Understanding the Effects of Anesthesia on Cortical Electrophysiological Recordings: A Scoping Review
Abstract
1. Introduction
2. Understanding the Mechanism of General Anesthesia: How Far Are We?
3. Effects of Different Anesthetics on Cortical Electrophysiological Recordings
3.1. Injectable Anesthetics
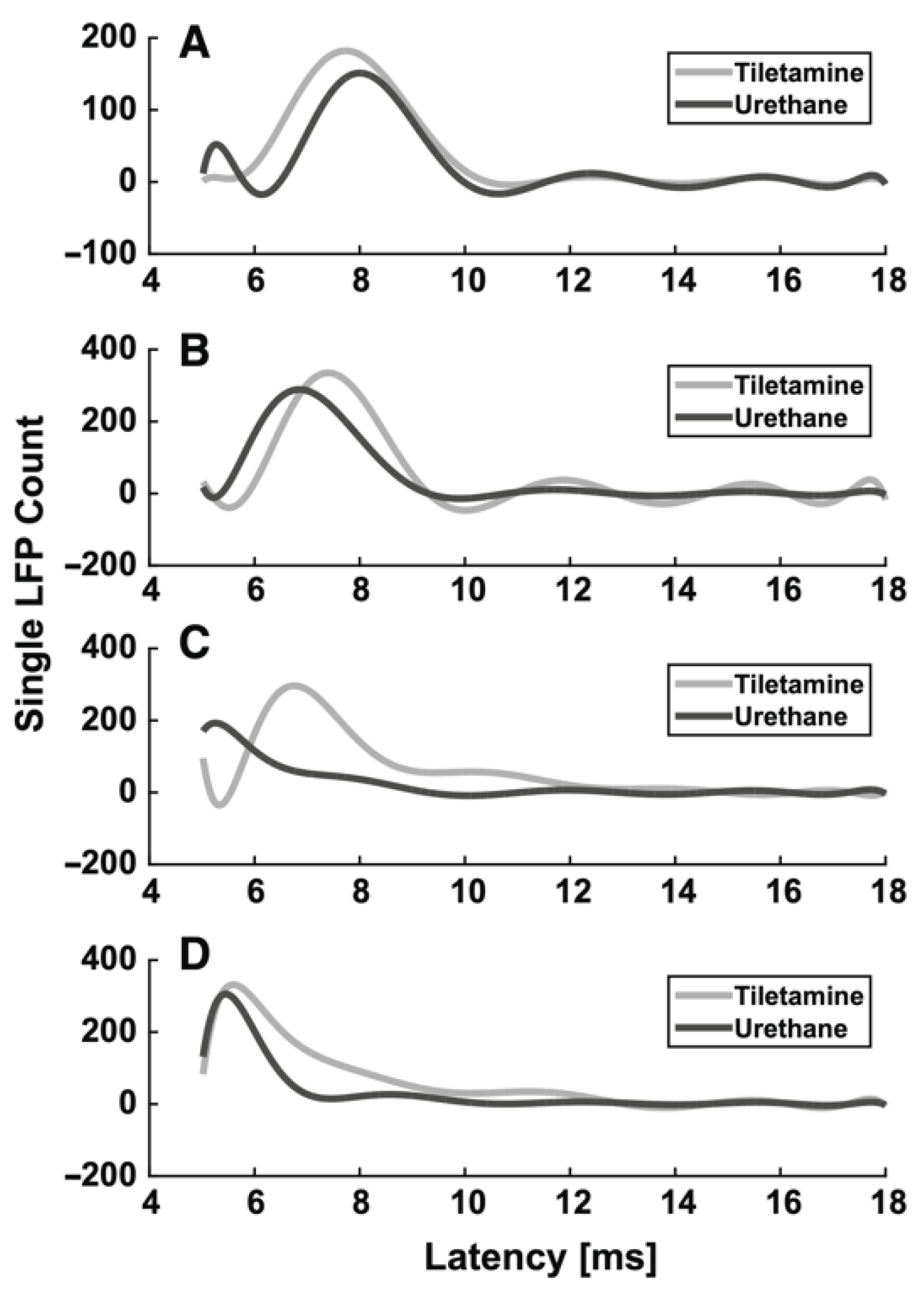
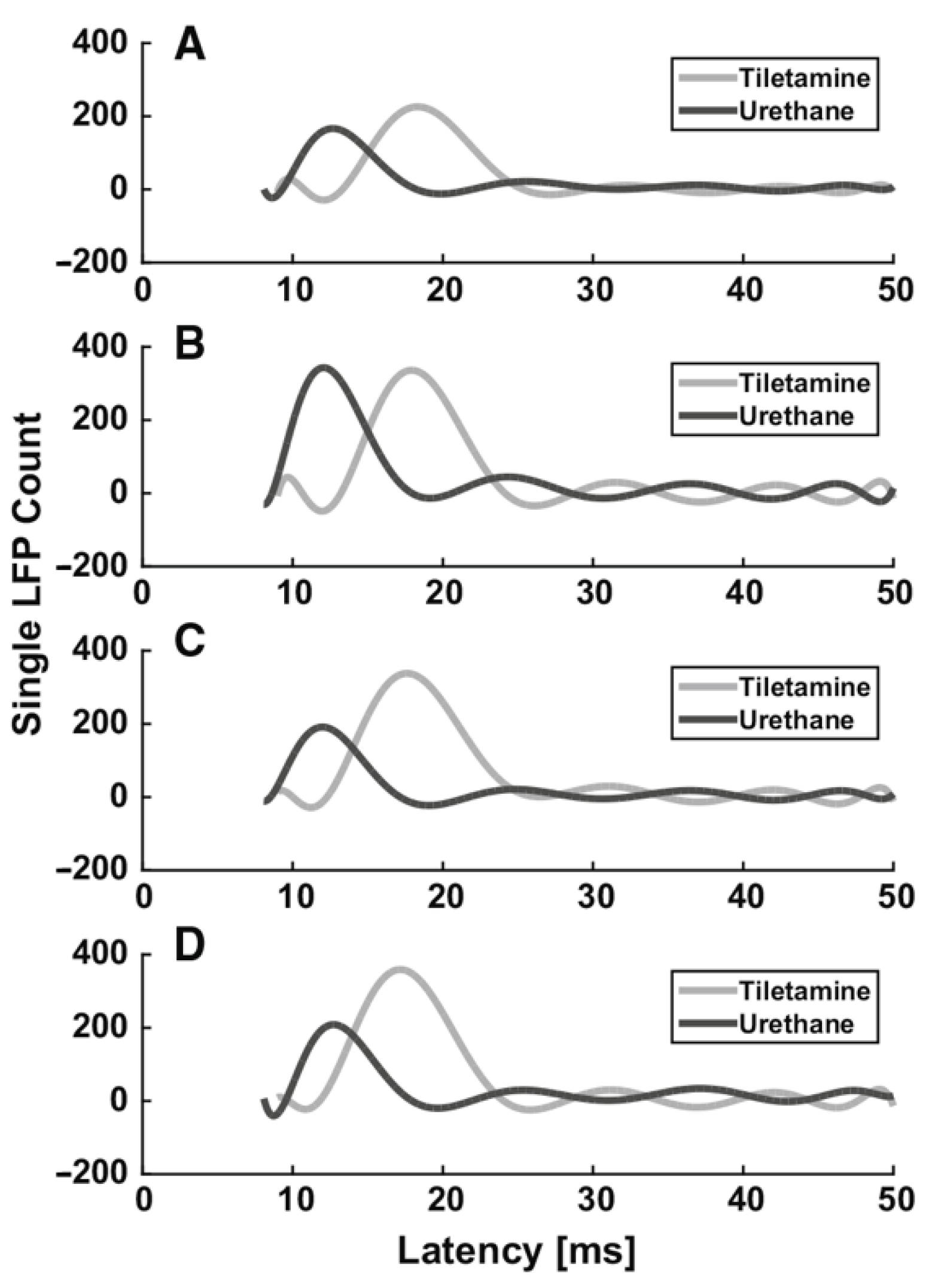
3.1.1. Urethane
3.1.2. Ketamine
3.1.3. Ketamine-Xylazine/Medetomidine
3.1.4. Dexmedetomidine
3.1.5. Medetomidine-Midazolam-Butorphanol (MMB)
3.1.6. Sodium Pentobarbital
3.1.7. Propofol
3.2. Volatile Anesthetics
3.2.1. Isoflurane
3.2.2. Sevoflurane
4. Conclusions and Future Perspectives
| Name | Mechanism of Action | Start and Duration of Action | Use in Rodents | Use in Humans | Side Effects | Major Effects on Rodents Cortical Recordings | References |
|---|---|---|---|---|---|---|---|
| Injectable anesthetics | |||||||
| Urethane | moderate GABAA, glycine and nACh receptors excitation, NR1A/NR2A NMDA and GluR1/GluR2 AMPA receptors inhibition | Start 1–5 min Duration hours | i.p. Induction: 1.5–2 g/kg body mass; additional doses: 10–20% of induction. i.v. 1.75 g/kg body mass | No | Mucus in the respiratory tract | Evoked activity: ↑ latency, ↓ SEP and VSDI amplitude (dose-dependent effect) Spontaneous activity: ↓ burst and firing rates, ↑ burst duration, ↑ number of spikes in a burst (dose-dependent effect) | [49] |
| Ketamine | NMDA channel antagonist; AMPA receptors upregulator and µ-type opioid receptors agonist; anesthesia, analgesia, amnesia, sedation | Start 1–5 min. Duration 15 min | Combined with xylazine/medetomidine. i.p. Induction: 40–100 mg/kg body mass; additional doses: 10–30% of induction | Yes | Laryngospasm, hypertension, tachycardia, hypersalivation, vomiting, psychiatric symptoms | Alone Evoked activity: ↑ LFP amplitude, ↑ long lasting LFP (compared to awake) With Xylazine: Evoked activity: LIV onset; Spontaneous activity: LV onset; ↓ frequency, ↑ amplitude (dose-dependent effect) | [114] |
| Xylazine | alpha2 adrenoceptor agonist; sedation, muscle relaxation, analgesia | Start 1–5 min. Duration 15 min | Combined with ketamine. i.p. Induction: 5–15 mg/kg body mass; additional doses: 30% of induction | No | In humans, it could cause central nervous system depression, respiratory depression, bradycardia, | see Ketamine | [115] |
| Medetomidine/Dexmedetomidine | alpha2 adrenoceptor agonist; sedation, anxiolysis, analgesia | Start: Combined with midazolam/butorphanol: Start: i.p. 25 min, s.c. 10 min | Combined with ketamine. i.p. Induction 0.5 mg/kg body mass Combined with midazolam/butorphanol. i.p. or s.c. 0.15–0.3 mg/kg body mass; additional doses (if needed for i.p.): 20% of induction | Yes 1 | Alone: Evoked activity: ↑ latency, ↓ amplitude, ↓ firing rates (dose/route-dependent effect), weak ↓ power of HF thalamocortical activity (dose-dependent) With Midazolam/Butorphanol: Spontaneous activity: ↓ LV firing rate | [116] | |
| Midazolam | GABA receptor positive allosteric modulator; anterograde, anxiolytic, sedation, hypnosis amnesia | Start: i.p. 25 min, s.c. 10 min combined with medetomidine/butorphanol | Combined with medetomidine/butorphanol. i.p. or s.c. Induction: 2–4 mg/kg body mass; additional doses (if needed for i.p.): 20% of induction | Yes | Respiratory depression, hypotension, agitation | see Medetomidine | [117] |
| Butorphanol | opioid; analgesia | Start: i.p. 25 min, s.c. 10 min combined with medetomidine/midazolam Duration: 45–60 min combined with medetomidine/midazolam | Combined with medetomidine/midazolam. i.p. or s.c. Induction: 2.5–5 mg/kg body mass; additional doses (if needed for i.p.): 20% of induction | Yes 2 | see Medetomidine | [118,119] | |
| Sodium pentobarbital | GABAA receptor agonist | Duration 120 min | i.p. Induction: 50 mg/kg body mass; additional doses not required | Yes 3 | Evoked activity: ↓ LFP and SU amplitude | [120] | |
| Propofol | GABA positive allosteric modulator; sedation, hypnosis, amnesia | i.v. 450–700 μg/kg/min | Yes | Respiratory depression, apnea, hypotension, painful injection | Evoked activity: ↑ latency, ↓ amplitude Spontaneous activity: ↓ high frequency oscillations | [121] | |
| Volatile anesthetics | |||||||
| Isoflurane | tandem pore K channels, Nav channels, NMDA, glycine and GABAA receptors agonist | Start 10 min, Duration: 5 min | Inhalation. Induction: 2–5%; maintenance: 1.5–2.5% | Yes | Respiratory depression, irregular heartbeat, low blood pressure | Evoked activity: ↑ latency, ↓ amplitude, ↓ firing rate (>variability of neuronal responses) (dose-dependent effect). Spontaneous activity: ↓ firing rate, ↓ delta-gamma bands (dose-dependent effect) | [122] |
| Sevoflurane | GABAA. glycine potentiation, NMDA and nACh inhibition | Inhalation. Induction: 1.6–2.8% | Yes | Agitation, bradycardia, hypotension, cough, vomiting | Spontaneous activity: ↓ beta-gamma coherence, ↑ slow delta power, ↑ delta coherence (dose-dependent effect) | [123] | |
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Brown, E.N.; Lydic, R.; Schiff, N.D. General anesthesia, sleep, and coma. N. Engl. J. Med. 2010, 363, 2638–2650. [Google Scholar] [CrossRef] [PubMed]
- Kurdi, M.S.; Ramaswamy, A.H. Anesthetizing animals: Similar to humans yet, peculiar? Anesth. Essays Res. 2015, 9, 298. [Google Scholar] [CrossRef] [PubMed]
- Mashour, G.A.; Alkire, M.T. Evolution of consciousness: Phylogeny, ontogeny, and emergence from general anesthesia. Proc. Natl. Acad. Sci. USA 2013, 110, 10357–10364. [Google Scholar] [CrossRef] [PubMed]
- Campoy, L.; Read, M. Small Animal Regional Anesthesia and Analgesia; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Gargiulo, S.; Greco, A.; Gramanzini, M.; Esposito, S.; Affuso, A.; Brunetti, A.; Vesce, G. Mice anesthesia, analgesia, and care, Part I: Anesthetic considerations in preclinical research. ILAR J. 2012, 53, E55–E69. [Google Scholar] [CrossRef]
- Chou, R.; Gordon, D.B.; de Leon-Casasola, O.A.; Rosenberg, J.M.; Bickler, S.; Brennan, T.; Carter, T.; Cassidy, C.L.; Chittenden, E.H.; Degenhardt, E. Management of postoperative pain: A clinical practice guideline from the American pain society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ committee on regional anesthesia, executive committee, and administrative council. J. Pain 2016, 17, 131–157. [Google Scholar]
- Dawkins, M. Animal Suffering: The Science of Animal Welfare; Springer: Dordrecht, The Netherlands, 2012. [Google Scholar]
- Flecknell, P.A.; Thomas, A.A. Comparative anesthesia and analgesia of laboratory animals. Vet. Anesth. Analg. 2015, 754. [Google Scholar] [CrossRef]
- Huang, Z.; Wang, Z.; Zhang, J.; Dai, R.; Wu, J.; Li, Y.; Liang, W.; Mao, Y.; Yang, Z.; Holland, G. Altered temporal variance and neural synchronization of spontaneous brain activity in anesthesia. Hum. Brain Mapp. 2014, 35, 5368–5378. [Google Scholar] [CrossRef]
- Hohlbaum, K.; Bert, B.; Dietze, S.; Palme, R.; Fink, H.; Thöne-Reineke, C. Impact of repeated anesthesia with ketamine and xylazine on the well-being of C57BL/6JRj mice. PLoS ONE 2018, 13, e0203559. [Google Scholar] [CrossRef]
- Garcia-Pereira, F. Epidural anesthesia and analgesia in small animal practice: An update. Vet. J. 2018, 242, 24–32. [Google Scholar] [CrossRef]
- Clar, D.T.; Richards, J.R. Anesthetic Gases. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Wolfe, R.C. Inhaled anesthetic agents. J. Perianesthesia Nurs. 2020, 35, 441–444. [Google Scholar] [CrossRef]
- Billard, V. Pharmacokinetic-pharmacodynamic relationship of anesthetic drugs: From modeling to clinical use. F1000Research 2015, 4. [Google Scholar] [CrossRef]
- Brown, E.N.; Pavone, K.J.; Naranjo, M. Multimodal general anesthesia: Theory and practice. Anesth. Analg. 2018, 127, 1246. [Google Scholar] [CrossRef] [PubMed]
- Field, R.R. The syringe driver: Continuous subcutaneous infusions in palliative care. Anesth. Analg. 2018, 126, 717. [Google Scholar] [CrossRef]
- Meyer, R.E.; Fish, R. Pharmacology of injectable anesthetics, sedatives, and tranquilizers. In Anesthesia and Analgesia in Laboratory Animals, 2nd ed.; Academic Press: San Diego, CA, USA, 2008. [Google Scholar]
- Miller, A.L.; Theodore, D.; Widrich, J. Inhalational Anesthetic. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Berry, S.H. Injectable anesthetics. Vet. Anesth. Analg. 2015, 277. [Google Scholar] [CrossRef]
- Buitrago, S.; Martin, T.E.; Tetens-Woodring, J.; Belicha-Villanueva, A.; Wilding, G.E. Safety and efficacy of various combinations of injectable anesthetics in BALB/c mice. J. Am. Assoc. Lab. Anim. Sci. 2008, 47, 11–17. [Google Scholar] [PubMed]
- Posner, L.P.; Burns, P. Injectable anaesthetic agents. Vet. Pharmacol. Ther. 2009, 266, 300. [Google Scholar]
- Jurd, R.; Arrasa, M.; Lambert, S.; Drexler, B.; Siegwart, R.; Crestani, F.; Zaugg, M.; Vogt, K.E.; Ledermann, B.; Antkowiak, B. General anesthetic actions in vivo strongly attenuated by a point mutation in the GABAA receptor β3 subunit. Faseb J. 2003, 17, 250–252. [Google Scholar] [CrossRef]
- Fish, R.E. Pharmacology of injectable anesthetics. In Anesthesia and Analgesia in Laboratory Animals; Elsevier: Amsterdam, The Netherlands, 1997; pp. 1–28. [Google Scholar]
- Forman, S.A.; Ishizawa, Y. Inhaled Anesthetic Pharmacokinetics: Uptake, Distribution, Metabolism, and toxicity. In Miller’s Anesthesia, 8th ed.; Saunders: Philadelphia, PA, USA, 2015; pp. 638–669. [Google Scholar]
- Campagna, J.A.; Miller, K.W.; Forman, S.A. Mechanisms of actions of inhaled anesthetics. N. Engl. J. Med. 2003, 348, 2110–2124. [Google Scholar] [CrossRef]
- Smith, R.A.; Porter, E.G.; Miller, K.W. The solubility of anesthetic gases in lipid bilayers. Biochim. Biophys. Acta (BBA)—Biomembr. 1981, 645, 327–338. [Google Scholar] [CrossRef]
- Behne, M.; Wilke, H.-J.; Harder, S. Clinical pharmacokinetics of sevoflurane. Clin. Pharmacokinet. 1999, 36, 13–26. [Google Scholar] [CrossRef]
- Schwarzkopf, K.; Schreiber, T.; Preussler, N.-P.; Gaser, E.; Hüter, L.; Bauer, R.; Schubert, H.; Karzai, W. Lung perfusion, shunt fraction, and oxygenation during one-lung ventilation in pigs: The effects of desflurane, isoflurane, and propofol. J. Cardiothorac. Vasc. Anesth. 2003, 17, 73–75. [Google Scholar] [CrossRef] [PubMed]
- Urban, B. The site of anesthetic action. In Modern Anesthetics; Springer: Berlin/Heidelberg, Germany, 2008; pp. 3–29. [Google Scholar]
- Grasshoff, C.; Rudolph, U.; Antkowiak, B. Molecular and systemic mechanisms of general anesthesia: The ‘multi-site and multiple mechanisms’ concept. Curr. Opin. Anesthesiol. 2005, 18, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Alkire, M.T.; Hudetz, A.G.; Tononi, G. Consciousness and anesthesia. Science 2008, 322, 876–880. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, U.; Antkowiak, B. Molecular and neuronal substrates for general anaesthetics. Nat. Rev. Neurosci. 2004, 5, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Kelz, M.B.; Mashour, G.A. The biology of general anesthesia from paramecium to primate. Curr. Biol. 2019, 29, R1199–R1210. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Vlisides, P.E.; Kelz, M.B.; Avidan, M.S.; Mashour, G.A. Dynamic cortical connectivity during general anesthesia in healthy volunteers. Anesthesiology 2019, 130, 870–884. [Google Scholar] [CrossRef]
- Kitamura, A.; Marszalec, W.; Yeh, J.Z.; Narahashi, T. Effects of halothane and propofol on excitatory and inhibitory synaptic transmission in rat cortical neurons. J. Pharmacol. Exp. Ther. 2003, 304, 162–171. [Google Scholar] [CrossRef]
- Franks, N.; Lieb, W. Molecular and cellular mechanisms of general anesthesia. Nature 1994, 367, 607–614. [Google Scholar] [CrossRef]
- De Sousa, S.L.; Dickinson, R.; Lieb, W.R.; Franks, N.P. Contrasting synaptic actions of the inhalational general anesthetics isoflurane and xenon. Anesth. J. Am. Soc. Anesth. 2000, 92, 1055–1066. [Google Scholar] [CrossRef]
- Urban, B.W.; Bleckwenn, M.; Barann, M. Interactions of anesthetics with their targets: Non-specific, specific or both? Pharmacol. Ther. 2006, 111, 729–770. [Google Scholar] [CrossRef]
- Forman, S.A.; Miller, K.W. Mapping general anesthetic sites in heteromeric gamma-aminobutyric acid type a receptors reveals a potential for targeting receptor subtypes. Anesth. Analg. 2016, 123, 1263. [Google Scholar] [CrossRef]
- Hemmings, H.C., Jr.; Riegelhaupt, P.M.; Kelz, M.B.; Solt, K.; Eckenhoff, R.G.; Orser, B.A.; Goldstein, P.A. Towards a comprehensive understanding of anesthetic mechanisms of action: A decade of discovery. Trends Pharmacol. Sci. 2019, 40, 464–481. [Google Scholar] [CrossRef] [PubMed]
- Fourati, Z.; Howard, R.J.; Heusser, S.A.; Hu, H.; Ruza, R.R.; Sauguet, L.; Lindahl, E.; Delarue, M. Structural basis for a bimodal allosteric mechanism of general anesthetic modulation in pentameric ligand-gated ion channels. Cell Rep. 2018, 23, 993–1004. [Google Scholar] [CrossRef] [PubMed]
- Minami, K.; Uezono, Y. Gq protein-coupled receptors as targets for anesthetics. Curr. Pharm. Des. 2006, 12, 1931–1937. [Google Scholar] [CrossRef] [PubMed]
- Pavel, M.A.; Petersen, E.N.; Wang, H.; Lerner, R.A.; Hansen, S.B. Studies on the mechanism of general anesthesia. Proc. Natl. Acad. Sci. USA 2020, 117, 13757–13766. [Google Scholar] [CrossRef]
- Sloan, T.B. Anesthetics and the brain. Anesthesiol. Clin. N. Am. 2002, 20, 265–292. [Google Scholar] [CrossRef]
- Voss, L.J.; Sleigh, J.W. A metabolic mechanism for anaesthetic suppression of cortical synaptic function in mouse brain slices—A pilot investigation. Int. J. Mol. Sci. 2020, 21, 4703. [Google Scholar] [CrossRef]
- Laredo, F. Injectable Anesthetics; Clinician’s Brief: Tulsa, OK, USA, 2015; pp. 27–32. [Google Scholar]
- Hara, K.; Harris, R.A. The anesthetic mechanism of urethane: The effects on neurotransmitter-gated ion channels. Anesth. Analg. 2002, 94, 313–318. [Google Scholar]
- Simons, D.J.; Carvell, G.E.; Hershey, A.E.; Bryant, D.P. Responses of barrel cortex neurons in awake rats and effects of urethane anesthesia. Exp. Brain Res. 1992, 91, 259–272. [Google Scholar] [CrossRef]
- Pagliardini, S.; Funk, G.D.; Dickson, C.T. Breathing and brain state: Urethane anesthesia as a model for natural sleep. Respir. Physiol. Neurobiol. 2013, 188, 324–332. [Google Scholar] [CrossRef]
- Maggi, C.; Meli, A. Suitability of urethane anesthesia for physiopharmacological investigations in various systems Part 1: General considerations. Experientia 1986, 42, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Koblin, D.D. Urethane: Help or hindrance? Anesth. Anelgesia 2002, 94. [Google Scholar] [CrossRef]
- Sceniak, M.P.; MacIver, M.B. Cellular actions of urethane on rat visual cortical neurons in vitro. J. Neurophysiol. 2006, 95, 3865–3874. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Puerta, V.; Yang, J.-W.; Siwek, M.E.; Kilb, W.; Sun, J.-J.; Luhmann, H.J. Propagation of spontaneous slow-wave activity across columns and layers of the adult rat barrel cortex in vivo. Brain Struct. Funct. 2016, 221, 4429–4449. [Google Scholar] [CrossRef] [PubMed]
- Clement, E.A.; Richard, A.; Thwaites, M.; Ailon, J.; Peters, S.; Dickson, C.T. Cyclic and sleep-like spontaneous alternations of brain state under urethane anesthesia. PLoS ONE 2008, 3, e2004. [Google Scholar] [CrossRef]
- Rojas, M.J.; Navas, J.A.; Rector, D.M. Evoked response potential markers for anesthetic and behavioral states. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 291, R189–R196. [Google Scholar] [CrossRef][Green Version]
- Hama, N.; Ito, S.-I.; Hirota, A. Optical imaging of the propagation patterns of neural responses in the rat sensory cortex: Comparison under two different anesthetic conditions. Neuroscience 2015, 284, 125–133. [Google Scholar] [CrossRef]
- Unichenko, P.; Yang, J.-W.; Kirischuk, S.; Kolbaev, S.; Kilb, W.; Hammer, M.; Krueger-Burg, D.; Brose, N.; Luhmann, H.J. Autism related neuroligin-4 knockout impairs intracortical processing but not sensory inputs in mouse barrel cortex. Cereb. Cortex 2018, 28, 2873–2886. [Google Scholar] [CrossRef]
- Erchova, I.A.; Lebedev, M.A.; Diamond, M.E. Somatosensory cortical neuronal population activity across states of anesthesia. Eur. J. Neurosci. 2002, 15, 744–752. [Google Scholar] [CrossRef]
- Devonshire, I.M.; Grandy, T.H.; Dommett, E.J.; Greenfield, S.A. Effects of urethane anesthesia on sensory processing in the rat barrel cortex revealed by combined optical imaging and electrophysiology. Eur. J. Neurosci. 2010, 32, 786–797. [Google Scholar] [CrossRef]
- Sha, O.; Hao, Y.; Cho, E.; Zhou, L. Clinical Applications and Side Effects of Ketamine. In Ketamine: Use and Abuse; CRC Press: Boca Raton, FL, USA, 2015; pp. 13–36. [Google Scholar]
- Kalmoe, M.C.; Janski, A.M.; Zorumski, C.F.; Nagele, P.; Palanca, B.J.; Conway, C.R. Ketamine and nitrous oxide: The evolution of NMDA receptor antagonists as antidepressant agents. J. Neurol. Sci. 2020, 412, 116778. [Google Scholar] [CrossRef] [PubMed]
- Zanos, P.; Moaddel, R.; Morris, P.J.; Georgiou, P.; Fischell, J.; Elmer, G.I.; Alkondon, M.; Yuan, P.; Pribut, H.J.; Singh, N.S. NMDAR inhibition-independent antidepressant actions of ketamine metabolites. Nature 2016, 533, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Hirota, K.; Kubota, T.; Ishihara, H.; Matsuki, A. The effects of nitrous oxide and ketamine on the bispectral index and 95% spectral edge frequency during propofol–fentanyl anesthesia. Eur. J. Anaesthesiol. 1999, 16, 779–783. [Google Scholar] [PubMed]
- Morgan, C.J.; Curran, H.V.; Drugs, I.S.C.o. Ketamine use: A review. Addiction 2012, 107, 27–38. [Google Scholar] [CrossRef]
- Kurdi, M.S.; Theerth, K.A.; Deva, R.S. Ketamine: Current applications in anesthesia, pain, and critical care. Anesth. Essays Res. 2014, 8, 283. [Google Scholar] [CrossRef]
- Dimitriadis, G.; Fransen, A.M.; Maris, E. Sensory and cognitive neurophysiology in rats. Part 2: Validation and demonstration. J. Neurosci. Methods 2014, 232, 47–57. [Google Scholar] [CrossRef]
- Dimitriadis, G.; Fransen, A.M.; Maris, E. Sensory and cognitive neurophysiology in rats, Part 1: Controlled tactile stimulation and micro-ECoG recordings in freely moving animals. J. Neurosci. Methods 2014, 232, 63–73. [Google Scholar] [CrossRef]
- Struck, M.B.; Andrutis, K.A.; Ramirez, H.E.; Battles, A.H. Effect of a short-term fast on ketamine–xylazine anesthesia in rats. J. Am. Assoc. Lab. Anim. Sci. 2011, 50, 344–348. [Google Scholar]
- Wixson, S.; White, W.; Hughes Jr, H.; Lang, C.; Marshall, W. A comparison of pentobarbital, fentanyl-droperidol, ketamine-xylazine and ketamine-diazepam anesthesia in adult male rats. Lab. Anim. Sci. 1987, 37, 726–730. [Google Scholar]
- Van Pelt, L. Ketamine and xylazine for surgical anesthesia in rats. J. Am. Vet. Med Assoc. 1977, 171, 842–844. [Google Scholar]
- Fontanini, A.; Bower, J.M. Variable coupling between olfactory system activity and respiration in ketamine/xylazine anesthetized rats. J. Neurophysiol. 2005, 93, 3573–3581. [Google Scholar] [CrossRef] [PubMed]
- Tandon, S.; Kambi, N.; Jain, N. Overlapping representations of the neck and whiskers in the rat motor cortex revealed by mapping at different anaesthetic depths. Eur. J. Neurosci. 2008, 27, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Ordek, G.; Groth, J.D.; Sahin, M. Differential effects of ketamine/xylazine anesthesia on the cerebral and cerebellar cortical activities in the rat. J. Neurophysiol. 2013, 109, 1435–1443. [Google Scholar] [CrossRef] [PubMed]
- Fiáth, R.; Kerekes, B.P.; Wittner, L.; Tóth, K.; Beregszászi, P.; Horváth, D.; Ulbert, I. Laminar analysis of the slow wave activity in the somatosensory cortex of anesthetized rats. Eur. J. Neurosci. 2016, 44, 1935–1951. [Google Scholar] [CrossRef] [PubMed]
- Bettinardi, R.G.; Tort-Colet, N.; Ruiz-Mejias, M.; Sanchez-Vives, M.V.; Deco, G. Gradual emergence of spontaneous correlated brain activity during fading of general anesthesia in rats: Evidences from fMRI and local field potentials. Neuroimage 2015, 114, 185–198. [Google Scholar] [CrossRef]
- Naaz, S.; Ozair, E. Dexmedetomidine in current anesthesia practice-a review. J. Clin. Diagn. Res. JCDR 2014, 8, GE01. [Google Scholar]
- Angel, A. Central neuronal pathways and the process of anesthesia. BJA Br. J. Anaesth. 1993, 71, 148–163. [Google Scholar] [CrossRef]
- Chen, B.-S.; Peng, H.; Wu, S.-N. Dexmedetomidine, an α2-adrenergic agonist, inhibits neuronal delayed-rectifier potassium current and sodium current. Br. J. Anaesth. 2009, 103, 244–254. [Google Scholar] [CrossRef]
- Hayton, S.; Kriss, A.; Muller, D. Comparison of the effects of four anaesthetic agents on somatosensory evoked potentials in the rat. Lab. Anim. 1999, 33, 243–251. [Google Scholar] [CrossRef]
- Li, B.-H.; Lohmann, J.S.; Schuler, H.G.; Cronin, A.J. Preservation of the cortical somatosensory-evoked potential during dexmedetomidine infusion in rats. Anesth. Analg. 2003, 96, 1155–1160. [Google Scholar] [CrossRef]
- Banks, M.; Moran, N.; Krause, B.; Grady, S.; Uhlrich, D.; Manning, K. Altered stimulus representation in rat auditory cortex is not causal for loss of consciousness under general anesthesia. Br. J. Anaesth. 2018, 121, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Plourde, G.; Arseneau, F. Attenuation of high-frequency (30–200 Hz) thalamocortical EEG rhythms as correlate of anaesthetic action: Evidence from dexmedetomidine. BJA: Br. J. Anaesth. 2017, 119, 1150–1160. [Google Scholar] [CrossRef] [PubMed]
- Shibuta, H.; Yamana, R.; Kashimoto, J.; Kamio, K.; Suda, A. Comparison of the anesthetic effect by the injection route of mixed anesthesia (medetomidine, midazolam and butorphanol) and the effect of this anesthetic agent on the respiratory function. J. Vet. Med Sci. 2019. [Google Scholar] [CrossRef] [PubMed]
- Osanai, H.; Tateno, T. Neural response differences in the rat primary auditory cortex under anesthesia with ketamine versus the mixture of medetomidine, midazolam and butorphanol. Hear. Res. 2016, 339, 69–79. [Google Scholar] [CrossRef]
- Peters, J.A.; Kirkness, E.F.; Callachan, H.; Lambert, J.J.; Turner, A.J. Modulation of the GABAA receptor by depressant barbiturates and pregnane steroids. Br. J. Pharmacol. 1988, 94, 1257–1269. [Google Scholar] [CrossRef]
- Johnson, A.B.; Sadiq, N.M. Pentobarbital. In StatPearls [Internet]; StatPearls: Treasure Island, FL, USA, 2020. [Google Scholar]
- Kuo, C.-C.; Lee, J.-C.; Chiou, R.-J.; Tsai, M.-L.; Yen, C.-T. Spatiotemporal changes of neuronal responses in the primary somatosensory cortex to Noxious tail stimulation in awake and pentobarbital-anesthetized rats. Chin. J. Physiol. 2015, 58, 332–342. [Google Scholar] [CrossRef]
- Belrose, J.C.; Noppens, R.R. Anesthesiology and cognitive impairment: A narrative review of current clinical literature. BMC Anesthesiol. 2019, 19, 1–12. [Google Scholar] [CrossRef]
- Vanlersberghe, C.; Camu, F. Propofol. In Modern Anesthetics; Springer: Berlin/Heidelberg, Germany, 2008; pp. 227–252. [Google Scholar]
- Hill-Venning, C.; Belelli, D.; Peters, J.A.; Lambert, J.J. Subunit-dependent interaction of the general anaesthetic etomidate with the γ-aminobutyric acid type A receptor. Br. J. Pharmacol. 1997, 120, 749–756. [Google Scholar] [CrossRef]
- Krasowski, M.D.; Koltchine, V.V.; Rick, C.E.; Ye, Q.; Finn, S.E.; Harrison, N.L. Propofol and other intravenous anesthetics have sites of action on the γ-aminobutyric acid type A receptor distinct from that for isoflurane. Mol. Pharmacol. 1998, 53, 530–538. [Google Scholar] [CrossRef]
- Siegwart, R.; Jurd, R.; Rudolph, U. Molecular determinants for the action of general anesthetics at recombinant α2β3γ2γ-aminobutyric acidA receptors. J. Neurochem. 2002, 80, 140–148. [Google Scholar] [CrossRef]
- Fowler, C.J. Possible involvement of the endocannabinoid system in the actions of three clinically used drugs. Trends Pharmacol. Sci. 2004, 25, 59–61. [Google Scholar] [CrossRef] [PubMed]
- Shafer, A.; Doze, V.A.; Shafer, S.L.; White, P.F. Pharmacokinetics and pharmacodynamics of propofol infusions during general anesthesia. Anesthesiol. J. Am. Soc. Anesthesiol. 1988, 69, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Purdon, P.; Pavone, K.; Akeju, O.; Smith, A.; Sampson, A.; Lee, J.; Zhou, D.; Solt, K.; Brown, E. The ageing brain: Age-dependent changes in the electroencephalogram during propofol and sevoflurane general anesthesia. Br. J. Anaesth. 2015, 115, i46–i57. [Google Scholar] [CrossRef] [PubMed]
- Sahinovic, M.M.; Struys, M.M.; Absalom, A.R. Clinical pharmacokinetics and pharmacodynamics of propofol. Clin. Pharmacokinet. 2018, 57, 1539–1558. [Google Scholar] [CrossRef] [PubMed]
- Klostermann, F.; Funk, T.; Vesper, J.; Siedenberg, R.; Curio, G. Propofol narcosis dissociates human intrathalamic and cortical high-frequency (>400 Hz) SEP components. Neuroreport 2000, 11, 2607–2610. [Google Scholar] [CrossRef]
- Landoni, G.; Lomivorotov, V.V.; Nigro Neto, C.; Monaco, F.; Pasyuga, V.V.; Bradic, N.; Lembo, R.; Gazivoda, G.; Likhvantsev, V.V.; Lei, C.; et al. Volatile anesthetics versus total intravenous anesthesia for cardiac surgery. N. Engl. J. Med. 2019, 380, 1214–1225. [Google Scholar] [CrossRef]
- Airaksinen, A.M.; Hekmatyar, S.K.; Jerome, N.; Niskanen, J.P.; Huttunen, J.K.; Pitkänen, A.; Kauppinen, R.A.; Gröhn, O.H. Simultaneous BOLD fMRI and local field potential measurements during kainic acid–induced seizures. Epilepsia 2012, 53, 1245–1253. [Google Scholar] [CrossRef]
- Noda, T.; Takahashi, H. Anesthetic effects of isoflurane on the tonotopic map and neuronal population activity in the rat auditory cortex. Eur. J. Neurosci. 2015, 42, 2298–2311. [Google Scholar] [CrossRef]
- Cho, D.; Shin, T.J.; Ham, J.; Choi, D.-H.; Kim, S.; Jeong, S.; Kim, H.-I.; Kim, J.G.; Lee, B. Differential modulation of thalamo-parietal interactions by varying depths of isoflurane anesthesia. PLoS ONE 2017, 12, e0175191. [Google Scholar] [CrossRef]
- Liu, X.; Zhu, X.-H.; Zhang, Y.; Chen, W. The change of functional connectivity specificity in rats under various anesthesia levels and its neural origin. Brain Topogr. 2013, 26, 363–377. [Google Scholar] [CrossRef]
- White, B.; Abbott, L.F.; Fiser, J. Suppression of cortical neural variability is stimulus-and state-dependent. J. Neurophysiol. 2012, 108, 2383–2392. [Google Scholar] [CrossRef] [PubMed]
- Sellers, K.K.; Bennett, D.V.; Hutt, A.; Williams, J.H.; Fröhlich, F. Awake vs. anesthetized: Layer-specific sensory processing in visual cortex and functional connectivity between cortical areas. J. Neurophysiol. 2015, 113, 3798–3815. [Google Scholar] [CrossRef] [PubMed]
- Aasebø, I.E.; Lepperød, M.E.; Stavrinou, M.; Nøkkevangen, S.; Einevoll, G.; Hafting, T.; Fyhn, M. Temporal processing in the visual cortex of the awake and anesthetized rat. eNeuro 2017, 4. [Google Scholar] [CrossRef]
- Abe, Y.; Tsurugizawa, T.; Le Bihan, D. Water diffusion closely reveals neural activity status in rat brain loci affected by anesthesia. PLoS Biol. 2017, 15, e2001494. [Google Scholar] [CrossRef] [PubMed]
- Guidera, J.A.; Taylor, N.E.; Lee, J.T.; Vlasov, K.Y.; Pei, J.; Stephen, E.P.; Mayo, J.P.; Brown, E.N.; Solt, K. Sevoflurane induces coherent slow-delta oscillations in rats. Front. Neural Circuits 2017, 11, 36. [Google Scholar] [CrossRef]
- Cecchetto, C.; Mahmud, M.; Vassanelli, S. Anesthesia effect on single local field potentials variability in rat barrel cortex: Preliminary results. In Proceedings of the 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015; pp. 4721–4724. [Google Scholar]
- Mahmud, M.; Cecchetto, C.; Vassanelli, S. An automated method for characterization of evoked single-trial local field potentials recorded from rat barrel cortex under mechanical whisker stimulation. Cogn. Comput. 2016, 8, 935–945. [Google Scholar] [CrossRef]
- Sanders, R.; Brian, D.; Maze, M. G-Protein-Coupled Receptors. In Modern Anesthetics; Springer: Berlin/Heidelberg, Germany, 2008; pp. 93–117. [Google Scholar]
- Wang, C.; Slikker, W., Jr. Strategies and experimental models for evaluating anesthetics: Effects on the developing nervous system. Anesth. Analg. 2008, 106, 1643–1658. [Google Scholar] [CrossRef]
- Colon, E.; Bittner, E.A.; Kussman, B.; McCann, M.E.; Soriano, S.; Borsook, D. Anesthesia, brain changes, and behavior: Insights from neural systems biology. Prog. Neurobiol. 2017, 153, 121–160. [Google Scholar] [CrossRef]
- Sun, L.-H.; Fan, Y.-Y.; Wang, X.; Zheng, H.-B. Pharmacodynamic elucidation of glutamate & dopamine in ketamine-induced anesthesia. Chem. Biol. Interact. 2020, 327, 109164. [Google Scholar]
- Nowacka, A.; Borczyk, M. Ketamine applications beyond anesthesia—A literature review. Eur. J. Pharmacol. 2019, 860, 172547. [Google Scholar] [CrossRef]
- Ruiz-Colón, K.; Chavez-Arias, C.; Díaz-Alcalá, J.E.; Martínez, M.A. Xylazine intoxication in humans and its importance as an emerging adulterant in abused drugs: A comprehensive review of the literature. Forensic Sci. Int. 2014, 240, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gertler, R.; Brown, H.C.; Mitchell, D.H.; Silvius, E.N. Dexmedetomidine: A novel sedative-analgesic agent. Bayl. Univ. Med Cent. Proc. 2001, 14, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Sivaramakrishnan, G.; Sridharan, K. Nitrous oxide and midazolam sedation: A systematic review and meta-analysis. Anesth. Prog. 2017, 64, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Bansal, P.; Jain, G. Control of shivering with clonidine, butorphanol, and tramadol under spinal anesthesia: A comparative study. Local Reg. Anesth. 2011, 4, 29. [Google Scholar] [CrossRef]
- Kirihara, Y.; Takechi, M.; Kurosaki, K.; Kobayashi, Y.; Saito, Y.; Takeuchi, T. Effects of an anesthetic mixture of medetomidine, midazolam, and butorphanol in rats—Strain difference and antagonism by atipamezole. Exp. Anim. 2016, 65, 27–36. [Google Scholar] [CrossRef]
- Maldifassi, M.C.; Baur, R.; Sigel, E. Functional sites involved in modulation of the GABAA receptor channel by the intravenous anesthetics propofol, etomidate and pentobarbital. Neuropharmacology 2016, 105, 207–214. [Google Scholar] [CrossRef]
- Bryson, H.M.; Fulton, B.R.; Faulds, D. Propofol. Drugs 1995, 50, 513–559. [Google Scholar] [CrossRef]
- Soltanizadeh, S.; Degett, T.H.; Gögenur, I. Outcomes of cancer surgery after inhalational and intravenous anesthesia: A systematic review. J. Clin. Anesth. 2017, 42, 19–25. [Google Scholar] [CrossRef]
- Brioni, J.D.; Varughese, S.; Ahmed, R.; Bein, B. A clinical review of inhalation anesthesia with sevoflurane: From early research to emerging topics. J. Anesth. 2017, 31, 764–778. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sorrenti, V.; Cecchetto, C.; Maschietto, M.; Fortinguerra, S.; Buriani, A.; Vassanelli, S. Understanding the Effects of Anesthesia on Cortical Electrophysiological Recordings: A Scoping Review. Int. J. Mol. Sci. 2021, 22, 1286. https://doi.org/10.3390/ijms22031286
Sorrenti V, Cecchetto C, Maschietto M, Fortinguerra S, Buriani A, Vassanelli S. Understanding the Effects of Anesthesia on Cortical Electrophysiological Recordings: A Scoping Review. International Journal of Molecular Sciences. 2021; 22(3):1286. https://doi.org/10.3390/ijms22031286
Chicago/Turabian StyleSorrenti, Vincenzo, Claudia Cecchetto, Marta Maschietto, Stefano Fortinguerra, Alessandro Buriani, and Stefano Vassanelli. 2021. "Understanding the Effects of Anesthesia on Cortical Electrophysiological Recordings: A Scoping Review" International Journal of Molecular Sciences 22, no. 3: 1286. https://doi.org/10.3390/ijms22031286
APA StyleSorrenti, V., Cecchetto, C., Maschietto, M., Fortinguerra, S., Buriani, A., & Vassanelli, S. (2021). Understanding the Effects of Anesthesia on Cortical Electrophysiological Recordings: A Scoping Review. International Journal of Molecular Sciences, 22(3), 1286. https://doi.org/10.3390/ijms22031286










