GeromiRs Are Downregulated in the Tumor Microenvironment during Colon Cancer Colonization of the Liver in a Murine Metastasis Model
Abstract
1. Introduction
2. Results
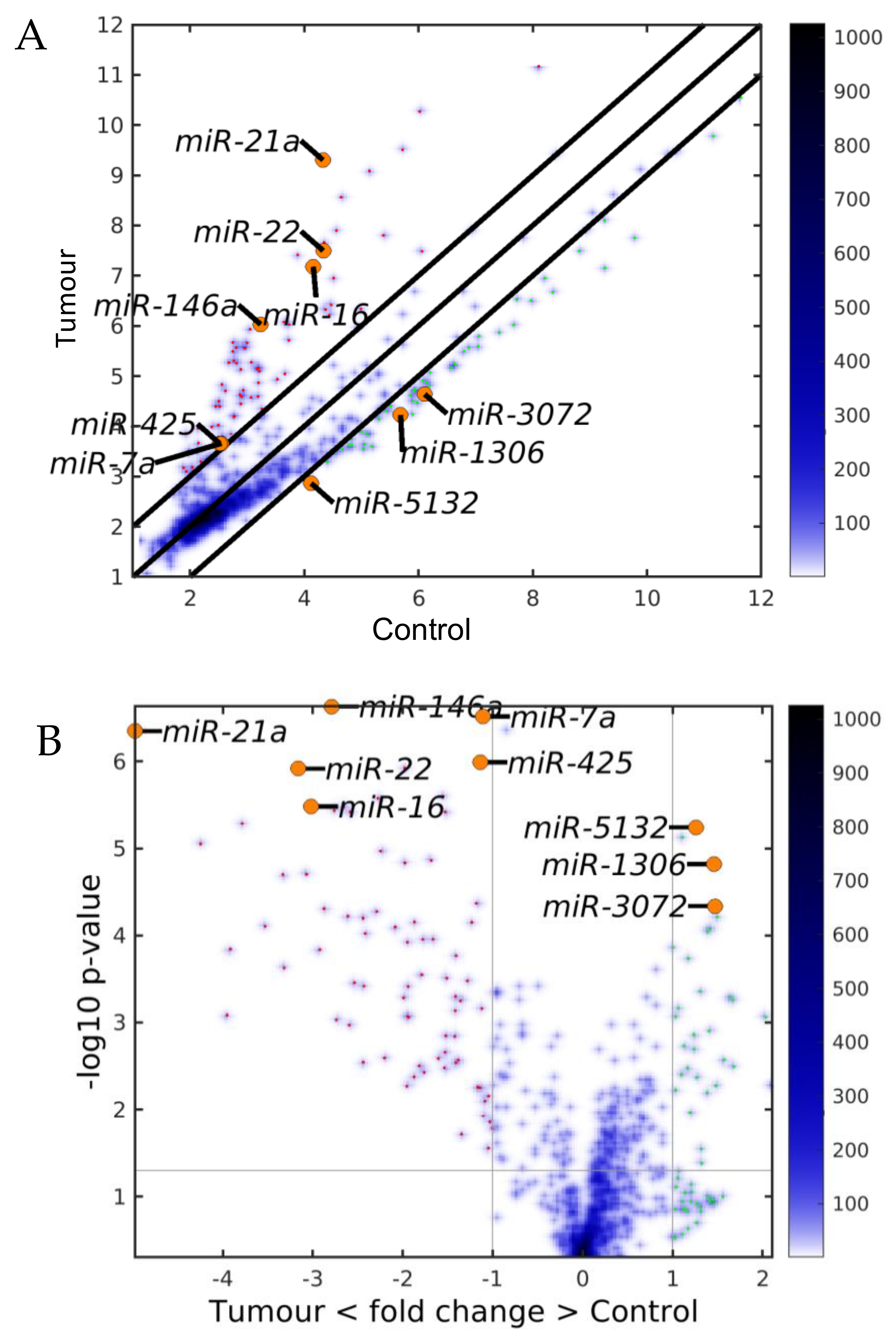
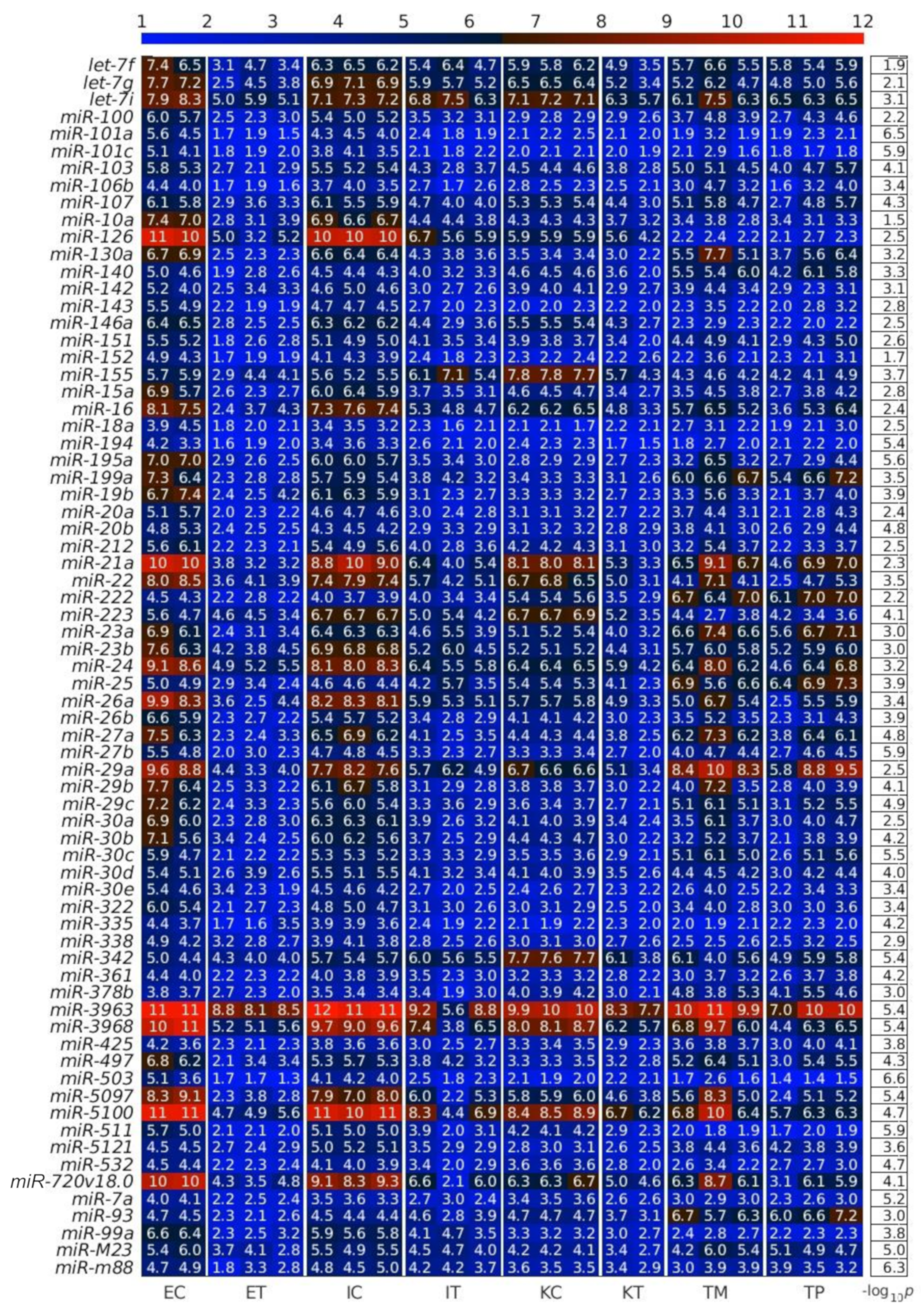
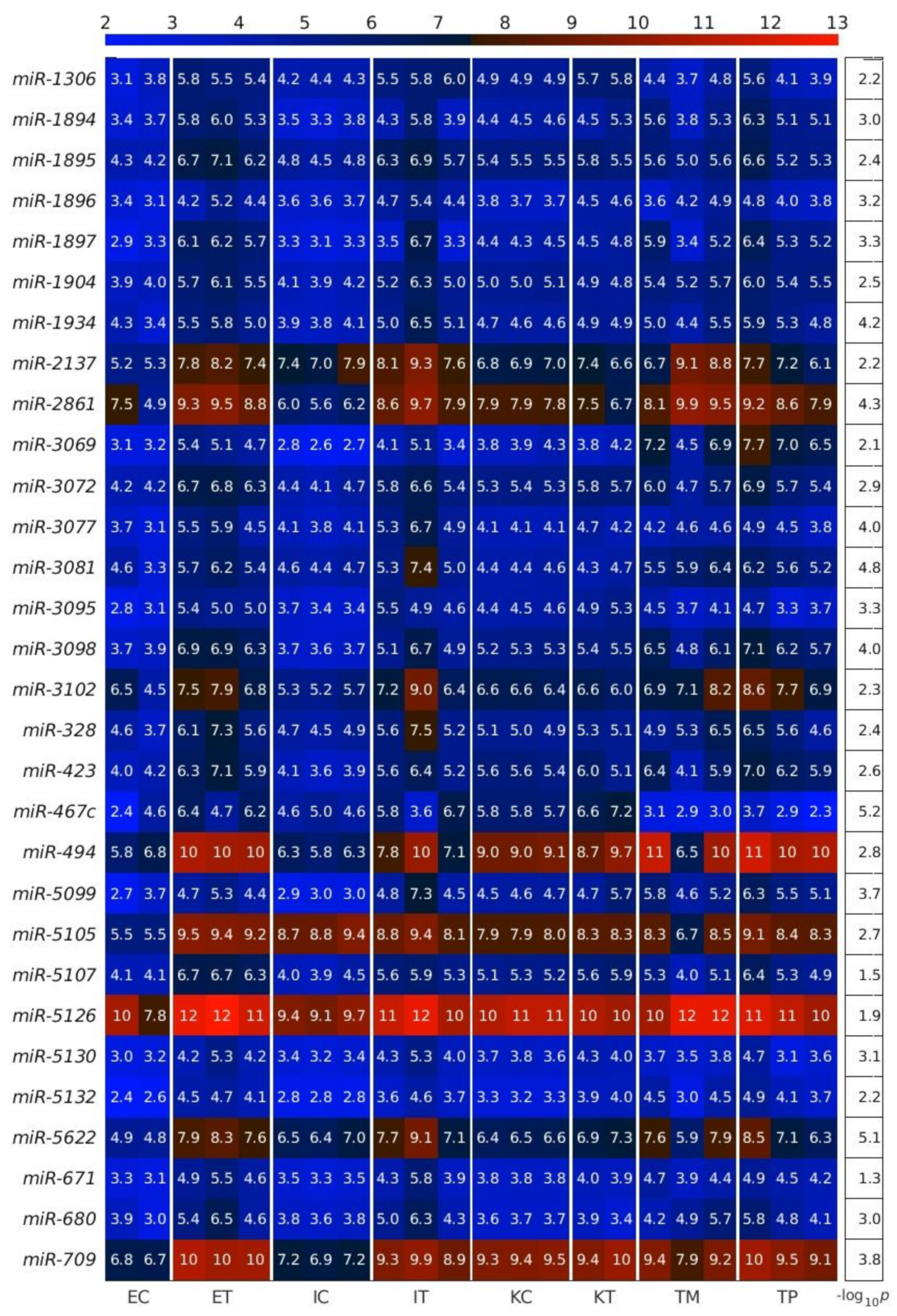
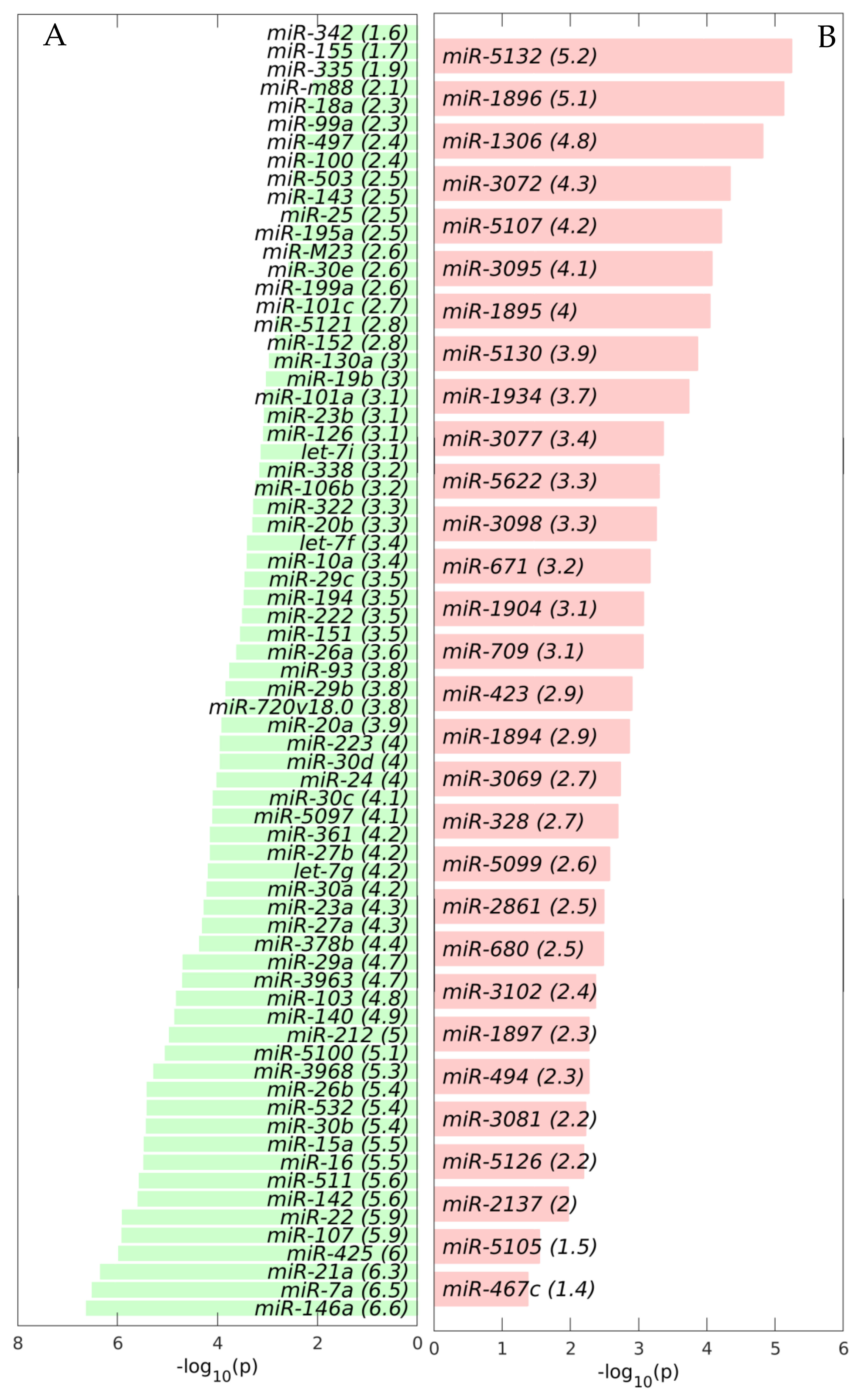
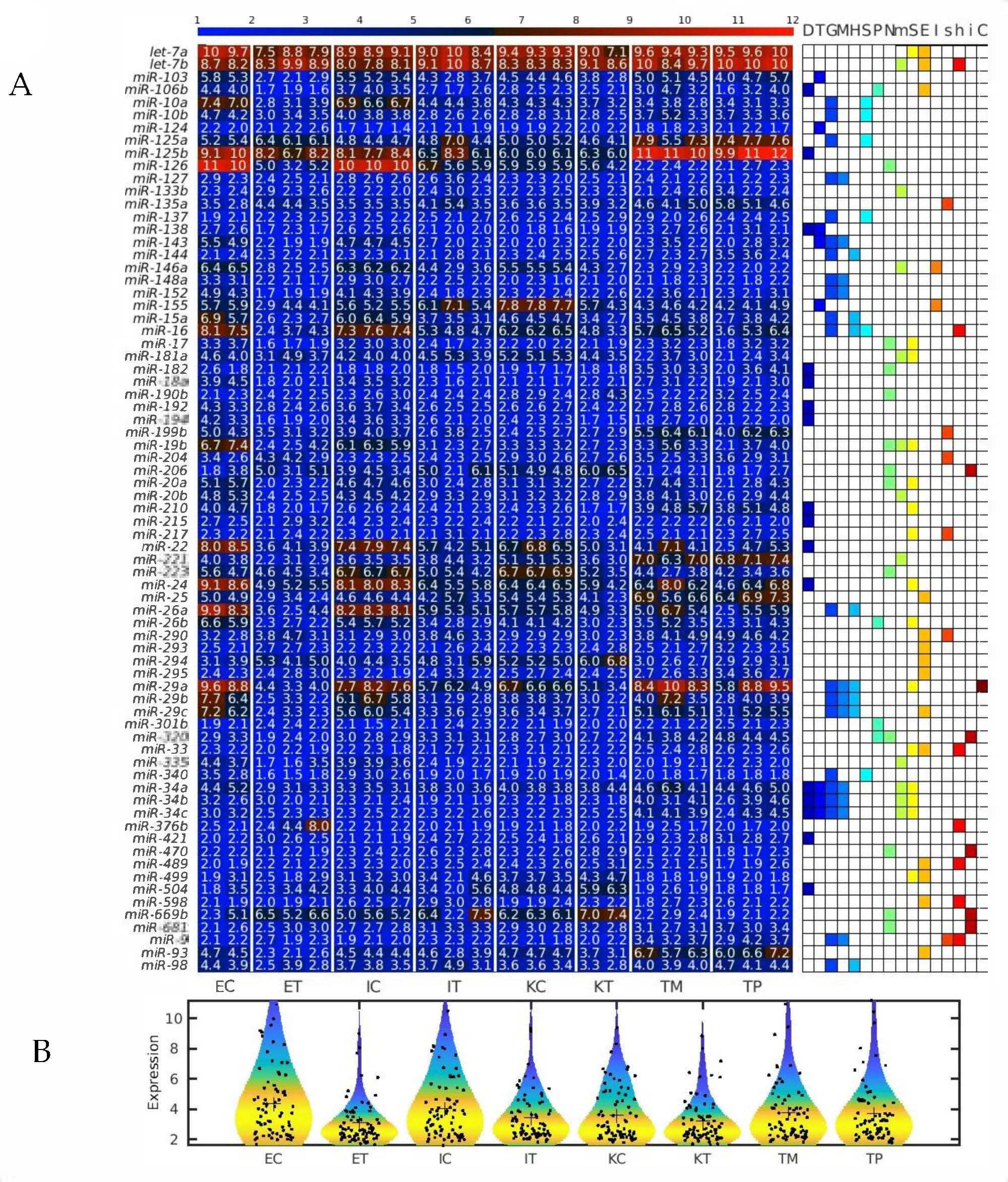
2.1. MiRNAs in the Liver TME Are Mainly Downregulated
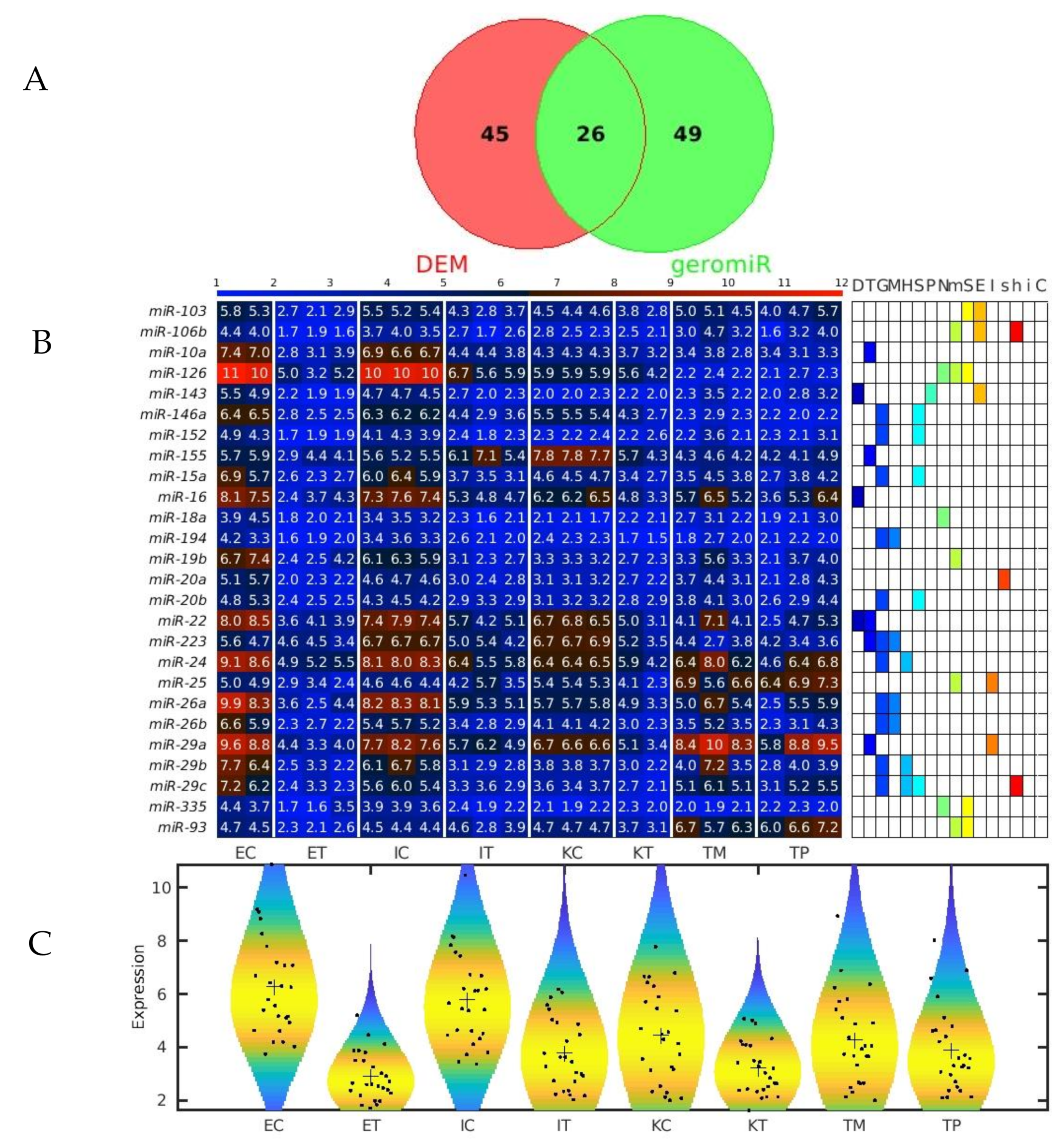
2.2. GeromiRs Are Significantly Downregulated in the TME
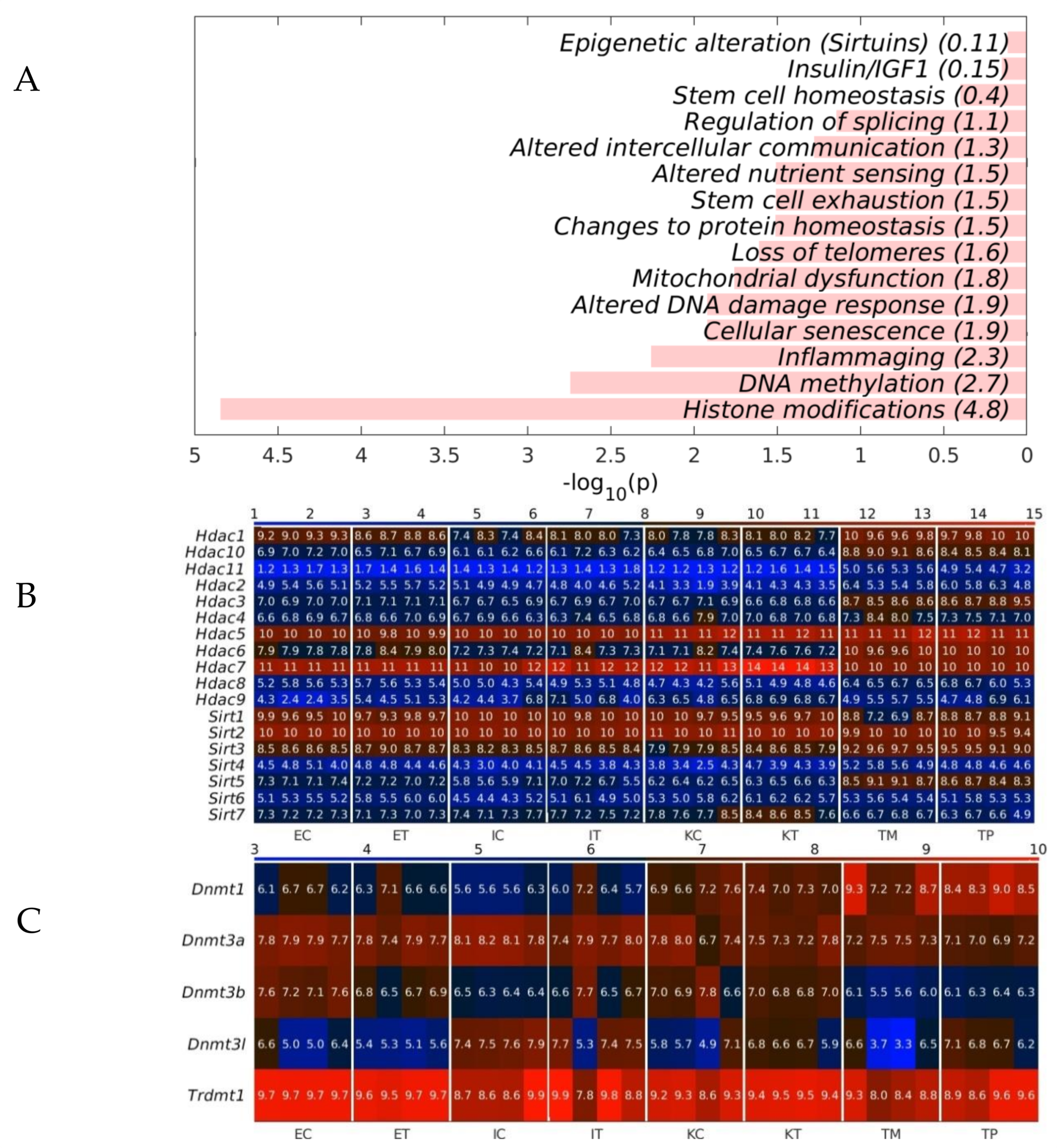
2.3. The GeromiRs Downregulated in the TME Are Significantly Involved in Histone Modifications and DNA Methylation
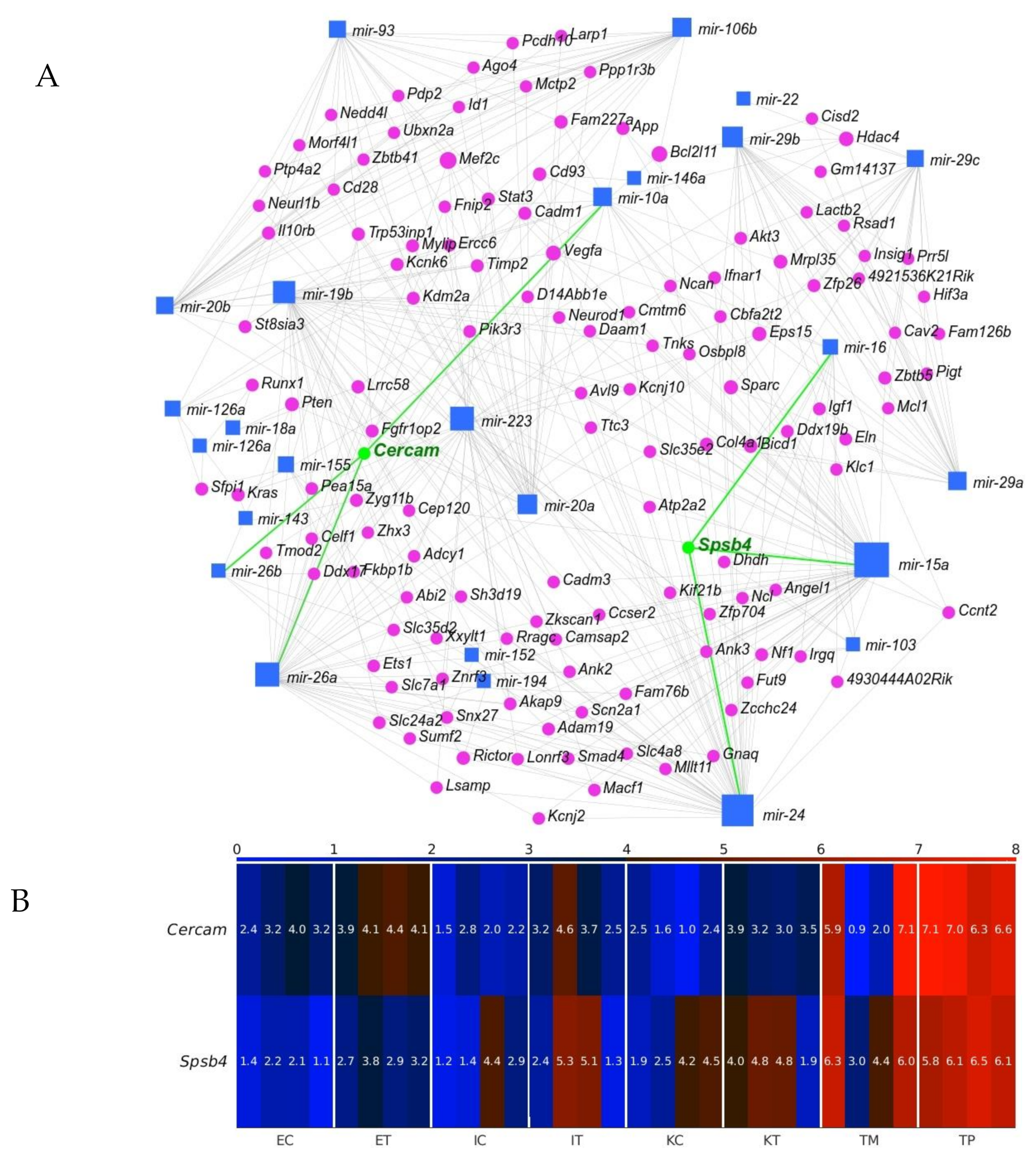
2.4. Cercam and Spsb4 Upregulated in the TME Cells Were Identified as an Element of the Network of Downregulation in the GeromiRs of TME Cells and Their Gene Targets
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Colorectal Cancer Cells
4.3. Control and Tumor-Activated Hepatic Cell Isolation and Culture
4.4. Omics Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dagogo-Jack, I.; Shaw, A.T. Tumour Heterogeneity and Resistance to Cancer Therapies. Nat. Rev. Clin. Oncol. 2018, 15, 81–94. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Key Statistics for Colorectal Cancer; American Cancer Society: Atlanta, GA, USA, 2021. [Google Scholar]
- Jin, L.; Han, B.; Siegel, E.; Cui, Y.; Giuliano, A.; Cui, X. Breast Cancer Lung Metastasis: Molecular Biology and Therapeutic Implications. Cancer Biol. Ther. 2018, 19, 858–868. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, M.; Haider, A.; Rashid, S.; Al-Nabet, A.D.M.H. Paget’s “Seed and Soil” Theory of Cancer Metastasis: An Idea Whose Time has Come. Adv. Anat. Pathol. 2019, 26, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Alpini, G.; Phillips, J.O.; Vroman, B.; LaRusso, N.F. Recent Advances in the Isolation of Liver Cells. Hepatology 1994, 20, 494–514. [Google Scholar] [CrossRef]
- Ma, L.; Hernandez, M.O.; Zhao, Y.; Mehta, M.; Tran, B.; Kelly, M.; Rae, Z.; Hernandez, J.M.; Davis, J.L.; Martin, S.P.; et al. Tumor Cell Biodiversity Drives Microenvironmental Reprogramming in Liver Cancer. Cancer Cell 2019, 36, 418–430.e6. [Google Scholar] [CrossRef]
- Papadimitriou, M.N.; Menter, D.G.; Konstantopoulos, K.; Nicolson, G.L.; McIntire, L.V. Integrin alpha4beta1/VCAM-1 Pathway Mediates Primary Adhesion of RAW117 Lymphoma Cells to Hepatic Sinusoidal Endothelial Cells Under Flow. Clin. Exp. Metastasis 1999, 17, 669–676. [Google Scholar] [CrossRef]
- Valcárcel, M.; Carrascal, T.; Crende, O.; Vidal-Vanaclocha, F. IL-18 Regulates Melanoma VLA-4 Integrin Activation through a Hierarchized Sequence of Inflammatory Factors. J. Investig. Dermatol. 2014, 134, 470–480. [Google Scholar] [CrossRef]
- Tsuchida, T.; Friedman, S.L. Mechanisms of Hepatic Stellate Cell Activation. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 397–411. [Google Scholar] [CrossRef]
- Hu, Y.B.; Ye, X.T.; Zhou, Q.Q.; Fu, R.Q. Sestrin 2 Attenuates Rat Hepatic Stellate Cell (HSC) Activation and Liver Fibrosis Via an mTOR/AMPK-Dependent Mechanism. Cell. Physiol. Biochem. 2018, 51, 2111–2122. [Google Scholar] [CrossRef]
- Fibbi, G.; Pucci, M.; D’Alessio, S.; Grappone, C.; Pellegrini, G.; Salzano, R.; Casini, A.; Milani, S.; Del Rosso, M. Transforming Growth Factor Beta-1 Stimulates Invasivity of Hepatic Stellate Cells by Engagement of the Cell-Associated Fibrinolytic System. Growth Factors 2001, 19, 87–100. [Google Scholar] [CrossRef]
- Marquez, J.; Fernandez-Piñeiro, I.; Araúzo-Bravo, M.J.; Poschmann, G.; Stühler, K.; Khatib, A.M.; Sanchez, A.; Unda, F.; Ibarretxe, G.; Bernales, I.; et al. Targeting Liver Sinusoidal Endothelial Cells with miR-20a-Loaded Nanoparticles Reduces Murine Colon Cancer Metastasis to the Liver. Int. J. Cancer 2018, 143, 709–719. [Google Scholar] [CrossRef]
- Sun, X.; He, Y.; Ma, T.T.; Huang, C.; Zhang, L.; Li, J. Participation of miR-200a in TGF-Β1-Mediated Hepatic Stellate Cell Activation. Mol. Cell. Biochem. 2014, 388, 11–23. [Google Scholar] [CrossRef]
- Saliminejad, K.; Khorram Khorshid, H.R.; Soleymani Fard, S.; Ghaffari, S.H. An Overview of microRNAs: Biology, Functions, Therapeutics, and Analysis Methods. J. Cell. Physiol. 2019, 234, 5451–5465. [Google Scholar] [CrossRef]
- Bayraktar, R.; Van Roosbroeck, K.; Calin, G.A. Cell-to-Cell Communication: MicroRNAs as Hormones. Mol. Oncol. 2017, 11, 1673–1686. [Google Scholar] [CrossRef]
- Putteeraj, M.; Fairuz, Y.M.; Teoh, S.L. MicroRNA Dysregulation in Alzheimer’s Disease. CNS Neurol. Disord. Drug Targets 2017, 16, 1000–1009. [Google Scholar] [CrossRef]
- Wojciechowska, A.; Braniewska, A.; Kozar-Kamińska, K. MicroRNA in Cardiovascular Biology and Disease. Adv. Clin. Exp. Med. 2017, 26, 865–874. [Google Scholar] [CrossRef]
- Acunzo, M.; Romano, G.; Wernicke, D.; Croce, C.M. MicroRNA and Cancer—A Brief Overview. Adv. Biol. Regul. 2015, 57, 1–9. [Google Scholar] [CrossRef]
- Esquela-Kerscher, A.; Slack, F.J. Oncomirs–microRNAs with a Role in Cancer. Nat. Rev. Cancer 2006, 6, 259–269. [Google Scholar] [CrossRef]
- Lopez-Camarillo, C.; Marchat, L.A.; Arechaga-Ocampo, E.; Perez-Plasencia, C.; Del Moral-Hernandez, O.; Castaneda-Ortiz, E.J.; Rodriguez-Cuevas, S. MetastamiRs: Non-Coding MicroRNAs Driving Cancer Invasion and Metastasis. Int. J. Mol. Sci. 2012, 13, 1347–1379. [Google Scholar] [CrossRef]
- Caravia, X.M.; Roiz-Valle, D.; Morán-Álvarez, A.; López-Otín, C. Functional Relevance of miRNAs in Premature Ageing. Mech. Ageing Dev. 2017, 168, 10–19. [Google Scholar] [CrossRef]
- Liu, Z.; Li, Y.; Luo, Q. Relationship between CT Signs and the Expression of miR-146a in Colon Cancer. Oncol. Lett. 2018, 16, 6598–6602. [Google Scholar] [CrossRef]
- Simanovich, E.; Brod, V.; Rahat, M.M.; Rahat, M.A. Function of miR-146a-5p in Tumor Cells as a Regulatory Switch between Cell Death and Angiogenesis: Macrophage Therapy Revisited. Front. Immunol. 2018, 8, 1931. [Google Scholar] [CrossRef]
- Xu, K.; Chen, Z.; Qin, C.; Song, X. miR-7 Inhibits Colorectal Cancer Cell Proliferation and Induces Apoptosis by Targeting XRCC2. Onco Targets Ther. 2014, 7, 325–332. [Google Scholar]
- Liu, Z.L.; Wang, H.; Liu, J.; Wang, Z.X. MicroRNA-21 (miR-21) Expression Promotes Growth, Metastasis, and Chemo- Or Radioresistance in Non-Small Cell Lung Cancer Cells by Targeting PTEN. Mol. Cell. Biochem. 2013, 372, 35–45. [Google Scholar] [CrossRef]
- Harries, L.W. MicroRNAs as Mediators of the Ageing Process. Genes 2014, 5, 656–670. [Google Scholar] [CrossRef]
- Ugalde, A.P.; Kwarciak, A.; Caravia, X.M.; López-Otín, C.; Ramsay, A.J. The Emergence of GeroMIRs: A Group of MicroRNAs Implicated in Aging. In MicroRNAs in Medicine; Lawrie, C.H., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014. [Google Scholar]
- Caravia, X.M.; López-Otín, C. Regulatory Roles of miRNAs in Aging. Adv. Exp. Med. Biol. 2015, 887, 213–230. [Google Scholar]
- Xia, J.; Zhang, W. Noncanonical microRNAs and Endogenous siRNAs in Lytic Infection of Murine Gammaherpesvirus. PLoS ONE 2012, 7, e47863. [Google Scholar] [CrossRef]
- Liu, X.; Zhou, F.; Yang, Y.; Wang, W.; Niu, L.; Zuo, D.; Li, X.; Hua, H.; Zhang, B.; Kou, Y.; et al. MiR-409-3p and MiR-1896 Co-Operatively Participate in IL-17-Induced Inflammatory Cytokine Production in Astrocytes and Pathogenesis of EAE Mice Via Targeting SOCS3/STAT3 Signaling. Glia 2019, 67, 101–112. [Google Scholar] [CrossRef]
- He, Z.J.; Li, W.; Chen, H.; Wen, J.; Gao, Y.F.; Liu, Y.J. miR-1306-3p Targets FBXL5 to Promote Metastasis of Hepatocellular Carcinoma through Suppressing Snail Degradation. Biochem. Biophys. Res. Commun. 2018, 504, 820–826. [Google Scholar] [CrossRef]
- Chang, L.; Zhou, G.; Soufan, O.; Xia, J. miRNet 2.0: Network-Based Visual Analytics for miRNA Functional Analysis and Systems Biology. Nucleic Acids Res. 2020, 48, W244–W251. [Google Scholar] [CrossRef]
- Uhlen, M.; Zhang, C.; Lee, S.; Sjöstedt, E.; Fagerberg, L.; Bidkhori, G.; Benfeitas, R.; Arif, M.; Liu, Z.; Edfors, F.; et al. A Pathology Atlas of the Human Cancer Transcriptome. Science 2017, 357, eaan2507. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Wang, H.; Chen, H.; Han, Y. SMARCD3 is a Potential Prognostic Marker and Therapeutic Target in CAFs. Aging 2020, 12, 20835–20861. [Google Scholar] [CrossRef] [PubMed]
- Wei, R.; Liu, H.; Li, C.; Guan, X.; Zhao, Z.; Ma, C.; Wang, X.; Jiang, Z. Computational Identification of 29 Colon and Rectal Cancer-Associated Signatures and their Applications in Constructing Cancer Classification and Prognostic Models. Front. Genet. 2020, 1, 1–17. [Google Scholar]
- Weiss, F.U.; Marques, I.J.; Woltering, J.M.; Vlecken, D.H.; Aghdassi, A.; Partecke, L.I.; Heidecke, C.D.; Lerch, M.M.; Bagowski, C.P. Retinoic Acid Receptor Antagonists Inhibit miR-10a Expression and Block Metastatic Behavior of Pancreatic Cancer. Gastroenterology 2009, 137, 2136–2137. [Google Scholar] [CrossRef]
- Agirre, X.; Jiménez-Velasco, A.; San José-Enériz, E.; Garate, L.; Bandrés, E.; Cordeu, L.; Aparicio, O.; Saez, B.; Navarro, G.; Vilas-Zornoza, A.; et al. Down-Regulation of Hsa-miR-10a in Chronic Myeloid Leukemia CD34+ Cells Increases USF2-Mediated Cell Growth. Mol. Cancer Res. 2008, 6, 1830–1840. [Google Scholar] [CrossRef]
- Huang, T.; Ren, K.; Ding, G.; Yang, L.; Wen, Y.; Peng, B.; Wang, G.; Wang, Z. miR-10a Increases the Cisplatin Resistance of Lung Adenocarcinoma Circulating Tumor Cells Via Targeting PIK3CA in the PI3K/Akt Pathway. Oncol. Rep. 2020, 43, 1906–1914. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, K.; Xu, Y.; Gao, Y.; Li, C.; Wang, R.; Chen, L. The Role of microRNA-26a in Human Cancer Progression and Clinical Application. Tumour Biol. 2016, 37, 7095–7108. [Google Scholar] [CrossRef]
- Zhang, Z.; Kim, K.; Li, X.; Moreno, M.; Sharp, T.; Goodheart, M.J.; Safe, S.; Dupuy, A.J.; Amendt, B.A. MicroRNA-26b Represses Colon Cancer Cell Proliferation by Inhibiting Lymphoid Enhancer Factor 1 Expression. Mol. Cancer Ther. 2014, 13, 1942–1951. [Google Scholar] [CrossRef]
- Shinjo, K.; Hara, K.; Nagae, G.; Umeda, T.; Katsushima, K.; Suzuki, M.; Murofushi, Y.; Umezu, Y.; Takeuchi, I.; Takahashi, S.; et al. A Novel Sensitive Detection Method for DNA Methylation in Circulating Free DNA of Pancreatic Cancer. PLoS ONE 2020, 15, e0233782. [Google Scholar] [CrossRef]
- Aqeilan, R.I.; Calin, G.A.; Croce, C.M. miR-15a and miR-16-1 in Cancer: Discovery, Function and Future Perspectives. Cell Death Differ. 2010, 17, 215–220. [Google Scholar] [CrossRef]
- Wang, S.; Liu, N.; Tang, Q.; Sheng, H.; Long, S.; Wu, W. MicroRNA-24 in Cancer: A Double Side Medal with Opposite Properties. Front. Oncol. 2020, 10, 553714. [Google Scholar] [CrossRef]
- Hayflick, L.; Moorhead, P.S. The Serial Cultivation of Human Diploid Cell Strains. Exp. Cell Res. 1961, 25, 585–621. [Google Scholar] [CrossRef]
- Harley, C.B.; Futcher, A.B.; Greider, C.W. Telomeres Shorten during Ageing of Human Fibroblasts. Nature 1990, 345, 458–460. [Google Scholar] [CrossRef]
- Shay, J.W.; Roninson, I.B. Hallmarks of Senescence in Carcinogenesis and Cancer Therapy. Oncogene 2004, 23, 2919–2933. [Google Scholar] [CrossRef]
- Sherr, C.J.; DePinho, R.A. Cellular Senescence: Mitotic Clock or Culture Shock? Cell 2000, 102, 407–410. [Google Scholar] [CrossRef]
- Wright, W.E.; Piatyszek, M.A.; Rainey, W.E.; Byrd, W.; Shay, J.W. Telomerase Activity in Human Germline and Embryonic Tissues and Cells. Dev. Genet. 1996, 18, 173–179. [Google Scholar] [CrossRef]
- Su, Y.H.; Huang, W.C.; Huang, T.H.; Huang, Y.J.; Sue, Y.K.; Huynh, T.T.; Hsiao, M.; Liu, T.Z.; Wu, A.T.; Lin, C.M. Folate Deficient Tumor Microenvironment Promotes Epithelial-to-Mesenchymal Transition and Cancer Stem-Like Phenotypes. Oncotarget 2016, 7, 33246–33256. [Google Scholar] [CrossRef]
- Charpin, C.; Garcia, S.; Bouvier, C.; Devictor, B.; Andrac, L.; Choux, R.; Lavaut, M. E-Cadherin Quantitative Immunocytochemical Assays in Breast Carcinomas. J. Pathol. 1997, 181, 294–300. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Y.S.; Mugiyanto, E.; Chang, W.C.; Yvonne Wan, Y.J. MiR-22 as a Metabolic Silencer and Liver Tumor Suppressor. Liver Res. 2020, 4, 74–80. [Google Scholar] [CrossRef]
- Yin, H.; Wang, Y.; Wu, Y.; Zhang, X.; Zhang, X.; Liu, J.; Wang, T.; Fan, J.; Sun, J.; Yang, A.; et al. EZH2-Mediated Epigenetic Silencing of miR-29/miR-30 Targets LOXL4 and Contributes to Tumorigenesis, Metastasis, and Immune Microenvironment Remodeling in Breast Cancer. Theranostics 2020, 10, 8494–8512. [Google Scholar] [CrossRef]
- Liu, T.; Xu, Z.; Ou, D.; Liu, J.; Zhang, J. The miR-15a/16 Gene Cluster in Human Cancer: A Systematic Review. J. Cell. Physiol. 2019, 234, 5496–5506. [Google Scholar] [CrossRef]
- Long, L.; Huang, G.; Zhu, H.; Guo, Y.; Liu, Y.; Huo, J. Down-Regulation of miR-138 Promotes Colorectal Cancer Metastasis Via Directly Targeting TWIST2. J. Transl. Med. 2013, 11, 275. [Google Scholar] [CrossRef]
- Bauer, K.M.; Hummon, A.B. Effects of the miR-143/-145 microRNA Cluster on the Colon Cancer Proteome and Transcriptome. J. Proteome Res. 2012, 11, 4744–4754. [Google Scholar] [CrossRef] [PubMed]
- Moser, A.R.; Pitot, H.C.; Dove, W.F. A Dominant Mutation that Predisposes to Multiple Intestinal Neoplasia in the Mouse. Science 1990, 247, 322–324. [Google Scholar] [CrossRef]
- Zhao, H.J.; Ren, L.L.; Wang, Z.H.; Sun, T.T.; Yu, Y.N.; Wang, Y.C.; Yan, T.T.; Zou, W.; He, J.; Zhang, Y.; et al. MiR-194 Deregulation Contributes to Colorectal Carcinogenesis Via Targeting AKT2 Pathway. Theranostics 2014, 4, 1193–1208. [Google Scholar] [CrossRef]
- Hellebrekers, D.M.; Melotte, V.; Viré, E.; Langenkamp, E.; Molema, G.; Fuks, F.; Herman, J.G.; Van Criekinge, W.; Griffioen, A.W.; van Engeland, M. Identification of Epigenetically Silenced Genes in Tumor Endothelial Cells. Cancer Res. 2007, 67, 4138–4148. [Google Scholar] [CrossRef]
- Bourassa, M.W.; Ratan, R.R. The Interplay between microRNAs and Histone Deacetylases in Neurological Diseases. Neurochem. Int. 2014, 77, 33–39. [Google Scholar] [CrossRef]
- Scott, G.K.; Mattie, M.D.; Berger, C.E.; Benz, S.C.; Benz, C.C. Rapid Alteration of microRNA Levels by Histone Deacetylase Inhibition. Cancer Res. 2006, 66, 1277–1281. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Yuan, P.; Wang, Y.; Hunsberger, J.G.; Elkahloun, A.; Wei, Y.; Damschroder-Williams, P.; Du, J.; Chen, G.; Manji, H.K. Evidence for Selective microRNAs and their Effectors as Common Long-Term Targets for the Actions of Mood Stabilizers. Neuropsychopharmacology 2009, 34, 1395–1405. [Google Scholar] [PubMed]
- Gerovska, D.; Larrinaga, G.; Solano-Iturri, J.D.; Márquez, J.; García Gallastegi, P.; Khatib, A.M.; Poschmann, G.; Stühler, K.; Armesto, M.; Lawrie, C.H.; et al. An Integrative Omics Approach Reveals Involvement of BRCA1 in Hepatic Metastatic Progression of Colorectal Cancer. Cancers 2020, 12, 2380. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; French, S.W.; Chau, T.; Liu, H.X.; Sheng, L.; Wei, F.; Stondell, J.; Garcia, J.C.; Du, Y.; Bowlus, C.L.; et al. RARβ Acts as both an Upstream Regulator and Downstream Effector of miR-22, which Epigenetically Regulates NUR77 to Induce Apoptosis of Colon Cancer Cells. FASEB J. 2019, 33, 2314–2326. [Google Scholar] [CrossRef]
- Frye, R.A. Phylogenetic Classification of Prokaryotic and Eukaryotic Sir2-Like Proteins. Biochem. Biophys. Res. Commun. 2000, 273, 793–798. [Google Scholar] [CrossRef]
- Lombard, D.B.; Schwer, B.; Alt, F.W.; Mostoslavsky, R. SIRT6 in DNA Repair, Metabolism and Ageing. J. Intern. Med. 2008, 263, 128–141. [Google Scholar] [CrossRef]
- Edgar, R.; Domrachev, M.; Lash, A.E. Gene Expression Omnibus: NCBI Gene Expression and Hybridization Array Data Repository. Nucleic Acids Res. 2002, 30, 207–210. [Google Scholar] [CrossRef]

| Hallmark | Harries 2014 [26] | Ugalde et al. 2014 [27] | Caravia and López-Otín 2015 [28] |
|---|---|---|---|
| Altered DNA damage response | miR-21, miR-24, miR-34a, miR-34b, miR-34c,miR-106b, miR-125b, miR-192, miR-194,miR-210, miR-215, miR-421, miR-504 | miR-18a, miR-22,miR-24, miR-29, miR-34a, miR-34b, miR-34c, miR-99, miR-138, miR-182, miR-210, miR-373, miR-421, miR-605 | let-7, miR-1, miR-16-1, miR-29, miR-34, miR-103, miR-124, miR-143, miR-145 |
| Loss of telomeres | miR-34a, miR-34b, miR-34c, miR-138, miR-155 | miR-155, miR-200, miR-498 | |
| DNA methylation | miR-9, miR-29a, miR-29b, miR-29c, miR-34a, miR-34b, miR-34c, miR-124a, miR-127, miR-143, miR-148a, miR-152, miR-200 | ||
| Histone modifications | miR-15a, miR-16, miR-26a, miR-29a, miR-29b, miR-29c, miR-98, miR-101, miR-144 | ||
| Regulation of splicing | miR-1, miR-7, miR-10a, miR-10b, miR-16, miR-124a, miR-125a, miR-137, miR-193a-3p, miR-340, miR-519 | ||
| Changes to protein homeostasis | miR-1, miR-26b, miR-106b, miR-301b, miR-320 | miR-9, miR-16, miR-17-5p, miR-34, miR-101, miR-130, miR-376b, miR-2016a, miR-E1108, miR-E1016 | |
| Altered nutrient sensing | miR-1, miR-17, miR-19b, miR-20a, miR-106a, miR-126, miR-190b, miR-206, miR-320,miR-486 | miR-1, miR-17, miR-19b, miR-20a, miR-106a, miR-145, miR-182, miR-206, miR-223, miR-320, miR-470, miR-669b, miR-681 | |
| Mitochondrial dysfunction | miR-34a, miR-145, miR-146a, miR-335 | let-7b, miR-19b, miR-20b,miR-34a, miR-34b, miR-34c, miR-106a, miR-133b, miR-146a, miR-181a, miR-221 | |
| Cellular senescence | let-7a, miR-17, miR-19b, miR-20a, miR-29a, miR-34a, miR-34b, miR-34c, miR-106a, miR-217, miR-369-3p, miR-371, miR-372, miR-373, miR-499 | miR-20a, miR-24, miR-146 | let-7, miR-21, miR-26b, miR-33, miR-181a, miR-210, miR-424 |
| Stem cell exhaustion | let-7a, miR-29c, miR-290, miR-291-3p, miR-292-3p, miR-293, miR-294, miR-295, miR-371, miR-369-3p, miR-499 | let-7b, miR-25, miR-33, miR-93, miR-106b, miR-141-3p, miR-486-5p, miR-489, miR-598 | |
| Inflammaging | miR-21, miR-146a, miR-155 | ||
| Epigenetic alterations (Sirtuins) | miR-9, miR-34, miR-135a, miR-181, miR-199b, miR-204, miR-217, miR-486, miR-519 | miR-9, miR-34, miR-135a, miR-191a, miR-191b, miR-199b, miR-204, miR-217, miR-290,miR-519 | |
| Stem cell homeostasis | let-7b, miR-33, miR-106b-25, miR-302, miR-486, miR-489, miR-598 | ||
| Insulin/IGF1 | lin-4, miR-1, miR-71, miR-145, miR-206, miR-239, miR-320, miR-470, miR-669b, miR-681 | ||
| Altered Intercellular Communication | let-7, miR-21, miR-29a, miR-71, miR-80 |
| Hallmark | GeromiRs |
|---|---|
| Altered DNA damage response | miR-106b, miR-125b, miR-138, miR-182, miR-18a, miR-192,miR-194, miR-210, miR-215, miR-22, miR-24, miR-34a, miR-34b, miR-34c, miR-421, miR-504 |
| Loss of telomeres | miR-103, miR-124, miR-138,miR-143, miR-155, miR-34a, miR-34b, miR-34c |
| DNA methylation | miR-127, miR-143, miR-148a,miR-152, miR-29a, miR-29b, miR-29c, miR-34a, miR-34b, miR-34c, miR-9 |
| Histone modifications | miR-144, miR-15a, miR-16, miR-26a, miR-29a, miR-29b, miR-29c, miR-98 |
| Regulation of splicing | miR-10a, miR-10b, miR-125a, miR-137, miR-16, miR-340 |
| Changes to protein homeostasis | miR-106b, miR-26b, miR-301b, miR-320 |
| Altered nutrient sensing | miR-106a, miR-126, miR-17, miR-182, miR-190b, miR-19b, miR-206,miR-20a, miR-223, miR-320, miR-470, miR-486, miR-669b, miR-681 |
| Mitochondrial dysfunction | let-7b, miR-106a, miR-133b, miR-146a, miR-181a,miR-19b, miR-20b, miR-221, miR-335, miR-34a, miR-34b, miR-34c |
| Cellular senescence | let-7a, miR-106a, miR-17, miR-181a, miR-19b, miR-20a, miR-210, miR-217,miR-24, miR-26b, miR-29a, miR-33, miR-34a, miR-34b, miR-34c, miR-499 |
| Stem cell exhaustion | let-7a, let-7b,miR-106b, miR-25, miR-290, miR-293, miR-294, miR-295, miR-29c, miR-33, miR-489, miR-499, miR-598,miR-93 |
| Inflammaging | miR-146a, miR-155 |
| Epigenetic alterations (Sirtuins) | miR-135a, miR-199b, miR-204, miR-217, miR-290, miR-486, miR-9 |
| Stem cell homeostasis | let-7b,miR-16, miR-33, miR-376b, miR-486, miR-489, miR-598, miR-9 |
| Insulin/IGF1 | miR-206, miR-320, miR-470, miR-669b, miR-681 |
| Altered Intercellular Communication | miR-29a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gerovska, D.; Garcia-Gallastegi, P.; Crende, O.; Márquez, J.; Larrinaga, G.; Unzurrunzaga, M.; Araúzo-Bravo, M.J.; Badiola, I. GeromiRs Are Downregulated in the Tumor Microenvironment during Colon Cancer Colonization of the Liver in a Murine Metastasis Model. Int. J. Mol. Sci. 2021, 22, 4819. https://doi.org/10.3390/ijms22094819
Gerovska D, Garcia-Gallastegi P, Crende O, Márquez J, Larrinaga G, Unzurrunzaga M, Araúzo-Bravo MJ, Badiola I. GeromiRs Are Downregulated in the Tumor Microenvironment during Colon Cancer Colonization of the Liver in a Murine Metastasis Model. International Journal of Molecular Sciences. 2021; 22(9):4819. https://doi.org/10.3390/ijms22094819
Chicago/Turabian StyleGerovska, Daniela, Patricia Garcia-Gallastegi, Olatz Crende, Joana Márquez, Gorka Larrinaga, Maite Unzurrunzaga, Marcos J. Araúzo-Bravo, and Iker Badiola. 2021. "GeromiRs Are Downregulated in the Tumor Microenvironment during Colon Cancer Colonization of the Liver in a Murine Metastasis Model" International Journal of Molecular Sciences 22, no. 9: 4819. https://doi.org/10.3390/ijms22094819
APA StyleGerovska, D., Garcia-Gallastegi, P., Crende, O., Márquez, J., Larrinaga, G., Unzurrunzaga, M., Araúzo-Bravo, M. J., & Badiola, I. (2021). GeromiRs Are Downregulated in the Tumor Microenvironment during Colon Cancer Colonization of the Liver in a Murine Metastasis Model. International Journal of Molecular Sciences, 22(9), 4819. https://doi.org/10.3390/ijms22094819






