Cartilage Homeostasis and Osteoarthritis
Abstract
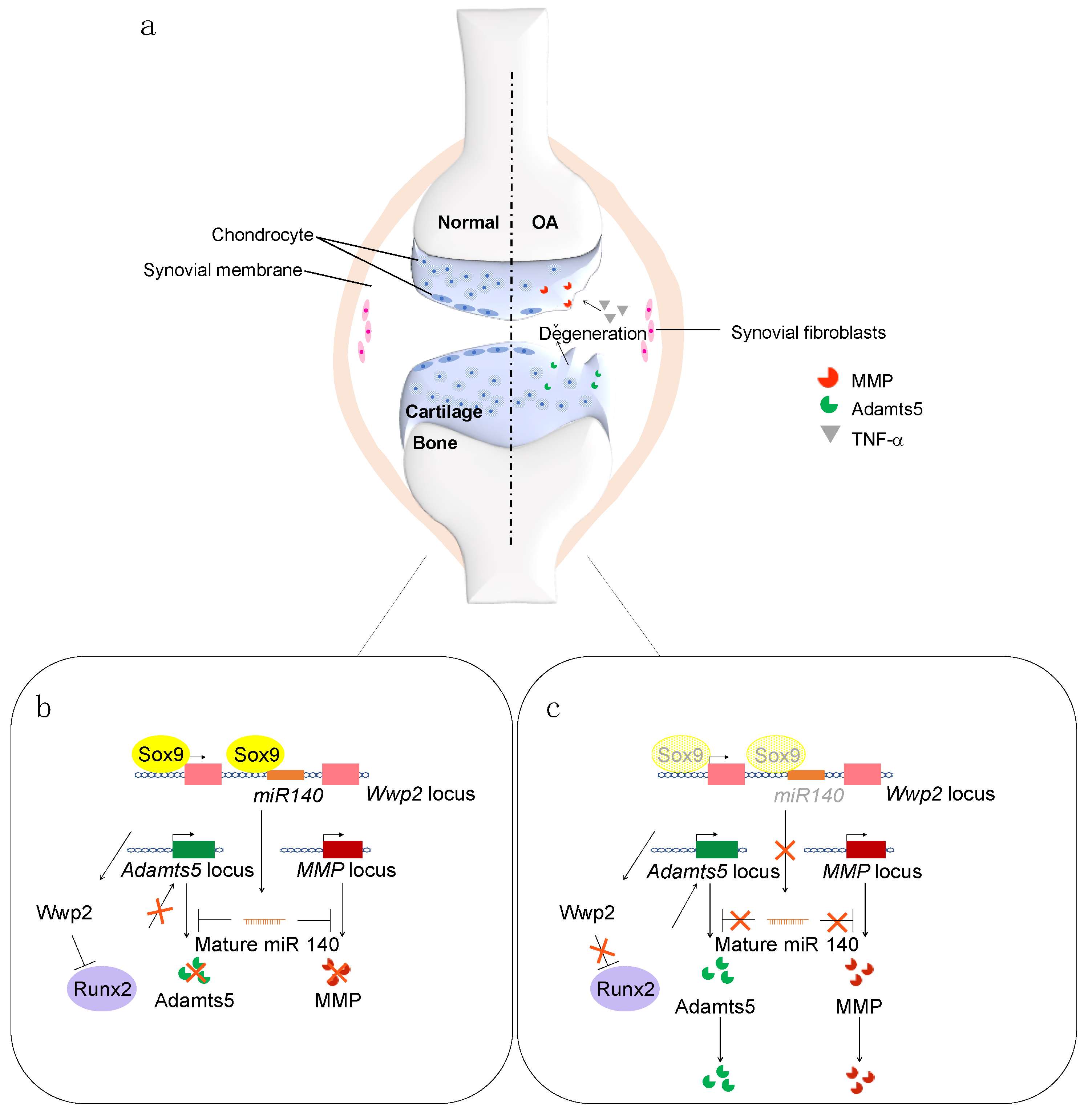
1. Introduction
2. The Role of Sox9 on Chondrogenesis
3. The Effect of Sox9 on Cartilage Homeostasis and OA
4. ncRNAs Involved in Cartilage Homeostasis and OA
4.1. MiRNAs
4.1.1. MiRNA-140
4.1.2. MiR-455
4.1.3. MiRNAs Regulating Hif-2α
4.1.4. Other miRNAs
4.2. CircRNAs
4.2.1. CircRNAs and Idiopathic Short Stature (ISS)
4.2.2. CircRNAs and OA
4.3. MiRNAs and Diseases
4.3.1. Disease-Related miRNAs
4.3.2. MiRNAs and Skeletal Dysplasia
4.3.3. Gain-of-Function Mutation of miR-140
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Acan | Aggrecan |
| ADAMTS | a disintegrin and metalloproteinases with thrombospondin motif |
| ceRNA | competing endogenous RNA |
| ChIP-Seq | chromatin immunoprecipitation sequencing |
| circRNA | circular RNA |
| Col2a1 | collagen type II alpha 1 |
| Col9a2 | collagen type IX alpha 2 |
| Col11a2 | collagen type XI alpha 2 |
| Col10a1 | collagen type X alpha 1 |
| Col27a1 | collagen type XXVII alpha 1 |
| ceRNA | competing endogenous RNA |
| CRISPR-ChIP-MS | CRISPR-ChIP-mass spectrometry |
| DMM | destabilization of the medial meniscus |
| DNMT | DNA methyltransferase |
| ECM | extracellular matrix |
| EPAS1 | endothelial PAS domain protein 1 |
| ES | embryonic stem |
| GAS5 | Growth-arrest-specific 5 |
| hADSC | human adipose-derived stem cell |
| HDAC | histone deacetylase |
| HMG | high-mobility group |
| HIF | hypoxia inducible factor |
| hMSC | human mesenchymal stem cell |
| ISS | idiopathic short stature |
| JAK3 | Janus kinase 3 |
| KO | knockout |
| lncRNA | long ncRNA |
| miR-140 | MiRNA-140 |
| miRNA | microRNA |
| MMP | matrix metalloproteinase |
| ncRNAs | noncoding RNAs |
| OA | Osteoarthritis |
| OARSI | Osteoarthritis Research Society International |
| PAK2 | P21-activated kinase |
| PARylation | poly(ADP-ribosyl)ation |
| piRNAs | Piwi-interacting RNAs |
| pre-miRNA | precursor miRNA |
| pri-miRNA | primary miRNA |
| RCSE | rib cage-specific enhancer |
| RISC | RNA-induced silencing complex |
| Runx2 | Runt-related transcription factor 2 |
| siRNA | small interfering RNA |
| snoRNA | small nucleolar RNA |
| Sox5 | SRY-box transcription factor 5 |
| Sox6 | SRY-box transcription factor 6 |
| Sox9 | SRY-box transcription factor 9 |
| tRF | tRNA-derived fragment |
| tRNA | transition RNA |
| TGFβ | transforming growth factor-β |
| VMA21 | vacuolar ATPase assembly factor 21 |
| WT | wild-type |
| Wwp2 | WW domain-containing protein 2 |
References
- Lotz, M.K. New Developments in Osteoarthritis. Posttraumatic Osteoarthritis: Pathogenesis and Pharmacological Treatment Options. Arthritis Res. Ther. 2010, 12, 211. [Google Scholar] [CrossRef]
- Hashimoto, M.; Nakasa, T.; Hikata, T.; Asahara, H. Molecular Network of Cartilage Homeostasis and Osteoarthritis. Med. Res. Rev. 2008, 28, 464–481. [Google Scholar] [CrossRef] [PubMed]
- Goldring, M.B.; Marcu, K.B. Cartilage Homeostasis in Health and Rheumatic Diseases. Arthritis Res. Ther. 2009, 11, 224. [Google Scholar] [CrossRef]
- Lotz, M. Osteoarthritis Year 2011 in Review: Biology. Osteoarthr. Cartil. 2012, 20, 192–196. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jiang, Y. Osteoarthritis Year in Review 2021: Biology. Osteoarthr. Cartil. 2022, 30, 207–215. [Google Scholar] [CrossRef]
- Mow, V.C.; Ratcliffe, A.; Robin Poole, A. Cartilage and Diarthrodial Joints as Paradigms for Hierarchical Materials and Structures. Biomaterials 1992, 13, 67–97. [Google Scholar] [CrossRef]
- Ratneswaran, A.; Rockel, J.S.; Kapoor, M. Understanding Osteoarthritis Pathogenesis: A Multiomics System-Based Approach. Curr. Opin. Rheumatol. 2020, 32, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, C.; Bay-Jensen, A.C.; Pap, T.; Dvir-Ginzberg, M.; Quasnichka, H.; Barrett-Jolley, R.; Mobasheri, A.; Henrotin, Y. Chondrocyte Secretome: A Source of Novel Insights and Exploratory Biomarkers of Osteoarthritis. Osteoarthr. Cartil. 2017, 25, 1199–1209. [Google Scholar] [CrossRef]
- Polacek, M.; Bruun, J.A.; Johansen, O.; Martinez, I. Differences in the Secretome of Cartilage Explants and Cultured Chondrocytes Unveiled by SILAC Technology. J. Orthop. Res. 2010, 28, 1040–1049. [Google Scholar] [CrossRef]
- Calamia, V.; Lourido, L.; Fernández-Puente, P.; Mateos, J.; Rocha, B.; Montell, E.; Vergés, J.; Ruiz-Romero, C.; Blanco, F.J. Secretome Analysis of Chondroitin Sulfate-Treated Chondrocytes Reveals Anti-Angiogenic, Anti-Inflammatory and Anti-Catabolic Properties. Arthritis Res. Ther. 2012, 14, R202. [Google Scholar] [CrossRef] [PubMed]
- Calamia, V.; Mateos, J.; Fernández-Puente, P.; Lourido, L.; Rocha, B.; Fernández-Costa, C.; Montell, E.; Vergés, J.; Ruiz-Romero, C.; Blanco, F.J. A Pharmacoproteomic Study Confirms the Synergistic Effect of Chondroitin Sulfate and Glucosamine. Sci. Rep. 2014, 4, 5069. [Google Scholar] [CrossRef]
- Catterall, J.B.; Rowan, A.D.; Sarsfield, S.; Saklatvala, J.; Wait, R.; Cawston, T.E. Development of a Novel 2D Proteomics Approach for the Identification of Proteins Secreted by Primary Chondrocytes after Stimulation by IL-1 and Oncostatin M. Rheumatology 2006, 45, 1101–1109. [Google Scholar] [CrossRef] [PubMed]
- Riffault, M.; Moulin, D.; Grossin, L.; Mainard, D.; Magdalou, J.; Vincourt, J.B. Label-Free Relative Quantification Applied to LC-MALDI Acquisition for Rapid Analysis of Chondrocyte Secretion Modulation. J. Proteom. 2015, 114, 263–273. [Google Scholar] [CrossRef]
- Polacek, M.; Bruun, J.A.; Elvenes, J.; Figenschau, Y.; Martinez, I. The Secretory Profiles of Cultured Human Articular Chondrocytes and Mesenchymal Stem Cells: Implications for Autologous Cell Transplantation Strategies. Cell Transplant. 2011, 20, 1381–1393. [Google Scholar] [CrossRef] [PubMed]
- Polacek, M.; Bruun, J.A.; Johansen, O.; Martinez, I. Comparative Analyses of the Secretome from Dedifferentiated and Redifferentiated Adult Articular Chondrocytes. Cartilage 2011, 2, 186–196. [Google Scholar] [CrossRef]
- Haglund, L.; Bernier, S.M.; Önnerfjord, P.; Recklies, A.D. Proteomic Analysis of the LPS-Induced Stress Response in Rat Chondrocytes Reveals Induction of Innate Immune Response Components in Articular Cartilage. Matrix Biol. 2008, 27, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Lourido, L.; Calamia, V.; Fernández-Puente, P.; Mateos, J.; Oreiro, N.; Blanco, F.J.; Ruiz-Romero, C. Secretome Analysis of Human Articular Chondrocytes Unravels Catabolic Effects of Nicotine on the Joint. Proteom. Clin. Appl. 2016, 10, 671–680. [Google Scholar] [CrossRef]
- Stenberg, J.; Rüetschi, U.; Skiöldebrand, E.; Kärrholm, J.; Lindahl, A. Quantitative Proteomics Reveals Regulatory Differences in the Chondrocyte Secretome from Human Medial and Lateral Femoral Condyles in Osteoarthritic Patients. Proteome Sci. 2013, 11, 43. [Google Scholar] [CrossRef]
- Taylor, D.W.; Ahmed, N.; Parreno, J.; Lunstrum, G.P.; Gross, A.E.; Diamandis, E.P.; Kandel, R.A. Collagen Type XII and Versican Are Present in the Early Stages of Cartilage Tissue Formation by Both Redifferentating Passaged and Primary Chondrocytes. Tissue Eng. Part A 2015, 21, 683–693. [Google Scholar] [CrossRef]
- Balakrishnan, L.; Bhattacharjee, M.; Ahmad, S.; Nirujogi, R.S.; Renuse, S.; Subbannayya, Y.; Marimuthu, A.; Srikanth, S.M.; Raju, R.; Dhillon, M.; et al. Differential Proteomic Analysis of Synovial Fluid from Rheumatoid Arthritis and Osteoarthritis Patients. Clin. Proteom. 2014, 11, 1. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.; Guo, J.; Luo, Y.; Zhang, W.; Cui, Y.; Wang, Q.; Zhang, Z.; Wang, T. Functional Proteomics Revealed IL-1β Amplifies TNF Downstream Protein Signals in Human Synoviocytes in a TNF-Independent Manner. Biochem. Biophys. Res. Commun. 2014, 450, 538–544. [Google Scholar] [CrossRef]
- Gobezie, R.; Kho, A.; Krastins, B.; Sarracino, D.A.; Thornhill, T.S.; Chase, M.; Millett, P.J.; Lee, D.M. High Abundance Synovial Fluid Proteome: Distinct Profiles in Health and Osteoarthritis. Arthritis Res. Ther. 2007, 9, R36. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, L.; Nirujogi, R.S.; Ahmad, S.; Bhattacharjee, M.; Manda, S.S.; Renuse, S.; Kelkar, D.S.; Subbannayya, Y.; Raju, R.; Goel, R.; et al. Poteomic Analysis of Human Osteoarthritis Synovial Fluid. Clin. Proteomics 2014, 11, 6. [Google Scholar] [CrossRef]
- Kamphorst, J.J.; Van Der Heijden, R.; DeGroot, J.; Lafeber, F.P.J.G.; Reijmers, T.H.; Van El, B.; Tjaden, U.R.; Van Der Greef, J.; Hankemeier, T. Profiling of Endogenous Peptides in Human Synovial Fluid by NanoLC-MS: Method Validation and Peptide Identification. J. Proteome Res. 2007, 6, 4388–4396. [Google Scholar] [CrossRef]
- Ritter, S.Y.; Subbaiah, R.; Bebek, G.; Crish, J.; Scanzello, C.R.; Krastins, B.; Sarracino, D.; Lopez, M.F.; Crow, M.K.; Aigner, T.; et al. Proteomic Analysis of Synovial Fluid from the Osteoarthritic Knee: Comparison with Transcriptome Analyses of Joint Tissues. Arthritis Rheum. 2013, 65, 981–992. [Google Scholar] [CrossRef] [PubMed]
- Yamagiwa, H.; Sarkar, G.; Charlesworth, M.C.; McCormick, D.J.; Bolander, M.E. Two-Dimensional Gel Electrophoresis of Synovial Fluid: Method for Detecting Candidate Protein Markers for Osteoarthritis. J. Orthop. Sci. 2003, 8, 482–490. [Google Scholar] [CrossRef]
- Russo, R.; Vassallo, V.; Stellavato, A.; Valletta, M.; Cimini, D.; Pedone, P.V.; Schiraldi, C.; Chambery, A. Differential Secretome Profiling of Human Osteoarthritic Synoviocytes Treated with Biotechnological Unsulfated and Marine Sulfated Chondroitins. Int. J. Mol. Sci. 2020, 21, 3746. [Google Scholar] [CrossRef]
- Lotz, M. Cytokines in Cartilage Injury and Repair. Clin. Orthop. Relat. Res. 2001, 391, S108–S115. [Google Scholar] [CrossRef] [PubMed]
- Goldring, M.B. The Role of the Chondrocyte in Osteoarthritis. Arthritis Rheum. 2000, 43, 1916–1926. [Google Scholar] [CrossRef]
- Gravallese, E.M.; Goldring, S.R. Cellular mechanisms and the role of cytokines in bone erosions in rheumatoid arthritis. Arthritis Rheum. 2000, 43, 2143–2151. [Google Scholar] [CrossRef]
- Hayami, T.; Pickarski, M.; Zhuo, Y.; Wesolowski, G.A.; Rodan, G.A.; Duong, L.T. Characterization of Articular Cartilage and Subchondral Bone Changes in the Rat Anterior Cruciate Ligament Transection and Meniscectomized Models of Osteoarthritis. Bone 2006, 38, 234–243. [Google Scholar] [CrossRef] [PubMed]
- Bleuel, J.; Zaucke, F.; Brüggemann, G.P.; Niehoff, A. Effects of Cyclic Tensile Strain on Chondrocyte Metabolism: A Systematic Review. PLoS ONE 2015, 10, e0119816. [Google Scholar] [CrossRef]
- Lotz, M.; Loeser, R.F. Effects of Aging on Articular Cartilage Homeostasis. Bone 2012, 51, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Loeser, R.F.; Collins, J.A.; Diekman, B.O. Ageing and the Pathogenesis of Osteoarthritis. Nat. Rev. Rheumatol. 2016, 12, 412–420. [Google Scholar] [CrossRef]
- McCulloch, K.; Litherland, G.J.; Rai, T.S. Cellular Senescence in Osteoarthritis Pathology. Aging Cell 2017, 16, 210–218. [Google Scholar] [CrossRef]
- Kurakazu, I.; Akasaki, Y.; Tsushima, H.; Sueishi, T.; Toya, M.; Kuwahara, M.; Uchida, T.; Lotz, M.K.; Nakashima, Y. TGFβ1 Signaling Protects Chondrocytes against Oxidative Stress via FOXO1–Autophagy Axis. Osteoarthr. Cartil. 2021, 29, 1600–1613. [Google Scholar] [CrossRef] [PubMed]
- Roemer, F.W.; Guermazi, A.; Felson, D.T.; Niu, J.; Nevitt, M.C.; Crema, M.D.; Lynch, J.A.; Lewis, C.E.; Torner, J.; Zhang, Y. Presence of MRI-Detected Joint Effusion and Synovitis Increases the Risk of Cartilage Loss in Knees without Osteoarthritis at 30-Month Follow-up: The MOST Study. Ann. Rheum. Dis. 2011, 70, 1804–1809. [Google Scholar] [CrossRef]
- Xie, Y.; Zinkle, A.; Chen, L.; Mohammadi, M. Fibroblast Growth Factor Signalling in Osteoarthritis and Cartilage Repair. Nat. Rev. Rheumatol. 2020, 16, 547–564. [Google Scholar] [CrossRef]
- Liu-Bryan, R.; Terkeltaub, R. Emerging Regulators of the Inflammatory Process in Osteoarthritis. Nat. Rev. Rheumatol. 2015, 11, 35–44. [Google Scholar] [CrossRef]
- Peffers, M.J.; Milner, P.I.; Tew, S.R.; Clegg, P.D. Regulation of SOX9 in Normal and Osteoarthritic Equine Articular Chondrocytes by Hyperosmotic Loading. Osteoarthr. Cartil. 2010, 18, 1502–1508. [Google Scholar] [CrossRef]
- Bian, Q.; Wang, Y.-J.; Liu, S.-F.; Li, Y.-P. Osteoarthritis: Genetic Factors, Animal Models, Mechanisms, and Therapies. Front. Biosci. 2012, 4, 74–100. [Google Scholar] [CrossRef]
- de Crombrugghe, B.; Lefebvre, V.; Nakashima, K. Regulatory Mechanisms in the Pathways of Cartilage and Bone Formation. Curr. Opin. Cell Biol. 2001, 13, 721–728. [Google Scholar] [CrossRef]
- Bi, W.; Deng, J.M.; Zhang, Z.; Behringer, R.R.; De Crombrugghe, B. Sox9 Is Required for Cartilage Formation. Nat. Genet. 1999, 22, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Bi, W.; Huang, W.; Whitworth, D.J.; Deng, J.M.; Zhang, Z.; Behringer, R.R.; De Crombrugghe, B. Haploinsufficiency of Sox9 Results in Defective Cartilage Primordia and Premature Skeletal Mineralization. Proc. Natl. Acad. Sci. USA 2001, 98, 6698–6703. [Google Scholar] [CrossRef]
- Cheah, K.S.E.; Lau, E.T.; Au, P.K.C.; Tam, P.P.L. Expression of the Mouse A1(II) Collagen Gene Is Not Restricted to Cartilage during Development. Development 1991, 111, 945–953. [Google Scholar] [CrossRef]
- Lefebvre, V.; Li, P.; De Crombrugghe, B. A New Long Form of Sox5 (L-Sox5), Sox6 and Sox9 Are Coexpressed in Chondrogenesis and Cooperatively Activate the Type II Collagen Gene. EMBO J. 1998, 17, 5718–5733. [Google Scholar] [CrossRef]
- Lefebvre, V.; Huang, W.; Harley, V.R.; Goodfellow, P.N.; de Crombrugghe, B. SOX9 Is a Potent Activator of the Chondrocyte-Specific Enhancer of the pro Alpha1(II) Collagen Gene. Mol. Cell. Biol. 1997, 17, 2336–2346. [Google Scholar] [CrossRef]
- Wright, E.; Hargrave, M.R.; Christiansen, J.; Cooper, L.; Kun, J.; Evans, T.; Gangadharan, U.; Greenfield, A.; Koopman, P. The Sry-Related Gene Sox9 Is Expressed during Chondrogenesis in Mouse Embryos. Nat. Genet. 1995, 9, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, T.; Kawaguchi, H.; Kamekura, S.; Ogata, N.; Mori, Y.; Nakamura, K.; Ikegawa, S.; Chung, U. Il Distinct Roles of Sox5, Sox6, and Sox9 in Different Stages of Chondrogenic Differentiation. J. Bone Miner. Metab. 2005, 23, 337–340. [Google Scholar] [CrossRef]
- Yamashita, S.; Miyaki, S.; Kato, Y.; Yokoyama, S.; Sato, T.; Barrionuevo, F.; Akiyama, H.; Scherer, G.; Takada, S.; Asahara, H. L-Sox5 and Sox6 Proteins Enhance Chondrogenic MiR-140 MicroRNA Expression by Strengthening Dimeric Sox9 Activity. J. Biol. Chem. 2012, 287, 22206–22215. [Google Scholar] [CrossRef]
- Lefrebvre, V.; de Crombrugghe, B. Toward Understanding S0X9 Function in Chondrocyte Differentiation. Matrix Biol. 1998, 16, 529–540. [Google Scholar] [CrossRef]
- Zhao, Q.; Eberspaecher, H.; Lefebvre, V.; De Crombrugghe, B. Parallel Expression of Sox9 and Col2a1 in Cells Undergoing Chondrogenesis. Dev. Dyn. 1997, 209, 377–386. [Google Scholar] [CrossRef]
- Chambers, M.G.; Kuffner, T.; Cowan, S.K.; Cheah, K.S.E.; Mason, R.M. Expression of Collagen and Aggrecan Genes in Normal and Osteoarthritic Murine Knee Joints. Osteoarthr. Cartil. 2002, 10, 51–61. [Google Scholar] [CrossRef]
- Haseeb, A.; Kc, R.; Angelozzi, M.; de Charleroy, C.; Rux, D.; Tower, R.J.; Yao, L.; da Silva, R.P.; Pacifici, M.; Qin, L.; et al. SOX9 Keeps Growth Plates and Articular Cartilage Healthy by Inhibiting Chondrocyte Dedifferentiation/ Osteoblastic Redifferentiation. Proc. Natl. Acad. Sci. USA 2021, 118, e2019152118. [Google Scholar] [CrossRef]
- Henry, S.P.; Jang, C.W.; Deng, J.M.; Zhang, Z.; Behringer, R.R.; De Crombrugghe, B. Generation of Aggrecan-CreERT2 Knockin Mice for Inducible Cre Activity in Adult Cartilage. Genesis 2009, 47, 805–814. [Google Scholar] [CrossRef]
- Oh, C.D.; Lu, Y.; Liang, S.; Mori-Akiyama, Y.; Chen, D.; De Crombrugghe, B.; Yasuda, H. SOX9 Regulates Multiple Genes in Chondrocytes, Including Genes Encoding ECM Proteins, ECM Modification Enzymes, Receptors, and Transporters. PLoS ONE 2014, 9, e107577. [Google Scholar] [CrossRef] [PubMed]
- Mochizuki, Y.; Chiba, T.; Kataoka, K.; Yamashita, S.; Sato, T.; Kato, T.; Takahashi, K.; Miyamoto, T.; Kitazawa, M.; Hatta, T.; et al. Combinatorial CRISPR/Cas9 Approach to Elucidate a Far-Upstream Enhancer Complex for Tissue-Specific Sox9 Expression. Dev. Cell 2018, 46, 794–806.e6. [Google Scholar] [CrossRef]
- Johnson, K.; Zhu, S.; Tremblay, M.S.; Payette, J.N.; Wang, J.; Bouchez, L.C.; Meeusen, S.; Althage, A.; Cho, C.Y.; Wu, X.; et al. A Stem Cell-Based Approach to Cartilage Repair. Science 2012, 336, 717–721. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, Y.; Wang, W.; Tu, B.; Zhu, Y.; Fan, C.; Li, Y. Overexpression of SOX9 Alleviates the Progression of Human Osteoarthritis in Vitro and in Vivo. Drug Des. Dev. Ther. 2019, 13, 2833–2842. [Google Scholar] [CrossRef]
- Kim, S.; Han, S.; Kim, Y.; Kim, H.S.; Gu, Y.R.; Kang, D.; Cho, Y.; Kim, H.; Lee, J.; Seo, Y.; et al. Tankyrase Inhibition Preserves Osteoarthritic Cartilage by Coordinating Cartilage Matrix Anabolism via Effects on SOX9 PARylation. Nat. Commun. 2019, 10, 4898. [Google Scholar] [CrossRef]
- Hattori, M. Finishing the Euchromatic Sequence of the Human Genome. Tanpakushitsu Kakusan Koso 2005, 50, 162–168. (In Japanese) [Google Scholar]
- Peffers, M.J.; Chabronova, A.; Balaskas, P.; Fang, Y.; Dyer, P.; Cremers, A.; Emans, P.J.; Feczko, P.Z.; Caron, M.M.; Welting, T.J.M. SnoRNA Signatures in Cartilage Ageing and Osteoarthritis. Sci. Rep. 2020, 10, 10641. [Google Scholar] [CrossRef]
- Green, J.A.; Ansari, M.Y.; Ball, H.C.; Haqqi, T.M. TRNA-Derived Fragments (TRFs) Regulate Post-Transcriptional Gene Expression via AGO-Dependent Mechanism in IL-1β Stimulated Chondrocytes. Osteoarthr. Cartil. 2020, 28, 1102–1110. [Google Scholar] [CrossRef]
- Balaskas, P.; Green, J.A.; Haqqi, T.M.; Dyer, P.; Kharaz, Y.A.; Fang, Y.; Liu, X.; Welting, T.J.M.; Peffers, M.J. Small Non-Coding Rnaome of Ageing Chondrocytes. Int. J. Mol. Sci. 2020, 21, 5675. [Google Scholar] [CrossRef]
- Zacharjasz, J.; Mleczko, A.M.; Bakowski, P.; Piontek, T.; Bakowska-żywicka, K. Small Noncoding Rnas in Knee Osteoarthritis: The Role of Micrornas and Trna-Derived Fragments. Int. J. Mol. Sci. 2021, 22, 5711. [Google Scholar] [CrossRef]
- Young, D.A.; Barter, M.J.; Soul, J. Osteoarthritis Year in Review: Genetics, Genomics, Epigenetics. Osteoarthr. Cartil. 2022, 30, 216–225. [Google Scholar] [CrossRef]
- Harfe, B.D.; McManus, M.T.; Mansfield, J.H.; Hornstein, E.; Tabin, C.J. The RNaseIII Enzyme Dicer Is Required for Morphogenesis but Not Patterning of the Vertebrate Limb. Proc. Natl. Acad. Sci. USA 2005, 102, 10898–10903. [Google Scholar] [CrossRef]
- Kobayashi, T.; Lu, J.; Cobb, B.S.; Rodda, S.J.; McMahon, A.P.; Schipani, E.; Merkenschlager, M.; Kronenberg, H.M. Dicer-Dependent Pathways Regulate Chondrocyte Proliferation and Differentiation. Proc. Natl. Acad. Sci. USA 2008, 105, 1949–1954. [Google Scholar] [CrossRef]
- Iliopoulos, D.; Malizos, K.N.; Oikonomou, P.; Tsezou, A. Integrative MicroRNA and Proteomic Approaches Identify Novel Osteoarthritis Genes and Their Collaborative Metabolic and Inflammatory Networks. PLoS ONE 2008, 3, e3740. [Google Scholar] [CrossRef] [PubMed]
- Cong, L.; Zhu, Y.; Tu, G. A Bioinformatic Analysis of MicroRNAs Role in Osteoarthritis. Osteoarthr. Cartil. 2017, 25, 1362–1371. [Google Scholar] [CrossRef]
- Wienholds, E.; Kloosterman, W.P.; Miska, E.; Alvarez-Saavedra, E.; Berezikov, E.; de Bruijn, E.; Horvitz, H.R.; Kauppinen, S.; Plasterk, R.H.A. MicroRNA Expression in Zebrafish Embryonic Development. Science 2005, 309, 310–311. [Google Scholar] [CrossRef]
- Tuddenham, L.; Wheeler, G.; Ntounia-Fousara, S.; Waters, J.; Hajihosseini, M.K.; Clark, I.; Dalmay, T. The Cartilage Specific MicroRNA-140 Targets Histone Deacetylase 4 in Mouse Cells. FEBS Lett. 2006, 580, 4214–4217. [Google Scholar] [CrossRef]
- Miyaki, S.; Nakasa, T.; Otsuki, S.; Grogan, S.P.; Higashiyama, R.; Inoue, A.; Kato, Y.; Sato, T.; Lotz, M.K.; Asahara, H. MicroRNA-140 Is Expressed in Differentiated Human Articular Chondrocytes and Modulates Interleukin-1 Responses. Arthritis Rheum. 2009, 60, 2723–2730. [Google Scholar] [CrossRef]
- Miyaki, S.; Sato, T.; Inoue, A.; Otsuki, S.; Ito, Y.; Yokoyama, S.; Kato, Y.; Takemoto, F.; Nakasa, T.; Yamashita, S.; et al. MicroRNA-140 Plays Dual Roles in Both Cartilage Development and Homeostasis. Genes Dev. 2010, 24, 1173–1185. [Google Scholar] [CrossRef]
- Bernassola, F.; Karin, M.; Ciechanover, A.; Melino, G. The HECT Family of E3 Ubiquitin Ligases: Multiple Players in Cancer Development. Cancer Cell 2008, 14, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Rotin, D.; Kumar, S. Physiological Functions of the HECT Family of Ubiquitin Ligases. Nat. Rev. Mol. Cell Biol. 2009, 10, 398–409. [Google Scholar] [CrossRef]
- Yang, J.; Qin, S.; Yi, C.; Ma, G.; Zhu, H.; Zhou, W.; Xiong, Y.; Zhu, X.; Wang, Y.; He, L.; et al. MiR-140 Is Co-Expressed with Wwp2-C Transcript and Activated by Sox9 to Target Sp1 in Maintaining the Chondrocyte Proliferation. FEBS Lett. 2011, 585, 2992–2997. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; He, X.; Kato, H.; Wakitani, S.; Kobayashi, T.; Watanabe, S.; Iida, A.; Tahara, H.; Warman, M.L.; Watanapokasin, R.; et al. Sox9 Is Upstream of MicroRNA-140 in Cartilage. Appl. Biochem. Biotechnol. 2012, 166, 64–71. [Google Scholar] [CrossRef]
- Han, Y.; Lefebvre, V. L-Sox5 and Sox6 Drive Expression of the Aggrecan Gene in Cartilage by Securing Binding of Sox9 to a Far-Upstream Enhancer. Mol. Cell. Biol. 2008, 28, 4999–5013. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Inloes, J.B.; Katagiri, T.; Kobayashi, T. Chondrocyte-Specific MicroRNA-140 Regulates Endochondral Bone Development and Targets Dnpep to Modulate Bone Morphogenetic Protein Signaling. Mol. Cell. Biol. 2011, 31, 3019–3028. [Google Scholar] [CrossRef]
- Zou, W.; Chen, X.; Shim, J.H.; Huang, Z.; Brady, N.; Hu, D.; Drapp, R.; Sigrist, K.; Glimcher, L.H.; Jones, D. The E3 Ubiquitin Ligase Wwp2 Regulates Craniofacial Development through Mono-Ubiquitylation of Goosecoid. Nat. Cell Biol. 2011, 13, 59–65. [Google Scholar] [CrossRef]
- Inui, M.; Mokuda, S.; Sato, T.; Tamano, M.; Takada, S.; Asahara, H. Dissecting the Roles of MiR-140 and Its Host Gene. Nat. Cell Biol. 2018, 20, 516–518. [Google Scholar] [CrossRef]
- Mokuda, S.; Nakamichi, R.; Matsuzaki, T.; Ito, Y.; Sato, T.; Miyata, K.; Inui, M.; Olmer, M.; Sugiyama, E.; Lotz, M.; et al. Wwp2 Maintains Cartilage Homeostasis through Regulation of Adamts5. Nat. Commun. 2019, 10, 2429. [Google Scholar] [CrossRef] [PubMed]
- Lalevée, S.; Lapaire, O.; Bühler, M. MiR455 Is Linked to Hypoxia Signaling and Is Deregulated in Preeclampsia. Cell Death Dis. 2014, 5, e1408. [Google Scholar] [CrossRef]
- Swingler, T.E.; Wheeler, G.; Carmont, V.; Elliott, H.R.; Barter, M.J.; Abu-Elmagd, M.; Donell, S.T.; Boot-Handford, R.P.; Hajihosseini, M.K.; Münsterberg, A.; et al. The Expression and Function of MicroRNAs in Chondrogenesis and Osteoarthritis. Arthritis Rheum. 2012, 64, 1909–1919. [Google Scholar] [CrossRef]
- Zhang, Z.; Kang, Y.; Zhang, Z.; Zhang, H.; Duan, X.; Liu, J.; Li, X.; Liao, W. Expression of MicroRNAs during Chondrogenesis of Human Adipose-Derived Stem Cells. Osteoarthr. Cartil. 2012, 20, 1638–1646. [Google Scholar] [CrossRef]
- Zhang, Z.; Hou, C.; Meng, F.; Zhao, X.; Zhang, Z.; Huang, G.; Chen, W.; Fu, M.; Liao, W. MiR-455-3p Regulates Early Chondrogenic Differentiation via Inhibiting Runx2. FEBS Lett. 2015, 589, 3671–3678. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Chen, L.; Zhang, Z.; Meng, F.; Huang, G.; Sheng, P.; Zhang, Z.; Liao, W. MicroRNA-455-3p Modulates Cartilage Development and Degeneration through Modification of Histone H3 Acetylation. Biochim. Biophys. Acta Mol. Cell Res. 2016, 1863, 2881–2891. [Google Scholar] [CrossRef]
- Hu, S.; Zhao, X.; Mao, G.; Zhang, Z.; Wen, X.; Zhang, C.; Liao, W.; Zhang, Z. MicroRNA-455-3p Promotes TGF-β Signaling and Inhibits Osteoarthritis Development by Directly Targeting PAK2. Exp. Mol. Med. 2019, 51, 1–13. [Google Scholar] [CrossRef]
- Sun, H.; Zhao, X.; Zhang, C.; Zhang, Z.; Lun, J.; Liao, W.; Zhang, Z. MiR-455-3p Inhibits the Degenerate Process of Chondrogenic Differentiation through Modification of DNA Methylation Article. Cell Death Dis. 2018, 9, 537. [Google Scholar] [CrossRef] [PubMed]
- Baskerville, S.; Bartel, D.P. Microarray Profiling of MicroRNAs Reveals Frequent Coexpression with Neighboring MiRNAs and Host Genes. RNA 2005, 11, 241–247. [Google Scholar] [CrossRef]
- Rodriguez, A.; Griffiths-Jones, S.; Ashurst, J.L.; Bradley, A. Identification of Mammalian MicroRNA Host Genes and Transcription Units. Genome Res. 2004, 14, 1902–1910. [Google Scholar] [CrossRef] [PubMed]
- Dudek, K.A.; Lafont, J.E.; Martinez-Sanchez, A.; Murphy, C.L. Type II Collagen Expression Is Regulated by Tissue-Specific MiR-675 in Human Articular Chondrocytes. J. Biol. Chem. 2010, 285, 24381–24387. [Google Scholar] [CrossRef]
- Ito, Y.; Matsuzaki, T.; Ayabe, F.; Mokuda, S.; Kurimoto, R.; Matsushima, T.; Tabata, Y.; Inotsume, M.; Tsutsumi, H.; Liu, L.; et al. Both MicroRNA-455-5p and -3p Repress Hypoxia-Inducible Factor-2α Expression and Coordinately Regulate Cartilage Homeostasis. Nat. Commun. 2021, 12, 4148. [Google Scholar] [CrossRef]
- Slezak-Prochazka, I.; Selvi, D.; Kroesen, B.J.; Van Den Berg, A. MicroRNAs, Macrocontrol: Regulation of MiRNA Processing. RNA 2010, 16, 1087–1095. [Google Scholar] [CrossRef]
- Trabucchi, M.; Briata, P.; Filipowicz, W.; Rosenfeld, M.G.; Ramos, A.; Gherzi, R. How to Control MiRNA Maturation? RNA Biol. 2009, 6, 536–540. [Google Scholar] [CrossRef][Green Version]
- Newman, M.A.; Hammond, S.M. Emerging Paradigms of Regulated MicroRNA Processing. Genes Dev. 2010, 24, 1086–1092. [Google Scholar] [CrossRef]
- Sakurai, K.; Furukawa, C.; Haraguchi, T.; Inada, K.I.; Shiogama, K.; Tagawa, T.; Fujita, S.; Ueno, Y.; Ogata, A.; Ito, M.; et al. MicroRNAs MiR-199a-5p and -3p Target the Brm Subunit of SWI/SNF to Generate a Double-Negative Feedback Loop in a Variety of Human Cancers. Cancer Res. 2011, 71, 1680–1689. [Google Scholar] [CrossRef]
- Lim, L.P.; Lau, N.C.; Garrett-Engele, P.; Grimson, A.; Schelter, J.M.; Castle, J.; Bartel, D.P.; Linsley, P.S.; Johnson, J.M. Microarray Analysis Shows That Some MicroRNAs Downregulate Large Numbers Of-Target MRNAs. Nature 2005, 433, 769–773. [Google Scholar] [CrossRef]
- Friedman, R.C.; Farh, K.K.H.; Burge, C.B.; Bartel, D.P. Most Mammalian MRNAs Are Conserved Targets of MicroRNAs. Genome Res. 2009, 19, 92–105. [Google Scholar] [CrossRef]
- Mataki, H.; Seki, N.; Mizuno, K.; Nohata, N.; Kamikawaji, K.; Kumamoto, T.; Koshizuka, K.; Goto, Y.; Inoue, H. Dual-Strand Tumor-Suppressor MicroRNA-145 (MiR-145-5p and MiR-145-3p) Coordinately Targeted MTDH in Lung Squamous Cell Carcinoma. Oncotarget 2016, 7, 72084–72098. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Kim, J.; Ryu, J.H.; Oh, H.; Chun, C.H.; Kim, B.J.; Min, B.H.; Chun, J.S. Hypoxia-Inducible Factor-2α Is a Catabolic Regulator of Osteoarthritic Cartilage Destruction. Nat. Med. 2010, 16, 687–693. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Fukai, A.; Mabuchi, A.; Ikeda, T.; Yano, F.; Ohba, S.; Nishida, N.; Akune, T.; Yoshimura, N.; Nakagawa, T.; et al. Transcriptional Regulation of Endochondral Ossification by HIF-2α during Skeletal Growth and Osteoarthritis Development. Nat. Med. 2010, 16, 678–686. [Google Scholar] [CrossRef]
- Zhou, K.; He, S.; Yu, H.; Pei, F.; Zhou, Z. Inhibition of Syndecan-4 Reduces Cartilage Degradation in Murine Models of Osteoarthritis through the Downregulation of HIF-2α by MiR-96-5p. Lab. Investig. 2021, 101, 1060–1070. [Google Scholar] [CrossRef]
- Guan, Y.-J.; Yang, X.; Wei, L.; Chen, Q. MiR-365: A Mechanosensitive MicroRNA Stimulates Chondrocyte Differentiation through Targeting Histone Deacetylase 4. FASEB J. 2011, 25, 4457–4466. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.S.; Park, S.J.; Lee, M.H.; Kim, H.A. MicroRNA-365 Regulates IL-1β-Induced Catabolic Factor Expression by Targeting HIF-2α in Primary Chondrocytes. Sci. Rep. 2017, 7, 17889. [Google Scholar] [CrossRef]
- Seidl, C.I.; Martinez-Sanchez, A.; Murphy, C.L. Derepression of MicroRNA-138 Contributes to Loss of the Human Articular Chondrocyte Phenotype. Arthritis Rheumatol. 2016, 68, 398–409. [Google Scholar] [CrossRef]
- Bluhm, B.; Ehlen, H.W.A.; Holzer, T.; Georgieva, V.S.; Heilig, J.; Pitzler, L.; Etich, J.; Bortecen, T.; Frie, C.; Probst, K.; et al. MiR-322 Stabilizes MEK1 Expression to Inhibit RAF/MEK/ERK Pathway Activation in Cartilage. Development 2017, 144, 3562–3577. [Google Scholar] [CrossRef]
- Marchand, A.; Atassi, F.; Mougenot, N.; Clergue, M.; Codoni, V.; Berthuin, J.; Proust, C.; Trégouët, D.A.; Hulot, J.S.; Lompré, A.M. MiR-322 Regulates Insulin Signaling Pathway and Protects against Metabolic Syndrome-Induced Cardiac Dysfunction in Mice. Biochim. Biophys. Acta Mol. Basis Dis. 2016, 1862, 611–621. [Google Scholar] [CrossRef]
- Boucherat, O.; Nadeau, V.; Be, F.; Charron, J.; Jeannotte, L.; Boucherat, O.; Nadeau, V.; Be, F.; Charron, J.; Jeannotte, L. Erratum to Crucial Requirement of Erk/Mapk Signaling in Respiratory Tract Development. Development 2015, 142, 3197–3211, Erratum in Development 2015, 141, 3801. [Google Scholar] [CrossRef]
- Coccia, E.M.; Cicala, C.; Charlesworth, A.; Ciccarelli, C.; Rossi, G.B.; Philipson, L.; Sorrentino, V. Regulation and Expression of a Growth Arrest-Specific Gene (Gas5) during Growth, Differentiation, and Development. Mol. Cell. Biol. 1992, 12, 3514–3521. [Google Scholar] [CrossRef]
- Song, J.; Ahn, C.; Chun, C.H.; Jin, E.J. A Long Non-Coding RNA, GAS5, Plays a Critical Role in the Regulation of MiR-21 during Osteoarthritis. J. Orthop. Res. 2014, 32, 1628–1635. [Google Scholar] [CrossRef]
- Liu, X.; She, Y.; Wu, H.; Zhong, D.; Zhang, J. Long Non-Coding RNA Gas5 Regulates Proliferation and Apoptosis in HCS-2/8 Cells and Growth Plate Chondrocytes by Controlling FGF1 Expression via MIR-21 Regulation. J. Biomed. Sci. 2018, 25, 18. [Google Scholar] [CrossRef]
- Ventura, A.; Young, A.G.; Winslow, M.M.; Lintault, L.; Meissner, A.; Erkeland, S.J.; Newman, J.; Bronson, R.T.; Crowley, D.; Stone, J.R.; et al. Targeted Deletion Reveals Essential and Overlapping Functions of the MiR-17∼92 Family of MiRNA Clusters. Cell 2008, 132, 875–886. [Google Scholar] [CrossRef]
- De Pontual, L.; Yao, E.; Callier, P.; Faivre, L.; Drouin, V.; Cariou, S.; Van Haeringen, A.; Geneviève, D.; Goldenberg, A.; Oufadem, M.; et al. Germline Deletion of the MiR-17∼92 Cluster Causes Skeletal and Growth Defects in Humans. Nat. Genet. 2011, 43, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.Y.; Li, S.; Jin, P.; Shang, T.; Sun, R.; Kang, Y.; Zhu, W.; Wang, Q.; Zhang, X.; Yin, F.; et al. Dual Functions of MicroRNA-17 in Maintaining Cartilage Homeostasis and Protection against Osteoarthritis. Nat. Commun. 2022, 13, 2447. [Google Scholar] [CrossRef]
- Khan, S.; Brougham, C.L.; Ryan, J.; Sahrudin, A.; O’Neill, G.; Wall, D.; Curran, C.; Newell, J.; Kerin, M.J.; Dwyer, R.M. MiR-379 Regulates Cyclin B1 Expression and Is Decreased in Breast Cancer. PLoS ONE 2013, 8, e68753. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zheng, W.; Li, D.; Zheng, J. MiR-379-5p Promotes Chondrocyte Proliferation via Inhibition of PI3K/Akt Pathway by Targeting YBX1 in Osteoarthritis. Cartilage 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.L.; Yang, L. Regulation of CircRNA Biogenesis. RNA Biol. 2015, 12, 381–388. [Google Scholar] [CrossRef]
- Zhang, X.O.; Wang, H.B.; Zhang, Y.; Lu, X.; Chen, L.L.; Yang, L. Complementary Sequence-Mediated Exon Circularization. Cell 2014, 159, 134–147. [Google Scholar] [CrossRef]
- Jeck, W.R.; Sharpless, N.E. Detecting and Characterizing Circular RNAs. Nat. Biotechnol. 2014, 32, 453–461. [Google Scholar] [CrossRef]
- Jeck, W.R.; Sorrentino, J.A.; Wang, K.; Slevin, M.K.; Burd, C.E.; Liu, J.; Marzluff, W.F.; Sharpless, N.E. Circular RNAs Are Abundant, Conserved, and Associated with ALU Repeats. RNA 2013, 19, 426. [Google Scholar] [CrossRef]
- Sanger, H.L.; Klotz, G.; Riesner, D.; Gross, H.J.; Kleinschmidt, A.K. Viroids Are Single Stranded Covalently Closed Circular RNA Molecules Existing as Highly Base Paired Rod like Structures. Proc. Natl. Acad. Sci. USA 1976, 73, 3852–3856. [Google Scholar] [CrossRef] [PubMed]
- Tu, C.; He, J.; Qi, L.; Ren, X.; Zhang, C.; Duan, Z.; Yang, K.; Wang, W.; Lu, Q.; Li, Z. Emerging Landscape of Circular RNAs as Biomarkers and Pivotal Regulators in Osteosarcoma. J. Cell. Physiol. 2020, 235, 9037–9058. [Google Scholar] [CrossRef]
- Meng, S.; Zhou, H.; Feng, Z.; Xu, Z.; Tang, Y.; Li, P.; Wu, M. CircRNA: Functions and Properties of a Novel Potential Biomarker for Cancer. Mol. Cancer 2017, 16, 94. [Google Scholar] [CrossRef]
- Lin, T.; Zhang, X.; Lu, Y.; Gong, L. Identification of Circular RNA Related to Inflammation-Induced Lymphangiogenesis by Microarray Analysis. DNA Cell Biol. 2019, 38, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Li, Y.; Niringiyumukiza, J.D.; Su, P.; Xiang, W. Circular RNA Involvement in Aging: An Emerging Player with Great Potential; Elsevier Ireland Ltd: Shannon, Ireland, 2019; Volume 178. ISBN 860278369 2605.
- Li, X.; Liu, C.X.; Xue, W.; Zhang, Y.; Jiang, S.; Yin, Q.F.; Wei, J.; Yao, R.W.; Yang, L.; Chen, L.L. Coordinated CircRNA Biogenesis and Function with NF90/NF110 in Viral Infection. Mol. Cell 2017, 67, 214–227.e7. [Google Scholar] [CrossRef]
- Liu, X.; Yan, C.; Deng, X.; Jia, J. Hsa_circularRNA_0079201 Suppresses Chondrocyte Proliferation and Endochondral Ossification by Regulating the MicroRNA-140-3p/SMAD2 Signaling Pathway in Idiopathic Short Stature. Int. J. Mol. Med. 2020, 46, 1993–2006. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Yang, H.H.; Sun, Z.G.; Tang, H.B.; Min, J.K. Whole-Transcriptome Sequencing of Knee Joint Cartilage from Osteoarthritis Patients. Bone Jt. Res. 2019, 8, 290–303. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, C.; Zhang, F.; Zhang, Y.; Ren, Z.; Lammi, M.J.; Guo, X. Screening for Differentially Expressed Circular RNAs in the Cartilage of Osteoarthritis Patients for Their Diagnostic Value. Genet. Test. Mol. Biomark. 2019, 23, 706–716. [Google Scholar] [CrossRef] [PubMed]
- Xiao, K.; Xia, Z.; Feng, B.; Bian, Y.; Fan, Y.; Li, Z.; Wu, Z.; Qiu, G.; Weng, X. Circular RNA Expression Profile of Knee Condyle in Osteoarthritis by Illumina HiSeq Platform. J. Cell. Biochem. 2019, 120, 17500–17511. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Du, D.; Chen, A.; Zhu, L. Circular RNA Expression Profile of Articular Chondrocytes in an IL-1β-Induced Mouse Model of Osteoarthritis. Gene 2018, 644, 20–26. [Google Scholar] [CrossRef]
- Li, X.; Xie, C.; Xiao, F.; Su, H.; Li, Z.; Weng, J.; Huang, Y.; He, P. Circular RNA Circ_0000423 Regulates Cartilage ECM Synthesis via Circ_0000423/MiRNA-27b-3p/MMP-13 Axis in Osteoarthritis. Aging 2022, 14, 3400–3415. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Yuan, B.; Pei, Z.; Zhang, K.; Ding, Z.; Zhu, S.; Wang, Y.; Guan, Z.; Cao, Y. Circ_0136474 and MMP-13 Suppressed Cell Proliferation by Competitive Binding to MiR-127-5p in Osteoarthritis. J. Cell. Mol. Med. 2019, 23, 6554–6564. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, Y.; Zhang, Y.; Wang, J.-J. CircRNA Hsa_circ_0005105 Upregulates NAMPT Expression and Promotes Chondrocyte Extracellular Matrix Degradation by Sponging MiR-26a. Cell Biol. Int. 2017, 41, 1283–1289. [Google Scholar] [CrossRef]
- Zhu, H.; Hu, Y.; Wang, C.; Zhang, X.; He, D. CircGCN1L1 Promotes Synoviocyte Proliferation and Chondrocyte Apoptosis by Targeting MiR-330-3p and TNF-α in TMJ Osteoarthritis. Cell Death Dis. 2020, 11, 284. [Google Scholar] [CrossRef]
- Wang, Z.; Rao, Z.; Wang, X.; Jiang, C.; Yang, Y. CircPhc3 Sponging MicroRNA-93-3p Is Involved in the Regulation of Chondrocyte Function by Mechanical Instability in Osteoarthritis. Int. J. Mol. Med. 2022, 49, 6. [Google Scholar] [CrossRef]
- Luobu, Z.; Wang, L.; Jiang, D.; Liao, T.; Luobu, C.; Qunpei, L. CircSCAPER Contributes to IL-1β-Induced Osteoarthritis in Vitro via MiR-140-3p/EZH2 Axis. Bone Jt. Res. 2022, 11, 61–72. [Google Scholar] [CrossRef]
- Yang, D.; Hu, X.; Chen, Y.; Wang, C. Circular RNA Derived from Vacuolar ATPase Assembly Factor VMA21 Suppresses Lipopolysaccharide-Induced Apoptosis of Chondrocytes in Osteoarthritis (OA) by Decreasing Mature MiR-103 Production. Mol. Biotechnol. 2022. [Google Scholar] [CrossRef]
- Yu, Z.; Cong, F.; Zhang, W.; Song, T.; Zhang, S.; Jiang, R. Circular RNA Circ_0020014 Contributes to Osteoarthritis Progression via MiR-613/ADAMTS5 Axis. Bosn. J. Basic Med. Sci. 2022, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Luo, J.; Zeng, S. Circ-LRP1B Functions as a Competing Endogenous RNA to Regulate Proliferation, Apoptosis and Oxidative Stress of LPS-Induced Human C28/I2 Chondrocytes. J. Bioenerg. Biomembr. 2022, 54, 93–108. [Google Scholar] [CrossRef]
- Man, G.; Yang, H.; Shen, K.; Zhang, D.; Zhang, J.; Wu, H.; Zhang, H.; Wang, J. Circular RNA RHOT1 Regulates MiR-142-5p/CCND1 to Participate in Chondrocyte Autophagy and Proliferation in Osteoarthritis. J. Immunol. Res. 2022, 2022, 4370873. [Google Scholar] [CrossRef] [PubMed]
- Wahafu, P.; Xu, A.; Zhao, B.; Tuo, Y.; Yang, J. Circ_0005526 Contributes to Interleukin-1β-Induced Chondrocyte Injury in Osteoarthritis via Upregulating Transcription Factor 4 by Interacting with MiR-142-5p. Bioengineered 2022, 13, 8407–8418. [Google Scholar] [CrossRef]
- He, M.; Jia, Z.; Wen, Y.; Chen, X. Circ_0043947 Contributes to Interleukin 1β-Induced Injury in Chondrocytes by Sponging MiR-671-5p to up-Regulate RTN3 Expression in Osteoarthritis Pathology. J. Orthop. Surg. Res. 2022, 17, 177. [Google Scholar] [CrossRef]
- Shen, L.; Ji, C.; Lin, J.; Yang, H. Regulation of CircADAMTS6-MiR-324-5p-PIK3R3 CeRNA Pathway May Be a Novel Mechanism of IL-1β-Induced Osteoarthritic Chondrocytes. J. Bone Miner. Metab. 2022, 40, 389–401. [Google Scholar] [CrossRef]
- Mencía, A.; Modamio-Høybjør, S.; Redshaw, N.; Morín, M.; Mayo-Merino, F.; Olavarrieta, L.; Aguirre, L.A.; Del Castillo, I.; Steel, K.P.; Dalmay, T.; et al. Mutations in the Seed Region of Human MiR-96 Are Responsible for Nonsyndromic Progressive Hearing Loss. Nat. Genet. 2009, 41, 609–613. [Google Scholar] [CrossRef]
- Mortier, G.R.; Cohn, D.H.; Cormier-Daire, V.; Hall, C.; Krakow, D.; Mundlos, S.; Nishimura, G.; Robertson, S.; Sangiorgi, L.; Savarirayan, R.; et al. Nosology and Classification of Genetic Skeletal Disorders: 2019 Revision. Am. J. Med. Genet. Part A 2019, 179, 2393–2419. [Google Scholar] [CrossRef] [PubMed]
- Kannu, P.; Campos-Xavier, A.B.; Hull, D.; Martinet, D.; Ballhausen, D.; Bonafé, L. Post-Axial Polydactyly Type A2, Overgrowth and Autistic Traits Associated with a Chromosome 13q31.3 Microduplication Encompassing MiR-17-92 and GPC5. Eur. J. Med. Genet. 2013, 56, 452–457. [Google Scholar] [CrossRef]
- Hemmat, M.; Rumple, M.J.; Mahon, L.W.; Strom, C.M.; Anguiano, A.; Talai, M.; Nguyen, B.; Boyar, F.Z. Short Stature, Digit Anomalies and Dysmorphic Facial Features Are Associated with the Duplication of MiR-17 ∼ 92 Cluster. Mol. Cytogenet. 2014, 7, 27. [Google Scholar] [CrossRef]
- Grigelioniene, G.; Suzuki, H.I.; Taylan, F.; Mirzamohammadi, F.; Borochowitz, Z.U.; Ayturk, U.M.; Tzur, S.; Horemuzova, E.; Lindstrand, A.; Weis, M.A.; et al. Gain-of-Function Mutation of MicroRNA-140 in Human Skeletal Dysplasia. Nat. Med. 2019, 25, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Rajewsky, N. The Evolution of Gene Regulation by Transcription Factors and MicroRNAs. Nat. Rev. Genet. 2007, 8, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Berezikov, E. Evolution of MicroRNA Diversity and Regulation in Animals. Nat. Rev. Genet. 2011, 12, 846–860. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fujii, Y.; Liu, L.; Yagasaki, L.; Inotsume, M.; Chiba, T.; Asahara, H. Cartilage Homeostasis and Osteoarthritis. Int. J. Mol. Sci. 2022, 23, 6316. https://doi.org/10.3390/ijms23116316
Fujii Y, Liu L, Yagasaki L, Inotsume M, Chiba T, Asahara H. Cartilage Homeostasis and Osteoarthritis. International Journal of Molecular Sciences. 2022; 23(11):6316. https://doi.org/10.3390/ijms23116316
Chicago/Turabian StyleFujii, Yuta, Lin Liu, Lisa Yagasaki, Maiko Inotsume, Tomoki Chiba, and Hiroshi Asahara. 2022. "Cartilage Homeostasis and Osteoarthritis" International Journal of Molecular Sciences 23, no. 11: 6316. https://doi.org/10.3390/ijms23116316
APA StyleFujii, Y., Liu, L., Yagasaki, L., Inotsume, M., Chiba, T., & Asahara, H. (2022). Cartilage Homeostasis and Osteoarthritis. International Journal of Molecular Sciences, 23(11), 6316. https://doi.org/10.3390/ijms23116316





