Lost-in-Translation of Metabolic Effects of Inorganic Nitrate in Type 2 Diabetes: Is Ascorbic Acid the Answer?
Abstract
1. Introduction
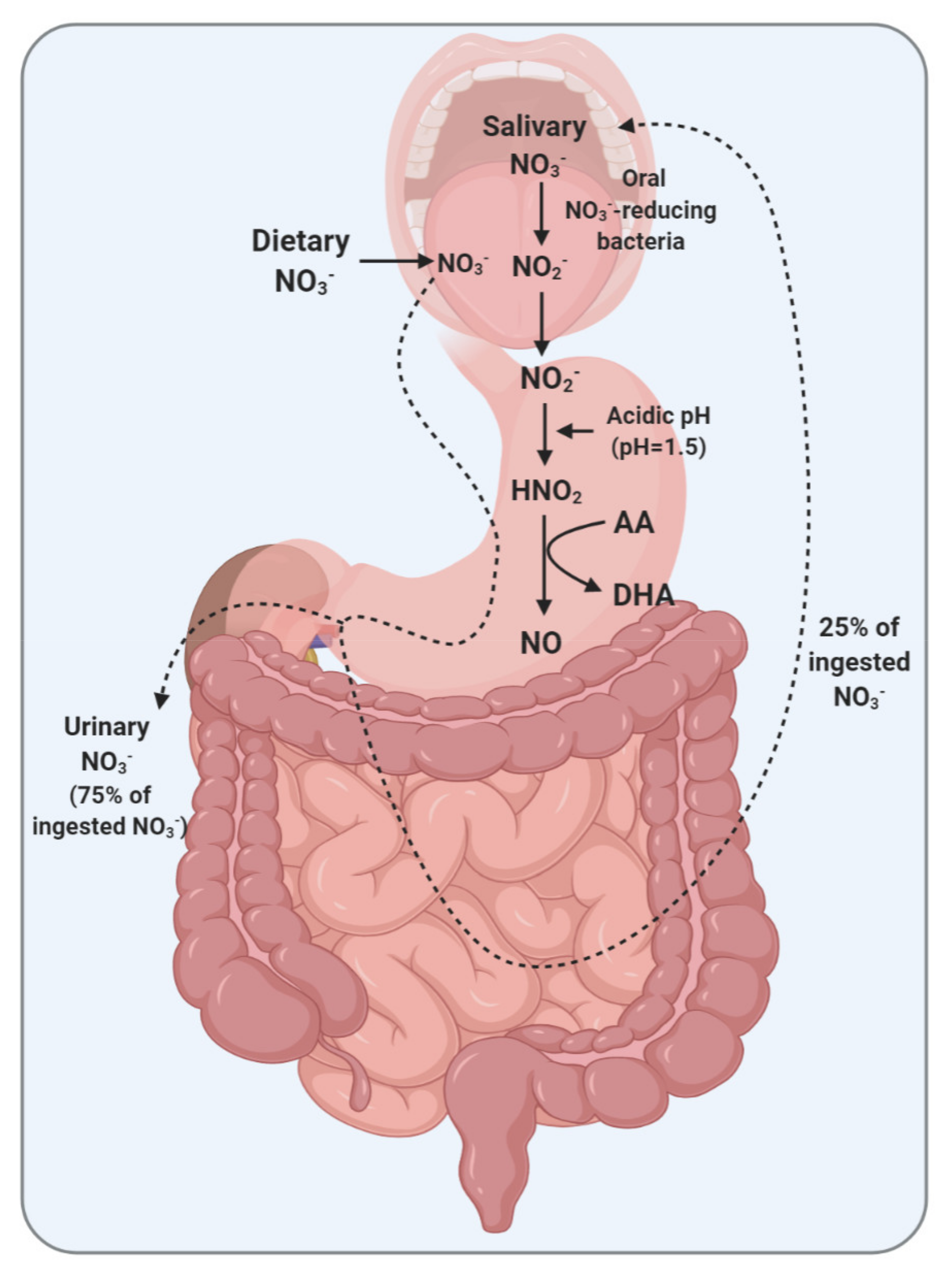
2. A Brief Overview of NO3−-NO2−-NO Pathway
3. Effects of Inorganic NO3− and NO2− in Type 2 Diabetes
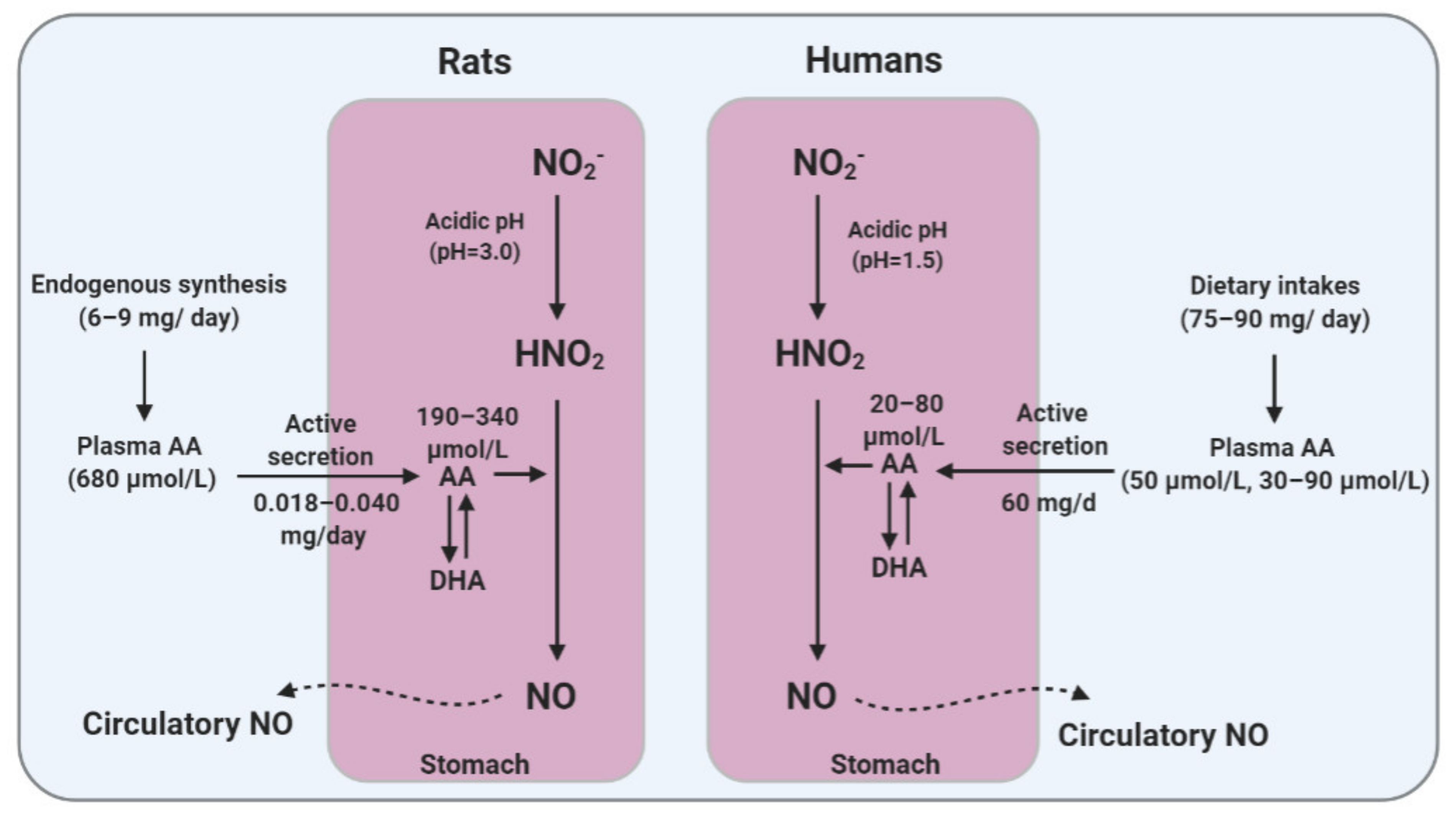
4. A Brief Overview of AA Metabolism: Differences between Animals and Humans
5. Gastric NO Generation: Critical Role of AA
5.1. Gastric Generation of NO
5.2. Gastric Secretion of AA
5.3. Role of AA in Gastric NO Generation
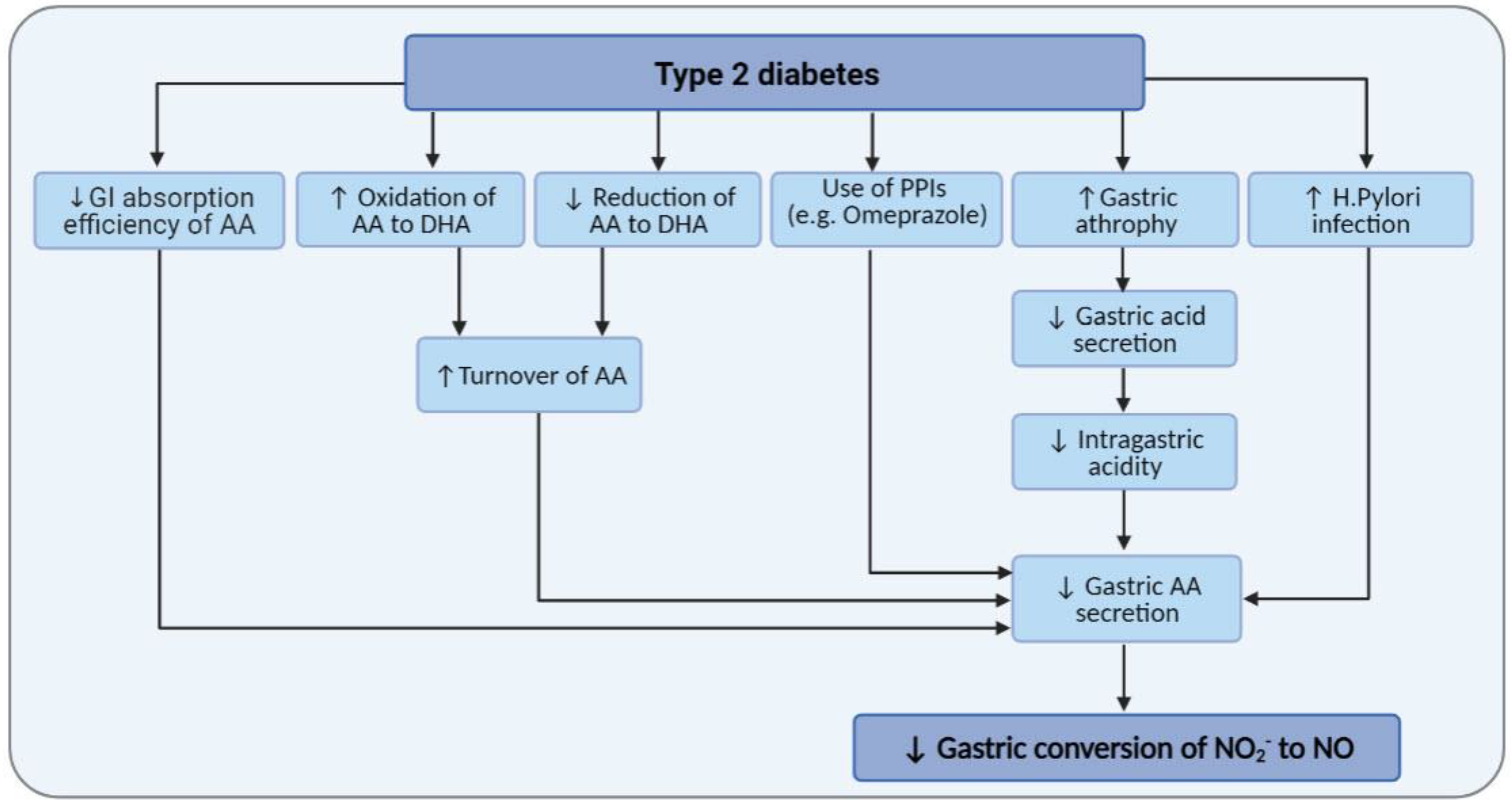
6. Diabetes and AA Metabolism
7. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lundberg, J.O.; Weitzberg, E.; Gladwin, M.T. The nitrate-nitrite-nitric oxide pathway in physiology and therapeutics. Nat. Rev. Drug Discov. 2008, 7, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, A.; Jeddi, S. Anti-obesity and anti-diabetic effects of nitrate and nitrite. Nitric Oxide Biol. Chem. 2017, 70, 9–24. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Carlstrom, M.; Weitzberg, E. Metabolic Effects of Dietary Nitrate in Health and Disease. Cell Metab. 2018, 28, 9–22. [Google Scholar] [CrossRef]
- Tessari, P.; Cecchet, D.; Cosma, A.; Vettore, M.; Coracina, A.; Millioni, R.; Iori, E.; Puricelli, L.; Avogaro, A.; Vedovato, M. Nitric Oxide Synthesis Is Reduced in Subjects With Type 2 Diabetes and Nephropathy. Diabetes 2010, 59, 2152–2159. [Google Scholar] [CrossRef]
- Natali, A.; Ribeiro, R.; Baldi, S.; Tulipani, A.; Rossi, M.; Venturi, E.; Mari, A.; Macedo, M.P.; Ferrannini, E. Systemic inhibition of nitric oxide synthesis in non-diabetic individuals produces a significant deterioration in glucose tolerance by increasing insulin clearance and inhibiting insulin secretion. Diabetologia 2013, 56, 1183–1191. [Google Scholar] [CrossRef]
- Gheibi, S.; Jeddi, S.; Carlström, M.; Gholami, H.; Ghasemi, A. Effects of long-term nitrate supplementation on carbohydrate metabolism, lipid profiles, oxidative stress, and inflammation in male obese type 2 diabetic rats. Nitric Oxide Biol. Chem. 2018, 75, 27–41. [Google Scholar] [CrossRef]
- Gheibi, S.; Bakhtiarzadeh, F.; Jeddi, S.; Farrokhfall, K.; Zardooz, H.; Ghasemi, A. Nitrite increases glucose-stimulated insulin secretion and islet insulin content in obese type 2 diabetic male rats. Nitric Oxide Biol. Chem. 2017, 64, 39–51. [Google Scholar] [CrossRef]
- Khalifi, S.; Rahimipour, A.; Jeddi, S.; Ghanbari, M.; Kazerouni, F.; Ghasemi, A. Dietary nitrate improves glucose tolerance and lipid profile in an animal model of hyperglycemia. Nitric Oxide Biol. Chem. 2015, 44, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, K.; Nakano, G.; Ehara, N.; Sonoda, K.; Ito, J.; Uchida, H.; Kobayashi, J. Dietary nitrite supplementation improves insulin resistance in type 2 diabetic KKA(y) mice. Nitric Oxide Biol. Chem. 2015, 44, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Carlstrom, M.; Larsen, F.J.; Nystrom, T.; Hezel, M.; Borniquel, S.; Weitzberg, E.; Lundberg, J.O. Dietary inorganic nitrate reverses features of metabolic syndrome in endothelial nitric oxide synthase-deficient mice. Proc. Natl. Acad. Sci. USA 2010, 107, 17716–17720. [Google Scholar] [CrossRef]
- Cordero-Herrera, I.; Guimarães, D.D.; Moretti, C.; Zhuge, Z.; Han, H.; McCann Haworth, S.; Uribe Gonzalez, A.E.; Andersson, D.C.; Weitzberg, E.; Lundberg, J.O.; et al. Head-to-head comparison of inorganic nitrate and metformin in a mouse model of cardiometabolic disease. Nitric Oxide Biol. Chem. 2020, 97, 48–56. [Google Scholar] [CrossRef]
- Siervo, M.; Lara, J.; Jajja, A.; Sutyarjoko, A.; Ashor, A.W.; Brandt, K.; Qadir, O.; Mathers, J.C.; Benjamin, N.; Winyard, P.G.; et al. Ageing modifies the effects of beetroot juice supplementation on 24-hour blood pressure variability: An individual participant meta-analysis. Nitric Oxide Biol. Chem. 2015, 47, 97–105. [Google Scholar] [CrossRef]
- Sambe, T.; Mason, R.P.; Dawoud, H.; Bhatt, D.L.; Malinski, T. Metformin treatment decreases nitroxidative stress, restores nitric oxide bioavailability and endothelial function beyond glucose control. Biomed. Pharmacother. 2018, 98, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Milsom, A.B.; Fernandez, B.O.; Garcia-Saura, M.F.; Rodriguez, J.; Feelisch, M. Contributions of nitric oxide synthases, dietary nitrite/nitrate, and other sources to the formation of NO signaling products. Antioxid Redox Signal 2012, 17, 422–432. [Google Scholar] [CrossRef]
- Carlström, M.; Liu, M.; Yang, T.; Zollbrecht, C.; Huang, L.; Peleli, M.; Borniquel, S.; Kishikawa, H.; Hezel, M.; Persson, A.E.G.; et al. Cross-talk Between Nitrate-Nitrite-NO and NO Synthase Pathways in Control of Vascular NO Homeostasis. Antioxid Redox Signal 2015, 23, 295–306. [Google Scholar] [CrossRef]
- Wong, W.T.; Cooke, J.P. Nutritional Impact on the Nitric Oxide Pathway. In Nitrite and Nitrate in Human Health and Disease; Springer: Berlin/Heidelberg, Germany, 2011; pp. 97–122. [Google Scholar]
- Bahadoran, Z.; Mirmiran, P.; Ghasemi, A.; Carlström, M.; Azizi, F.; Hadaegh, F. Vitamin C intake modify the impact of dietary nitrite on the incidence of type 2 diabetes: A 6-year follow-up in Tehran Lipid and Glucose Study. Nitric Oxide Biol. Chem. 2017, 62, 24–31. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Carlström, M.; Ghasemi, A.; Mirmiran, P.; Azizi, F.; Hadaegh, F. Total antioxidant capacity of the diet modulates the association between habitual nitrate intake and cardiovascular events: A longitudinal follow-up in Tehran Lipid and Glucose Study. Nutr. Metab. 2018, 15, 19. [Google Scholar] [CrossRef]
- Hord, N.G.; Tang, Y.; Bryan, N.S. Food sources of nitrates and nitrites: The physiologic context for potential health benefits. Am. J. Clin. Nutr. 2009, 90, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Gangolli, S.D.; Van Den Brandt, P.A.; Feron, V.J.; Janzowsky, C.; Koeman, J.H.; Speijers, G.J.; Spiegelhalder, B.; Walker, R.; Wishnok, J.S. Nitrate, nitrite and N-nitroso compounds. Eur. J. Pharmacol. Environ. Toxicol. Pharmacol. 1994, 292, 1–38. [Google Scholar] [CrossRef]
- Schultz, D.S.; Deen, W.M.; Karel, S.F.; Wagner, D.A.; Tannenbaum, S.R. Pharmacokinetics of nitrate in humans: Role of gastrointestinal absorption and metabolism. Carcinogenesis 1985, 6, 847–852. [Google Scholar] [CrossRef] [PubMed]
- Wagner, D.A.; Schultz, D.S.; Deen, W.M.; Young, V.R.; Tannenbaum, S.R. Metabolic fate of an oral dose of 15N-labeled nitrate in humans: Effect of diet supplementation with ascorbic acid. Cancer Res. 1983, 43, 1921–1925. [Google Scholar] [PubMed]
- Ellen, G.; Schuller, P.L.; Bruijns, E.; Froeling, P.G.; Baadenhuijsen, H.U. Volatile N-nitrosamines, nitrate and nitrite in urine and saliva of healthy volunteers after administration of large amounts of nitrate. IARC Sci. Publ. 1982, 41, 365–378. [Google Scholar]
- Radomski, J.L.; Palmiri, C.; Hearn, W.L. Concentrations of nitrate in normal human urine and the effect of nitrate ingestion. Toxicol. Appl. Pharmacol. 1978, 45, 63–68. [Google Scholar] [CrossRef]
- Pannala, A.S.; Mani, A.R.; Spencer, J.P.; Skinner, V.; Bruckdorfer, K.R.; Moore, K.P.; Rice-Evans, C.A. The effect of dietary nitrate on salivary, plasma, and urinary nitrate metabolism in humans. Free Radic. Biol. Med. 2003, 34, 576–584. [Google Scholar] [CrossRef]
- Saul, R.L.; Kabir, S.H.; Cohen, Z.; Bruce, W.R.; Archer, M.C. Reevaluation of Nitrate and Nitrite Levels in the Human Intestine. Cancer Res. 1981, 41, 2280–2283. [Google Scholar]
- Mitchell, H.; Shonle, H.; Grindley, H. The origin of the nitrates in the urine. J. Biol. Chem. 1916, 24, 461–490. [Google Scholar] [CrossRef]
- Spiegelhalder, B.; Eisenbrand, G.; Preussmann, R. Influence of dietary nitrate on nitrite content of human saliva: Possible relevance to in vivo formation of N-nitroso compounds. Food Cosmet. Toxicol. 1976, 14, 545–548. [Google Scholar] [CrossRef]
- Qin, L.; Liu, X.; Sun, Q.; Fan, Z.; Xia, D.; Ding, G.; Ong, H.L.; Adams, D.; Gahl, W.A.; Zheng, C.; et al. Sialin (SLC17A5) functions as a nitrate transporter in the plasma membrane. Proc. Natl. Acad. Sci. USA 2012, 109, 13434–13439. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Govoni, M. Inorganic nitrate is a possible source for systemic generation of nitric oxide. Free Radic. Biol. Med. 2004, 37, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Montenegro, M.F.; Sundqvist, M.L.; Nihlén, C.; Hezel, M.; Carlström, M.; Weitzberg, E.; Lundberg, J.O. Profound differences between humans and rodents in the ability to concentrate salivary nitrate: Implications for translational research. Redox Biol. 2016, 10, 206–210. [Google Scholar] [CrossRef]
- Witter, J.P.; Balish, E. Distribution and metabolism of ingested NO3− and NO2− in germfree and conventional-flora rats. Appl. Environ. Microbiol. 1979, 38, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Walker, R. The metabolism of dietary nitrites and nitrates. Biochem. Soc. Trans. 1996, 24, 780–785. [Google Scholar] [CrossRef] [PubMed]
- Doel, J.J.; Benjamin, N.; Hector, M.P.; Rogers, M.; Allaker, R.P. Evaluation of bacterial nitrate reduction in the human oral cavity. Eur. J. Oral Sci. 2005, 113, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Duncan, C.; Townend, J.; Killham, K.; Smith, L.M.; Johnston, P.; Dykhuizen, R.; Kelly, D.; Golden, M.; Benjamin, N.; et al. Nitrate-reducing bacteria on rat tongues. Appl. Environ. Microbiol. 1997, 63, 924–930. [Google Scholar] [CrossRef] [PubMed]
- Kapil, V.; Haydar, S.M.; Pearl, V.; Lundberg, J.O.; Weitzberg, E.; Ahluwalia, A. Physiological role for nitrate-reducing oral bacteria in blood pressure control. Free Radic. Biol. Med. 2013, 55, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Petersson, J.; Carlström, M.; Schreiber, O.; Phillipson, M.; Christoffersson, G.; Jägare, A.; Roos, S.; Jansson, E.A.; Persson, A.E.; Lundberg, J.O.; et al. Gastroprotective and blood pressure lowering effects of dietary nitrate are abolished by an antiseptic mouthwash. Free Radic. Biol. Med. 2009, 46, 1068–1075. [Google Scholar] [CrossRef]
- Govoni, M.; Jansson, E.A.; Weitzberg, E.; Lundberg, J.O. The increase in plasma nitrite after a dietary nitrate load is markedly attenuated by an antibacterial mouthwash. Nitric Oxide Biol. Chem. 2008, 19, 333–337. [Google Scholar] [CrossRef]
- Hyde, E.R.; Luk, B.; Cron, S.; Kusic, L.; McCue, T.; Bauch, T.; Kaplan, H.; Tribble, G.; Petrosino, J.F.; Bryan, N.S. Characterization of the rat oral microbiome and the effects of dietary nitrate. Free Radic. Biol. Med. 2014, 77, 249–257. [Google Scholar] [CrossRef]
- McKnight, G.M.; Smith, L.M.; Drummond, R.S.; Duncan, C.W.; Golden, M.; Benjamin, N. Chemical synthesis of nitric oxide in the stomach from dietary nitrate in humans. Gut 1997, 40, 211–214. [Google Scholar] [CrossRef]
- Iijima, K.; Fyfe, V.; McColl, K.E. Studies of nitric oxide generation from salivary nitrite in human gastric juice. Scand. J. Gastroenterol. 2003, 38, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Bahadoran, Z.; Mirmiran, P.; Ghasemi, A. Role of Nitric Oxide in Insulin Secretion and Glucose Metabolism. Trends Endocrinol. Metab. TEM 2020, 31, 118–130. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.-N.; Tain, Y.-L. Regulation of Nitric Oxide Production in the Developmental Programming of Hypertension and Kidney Disease. Int. J. Mol. Sci. 2019, 20, 681. [Google Scholar] [CrossRef]
- Chen, J.-Y.; Ye, Z.-X.; Wang, X.-F.; Chang, J.; Yang, M.-W.; Zhong, H.-H.; Hong, F.-F.; Yang, S.-L. Nitric oxide bioavailability dysfunction involves in atherosclerosis. Biomed. Pharmacother. 2018, 97, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Han, M.; Rezaei, A.; Li, D.; Wu, G.; Ma, X. L-Arginine Modulates Glucose and Lipid Metabolism in Obesity and Diabetes. Curr. Protein Pept. Sci. 2017, 18, 599–608. [Google Scholar] [CrossRef]
- Piatti, P.M.; Monti, L.D.; Valsecchi, G.; Magni, F.; Setola, E.; Marchesi, F.; Galli-Kienle, M.; Pozza, G.; Alberti, K.G. Long-term oral L-arginine administration improves peripheral and hepatic insulin sensitivity in type 2 diabetic patients. Diabetes Care 2001, 24, 875–880. [Google Scholar] [CrossRef] [PubMed]
- Azizi, S.; Mahdavi, R.; Vaghef-Mehrabany, E.; Maleki, V.; Karamzad, N.; Ebrahimi-Mameghani, M. Potential roles of Citrulline and watermelon extract on metabolic and inflammatory variables in diabetes mellitus, current evidence and future directions: A systematic review. Clin. Exp. Pharmacol. Physiol. 2020, 47, 187–198. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Weitzberg, E. NO generation from inorganic nitrate and nitrite: Role in physiology, nutrition and therapeutics. Arch. Pharmacal Res. 2009, 32, 1119–1126. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Ghasemi, A.; Mirmiran, P.; Azizi, F.; Hadaegh, F. Beneficial effects of inorganic nitrate/nitrite in type 2 diabetes and its complications. Nutr. Metab. 2015, 12, 16. [Google Scholar] [CrossRef]
- McNally, B.; Griffin, J.L.; Roberts, L.D. Dietary inorganic nitrate: From villain to hero in metabolic disease? Mol. Nutr. Food Res. 2016, 60, 67–78. [Google Scholar] [CrossRef]
- Walker, H.A.; McGing, E.; Fisher, I.; Böger, R.H.; Bode-Böger, S.M.; Jackson, G.; Ritter, J.M.; Chowienczyk, P.J. Endothelium-dependent vasodilation is independent of the plasma L-arginine/ADMA ratio in men with stable angina: Lack of effect of oral l-arginine on endothelial function, oxidative stress and exercise performance. J. Am. Coll. Cardiol. 2001, 38, 499–505. [Google Scholar] [CrossRef]
- Schulman, S.P.; Becker, L.C.; Kass, D.A.; Champion, H.C.; Terrin, M.L.; Forman, S.; Ernst, K.V.; Kelemen, M.D.; Townsend, S.N.; Capriotti, A.; et al. L-arginine therapy in acute myocardial infarction: The Vascular Interaction With Age in Myocardial Infarction (VINTAGE MI) randomized clinical trial. JAMA 2006, 295, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.M., Jr. Regulation of enzymes of urea and arginine synthesis. Annu. Rev. Nutr. 1992, 12, 81–101. [Google Scholar] [CrossRef]
- Cynober, L.; Le Boucher, J.; Vasson, M.-P. Arginine metabolism in mammals. J. Nutr. Biochem. 1995, 6, 402–413. [Google Scholar] [CrossRef]
- Adams, M.R.; Forsyth, C.J.; Jessup, W.; Robinson, J.; Celermajer, D.S. Oral L-arginine inhibits platelet aggregation but does not enhance endothelium-dependent dilation in healthy young men. J. Am. Coll. Cardiol. 1995, 26, 1054–1061. [Google Scholar] [CrossRef]
- Mohan, S.; Wu, C.C.; Shin, S.; Fung, H.L. Continuous exposure to L-arginine induces oxidative stress and physiological tolerance in cultured human endothelial cells. Amino Acids 2012, 43, 1179–1188. [Google Scholar] [CrossRef]
- Nyström, T.; Ortsäter, H.; Huang, Z.; Zhang, F.; Larsen, F.J.; Weitzberg, E.; Lundberg, J.O.; Sjöholm, Å. Inorganic nitrite stimulates pancreatic islet blood flow and insulin secretion. Free Radic. Biol. Med. 2012, 53, 1017–1023. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, A.; Afzali, H.; Jeddi, S. Effect of oral nitrite administration on gene expression of SNARE proteins involved in insulin secretion from pancreatic islets of male type 2 diabetic rats. Biomed. J. 2021, in press. [Google Scholar] [CrossRef]
- Roberts, L.D.; Ashmore, T.; Kotwica, A.O.; Murfitt, S.A.; Fernandez, B.O.; Feelisch, M.; Murray, A.J.; Griffin, J.L. Inorganic nitrate promotes the browning of white adipose tissue through the nitrate-nitrite-nitric oxide pathway. Diabetes 2015, 64, 471–484. [Google Scholar] [CrossRef]
- Gheibi, S.; Jeddi, S.; Carlstrom, M.; Kashfi, K.; Ghasemi, A. Hydrogen sulfide potentiates the favorable metabolic effects of inorganic nitrite in type 2 diabetic rats. Nitric Oxide Biol. Chem. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kapil, V.; Khambata, R.; Jones, D.; Rathod, K.; Primus, C.; Massimo, G.; Fukuto, J.; Ahluwalia, A. The Noncanonical Pathway for In Vivo Nitric Oxide Generation: The Nitrate-Nitrite-Nitric Oxide Pathway. Pharmacol. Rev. 2020, 72, 692–766. [Google Scholar] [CrossRef]
- Jeddi, S.; Yousefzadeh, N.; Afzali, H.; Ghasemi, A. Long-term nitrate administration increases expression of browning genes in epididymal adipose tissue of male type 2 diabetic rats. Gene 2021, 766, 145155. [Google Scholar] [CrossRef]
- Tian, R.; Peng, R.; Yang, Z.; Peng, Y.-Y.; Lu, N. Supplementation of dietary nitrate attenuated oxidative stress and endothelial dysfunction in diabetic vasculature through inhibition of NADPH oxidase. Nitric Oxide Biol. Chem. 2020, 96, 54–63. [Google Scholar] [CrossRef]
- Aggarwal, H.; Pathak, P.; Singh, P.; Gayen, J.R.; Jagavelu, K.; Dikshit, M. Systemic Insulin Resistance and Metabolic Perturbations in Chow Fed Inducible Nitric Oxide Synthase Knockout Male Mice: Partial Reversal by Nitrite Supplementation. Antioxidants 2020, 9, 736. [Google Scholar] [CrossRef]
- Norouzirad, R.; Gholami, H.; Ghanbari, M.; Hedayati, M.; González-Muniesa, P.; Jeddi, S.; Ghasemi, A. Dietary inorganic nitrate attenuates hyperoxia-induced oxidative stress in obese type 2 diabetic male rats. Life Sci. 2019, 230, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Torregrossa, A.C.; Potts, A.; Pierini, D.; Aranke, M.; Garg, H.K.; Bryan, N.S. Dietary nitrite improves insulin signaling through GLUT4 translocation. Free Radic. Biol. Med. 2014, 67, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Cermak, N.M.; Hansen, D.; Kouw, I.W.; van Dijk, J.W.; Blackwell, J.R.; Jones, A.M.; Gibala, M.J.; van Loon, L.J. A single dose of sodium nitrate does not improve oral glucose tolerance in patients with type 2 diabetes mellitus. Nutr. Res. 2015, 35, 674–680. [Google Scholar] [CrossRef]
- Gilchrist, M.; Winyard, P.G.; Fulford, J.; Anning, C.; Shore, A.C.; Benjamin, N. Dietary nitrate supplementation improves reaction time in type 2 diabetes: Development and application of a novel nitrate-depleted beetroot juice placebo. Nitric Oxide Biol. Chem. 2014, 40, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, M.; Winyard, P.G.; Aizawa, K.; Anning, C.; Shore, A.; Benjamin, N. Effect of dietary nitrate on blood pressure, endothelial function, and insulin sensitivity in type 2 diabetes. Free Radic. Biol. Med. 2013, 60, 89–97. [Google Scholar] [CrossRef]
- Faconti, L.; Mills, C.E. Cardiac effects of 6 months’ dietary nitrate and spironolactone in patients with hypertension and with/at risk of type 2 diabetes, in the factorial design, double-blind, randomized controlled VaSera trial. Br. J. Clin. Pharmacol. 2019, 85, 169–180. [Google Scholar] [CrossRef]
- Mills, C.E.; Govoni, V.; Faconti, L.; Casagrande, M.L.; Morant, S.V.; Crickmore, H.; Iqbal, F.; Maskell, P.; Masani, A.; Nanino, E. A randomised, factorial trial to reduce arterial stiffness independently of blood pressure: Proof of concept? The VaSera trial testing dietary nitrate and spironolactone. Br. J. Clin. Pharmacol. 2020, 86, 891–902. [Google Scholar] [CrossRef]
- Soin, A.; Bock, G.; Giordano, A.; Patel, C.; Drachman, D. A Randomized, Double-Blind Study of the Effects of a Sustained Release Formulation of Sodium Nitrite (SR-nitrite) on Patients with Diabetic Neuropathy. Pain Physician 2018, 21, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Senefeld, J.W.; Wiggins, C.C.; Regimbal, R.J.; Dominelli, P.B.; Baker, S.E.; Joyner, M.J. Ergogenic Effect of Nitrate Supplementation: A Systematic Review and Meta-analysis. Med. Sci. Sports Exerc. 2020, 52, 2250–2261. [Google Scholar] [CrossRef] [PubMed]
- Van De Walle, G.P.; Vukovich, M.D. The Effect of Nitrate Supplementation on Exercise Tolerance and Performance: A Systematic Review and Meta-Analysis. J. Strength Cond. Res. 2018, 32, 1796–1808. [Google Scholar] [CrossRef]
- Li, D.; Nishi, S.K.; Jovanovski, E.; Zurbau, A.; Komishon, A.; Mejia, S.B.; Khan, T.A.; Sievenpiper, J.L.; Milicic, D.; Jenkins, A.; et al. Repeated administration of inorganic nitrate on blood pressure and arterial stiffness: A systematic review and meta-analysis of randomized controlled trials. J. Hypertens. 2020, 38, 2122–2140. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Norouzirad, R.; Mirmiran, P.; Gaeini, Z.; Jeddi, S.; Shokri, M.; Azizi, F.; Ghasemi, A. Effect of inorganic nitrate on metabolic parameters in patients with type 2 diabetes: A 24-week randomized double-blind placebo-controlled clinical trial. Nitric Oxide Biol. Chem. 2021, 107, 58–65. [Google Scholar] [CrossRef]
- Shepherd, A.I.; Gilchrist, M.; Winyard, P.G.; Jones, A.M.; Hallmann, E.; Kazimierczak, R.; Rembialkowska, E.; Benjamin, N.; Shore, A.C.; Wilkerson, D.P. Effects of dietary nitrate supplementation on the oxygen cost of exercise and walking performance in individuals with type 2 diabetes: A randomized, double-blind, placebo-controlled crossover trial. Free Radic. Biol. Med. 2015, 86, 200–208. [Google Scholar] [CrossRef]
- Mohler, E.R., 3rd; Hiatt, W.R.; Gornik, H.L.; Kevil, C.G.; Quyyumi, A.; Haynes, W.G.; Annex, B.H. Sodium nitrite in patients with peripheral artery disease and diabetes mellitus: Safety, walking distance and endothelial function. Vasc. Med. 2014, 19, 9–17. [Google Scholar] [CrossRef]
- Greenway, F.L.; Predmore, B.L.; Flanagan, D.R.; Giordano, T.; Qiu, Y.; Brandon, A.; Lefer, D.J.; Patel, R.P.; Kevil, C.G. Single-dose pharmacokinetics of different oral sodium nitrite formulations in diabetes patients. Diabetes Technol. Ther. 2012, 14, 552–560. [Google Scholar] [CrossRef]
- Linster, C.L.; Van Schaftingen, E. Vitamin C. Biosynthesis, recycling and degradation in mammals. FEBS J. 2007, 274, 1–22. [Google Scholar] [CrossRef]
- Levine, M.; Padayatty, S.J.; Espey, M.G. Vitamin C: A concentration-function approach yields pharmacology and therapeutic discoveries. Adv. Nutr. 2011, 2, 78–88. [Google Scholar] [CrossRef] [PubMed]
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific Opinion on Dietary Reference Values for vitamin C. EFSA J. 2013, 11, 3418–3468. [Google Scholar] [CrossRef]
- Frei, B.; Traber, M.G. The new US Dietary Reference Intakes for vitamins C and E. Redox Rep. 2001, 6, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.S. Biomarkers for establishing a tolerable upper intake level for vitamin C. Nutr. Rev. 1999, 57, 71–77. [Google Scholar] [CrossRef]
- Van Gorkom, G.N.Y.; Lookermans, E.L.; Van Elssen, C.H.M.J.; Bos, G.M.J. The Effect of Vitamin C (Ascorbic Acid) in the Treatment of Patients with Cancer: A Systematic Review. Nutrients 2019, 11, 977. [Google Scholar] [CrossRef] [PubMed]
- Ashor, A.W.; Werner, A.D.; Lara, J.; Willis, N.D.; Mathers, J.C.; Siervo, M. Effects of vitamin C supplementation on glycaemic control: A systematic review and meta-analysis of randomised controlled trials. Eur. J. Clin. Nutr. 2017, 71, 1371–1380. [Google Scholar] [CrossRef]
- Mason, S.A.; Keske, M.A. Effects of Vitamin C Supplementation on Glycemic Control and Cardiovascular Risk Factors in People With Type 2 Diabetes: A GRADE-Assessed Systematic Review and Meta-analysis of Randomized Controlled Trials. Diabetes Care 2021, 44, 618–630. [Google Scholar] [CrossRef]
- Tsukaguchi, H.; Tokui, T.; Mackenzie, B.; Berger, U.V.; Chen, X.Z.; Wang, Y.; Brubaker, R.F.; Hediger, M.A. A family of mammalian Na+-dependent L-ascorbic acid transporters. Nature 1999, 399, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.J.; Johnson, D.; Jarvis, S.M. Vitamin C transport systems of mammalian cells. Mol. Membr. Biol. 2001, 18, 87–95. [Google Scholar] [CrossRef]
- Levine, M.; Conry-Cantilena, C.; Wang, Y.; Welch, R.W.; Washko, P.W.; Dhariwal, K.R.; Park, J.B.; Lazarev, A.; Graumlich, J.F.; King, J.; et al. Vitamin C pharmacokinetics in healthy volunteers: Evidence for a recommended dietary allowance. Proc. Natl. Acad. Sci. USA 1996, 93, 3704–3709. [Google Scholar] [CrossRef]
- Sage, J.M.; Carruthers, A. Human erythrocytes transport dehydroascorbic acid and sugars using the same transporter complex. Am. J. Physiol. Cell Physiol. 2014, 306, C910–C917. [Google Scholar] [CrossRef]
- Corpe, C.P.; Eck, P.; Wang, J.; Al-Hasani, H.; Levine, M. Intestinal dehydroascorbic acid (DHA) transport mediated by the facilitative sugar transporters, GLUT2 and GLUT8. J. Biol. Chem. 2013, 288, 9092–9101. [Google Scholar] [CrossRef]
- Rumsey, S.C.; Kwon, O.; Xu, G.W.; Burant, C.F.; Simpson, I.; Levine, M. Glucose transporter isoforms GLUT1 and GLUT3 transport dehydroascorbic acid. J. Biol. Chem. 1997, 272, 18982–18989. [Google Scholar] [CrossRef] [PubMed]
- Washko, P.W.; Wang, Y.; Levine, M. Ascorbic acid recycling in human neutrophils. J. Biol. Chem. 1993, 268, 15531–15535. [Google Scholar] [CrossRef]
- Padayatty, S.J.; Levine, M. Vitamin C: The known and the unknown and Goldilocks. Oral Dis. 2016, 22, 463–493. [Google Scholar] [CrossRef] [PubMed]
- Drouin, G.; Godin, J.-R.; Pagé, B. The genetics of vitamin C loss in vertebrates. Curr. Genom. 2011, 12, 371–378. [Google Scholar] [CrossRef]
- Ginter, E. Endogenous ascorbic acid synthesis and recommended dietary allowances for vitamin C. Am. J. Clin. Nutr. 1981, 34, 1448–1451. [Google Scholar] [CrossRef] [PubMed]
- Iwama, M.; Amano, A.; Shimokado, K.; Maruyama, N.; Ishigami, A. Ascorbic acid levels in various tissues, plasma and urine of mice during aging. J. Nutr. Sci. Vitaminol. 2012, 58, 169–174. [Google Scholar] [CrossRef]
- Kim, H.; Bae, S.; Yu, Y.; Kim, Y.; Kim, H.-R.; Hwang, Y.-I.; Kang, J.S.; Lee, W.J. The analysis of vitamin C concentration in organs of gulo(-/-) mice upon vitamin C withdrawal. Immune Netw. 2012, 12, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Van der Loo, B.; Bachschmid, M.; Spitzer, V.; Brey, L.; Ullrich, V.; Lüscher, T.F. Decreased plasma and tissue levels of vitamin C in a rat model of aging: Implications for antioxidative defense. Biochem. Biophys. Res. Commun. 2003, 303, 483–487. [Google Scholar] [CrossRef]
- Benjamin, N.; McKnight, G. Implications for Nitrate Intake. In Managing Risks of Nitrates to Humans and the Environment; Wilson, W.S., Ball, A.S., Hinton, R.H., Eds.; The Royal Society of Chemistry: Cambridge, UK, 1999; pp. 281–288. [Google Scholar]
- Monsen, E.R. Dietary reference intakes for the antioxidant nutrients: Vitamin C, vitamin E, selenium, and carotenoids. J. Acad. Nutr. Diet. 2000, 100, 637. [Google Scholar]
- Brubacher, D.; Moser, U.; Jordan, P. Vitamin C concentrations in plasma as a function of intake: A meta-analysis. Int. J. Vitam. Nutr. Res. 2000, 70, 226–237. [Google Scholar] [CrossRef]
- Helser, M.A.; Hotchkiss, J.H.; Roe, D.A. Influence of fruit and vegetable juices on the endogenous formation of N-nitrosoproline and N-nitrosothiazolidine-4-carboxylic acid in humans on controlled diets. Carcinogenesis 1992, 13, 2277–2280. [Google Scholar] [CrossRef]
- Kallner, A.; Hartmann, D.; Hornig, D. On the absorption of ascorbic acid in man. Int. J. Vitam. Nutr. Res. 1977, 47, 383–388. [Google Scholar]
- Kallner, A.; Hartmann, D.; Hornig, D. Steady-state turnover and body pool of ascorbic acid in man. Am. J. Clin. Nutr. 1979, 32, 530–539. [Google Scholar] [CrossRef] [PubMed]
- Tuo, B.-G.; Yan, Y.-H.; Ge, Z.-L.; Ou, G.-W.; Zhao, K. Ascorbic acid secretion in the human stomach and the effect of gastrin. World J. Gastroenterol. 2000, 6, 704–708. [Google Scholar] [CrossRef]
- Burns, J.; Mosbach, E.; Schulenberg, S. Ascorbic acid synthesis in normal and drug-treated rats, studied with L-ascorbic-l-C14 acid. J. Biol. Chem. 1954, 207, 679–687. [Google Scholar] [CrossRef]
- Kallner, A. Requirement for vitamin C based on metabolic studies. Ann. N. Y. Acad. Sci. 1987, 498, 418–423. [Google Scholar] [CrossRef]
- Corpe, C.P.; Tu, H.; Eck, P.; Wang, J.; Faulhaber-Walter, R.; Schnermann, J.; Margolis, S.; Padayatty, S.; Sun, H.; Wang, Y.; et al. Vitamin C transporter Slc23a1 links renal reabsorption, vitamin C tissue accumulation, and perinatal survival in mice. J. Clin. Investig. 2010, 120, 1069–1083. [Google Scholar] [CrossRef]
- Lundberg, J.O.N.; Weitzberg, E.; Lundberg, J.M.; Alving, K. Intragastric nitric oxide production in humans: Measurements in expelled air. Gut 1994, 35, 1543–1546. [Google Scholar] [CrossRef] [PubMed]
- Iijima, K.; Henry, E.; Moriya, A.; Wirz, A.; Kelman, A.W.; McColl, K.E.L. Dietary nitrate generates potentially mutagenic concentrations of nitric oxide at the gastroesophageal junction. Gastroenterology 2002, 122, 1248–1257. [Google Scholar] [CrossRef] [PubMed]
- Moriya, A.; Grant, J.; Mowat, C.; Williams, C.; Carswell, A.; Preston, T.; Anderson, S.; Iijima, K.; McColl, K.E. In vitro studies indicate that acid catalysed generation of N-nitrosocompounds from dietary nitrate will be maximal at the gastro-oesophageal junction and cardia. Scand. J. Gastroenterol. 2002, 37, 253–261. [Google Scholar] [CrossRef]
- Suzuki, H.; Iijima, K.; Moriya, A.; McElroy, K.; Scobie, G.; Fyfe, V.; McColl, K.E.L. Conditions for acid catalysed luminal nitrosation are maximal at the gastric cardia. Gut 2003, 52, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
- Sobala, G.M.; Schorah, C.J.; Sanderson, M.; Dixon, M.F.; Tompkins, D.S.; Godwin, P.; Axon, A.T.R. Ascorbic acid in the human stomach. Gastroenterology 1989, 97, 357–363. [Google Scholar] [CrossRef]
- Waring, A.J.; Drake, I.M.; Schorah, C.J.; White, K.L.; Lynch, D.A.; Axon, A.T.; Dixon, M.F. Ascorbic acid and total vitamin C concentrations in plasma, gastric juice, and gastrointestinal mucosa: Effects of gastritis and oral supplementation. Gut 1996, 38, 171–176. [Google Scholar] [CrossRef]
- Sobala, G.M.; Pignatelli, B.; Schorah, C.J.; Bartsch, H.; Sanderson, M.; Dixon, M.F.; Shires, S.; King, R.F.; Axon, A.T. Levels of nitrite, nitrate, N-nitroso compounds, ascorbic acid and total bile acids in gastric juice of patients with and without precancerous conditions of the stomach. Carcinogenesis 1991, 12, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Waring, A.J.; Schorah, C.J. Transport of ascorbic acid in gastric epithelial cells in vitro. Clin. Chim. Acta Int. J. Clin. Chem. 1998, 275, 137–149. [Google Scholar] [CrossRef]
- Schorah, C.J.; Sobala, G.M.; Sanderson, M.; Collis, N.; Primrose, J.N. Gastric juice ascorbic acid: Effects of disease and implications for gastric carcinogenesis. Am. J. Clin. Nutr. 1991, 53, 287s–293s. [Google Scholar] [CrossRef] [PubMed]
- Rood, J.C.; Ruiz, B.; Fontham, E.T.; Malcom, G.T.; Hunter, F.M.; Sobhan, M.; Johnson, W.D.; Correa, P. Helicobacter pylori-associated gastritis and the ascorbic acid concentration in gastric juice. Nutr. Cancer 1994, 22, 65–72. [Google Scholar] [CrossRef]
- Rathbone, B.J.; Johnson, A.W.; Wyatt, J.I.; Kelleher, J.; Heatley, R.V.; Losowsky, M.S. Ascorbic acid: A factor concentrated in human gastric juice. Clin. Sci. 1989, 76, 237–241. [Google Scholar] [CrossRef]
- Mowat, C.; Carswell, A.; Wirz, A.; McColl, K.E.L. Omeprazole and dietary nitrate independently affect levels of vitamin C and nitrite in gastric juice. Gastroenterology 1999, 116, 813–822. [Google Scholar] [CrossRef]
- Zhang, Z.W.; Patchett, S.E.; Perrett, D.; Katelaris, P.H.; Domizio, P.; Farthing, M.J.G. The relation between gastric vitamin C concentrations, mucosal histology, and CagA seropositivity in the human stomach. Gut 1998, 43, 322–326. [Google Scholar] [CrossRef]
- Muto, N.; Eguchi, R.; Akagi, Y.; Itoh, N.; Tanaka, K. Cholecystokinin stimulates ascorbic acid secretion through its specific receptor in the perfused stomach of rats. Res. Commun. Mol. Pathol. Pharmacol. 1998, 101, 127–136. [Google Scholar] [PubMed]
- Muto, N.; Ohta, T.; Suzuki, T.; Itoh, N.; Tanaka, K. Evidence for the involvement of a muscarinic receptor in ascorbic acid secretion in the rat stomach. Biochem. Pharmacol. 1997, 53, 553–559. [Google Scholar] [CrossRef]
- Breidenbach, A.W.; Cambel, P.; Ray, F.E. Gastric Ascorbic Acid in the Gastritic Rat. Proc. Soc. Exp. Biol. Med. 1952, 80, 144–146. [Google Scholar] [CrossRef]
- Licht, W.R.; Tannenbaum, S.R.; Deen, W.M. Use of ascorbic acid to inhibit nitrosation: Kinetic and mass transfer considerations for an in vitro system. Carcinogenesis 1988, 9, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Mowat, C.; McColl, K.E.L. Alterations in intragastric nitrite and vitamin C levels during acid inhibitory therapy. Best Pract. Res. Clin. Gastroenterol. 2001, 15, 523–537. [Google Scholar] [CrossRef]
- Okazaki, K.; Ishii, Y.; Kitamura, Y.; Maruyama, S.; Umemura, T.; Miyauchi, M.; Yamagishi, M.; Imazawa, T.; Nishikawa, A.; Yoshimura, Y. Dose-dependent promotion of rat forestomach carcinogenesis by combined treatment with sodium nitrite and ascorbic acid after initiation with N-methyl-N′-nitro-N-nitrosoguanidine: Possible contribution of nitric oxide-associated oxidative DNA damage. Cancer Sci. 2006, 97, 175–182. [Google Scholar] [CrossRef] [PubMed]
- McLennan, S.; Yue, D.K.; Fisher, E.; Capogreco, C.; Heffernan, S.; Ross, G.R.; Turtle, J.R. Deficiency of Ascorbic Acid in Experimental Diabetes: Relationship With Collagen and Polyol Pathway Abnormalities. Diabetes 1988, 37, 359–361. [Google Scholar] [CrossRef]
- Wilson, R.; Willis, J.; Gearry, R.; Skidmore, P.; Fleming, E.; Frampton, C.; Carr, A. Inadequate Vitamin C Status in Prediabetes and Type 2 Diabetes Mellitus: Associations with Glycaemic Control, Obesity, and Smoking. Nutrients 2017, 9, 997. [Google Scholar] [CrossRef]
- Christie-David, D.; Gunton, J. Vitamin c deficiency and diabetes mellitus-easily missed? Diabet. Med. A J. Br. Diabet. Assoc. 2017, 34, 294. [Google Scholar] [CrossRef]
- Lysy, J.; Zimmerman, J. Ascorbic acid status in diabetes mellitus. Nutr. Res. 1992, 12, 713–720. [Google Scholar] [CrossRef]
- Seghieri, G.; Martinoli, L.; di Felice, M.; Anichini, R.; Fazzini, A.; Ciuti, M.; Miceli, M.; Gaspa, L.; Franconi, F. Plasma and platelet ascorbate pools and lipid peroxidation in insulin-dependent diabetes mellitus. Eur. J. Clin. Investig. 1998, 28, 659–663. [Google Scholar] [CrossRef] [PubMed]
- Som, S.; Basu, S.; Mukherjee, D.; Deb, S.; Choudhury, P.R.; Mukherjee, S.; Chatterjee, S.N.; Chatterjee, I.B. Ascorbic acid metabolism in diabetes mellitus. Metab. Clin. Exp. 1981, 30, 572–577. [Google Scholar] [CrossRef]
- Sinclair, A.; Girling, A.; Gray, L.; Le Guen, C.; Lunec, J.; Barnett, A. Disturbed handling of ascorbic acid in diabetic patients with and without microangiopathy during high dose ascorbate supplementation. Diabetologia 1991, 34, 171–175. [Google Scholar] [CrossRef]
- Du, Y.T.; Rayner, C.K.; Jones, K.L.; Talley, N.J.; Horowitz, M. Gastrointestinal Symptoms in Diabetes: Prevalence, Assessment, Pathogenesis, and Management. Diabetes Care 2018, 41, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Devrajani, B.R.; Shah, S.Z.; Soomro, A.A.; Devrajani, T. Type 2 diabetes mellitus: A risk factor for Helicobacter pylori infection: A hospital based case-control study. Int. J. Diabetes Dev. Ctries. 2010, 30, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Boehme, M.W.; Autschbach, F.; Ell, C.; Raeth, U. Prevalence of silent gastric ulcer, erosions or severe acute gastritis in patients with type 2 diabetes mellitus—A cross-sectional study. Hepato Gastroenterol. 2007, 54, 643–648. [Google Scholar]
- Hasler, W.L.; Coleski, R.; Chey, W.D.; Koch, K.L.; McCallum, R.W.; Wo, J.M.; Kuo, B.; Sitrin, M.D.; Katz, L.A.; Hwang, J.; et al. Differences in intragastric pH in diabetic vs. idiopathic gastroparesis: Relation to degree of gastric retention. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 294, G1384–G1391. [Google Scholar] [CrossRef]
- Freeman, J.T.; Hafkesbring, R. Comparative studies of ascorbic acid levels in gastric secretion and blood. III. Gastrointestinal diseases. Gastroenterology 1957, 32, 878–886. [Google Scholar] [CrossRef]
- Ashor, A.W.; Shannon, O.M.; Werner, A.-D.; Scialo, F.; Gilliard, C.N.; Cassel, K.S.; Seal, C.J.; Zheng, D.; Mathers, J.C.; Siervo, M. Effects of inorganic nitrate and vitamin C co-supplementation on blood pressure and vascular function in younger and older healthy adults: A randomised double-blind crossover trial. Clin. Nutr. 2020, 39, 708–717. [Google Scholar] [CrossRef]
- Bednar, C.; Kies, C. Nitrate and vitamin C from fruits and vegetables: Impact of intake variations on nitrate and nitrite excretions of humans. Plant Foods Hum. Nutr. 1994, 45, 71–80. [Google Scholar] [CrossRef] [PubMed]
| Author | Model | Treatment | Outcomes |
|---|---|---|---|
| Jeddi et al., 2021 [62] | High-fat diet + low-dose of STZ (30 mg/kg body weight), male rats | 100 mg/L NaNO3 in drinking water for 6 months | ↓ Serum glucose by 13% ↓ Serum insulin by 23% ↑ cGMP level in epididymal adipose tissue by 85% ↑ Adipocyte density by 193% (epididymal adipose tissue) ↓ Adipocyte area by 53% (epididymal adipose tissue) ↑ Expression of browning genes in epididymal adipose tissue (↑ mRNA and protein levels of PPAR-γ, PGC1-α, and UCP-1 to their normal values) |
| Tian et al., 2020 [63] | High-fat diet + low dose of STZ (20 mg/kg body weight), male mice | 255 mg/L NaNO3 in drinking water for 8 weeks | ↓ Fasting glucose Prevention of impaired glucose tolerance (measured by IP-GTT), Prevention of insulin resistance (measured by IP-ITT) ↓ Systolic blood pressure ↓Vascular oxidative stress (↓ROS formation) ↓ NADPH oxidase activity via induction of HO-1 and reduction in p47phox expression Improvement of endothelial function (ACh-mediated vascular relaxation) Improvement of inflammation and dyslipidemia ↓ Development of aortic atherosclerosis |
| Aggarwal et al., 2020 [64] | Insulin-resistant iNOS−/− male mice | 50 mg/L NaNO2 in drinking water for 5 weeks | Improved glucose tolerance (measured by IP-GTT) Improved insulin resistance (measured by IP-ITT) Partially reversed up-regulated gluconeogenesis (↓ expression of PEPCK, G6P, and PC) Restored total Akt (PKB) expression in the liver and adipose tissue Restored decreased Akt-1/2/3 phosphorylation (Ser473) in the liver Improved insulin signaling in the adipose tissue |
| Norouzirad et al., 2019 [65] | High-fat diet + low dose of STZ (30 mg/kg body weight), male rats | 100 mg/L NaNO3 in drinking water for 5 weeks | ↓ Fasting glucose ↓ Gluconeogenesis (measured by IP-PTT) Improved glucose tolerance Restored CAT activity to near normal value Restored elevated TOS to near normal value Restored decreased TAC levels to near normal value ↑ Serum SOD, GSH, and GSH-to-GSSG ratio |
| Gheibi et al., 2018 [6] | High-fat diet + low dose of STZ (25 mg/g body weight), male rats | 100 mg/L NaNO3 in drinking water for 8 weeks | ↓ Serum glucose and insulin, ↔ HbA1c ↑ Glucose tolerance (measured by IP-GTT) ↑ Insulin sensitivity (measured by QUICKI) ↓ Gluconeogenesis (measured by IP-PTT) ↑ GLUT4 mRNA expression and protein levels in the soleus muscle by 215% and 17% ↑ GLUT4 mRNA expression and protein levels in the epididymal adipose tissue by 344% and 22% ↔ GSIS, islet insulin content ↑ Serum CAT activity, ↓ Serum IL-1β ↔ Serum TBARS ↓ Elevated iNOS mRNA expression in the soleus muscle and epididymal adipose tissue |
| Gheibi et al., 2017 [7] | High-fat diet + low dose of STZ (30 mg/kg body weight), male rats | 50 mg/L NaNO2 in drinking water for 8 weeks | ↑ GSIS (by 34%), ↔ BIS ↑ Protein levels of GLUT4 in the soleus muscle and epididymal adipose tissue by 22% and 26% Improved glucose tolerance (measured by IP-GTT) and insulin sensitivity (measured by IP-ITT and QUICKI) ↓ Insulin resistance (measured by HOMA-IR) ↓ Fasting serum glucose and insulin, ↔ HbA1c Restored pancreatic insulin content to 73% of controls (68.2 ± 6.4 vs. 117 ± 6.0 pmol/mg protein) Restored elevated serum levels of TC, TG, and LDL-C ↔ HDL-C |
| Ohtake et al., 2015 [9] | KKAy diabetic male mice | 50 and 150 mg/L nitrite in drinking water for 10 weeks | ↓ Fasting glucose ↓ Insulin resistance (measured by HOMA-IR) Improved glucose tolerance (measured by IP-GTT) ↑GLUT4 expression on the cell membrane of the skeletal muscle |
| Khalifi et al., 2015 [8] | STZ (65 mg/kg) + nicotinamide (95 mg/kg), male rats | 100 mg/L NaNO3 in drinking water for 8 weeks | Improved glucose tolerance (measured as IV-GTT) ↓ Serum TC (23.6%), TG (24.2%), and LDL-C (28.8%) ↑ Serum HDL-C (42.4%) Restored TAC and CAT levels to normal values |
| Jiang et al., 2014 [66] | db/db diabetic male mice | 50 mg/L NaNO2 in drinking water for 4 weeks | ↓ Fasting glucose (by 35%) ↓ Plasma insulin |
| Carlstrom et al., 2010 [10] | eNOS-deficient female mice | 85 mg/L NaNO3 in drinking water for 8–10 weeks | ↓ HbA1c, Fasting glucose ↓ Pro-insulin to insulin ratio ↑ Glucose tolerance (measured by IP-GTT) |
| Study | Intervention | Outcomes |
|---|---|---|
| Bahadoran et al., 2021 [76] | NO3−-rich beetroot powder (250 mg/day NO3−), for 24 weeks | ↔ Fasting glucose, HbA1c, insulin, C-peptide ↔ HOMA-IR, QUICKI ↔ Serum lipid parameters ↔ Serum ALT, AST, ALP, GGT ↔ Serum creatinine and uric acid ↔ Urinary creatinine and albumin |
| Faconti et al., 2019 [70] and Mills et al. [71] | NO3−-containing beetroot juice (279 mg/day NO3−), for 24 weeks | ↔ SBP, DBP ↔ Arterial stiffness ↔ Fasting glucose, HbA1c ↓ Left ventricular end-diastolic and end-systolic volume |
| Soin et al., 2018 [72] | 40 and 80 mg/day sustained-release formulation NaNO2, for 12 weeks | ↔ HbA1c Improvement of neuropathic pain |
| Shepherd et al., 2015 [77] | 70 mL/day NO3−-containing beetroot juice (398 mg/day NO3−), for 4 days | ↔ SBP, DBP ↔ Oxygen cost of exercise ↔ Walking performance (6-min walk test) |
| Cermak et al., 2015 [67] | An acute dose of NaNO3 (12.75 mg/kg body weight) | ↔ Postprandial glucose and insulin response to 75-g glucose ↑ OGIS index ↔ HOMA-IR |
| Mohler et al., 2014 [78] | 40 and 80 mg/day NaNO2, for 10 weeks | ↑ FMD at dose of 80 mg/day |
| Gilchrist et al., 2014 [68] | 250 mL/day beetroot juice (465 mg/d NO3−), for 2 weeks | ↔ Fasting glucose, HbA1c ↔ Cognitive function Improvement in simple reaction time |
| Gilchrist et al., 2013 [69] | 250 mL/day beetroot juice (465 mg/d NO3−), for 2 weeks | ↔ SBP, DBP ↔ Macro-(FMD) and micro-(ACh-induced vasodilation) vascular function ↔ Insulin sensitivity (hyperinsulinemic-euglycemic clamp technique) |
| Greenway et al., 2012 [79] | An acute dose of 80 mg of NaNO2 (IR and EC formulation) | ↓ SPB and DBP in IR ↔ SPB and DBP in EC |
| Parameter | Human [81,82,83,84,90,102,103,104,105,106,107] | Rat [100,108,109] | Mouse [98,99,110] |
|---|---|---|---|
| Sources of AA | Dietary intake | Glycogen catabolism | Glycogen catabolism |
| Endogenous production rate (mg/day) | 0 | 6–9 | 12.5 |
| Exogenous requirement (mg/day) | To prevent scurvy = 60 To maintain plasma AA > 50 µmol/L = 100 RDA = 75 and 90 for adult women and men To prevent formation of harmful nitrosamines = 200 UL = 2000–6000 | 0 | 0 |
| Absorption rate of exogenous sources | 70–90% (dependent to ingested amounts) | – | – |
| Body pool (mg/100 g) | 2 | 9–12 | 12–15 |
| Fractional turnover (% of body pool catabolized daily) | 3 | 24–29 | 60–90 |
| Urinary excretion | 25% of intake (10–87% dependent to ingested amounts) | 13–17% of synthesized value (0.33–0.46 mg/100 g/day) | 10–17% in male (0.4–0.6 mg/day) 5–8% in female (0.2–0.3 mg/day) |
| Plasma concentration (µmol/L) | 50 (range 30–90) | 680 | 60–90 |
| Mechanisms of tissue uptake | SVCT1 and SVCT2 | SVCT1 and SVCT2 | SVCT1 and SVCT2 |
| Gastric secretion of AA (mg/day) | 60 | Basal = 0.018–0.040; Carbachol-induced = 0.28 ± 0.17 | – |
| Unknown mechanisms | Active secretion regulated by muscarinic receptor-associated cholinergic stimulation and CCK receptor-associated humoral stimulation | – | |
| Intragastric concentration | 20–80 µmol/L | 190–340 µmol/L in gastric juice (1260 and 658 µmol/100 g, in the glandular stomach and the forestomach) | –— |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bahadoran, Z.; Mirmiran, P.; Kashfi, K.; Ghasemi, A. Lost-in-Translation of Metabolic Effects of Inorganic Nitrate in Type 2 Diabetes: Is Ascorbic Acid the Answer? Int. J. Mol. Sci. 2021, 22, 4735. https://doi.org/10.3390/ijms22094735
Bahadoran Z, Mirmiran P, Kashfi K, Ghasemi A. Lost-in-Translation of Metabolic Effects of Inorganic Nitrate in Type 2 Diabetes: Is Ascorbic Acid the Answer? International Journal of Molecular Sciences. 2021; 22(9):4735. https://doi.org/10.3390/ijms22094735
Chicago/Turabian StyleBahadoran, Zahra, Parvin Mirmiran, Khosrow Kashfi, and Asghar Ghasemi. 2021. "Lost-in-Translation of Metabolic Effects of Inorganic Nitrate in Type 2 Diabetes: Is Ascorbic Acid the Answer?" International Journal of Molecular Sciences 22, no. 9: 4735. https://doi.org/10.3390/ijms22094735
APA StyleBahadoran, Z., Mirmiran, P., Kashfi, K., & Ghasemi, A. (2021). Lost-in-Translation of Metabolic Effects of Inorganic Nitrate in Type 2 Diabetes: Is Ascorbic Acid the Answer? International Journal of Molecular Sciences, 22(9), 4735. https://doi.org/10.3390/ijms22094735







