Annexins A2, A6 and Fetuin-A Affect the Process of Mineralization in Vesicles Derived from Human Osteoblastic hFOB 1.19 and Osteosarcoma Saos-2 Cells
Abstract
1. Introduction
2. Results
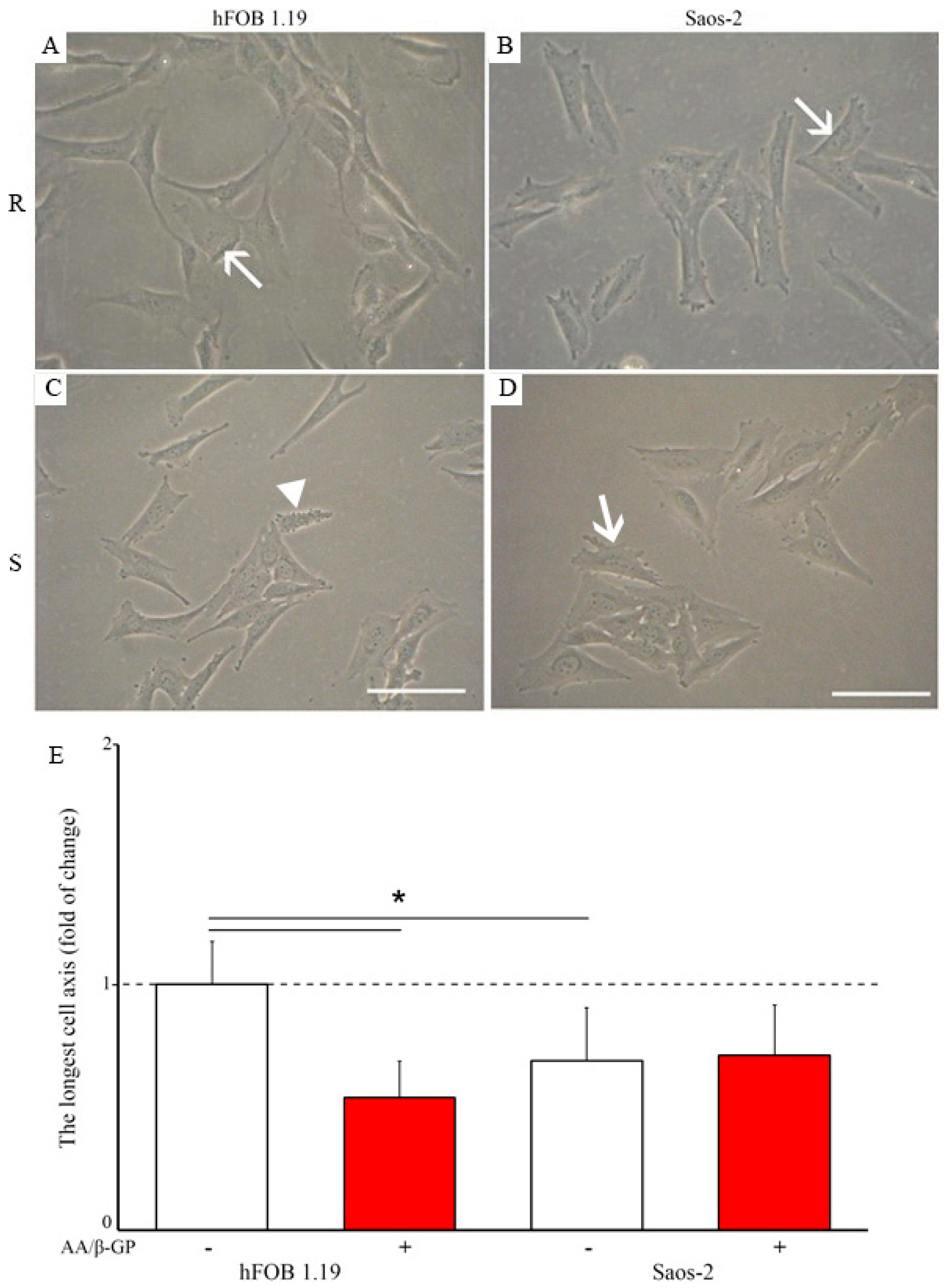
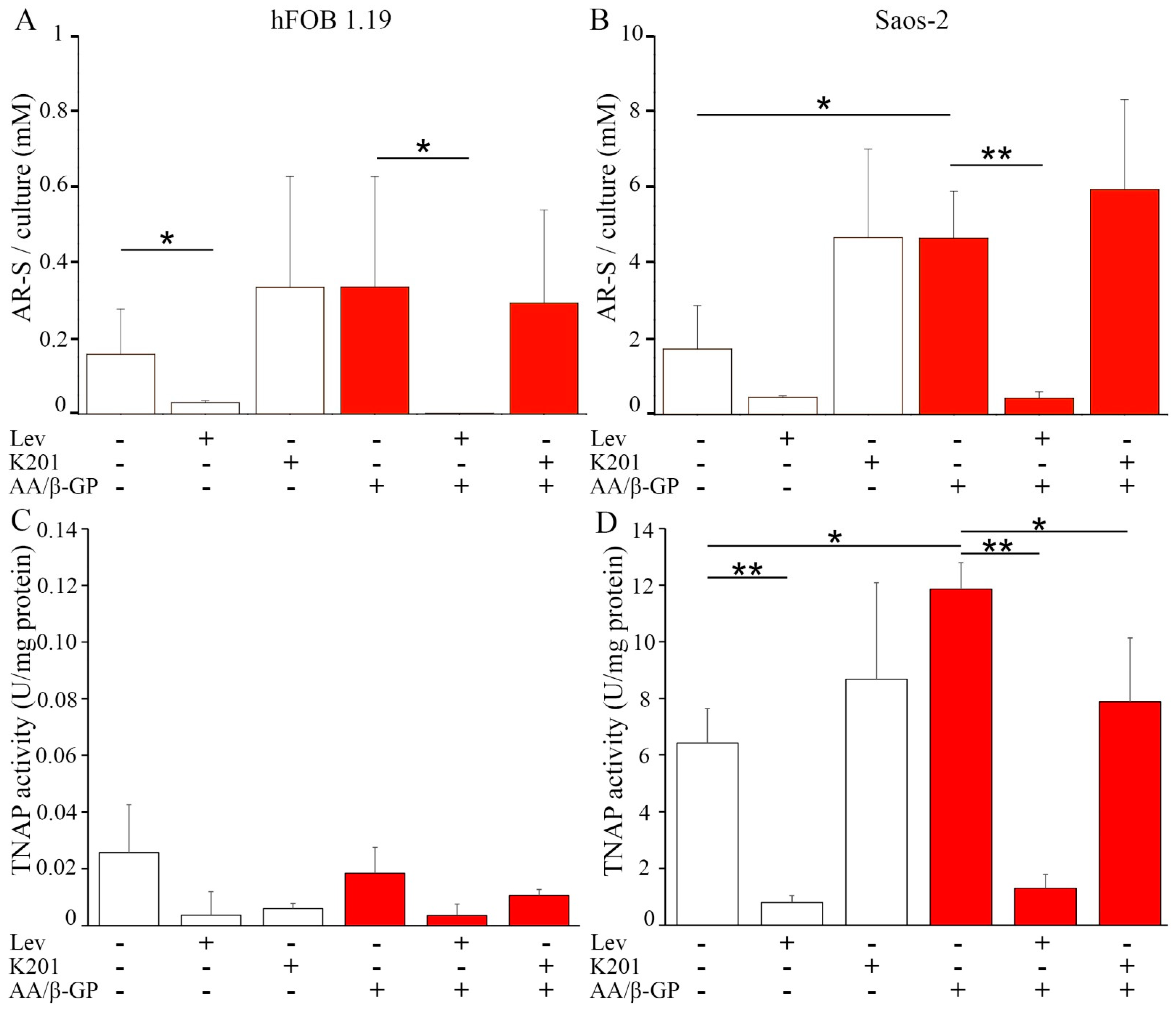
2.1. Characterization of the Mineralization Process in Human Fetal Osteoblastic Cell Line (hFOB 1.19 Cells) and Osteosarcoma Cell Lina (Saos-2 Cells)
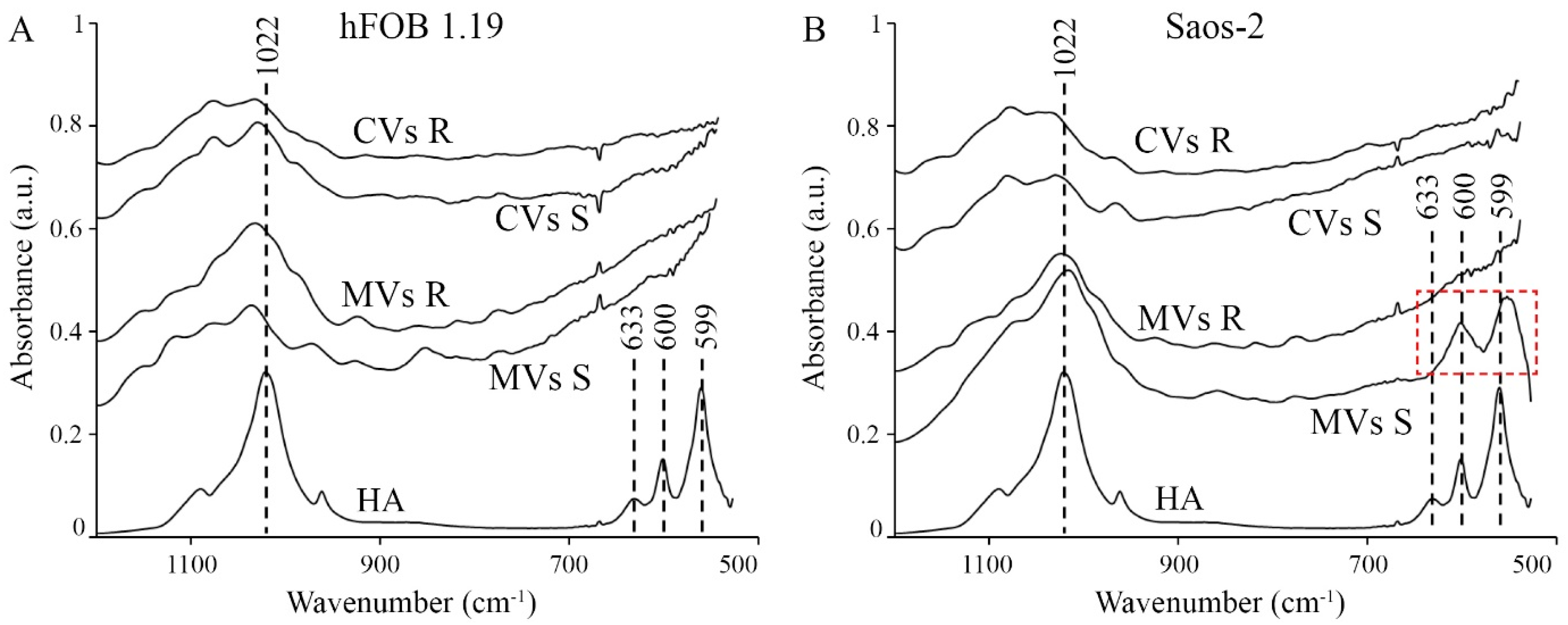
2.2. Analysis of Vesicular Fractions during Mineral Formation by Osteoblastic hFOB 1.19 and Osteosarcoma Saos-2 Cells
2.3. Protein Profiles of Mineralizing Osteoblastic hFOB 1.19 and Osteosarcoma Saos-2 Cells
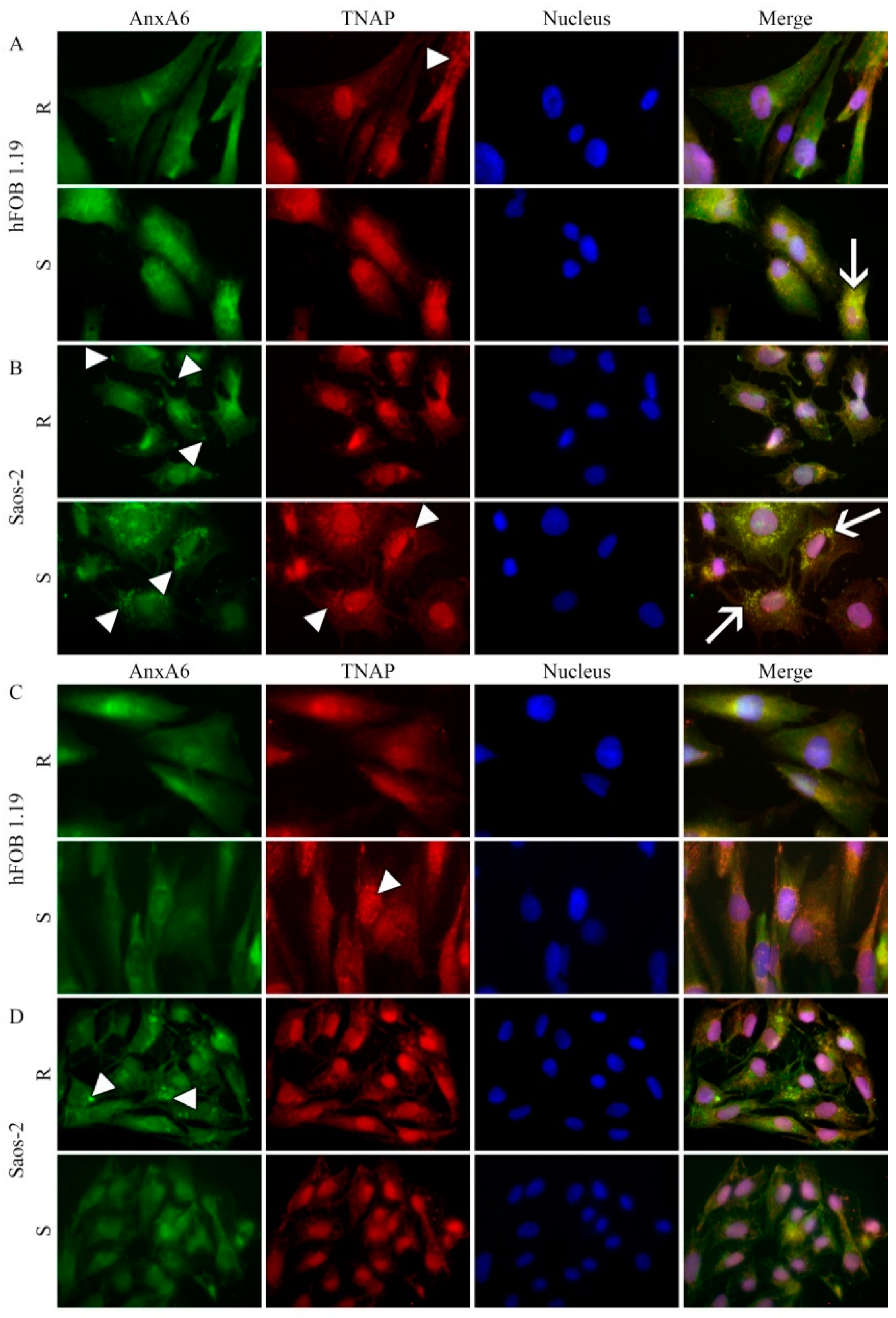
2.4. Protein Distributions in Resting and Stimulated hFOB 1.19 and Saos-2 Cells
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Treatment
4.2. Calcium Minerals Detection
4.3. Collagenase-Treatment, Vesicular Fractions, and Electron Microscopy with EDX
4.4. IR Spectra of Minerals Formed by Vesicular Fractions
4.5. Cell Lysis and TNAP Activity Assay
4.6. SDS-PAGE and Immunoblot Analysis
4.7. Immunochemistry and Fluorescent Microscopy with ApoTome
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Aghajanian, P.; Mohan, S. The art of building bone: Emerging role of chondrocyte-to-osteoblast transdiferentiation in endochondral ossification. Bone Res. 2018, 6, 19. [Google Scholar] [CrossRef] [PubMed]
- Anderson, H.C. Matrix vesicles and calcification. Curr. Rheumatol. Rep. 2003, 5, 222–226. [Google Scholar] [CrossRef]
- Bottini, M.; Mebarek, S.; Anderson, K.L.; Strzelecka-Kiliszek, A.; Bozycki, L.; Simão, A.M.S.; Bolean, M.; Ciancaglini, P.; Pikula, J.B.; Pikula, S.; et al. Matrix vesicles from chondrocytes and osteoblasts: Their biogenesis, properties, functions and biomimetic models. Biochim. Biophys. Acta (BBA) Gen. Subj. 2018, 1862, 532–546. [Google Scholar] [CrossRef]
- Golub, E.E. Role of matrix vesicles in biomineralization. Biochim. Biophys. Acta (BBA) Gen. Subj. 2009, 1790, 1592–1598. [Google Scholar] [CrossRef]
- Wuthier, R.E.; Lipscomb, G.F. Matrix vesicles: Structure, composition, formation and function in calcification. Front. Biosci. 2011, 16, 2812–2902. [Google Scholar] [CrossRef]
- Thouverey, C.; Strzelecka-Kiliszek, A.; Balcerzak, M.; Buchet, R.; Pikula, S. Matrix vesicles originate from apical membrane microvilli of mineralizing osteoblast-like Saos-2 cells. J. Cell. Biochem. 2009, 106, 127–138. [Google Scholar] [CrossRef]
- Thouverey, C.; Malinowska, A.; Balcerzak, M.; Strzelecka-Kiliszek, A.; Buchet, R.; Dadlez, M.; Pikula, S. Proteomic characterization of biogenesis and functions of matrix vesicles released from mineralizing human osteoblast-like cells. J. Proteom. 2011, 74, 1123–1134. [Google Scholar] [CrossRef]
- Cui, L.; Houston, D.A.; Farquharson, C.; MacRae, V.E. Characterisation of matrix vesicles in skeletal and soft tissue mineralisation. Bone 2016, 87, 147–158. [Google Scholar] [CrossRef]
- Strzelecka-Kiliszek, A.; Bozycki, L.; Mebarek, S.; Buchet, R.; Pikula, S. Characteristics of minerals in matrix vesicles produced by human osteoblasts hFOB 1.19 and osteosarcoma Saos-2 cells stimulated for mineralization. J. Inorg. Biochem. 2017, 171, 100–107. [Google Scholar] [CrossRef]
- Strzelecka-Kiliszek, A.; Romiszewska, M.; Bozycki, L.; Mebarek, S.; Bandorowicz-Pikula, J.; Buchet, R.; Pikula, S. Src and ROCK Kinases Differentially Regulate Mineralization of Human Osteosarcoma Saos-2 Cells. Int. J. Mol. Sci. 2019, 20, 2872. [Google Scholar] [CrossRef]
- Kirsch, T.; Wuthier, R.E. Stimulation of calcification of growth plate cartilage matrix vesicles by binding to type II and X collagens. J. Biol. Chem. 1994, 269, 11462–11469. [Google Scholar] [CrossRef]
- Ciancaglini, P.; Yadav, M.C.; Simão, A.M.S.; Narisawa, S.; Pizauro, J.M.; Farquharson, C.; Hoylaerts, M.F.; Millán, J.L. Kinetic Analysis of Substrate Utilization by Native and TNAP-, NPP1- or PHOSPHO1-Deficient Matrix Vesicles. J. Bone Miner. Res. 2009, 25, 716–737. [Google Scholar] [CrossRef]
- Millán, J.L. The Role of Phosphatases in the Initiation of Skeletal Mineralization. Calcif. Tissue Int. 2013, 93, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Hessle, L.; Johnson, K.A.; Anderson, H.C.; Narisawa, S.; Sali, A.; Goding, J.W.; Terkeltaub, R.; Millán, J.L. Tissue-nonspecific alkaline phosphatase and plasma cell membrane glycoprotein-1 are central antagonistic regulators of bone mineralization. Proc. Natl. Acad. Sci. USA 2002, 99, 9445–9449. [Google Scholar] [CrossRef] [PubMed]
- Thouverey, C.; Bechkoff, G.; Pikula, S.; Buchet, R. Inorganic pyrophosphate as a regulator of hydroxyapatite or calcium pyrophosphate dihydrate mineral deposition by matrix vesicles. Osteoarthr. Cartil. 2009, 17, 64–72. [Google Scholar] [CrossRef]
- Wang, W.; Xu, J.; Kirsch, T. Annexin-mediated Ca2+ Influx Regulates Growth Plate Chondrocyte Maturation and Apoptosis. J. Biol. Chem. 2003, 278, 3762–3769. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Xu, J.; Kirsch, T. Annexin V and terminal differentiation of growth plate chondrocytes. Exp. Cell Res. 2005, 305, 156–165. [Google Scholar] [CrossRef]
- Genetos, D.C.; Wong, A.; Weber, T.J.; Karin, N.J.; Yellowley, C.E. Impaired Osteoblast Differentiation in Annexin A2- and -A5-Deficient Cells. PLoS ONE 2014, 9, e107482. [Google Scholar] [CrossRef]
- Bolean, M.; Simão, A.M.S.; Kiffer-Moreira, T.; Hoylaerts, M.F.; Millán, J.L.; Itri, R.; Ciancaglini, P. Proteoliposomes with the ability to transport Ca2+ into the vesicles and hydrolyze phosphosubstrates on their surface. Arch. Biochem. Biophys. 2015, 584, 79–89. [Google Scholar] [CrossRef]
- Grieve, A.G.; Moss, S.E.; Hayes, M.J. Annexin A2 at the Interface of Actin and Membrane Dynamics: A Focus on Its Roles in Endocytosis and Cell Polarization. Int. J. Cell Biol. 2012, 2012, 1–11. [Google Scholar] [CrossRef]
- Cmoch, A.; Strzelecka-Kiliszek, A.; Palczewska, M.; Groves, P.; Pikula, S. Matrix vesicles isolated from mineralization-competent Saos-2 cells are selectively enriched with annexins and S100 proteins. Biochem. Biophys. Res. Commun. 2011, 412, 683–687. [Google Scholar] [CrossRef]
- Sekrecka, A.; Balcerzak, M.; Thouverey, C.; Buchet, R.; Pikula, S. Annexin in mineralization process. Postępy Biochem. 2007, 53, 159–163. [Google Scholar]
- Chen, N.X.; O’Neill, K.D.; Chen, X.; Moe, S.M. Annexin-Mediated Matrix Vesicle Calcification in Vascular Smooth Muscle Cells. J. Bone Miner. Res. 2008, 23, 1798–1805. [Google Scholar] [CrossRef]
- Genge, B.R.; Wu, L.N.; Wuthier, R.E. Mineralization of annexin-5-containing lipid-calcium-phosphate complexes: Modulation by varying lipid composition and incubation with cartilage collagens. J. Biol. Chem. 2008, 283, 9737–9748. [Google Scholar] [CrossRef]
- Kirsch, T.; Harrison, G.; Golub, E.E.; Nah, H.-D. The Roles of Annexins and Types II and X Collagen in Matrix Vesicle-mediated Mineralization of Growth Plate Cartilage. J. Biol. Chem. 2000, 275, 35577–35583. [Google Scholar] [CrossRef] [PubMed]
- Golczak, M.; Kicinska, A.; Bandorowicz-Pikula, J.; Buchet, R.; Szewczyk, A.; Pikula, S. Acidic pH-induced folding of annexin VI is a prerequisite for its insertion into lipid bilayers and formation of ion channels by the protein molecules. FASEB J. 2001, 15, 1083–1085. [Google Scholar] [PubMed]
- Veschi, E.A.; Bolean, M.; Strzelecka-Kiliszek, A.; Bandorowicz-Pikula, J.; Pikula, S.; Granjon, T.; Mebarek, S.; Magne, D.; Ramos, A.P.; Rosato, N.; et al. Localization of Annexin A6 in Matrix Vesicles During Physiological Mineralization. Int. J. Mol. Sci. 2020, 21, 1367. [Google Scholar] [CrossRef] [PubMed]
- Rochette, C.N.; Rosenfeldt, S.; Heiss, A.; Narayanan, T.; Ballauff, M.; Jahnen-Dechent, W. A Shielding Topology Stabilizes the Early Stage Protein-Mineral Complexes of Fetuin-A and Calcium Phosphate: A Time-Resolved Small-Angle X-ray Study. ChemBioChem 2009, 10, 735–740. [Google Scholar] [CrossRef]
- Herrmann, M.; Kinkeldey, A.; Jahnen-Dechent, W. Fetuin-A Function in Systemic Mineral Metabolism. Trends Cardiovasc. Med. 2012, 22, 197–201. [Google Scholar] [CrossRef]
- Mori, K.; Emoto, M.; Inaba, M. Fetuin-A: A multifunctional protein. Recent Patents Endocr. Metab. Immune Drug Discov. 2011, 5, 124–146. [Google Scholar] [CrossRef]
- Wang, H.; Sama, A.E. Anti-inflammatory role of fetuin-A in injury and infection. Curr. Mol. Med. 2012, 12, 625–633. [Google Scholar] [CrossRef]
- Szweras, M.; Liu, D.; Partridge, E.A.; Pawling, J.; Sukhu, B.; Clokie, C.; Jahnen-Dechent, W.; Tenenbaum, H.C.; Swallow, C.J.; Grynpas, M.D.; et al. Alpha 2-HS glycoprotein/fetuin, a transforming growth factor-beta/bone morphogenetic protein antagonist, regulates postnatal bone growth and remodeling. J. Biol. Chem. 2002, 277, 19991–19997. [Google Scholar] [CrossRef]
- Chen, N.X.; O’Neill, K.D.; Chen, X.; Duan, D.; Wang, E.; Sturek, M.S.; Edwards, J.M.; Moe, S.M. Fetuin-A uptake in bovine vascular smooth muscle cells is calcium dependent and mediated by annexins. Am. J. Physiol.-Ren. Physiol. 2007, 292, F599–F606. [Google Scholar] [CrossRef] [PubMed]
- Kundranda, M.N.; Ray, S.; Saria, M.; Friedman, D.; Matrisian, L.M.; Lukyanov, P.; Ochieng, J. Annexins expressed on the cell surface serve as receptors for adhesion to immobilized fetuin-A. Biochim. Biophys. Acta (BBA) Bioenerg. 2004, 1693, 111–123. [Google Scholar] [CrossRef] [PubMed]
- Bommanavar, S.; Hosmani, J.; Togoo, R.A.; Baeshen, H.A.; Raj, A.T.; Patil, S.; Bhandi, S.; Birkhed, D. Role of matrix vesicles and crystal ghosts in bio-mineralization. J. Bone Miner. Metab. 2020, 38, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Kirsch, T. Retinoic acid stimulates annexin-mediated growth plate chondrocyte mineralization. J. Cell Biol. 2002, 157, 1061–1069. [Google Scholar] [CrossRef]
- Minashima, T.; Small, W.; Moss, S.E.; Kirsch, T. Intracellular Modulation of Signaling Pathways by Annexin A6 Regulates Terminal Differentiation of Chondrocytes. J. Biol. Chem. 2012, 287, 14803–14815. [Google Scholar] [CrossRef]
- Hasegawa, T. Ultrastructure and biological function of matrix vesicles in bone mineralization. Histochem. Cell Biol. 2018, 149, 289–304. [Google Scholar] [CrossRef]
- Vaingankar, S.M.; Fitzpatrick, T.A.; Johnson, K.; Goding, J.W.; Maurice, M.; Terkeltaub, R. Subcellular targeting and function of osteoblast nucleotide pyrophosphatase phosphodiesterase 1. Am. J. Physiol. Cell Physiol. 2004, 286, C1177–C1187. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Towbin, H.; Staehelin, T.; Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc. Natl. Acad. Sci. USA 1979, 76, 4350–4354. [Google Scholar] [CrossRef] [PubMed]
- Kraus, D. Consolidated data analysis and presentation using an open-source add-in for the Microsoft Excel® spreadsheet software. Med. Writ. 2014, 23, 25–28. [Google Scholar] [CrossRef]

| Vesicle Type | Ca/P Ratio | ||||
|---|---|---|---|---|---|
| hFOB 1.19 Cells | Saos-2 Cells | ||||
| R | CVs | 0.59 ± 0.17 |  | 0.92 ± 0.57 |  |
| MVs | 0.11 ± 0.06 | 0.19 ± 0.03 | |||
| S | CVs | 0.15 ± 0.06 | 0.31 ± 0.09 | ||
| MVs | 0.28 ± 0.25 | 1.06 ± 0.22 | |||
| Inhibitor Type | Protein Type | Relative Co-Localization Area (%) | ||||
|---|---|---|---|---|---|---|
| hFOB 1.19 Cells | Saos-2 Cells | |||||
| R | - | AnxA6/TNAP | 12.93 ± 1.49 | 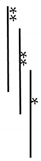 | 12.60 ± 0.88 | 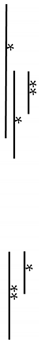 |
| AnxA2/TNAP | 10.86 ± 1.55 | 16.59 ± 1.20 | ||||
| FetuA/TNAP | 12.51 ± 2.11 | 16.61 ± 1.84 | ||||
| FetuA/β-actin | 13.40 ± 1.79 | 9.03 ± 0.43 | ||||
| Lev | AnxA6/TNAP | 31.72 ± 3.65 | 17.93 ± 0.83 | |||
| FetuA/β-actin | 33.33 ± 4.61 | 14.69 ± 2.28 | ||||
| K201 | AnxA6/TNAP | 19.46 ± 1.39 | 24.37 ± 4.26 | |||
| FetuA/β-actin | 20.87 ± 0.68 | 12.82 ± 0.67 | ||||
| S | - | AnxA6/TNAP | 17.40 ± 1.21 | 22.54 ± 1.49 | ||
| AnxA2/TNAP | 16.03 ± 2.33 | 21.98 ± 5.09 | ||||
| FetuA/TNAP | 17.79 ± 0.46 | 22.23 ± 3.78 | ||||
| FetuA/β-actin | 18.24 ± 0.79 | 11.90 ± 1.53 | ||||
| Lev | AnxA6/TNAP | 24.27 ± 3.43 | 21.57 ± 1.54 | |||
| FetuA/β-actin | 21.68 ± 0.20 | 19.23 ± 0.69 | ||||
| K201 | AnxA6/TNAP | 16.02 ± 0.96 | 18.39 ± 1.31 | |||
| FetuA/β-actin | 14.59 ± 2.87 | 23.25 ± 4.90 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bozycki, L.; Mroczek, J.; Bessueille, L.; Mebarek, S.; Buchet, R.; Pikula, S.; Strzelecka-Kiliszek, A. Annexins A2, A6 and Fetuin-A Affect the Process of Mineralization in Vesicles Derived from Human Osteoblastic hFOB 1.19 and Osteosarcoma Saos-2 Cells. Int. J. Mol. Sci. 2021, 22, 3993. https://doi.org/10.3390/ijms22083993
Bozycki L, Mroczek J, Bessueille L, Mebarek S, Buchet R, Pikula S, Strzelecka-Kiliszek A. Annexins A2, A6 and Fetuin-A Affect the Process of Mineralization in Vesicles Derived from Human Osteoblastic hFOB 1.19 and Osteosarcoma Saos-2 Cells. International Journal of Molecular Sciences. 2021; 22(8):3993. https://doi.org/10.3390/ijms22083993
Chicago/Turabian StyleBozycki, Lukasz, Joanna Mroczek, Laurence Bessueille, Saida Mebarek, René Buchet, Slawomir Pikula, and Agnieszka Strzelecka-Kiliszek. 2021. "Annexins A2, A6 and Fetuin-A Affect the Process of Mineralization in Vesicles Derived from Human Osteoblastic hFOB 1.19 and Osteosarcoma Saos-2 Cells" International Journal of Molecular Sciences 22, no. 8: 3993. https://doi.org/10.3390/ijms22083993
APA StyleBozycki, L., Mroczek, J., Bessueille, L., Mebarek, S., Buchet, R., Pikula, S., & Strzelecka-Kiliszek, A. (2021). Annexins A2, A6 and Fetuin-A Affect the Process of Mineralization in Vesicles Derived from Human Osteoblastic hFOB 1.19 and Osteosarcoma Saos-2 Cells. International Journal of Molecular Sciences, 22(8), 3993. https://doi.org/10.3390/ijms22083993








