Effects of Cannabidiol and Beta-Caryophyllene Alone or in Combination in a Mouse Model of Permanent Ischemia
Abstract
1. Introduction
2. Results
2.1. Descriptive and Statistical Findings
2.1.1. Infarct Size
- I.
- Treatment with BCP, CBD, and their combination led to a significant decrease in infarct size.
- II.
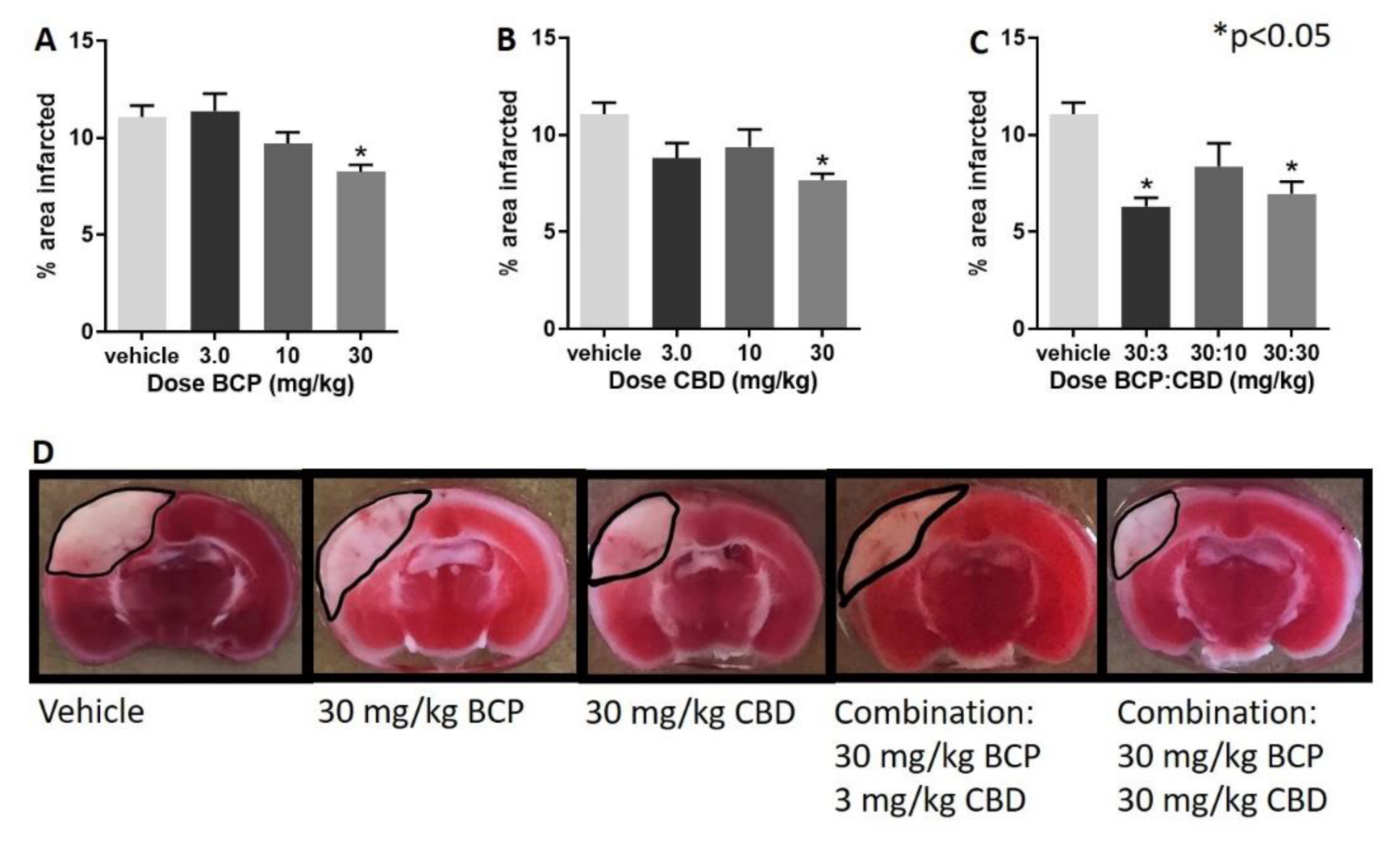
- Following administration of BCP, one-way ANOVA test revealed a significant effect of BCP (F (3, 28) = 5.237, p = 0.005), with Dunnett’s multiple comparison showing a significant effect at the 30 mg/kg dose (Figure 1A).
- III.
- Following administration of CBD, one-way ANOVA revealed a significant effect of CBD (F (3, 27) = 3.974, p = 0.018), with Dunnett’s multiple comparison test showing a significant effect at the 30 mg/kg dose.
- IV.
- Following administration of BCP:CBD combinations, one-way ANOVA revealed a significant effect of BCP (F (3, 27) = 7.182, p = 0.001), with Dunnett’s multiple comparison test showing a significant effect at the 30:3 and 30:30 BCP:CBD mg/kg doses (Figure 1C).
- V.
- Figure 1D shows the location and shape of the infarct in coronal section and its attenuation by BCP, CBD, and the combination in representative mice. The center of the lesion is in parietal cortex area 1 (primary somatosensory cortex) at approximately –2.0 mm from Bregma, and the extent of the infarct can be observed in the coronal slices from approximately 0.20 mm rostral to Bregma to approximately –2.80 mm caudal to Bregma. The infarct spreads to the border of the corpus callosum but is not observed in deeper tissue such as the CA1-3 fields of the hippocampus as measured with the TTC staining. Interestingly, BCP treatment led to more shallow but broader damage, CBD treatment led to a narrower, but deep, infarct, and the combinations combined these two characteristics with a more narrow and shallow range of damage.
2.1.2. Microglia Analysis
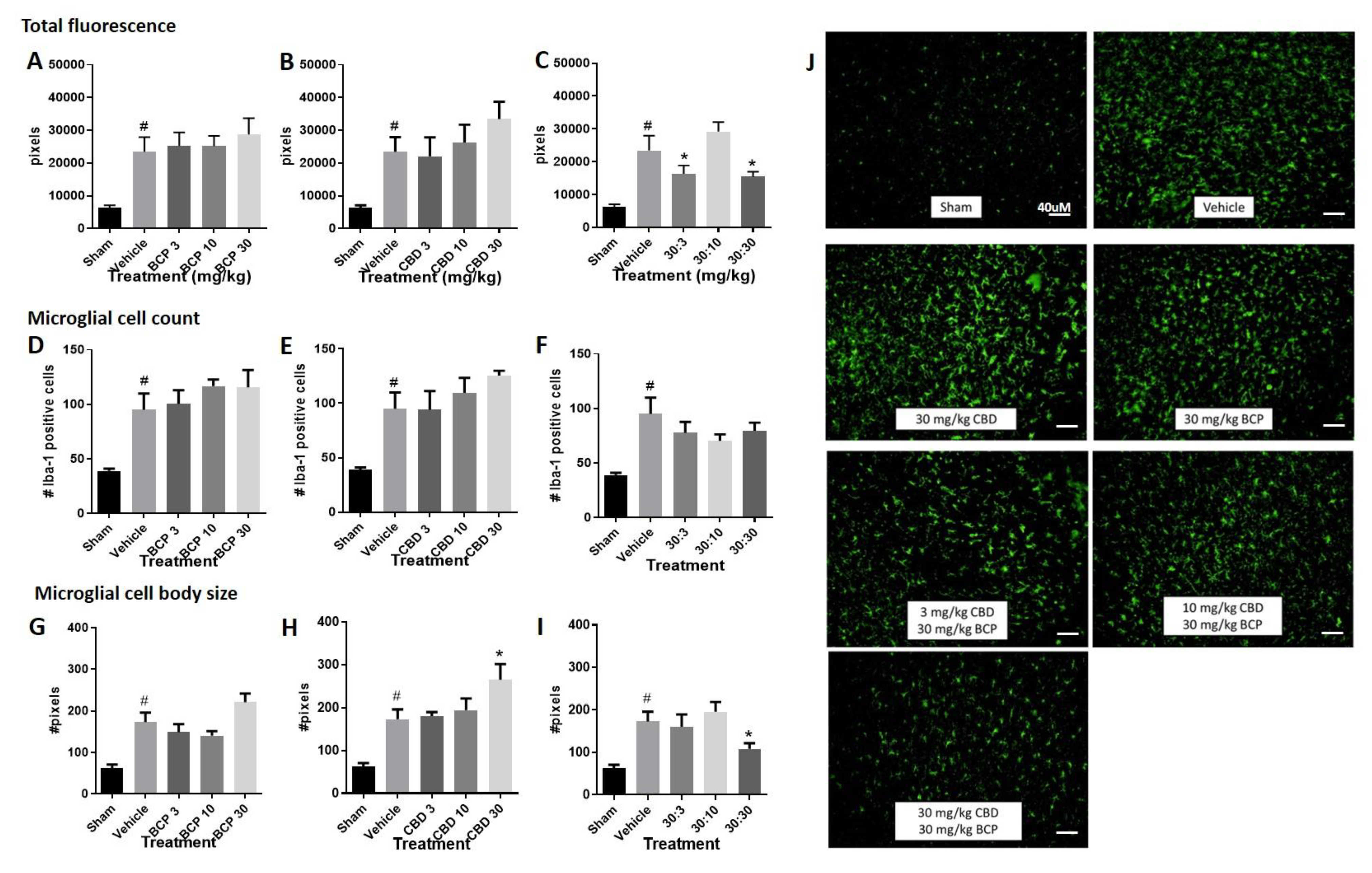
- Total fluorescence: Increased immunolabeling for Iba-1 was observed in the stroke animals compared to sham control (Figure 2A–C, Student t-test sham versus vehicle, p < 0.05). The Iba1 antibody is commonly used as a marker of microglia activation in staining and immunohistochemistry, given that ionized calcium binding adaptor molecule 1 (Iba1) is a microglia/macrophage-specific calcium-binding protein with actin-bundling activity that participates in membrane ruffling and phagocytosis in activated microglia.
- (1)
- Treatment with BCP or CBD alone did not significantly alter total fluorescence compared to the vehicle-treated stroked mice. One-way ANOVAs compared treatments in stroked animals, sham animals were excluded in the analysis. One way ANOVA for BCP was (F (3, 33) < 1.0), and one-way ANOVA for CBD was (F (3, 30) < 1.0) (Figure 2A,B).
- (2)
- However, treatment with the BCP:CBD combination led to a significant reduction in total immunofluorescence compared to vehicle-treated stroked mice, (F (3, 32) = 4.483, p = 0.01), with Dunnett’s multiple comparison test showing a significant effect at the 30:3 and 30:30 BCP:CBD mg/kg doses (Figure 2C).
- Microglial cell count. Increased Iba-1 positive cell number was observed in the stroke animals compared to sham control. (Figure 2D–F, Student t-test sham versus vehicle, p < 0.05).
- (1)
- Treatment with BCP or CBD alone did not significantly alter total microglial cell number compared to the vehicle-treated stroked mice. One way ANOVA for BCP was (F (3, 31) < 1.0), and one way ANOVA for CBD was (F (3, 30) < 1.0) (Figure 2D,E).
- (2)
- Treatment with the BCP:CBD combination also did not alter total microglial cell number compared to vehicle-treated stroked mice (F (3, 30) = 1.042, p = 0.388) (Figure 2F).
- Microglial cell body size. Increased cell body size was observed in the stroke animals compared to sham controls. (Figure 2G–I, Student t-test sham versus vehicle, p < 0.05).
- (1)
- Treatment with BCP produced a significant increase in average cell body size as compared with vehicle-treated stroked mice (F (3, 32) = 3.593, p = 0.0241) (Figure 2G), with Dunnett’s multiple comparison test showing no significant effect at any dose.
- (2)
- Treatment with CBD produced a significant increase in average cell body size as compared with vehicle-treated stroked mice (F (3, 32) = 2.728, p = 0.05) (Figure 2H), with Dunnett’s multiple comparison test showing a significant effect at the 3.0 and 30 mg/kg doses.
- (3)
- However, treatment with the BCP:CBD combination led to a significant reduction in average cell body size as compared with vehicle-treated stroked mice, (F (3, 32) = 6.319, p = 0.002), with Dunnett’s multiple comparison test showing a significant effect at the 30:30 BCP:CBD mg/kg doses (Figure 2I).
2.1.3. Grip Strength Behavior
- (1)
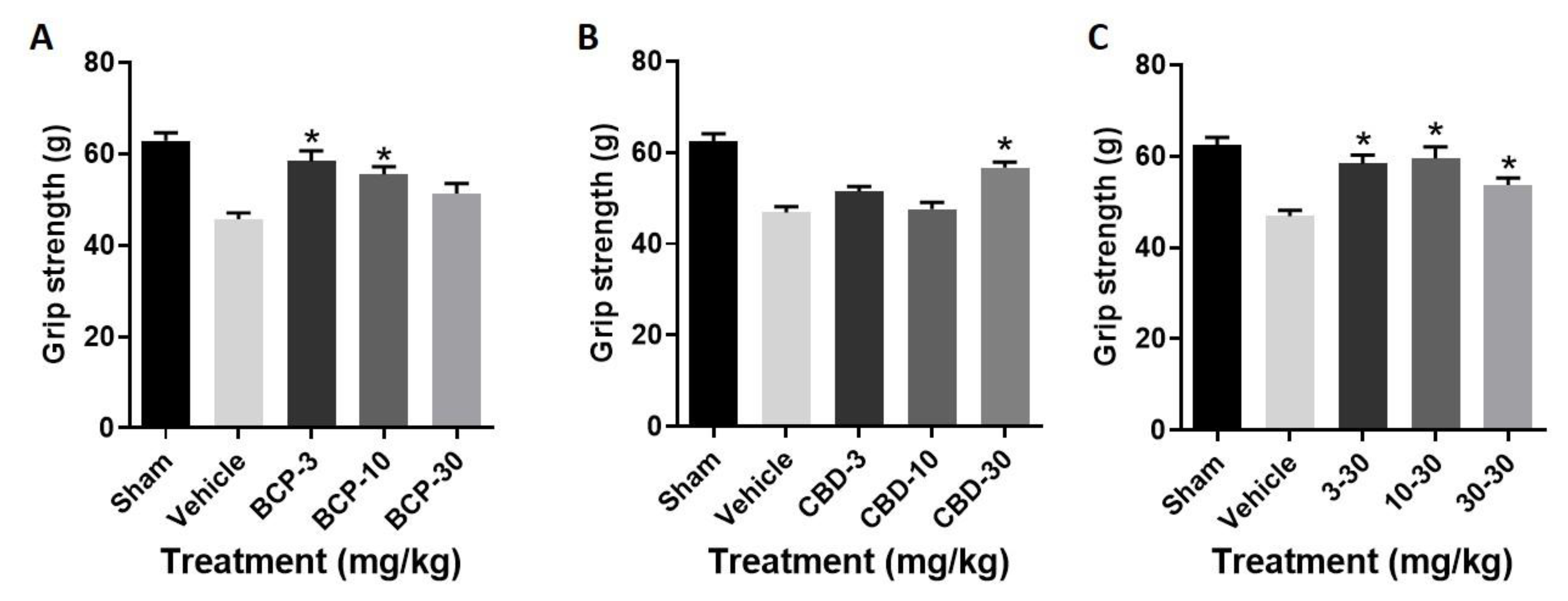
- Treatment with BCP produced a significant improvement in grip strength as compared with stroke (F (3, 28) = 9.926, p < 0.0001) (Figure 3A), with Dunnett’s multiple comparisons test showing significance at the 3.0 and 10 mg/kg doses.
- (2)
- Treatment with CBD produced a significant improvement in grip strength as compared with stroke (F (3, 28) = 13.28, p < 0.0001) (Figure 3B), with Dunnett’s multiple comparisons test showing significance at the 30 mg/kg dose.
- (3)
- Treatment with the BCP:CBD combination produced a significant improvement in grip strength as compared with stroke (F (3, 28) = 8.535, p = p < 0.0001) (Figure 3C), with Dunnett’s multiple comparisons test showing significance at the 3.0, 10, and 30 mg/kg doses.
2.2. Figures, Tables and Schemes
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Induction of Stroke: Photothrombosis Model
4.3. Grip Strength Testing
4.4. Immunohistochemistry Staining
4.5. Drugs
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Benjamin, E.J.; Blaha, M.J.; Chiuve, S.E.; Cushman, M.; Das, S.R.; de Ferranti, S.D.; Floyd, J.; Fornage, M.; Gillespie, C.; Isasi, C.R.; et al. Heart disease and stroke statistics—2017 update a report from the American heart association. Circulation 2017, 135, e146–e603. [Google Scholar] [CrossRef] [PubMed]
- Heuschmann, P.U.; Wiedmann, S.; Wellwood, I.; Rudd, A.; Di Carlo, A.; Bejot, Y.; Ryglewicz, D.; Rastenyte, D.; Wolfe, C.D. Three-month stroke outcome: The European Registers of Stroke (EROS) Investigators. Neurology 2011, 76, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Jauch, E.C.; Saver, J.L.; Adams, H.P., Jr.; Bruno, A.; Connors, J.J.; Demaerschalk, B.M.; Khatri, P.; McMullan, P.W., Jr.; Qureshi, A.I.; Rosenfield, K.; et al. Guidelines for the early management of patients with acute ischemic stroke: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2013, 44, 870–947. [Google Scholar] [CrossRef] [PubMed]
- Courties, G.; Herisson, F.; Sager, H.B.; Heidt, T.; Ye, Y.; Wei, Y.; Sun, Y.; Severe, N.; Dutta, P.; Scharff, J.; et al. Ischemic Stroke Activates Hematopoietic Bone Marrow Stem Cells. Circ. Res. 2015, 116, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Iadecola, C.; Anrather, J. The immunology of stroke: From mechanisms to translation. Nat. Med. 2011, 17, 796–808. [Google Scholar] [CrossRef]
- Möller, K.; Boltze, J.; Pösel, C.; Seeger, J.; Stahl, T.; Wagner, D.-C. Sterile Inflammation after Permanent Distal MCA Occlusion in Hypertensive Rats. Br. J. Pharmacol. 2014, 34, 307–315. [Google Scholar] [CrossRef]
- Jin, Q.; Cheng, J.; Liu, Y.; Wu, J.; Wang, X.; Wei, S.; Zhou, X.; Qin, Z.; Jia, J.; Zhen, X. Improvement of functional recovery by chronic metformin treatment is associated with enhanced alternative activation of microglia/macrophages and increased angiogenesis and neurogenesis following experimental stroke. Brain Behav. Immun. 2014, 40, 131–142. [Google Scholar] [CrossRef]
- Qin, C.; Fan, W.-H.; Liu, Q.; Shang, K.; Murugan, M.; Wu, L.-J.; Wang, W.; Tian, D.-S. Fingolimod Protects Against Ischemic White Matter Damage by Modulating Microglia Toward M2 Polarization via STAT3 Pathway. Stroke 2017, 48, 3336–3346. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, J.; Wang, Y.; Yang, G.-Y. The biphasic function of microglia in ischemic stroke. Prog. Neurobiol. 2017, 157, 247–272. [Google Scholar] [CrossRef]
- Charriaut-Marlangue, C.; Leconte, C.; Csaba, Z.; Chafa, L.; Pansiot, J.; Talatizi, M.; Simon, K.; Moretti, R.; Marchand-Leroux, C.; Baud, O.; et al. Sex differences in the effects of PARP inhibition on microglial phenotypes following neonatal stroke. Brain Behav. Immun. 2018, 73, 375–389. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Martin, B.R.; Adler, M.W.; Razdan, R.K.; Jallo, J.I.; Tuma, R.F. Cannabinoid CB2 receptor activation decreases cerebral infarction in a mouse focal ischemia/reperfusion model. J. Cereb. Blood Flow Metab. 2007, 27, 1387–1396. [Google Scholar] [CrossRef]
- Zhang, M.; Martin, B.R.; Adler, M.W.; Razdan, R.K.; Ganea, D.; Tuma, R.F. Modulation of the balance between cannabinoid CB(1) and CB(2) receptor activation during cerebral ischemic/reperfusion injury. Neuroscience 2008, 152, 753–760. [Google Scholar] [CrossRef]
- Zhang, M.; Mahadevan, A.; Amere, M.; Li, H.; Ganea, D.; Tuma, R.F. Unique Effects of Compounds Active at Both Cannabinoid and Serotonin Receptors During Stroke. Transl. Stroke Res. 2012, 3, 348–356. [Google Scholar] [CrossRef]
- Ronca, R.D.; Myers, A.M.; Ganea, D.; Tuma, R.F.; Walker, E.A.; Ward, S.J. A selective cannabinoid CB2 agonist attenuates damage and improves memory retention following stroke in mice. Life Sci. 2015, 138, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Almogi-Hazan, O.; Or, R. Cannabis, the Endocannabinoid System and Immunity-the Journey from the Bedside to the Bench and Back. Int. J. Mol. Sci. 2020, 21, 4448. [Google Scholar] [CrossRef] [PubMed]
- Huestis, M.A.; Gorelick, D.A.; Heishman, S.J.; Preston, K.L.; Nelson, R.A.; Moolchan, E.T.; Frank, R.A. Blockade of Effects of Smoked Marijuana by the CB1-Selective Cannabinoid Receptor Antagonist SR141716. Arch. Gen. Psychiatry 2001, 58, 322–328. [Google Scholar] [CrossRef]
- Ni, X.; Geller, E.B.; Eppihimer, M.J.; Eisenstein, T.K.; Adler, M.W.; Tuma, R.F. Win 55212-2, a cannabinoid receptor agonist, attenuates leukocyte/endothelial interactions in an experimental autoimmune encephalomyelitis model. Mult. Scler. J. 2004, 10, 158–164. [Google Scholar] [CrossRef]
- Adhikary, S.; Li, H.; Heller, J.; Skarica, M.; Zhang, M.; Ganea, D.; Tuma, R.F. Modulation of Inflammatory Responses by a Cannabinoid-2–Selective Agonist after Spinal Cord Injury. J. Neurotrauma 2011, 28, 2417–2427. [Google Scholar] [CrossRef] [PubMed]
- Fechtner, S.; Singh, A.K.; Srivastava, I.; Szlenk, C.T.; Muench, T.R.; Natesan, S.; Ahmed, S. Cannabinoid Receptor 2 Agonist JWH-015 Inhibits Interleukin-1beta-Induced Inflammation in Rheumatoid Arthritis Synovial Fibroblasts and in Adjuvant Induced Arthritis Rat via Glucocorticoid Receptor. Front. Immunol. 2019, 10, 1027. [Google Scholar] [CrossRef]
- Sharma, C.; Al Kaabi, J.M.; Nurulain, S.M.; Goyal, S.N.; Kamal, M.A.; Ojha, S. Polypharmacological Properties and Therapeutic Potential of β-Caryophyllene: A Dietary Phytocannabinoid of Pharmaceutical Promise. Curr. Pharm. Des. 2016, 22, 3237–3264. [Google Scholar] [CrossRef]
- Gertsch, J.; Leonti, M.; Raduner, S.; Racz, I.; Chen, J.Z.; Xie, X.Q.; Altmann, K.H.; Karsak, M.; Zimmer, A. Beta-caryophyllene is a dietary cannabinoid. Proc. Natl. Acad. Sci. USA 2008, 105, 9099–9104. [Google Scholar] [CrossRef] [PubMed]
- Picciolo, G.; Pallio, G.; Altavilla, D.; Vaccaro, M.; Oteri, G.; Irrera, N.; Squadrito, F. β-Caryophyllene Reduces the Inflammatory Phenotype of Periodontal Cells by Targeting CB2 Receptors. Biomedicines 2020, 8, 164. [Google Scholar] [CrossRef]
- Meeran, M.N.; Al Taee, H.; Azimullah, S.; Tariq, S.; Adeghate, E.; Ojha, S. β-Caryophyllene, a natural bicyclic sesquiterpene attenuates doxorubicin-induced chronic cardiotoxicity via activation of myocardial cannabinoid type-2 (CB(2)) receptors in rats. Chem. Interact. 2019, 304, 158–167. [Google Scholar] [CrossRef]
- Yang, M.; Lv, Y.; Tian, X.; Lou, J.; An, R.; Zhang, Q.; Li, M.; Xu, L.; Dong, Z. Neuroprotective Effect of β-Caryophyllene on Cerebral Ischemia-Reperfusion Injury via Regulation of Necroptotic Neuronal Death and Inflammation: In Vivo and in Vitro. Front. Neurosci. 2017, 11, 583. [Google Scholar] [CrossRef]
- Tian, X.; Liu, H.; Xiang, F.; Xu, L.; Dong, Z. beta-Caryophyllene protects against ischemic stroke by promoting polarization of microglia toward M2 phenotype via the TLR4 pathway. Life Sci. 2019, 237, 116915. [Google Scholar] [CrossRef] [PubMed]
- Hampson, A.J.; Grimaldi, M.; Axelrod, J.; Wink, D. Cannabidiol and (-)Delta9-tetrahydrocannabinol are neuroprotective antioxidants. Proc. Natl. Acad. Sci. USA 1998, 95, 8268–8273. [Google Scholar] [CrossRef]
- Oberbarnscheidt, T.; Miller, N.S. The Impact of Cannabidiol on Psychiatric and Medical Conditions. J. Clin. Med. Res. 2020, 12, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and Anti-Inflammatory Properties of Cannabidiol. Antioxidants 2019, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Kicman, A.; Toczek, M. The Effects of Cannabidiol, a Non-Intoxicating Compound of Cannabis, on the Cardiovascular System in Health and Disease. Int. J. Mol. Sci. 2020, 21, 6740. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, K.; Mishima, K.; Abe, K.; Hasebe, N.; Takamatsu, F.; Yasuda, H.; Ikeda, T.; Inui, K.; Egashira, N.; Iwasaki, K.; et al. Cannabidiol prevents infarction via the non-CB1 cannabinoid receptor mechanism. NeuroReport 2004, 15, 2381–2385. [Google Scholar] [CrossRef] [PubMed]
- Mishima, K.; Hayakawa, K.; Abe, K.; Ikeda, T.; Egashira, N.; Iwasaki, K.; Fujiwara, M. Cannabidiol prevents cerebral infarction via a serotonergic 5-hydroxytryptamine1A receptor-dependent mechanism. Stroke 2005, 36, 1077–1082. [Google Scholar] [CrossRef]
- Hayakawa, K.; Mishima, K.; Nozako, M.; Hazekawa, M.; Irie, K.; Fujioka, M.; Orito, K.; Abe, K.; Hasebe, N.; Egashira, N.; et al. Delayed treatment with cannabidiol has a cerebroprotective action via a cannabinoid receptor-independent myeloperoxidase-inhibiting mechanism. J. Neurochem. 2007, 102, 1488–1496. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, K.; Mishima, K.; Fujiwara, M. Therapeutic Potential of Non-Psychotropic Cannabidiol in Ischemic Stroke. Pharmaceuticals 2010, 3, 2197–2212. [Google Scholar] [CrossRef]
- Ward, S.J.; Castelli, F.; Reichenbach, Z.W.; Tuma, R.F. Surprising outcomes in cannabinoid CB1/CB2 receptor double knockout mice in two models of ischemia. Life Sci. 2018, 195, 1–5. [Google Scholar] [CrossRef]
- Zhang, M.; Adler, M.W.; Abood, M.E.; Ganea, D.; Jallo, J.; Tuma, R.F. CB2 receptor activation attenuates microcirculatory dysfunction during cerebral ischemic/reperfusion injury. Microvasc. Res. 2009, 78, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Martin, B.R.; Adler, M.W.; Razdan, R.J.; Kong, W.; Ganea, D.; Tuma, R.F. Modulation of Cannabinoid Receptor Activation as a Neuroprotective Strategy for EAE and Stroke. J. Neuroimmune Pharmacol. 2009, 4, 249–259. [Google Scholar] [CrossRef]
- Chen, S.; Wang, Y.; Wang, X.; He, M.; Zhang, L.; Dong, Z. PKA-Dependent Membrane Surface Recruitment of CI-AMPARs Is Crucial for BCP-Mediated Protection Against Post-acute Ischemic Stroke Cognitive Impairment. Front. Neurol. 2020, 11, 566067. [Google Scholar] [CrossRef]
- Zarruk, J.G.; Fernández-López, D.; García-Yébenes, I.; García-Gutiérrez, M.S.; Vivancos, J.; Nombela, F.; Torres, M.; Burguete, M.C.; Manzanares, J.; Lizasoain, I.; et al. Cannabinoid type 2 receptor activation downregulates stroke-induced classic and alternative brain macrophage/microglial activation concomitant to neuroprotection. Stroke 2012, 43, 211–219. [Google Scholar] [CrossRef]
- Khaksar, S.; Bigdeli, M.R. Intra-cerebral cannabidiol infusion-induced neuroprotection is partly associated with the TNF-α/TNFR1/NF-kB pathway in transient focal cerebral ischaemia. Brain Inj. 2017, 31, 1932–1943. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.A.; Meyer, E.; Soares, L.M.; Milani, H.; Guimarães, F.S.; de Oliveira, R.M.W. Cannabidiol reduces neuroinflammation and promotes neuroplasticity and functional recovery after brain ischemia. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2017, 75, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Toth, C.C.; Jedrzejewski, N.M.; Ellis, C.L.; Frey, W.H. Cannabinoid-Mediated Modulation of Neuropathic Pain and Microglial Accumulation in a Model of Murine Type I Diabetic Peripheral Neuropathic Pain. Mol. Pain 2010, 6, 16. [Google Scholar] [CrossRef]
- Mohammed, N.; Ceprian, M.; Jimenez, L.; Pazos, M.R.; Martínez-Orgado, J. Neuroprotective Effects of Cannabidiol in Hypoxic Ischemic Insult. The Therapeutic Window in Newborn Mice. CNS Neurol. Disord.-Drug Targets 2017, 16, 102–108. [Google Scholar] [CrossRef]
- Li, H.; Kong, W.; Chambers, C.R.; Yu, D.; Ganea, D.; Tuma, R.F.; Ward, S.J. The non-psychoactive phytocannabinoid cannabidiol (CBD) attenuates pro-inflammatory mediators, T cell infiltration, and thermal sensitivity following spinal cord injury in mice. Cell. Immunol. 2018, 329, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Watson, B.D.; Dietrich, W.D.; Busto, R.; Wachtel, M.S.; Ginsberg, M.D. Induction of reproducible brain infarction by photochemically initiated thrombosis. Ann. Neurol. 1985, 17, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-K.; Park, M.-S.; Kim, Y.-S.; Moon, K.-S.; Joo, S.-P.; Kim, T.-S.; Kim, J.-H.; Kim, S.-H. Photochemically induced cerebral ischemia in a mouse model. Surg. Neurol. 2007, 67, 620–625. [Google Scholar] [CrossRef] [PubMed]
- Deacon, R.M. Measuring the Strength of Mice. J. Vis. Exp. 2013, 76, e2610. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yokubaitis, C.G.; Jessani, H.N.; Li, H.; Amodea, A.K.; Ward, S.J. Effects of Cannabidiol and Beta-Caryophyllene Alone or in Combination in a Mouse Model of Permanent Ischemia. Int. J. Mol. Sci. 2021, 22, 2866. https://doi.org/10.3390/ijms22062866
Yokubaitis CG, Jessani HN, Li H, Amodea AK, Ward SJ. Effects of Cannabidiol and Beta-Caryophyllene Alone or in Combination in a Mouse Model of Permanent Ischemia. International Journal of Molecular Sciences. 2021; 22(6):2866. https://doi.org/10.3390/ijms22062866
Chicago/Turabian StyleYokubaitis, Cody G., Hassan N. Jessani, Hongbo Li, Allison K. Amodea, and Sara Jane Ward. 2021. "Effects of Cannabidiol and Beta-Caryophyllene Alone or in Combination in a Mouse Model of Permanent Ischemia" International Journal of Molecular Sciences 22, no. 6: 2866. https://doi.org/10.3390/ijms22062866
APA StyleYokubaitis, C. G., Jessani, H. N., Li, H., Amodea, A. K., & Ward, S. J. (2021). Effects of Cannabidiol and Beta-Caryophyllene Alone or in Combination in a Mouse Model of Permanent Ischemia. International Journal of Molecular Sciences, 22(6), 2866. https://doi.org/10.3390/ijms22062866




