Therapeutic Effect and Mechanisms of Essential Oils in Mood Disorders: Interaction between the Nervous and Respiratory Systems
Abstract
1. Introduction
2. Therapeutic Effects of EOs
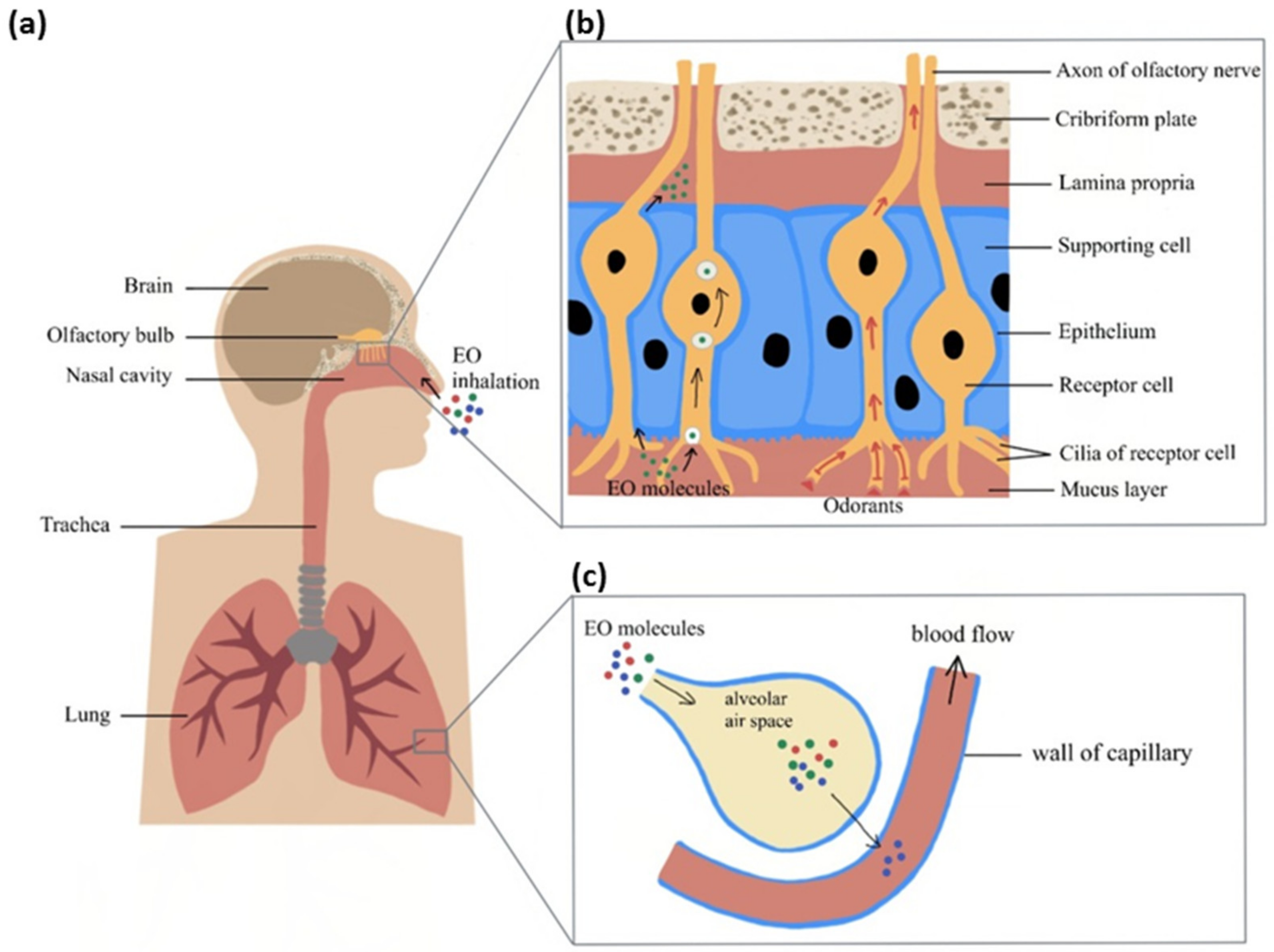
3. Absorption of EO Molecules through Inhalation (Mechanisms behind the Effect of EOs on Brain)
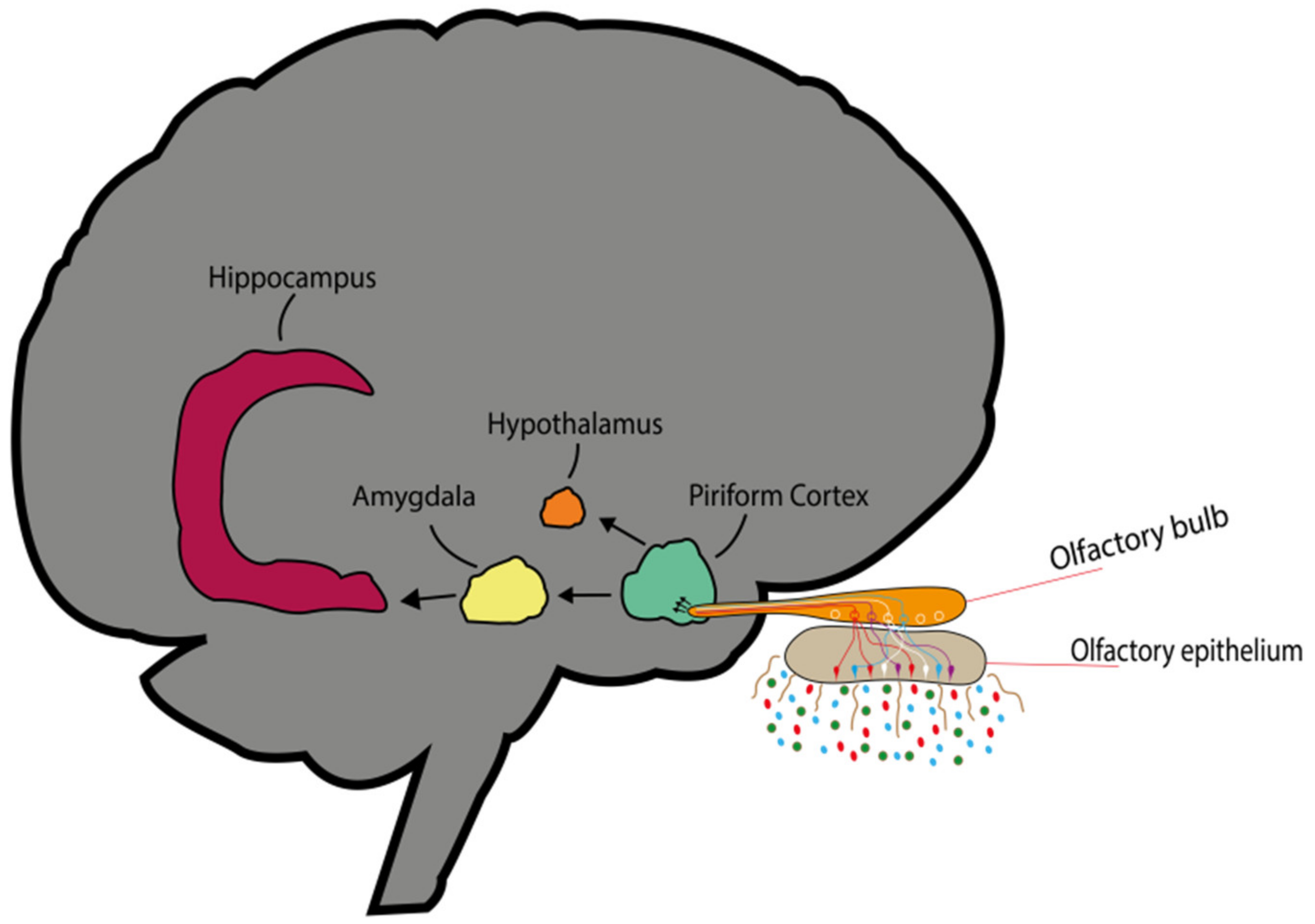
3.1. Transmission of Olfactory Odorant Signal (Through Activation of Nasal Olfactory Chemoreceptors)
3.2. Chemical Transport of Molecules (Direct Penetration of EO Molecules via Neuronal Pathway)
3.3. Respiratory System and Central Nervous System (Alveolar Absorption of EO Molecules into the Blood Circulation with Subsequent Effect on Brain)
4. Effect of EO on Cellular/Molecular Events
4.1. Regulation of Monoamines
4.2. Neurogenesis and Neurotrophic Factors
4.3. Regulation of the Neuroendocrine System
4.4. Other Possible Mechanisms: Oxidative Stress and Inflammation
5. Future Studies
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 5-HT | serotonin |
| ACTH | adrenocorticotropic hormone |
| BBB | blood–brain barrier |
| BDNF | brain-derived neurotrophic factor |
| BEO | bergamot EO |
| CC-EO | cinnamomum cassia presl |
| CNS | central nervous system |
| CREB | cAMP-response element binding |
| CRH | corticotrophin-releasing hormone |
| DA | dopamine |
| EO | essential oil |
| EPM | elevated plus maze test |
| FRSA | free radical scavenging activity |
| FST | forced swimming test |
| GABA | γ-aminobutyric acid |
| GSH | glutathione |
| HPA axis | hypothalamic–pituitary–adrenal axis |
| LPS | lipopolysaccharide |
| MAPK | mitogen-activated protein kinase |
| MDA | malondialdehyde |
| MJ | methyl jasmonate |
| NAc | nucleus accumbens |
| OFT | open field test |
| OSN | olfactory sensory neuron |
| ROS | reactive oxygen species |
| TNF-α | tumour necrosis factor-alpha |
| TRP | transient receptor potential channels |
| VTA | ventral tegmental area |
| YYO | ylang-ylang EO |
References
- Jaradat, N.A.; Al Zabadi, H.; Rahhal, B.; Hussein, A.M.; Mahmoud, J.S.; Mansour, B.; Khasati, A.I.; Issa, A. The effect of inhalation of Citrus sinensis flowers and Mentha spicata leave essential oils on lung function and exercise performance: A quasi-experimental uncontrolled before-and-after study. J. Int. Soc. Sports Nutr. 2016, 13, 36. [Google Scholar] [CrossRef] [PubMed]
- Lakhan, S.E.; Sheafer, H.; Tepper, D. The Effectiveness of Aromatherapy in Reducing Pain: A Systematic Review and Meta-Analysis. Pain Res. Treat. 2016, 2016, 8158693. [Google Scholar] [CrossRef]
- Cooke, B.; Ernst, E. Aromatherapy: A systematic review. Br. J. Gen. Pract. 2000, 50, 493–496. [Google Scholar]
- Hay, I.C.; Jamieson, M.; Ormerod, A.D. Randomized trial of aromatherapy. Successful treatment for alopecia areata. Arch. Dermatol. 1998, 134, 1349–11352. [Google Scholar] [CrossRef] [PubMed]
- De Groot, A.C.; Schmidt, E. Essential Oils, Part III: Chemical Composition. Dermatitis 2016, 27, 161–169. [Google Scholar]
- Koyama, S.; Heinbockel, T. The Effects of Essential Oils and Terpenes in Relation to Their Routes of Intake and Application. Int. J. Mol. Sci. 2020, 21, 1558. [Google Scholar] [CrossRef] [PubMed]
- Petrovic, J.; Stojkovic, D.; Sokovic, M. Terpene core in selected aromatic and edible plants: Natural health improving agents. Adv. Food Nutr. Res. 2019, 90, 423–451. [Google Scholar] [PubMed]
- Burnett, K.M.; Solterbeck, L.A.; Strapp, C.M. Scent and mood state following an anxiety-provoking task. Psychol. Rep. 2004, 95, 707–722. [Google Scholar] [CrossRef]
- Tsang, H.W.; Cheung, W.M.; Chan, A.H.; Fung, K.M.; Leung, A.Y.; Au, D.W. A pilot evaluation on a stress management programme using a combined approach of cognitive behavioural therapy (CBT) and complementary and alternative medicine (CAM) for elementary school teachers. Stress Health 2015, 31, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Fung, J.K.K.; Tsang, H.W. Management of behavioural and psychological symptoms of dementia by an aroma-massage with acupressure treatment protocol: A randomised clinical trial. J. Clin. Nurs. 2018, 27, 1812–1825. [Google Scholar] [CrossRef] [PubMed]
- Penninx, B.W.; Beekman, A.T.; Honig, A.; Deeg, D.J.; Schoevers, R.A.; van Eijk, J.Z.; van Tilburg, W. Depression and cardiac mortality: Results from a community-based longitudinal study. Arch. Gen. Psychiatry 2001, 58, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Karan, N.B. Influence of lavender oil inhalation on vital signs and anxiety: A randomized clinical trial. Physiol. Behav. 2019, 211, 112676. [Google Scholar] [CrossRef] [PubMed]
- Fung, J.K.; Tsang, H.W.; Chung, R.C. A systematic review of the use of aromatherapy in treatment of behavioral problems in dementia. Geriatr. Gerontol. Int. 2012, 12, 372–382. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.L.; Wu, Y.; Tsang, H.W.; Leung, A.Y.; Cheung, W.M. A systematic review on the anxiolytic effects of aromatherapy in people with anxiety symptoms. J. Altern. Complement. Med. 2011, 17, 101–108. [Google Scholar] [CrossRef]
- Sanchez-Vidana, D.I.; Ngai, S.P.; He, W.; Chow, J.K.; Lau, B.W.; Tsang, H.W. The Effectiveness of Aromatherapy for Depressive Symptoms: A Systematic Review. Evid. Based Complement Alternat. Med. 2017, 2017, 5869315. [Google Scholar] [CrossRef]
- Yim, V.W.; Ng, A.K.; Tsang, H.W.; Leung, A.Y. A review on the effects of aromatherapy for patients with depressive symptoms. J. Altern. Complement. Med. 2009, 15, 187–195. [Google Scholar] [CrossRef]
- Fayazi, S.; Babashahi, M.; Rezaei, M. The effect of inhalation aromatherapy on anxiety level of the patients in preoperative period. Iran. J. Nurs. Midwifery Res. 2011, 16, 278–283. [Google Scholar] [PubMed]
- Lehrner, J.; Marwinski, G.; Lehr, S.; Johren, P.; Deecke, L. Ambient odors of orange and lavender reduce anxiety and improve mood in a dental office. Physiol. Behav. 2005, 86, 92–95. [Google Scholar] [CrossRef] [PubMed]
- Senturk, A.; Tekinsoy Kartin, P. The Effect of Lavender Oil Application via Inhalation Pathway on Hemodialysis Patients’ Anxiety Level and Sleep Quality. Holist Nurs. Pract. 2018, 32, 324–335. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, H.; Mardani, A.; Basirinezhad, M.H.; Hamidzadeh, A.; Eskandari, F. The effects of Lavender and Chamomile essential oil inhalation aromatherapy on depression, anxiety and stress in older community-dwelling people: A randomized controlled trial. Explore (N. Y.) 2021. [Google Scholar] [CrossRef]
- Goes, T.C.; Antunes, F.D.; Alves, P.B.; Teixeira-Silva, F. Effect of sweet orange aroma on experimental anxiety in humans. J. Altern. Complement. Med. 2012, 18, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Asakura, H.; Hayashi, T. Effects of olfactory stimulation from the fragrance of the Japanese citrus fruit yuzu (Citrus junos Sieb. ex Tanaka) on mood states and salivary chromogranin A as an endocrinologic stress marker. J. Altern. Complement. Med. 2014, 20, 500–506. [Google Scholar] [CrossRef]
- Watanabe, E.; Kuchta, K.; Kimura, M.; Rauwald, H.W.; Kamei, T.; Imanishi, J. Effects of bergamot (Citrus bergamia (Risso) Wright & Arn.) essential oil aromatherapy on mood states, parasympathetic nervous system activity, and salivary cortisol levels in 41 healthy females. Forsch. Komplementmed. 2015, 22, 43–49. [Google Scholar] [PubMed]
- McKay, D.L.; Blumberg, J.B. A review of the bioactivity and potential health benefits of chamomile tea (Matricaria recutita L.). Phytother. Res. 2006, 20, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Moss, L.; Rouse, M.; Wesnes, K.A.; Moss, M. Differential effects of the aromas of Salvia species on memory and mood. Hum. Psychopharmacol. 2010, 25, 388–396. [Google Scholar] [CrossRef]
- Conrad, P.; Adams, C. The effects of clinical aromatherapy for anxiety and depression in the high risk postpartum woman–A pilot study. Complement. Ther. Clin. Pract. 2012, 18, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Song, E.J.; Lee, M.Y. Effects of Aromatherapy on Stress Responses, Autonomic Nervous System Activity and Blood Pressure in the Patients Undergoing Coronary Angiography: A Non-Randomized Controlled Trial. J. Korean Acad. Nurs. 2018, 48, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Belovicova, K.; Bogi, E.; Csatlosova, K.; Dubovicky, M. Animal tests for anxiety-like and depression-like behavior in rats. Interdiscip. Toxicol. 2017, 10, 40–43. [Google Scholar] [CrossRef]
- Slattery, D.A.; Cryan, J.F. Using the rat forced swim test to assess antidepressant-like activity in rodents. Nat. Protoc. 2012, 7, 1009–1014. [Google Scholar] [CrossRef]
- Chioca, L.R.; Antunes, V.D.; Ferro, M.M.; Losso, E.M.; Andreatini, R. Anosmia does not impair the anxiolytic-like effect of lavender essential oil inhalation in mice. Life Sci. 2013, 92, 971–975. [Google Scholar] [CrossRef] [PubMed]
- Chioca, L.R.; Ferro, M.M.; Baretta, I.P.; Oliveira, S.M.; Silva, C.R.; Ferreira, J.; Losso, E.M.; Andreatini, R. Anxiolytic-like effect of lavender essential oil inhalation in mice: Participation of serotonergic but not GABAA/benzodiazepine neurotransmission. J. Ethnopharmacol. 2013, 147, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Komiya, M.; Takeuchi, T.; Harada, E. Lemon oil vapor causes an anti-stress effect via modulating the 5-HT and DA activities in mice. Behav. Brain Res. 2006, 172, 240–249. [Google Scholar] [CrossRef]
- Zhang, N.; Zhang, L.; Feng, L.; Yao, L. Cananga odorata essential oil reverses the anxiety induced by 1-(3-chlorophenyl) piperazine through regulating the MAPK pathway and serotonin system in mice. J. Ethnopharmacol. 2018, 219, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.W.; Li, R.P.; Li, M.; Wang, S.Y.; Zhang, X.; Niu, X.X.; Li, W.; Yan, L.; Wang, Y.; Fu, Q.; et al. Antidepressant-like effect of essential oil of Perilla frutescens in a chronic, unpredictable, mild stress-induced depression model mice. Chin. J. Nat. Med. 2014, 12, 753–759. [Google Scholar] [CrossRef]
- Sanchez-Vidana, D.I.; Po, K.K.; Fung, T.K.; Chow, J.K.; Lau, W.K.; So, P.K.; Lau, B.W.; Tsang, H.W. Lavender essential oil ameliorates depression-like behavior and increases neurogenesis and dendritic complexity in rats. Neurosci. Lett. 2019, 701, 180–192. [Google Scholar] [CrossRef]
- Rojas, L.B.; Visbal, T.; Morillo, M.; de Rojas, Y.C.; Arzola, J.C.; Usubillaga, A. The volatile constituents of Salvia leucantha. Nat. Prod. Commun. 2010, 5, 937–938. [Google Scholar] [CrossRef] [PubMed]
- Tsang, H.W.; Lo, S.C.; Chan, C.C.; Ho, T.Y.; Fung, K.M.; Chan, A.H.; Au, D.W. Neurophysiological and behavioural effects of lavender oil in rats with experimentally induced anxiety. Flavour Fragr. J. 2013, 28, 168–173. [Google Scholar] [CrossRef]
- Lizarraga-Valderrama, L.R. Effects of essential oils on central nervous system: Focus on mental health. Phytother. Res. 2021, 35, 657–679. [Google Scholar] [CrossRef]
- Zhang, N.; Zhang, L.; Feng, L.; Yao, L. The anxiolytic effect of essential oil of Cananga odorata exposure on mice and determination of its major active constituents. Phytomedicine 2016, 23, 1727–1734. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.J.; Heinbockel, T. Essential Oils and Their Constituents Targeting the GABAergic System and Sodium Channels as Treatment of Neurological Diseases. Molecules 2018, 23, 1061. [Google Scholar] [CrossRef] [PubMed]
- Coelho, L.S.; Correa-Netto, N.F.; Masukawa, M.Y.; Lima, A.C.; Maluf, S.; Linardi, A.; Santos-Junior, J.G. Inhaled Lavandula angustifolia essential oil inhibits consolidation of contextual- but not tone-fear conditioning in rats. J. Ethnopharmacol. 2018, 215, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Hocayen, P.A.S.; Wendler, E.; Vecchia, D.D.; Kanazawa, L.K.S.; Issy, A.C.; Del Bel, E.; Andreatini, R. The nitrergic neurotransmission contributes to the anxiolytic-like effect of Citrus sinensis essential oil in animal models. Phytother. Res. 2019, 33, 901–909. [Google Scholar] [CrossRef] [PubMed]
- Koo, B.S.; Park, K.S.; Ha, J.H.; Park, J.H.; Lim, J.C.; Lee, D.U. Inhibitory effects of the fragrance inhalation of essential oil from Acorus gramineus on central nervous system. Biol. Pharm. Bull. 2003, 26, 978–982. [Google Scholar] [CrossRef]
- Cioanca, O.; Hritcu, L.; Mihasan, M.; Trifan, A.; Hancianu, M. Inhalation of coriander volatile oil increased anxiolytic-antidepressant-like behaviors and decreased oxidative status in beta-amyloid (1-42) rat model of Alzheimer’s disease. Physiol. Behav. 2014, 131, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Lim, E.J.; Zhao, R.J.; Oh, S.R.; Jung, J.W.; Ahn, E.M.; Lee, E.S.; Koo, J.S.; Kim, H.Y.; Chang, S.; et al. Effect of the fragrance inhalation of essential oil from Asarum heterotropoides on depression-like behaviors in mice. BMC Complement. Altern. Med. 2015, 15, 43. [Google Scholar] [CrossRef] [PubMed]
- De Almeida, R.N.; Motta, S.C.; de Brito Faturi, C.; Catallani, B.; Leite, J.R. Anxiolytic-like effects of rose oil inhalation on the elevated plus-maze test in rats. Pharmacol. Biochem. Behav. 2004, 77, 361–364. [Google Scholar] [CrossRef]
- Villareal, M.O.; Ikeya, A.; Sasaki, K.; Arfa, A.B.; Neffati, M.; Isoda, H. Anti-stress and neuronal cell differentiation induction effects of Rosmarinus officinalis L. essential oil. BMC Complement. Altern. Med. 2017, 17, 549. [Google Scholar] [CrossRef]
- Kotlik, B.A.; Petrova, L.M.; Red’ko, A.I.; Kharitonov, E. Sources of equipment measurement error in stimulation electromyography. Fiziol. Cheloveka 1986, 12, 691–694. [Google Scholar]
- Zimmerman, W.B. Sleep mentation and auditory awakening thresholds. Psychophysiology 1970, 6, 540–549. [Google Scholar] [CrossRef]
- Llorens, J. The physiology of taste and smell: How and why we sense flavors. Water Sci. Technol. 2004, 49, 1–10. [Google Scholar] [CrossRef]
- Lee, A.J.; Ling, R.S. Improved Cementing Techniques. Instr. Course Lect. 1981, 30, 407–413. [Google Scholar]
- Hintiryan, H.; Gou, L.; Zingg, B.; Yamashita, S.; Lyden, H.M.; Song, M.Y.; Grewal, A.K.; Zhang, X.; Toga, A.W.; Dong, H.W. Comprehensive connectivity of the mouse main olfactory bulb: Analysis and online digital atlas. Front Neuroanat. 2012, 6, 30. [Google Scholar] [CrossRef]
- Saive, A.L.; Royet, J.P.; Plailly, J. A review on the neural bases of episodic odor memory: From laboratory-based to autobiographical approaches. Front Behav. Neurosci. 2014, 8, 240. [Google Scholar] [CrossRef] [PubMed]
- Komori, T.; Fujiwara, R.; Tanida, M.; Nomura, J. Potential antidepressant effects of lemon odor in rats. Eur. Neuropsychopharmacol. 1995, 5, 477–480. [Google Scholar] [CrossRef]
- Fujiwara, R.; Komori, T.; Noda, Y.; Kuraoka, T.; Shibata, H.; Shizuya, K.; Miyahara, S.; Ohmori, M.; Nomura, J.; Yokoyama, M.M. Effects of a long-term inhalation of fragrances on the stress-induced immunosuppression in mice. Neuroimmunomodulation 1998, 5, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Aqrabawi, A.J.; Kim, J.C. Hippocampal projections to the anterior olfactory nucleus differentially convey spatiotemporal information during episodic odour memory. Nat. Commun. 2018, 9, 2735. [Google Scholar] [CrossRef]
- Stephens, M.A.; Wand, G. Stress and the HPA axis: Role of glucocorticoids in alcohol dependence. Alcohol. Res. 2012, 34, 468–483. [Google Scholar] [PubMed]
- Gordan, R.; Gwathmey, J.K.; Xie, L.H. Autonomic and endocrine control of cardiovascular function. World J. Cardiol. 2015, 7, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.A.; Leon, M. Chemotopic odorant coding in a mammalian olfactory system. J. Comp. Neurol. 2007, 503, 1–34. [Google Scholar] [CrossRef]
- Su, C.Y.; Menuz, K.; Carlson, J.R. Olfactory perception: Receptors, cells, and circuits. Cell 2009, 139, 45–59. [Google Scholar] [CrossRef] [PubMed]
- Kobayakawa, K.; Kobayakawa, R.; Matsumoto, H.; Oka, Y.; Imai, T.; Ikawa, M.; Okabe, M.; Ikeda, T.; Itohara, S.; Kikusui, T.; et al. Innate versus learned odour processing in the mouse olfactory bulb. Nature 2007, 450, 503–508. [Google Scholar] [CrossRef]
- Illum, L. Is nose-to-brain transport of drugs in man a reality? J. Pharm. Pharmacol. 2004, 56, 3–17. [Google Scholar] [CrossRef]
- Selvaraj, K.; Gowthamarajan, K.; Karri, V. Nose to brain transport pathways an overview: Potential of nanostructured lipid carriers in nose to brain targeting. Artif. Cells Nanomed. Biotechnol. 2018, 46, 2088–2095. [Google Scholar] [CrossRef]
- Kamei, N. Nose-to-Brain Delivery of Peptide Drugs Enhanced by Coadministration of Cell-penetrating Peptides: Therapeutic Potential for Dementia. Yakugaku Zasshi 2017, 137, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Lochhead, J.J.; Thorne, R.G. Intranasal delivery of biologics to the central nervous system. Adv. Drug Deliv. Rev. 2012, 64, 614–628. [Google Scholar] [CrossRef] [PubMed]
- Crowe, T.P.; Greenlee, M.H.W.; Kanthasamy, A.G.; Hsu, H.W. Mechanism of intranasal drug delivery directly to the brain. Life Sci. 2018, 195, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Ohmori, A.; Shinomiya, K.; Utsu, Y.; Tokunaga, S.; Hasegawa, Y.; Kamei, C. Effect of santalol on the sleep-wake cycle in sleep-disturbed rats. Nihon Shinkei Seishin Yakurigaku Zasshi 2007, 27, 167–171. [Google Scholar] [PubMed]
- Hwang, J.H. The effects of the inhalation method using essential oils on blood pressure and stress responses of clients with essential hypertension. Taehan Kanho Hakhoe Chi. 2006, 36, 1123–1134. [Google Scholar] [CrossRef]
- Maddocks-Jennings, W.; Wilkinson, J.M. Aromatherapy practice in nursing: Literature review. J. Adv. Nurs. 2004, 48, 93–103. [Google Scholar] [CrossRef]
- Woronuk, G.; Demissie, Z.; Rheault, M.; Mahmoud, S. Biosynthesis and therapeutic properties of Lavandula essential oil constituents. Planta. Med. 2011, 77, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Faturi, C.B.; Leite, J.R.; Alves, P.B.; Canton, A.C.; Teixeira-Silva, F. Anxiolytic-like effect of sweet orange aroma in Wistar rats. Prog. Neuropsychopharmacol. Biol. Psychiatry 2010, 34, 605–609. [Google Scholar] [CrossRef]
- Homma, I.; Masaoka, Y. Breathing rhythms and emotions. Exp. Physiol. 2008, 93, 1011–1021. [Google Scholar] [CrossRef]
- Masaoka, Y.; Izumizaki, M.; Homma, I. Where is the rhythm generator for emotional breathing? Prog. Brain Res. 2014, 209, 367–377. [Google Scholar]
- Mrozek, S.; Constantin, J.M.; Geeraerts, T. Brain-lung crosstalk: Implications for neurocritical care patients. World J. Crit. Care Med. 2015, 4, 163–178. [Google Scholar] [CrossRef]
- Rodriguez-Gonzalez, R.; Ramos-Nuez, A.; Martin-Barrasa, J.L.; Lopez-Aguilar, J.; Baluja, A.; Alvarez, J.; Rocco, P.R.; Pelosi, P.; Villar, J. Endotoxin-induced lung alveolar cell injury causes brain cell damage. Exp. Biol. Med. (Maywood) 2015, 240, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Gibb, T.D.; Sidles, J.A.; Harryman, D.T.; McQuade, K.J.; Matsen, F.A. The effect of capsular venting on glenohumeral laxity. Clin. Orthop. Relat. Res. 1991, 120–127. [Google Scholar] [PubMed]
- Ho, R.T.; Chan, J.S.; Wang, C.W.; Lau, B.W.; So, K.F.; Yuen, L.P.; Sham, J.S.; Chan, C.L. A randomized controlled trial of qigong exercise on fatigue symptoms, functioning, and telomerase activity in persons with chronic fatigue or chronic fatigue syndrome. Ann. Behav. Med. 2012, 44, 160–170. [Google Scholar] [CrossRef]
- Po, K.K.; Leung, J.W.; Chan, J.N.; Fung, T.K.; Sanchez-Vidana, D.I.; Sin, E.-L.; So, F.; Lau, B.W.; Siu, A.M. Protective effect of Lycium Barbarum polysaccharides on dextromethorphan-induced mood impairment and neurogenesis suppression. Brain Res. Bull. 2017, 134, 10–17. [Google Scholar] [CrossRef]
- Po, K.T.; Siu, A.M.; Lau, B.W.; Chan, J.N.; So, K.F.; Chan, C.C. Repeated, high-dose dextromethorphan treatment decreases neurogenesis and results in depression-like behavior in rats. Exp. Brain Res. 2015, 233, 2205–2214. [Google Scholar] [CrossRef] [PubMed]
- Albert, P.R.; Benkelfat, C.; Descarries, L. The neurobiology of depression--revisiting the serotonin hypothesis. I. Cellular and molecular mechanisms. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2012, 367, 2378–2381. [Google Scholar] [CrossRef] [PubMed]
- Xiong, M.; Li, Y.; Tang, P.; Zhang, Y.; Cao, M.; Ni, J.; Xing, M. Effectiveness of Aromatherapy Massage and Inhalation on Symptoms of Depression in Chinese Community-Dwelling Older Adults. J. Altern. Complement. Med. 2018, 24, 717–724. [Google Scholar] [CrossRef]
- Zhong, Y.; Zheng, Q.; Hu, P.; Huang, X.; Yang, M.; Ren, G.; Du, Q.; Luo, J.; Zhang, K.; Li, J.; et al. Sedative and hypnotic effects of compound Anshen essential oil inhalation for insomnia. BMC Complement. Altern. Med. 2019, 19, 306. [Google Scholar] [CrossRef]
- Rombola, L.; Scuteri, D.; Watanabe, C.; Sakurada, S.; Hamamura, K.; Sakurada, T.; Tonin, P.; Corasaniti, M.T.; Bagetta, G.; Morrone, L.A. Role of 5-HT1A Receptor in the Anxiolytic-Relaxant Effects of Bergamot Essential Oil in Rodent. Int. J. Mol. Sci. 2020, 21, 2597. [Google Scholar] [CrossRef]
- Belujon, P.; Grace, A.A. Dopamine System Dysregulation in Major Depressive Disorders. Int. J. Neuropsychopharmacol. 2017, 20, 1036–1046. [Google Scholar] [CrossRef] [PubMed]
- Nestler, E.J.; Carlezon, W.A., Jr. The mesolimbic dopamine reward circuit in depression. Biol. Psychiatry 2006, 59, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Xu, H.; Liu, Y.; He, H.; Li, G. Vanillin-induced amelioration of depression-like behaviors in rats by modulating monoamine neurotransmitters in the brain. Psychiatry Res. 2015, 225, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Ruan, L.; Lau, B.W.; Wang, J.; Huang, L.; Zhuge, Q.; Wang, B.; Jin, K.; So, K.F. Neurogenesis in neurological and psychiatric diseases and brain injury: From bench to bedside. Prog. Neurobiol. 2014, 115, 116–137. [Google Scholar] [CrossRef]
- Malberg, J.E.; Eisch, A.J.; Nestler, E.J.; Duman, R.S. Chronic antidepressant treatment increases neurogenesis in adult rat hippocampus. J. Neurosci. 2000, 20, 9104–9110. [Google Scholar] [CrossRef]
- Lee, J.C.; Yau, S.Y.; Lee, T.M.C.; Lau, B.W.; So, K.F. Voluntary Wheel Running Reverses the Decrease in Subventricular Zone Neurogenesis Caused by Corticosterone. Cell Transplant. 2016, 25, 1979–1986. [Google Scholar] [CrossRef]
- DeCarolis, N.A.; Eisch, A.J. Hippocampal neurogenesis as a target for the treatment of mental illness: A critical evaluation. Neuropharmacology 2010, 58, 884–893. [Google Scholar] [CrossRef] [PubMed]
- Santarelli, L.; Saxe, M.; Gross, C.; Surget, A.; Battaglia, F.; Dulawa, S.; Weisstaub, N.; Lee, J.; Duman, R.; Arancio, O. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science 2003, 301, 805–809. [Google Scholar] [CrossRef]
- Ayuob, N.N. Evaluation of the antidepressant-like effect of musk in an animal model of depression: How it works. Anat. Sci. Int. 2017, 92, 539–553. [Google Scholar] [CrossRef] [PubMed]
- Huang, E.J.; Reichardt, L.F. Trk receptors: Roles in neuronal signal transduction. Annu. Rev. Biochem. 2003, 72, 609–642. [Google Scholar] [CrossRef] [PubMed]
- Neto, F.L.; Borges, G.; Torres-Sanchez, S.; Mico, J.A.; Berrocoso, E. Neurotrophins role in depression neurobiology: A review of basic and clinical evidence. Curr. Neuropharmacol. 2011, 9, 530–552. [Google Scholar] [CrossRef]
- Shimizu, E.; Hashimoto, K.; Watanabe, H.; Komatsu, N.; Okamura, N.; Koike, K.; Shinoda, N.; Nakazato, M.; Kumakiri, C.; Okada, S.; et al. Serum brain-derived neurotrophic factor (BDNF) levels in schizophrenia are indistinguishable from controls. Neurosci. Lett. 2003, 351, 111–114. [Google Scholar] [CrossRef]
- Wu, J.J.; Cui, Y.; Yang, Y.S.; Kang, M.S.; Jung, S.C.; Park, H.R.; Yeun, H.Y.; Jang, W.J.; Lee, S.; Kwak, Y.S.; et al. Modulatory effects of aromatherapy massage intervention on electroencephalogram, psychological assessments, salivary cortisol and plasma brain-derived neurotrophic factor. Complement. Ther. Med. 2014, 22, 456–462. [Google Scholar] [CrossRef]
- Okuda, M.; Fujita, Y.; Takada-Takatori, Y.; Sugimoto, H.; Urakami, K. Aromatherapy improves cognitive dysfunction in senescence-accelerated mouse prone 8 by reducing the level of amyloid beta and tau phosphorylation. PLoS ONE 2020, 15, 0240378. [Google Scholar] [CrossRef]
- Shinomiya, M.; Kawamura, K.; Tanida, E.; Nagoshi, M.; Motoda, H.; Kasanami, Y.; Hiragami, F.; Kano, Y. Neurite outgrowth of PC12 mutant cells induced by orange oil and d-limonene via the p38 MAPK pathway. Acta. Med. Okayama 2012, 66, 111–118. [Google Scholar] [PubMed]
- Heydari, N.; Abootalebi, M.; Jamalimoghadam, N.; Kasraeian, M.; Emamghoreishi, M.; Akbarzaded, M. Evaluation of aromatherapy with essential oils of Rosa damascena for the management of premenstrual syndrome. Int. J. Gynaecol. Obstet. 2018, 142, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Berihanova, R.R.; Minenko, I.A. Possibilities of complex non-drug programs in the correction of psychoemotional disorders of menopause in patients with metabolic syndrome. Vopr. Kurortol. Fizioter. Lech. Fiz. Kult. 2019, 96, 50–59. [Google Scholar] [CrossRef]
- Berihanova, R.R.; Minenko, I.A. Hormonal profile of women with metabolic syndrome in the background of multimodal non-medicinal correction of climacteric disorders. Adv. Gerontol. 2020, 33, 721–728. [Google Scholar]
- Chan, J.N.; Lee, J.C.; Lee, S.S.; Hui, K.K.; Chan, A.H.; Fung, T.K.; Sanchez-Vidana, D.I.; Lau, B.W.; Ngai, S.P. Interaction Effect of Social Isolation and High. Dose Corticosteroid on Neurogenesis and Emotional Behavior. Front Behav. Neurosci. 2017, 11, 18. [Google Scholar] [CrossRef]
- Atsumi, T.; Tonosaki, K. Smelling lavender and rosemary increases free radical scavenging activity and decreases cortisol level in saliva. Psychiatry Res. 2007, 150, 89–96. [Google Scholar] [CrossRef]
- Shiina, Y.; Funabashi, N.; Lee, K.; Toyoda, T.; Sekine, T.; Honjo, S.; Hasegawa, R.; Kawata, T.; Wakatsuki, Y.; Hayashi, S.; et al. Relaxation effects of lavender aromatherapy improve coronary flow velocity reserve in healthy men evaluated by transthoracic Doppler echocardiography. Int. J. Cardiol. 2008, 129, 193–197. [Google Scholar] [CrossRef]
- Toda, M.; Morimoto, K. Effect of lavender aroma on salivary endocrinological stress markers. Arch. Oral. Biol. 2008, 53, 964–968. [Google Scholar] [CrossRef]
- Chen, P.J.; Chou, C.C.; Yang, L.; Tsai, Y.L.; Chang, Y.C.; Liaw, J.J. Effects of Aromatherapy Massage on Pregnant Women’s Stress and Immune Function: A Longitudinal, Prospective, Randomized Controlled Trial. J. Altern. Complement. Med. 2017, 23, 778–786. [Google Scholar] [CrossRef]
- Field, T.; Field, T.; Cullen, C.; Largie, S.; Diego, M.; Schanberg, S.; Kuhn, C. Lavender bath oil reduces stress and crying and enhances sleep in very young infants. Early Hum. Dev. 2008, 84, 399–401. [Google Scholar] [CrossRef]
- Stringer, J.; Swindell, R.; Dennis, M. Massage in patients undergoing intensive chemotherapy reduces serum cortisol and prolactin. Psychooncology 2008, 17, 1024–1031. [Google Scholar] [CrossRef]
- Pasyar, N.; Rambod, M.; Araghi, F. The effect of bergamot orange essence on anxiety, salivary cortisol, and alpha amylase in patients prior to laparoscopic cholecystectomy: A controlled trial study. Complement Ther. Clin. Pract. 2020, 39, 101153. [Google Scholar] [CrossRef] [PubMed]
- Kawai, E.; Takeda, R.; Ota, A.; Morita, E.; Imai, D.; Suzuki, Y.; Yokoyama, H.; Ueda, S.Y.; Nakahara, H.; Miyamoto, T.; et al. Increase in diastolic blood pressure induced by fragrance inhalation of grapefruit essential oil is positively correlated with muscle sympathetic nerve activity. J. Physiol. Sci. 2020, 70, 2. [Google Scholar] [CrossRef] [PubMed]
- Saiyudthong, S.; Marsden, C.A. Acute effects of bergamot oil on anxiety-related behaviour and corticosterone level in rats. Phytother. Res. 2011, 25, 858–862. [Google Scholar] [CrossRef] [PubMed]
- Takemoto, H.; Omameuda, Y.; Ito, M.; Fukuda, T.; Kaneko, S.; Akaike, A.; Kobayashi, Y. Inhalation administration of valerena-4,7(11)-diene from Nardostachys chinensis roots ameliorates restraint stress-induced changes in murine behavior and stress-related factors. Biol. Pharm. Bull. 2014, 37, 1050–1055. [Google Scholar] [CrossRef]
- Adebesin, A.; Adeoluwa, O.A.; Eduviere, A.T.; Umukoro, S. Methyl jasmonate attenuated lipopolysaccharide-induced depressive-like behaviour in mice. J. Psychiatr. Res. 2017, 94, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Hawken, P.A.; Fiol, C.; Blache, D. Genetic differences in temperament determine whether lavender oil alleviates or exacerbates anxiety in sheep. Physiol. Behav. 2012, 105, 1117–1123. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, S.; Nagappa, A.N.; Patil, C.R. Role of oxidative stress in depression. Drug Discov. Today 2020, 25, 1270–1276. [Google Scholar] [CrossRef]
- Rossetti, A.C.; Paladini, M.S.; Riva, M.A.; Molteni, R. Oxidation-reduction mechanisms in psychiatric disorders: A novel target for pharmacological intervention. Pharmacol. Ther. 2020, 210, 107520. [Google Scholar] [CrossRef]
- Chou, S.T.; Chang, W.L.; Chang, C.T.; Hsu, S.L.; Lin, Y.C.; Shih, Y. Cinnamomum cassia essential oil inhibits alpha-MSH-induced melanin production and oxidative stress in murine B16 melanoma cells. Int. J. Mol. Sci. 2013, 14, 19186–19201. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, D.P.; de Almeida Soares Hocayen, P.; Andrade, L.N.; Andreatini, R. A Systematic Review of the Anxiolytic-Like Effects of Essential Oils in Animal Models. Molecules 2015, 20, 1860–1862. [Google Scholar] [CrossRef] [PubMed]
- Heydari, S.; Jooyandeh, H.; Alizadeh Behbahani, B.; Noshad, M. The impact of Qodume Shirazi seed mucilage-based edible coating containing lavender essential oil on the quality enhancement and shelf life improvement of fresh ostrich meat: An. experimental and modeling study. Food Sci. Nutr. 2020, 8, 6497–6512. [Google Scholar] [CrossRef]

| EO(s) Scientific Name (Common Name) | Author (Year) | Results | |
|---|---|---|---|
| Lavandula | |||
| Lavandula angustifolia (Lavender) | Burnett et al. (2004) | Anxiolytic effect | [8] |
| Lehrner et al. (2005) | Reduced anxiety; positive effect on mood; higher level of calmness | [18] | |
| Fayazi et al. (2011) | Anxiolytic effect | [17] | |
| Senturk et al. (2018) | Anxiolytic effect | [19] | |
| Karan et al. (2019) | Blood pressure control; anxiolytic effect; respiratory relaxation | [12] | |
| Ebrahimi et al. (2021) | Antidepressant effect; anxiolytic effect; reduced stress | [20] | |
| Citrus | |||
| Citrus sinensis (Sweet orange) | Lehrner et al. (2005) | Anxiolytic effect; positive effect on mood | [18] |
| Goes et al. (2012) | Anxiolytic effect | [21] | |
| Citrus junos (Yuzu) | Matsumoto et al. (2014) | Anxiolytic effect; antidepressant effect | [22] |
| Citrus bergamia (Bergamot) | Watanabe et al. (2015) | Reduced salivary cortisol level | [23] |
| Matricaria | |||
| Matricaria chamomilla (Chamomile) | McKay et al. (2006) | Antidepressant effect; anxiolytic effect | [24] |
| Ebrahimi et al. (2021) | Antidepressant effect; anxiolytic effect; reduced stress | [20] | |
| Salvia | |||
| Salvia rosmarinus (Rosemary) | Burnett et al. (2004) | Anxiolytic effect | [8] |
| Salvia officinalis (Sage) | Muss et al. (2010 | Positive effect on mood | [25] |
| Salvia lavandulaefolia (Spanish sage) | Muss et al. (2010) | Positive effect on mood | [25] |
| Mixture | |||
| Lavandula angustifolia (Lavender) + Rosa damascena (Damascus Rose) | Conrad et al. (2012) | Anxiolytic effect; antidepressant effect | [26] |
| Lavandula angustifolia (Lavender) Cananga odorata (Ylang-ylang) + Citrus aurantium (Neroli) | Song et al. (2017) | Anxiolytic effect | [27] |
| EO(s) Scientific Name (Common Name) | Author (Year) | Animal | Behaviour Outcome (a) | Secondary Outcome (b) | Results | |
|---|---|---|---|---|---|---|
| Lavandula | ||||||
| Lavandula angustifolia (Lavender) | Chioca et al. (2013) | Mice | MBT, OFT | (a) Increase in locomotor activity (b) - | [30] | |
| Chioca et al. (2013) | Mice | EPM | 5-HT | (a) Increase in open arm timing (b) Increase of 5-HT level | [31] | |
| Coelho et al. (2014) | Rats | CFT | c-Fos | (a) Decrease in freezing response (b) Increase in c-Fos expression | [41] | |
| Sanchez-Vidana et al. (2019) | Rats | OFT, FST | DCX, BDNF | (a) Increase in locomotor activity; decrease in immobility timing (b) Increase in DCX expression and BDNF level | [35] | |
| Citrus | ||||||
| Citrus limon (Lemon) | Komiya et al. (2006) | Mice | EPM, OFT FST | DA, 5-HT | (a) Increase in open arm timing and locomotor activity; decrease in immobility timing (b) Increase of 5-HT and DA levels | [32] |
| Citrus sinensis (Sweet orange) | Hocayen et al. (2019) | Mice | MBT, OFT, Light/dark test | NADPH-d | (a) Increase in locomotor activity and spending time in bright area (b) Decrease of NADPH cells | [42] |
| Other | ||||||
| Acorus gramineus (Japanese sweet flag) | Koo et al. (2003) | Mice | NADPH-d | (a) - (b) Decrease of NADPH cells | [43] | |
| Perilla frutescens (Perilla) | Ji et al. (2014) | Mice | OFT, FST, TST | 5-HT, 5-HIAA | (a) Decrease in immobility timing (b) Increase of 5-HT and 5-HIAA levels | [34] |
| Coriandrum sativum (Coriander) | Cioanca et al. (2014) | Rats | EPM, FST | GSH | (a) Increase in open arm timing; decrease in immobility timing (b) Increase of GSH | [44] |
| Asarum caudatum (Asarum) | Park et al. (2015) | Mice | FST, TST | CRF, 5-HT | (a) Decrease in immobility timing (b) Decrease of CRF; increase of 5-HT level | [45] |
| Rosa disambiguation (Rose) | Villareal et al. (2017) | Rats | EPM | (a) Increase in open arm timing (b) - | [46] | |
| Rosmarinus officinalis (Rosemary) | Villareal et al. (2017) | Mice | TST | DA, Cort | (a) Decrease in immobility timing (b) Decrease of serum Cort level and increase of brain DA levels | [47] |
| Cananga odorata (Ylang ylang) | Zhang et al. (2018) | Mice | EPM | 5-HT | (a) Increase in open arm timing and locomotor activity (b) Increase of 5-HT level | [33] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fung, T.K.H.; Lau, B.W.M.; Ngai, S.P.C.; Tsang, H.W.H. Therapeutic Effect and Mechanisms of Essential Oils in Mood Disorders: Interaction between the Nervous and Respiratory Systems. Int. J. Mol. Sci. 2021, 22, 4844. https://doi.org/10.3390/ijms22094844
Fung TKH, Lau BWM, Ngai SPC, Tsang HWH. Therapeutic Effect and Mechanisms of Essential Oils in Mood Disorders: Interaction between the Nervous and Respiratory Systems. International Journal of Molecular Sciences. 2021; 22(9):4844. https://doi.org/10.3390/ijms22094844
Chicago/Turabian StyleFung, Timothy K. H., Benson W. M. Lau, Shirley P. C. Ngai, and Hector W. H. Tsang. 2021. "Therapeutic Effect and Mechanisms of Essential Oils in Mood Disorders: Interaction between the Nervous and Respiratory Systems" International Journal of Molecular Sciences 22, no. 9: 4844. https://doi.org/10.3390/ijms22094844
APA StyleFung, T. K. H., Lau, B. W. M., Ngai, S. P. C., & Tsang, H. W. H. (2021). Therapeutic Effect and Mechanisms of Essential Oils in Mood Disorders: Interaction between the Nervous and Respiratory Systems. International Journal of Molecular Sciences, 22(9), 4844. https://doi.org/10.3390/ijms22094844






