Sodium Salicylate Influences the Pseudomonas aeruginosa Biofilm Structure and Susceptibility Towards Silver
Abstract
1. Introduction
2. Results
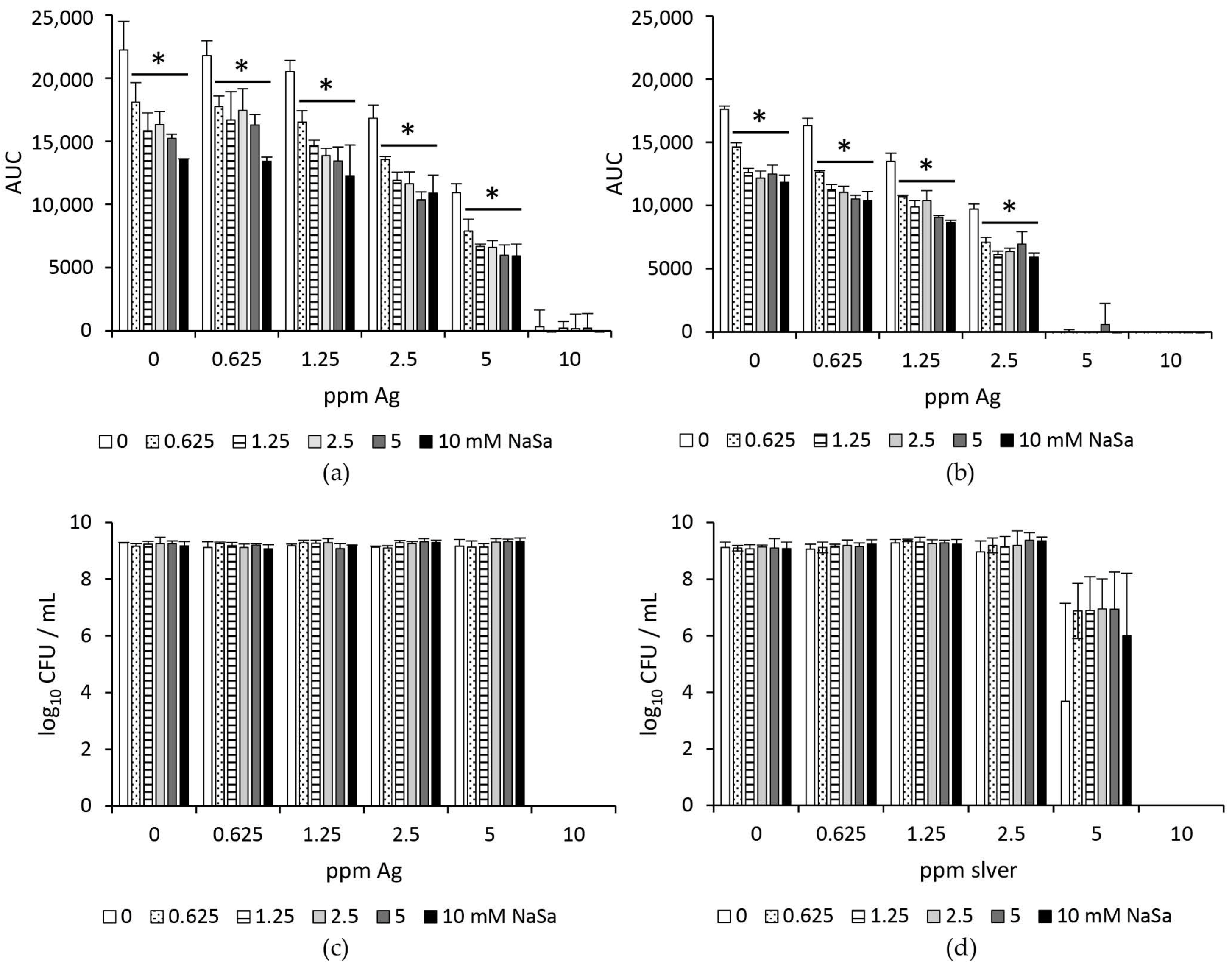
2.1. Increased Silver Susceptibility of Biofilms Formed by Pseudomonas aeruginosa in the Presence of NaSa
2.2. Silver and NaSa Suppress the Growth but Not the Viability of Pseudomonas aeruginosa
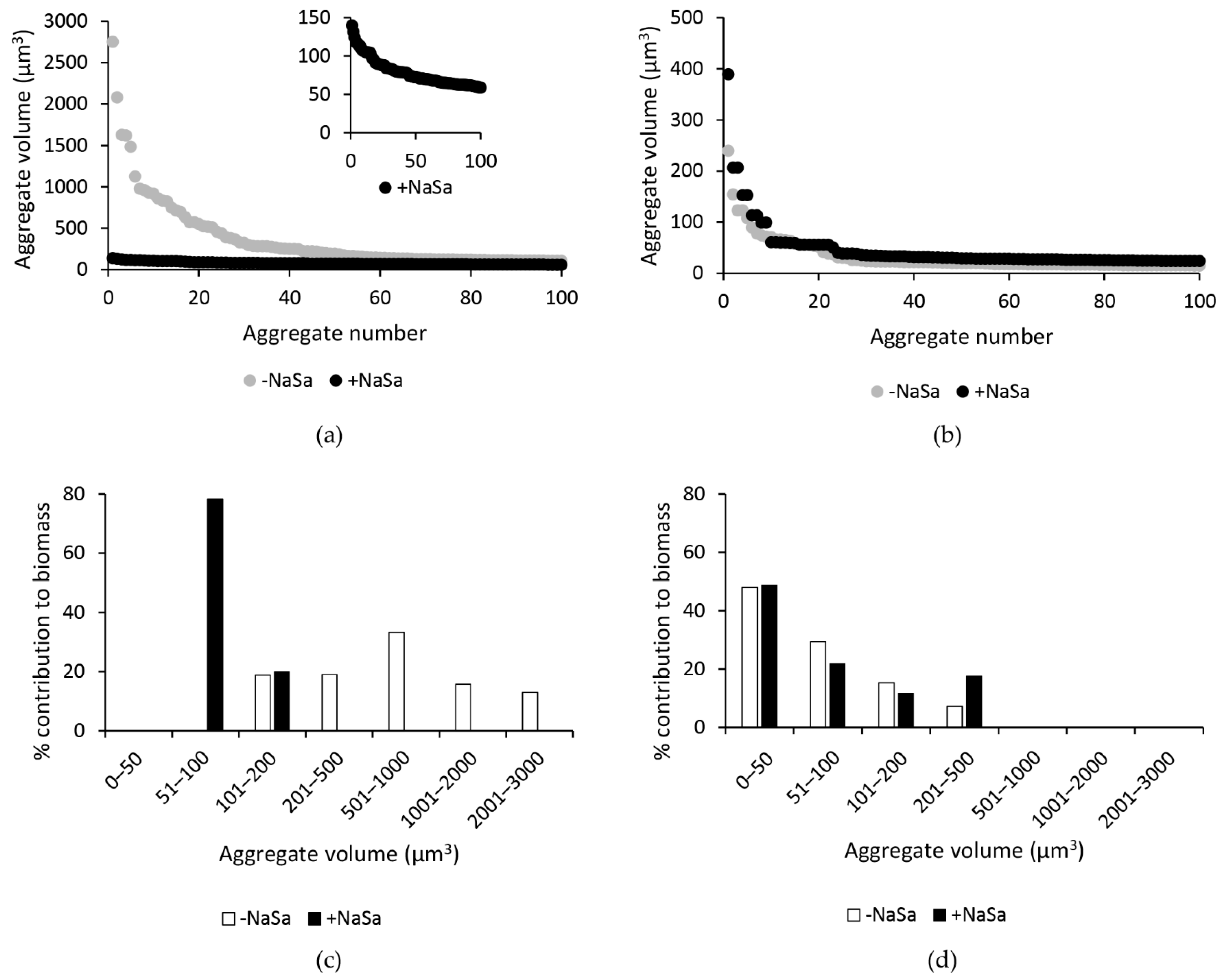
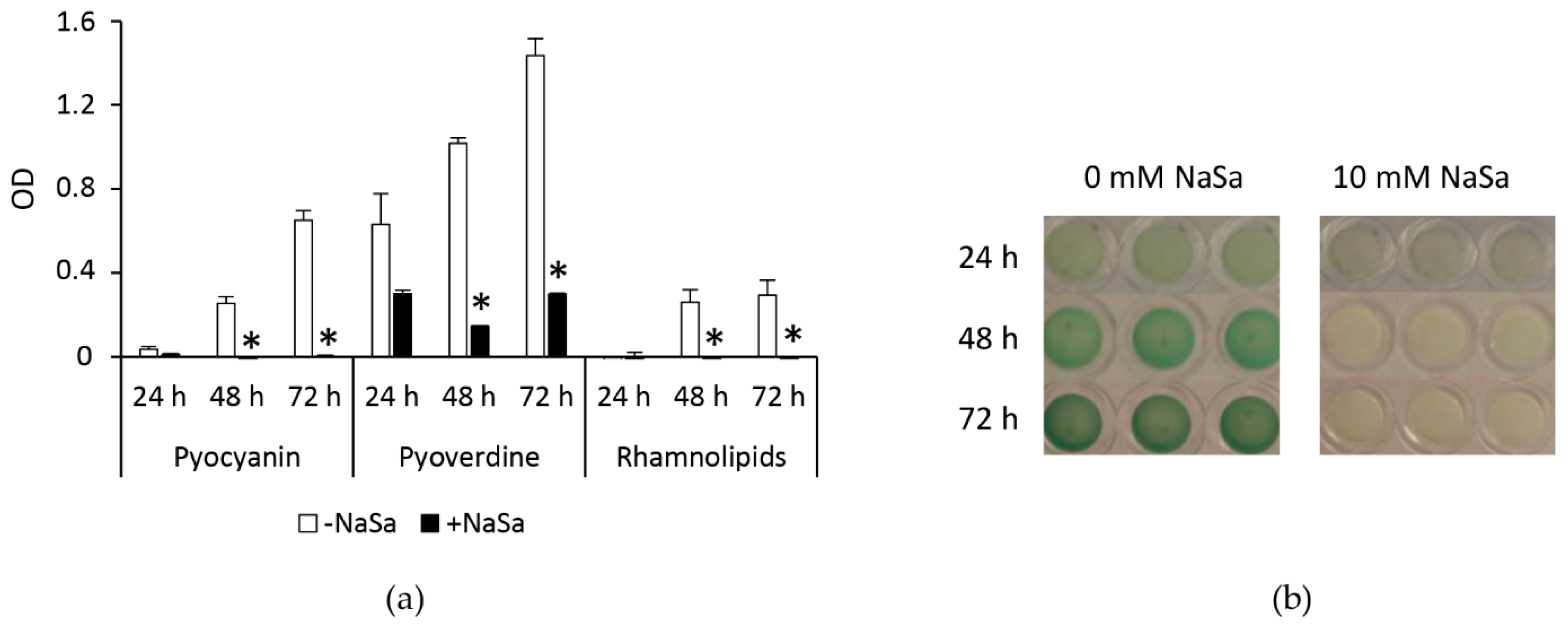
2.3. NaSa Reduces Pseudomonas aeruginosa Biofilm Formation, Cell Aggregation, and Virulence Factor Production
3. Discussion
4. Materials and Methods
4.1. Pseudomonas aeruginosa Strains and Culture Conditions
4.2. Pseudomonas aeruginosa Biofilm Formation and Susceptibility Towards Silver in the Presence or Absence of NaSa
4.3. Pseudomonas aeruginosa Biofilm Virulence Factor Production
4.4. Planktonic Cells of Pseudomonas aeruginosa
4.5. Pseudomonas aeruginosa Biofilm Architecture
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Ag | Silver |
| ASA | Acetylsalicylic acid |
| BBC | Biofilm bactericidal concentration |
| CF | Cystic fibrosis |
| CFU | Colony forming units |
| CLSM | Confocal laser scanning microscopy |
| eDNA | Extracellular DNA |
| EPS | Extracellular polymeric substances |
| MBC | Minimum bactericidal concentration |
| MIC | Minimum inhibitory concentration |
| OD | Optical density |
| SA | Salicylic acid |
| SWF | Simulated Wound Fluid |
| NaSa | Sodium salicylate |
| QS | Quorum sensing |
| QSIs | Quorum sensing inhibitors |
References
- Gottrup, F. A specialized wound-healing center concept: Importance of a multidisciplinary department structure and surgical treatment facilities in the treatment of chronic wounds. Am. J. Surg. 2004, 187, S38–S43. [Google Scholar] [CrossRef]
- Schultz, G.; Bjarnsholt, T.; James, G.A.; Leaper, D.J.; McBain, A.J.; Malone, M.; Stoodley, P.; Swanson, T.; Tachi, M.; Wolcott, R.D. Consensus guidelines for the identification and treatment of biofilms in chronic non-healing wounds. Wound Repair Regen 2017, 25, 744–757. [Google Scholar] [CrossRef] [PubMed]
- Flemming, H.-C.; Wingender, J. The biofilm matrix. Nat. Rev. Microbiol. 2010, 8, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Malone, M.; Bjarnsholt, T.; McBain, A.; James, G.; Stoodley, P.; Leaper, D.; Tachi, M.; Schultz, G.; Swanson, T.; Wolcott, R. The prevalence of biofilms in chronic wounds: A systematic review and meta-analysis of published data. J. Wound Care 2017, 26, 20–25. [Google Scholar] [CrossRef]
- Salisbury, A.M.; Woo, K.; Sarkar, S.; Schultz, G.; Malone, M.; Mayer, D.O.; Percival, S.L. Tolerance of bio-films to antimicrobials and significance to antibiotic resistance in wounds. Surg. Technol. Int. 2018, 33, 59–66. [Google Scholar]
- Sugden, R.; Kelly, R.; Davies, S. Combatting antimicrobial resistance globally. Nat. Microbiol. 2016, 1, 16187. [Google Scholar] [CrossRef]
- Wolcott, R.D.; Hanson, J.D.; Rees, E.J.; Koenig, L.D.; Phillips, C.D.; Wolcott, R.A.; Cox, S.B.; White, J.S. Analysis of the chronic wound microbiota of 2963 patients by 16S rDNA pyrosequencing. Wound Repair Regen. 2016, 24, 163–174. [Google Scholar] [CrossRef]
- Gjødsbøl, K.; Christensen, J.J.; Karlsmark, T.; Jørgensen, B.; Klein, B.M.; Krogfelt, K.A. Multiple bacterial species reside in chronic wounds: A longitudinal study. Int. Wound J. 2006, 3, 225–231. [Google Scholar] [CrossRef]
- Lee, J.; Zhang, L.-H. The hierarchy quorum sensing network in Pseudomonas aeruginosa. Protein Cell 2015, 6, 26–41. [Google Scholar] [CrossRef]
- Cheung, A.L.; Bayer, A.S.; Zhang, G.; Gresham, H.; Xiong, Y.-Q. Regulation of virulence determinants in vitro and in vivo in Staphylococcus aureus. FEMS Immunol. Med Microbiol. 2004, 40, 1–9. [Google Scholar] [CrossRef]
- Davies, D.G.; Parsek, M.R.; Pearson, J.P.; Iglewski, B.H.; Costerton, J.W.; Greenberg, E.P. The involvement of cell-to-cell signals in the development of a bacterial biofilm. Science 1998, 280, 295–298. [Google Scholar] [CrossRef] [PubMed]
- De Kievit, T.R. Quorum sensing in Pseudomonas aeruginosa biofilms. Environ. Microbiol. 2009, 11, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Shrout, J.D.; Tolker-Nielsen, T.; Givskov, M.; Parsek, M.R. The contribution of cell-cell signaling and motility to bacterial biofilm formation. MRS Bull. 2011, 36, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Rémy, B.; Mion, S.; Plener, L.; Elias, M.; Chabrière, E.; Daudé, D. Interference in bacterial quorum sensing: A biopharmaceutical perspective. Front. Pharmacol. 2018, 9, 203. [Google Scholar] [CrossRef] [PubMed]
- Brackman, G.; Cos, P.; Maes, L.; Nelis, H.J.; Coenye, T. Quorum sensing inhibitors increase the susceptibility of bacterial biofilms to antibiotics in vitro and in vivo. Antimicrob. Agents Chemother. 2011, 55, 2655–2661. [Google Scholar] [CrossRef] [PubMed]
- El-Mowafy, S.A.; Abd El Galil, K.H.; El-Messery, S.M.; Shaaban, M. Aspirin is an efficient inhibitor of quorum sensing, virulence and toxins in Pseudomonas aeruginosa. Microb. Pathog. 2014, 74, 25–32. [Google Scholar] [CrossRef]
- Yang, L.; Rybtke, M.T.; Jakobsen, T.H.; Hentzer, M.; Bjarnsholt, T.; Givskov, M.; Tolker-Nielsen, T. Computer-aided identification of recognized drugs as Pseudomonas aeruginosa quorum-sensing inhibitors. Antimicrob. Agents Chemother. 2009, 53, 2432–2443. [Google Scholar] [CrossRef]
- Chow, S.; Gu, K.; Jiang, L.; Nassour, A. Salicylic acid affects swimming, twitching and swarming motility in Pseudomonas aeruginosa, resulting in decreased biofilm formation. J. Exp. Microbiol. Immunol. 2011, 15, 22–29. [Google Scholar]
- Yalkowsky, S.H.; Banerjee, S. Aqueous Solubility: Methods of Estimation for Organic Compounds; Marcel Dekker: New York, NY, USA, 1992. [Google Scholar]
- Gerner, E.; Almqvist, S.; Werthen, M.; Trobos, M. Sodium salicylate interferes with quorum-sensing-regulated virulence in chronic wound isolates of Pseudomonas aeruginosa in simulated wound fluid. J. Med. Microbiol. 2020, 69, 767–780. [Google Scholar] [CrossRef]
- Dissemond, J.; Böttrich, J.G.; Braunwarth, H.; Hilt, J.; Wilken, P.; Münter, K. Evidence for silver in wound care—meta-analysis of clinical studies from 2000–2015. J. Dtsch. Dermatol. Ges. 2017, 15, 524–535. [Google Scholar] [CrossRef]
- Werthen, M.; Henriksson, L.; Jensen, P.O.; Sternberg, C.; Givskov, M.; Bjarnsholt, T. An in vitro model of bacterial infections in wounds and other soft tissues. APMIS 2010, 118, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Soukarieh, F.; Williams, P.; Stocks, M.J.; Cámara, M. Pseudomonas aeruginosa quorum sensing systems as drug discovery targets: Current position and future perspectives. J. Med. Chem. 2018, 61, 10385–10402. [Google Scholar] [CrossRef] [PubMed]
- Reuter, K.; Steinbach, A.; Helms, V. Interfering with bacterial quorum sensing. Perspect. Med. Chem. 2016, 8, PMC.S13209-15. [Google Scholar] [CrossRef] [PubMed]
- Hentzer, M.; Wu, H.; Andersen, J.B.; Riedel, K.; Rasmussen, T.B.; Bagge, N.; Kumar, N.; Schembri, M.A.; Song, Z.; Kristoffersen, P.; et al. Attenuation of Pseudomonas aeruginosa virulence by quorum sensing inhibitors. EMBO J. 2003, 22, 3803–3815. [Google Scholar] [CrossRef] [PubMed]
- Bjarnsholt, T.; Jensen, P.Ø.; Rasmussen, T.B.; Christophersen, L.; Calum, H.; Hentzer, M.; Hougen, H.-P.; Rygaard, J.; Moser, C.; Eberl, L.; et al. Garlic blocks quorum sensing and promotes rapid clearing of pulmonary Pseudomonas aeruginosa infections. Microbiology 2005, 151, 3873–3880. [Google Scholar] [CrossRef]
- Tarnuzzer, R.W.; Schultz, G.S. Biochemical analysis of acute and chronic wound environments. Wound Repair Regen. 1996, 4, 321–325. [Google Scholar] [CrossRef]
- Aiba-Kojima, E.; Tsuno, N.H.; Inoue, K.; Matsumoto, D.; Shigeura, T.; Sato, T.; Suga, H.; Kato, H.; Nagase, T.; Gonda, K.; et al. Characterization of wound drainage fluids as a source of soluble factors associated with wound healing: Comparison with platelet-rich plasma and potential use in cell culture. Wound Repair Regen. 2007, 15, 511–520. [Google Scholar] [CrossRef]
- Trainor, G.L. The importance of plasma protein binding in drug discovery. Expert Opin. Drug Discov. 2007, 2, 51–64. [Google Scholar] [CrossRef]
- Ghuman, J.; Zunszain, P.A.; Petitpas, I.; Bhattacharya, A.A.; Otagiri, M.; Curry, S.H. Structural basis of the drug-binding specificity of human serum albumin. J. Mol. Biol. 2005, 353, 38–52. [Google Scholar] [CrossRef]
- Kruczek, C.; Qaisar, U.; Colmer-Hamood, J.A.; Hamood, A.N. Serum influences the expression of Pseudo-monas aeruginosa quorum-sensing genes and QS-controlled virulence genes during early and late stages of growth. MicrobiologyOpen 2014, 3, 64–79. [Google Scholar] [CrossRef]
- Smith, A.C.; Rice, A.; Sutton, B.; Gabrilska, R.; Wessel, A.K.; Whiteley, M.; Rumbaugh, K.P. Albumin inhibits pseudomonas aeruginosa quorum sensing and alters polymicrobial interactions. Infect. Immun. 2017, 85, e00116-17. [Google Scholar] [CrossRef] [PubMed]
- Price, C.T.; Lee, I.R.; Gustafson, J.E. The effects of salicylate on bacteria. Int. J. Biochem. Cell Biol. 2000, 32, 1029–1043. [Google Scholar] [CrossRef]
- Wang, T.; El Meouche, I.; Dunlop, M.J. Bacterial persistence induced by salicylate via reactive oxygen species. Sci. Rep. 2017, 7, 43839. [Google Scholar] [CrossRef] [PubMed]
- Bandara, M.; Sankaridurg, P.; Zhu, H.; Hume, E.; Willcox, M. Effect of salicylic acid on the membrane proteome and virulence of pseudomonas aeruginosa. Invest. Ophthalmol. Vis. Sci. 2016, 57, 1213–1220. [Google Scholar] [CrossRef]
- Christensen, G.D.; Simpson, W.A.; Younger, J.J.; Baddour, L.M.; Barrett, F.F.; Melton, D.M.; Beachey, E.H. Adherence of coagulase-negative staphylococci to plastic tissue culture plates: A quantitative model for the adherence of staphylococci to medical devices. J. Clin. Microbiol. 1985, 22, 996–1006. [Google Scholar] [CrossRef]
- Stewart, P.S. Antimicrobial tolerance in biofilms. Microbiol. Spectr. 2015, 3, 3. [Google Scholar] [CrossRef]
- Lewis, K. Persister cells, dormancy and infectious disease. Nat. Rev. Microbiol. 2006, 5, 48–56. [Google Scholar] [CrossRef]
- Donlan, R.M. Role of biofilms in antimicrobial resistance. ASAIO J. 2000, 46, S47–S52. [Google Scholar] [CrossRef]
- Higgins, S.; Heeb, S.; Rampioni, G.; Fletcher, M.P.; Williams, P.; Cámara, M. Differential regulation of the phenazine biosynthetic operons by quorum sensing in Pseudomonas aeruginosa PAO1-N. Front. Cell. Infect. Microbiol. 2018, 8, 252. [Google Scholar] [CrossRef]
- Ramos, I.; Dietrich, L.E.P.; Price-Whelan, A.; Newman, D.K. Phenazines affect biofilm formation by Pseudomonas aeruginosa in similar ways at various scales. Res. Microbiol. 2010, 161, 187–191. [Google Scholar] [CrossRef]
- Davey, M.E.; Caiazza, N.C.; O’Toole, G.A. Rhamnolipid surfactant production affects biofilm architecture in Pseudomonas aeruginosa PAO1. J. Bacteriol. 2003, 185, 1027–1036. [Google Scholar] [CrossRef] [PubMed]
- Pamp, S.J.; Tolker-Nielsen, T. Multiple roles of biosurfactants in structural biofilm development by Pseudomonas aeruginosa. J. Bacteriol. 2007, 189, 2531–2539. [Google Scholar] [CrossRef] [PubMed]
- Banin, E.; Vasil, M.L.; Greenberg, E.P. Iron and pseudomonas aeruginosa biofilm formation. Proc. Natl. Acad. Sci. USA 2005, 102, 11076–11081. [Google Scholar] [CrossRef] [PubMed]
- Fazli, M.; Almblad, H.; Rybtke, M.L.; Givskov, M.; Eberl, L.; Tolker-Nielsen, T. Regulation of biofilm formation in Pseudomonas and Burkholderia species. Environ. Microbiol. 2014, 16, 1961–1981. [Google Scholar] [CrossRef]
- Zimmermann, P.; Curtis, N. The effect of aspirin on antibiotic susceptibility. Expert Opin. Ther. Targets 2018, 22, 967–972. [Google Scholar] [CrossRef]
- Feltner, J.B.; Wolter, D.J.; Pope, C.E.; Groleau, M.C.; Smalley, N.E.; Greenberg, E.P.; Mayer-Hamblett, N.; Burns, J.; Déziel, E.; Hoffman, L.R.; et al. LasR variant cystic fibrosis isolates reveal an adaptable quorum-sensing hierarchy in Pseudomonas aeruginosa. mBio 2016, 7, e01513-16. [Google Scholar] [CrossRef]
- Hamood, A.N.; Griswold, J.; Colmer, J. Characterization of elastase-deficient clinical isolates of Pseudomonas aeruginosa. Infect. Immun. 1996, 64, 3154–3160. [Google Scholar] [CrossRef]
- Vanderwoude, J.; Fleming, D.; Azimi, S.; Trivedi, U.; Rumbaugh, K.P.; Diggle, S.P. The evolution of virulence in Pseudomonas aeruginosa during chronic wound infection. Proc. Biol. Sci. 2020, 287, 20202272. [Google Scholar] [CrossRef]
- Lansdown, A.B.; Williams, A. How safe is silver in wound care? J. Wound Care 2004, 13, 131–136. [Google Scholar] [CrossRef]
- Graham, C. The role of silver in wound healing. Br. J. Nurs. 2005, 14, S22–S28. [Google Scholar] [CrossRef]
- Palmer, B.F.; Clegg, D.J. Salicylate Toxicity. N. Engl. J. Med. 2020, 382, 2544–2555. [Google Scholar] [CrossRef] [PubMed]
- Madan, R.K.; Levitt, J. A review of toxicity from topical salicylic acid preparations. J. Am. Acad. Dermatol. 2014, 70, 788–792. [Google Scholar] [CrossRef] [PubMed]
- Schmidtchen, A.; Wolff, H.; Hansson, C. Differential proteinase expression by Pseudomonas aeruginosa derived from chronic leg ulcers. Acta Derm Venereol 2002, 81, 406–409. [Google Scholar] [CrossRef] [PubMed]
- Bjarnsholt, T.; Jensen, P.Ø.; Burmølle, M.; Hentzer, M.; Haagensen, J.A.J.; Hougen, H.P.; Calum, H.; Madsen, K.G.; Moser, C.; Molin, S.; et al. Pseudomonas aeruginosa tolerance to tobramycin, hydrogen peroxide and polymorphonuclear leukocytes is quorum-sensing dependent. Microbiology (Reading) 2005, 151 Pt 2, 373–383. [Google Scholar] [CrossRef]
- Klausen, M.; Heydorn, A.; Ragas, P.; Lambertsen, L.; Aaes-Jørgensen, A.; Molin, S.; Tolker-Nielsen, T. Biofilm formation by Pseudomonas aeruginosa wild type, flagella and type IV pili mutants. Mol. Microbiol. 2003, 48, 1511–1524. [Google Scholar] [CrossRef]
- O’Toole, G.A. Microtiter dish biofilm formation assay. J. Vis. Exp. 2011, 47, e2437. [Google Scholar] [CrossRef]
- Koch, A.K.; Käppeli, O.; Fiechter, A.; Reiser, J. Hydrocarbon assimilation and biosurfactant production in Pseudomonas aeruginosa mutants. J. Bacteriol. 1991, 173, 4212–4219. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Bolte, S.; Cordelières, F.P. A guided tour into subcellular colocalization analysis in light microscopy. J. Microsc. 2006, 224, 213–232. [Google Scholar] [CrossRef]
- Royer, L.A.; Weigert, M.; Günther, U.; Maghelli, N.; Jug, F.; Sbalzarini, I.F.; Myers, E.W. ClearVolume: Open-source live 3D visualization for light-sheet microscopy. Nat. Methods 2015, 12, 480–481. [Google Scholar] [CrossRef]



| Strain Name | Origin and Properties | Reference |
|---|---|---|
| 2 (M20.447) 5 (M20.450) | Clinical wound isolates of P. aeruginosa, with strain 2 and 5 exhibiting low and high in virulence factor and QS signal production, respectively | [20,54] |
| PAO1 wt | Reference P. aeruginosa strain (Pseudomonas Genetic Stock Center; strain PAO0001) | |
| PAO1 ∆lasR ∆rhlR | PAO1 lacking the QS signal receptors lasR and rhlR | [55] |
| PAO1 wt-GFP | PAO1 tagged with eGFP in a mini-Tn7 construct, Gmr | [56] |
| PAO1 ∆lasR∆rhlR-GFP | PAO1 ∆lasR ∆rhlR tagged with GFP expressed on plasmid pMRP9 | [55] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gerner, E.; Almqvist, S.; Thomsen, P.; Werthén, M.; Trobos, M. Sodium Salicylate Influences the Pseudomonas aeruginosa Biofilm Structure and Susceptibility Towards Silver. Int. J. Mol. Sci. 2021, 22, 1060. https://doi.org/10.3390/ijms22031060
Gerner E, Almqvist S, Thomsen P, Werthén M, Trobos M. Sodium Salicylate Influences the Pseudomonas aeruginosa Biofilm Structure and Susceptibility Towards Silver. International Journal of Molecular Sciences. 2021; 22(3):1060. https://doi.org/10.3390/ijms22031060
Chicago/Turabian StyleGerner, Erik, Sofia Almqvist, Peter Thomsen, Maria Werthén, and Margarita Trobos. 2021. "Sodium Salicylate Influences the Pseudomonas aeruginosa Biofilm Structure and Susceptibility Towards Silver" International Journal of Molecular Sciences 22, no. 3: 1060. https://doi.org/10.3390/ijms22031060
APA StyleGerner, E., Almqvist, S., Thomsen, P., Werthén, M., & Trobos, M. (2021). Sodium Salicylate Influences the Pseudomonas aeruginosa Biofilm Structure and Susceptibility Towards Silver. International Journal of Molecular Sciences, 22(3), 1060. https://doi.org/10.3390/ijms22031060








