Tibolone Pre-Treatment Ameliorates the Dysregulation of Protein Translation and Transport Generated by Palmitic Acid-Induced Lipotoxicity in Human Astrocytes: A Label-Free MS-Based Proteomics and Network Analysis
Abstract
1. Introduction
2. Results
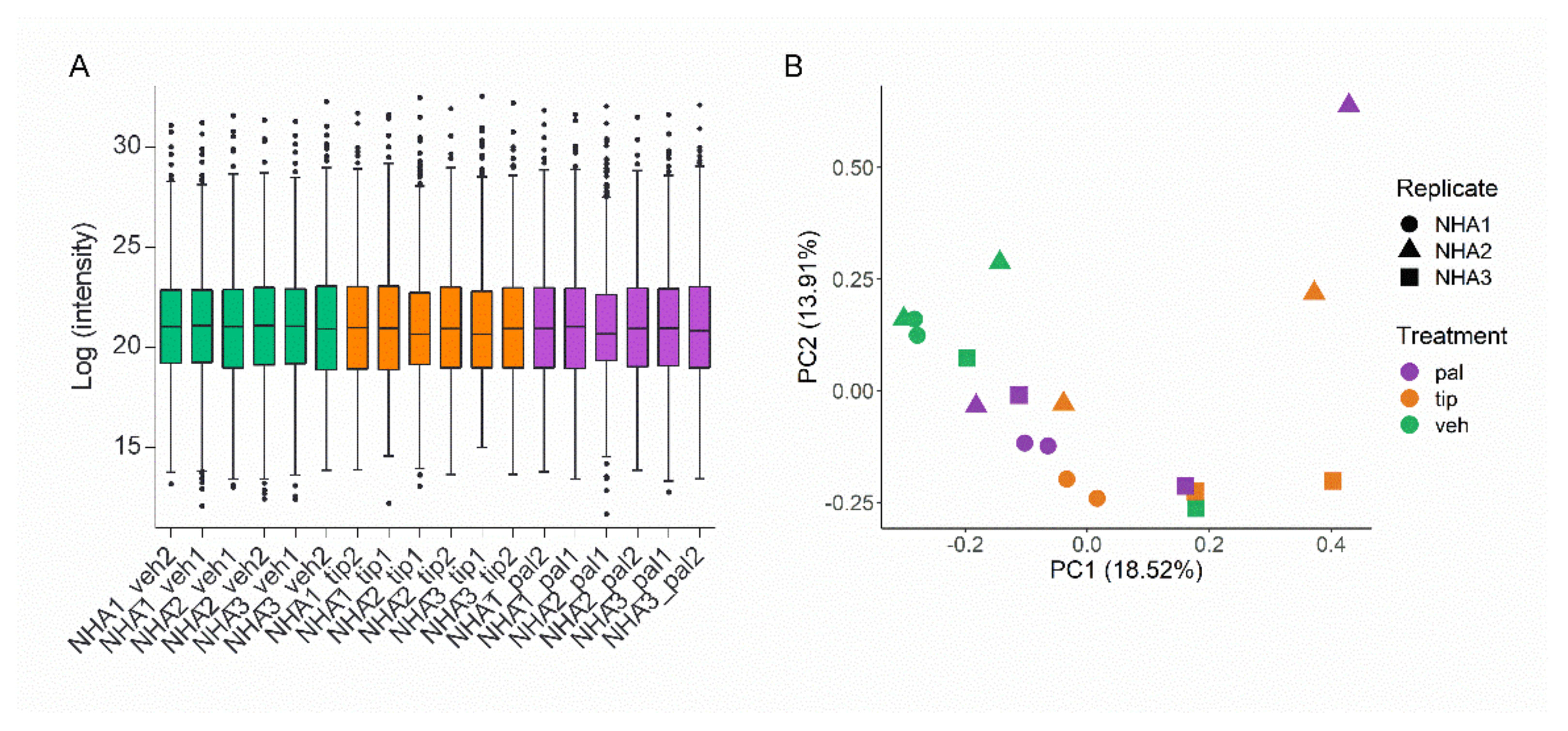
2.1. Proteomic-Wide Profiling of Palmitic Acid-Exposed Astrocytes
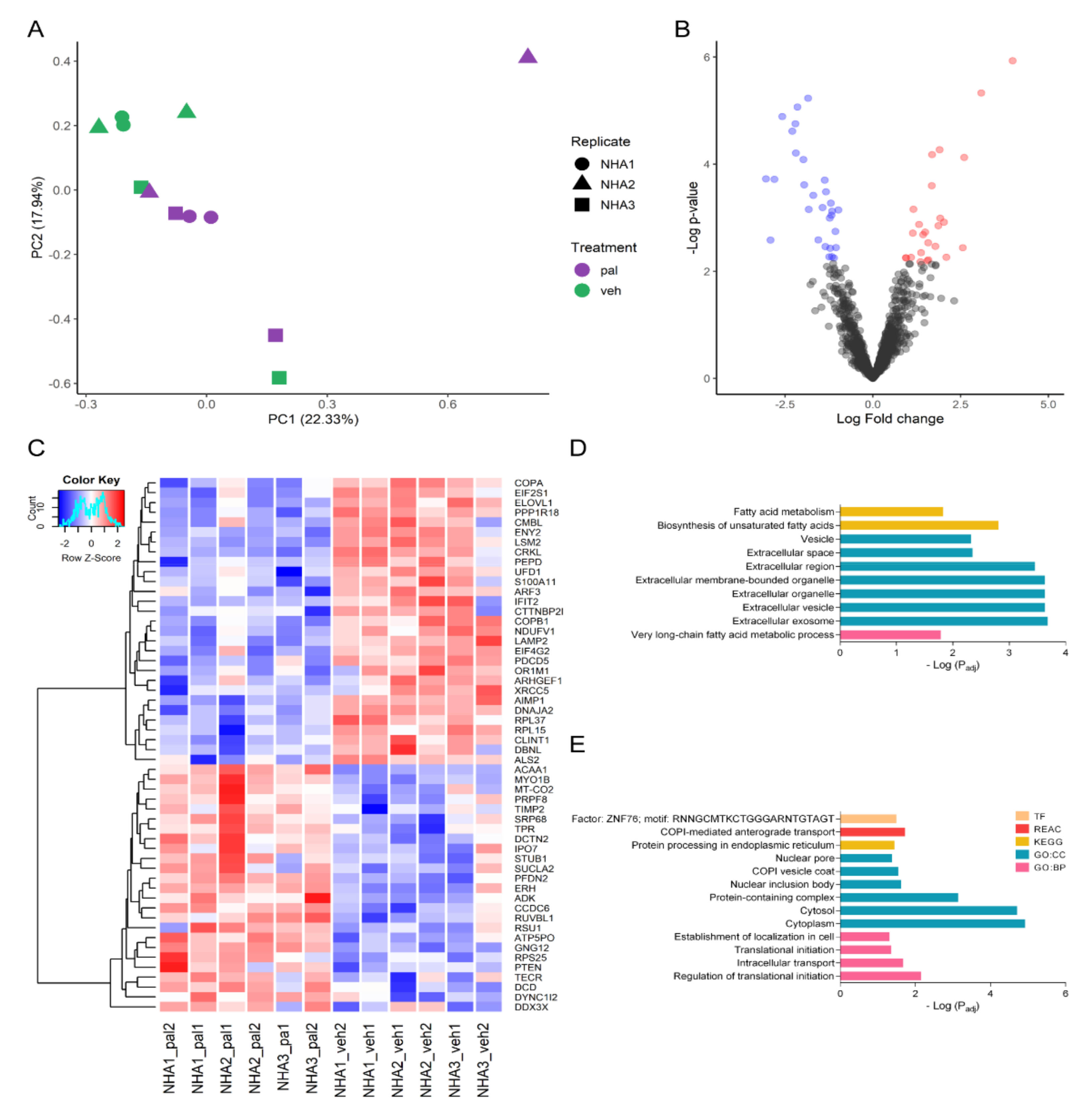
2.2. Pal Reduces the Expression of Proteins Related to Transport, Transcription, and Translation Processes
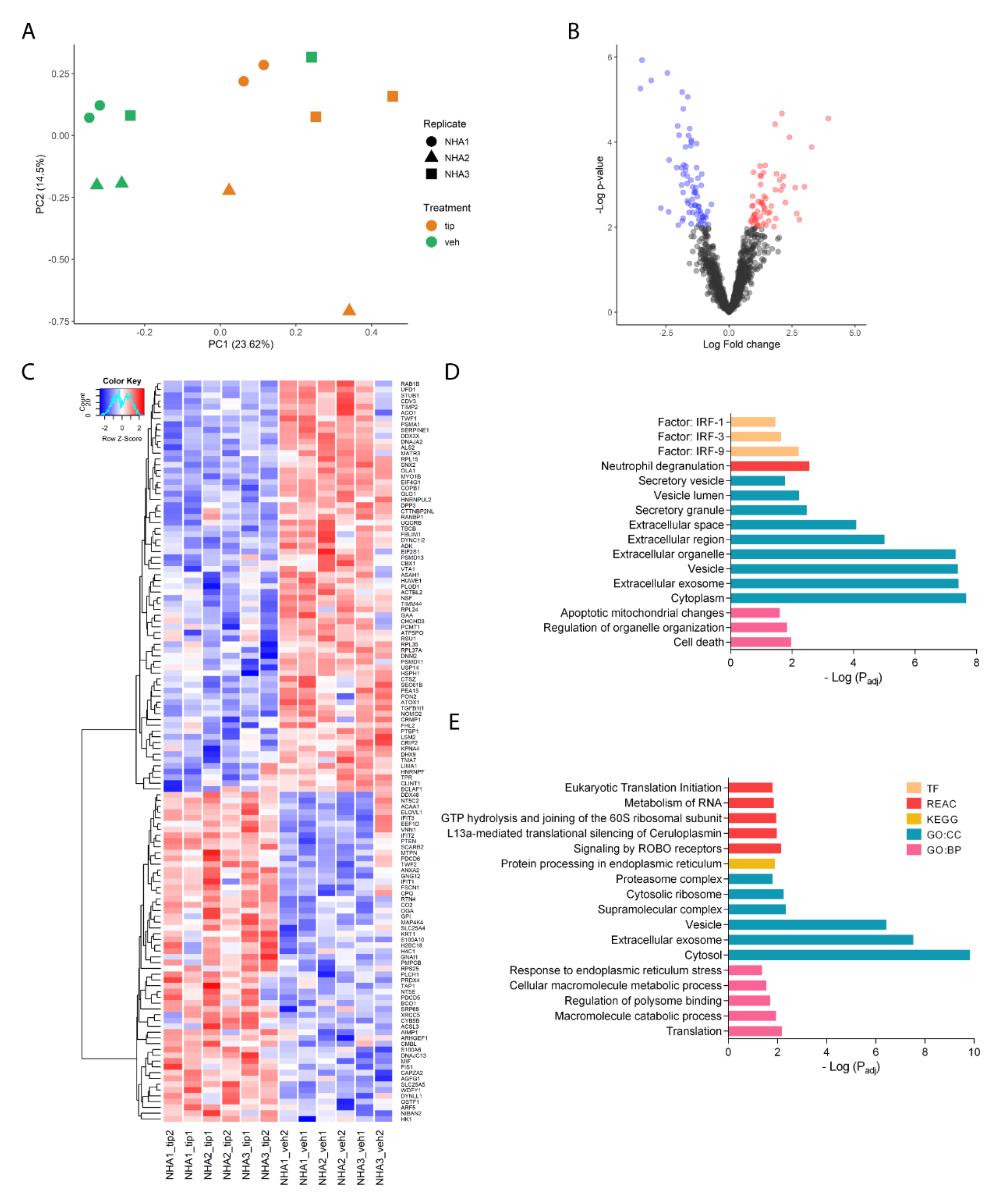
2.3. Tibolone Regulates the Expression of Proteins Related to Translation, Transport, and Immune Response
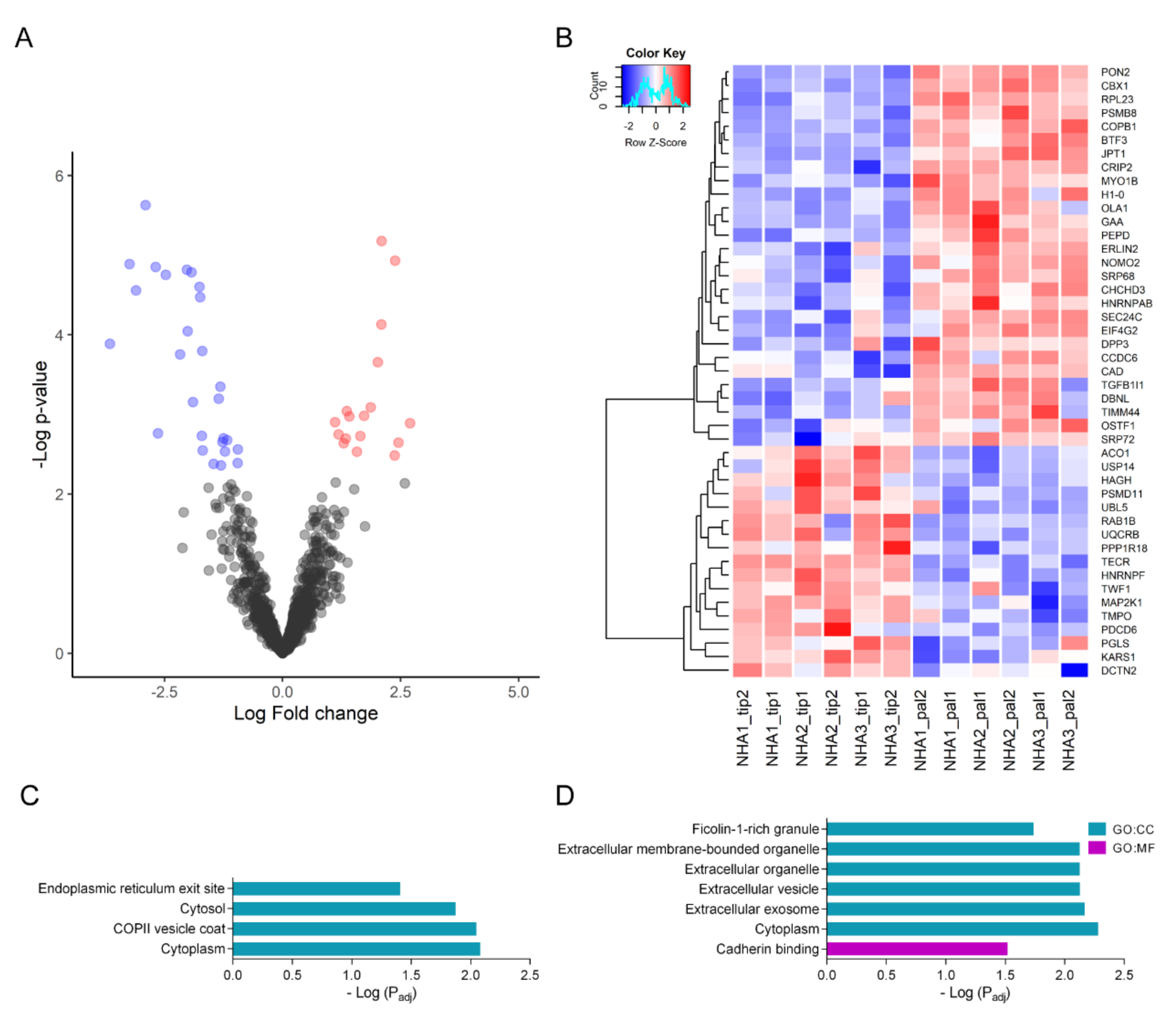
2.4. Tibolone Restores Expression Levels in Transport, Translation, and Apoptosis-Related Proteins
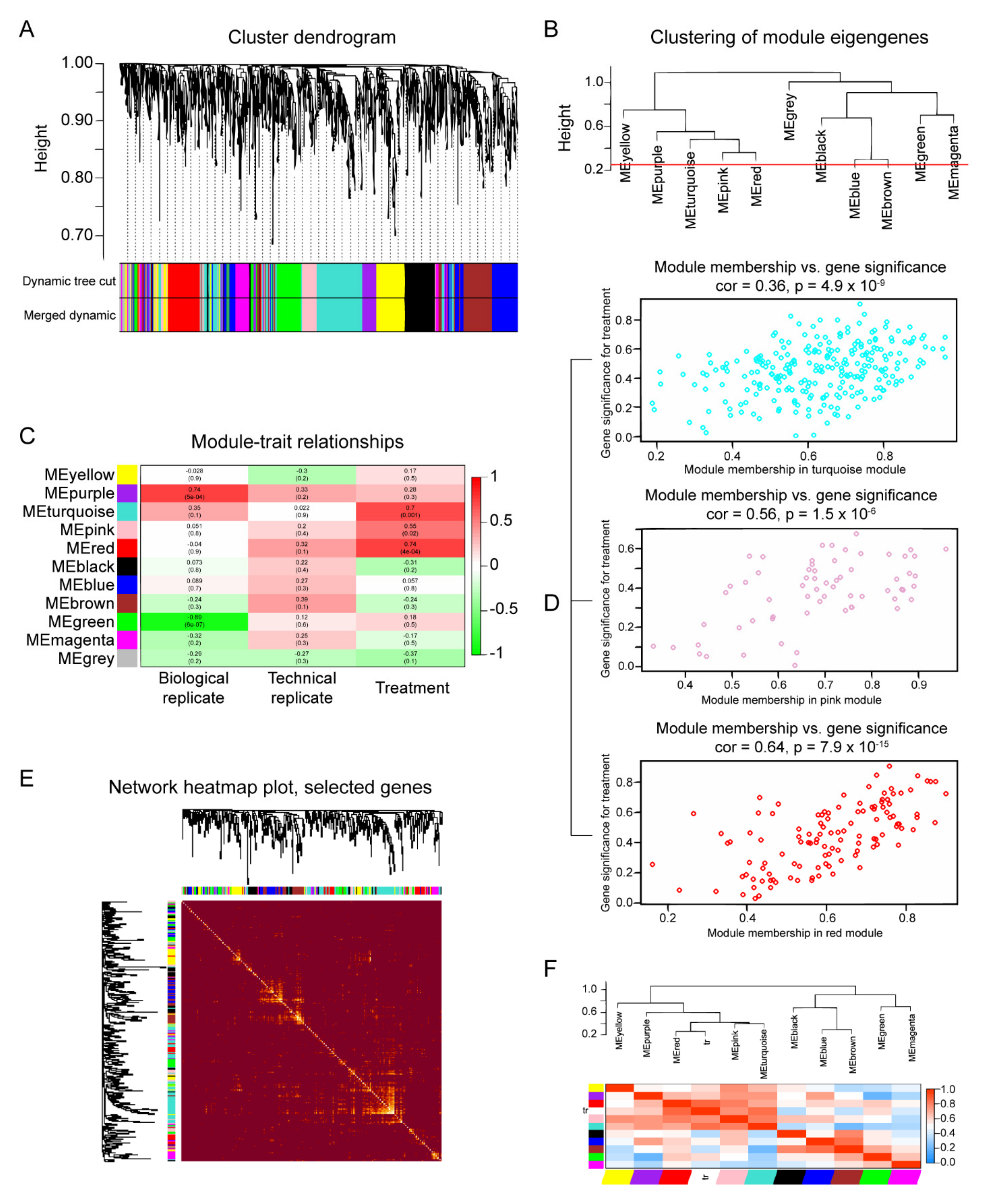
2.5. ARF3 and IPO7 as Key Proteins in the Lipotoxic Scenario Triggered by Pal
2.6. Experimental Validation
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Tibolone Pre-Treatment
4.3. Palmitic Acid Treatment
4.4. Protein Extraction and Quantification
4.5. Protein Digestion and Load in the Q Exactive
4.6. Raw Files Processing for Protein Identification
4.7. Normalization and Statistics for Relative Quantification
4.8. Differential Expression Analysis
4.9. Functional Enrichment Analysis
4.10. Weighted Co-Expression Network Analysis
4.11. Experimental Validation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO Obesity. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 12 January 2021).
- Gupta, S.; Knight, A.G.; Gupta, S.; Keller, J.N.; Bruce-Keller, A.J. Saturated long-chain fatty acids activate inflammatory signaling in astrocytes. J. Neurochem. 2012, 120, 1060–1071. [Google Scholar] [CrossRef] [PubMed]
- Shamim, A.; Mahmood, T.; Ahsan, F.; Kumar, A.; Bagga, P. Lipids: An insight into the neurodegenerative disorders. Clin. Nutr. Exp. 2018, 20, 1–19. [Google Scholar] [CrossRef]
- Anderson, G.; Rodriguez, M.; Reiter, R.J. Multiple sclerosis: Melatonin, orexin, and ceramide interact with platelet activation coagulation factors and gut-microbiome-derived butyrate in the circadian dysregulation of mitochondria in glia and immune cells. Int. J. Mol. Sci. 2019, 20, 5500. [Google Scholar] [CrossRef] [PubMed]
- Cakir, I.; Nillni, E.A. Endoplasmic Reticulum Stress, the Hypothalamus, and Energy Balance. Trends Endocrinol. Metab. 2019, 30, 163–176. [Google Scholar] [CrossRef]
- González-Giraldo, Y.; Garzón-Benitez, A.V.; Forero, D.A.; Barreto, G.E. TERT inhibition leads to reduction of IL-6 expression induced by palmitic acid and interferes with the protective effects of tibolone in an astrocytic cell model. J. Neuroendocrinol. 2019, 31, e12768. [Google Scholar] [CrossRef]
- Korbecki, J.; Bajdak-Rusinek, K. The effect of palmitic acid on inflammatory response in macrophages: An overview of molecular mechanisms. Inflamm. Res. 2019, 68, 915–932. [Google Scholar] [CrossRef]
- Melo, H.M.; Santos, L.E.; Ferreira, S.T. Diet-Derived Fatty Acids, Brain Inflammation, and Mental Health. Front. Neurosci. 2019, 13, 265. [Google Scholar] [CrossRef]
- Melo, H.M.; da Silva, G.S.S.; Sant’Ana, M.R.; Teixeira, C.V.L.; Clarke, J.R.; Miya Coreixas, V.S.; de Melo, B.C.; Fortuna, J.T.S.; Forny-Germano, L.; Ledo, J.H.; et al. Palmitate Is Increased in the Cerebrospinal Fluid of Humans with Obesity and Induces Memory Impairment in Mice via Pro-inflammatory TNF-α. Cell Rep. 2020, 30, 2180–2194.e8. [Google Scholar] [CrossRef]
- Liddelow, S.A.; Sofroniew, M.V. Astrocytes usurp neurons as a disease focus. Nat. Neurosci. 2019, 22, 512–513. [Google Scholar] [CrossRef]
- Lee, H.G.; Wheeler, M.A.; Quintana, F.J. Function and therapeutic value of astrocytes in neurological diseases. Nat. Rev. Drug Discov. 2022, 21, 339–358. [Google Scholar] [CrossRef]
- Carta, G.; Murru, E.; Banni, S.; Manca, C. Palmitic Acid: Physiological Role, Metabolism and Nutritional Implications. Front. Physiol. 2017, 8, 902. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.; Melrose, J.; Chan, C. Involvement of astroglial ceramide in palmitic acid-induced Alzheimer-like changes in primary neurons. Eur. J. Neurosci. 2007, 26, 2131–2141. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Rodriguez, A.; Acaz-Fonseca, E.; Boya, P.; Arevalo, M.A.; Garcia-Segura, L.M. Lipotoxic Effects of Palmitic Acid on Astrocytes Are Associated with Autophagy Impairment. Mol. Neurobiol. 2019, 56, 1665–1680. [Google Scholar] [CrossRef] [PubMed]
- Unger, R.H.; Clark, G.O.; Scherer, P.E.; Orci, L. Lipid homeostasis, lipotoxicity and the metabolic syndrome. Biochim. Biophys. Acta—Mol. Cell Biol. Lipids 2010, 1801, 209–214. [Google Scholar] [CrossRef]
- Okun, E.; Griffioen, K.J.; Lathia, J.D.; Tang, S.-C.; Mattson, M.P.; Arumugam, T. V Toll-like receptors in neurodegeneration. Brain Res. Rev. 2009, 59, 278–292. [Google Scholar] [CrossRef]
- Wong, K.L.; Wu, Y.R.; Cheng, K.S.; Chan, P.; Cheung, C.W.; Lu, D.Y.; Su, T.H.; Liu, Z.M.; Leung, Y.M. Palmitic acid-induced lipotoxicity and protection by (+)-catechin in rat cortical astrocytes. Pharmacol. Rep. 2014, 66, 1106–1113. [Google Scholar] [CrossRef]
- van Horssen, J.; van Schaik, P.; Witte, M. Inflammation and mitochondrial dysfunction: A vicious circle in neurodegenerative disorders? Neurosci. Lett. 2019, 710, 132931. [Google Scholar] [CrossRef]
- Liu, M.; Kelley, M.H.; Herson, P.S.; Hurn, P.D. Neuroprotection of sex steroids. Minerva Endocrinol. 2010, 35, 127–143. [Google Scholar] [CrossRef]
- Acaz-Fonseca, E.; Sanchez-Gonzalez, R.; Azcoitia, I.; Arevalo, M.A.; Garcia-Segura, L.M. Role of astrocytes in the neuroprotective actions of 17β-estradiol and selective estrogen receptor modulators. Mol. Cell. Endocrinol. 2014, 389, 48–57. [Google Scholar] [CrossRef]
- Arevalo, M.A.; Santos-Galindo, M.; Lagunas, N.; Azcoitia, I.; Garcia-Segura, L.M. Selective estrogen receptor modulators as brain therapeutic agents. J. Mol. Endocrinol. 2011, 46, R1–R9. [Google Scholar] [CrossRef]
- Karki, P.; Webb, A.; Zerguine, A.; Choi, J.; Son, D.S.; Lee, E. Mechanism of raloxifene-induced upregulation of glutamate transporters in rat primary astrocytes. Glia 2014, 62, 1270–1283. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Rodriguez, A.B.; Ávila-Rodriguez, M.; Vega-Vela, N.E.; Capani, F.; Gonzalez, J.; García-Segura, L.M.; Barreto, G.E. Neuroprotection by Exogenous Estrogenic Compounds Following Traumatic Brain Injury. Estrogen Eff. Trauma. Brain Inj. 2015, 73–90. [Google Scholar] [CrossRef]
- Ávila Rodriguez, M.; Garcia-Segura, L.M.; Cabezas, R.; Torrente, D.; Capani, F.; Gonzalez, J.; Barreto, G.E. Tibolone protects T98G cells from glucose deprivation. J. Steroid Biochem. Mol. Biol. 2014, 144, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Pinto-Almazán, R.; Rivas-Arancibia, S.; Farfán-García, E.D.; Rodríguez-Martínez, E.; Guerra-Araiza, C. Neuroprotective effects of tibolone against oxidative stress induced by ozone exposure. Rev. Neurol. 2014, 58, 441–448. [Google Scholar] [PubMed]
- Crespo-Castrillo, A.; Yanguas-Casás, N.; Arevalo, M.A.; Azcoitia, I.; Barreto, G.E.; Garcia-Segura, L.M. The Synthetic Steroid Tibolone Decreases Reactive Gliosis and Neuronal Death in the Cerebral Cortex of Female Mice After a Stab Wound Injury. Mol. Neurobiol. 2018, 55, 8651–8667. [Google Scholar] [CrossRef]
- Del Río, J.P.; Molina, S.; Hidalgo-Lanussa, O.; Garcia-Segura, L.M.; Barreto, G.E. Tibolone as Hormonal Therapy and Neuroprotective Agent. Trends Endocrinol. Metab. 2020, 31, 742–759. [Google Scholar] [CrossRef]
- Reed, M.J.; Kloosterboer, H.J. Tibolone: A selective tissue estrogenic activity regulator. Maturitas 2004, 48, 54–56. [Google Scholar] [CrossRef]
- Pinto-Almazán, R.; Segura-Uribe, J.J.; Farfán-García, E.D.; Guerra-Araiza, C. Effects of Tibolone on the Central Nervous System: Clinical and Experimental Approaches. Biomed. Res. Int. 2017, 2017, 8630764. [Google Scholar] [CrossRef]
- Arevalo, M.-A.; Santos-Galindo, M.; Bellini, M.-J.; Azcoitia, I.; Garcia-Segura, L.M. Actions of estrogens on glial cells: Implications for neuroprotection. Biochim. Biophys. Acta—Gen. Subj. 2010, 1800, 1106–1112. [Google Scholar] [CrossRef]
- Karki, P.; Smith, K.; Johnson, J.; Lee, E. Astrocyte-derived growth factors and estrogen neuroprotection: Role of transforming growth factor-β in estrogen-induced upregulation of glutamate transporters in astrocytes. Mol. Cell. Endocrinol. 2014, 389, 58–64. [Google Scholar] [CrossRef]
- Dietrich, A.K.; Humphreys, G.I.; Nardulli, A.M. Expression of estrogen receptor α in the mouse cerebral cortex. Mol. Cell. Endocrinol. 2015, 406, 19–26. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Avila-Rodriguez, M.; Garcia-Segura, L.M.; Hidalgo-lanussa, O.; Baez, E.; Gonzalez, J.; Barreto, G.E. Tibolone protects astrocytic cells from glucose deprivation through a mechanism involving estrogen receptor beta and the upregulation of neuroglobin expression. Mol. Cell. Endocrinol. 2016, 433, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Kloosterboer, H.J. Tissue-selectivity: The mechanism of action of tibolone. Maturitas 2004, 48, 30–40. [Google Scholar] [CrossRef] [PubMed]
- De Aguilar, J.L.G.; González De Aguilar, J.-L. Lipid Biomarkers for Amyotrophic Lateral Sclerosis. Front. Neurol. 2019, 10 (Suppl. 1), 284. [Google Scholar] [CrossRef]
- Martin-Jiménez, C.; González, J.; Vesga, D.; Aristizabal, A.; Barreto, G.E. Tibolone Ameliorates the Lipotoxic Effect of Palmitic Acid in Normal Human Astrocytes. Neurotox. Res. 2020, 38, 585–595. [Google Scholar] [CrossRef]
- González-Giraldo, Y.; Forero, D.A.; Echeverria, V.; Garcia-Segura, L.M.; Barreto, G.E. Tibolone attenuates inflammatory response by palmitic acid and preserves mitochondrial membrane potential in astrocytic cells through estrogen receptor beta. Mol. Cell. Endocrinol. 2019, 486, 65–78. [Google Scholar] [CrossRef]
- Vesga-Jiménez, D.J.; Martin, C.; Barreto, G.E.; Aristizábal-Pachón, A.F.; Pinzón, A.; González, J. Fatty Acids: An Insight into the Pathogenesis of Neurodegenerative Diseases and Therapeutic Potential. Int. J. Mol. Sci. 2022, 23, 2577. [Google Scholar] [CrossRef]
- McArdle, A.J.; Menikou, S. What is proteomics? Arch. Dis. Child. Educ. Pract. Ed. 2020, 106, 178–181. [Google Scholar] [CrossRef]
- Dupree, E.J.; Jayathirtha, M.; Yorkey, H.; Mihasan, M.; Petre, B.A.; Darie, C.C. A critical review of bottom-up proteomics: The good, the bad, and the future of this field. Proteomes 2020, 8, 14. [Google Scholar] [CrossRef]
- Karpievitch, Y.V.; Dabney, A.R.; Smith, R.D. Normalization and missing value imputation for label-free LC-MS analysis. BMC Bioinform. 2012, 13 (Suppl. 1), S5. [Google Scholar] [CrossRef]
- Gaudet, P.; Livstone, M.S.; Lewis, S.E.; Thomas, P.D. Phylogenetic-based propagation of functional annotations within the Gene Ontology consortium. Brief. Bioinform. 2011, 12, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Jäkel, S.; Görlich, D. Importin β, transportin, RanBP5 and RanBP7 mediate nuclear import of ribosomal proteins in mammalian cells. EMBO J. 1998, 17, 4491–4502. [Google Scholar] [CrossRef] [PubMed]
- Jäkel, S.; Albig, W.; Kutay, U.; Bischoff, F.R.; Schwamborn, K.; Doenecke, D.; Görlich, D. The importin β/importin 7 heterodimer is a functional nuclear import receptor for histone H1. EMBO J. 1999, 18, 2411–2423. [Google Scholar] [CrossRef]
- Zhao, Y.; Lang, G.; Ito, S.; Bonnet, J.; Metzger, E.; Sawatsubashi, S.; Suzuki, E.; Le Guezennec, X.; Stunnenberg, H.G.; Krasnov, A.; et al. A TFTC/STAGA module mediates histone H2A and H2B deubiquitination, coactivates nuclear receptors, and counteracts heterochromatin silencing. Mol. Cell 2008, 29, 92–101. [Google Scholar] [CrossRef]
- Slobodin, B.; Han, R.; Calderone, V.; Vrielink, J.A.F.O.; Loayza-Puch, F.; Elkon, R.; Agami, R. Transcription Impacts the Efficiency of mRNA Translation via Co-transcriptional N6-adenosine Methylation. Cell 2017, 169, 326–337.e12. [Google Scholar] [CrossRef] [PubMed]
- Kar, A.N.; MacGibeny, M.A.; Gervasi, N.M.; Gioio, A.E.; Kaplan, B.B. Intra-axonal synthesis of eukaryotic translation initiation factors regulates local protein synthesis and axon growth in rat sympathetic neurons. J. Neurosci. 2013, 33, 7165–7174. [Google Scholar] [CrossRef]
- Bogie, J.F.J.; Haidar, M.; Kooij, G.; Hendriks, J.J.A. Fatty acid metabolism in the progression and resolution of CNS disorders. Adv. Drug Deliv. Rev. 2020, 159, 198–213. [Google Scholar] [CrossRef]
- Flores-León, M.; Alcaraz, N.; Pérez-Domínguez, M.; Torres-Arciga, K.; Rebollar-Vega, R.; De la Rosa-Velázquez, I.A.; Arriaga-Canon, C.; Herrera, L.A.; Arias, C.; González-Barrios, R. Transcriptional Profiles Reveal Deregulation of Lipid Metabolism and Inflammatory Pathways in Neurons Exposed to Palmitic Acid. Mol. Neurobiol. 2021, 58, 4639–4651. [Google Scholar] [CrossRef]
- Rodríguez, M.; Pintado, C.; Torrillas-de la Cal, R.; Moltó, E.; Gallardo, N.; Andrés, A.; Arribas, C. Ageing alters the lipid sensing process in the hypothalamus of Wistar rats. Effect of food restriction. Nutr. Neurosci. 2021, 1–15. [Google Scholar] [CrossRef]
- Schönfeld, P.; Reiser, G. How the brain fights fatty acids’ toxicity. Neurochem. Int. 2021, 148, 105050. [Google Scholar] [CrossRef]
- Dozio, V.; Sanchez, J.C. Profiling the proteomic inflammatory state of human astrocytes using DIA mass spectrometry 06 Biological Sciences 0601 Biochemistry and Cell Biology. J. Neuroinflamm. 2018, 15, 331. [Google Scholar] [CrossRef] [PubMed]
- Herland, A.; Maoz, B.M.; FitzGerald, E.A.; Grevesse, T.; Vidoudez, C.; Sheehy, S.P.; Budnik, N.; Dauth, S.; Mannix, R.; Budnik, B.; et al. Proteomic and Metabolomic Characterization of Human Neurovascular Unit Cells in Response to Methamphetamine. Adv. Biosyst. 2020, 4, 1900230. [Google Scholar] [CrossRef] [PubMed]
- Dozio, V.; Daali, Y.; Desmeules, J.; Sanchez, J.C. Deep proteomics and phosphoproteomics reveal novel biological pathways perturbed by morphine, morphine-3-glucuronide and morphine-6-glucuronide in human astrocytes. J. Neurosci. Res. 2022, 100, 220–236. [Google Scholar] [CrossRef] [PubMed]
- Boye, E.; Grallert, B. eIF2α phosphorylation and the regulation of translation. Curr. Genet. 2020, 66, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.L.; Sonenberg, N.; Parker, R. Neuronal Regulation of eIF2α; Function in Health and Neurological Disorders. Trends Mol. Med. 2018, 24, 575–589. [Google Scholar] [CrossRef]
- Mikami, S.; Masutani, M.; Sonenberg, N.; Yokoyama, S.; Imataka, H. An efficient mammalian cell-free translation system supplemented with translation factors. Protein Expr. Purif. 2006, 46, 348–357. [Google Scholar] [CrossRef]
- Hernández-Ortega, K.; Garcia-Esparcia, P.; Gil, L.; Lucas, J.J.; Ferrer, I. Altered Machinery of Protein Synthesis in Alzheimer’s: From the Nucleolus to the Ribosome. Brain Pathol. 2016, 26, 593–605. [Google Scholar] [CrossRef]
- Rocchio, F.; Tapella, L.; Manfredi, M.; Chisari, M.; Ronco, F.; Ruffinatti, F.A.; Conte, E.; Canonico, P.L.; Sortino, M.A.; Grilli, M.; et al. Gene expression, proteome and calcium signaling alterations in immortalized hippocampal astrocytes from an Alzheimer’s disease mouse model. Cell Death Dis. 2019, 10, 24. [Google Scholar] [CrossRef]
- Deshpande, P.; Flinkman, D.; Hong, Y.; Goltseva, E.; Siino, V.; Sun, L.; Peltonen, S.; Elo, L.L.; Kaasinen, V.; James, P.; et al. Protein synthesis is suppressed in sporadic and familial Parkinson’s disease by LRRK2. FASEB J. 2020, 34, 14217–14233. [Google Scholar] [CrossRef]
- Shang, Y.; Xu, X.; Duan, X.; Guo, J.; Wang, Y.; Ren, F.; He, D.; Chang, Z. Hsp70 and Hsp90 oppositely regulate TGF-β signaling through CHIP/Stub1. Biochem. Biophys. Res. Commun. 2014, 446, 387–392. [Google Scholar] [CrossRef]
- Ortiz-Rodriguez, A.; Arevalo, M.A. The contribution of astrocyte autophagy to systemic metabolism. Int. J. Mol. Sci. 2020, 21, 2479. [Google Scholar] [CrossRef] [PubMed]
- Wakashima, T.; Abe, K.; Kihara, A. Dual Functions of the Trans-2-Enoyl-CoA Reductase TER in the Sphingosine 1-Phosphate Metabolic Pathway and in Fatty Acid Elongation. J. Biol. Chem. 2014, 289, 24736–24748. [Google Scholar] [CrossRef] [PubMed]
- Braverman, N.; Eichler, F. Peroxisomal Disorders and Neurological Disease. In Encyclopedia of Neuroscience; Squire, L.R., Ed.; Academic Press: Oxford, UK, 2009; pp. 579–588. ISBN 978-0-08-045046-9. [Google Scholar]
- Obinata, H.; Hla, T. Sphingosine 1-phosphate and inflammation. Int. Immunol. 2019, 31, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Doyon, Y.; Selleck, W.; Lane, W.S.; Tan, S.; Côté, J. Structural and Functional Conservation of the NuA4 Histone Acetyltransferase Complex from Yeast to Humans. Mol. Cell. Biol. 2004, 24, 1884–1896. [Google Scholar] [CrossRef] [PubMed]
- Gartner, W.; Rossbacher, J.; Zierhut, B.; Daneva, T.; Base, W.; Weissel, M.; Waldhäusl, W.; Pasternack, M.S.; Wagner, L. The ATP-dependent helicase RUVBL1/TIP49a associates with tubulin during mitosis. Cell Motil. Cytoskelet. 2003, 56, 79–93. [Google Scholar] [CrossRef]
- McGourty, C.A.; Akopian, D.; Walsh, C.; Gorur, A.; Werner, A.; Schekman, R.; Bautista, D.; Rape, M. Regulation of the CUL3 Ubiquitin Ligase by a Calcium-Dependent Co-adaptor. Cell 2016, 167, 525–538.e14. [Google Scholar] [CrossRef]
- Okumura, M.; Ichioka, F.; Kobayashi, R.; Suzuki, H.; Yoshida, H.; Shibata, H.; Maki, M. Penta-EF-hand protein ALG-2 functions as a Ca2+-dependent adaptor that bridges Alix and TSG101. Biochem. Biophys. Res. Commun. 2009, 386, 237–241. [Google Scholar] [CrossRef]
- Takeshi, T.; Kojima, K.; Zhang, W.; Sasaki, K.; Ito, M.; Suzuki, H.; Kawasaki, M.; Wakatsuki, S.; Takahara, T.; Shibata, H.; et al. Structural analysis of the complex between Penta-Ef-Hand Alg-2 protein and sec31a peptide reveals a novel target recognition mechanism of Alg-2. Int. J. Mol. Sci. 2015, 16, 3677–3699. [Google Scholar] [CrossRef]
- Lee, J.H.; Rho, S.B.; Chun, T. Programmed cell death 6 (PDCD6) protein interacts with death-associated protein kinase 1 (DAPk1): Additive effect on apoptosis via caspase-3 dependent pathway. Biotechnol. Lett. 2005, 27, 1011–1015. [Google Scholar] [CrossRef]
- Quijano, C.; Trujillo, M.; Castro, L.; Trostchansky, A. Interplay between oxidant species and energy metabolism. Redox Biol. 2016, 8, 28–42. [Google Scholar] [CrossRef]
- Lushchak, O.V.; Piroddi, M.; Galli, F.; Lushchak, V.I. Aconitase post-translational modification as a key in linkage between Krebs cycle, iron homeostasis, redox signaling, and metabolism of reactive oxygen species. Redox Rep. 2014, 19, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Laakkonen, E.K.; Soliymani, R.; Karvinen, S.; Kaprio, J.; Kujala, U.M.; Baumann, M.; Sipilä, S.; Kovanen, V.; Lalowski, M. Estrogenic regulation of skeletal muscle proteome: A study of premenopausal women and postmenopausal MZ cotwins discordant for hormonal therapy. Aging Cell 2017, 16, 1276–1287. [Google Scholar] [CrossRef] [PubMed]
- Pichlmair, A.; Lassnig, C.; Eberle, C.A.; Górna, M.W.; Baumann, C.L.; Burkard, T.R.; Búrckstúmmer, T.; Stefanovic, A.; Krieger, S.; Bennett, K.L.; et al. IFIT1 is an antiviral protein that recognizes 5′-triphosphate RNA. Nat. Immunol. 2011, 12, 624–630. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Li, B.; Zhu, H.Q.; Song, M.G.; Pan, X.R.; Jia, P.M.; Peng, L.L.; Dou, A.X.; Chen, G.Q.; Chen, S.J.; et al. RIG-G as a key of the antiproliferative activity of interferon-related pathways through enhancing p21 and p27 proteins. Proc. Natl. Acad. Sci. USA 2006, 103, 16448–16453. [Google Scholar] [CrossRef]
- Stawowczyk, M.; Van Scoy, S.; Kumar, K.P.; Reich, N.C. The interferon stimulated gene 54 promotes apoptosis. J. Biol. Chem. 2011, 286, 7257–7266. [Google Scholar] [CrossRef]
- Kaneko, S.; Chena, X.; Lua, P.; Yao, X.; Wright, T.G.; Rajurkar, M.; Kariya, K.I.; Mao, J.; Ip, T.; Xu, L. Smad inhibition by the Ste20 kinase misshapen. Proc. Natl. Acad. Sci. USA 2011, 108, 11127–11132. [Google Scholar] [CrossRef]
- Yue, J.; Xie, M.; Gou, X.; Lee, P.; Schneider, M.D.; Wu, X. Microtubules Regulate Focal Adhesion Dynamics through MAP4K4. Dev. Cell 2014, 31, 572–585. [Google Scholar] [CrossRef]
- Liu, A.W.; Cai, J.; Zhao, X.L.; Jiang, T.H.; He, T.F.; Fu, H.Q.; Zhu, M.H.; Zhang, S.H. ShRNA-targeted MAP4K4 inhibits hepatocellular carcinoma growth. Clin. Cancer Res. 2011, 17, 710–720. [Google Scholar] [CrossRef]
- Ensenat, D.; Yao, Z.; Wang, X.S.; Kori, R.; Zhou, G.; Lee, S.C.; Tan, T.H. A Novel Src Homology 3 Domain-containing Adaptor Protein, HIP-55, That Interacts with Hematopoietic Progenitor Kinase 1. J. Biol. Chem. 1999, 274, 33945–33950. [Google Scholar] [CrossRef]
- Dhanasekaran, D.N.; Reddy, E.P. JNK signaling in apoptosis. Oncogene 2008, 27, 6245–6251. [Google Scholar] [CrossRef]
- Rocha-Perugini, V.; Gordon-Alonso, M.; Sánchez-Madrid, F. Role of drebrin at the immunological synapse. In Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2017; Volume 1006, pp. 271–280. [Google Scholar] [CrossRef]
- Lakkaraju, A.K.K.; Luyet, P.P.; Parone, P.; Falguières, T.; Strub, K. Inefficient targeting to the endoplasmic reticulum by the signal recognition particle elicits selective defects in post-ER membrane trafficking. Exp. Cell Res. 2007, 313, 834–847. [Google Scholar] [CrossRef] [PubMed]
- Grotwinkel, J.T.; Wild, K.; Segnitz, B.; Sinning, I. SRP RNA remodeling by SRP68 explains its role in protein translocation. Science 2014, 344, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Mary, C.; Scherrer, A.; Huck, L.; Lakkaraju, A.K.K.; Thomas, Y.; Johnson, A.E.; Strub, K. Residues in SRP9/14 essential for elongation arrest activity of the signal recognition particle define a positively charged functional domain on one side of the protein. Rna 2010, 16, 969–979. [Google Scholar] [CrossRef] [PubMed]
- Siegel, V.; Walter, P. Elongation arrest is not a prerequisite for secretory protein translocation across the microsomal membrane. J. Cell Biol. 1985, 100, 1913–1921. [Google Scholar] [CrossRef]
- Yang, W.; Shi, J.; Zhou, Y.; Liu, T.; Zhan, F.; Zhang, K.; Liu, N. Integrating proteomics and transcriptomics for the identification of potential targets in early colorectal cancer. Int. J. Oncol. 2019, 55, 439–450. [Google Scholar] [CrossRef]
- Golomb, L.; Bublik, D.R.; Wilder, S.; Nevo, R.; Kiss, V.; Grabusic, K.; Volarevic, S.; Oren, M. Importin 7 and exportin 1 Link c-Myc and p53 to regulation of ribosomal Biogenesis. Mol. Cell 2012, 45, 222–232. [Google Scholar] [CrossRef] [PubMed]
- Bursac, S.; Brdovcak, M.C.; Donati, G.; Volarevic, S. Activation of the tumor suppressor p53 upon impairment of ribosome biogenesis. Biochim. Biophys. Acta–Mol. Basis Dis. 2014, 1842, 817–830. [Google Scholar] [CrossRef]
- Fumagalli, S.; Ivanenkov, V.V.; Teng, T.; Thomas, G. Suprainduction of p53 by disruption of 40S and 60S ribosome biogenesis leads to the activation of a novel G2/M checkpoint. Genes Dev. 2012, 26, 1028–1040. [Google Scholar] [CrossRef]
- Lin, A.; McNally, J.; Wool, I.G. The primary structure of rat liver ribosomal protein L37. Homology with yeast and bacterial ribosomal proteins. J. Biol. Chem. 1983, 258, 10664–10671. [Google Scholar] [CrossRef]
- Evans, H.T.; Benetatos, J.; van Roijen, M.; Bodea, L.L.-G.; Götz, J. Decreased synthesis of ribosomal proteins in tauopathy revealed by non-canonical amino acid labelling. EMBO J. 2019, 38, e101174. [Google Scholar] [CrossRef]
- Yu, J.; Chia, J.; Canning, C.A.; Jones, C.M.; Bard, F.A.; Virshup, D.M. WLS Retrograde transport to the endoplasmic reticulum during Wnt secretion. Dev. Cell 2014, 29, 277–291. [Google Scholar] [CrossRef] [PubMed]
- Staples, C.J.; Myers, K.N.; Beveridge, R.D.D.; Patil, A.A.; Howard, A.E.; Barone, G.; Lee, A.J.X.; Swanton, C.; Howell, M.; Maslen, S.; et al. Ccdc13 is a novel human centriolar satellite protein required for ciliogenesis and genome stability. J. Cell Sci. 2014, 127, 2910–2919. [Google Scholar] [CrossRef] [PubMed]
- Teuling, E.; van Dis, V.; Wulf, P.S.; Haasdijk, E.D.; Akhmanova, A.; Hoogenraad, C.C.; Jaarsma, D. A novel mouse model with impaired dynein/dynactin function develops amyotrophic lateral sclerosis (ALS)-like features in motor neurons and improves lifespan in SOD1-ALS mice. Hum. Mol. Genet. 2008, 17, 2849–2862. [Google Scholar] [CrossRef]
- De Vos, K.J.; Hafezparast, M. Neurobiology of axonal transport defects in motor neuron diseases: Opportunities for translational research? Neurobiol. Dis. 2017, 105, 283–299. [Google Scholar] [CrossRef] [PubMed]
- Lacy, P. Mechanisms of degranulation in neutrophils. Allergy Asthma Clin. Immunol. 2006, 2, 98–108. [Google Scholar] [CrossRef]
- Liu, L.; Martin, R.; Kohler, G.; Chan, C. Palmitate induces transcriptional regulation of BACE1 and presenilin by STAT3 in neurons mediated by astrocytes. Exp. Neurol. 2013, 248, 482–490. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hickson-Bick, D.L.M.; Sparagna, G.C.; Buja, L.M.; McMillin, J.B. Palmitate-induced apoptosis in neonatal cardiomyocytes is not dependent on the generation of ROS. Am. J. Physiol. Circ. Physiol. 2002, 282, H656–H664. [Google Scholar] [CrossRef]
- González-Giraldo, Y.; Garcia-Segura, L.M.; Echeverria, V.; Barreto, G.E. Tibolone Preserves Mitochondrial Functionality and Cell Morphology in Astrocytic Cells Treated with Palmitic Acid. Mol. Neurobiol. 2018, 55, 4453–4462. [Google Scholar] [CrossRef]
- Huber, W.; Von Heydebreck, A.; Sültmann, H.; Poustka, A.; Vingron, M. Variance stabilization applied to microarray data calibration and to the quantification of differential expression. Bioinformatics 2002, 18, S96–S104. [Google Scholar] [CrossRef]
- Välikangas, T.; Suomi, T.; Elo, L.L. A comprehensive evaluation of popular proteomics software workflows for label-free proteome quantification and imputation. Brief. Bioinform. 2018, 19, 1344–1355. [Google Scholar] [CrossRef]
- Elo, L.L.; Filén, S.; Lahesmaa, R.; Aittokallio, T. Reproducibility-optimized test statistic for ranking genes in microarray studies. IEEE/ACM Trans. Comput. Biol. Bioinforma. 2008, 5, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Suomi, T.; Seyednasrollah, F.; Jaakkola, M.K.; Faux, T.; Elo, L.L. ROTS: An R package for reproducibility-optimized statistical testing. PLoS Comput. Biol. 2017, 13, e1005562. [Google Scholar] [CrossRef] [PubMed]
- Storey, J.; Bass, A.; Dabney, A.; Robinson, D. Qvalue: Q-Value Estimation for False Discovery Rate Control. 2019. Available online: https://github.com/StoreyLab/qvalue (accessed on 15 October 2020).
- Reimand, J.; Isserlin, R.; Voisin, V.; Kucera, M.; Tannus-Lopes, C.; Rostamianfar, A.; Wadi, L.; Meyer, M.; Wong, J.; Xu, C.; et al. Pathway enrichment analysis and visualization of omics data using g:Profiler, GSEA, Cytoscape and EnrichmentMap. Nat. Protoc. 2019, 14, 482–517. [Google Scholar] [CrossRef] [PubMed]
- Raudvere, U.; Kolberg, L.; Kuzmin, I.; Arak, T.; Adler, P.; Peterson, H.; Vilo, J. G:Profiler: A web server for functional enrichment analysis and conversions of gene lists (2019 update). Nucleic Acids Res. 2019, 47, W191–W198. [Google Scholar] [CrossRef]
- Langfelder, P.; Horvath, S. WGCNA: An R package for weighted correlation network analysis. BMC Bioinform. 2008, 9, 559. [Google Scholar] [CrossRef]
- Bader, G.D.; Hogue, C.W.V. An automated method for finding molecular complexes in large protein interaction networks. BMC Bioinform. 2003, 4, 2. [Google Scholar] [CrossRef]
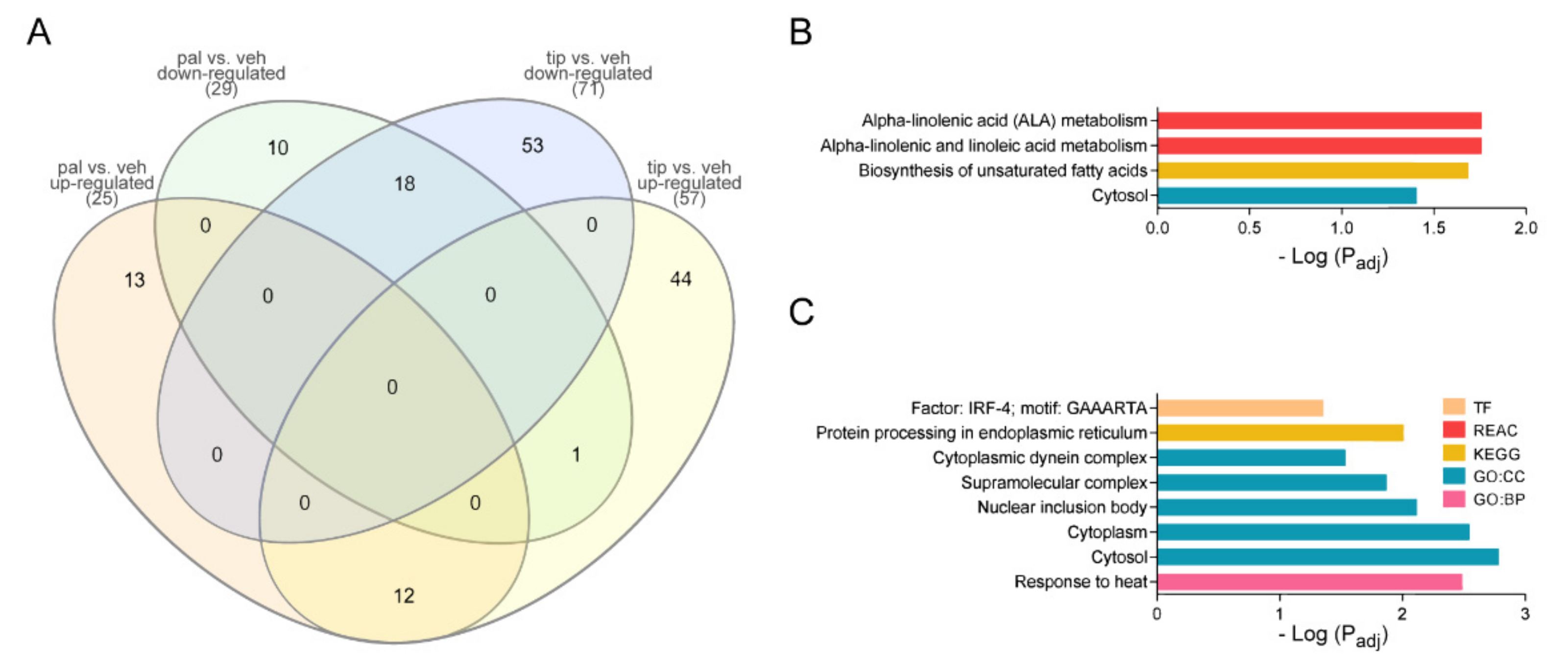


| Shared Down | Shared Up |
|---|---|
| ADK | ACAA1 |
| ALS2 | AIMP1 |
| ATP5PO | ARHGEF1 |
| CLINT1 | CMBL |
| COPB1 | ELOVL1 |
| CTTNBP2NL | GNG12 |
| DDX3X | IFIT2 |
| DNAJA2 | MT-CO2 |
| DYNC1I2 | PDCD5 |
| EIF2S1 | PTEN |
| LSM2 | RPS25 |
| MYO1B | XRCC5 |
| RPL15 | |
| RSU1 | |
| SRP68 | |
| STUB1 | |
| TIMP2 | |
| TPR | SRP68 (up-regulated in tip vs. veh) |
| UFD1 |
| Upregulated | Downregulated | ||||
|---|---|---|---|---|---|
| pal vs. veh | tip vs. veh | tip vs. pal | pal vs. veh | tip vs. veh | tip vs. pal |
| ARF3 | MAP4K4 HGK KIAA0687 NIK | None | ALS2 ALS2CR6 KIAA1563 | ALS2 ALS2CR6 KIAA1563 | CHCHD3 MIC19 MINOS3 |
| PTEN MMAC1 TEP1 | PLCH1 KIAA1069 PLCL3 | STUB1 CHIP PP1131 | TGFB1I1 ARA55 | TGFB1I1 ARA55 | |
| PDCD6 ALG2 | ADK | CHCHD3 MIC19 MINOS3 | ACO1 IREB1 | ||
| CAPZA2 | DNAJA2 CPR3 HIRIP4 | MYO1B | TWF1 PTK9 | ||
| TWF2 PTK9L MSTP011 | TIMP2 | STUB1 CHIP PP1131 | DPP3 | ||
| PTEN MMAC1 TEP1 | IPO7 RANBP7 | EIF4G1 EIF4F EIF4G EIF4GI | OLA1 GTPBP9 PRO2455 PTD004 | ||
| NT5C2 NT5B NT5CP PNT5 | UFD1 UFD1L | TIMP2 | GAA | ||
| CPQ LCH1 PGCP | MYO1B | DNAJA2 CPR3 HIRIP4 | |||
| DDX46 KIAA0801 | ADK | ||||
| IFIT3 CIG-49 IFI60 IFIT4 ISG60 | TWF1 PTK9 | ||||
| VNN1 | DDX3X DBX DDX3 | ||||
| GNG12 | OLA1 GTPBP9 PRO2455 PTD004 | ||||
| SLC25A4 ANT1 | DPP3 | ||||
| IFIT2 CIG-42 G10P2 IFI54 ISG54 | RPL15 EC45 TCBAP0781 | ||||
| UFD1 UFD1L | |||||
| PSMD11 | |||||
| SERPINE1 PAI1 PLANH1 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vesga-Jiménez, D.J.; Martín-Jiménez, C.A.; Grismaldo Rodríguez, A.; Aristizábal-Pachón, A.F.; Pinzón, A.; Barreto, G.E.; Ramírez, D.; González, J. Tibolone Pre-Treatment Ameliorates the Dysregulation of Protein Translation and Transport Generated by Palmitic Acid-Induced Lipotoxicity in Human Astrocytes: A Label-Free MS-Based Proteomics and Network Analysis. Int. J. Mol. Sci. 2022, 23, 6454. https://doi.org/10.3390/ijms23126454
Vesga-Jiménez DJ, Martín-Jiménez CA, Grismaldo Rodríguez A, Aristizábal-Pachón AF, Pinzón A, Barreto GE, Ramírez D, González J. Tibolone Pre-Treatment Ameliorates the Dysregulation of Protein Translation and Transport Generated by Palmitic Acid-Induced Lipotoxicity in Human Astrocytes: A Label-Free MS-Based Proteomics and Network Analysis. International Journal of Molecular Sciences. 2022; 23(12):6454. https://doi.org/10.3390/ijms23126454
Chicago/Turabian StyleVesga-Jiménez, Diego Julián, Cynthia A. Martín-Jiménez, Adriana Grismaldo Rodríguez, Andrés Felipe Aristizábal-Pachón, Andrés Pinzón, George E. Barreto, David Ramírez, and Janneth González. 2022. "Tibolone Pre-Treatment Ameliorates the Dysregulation of Protein Translation and Transport Generated by Palmitic Acid-Induced Lipotoxicity in Human Astrocytes: A Label-Free MS-Based Proteomics and Network Analysis" International Journal of Molecular Sciences 23, no. 12: 6454. https://doi.org/10.3390/ijms23126454
APA StyleVesga-Jiménez, D. J., Martín-Jiménez, C. A., Grismaldo Rodríguez, A., Aristizábal-Pachón, A. F., Pinzón, A., Barreto, G. E., Ramírez, D., & González, J. (2022). Tibolone Pre-Treatment Ameliorates the Dysregulation of Protein Translation and Transport Generated by Palmitic Acid-Induced Lipotoxicity in Human Astrocytes: A Label-Free MS-Based Proteomics and Network Analysis. International Journal of Molecular Sciences, 23(12), 6454. https://doi.org/10.3390/ijms23126454









