Hypomethylation of CLDN4 Gene Promoter Is Associated with Malignant Phenotype in Urinary Bladder Cancer
Abstract
:1. Introduction
2. Results
2.1. DNA Methylation in the CLDN4 Promoter in BUCs
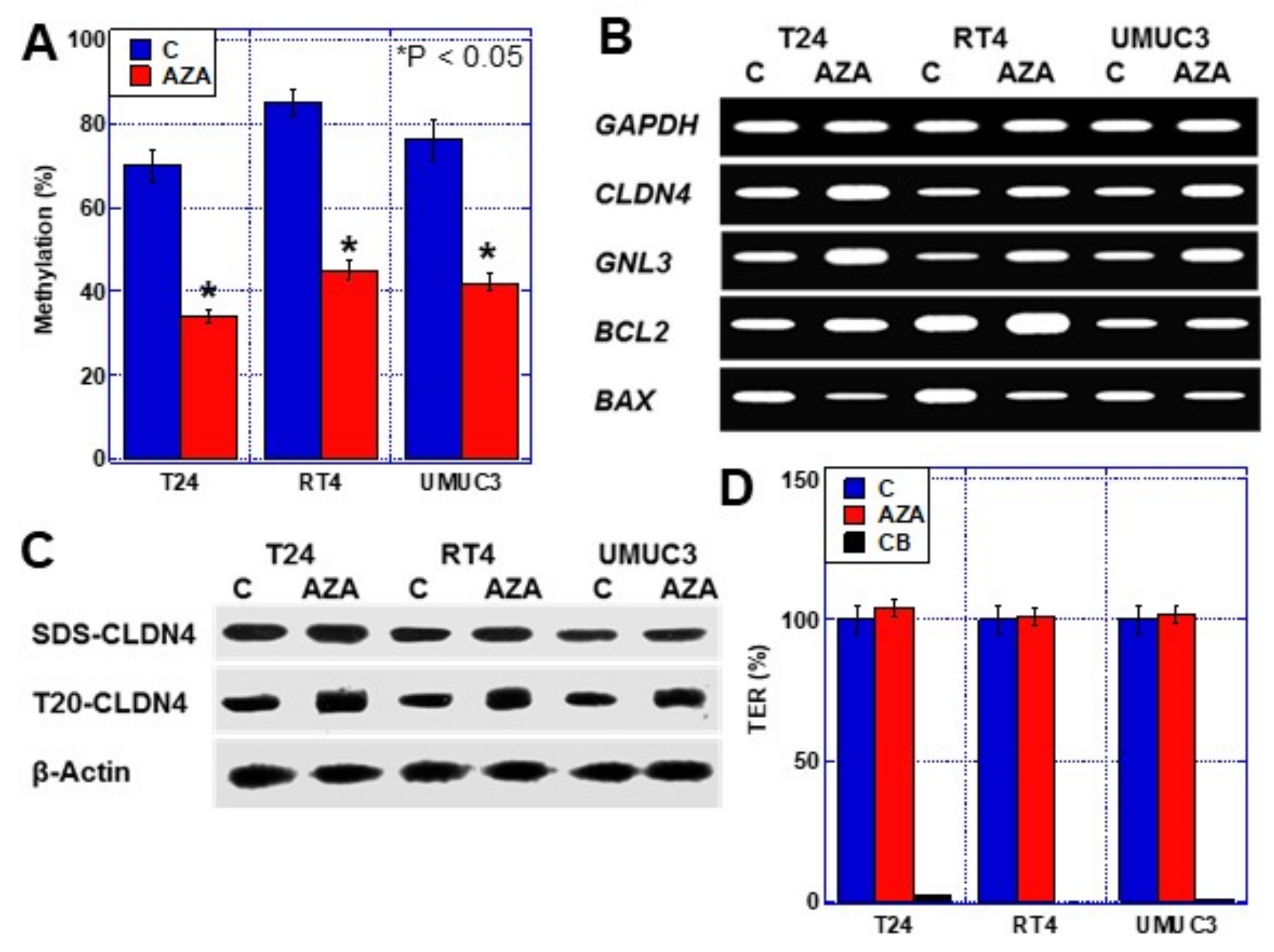
2.2. CLDN4 Promoter DNA Methylation and Gene Expression
2.3. Effects of CLDN4 Promoter DNA Demethylation
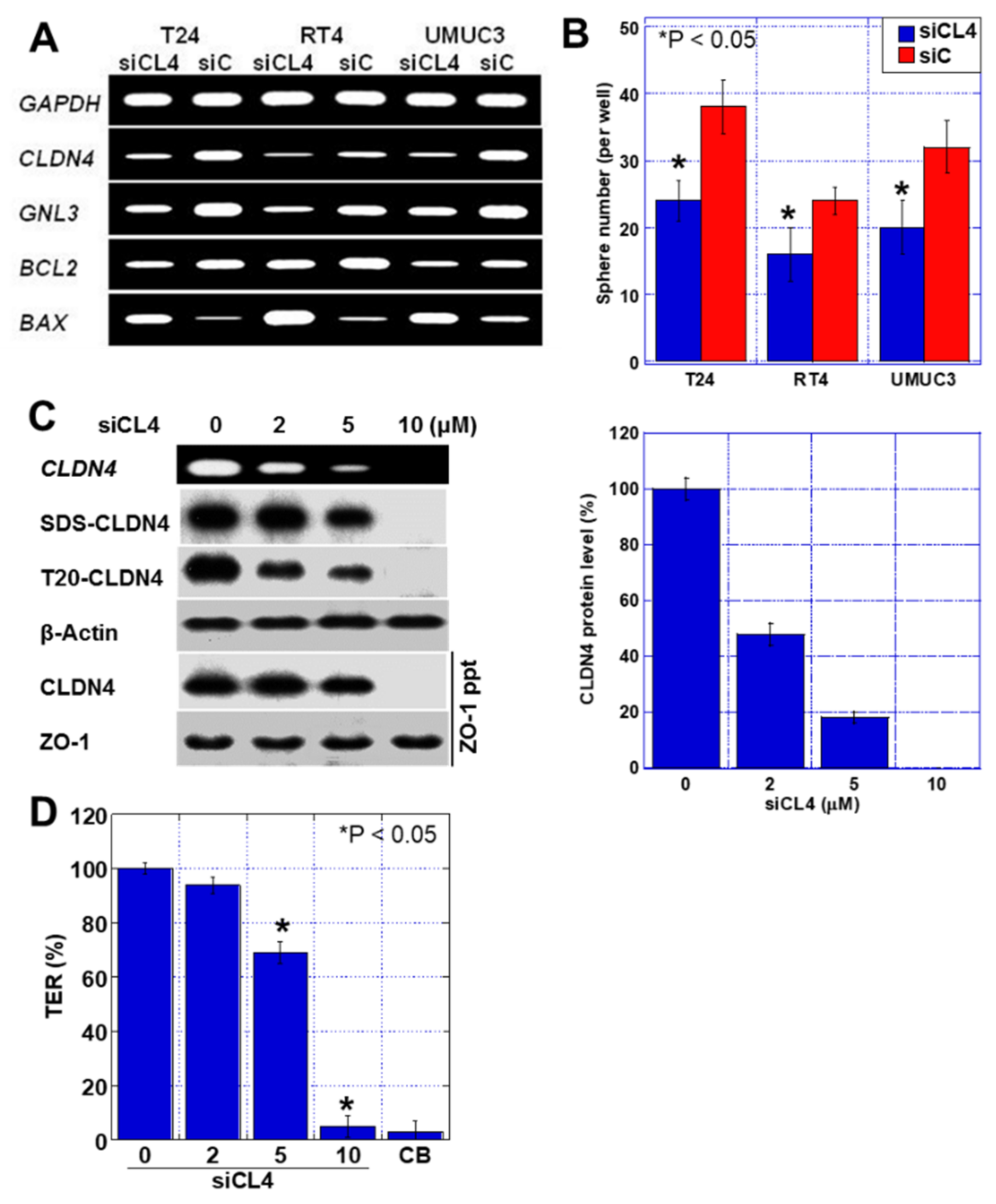
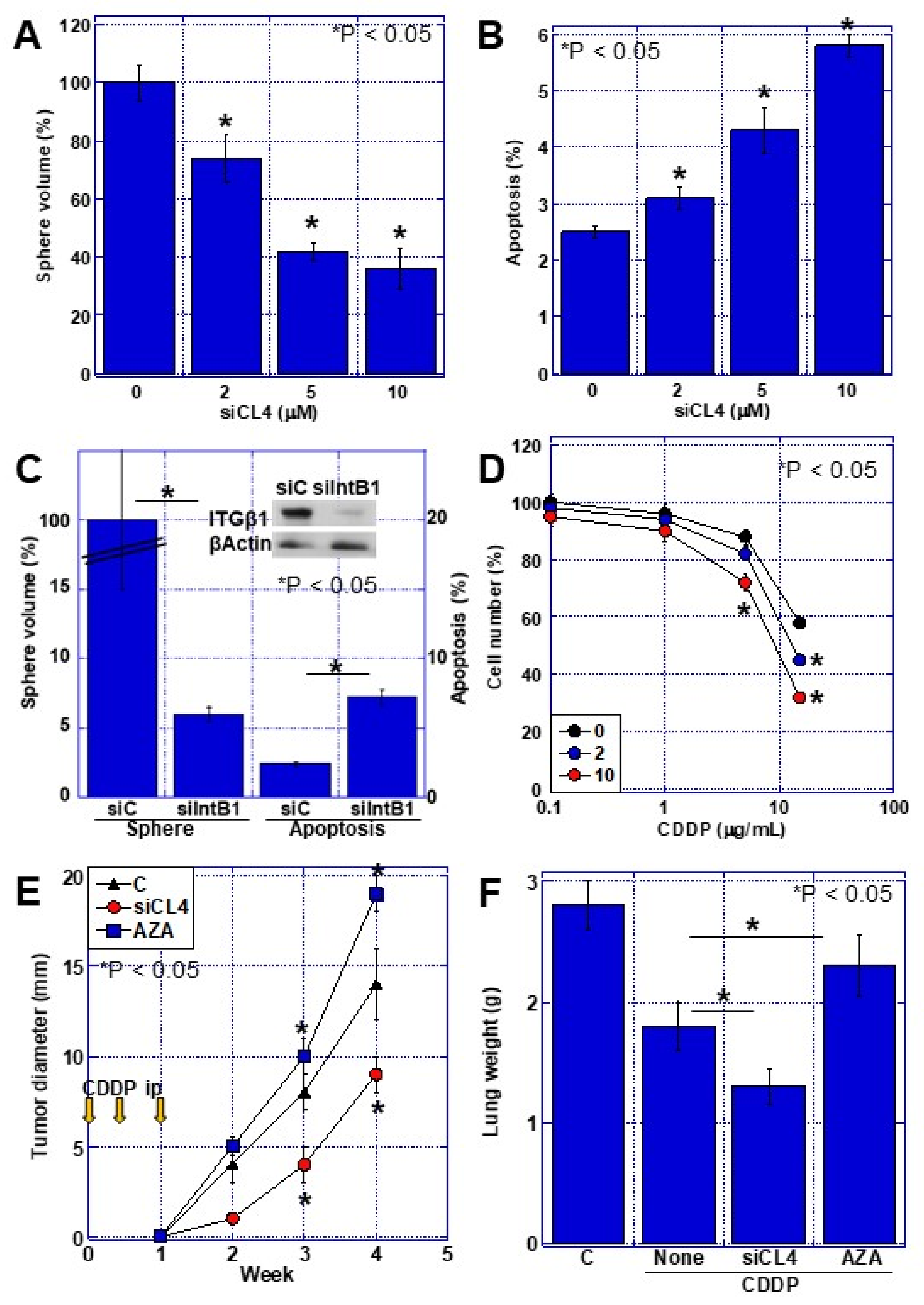
2.4. Effects of CLDN4 Knockdown
2.5. Non-Tight Junction CLDN4 Binding to Integrinβ1
2.6. Effects of Integrin Activation by CLDN4
3. Discussion
4. Materials and Methods
4.1. Surgical Specimens
4.2. Cells and Reagents
4.3. DNA Extraction
4.4. The Primer Set for Pyrosequencing
4.5. Bisulfite Pyrosequencing
4.6. Small Interfering RNA
4.7. Enzyme-Linked Immunosorbent Assay (ELISA)
4.8. Immunohistochemistry
4.9. Protein Extraction
4.10. Immunoblot Analysis
4.11. Immunoprecipitation
4.12. Affinity Assay
4.13. Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
4.14. Sphere Assay
4.15. Apoptosis Assay
4.16. Transepithelial Electroresistance (TER) Assay
4.17. Animals
4.18. Animal Tumor Models
4.19. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TJ | tight junction |
| CLDN4 | claudin-4 |
| BUC | bladder urothelial carcinoma |
| EMT | epithelial-mesenchymal transition |
| AZA | aza-2′-deoxycytidine |
| SDS | sodium dodecyl sulfate |
| T20 | tween 20 |
| TER | transepithelial electric resistance |
| PFO | perfluoro-octanoic acid |
| CDDP | cisplatin |
References
- Turksen, K.; Troy, T.C. Junctions gone bad: Claudins and loss of the barrier in cancer. Biochim. Biophys. Acta 2011, 1816, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Escudero-Esparza, A.; Jiang, W.G.; Martin, T.A. The Claudin family and its role in cancer and metastasis. Front. Biosci. 2011, 16, 1069–1083. [Google Scholar] [CrossRef] [PubMed]
- Kuwada, M.; Chihara, Y.; Luo, Y.; Li, X.; Nishiguchi, Y.; Fujiwara, R.; Sasaki, T.; Fujii, K.; Ohmori, H.; Fujimoto, K.; et al. Pro-chemotherapeutic effects of antibody against extracellular domain of claudin-4 in bladder cancer. Cancer Lett. 2015, 369, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara-Tani, R.; Sasaki, T.; Luo, Y.; Goto, K.; Kawahara, I.; Nishiguchi, Y.; Kishi, S.; Mori, S.; Ohmori, H.; Kondoh, M.; et al. Anti-claudin-4 extracellular domain antibody enhances the antitumoral effects of chemotherapeutic and antibody drugs in colorectal cancer. Oncotarget 2018, 9, 37367–37378. [Google Scholar] [CrossRef]
- Nishiguchi, Y.; Fujiwara-Tani, R.; Sasaki, T.; Luo, Y.; Ohmori, H.; Kishi, S.; Mori, S.; Goto, K.; Yasui, W.; Sho, M.; et al. Targeting claudin-4 enhances CDDP-chemosensitivity in gastric cancer. Oncotarget 2019, 10, 2189–2202. [Google Scholar] [CrossRef]
- Sasaki, T.; Fujiwara-Tani, R.; Kishi, S.; Mori, S.; Luo, Y.; Ohmori, H.; Kawahara, I.; Goto, K.; Nishiguchi, Y.; Mori, T.; et al. Targeting claudin-4 enhances chemosensitivity of pancreatic ductal carcinomas. Cancer Med. 2019, 8, 6700–6708. [Google Scholar] [CrossRef]
- Swisshelm, K.; Macek, R.; Kubbies, M. Role of claudins in tumorigenesis. Adv. Drug Deliv. Rev. 2005, 57, 919–928. [Google Scholar] [CrossRef]
- Kwon, M.J. Emerging roles of claudins in human cancer. Int. J. Mol. Sci. 2013, 14, 18148–18180. [Google Scholar] [CrossRef]
- Fumarola, S.; Cecati, M.; Sartini, D.; Ferretti, G.; Milanese, G.; Galosi, A.B.; Pozzi, V.; Campagna, R.; Morresi, C.; Emanuelli, M.; et al. Bladder Cancer Chemosensitivity is Affected by Paraoxonase-2 Expression. Antioxidants 2020, 9, 175. [Google Scholar] [CrossRef]
- Cavalli, G.; Heard, E. Advances in epigenetics link genetics to the environment and disease. Nature 2019, 571, 489–499. [Google Scholar] [CrossRef]
- Feinberg, A.P. The Key Role of Epigenetics in Human Disease Prevention and Mitigation. N. Eng. J. Med. 2018, 378, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Kanwal, R.; Gupta, K.; Gupta, S. Cancer epigenetics: An introduction. Methods Mol. Biol. 2015, 1238, 3–25. [Google Scholar] [PubMed]
- Nebbioso, A.; Tambaro, F.P.; Dell’Aversana, C.; Altucci, L. Cancer epigenetics: Moving forward. PLoS Genet. 2018, 14, e1007362. [Google Scholar] [CrossRef] [PubMed]
- Kwon, M.J.; Kim, S.H.; Jeong, H.M.; Jung, H.S.; Kim, S.S.; Lee, J.E.; Gye, M.C.; Erkin, O.C.; Koh, S.S.; Choi, Y.L.; et al. Claudin-4 overexpression is associated with epigenetic derepression in gastric carcinoma. Lab. Investig. 2011, 91, 1652–1667. [Google Scholar] [CrossRef]
- Boireau, S.; Buchert, M.; Samuel, M.S.; Pannequin, J.; Ryan, J.L.; Choquet, A.; Chapuis, H.; Rebillard, X.; Avances, C.; Ernst, M.; et al. DNA-methylation-dependent alterations of claudin-4 expression in human bladder carcinoma. Carcinogenesis 2007, 28, 246–258. [Google Scholar] [CrossRef]
- Hahn-Stromberg, V.; Askari, S.; Ahmad, A.; Befekadu, R.; Nilsson, T.K. Expression of claudin 1, claudin 4, and claudin 7 in colorectal cancer and its relation with CLDN DNA methylation patterns. Tumour Biol. 2017, 39, 1010428317697569. [Google Scholar] [CrossRef]
- Litkouhi, B.; Kwong, J.; Lo, C.M.; Smedley, J.G., 3rd; McClane, B.A.; Aponte, M.; Gao, Z.; Sarno, J.L.; Hinners, J.; Welch, W.R.; et al. Claudin-4 overexpression in epithelial ovarian cancer is associated with hypomethylation and is a potential target for modulation of tight junction barrier function using a C-terminal fragment of Clostridium perfringens enterotoxin. Neoplasia 2007, 9, 304–314. [Google Scholar] [CrossRef]
- Edge, S.; Byrd, D.R.; Compton, C.C.; Fritz, A.G.; Greene, F.L.; Trotti, A. AJCC Cancer Staging Manual, 7th ed.; Springer: New York, NY, USA, 2010. [Google Scholar]
- Lu, Z.; Kim, D.H.; Fan, J.; Lu, Q.; Verbanac, K.; Ding, L.; Renegar, R.; Chen, Y.H. A non-tight junction function of claudin-7-Interaction with integrin signaling in suppressing lung cancer cell proliferation and detachment. Mol. Cancer 2015, 14, 120. [Google Scholar] [CrossRef]
- Hasegawa, K.; Wakino, S.; Simic, P.; Sakamaki, Y.; Minakuchi, H.; Fujimura, K.; Hosoya, K.; Komatsu, M.; Kaneko, Y.; Kanda, T.; et al. Renal tubular Sirt1 attenuates diabetic albuminuria by epigenetically suppressing Claudin-1 overexpression in podocytes. Nat. Med. 2013, 19, 1496–1504. [Google Scholar] [CrossRef]
- Di Cello, F.; Cope, L.; Li, H.; Jeschke, J.; Wang, W.; Baylin, S.B.; Zahnow, C.A. Methylation of the claudin 1 promoter is associated with loss of expression in estrogen receptor positive breast cancer. PLoS ONE 2013, 8, e68630. [Google Scholar]
- Hichino, A.; Okamoto, M.; Taga, S.; Akizuki, R.; Endo, S.; Matsunaga, T.; Ikari, A. Down-regulation of Claudin-2 Expression and Proliferation by Epigenetic Inhibitors in Human Lung Adenocarcinoma A549 Cells. J. Biol. Chem. 2017, 292, 2411–2421. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yu, W.; Chen, S.; Chen, Y.; Chen, L.; Zhang, S. Methylation of the claudin3 promoter predicts the prognosis of advanced gastric adenocarcinoma. Oncol. Rep. 2018, 40, 49–60. [Google Scholar] [PubMed]
- Kwon, M.J.; Kim, S.S.; Choi, Y.L.; Jung, H.S.; Balch, C.; Kim, S.H.; Song, Y.S.; Marquez, V.E.; Nephew, K.P.; Shin, Y.K. Derepression of CLDN3 and CLDN4 during ovarian tumorigenesis is associated with loss of repressive histone modifications. Carcinogenesis 2010, 31, 974–983. [Google Scholar] [CrossRef] [PubMed]
- Honda, H.; Pazin, M.J.; D’Souza, T.; Ji, H.; Morin, P.J. Regulation of the CLDN3 gene in ovarian cancer cells. Cancer Biol. Ther. 2007, 6, 1733–1742. [Google Scholar] [CrossRef]
- Nishikiori, N.; Sawada, N.; Ohguro, H. Prevention of murine experimental corneal trauma by epigenetic events regulating claudin 6 and claudin 9. JPN J. Ophthalmol. 2008, 52, 195–203. [Google Scholar] [CrossRef]
- Kudinov, A.E.; Deneka, A.; Nikonova, A.S.; Beck, T.N.; Ahn, Y.H.; Liu, X.; Martinez, C.F.; Schultz, F.A.; Reynolds, S.; Yang, D.H.; et al. Musashi-2 (MSI2) supports TGF-β signaling and inhibits claudins to promote non-small cell lung cancer (NSCLC) metastasis. Proc. Natl. Acad. Sci. USA 2016, 113, 6955–6960. [Google Scholar] [CrossRef]
- Li, J.; Zhou, C.; Ni, S.; Wang, S.; Ni, C.; Yang, P.; Ye, M. Methylated claudin-11 associated with metastasis and poor survival of colorectal cancer. Oncotarget 2017, 8, 96249–96262. [Google Scholar] [CrossRef]
- Kyuno, D.; Yamaguchi, H.; Ito, T.; Kono, T.; Kimura, Y.; Imamura, M.; Konno, T.; Hirata, K.; Sawada, N.; Kojima, T. Targeting tight junctions during epithelial to mesenchymal transition in human pancreatic cancer. World J. Gastroenterol. 2014, 20, 10813–10824. [Google Scholar] [CrossRef]
- Nakashima, C.; Yamamoto, K.; Kishi, S.; Sasaki, T.; Ohmori, H.; Fujiwara-Tani, R.; Mori, S.; Kawahara, I.; Nishiguchi, Y.; Mori, T.; et al. Clostridium perfringens enterotoxin induces claudin-4 to activate YAP in oral squamous cell carcinomas. Oncotarget 2020, 11, 309–321. [Google Scholar] [CrossRef]
- Fujiwara-Tani, R.; Fujii, K.; Mori, S.; Kishi, S.; Sasaki, T.; Ohmori, H.; Nakashima, C.; Kawahara, I.; Nishiguchi, Y.; Mori, T.; et al. Role of Clostridium perfringens Enterotoxin on YAP Activation in Colonic Sessile Serrated Adenoma/Polyps with Dysplasia. Int. J. Mol. Sci. 2020, 21, 3840. [Google Scholar] [CrossRef]
- Fredriksson, K.; Van Itallie, C.M.; Aponte, A.; Gucek, M.; Tietgens, A.J.; Anderson, J.M. Proteomic analysis of proteins surrounding occludin and claudin-4 reveals their proximity to signaling and trafficking networks. PLoS ONE 2015, 10, e0117074. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Lu, Q.; Chen, Y.H. Claudin-7 modulates cell-matrix adhesion that controls cell migration, invasion and attachment of human HCC827 lung cancer cells. Oncol. Lett. 2019, 17, 2890–2896. [Google Scholar] [CrossRef] [PubMed]
- Gardelli, C.; Russo, L.; Cipolla, L.; Moro, M.; Andriani, F.; Rondinone, O.; Nicotra, F.; Sozzi, G.; Bertolini, G.; Roz, L. Differential glycosylation of collagen modulates lung cancer stem cell subsets through β1 integrin-mediated interactions. Cancer Sci. 2021, 112, 217–230. [Google Scholar] [CrossRef]
- Moon, J.H.; Rho, Y.S.; Lee, S.H.; Koo, B.S.; Lee, H.J.; Do, S.I.; Cho, J.H.; Eun, Y.G.; Park, M.W.; Shin, H.A.; et al. Role of integrin β1 as a biomarker of stemness in head and neck squamous cell carcinoma. Oral. Oncol. 2019, 96, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Schulz, W.A. DNA methylation in urological malignancies. Int. J. Oncol. 1998, 13, 151–167. [Google Scholar]
- Wang, C.; Jiang, X.; Huang, B.; Zhou, W.; Cui, X.; Zheng, C.; Liu, F.; Bi, J.; Zhang, Y.; Luo, H.; et al. Inhibition of matrix stiffness relating integrin β1 signaling pathway inhibits tumor growth in vitro and in hepatocellular cancer xenografts. BMC Cancer 2021, 21, 1276. [Google Scholar] [CrossRef]
- Campagna, R.; Pozzi, V.; Spinelli, G.; Sartini, D.; Milanese, G.; Galosi, A.B.; Emanuelli, M. The Utility of Nicotinamide N-Methyltransferase as a Potential Biomarker to Predict the Oncological Outcomes for Urological Cancers: An Update. Biomolecules 2021, 11, 1214. [Google Scholar] [CrossRef]
- Zhang, J.; Zhu, Y.; Wang, Y.; Fu, Q.; Xie, H.; Liu, Z.; Fu, H.; Cao, Y.; Xu, J.; Dai, B. Prognostic and Predictive Value of O(6)-methylguanine Methyltransferase for Chemotherapy in Patients with Muscle-Invasive Bladder Cancer. Ann. Surg. Oncol. 2018, 25, 342–348. [Google Scholar] [CrossRef]
- Pozzi, V.; Di Ruscio, G.; Sartini, D.; Campagna, R.; Seta, R.; Fulvi, P.; Vici, A.; Milanese, G.; Brandoni, G.; Galosi, A.B.; et al. Clinical performance and utility of a NNMT-based urine test for bladder cancer. Int. J. Biol. Markers 2018, 33, 94–101. [Google Scholar] [CrossRef]
- Kuniyasu, H.; Yano, S.; Sasaki, T.; Sasahira, T.; Sone, S.; Ohmori, H. Colon cancer cell-derived high mobility group 1/amphoterin induces growth inhibition and apoptosis in macrophages. Am. J. Pathol. 2005, 166, 751–760. [Google Scholar] [CrossRef]
- Kuniyasu, H.; Luo, Y.; Fujii, K.; Sasahira, T.; Moriwaka, Y.; Tatsumoto, N.; Sasaki, T.; Yamashita, Y.; Ohmori, H. CD10 enhances metastasis of colorectal cancer by abrogating the anti-tumoural effect of methionine-enkephalin in the liver. Gut 2010, 59, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Kuniyasu, H.; Oue, N.; Wakikawa, A.; Shigeishi, H.; Matsutani, N.; Kuraoka, K.; Ito, R.; Yokozaki, H.; Yasui, W. Expression of receptors for advanced glycation end-products (RAGE) is closely associated with the invasive and metastatic activity of gastric cancer. J. Pathol. 2002, 196, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Mitic, L.L.; Unger, V.M.; Anderson, J.M. Expression, solubilization, and biochemical characterization of the tight junction transmembrane protein claudin-4. Protein Sci. 2003, 12, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Kuniyasu, H.; Oue, N.; Tsutsumi, M.; Tahara, E.; Yasui, W. Heparan Sulfate Enhances Invasion by Human Colon Carcinoma Cell Lines through Expression of CD44 Variant Exon 3. Clin. Cancer Res. 2001, 7, 4067–4072. [Google Scholar]
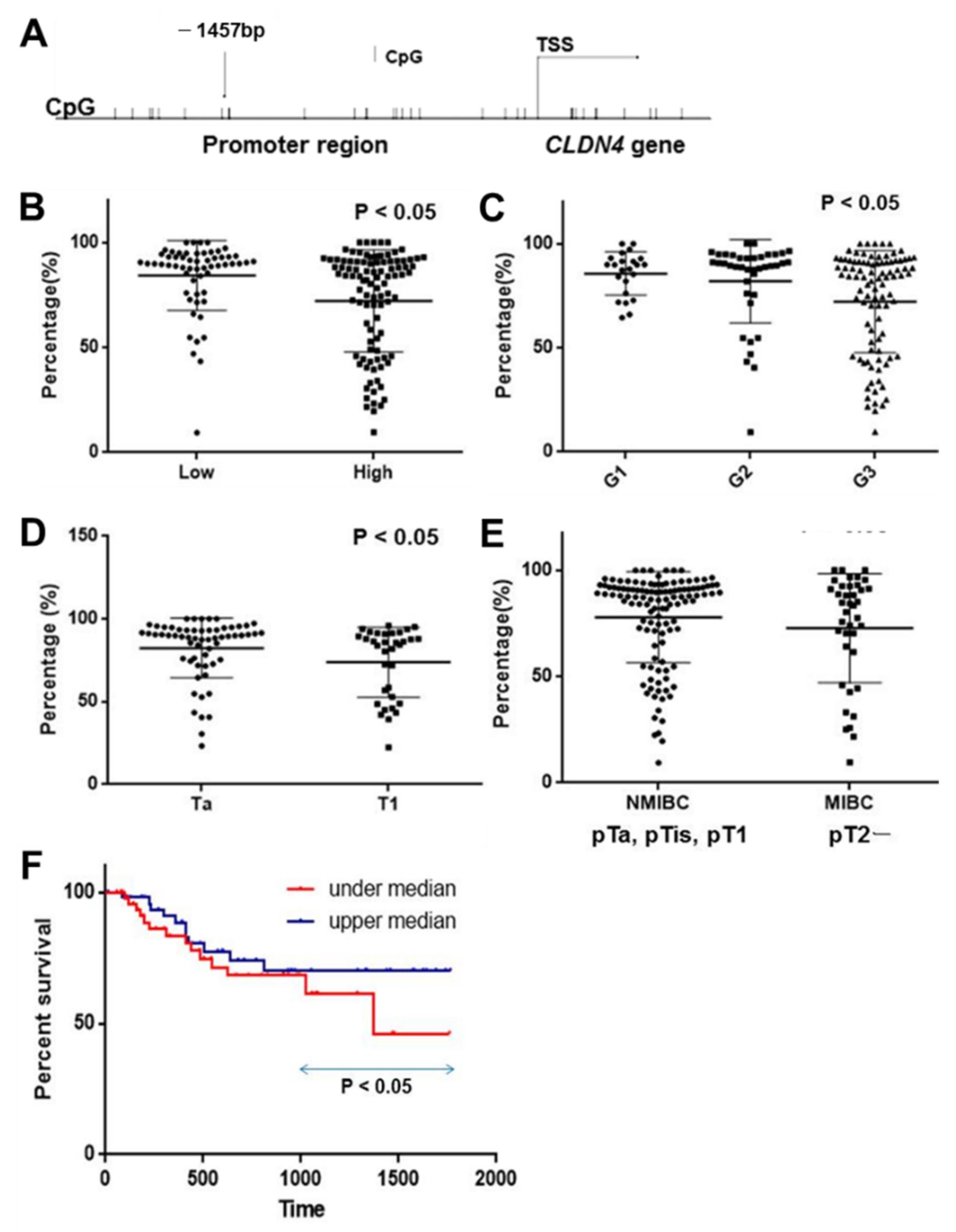


| Parameter | n | Methylation (%) | p | |
|---|---|---|---|---|
| Normal urothelium | 20 | 92 ± 5 | ||
| BUC | 157 | 72 ± 19 | <0.0001 | |
| Grade (1) | G1 | 23 | 81 ± 12 | |
| G2 | 39 | 77 ± 24 | ||
| G3 | 95 | 68 ± 20 | 0.0057 | |
| pT (2) | pTa | 61 | 82 ± 19 | |
| pTis | 23 | 75 ± 20 | ||
| pT1 | 34 | 73 ± 23 | ||
| pT2 | 39 | 69 ± 23 | 0.0215 | |
| Muscle | NMIBC | 118 | 78 ± 21 | |
| invasion | MIBC | 39 | 69 ± 23 | 0.0249 |
| Parameter | CLDN4 Methylation (1) | p | ||
|---|---|---|---|---|
| High | Low | |||
| Number | 11 | 12 | ||
| Grade (2) | Low | 1 | 0 | NS |
| High | 10 | 12 | ||
| pT (3) | pT1 | 10 | 10 | NS |
| pT2 | 1 | 2 | ||
| INF (4) | a/b | 11 | 5 | 0.0046 |
| c | 0 | 7 | ||
| Ly/V (5) | - | 11 | 4 | 0.0006 |
| + | 0 | 9 | ||
| Reurrence (6) | 3 | 10 | 0.0123 | |
| Antibody | Species | Provider | City and Country |
|---|---|---|---|
| CLDN4 | human | clone 4D3 | [3] |
| ZO-1 | human | Abcam | Cambridge, MA, USA |
| ITGβ1 | human | Santa-Cruz Biotechnology | Santa Cruz, CA, USA |
| pFAK (pTyr397) | human | Boster Immunoleader | Pleasanton, CA, USA |
| FAK | human | Santa-Cruz Biotechnology | Santa Cruz, CA, USA |
| Ki-67 | human | DAKO-Agilent | Santa Clara, CA, USA |
| CD44v9 | human | Cosmobio | Carlsbad, CA, USA |
| Nucleostemin | human | Santa-Cruz Biotechnology | Santa Cruz, CA, USA |
| E-cadherin | human | DAKO-Agilent | Santa Clara, CA, USA |
| βActin | human | Zymed Laboratories Inc. | South San Francisco, CA, USA |
| ELISA | Species | Provider | City and country |
| CLDN4 | human | MyBiosource, Inc. | San Diego, CA, USA |
| CLDN7 | Human | Wuhan Huamei Biotech | Wuhan, China |
| Ki-67 | human | Abcam | Cambridge, MA, USA |
| GNL3 | human | antibodies-online GmbH | Aachen, Germany |
| CD44v9 | human | Elabscience | Houston, TX, USA |
| CDH1 | human | Abcam | Cambridge, MA, USA |
| SNAIL | human | Biocompare | South San Francisco, CA, USA |
| Vimentin | human | Abcam | Cambridge, MA, USA |
| Gene Symbol | Species | Accession No. | Site | Sequence |
|---|---|---|---|---|
| CLDN4 | human | NM_001305.4 | Upper | CTC CAT GGG GCT ACA GGT AA |
| Lower | AGC AGC GAG TCG TAC ACC TT | |||
| GNL3 | human | BC001024.2 | Upper | ATT GCC AAC AGT GGT GTT CA |
| Lower | AAT GGC TTT GCT GCA AGT TT | |||
| BCL2 | human | M13994.1 | Upper | ACG ACA ACC GGG AGA TAG TG |
| Lower | CAT CCC AGC CTC CGT TAT CC | |||
| BAX | human | L22473.1 | Upper | CAT GAA GAC AGG GGC CCT TT |
| Lower | CTT CCA GAT GGT GAG CGA GG | |||
| GAPDH | human | BC025925.1 | Upper | GAG TCA ACG GAT TTG GTC GT |
| Lower | TTG ATT TTG GAG GGA TCT CG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maesaka, F.; Kuwada, M.; Horii, S.; Kishi, S.; Fujiwara-Tani, R.; Mori, S.; Fujii, K.; Mori, T.; Ohmori, H.; Owari, T.; et al. Hypomethylation of CLDN4 Gene Promoter Is Associated with Malignant Phenotype in Urinary Bladder Cancer. Int. J. Mol. Sci. 2022, 23, 6516. https://doi.org/10.3390/ijms23126516
Maesaka F, Kuwada M, Horii S, Kishi S, Fujiwara-Tani R, Mori S, Fujii K, Mori T, Ohmori H, Owari T, et al. Hypomethylation of CLDN4 Gene Promoter Is Associated with Malignant Phenotype in Urinary Bladder Cancer. International Journal of Molecular Sciences. 2022; 23(12):6516. https://doi.org/10.3390/ijms23126516
Chicago/Turabian StyleMaesaka, Fumisato, Masaomi Kuwada, Shohei Horii, Shingo Kishi, Rina Fujiwara-Tani, Shiori Mori, Kiyomu Fujii, Takuya Mori, Hitoshi Ohmori, Takuya Owari, and et al. 2022. "Hypomethylation of CLDN4 Gene Promoter Is Associated with Malignant Phenotype in Urinary Bladder Cancer" International Journal of Molecular Sciences 23, no. 12: 6516. https://doi.org/10.3390/ijms23126516
APA StyleMaesaka, F., Kuwada, M., Horii, S., Kishi, S., Fujiwara-Tani, R., Mori, S., Fujii, K., Mori, T., Ohmori, H., Owari, T., Miyake, M., Nakai, Y., Tanaka, N., Bhawal, U. K., Luo, Y., Kondoh, M., Fujimoto, K., & Kuniyasu, H. (2022). Hypomethylation of CLDN4 Gene Promoter Is Associated with Malignant Phenotype in Urinary Bladder Cancer. International Journal of Molecular Sciences, 23(12), 6516. https://doi.org/10.3390/ijms23126516








