Metabotropic Glutamate Receptor Blockade Reduces Preservation Damage in Livers from Donors after Cardiac Death
Abstract
1. Introduction
2. Results
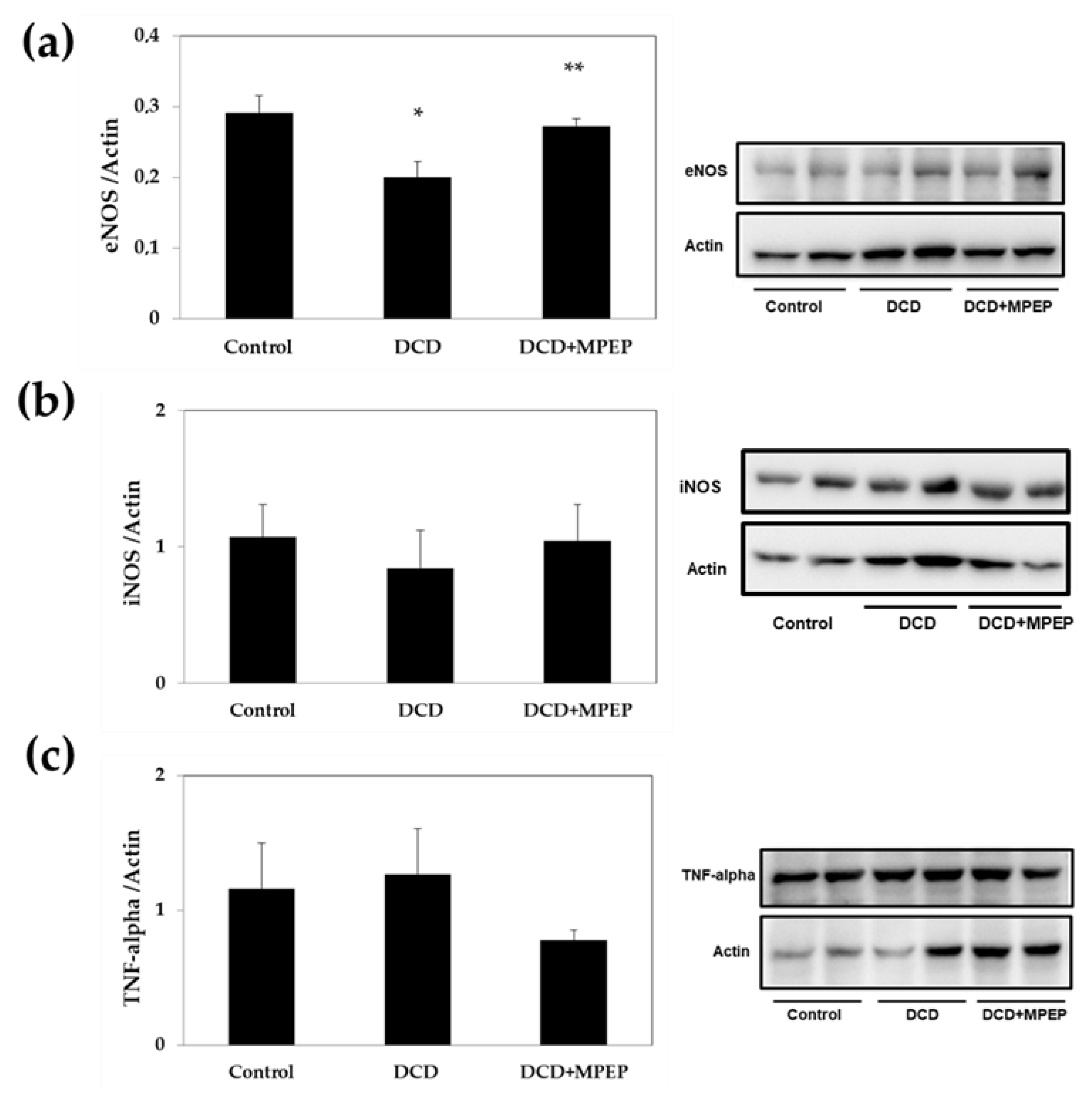
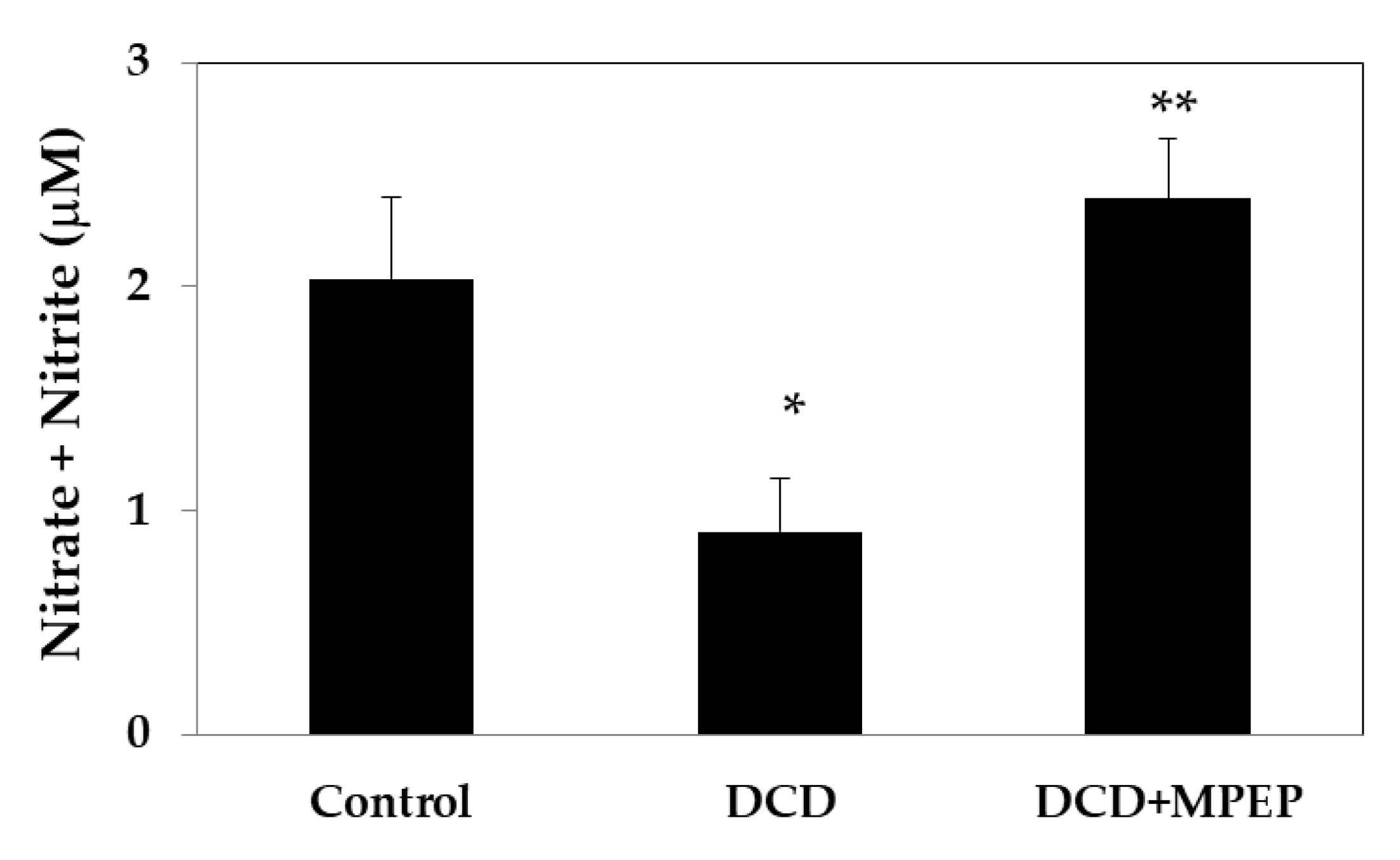
2.1. Hepatic eNOS, iNOS, and TNF-Alpha Content after Cold-Storage Preservation
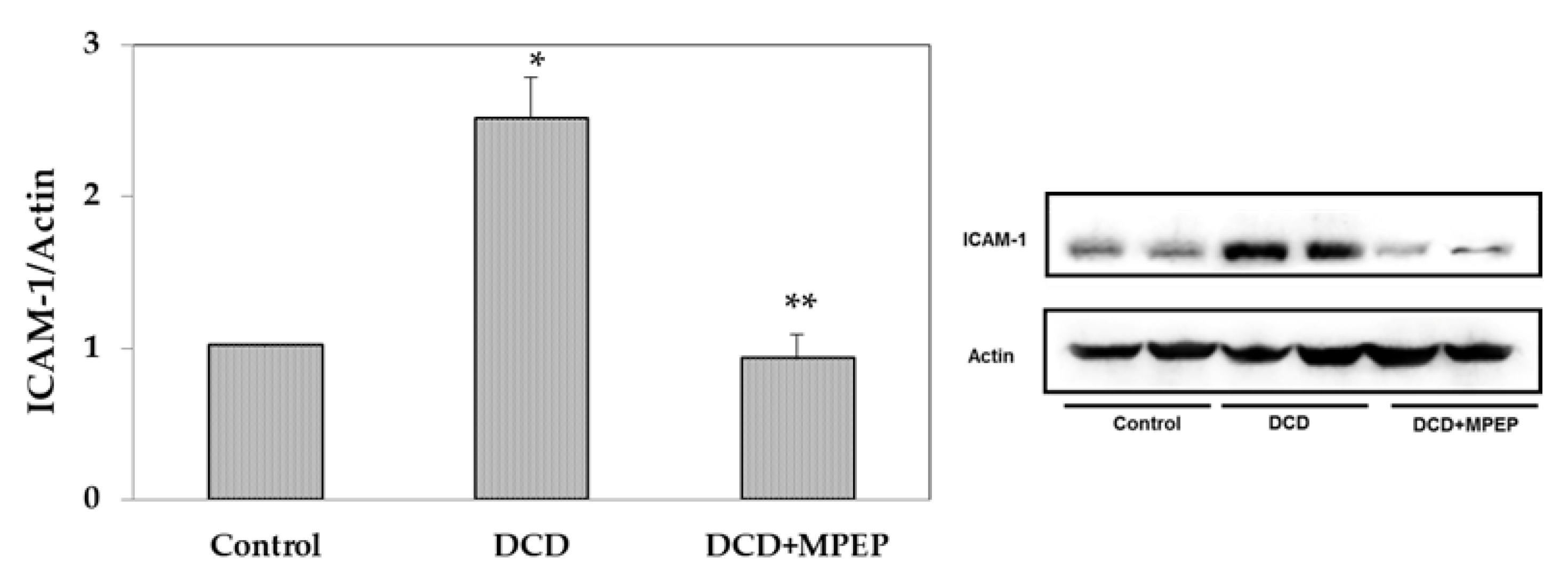
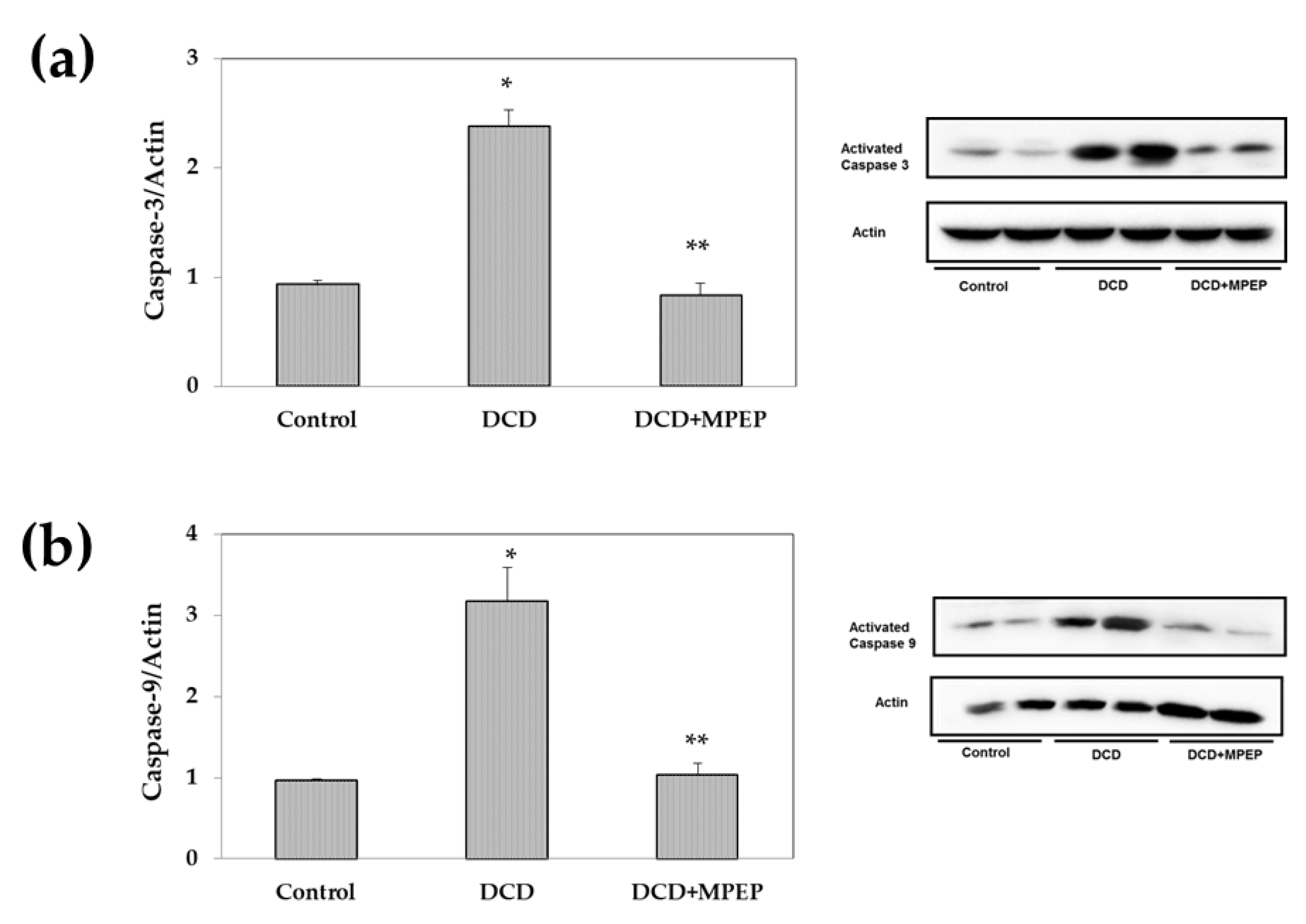
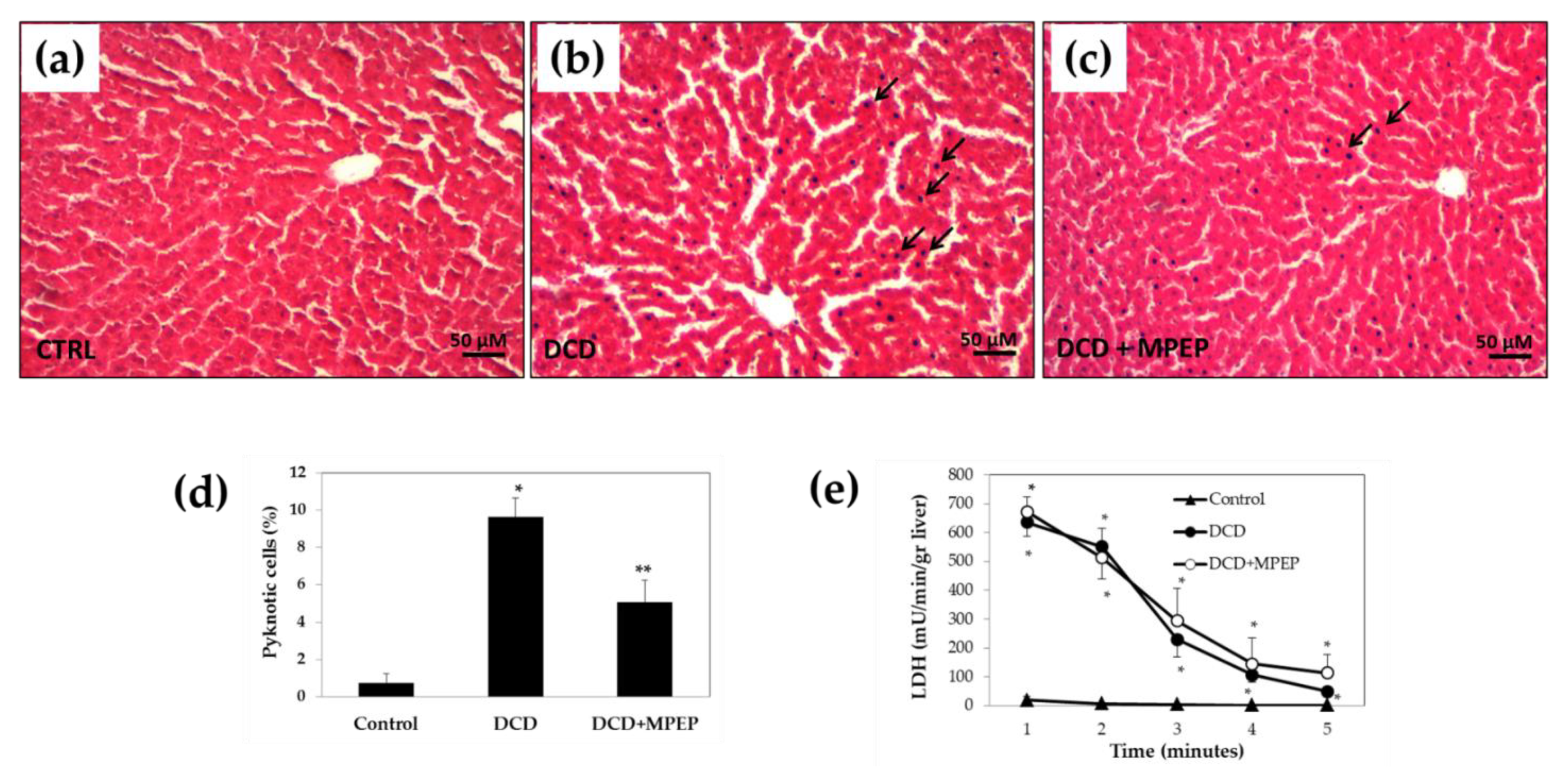
2.2. Effects of MPEP on ICAM-1 and Apoptosis
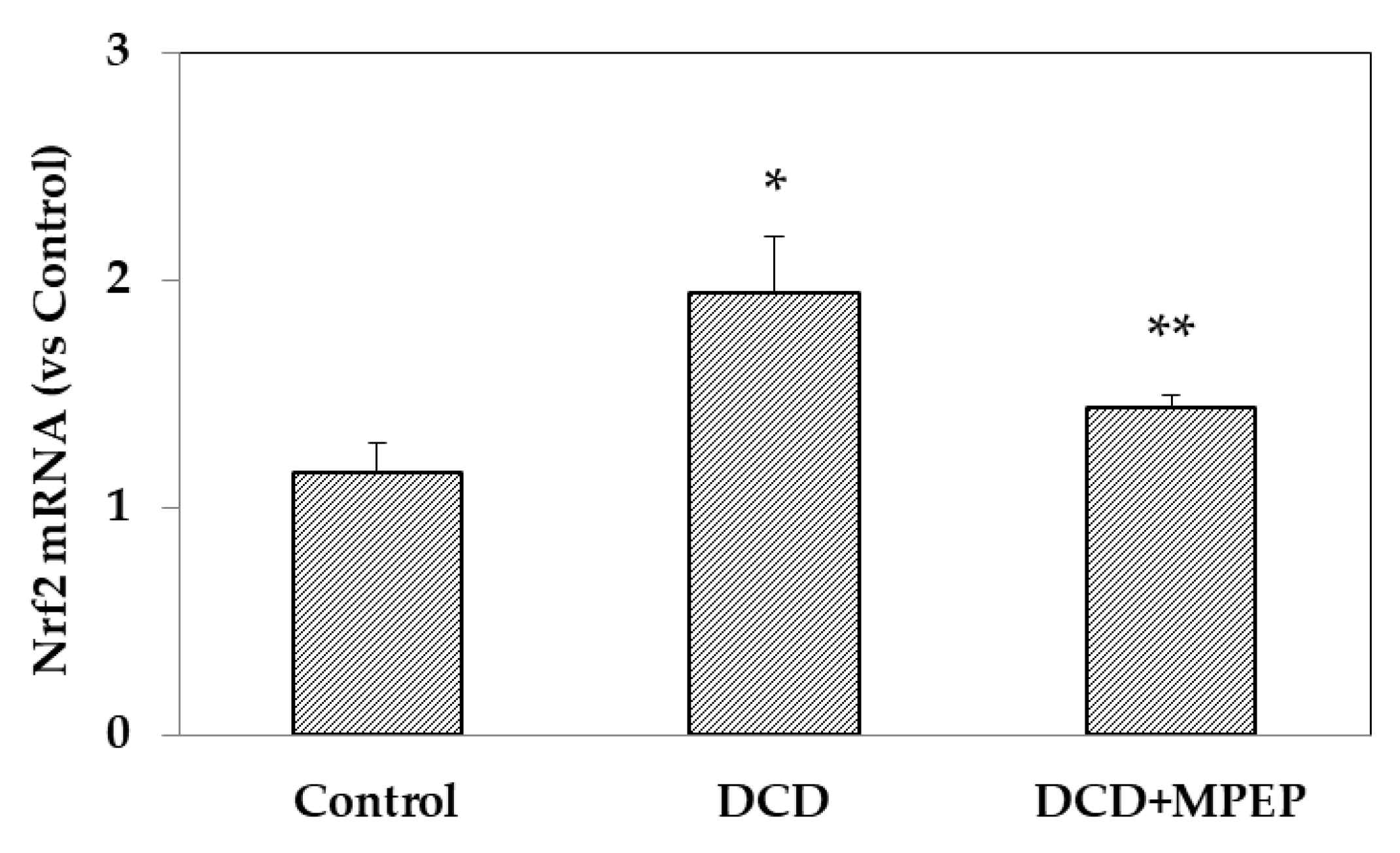
2.3. Effects of MPEP on Nrf2 mRNA
2.4. Hepatocellular Injury of Livers from DCDs
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Animals and Surgery
4.3. Western Blot Assay
4.4. RT-PCR
4.5. Histological Analysis
4.6. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ferrigno, A.; Berardo, C.; Di Pasqua, L.G.; Siciliano, V.; Richelmi, P.; Vairetti, M. Localization and role of metabotropic glutamate receptors subtype 5 in the gastrointestinal tract. World J. Gastroenterol. 2017, 23, 4500–4507. [Google Scholar] [CrossRef]
- Storto, M.; de Grazia, U.; Knöpfel, T.; Canonico, P.L.; Copani, A.; Richelmi, P.; Nicoletti, F.; Vairetti, M. Selective blockade of mGlu5 metabotropic glutamate receptors protects rat hepatocytes against hypoxic damage. Hepatology 2000, 31, 649–655. [Google Scholar] [CrossRef]
- Storto, M.; Ngomba, R.T.; Battaglia, G.; Freitas, I.; Griffini, P.; Richelmi, P.; Nicoletti, F.; Vairetti, M.; Teke Ngomba, R.; Battaglia, G.; et al. Selective blockade of mGlu5 metabotropic glutamate receptors is protective against acetaminophen hepatotoxicity in mice. J. Hepatol. 2003, 38, 179–187. [Google Scholar] [CrossRef]
- Storto, M.; Battaglia, G.; Gradini, R.; Bruno, V.; Nicoletti, F.; Vairetti, M. Mouse hepatocytes lacking mGlu5 metabotropic glutamate receptors are less sensitive to hypoxic damage. Eur. J. Pharmacol. 2004, 497, 25–27. [Google Scholar] [CrossRef]
- Ferrigno, A.; Berardo, C.; Di Pasqua, L.G.; Siciliano, V.; Richelmi, P.; Nicoletti, F.; Vairetti, M. Selective blockade of the metabotropic glutamate receptor mGluR5 protects mouse livers in in vitro and ex vivo models of ischemia reperfusion injury. Int. J. Mol. Sci. 2018, 19, 314. [Google Scholar] [CrossRef] [PubMed]
- Bodzin, A.S.; Baker, T.B. Liver Transplantation Today: Where We Are Now and Where We Are Going. Liver Transplant. 2018, 24, 1470–1475. [Google Scholar] [CrossRef] [PubMed]
- Manyalich, M.; Nelson, H.; Delmonico, F.L. The need and opportunity for donation after circulatory death worldwide. Curr. Opin. Organ Transplant. 2018, 23, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Apinyachon, W.; Xia, W.; Hong, J.C.; Busuttil, R.W.; Steadman, R.H.; Xia, V.W. Perioperative complications in liver transplantation using donation after cardiac death grafts: A propensity-matched study. Liver Transplant. 2014, 20, 823–830. [Google Scholar] [CrossRef]
- De Carlis, R.; Buscemi, V.; Checchini, G.; Frassoni, S.; Bagnardi, V.; Pagnanelli, M.; Lauterio, A.; De Carlis, L. Liver transplantation from brain-dead donors on mechanical circulatory support: A systematic review of the literature. Transpl. Int. 2021, 34, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Kalisvaart, M.; Perera, M.T.P.R. Using Marginal Grafts for Liver Transplantation: The Balance of Risk. J. Investig. Surg. 2020, 33, 565–567. [Google Scholar] [CrossRef]
- Zaouali, M.A.; Abdennebi, H.B.; Padrissa-Altés, S.; Mahfoudh-Boussaid, A.; Roselló-Catafau, J. Pharmacological strategies against cold ischemia reperfusion injury. Expert Opin. Pharmacother. 2010, 11, 537–555. [Google Scholar] [CrossRef] [PubMed]
- Dutkowski, P.; Linecker, M.; Deoliveira, M.L.; Müllhaupt, B.; Clavien, P.A. Challenges to liver transplantation and strategies to improve outcomes. Gastroenterology 2015, 148, 307–323. [Google Scholar] [CrossRef] [PubMed]
- Abdennebi, H.B.; Zaoualí, M.A.; Alfany-Fernandez, I.; Tabka, D.; Roselló-Catafau, J. How to protect liver graft with nitric oxide. World J. Gastroenterol. 2011, 17, 2879. [Google Scholar] [CrossRef] [PubMed]
- Varadarajan, R.; Golden-Mason, L.; Young, L.; McLoughlin, P.; Nolan, N.; McEntee, G.; Traynor, O.; Geoghegan, J.; Hegarty, J.E.; O’Farrelly, C. Nitric oxide in early ischaemia reperfusion injury during human orthotopic liver transplantation. Transplantation 2004, 78, 250–256. [Google Scholar] [CrossRef]
- El-Wahsh, M.; Fuller, B.; Davidson, B.; Rolles, K. Hepatic cold hypoxia and oxidative stress: Implications for ICAM-1 expression and modulation by glutathione during experimental isolated liver preservation. Cryobiology 2003, 47, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Mosbah, I.B.; Zaouali, M.; Martel, C.; Bjaoui, M.; Abdennebi, H.B.; Hotter, G.; Brenner, C.; Roselló-Catafau, J. IGL-1 solution reduces endoplasmic reticulum stress and apoptosis in rat liver transplantation. Cell Death Dis. 2012, 3, e279. [Google Scholar] [CrossRef]
- Wang, H.B.; Wang, W.Q.; Wu, Q.J.; Hou, Y.J.; Li, H.X.; Yang, H.J.; Yang, M.F.; Sun, B.L.; Zhang, Z.Y. Negative Allosteric Modulator of mGluR1 Improves Long-Term Neurologic Deficits after Experimental Subarachnoid Hemorrhage. ACS Chem. Neurosci. 2020, 11, 2869–2880. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.W.; Zhao, L.; Zhou, M.; Wang, H.; Yang, N.; Dai, S.S. Transplantation with mGluR5 deficiency bone marrow displays antidepressant-like effect in C57BL/6J mice. Brain. Behav. Immun. 2019, 79, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Bejaoui, M.; Pantazi, E.; Folch-Puy, E.; Baptista, P.M.; García-Gil, A.; Adam, R.; Roselló-Catafau, J. Emerging concepts in liver graft preservation. World J. Gastroenterol. 2015, 21, 396–407. [Google Scholar] [CrossRef]
- Zhang, B.; Liu, Q.H.; Zhou, C.J.; Hu, M.Z.; Qian, H.X. Protective effect of eNOS overexpression against ischemia/reperfusion injury in small-for-size liver transplantation. Exp. Ther. Med. 2016, 12, 3181–3188. [Google Scholar] [CrossRef] [PubMed]
- Russo, L.; Gracia-Sancho, J.; García-Calderó, H.; Marrone, G.; García-Pagán, J.C.; García-Cardeña, G.; Bosch, J. Addition of simvastatin to cold storage solution prevents endothelial dysfunction in explanted rat livers. Hepatology 2012, 55, 921–930. [Google Scholar] [CrossRef] [PubMed]
- Lopez, A.; Panisello-Rosello, A.; Castro-Benitez, C.; Adam, R. Glycocalyx preservation and NO production in fatty livers—The protective role of high molecular polyethylene glycol in cold ischemia injury. Int. J. Mol. Sci. 2018, 19, 2375. [Google Scholar] [CrossRef]
- Zaoualí, M.A.; Reiter, R.J.; Padrissa-Altés, S.; Boncompagni, E.; García, J.J.; Ben Abnennebi, H.; Freitas, I.; García-Gil, F.A.; Rosello-Catafau, J. Melatonin protects steatotic and nonsteatotic liver grafts against cold ischemia and reperfusion injury. J. Pineal Res. 2011, 50, 213–221. [Google Scholar] [CrossRef]
- Duranski, M.R.; Elrod, J.W.; Calvert, J.W.; Bryan, N.S.; Feelisch, M.; Lefer, D.J. Genetic overexpression of eNOS attenuates hepatic ischemia-reperfusion injury. Am. J. Physiol. Hear. Circ. Physiol. 2006, 291, H2980–H2986. [Google Scholar] [CrossRef] [PubMed]
- Selzner, N.; Rudiger, H.; Graf, R.; Clavien, P.A. Protective strategies against ischemic injury of the liver. Gastroenterology 2003, 125, 917–936. [Google Scholar] [CrossRef]
- Wei, Y.; Chen, P.; de Bruyn, M.; Zhang, W.; Bremer, E.; Helfrich, W. Carbon monoxide-Releasing Molecule-2 (CORM-2) attenuates acute hepatic ischemia reperfusion injury in rats. BMC Gastroenterol. 2010, 10, 42. [Google Scholar] [CrossRef]
- Lopez-Neblina, F.; Toledo, A.H.; Toledo-Pereyra, L.H. Molecular biology of apoptosis in ischemia and reperfusion. J. Investig. Surg. 2005, 18, 335–350. [Google Scholar] [CrossRef]
- Von Heesen, M.; Müller, S.; Keppler, U.; Strowitzki, M.J.; Scheuer, C.; Schilling, M.K.; Menger, M.D.; Moussavian, M.R. Preconditioning by Cilostazol protects against cold hepatic ischemia-reperfusion injury. Ann. Transplant. 2015, 20, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Ho, E.; Karimi Galougahi, K.; Liu, C.C.; Bhindi, R.; Figtree, G.A. Biological markers of oxidative stress: Applications to cardiovascular research and practice. Redox Biol. 2013, 1, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Xue, S.; He, W.; Zeng, X.; Tang, Z.; Feng, S.; Zhong, Z.; Xiong, Y.; Wang, Y.; Ye, Q. Hypothermic machine perfusion attenuates ischemia/reperfusion injury against rat livers donated after cardiac death by activating the Keap1/Nrf2-ARE signaling pathway. Mol. Med. Rep. 2018, 18, 815–826. [Google Scholar] [CrossRef] [PubMed]
- Giraud, S.; Kerforne, T.; Zely, J.; Ameteau, V.; Couturier, P.; Tauc, M.; Hauet, T. The inhibition of eIF5A hypusination by GC7, a preconditioning protocol to prevent brain death-induced renal injuries in a preclinical porcine kidney transplantation model. Am. J. Transplant. 2020, 20, 3326–3340. [Google Scholar] [CrossRef] [PubMed]
- Peralta, C.; Jiménez-Castro, M.B.; Gracia-Sancho, J. Hepatic ischemia and reperfusion injury: Effects on the liver sinusoidal milieu. J. Hepatol. 2013, 59, 1094–1106. [Google Scholar] [CrossRef] [PubMed]
- Bessems, M.; Doorschodt, B.M.; van Marle, J.; Vreeling, H.; Meijer, A.J.; van Gulik, T.M. Improved machine perfusion preservation of the non-heart-beating donor rat liver using Polysol: A new machine perfusion preservation solution. Liver Transpl. 2005, 11, 1379–1388. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, B.; Manas, M.D.; Jaques, B.C.; Talbot, D. Ischemic Cholangiopathy After Liver Transplantation From Controlled Non–Heart-Beating Donors–A Single-Center Experience. Transplant. Proc. 2007, 39, 2793–2795. [Google Scholar] [CrossRef] [PubMed]
- Panisello-Roselló, A.; Alva, N.; Flores, M.; Lopez, A.; Benítez, C.C.; Folch-Puy, E.; Rolo, A.; Palmeira, C.; Adam, R.; Carbonell, T.; et al. Aldehyde dehydrogenase 2 (ALDH2) in rat fatty liver cold ischemia injury. Int. J. Mol. Sci. 2018, 19, 2479. [Google Scholar] [CrossRef] [PubMed]
- Ferrigno, A.; Rizzo, V.; Boncompagni, E.; Bianchi, A.; Gringeri, E.; Neri, D.; Richelmi, P.; Freitas, I.; Cillo, U.; Vairetti, M. Machine perfusion at 20??C reduces preservation damage to livers from non-heart beating donors. Cryobiology 2011, 62, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Chomczynski, P.; Mackey, K. Substitution of chloroform by bromo-chloropropane in the single-step method of RNA isolation. Anal. Biochem. 1995, 225, 163–164. [Google Scholar] [CrossRef] [PubMed]
- Berardo, C.; Siciliano, V.; Di Pasqua, L.G.; Richelmi, P.; Vairetti, M.; Ferrigno, A. Comparison between lipofectamine RNAiMAX and genmute transfection agents in two cellular models of human hepatoma. Eur. J. Histochem. 2019, 63, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. Primer Sequence Disclosure: A Clarification of the MIQE Guidelines. Clin. Chem. 2011, 57, 919–921. [Google Scholar] [CrossRef]
| Gene | Amplicon Sequence |
|---|---|
| Nrf2 (Nfe2l2) | TACCCCAAGATCTATGTCTTCCTCCAAAGGATGTCAATCAAATCCAT GTCCTGCTGGGACTGTAGTCCTGGCGGTGGAATTCCAAGTCCATCAT GCTGAGGGCGGACGCTGCGCTAGGGC |
| GAPDH | TGATGGCAACAATGTCCACTTTGTCACAAGAGAAGGCAGCCCTGGA ACCAGGCGTCCGATACGGCCAAATCCGTTCACACCGACTTCACCATCTTGTCTATGAGACGAGGCTGGCACTGCACAAGAATGCGGCTGTCTCTA |
| USP28 | CCGAGACGGGTCTGAAGCAGGGCTTATTAAGGCATTTCATGAAGAGTACTCCAGGCTCTATCAGCTTGCCAAAGAGACACCCACCTCTCACAGTGATCC |
| HPRT-1 | TTCATGCAAAAGCTTTACTAAGTAGATGGCCACAGGACTAGAACGT CTGCTAGTTCTTTACTGGCCACATCAACAGGACTCTTGTAGATTCAAC TTGCCGCTGTCTTTT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Pasqua, L.G.; Berardo, C.; Cagna, M.; Verta, R.; Collotta, D.; Nicoletti, F.; Ferrigno, A.; Collino, M.; Vairetti, M. Metabotropic Glutamate Receptor Blockade Reduces Preservation Damage in Livers from Donors after Cardiac Death. Int. J. Mol. Sci. 2021, 22, 2234. https://doi.org/10.3390/ijms22052234
Di Pasqua LG, Berardo C, Cagna M, Verta R, Collotta D, Nicoletti F, Ferrigno A, Collino M, Vairetti M. Metabotropic Glutamate Receptor Blockade Reduces Preservation Damage in Livers from Donors after Cardiac Death. International Journal of Molecular Sciences. 2021; 22(5):2234. https://doi.org/10.3390/ijms22052234
Chicago/Turabian StyleDi Pasqua, Laura Giuseppina, Clarissa Berardo, Marta Cagna, Roberta Verta, Debora Collotta, Ferdinando Nicoletti, Andrea Ferrigno, Massimo Collino, and Mariapia Vairetti. 2021. "Metabotropic Glutamate Receptor Blockade Reduces Preservation Damage in Livers from Donors after Cardiac Death" International Journal of Molecular Sciences 22, no. 5: 2234. https://doi.org/10.3390/ijms22052234
APA StyleDi Pasqua, L. G., Berardo, C., Cagna, M., Verta, R., Collotta, D., Nicoletti, F., Ferrigno, A., Collino, M., & Vairetti, M. (2021). Metabotropic Glutamate Receptor Blockade Reduces Preservation Damage in Livers from Donors after Cardiac Death. International Journal of Molecular Sciences, 22(5), 2234. https://doi.org/10.3390/ijms22052234










