allergic airway inflammation in an asthma model (Review). Exp. Ther. Med. 2020, 19, 809–816. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, W.; Shu, Q.; Fan, J. Neural regulation of interactions between group 2 Innate Lymphoid Cells and pulmonary immune cells. Front. Immunol. 2020, 11, 576929. [Google Scholar] [CrossRef]
- Wallrapp, A.; Riesenfeld, S.J.; Burkett, P.R.; Abdulnour, R.E.; Nyman, J.; Dionne, D.; Hofree, M.; Cuoco, M.S.; Rodman, C.; Farouq, D.; et al. The neuropeptide NMU amplifies ILC2-driven allergic lung inflammation. Nature 2017, 549, 351–356. [Google Scholar] [CrossRef]
- Ye, Y.; Hue, L. The potential role of Neuromedin U in human type-2 immunity. Eur. Respir. J. 2019, 54 (Suppl. S63), OA1626. [Google Scholar] [CrossRef]
- Moriyama, S.; Brestoff, J.R.; Flamar, A.L.; Moeller, J.B.; Klose, C.S.N.; Rankin, L.C.; Naomi, A.; Yudanin, N.A.; Monticelli, L.A.; Putzel, G.G.; et al. Beta2- adrenergic receptor-mediated negative regulation of group 2 innate lymphoid cell responses. Science 2018, 359, 1056–1061. [Google Scholar] [CrossRef] [Green Version]
- Pongratz, G.; Straub, R.H. The sympathetic nervous response in inflammation. Arthritis Res. Ther. 2014, 16, 504. [Google Scholar] [CrossRef] [Green Version]
- Pinho-Ribeiro, F.A.; Baddal, B.; Haarsma, R.; O’Seaghdha, M.; Yang, N.J.; Blake, K.J.; Portley, M.; Verri, W.A.; Dale, J.B.; Wessels, M.R.; et al. Blocking neuronal signaling to immune cells treats streptococcal invasive infection. Cell 2018, 173, 1083–1097. [Google Scholar] [CrossRef] [Green Version]
- Baral, P.; Umans, B.D.; Li, L.; Wallrapp, A.; Bist, M.; Kirschbaum, T.; Wei, Y.; Zhou, Y.; Kuchroo, V.L.; Burkett, P.R.; et al. Nociceptor sensory neurons suppress neutrophil and γδTcell responses in bacterial lung infections and lethal pneumonia. Nat. Med. 2018, 24, 417–426. [Google Scholar] [CrossRef]
- Roumier, A.; Béchade, C.; Maroteaux, L. Serotonin and the immune system. In Serotonin the Mediator That Spans Evolution; Pilowsky, P.M., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 181–196. [Google Scholar] [CrossRef]
- Herr, N.; Bode, C.; Duerschmied, D. The effects of serotonin in immune cells. Front. Cardiovasc. Med. 2017, 4, 48. [Google Scholar] [CrossRef] [Green Version]
- Švajger, U.; Rožman, P. Induction of tolerogenic dendritic cells by endogenous biomolecules: An Update. Front. Immunol. 2018, 9, 2482. [Google Scholar] [CrossRef] [Green Version]
- Szabo, A.; Gogolak, P.; Koncz, G.; Foldvari, Z.; Pazmandi, K.; Miltner, M.; Poliska, S.; Bacsi, A.; Djurovic, S.; Rajnavolgyi, E.; et al. Immunomodulatory capacity of the serotonin receptor 5-HT2B in a subset of human dendritic cells. Sci. Rep. 2018, 8, 1765. [Google Scholar] [CrossRef] [Green Version]
- Bhandage, A.K.; Barragan, A. GABAergic signaling by cells of the immune system: More the rule than the exception. Cell Mol. Life Sci. 2021, 78, 5667–5679. [Google Scholar] [CrossRef]
- Dimic, D.; Timotijevic, L.; Nicolic, I.; Zdravkovic, A.; Nikcevic, L.; Dimic, N.; Simeunovic, I. Effects of GABA on lung function in asthmatics after methacholine inhalation. Eur. Respir. J. 2017, 50 (Suppl. S61), PA3574. [Google Scholar] [CrossRef]
- Ngo, D.-H.; Vo, T.S. An updated review on pharmaceutical properties of gamma-aminobutyric acid. Molecules 2019, 24, 2678. [Google Scholar] [CrossRef] [Green Version]
- Sui, P.; Wiesner, D.L.; Jinhao Xu, J.; Zhang, Y.; Lee, J.; Van Dyken, S.; Ashua, A.; Chuyue Yu, C.; Klein, B.S.; Locksley, R.M.; et al. Pulmonary neuroendocrine cells amplify allergic asthma responses. Science 2018, 360. [Google Scholar] [CrossRef] [Green Version]
- Breitinger, U.; Breitinger, H.-G. Modulators of the inhibitory glycine receptor. ACS Chem. Neurosci. 2020, 11, 1706–1725. [Google Scholar] [CrossRef]
- Razak, M.A.; Begum, P.S.; Viswanath, B.; Rajagopal, S. Multifarious beneficial effect of nonessential amino acid, glycine: A review. Oxidative Med. Cell. Longev. 2017, 2017, 1716701. [Google Scholar] [CrossRef]
- Weinberg, J.M.; Venkatachalam, M.A.; Bienholz, A. The role of glycine in regulated cell death. Cell Mol. Life Sci. 2016, 73, 2285–2308. [Google Scholar] [CrossRef] [Green Version]
- Li, T.; Wang, P.; Wang, S.C.; Wang, Y.-F. Approaches mediating oxytocin regulation of the immune system. Front. Immunol. 2017, 7, 693. [Google Scholar] [CrossRef]
- Moberg, K.U.; Handlin, L.; Kendall-Tackett, K.; Petersson, M. Oxytocin is a principal hormone that exerts part of its effects by active fragments. Med.Hypotheses 2019, 133, 109394. [Google Scholar] [CrossRef]
- Wang, Y.-F. Center role of the oxytocin-secreting system in neuroendocrine-immune network revisited. J. Clin. Exp. Neuroimmunol. 2016, 1, 102. [Google Scholar]
- Gotovina, J.; Pranger, C.L.; Jensen, A.N.; Wagner, S.; Kothgassner, O.D.; Mothes-Luksch, N.; Palme, R.; Larenas-Linnemann, D.; Singh, J.; Mosges, R.; et al. Elevated oxytocin and noradrenaline indicate higher stress levels in allergic rhinitis patients: Implications for the skin prick diagnosis in a pilot study. PLoS ONE 2018, 13, e0196879. [Google Scholar] [CrossRef] [Green Version]
- Nagashima, H.; Mahlakoiv, T.; Shih, H.Y.; Davis, F.P.; Meylan, F.; Huang, Y.; Harrison, O.J.; Yao, C.; Mikami, Y.; Urban, J.F., Jr.; et al. Neuropeptide CGRP limits group 2 innate lymphoid cell responses and constrains type 2 inflammation. Immunity 2019, 51, 682–695.e686. [Google Scholar] [CrossRef]
- Wallrapp, A.; Burkett, P.R.; Riesenfeld, S.J.; Kim, S.J.; Christian, E.; Abdulnour, R.E.; Thakore, P.I.; Schnell, A.; Lambden, C.; Herbst, R.; et al. Calcitonin Gene-Related Peptide negatively regulates alarmin-driven type 2 Innate Lymphoid Cell responses. Immunity 2019, 51, 709–723.e706. [Google Scholar] [CrossRef]
- Ochoa-Callejero, L.; Garcia-Sanmartin, J.; Villoslada-Blanco, P.; Iniguez, M.; Perez-Matute, P.; Pujadas, E.; Fowkes, M.E.; Brody, R.; Oteo, J.A.; Martínez, A. Circulating levels of calcitonin gene-related peptide are lower in COVID-19 patients. J. Endocr. Soc. 2021, 5. [Google Scholar] [CrossRef]
- Audrit, K.J.; Delventhal, L.; Aydin, O.; Nassenstein, C. The nervous system of airways and its remodeling in inflammatory lung diseases. Cell Tissue Res. 2017, 367, 571–590. [Google Scholar] [CrossRef]
- Verma, A.K.; Manohar, M.; Venkateshaiah, S.U.; Mishra, A. Neuroendocrine cells derived chemokine vasoactive intestinal polypeptide (VIP) in allergic diseases. Cytokine Growth Factor Rev. 2017, 38, 37–48. [Google Scholar] [CrossRef]
- Sallaberry, C.A.; Astern, L. The endocannabinoid system, our universal regulator. J. Young Investig. 2018, 34, 48–55. [Google Scholar] [CrossRef] [Green Version]
- Dhital, S.; Stokes, J.V.; Park, N.; Seo, K.S.; Kaplan, B.L.F. Cannabidiol (CBD) induces functional Tregs in response to low-level T cell activation. Cell. Immunol. 2017, 312, 25–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garcia-Garcia, L.; Olle, L.; Martin, M.; Roca-Ferrer, J.; Munoz-Cano, R. Adenosine signaling in mast cells and allergic diseases. Int. J. Mol. Sci. 2021, 22, 5203. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, Y.; Yanase, Y.; Irifuku, R.; Ishii, K.; Kawaguchi, T.; Takahagi, S.; Hide, I.; Hide, M. The role of adenosine for IgE receptor-dependent degranulation of human peripheral basophils and skin mast cells. Allergol. Int. 2018, 67, 524–526. [Google Scholar] [CrossRef] [PubMed]
- Silva-Vilches, C.; Ring, S.; Mahnke, K. ATP and its metabolite adenosine as regulators of dendritic cell activity. Front. Immunol. 2018, 9, 2581. [Google Scholar] [CrossRef]
- Sha, J.; Meng, C.; Li, L.; Xiu, Q.; Zhu, D. Correlation of serum β-Endorphin and the quality of life in allergic rhinitis. Dis.Markers 2016, 2016, 2025418. [Google Scholar] [CrossRef] [Green Version]
- Shrihari, T.G. Endorphins—A natural healer. J. Cancer Prev. Curr. Res. 2018, 9, 223–234. [Google Scholar] [CrossRef]
- Liang, X.; Liu, R.; Chen, C.; Ji, F.; Li, T. Opioid system modulates the immune function: A review. Transl. Perioper. Pain Med. 2016, 1, 5–13. [Google Scholar]
- Manti, S.; Brown, P.; Perez, M.K.; Piedimonte, G. The role of neurotrophins in inflammation and allergy. Vitam. Horm. 2017, 104, 313–341. [Google Scholar] [CrossRef]
- Prakash, Y.S.; Martin, R.J. Brain-derived neurotrophic factor in the airways. Pharmacol. Ther. 2014, 143, 74–86. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, T.; Fajt, M.L.; Trudeau, J.B.; Voraphani, N.; Hu, H.; Zhou, X.; Holguin, F.; Wenze, S.E. Brain-derived neurotrophic factor expression in asthma. Association with severity and type 2 inflammatory processes. Am. J. Respir. Cell Mol. Biol. 2015, 53, 844–852. [Google Scholar] [CrossRef] [Green Version]
- Kannampalli, P.; Sengupta, J.N. Role of principal ionotropic and metabotropic receptors in visceral pain. J. Neurogastroenterol. Motil. 2015, 21, 147–158. [Google Scholar] [CrossRef] [Green Version]
- Foster, S.R.; Hauser, A.S.; Vedel, L.; Strachan, R.T.; Huang, X.-P.; Gavin, A.C.; Shah, S.D.; Nayak, A.P.; Haugaard-Kedstro, L.M.; Penn, R.B.; et al. Discovery of human signaling systems: Pairing peptides to G protein-coupled receptors. Cell 2019, 179, 895–908.e21. [Google Scholar] [CrossRef] [Green Version]
- Breiteneder, H.; Diamant, Z.; Eiwegger, T.; Fokkens, W.J.; Traidl-Hoffmann, C.; Nadeau, K.; O’Hehir, R.E.; O’Mahony, L.; Pfaar, O.; Torres, M.J.; et al. Future research trends in understanding the mechanisms underlying allergic diseases for improved patient care. Allergy 2019, 74, 2293–2311. [Google Scholar] [CrossRef]
- Liu, N.; Wang, J.; Zhang, M. Identification of urinary peptides associated with allergic rhinitis. Rev. Française D’allergologie 2021, 61, 583–591. [Google Scholar] [CrossRef]
- Vizuet-de-Rueda, J.C.; Montero-Vargas, J.M.; Galván-Morales, M.A.; Porras-Gutiérrez-de-Velasco, R.; Teran, L.M. Current insights on the impact of proteomics in respiratory allergies. Int. J. Mol. Sci. 2022, 23, 5703. [Google Scholar] [CrossRef]
- Tomazic, P.V.; Darnhofer, B.; Birner-Gruenberger, R. Nasal mucus proteome and its involvement in allergic rhinitis. Expert Rev. Proteom. 2020, 17, 191–199. [Google Scholar] [CrossRef] [Green Version]
- Le, D.D. Neuroimmune Interactions in Allergic Airway Diseases: Studies in Mouse Models and Humans. Ph.D. Thesis, Abteilungfür Transplantations- und Infektionsimmunologie der MedizinischenFakultät der Universität des Saarlandes, Homburg, Germany, 2017; 100p. [Google Scholar]
- Husna, S.M.N.; Tan, H.-T.T.; MdShukri, N.; Ashari, N.S.M.; Wong, K.K. Nasal epithelial barrier integrity and tight junctions disruption in allergic rhinitis: Overview and pathogenic insights. Front. Immunol. 2021, 12, 663626. [Google Scholar] [CrossRef]
- Deprez, M.; Zaragosi, L.E.; Truchi, M.; Becavin, C.; Ruiz García, S.; Marie-Jeanne, A.; Plaisant, M.; Magnone, V.; Lebrigand, K.; Abelanet, S.; et al. A single-cell atlas of the human healthy airways. Am. J. Respir. Crit. Care Med. 2020, 202, 1636–1645. [Google Scholar] [CrossRef]
- Podlesnaja, M.; Pilmane, M.; Sumeraga, G. Cytokines, proliferation markers, antimicrobial factors and neuropeptide-containing innervation in human nasal mucosa after rhinoseptoplasty procedure. Med. Sci. 2021, 9, 25. [Google Scholar] [CrossRef]
- Krohn, I.K.; Sven, F.; Seys, G.L.; Jonckheere, A.-C.; de Casterlé, I.D.; Ceuppens, J.L.; Steelant, B.; Hellings, P.W. Nasal epithelial barrier dysfunction increases sensitization and mast cell degranulation in the absence of allergic inflammation. Allergy 2020, 75, 1155–1164. [Google Scholar] [CrossRef]
- Kohanski, M.A.; Workman, A.D.; Patel, N.N.; Hung, L.Y.; Shtraks, J.P.; Chen, B.; Blasetti, M.; Doghramji, L.; Kennedy, D.W.; Adappa, N.D. Solitary chemosensory cells are a primary epithelial source of IL-25 in patients with chronic rhinosinusitis with nasal polyps. J. Allergy Clin. Immunol. 2018, 142, 460–469.e7. [Google Scholar] [CrossRef]
- Bousquet, J.; Anto, J.M.; Bachert, C.; Baiardini, I.; Bosnic-Anticevich, S.; Canonica, G.W.; Melén, E.; Palomares, O.; Scadding, G.K.; Togias, A.; et al. Allergic rhinitis. Nat. Rev. Dis. Primers 2020, 6, 95. [Google Scholar] [CrossRef]
- Passali, D.; Cingi, C.; Staffa, P.; Passali, F.; Muluk, N.B.; Bellussi, M.L. The international study of the allergic rhinitis survey: Outcomes from 4 geographical regions. Asia Pac. Allergy 2018, 8, e7. [Google Scholar] [CrossRef] [Green Version]
- Romano, M.R.; James, S.; Farrington, E.; Perry, R.; Elliott, L. The impact of perennial allergic rhinitis with/without allergic asthma on sleep, work and activity level. Allergy Asthma Clin. Immunol. 2020, 16, 12. [Google Scholar] [CrossRef]
- Eguiluz-Gracia, I.; Perez-Sanchez, N.; Bogas, G.; Campo, P.; Rondon, C. How to diagnose and treat local allergic rhinitis: A challenge for clinicians. J. Clin. Med. 2019, 8, 1062. [Google Scholar] [CrossRef] [Green Version]
- Maoz-Segal, R.; Machnes-Maayan, D.; Veksler-Offengenden, I.; Frizinsky, S.; Hajyahia, S.; Agmon-Levin, N. Local allergic rhinitis: An old story but a new entity. In Rhinosinusitis; Gendeh, B.S., Turkalj, M., Eds.; IntechOpen: London, UK, 2019; pp. 1–9. [Google Scholar] [CrossRef] [Green Version]
- Campo, P.; Eguiluz-Gracia, I.; Plaza-Serón, M.C.; Salas, M.; Rodríguez, M.J.; Perez-Sanchez, N.; González, M.; Molina, A.; Mayorga, C.; Torres, M.J.; et al. Bronchial asthma triggered by house dust mites in patients with local allergic rhinitis. Allergy 2019, 74, 1502–1510. [Google Scholar] [CrossRef]
- Yamana, Y.; Fukuda, K.; Ko, R.; Uchio, E. Local allergic conjunctivitis: A phenotype of allergic conjunctivitis. Int. Ophthalmol. 2019, 39, 2539–2544. [Google Scholar] [CrossRef] [Green Version]
- Testera-Montes, A.; Salas, M.; Palomares, F.; Ariza, A.; Torres, M.J.; Rondón, C.; Eguiluz-Gracia, I. Local respiratory allergy: From rhinitis phenotype to disease spectrum. Front. Immunol. 2021, 12, 691964. [Google Scholar] [CrossRef]
- Bellanti, J.A.; Settipane, R.A. The atopic disorders and atopy… “strange diseases” now better defined! Allergy Asthma Proc. 2017, 38, 241–242. [Google Scholar] [CrossRef] [Green Version]
- Bellinger, D.L.; Lorton, D. Autonomic regulation of cellular immune function. Auton. Neurosci. 2014, 182, 15–41. [Google Scholar] [CrossRef] [PubMed]
- Padro, C.J.; Sanders, V.M. Neuroendocrine regulation of inflammation. Semin. Immunol. 2014, 26, 357–368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Collin, M.; Bigley, V. Human dendritic cell subsets: An update. Immunology 2018, 154, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Takenaka, M.C.; Guereschi, M.G.; Basso, A.S. Neuroimmune interactions: Dendritic cell modulation by the sympathetic nervous system. Semin. Immunopathol. 2017, 39, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Holst, K.; Guseva, D.; Schindler, S.; Sixt, M.; Braun, A.; Chopra, H.; Pabst, O.; Ponimaskin, E. The serotonin receptor 5-HT7R regulates the morphology and migratory properties of dendritic cells. J. Cell Sci. 2015, 128, 2866–2880. [Google Scholar] [CrossRef] [Green Version]
- Ross, E.A.; Devitt, A.; Johnson, J.R. Macrophages: The good, the bad, and the gluttony. Front. Immunol. 2021, 12, 708186. [Google Scholar] [CrossRef]
- Abdelaziz, M.H.; Abdelwahab, S.F.; Wan, J.; Cai, W.; Huixuan, W.; Jianjun, C.; Kumar, K.D.; Vasudevan, A.; Sadek, S.; Su, Z.; et al. Alternatively activated macrophages; a double-edged sword in allergic asthma. J. Transl. Med. 2020, 18, 58. [Google Scholar] [CrossRef]
- Shrivastava, R.; Shukla, N. Attributes of alternatively activated (M2) macrophages. Life Sci. 2019, 224, 222–231. [Google Scholar] [CrossRef]
- Yao, A.; Wilson, J.A.; Ball, S.L. Autonomic nervous system dysfunction and sinonasal symptoms. Allergy Rhinol. 2018, 9, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Zheng, H.; Zhang, Y.; Pan, J.; Liu, N.; Qin, L.; Liu, M.; Wang, T. The role of type 2 innate lymphoid cells in allergic diseases. Front. Immunol. 2021, 12, 586078. [Google Scholar] [CrossRef]
- Pasha, M.A.; Patel, G.; Hopp, R.; Yang, Q. Role of innate lymphoid cells in allergic diseases. Allergy Asthma Proc. 2019, 40, 138–145. [Google Scholar] [CrossRef]
- Huber, M.; Cato, A.C.B.; Ainooson, G.K.; Freichel, M.; Tsvilovskyy, V.; Jessberger, R.; Riedlinger, E.; Sommerhoff, C.P.; Bischoff, S.C. Regulation of the pleiotropic effects of tissue-resident mast cells. J. Allergy Clin. Immunol. 2019, 144, S31–S45. [Google Scholar] [CrossRef] [Green Version]
- Varricchi, G.; Rossi, F.W.; Galdiero, M.R.; Granata, F.; Criscuolo, G.; Spadaro, G.; de Paulis, A.; Marone, G. Physiological roles of mast cells: Collegium InternationaleAllergologicum Update 2019. Int. Arch. Allergy Immunol. 2019, 179, 247–261. [Google Scholar] [CrossRef]
- Beck, S.C.; Wilding, T.; Buka, R.J.; Baretto, R.L.; Huissoon, A.P.; Krishna, M.T. Biomarkers in human anaphylaxis: A critical appraisal of current evidence and perspectives. Front. Immunol. 2019, 10, 494. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Shi, X.; Li, X.; Zou, J.; Zhou, C.; Liu, W.; Shao, H.; Chen, H.; Shi, L. Neurotransmitter and neuropeptide regulation of mast cell function: A systematic review. J. Neuroinflamm. 2020, 17, 356. [Google Scholar] [CrossRef]
- Ramirez, G.A.; Yacoub, M.-R.; Ripa, M.; Mannina, D.; Gariddi, A.; Saporiti, N.; Ciceri, F.; Castagna, A.; Giselda Colombo, G.; Dagna, D. Eosinophils from physiology to disease: A comprehensive review. BioMed Res. Int. 2018, 9095275. [Google Scholar] [CrossRef] [Green Version]
- Fettrelet, T.; Gigon, L.; Karaulov, A.; Yousefi, S.; Simon, H.-U. The enigma of eosinophil degranulation. Int. J. Mol. Sci. 2021, 22, 7091. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, Z.; Wen, X.; Huang, G.; Nian, S.; Li, L.; Guo, X.; Ye, Y.; Yuan, Q. The onset, development and pathogenesis of severe neutrophilic asthma. Immunol. Cell Biol. 2022, 100, 144–159. [Google Scholar] [CrossRef]
- Lugogo, N.L.; Akuthota, P. Type 2 biomarkers in asthma: Yet another reflection of heterogeneity. J. Allergy Clin. Immunol. Pract 2021, 9, 1276–1277. [Google Scholar] [CrossRef]
- Polak, D.; Hafner, C.; Briza, P.; Kitzmuller, C.; Elbe-Burger, A.; Samadi, N.; Gschwandtner, M.; Pfützner, W.; Zlabinger, G.J.; Jahn-Schmid, B.; et al. A novel role for neutrophils in IgE-mediated allergy: Evidence for antigen presentation in late-phase reactions. J. Allergy Clin. Immunol. 2019, 143, 1143–1152. [Google Scholar] [CrossRef] [Green Version]

| Counterparts | Organs/Tissues | Cells | Particular Phenomena |
|---|---|---|---|
| Neuronal | CNS, spinal cord, and non-neuronal subsystem | Neurons and nerve fibers (somatosensory, viscerosensory, sympathetic, parasympathetic, and enteric) [18,19] Neuroglia Neuron-derived extracellular vesicles [20] | “Neurogenic” inflammation [18,21] |
| Neuroendocrine | Hypothalamus, pituitary gland, and peripheral endocrine glands (i.e., HPA axis on the whole) | Endocrine gland cells NEC [11,22,23] | General adaptation syndrome [16] |
| Immune | Primary, secondary, and tertiary organs | Immune cells (lymphocytes and non-lymphoid immune cells) Immune cell-derived extracellular vesicles [24] Barrier epithelium’s cells (ciliated, deuterosomal, goblet, club, basal, suprabasal, tuft cells, ionocytes, etc. [7,10] | Innate immunity Pathways of adaptive immune responses Immune (allergen) tolerance [25,26] |
| Neuro Molecule | Some Receptors | Category and Prevalent Source | Predominant Activity in Relation to Allergen Tolerance | References |
|---|---|---|---|---|
| Acetylcholine (ACh) | a7nAchR; M1AchR-M4AchR | Excitatory neurotransmitter (parasympathetic neurons, NEC, lymphocytes, monocytes) | Pro-immunogenic | [18,22,34,35,36,37] |
| Dopamine (DA) | D1-D5 | “Critical” excitatory and inhibitory neurotransmitter [2] (sympathetic neurons, lymphocytes, dendritic cells (DCs), macrophages, neutrophils) | Pro-immunogenic and ambivalent | [2,27,29,38,39] |
| L-glutamate (L-Glu) | mGluRs-iGluRs | “Critical” excitatory neurotransmitter [2] (neurons, neuroglia, and other non-neuronal cells) | Pro-immunogenic | [2,27,28,40,41] |
| Histamine | H1-H4 | Excitatory neurotransmitter (neurons, mast cells, basophils, T cells, enterochromaffin tissue) | Pro-immunogenic | [42,43,44,45,46] |
| Arginine-vasopressin | AV1AR, AV1BR, AV2R | Neurohormone, or a peptide hormone (the posterior pituitary gland) | Pro-immunogenic | [47,48,49] |
| Melatonin | MT1-MT3 | Neurohormone, or a peptide hormone (the pineal gland) | Pro-immunogenic | [50,51,52,53] |
| Substance P (SP) | NK1R-NK3R | “Critical” neuropeptide [2] (sensory neurons, microglia, lymphocytes, DCs, macrophages, eosinophils) | Pro-immunogenic | [5,6,13,27,54,55,56,57] |
| Neuromedin U (NMU) | NMUR1-NMUR2 | Neuropeptide (parasympathetic sensory neurons, NEC) | Pro-immunogenic | [5,6,13,17,22,58,59,60,61,62] |
| Norepinephrine (NE) | β2AR; αAR | Excitatory neurotransmitter (sympathetic neurons, adrenal medulla, lymphocytes, NK cells, monocytes, macrophages, NEC) | Protolerogenic | [22,27,63,64,65,66] |
| Serotonin (5-hydroxytryptamine) | 5-HT1-5-HT7 | “Critical” inhibitory neurotransmitter [2] (central and enteric neurons, enterochromaffin tissue, NEC) | Protolerogenic | [2,27,67,68,69,70] |
| γ Aminobutyric acid (GABA) | GABAA-GABAB | Inhibitory neurotransmitter (neurons, T cells, macrophages, NEC) | Protolerogenic | [27,71,72,73,74] |
| Glycine | GlyRs | Inhibitory neurotransmitter (various neurons) | Protolerogenic | [75,76,77] |
| Oxytocin | OXTR | Neurohormone, or a peptide hormone (the posterior pituitary gland) | Protolerogenic | [78,79,80,81] |
| Calcitonin-gene-related peptide (CGRP) | CLRs | Neuropeptide (sensory neurons, T cells, B cells, NEC, the thyroid gland) | Protolerogenic and ambivalent | [3,6,17,18,22,27,60,82,83,84] |
| Vasoactive intestinal peptide (VIP) | VPAC1-VPAC2 | Neuropeptide (parasympathetic sensory neurons, NEC, the gut) | Protolerogenic and ambivalent | [5,6,13,17,18,22,27,60,85,86] |
| Endocannabinoids | CB1-CB2 | Atypical neurotransmitters (various neurons) | Protolerogenic | [27,87,88] |
| Adenosine ATP | A1-A3; P1, P2X, P2Y | Atypical neurotransmitters (various cells, a DAMP) | Protolerogenic | [27,89,90,91] |
| Endorphins | μ, δ, κ opioid Rs | Peptides (the pituitary gland, thymus, lymphocytes) | Protolerogenic | [27,92,93,94] |
| Nerve growth factor (NGF) Brain-derived neurotrophic factor (BDNF) Neurotrophin 3 Neurotrophin 4 | TrkA-TrkC; p75NTR | Neurotrophins (sensory neurons, B cells, monocytes, mast cells, eosinophils, keratinocytes, smooth-muscle cells, NEC) | Pro-immunogenic or protolerogenic | [18,32,95,96,97] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klimov, V.; Cherevko, N.; Klimov, A.; Novikov, P. Neuronal-Immune Cell Units in Allergic Inflammation in the Nose. Int. J. Mol. Sci. 2022, 23, 6938. https://doi.org/10.3390/ijms23136938
Klimov V, Cherevko N, Klimov A, Novikov P. Neuronal-Immune Cell Units in Allergic Inflammation in the Nose. International Journal of Molecular Sciences. 2022; 23(13):6938. https://doi.org/10.3390/ijms23136938
Chicago/Turabian StyleKlimov, Vladimir, Natalia Cherevko, Andrew Klimov, and Pavel Novikov. 2022. "Neuronal-Immune Cell Units in Allergic Inflammation in the Nose" International Journal of Molecular Sciences 23, no. 13: 6938. https://doi.org/10.3390/ijms23136938
APA StyleKlimov, V., Cherevko, N., Klimov, A., & Novikov, P. (2022). Neuronal-Immune Cell Units in Allergic Inflammation in the Nose. International Journal of Molecular Sciences, 23(13), 6938. https://doi.org/10.3390/ijms23136938





