The Role of LGR4 (GPR48) in Normal and Cancer Processes
Abstract
1. Introduction
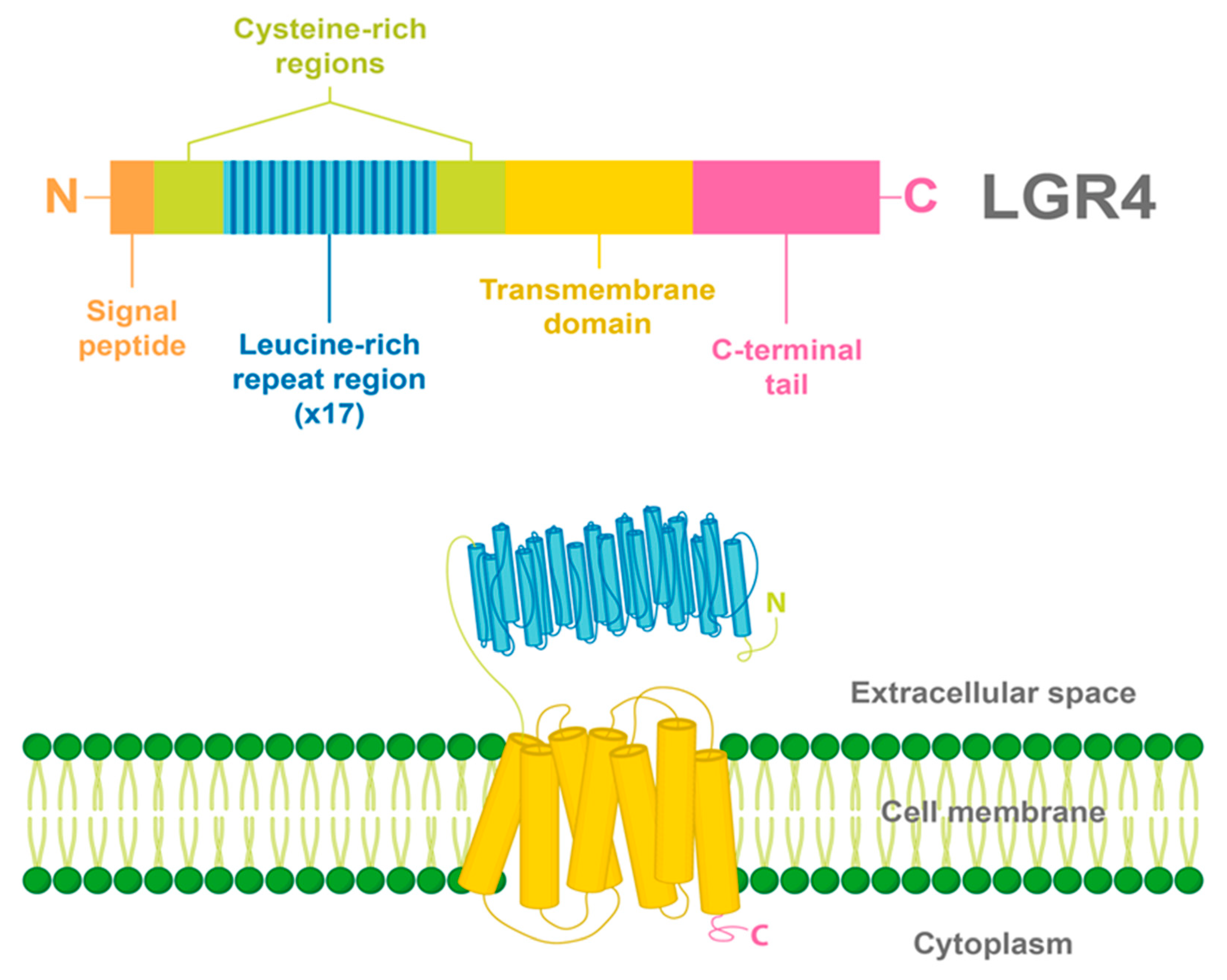
2. LGR4 Characterization
3. Ligands and Canonical Signaling Pathways Regulated by LGR4
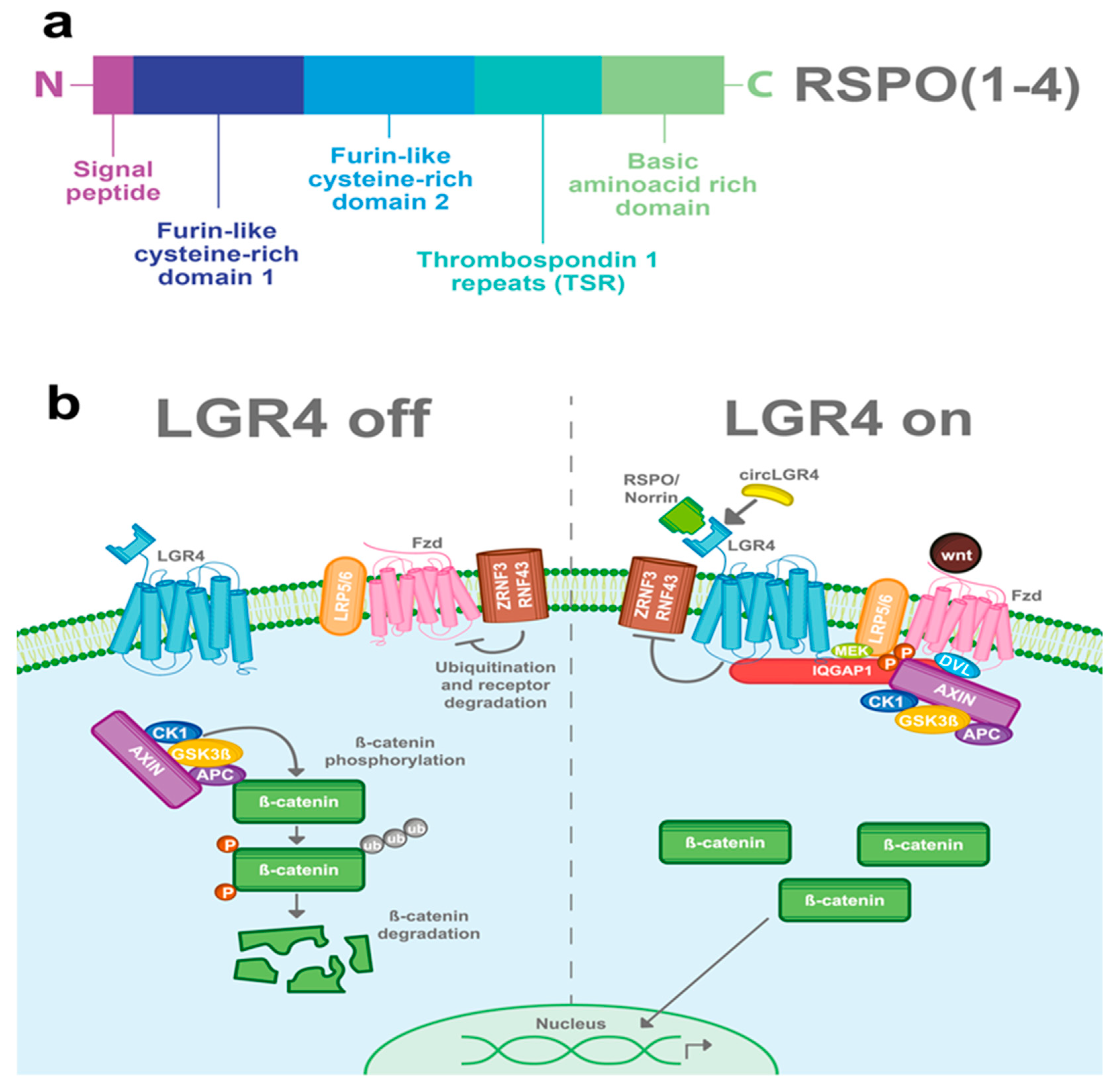
3.1. R-Spondins (RSPOs)
3.2. Norrin
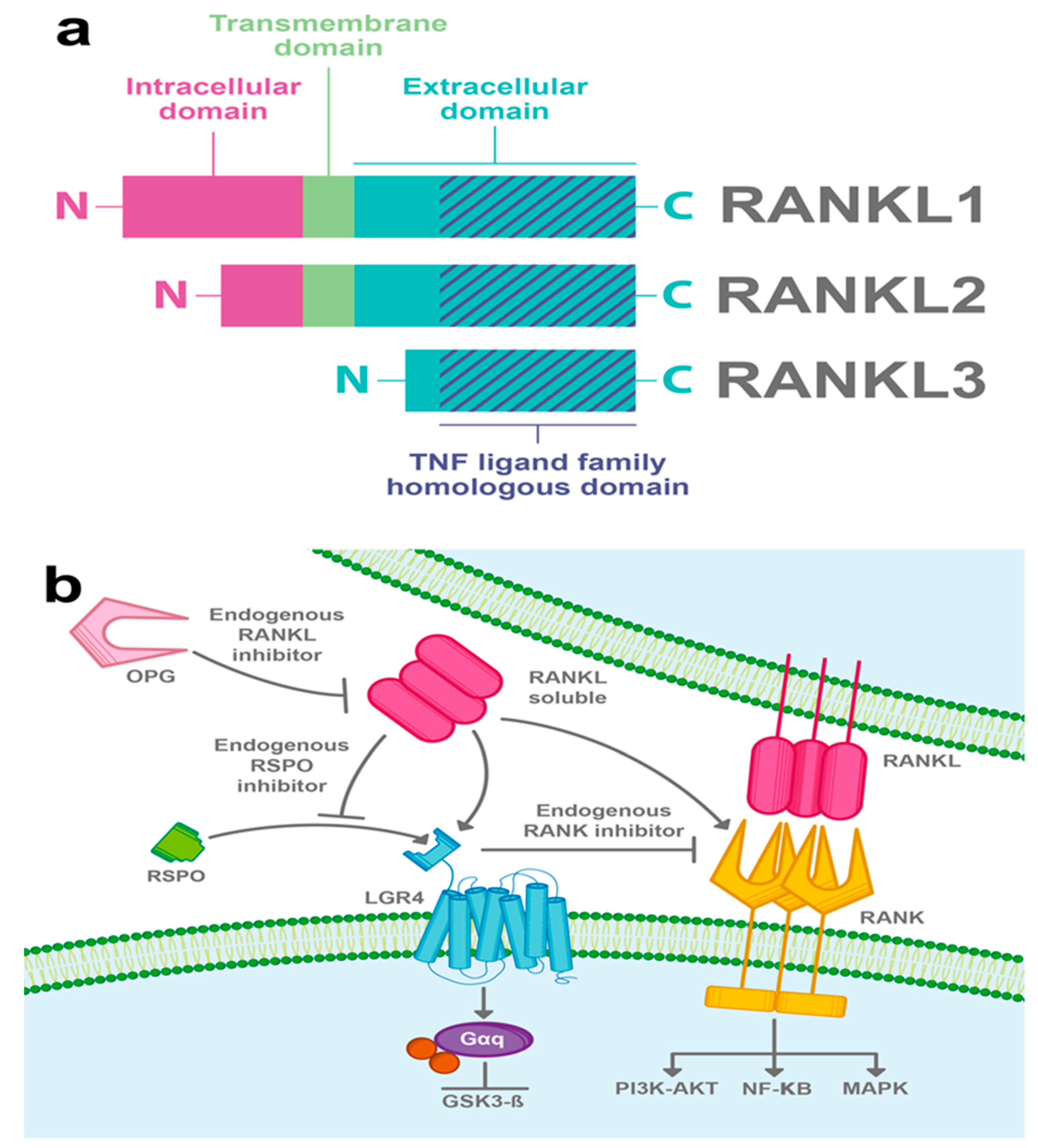
3.3. RANKL
3.4. circLGR4
4. LGR4 Regulation through microRNAs
5. LGR4 in Normal Tissues
5.1. Male Reproductive Tract
5.2. Female Reproductive Tract
5.3. Eye
5.4. Intestine
5.5. Mammary Gland
5.6. Kidney
5.7. Liver
5.8. Epidermis
5.9. Teeth
5.10. Bone
5.11. Additional Functions
6. LGR4 in Cancer
6.1. Breast Cancer
6.2. Colorectal Cancer
6.3. Lung Cancer
6.4. Oral Cancer
6.5. Prostate Cancer
6.6. Skin Cancer
6.7. Other Cancers
7. LGR4 Function in Cancer Stem Cells
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ALDH | Aldehyde dehydrogenase |
| AML | Acute myeloid leukemia |
| APC | Adenomatous polyposis coli |
| ATF4 | Activating transcription factor 4 |
| BMP | Bone morphogenetic protein |
| cAMP | Cyclic adenosine monophosphate |
| CAT | Catalase |
| CK1 | Casein kinase 1 |
| CREB | cAMP response element-binding protein |
| CSCs | Cancer stem cells |
| DSS | Dextran sodium sulfate |
| DVL | Dishevelled |
| EDAR | Ectodysplasin A receptor |
| EGFR | Epidermal growth factor receptor |
| ELF3 | E74 like ETS transcription factor 3 |
| EMT | Epithelial-mesenchymal transition |
| ERK | Extracellular signal regulated kinase |
| ESR1 | Estrogen receptor 1 |
| FAK | Focal adhesion kinase |
| FSH | Follicle-stimulating hormone |
| FZD | Frizzled |
| GPCR | G protein-coupled receptor |
| GSK3β | Glycogen synthase kinase 3 beta |
| HB-EGF | Heparin-binding EGF-like growth factor |
| IL-6 | Interleukin 6 |
| INSL3 | Insulin-like peptide 3 |
| IQGAP1 | IQ motif containing GTPase activating protein 1 |
| JMJD2A | Jumonji domain-containing protein 2A |
| LEF1 | Lymphoid enhancer binding factor 1 |
| LGRs | Leucine-rich repeats containing G protein-coupled receptors |
| LH | Luteinizing hormone |
| LIF | Lymphoid enhancer-binding factor 1 |
| LRP | Low-density lipoprotein receptor-related protein |
| LRR | Leucin-rich repeats |
| MAPK | Mitogen-activated protein kinase |
| MMP2 | Matrix metalloproteinase 2 |
| NFATC1 | Nuclear factor of activated T-cells, cytoplasmic 1 |
| NF-κB | Nuclear factor of activated T-cells, cytoplasmic 1 |
| OLFM4 | Olfactomedin 4 |
| OPG | Osteoprotegerin |
| PDK | Polycistic kidney disease |
| PI3K | Phosphatidylinositol 3-kinase |
| PITX2 | Paired Like homeodomain 2 |
| PR | Progesterone receptor |
| PSA | Prostate-specific antigen |
| RANK | Receptor activator of NF-kappa B |
| RANKL | Receptor activator of NF-kappa B ligant |
| RNF43 | Ring finger protein 43 |
| RSPOs | R-spondins |
| RXFP | Relaxin family pepetide receptor |
| SHH | Sonic hedgehog |
| SOD1 | Superoxide dismutase-1 |
| STAT3 | Signal transucer and activator of transcription 3 |
| TAMs | Tumor-associated macrophages |
| TCF | T-cell factor |
| TGF-β | Transforming growth factor beta |
| TLR | Toll-like receptor |
| TNF | Tumor necrosis factor |
| TSH | Thyroid-stimulating hormone |
| TSR | Thrombospondin 1 repeat domain |
| ZNRF3 | Zinc and ring finger |
References
- Weinberg, R.A. The Biology of Cancer, 2nd ed.; Garland Science, Taylor and Francis Group: New York, NY, USA; London, UK, 2013; ISBN 9780429258794. [Google Scholar]
- Krauss, G. Biochemistry of Signal Transduction and Regulation; Wiley: Hoboken, NJ, USA, 2003; ISBN 9783527601868. [Google Scholar]
- Bar-Shavit, R.; Maoz, M.; Kancharla, A.; Nag, J.K.; Agranovich, D.; Grisaru-Granovsky, S.; Uziely, B. G protein-coupled receptors in cancer. Int. J. Mol. Sci. 2016, 17, 1320. [Google Scholar] [CrossRef] [PubMed]
- Van Loy, T.; Vandersmissen, H.P.; Van Hiel, M.B.; Poels, J.; Verlinden, H.; Badisco, L.; Vassart, G.; Vanden Broeck, J. Comparative genomics of leucine-rich repeats containing G protein-coupled receptors and their ligands. Gen. Comp. Endocrinol. 2008, 155, 14–21. [Google Scholar] [CrossRef]
- Petrie, E.J.; Lagaida, S.; Sethi, A.; Bathgate, R.A.D.; Gooley, P.R. In a class of their own-RXFP1 and RXFP2 are unique members of the LGR family. Front. Endocrinol. 2015, 6, 137. [Google Scholar] [CrossRef]
- Carmon, K.S.; Gong, X.; Lin, Q.; Thomas, A.; Liu, Q. R-spondins function as ligands of the orphan receptors LGR4 and LGR5 to regulate Wnt/β-catenin signaling. Proc. Natl. Acad. Sci. USA 2011, 108, 11452–11457. [Google Scholar] [CrossRef]
- Ruffner, H.; Sprunger, J.; Charlat, O.; Leighton-Davies, J.; Grosshans, B.; Salathe, A.; Zietzling, S.; Beck, V.; Therier, M.; Isken, A.; et al. R-spondin potentiates Wnt/β-Catenin signaling through orphan receptors LGR4 and LGR5. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Deng, C.; Reddy, P.; Cheng, Y.; Luo, C.-W.; Hsiao, C.-L.; Hsueh, A.J.W. Multi-functional norrin is a ligand for the LGR4 receptor. J. Cell Sci. 2013, 126, 2060–2068. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Yang, Z.; Ma, Y.; Yue, Z.; Lin, H.; Qu, G.; Huang, J.; Dai, W.; Li, C.; Zheng, C.; et al. LGR4 is a receptor for RANKL and negatively regulates osteoclast differentiation and bone resorption. Nat. Med. 2016, 22, 539–546. [Google Scholar] [CrossRef]
- Zhi, X.; Zhang, J.; Cheng, Z.; Bian, L.; Qin, J. circLgr4 drives colorectal tumorigenesis and invasion through Lgr4-targeting peptide. Int. J. Cancer 2019. [Google Scholar] [CrossRef] [PubMed]
- Glinka, A.; Dolde, C.; Kirsch, N.; Huang, Y.L.; Kazanskaya, O.; Ingelfinger, D.; Boutros, M.; Cruciat, C.M.; Niehrs, C. LGR4 and LGR5 are R-spondin receptors mediating Wnt/β-catenin and Wnt/PCP signalling. EMBO Rep. 2011, 12, 1055–1061. [Google Scholar] [CrossRef]
- Hsu, S.Y.; Liang, S.-G.; Hsueh, A.J.W. Characterization of Two LGR Genes Homologous to Gonadotropin and Thyrotropin Receptors with Extracellular Leucine-Rich Repeats and a G Protein-Coupled, Seven-Transmembrane Region. Mol. Endocrinol. 1998, 12, 1830–1845. [Google Scholar] [CrossRef]
- Loh, E.D.; Broussard, S.R.; Liu, Q.; Copeland, N.G.; Gilbert, D.J.; Jenkins, N.A.; Kolakowski, L.F., Jr. Chromosomal localization of GPR48, a novel glycoprotein hormone receptor like GPCR, in human and mouse with radiation hybrid and interspecific backcross mapping. Cytogenet. Genome Res. 2000, 89, 2–5. [Google Scholar] [CrossRef]
- Loh, E.D.; Broussard, S.R.; Kolakowski, L.F. Molecular characterization of a novel glycoprotein hormone G-protein-coupled receptor. Biochem. Biophys. Res. Commun. 2001, 282, 757–764. [Google Scholar] [CrossRef]
- Yi, J.; Xiong, W.; Gong, X.; Bellister, S.; Ellis, L.M.; Liu, Q. Analysis of LGR4 Receptor Distribution in Human and Mouse Tissues. PLoS ONE 2013, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.R.; Yoon, J.K. The R-spondin family of proteins: Emerging regulators of WNT signaling. Int. J. Biochem. Cell Biol. 2012, 100, 130–134. [Google Scholar] [CrossRef]
- Raslan, A.A.; Yoon, J.K. R-spondins: Multi-mode WNT signaling regulators in adult stem cells. Int. J. Biochem. Cell Biol. 2019, 106, 26–34. [Google Scholar] [CrossRef]
- Wang, D.; Huang, B.; Zhang, S.; Yu, X.; Wu, W.; Wang, X. Structural basis for R-spondin receptors. Genes Dev. 2013, 1339–1344. [Google Scholar] [CrossRef] [PubMed]
- De Lau, W.; Barker, N.; Low, T.Y.; Koo, B.; Li, V.S.W.; Teunissen, H.; Kujala, P.; Haegebarth, A.; Peters, P.J.; Van De Wetering, M.; et al. Lgr5 homologues associate with Wnt receptors and mediate R-spondin signalling. Nature 2011, 476, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.X.; Xie, Y.; Zhang, Y.; Zhang, O.; Oster, E.; Avello, M.; Lei, H.; Mickanin, C.; Liu, D.; Ruffner, H.; et al. ZNRF3 promotes Wnt receptor turnover in an R-spondin-sensitive manner. Nature 2012, 485, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.C.; De Lau, W.; Madoori, P.K.; Forneris, F.; Granneman, J.C.M.; Clevers, H.; Gros, P. Structures of Wnt-Antagonist ZNRF3 and Its Complex with R-Spondin 1 and Implications for Signaling. PLoS ONE 2013, 8, 1–10. [Google Scholar] [CrossRef]
- Xie, Y.; Zamponi, R.; Charlat, O.; Ramones, M.; Swalley, S.; Jiang, X.; Rivera, D.; Tschantz, W.; Lu, B.; Quinn, L.; et al. Interaction with both ZNRF3 and LGR4 is required for the signalling activity of R-spondin. EMBO Rep. 2013, 14, 1120–1126. [Google Scholar] [CrossRef]
- Moad, H.E.; Pioszak, A.A. Reconstitution of R-spondin:LGR4:ZNRF3 adult stem cell growth factor signaling complexes with recombinant proteins produced in escherichia coli. Biochemistry 2013, 52, 7295–7304. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, W.; Mulholland, M.W. LGR4 and Its Role in Intestinal Protection and Energy Metabolism. Front. Endocrinol. 2015, 6, 1–9. [Google Scholar] [CrossRef]
- Carmon, K.S.; Gong, X.; Yi, J.; Thomas, A.; Liu, Q. RSPO-LGR4 functions via IQGAP1 to potentiate Wnt signaling. Proc. Natl. Acad. Sci. USA 2014, 111. [Google Scholar] [CrossRef]
- Komiya, Y.; Habas, R. Wnt signal transduction pathways. Organogenesis 2008, 4, 68–75. [Google Scholar] [CrossRef]
- Duchartre, Y.; Kim, Y.-M.; Kahn, M. The Wnt signaling pathway in cancer. Crit. Rev. Oncol. Hematol. 2016, 99, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Braunger, B.M.; Tamm, E.R. The Different Functions of Norrin. In Advances in Experimental Medicine and Biology; Springer: Berlin/Heidelberg, Germany, 2012; Volume 801, pp. 679–683. ISBN 978-1-4614-0630-3. [Google Scholar]
- Renema, N.; Navet, B.; Heymann, M.-F.; Lezot, F.; Heymann, D. RANK-RANKL signalling in cancer. Biosci. Rep. 2016, 36, e00366. [Google Scholar] [CrossRef]
- Ikeda, T.; Kasai, M.; Utsuyama, M.; Hirokawa, K. Determination of three isoforms of the receptor activator of nuclear factor-κB ligand and their differential expression in bone and thymus. Endocrinology 2001, 142, 1419–1426. [Google Scholar] [CrossRef]
- Saliminejad, K.; Khorram Khorshid, H.R.; Soleymani Fard, S.; Ghaffari, S.H. An overview of microRNAs: Biology, functions, therapeutics, and analysis methods. J. Cell. Physiol. 2019, 234, 5451–5465. [Google Scholar] [CrossRef]
- Cong, F.; Wu, N.; Tian, X.; Fan, J.; Liu, J.; Song, T.; Fu, H. MicroRNA-34c promotes osteoclast differentiation through targeting LGR4. Gene 2017, 610, 1–8. [Google Scholar] [CrossRef]
- Wu, J.; Li, X.; Li, D.; Ren, X.; Li, Y.; Herter, E.K.; Qian, M.; Toma, M.A.; Wintler, A.M.; Sérézal, I.G.; et al. MicroRNA-34 Family Enhances Wound Inflammation by Targeting LGR4. J. Investig. Dermatol. 2020, 140, 465–476.e11. [Google Scholar] [CrossRef]
- Hou, Q.; Zhou, L.; Tang, J.; Ma, N.; Xu, A.; Tang, J.; Zheng, D.; Chen, X.; Chen, F.; Dong, X.D.; et al. LGR4 Is a Direct Target of MicroRNA-34a and modulates the proliferation and migration of retinal pigment epithelial ARPE-19 Cells. PLoS ONE 2016, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xu, X. MicroRNA-137 dysregulation predisposes to osteoporotic fracture by impeding ALP activity and expression via suppression of leucine-rich repeat-containing G-protein-coupled receptor 4 expression. Int. J. Mol. Med. 2018, 42, 1026–1033. [Google Scholar] [CrossRef]
- Dong, Y.; Zheng, Y.; Xiao, J.; Zhu, C.; Zhao, M. MicroRNA let-7b induces lens epithelial cell apoptosis by targeting leucine-rich repeat containing G protein-coupled receptor 4 (Lgr4) in age-related cataract. Exp. Eye Res. 2016, 147, 98–104. [Google Scholar] [CrossRef]
- Li, F.; Gu, C.; Tian, F.; Jia, Z.; Meng, Z. miR-218 impedes IL-6-induced prostate cancer cell proliferation and invasion via suppression of LGR4 expression. Oncol. Rep. 2016, 35, 2859–2865. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liang, F.; Yue, J.; Liu, P.; Wang, J.; Wang, Z.; Li, H.; Cheng, D.; Du, J.; Zhang, K.; et al. MicroRNA-137 regulates hypoxia-mediated migration and epithelial.mesenchymal transition in prostate cancer by targeting LGR4 via the EGFR/ERK signaling pathway. Int. J. Oncol. 2020, 57, 540–549. [Google Scholar] [CrossRef]
- Yang, D.; Li, J.; Xu, Q.; Xia, T.; Xia, J. Inhibitory Effect of MiR-449b on Cancer Cell Growth and Invasion through LGR4 in Non-Small-Cell Lung Carcinoma. Curr. Med. Sci. 2018, 38, 582–589. [Google Scholar] [CrossRef]
- Mazerbourg, S.; Bouley, D.M.; Sudo, S.; Klein, C.A.; Zhang, J.V.; Kawamura, K.; Goodrich, L.V.; Rayburn, H.; Tessier-Lavigne, M.; Hsueh, A.J.W. Leucine-Rich Repeat-Containing, G Protein-Coupled Receptor 4 Null Mice Exhibit Intrauterine Growth Retardation Associated with Embryonic and Perinatal Lethality. Mol. Endocrinol. 2004, 18, 2241–2254. [Google Scholar] [CrossRef] [PubMed]
- Mendive, F.; Laurent, P.; Van Schoore, G.; Skarnes, W.; Pochet, R.; Vassart, G. Defective postnatal development of the male reproductive tract in LGR4 knockout mice. Dev. Biol. 2006, 290, 421–434. [Google Scholar] [CrossRef]
- Hoshii, T.; Takeo, T.; Nakagata, N.; Takeya, M.; Araki, K.; Yamamura, K. LGR4 Regulates the Postnatal Development and Integrity of Male Reproductive Tracts in Mice1. Biol. Reprod. 2007, 76, 303–313. [Google Scholar] [CrossRef]
- Lambot, M.H.; Mendive, F.; Laurent, P.; Schoore, G.V.A.N.; Noe, J.; Vanderhaeghen, P.; Vassart, G. Three-Dimensional Reconstruction of Efferent Ducts in Wild-Type and Lgr4 Knock-Out Mice. Anat. Rec. Adv. Integr. Anat. Evol. Biol. 2009, 603, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Liu, S.; Guan, Y.; Pan, H.; Guan, X.; Qiu, Z.; Li, L.; Gao, N.; Zhao, Y.; Li, X.; et al. Lgr4-mediated Wnt/β -catenin signaling in peritubular myoid cells is essential for spermatogenesis. Development 2013, 1761, 1751–1761. [Google Scholar] [CrossRef]
- Luo, W.; Rodriguez, M.; Valdez, J.M.; Zhu, X.; Tan, K.; Li, D.; Siwko, S.; Xin, L.; Liu, M. Lgr4 is a key regulator of prostate development and prostate stem cell differentiation. Stem Cells 2013, 31, 2492–2505. [Google Scholar] [CrossRef] [PubMed]
- Krusche, C.A.; Kroll, T.; Beier, H.M.; Classen-Linke, I. Expression of leucine-rich repeat-containing G-protein-coupled receptors in the human cyclic endometrium. Fertil. Steril. 2007, 87, 1428–1437. [Google Scholar] [CrossRef] [PubMed]
- Mohri, Y.; Umezu, T.; Hidema, S.; Tomisawa, H.; Akamatsu, A.; Kato, S.; Nawa, A.; Nishimori, K. Reduced fertility with impairment of early-stage embryos observed in mice lacking Lgr4 in epithelial tissues. Fertil. Steril. 2010, 94, 2878–2881. [Google Scholar] [CrossRef]
- De Oliveira, V.; Schaefer, J.; Calder, M.; Lydon, J.P.; Demayo, F.J.; Bhattacharya, M.; Radovick, S.; Babwah, A.V. Uterine Gαq/11 signaling, in a progesterone-dependent manner, critically regulates the acquisition of uterine receptivity in the female mouse. FASEB J. 2019, 33, 9374–9387. [Google Scholar] [CrossRef] [PubMed]
- Sone, M.; Oyama, K.; Mohri, Y.; Hayashi, R.; Clevers, H.; Nishimori, K. LGR4 expressed in uterine epithelium is necessary for uterine gland development and contributes to decidualization in mice. FASEB J. 2013, 27, 4917–4928. [Google Scholar] [CrossRef]
- Taylor, P.; Kida, T.; Oyama, K.; Sone, M.; Koizumi, M.; Hidema, S. Lgr4 is required for endometrial receptivity acquired through ovarian hormone signaling. Biosci. Biotechnol. Biochem. 2014, 78, 1813–1816. [Google Scholar] [CrossRef]
- Koizumi, M.; Oyama, K.; Yamakami, Y.; Kida, T.; Satoh, R.; Kato, S.; Oe, T.; Goto, T.; Clevers, H.; Nawa, A.; et al. Lgr4 Controls Specialization of Female Gonads in Mice. Biol. Reprod. 2015, 93, 1–11. [Google Scholar] [CrossRef]
- Pan, H.; Cui, H.; Liu, S.; Qian, Y.; Wu, H.; Li, L.; Guan, Y.; Guan, X.; Zhang, L.; Fan, H.Y.; et al. Lgr4 gene regulates corpus luteum maturation through modulation of the WNT-mediated EGFR-ERK signaling pathway. Endocrinology 2014, 155, 3624–3637. [Google Scholar] [CrossRef] [PubMed]
- Kato, S.; Mohri, Y.; Matsuo, T.; Ogawa, E.; Umezawa, A.; Okuyama, R.; Nishimori, K. Eye-open at birth phenotype with reduced keratinocyte motility in LGR4 null mice. FEBS Lett. 2007, 581, 4685–4690. [Google Scholar] [CrossRef][Green Version]
- Jin, C.; Yin, F.; Lin, M.; Li, H.; Wang, Z.; Weng, J. GPR48 Regulates Epithelial Cell Proliferation and Migration by Activating EGFR during Eyelid Development. Investig. Opthalmol. Vis. Sci. 2008, 49, 4245–4253. [Google Scholar] [CrossRef]
- Siwko, S.; Lai, L.; Weng, J.; Liu, M. Lgr4 in ocular development and glaucoma. J. Ophthalmol. 2013, 2013, 1–9. [Google Scholar] [CrossRef]
- Weng, J.; Luo, J.; Cheng, X.; Jin, C.; Zhou, X.; Qu, J.; Tu, L.; Ai, D.; Li, D.; Wang, J.; et al. Deletion of G protein-coupled receptor 48 leads to ocular anterior segment dysgenesis (ASD) through down-regulation of Pitx2. Proc. Natl. Acad. Sci. USA 2008, 105, 6081–6086. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Hou, Q.; Dong, X.D.; Wang, Z.; Chen, X.; Zheng, D.; Zhou, L.; He, C.; Liu, M.; Tu, L.L.; et al. Targeted deletion of the murine Lgr4 gene decreases lens epithelial cell resistance to oxidative stress and induces age-related cataract formation. PLoS ONE 2015, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mustata, R.C.; Van Loy, T.; Lefort, A.; Libert, F.; Strollo, S.; Vassart, G.; Garcia, M.I. Lgr4 is required for Paneth cell differentiation and maintenance of intestinal stem cells ex vivo. EMBO Rep. 2011, 12, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Farin, H.F.; Van Es, J.H.; Clevers, H. Redundant sources of Wnt regulate intestinal stem cells and promote formation of paneth cells. Gastroenterology 2012, 143, 1518–1529.e7. [Google Scholar] [CrossRef] [PubMed]
- Kinzel, B.; Pikiolek, M.; Orsini, V.; Sprunger, J.; Isken, A.; Zietzling, S.; Desplanches, M.; Dubost, V.; Breustedt, D.; Valdez, R.; et al. Functional roles of Lgr4 and Lgr5 in embryonic gut, kidney and skin development in mice. Dev. Biol. 2014, 390, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Mustata, R.C.; Vasile, G.; Fernandez-Vallone, V.; Strollo, S.; Lefort, A.; Libert, F.; Monteyne, D.; Pérez-Morga, D.; Vassart, G.; Garcia, M.I. Identification of Lgr5-Independent Spheroid-Generating Progenitors of the Mouse Fetal Intestinal Epithelium. Cell Rep. 2013, 5, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Qian, Y.; Li, L.; Wei, G.; Guan, Y.; Pan, H.; Guan, X.; Zhang, L.; Lu, X.; Zhao, Y.; et al. Lgr4 gene deficiency increases susceptibility and severity of dextran sodium sulfate-induced inflammatory bowel disease in mice. J. Biol. Chem. 2013, 288, 8794–8803. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Dong, J.; Li, D.; Lai, L.; Siwko, S.; Li, Y.; Liu, M. Lgr4 regulates mammary gland development and stem cell activity through the pluripotency transcription factor Sox2. Stem Cells 2013, 31, 1921–1931. [Google Scholar] [CrossRef] [PubMed]
- Oyama, K.; Mohri, Y.; Sone, M.; Nawa, A.; Nishimori, K. Conditional knockout of Lgr4 leads to impaired ductal elongation and branching morphogenesis in mouse mammary glands. Sex. Dev. 2011, 5, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Kato, S.; Matsubara, M.; Matsuo, T.; Mohri, Y.; Kazama, I.; Hatano, R.; Umezawa, A.; Nishimori, K. Leucine-rich repeat-containing G protein-coupled receptor-4 (LGR4, Gpr48) is essential for renal development in mice. Nephron Exp. Nephrol. 2006, 104, 63–76. [Google Scholar] [CrossRef]
- Mohri, Y.; Oyama, K.; Akamatsu, A.; Kato, S.; Nishimori, K. Lgr4-deficient mice showed premature differentiation of ureteric bud with reduced expression of Wnt effector Lef1 and Gata3. Dev. Dyn. 2011, 240, 1626–1634. [Google Scholar] [CrossRef]
- Mohri, Y.; Oyama, K.; Sone, M.; Akamatsu, A.; Nishimori, K. LGR4 is required for the cell survival of the peripheral mesenchyme at the embryonic stages of nephrogenesis. Biosci. Biotechnol. Biochem. 2012, 76, 888–891. [Google Scholar] [CrossRef] [PubMed]
- Dang, Y.; Liu, B.; Xu, P.; Zhu, P.; Zhai, Y.; Liu, M.; Ye, X. Gpr48 Deficiency Induces Polycystic Kidney Lesions and Renal Fibrosis in Mice by Activating Wnt Signal Pathway. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Li, Z.; Liu, S.; Lou, J.; Mulholland, M.; Zhang, W. LGR4 protects hepatocytes from injury in mouse. Am. J. Physiol. Gastrointest. Liver Physiol. 2019, 316, G123–G131. [Google Scholar] [CrossRef]
- Liu, S.; Yin, Y.; Yu, R.; Li, Y.; Zhang, W. R-spondin3-LGR4 signaling protects hepatocytes against DMOG-induced hypoxia/reoxygenation injury through activating β-catenin. Biochem. Biophys. Res. Commun. 2018, 499, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Planas-Paz, L.; Orsini, V.; Boulter, L.; Calabrese, D.; Pikiolek, M.; Nigsch, F.; Xie, Y.; Roma, G.; Donovan, A.; Mart, P.; et al. The RSPO-LGR4/5-ZNRF3/RNF43 module controls liver zonation and size. Nat. Cell Biol. 2016, 18, 467–479. [Google Scholar] [CrossRef]
- Mohri, Y.; Kato, S.; Umezawa, A.; Okuyama, R.; Nishimori, K. Impaired hair placode formation with reduced expression of hair follicle-related genes in mice lacking Lgr4. Dev. Dyn. 2008, 237, 2235–2242. [Google Scholar] [CrossRef] [PubMed]
- Żak, M.; Van Oort, T.; Hendriksen, F.G.; Garcia, M.I.; Vassart, G.; Grolman, W. LGR4 and LGR5 regulate hair cell differentiation in the sensory epithelium of the developing mouse cochlea. Front. Cell. Neurosci. 2016, 10, 1–15. [Google Scholar] [CrossRef]
- Wang, Z.; Jin, C.; Li, H.; Li, C.; Hou, Q.; Liu, M.; Da, X.; Dong, E.; Tu, L. GPR48-Induced keratinocyte proliferation occurs through HB-EGF mediated EGFR transactivation. FEBS Lett. 2010, 584, 4057–4062. [Google Scholar] [CrossRef]
- Yamakami, Y.; Kohashi, K.; Oyama, K.; Mohri, Y.; Hidema, S.; Nishimori, K. LGR4 is required for sequential molar development. Biochem. Biophys. Rep. 2016, 8, 174–183. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhou, M.; Guo, S.; Yuan, L.; Zhang, Y.; Zhang, M.; Chen, H.; Lu, M.; Yang, J.; Ma, J. Blockade of LGR4 inhibits proliferation and odonto/osteogenic differentiation of stem cells from apical papillae. J. Mol. Histol. 2017, 48, 389–401. [Google Scholar] [CrossRef]
- Luo, J.; Zhou, W.; Zhou, X.; Li, D.; Weng, J.; Yi, Z.; Cho, S.G.; Li, C.; Yi, T.; Wu, X.; et al. Regulation of bone formation and remodeling by G-protein-coupled receptor 48. Development 2009, 136, 2747–2756. [Google Scholar] [CrossRef]
- Zhu, C.; Zheng, X.; Yang, Y.; Li, B.; Wang, Y.; Jiang, S.; Jiang, L. LGR4 acts as a key receptor for R-spondin 2 to promote osteogenesis through Wnt signaling pathway. Cell. Signal. 2016, 28, 989–1000. [Google Scholar] [CrossRef]
- Pawaputanon Na Mahasarakham, C.; Ezura, Y.; Kawasaki, M.; Smriti, A.; Moriya, S.; Yamada, T.; Izu, Y.; Nifuji, A.; Nishimori, K.; Izumi, Y.; et al. BMP-2 Enhances Lgr4 Gene Expression in Osteoblastic Cells. J. Cell. Physiol. 2016, 231, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Pawaputanon Na Mahasarakham, C.; Izu, Y.; Nishimori, K.; Izumi, Y.; Noda, M.; Ezura, Y. Lgr4 Expression in Osteoblastic Cells Is Suppressed by Hydrogen Peroxide Treatment. J. Cell. Physiol. 2016, 232, 1761–1766. [Google Scholar] [CrossRef]
- Sun, P.; Siwko, S. Loss of Lgr4 inhibits differentiation, migration and apoptosis, and promotes proliferation in bone mesenchymal stem cells. J. Cell. Physiol. 2018, 234, 10855–10867. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.; Zheng, X.; Zhu, C.; Li, B.; Wang, Y.; Jiang, S. Evidence of the Role of R-Spondin 1 and Its Receptor Lgr4 in the Transmission of Mechanical Stimuli to Biological Signals for Bone Formation. Int. J. Mol. Sci. 2017, 28, 564. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhang, P.; Liu, Y.; Lv, L.; Zhang, X.; Liu, H. RSPO3-LGR4 Regulates Osteogenic Differentiation Of Human Adipose- Derived Stem Cells Via ERK/FGF Signalling. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.; Hill, D.R.; Kumar, N.; Huang, S.; Chin, A.M.; Dye, B.R.; Nagy, M.S.; Verzi, M.P.; Spence, J.R. LGR4 and LGR5 Function Redundantly During Human Endoderm Differentiation. Cell. Mol. Gastroenterol. Hepatol. 2016, 2, 648–662.e8. [Google Scholar] [CrossRef]
- Yamashita, R.; Takegawa, Y.; Sakumoto, M.; Nakahara, M.; Kawazu, H.; Hoshii, T.; Araki, K.; Yokouchi, Y.; Yamamura, K. Defective Development of the Gall Bladder and Cystic Duct in Lgr4–Hypomorphic Mice. Dev. Dyn. 2009, 993–1000. [Google Scholar] [CrossRef] [PubMed]
- Van Schoore, G.; Mendive, F.; Pochet, R.; Vassart, G. Expression pattern of the orphan receptor LGR4/GPR48 gene in the mouse. Histochem. Cell Biol. 2005, 124, 35–50. [Google Scholar] [CrossRef]
- Yi, T.; Weng, J.; Siwko, S.; Luo, J.; Li, D.; Liu, M. LGR4/GPR48 Inactivation Leads to Aniridia-Genitourinary Anomalies-Mental Retardation Syndrome Defects. J. Biol. Chem. 2014, 289, 8767–8780. [Google Scholar] [CrossRef] [PubMed]
- Styrkarsdottir, U.; Thorleifsson, G.; Sulem, P.; Gudbjartsson, D.F.; Sigurdsson, A.; Jonasdottir, A.; Jonasdottir, A.; Oddsson, A.; Helgason, A.; Magnusson, O.T.; et al. Nonsense mutation in the LGR4 gene is associated with several human diseases and other traits. Nature 2013, 497, 517–520. [Google Scholar] [CrossRef]
- Mancini, A.; Howard, S.R.; Marelli, F.; Cabrera, C.P.; Barnes, M.R.; Sternberg, M.J.E.; Leprovots, M.; Hadjidemetriou, I.; Monti, E.; David, A.; et al. LGR4 deficiency results in delayed puberty through impaired Wnt/β-catenin signaling. JCI Insight 2020, 5. [Google Scholar] [CrossRef]
- Han, X.H.; Jin, Y.-R.; Tan, L.; Kosciuk, T.; Lee, J.-S.; Yoon, J.K. Regulation of the Follistatin Gene by RSPO-LGR4 Signaling via Activation of the WNT/-Catenin Pathway in Skeletal Myogenesis. Mol. Cell. Biol. 2014, 34, 752–764. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, F.; Massa, F.; Motamedi, F.J.; Vidal, V.; Rocha, A.S.; Gregoire, E.P.; Cai, C.-L.; Wagner, K.D.; Schedl, A. Myocardial-specific R-spondin3 drives proliferation of the coronary stems primarily through the Leucine Rich Repeat G Protein coupled receptor LGR4. Dev. Biol. 2018, 441, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Luo, J.; Luo, W.; Weng, J.; Wang, Z.; Li, B.; Li, D.; Liu, M. Inactivation of G-protein-coupled Receptor 48 (Gpr48/Lgr4) Impairs Definitive Erythropoiesis at Midgestation through Down-regulation of the ATF4 Signaling Pathway. J. Biol. Chem. 2008, 283, 36687–36697. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, H.; Guo, J.; Gao, J.; Wang, M.; Xia, M.; Wen, Y.; Su, P.; Yang, M.; Liu, M.; et al. LGR4, Not LGR5, Enhances hPSC Hematopoiesis by Facilitating Mesoderm Induction via TGF-Beta Signaling Activation. Cell Rep. 2020, 31, 107600. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Zhang, Z.; Chen, L.; Yu, H.; Guo, S.; Xu, Y.; Oudit, G.Y.; Zhang, Y.; Chang, Q.; Song, B.; et al. Peptides Ascending aortic adventitial remodeling and fibrosis are ameliorated with Apelin-13 in rats after TAC via suppression of the miRNA-122 and LGR4-β-catenin signaling. Peptides 2016, 86, 85–94. [Google Scholar] [CrossRef]
- Huang, C.-K.; Dai, D.; Xie, H.; Zhu, Z.; Hu, J.; Su, M.; Liu, M.; Lu, L.; Shen, W.; Ning, G.; et al. Lgr4 Governs a Pro-Inflammatory Program in Macrophages to Antagonize Post-Infarction Cardiac Repair. Circ. Res. 2020, 127, 953–973. [Google Scholar] [CrossRef]
- Ge, J.J.; Lu, W.K.; Zhu, N.; Gao, X.J.; Yang, F.Q.; Wu, X.Z. Effect of LGR4 on synovial cells and inflammatory factors in rats with traumatic osteoarthritis. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 6783–6790. [Google Scholar] [CrossRef]
- Du, B.; Luo, W.; Li, R.; Tan, B.; Han, H.; Lu, X.; Li, D.; Qian, M.; Zhang, D.; Zhao, Y.; et al. Lgr4/Gpr48 negatively regulates TLR2/4-associated pattern recognition and innate immunity by targeting CD14 expression. J. Biol. Chem. 2013, 288, 15131–15141. [Google Scholar] [CrossRef]
- Hohenhaus, D.M.; Schaale, K.; Le Cao, K.A.; Seow, V.; Iyer, A.; Fairlie, D.P.; Sweet, M.J. An mRNA atlas of G protein-coupled receptor expression during primary human monocyte/macrophage differentiation and lipopolysaccharide-mediated activation identifies targetable candidate regulators of inflammation. Immunobiology 2013, 218, 1345–1353. [Google Scholar] [CrossRef]
- Guan, X.; Duan, Y.; Zeng, Q.; Pan, H.; Qian, Y.; Li, D.; Cao, X.; Liu, M. Lgr4 Protein Deficiency Induces Ataxia-like Phenotype in Mice and Impairs Long Term Depression at Cerebellar Parallel Fiber-Purkinje Cell Synapses. J. Biol. Chem. 2014, 289, 26492–26504. [Google Scholar] [CrossRef]
- Li, J.Y.; Chai, B.; Zhang, W.; Fritze, D.M.; Zhang, C.; Mulholland, M.W. LGR4 and its ligands, R-spondin 1 and R-spondin 3, regulate food intake in the hypothalamus of male rats. Endocrinology 2014, 155, 429–440. [Google Scholar] [CrossRef]
- Otsuka, A.; Jinguji, A.; Maejima, Y.; Kasahara, Y.; Shimomura, K.; Hidema, S.; Nishimori, K. LGR4 is essential for R-spondin1-mediated suppression of food intake via pro-opiomelanocortin. Biosci. Biotechnol. Biochem. 2019, 83, 1336–1342. [Google Scholar] [CrossRef]
- Sun, Y.; Hong, J.; Chen, M.; Ke, Y.; Zhao, S.; Liu, W.; Ma, Q.; Shi, J.; Zou, Y.; Ning, T.; et al. Ablation of Lgr4 enhances energy adaptation in skeletal muscle via activation of Ampk/Sirt1/Pgc1α pathway. Biochem. Biophys. Res. Commun. 2015, 464, 396–400. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Gao, Y.; Zhang, L.; Yin, Y.; Zhang, W. Rspo1/Rspo3-LGR4 signaling inhibits hepatic cholesterol synthesis through the AMPKα-SREBP2 pathway. FASEB J. 2020, 34, 14946–14959. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, X.; Wang, J.; Chen, M.; Fan, N.; Ma, Q.; Liu, R.; Wang, R.; Li, X.; Liu, M.; et al. LGR4 acts as a link between the peripheral circadian clock and lipid metabolism in liver. J. Mol. Endocrinol. 2014, 52, 133–144. [Google Scholar] [CrossRef]
- Zou, Y.; Ning, T.; Shi, J.; Chen, M.; Ding, L.; Huang, Y.; Kauderer, S.; Xu, M.; Cui, B.; Bi, Y.; et al. Association of a gain-of-function variant in LGR4 with central obesity. Obesity 2017, 25, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, X.; Ke, Y.; Lu, Y.; Wang, F.; Fan, N.; Sun, H.; Zhang, H.; Liu, R.; Yang, J.; et al. GPR48 Increases Mineralocorticoid Receptor Gene Expression. J. Am. Soc. Nephrol. 2012, 23, 281–293. [Google Scholar] [CrossRef]
- Li, B.; Yao, Q.; Guo, S.; Ma, S.; Dong, Y.; Xin, H.; Wang, H.; Liu, L.; Chang, W.; Zhang, Y. Type 2 diabetes with hypertensive patients results in changes to features of adipocytokines: Leptin, Irisin, LGR4, and Sfrp5. Clin. Exp. Hypertens. 2019, 41, 645–650. [Google Scholar] [CrossRef]
- Global Cancer Observatory. Available online: https://gco.iarc.fr/ (accessed on 21 January 2021).
- Yue, Z.; Yuan, Z.; Zeng, L.; Wang, Y.; Lai, L.; Li, J.; Sun, P.; Xue, X.; Qi, J. LGR4 modulates breast cancer initiation, metastasis, and cancer stem cells. FASEB J. 2018, 32, 2422–2437. [Google Scholar] [CrossRef]
- Zhu, Y.; Xu, L.; Chen, M.; Ma, H.; Lou, F. GPR48 Promotes Multiple Cancer Cell Proliferation via Activation of Wnt Signaling. Asian Pac. J. Cancer Prev. 2013, 14, 4775–4778. [Google Scholar] [CrossRef]
- Gao, Y.; Kitagawa, K.; Hiramatsu, Y.; Kikuchi, H.; Isobe, T.; Shimada, M.; Uchida, C.; Hattori, T.; Oda, T.; Nakayama, K.; et al. Up-regulation of GPR48 induced by down-regulation of p27Kip1 enhances carcinoma cell invasiveness and metastasis. Cancer Res. 2006, 66, 11623–11631. [Google Scholar] [CrossRef]
- Wu, J.; Xie, N.; Xie, K.; Zeng, J.; Cheng, L.; Lei, Y.; Liu, Y.; Song, L.; Dong, D.; Chen, Y.; et al. GPR48, a poor prognostic factor, promotes tumor metastasis and activates β -catenin/TCF signaling in colorectal cancer. Carcinogenesis 2013, 34, 2861–2869. [Google Scholar] [CrossRef]
- Gugger, M.; White, R.; Song, S.; Waser, B.; Cescato, R.; Rivière, P.; Reubi, J.C. GPR87 is an overexpressed G-protein coupled receptor in squamous cell carcinoma of the lung. Dis. Markers 2008, 24, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Yi, J.; Carmon, K.S.; Crumbley, C.A.; Xiong, W.; Thomas, A.; Fan, X.; Guo, S.; An, Z.; Chang, J.T.; et al. Aberrant RSPO3-LGR4 signaling in Keap1-deficient lung adenocarcinomas promotes tumor aggressiveness. Oncogene 2015, 34, 4692–4701. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Song, Y.; Ling, Z.; Li, Y.; Ren, X.; Yang, J.; Wang, Z.; Xia, J.; Zhang, W.; Cheng, B. R-spondin 2-LGR4 system regulates growth, migration and invasion, epithelial-mesenchymal transition and stem-like properties of tongue squamous cell carcinoma via Wnt/β-catenin signaling. EBioMedicine 2019, 44, 275–288. [Google Scholar] [CrossRef]
- Al-Samadi, A.; Salo, T. Understanding the role of the R-spondin 2-LGR4 system in tongue squamous cell carcinoma progression. EBioMedicine 2019, 44, 8–9. [Google Scholar] [CrossRef]
- Luo, W.; Tan, P.; Rodriguez, M.; He, L.; Tan, K.; Zeng, L.; Siwko, S.; Liu, M. Leucine-rich repeat–containing G protein– coupled receptor 4 (Lgr4) is necessary for prostate cancer metastasis via epithelial–mesenchymal transition. J. Biol. Chem. 2017, 292, 15525–15537. [Google Scholar] [CrossRef]
- Liang, F.; Yue, J.; Wang, J. GPCR48/LGR4 promotes tumorigenesis of prostate cancer via PI3K/Akt signaling pathway. Med. Oncol. 2015, 32. [Google Scholar] [CrossRef]
- Zhang, J.; Li, Q.; Zhang, S.; Xu, Q.; Wang, T. Lgr4 promotes prostate tumorigenesis through the Jmjd2a/AR signaling pathway. Exp. Cell Res. 2016, 349, 77–84. [Google Scholar] [CrossRef]
- Liang, F.; Zhang, H.; Cheng, D.; Gao, H.; Wang, J.; Yue, J.; Zhang, N.; Wang, J.; Wang, Z.; Zhao, B. Ablation of LGR4 signaling enhances radiation sensitivity of prostate cancer cells. Life Sci. 2021, 265, 118737. [Google Scholar] [CrossRef]
- Hou, Q.; Han, S.; Yang, L.; Chen, S.; Chen, J.; Ma, N.; Wang, C.; Tang, J.; Chen, X.; Chen, F.; et al. The interplay of microRNA-34a, LGR4, EMT-associated factors, and MMP2 in regulating uveal melanoma cells. Investig. Ophthalmol. Vis. Sci. 2019, 60, 4503–4510. [Google Scholar] [CrossRef]
- Xu, P.; Dang, Y.; Wang, L.; Liu, X.; Ren, X.; Gu, J.; Liu, M.; Dai, X.; Ye, X. Lgr4 is crucial for skin carcinogenesis by regulating MEK/ERK and Wnt/b -catenin signaling pathways. Cancer Lett. 2016, 383, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Liang, G.; Du, P.; Liu, Y. Lgr4 Promotes Glioma Cell Proliferation through Activation of Wnt Signaling. Asian Pac. J. Cancer Prev. 2013, 14, 4907–4911. [Google Scholar] [CrossRef]
- Kang, Y.E.; Kim, J.; Kim, K.S.; Chang, J.Y.; Lee, J.; Yi, S.; Kim, H.W.; Kim, J.T.; Lee, K.; Choi, M.J.; et al. Upregulation of RSPO2-GPR48/LGR4 signaling in papillary thyroid carcinoma contributes to tumor progression. Oncotarget 2017, 8, 114980–114994. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Ji, N.; Yi, J.; Lv, J.; Yuan, J.; Lin, Z.; Liu, L.; Feng, X. LGR4 overexpression is associated with clinical parameters and poor prognosis of serous ovarian cancer. Cancer Biomarkers 2020, 28, 65–72. [Google Scholar] [CrossRef]
- Liu, J.; Wei, W.; Guo, C.A.; Han, N.; Pan, J.F.; Fei, T.; Yan, Z.Q. Stat3 upregulates leucine-rich repeat-containing G protein-coupled receptor 4 expression in osteosarcoma cells. Biomed Res. Int. 2013, 2013. [Google Scholar] [CrossRef]
- Steffen, J.S.; Simon, E.; Warneke, V.; Balschun, K.; Ebert, M.; Röcken, C. LGR4 and LGR6 are differentially expressed and of putative tumor biological significance in gastric carcinoma. Virchows Arch. 2012, 461, 355–365. [Google Scholar] [CrossRef]
- Wang, Z.; Yin, P.; Sun, Y.; Na, L.; Gao, J.; Wang, W.; Zhao, C. LGR4 maintains HGSOC cell epithelial phenotype and stem-like traits. Gynecol. Oncol. 2020, 159, 839–849. [Google Scholar] [CrossRef]
- Tan, B.; Shi, X.; Zhang, J.; Qin, J.; Zhang, N.; Ren, H.; Qian, M.; Siwko, S.; Carmon, K.; Liu, Q.; et al. Inhibition of RSPO-LGR4 facilitates checkpoint blockade therapy by switching macrophage polarization. Cancer Res. 2018, 78, 4929–4942. [Google Scholar] [CrossRef]
- Al-Hajj, M.; Clarke, M.F. Self-renewal and solid tumor stem cells. Oncogene 2004, 23, 7274–7282. [Google Scholar] [CrossRef]
- Fulawka, L.; Donizy, P.; Halon, A. Cancer stem cells—the current status of an old concept: Literature review and clinical approaches. Biol. Res. 2014, 47, 66. [Google Scholar] [CrossRef] [PubMed]
- Salik, B.; Yi, H.; Hassan, N.; Santiappillai, N.; Vick, B.; Connerty, P.; Duly, A.; Trahair, T.; Woo, A.J.; Beck, D.; et al. Targeting RSPO3-LGR4 Signaling for Leukemia Stem Cell Eradication in Acute Myeloid Leukemia. Cancer Cell 2020, 38, 263–278.e6. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ordaz-Ramos, A.; Rosales-Gallegos, V.H.; Melendez-Zajgla, J.; Maldonado, V.; Vazquez-Santillan, K. The Role of LGR4 (GPR48) in Normal and Cancer Processes. Int. J. Mol. Sci. 2021, 22, 4690. https://doi.org/10.3390/ijms22094690
Ordaz-Ramos A, Rosales-Gallegos VH, Melendez-Zajgla J, Maldonado V, Vazquez-Santillan K. The Role of LGR4 (GPR48) in Normal and Cancer Processes. International Journal of Molecular Sciences. 2021; 22(9):4690. https://doi.org/10.3390/ijms22094690
Chicago/Turabian StyleOrdaz-Ramos, Alejandro, Victor Hugo Rosales-Gallegos, Jorge Melendez-Zajgla, Vilma Maldonado, and Karla Vazquez-Santillan. 2021. "The Role of LGR4 (GPR48) in Normal and Cancer Processes" International Journal of Molecular Sciences 22, no. 9: 4690. https://doi.org/10.3390/ijms22094690
APA StyleOrdaz-Ramos, A., Rosales-Gallegos, V. H., Melendez-Zajgla, J., Maldonado, V., & Vazquez-Santillan, K. (2021). The Role of LGR4 (GPR48) in Normal and Cancer Processes. International Journal of Molecular Sciences, 22(9), 4690. https://doi.org/10.3390/ijms22094690





