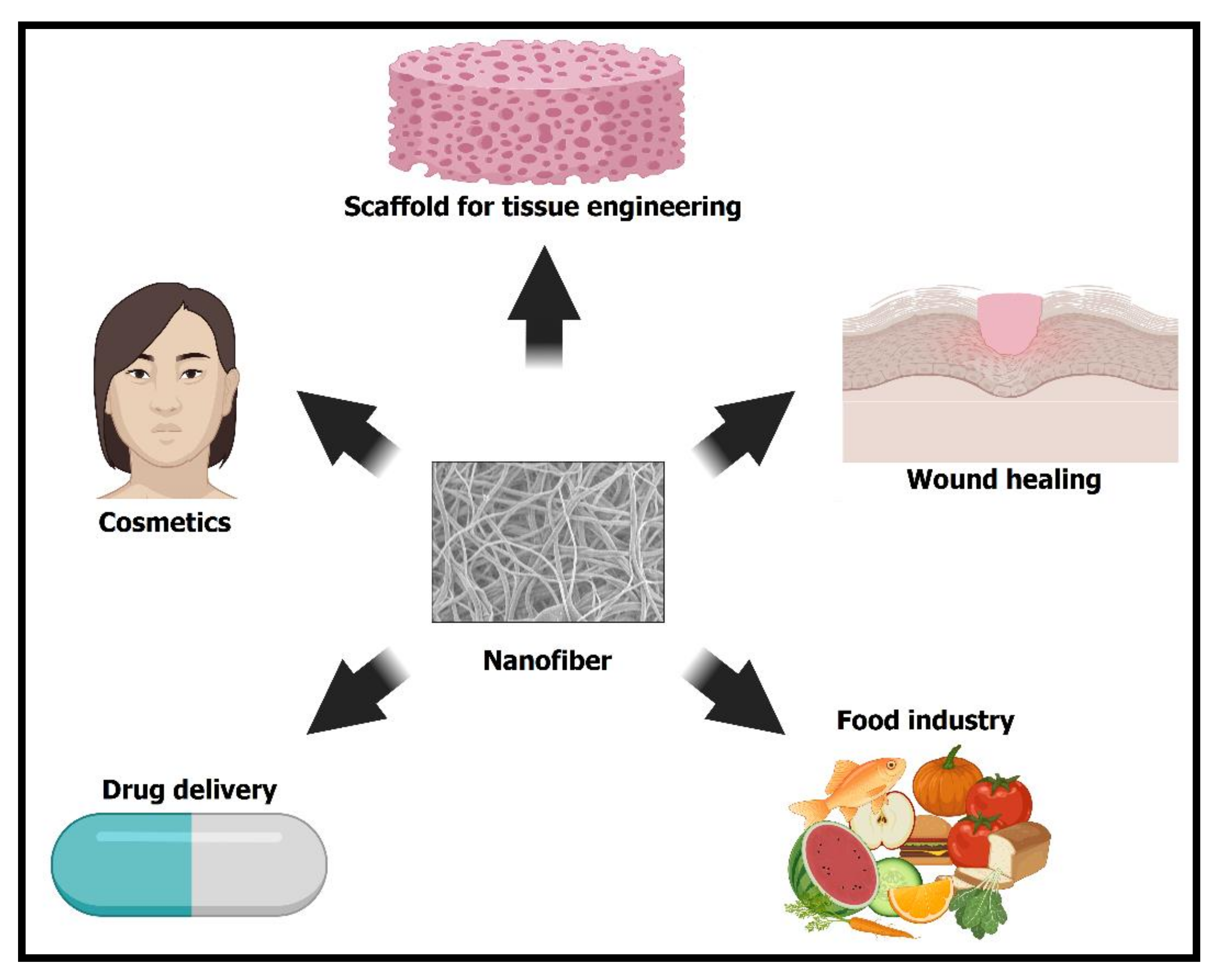
Prospects of Polymeric Nanofibers Loaded with Essential Oils for Biomedical and Food-Packaging Applications
Abstract
1. Introduction
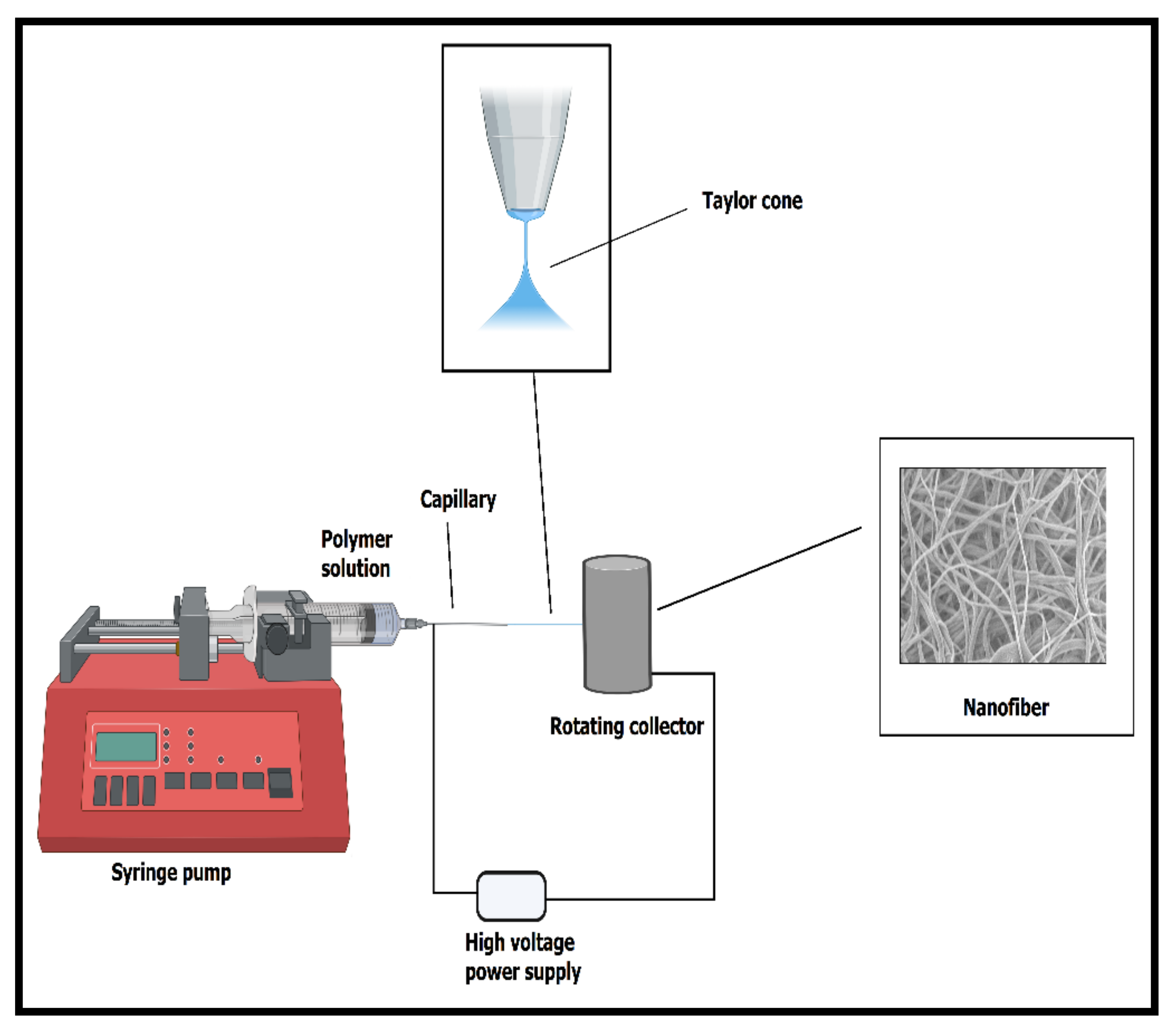
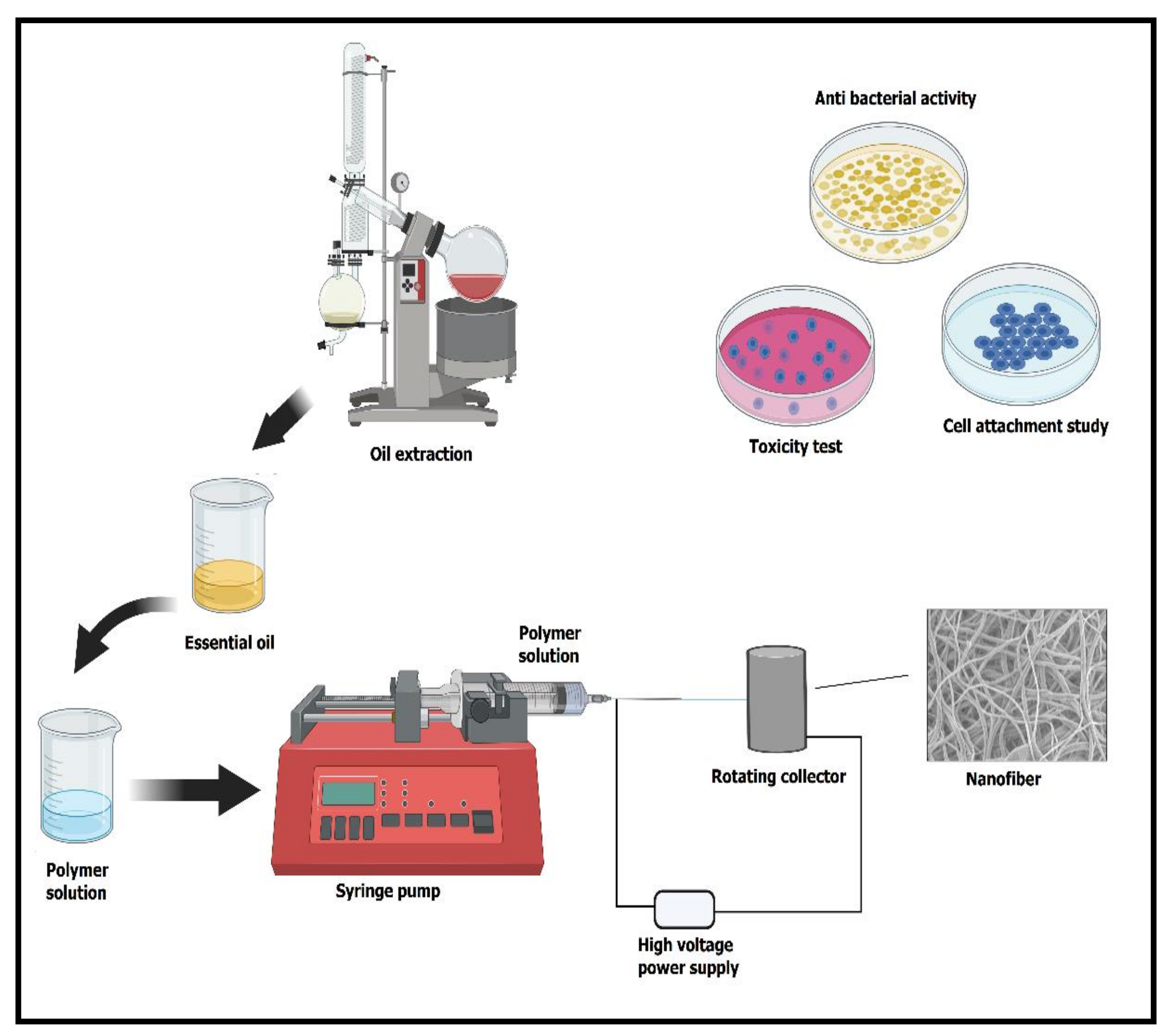
2. The Essential Process of Electrospinning to Form Nanofibers
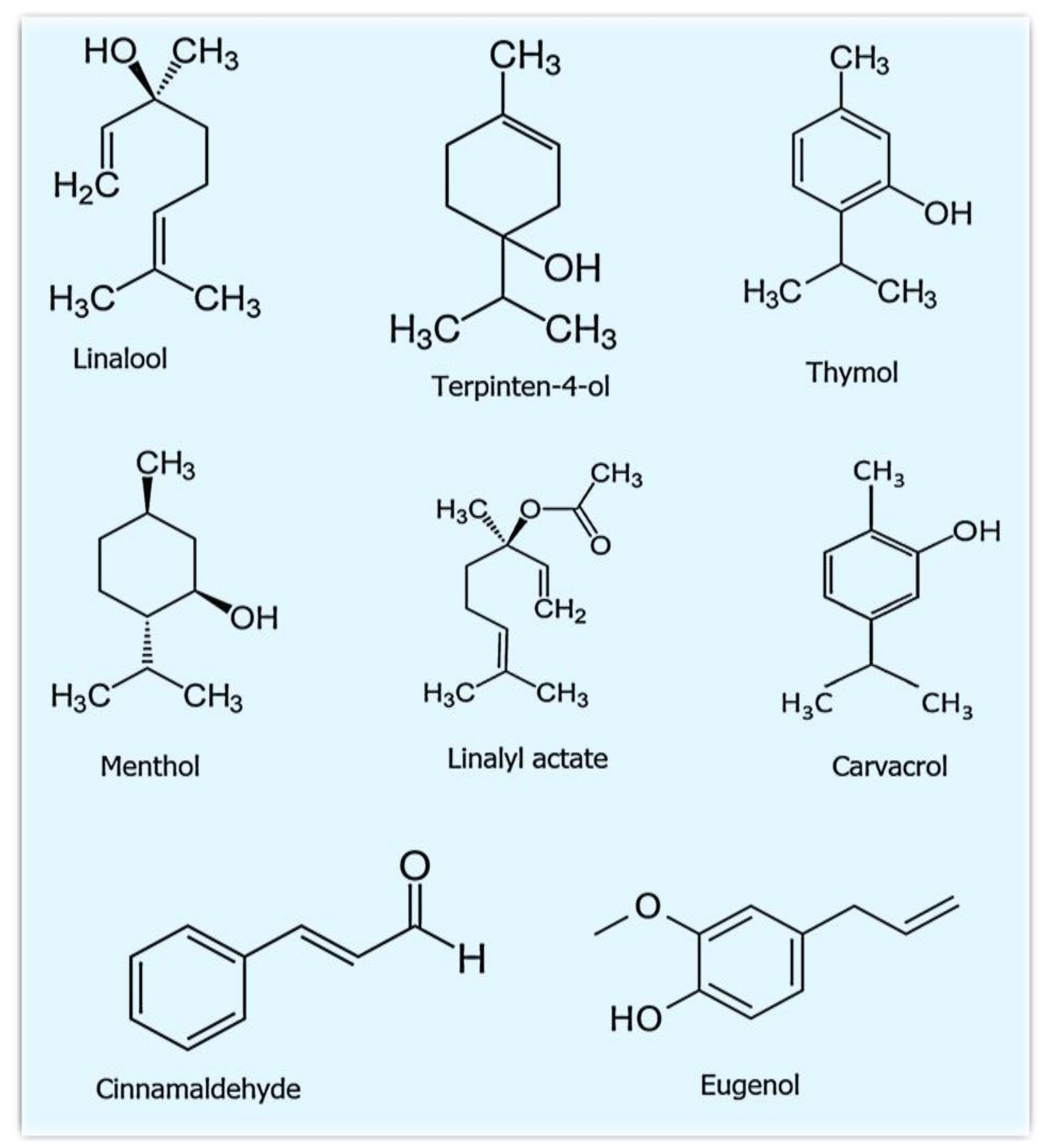
3. Essential Oils That Are Used to Modify the Nanofibers for Advanced Delivery
4. The Rationale of Encapsulation of Essential Oils into Polymeric Nanofibers
4.1. Encapsulation of Lavender Oil
4.2. Encapsulation of Thyme Essential Oil into Nanofiber Scaffolds
4.3. Encapsulation of Cinnamon Oil into Nanofibers
4.4. Encapsulation of Tea Tree Oil
4.5. Encapsulation of Peppermint Oil
4.6. Encapsulation of Clove Essential Oil
4.7. Oregano Essential Oil
4.8. Rosemary Essential Oil
4.9. Encapsulation of Ginger Essential Oil
4.10. Other Less-Common Essential Oils That Are Delivered While Encapsulating Them in Nanofibers
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Rochfort, S.; Panozzo, J. Phytochemicals for health, the role of pulses. J. Agric. Food Chem. 2007, 55, 7981–7994. [Google Scholar] [CrossRef]
- Ranjan, A.; Ramachandran, S.; Gupta, N.; Kaushik, I.; Wright, S.; Srivastava, S.; Das, H.; Srivastava, S.; Prasad, S.; Srivastava, S.K. Molecular Sciences Role of Phytochemicals in Cancer Prevention. Int. J. Mol. Sci. 2019, 20, 4981. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Colletti, A. Role of Phytochemicals in the Management of Metabolic Syndrome. In Phytomedicine; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Patra, A.K. An Overview of Antimicrobial Properties of Different Classes of Phytochemicals. In Dietary Phytochemicals and Microbes; Springer: Amsterdam, The Netherlands, 2012; pp. 1–32. [Google Scholar]
- Monte, J.; Abreu, A.C.; Borges, A.; Simões, L.C.; Simões, M. Antimicrobial Activity of Selected Phytochemicals against Escherichia Coli and Staphylococcus Aureus and Their Biofilms. Pathogenes 2014, 3, 473–498. [Google Scholar] [CrossRef] [PubMed]
- Maiyo, Z.C.; Ngure, R.M.; Matasyoh, J.C.; Chepkorir, R. Phytochemical constituents and antimicrobial activity of leaf extracts of three Amaranthus plant species. Afr. J. Biotechnol. 2010, 9, 3178–3182. [Google Scholar]
- Martinez, J.L. Environmental pollution by antibiotics and by antibiotic resistance determinants. Environ. Pollut. 2009, 157, 2893–2902. [Google Scholar] [CrossRef] [PubMed]
- Larsson, D.G.J. Antibiotics in the Environment. Upsala J. Med. Sci. 2014, 119, 108–112. [Google Scholar] [CrossRef]
- Grenni, P.; Ancona, V.; Caracciolo, A.B. Ecological effects of antibi-otics on natural ecosystems: A review. Microchem. J. 2018, 136, 25–39. [Google Scholar] [CrossRef]
- Sridhar, R.; Lakshminarayanan, R.; Madhaiyan, K.; Bharathi, V.A.; Hsiu, K.; Lim, C.; Ramakrishna, S. Electrosprayed Nanoparticles and Electrospun Nanofibers Based on Natural Materials: Applications in Tissue Regeneration, Drug Delivery and Pharmaceuticals. Chem. Soc. Rev. 2015, 44, 790–814. [Google Scholar] [CrossRef]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antimicrobial Activity of Essential Oils and Other Plant Extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar] [CrossRef] [PubMed]
- Kä, M.P.; Hopia, A.I.; Vuorela, H.J.; Rauha, J.-P.; Pihlaja, K.; Kujala, T.S.; Heinonen, M. Antioxidant Activity of Plant Extracts Containing Phenolic Compounds. ACS Publ. 1999, 47, 3954–3962. [Google Scholar]
- Friedman, M. Antibiotic-Resistant Bacteria: Prevalence in Food and Inactivation by Food-Compatible Compounds and Plant Extracts. J. Agric. Food Chem. 2015, 63, 3805–3822. [Google Scholar] [CrossRef]
- Dorman, H.J.D.; Deans, S.G. Antimicrobial Agents from Plants: Antibacterial Activity of Plant Volatile Oils. J. Appl. Microbiol. 2000, 88, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Li, Y.; Chen, T. Techniques for Fabrication and Construction of Three-Dimensional Scaffolds for Tissue Engineering. Int. J. Nanomed. 2013, 8, 337–350. [Google Scholar] [CrossRef] [PubMed]
- Stojanovska, E.; Canbay, E.; Pampal, E.S.; Calisir, M.D.; Agma, O.; Polat, Y.; Simsek, R.; Gundogdu, S.; Akgul, Y.; Kilic, A. A Review on Non-Electro Nanofibre Spinning Techniques. RSC Adv. 2016, 6, 83783–83801. [Google Scholar] [CrossRef]
- Beachley, V.; Wen, X. Polymer Nanofibrous Structures: Fabrication, Biofunctionalization, and Cell Interactions. Prog. Polym. Sci. 2010, 35, 868–892. [Google Scholar] [CrossRef]
- Li, W.-J.; Shanti, R.M.; Tuan, R.S. Electrospinning Technology for Nanofibrous Scaffolds in Tissue Engineering. In Nanotechnologies for the Life Sciences; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2007. [Google Scholar]
- Zhang, X.; Lu, Y. Polymer Reviews Centrifugal Spinning: An Alternative Approach to Fabricate Nanofibers at High Speed and Low Cost Centrifugal Spinning: An Alternative Approach to Fabricate Nanofibers at High Speed and Low Cost. Polym. Rev. 2014, 54, 677–701. [Google Scholar] [CrossRef]
- Ramakrishna, S. An Introduction to Electrospinning and Nanofibers; World Scientific: Singapore, 2005. [Google Scholar]
- Kumar, P. Effect of Collector on Electrospinning to Fabricate Aligned Nanofiber Bio-Medical Engineering. Ph.D. Thesis, National Institutes of Technology, Calicut, India, 2011. [Google Scholar]
- Wang, J.; Nain, A.S. Suspended Micro/Nanofiber Hierarchical Biological Scaffolds Fabricated Using Non-Electrospinning STEP Technique. Langmuir 2014, 30, 13641–13649. [Google Scholar] [CrossRef]
- Naskar, D.; Bhattacharjee, P.; Ghosh, A.K.; Mandal, M.; Kundu, S.C. Carbon Nanofiber Reinforced Nonmulberry Silk Protein Fibroin Nanobiocomposite for Tissue Engineering Applications. ACS Appl. Mater. Interfaces 2017, 9, 19356–19370. [Google Scholar] [CrossRef]
- Xu, W.; Song, Q.; Xu, J.F.; Serpe, M.J.; Zhang, X. Supramolecular Hydrogels Fabricated from Supramonomers: A Novel Wound Dressing Material. ACS Appl. Mater. Interfaces 2017, 9, 11368–11372. [Google Scholar] [CrossRef]
- Hu, X.; Liu, S.; Zhou, G.; Huang, Y.; Xie, Z.; Jing, X. Electrospinning of Polymeric Nanofibers for Drug Delivery Applications. J. Controll. Rel. 2014, 185, 12–21. [Google Scholar] [CrossRef]
- Ho Jung, Y.; Yong Kim, H.; Rae Lee, D.; Young Park, S.; Seob Khil, M. Characterization of PVOH Nonwoven Mats Prepared from Surfactant-Polymer System via Electrospinning. Macromol. Res. 2005, 13, 385–390. [Google Scholar] [CrossRef]
- Doshi, J.; Reneker, D.H. Electrospinning process and applications of electrospun fibers. J. Electrostat. 1995, 35, 151–160. [Google Scholar] [CrossRef]
- Wannes, W.A.; Mhamdi, B.; Sriti, J.; Jemia, M.B.; Ouchikh, O.; Hamdaoui, G.; Kchouk, M.E.; Marzouk, B. Antioxidant activities of the essential oils and methanol extracts from myrtle (Myrtus communis var. italica L.) leaf, stem and flower. Food Chem. Toxicol. 2010, 48, 1362–1370. [Google Scholar] [CrossRef]
- Pandey, R.; Kalra, A.; Tandon, S.; Mehrotra, N.; Singh, H.N.; Kumar, S. Essential Oils as Potent Sources of Nematicidal Compounds. J. Phytopathol. 2000, 148, 501–502. [Google Scholar] [CrossRef]
- Pessoa, L.M.; Morais, S.M.; Bevilaqua, C.M.L.; Luciano, J.H.S. Anthelmintic activity of essential oil of Ocimum gratissimum Linn. and eugenol against Haemonchus contortus. Vet. Parasitol. 2002, 109, 59–63. [Google Scholar] [CrossRef]
- Karpouhtsis, I.; Pardali, E.; Feggou, E.; Kokkini, S.; Scouras, Z.G.; Mavragani-Tsipidou, P. Insecticidal and Genotoxic Activities of Oregano Essential Oils. J. Agric. Food Chem. 1998, 46, 1111–1115. [Google Scholar] [CrossRef]
- Konstantopoulou, I.; Vassilopoulou, L.; Mavragani-Tsipidou, P.; Scouras, Z.G. Insecticidal effects of essential oils. A study of the effects of essential oils extracted from eleven greek aromatic plants on Drosophila auraria. Experientia 1992, 48, 616–619. [Google Scholar] [CrossRef] [PubMed]
- Kasper, S.; Anghelescu, I.; Dienel, A. Efficacy of orally administered Silexan in patients with anxiety-related restlessness and disturbed sleep–A randomized, placebo-controlled trial. Eur. Neuropsychopharmacol. 2015, 25, 1960–1967. [Google Scholar] [CrossRef]
- Pergentino De Sousa, D.; De, P.; Soares Hocayen, A.; Andrade, L.N.; Andreatini, R. Molecules A Systematic Review of the Anxiolytic-Like Effects of Essential Oils in Animal Models. Molecules 2015, 20, 18620–18660. [Google Scholar] [CrossRef] [PubMed]
- Uehleke, B.; Stange, R. Phase II Trial on the Effects of Silexan in Patients with Neurasthenia, Post-Traumatic Stress Disorder or Somatization Disorder Network-Pharmacology: Systematic Review to Analyse the Description of Herbal Drugs in Meta-Analyses View Project. Phytomed. Int. J. Phyther. Phytopharm. 2012, 19, 665–671. [Google Scholar]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils–A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Mendes, S.S.; Bomfim, R.R.; Jesus, H.C.R.; Alves, P.B.; Blank, A.F.; Estevam, C.S.; Antoniolli, A.R.; Thomazzi, S.M. Evaluation of the analgesic and anti-inflammatory effects of the essential oil of Lippia gracilis leaves. J. Ethnopharmacol. 2010, 129, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Pant, B.; Park, M.; Ojha, G.P.; Kim, D.-U.; Kim, H.-Y.; Park, S.-J. International Journal of Polymeric Materials and Polymeric Biomaterials Electrospun Salicylic Acid/Polyurethane Composite Nanofibers for Biomedical Applications Electrospun Salicylic Acid/Polyurethane Composite Nanofibers for Biomedical Applications. Int. J. Polym. Mater. Polym. Biomater. 2018, 67, 739–744. [Google Scholar] [CrossRef]
- Pant, B.; Pant, H.R.; Barakat, N.A.; Park, M.; Jeon, K.; Choi, Y.; Kim, H.-Y. Carbon nanofibers decorated with binary semiconductor (TiO2/ZnO) nanocomposites for the effective removal of organic pollutants and the enhancement of antibacterial activities. Ceram. Int. 2013, 39, 7029–7035. [Google Scholar] [CrossRef]
- Ates, B.; Koytepe, S.; Ulu, A.; Gurses, C.; Thakur, V.K. Chemistry, Structures, and Advanced Applications of Nanocomposites from Biorenewable Resources. Chem. Rev. 2020, 120, 9304–9362. [Google Scholar] [CrossRef]
- Zhang, Z.; Zheng, Y.; Zhang, L.; Prasath Mani, M.; Kumar Jaganathan, S. In Vitro Blood Compatibility and Bone Mineralization Aspects of Polymeric Scaffold Laden with Essential Oil and Metallic Particles for Bone Tissue Engineering. Int. J. Polym. Anal. Charact. 2019, 24, 504–516. [Google Scholar] [CrossRef]
- Eğri, Ö. Cumhuriyet Science Journal Production of Lavender Oil Loaded Antibacterial Polymeric Membranes. Cumhur. Sci. J. 2020, 41, 2587–2680. [Google Scholar] [CrossRef]
- Dadras Chomachayi, M.; Solouk, A.; Akbari, S.; Sadeghi, D.; Mirahmadi, F.; Mirzadeh, H. Electrospun Nanofibers Comprising of Silk Fibroin/Gelatin for Drug Delivery Applications: Thyme Essential Oil and Doxycycline Monohydrate Release Study. J. Biomed. Mater. Res. Part A 2018, 106, 1092–1103. [Google Scholar] [CrossRef] [PubMed]
- Koushki, P.; Bahrami, S.H.; Ranjbar-Mohammadi, M. Coaxial Nanofibers from Poly(Caprolactone)/Poly(Vinyl Alcohol)/Thyme and Their Antibacterial Properties. J. Ind. Text. 2018, 47, 834–852. [Google Scholar] [CrossRef]
- Fonseca, L.M.; Radünz, M.; dos Santos Hackbart, H.C.; da Silva, F.T.; Camargo, T.M.; Bruni, G.P.; Monks, J.L.F.; da Rosa Zavareze, E.; Dias, A.R.G. Electrospun Potato Starch Nanofibers for Thyme Essential Oil Encapsulation: Antioxidant Activity and Thermal Resistance. J. Sci. Food Agric. 2020, 100, 4263–4271. [Google Scholar] [CrossRef]
- Son, B.C.; Park, C.H.; Kim, C.S. Fabrication of Antimicrobial Nanofiber Air Filter Using Activated Carbon and Cinnamon Essential Oil. J. Nanosci. Nanotechnol. 2020, 20, 4376–4380. [Google Scholar] [CrossRef] [PubMed]
- Mahdavi, V.; Rafiee-Dastjerdi, H.; Asadi, A.; Razmjou, J.; Achachlouei, B.F.; Kamita, S.G. Effective Management of the Phthorimaea Operculella (Zeller) Using PVA Nanofibers Loaded with Cinnamomum Zeylanicum Essential Oil. Am. J. Potato Res. 2017, 94, 647–657. [Google Scholar] [CrossRef]
- Ranjbaryan, S.; Pourfathi, B.; Almasi, H. Reinforcing and release controlling effect of cellulose nanofiber in sodium caseinate films activated by nanoemulsified cinnamon essential oil. Food Packag. Shelf Life 2019, 21, 100341. [Google Scholar] [CrossRef]
- Wen, P.; Zhu, D.-H.; Wu, H.; Zong, M.-H.; Jing, Y.-R.; Han, S.-Y. Encapsulation of Cinnamon Essential Oil in Electrospun Nanofibrous Film for Active Food Packaging. Food Control 2016, 59, 366–376. [Google Scholar] [CrossRef]
- Lee, J.Y.; Jang, S.; Aguilar, L.E.; Park, C.H.; Kim, C.S. Structural Packaging Technique Using Biocompatible Nanofiber with Essential Oil to Prolong the Shelf-Life of Fruit. J. Nanosci. Nanotechnol. 2019, 19, 2228–2231. [Google Scholar] [CrossRef] [PubMed]
- Rieger, K.A.; Schiffman, J.D. Electrospinning an Essential Oil: Cinnamaldehyde Enhances the Antimicrobial Efficacy of Chitosan/Poly(Ethylene Oxide) Nanofibers. Carbohydr. Polym. 2014, 113, 561–568. [Google Scholar] [CrossRef]
- Güler, H.K.; Cengiz, F.; Emel, Ç.; Çetin, S.; Kesici Güler, H.; Çallıoğlu, C.; Çetin, E.S. Antibacterial PVP/Cinnamon Essential Oil Nanofibers by Emulsion Electrospinning Antibacterial PVP/Cinnamon Essential Oil Nanofibers by Emulsion Electrospinning. J. Text. Inst. 2018, 110, 302–310. [Google Scholar] [CrossRef]
- Cui, H.; Bai, M.; Li, C.; Liu, R.; Lin, L. Fabrication of Chitosan Nanofibers Containing Tea Tree Oil Liposomes against Salmonella Spp. in Chicken. LWT 2018, 96, 671–678. [Google Scholar] [CrossRef]
- Suganya Bharathi, B.; Stalin, T. Cerium Oxide and Peppermint Oil Loaded Polyethylene Oxide/Graphene Oxide Electrospun Nanofibrous Mats as Antibacterial Wound Dressings. Mater. Today Commun. 2019, 21, 100664. [Google Scholar] [CrossRef]
- Sahal, G.; Nasseri, B.; Ebrahimi, A.; Bilkay, I.S. Electrospun Essential Oil-Polycaprolactone Nanofibers as Antibiofilm Surfaces against Clinical Candida Tropicalis Isolates. Biotechnol. Lett. 2019, 41, 511–522. [Google Scholar] [CrossRef]
- Liakos, I.L.; Holban, A.M.; Carzino, R.; Lauciello, S.; Grumezescu, A.M. Electrospun Fiber Pads of Cellulose Acetate and Essential Oils with Antimicrobial Activity. Nanomaterial 2017, 7, 84. [Google Scholar] [CrossRef] [PubMed]
- Souza, V.G.L.; Pires, J.R.A.; Érica, T.V.; Coelhoso, I.M.; Duarte, M.P.; Fernando, A.L. Shelf Life Assessment of Fresh Poultry Meat Packaged in Novel Bionanocomposite of Chitosan/Montmorillonite Incorporated with Ginger Essential Oil. Coatings 2018, 8, 177. [Google Scholar] [CrossRef]
- Reneker, D.H.; Chun, I. Nanometre Diameter Fibres of Polymer, Produced by Electrospinning. Nanotechnology 1996, 7, 216–223. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef]
- Zargham, S.; Bazgir, S.; Tavakoli, A.; Rashidi, A.S.; Damerchely, R. The Effect of Flow Rate on Morphology and Deposition Area of Electrospun Nylon 6 Nanofiber. J. Eng. Fiber. Fabr. 2012, 7, 42–49. [Google Scholar] [CrossRef]
- Li, H.; Xu, Y.; Xu, H.; Chang, J. Electrospun membranes: Control of the structure and structure related applications in tissue regeneration and drug delivery. J. Mater. Chem. B 2014, 2, 5492–5510. [Google Scholar] [CrossRef]
- Han, D.; Cheung, K.C. Biodegradable Cell-Seeded Nanofiber Scaffolds for Neural Repair. Polymers 2011, 3, 1684–1733. [Google Scholar] [CrossRef]
- Kakinoki, S.; Uchida, S.; Ehashi, T.; Murakami, A.; Yamaoka, T. Surface Modification of Poly(L-lactic acid) Nanofiber with Oligo(D-lactic acid) Bioactive-Peptide Conjugates for Peripheral Nerve Regeneration. Polymers 2011, 3, 820–832. [Google Scholar] [CrossRef]
- Pant, B.; Park, M.; Park, S.J. Drug Delivery Applications of Core-Sheath Nanofibers Prepared by Coaxial Electrospinning: A Review. Pharmaceutics 2019, 11, 305. [Google Scholar] [CrossRef]
- Mirjalili, M.; Zohoori, S. Review for Application of Electrospinning and Electrospun Nanofibers Technology in Textile Industry. J. Nanostructure Chem. 2016, 6, 207–213. [Google Scholar] [CrossRef]
- Mele, E.; Bayer, I.S.; Nanni, G.; Heredia-Guerrero, J.A.; Ruffilli, R.; Ayadi, F.; Marini, L.; Cingolani, R.; Athanassiou, A. Biomimetic Approach for Liquid Encapsulation with Nanofibrillar Cloaks. Langmuir 2014, 30, 2896–2902. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Meng, J.; Wang, X.; Han, C.; Yan, M.; Zhao, K.; Xu, X.; Ren, W.; Zhao, Y.; Xu, L.; et al. General Synthesis of Complex Nanotubes by Gradient Electrospinning and Controlled Pyrolysis. Nat. Commun. 2015, 6, 7402. [Google Scholar] [CrossRef]
- Ren, X.; Ying, P.; Yang, Z.; Shang, M.; Hou, H.; Gao, F. Foaming-Assisted Electrospinning of Large-Pore Mesoporous ZnO Nanofibers with Tailored Structures and Enhanced Photocatalytic Activity. RSC Adv. 2015, 5, 16361–16367. [Google Scholar] [CrossRef]
- Peng, S.; Li, L.; Hu, Y.; Srinivasan, M.; Cheng, F.; Chen, J.; Ramakrishna, S. Fabrication of Spinel One-Dimensional Architectures by Single-Spinneret Electrospinning for Energy Storage Applications. ACS Nano 2015, 9, 1945–1954. [Google Scholar] [CrossRef] [PubMed]
- Guenther, E.; Althausen, D. The Essential Oils; Van Nostrand: New York, NY, USA, 1948. [Google Scholar]
- Li, K.K.; Yin, S.W.; Yang, X.Q.; Tang, C.H.; Wei, Z.H. Fabrication and Characterization of Novel Antimicrobial Films Derived from Thymol-Loaded Zein-Sodium Caseinate (SC) Nanoparticles. J. Agric. Food Chem. 2012, 60, 11592–11600. [Google Scholar] [CrossRef]
- Lombrea, A.; Antal, D.; Ardelean, F.; Avram, S.; Pavel, I.Z.; Vlaia, L.; Mut, A.-M.; Diaconeasa, Z.; Dehelean, C.A.; Soica, C.; et al. Molecular Sciences A Recent Insight Regarding the Phytochemistry and Bioactivity of Origanum Vulgare L. Essential Oil. Int. J. Mol. Sci. 2020, 2020, 9653. [Google Scholar]
- Chorianopoulos, N.G.; Giaouris, E.D.; Skandamis, P.N.; Haroutounian, S.A.; Nychas, G.J.E. Disinfectant Test against Monoculture and Mixed-Culture Biofilms Composed of Technological, Spoilage and Pathogenic Bacteria: Bactericidal Effect of Essential Oil and Hydrosol of Satureja Thymbra and Comparison with Standard Acid-Base Sanitizers. J. Appl. Microbiol. 2008, 104, 1586–1596. [Google Scholar] [CrossRef]
- Burt, S.; Reinders, R. Antibacterial activity of selected plant essential oils against Escherichia coli O157:H7. Lett. Appl. Microbiol. 2003, 36, 162–167. [Google Scholar] [CrossRef]
- De Martino, L.; De Feo, V.; Nazzaro, F. Molecules Chemical Composition and in Vitro Antimicrobial and Mutagenic Activities of Seven Lamiaceae Essential Oils. Molecules 2009, 14, 4213–4230. [Google Scholar] [CrossRef] [PubMed]
- Russo, M.; Galletti, G.C.; Bocchini, P.; Carnacini, A. Essential Oil Chemical Composition of Wild Populations of Italian Oregano Spice (Origanum Vulgare Ssp. Hirtum (Link) Ietswaart): A Preliminary Evaluation of Their Use in Chemotaxonomy by Cluster Analysis. 1. Inflorescences. J. Agric. Food Chem. 1998, 46, 3741–3746. [Google Scholar] [CrossRef]
- Senatore, F. Influence of Harvesting Time on Yield and Composition of the Essential Oil of a Thyme (Thymus Pulegioides L.) Growing Wild in Campania (Southern Italy). J. Agric. Food Chem. 1996, 44, 1327–1332. [Google Scholar] [CrossRef]
- Bauer, K.; Garbe, D.; Surburg, H. Common Fragrance and Flavor Materials: Preparation, Properties and Uses; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- Cosentino, S.; Tuberoso, C.I.G.; Pisano, B.; Satta, M.; Mascia, V.; Arzedi, E.; Palmas, F. In-Vitro Antimicrobial Activity and Chemical Composition of Sardinian Thymus Essential Oils. Lett. Appl. Microbiol. 1999, 29, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Angioni, A.; Barra, A.; Coroneo, V.; Dessi, S.; Cabras, P. Chemical Composition, Seasonal Variability, and Antifungal Activity of Lavandula Stoechas L. Ssp. Stoechas Essential Oils from Stem/Leaves and Flowers. J. Agric. Food Chem. 2006, 54, 4364–4370. [Google Scholar] [CrossRef] [PubMed]
- Masotti, V.R.Ä.; Juteau, F.; Bessie, Å.M.J.R.; Viano, J. Seasonal and Phenological Variations of the Essential Oil from the Narrow Endemic Species Artemisia Molinieri and Its Biological Activities. ACS Publ. 2003, 51, 7115–7121. [Google Scholar]
- Ojeda, T. Polymers and the Environment. In Polymer Science; IntechOpen: London, UK, 2013; Volume 23. [Google Scholar]
- Stevens, E.S. What makes green plastics green? Biocycle 2003, 44, 24–27. [Google Scholar]
- Al-Salem, S.M.; Lettieri, P.; Baeyens, J. Recycling and Recovery Routes of Plastic Solid Waste (PSW): A Review. Waste Manag. 2009, 29, 2625–2643. [Google Scholar] [CrossRef]
- Isman, M.B.; Miresmailli, S.; MacHial, C. Commercial Opportunities for Pesticides Based on Plant Essential Oils in Agriculture, Industry and Consumer Products. Phytochem. Rev. 2011, 10, 197–204. [Google Scholar] [CrossRef]
- Ramos, M.; Jiménez, A.; Peltzer, M.; Garrigós, M.C. Characterization and antimicrobial activity studies of polypropylene films with carvacrol and thymol for active packaging. J. Food Eng. 2012, 109, 513–519. [Google Scholar] [CrossRef]
- Ghormade, V.; Deshpande, M.V.; Paknikar, K.M. Perspectives for nano-biotechnology enabled protection and nutrition of plants. Biotechnol. Adv. 2011, 29, 792–803. [Google Scholar] [CrossRef]
- Eltayeb, M.; Bakhshi, P.K.; Stride, E.; Edirisinghe, M. Preparation of solid lipid nanoparticles containing active compound by electrohydrodynamic spraying. Food Res. Int. 2013, 53, 88–95. [Google Scholar] [CrossRef]
- Anjali, C.; Sharma, Y.; Mukherjee, A.; Chandrasekaran, N. Neem Oil (Azadirachta Indica) Nanoemulsion-a Potent Larvicidal Agent against Culex Quinquefasciatus. Pest Manag. Sci. 2012, 68, 158–163. [Google Scholar] [CrossRef]
- Hajiali, H.; Summa, M.; Russo, D.; Armirotti, A.; Brunetti, V.; Bertorelli, R.; Athanassiou, A.; Mele, E. Alginate–Lavender Nanofibers with Antibacterial and Anti-Inflammatory Activity to Effectively Promote Burn Healing. J. Mater. Chem. B 2016, 4, 1686–1695. [Google Scholar] [CrossRef]
- Kataria, K.; Gupta, A.; Rath, G.; Mathur, R.; Dhakate, S. In vivo wound healing performance of drug loaded electrospun composite nanofibers transdermal patch. Int. J. Pharm. 2014, 469, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, N.; Amin, M.C.I.M.; Pandey, M.; Ahmad, N.; Rajab, N.F. Bacterial cellulose/acrylic acid hydrogel synthesized via electron beam irradiation: Accelerated burn wound healing in an animal model. Carbohydr. Polym. 2014, 114, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yang, D.; Chen, X.; Xu, Q.; Lu, F.; Nie, J. Electrospun Water-Soluble Carboxyethyl Chitosan/Poly(Vinyl Alcohol) Nanofibrous Membrane as Potential Wound Dressing for Skin Regeneration. Biomacromolecule 2008, 9, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Childs, C.; Edwards-Jones, V.; Heathcote, D.; Dawson, M.; Davenport, P. Patterns of Staphylococcus aureus colonization, toxin production, immunity and illness in burned children. Burns 1994, 20, 514–521. [Google Scholar] [CrossRef]
- Church, D.; Elsayed, S.; Reid, O.; Winston, B.; Lindsay, R. Burn Wound Infections. Clin. Microbiol. Rev. 2006, 19, 403–434. [Google Scholar] [CrossRef] [PubMed]
- Prusinowska, R.; Śmigielski, K.B. Composition, biological properties and therapeutic effects of lavender (Lavandula angustifolia L). A review. Herba Pol. 2014, 60, 56–66. [Google Scholar] [CrossRef]
- Inouye, S.; Takizawa, T.; Yamaguchi, H. Antibacterial activity of essential oils and their major constituents against respiratory tract pathogens by gaseous contact. J. Antimicrob. Chemother. 2001, 47, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Sokovic, M.; Marin, P.D.; Brkic, D.; van Griensven, L.J. Chemical composition and antibacterial activity of essential oils against human pathogenic bacteria. Food 2008, 1, 220–226. [Google Scholar]
- Prashar, A.; Locke, I.C.; Evans, C.S. Cytotoxicity of Lavender Oil and Its Major Components to Human Skin Cells. Cell Prolif. 2004, 37, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Sequeira, R.S.; Miguel, S.P.; Cabral, C.S.; Moreira, A.F.; Ferreira, P.; Correia, I.J. Development of a poly(vinyl alcohol)/lysine electrospun membrane-based drug delivery system for improved skin regeneration. Int. J. Pharm. 2019, 570, 118640. [Google Scholar] [CrossRef] [PubMed]
- Adderley, U.J. Managing Wound Exudate and Promoting Healing. Br. J. Community Nurs. 2010, 15, 15–20. [Google Scholar] [CrossRef]
- Danh, L.T.; Triet, N.D.A.; Han, L.T.N.; Zhao, J.; Mammucari, R.; Foster, N. Antioxidant activity, yield and chemical composition of lavender essential oil extracted by supercritical CO2. J. Supercrit. Fluids 2012, 70, 27–34. [Google Scholar] [CrossRef]
- Balasubramanian, K.; Kodam, K.M. Encapsulation of therapeutic lavender oil in an electrolyte assisted polyacrylonitrile nanofibres for antibacterial applications. RSC Adv. 2014, 4, 54892–54901. [Google Scholar] [CrossRef]
- Li, D.; Xia, Y. Electrospinning of Nanofibers: Reinventing the Wheel? Adv. Mater. 2004, 16, 1151–1170. [Google Scholar] [CrossRef]
- Fong, H.; Chun, I.; Reneker, D. Beaded nanofibers formed during electrospinning. Polymer 1999, 40, 4585–4592. [Google Scholar] [CrossRef]
- Yu, Z.; He, B.; Long, C.; Liu, R.; Sheng, M.; Wang, G.; Tang, J.Z.; Gu, Z. Synthesis, Characterization, and Drug Delivery of Amphiphilic Poly{(Lactic Acid)-Co-[(Glycolic Acid)-Alt-(L-Glutamic Acid)]}-g-Poly(Ethylene Glycol). Macromol. Res. 2012, 20, 250–258. [Google Scholar] [CrossRef]
- Sofi, H.S.; Akram, T.; Tamboli, A.H.; Majeed, A.; Shabir, N.; Sheikh, F.A. Novel lavender oil and silver nanoparticles simultaneously loaded onto polyurethane nanofibers for wound-healing applications. Int. J. Pharm. 2019, 569, 118590. [Google Scholar] [CrossRef]
- Lee, S.J.; Heo, D.N.; Moon, J.-H.; Ko, W.-K.; Lee, J.B.; Bae, M.S.; Park, S.W.; Kim, J.E.; Lee, D.H.; Kim, E.-C.; et al. Electrospun chitosan nanofibers with controlled levels of silver nanoparticles. Preparation, characterization and antibacterial activity. Carbohydr. Polym. 2014, 111, 530–537. [Google Scholar] [CrossRef]
- GhavamiNejad, A.; Rajan Unnithan, A.; Ramachandra Kurup Sasikala, A.; Samarikhalaj, M.; Thomas, R.G.; Jeong, Y.Y.; Nasseri, S.; Murugesan, P.; Wu, D.; Hee Park, C.; et al. Mussel-Inspired Electrospun Nanofibers Functionalized with Size-Controlled Silver Nanoparticles for Wound Dressing Application. ACS Appl. Mater. Interfaces 2015, 7, 12176–12183. [Google Scholar] [CrossRef]
- Sheikh, F.A.; Barakat, N.A.M.; Kanjwal, M.A.; Chaudhari, A.A.; Jung, I.-H.; Lee, J.H.; Kim, H.Y. Electrospun antimicrobial polyurethane nanofibers containing silver nanoparticles for biotechnological applications. Macromol. Res. 2009, 17, 688–696. [Google Scholar] [CrossRef]
- Lara, H.H.; Garza-Treviño, E.N.; Ixtepan-Turrent, L.; Singh, D.K. Silver Nanoparticles Are Broad-Spectrum Bactericidal and Virucidal Compounds. J. Nanobiotechnol. 2011, 9, 30. [Google Scholar] [CrossRef]
- Cavanagh, H.M.A.; Wilkinson, J.M. Biological Activities of Lavender Essential Oil. Wiley Online Libr. 2002, 16, 301–308. [Google Scholar] [CrossRef]
- Liakos, I.; Rizzello, L.; Scurr, D.J.; Pompa, P.P.; Bayer, I.S.; Athanassiou, A. All-natural composite wound dressing films of essential oils encapsulated in sodium alginate with antimicrobial properties. Int. J. Pharm. 2014, 463, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Pant, B.; Park, M.; Park, S.-J. One-Step Synthesis of Silver Nanoparticles Embedded Polyurethane Nano-Fiber/Net Structured Membrane as an Effective Antibacterial Medium. Polymers 2019, 11, 1185. [Google Scholar] [CrossRef] [PubMed]
- Pant, B.; Pant, H.R.; Pandeya, D.R.; Panthi, G.; Nam, K.T.; Hong, S.T.; Kim, C.S.; Kim, H.Y. Characterization and antibacterial properties of Ag NPs loaded nylon-6 nanocomposite prepared by one-step electrospinning process. Colloids Surfaces A Physicochem. Eng. Asp. 2012, 395, 94–99. [Google Scholar] [CrossRef]
- Vafania, B.; Fathi, M.; Soleimanian-Zad, S. Nanoencapsulation of thyme essential oil in chitosan-gelatin nanofibers by nozzle-less electrospinning and their application to reduce nitrite in sausages. Food Bioprod. Process. 2019, 116, 240–248. [Google Scholar] [CrossRef]
- Helander, I.M.; Alakomi, H.L.; Latva-Kala, K.; Mattila-Sandholm, T.; Pol, I.; Smid, E.J.; Gorris, L.G.M.; Von Wright, A. Characterization of the Action of Selected Essential Oil Components on Gram-Negative Bacteria. J. Agric. Food Chem. 1998, 46, 3590–3595. [Google Scholar] [CrossRef]
- Lambert, R.J.W.; Skandamis, P.N.; Coote, P.J.; Nychas, G.J.E. A Study of the Minimum Inhibitory Concentration and Mode of Action of Oregano Essential Oil, Thymol and Carvacrol. J. Appl. Microbiol. 2001, 91, 453–462. [Google Scholar] [CrossRef]
- Lin, L.; Zhu, Y.; Cui, H. Electrospun thyme essential oil/gelatin nanofibers for active packaging against Campylobacter jejuni in chicken. LWT 2018, 97, 711–718. [Google Scholar] [CrossRef]
- Skarp, C.; Hänninen, M.-L.; Rautelin, H. Campylobacteriosis: The role of poultry meat. Clin. Microbiol. Infect. 2016, 22, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Ma, C.; Li, C.; Lin, L. Enhancing the antibacterial activity of thyme oil against Salmonella on eggshell by plasma-assisted process. Food Control 2016, 70, 183–190. [Google Scholar] [CrossRef]
- Lu, H.; Zou, L.; Xu, Y.; Li, Y.V. Controlled Dispersion of Multiwalled Carbon Nanotubes Modified by Hyperbranched Polylysine. J. Appl. Polym. Sci. 2018, 135, 46249. [Google Scholar] [CrossRef]
- Rizwan, M.; Yahya, R.; Hassan, A.; Yar, M.; Anita Omar, R.; Azari, P.; Danial Azzahari, A.; Selvanathan, V.; Rageh Al-Maleki, A.; Venkatraman, G. Synthesis of a Novel Organosoluble, Biocompatible, and Antibacterial Chitosan Derivative for Biomedical Applications. J. Appl. Polym. Sci. 2018, 135, 135. [Google Scholar] [CrossRef]
- López, P.; Sánchez, C.; Batlle, R.; Nerín, C. Development of Flexible Antimicrobial Films Using Essential Oils as Active Agents. J. Agric. Food Chem. 2007, 55, 8814–8824. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Y.; Zhu, Z.; Jiao, X.; Shang, Y.; Wen, Y. Encapsulation of Thymol in Biodegradable Nanofiber via Coaxial Eletrospinning and Applications in Fruit Preservation. ACS Publ. 2019, 67, 1736–1741. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Markel, D.C.; Yu, X.; Shi, T.; Ren, W. Coaxial PCL/PVA Electrospun Nanofibers: Osseointegration Enhancer and Controlled Drug Release Device Erythromycin-Doped Nanofiber Coating to Increase Implant Longevity View Project Infection View Project Coaxial PCL/PVA Electrospun Nanofibers: Osseointegration Enhancer and Controlled Drug Release Device Coaxial PCL/PVA Electrospun Nanofibers: Osseointegration Enhancer and Controlled Drug Release Device. Biofabrication 2013, 5, 35006–35017. [Google Scholar]
- Singh, R.; Ahmed, F.; Polley, P.; Giri, J. Fabrication and Characterization of Core-Shell Nanofibers Using a Next-Generation Airbrush for Biomedical Applications. ACS Appl. Mater. Interfaces 2018, 10, 41924–41934. [Google Scholar] [CrossRef]
- Lin, L.; Liao, X.; Cui, H. Cold plasma treated thyme essential oil/silk fibroin nanofibers against Salmonella Typhimurium in poultry meat. Food Packag. Shelf Life 2019, 21, 100337. [Google Scholar] [CrossRef]
- Cui, H.; Bai, M.; Yuan, L.; Surendhiran, D.; Lin, L. Sequential effect of phages and cold nitrogen plasma against Escherichia coli O157:H7 biofilms on different vegetables. Int. J. Food Microbiol. 2018, 268, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Rajabi, M.; Firouzi, M.; Hassannejad, Z.; Haririan, I. Fabrication and Characterization of Electrospun Laminin-Functionalized Silk Fibroin/Poly (Ethylene Oxide) Nanofibrous Scaffolds for Peripheral Nerve Regeneration Performance Evaluation of Poly (L-Lactide-Co-D, L-Lactide)/Poly (Acrylic Acid) Blends and Their Nanofibers for Tissue Engineering Applications View Project. Artic. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 106, 1595–1604. [Google Scholar]
- Wen, P.; Zhu, D.-H.; Feng, K.; Liu, F.-J.; Lou, W.-Y.; Li, N.; Zong, M.-H.; Wu, H. Fabrication of electrospun polylactic acid nanofilm incorporating cinnamon essential oil/β-cyclodextrin inclusion complex for antimicrobial packaging. Food Chem. 2016, 196, 996–1004. [Google Scholar] [CrossRef] [PubMed]
- Pasquet, J.; Chevalier, Y.; Couval, E.; Bouvier, D.; Noizet, G.; Morlière, C.; Bolzinger, M.-A. Antimicrobial activity of zinc oxide particles on five micro-organisms of the Challenge Tests related to their physicochemical properties. Int. J. Pharm. 2014, 460, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Celebioglu, A.; Umu, O.C.; Tekinay, T.; Uyar, T. Antibacterial electrospun nanofibers from triclosan/cyclodextrin inclusion complexes. Colloids Surfaces B Biointerfaces 2014, 116, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Venkitasamy, C.; Pan, Z.; Wang, W. Recent developments on umami ingredients of edible mushrooms—A review. Trends Food Sci. Technol. 2013, 33, 78–92. [Google Scholar] [CrossRef]
- Pan, J.; Ai, F.; Shao, P.; Chen, H.; Gao, H. Development of Polyvinyl Alcohol/β-Cyclodextrin Antimicrobial Nanofibers for Fresh Mushroom Packaging. Food Chem. 2019, 300, 125249. [Google Scholar] [CrossRef]
- Fu, J.; Qiao, J.L.; Ma, J.X. Highly Stable Solid Alkaline ElectrolyteMembranes fromPoly (vinyl alcohol)/Poly (vinyl pyrrolidone) Based on Chemical Cross-Linking. Acta Phys.-Chim. Sin. 2010, 26, 2975–2981. [Google Scholar]
- Cerniglia, C.E.; Kotarski, S. Approaches in the Safety Evaluations of Veterinary Antimicrobial Agents in Food to Determine the Effects on the Human Intestinal Microflora. J. Vet. Pharmacol. Ther. 2005, 28, 3–20. [Google Scholar] [CrossRef]
- Cevallos, P.A.P.; Buera, M.P.; Elizalde, B.E. Encapsulation of cinnamon and thyme essential oils components (cinnamaldehyde and thymol) in β-cyclodextrin: Effect of interactions with water on complex stability. J. Food Eng. 2010, 99, 70–75. [Google Scholar] [CrossRef]
- Topuz, F.; Uyar, T. Electrospinning of nanocomposite nanofibers from cyclodextrin and laponite. Compos. Commun. 2019, 12, 33–38. [Google Scholar] [CrossRef]
- Lin, L.; Dai, Y.; Cui, H. Antibacterial poly(ethylene oxide) electrospun nanofibers containing cinnamon essential oil/beta-cyclodextrin proteoliposomes. Carbohydr. Polym. 2017, 178, 131–140. [Google Scholar] [CrossRef]
- Séon-Lutz, M.; Couffin, A.-C.; Vignoud, S.; Schlatter, G.; Hébraud, A. Electrospinning in water and in situ crosslinking of hyaluronic acid/cyclodextrin nanofibers: Towards wound dressing with controlled drug release. Carbohydr. Polym. 2019, 207, 276–287. [Google Scholar] [CrossRef]
- Xu, W.; Wu, C. The impact of pulsed light on decontamination, quality, and bacterial attachment of fresh raspberries. Food Microbiol. 2016, 57, 135–143. [Google Scholar] [CrossRef]
- Ajayi, O.; Obadina, A.; Idowu, M.; Adegunwa, M.; Kajihausa, O.; Sanni, L.; Asagbra, Y.; Ashiru, B.; Tomlins, K. Effect of Packaging Materials on the Chemical Composition and Microbiological Quality of Edible Mushroom (Pleurotus Ostreatus) Grown on Cassava Peels. Food Sci. Nutr. 2015, 3, 284–291. [Google Scholar] [CrossRef] [PubMed]
- Nazari, M.; Majdi, H.; Milani, M.; Abbaspour-Ravasjani, S.; Hamishehkar, H.; Lim, L.-T. Cinnamon nanophytosomes embedded electrospun nanofiber: Its effects on microbial quality and shelf-life of shrimp as a novel packaging. Food Packag. Shelf Life 2019, 21, 100349. [Google Scholar] [CrossRef]
- Kamoun, E.A.; Chen, X.; Eldin, M.S.M.; Kenawy, E.-R.S. Crosslinked poly(vinyl alcohol) hydrogels for wound dressing applications: A review of remarkably blended polymers. Arab. J. Chem. 2015, 8, 1–14. [Google Scholar] [CrossRef]
- Han, J.; Lei, T.; Wu, Q. High-water-content mouldable polyvinyl alcohol-borax hydrogels reinforced by well-dispersed cellulose nanoparticles: Dynamic rheological properties and hydrogel formation mechanism. Carbohydr. Polym. 2014, 102, 306–316. [Google Scholar] [CrossRef] [PubMed]
- Adjouman, Y.D.; Nindjin, C.; Tetchi, F.A.; Dalcq, A.C.; Amani, N.G.; Sindic, M. Water vapor permeability of edible films based on improved Cassava (Manihot esculenta Crantz) native starches. J. Food Process. Technol. 2017, 8, 665. [Google Scholar]
- Andreuccetti, C.; Carvalho, R.A.; Galicia-García, T.; Martínez-Bustos, F.; Grosso, C.R. Effect of surfactants on the functional properties of gelatin-based edible films. J. Food Eng. 2011, 103, 129–136. [Google Scholar] [CrossRef]
- Mohammadi, M.; Mirabzadeh, S.; Shahvalizadeh, R.; Hamishehkar, H. Development of novel active packaging films based on whey protein isolate incorporated with chitosan nanofiber and nano-formulated cinnamon oil. Int. J. Biol. Macromol. 2020, 149, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Zhang, Y.; Critzer, F.; Davidson, P.M.; Zivanovic, S.; Zhong, Q. Physical, mechanical, and antimicrobial properties of chitosan films with microemulsions of cinnamon bark oil and soybean oil. Food Hydrocoll. 2016, 52, 533–542. [Google Scholar] [CrossRef]
- Fang, D.; Deng, Z.; Jung, J.; Hu, Q.; Zhao, Y. Mushroom Polysaccharides-Incorporated Cellulose Nanofiber Films with Improved Mechanical, Moisture Barrier, and Antioxidant Properties. J. Appl. Polym. Sci. 2018, 135. [Google Scholar] [CrossRef]
- Arfat, Y.A.; Benjakul, S.; Prodpran, T.; Sumpavapol, P.; Songtipya, P. Properties and antimicrobial activity of fish protein isolate/fish skin gelatin film containing basil leaf essential oil and zinc oxide nanoparticles. Food Hydrocoll. 2014, 41, 265–273. [Google Scholar] [CrossRef]
- Jiang, Y.; Sokorai, K.; Pyrgiotakis, G.; Demokritou, P.; Li, X.; Mukhopadhyay, S.; Jin, T.; Fan, X. Cold Plasma-Activated Hydrogen Peroxide Aerosol Inactivates Escherichia Coli O157:H7, Salmonella Typhimurium, and Listeria Innocua and Maintains Quality of Grape Tomato, Spinach and Cantaloupe. Int. J. Food Microbiol. 2017, 249, 53–60. [Google Scholar] [CrossRef]
- Campion, A.M.; Morrissey, R.M.; Field, D.; Cotter, P.D.; Hill, C.; Ross, R.P. Use of enhanced nisin derivatives in combination with food-grade oils or citric acid to control Cronobacter sakazakii and Escherichia coli O157:H7. Food Microbiol. 2017, 65, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Bai, M.; Lin, L. Plasma-Treated Poly(Ethylene Oxide) Nanofibers Containing Tea Tree Oil/Beta-Cyclodextrin Inclusion Complex for Antibacterial Packaging. Carbohydr. Polym. 2018, 179, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Maslakci, N.N.; Ulusoy, S.; Oksuz, A.U. Investigation of the effects of plasma-treated chitosan electrospun fibers onto biofilm formation. Sensors Actuators B Chem. 2017, 246, 887–895. [Google Scholar] [CrossRef]
- Surucu, S.; Masur, K.; Sasmazel, H.T.; Von Woedtke, T.; Weltmann, K.D. Atmospheric plasma surface modifications of electrospun PCL/chitosan/PCL hybrid scaffolds by nozzle type plasma jets for usage of cell cultivation. Appl. Surf. Sci. 2016, 385, 400–409. [Google Scholar] [CrossRef]
- Cui, H.; Li, W.; Lin, L. Bacterial protease-triggered clove oil release from proteoliposomes against S. aureus biofilms on dried soybean curd. RSC Adv. 2016, 6, 34833–34840. [Google Scholar] [CrossRef]
- Cui, H.; Yuan, L.; Li, W.; Lin, L. Edible Film Incorporated with Chitosan and Artemisia Annua Oil Nanoliposomes for Inactivation of Escherichia Coli O157:H7 on Cherry Tomato. Int. J. Food Sci. Technol. 2017, 52, 687–698. [Google Scholar] [CrossRef]
- Lee, J.Y.; Lee, J.; Ko, S.W.; Son, B.C.; Lee, J.H.; Kim, C.S.; Park, C.H. Fabrication of antibacterial nanofibrous membrane infused with essential oil extracted from tea tree for packaging applications. Polymers 2020, 12, 125. [Google Scholar] [CrossRef]
- Strotmann, U.; Eglsäer, H.; Pagga, U. Development and evaluation of a growth inhibition test with sewage bacteria for assessing bacterial toxicity of chemical compounds. Chemosphere 1994, 28, 755–766. [Google Scholar] [CrossRef]
- Strotmann, U.J.; Pagga, U. A growth inhibition test with sewage bacteria—Results of an international ring test 1995. Chemosphere 1996, 32, 921–933. [Google Scholar] [CrossRef]
- Tarus, B.K.; Mwasiagi, J.I.; Fadel, N.; Aloufy, A.; Mwasiagi, J.I.; Al-Oufy, A.; Elmessiry, M. Electrospun Cellulose Acetate and Poly(Vinyl Chloride) Nanofiber Mats Containing Silver Nanoparticles for Antifungi Packaging. SN Appl. Sci. 2019, 1, 1–12. [Google Scholar] [CrossRef]
- Atarés, L.; Chiralt, A. Essential oils as additives in biodegradable films and coatings for active food packaging. Trends Food Sci. Technol. 2016, 48, 51–62. [Google Scholar] [CrossRef]
- Sánchez-González, L.; González-Martínez, C.; Chiralt, A.; Cháfer, M. Physical and antimicrobial properties of chitosan—Tea tree essential oil composite films. J. Food Eng. 2010, 98, 443–452. [Google Scholar] [CrossRef]
- Silveira, M.P.; Silva, H.C.; Pimentel, I.C.; Poitevin, C.G.; da Costa Stuart, A.K.; Carpiné, D.; de Matos Jorge, L.M.; Jorge, R.M.M. Development of Active Cassava Starch Cellulose Nanofiber-Based Films Incorporated with Natural Antimicrobial Tea Tree Essential Oil. J. Appl. Polym. Sci. 2020, 137, 137. [Google Scholar] [CrossRef]
- Jiménez, A.; Fabra, M.J.; Talens, P.; Chiralt, A. Edible and Biodegradable Starch Films: A Review. Food Bioprocess Technol. 2012, 5, 2058–2076. [Google Scholar] [CrossRef]
- Azeredo, H.M.; Rosa, M.F.; Mattoso, L.H.C. Nanocellulose in bio-based food packaging applications. Ind. Crop. Prod. 2017, 97, 664–671. [Google Scholar] [CrossRef]
- Muhammad Shamsuddin, I.; Ahmad Jafar, J.; Sadiq Abdulrahman Shawai, A.; Yusuf, S.; Lateefah, M.; Aminu, I. Bioplastics as Better Alternative to Petroplastics and Their Role in National Sustainability: A Review. Adv. Biosci. Bioeng. 2017, 5, 63–70. [Google Scholar] [CrossRef]
- Panashe, J.A.; Danyuo, Y. Recycling of Plastic Waste Materials: Mechanical Properties and Implications for Road Construction. MRS Adv. 2020, 5, 1305–1312. [Google Scholar] [CrossRef]
- Vogler, E.A. Structure and reactivity of water at biomaterial surfaces. Adv. Colloid Interface Sci. 1998, 74, 69–117. [Google Scholar] [CrossRef]
- Mali, S.; Grossmann, M.V.E.; Yamashita, F. Filmes de amido: Produção, propriedades e potencial de utilização. Semin. Cien. Agrár. 2010, 31, 137. [Google Scholar] [CrossRef]
- Marques, G.S.; de Carvalho, G.R.; Marinho, N.P.; de Muniz, G.I.B.; de Matos Jorge, L.M.; Jorge, R.M.M. Production and Characterization of Starch-Based Films Reinforced by Ramie Nanofibers (Boehmeria Nivea). J. Appl. Polym. Sci. 2019, 136, 136. [Google Scholar] [CrossRef]
- Carpiné, D.; Dagostin, J.L.A.; De Andrade, E.F.; Bertan, L.C.; Mafra, M.R. Effect of the natural surfactant Yucca schidigera extract on the properties of biodegradable emulsified films produced from soy protein isolate and coconut oil. Ind. Crop. Prod. 2016, 83, 364–371. [Google Scholar] [CrossRef]
- Debiagi, F.; Kobayashi, R.K.; Nakazato, G.; Panagio, L.A.; Mali, S. Biodegradable active packaging based on cassava bagasse, polyvinyl alcohol and essential oils. Ind. Crop. Prod. 2014, 52, 664–670. [Google Scholar] [CrossRef]
- Ge, Y.; Ge, M. Distribution of Melaleuca Alternifolia Essential Oil in Liposomes with Tween 80 Addition and Enhancement of in Vitro Antimicrobial Effect. J. Exp. Nanosci. 2016, 11, 345–358. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Griffin, S.G.; Markham, J.L.; Leach, D.N. An Agar Dilution Method for the Determination of the Minimum Inhibitory Concentration of Essential Oils. J. Essent. Oil Res. 2000, 12, 249–255. [Google Scholar] [CrossRef]
- Unalan, I.; Slavik, B.; Buettner, A.; Goldmann, W.H.; Frank, G.; Boccaccini, A.R. Physical and Antibacterial Properties of Peppermint Essential Oil Loaded Poly (ε-Caprolactone) (PCL) Electrospun Fiber Mats for Wound Healing. Front. Bioeng. Biotechnol. 2019, 7, 346. [Google Scholar] [CrossRef] [PubMed]
- Reverchon, E.; Ambruosi, A.; Senatore, F. Isolation of Peppermint Oil Using Supercritical Co2 Extraction. Flavour Fragr. J. 1994, 9, 19–23. [Google Scholar] [CrossRef]
- Rohloff, J. Monoterpene Composition of Essential Oil from Peppermint (Mentha x Piperita L.) with Regard to Leaf Position Using Solid-Phase Microextraction and Gas Chromatography/Mass Spectrometry Analysis. J. Agric. Food Chem. 1999, 47, 3782–3786. [Google Scholar] [CrossRef]
- Schulz, H.; Baranska, M.; Belz, H.-H.; Rösch, P.; A Strehle, M.; Popp, J. Chemotaxonomic characterisation of essential oil plants by vibrational spectroscopy measurements. Vib. Spectrosc. 2004, 35, 81–86. [Google Scholar] [CrossRef]
- Rösch, P.; Kiefer, W.; Popp, J. Chemotaxonomy of mints of genus Mentha by applying Raman spectroscopy. Biopolym. Orig. Res. Biomol. 2002, 67, 358–361. [Google Scholar]
- Vargas Jentzsch, P.; Ramos, L.A.; Ciobotă, V. Handheld Raman Spectroscopy for the Distinction of Essential Oils Used in the Cosmetics Industry. Cosmetics 2015, 2, 162–176. [Google Scholar] [CrossRef]
- Wesełucha-Birczyńska, A.; Świętek, M.; Sołtysiak, E.; Galiński, P.; Płachta, Ł.; Piekara, K.; Błażewicz, M. Raman Spectroscopy and the Material Study of Nanocomposite Membranes from Poly(ε-Caprolactone) with Biocompatibility Testing in Osteoblast-like Cells. Analyst 2015, 140, 2311. [Google Scholar] [CrossRef]
- Jin, G.; Prabhakaran, M.P.; Kai, D.; Annamalai, S.K.; Arunachalam, K.D.; Ramakrishna, S. Tissue Engineered Plant Extracts as Nanofibrous Wound Dressing. Biomaterials 2013, 34, 724–734. [Google Scholar] [CrossRef]
- McKay, D.L.; Blumberg, J.B. A Review of the Bioactivity and Potential Health Benefits of Peppermint Tea (Mentha Piperita L.). Phytother. Res. 2006, 20, 619–633. [Google Scholar] [CrossRef]
- Jaganathan, S.K.; Mani, M.P.; Khudzari, A.Z.M. Electrospun Combination of Peppermint Oil and Copper Sulphate with Conducive Physico-Chemical properties for Wound Dressing Applications. Polymers 2019, 11, 586. [Google Scholar] [CrossRef]
- Jaganathan, S.K.; Mani, M.P.; Palaniappan, S.K.; Rathanasamy, R. Fabrication and characterisation of nanofibrous polyurethane scaffold incorporated with corn and neem oil using single stage electrospinning technique for bone tissue engineering applications. J. Polym. Res. 2018, 25, 146. [Google Scholar] [CrossRef]
- Detta, N.; Errico, C.; Dinucci, D.; Puppi, D.; Clarke, D.A.; Reilly, G.C.; Chiellini, F. Novel electrospun polyurethane/gelatin composite meshes for vascular grafts. J. Mater. Sci. Mater. Med. 2010, 21, 1761–1769. [Google Scholar] [CrossRef]
- Amna, T.; Hassan, M.; Yang, J.; Khil, M.-S.; Song, K.-D.; Hwang, I.; Oh, J.-D. Virgin olive oil blended polyurethane micro/nanofibers ornamented with copper oxide nanocrystals for biomedical applications. Int. J. Nanomed. 2014, 9, 891–898. [Google Scholar] [CrossRef]
- Jaganathan, S.K.; Mani, M.P. Electrospun polyurethane nanofibrous composite impregnated with metallic copper for wound-healing application. 3 Biotech 2018, 8, 327. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.H.; Kim, M.J.; Ryu, S.J.; Ki, C.S.; Park, Y.H. Effect of Fiber Diameter on Surface Morphology, Mechanical Property, and Cell Behavior of Electrospun Poly(ε-Caprolactone) Mat. Fibers Polym. 2016, 17, 1033–1042. [Google Scholar] [CrossRef]
- Jaganathan, S.K.; Mani, M.P. Single-stage synthesis of electrospun polyurethane scaffold impregnated with zinc nitrate nanofibers for wound healing applications. J. Appl. Polym. Sci. 2019, 136, 136. [Google Scholar] [CrossRef]
- Jaganathan, S.K.; Balaji, A.; Ismail, A.F.; Rajasekar, R. Fabrication and hemocompatibility assessment of novel polyurethane-based bio-nanofibrous dressing loaded with honey and Carica papaya extract for the management of burn injuries. Int. J. Nanomed. 2016, 11, 4339–4355. [Google Scholar] [CrossRef]
- Pitz, H.D.S.; Pereira, A.; Blasius, M.B.; Voytena, A.P.L.; Affonso, R.C.L.; Fanan, S.; Trevisan, A.C.D.; Ribeiro-Do-Valle, R.M.; Maraschin, M. In VitroEvaluation of the Antioxidant Activity and Wound Healing Properties of Jaboticaba (Plinia peruviana) Fruit Peel Hydroalcoholic Extract. Oxidative Med. Cell. Longev. 2016, 2016, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Hosseinkhani, H.; Hosseinkhani, M.; Khademhosseini, A.; Yokoyama, Y.; Estrada, G.G.; Kobayashi, H. Quantitative Analysis of Cell Adhesion on Aligned Micro- and Nanofibers. J. Biomed. Mater. Res. Part A 2008, 84, 291–299. [Google Scholar] [CrossRef]
- Nandakumar, V.; Suresh, G.; Chittaranjan, S.; Doble, M. Synthesis and Characterization of Hydrophilic High Glycolic Acid− Poly(DL-Lactic-Co-Glycolic Acid)/Polycaprolactam/Polyvinyl Alcohol Blends and Their Biomedical Application as a Ureteral Material. ACS Publ. 2012, 52, 751–760. [Google Scholar] [CrossRef]
- Cui, H.; Zhao, C.; Lin, L. The specific antibacterial activity of liposome-encapsulated Clove oil and its application in tofu. Food Control 2015, 56, 128–134. [Google Scholar] [CrossRef]
- Mulla, M.; Ahmed, J.; Al-Attar, H.; Castro-Aguirre, E.; Arfat, Y.A.; Auras, R. Antimicrobial efficacy of clove essential oil infused into chemically modified LLDPE film for chicken meat packaging. Food Control 2017, 73, 663–671. [Google Scholar] [CrossRef]
- Cui, H.; Bai, M.; Rashed, M.M.; Lin, L. The antibacterial activity of clove oil/chitosan nanoparticles embedded gelatin nanofibers against Escherichia coli O157:H7 biofilms on cucumber. Int. J. Food Microbiol. 2018, 266, 69–78. [Google Scholar] [CrossRef]
- Agheb, M.; Dinari, M.; Rafienia, M.; Salehi, H. Novel electrospun nanofibers of modified gelatin-tyrosine in cartilage tissue engineering. Mater. Sci. Eng. C 2017, 71, 240–251. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Lim, S.; Birajdar, M.S.; Lee, S.-H.; Park, H. Fabrication of FGF-2 immobilized electrospun gelatin nanofibers for tissue engineering. Int. J. Biol. Macromol. 2016, 93, 1559–1566. [Google Scholar] [CrossRef]
- Unalan, I.; Endlein, S.J.; Slavik, B.; Buettner, A.; Goldmann, W.H.; Detsch, R.; Boccaccini, A.R. Evaluation of Electrospun Poly(ε-Caprolactone)/Gelatin Nanofiber Mats Containing Clove Essential Oil for Antibacterial Wound Dressing. Pharmaceutics 2019, 11, 570. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.; Harding, K.G. Bacteria and wound healing. Curr. Opin. Infect. Dis. 2004, 17, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Priya, S.G.; Jungvid, H.; Kumar, A. Skin Tissue Engineering for Tissue Repair and Regeneration. Tissue Eng. Part B Rev. 2008, 14, 105–118. [Google Scholar] [CrossRef]
- Silva, N.; Júnior, A.F. Biological properties of medicinal plants: A review of their antimicrobial activity. J. Venom. Anim. Toxins Incl. Trop. Dis. 2010, 16, 402–413. [Google Scholar] [CrossRef]
- Sadri, M.; Arab-Sorkhi, S.; Vatani, H.; Bagheri-Pebdeni, A. New Wound Dressing Polymeric Nanofiber Containing Green Tea Extract Prepared by Electrospinning Method. Fibers Polym. 2015, 16, 1742–1750. [Google Scholar] [CrossRef]
- Chemat, F.; Vian, M.A.; Cravotto, G. Green Extraction of Natural Products: Concept and Principles. Int. J. Mol. Sci. 2012, 13, 8615–8627. [Google Scholar] [CrossRef]
- Liverani, L.; Boccaccini, A.R. Versatile Production of Poly(Epsilon-Caprolactone) Fibers by Electrospinning Using Benign Solvents. Nanomaterials 2016, 6, 75. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.-C.; Siedlecki, C.A. Effects of surface wettability and contact time on protein adhesion to biomaterial surfaces. Biomaterials 2007, 28, 3273–3283. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, B.; Marques, A.; Ramos, C.; Neng, N.R.; Nogueira, J.M.; Saraiva, J.A.; Nunes, M.L. Chemical composition and antibacterial and antioxidant properties of commercial essential oils. Ind. Crop. Prod. 2013, 43, 587–595. [Google Scholar] [CrossRef]
- Khan, A.U.R.; Nadeem, M.; Bhutto, M.A.; Yu, F.; Xie, X.; El-Hamshary, H.; El-Faham, A.; Ibrahim, U.A.; Mo, X. Physico-Chemical and Biological Evaluation of PLCL/SF Nanofibers Loaded with Oregano Essential Oil. Pharmaceutics 2019, 11, 386. [Google Scholar] [CrossRef] [PubMed]
- Bagherzadeh, R.; Najar, S.S.; Latifi, M.; Tehran, M.A.; Kong, L. A Theoretical Analysis and Prediction of Pore Size and Pore Size Distribution in Electrospun Multilayer Nanofibrous Materials. J. Biomed. Mater. Res. Part A 2013, 101, 2107–2117. [Google Scholar] [CrossRef]
- Aderibigbe, B.A.; Buyana, B. Alginate in Wound Dressings. Pharmaceutics 2018, 10, 42. [Google Scholar]
- Kumar, P.T.S.; Lakshmanan, V.-K.; Biswas, R.; Nair, S.V.; Jayakumar, R. Synthesis and Biological Evaluation of Chitin Hydrogel/Nano ZnO Composite Bandage as Antibacterial Wound Dressing. J. Biomed. Nanotechnol. 2012, 8, 891–900. [Google Scholar] [CrossRef]
- Zhang, K.; Wang, H.; Huang, C.; Su, Y.; Mo, X.; Ikada, Y. Fabrication of Silk Fibroin Blended P(LLA-CL) Nanofibrous Scaffolds for Tissue Engineering. J. Biomed. Mater. Res. Part A 2010, 93, 984–993. [Google Scholar] [CrossRef]
- Kijeńska, E.; Prabhakaran, M.P.; Swieszkowski, W.; Kurzydlowski, K.J.; Ramakrishna, S. Electrospun bio-composite P (LLA-CL)/collagen I/collagen III scaffolds for nerve tissue engineering. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100, 1093–1102. [Google Scholar] [CrossRef] [PubMed]
- Avila-Sosa, R.; Hernández-Zamoran, E.; López-Mendoza, I.; Palou, E.; Munguía, M.T.J.; Nevarez-Moorillon, G.V.; López-Malo, A. Fungal Inactivation by Mexican Oregano (Lippia berlandieri Schauer) Essential Oil Added to Amaranth, Chitosan, or Starch Edible Films. J. Food Sci. 2010, 75, M127–M133. [Google Scholar] [CrossRef] [PubMed]
- Acosta, S.; Chiralt, A.; Santamarina, P.; Rosello, J.; González-Martínez, C.; Cháfer, M. Antifungal films based on starch-gelatin blend, containing essential oils. Food Hydrocoll. 2016, 61, 233–240. [Google Scholar] [CrossRef]
- Hu, X.; Jia, X.; Zhi, C.; Jin, Z.; Miao, M. Improving the properties of starch-based antimicrobial composite films using ZnO-chitosan nanoparticles. Carbohydr. Polym. 2019, 210, 204–209. [Google Scholar] [CrossRef]
- Li, J.; Ye, F.; Lei, L.; Zhao, G. Combined effects of octenylsuccination and oregano essential oil on sweet potato starch films with an emphasis on water resistance. Int. J. Biol. Macromol. 2018, 115, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Alsammarraie, F.K.; Nayigiziki, F.X.; Wang, W.; Vardhanabhuti, B.; Mustapha, A.; Lin, M. Effect and mechanism of cellulose nanofibrils on the active functions of biopolymer-based nanocomposite films. Food Res. Int. 2017, 99, 166–172. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Preparation of carrageenan-based functional nanocomposite films incorporated with melanin nanoparticles. Colloids Surfaces B Biointerfaces 2019, 176, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhou, M.; Cheng, G.; Cheng, F.; Lin, Y.; Zhu, P.-X. Fabrication and characterization of starch-based nanocomposites reinforced with montmorillonite and cellulose nanofibers. Carbohydr. Polym. 2019, 210, 429–436. [Google Scholar] [CrossRef]
- Davachi, S.M.; Shekarabi, A.S. Preparation and characterization of antibacterial, eco-friendly edible nanocomposite films containing Salvia macrosiphon and nanoclay. Int. J. Biol. Macromol. 2018, 113, 66–72. [Google Scholar] [CrossRef]
- Aguilar-Sánchez, R.; Munguía-Pérez, R.; Reyes-Jurado, F.; Navarro-Cruz, A.R.; Cid-Pérez, T.S.; Hernández-Carranza, P.; Beristain-Bauza, S.D.C.; Ochoa-Velasco, C.E.; Avila-Sosa, R. Structural, Physical, and Antifungal Characterization of Starch Edible Films Added with Nanocomposites and Mexican Oregano (Lippia berlandieri Schauer) Essential Oil. Molecules 2019, 24, 2340. [Google Scholar]
- Flaker, C.H.; Lourenço, R.V.; Bittante, A.M.; Sobral, P.J. Gelatin-based nanocomposite films: A study on montmorillonite dispersion methods and concentration. J. Food Eng. 2015, 167, 65–70. [Google Scholar] [CrossRef]
- Brigatti, M.; Galan, E.; Theng, B. Chapter 2 Structures and Mineralogy of Clay Minerals. Dev. Clay Sci. 2006, 1, 19–86. [Google Scholar]
- Escamilla-García, M.; Calderón-Domínguez, G.; Chanona-Pérez, J.J.; Mendoza-Madrigal, A.G.; Di Pierro, P.; García-Almendárez, B.E.; Amaro-Reyes, A.; Regalado-González, C. Physical, Structural, Barrier, and Antifungal Characterization of Chitosan–Zein Edible Films with Added Essential Oils. Int. J. Mol. Sci. 2017, 18, 2370. [Google Scholar] [CrossRef]
- Srinivasa, P.C.; Tharanathan, R.N. Chitin/Chitosan—Safe, Ecofriendly Packaging Materials with Multiple Potential Uses. Food Rev. Int. 2007, 23, 53–72. [Google Scholar] [CrossRef]
- Ponce, A.G.; Roura, S.I.; del Valle, C.E.; Moreira, M.R. Antimicrobial and antioxidant activities of edible coatings enriched with natural plant extracts: In vitro and in vivo studies. Postharvest Biol. Technol. 2008, 49, 294–300. [Google Scholar] [CrossRef]
- Alizadeh-Sani, M.; Khezerlou, A.; Ehsani, A. Fabrication and characterization of the bionanocomposite film based on whey protein biopolymer loaded with TiO2 nanoparticles, cellulose nanofibers and rosemary essential oil. Ind. Crop. Prod. 2018, 124, 300–315. [Google Scholar] [CrossRef]
- Sothornvit, R.; Hong, S.-I.; An, D.J.; Rhim, J.-W. Effect of clay content on the physical and antimicrobial properties of whey protein isolate/organo-clay composite films. LWT 2010, 43, 279–284. [Google Scholar] [CrossRef]
- Zolfi, M.; Khodaiyan, F.; Mousavi, M.; Hashemi, M. The improvement of characteristics of biodegradable films made from kefiran–whey protein by nanoparticle incorporation. Carbohydr. Polym. 2014, 109, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Almasi, H.; Ghanbarzadeh, B.; Entezami, A.A. Physicochemical properties of starch–CMC–nanoclay biodegradable films. Int. J. Biol. Macromol. 2010, 46, 1–5. [Google Scholar] [CrossRef]
- Li, H.; Pu, Y.; Kumar, R.; Ragauskas, A.J.; Wyman, C.E. Investigation of Lignin Deposition on Cellulose during Hydrothermal Pretreatment, Its Effect on Cellulose Hydrolysis, and Underlying Mechanisms. Biotechnol. Bioeng. 2014, 111, 485–492. [Google Scholar] [CrossRef]
- Zolfi, M.; Khodaiyan, F.; Mousavi, M.; Hashemi, M. Development and characterization of the kefiran-whey protein isolate-TiO2 nanocomposite films. Int. J. Biol. Macromol. 2014, 65, 340–345. [Google Scholar] [CrossRef]
- Sani, M.A.; Ehsani, A.; Hashemi, M. Whey protein isolate/cellulose nanofibre/TiO 2 nanoparticle/rosemary essential oil nanocomposite film: Its effect on microbial and sensory quality of lamb meat and growth of common foodborne pathogenic bacteria during refrigeration. Int. J. Food Microbiol. 2017, 251, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Amjadi, S.; Almasi, H.; Ghorbani, M.; Ramazani, S. Reinforced ZnONPs/ rosemary essential oil-incorporated zein electrospun nanofibers by κ-carrageenan. Carbohydr. Polym. 2020, 232, 115800. [Google Scholar] [CrossRef] [PubMed]
- Torres-Giner, S.; Ocio, M.J.; Lagaron, J.M. Novel antimicrobial ultrathin structures of zein/chitosan blends obtained by electrospinning. Carbohydr. Polym. 2009, 77, 261–266. [Google Scholar] [CrossRef]
- Nouri, A.; Yaraki, M.T.; Ghorbanpour, M.; Wang, S. Biodegradable κ-carrageenan/nanoclay nanocomposite films containing Rosmarinus officinalis L. extract for improved strength and antibacterial performance. Int. J. Biol. Macromol. 2018, 115, 227–235. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Carrageenan-based antimicrobial bionanocomposite films incorporated with ZnO nanoparticles stabilized by melanin. Food Hydrocoll. 2019, 90, 500–507. [Google Scholar] [CrossRef]
- Sekar, A.D.; Kumar, V.; Muthukumar, H.; Gopinath, P.; Matheswaran, M. Electrospinning of Fe-doped ZnO nanoparticles incorporated polyvinyl alcohol nanofibers for its antibacterial treatment and cytotoxic studies. Eur. Polym. J. 2019, 118, 27–35. [Google Scholar] [CrossRef]
- Ignatova, M.; Manolova, N.; Rashkov, I.; Markova, N. Quaternized chitosan/κ-carrageenan/caffeic acid–coated poly(3-hydroxybutyrate) fibrous materials: Preparation, antibacterial and antioxidant activity. Int. J. Pharm. 2016, 513, 528–537. [Google Scholar] [CrossRef] [PubMed]
- Fabra, M.J.; López-Rubio, A.; Lagaron, J.M. Use of the electrohydrodynamic process to develop active/bioactive bilayer films for food packaging applications. Food Hydrocoll. 2016, 55, 11–18. [Google Scholar] [CrossRef]
- Abdollahi, M.; Rezaei, M.; Farzi, G. A novel active bionanocomposite film incorporating rosemary essential oil and nanoclay into chitosan. J. Food Eng. 2012, 111, 343–350. [Google Scholar] [CrossRef]
- Goonoo, N.; Khanbabaee, B.; Steuber, M.; Bhaw-Luximon, A.; Jonas, U.; Pietsch, U.; Jhurry, D.; Schönherr, H. κ-Carrageenan Enhances the Biomineralization and Osteogenic Differentiation of Electrospun Polyhydroxybutyrate and Polyhydroxybutyrate Valerate Fibers. Biomacromolecules 2017, 18, 1563–1573. [Google Scholar] [CrossRef]
- Kumar, S.; Rajan Unnithan, A.; Gnanasekaran, G.; Sathishkumar, Y.; Lee, Y.S.; Kim, C.S. Electrospun Antibacterial Polyurethane-Cellulose Acetate-Zein Composite Mats for Wound Dressing. Carbohydr. Polym. 2014, 102, 884–892. [Google Scholar]
- Amjadi, S.; Emaminia, S.; Davudian, S.H.; Pourmohammad, S.; Hamishehkar, H.; Roufegarinejad, L. Preparation and characterization of gelatin-based nanocomposite containing chitosan nanofiber and ZnO nanoparticles. Carbohydr. Polym. 2019, 216, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Khaledian, Y.; Pajohi-Alamoti, M.; Bazargani-Gilani, B. Development of cellulose nanofibers coating incorporated with ginger essential oil and citric acid to extend the shelf life of ready-to-cook barbecue chicken. J. Food Process. Preserv. 2019, 43, 43. [Google Scholar] [CrossRef]
- Ercolini, D.; Russo, F.; Torrieri, E.; Masi, P.; Villani, F. Changes in the Spoilage-Related Microbiota of Beef during Refrigerated Storage under Different Packaging Conditions. Appl. Environ. Microbiol. 2006, 72, 4663–4671. [Google Scholar] [CrossRef]
- Frangos, L.; Pyrgotou, N.; Giatrakou, V.; Ntzimani, A.; Savvaidis, I. Combined effects of salting, oregano oil and vacuum-packaging on the shelf-life of refrigerated trout fillets. Food Microbiol. 2010, 27, 115–121. [Google Scholar] [CrossRef]
- Krishnan, K.R.; Babuskin, S.; Babu, P.A.S.; Sasikala, M.; Sabina, K.; Archana, G.; Sivarajan, M.; Sukumar, M. Antimicrobial and antioxidant effects of spice extracts on the shelf life extension of raw chicken meat. Int. J. Food Microbiol. 2014, 171, 32–40. [Google Scholar] [CrossRef]
- Tattari, S.; Kota, N.; Technical Officer, S.; Nimgulkar MPharm Scholar, C.; Polasa, K.; Panpatil, V.V.; Nimgulkar, C. In Vitro Evaluation on Antioxidant and Antimicrobial Activity of Spice Extracts of Ginger, Turmeric and Garlic. J. Pharmacogn. Phytochem. 2013, 2, 143–148. [Google Scholar]
- Sasidharan, I.; Menon, A.N. Comparative chemical composition and antimicrobial activity fresh & dry ginger oils (Zingiber officinale Roscoe). Int. J. Curr. Pharm. Res. 2010, 2, 40–43. [Google Scholar]
- Sivasothy, Y.; Chong, W.K.; Hamid, A.; Eldeen, I.M.; Sulaiman, S.F.; Awang, K. Essential oils of Zingiber officinale var. rubrum Theilade and their antibacterial activities. Food Chem. 2011, 124, 514–517. [Google Scholar] [CrossRef]
- Alvarado, C.; Brooks, J.C.; Echeverry, A.; Brashears, M.M. Validation of a Lactic Acid-and Citric Acid-Based Antimicrobial Product for the Reduction of Escherichia Coli O157:H7 and Salmonella on Beef Tips and Whole Chicken Carcasses Internalization and Thermal Susceptibility of Shiga Toxin-Producing Escherichia Coli (STEC) in Marinated Beef Products View Project Beef Flavor View Project. Artic. J. Food Prot. 2009, 72, 2208–2211. [Google Scholar]
- Sánchez-Ortega, I.; García-Almendárez, B.E.; Santos-López, E.M.; Amaro-Reyes, A.; Barboza-Corona, J.E.; Regalado, C. Antimicrobial Edible Films and Coatings for Meat and Meat Products Preservation. Sci. World J. 2014, 2014, 1–18. [Google Scholar] [CrossRef]
- Wu, Y.; Luo, X.; Li, W.; Song, R.; Li, J.; Li, Y.; Li, B.; Liu, S. Green and Biodegradable Composite Films with Novel Antimicrobial Performance Based on Cellulose. Food Chem. 2016, 197, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Bag, A.; Chattopadhyay, R.R. Evaluation of Synergistic Antibacterial and Antioxidant Efficacy of Essential Oils of Spices and Herbs in Combination. PLoS ONE 2015, 10, e0131321. [Google Scholar] [CrossRef]
- Tang, Y.; Zhou, Y.; Lan, X.; Huang, D.; Luo, T.; Ji, J.; Mafang, Z.; Miao, X.; Wang, H.; Wang, W. Electrospun Gelatin Nanofibers Encapsulated with Peppermint and Chamomile Essential Oils as Potential Edible Packaging. J. Agric. Food Chem. 2019, 67, 2227–2234. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.; Benjakul, S.; Prodpran, T.; Agustini, T.W. Physico-mechanical and antimicrobial properties of gelatin film from the skin of unicorn leatherjacket incorporated with essential oils. Food Hydrocoll. 2012, 28, 189–199. [Google Scholar] [CrossRef]
- Celebioglu, A.; Irem Yildiz, Z.; Uyar, T. Thymol/Cyclodextrin Inclusion Complex Nanofibrous Webs: Enhanced Water Solubility, High Thermal Stability and Antioxidant Property of Thymol. Food Res. Int. 2018, 106, 280–290. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Deng, L.; Zhang, C.; Feng, F.; Zhang, H. Tunable Physical Properties of Ethylcellulose/Gelatin Composite Nanofibers by Electrospinning. J. Agric. Food Chem. 2018, 66, 1907–1915. [Google Scholar] [CrossRef]
- Figueroa-Lopez, K.J.; Castro-Mayorga, J.L.; Andrade-Mahecha, M.M.; Cabedo, L.; Lagaron, J.M. Antibacterial and Barrier Properties of Gelatin Coated by Electrospun Polycaprolactone Ultrathin Fibers Containing Black Pepper Oleoresin of Interest in Active Food Biopackaging Applications. Nanomaterials 2018, 8, 199. [Google Scholar] [CrossRef] [PubMed]
- Luchese, C.L.; Pavoni, J.M.F.; dos Santos, N.Z.; Quines, L.K.; Pollo, L.D.; Spada, J.C.; Tessaro, I.C. Effect of Chitosan Addition on the Properties of Films Prepared with Corn and Cassava Starches. J. Food Sci. Technol. 2018, 55, 2963–2973. [Google Scholar] [CrossRef]
- Khazaei, N.; Esmaiili, M.; Djomeh, Z.E.; Ghasemlou, M.; Jouki, M. Characterization of new biodegradable edible film made from basil seed (Ocimum basilicum L.) gum. Carbohydr. Polym. 2014, 102, 199–206. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; De Martino, L.; Coppola, R.; De Feo, V. Effect of Essential Oils on Pathogenic Bacteria. Pharmaceuticals 2013, 6, 1451–1474. [Google Scholar] [CrossRef]
- Higueras, L.; López-Carballo, G.; Gavara, R.; Hernández-Muñoz, P. Reversible Covalent Immobilization of Cinnamaldehyde on Chitosan Films via Schiff Base Formation and Their Application in Active Food Packaging. Food Bioprocess Technol. 2014, 8, 526–538. [Google Scholar] [CrossRef]
- Ohtsu, N.; Kohari, Y.; Gotoh, M.; Yamada, R.; Nagata, Y.; Murata, M. Utilization of the Japanese Peppermint Herbal Water Byproduct of Steam Distillation as an Antimicrobial Agent. J. Oleo Sci. 2018, 67, 1227–1233. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Zhang, J.; Fan, J.; Clark, J.; Shen, P.; Li, Y.; Zhang, C. Microwave assisted extraction of phenolic compounds from four economic brown macroalgae species and evaluation of their antioxidant activities and inhibitory effects on α-amylase, α-glucosidase, pancreatic lipase and tyrosinase. Food Res. Int. 2018, 113, 288–297. [Google Scholar] [CrossRef]
- Zhou, Y.; Miao, X.; Lan, X.; Luo, J.; Luo, T.; Zhong, Z.; Gao, X.; Mafang, Z.; Ji, J.; Wang, H.; et al. Angelica Essential Oil Loaded Electrospun Gelatin Nanofibers for Active Food Packaging Application. Polymers 2020, 12, 299. [Google Scholar] [CrossRef]
- Siracusa, V.; Romani, S.; Gigli, M.; Mannozzi, C.; Cecchini, J.P.; Tylewicz, U.; Lotti, N. Characterization of Active Edible Films based on Citral Essential Oil, Alginate and Pectin. Materials 2018, 11, 1980. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, A.; Shaw, N.B.; Murphy, S.C.; Van De Vis, J.W.; Van Pelt-Heerschap, H.; Kerry, J.P. Extraction of Collagen from Fish Skins and Its Use in the Manufacture of Biopolymer Films. J. Aquat. Food Prod. Technol. 2006, 15, 21–32. [Google Scholar] [CrossRef]
- Turi, C.E.; Murch, S.J. Targeted and Untargeted Phytochemistry of Ligusticum canbyi: Indoleamines, Phthalides, Antioxidant Potential, and Use of Metabolomics as a Hypothesis-Generating Technique for Compound Discovery. Planta Medica 2013, 79, 1370–1379. [Google Scholar] [CrossRef] [PubMed]
- Kanmani, P.; Rhim, J.-W. Physical, mechanical and antimicrobial properties of gelatin based active nanocomposite films containing AgNPs and nanoclay. Food Hydrocoll. 2014, 35, 644–652. [Google Scholar] [CrossRef]
- Lin, L.; Mao, X.; Sun, Y.; Rajivgandhi, G.; Cui, H. Antibacterial Properties of Nanofibers Containing Chrysanthemum Essential Oil and Their Application as Beef Packaging. Int. J. Food Microbiol. 2019, 292, 21–30. [Google Scholar] [CrossRef]
- Clara, R.; Langhans, W.; Mansouri, A. Oleic acid stimulates glucagon-like peptide-1 release from enteroendocrine cells by modulating cell respiration and glycolysis. Metabolism 2016, 65, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Rieger, K.A.; Birch, N.P.; Schiffman, J.D. Designing electrospun nanofiber mats to promote wound healing—A review. J. Mater. Chem. B 2013, 1, 4531–4541. [Google Scholar] [CrossRef] [PubMed]
- Chouliara, E.; Karatapanis, A.; Savvaidis, I.; Kontominas, M. Combined effect of oregano essential oil and modified atmosphere packaging on shelf-life extension of fresh chicken breast meat, stored at 4 °C. Food Microbiol. 2007, 24, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Menon, A.N.; Padmakumari, K.P. Studies on Essential Oil Composition of Cultivars of Black Pepper (Piper Nigrum L.)—V. J. Essent. Oil Res. 2005, 17, 153–155. [Google Scholar] [CrossRef]

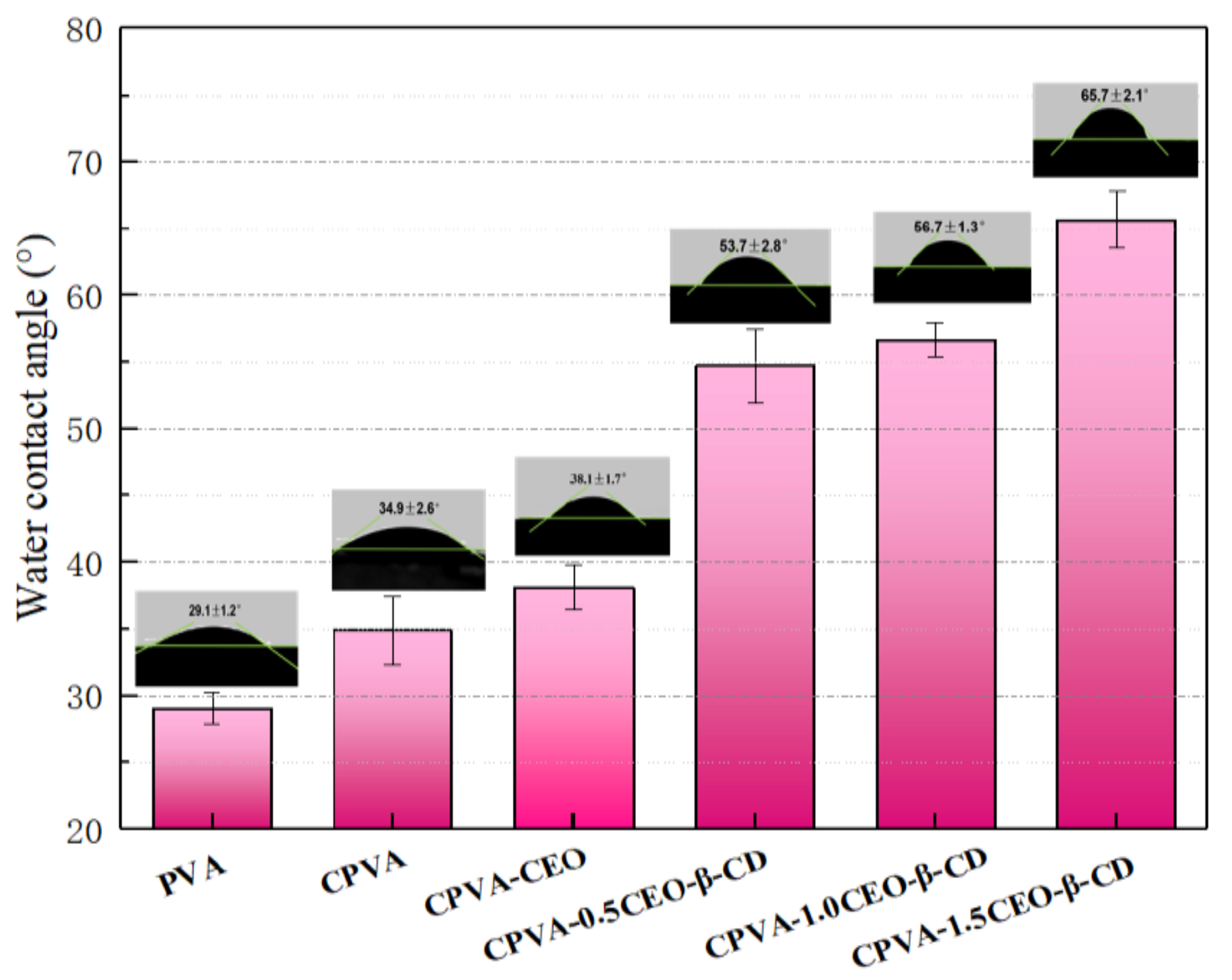
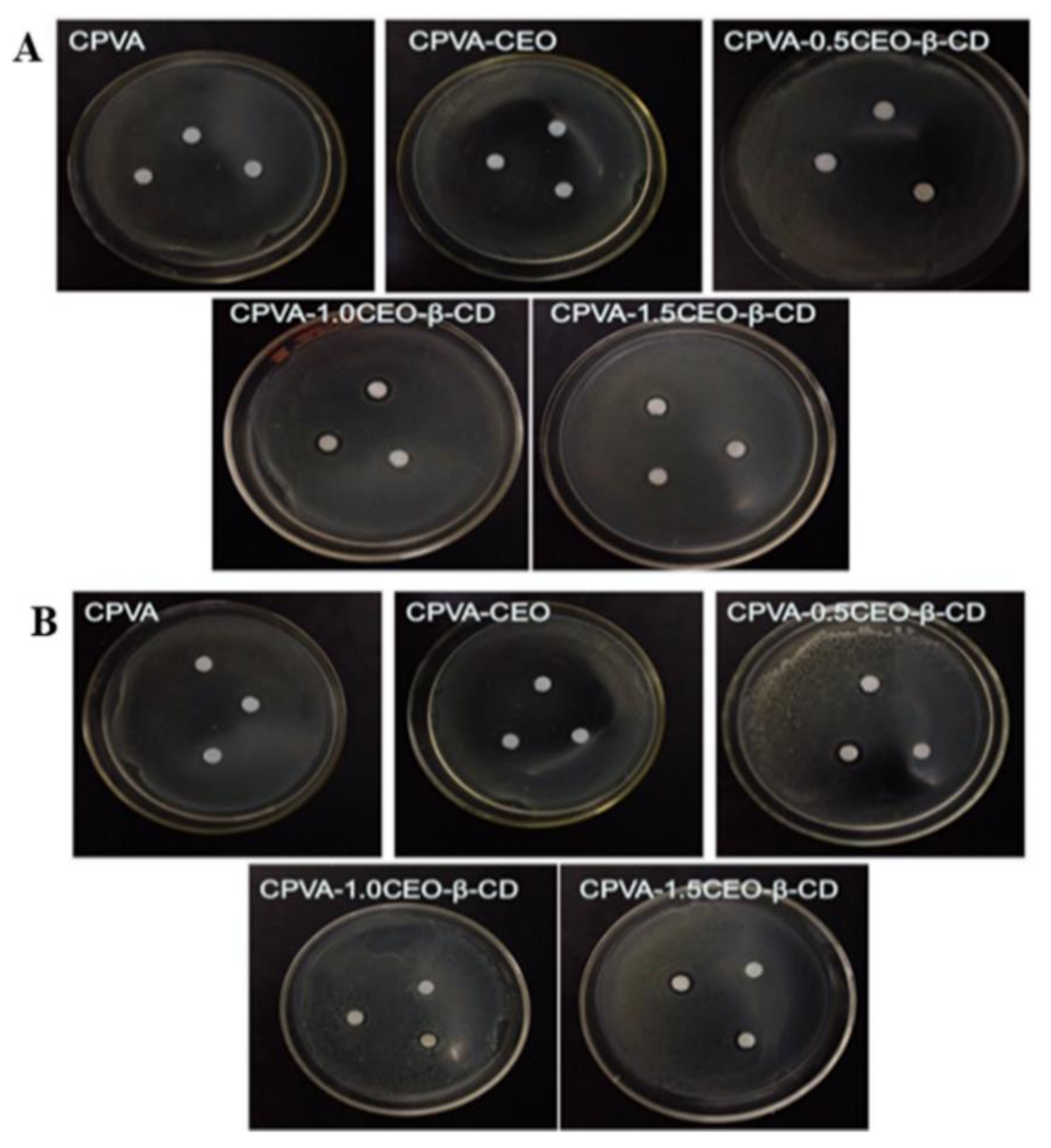
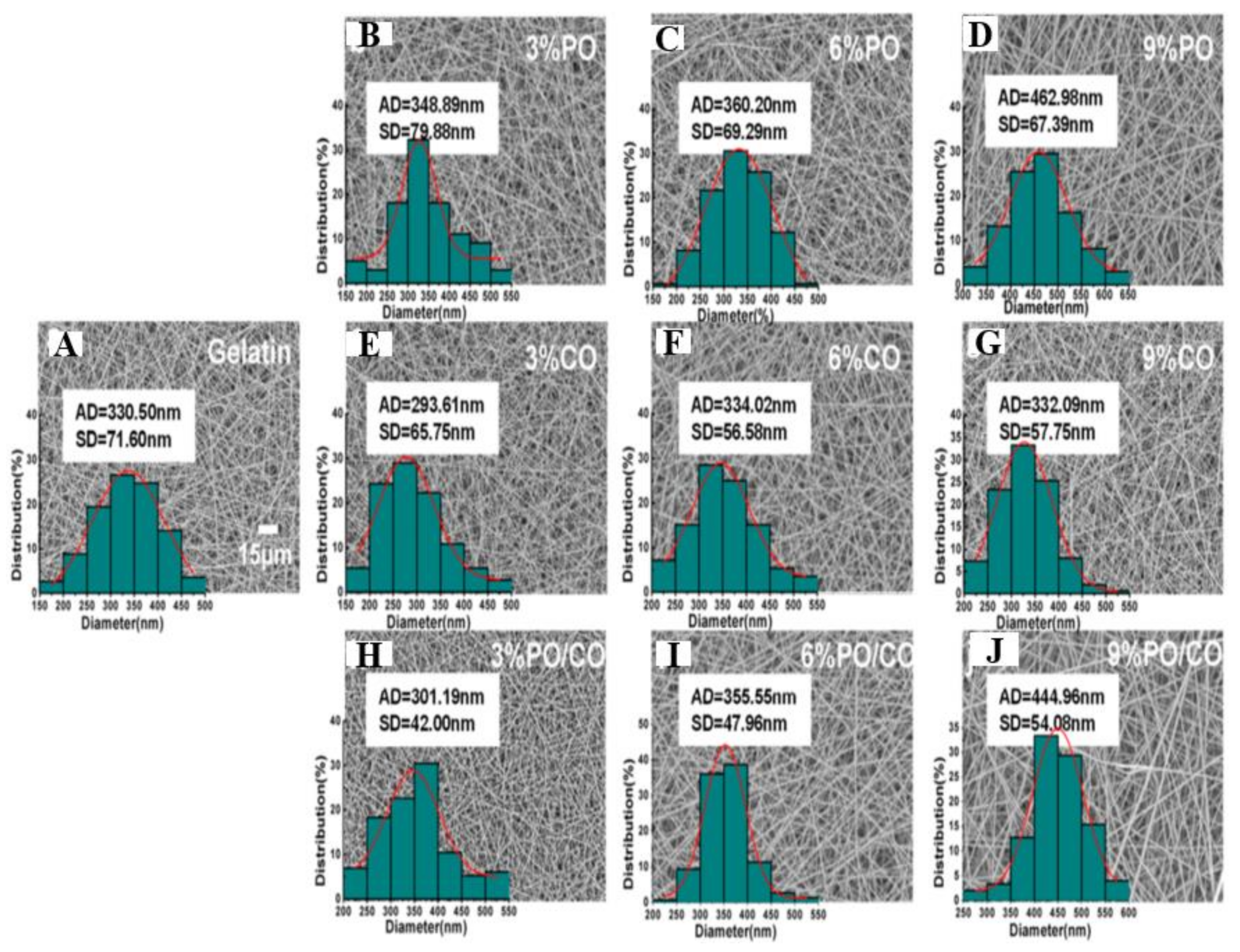
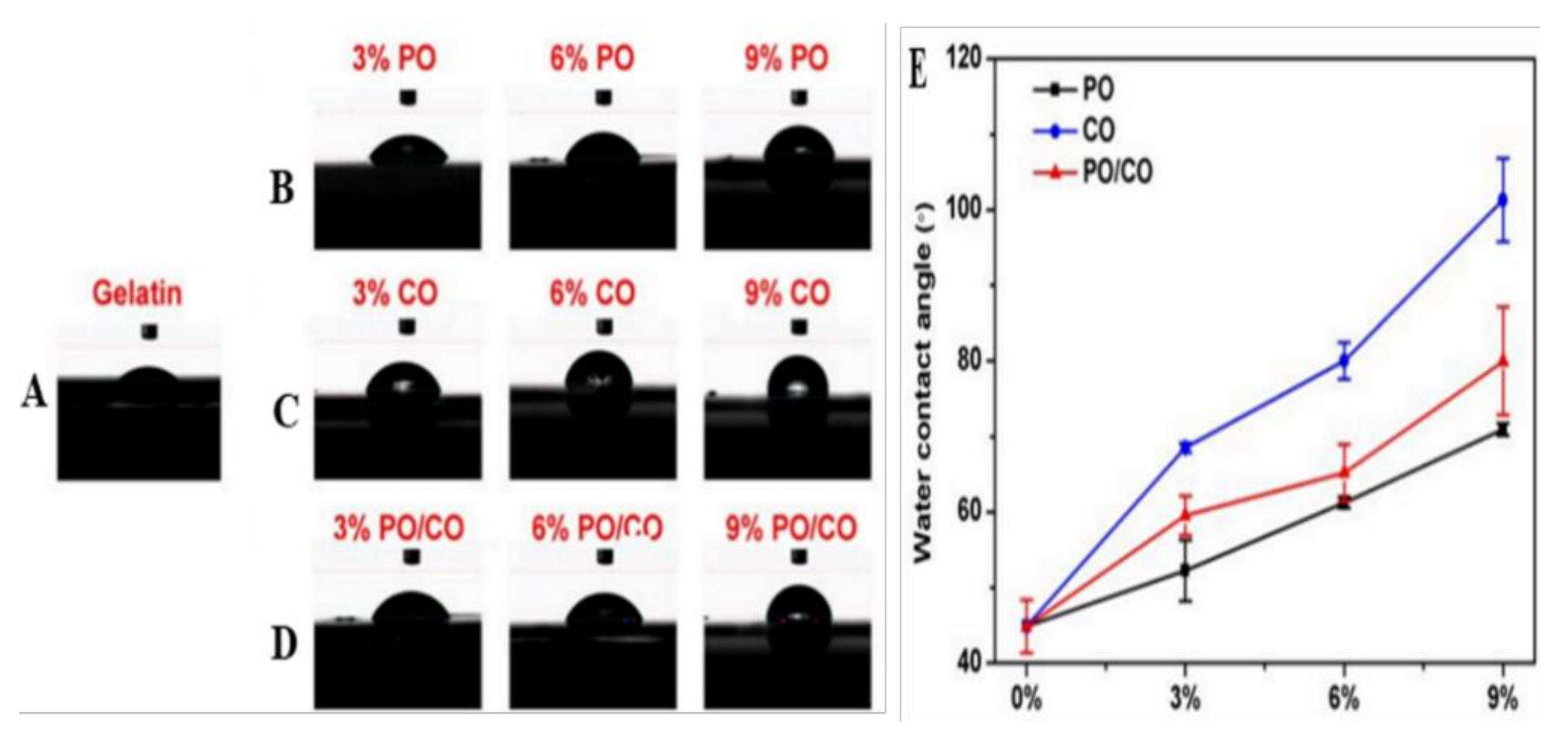

| Essential Oil | Polymer | Utilization | Ref |
|---|---|---|---|
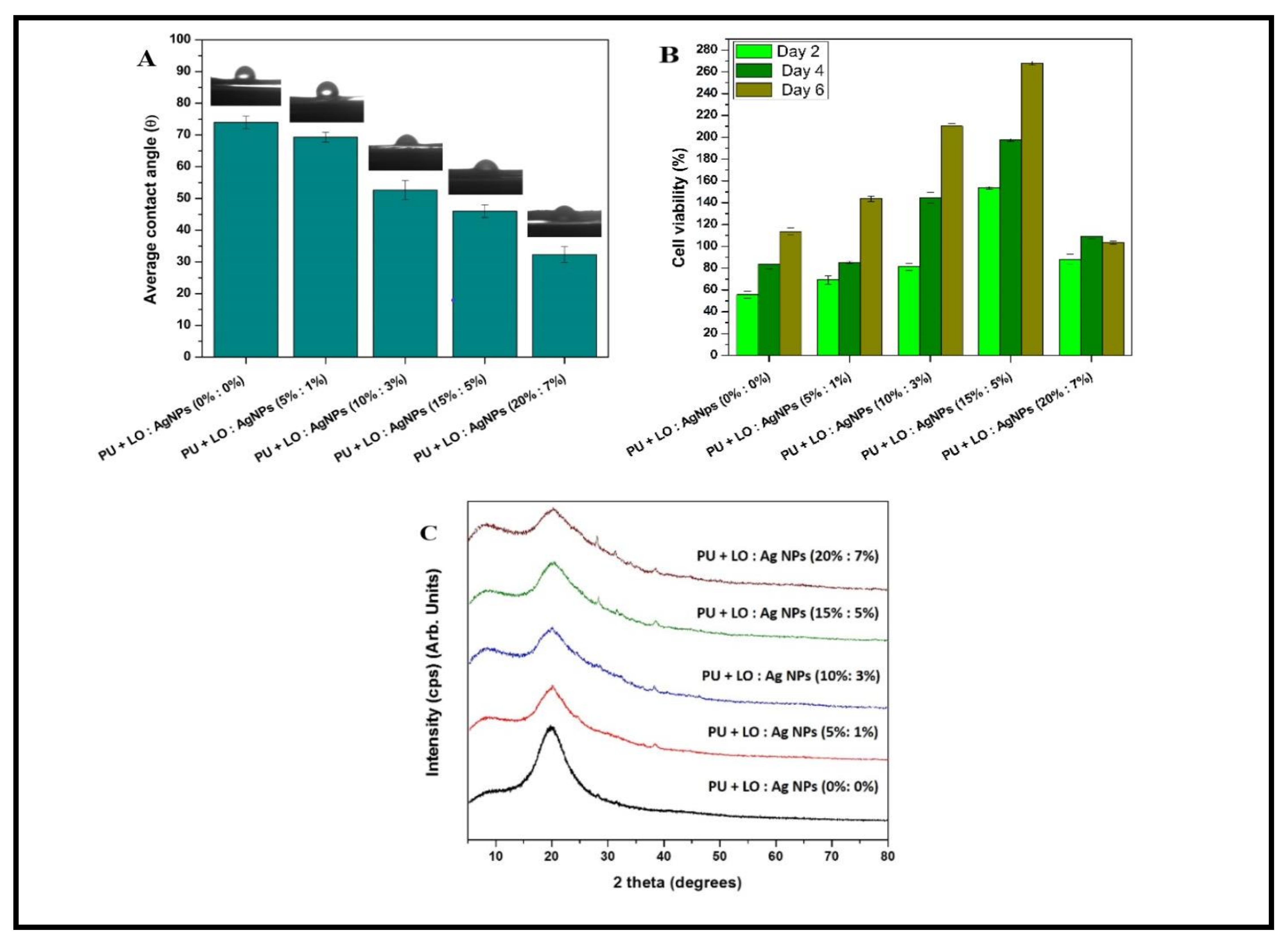
| Lavender (LO) | Polyurethane (PU) | After the encapsulation of LO and cobalt nitrate, the mechanical properties of electrospun PU scaffolds improved, and therefore were beneficial for bone-tissue engineering. It was seen that nanofibers were harmless to human dermal fibroblasts analyzed through MTS (5-(3-carboxymethoxyphenyl)-2-(4,5-dimethyl-thiazoly)-3-(4-sulfophenyl) tetrazolium assay. | [41] |
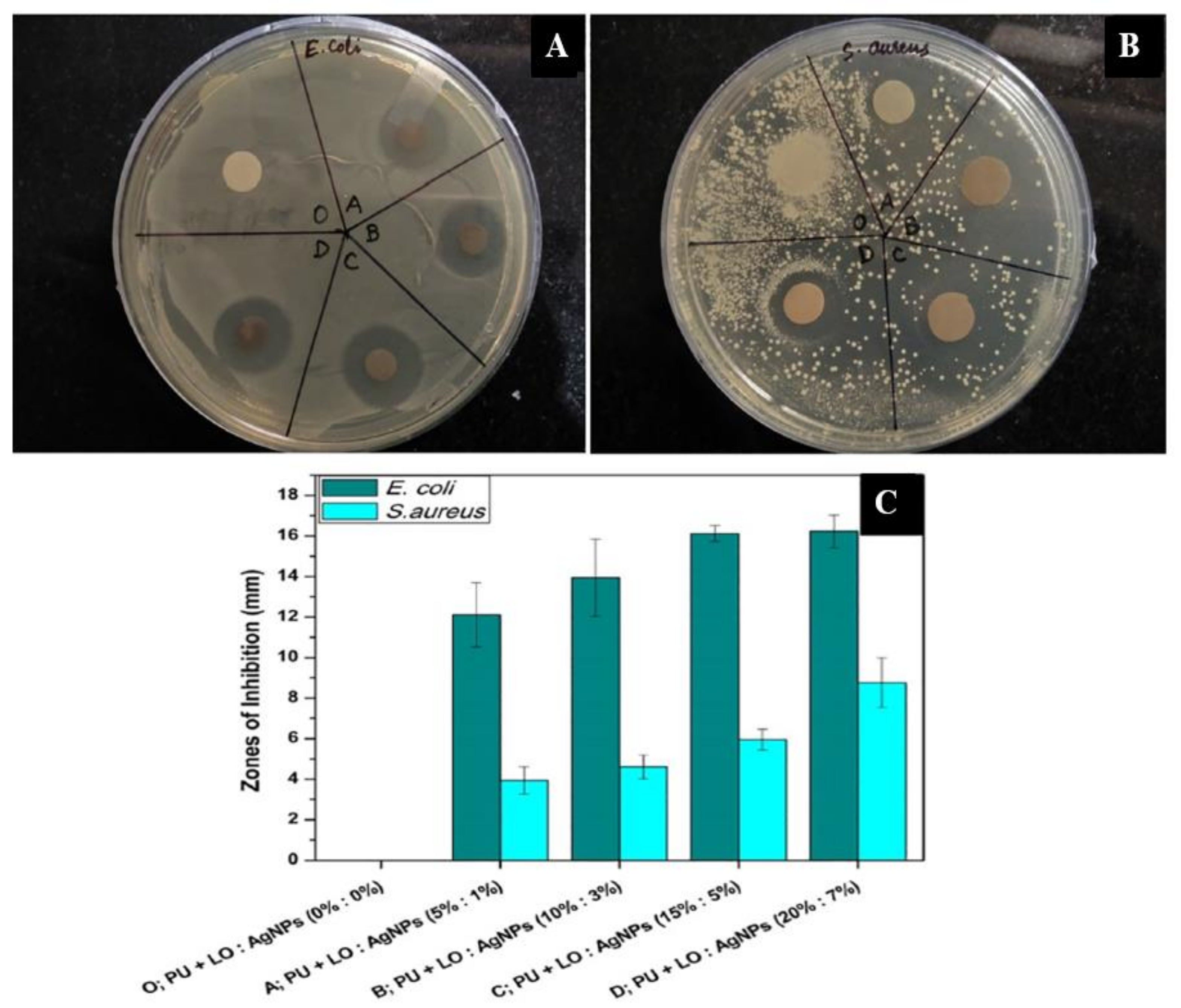
| Lavender (LO) | Polycaprolactone (PCL)/Polyethylene glycol (PEG) | This LO encapsulating PCL/PEG nanofibers generated through electrospinning showed a zone of inhibition against Staphylococcus aureus and Escherichia coli. | [42] |
| Thyme (TEO) | Silk fibroin (SF)/Gelatin (GEL) | SF/GEL nanofibers loaded with TEO and doxycycline monohydrate fabricated by electrospinning showed a larger prevention zone against Staphylococcus aureus and Klebsiella pneumoniae. In addition, the MTT (3-(4,5-dimethylthiazol-2-yl)-2–5-diphenyltetrazolium bromide) assay revealed that this nanofiber was not harmful to mouse fibroblast L929 cells. | [43] |
| Thyme (TEO) | Polycaprolactone (PCL)/Polyvinyl alcohol (PVA) | Coaxial electrospun core-shell nanofibers incorporated with TEO showed the highest antibacterial activity against Staphylococcus aureus and Escherichia coli. Thyme-extract encapsulated nanofibers could be used as potential wound-healing material and are promising materials to treat surfaces that contain pathogenic microorganisms. | [44] |
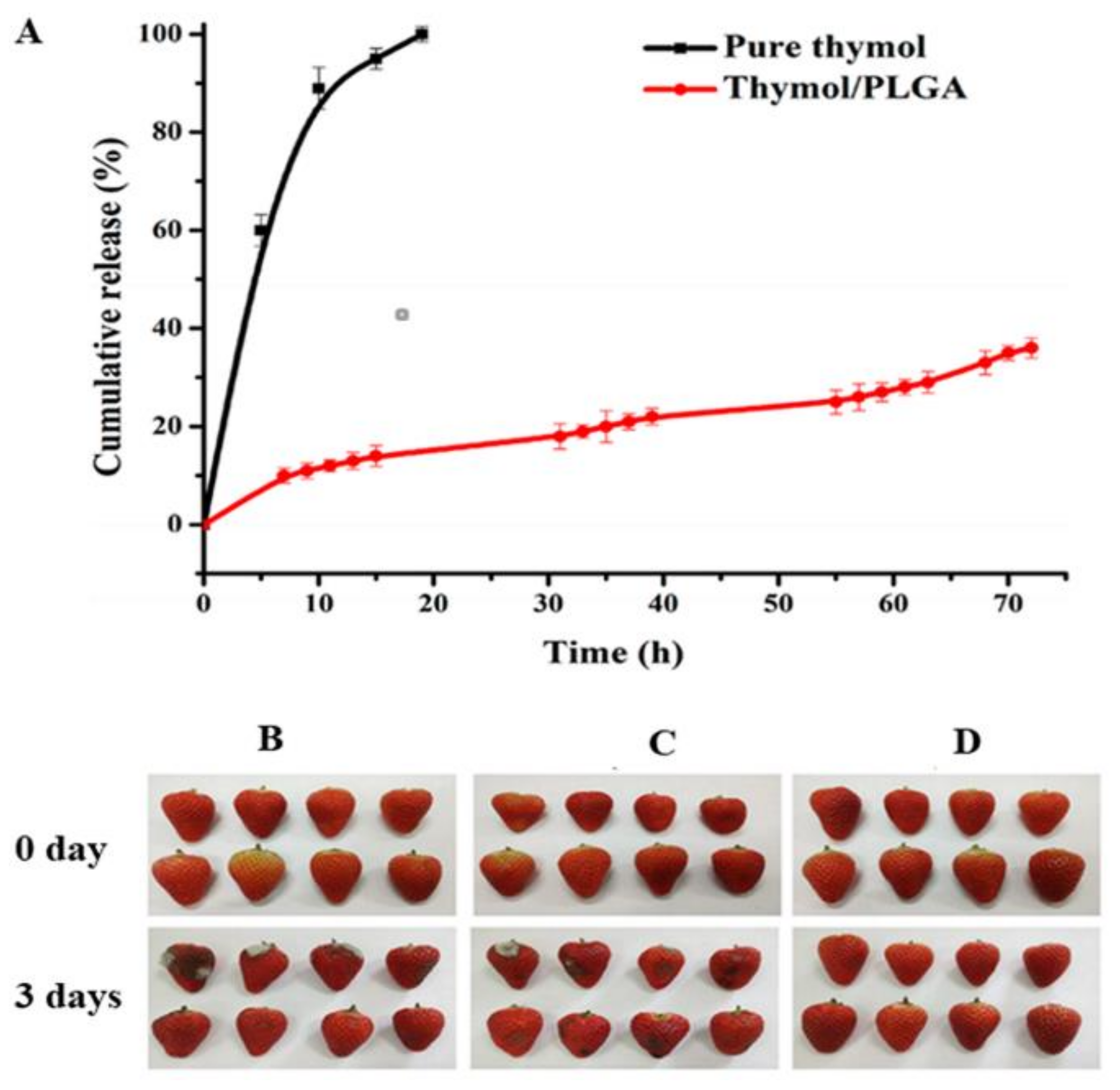
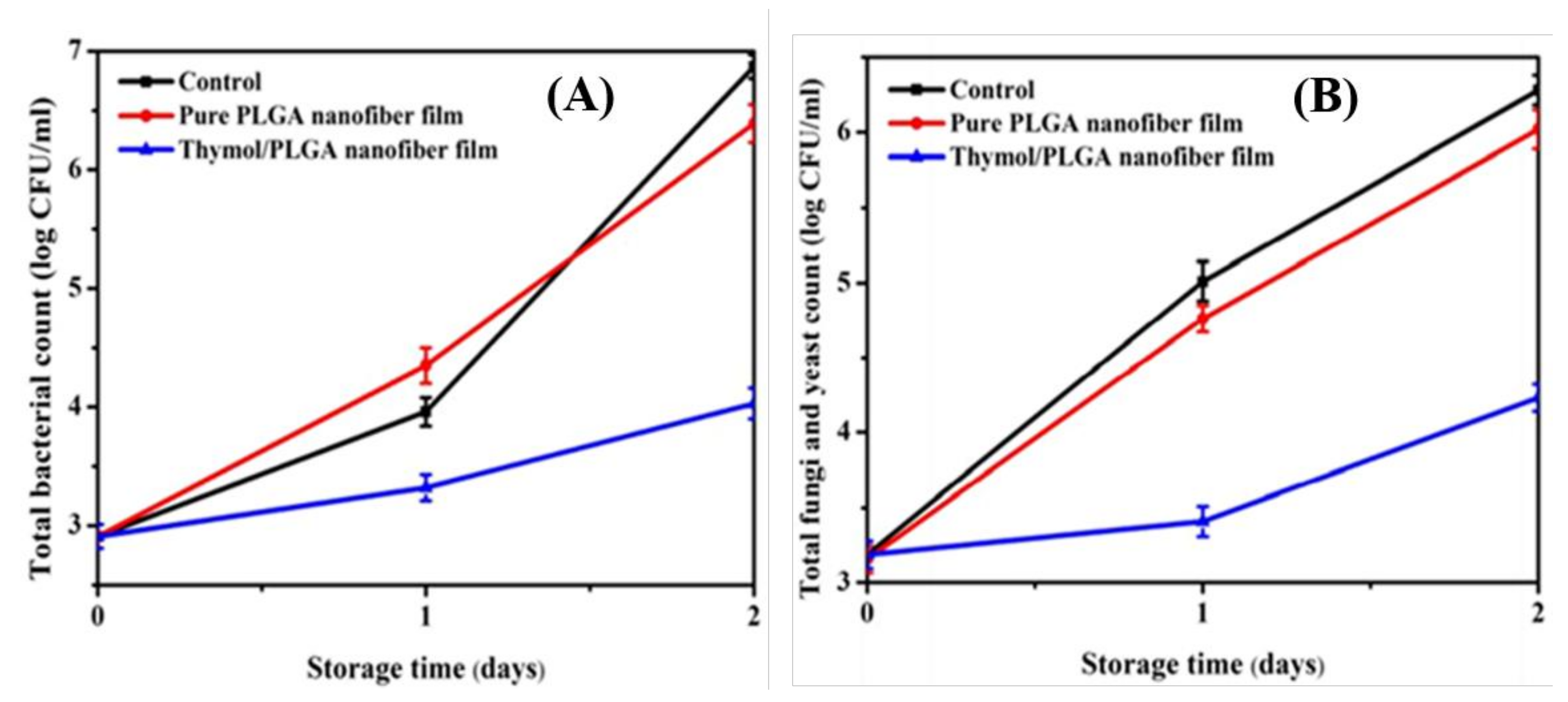
| Thyme (TEO) | Potato starch | Thermal stability of TEO encapsulating potato-starch nanofibers fabricated via electrospinning has been upgraded and can be applied in food products or packaging that requires high temperatures during their production. It can also be used as natural antioxidants in food products because 50% of this oil’s total phenolic compounds are retained after applying a thermal treatment of 100 °C. The antioxidant activity of the oil mentioned above is related to its significant content of phenolic compounds. | [45] |
| Cinnamon (CEO) | Polyurethane (PU) | Electrospinning-fabricated PU nanofiber loaded with CEO and activated carbon inhibited Escherichia coli and Staphylococcus aureus growth. Thus, the nanofiber air filter was more efficient. This nanofibrous air-filter media can be applied in various areas such as antibacterial fibers, personal masks, and air purifiers. | [46] |
| Cinnamon (CEO) | Polyvinyl alcohol (PVA) | Fumigant bioassays revealed that CEO inside nanofibers generated using electrospinning was more toxic than free CEO against all stages (e.g., male and female adults) of Phthorimaea operculella, one of the most common insect pests of cultivated potato. Accordingly, this nanofiber was effective in protecting horticulture extracts from pests during storage. | [47] |
| Cinnamon (CEO) | Sodium caseinate (SC)/Cellulose | The SC nanocomposite containing CEO and cellulose nanofiber prepared by the solvent-casting method was found to be successful in prolonging the shelf life and maintaining the quality of dry and oxidation sensitive foods such as nuts, spices, and bread and cereal products. | [48] |
| Cinnamon (CEO) | Polyvinyl alcohol (PVA) | Biodegradable electrospun PVA/CEO/β-cyclodextrin nanofibrous film showed excellent antimicrobial activity against Escherichia coli and Staphylococcus aureus. It could effectively prolong the shelf-life of strawberries, thus being applicable in active food packaging. | [49] |
| Cinnamon (CEO) | Polyurethane (PU) | Nanofibers fabricated by electrospinning were nontoxic, as they enhanced the growth of NIH 3T3 fibroblasts. They inhibited the growth of Staphylococcus aureus and Escherichia coli. They also reduced the chances of fruit decay. | [50] |
| Cinnamal- dehyde | Chitosan/ Polyethylene oxide (PEO) | Chitosan/citric acid/PEO nanofiber mats manufactured by electrospinning could act as delivery vehicles for this oil, potentially eliminating pseudomonas infections. | [51] |
| Cinnamon (CEO) | Polyvinyl pyrroli-done (PVP) | This nanofiber prepared by emulsion electrospinning showed good antibacterial effects against Staphylococcus aureus, Escherichia coli, and Candida albicans with 2, 3, and 4 wt % CEO. | [52] |
| Tea tree oil (TTO) | Chitosan | Electrospinning-prepared nanofiber membrane added with liposome-encapsulated TTO showed the maximum inhibition zone against Salmonella enteritidis and Salmonella typhimurium. It did not corrupt the sensory properties of chicken meat. Therefore, this nanofiber was conducive to extending the shelf life of chicken meat. | [53] |
| Peppermint (PO) | Polyethylene oxide (PEO)/Graphene oxide | Electrospinning-prepared nanofibrous mat having cerium oxide and PO exhibited prolonged antibacterial activity against Staphylococcus aureus and Escherichia coli due to the surface charge of cerium oxide and antibacterial properties of PO. Moreover, the in vitro MTT assay revealed that the nanofibrous mat exhibited low cytotoxicity toward L929 fibroblasts. The histological evaluations demonstrated that this nanofibrous mat accelerated re-epithelialization and collagen deposition, making it a potential candidate to be applied as a wound dressing to prevent skin infections. It has been shown that graphene oxide-filled nanofibrous scaffolds possess a porous structure and can maintain a moist environment around the wound, thereby facilitating the wound-healing process. | [54] |
| Red thyme/Clove oil (CLV) | Polycaprolactone (PCL) | PCL nanofibers added with CLV and red thyme essential oils prepared by electrospinning could be used as biofilm inhibitive agents on surfaces of biomaterials that are frequently contaminated by Candida tropicalis. | [55] |
| Rosemary (REO)/ Oregano (OEO) | Cellulose acetate (CA) | Electrospun CA nanofiber loaded with REO and OEO had good antimicrobial properties against three common microbial species: the bacteria Staphylococcus aureus and Escherichia coli, and the yeast Candida albicans. This work suggested that OEO was more potent than REO against the three studied microbes, possibly due to the immense microbial role of OEO molecules, such as carvacrol and thymol. | [56] |
| Ginger (GEO) | Chitosan | Chitosan bio-nanocomposite incorporated with sodium montmorillonite and GEO reduced the contamination and therefore improved the shelf life of poultry meat. This fiber can fulfill consumers’ demand for healthier and less chemically modified food products. It maintained the color, flavor, and PH value, and lessened the microbial contamination of meat products wrapped with this fiber. | [57] |
| Essential Oil | Main Components |
|---|---|
| Lavender | Linalool, Linalyl acetate, Camphor, and Eucalyptol |
| Thyme | Thymol, Gamma-terpinene, Para-cymene, and Carvacrol |
| Cinnamon | Cinnamaldehyde, Trans-cinnamyl acetate, Eugenol, and Camphor |
| Tea tree | Terpinen-4-ol, Gamma terpinene, Alpha-terpinene, and 1,8-Cineole |
| Peppermint | Menthol, Menthofuran, Menthyl acetate, and Menthone |
| Clove | Eugenol, Eugenyl acetate, Benzyl alcohol, and Beta-caryophyllene |
| Oregano vulgare | Carvacrol, Thymol, Beta-fenchyl alcohol, and Gamma-terpinene |
| Mexican oregano | Thymol, Carvacrol, Cineole 1–8, and Para-cymene |
| Rosemary | Cineole, Camphor, Alpha-pinene, and Camphene |
| Ginger roots | Alpha-zingiberene, Geranial, (Z)-citral, and Beta-cedrene |
| Fresh ginger rhizomes | Alpha-zingiberene, Beta-sesquiphellandrene, Trans-gamma-cadinene, and Geranial. |
| Chamomile | Alpha-bisabolol oxide A, Chamazulene, n-Octanal, and 1,8-Cineole |
| Angelica | (Z)-ligustilide, E-3-butylidene phthalide, (Z)- β-ocimene, and Gamma-terpinene |
| Chrysanthemum | Borneol, Beta-silenene, Camphor, and Guaia-3,9-diene |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rather, A.H.; Wani, T.U.; Khan, R.S.; Pant, B.; Park, M.; Sheikh, F.A. Prospects of Polymeric Nanofibers Loaded with Essential Oils for Biomedical and Food-Packaging Applications. Int. J. Mol. Sci. 2021, 22, 4017. https://doi.org/10.3390/ijms22084017
Rather AH, Wani TU, Khan RS, Pant B, Park M, Sheikh FA. Prospects of Polymeric Nanofibers Loaded with Essential Oils for Biomedical and Food-Packaging Applications. International Journal of Molecular Sciences. 2021; 22(8):4017. https://doi.org/10.3390/ijms22084017
Chicago/Turabian StyleRather, Anjum Hamid, Taha Umair Wani, Rumysa Saleem Khan, Bishweshwar Pant, Mira Park, and Faheem A. Sheikh. 2021. "Prospects of Polymeric Nanofibers Loaded with Essential Oils for Biomedical and Food-Packaging Applications" International Journal of Molecular Sciences 22, no. 8: 4017. https://doi.org/10.3390/ijms22084017
APA StyleRather, A. H., Wani, T. U., Khan, R. S., Pant, B., Park, M., & Sheikh, F. A. (2021). Prospects of Polymeric Nanofibers Loaded with Essential Oils for Biomedical and Food-Packaging Applications. International Journal of Molecular Sciences, 22(8), 4017. https://doi.org/10.3390/ijms22084017








