Chemical Probes and Activity-Based Protein Profiling for Cancer Research
Abstract
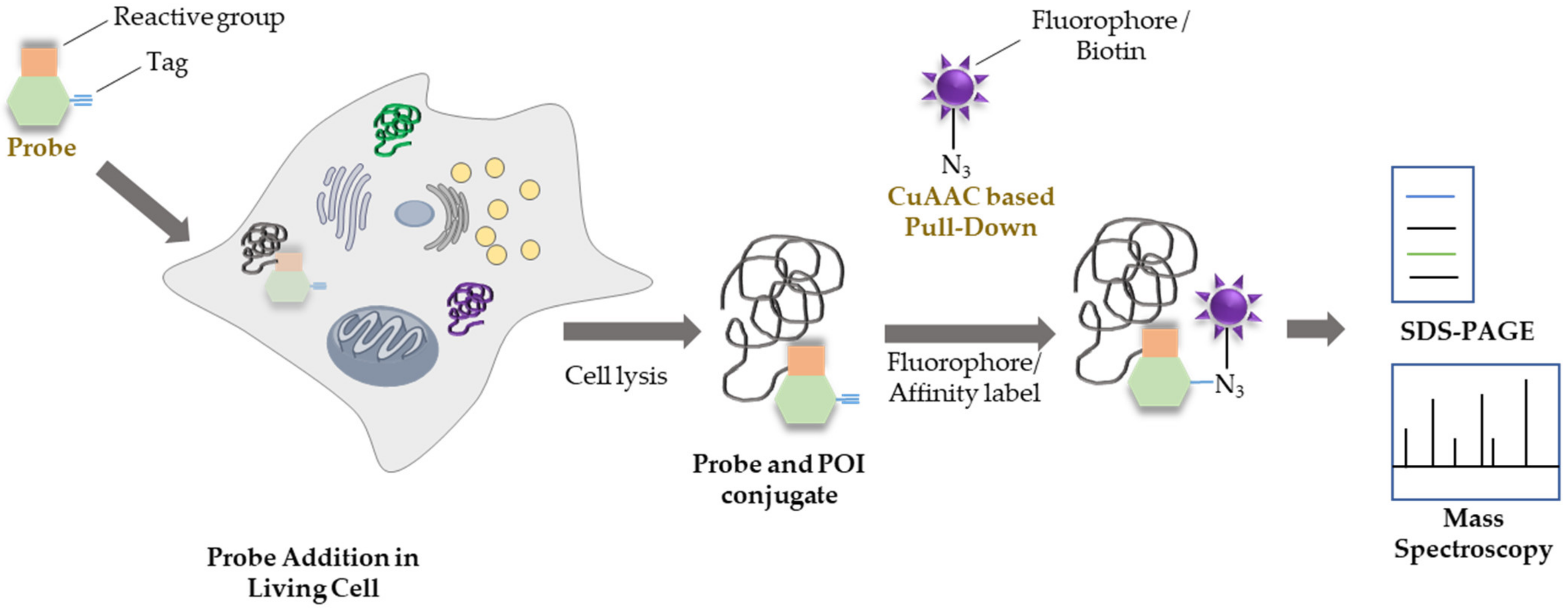
:1. Chemical Probes and Activity-Based Profiling
2. Transferase Activity Probe
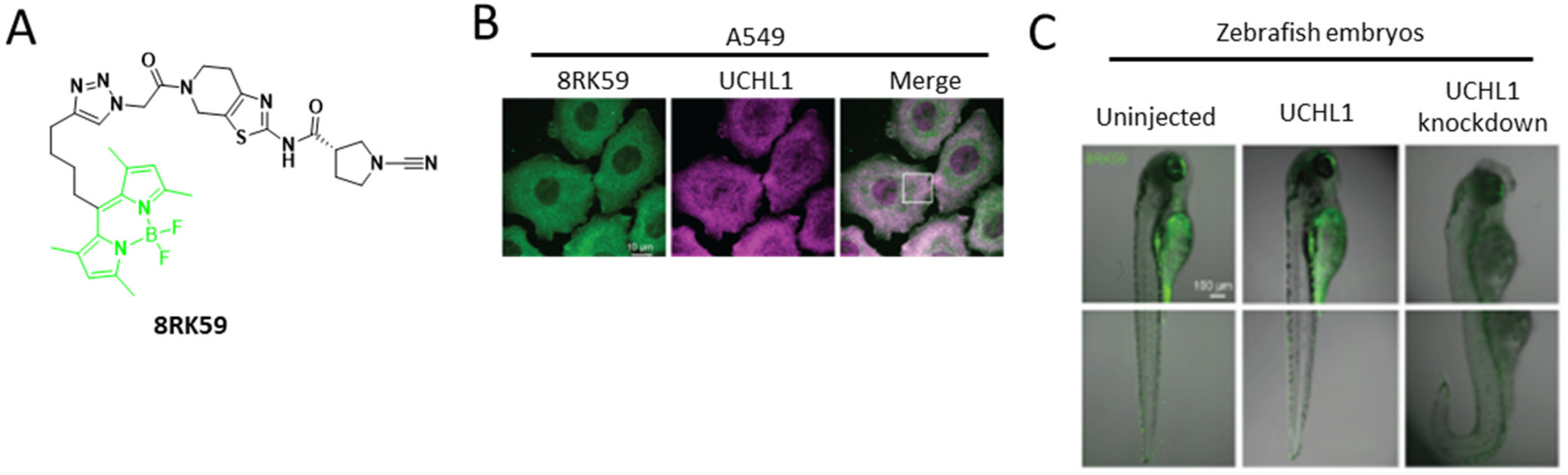
3. Hydrolase Activity Probe
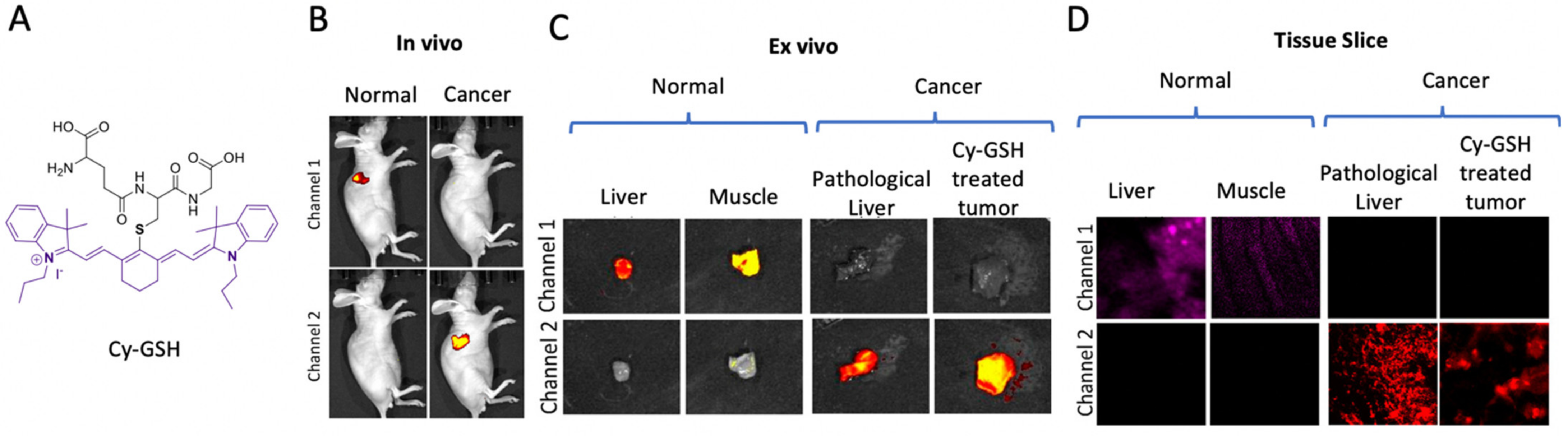
4. Oxidoreductase Probe
5. Epigenetics Regulator Activity Probe
6. Anticancer Activity of Probes
7. Immune Cell Activity Probe
8. Chemical Probe for Photodynamic Therapy
9. Current Challenges and Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Workman, P.; Collins, I. Probing the probes: Fitness factors for small molecule tools. Chem. Biol. 2010, 17, 561–577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagner, B.K. CHAPTER 1 Introduction to chemical probes. In The Discovery and Utility of Chemical Probes in Target Discovery; The Royal Society of Chemistry: London, UK, 2021; pp. 1–13. [Google Scholar]
- Gerry, C.J.; Schreiber, S.L. Chemical probes and drug leads from advances in synthetic planning and methodology. Nat. Rev. Drug Discov. 2018, 17, 333–352. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Fu, H.A.; Ivanov, A.A. Online informatics resources to facilitate cancer target and chemical probe discovery. RSC Med. Chem. 2020, 11, 611–624. [Google Scholar] [CrossRef] [PubMed]
- Nomura, D.K.; Dix, M.M.; Cravatt, B.F. Activity-based protein profiling for biochemical pathway discovery in cancer. Nat. Rev. Cancer 2010, 10, 630–638. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Tian, Y.; Wang, M.; Wang, M.; Sun, G.B.; Sun, X.B. Advanced activity-based protein profiling application strategies for drug development. Front. Pharmacol. 2018, 9, 353. [Google Scholar] [CrossRef]
- Benns, H.J.; Wincott, C.J.; Tate, E.W.; Child, M.A. Activity- and reactivity-based proteomics: Recent technological advances and applications in drug discovery. Curr. Opin. Chem. Biol. 2021, 60, 20–29. [Google Scholar] [CrossRef]
- Burton, N.R.; Kim, P.; Backus, K.M. Photoaffinity labelling strategies for mapping the small molecule-protein interactome. Org. Biomol. Chem. 2021, 19, 7792–7809. [Google Scholar] [CrossRef]
- Yang, Y.; Song, H.P.; He, D.; Zhang, S.; Dai, S.Z.; Lin, S.X.; Meng, R.; Wang, C.; Chen, P.R. Genetically encoded protein photocrosslinker with a transferable mass spectrometry-identifiable label. Nat. Commun. 2016, 7, 12299. [Google Scholar] [CrossRef]
- Meyer, J.P.; Adumeau, P.; Lewis, J.S.; Zeglis, B.M. Click chemistry and radiochemistry: The first 10 years. Bioconjugate Chem. 2016, 27, 2791–2807. [Google Scholar] [CrossRef] [Green Version]
- Li, J.N.; Fang, B.; Kinose, F.; Bai, Y.; Kim, J.Y.; Chen, Y.A.; Rix, U.; Koomen, J.M.; Haura, E.B. Target identification in small cell lung cancer via integrated phenotypic screening and activity based protein profiling. Mol. Cancer Ther. 2016, 15, 334–342. [Google Scholar] [CrossRef] [Green Version]
- Li, K.; Xu, S.; Xiong, M.Y.; Huan, S.Y.; Yuan, L.; Zhang, X.B. Molecular engineering of organic-based agents for in situ bioimaging and phototherapeutics. Chem. Soc. Rev. 2021, 50, 11766–11784. [Google Scholar] [CrossRef]
- Miller, M.A.; Kim, E.; Cuccarese, M.F.; Plotkin, A.L.; Prytyskach, M.; Kohler, R.H.; Pittet, M.J.; Weissleder, R. Near infrared imaging of Mer tyrosine kinase (MERTK) using MERi-SiR reveals tumor associated macrophage uptake in metastatic disease. Chem. Commun. 2018, 54, 42–45. [Google Scholar] [CrossRef]
- Soethoudt, M.; Stolze, S.C.; Westphal, M.V.; van Stralen, L.; Martella, A.; van Rooden, E.J.; Guba, W.; Varga, Z.V.; Deng, H.; van Kasteren, S.I.; et al. Selective photoaffinity probe that enables assessment of cannabinoid CB2 receptor expression and ligand engagement in human cells. J. Am. Chem. Soc. 2018, 140, 6067–6075. [Google Scholar] [CrossRef] [Green Version]
- Grant, E.K.; Fallon, D.J.; Eberl, H.C.; Fantom, K.G.M.; Zappacosta, F.; Messenger, C.; Tomkinson, N.C.O.; Bush, J.T. A Photoaffinity displacement assay and probes to study the cyclin-dependent kinase family. Angew. Chem. Int. Ed. 2019, 58, 17322–17327. [Google Scholar] [CrossRef]
- Deng, H.; Lei, Q.; Shang, W.; Li, Y.; Bi, L.; Yang, N.; Yu, Z.; Li, W. Potential applications of clickable probes in EGFR activity visualization and prediction of EGFR-TKI therapy response for NSCLC patients. Eur. J. Med. Chem. 2022, 230, 114100. [Google Scholar] [CrossRef]
- Liu, S.N.; Song, W.J.; Gao, X.Q.; Su, Y.X.; Gao, E.; Gao, Q.Z. Discovery of nonpeptide, reversible HER1/HER2 dual-targeting small-molecule inhibitors as near-infrared fluorescent probes for efficient tumor detection, diagnostic imaging, and drug screening. Anal. Chem. 2019, 91, 1507–1515. [Google Scholar] [CrossRef]
- Wang, X.; Ma, N.; Wu, R.; Ding, K.; Li, Z.Q. A novel reactive turn-on probe capable of selective profiling and no-wash imaging of Bruton’s tyrosine kinase in live cells. Chem. Commun. 2019, 55, 3473–3476. [Google Scholar] [CrossRef]
- Tian, Z.H.; Tian, X.G.; Feng, L.; Tian, Y.; Huo, X.K.; Zhang, B.J.; Deng, S.; Ma, X.C.; Cui, J.N. A highly sensitive and selective two-photon fluorescent probe for glutathione S-transferase detection and imaging in living cells and tissues. J. Mater. Chem. B 2019, 7, 4983–4989. [Google Scholar] [CrossRef]
- He, N.; Bai, S.; Huang, Y.; Xing, Y.L.; Chen, L.X.; Yu, F.B.; Lv, C.J. Evaluation of glutathione S-transferase inhibition effects on idiopathic pulmonary fibrosis therapy with a near-infrared fluorescent probe in cell and mice models. Anal. Chem. 2019, 91, 5424–5432. [Google Scholar] [CrossRef]
- Zhang, J.; Jin, Z.; Hu, X.X.; Meng, H.M.; Li, J.; Zhang, X.B.; Liu, H.W.; Deng, T.G.; Yao, S.; Feng, L.L. Efficient two-photon fluorescent probe for glutathione S-transferase detection and imaging in drug-induced liver injury sample. Anal. Chem. 2017, 89, 8097–8103. [Google Scholar] [CrossRef]
- Zhu, W.C.; Qian, X.L.; Wang, J.; Yu, H.; Liu, W.W.; Wang, H.Y.; Liu, Y. Near-infrared frequency upconversion probes for monitoring glutathione S-transferase to evaluate acute liver injury. Sensor. Actuat. B-Chem. 2021, 347, 130640. [Google Scholar] [CrossRef]
- Qin, Y.; Peng, C.Y.; Yang, W.; Fan, J.L.; Sheng, W.B.; Yi, P.; Qiu, Y.X.; Yu, H.H.; Jiang, S.; Wang, W.; et al. A bi-functional fluorescent probe for visualized and rapid natural drug screening via GSTs activity monitoring. Sensor. Actuat. B-Chem. 2021, 328, 129047. [Google Scholar] [CrossRef]
- Zhu, W.C.; Yu, H.; Qian, X.L.; Lu, K.; Zhao, C.; Zhang, Y.Y.; Wang, H.Y.; Liu, Y. Near-infrared frequency upconversion probe for revealing the relationship between glutathione S-transferase and drug-resistance. Anal. Chim. Acta 2021, 1181, 338920. [Google Scholar] [CrossRef]
- Navarrete, M.; Ho, J.; Dwivedi, R.C.; Choi, N.; Ezzati, P.; Spicer, V.; Arora, R.C.; Rigatto, C.; Wilkins, J.A. Activity-based protein profiling of intraoperative serine hydrolase activities during cardiac surgery. J. Proteome Res. 2018, 17, 3547–3556. [Google Scholar] [CrossRef]
- Kooij, R.; Liu, S.H.; Sapmaz, A.; Xin, B.T.; Janssen, G.M.C.; van Veelen, P.A.; Ovaa, H.; ten Dijke, P.; Geurink, P.P. Small-molecule activity-based probe for monitoring ubiquitin c-terminal hydrolase L1 (UCHL1) activity in live cells and zebrafish embryos. J. Am. Chem. Soc. 2020, 142, 16825–16841. [Google Scholar] [CrossRef]
- Panyain, N.; Godinat, A.; Lanyon-Hogg, T.; Lachiondo-Ortega, S.; Will, E.J.; Soudy, C.; Mondal, M.; Mason, K.; Elkhalifa, S.; Smith, L.M.; et al. Discovery of a potent and selective covalent inhibitor and activity-based probe for the deubiquitylating enzyme UCHL1, with antifibrotic activity (vol 142, pg 12020, 2020). J. Am. Chem. Soc. 2020, 142, 15199. [Google Scholar] [CrossRef]
- Chang, J.W.; Bhuiyan, M.; Tsai, H.M.; Zhang, H.N.J.; Li, G.; Fathi, S.; McCutcheon, D.C.; Leoni, L.; Freifelder, R.; Chen, C.T.; et al. In vivo imaging of the tumor-associated enzyme NCEH1 with a covalent PET probe. Angew. Chem. Int. Ed. 2020, 59, 15161–15165. [Google Scholar] [CrossRef]
- Fan, W.; Zhang, W.; Alshehri, S.; Garrison, J.C. Examination of the impact molecular charge has on NTSR1-targeted agents incorporated with cysteine protease inhibitors. Eur. J. Med. Chem. 2022, 234, 114241. [Google Scholar] [CrossRef]
- Blum, G.; von Degenfeld, G.; Merchant, M.J.; Blau, H.M.; Bogyo, M. Noninvasive optical imaging of cysteine protease activity using fluorescently quenched activity-based probes. Nat. Chem. Biol. 2007, 3, 668–677. [Google Scholar] [CrossRef]
- Nam, H.Y.; Song, D.; Eo, J.; Choi, N.E.; Hong, J.A.; Hong, K.T.; Lee, J.S.; Seo, J.; Lee, J. Activity-based probes for the high temperature requirement a serine proteases. ACS Chem. Biol. 2020, 15, 2346–2354. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Mendowicz, R.J.; Verhelst, S.H.L. Tagged benzoxazin-4-ones as novel activity-based probes for serine proteases. Chembiochem 2021, 22, 1578–1581. [Google Scholar] [CrossRef] [PubMed]
- Li, L.H.; Shi, W.; Wu, X.F.; Gong, Q.Y.; Li, X.H.; Ma, H.M. Monitoring gamma-glutamyl transpeptidase activity and evaluating its inhibitors by a water-soluble near-infrared fluorescent probe. Biosens. Bioelectron. 2016, 81, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Juan, O.Y.; Li, Y.F.; Jiang, W.L.; He, S.Y.; Liu, H.W.; Li, C.Y. Fluorescence-guided cancer diagnosis and surgery by a zero cross talk ratiometric near-infrared gamma-glutamyltranspeptidase fluorescent probe. Anal. Chem. 2019, 91, 1056–1063. [Google Scholar] [CrossRef]
- Xue, H.X.; Lu, J.Y.; Yan, H.W.; Huang, J.; Luo, H.B.; Wong, M.S.; Gao, Y.Q.; Zhang, X.L.; Guo, L. gamma-Glutamyl transpeptidase-activated indole-quinolinium based cyanine as a fluorescence turn-on nucleolus-targeting probe for cancer cell detection and inhibition. Talanta 2022, 237, 122898. [Google Scholar] [CrossRef]
- Huang, Z.; An, R.B.; Wei, S.X.; Wang, J.F.; Ye, D.J. Noninvasive ratiometric fluorescence imaging of gamma-glutamyltransferase activity using an activatable probe. Analyst 2021, 146, 1865–1871. [Google Scholar] [CrossRef]
- Li, Z.; Li, Y.; Liu, Y.; Zheng, Y.; Shen, J.; He, X.; Peng, B. One-step activated fluorescence bioimaging of γ-glutamyltransferase activity in living cancer cells based on chloro-rhodamine probe. Dyes Pigment. 2022, 199, 109962. [Google Scholar] [CrossRef]
- Li, H.D.; Li, Y.Q.; Yao, Q.C.; Fan, J.L.; Sun, W.; Long, S.; Shao, K.; Du, J.J.; Wang, J.Y.; Peng, X.J. In situ imaging of aminopeptidase N activity in hepatocellular carcinoma: A migration model for tumour using an activatable two-photon NIR fluorescent probe. Chem. Sci. 2019, 10, 1619–1625. [Google Scholar] [CrossRef] [Green Version]
- Gu, K.Z.; Liu, Y.J.; Guo, Z.Q.; Lian, C.; Yan, C.X.; Shi, P.; Tian, H.; Zhu, W.H. In situ ratiometric quantitative tracing of intracellular leucine aminopeptidase activity via an activatable near-infrared fluorescent. probe. ACS Appl. Mater. Interfaces 2016, 8, 26622–26629. [Google Scholar] [CrossRef]
- Chai, Y.; Gao, Y.T.; Xiong, H.W.; Lv, W.Q.; Yang, G.C.; Lu, C.F.; Nie, J.Q.; Ma, C.; Chen, Z.X.; Ren, J.; et al. A near-infrared fluorescent probe for monitoring leucine aminopeptidase in living cells. Analyst 2019, 144, 463–467. [Google Scholar] [CrossRef]
- Liu, X.J.; Zhang, Q.Y.; Wang, F.L.; Jiang, J.H. A near infrared fluorescent probe for the detection and imaging of prolyl aminopeptidase activity in living cells. Analyst 2019, 144, 5980–5985. [Google Scholar] [CrossRef]
- Pan, S.J.; Jang, S.Y.; Liew, S.S.; Fu, J.Q.; Wang, D.Y.; Lee, J.S.; Yao, S.Q. A vinyl sulfone-based fluorogenic probe capable of selective labeling of PHGDH in live mammalian cells. Angew. Chem. Int. Ed. 2018, 57, 579–583. [Google Scholar] [CrossRef]
- Somasundaram, V.; Basudhar, D.; Bharadwaj, G.; No, J.H.; Ridnour, L.A.; Cheng, R.Y.S.; Fujita, M.; Thomas, D.D.; Anderson, S.K.; McVicar, D.W.; et al. Molecular mechanisms of nitric oxide in cancer progression, signal transduction, and metabolism. Antioxid. Redox Signal. 2019, 30, 1124–1143. [Google Scholar] [CrossRef]
- Wang, C.; Song, X.B.; Han, Z.; Li, X.Y.; Xu, Y.P.; Xiao, Y. Monitoring nitric oxide in subcellular compartments by hybrid probe based on rhodamine spirolactam and SNAP-tag. ACS Chem. Biol. 2016, 11, 2033–2040. [Google Scholar] [CrossRef]
- Reczek, C.R.; Chandel, N.S. ROS promotes cancer cell survival through calcium signaling. Cancer Cell 2018, 33, 949–951. [Google Scholar] [CrossRef] [Green Version]
- Gao, M.; Li, Y.X.; Chen, X.H.; Li, S.W.; Ren, L.; Tang, B.Z. Aggregation-induced emission probe for light-up and in situ detection of calcium ions at high concentration. ACS Appl. Mater. Interfaces 2018, 10, 14410–14417. [Google Scholar] [CrossRef]
- Wang, Z.; Jia, J.; Huo, F.; Zhang, Y.; Chao, J. A fluorescent probe for monitoring Cys fluctuations in the oxidative stress environment simulated by Cu2+ or H2O2. Bioorg. Chem. 2022, 120, 105618. [Google Scholar] [CrossRef]
- Li, Z.; Ren, T.B.; Zhang, X.X.; Xu, S.; Gong, X.Y.; Yang, Y.; Ke, G.L.; Yuan, L.; Zhang, X.B. Precipitated fluorophore-based probe for accurate detection of mitochondrial analytes. Anal. Chem. 2021, 93, 2235–2243. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, Y.; He, H.S.; Wang, S.F.; Lei, Z.H.; Zhang, F. ROS/RNS and base dual activatable merocyanine-based NIR-II fluorescent molecular probe for in vivo biosensing. Angew. Chem. Int. Ed. 2021, 60, 26337–26341. [Google Scholar] [CrossRef]
- Ramesh, M.; Rajasekhar, K.; Gupta, K.; Babagond, V.; Saini, D.K.; Govindaraju, T. A matrix targeted fluorescent probe to monitor mitochondrial dynamics. Org. Biomol. Chem. 2021, 19, 801–808. [Google Scholar] [CrossRef]
- Li, Z.P.; Li, S.Q.; Lv, H.X.; Shen, J.L.; He, X.J.; Peng, B. BODIPY-based rapid response fluorescence probe for sensing and bioimaging endogenous superoxide anion in living cells. Spectrochim. Acta A 2022, 269, 120766. [Google Scholar] [CrossRef]
- Wang, L.F.; Liu, J.; Zhang, H.X.; Guo, W. Discrimination between cancerous and normal cells/tissues enabled by a near-infrared fluorescent HClO probe. Sens. Actuat. B-Chem. 2021, 334, 129602. [Google Scholar] [CrossRef]
- Zhang, K.J.; Liu, Z.Y.; Yao, Y.W.; Qiu, Y.T.; Li, F.; Chen, D.; Hamilton, D.J.; Li, Z.; Jiang, S. Structure-based design of a selective class I histone deacetylase (HDAC) near-infrared (NIR) probe for epigenetic regulation detection in triple-negative breast cancer (TNBC). J. Med. Chem. 2021, 64, 4020–4033. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Du, Y.; Liang, Q.; Cheng, Z.; Tian, J. Development of a novel histone deacetylase-targeted near-infrared probe for hepatocellular carcinoma imaging and fluorescence image-guided surgery. Mol. Imaging Biol. 2020, 22, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Du, W.; Zhang, T.; Liang, G. A Bioluminescent probe for simultaneously imaging esterase and histone deacetylase activity in a tumor. Anal. Chem. 2020, 92, 15275–15279. [Google Scholar] [CrossRef]
- Li, M.; Liu, J.; Chen, X.F.; Dang, Y.J.; Shao, Y.; Xu, Z.A.; Zhang, W. An activatable and tumor-targeting NIR fluorescent probe for imaging of histone deacetylase 6 in cancer cells and in vivo. Chem. Commun. 2022, 58, 1938–1941. [Google Scholar] [CrossRef]
- Ma, N.; Zhang, Z.M.; Lee, J.S.; Cheng, K.; Lin, L.G.; Zhang, D.M.; Hao, P.L.; Ding, K.; Ye, W.C.; Li, Z.Q. Affinity-based protein profiling reveals cellular targets of photoreactive anticancer inhibitors. ACS Chem. Biol. 2019, 14, 2546–2552. [Google Scholar] [CrossRef]
- Xu, J.Q.; Zhang, Z.; Lin, L.G.; Sun, H.Y.; White, L.V.; Ding, K.; Li, Z.Q. Quantitative proteomics reveals cellular off-targets of a DDR1 inhibitor. ACS Med. Chem. Lett. 2020, 11, 535–540. [Google Scholar] [CrossRef]
- Ding, X.R.; Li, Y.M.; Li, J.; Yin, Y.M. OSW-1 inhibits tumor growth and metastasis by NFATc2 on triple-negative breast cancer. Cancer Med.-US 2020, 9, 5558–5569. [Google Scholar] [CrossRef]
- Du, X.; Qian, C.; Jin, X. Effects of mitochondrial DNA deletion and OSW-1 on PI3K-AKT signaling pathway PCR array in SK-Hep1 cells. Chin. Med. 2019, 10, 13. [Google Scholar] [CrossRef] [Green Version]
- Yamada, R.; Hiraizumi, M.; Narita, S.; Sakurai, K. Two-step synthesis of a clickable photoaffinity probe from an anticancer saponin OSW-1 and its photochemical reactivity. Asian J. Org. Chem. 2016, 5, 330–334. [Google Scholar] [CrossRef]
- Lang, W.J.; Yuan, C.N.; Zhu, B.W.; Pan, S.J.; Liu, J.; Luo, J.; Nie, S.K.; Zhu, Q.; Lee, J.S.; Ge, J.Y. Expanding the "minimalist" small molecule tagging approach to different bioactive compounds. Org. Biomol. Chem. 2019, 17, 3010–3017. [Google Scholar] [CrossRef]
- Zhang, W.D.; Zhou, Y.B.; Li, D.L.; Ma, T. Near-infrared fluorescent probe with large stokes shift for detecting Human Neutrophil elastase in living cells. Spectrochim. Acta A 2021, 252, 119533. [Google Scholar] [CrossRef]
- Cao, T.; Teng, Z.D.; Zheng, L.; Qian, J.; Ma, H.; Wang, J.M.; Qin, W.W.; Guo, H.C. Activity-based ratiometric fluorescent small-molecule probe for endogenously monitoring neutrophil elastase in living cells. Anal. Chim. Acta 2020, 1127, 295–302. [Google Scholar] [CrossRef]
- Gao, M.; Lee, S.H.; Park, S.H.; Ciaramicoli, L.M.; Kwon, H.Y.; Cho, H.; Jeong, J.; Chang, Y.T. Neutrophil-selective fluorescent probe development through metabolism-oriented live-cell distinction. Angew. Chem. Int. Ed. 2021, 60, 23743–23749. [Google Scholar] [CrossRef]
- Kim, T.I.; Hwang, B.; Lee, B.; Bae, J.; Kim, Y. Selective monitoring and imaging of eosinophil peroxidase activity with a J-Aggregating probe. J. Am. Chem. Soc. 2018, 140, 11771–11776. [Google Scholar] [CrossRef]
- Yin, L.; Sun, H.; Zhao, M.; Wang, A.N.; Qiu, S.S.; Gao, Y.J.; Ding, J.N.; Ji, S.J.; Shi, H.B.; Gao, M.Y. Rational design and synthesis of a metalloproteinase-activatable probe for dual-modality imaging of metastatic lymph nodes in vivo. J. Org. Chem. 2019, 84, 6126–6133. [Google Scholar] [CrossRef]
- Zhu, D.S.; Guo, H.J.; Chang, Y.; Ni, Y.; Li, L.; Zhang, Z.M.; Hao, P.L.; Xu, Y.; Ding, K.; Li, Z.Q. Cell- and tissue-based proteome profiling and dual imaging of apoptosis markers with probes derived from venetoclax and idasanutlin. Angew. Chem. Int. Ed. 2018, 57, 9284–9289. [Google Scholar] [CrossRef]
- Zhou, Z.Y.; Zalutsky, M.R.; Chitneni, S.K. Fluorine-18 labeling of the MDM2 inhibitor RG7388 for PET imaging: Chemistry and preliminary evaluation. Mol. Pharmaceut. 2021, 18, 3871–3881. [Google Scholar] [CrossRef]
- Linden, G.; Zhang, L.; Pieck, F.; Linne, U.; Kosenkov, D.; Tonner, R.; Vazquez, O. Conditional singlet oxygen generation through a bioorthogonal DNA-targeted tetrazine reaction. Angew. Chem. Int. Ed. 2019, 58, 12868–12873. [Google Scholar] [CrossRef]
- Clavadetscher, J.; Hoffmann, S.; Lilienkampf, A.; Mackay, L.; Yusop, R.M.; Rider, S.A.; Mullins, J.J.; Bradley, M. Copper catalysis in living systems and in situ drug synthesis. Angew. Chem. Int. Ed. 2016, 55, 15662–15666. [Google Scholar] [CrossRef]
- Yao, Q.X.; Lin, F.; Fan, X.Y.; Wang, Y.P.; Liu, Y.; Liu, Z.F.; Jiang, X.Y.; Chen, P.R.; Gao, Y. Synergistic enzymatic and bioorthogonal reactions for selective prodrug activation in living systems. Nat. Commun. 2018, 9, 5032. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.Y.; Chang, J.J.; Shi, M.W.; Pan, W.; Li, N.; Tang, B. A dual-targeted organic photothermal agent for enhanced photothermal therapy. Angew. Chem. Int. Ed. 2019, 58, 1057–1061. [Google Scholar] [CrossRef]
- Rubtsova, N.I.; Hart, M.C.; Arroyo, A.D.; Osharovich, S.A.; Liebov, B.K.; Miller, J.; Yuan, M.; Cochran, J.M.; Chong, S.; Yodh, A.G.; et al. NIR fluorescent imaging and photodynamic therapy with a novel theranostic phospholipid probe for triple-negative breast cancer cells. Bioconjugate Chem. 2021, 32, 1852–1863. [Google Scholar] [CrossRef]
- Shi, M.W.; Fu, Z.L.; Pan, W.; Chen, Y.Y.; Wang, K.Y.; Zhou, P.; Li, N.; Tang, B. A protein-binding molecular photothermal agent for tumor ablation. Angew. Chem. Int. Ed. 2021, 60, 13564–13568. [Google Scholar] [CrossRef]
- Jiang, G.Y.; Li, C.B.; Liu, X.; Chen, Q.Q.; Li, X.K.; Gu, X.G.; Zhang, P.F.; Lai, Q.F.; Wang, J.G. Lipid droplet-targetable fluorescence guided photodynamic therapy of cancer cells with an activatable AIE-active fluorescent probe for hydrogen peroxide. Adv. Opt. Mater. 2020, 8, 2001119. [Google Scholar] [CrossRef]
- Siriwibool, S.; Kaekratoke, N.; Chansaenpak, K.; Siwawannapong, K.; Panajapo, P.; Sagarik, K.; Noisa, P.; Lai, R.Y.; Kamkaew, A. Near-infrared fluorescent pH responsive probe for targeted photodynamic cancer therapy. Sci. Rep. 2020, 10, 1283. [Google Scholar] [CrossRef] [Green Version]
- Yuan, J.; Ren, T.B.; Xu, S.; Wang, C.J.; Zhang, X.B.; Yuan, L. A unique multifunctional luminescent probe for self-monitoring photodynamic therapy by detecting H2S in cancer cells. ACS Appl. Bio Mater. 2021, 4, 6016–6022. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.W.; Zhang, J.W.; Shabat, D.R.; Fan, J.L.; Peng, X.J. Near-infrared chemiluminescent probe for real-time monitoring singlet oxygen in cells and mice model. ACS Sens. 2020, 5, 3158–3164. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.; Lee, J.S.; Hao, P.L.; Yao, S.Q.; Ding, K.; Li, Z.Q. Tetrazole-based probes for integrated phenotypic screening, affinity-based proteome profiling, and sensitive detection of a cancer biomarker. Angew. Chem. Int. Ed. 2017, 56, 15044–15048. [Google Scholar] [CrossRef] [PubMed]
- Vu, V.; Szewczyk, M.M.; Nie, D.Y.; Arrowsmith, C.H.; Barsyte-Lovejoy, D. Validating small molecule chemical probes for biological discovery. Annu. Rev. Biochem. 2022, 91. [Google Scholar] [CrossRef] [PubMed]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al Mazid, M.F.; Park, S.B.; Cheekatla, S.R.; Murale, D.P.; Shin, K.H.; Lee, J.-S. Chemical Probes and Activity-Based Protein Profiling for Cancer Research. Int. J. Mol. Sci. 2022, 23, 5936. https://doi.org/10.3390/ijms23115936
Al Mazid MF, Park SB, Cheekatla SR, Murale DP, Shin KH, Lee J-S. Chemical Probes and Activity-Based Protein Profiling for Cancer Research. International Journal of Molecular Sciences. 2022; 23(11):5936. https://doi.org/10.3390/ijms23115936
Chicago/Turabian StyleAl Mazid, Mohammad Faysal, Seung Bin Park, Subba Rao Cheekatla, Dhiraj P. Murale, Kyung Ho Shin, and Jun-Seok Lee. 2022. "Chemical Probes and Activity-Based Protein Profiling for Cancer Research" International Journal of Molecular Sciences 23, no. 11: 5936. https://doi.org/10.3390/ijms23115936
APA StyleAl Mazid, M. F., Park, S. B., Cheekatla, S. R., Murale, D. P., Shin, K. H., & Lee, J.-S. (2022). Chemical Probes and Activity-Based Protein Profiling for Cancer Research. International Journal of Molecular Sciences, 23(11), 5936. https://doi.org/10.3390/ijms23115936








