Is Inflammation a Friend or Foe for Orthodontic Treatment?: Inflammation in Orthodontically Induced Inflammatory Root Resorption and Accelerating Tooth Movement
Abstract
1. Introduction
2. Optimal Orthodontic Force
3. Inflammatory Mediators in Orthodontic Tooth Movement
3.1. Prostaglandins (PGs)
3.2. Cytokines
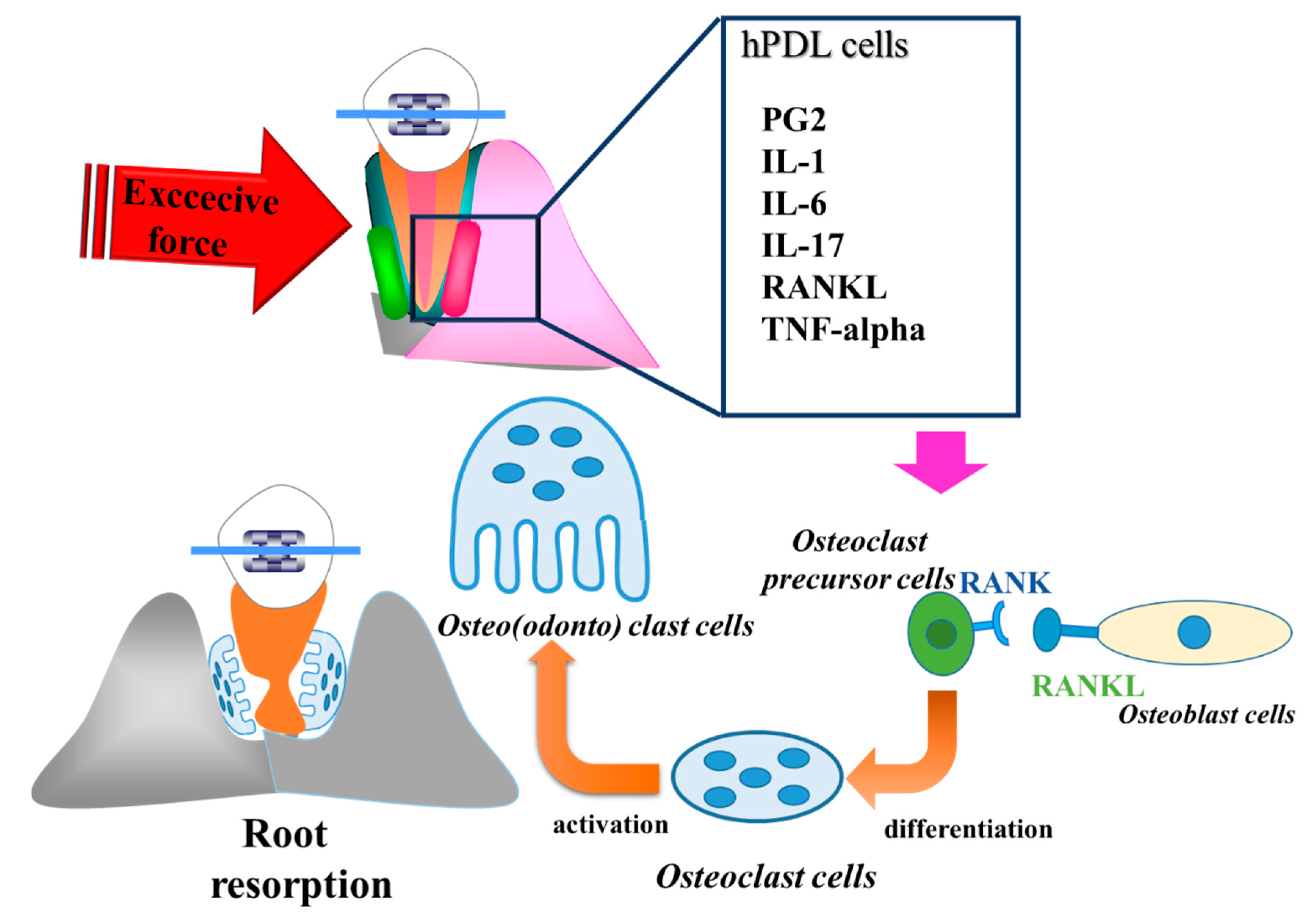
4. Inflammation of Orthodontically Induced Inflammatory Root Resorption (OIIRR) and Inflammation—Is Inflammation a Foe?
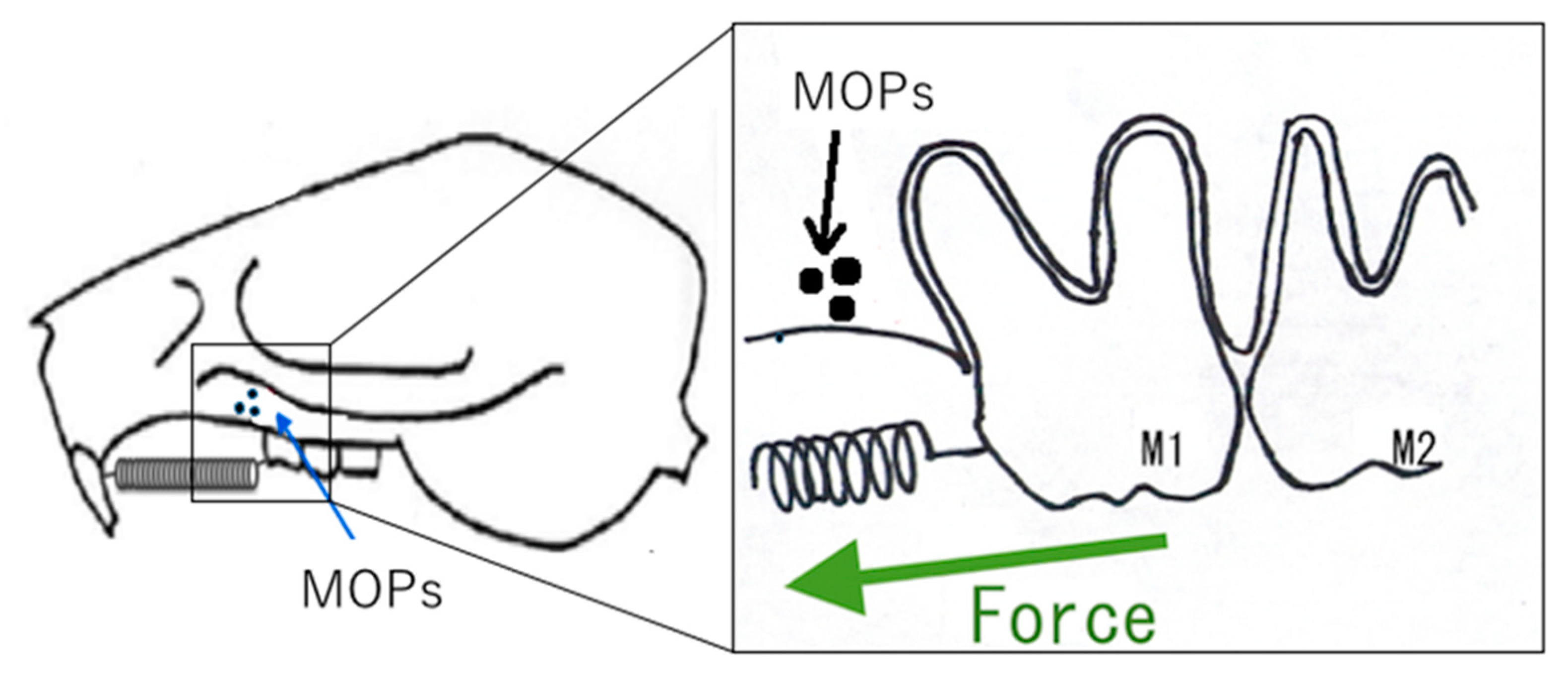
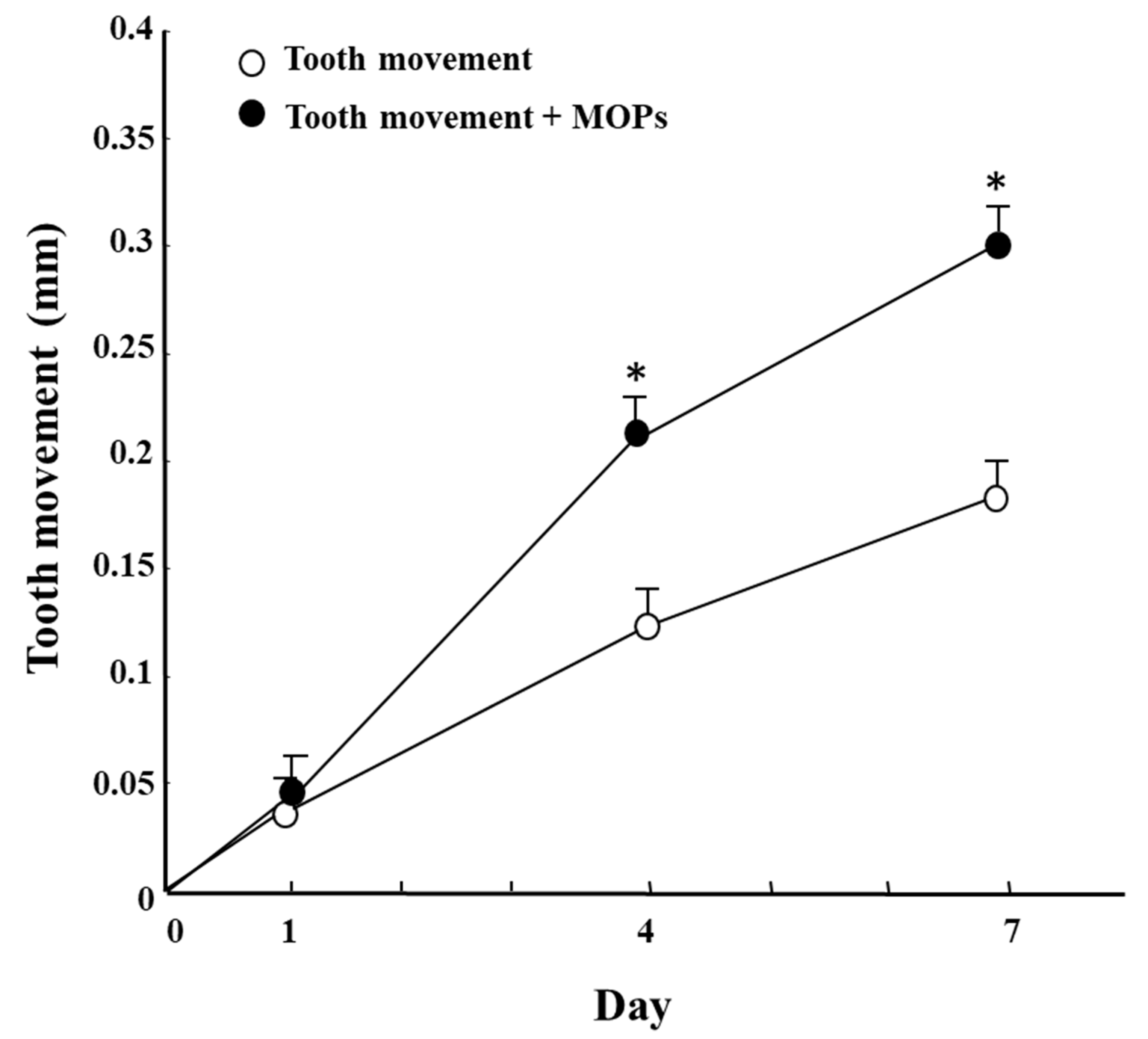
5. Accelerating Orthodontic Tooth Movement (AOTM) and Inflammation—Is Inflammation a Friend?
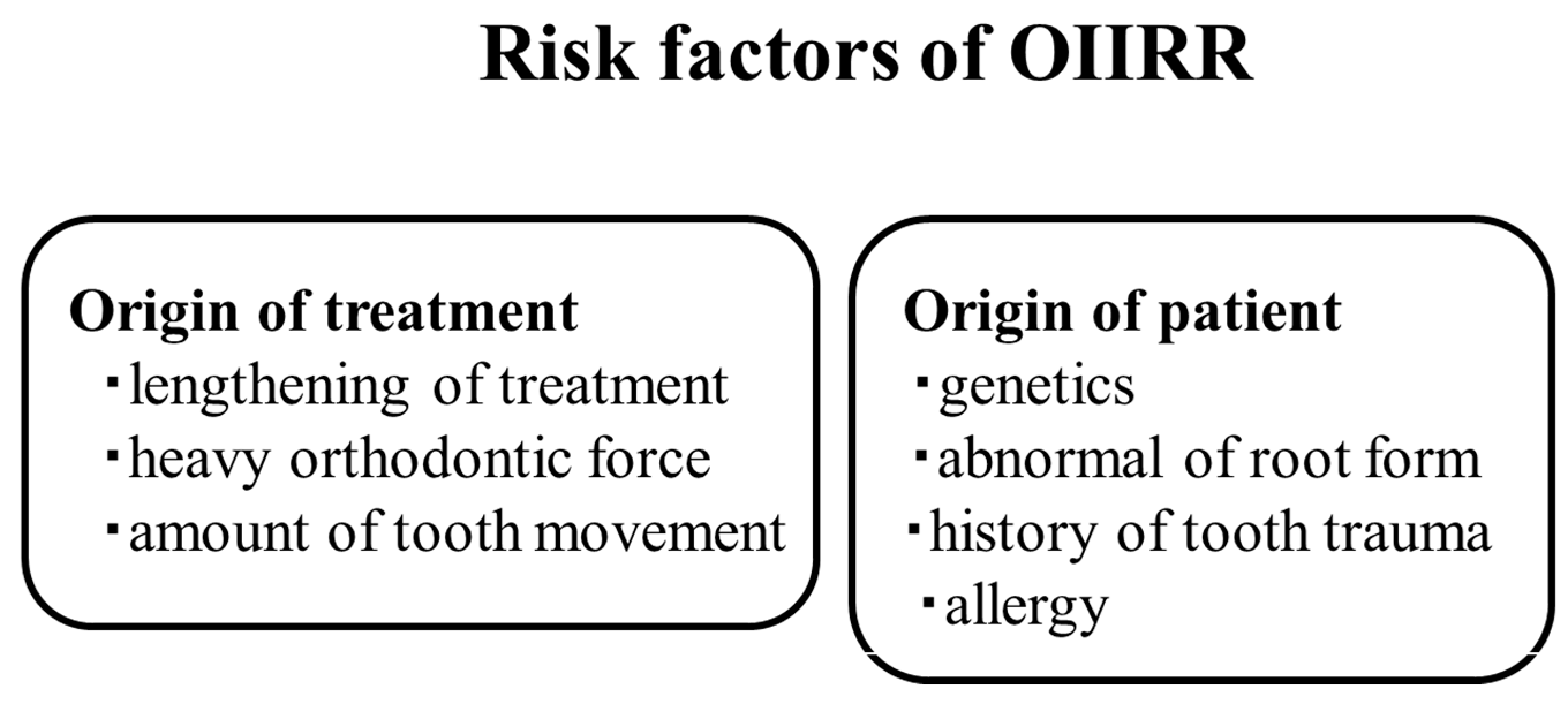
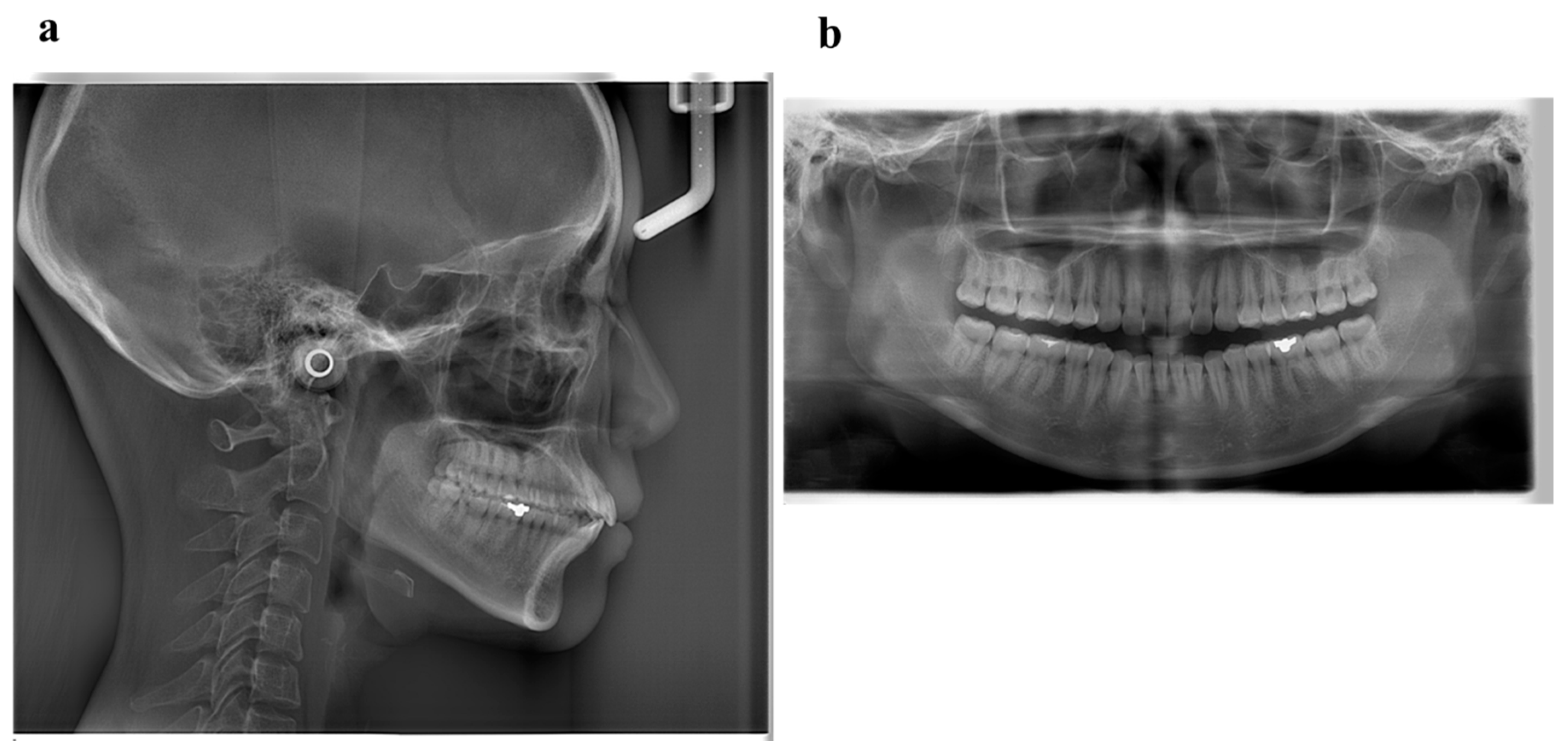
6. Diagnosis and Etiology
6.1. Diagnosis
6.2. Treatment Objectives
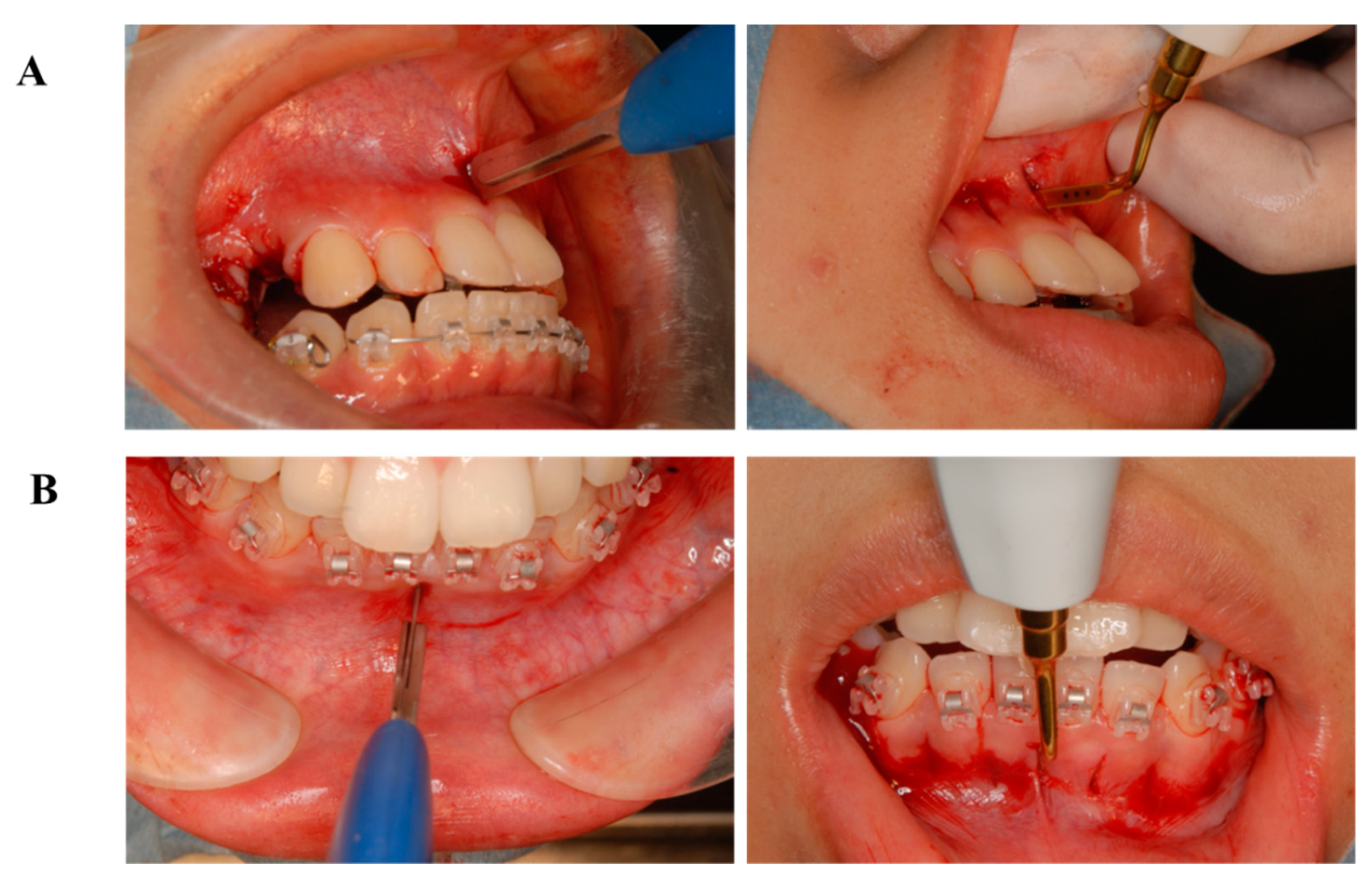
6.3. Treatment Alternatives
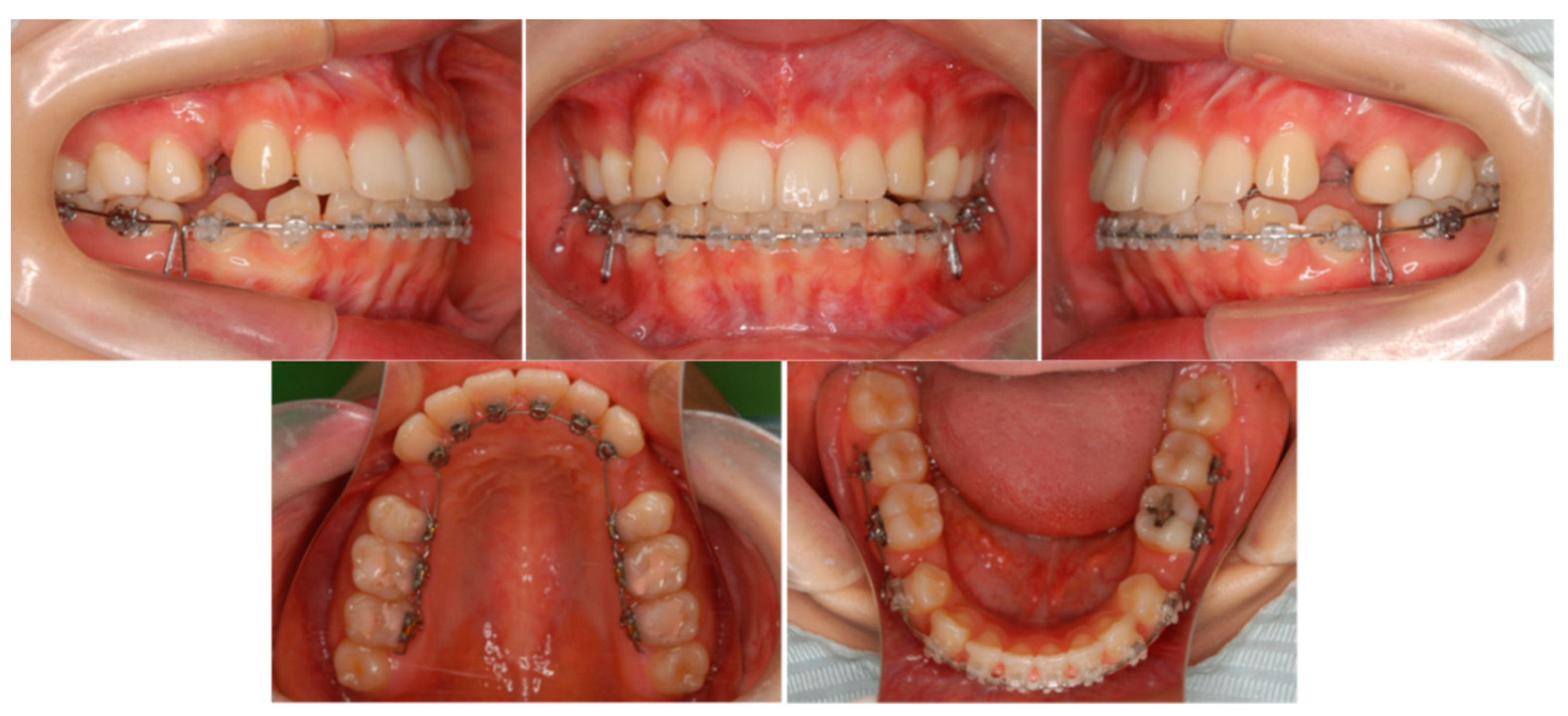
6.4. Treatment Progress
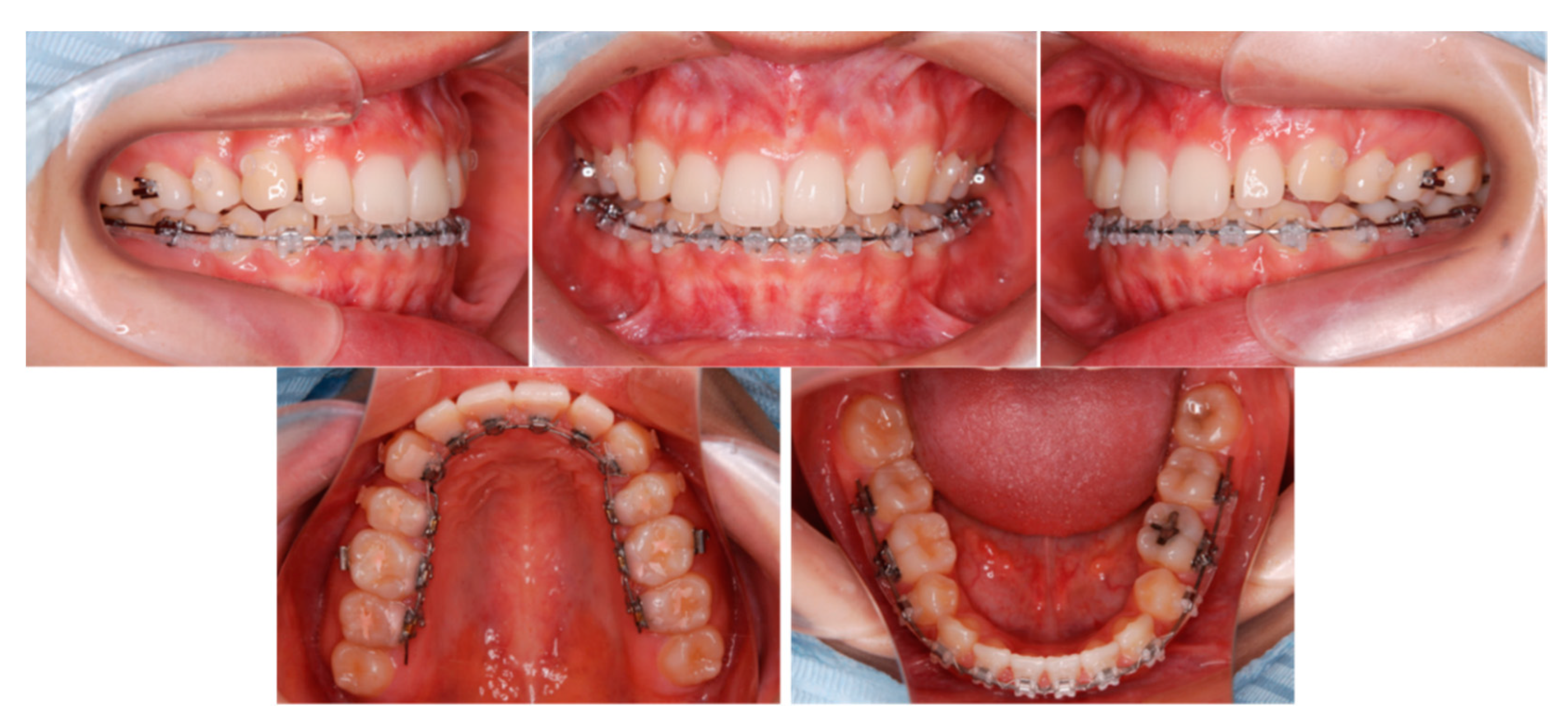
6.5. Treatment Results
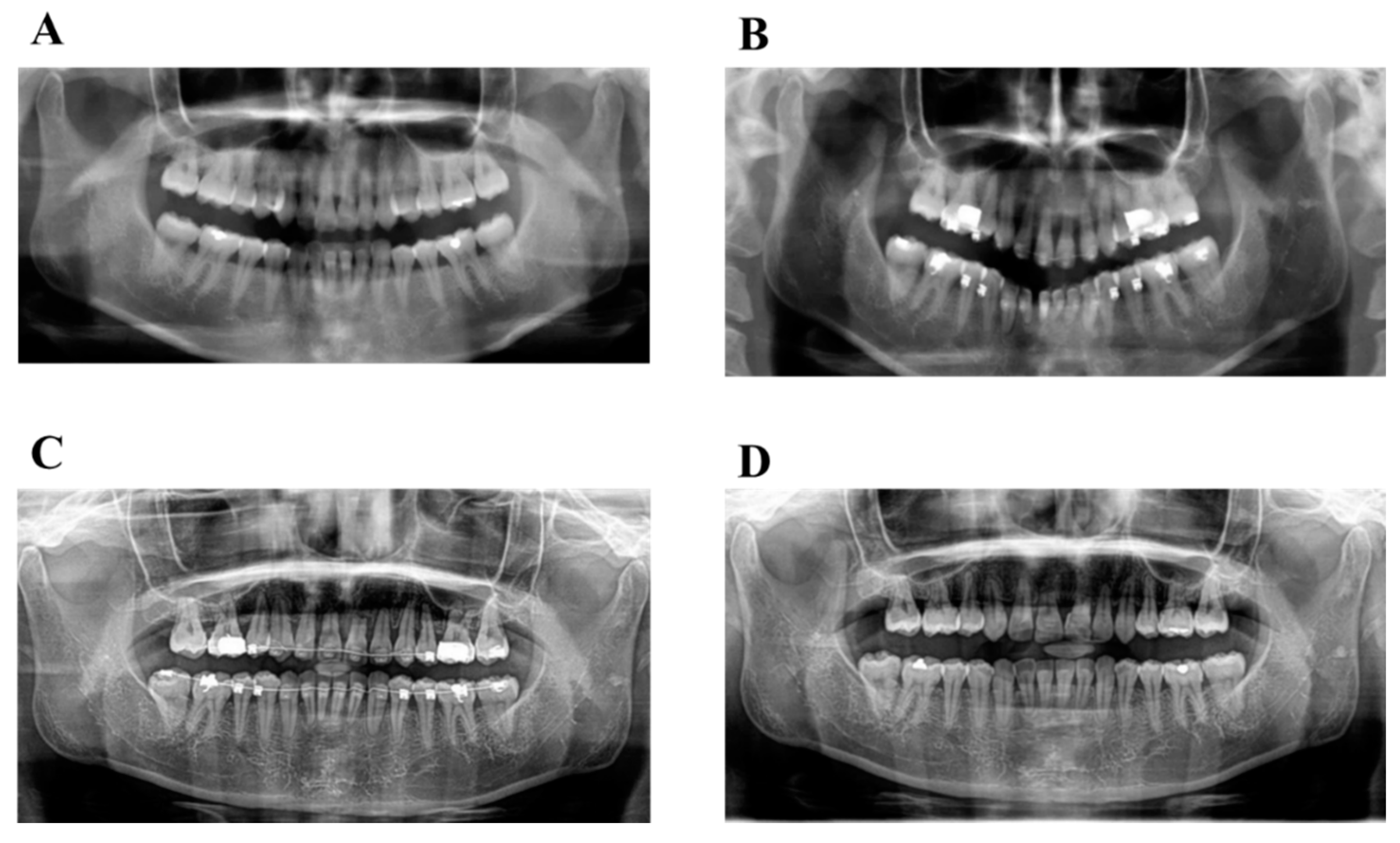
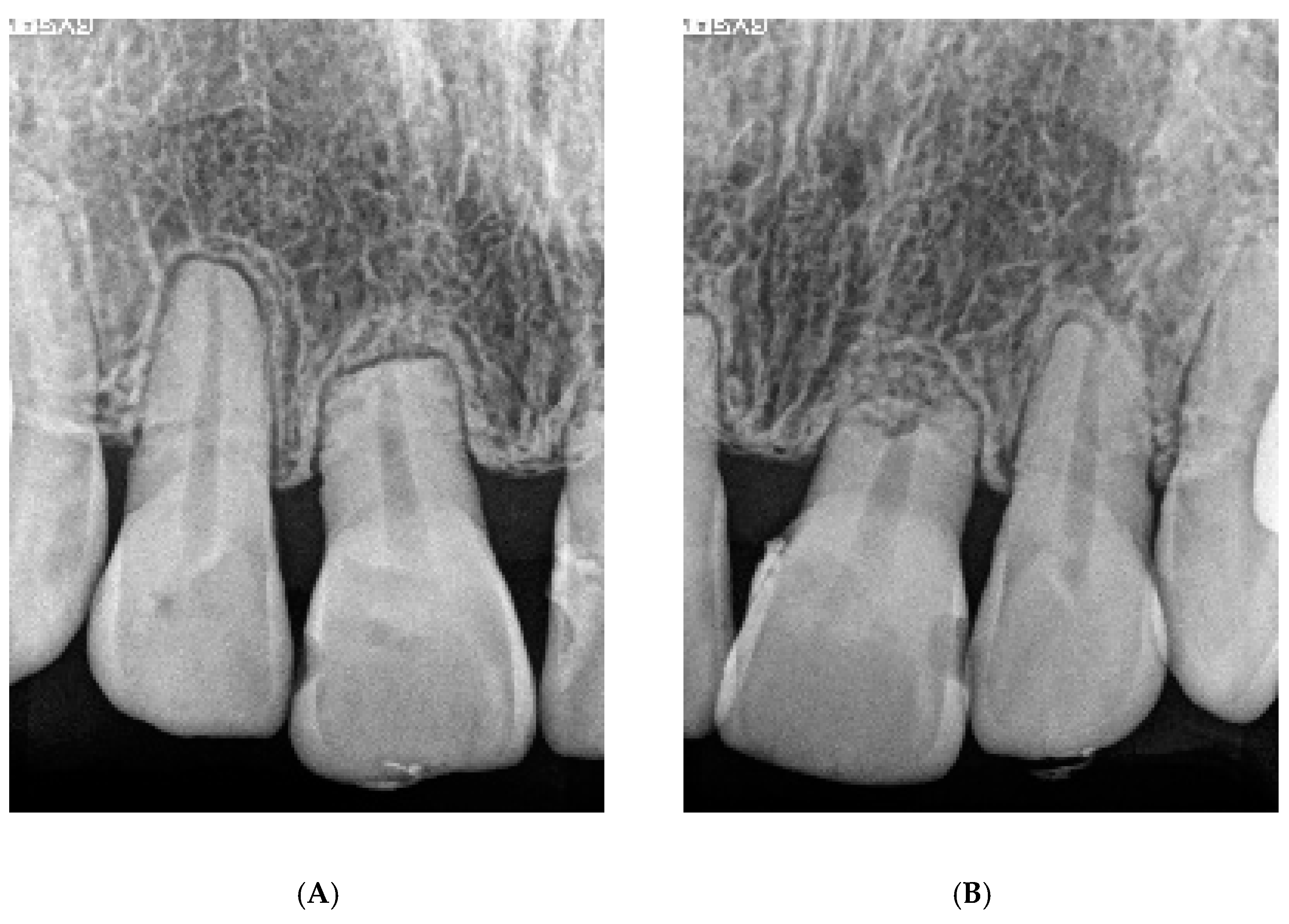
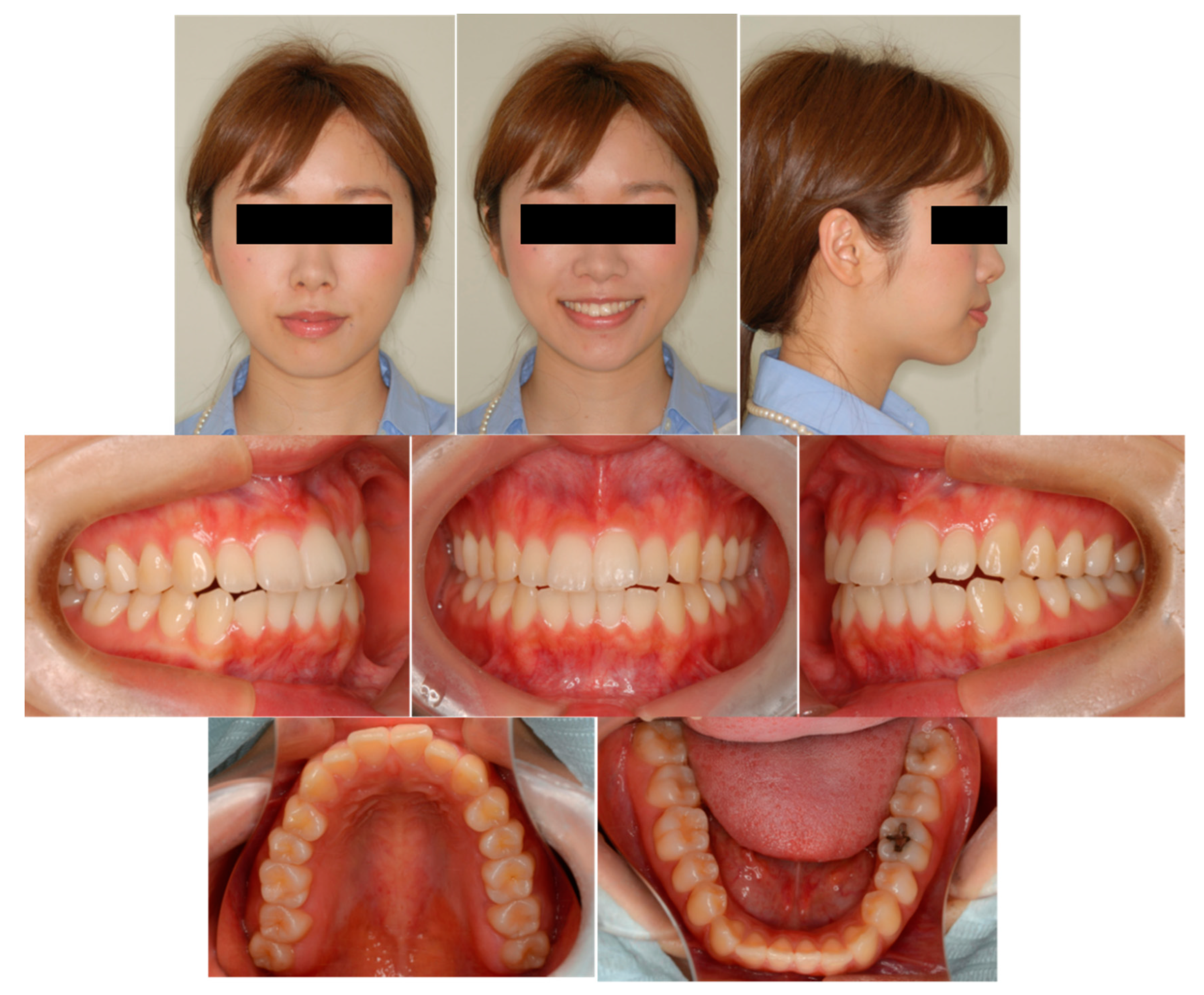
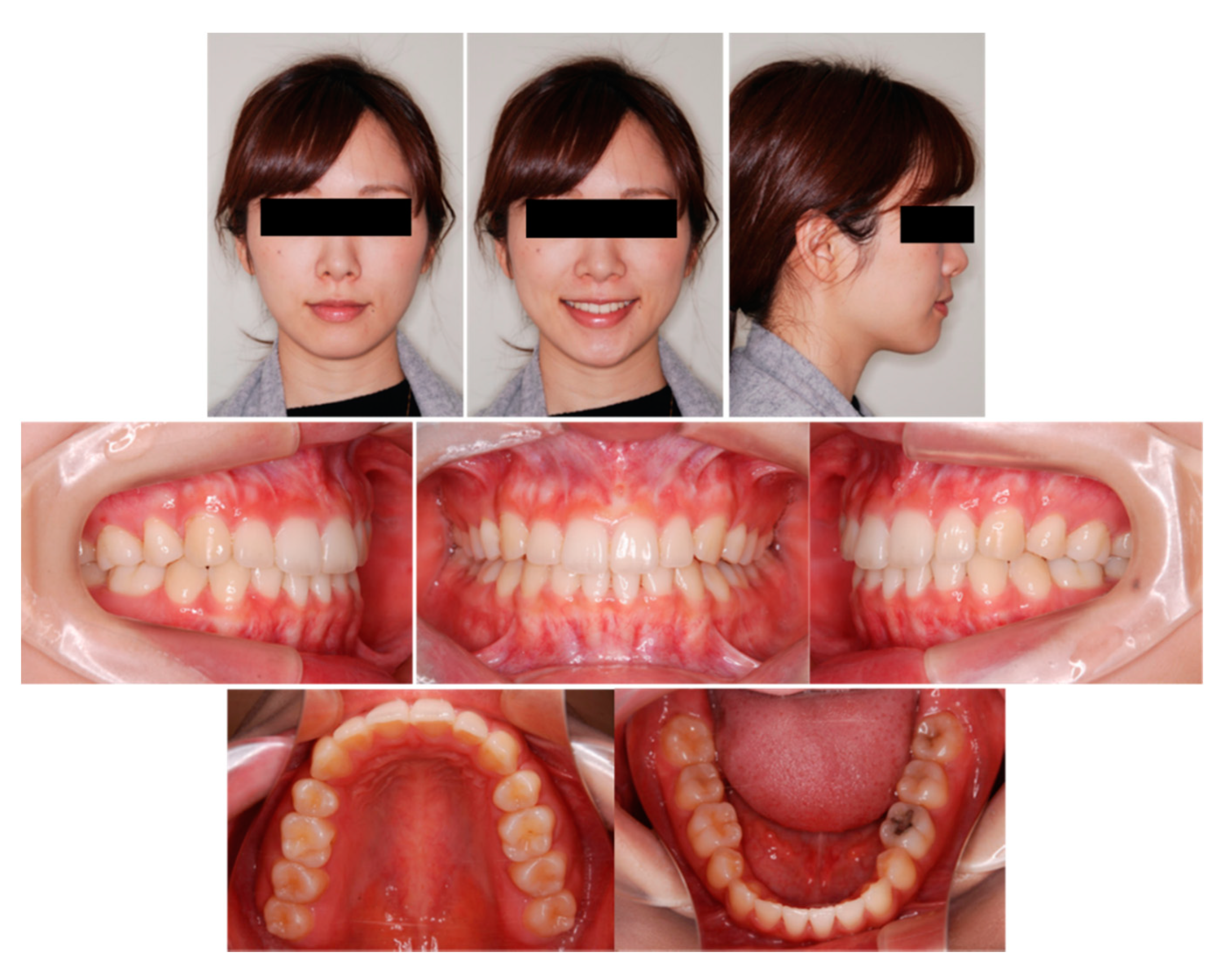
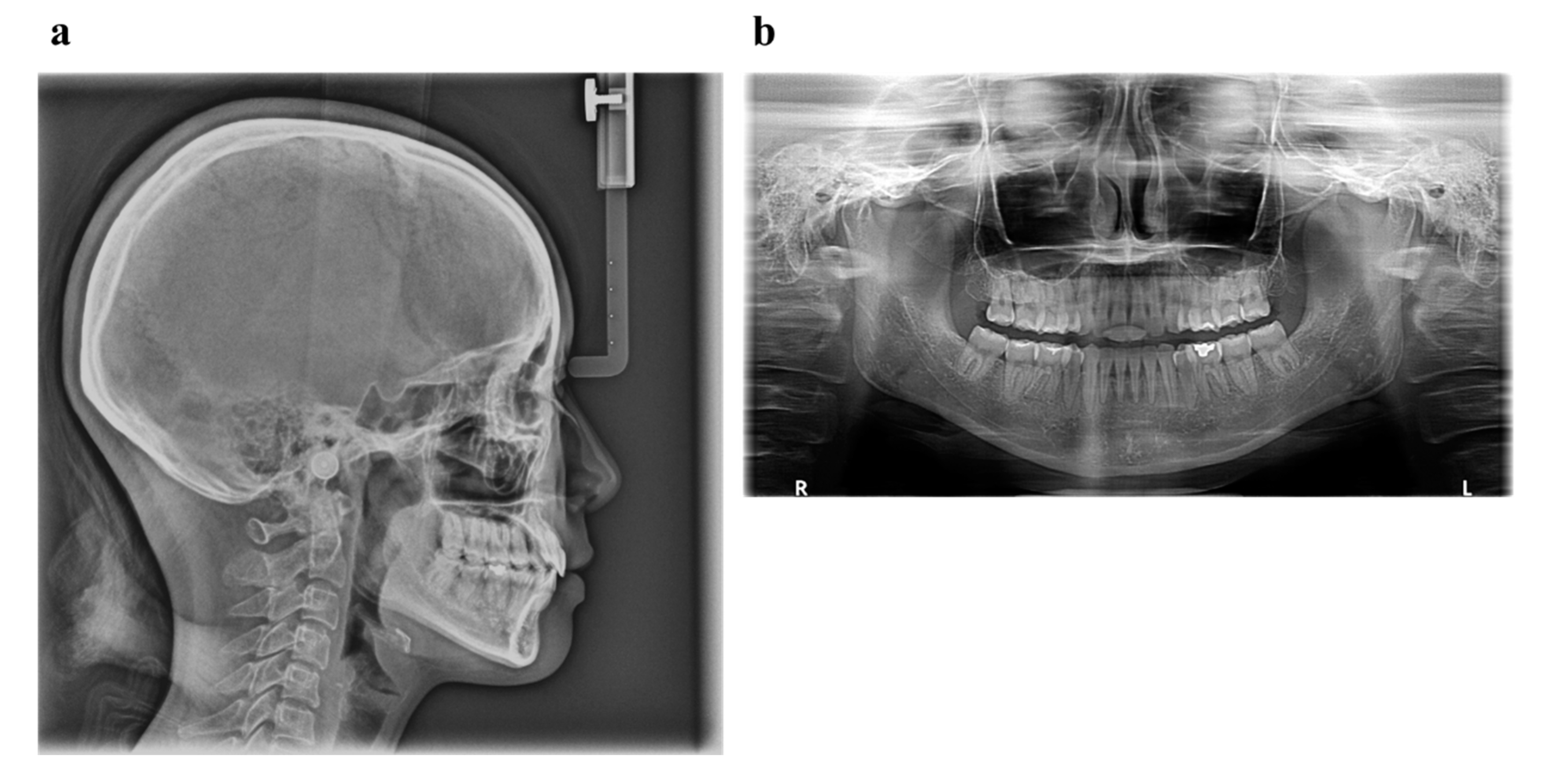
6.6. Case Summary
7. Conclusions
Author Contributions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| OIIRR | orthodontically induced inflammatory root resorption |
| AOTM | accelerating orthodontic tooth movement |
| EARR | external apical root resorption |
| PDL | periodontal ligament |
| RAP | the regional acceleratory phenomenon |
| M-CSF | macrophage-colony stimulating factor |
| RANKL | receptor activator of nuclear factor ligand |
| OTM | orthodontic tooth movement |
| MOPs | micro-osteoperforations |
References
- Vandevska-Radunovic, V. Neural modulation of inflammatory reactions in dental tissues incident to orthodontic tooth movement. A review of the literature. Eur. J. Orthod. 1999, 21, 231–247. [Google Scholar] [CrossRef]
- Davidovitch, Z.; Nicolay, O.F.; Ngan, P.W.; Shanfeld, J.L. Neurotransmitters, cytokines, and the control of alveolar bone remodeling in orthodontics. Dent. Clin. N. Am. 1988, 32, 411–435. [Google Scholar] [PubMed]
- Brezniak, N.; Wasserstein, A. Orthodontically induced inflammatory root resorption. Part II: The clinical aspects. Angle Orthod. 2002, 72, 180–184. [Google Scholar]
- Köle, H. Surgical operations on the alveolar ridge to correct occlusal abnormalities. Oral Surg. Oral Med. Oral Pathol. 1959, 12, 515–529. [Google Scholar] [CrossRef]
- Köle, H. Surgical operations on the alveolar ridge to correct occlusal abnormalities. Oral Surg. Oral Med. Oral Pathol. 1959, 12, 277–288. [Google Scholar] [CrossRef]
- Köle, H. Surgical operations on the alveolar ridge to correct occlusal abnormalities. Oral Surg. Oral Med. Oral Pathol. 1959, 12, 413–420. [Google Scholar] [CrossRef]
- Wilcko, W.M.; Wilcko, T.; Bouquot, J.E.; Ferguson, D.J. Rapid orthodontics with alveolar reshaping: Two case reports of decrowding. Int. J. Periodontics Restor. Dent. 2001, 21, 9–19. [Google Scholar]
- Wilcko, M.T.; Wilcko, W.M.; Pulver, J.J.; Bissada, N.F.; Bouquot, J.E. Accelerated Osteogenic Orthodontics Technique: A 1-Stage Surgically Facilitated Rapid Orthodontic Technique with Alveolar Augmentation. J. Oral Maxillofac. Surg. 2009, 67, 2149–2159. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Maltha, J.C.; Kuijpers-Jagtman, A.M. Optimum force magnitude for orthodontic tooth movement: A systematic literature review. Angle Orthod. 2003, 73, 86–92. [Google Scholar]
- Krishnan, V.; Davidovitch, Z. Cellular, molecular, and tissue-level reactions to orthodontic force. Am. J. Orthod. Dentofac. Orthop. 2006, 129, 469.e1–469.e32. [Google Scholar] [CrossRef]
- Toms, S.R.; Lemons, J.E.; Bartolucci, A.A.; Eberhardt, A.W. Nonlinear stress-strain behavior of periodontal ligament under orthodontic loading. Am. J. Orthod. Dentofac. Orthop. 2002, 122, 174–179. [Google Scholar] [CrossRef]
- Schwarz, A.M. Tissue changes incidental to orthodontic tooth movement. Int. J. Orthod. Oral Surg. Radiogr. 1932, 18, 331–352. [Google Scholar] [CrossRef]
- Oppenheim, A. Human tissue response to orthodontic intervention of short and long duration. Am. J. Orthod. Oral Surg. 1942, 28, 263–301. [Google Scholar] [CrossRef]
- Reitan, K. Clinical and histologic observations on tooth movement during and after orthodontic treatment. Am. J. Orthod. 1967, 53, 721–745. [Google Scholar] [CrossRef]
- Proffit, W.R. Contemporary Orthodontics; Year Book Inc.: St Louis, CA, USA; Elsevier: Mosby, MO, USA, 1999. [Google Scholar]
- Yamaguchi, M.; Garlet, G.P. The role of inflammation in defining the type and pattern of tissue response in orthodontic tooth movement. In Biological Mechanisms of Tooth Movement; Wiley: Hoboken, NJ, USA, 2015; pp. 121–137. [Google Scholar]
- Mitchell, J.A.; Larkin, S.; Williams, T.J. Cyclooxygenase-2: Regulation and relevance in inflammation. Biochem. Pharmacol. 1995, 50, 1535–1542. [Google Scholar] [CrossRef]
- Vane, J.R.; Bakhle, Y.S.; Botting, R.M. Cyclooxygenases 1 and 2. Annu. Rev. Pharmacol. Toxicol. 1998, 38, 97–120. [Google Scholar] [CrossRef]
- Feng, L.; Xia, Y.; Garcia, G.E.; Hwang, D.; Wilson, C.B. Involvement of reactive oxygen intermediates in cyclooxygenase-2 expression induced by interleukin-1, tumor necrosis factor-alpha, and lipopolysaccharide. J. Clin. Investig. 1995, 95, 1669–1675. [Google Scholar] [CrossRef]
- Howell, T.H.; Williams, R.C. Nonsteroidal Antiinflammatory Drugs as Inhibitors of Periodontal Disease Progression. Crit. Rev. Oral Biol. Med. 1993, 4, 177–196. [Google Scholar] [CrossRef]
- Offenbacher, S.; Heasman, P.A.; Collins, J.G. Modulation of host PGE2 secretion as a determinant of periodontal disease expression. J. Periodontol. 1993, 64, 432–444. [Google Scholar]
- Lohinai, Z.; Stachlewitz, R.; Székely, A.; Fehér, E.; Dézsi, L.; Szabó, C. Evidence for the expression of cyclooxygenase-2 enzyme in periodontitis. Life Sci. 2001, 70, 279–290. [Google Scholar] [CrossRef]
- Ngan, P.W.; Saito, S.; Saito, M.; Lanese, R.; Shanfeld, J.; Davidovitch, Z. The interactive effects of mechanical stress and interleukin-1 beta on prostaglandin E and cyclic AMP production in human periodontal ligament fibroblasts in vitro: Comparison with cloned osteoblastic cells of mouse (MC3T3-E1). Arch Oral Biol. 1990, 35, 717–725. [Google Scholar] [CrossRef]
- Shetty, N.; Patil, A.K.; Ganeshkar, S.V.; Hegde, S. Comparison of the effects of ibuprofen and acetaminophen on PGE2 levels in the GCF during orthodontic tooth movement: A human study. Prog. Orthod. 2013, 14, 6. [Google Scholar] [CrossRef] [PubMed]
- Leiker, B.J.; Nanda, R.S.; Currier, G.; Howes, R.I.; Sinha, P.K. The effects of exogenous prostaglandins on orthodontic tooth movement in rats. Am. J. Orthod. Dentofac. Orthop. 1995, 108, 380–388. [Google Scholar] [CrossRef]
- Yamasaki, K.; Shibata, Y.; Imai, S.; Tani, Y.; Shibasaki, Y.; Fukuhara, T. Clinical application of prostaglandin E1 (PGE1) upon orthodontic tooth movement. Am. J. Orthod. 1984, 85, 508–518. [Google Scholar] [CrossRef]
- Chung, C.J.; Baik, H.-S.; Soma, K. Bone formation and tooth movement are synergistically enhanced by administration of EP4 agonist. Am. J. Orthod. Dentofac. Orthop. 2007, 132, 427.e13–427.e20. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, K.; Miura, F.; Suda, T. Prostaglandin as a Mediator of Bone Resorption Induced by Experimental Tooth Movement in Rats. J. Dent. Res. 1980, 59, 1635–1642. [Google Scholar] [CrossRef]
- Dinarello, C.A. Interleukin-1 and Its Biologically Related Cytokines. Adv. Immunol. 1989, 44, 153–205. [Google Scholar] [CrossRef]
- Heinrich, P.C.; Castell, J.V.; Andus, T. Interleukin-6 and the acute phase response. Biochem. J. 1990, 265, 621–636. [Google Scholar] [CrossRef]
- Standiford, T. Anti-inflammatory Cytokines and Cytokine Antagonists. Curr. Pharm. Des. 2000, 6, 633–649. [Google Scholar] [CrossRef]
- Dinarello, C.A. Interleukin-1. Dig Dis Sci. 1988, 33, 25S–35S. [Google Scholar] [CrossRef]
- Gowen, M.; Wood, D.D.; Ihrie, E.J.; McGuire, M.K.B.; Russell, R.G.G. An interleukin 1 like factor stimulates bone resorption in vitro. Nat. Cell Biol. 1983, 306, 378–380. [Google Scholar] [CrossRef] [PubMed]
- Heath, J.K.; Saklatvala, J.; Meikle, M.C.; Atkinson, S.J.; Reynolds, J.J. Pig interleukin 1 (catabolin) is a potent stimulator of bone resorption in vitro. Calcif Tissue Int 1985, 37, 95–97. [Google Scholar] [CrossRef]
- Stashenko, P.; Obernesser, M.S.; Dewhirst, F.E. Effect of immune Cytokines on Bone. Immunol. Investig. 1989, 18, 239–249. [Google Scholar] [CrossRef]
- Lo, Y.J.; Liu, C.M.; Wong, M.Y.; Hou, L.T.; Chang, W.K. Interleukin 1beta-secreting cells in inflamed gingival tissue of adult periodontitis patients. Cytokine 1999, 11, 626–633. [Google Scholar]
- Kanda-Nakamura, C.; Izumi, Y.; Sueda, T. Increased Expression of Interleukin-1 Receptors on Fibroblasts Derived From Inflamed Gingiva. J. Periodontol. 1996, 67, 1267–1273. [Google Scholar] [CrossRef]
- Ishimi, Y.; Miyaura, C.; Jin, C.H.; Akatsu, T.; Abe, E.; Nakamura, Y.; Yamaguchi, A.; Yoshiki, S.; Matsuda, T.; Hirano, T. IL-6 is produced by osteoblasts and induces bone resorption. J. Immunol. 1990, 145, 3297–3303. [Google Scholar]
- Kurihara, N.; Bertolini, D.; Suda, T.; Akiyama, Y.; Roodman, G.D. IL-6 stimulates osteoclast-like multinucleated cell formation in long term human marrow cultures by inducing IL-1 release. J. Immunol. 1990, 144, 4226–4230. [Google Scholar]
- Löwik, C.; Van Der Pluijm, G.; Bloys, H.; Hoekman, K.; Bijvoet, O.; Aarden, L.; Papapoulos, S. Parathyroid hormone (PTH) and PTH-like protein (PLP) stimulate interleukin-6 production by osteogenic cells: A possible role of interleukin-6 in osteoclastogenesis. Biochem. Biophys. Res. Commun. 1989, 162, 1546–1552. [Google Scholar] [CrossRef]
- Bartold, P.M.; Haynes, D.R. Interleukin-6 production by human gingival fibroblasts. J. Periodontal Res. 1991, 26, 339–345. [Google Scholar] [CrossRef] [PubMed]
- May, L.T.; Ghrayeb, J.; Santhanam, U.; Tatter, S.B.; Sthoeger, Z.; Helfgott, D.C.; Chiorazzi, N.; Grieninger, G.; Sehgal, P.B. Synthesis and secretion of multiple forms of beta 2-interferon/B-cell differentiation factor 2/hepatocyte-stimulating factor by human fibroblasts and monocytes. J. Biol. Chem. 1988, 263, 7760–7766. [Google Scholar] [CrossRef]
- Irwin, C.R.; Myrillas, T.T. The role of IL-6 in the pathogenesis of periodontal disease. Oral Dis. 2008, 4, 43–47. [Google Scholar] [CrossRef]
- Yakovlev, E.; Kalichman, I.; Pisanti, S.; Shoshan, S.; Barak, V. Levels of Cytokines and Collagen Type I and Type III as a Function of Age in Human Gingivitis. J. Periodontol. 1996, 67, 788–793. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, N.; Ogura, N.; Yamaguchi, M.; Goseki, T.; Shibata, Y.; Abiko, Y.; Iwasawa, T.; Takiguchi, H. Stimulation by interleukin-1 of interleukin-6 production by human periodontal ligament cells. Arch. Oral Biol. 1992, 37, 743–748. [Google Scholar] [CrossRef]
- Yao, Z.; Painter, S.L.; Fanslow, W.C.; Ulrich, D.; MacDuff, B.M.; Spriggs, M.K.; Armitage, R.J. Human IL-17: A novel cytokine derived from T cells. J. Immunol. 1995, 155, 5483–5486. [Google Scholar]
- Kotake, S.; Udagawa, N.; Takahashi, N.; Matsuzaki, K.; Itoh, K.; Ishiyama, S.; Saito, S.; Inoue, K.; Kamatani, N.; Gillespie, M.T.; et al. IL-17 in synovial fluids from patients with rheumatoid arthritis is a potent stimulator of osteoclastogenesis. J. Clin. Investig. 1999, 103, 1345–1352. [Google Scholar] [CrossRef] [PubMed]
- Ishizu, T.; Osoegawa, M.; Mei, F.-J.; Kikuchi, H.; Tanaka, M.; Takakura, Y.; Minohara, M.; Murai, H.; Mihara, F.; Taniwaki, T.; et al. Intrathecal activation of the IL-17/IL-8 axis in opticospinal multiple sclerosis. Brain 2005, 128, 988–1002. [Google Scholar] [CrossRef] [PubMed]
- Molet, S.; Hamid, Q.; Davoineb, F.; Nutku, E.; Tahaa, R.; Pagé, N.; Olivenstein, R.; Elias, J.; Chakir, J. IL-17 is increased in asthmatic airways and induces human bronchial fibroblasts to produce cytokines. J. Allergy Clin. Immunol. 2001, 108, 430–438. [Google Scholar] [CrossRef]
- Yago, T.; Nanke, Y.; Ichikawa, N.; Kobashigawa, T.; Mogi, M.; Kamatani, N.; Kotake, S. IL-17 induces osteoclastogenesis from human monocytes alone in the absence of osteoblasts, which is potently inhibited by anti-TNF-α antibody: A novel mechanism of osteoclastogenesis by IL-17. J. Cell. Biochem. 2009, 108, 947–955. [Google Scholar] [CrossRef]
- Johnson, R.; Wood, N.; Serio, F. Interleukin-11 and IL-17 and the Pathogenesis of Periodontal Disease. J. Periodontol. 2004, 75, 37–43. [Google Scholar] [CrossRef]
- Honda, T.; Aoki, Y.; Takahashi, N.; Maekawa, T.; Nakajima, T.; Ito, H.; Tabeta, K.; Okui, T.; Kajita, K.; Domon, H.; et al. Elevated expression of IL-17 and IL-12 genes in chronic inflammatory periodontal disease. Clin. Chim. Acta 2008, 395, 137–141. [Google Scholar] [CrossRef]
- Roberts, F.A.; McCaffery, K.A.; Michalek, S.M. Profile of Cytokine mRNA Expression in Chronic Adult Periodontitis. J. Dent. Res. 1997, 76, 1833–1839. [Google Scholar] [CrossRef] [PubMed]
- Rossomando, E.; Kennedy, J.; Hadjimichael, J. Tumour necrosis factor alpha in gingival crevicular fluid as a possible indicator of periodontal disease in humans. Arch. Oral Biol. 1990, 35, 431–434. [Google Scholar] [CrossRef]
- Stashenko, P.; Jandinski, J.J.; Fujiyoshi, P.; Rynar, J.; Socransky, S.S. Tissue Levels of Bone Resorptive Cytokines in Periodontal Disease. J. Periodontol. 1991, 62, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Takeichi, O.; Saito, I.; Tsurumachi, T.; Moro, I.; Saito, T. Expression of Inflammatory Cytokine Genes In Vivo by Human Alveolar Bone-Derived Polymorphonuclear Leukocytes Isolated from Chronically Inflamed Sites of Bone Resorption. Calcif. Tissue Int. 1996, 58, 244–248. [Google Scholar] [CrossRef]
- Gašperšič, R.; Štiblar-MartinČiČ, D.; Osredkar, J.; Skalerič, U. Influence of subcutaneous administration of recombinant TNF-α on ligature-induced periodontitis in rats. J. Periodontal Res. 2003, 38, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-J.; Kang, I.-K.; Chung, C.-P.; Choi, S.-M. The subgingival microflora and gingival crevicular fluid cytokines in refractory periodontitis. J. Clin. Periodontol. 1995, 22, 885–890. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, D.E.; Hariyani, N.; Indrawati, R.; Ridwan, R.D.; Diyatri, I. Cytokines and Chemokines in Periodontitis. Eur. J. Dent. 2020, 14, 483–495. [Google Scholar] [CrossRef]
- Saito, M.; Saito, S.; Ngan, P.W.; Shanfeld, J.; Davidovitch, Z. Interleukin 1 beta and prostaglandin E are involved in the response of periodontal cells to mechanical stress in vivo and in vitro. Am. J. Orthod. Dentofac. Orthop. 1991, 99, 226–240. [Google Scholar] [CrossRef]
- Baba, S.; Kuroda, N.; Arai, C.; Nakamura, Y.; Sato, T. Immunocompetent cells and cytokine expression in the rat periodontal ligament at the initial stage of orthodontic tooth movement. Arch. Oral Biol. 2011, 56, 466–473. [Google Scholar] [CrossRef]
- VanSant, L.; De Llano-Pérula, M.C.; Verdonck, A.; Willems, G. Expression of biological mediators during orthodontic tooth movement: A systematic review. Arch. Oral Biol. 2018, 95, 170–186. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Wang, C.-L.; Koyama, Y.; Mitsui, N.; Shionome, C.; Sanuki, R.; Suzuki, N.; Mayahara, K.; Shimizu, N.; Maeno, M. Compressive force stimulates the gene expression of IL-17s and their receptors in MC3T3-E1 cells. Connect. Tissue Res. 2010, 51, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Bletsa, A.; Berggreen, E.; Brudvik, P. Interleukin-1? and tumor necrosis factor-? expression during the early phases of orthodontic tooth movement in rats. Eur. J. Oral Sci. 2006, 114, 423–429. [Google Scholar] [CrossRef]
- Garlet, T.P.; Coelho, U.; Silva, J.S.; Garlet, G.P. Cytokine expression pattern in compression and tension sides of the periodontal ligament during orthodontic tooth movement in humans. Eur. J. Oral Sci. 2007, 115, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Vissink, A. Cytokines in crevicular fluid and orthodontic tooth movement. Eur. J. Oral Sci. 2008, 116, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Jayaprakash, P.K.; Basavanna, J.M.; Grewal, H.; Modi, P.; Sapawat, P.; Bohara, P.D. Elevated levels of Interleukin (IL)-1beta, IL-6, tumor necrosis factor-alpha, epidermal growth factor, and beta2-microglobulin levels in gingival crevicular fluid during human Orthodontic tooth movement (OTM). J. Fam. Med. Prim. Care 2019, 8, 1602–1606. [Google Scholar] [CrossRef]
- Padisar, P.; Hashemi, R.; Naseh, M.; Nikfarjam, B.A.; Mohammadi, M. Assessment of tumor necrosis factor alpha (TNFalpha) and interleukin 6 level in gingival crevicular fluid during orthodontic tooth movement: A randomized split-mouth clinical trial. Electron. Physician 2018, 10, 7146–7154. [Google Scholar] [CrossRef]
- Lin, T.; Yang, L.; Zheng, W.; Zhang, B. Th17 Cytokines and its Correlation with Receptor Activator of Nuclear Factor kappa B Ligand During Orthodontic Tooth Movement. Iran J. Immunol. 2020, 17, 137–143. [Google Scholar]
- Lacey, D.; Timms, E.; Tan, H.-L.; Kelley, M.; Dunstan, C.; Burgess, T.; Elliott, R.; Colombero, A.; Elliott, G.; Scully, S.; et al. Osteoprotegerin Ligand Is a Cytokine that Regulates Osteoclast Differentiation and Activation. Cell 1998, 93, 165–176. [Google Scholar] [CrossRef]
- Yasuda, H.; Shima, N.; Nakagawa, N.; Yamaguchi, K.; Kinosaki, M.; Mochizuki, S.-I.; Tomoyasu, A.; Yano, K.; Goto, M.; Murakami, A.; et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc. Natl. Acad. Sci. USA 1998, 95, 3597–3602. [Google Scholar] [CrossRef] [PubMed]
- Haynes, D.R.; Atkins, G.J.; Loric, M.; Crotti, T.N.; Geary, S.M.; Findlay, D.M. Bidirectional signaling between stromal and hemopoietic cells regulates interleukin-1 expression during human osteoclast formation. Bone 1999, 25, 269–278. [Google Scholar] [CrossRef]
- Yasuda, H.; Shima, N.; Nakagawa, N.; Mochizuki, S.-I.; Yano, K.; Fujise, N.; Sato, Y.; Goto, M.; Yamaguchi, K.; Kuriyama, M.; et al. Identity of Osteoclastogenesis Inhibitory Factor (OCIF) and Osteoprotegerin (OPG): A Mechanism by which OPG/OCIF Inhibits Osteoclastogenesisin Vitro1. Endocrinology 1998, 139, 1329–1337. [Google Scholar] [CrossRef] [PubMed]
- Bucay, N.; Sarosi, I.; Dunstan, C.R.; Morony, S.; Tarpley, J.; Capparelli, C.; Scully, S.; Tan, H.L.; Xu, W.; Lacey, D.L.; et al. osteoprotegerin-deficient mice develop early onset osteoporosis and arterial calcification. Genes Dev. 1998, 12, 1260–1268. [Google Scholar] [CrossRef]
- Kong, Y.-Y.; Yoshida, H.; Sarosi, I.; Tan, H.-L.; Timms, E.; Capparelli, C.; Morony, S.; Oliveira-Dos-Santos, A.J.; Van, G.; Itie, A.; et al. OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymph-node organogenesis. Nat. Cell Biol. 1999, 397, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, A.; Amizuka, N.; Irie, K.; Murakami, A.; Fujise, N.; Kanno, T.; Sato, Y.; Nakagawa, N.; Yasuda, H.; Mochizuki, S.; et al. Severe osteoporosis in mice lacking osteoclastogenesis inhibitory factor/osteoprotegerin. Biochem. Biophys. Res. Commun. 1998, 247, 610–615. [Google Scholar] [CrossRef] [PubMed]
- Crotti, T.N.; Smith, M.D.; Findlay, D.M.; Zreiqat, H.; Ahern, M.J.; Weedon, H.; Hatzinikolous, G.; Capone, M.; Holding, C.; Haynes, D.R. Factors regulating osteoclast formation in human tissues adjacent to peri-implant bone loss: Expression of receptor activator NFkappaB, RANK ligand and osteoprotegerin. Biomaterials 2004, 25, 565–573. [Google Scholar] [CrossRef]
- Crotti, T.; Smith, M.D.; Hirsch, R.; Soukoulis, S.; Weedon, H.; Capone, M.; Ahern, M.J.; Haynes, D. Receptor activator NF kappaB ligand (RANKL) and osteoprotegerin (OPG) protein expression in periodontitis. J. Periodont Res. 2003, 38, 380–387. [Google Scholar] [CrossRef]
- Crotti, T.N.; Smith, M.D.; Weedon, H.; Ahern, M.J.; Findlay, D.M.; Kraan, M.; Tak, P.P.; Haynes, D.R. Receptor activator NF-kappaB ligand (RANKL) expression in synovial tissue from patients with rheumatoid arthritis, spondyloarthropathy, osteoarthritis, and from normal patients: Semiquantitative and quantitative analysis. Ann. Rheum. Dis. 2002, 61, 1047–1054. [Google Scholar] [CrossRef]
- Haynes, D.R.; Barg, E.; Crotti, T.N.; Holding, C.; Weedon, H.; Atkins, G.J.; Zannetino, A.; Ahern, M.J.; Coleman, M.; Roberts-Thomson, P.J.; et al. Osteoprotegerin expression in synovial tissue from patients with rheumatoid arthritis, spondyloarthropathies and osteoarthritis and normal controls. Rheumatology 2003, 42, 123–134. [Google Scholar] [CrossRef]
- Liu, D.; Xu, J.K.; Figliomeni, L.; Huang, L.; Pavlos, N.J.; Rogers, M.; Tan, A.; Price, P.; Zheng, M.H. Expression of RANKL and OPG mRNA in periodontal disease: Possible involvement in bone destruction. Int. J. Mol. Med. 2003, 11, 17–21. [Google Scholar] [CrossRef]
- Ogasawara, T.; Yoshimine, Y.; Kiyoshima, T.; Kobayashi, I.; Matsuo, K.; Akamine, A.; Sakai, H. In situ expression of RANKL, RANK, osteoprotegerin and cytokines in osteoclasts of rat periodontal tissue. J. Periodontal Res. 2003, 39, 42–49. [Google Scholar] [CrossRef]
- Feldmann, M.; Brennan, F.M.; Maini, R.N. Role of Cytokines in Rheumatoid Arthritis. Annu. Rev. Immunol. 1996, 14, 397–440. [Google Scholar] [CrossRef]
- Suda, T.; Takahashi, N.; Udagawa, N.; Jimi, E.; Gillespie, M.T.; Martin, T.J. Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr. Rev. 1999, 20, 345–357. [Google Scholar] [CrossRef] [PubMed]
- De Molon, R.S.; Park, C.H.; Jin, Q.; Sugai, J.; Cirelli, J.A. Characterization of ligature-induced experimental periodontitis. Microsc. Res. Tech. 2018, 81, 1412–1421. [Google Scholar] [CrossRef]
- Kanzaki, H.; Chiba, M.; Shimizu, Y.; Mitani, H. Periodontal ligament cells under mechanical stress induce osteoclastogenesis by receptor activator of nuclear factor kappaB ligand up-regulation via prostaglandin E2 synthesis. J. Bone. Miner. Res. 2002, 17, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Nishijima, Y.; Yamaguchi, M.; Kojima, T.; Aihara, N.; Nakajima, R.; Kasai, K. Levels of RANKL and OPG in gingival crevicular fluid during orthodontic tooth movement and effect of compression force on releases from periodontal ligament cells in vitro. Orthod. Craniofacial Res. 2006, 9, 63–70. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Aihara, N.; Kojima, T.; Kasai, K. RANKL Increase in Compressed Periodontal Ligament Cells from Root Resorption. J. Dent. Res. 2006, 85, 751–756. [Google Scholar] [CrossRef]
- Aihara, N.; Otsuka, A.; Yamaguchi, M. Localization of RANKL and cathepsin K, B, and L in rat periodontal tissues during experimental tooth movement. Orthod. Waves. 2005, 64, 107–113. [Google Scholar]
- Menezes, R.; Garlet, T.P.; Letra, A.; Bramante, C.M.; Campanelli, A.P.; Figueira, R.D.C.; Sogayar, M.C.; Granjeiro, J.M.; Garlet, G.P. Differential Patterns of Receptor Activator of Nuclear Factor Kappa B Ligand/Osteoprotegerin Expression in Human Periapical Granulomas: Possible Association with Progressive or Stable Nature of the Lesions. J. Endod. 2008, 34, 932–938. [Google Scholar] [CrossRef]
- Kanzaki, H.; Chiba, M.; Takahashi, I.; Haruyama, N.; Nishimura, M.; Mitani, H. Local OPG Gene Transfer to Periodontal Tissue Inhibits Orthodontic Tooth Movement. J. Dent. Res. 2004, 83, 920–925. [Google Scholar] [CrossRef] [PubMed]
- Kanzaki, H.; Chiba, M.; Arai, K.I.; Takahashi, I.; Haruyama, N.; Nishimura, M.; Mitani, H. Local RANKL gene transfer to the periodontal tissue accelerates orthodontic tooth movement. Gene Ther. 2006, 13, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M. RANK/RANKL/OPG during orthodontic tooth movement. Orthod. Craniofacial Res. 2009, 12, 113–119. [Google Scholar] [CrossRef]
- Chan, E. Physical properties of root cementum: Part5. Volumetic analysis of root resorption craters after application of light and heavy orthodontic forces. Am. J. Orthod. Dentofac. Orhop. 2005, 12, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Levander, E.; Malmgren, O.; Stenback, K. Apical root resorption during orthodontic treatment of patients with multiple aplasia: A study of maxillary incisors. Eur. J. Orthod. 1998, 20, 427–434. [Google Scholar] [CrossRef]
- Al-Qawasmi, R.; Hartsfield, J.J.; Everett, E.; Flury, L.; Liu, L.; Foroud, T.; Macri, J.; Roberts, W. Genetic Predisposition to External Apical Root Resorption in Orthodontic Patients: Linkage of Chromosome-18 Marker. J. Dent. Res. 2003, 82, 356–360. [Google Scholar] [CrossRef] [PubMed]
- Reitan, K. Tissue reaction as related to the age factor. Dental Record 1954, 74, 271–278. [Google Scholar]
- Malmgren, O.; Goldson, L.; Hill, C.; Orwin, A.; Petrini, L.; Lundberg, M. Root resorption after orthodontic treatment of traumatized teeth. Am. J. Orthod. 1982, 82, 487–491. [Google Scholar] [CrossRef]
- Shimizu, M.; Yamaguchi, M.; Fujita, S.; Utsunomiya, T.; Yamamoto, H.; Kasai, K. Interleukin-17/T-helper 17 cells in an atopic dermatitis mouse model aggravate orthodontic root resorption in dental pulp. Eur. J. Oral Sci. 2013, 121, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Ukai, T.; Kaneko, T.; Yoshinaga, M.; Yokoyama, M.; Ozaki, Y.; Hara, Y. T cells are able to promote lipopolysaccharide-induced bone resorption in mice in the absence of B cells. J. Periodontal Res. 2008, 43, 549–555. [Google Scholar]
- Asano, M.; Yamaguchi, M.; Nakajima, R.; Fujita, S.; Utsunomiya, T.; Yamamoto, H.; Kasai, K. IL-8 and MCP-1 induced by excessive orthodontic force mediates odontoclastogenesis in periodontal tissues. Oral Dis. 2010, 17, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Curl, L.; Sampson, W. The presence of TNF-alpha and TNFR1 in aseptic root resorption. A preliminary study. Aust. Orthod. J. 2011, 27, 102–109. [Google Scholar]
- Diercke, K.; Kohl, A.; Lux, C.J.; Erber, R. IL-1beta and compressive forces lead to a significant induction of RANKL-expression in primary human cementoblasts. J. Orofac. Orthop. 2012, 73, 397–412. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Sringkarnboriboon, S.; Ono, T. Proinflammatory mediators related to orthodontically induced periapical root resorption in rat mandibular molars. Eur. J. Orthod. 2017, 39, 686–691. [Google Scholar] [CrossRef] [PubMed]
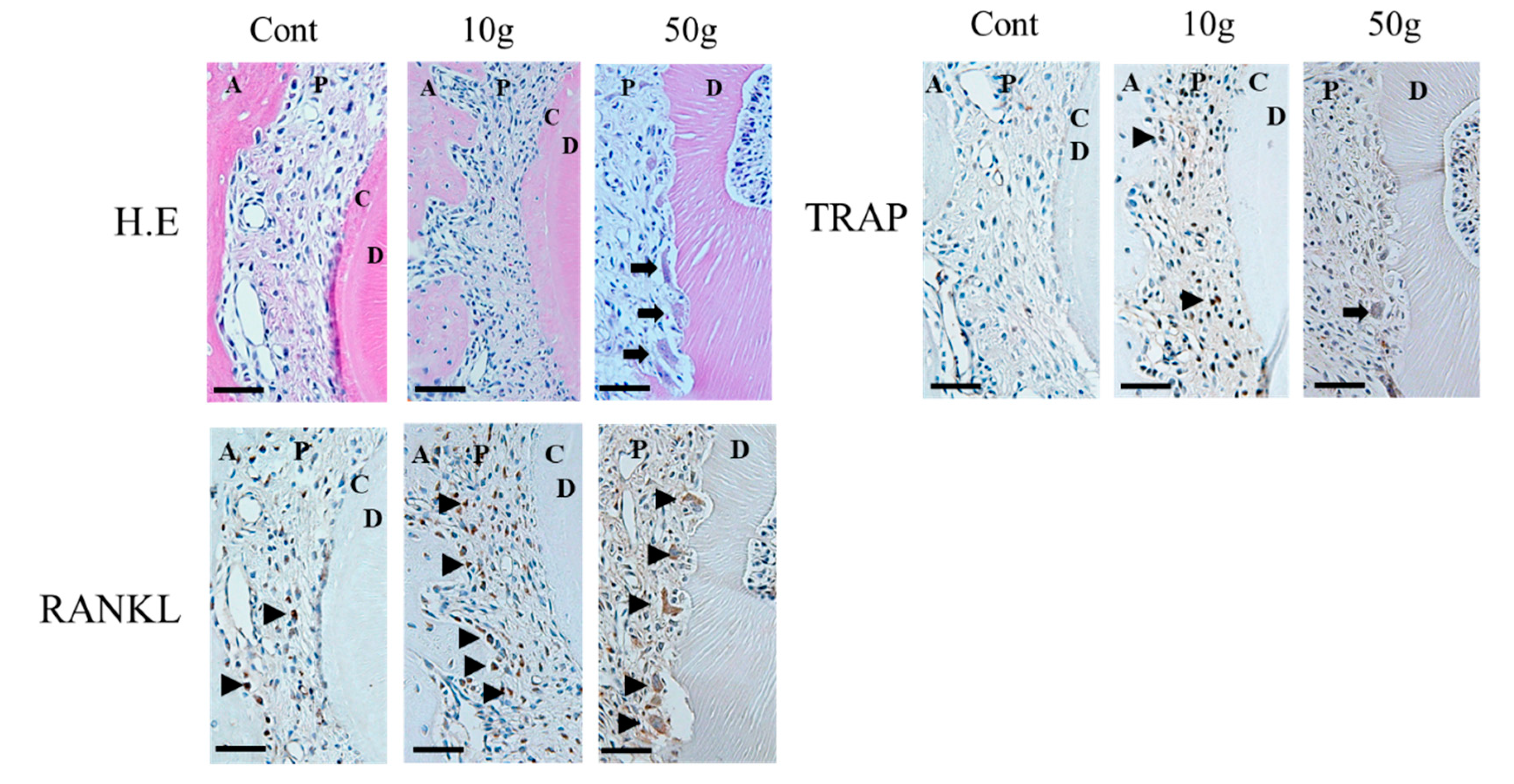
- Low, E.; Zoellner, H.; Kharbanda, O.P.; Darendeliler, M.A. Expression of mRNA for osteoprotegerin and receptor activator of nuclear factor kappa beta ligand (RANKL) during root resorption induced by the application of heavy orthodontic forces on rat molars. Am. J. Orthod. Dentofac. Orthop. 2005, 128, 497–503. [Google Scholar] [CrossRef]
- Yamada, K.; Yamaguchi, M.; Asano, M.; Fujita, S.; Kobayashi, R.; Kasai, K. Th17-cells in atopic dermatitis stimulate orthodontic root resorption. Oral Dis. 2012, 19, 683–693. [Google Scholar] [CrossRef]
- Hayashi, N.; Yamaguchi, M.; Nakajima, R.; Utsunomiya, T.; Yamamoto, H.; Kasai, K. T-helper 17 cells mediate the osteo/odontoclastogenesis induced by excessive orthodontic forces. Oral Dis. 2012, 18, 375–388. [Google Scholar] [CrossRef]
- Kikuta, J.; Yamaguchi, M.; Shimizu, M.; Yoshino, T.; Kasai, K. Notch Signaling Induces Root Resorption via RANKL and IL-6 from hPDL Cells. J. Dent. Res. 2014, 94, 140–147. [Google Scholar] [CrossRef]
- Tsukada, M.; Takagi, K.; Iwane, T.; Kikuta, J.; Shimizu, M.; Hikida, T.; Nakayama, E.; Kasai, K. TGF-β1 induces orthodontic root resorption through RANKL and IL-6 production from hPDL cells. Int. J. Oral-Medical Sci. 2021, in press. [Google Scholar]
- Minato, Y.; Yamaguchi, M.; Shimizu, M.; Kikuta, J.; Hikida, T.; Hikida, M.; Suemitsu, M.; Kuyama, K.; Kasai, K. Effect of caspases and RANKL induced by heavy force in orthodontic root resorption. Korean J. Orthod. 2018, 48, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Iwane, T.; Kikuta, J. Notch Signaling Response to Heavy Compression Force Induces Orthodontic Root Resorption via RANKL and IL-6 from Cementoblasts. Int. J. Oral-Med Sci. 2020, 18, 287–295. [Google Scholar] [CrossRef]
- Acar, A.; Canyürek, U.; Kocaaga., M.; Erverdi, N. Continuous vs. discontinuous force application and root resorption. Angle Orthod. 1999, 69, 159–163. [Google Scholar]
- Maltha, J.C.; van Leeuwen, E.J.; Dijkman, G.E.; Kuijpers-Jagtman, A.M. Incidence and severity of root resorption in orthodontically moved premolars in dogs. Orthod. Craniofacial Res. 2004, 7, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Hikida, T.; Yamaguchi, M.; Shimizu, M.; Kikuta, J.; Yoshino, T.; Kasai, K. Comparisons of orthodontic root resorption under heavy and jiggling reciprocating forces during experimental tooth movement in a rat model. Korean J. Orthod. 2016, 46, 228–241. [Google Scholar] [CrossRef]
- Matsuda, Y.; Motokawa, M.; Kaku, M.; Sumi, H.; Tanne, K.; Tanimoto, K. RANKL and OPG expression: Jiggling force affects root resorption in rats. Angle Orthod. 2016, 87, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Ueda, M.; Hikida, T.; Shimizu, M.; Kikuta, J.; Takagi, K.; Tsukada, M.; Yamaguchi, M. Involvement of interleukins-17 and -34 in exacerbated orthodontic root resorption by jiggling force during rat experimental tooth movement. J. World Fed. Orthod. 2020, 9, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Kau, C.H.; Kantarci, A.; Shaughnessy, T.; Vachiramon, A.; Santiwong, P.; De La Fuente, A.; Skrenes, D.; Ma, D.; Brawn, P. Photobiomodulation accelerates orthodontic alignment in the early phase of treatment. Prog. Orthod. 2013, 14, 30. [Google Scholar] [CrossRef] [PubMed]
- McGorray, S.P.; Dolce, C.; Kramer, S.; Stewart, D.; Wheeler, T.T. A randomized, placebo-controlled clinical trial on the effects of recombinant human relaxin on tooth movement and short-term stability. Am. J. Orthod. Dentofac. Orthop. 2012, 141, 196–203. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Hayashi, M.; Fujita, S.; Yoshida, T.; Utsunomiya, T.; Yamamoto, H.; Kasai, K. Low-energy laser irradiation facilitates the velocity of tooth movement and the expressions of matrix metalloproteinase-9, cathepsin K, and alpha(v) beta(3) integrin in rats. Eur. J. Orthod. 2010, 32, 131–139. [Google Scholar] [CrossRef]
- Nimeri, G.; Kau, C.H.; Abou-Kheir, N.S.; Corona, R. Acceleration of tooth movement during orthodontic treatment: A frontier in orthodontics. Prog. Orthod. 2013, 14, 42. [Google Scholar] [CrossRef]
- Frost, H.M. The regional acceleratory phenomenon: A review. Henry Ford Hosp. Med. J. 1983, 31, 3–9. [Google Scholar]
- Shih, M.; Norrdin, R. Regional acceleration of remodeling during healing of bone defects in beagles of various ages. Bone 1985, 6, 377–379. [Google Scholar] [CrossRef]
- Yaffe, A.; Fine, N.; Binderman, I. Regional Accelerated Phenomenon in the Mandible Following Mucoperiosteal Flap Surgery. J. Periodontol. 1994, 65, 79–83. [Google Scholar] [CrossRef]
- Lee, W.; Karapetyan, G.; Moats, R.; Yamashita, D.-D.; Moon, H.-B.; Ferguson, D.; Yen, S. Corticotomy-/Osteotomy-assisted Tooth Movement microCTs Differ. J. Dent. Res. 2008, 87, 861–867. [Google Scholar] [CrossRef]
- Frost, H.M. The biology of fracture healing. An overview for clinicians. Part II. Clin. Orthop. Relat. Res. 1989, 248, 283–309. [Google Scholar]
- Wang, L.; Lee, W.; Lei, D.-L.; Liu, Y.-P.; Yamashita, D.-D.; Yen, S.L.-K. Tisssue responses in corticotomy- and osteotomy-assisted tooth movements in rats: Histology and immunostaining. Am. J. Orthod. Dentofac. Orthop. 2009, 136, 770.e1–770.e11. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, K.; Shibata, Y.; Fukuhara, T. The Effect of Prostaglandins on Experimental Tooth Movement in Monkeys (Macaca fuscata). J. Dent. Res. 1982, 61, 1444–1446. [Google Scholar] [CrossRef]
- Iino, S.; Sakoda, S.; Miyawaki, S. An Adult Bimaxillary Protrusion Treated with Corticotomy-Facilitated Orthodontics and Titanium Miniplates. Angle Orthod. 2006, 76, 1074–1082. [Google Scholar] [CrossRef]
- Zhou, Y.; He, X.; Zhang, D. Study of bone remodeling in corticotomy-assisted orthodontic tooth movement in rats. J. Cell. Biochem. 2019, 120, 15952–15962. [Google Scholar] [CrossRef]
- Zou, M.; Li, C.; Zheng, Z. Remote Corticotomy Accelerates Orthodontic Tooth Movement in a Rat Model. BioMed Res. Int. 2019, 2019, 4934128. [Google Scholar] [CrossRef]
- Sugimori, T.; Yamaguchi, M.; Shimizu, M.; Kikuta, J.; Hikida, T.; Hikida, M.; Murakami, Y.; Suemitsu, M.; Kuyama, K.; Kasai, K. Micro-osteoperforations accelerate orthodontic tooth movement by stimulating periodontal ligament cell cycles. Am. J. Orthod. Dentofac. Orthop. 2018, 154, 788–796. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Sugimori, T.; Imamura, R.; Kasai, K. Gene expression profiling of rat periodontal ligament on stimulating with micro-osteoperforations (Japanese). J. Jpn. Assoc. Adult Orthod. 2016, 23, 2–5. [Google Scholar]


| Mechanical Stress | Cell | Inflammatory Mediator | Ref. |
|---|---|---|---|
| Compression force | Human PDL cells | IL-1, PGE | [60] |
| Compression force | Human PDL cells | IL-6 | [94] |
| Compression force | MC3T3-E1 cells | IL-17 | [63] |
| Compression force | Human PDL cells | RANKL/OPG | [86] |
| Compression force | Human PDL cells | RANKL/OPG | [87] |
| Type of TM | Animal | Inflammatory Mediator | Ref. |
|---|---|---|---|
| Canin Tip | Cat | IL-1, PGE | [60] |
| Molar Tip | Rat | TNF-α, IL-1β | [61] |
| Molar Tip | Rat | TNF-α, IL-1α | [64] |
| Rapid maxillary expansion | Human | TNF-α, RANKL | [65] |
| Molar Tip | Rat | RANKL/OPG | [89] |
| Human periapical granulomas | Human | RANKL/OPG | [90] |
| Tooth | Collection Side | Infammatory Mediator | Ref. |
|---|---|---|---|
| Canine | Pressure | PGE2, IL-1β | [66] |
| Canine | Pressure | IL-1β, IL-6, TNF-α | [67] |
| Canine | Pressure | IL-6, TNF-α | [68] |
| First premolar | Tension and pressure | IL-17 | [69] |
| Canine | Pressure | RANKL/OPG | [87] |
| Gene Symbol | Change (Fold) |
|---|---|
| CDC23 | 32.2 |
| CDC7 | 12.1 |
| MCM5 | 52.3 |
| MCM7 | 19.0 |
| Measurement | Normal (Japanese Woman) | Pretreatment (29 y 19 m) | Posttreatment (31y 2m) |
|---|---|---|---|
| SNA (° ) | 81.3 ± 2.7 | 80.0 | 80.0 |
| SNB (° ) | 78.6 ± 2.7 | 79.5 | 79.0 |
| ANB (° ) | 2.6 ± 1.1 | 0.5 | 1.0 |
| FMA (° ) | 26.3 ± 4.1 | 24.0 | 24.0 |
| FMIA (° ) | 56.9 ± 6.4 | 52.0 | 61.0 |
| IMPA (° ) | 96.8 ± 6.4 | 104.0 | 95.0 |
| U1-FH (° ) | 112.1 ± 4.2 | 126.5 | 115.5 |
| U1-L1 (° ) | 123.5 ± 5.5 | 105.0 | 125.0 |
| U1-A-Pog (mm) | 6.2 ± 1.5 | 11.2 | 4.6 |
| L1-A-Pog (mm) | 3.0 ± 1.5 | 8.1 | 2.3 |
| Gonial angle (° ) | 118.8 ± 6.1 | 117.5 | 117.5 |
| E-line: Upper (mm) | 1.4 ± 2.0 | −0.5 | −2.0 |
| E-line: Lower (mm) | 1.4 ± 2.0 | +0.3 | −0.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamaguchi, M.; Fukasawa, S. Is Inflammation a Friend or Foe for Orthodontic Treatment?: Inflammation in Orthodontically Induced Inflammatory Root Resorption and Accelerating Tooth Movement. Int. J. Mol. Sci. 2021, 22, 2388. https://doi.org/10.3390/ijms22052388
Yamaguchi M, Fukasawa S. Is Inflammation a Friend or Foe for Orthodontic Treatment?: Inflammation in Orthodontically Induced Inflammatory Root Resorption and Accelerating Tooth Movement. International Journal of Molecular Sciences. 2021; 22(5):2388. https://doi.org/10.3390/ijms22052388
Chicago/Turabian StyleYamaguchi, Masaru, and Shinichi Fukasawa. 2021. "Is Inflammation a Friend or Foe for Orthodontic Treatment?: Inflammation in Orthodontically Induced Inflammatory Root Resorption and Accelerating Tooth Movement" International Journal of Molecular Sciences 22, no. 5: 2388. https://doi.org/10.3390/ijms22052388
APA StyleYamaguchi, M., & Fukasawa, S. (2021). Is Inflammation a Friend or Foe for Orthodontic Treatment?: Inflammation in Orthodontically Induced Inflammatory Root Resorption and Accelerating Tooth Movement. International Journal of Molecular Sciences, 22(5), 2388. https://doi.org/10.3390/ijms22052388






