The Laccase Gene Family Mediate Multi-Perspective Trade-Offs during Tea Plant (Camellia sinensis) Development and Defense Processes
Abstract
:1. Introduction
2. Results
2.1. Molecular Cloning, Phylogenetic Analysis and Chromosomal Distribution of CsLACs
2.2. Analysis of Conserved Domains, Gene Architecture and Promoter Elements of CsLACs
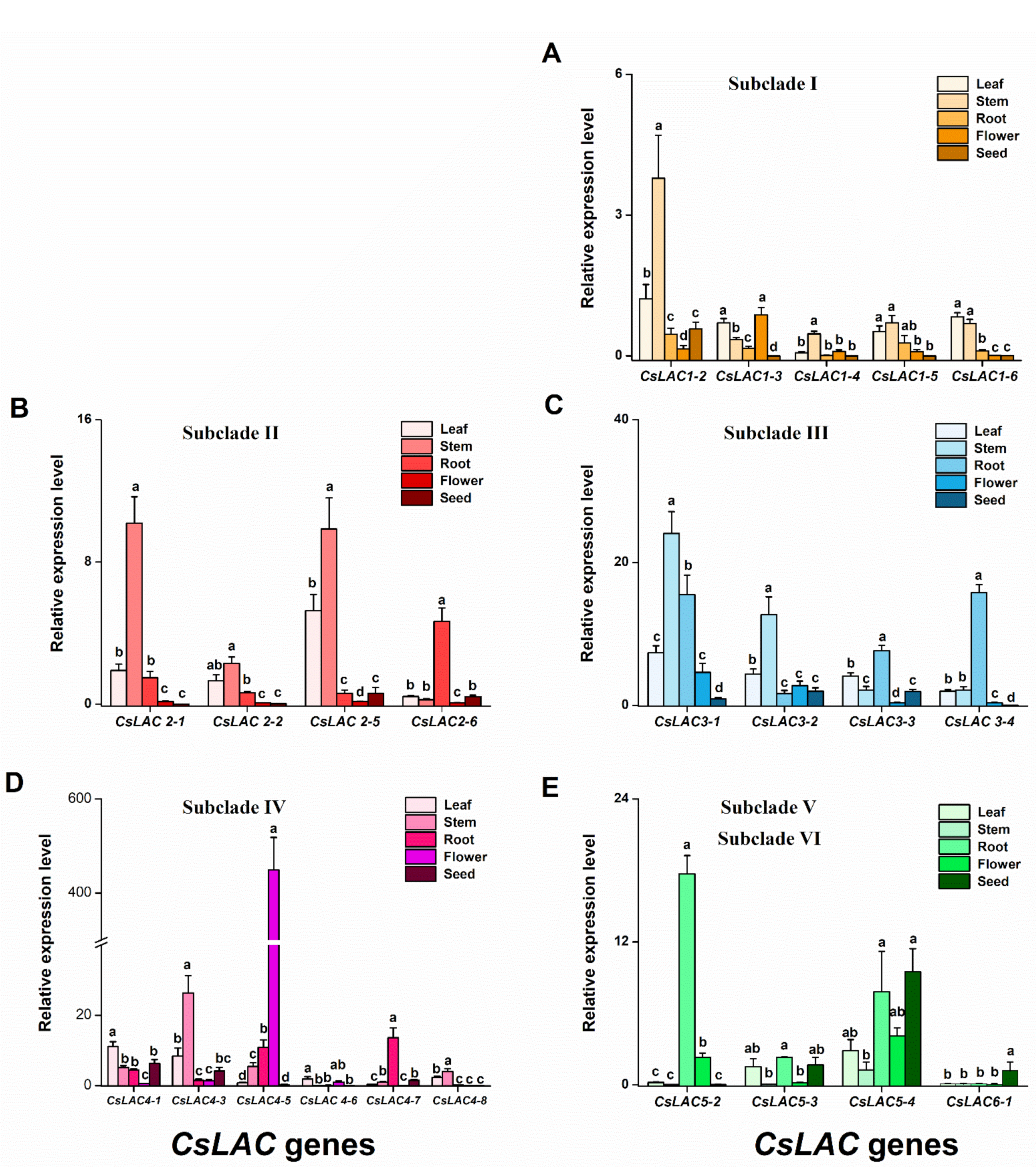
2.3. Expression Patterns of CsLACs during Leaf Development and in Different Tissues
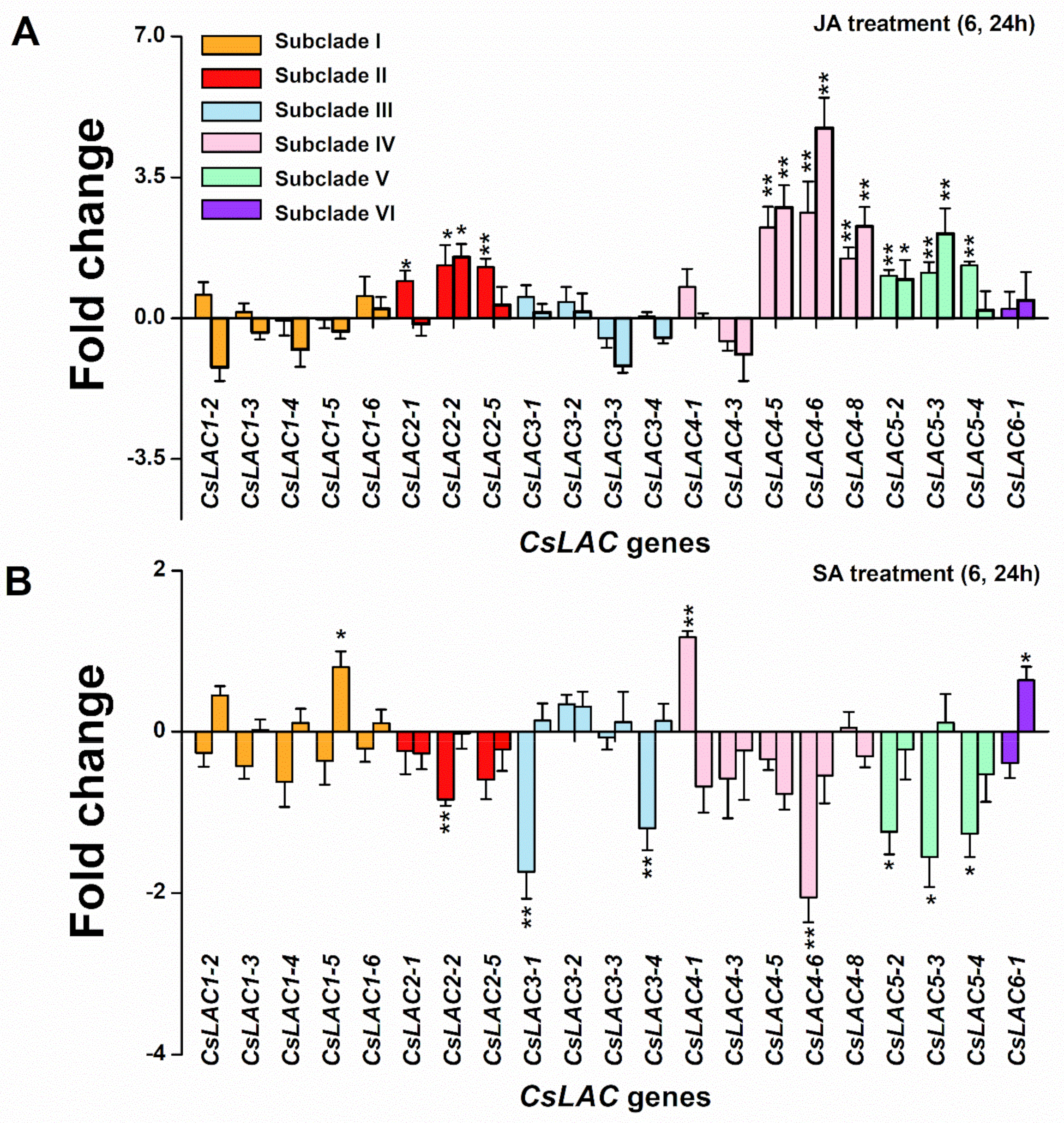
2.4. Expression Profiles of CsLACs in Response to Exogenous Application of JA and SA
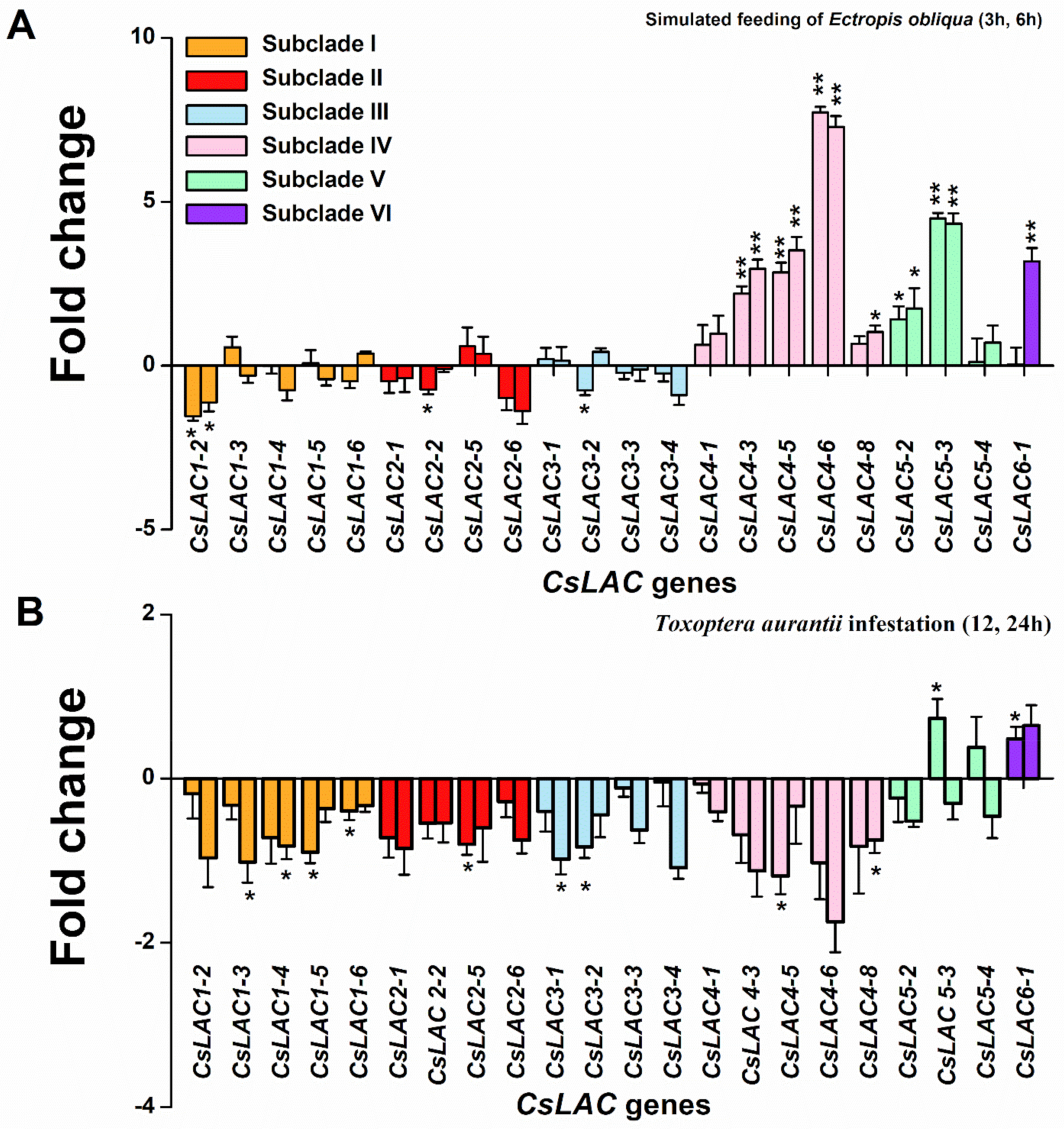
2.5. Expression Profiles of CsLACs in Response to Infestation by Herbivores
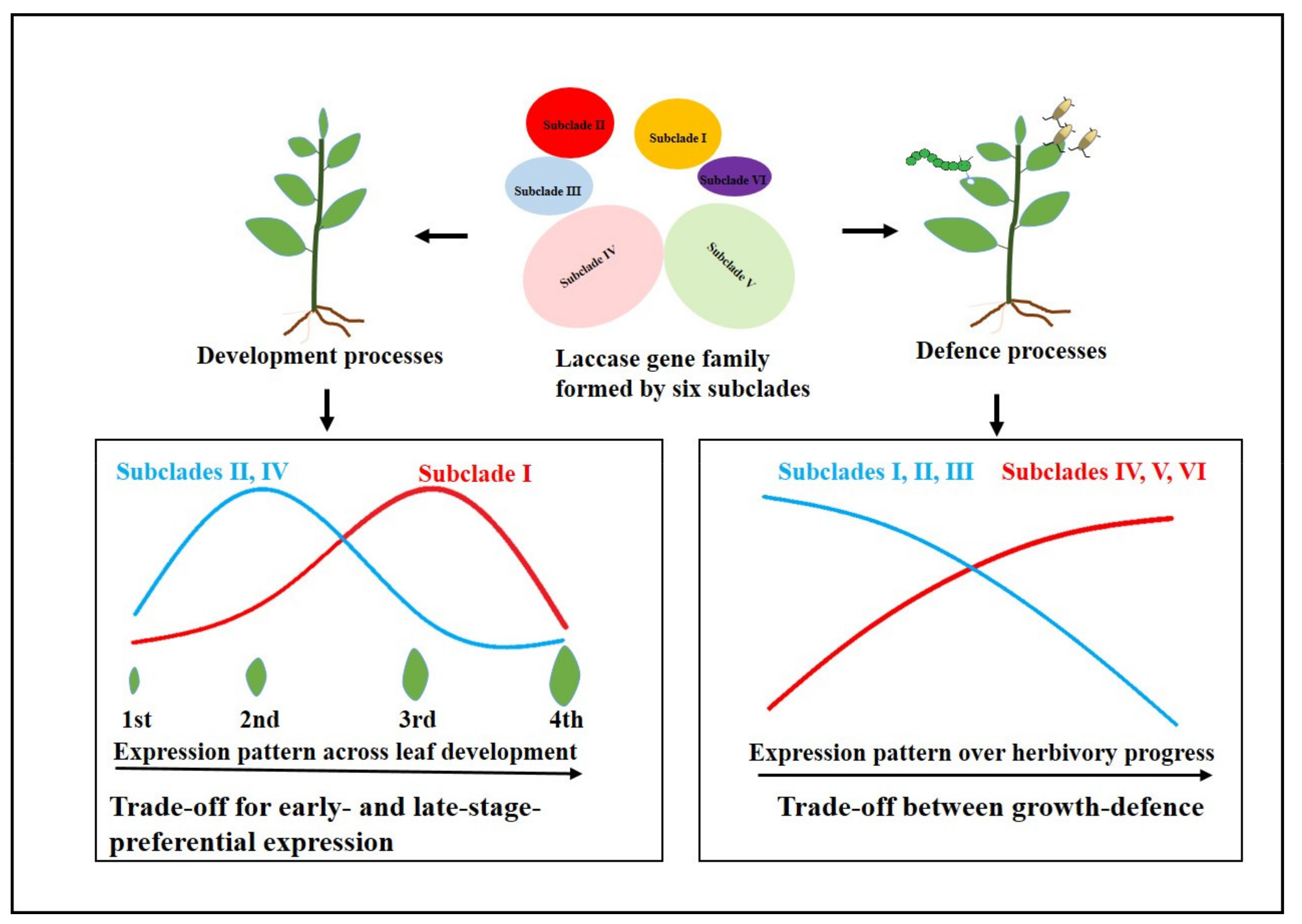
3. Discussion
4. Materials and Methods
4.1. Plants and Insects
4.2. Regurgitant Collection
4.3. Confirmation of the CsLACs Gene Family
4.4. Phylogenetic Analysis and Distribution Pattern of CsLACs
4.5. Analysis of Gene Structure, Conserved Motif and Cis-Elements
4.6. Plant Treatments
4.6.1. Different Tissues and Leaves at Different Stages of Maturity
4.6.2. Simulated Feeding of E. obliqua
4.6.3. T. Aurantii Infestation
4.6.4. JA and SA Treatment
4.7. RNA Sequencing and Data Processing
4.8. qRT-PCR Analysis
4.9. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Erb, M.; Reymond, P. Molecular interactions between plants and insect herbivores. Annu. Rev. Plant Biol. 2019, 70, 527–557. [Google Scholar] [CrossRef] [Green Version]
- Heil, M.; Baldwin, I.T. Fitness costs of induced resistance: Emerging experimental support for a slippery concept. Trends Plant Sci. 2002, 7, 61–67. [Google Scholar] [CrossRef]
- Wang, W.; Zhou, P.; Mo, X.; Hu, L.; Jin, N.; Chen, X.; Yu, Z.; Meng, J.; Erb, M.; Shang, Z.; et al. Induction of defense in cereals by 4-fluorophenoxyacetic acid suppresses insect pest populations and increases crop yields in the field. Proc. Natl. Acad. Sci. USA 2020, 117, 12017–12028. [Google Scholar] [CrossRef]
- Garcia, A.; Martinez, M.; Diaz, I.; Santamaria, M.E. The price of the induced defense against pests: A meta-analysis. Front. Plant Sci. 2021, 11, 615122. [Google Scholar] [CrossRef]
- Figueroa-Macías, J.P.; García, Y.C.; Núñez, M.; Díaz, K.; Olea, A.F.; Espinoza, L. Plant growth-defense trade-offs: Molecular processes leading to physiological changes. Int. J. Mol. Sci. 2021, 22, 693. [Google Scholar] [CrossRef]
- Mot, A.C.; Silaghi-Dumitrescu, R. Laccases: Complex architectures for one-electron oxidations. Biochemistry 2012, 77, 1395–1407. [Google Scholar] [CrossRef]
- Arregui, L.; Ayala, M.; Gómez-Gil, X.; Gutiérrez-Soto, G.; Hernández-Luna, C.E.; Santos, M.H.D.L.; Levin, L.; Rojo-Domínguez, A.; Romero-Martínez, D.; Saparrat, M.C.N.; et al. Laccases: Structure, function, and potential application in water bioremediation. Microb. Cell Factories 2019, 18, 200. [Google Scholar] [CrossRef]
- Turlapati, P.V.; Kim, K.-W.; Davin, L.B.; Lewis, N.G. The laccase multigene family in Arabidopsis thaliana: Towards addressing the mystery of their gene function(s). Planta 2011, 233, 439–470. [Google Scholar] [CrossRef]
- Berthet, S.; Demont-Caulet, N.; Pollet, B.; Bidzinski, P.; Cézard, L.; Le Bris, P.; Borrega, N.; Hervé, J.; Blondet, E.; Balzergue, S.; et al. Disruption of LACCASE4 and 17 results in tissue-specific alterations to lignification of Arabidopsis thaliana stems. Plant Cell 2011, 23, 1124–1137. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Q.; Nakashima, J.; Chen, F.; Yin, Y.; Fu, C.; Yun, J.; Shao, H.; Wang, X.; Wang, Z.-Y.; Dixon, R.A. LACCASE is necessary and nonredundant with PEROXIDASE for lignin polymerization during vascular development in Arabidopsis. Plant Cell 2013, 25, 3976–3987. [Google Scholar] [CrossRef] [Green Version]
- Liu, Q.; Luo, L.; Wang, X.; Shen, Z.; Zheng, L. Comprehensive analysis of rice laccase gene (OsLAC) family and ectopic expression of OsLAC10 enhances tolerance to copper stress in Arabidopsis. Int. J. Mol. Sci. 2017, 18, 209. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Min, L.; Yang, X.; Jin, S.; Zhang, L.; Li, Y.; Ma, Y.; Qi, X.; Li, D.; Liu, H.; et al. Laccase GhLac1 modulates broad-spectrum biotic stress tolerance via manipulating phenylpropanoid pathway and jasmonic acid synthesis. Plant Physiol. 2018, 176, 1808–1823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Wu, L.; Wang, X.; Chen, B.; Zhao, J.; Cui, J.; Li, Z.; Yang, J.; Wu, L.; Wu, J.; et al. The cotton laccase gene GhLAC15 enhances Verticillium wilt resistance via an increase in defence-induced lignification and lignin components in the cell walls of plants. Mol. Plant Pathol. 2019, 20, 309–322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, X.; Li, G.; Ma, C.; Abdullah, M.; Zhang, J.; Zhao, H.; Jin, Q.; Cai, Y.; Lin, Y. Comprehensive genome-wide analysis of the pear (Pyrus bretschneideri) laccase gene (PbLAC) family and functional identification of PbLAC1 involved in lignin biosynthesis. PLoS ONE 2019, 14, e0210892. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pourcel, L.; Routaboul, J.-M.; Kerhoas, L.; Caboche, M.; Lepiniec, L.; Debeaujon, I. TRANSPARENT TESTA10 Encodes a laccase-like enzyme involved in oxidative polymerization of flavonoids in Arabidopsis seed coat. Plant Cell 2005, 17, 2966–2980. [Google Scholar] [CrossRef] [Green Version]
- Cai, X.; Davis, E.J.; Ballif, J.; Liang, M.; Bushman, E.; Haroldsen, V.; Torabinejad, J.; Wu, Y. Mutant identification and characterization of the laccase gene family in Arabidopsis. J. Exp. Bot. 2006, 57, 2563–2569. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.C.; Yu, Y.; Wang, C.Y.; Li, Z.Y.; Liu, Q.; Xu, J.; Liao, J.; Wang, X.; Qu, L.; Chen, F.; et al. Overexpression of microRNA OsmiR397 improves rice yield by increasing grain size and promoting panicle branching. Nat. Biotechnol. 2013, 31, 848–852. [Google Scholar] [CrossRef]
- Soni, N.; Hegde, N.; Dhariwal, A.; Kushalappa, A.C. Role of laccase gene in wheat NILs differing at QTL-Fhb1 for resistance against Fusarium head blight. Plant Sci. 2020, 298, 110574. [Google Scholar] [CrossRef]
- Liang, M.; Haroldsen, V.; Cai, X.; Wu, Y. Expression of a putative laccase gene, ZmLAC1, in maize primary roots under stress. Plant Cell Environ. 2006, 29, 746–753. [Google Scholar] [CrossRef]
- Liu, M.; Dong, H.; Wang, M.; Liu, Q. Evolutionary divergence of function and expression of laccase genes in plants. J. Genet. 2020, 99, 23. [Google Scholar] [CrossRef]
- Chen, J.D.; Zheng, C.; Ma, J.Q.; Jiang, C.K.; Ercisli, S.; Yao, M.Z.; Chen, L. The chromosome-scale genome reveals the evolution and diversification after the recent tetraploidization event in tea plant. Hortic. Res. 2020, 7, 63. [Google Scholar] [CrossRef] [PubMed]
- Xia, E.; Tong, W.; Hou, Y.; An, Y.; Chen, L.; Wu, Q.; Liu, Y.; Yu, J.; Li, F.; Li, R.; et al. The reference genome of tea plant and resequencing of 81 diverse accessions provide insights into its genome evolution and adaptation. Mol. Plant 2020, 13, 1013–1026. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Yu, Z.; Liu, X.; Zeng, L.; Cheng, S.; Li, J.; Tang, J.; Yang, Z. Effect of major tea insect attack on formation of quality-related nonvolatile specialized metabolites in tea (Camellia sinensis) leaves. J. Agric. Food Chem. 2019, 67, 6716–6724. [Google Scholar] [CrossRef]
- Yang, H.; Wang, Y.; Li, L.; Li, F.; He, Y.; Wu, J.; Wei, C. Transcriptomic and phytochemical analyses reveal root-mediated resource-based defense response to leaf herbivory by Ectropis oblique in tea plant (Camellia sinensis). J. Agric. Food Chem. 2019, 67, 5465–5476. [Google Scholar] [CrossRef]
- War, A.R.; Paulraj, M.G.; Ahmad, T.; Buhroo, A.A.; Hussain, B.; Ignacimuthu, S.; Sharma, H.C. Mechanisms of plant defense against insect herbivores. Plant Signal. Behav. 2012, 7, 1306–1320. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Feng, J.; Jia, W.; Fan, P.; Bao, H.; Li, S.; Li, Y. Genome-wide identification of Sorghum bicolor laccases reveals potential targets for lignin modification. Front. Plant Sci. 2017, 8, 714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCaig, B.C.; Meagher, R.B.; Dean, J.F.D. Gene structure and molecular analysis of the laccase-like multicopper oxidase (LMCO) gene family in Arabidopsis thaliana. Planta 2005, 221, 619–636. [Google Scholar] [CrossRef]
- Balasubramanian, V.K.; Rai, K.M.; Thu, S.W.; Hii, M.M.; Mendu, V. Genome-wide identification of multifunctional laccase gene family in cotton (Gossypium spp.); expression and biochemical analysis during fiber development. Sci. Rep. 2016, 6, 34309. [Google Scholar] [CrossRef] [Green Version]
- Xu, X.; Zhou, Y.; Wang, B.; Ding, L.; Wang, Y.; Luo, L.; Zhang, Y.; Kong, W. Genome-wide identification and characterization of laccase gene family in Citrus sinensis. Gene 2018, 689, 114–123. [Google Scholar] [CrossRef]
- Wang, Q.; Li, G.; Zheng, K.; Zhu, X.; Ma, J.; Wang, D.; Tang, K.; Feng, X.; Leng, J.; Yu, H.; et al. The soybean laccase gene family: Evolution and possible roles in plant defense and stem strength selection. Genes 2019, 10, 701. [Google Scholar] [CrossRef] [Green Version]
- Simões, M.S.; Carvalho, G.G.; Ferreira, S.; Hernandes-Lopes, J.; De Setta, N.; Cesarino, I. Genome-wide characterization of the laccase gene family in Setaria viridis reveals members potentially involved in lignification. Planta 2020, 251, 46. [Google Scholar] [CrossRef]
- Cannon, S.B.; Mitra, A.; Baumgarten, A.; Young, N.D.; May, G. The roles of segmental and tandem gene duplication in the evolution of large gene families in Arabidopsis thaliana. BMC Plant Biol. 2004, 4, 10. [Google Scholar] [CrossRef] [Green Version]
- Cesarino, I.; Araújo, P.; Mayer, J.L.S.; Vicentini, R.; Berthet, S.; Demedts, B.; Vanholme, B.; Boerjan, W.; Mazzafera, P. Expression of SofLAC, a new laccase in sugarcane, restores lignin content but not S:G ratio of Arabidopsis lac17 mutant. J. Exp. Bot. 2013, 64, 1769–1781. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sterjiades, R.; Dean, J.; Gamble, G.; Himmelsbach, D.; Eriksson, K.-E. Extracellular laccases and peroxidases from sycamore maple (Acer pseudoplatanus) cell-suspension cultures. Planta 1993, 190, 75–87. [Google Scholar] [CrossRef]
- Hoffmann, N.; Benske, A.; Betz, H.; Schuetz, M.; Samuels, A.L. Laccases and peroxidases co-localize in lignified secondary cell walls throughout stem development. Plant Physiol. 2020, 184, 806–822. [Google Scholar] [CrossRef] [PubMed]
- Bürger, M.; Chory, J. Stressed out about hormones: How plants orchestrate immunity. Cell Host Microbe 2019, 26, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Liu, M.; Erb, M.; Glauser, G.; Zhang, J.; Li, X.; Sun, X. Indole primes defence signalling and increases herbivore resistance in tea plants. Plant Cell Environ. 2021, 44, 1165–1177. [Google Scholar] [CrossRef]
- Ullah, C.; Tsai, C.; Unsicker, S.; Xue, L.; Reichelt, M.; Gershenzon, J.; Hammerbacher, A. Salicylic acid activates poplar defense against the biotrophic rust fungus Melampsora larici-populina via increased biosynthesis of catechin and proanthocyanidins. New Phytol. 2019, 221, 960–975. [Google Scholar] [CrossRef] [Green Version]
- Fu, W.; Jin, G.; Jimenez-Aleman, G.H.; Wang, X.; Song, J.; Li, S.; Lou, Y.; Li, R. The jasmonic acid-amino acid conjugates JA-Val and JA-Leu are involved in rice resistance to herbivores. Plant Cell Environ. 2021. [Google Scholar] [CrossRef] [PubMed]
- Constabel, C.P.; Barbehenn, R. Defensive roles of polyphenol oxidase in plants. In Induced Plant Resistance to Herbivory; Schaller, A., Ed.; Springer: Dordrecht, The Netherlands, 2008; pp. 253–270. [Google Scholar]
- Zhang, J.; Zhang, X.; Ye, M.; Li, X.W.; Lin, S.B.; Sun, X.L. The jasmonic acid pathway positively regulates the polyphenol oxidase-based defense against tea geometrid caterpillars in the tea plant (Camellia sinensis). J. Chem. Ecol. 2020, 46, 308–316. [Google Scholar] [CrossRef]
- Zhang, J.; Sun, X. Recent advances in polyphenol oxidase-mediated plant stress responses. Phytochemistry 2021, 181, 112588. [Google Scholar] [CrossRef]
- Glazebrook, J. Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu. Rev. Phytopathol. 2005, 43, 205–227. [Google Scholar] [CrossRef] [PubMed]
- Niu, Z.; Li, G.; Hu, H.; Lv, J.; Zheng, Q.; Liu, J.; Wan, D. A gene that underwent adaptive evolution, LAC2 (LACCASE), in Populus euphratica improves drought tolerance by improving water transport capacity. Hortic. Res. 2021, 8, 88. [Google Scholar] [CrossRef] [PubMed]
- Lan, Z.; Krosse, S.; Achard, P.; van Dam, N.; Bede, J.C. DELLA proteins modulateArabidopsisdefences induced in response to caterpillar herbivory. J. Exp. Bot. 2014, 65, 571–583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, R.; Zhang, J.; Li, J.; Zhou, G.; Wang, Q.; Bian, W.; Erb, M.; Lou, Y. Prioritizing plant defence over growth through WRKY regulation facilitates infestation by non-target herbivores. eLife 2015, 4, e04805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Luo, T.; Wang, W.; Cao, T.; Li, R.; Lou, Y. Silencing OsSLR1enhances the resistance of rice to the brown planthopper Nilaparvata lugens. Plant Cell Environ. 2017, 40, 2147–2159. [Google Scholar] [CrossRef]
- Panda, S.; Kazachkova, Y.; Aharoni, A. Catch-22 in specialized metabolism: Balancing defense and growth. J. Exp. Bot. 2021, 72, 6027–6041. [Google Scholar] [CrossRef]
- Chen, D.; Li, J.; Jiao, F.; Wang, Q.; Li, J.; Pei, Y.; Zhao, M.; Song, X.; Guo, X. ZmACY-1 antagonistically regulates growth and stress responses in Nicotiana benthamiana. Front. Plant Sci. 2021, 12, 593001. [Google Scholar] [CrossRef]
- Cai, W.; Yang, S.; Wu, R.; Cao, J.; Shen, L.; Guan, D.; Shuilin, H. Pepper NAC-type transcription factor NAC2c balances the trade-off between growth and defense responses. Plant Physiol. 2021, 186, 2169–2189. [Google Scholar] [CrossRef]
- Yang, Z.W.; Duan, X.N.; Jin, S.; Li, X.W.; Chen, Z.M.; Ren, B.Z.; Sun, X.L. Regurgitant derived from the tea geometrid Ectropis obliqua suppresses wound-induced polyphenol oxidases activity in tea plants. J. Chem. Ecol. 2013, 39, 744–751. [Google Scholar] [CrossRef]
- Li, X.; Zhang, J.; Lin, S.; Xing, Y.; Zhang, X.; Ye, M.; Chang, Y.; Guo, H.; Sun, X. (+)-Catechin, epicatechin and epigallocatechin are important inducible defensive compounds against Ectropis grisescens in tea plants. Plant Cell Environ. 2021. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, 884–890. [Google Scholar] [CrossRef] [PubMed]
- Patro, R.; Duggal, G.; Love, M.I.; Irizarry, R.A.; Kingsford, C. Salmon provides fast and bias-aware quantification of transcript expression. Nat. Methods 2017, 14, 417–419. [Google Scholar] [CrossRef] [Green Version]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
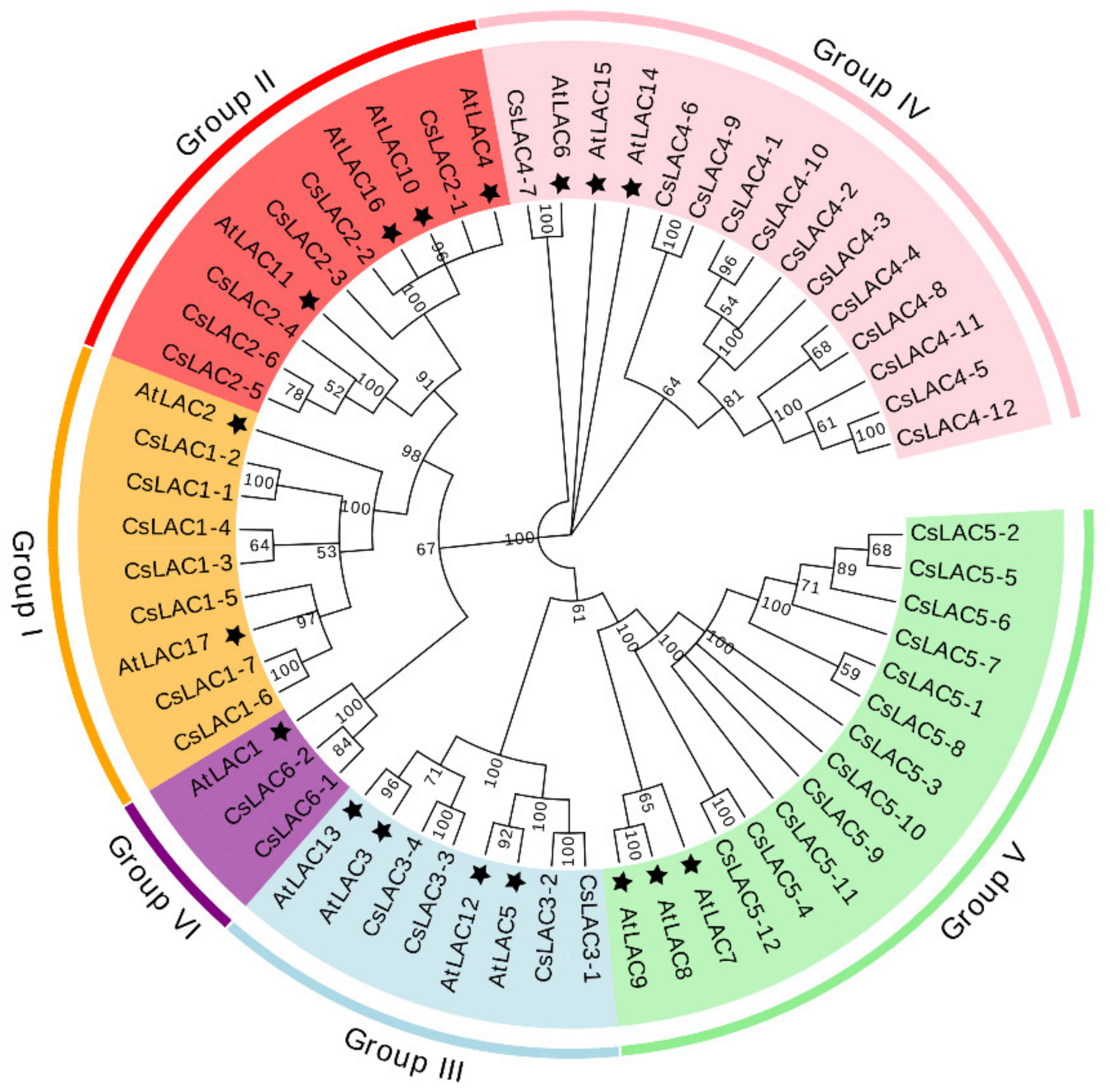
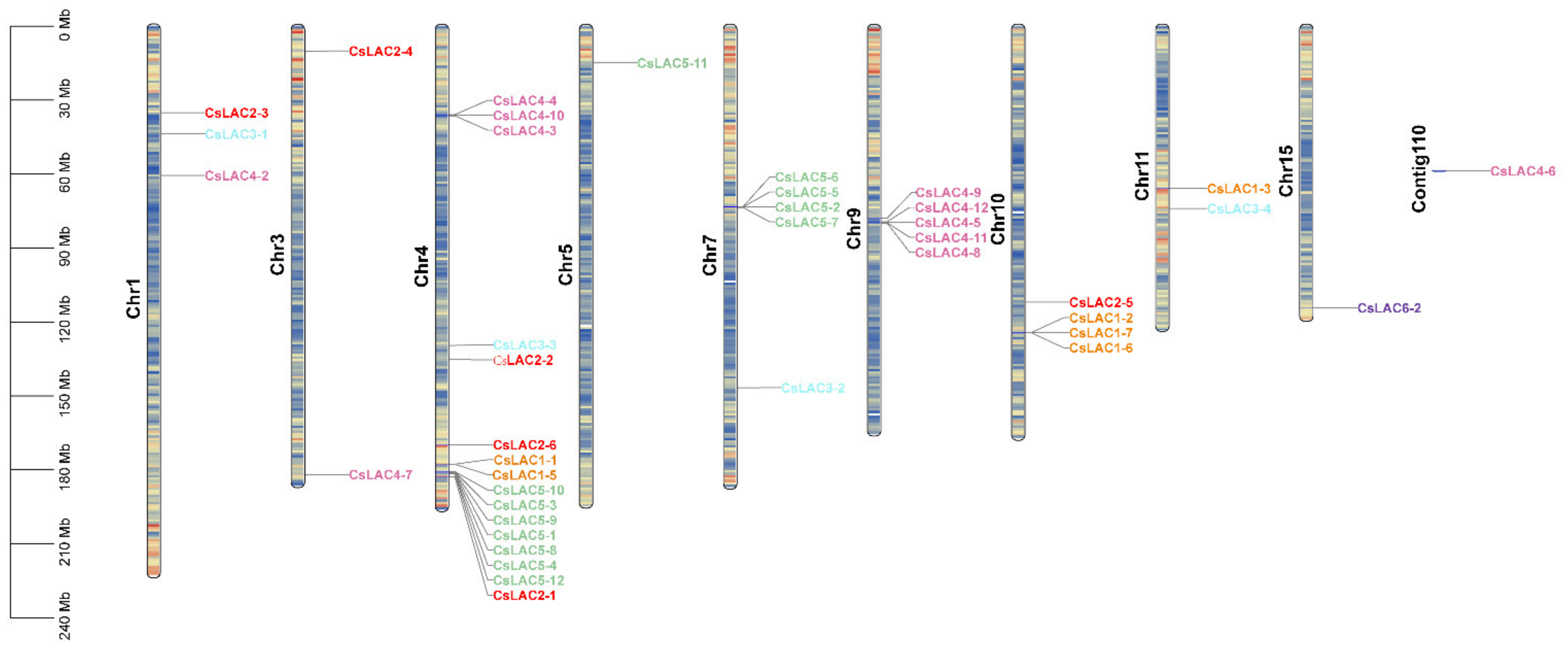
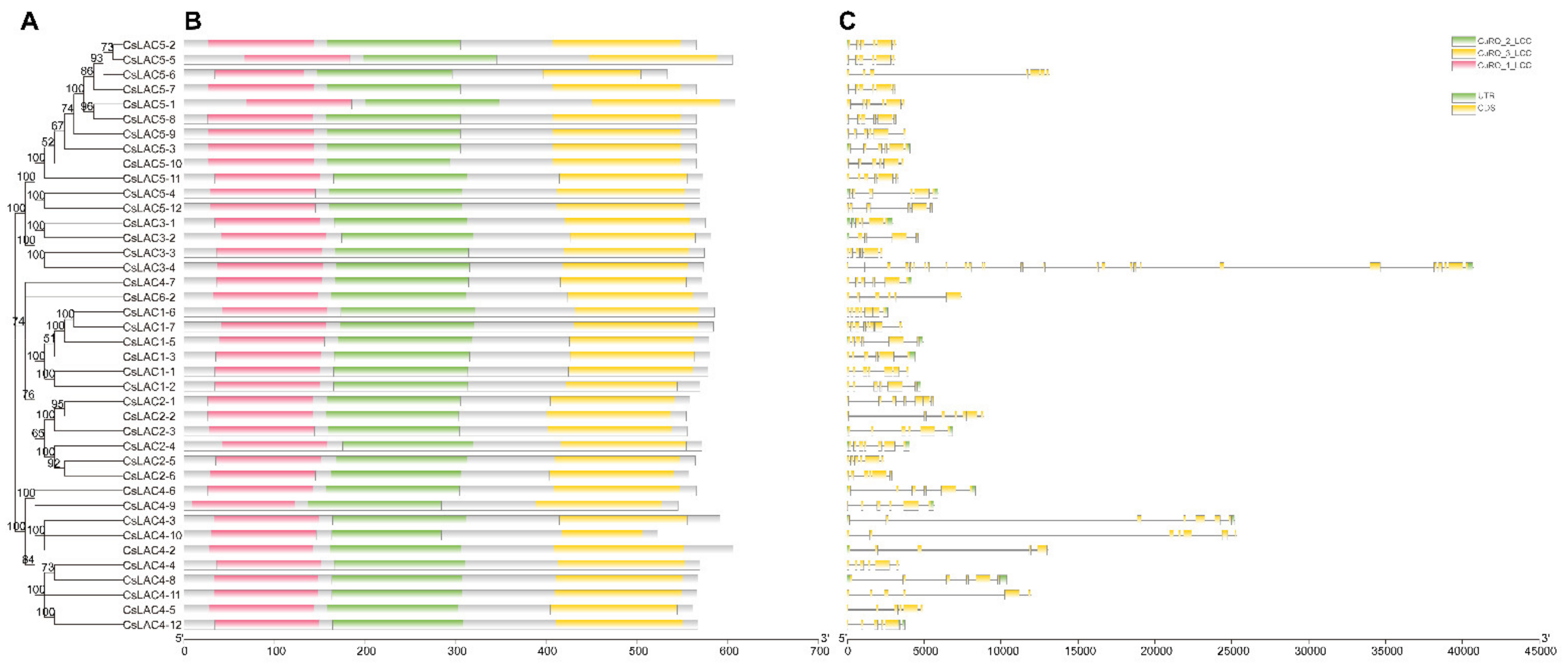
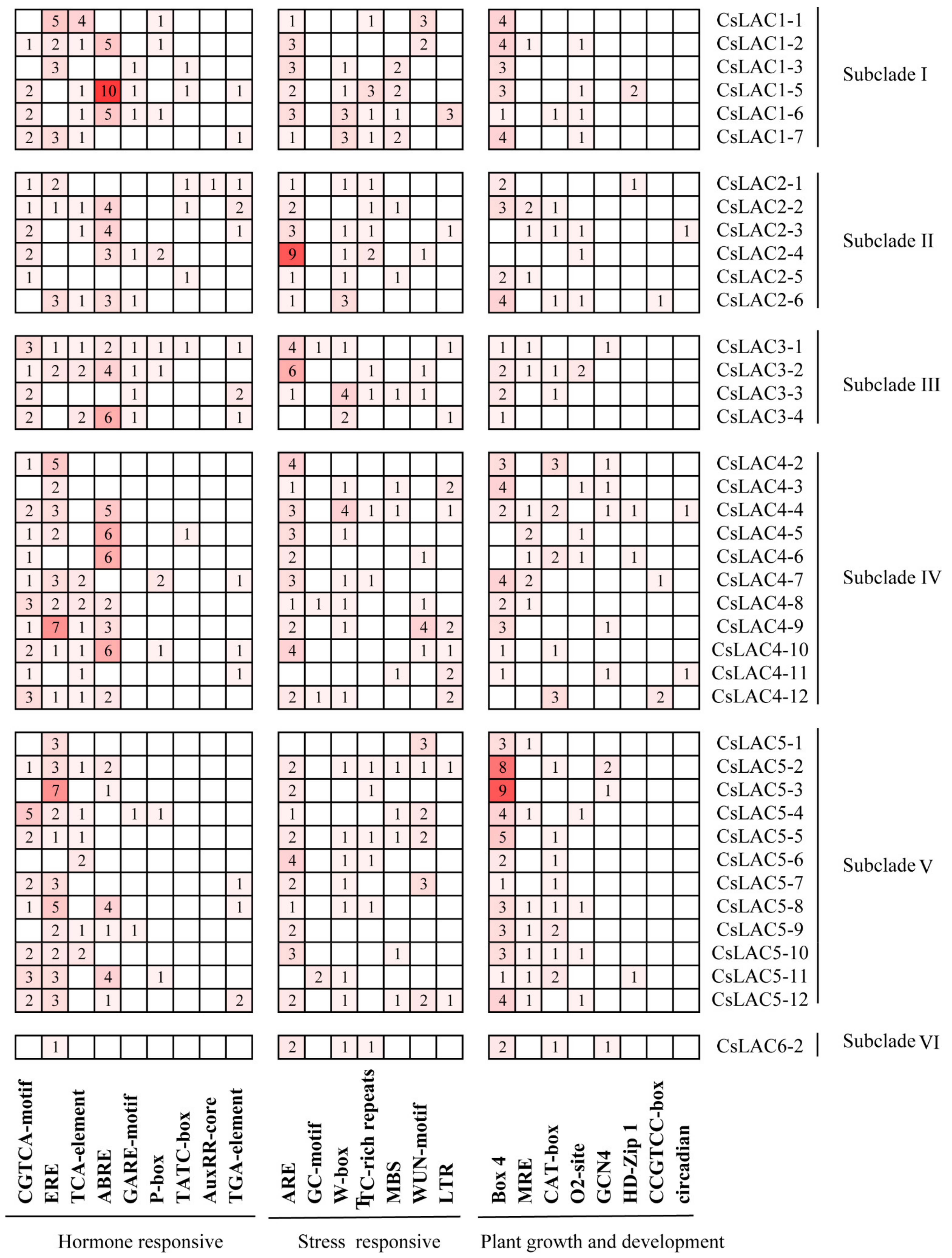

| Name | Accession Number | Intron | Extron | ORF Size (bp) | Protein | Sub-Cellular Localization | ||
|---|---|---|---|---|---|---|---|---|
| Amino Acid Residues | MW (Da) | pI | ||||||
| CsLAC1-1 | CSS0050170 | 6 | 7 | 1734 | 578 | 64.230 | 9.54 | Secretory |
| CsLAC1-2 | CSS0030617 | 5 | 6 | 1710 | 569 | 62.934 | 9.47 | Secretory |
| CsLAC1-3 | CSS0040822 | 5 | 6 | 1740 | 580 | 63.814 | 9.30 | Secretory |
| CsLAC1-4 | TEA004738.1 | 5 | 6 | 1734 | 578 | 63.801 | 8.81 | Secretory |
| CsLAC1-5 | CSS0032027 | 5 | 6 | 1737 | 579 | 64.134 | 8.90 | Secretory |
| CsLAC1-6 | CSS0029337 | 6 | 7 | 1755 | 585 | 64.656 | 9.14 | Secretory |
| CsLAC1-7 | CSS0045289 | 7 | 8 | 1755 | 584 | 64.398 | 9.02 | Secretory |
| CsLAC2-1 | CSS0030904 | 6 | 7 | 1674 | 558 | 60.821 | 9.34 | Secretory |
| CsLAC2-2 | CSS0014129 | 6 | 7 | 1665 | 554 | 61.137 | 8.93 | Secretory |
| CsLAC2-3 | CSS0015036 | 5 | 6 | 1668 | 555 | 61.106 | 8.34 | Secretory |
| CsLAC2-4 | CSS0047304 | 6 | 7 | 1713 | 571 | 63.545 | 8.99 | Secretory |
| CsLAC2-5 | CSS0001101 | 5 | 6 | 1692 | 564 | 63.073 | 8.25 | Secretory |
| CsLAC2-6 | CSS0005481 | 5 | 6 | 1671 | 557 | 62.160 | 9.12 | Secretory |
| CsLAC3-1 | CSS0035921 | 5 | 6 | 1728 | 575 | 62.560 | 6.13 | Secretory |
| CsLAC3-2 | CSS0017559 | 4 | 5 | 1746 | 581 | 63.949 | 9.18 | Secretory |
| CsLAC3-3 | CSS0041657 | 5 | 6 | 1722 | 574 | 63.758 | 6.30 | Secretory |
| CsLAC3-4 | CSS0009670 | 5 | 6 | 1719 | 573 | 64.121 | 9.05 | Secretory |
| CsLAC4-1 | NOT FOUND | - | - | 1335 | 444 | 49.38 | 5.51 | Non-secretory |
| CsLAC4-2 | CSS0038356 | 4 | 5 | 1821 | 606 | 67.629 | 5.26 | Secretory |
| CsLAC4-3 | CSS0043663 | 6 | 7 | 1773 | 591 | 66.744 | 5.27 | Secretory |
| CsLAC4-4 | CSS0043918 | 5 | 6 | 1707 | 569 | 63.872 | 8.95 | Secretory |
| CsLAC4-5 | CSS0007135 | 5 | 6 | 1683 | 561 | 62.828 | 5.16 | Secretory |
| CsLAC4-6 | CSS0022921 | 5 | 6 | 1767 | 589 | 66.331 | 7.62 | Non-secretory |
| CsLAC4-7 | CSS0004662 | 5 | 6 | 1713 | 571 | 63.469 | 7.22 | Secretory |
| CsLAC4-8 | CSS0047533 | 3 | 4 | 1701 | 567 | 62.980 | 8.52 | Secretory |
| CsLAC4-9 | CSS0036236 | 5 | 6 | 1638 | 545 | 61.148 | 6.09 | Non-secretory |
| CsLAC4-10 | CSS0025249 | 6 | 7 | 1544 | 522 | 59.050 | 5.33 | Secretory |
| CsLAC4-11 | CSS0039645 | 5 | 6 | 1701 | 566 | 63.180 | 8.30 | Secretory |
| CsLAC4-12 | CSS0046037 | 5 | 6 | 1425 | 474 | 53.109 | 5.05 | Non-secretory |
| CsLAC5-1 | CSS0013370 | 5 | 6 | 1824 | 608 | 67.871 | 6.53 | Non-secretory |
| CsLAC5-2 | CSS0010920 | 5 | 6 | 1695 | 565 | 62.439 | 6.33 | Secretory |
| CsLAC5-3 | CSS0010479 | 5 | 6 | 1698 | 565 | 62.148 | 6.18 | Secretory |
| CsLAC5-4 | CSS0020412 | 5 | 6 | 1707 | 569 | 62.319 | 7.34 | Secretory |
| CsLAC5-5 | CSS0045107 | 5 | 6 | 1698 | 565 | 62.463 | 6.33 | Secretory |
| CsLAC5-6 | CSS0048878 | 6 | 7 | 1602 | 533 | 58.490 | 6.14 | Secretory |
| CsLAC5-7 | CSS0008882 | 5 | 6 | 1695 | 564 | 62.714 | 6.08 | Secretory |
| CsLAC5-8 | CSS0010391 | 5 | 6 | 1701 | 566 | 62.778 | 6.59 | Secretory |
| CsLAC5-9 | CSS0030703 | 5 | 6 | 1698 | 565 | 62.411 | 6.88 | Secretory |
| CsLAC5-10 | CSS0044116 | 5 | 6 | 1698 | 565 | 62.360 | 8.31 | Secretory |
| CsLAC5-11 | CSS0013475 | 5 | 6 | 1719 | 572 | 63.498 | 7.07 | Secretory |
| CsLAC5-12 | CSS0023848 | 5 | 6 | 1710 | 569 | 62.247 | 6.71 | Secretory |
| CsLAC6-1 | TEA021330 | 4 | 5 | 1737 | 579 | 64.576 | 6.82 | Secretory |
| CsLAC6-2 | CSS0019151 | 5 | 6 | 1725 | 574 | 63.731 | 6.36 | Secretory |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, Y.; Xing, Y.; Liu, F.; Zhang, X.; Li, X.; Zhang, J.; Sun, X. The Laccase Gene Family Mediate Multi-Perspective Trade-Offs during Tea Plant (Camellia sinensis) Development and Defense Processes. Int. J. Mol. Sci. 2021, 22, 12554. https://doi.org/10.3390/ijms222212554
Yu Y, Xing Y, Liu F, Zhang X, Li X, Zhang J, Sun X. The Laccase Gene Family Mediate Multi-Perspective Trade-Offs during Tea Plant (Camellia sinensis) Development and Defense Processes. International Journal of Molecular Sciences. 2021; 22(22):12554. https://doi.org/10.3390/ijms222212554
Chicago/Turabian StyleYu, Yongchen, Yuxian Xing, Fengjing Liu, Xin Zhang, Xiwang Li, Jin Zhang, and Xiaoling Sun. 2021. "The Laccase Gene Family Mediate Multi-Perspective Trade-Offs during Tea Plant (Camellia sinensis) Development and Defense Processes" International Journal of Molecular Sciences 22, no. 22: 12554. https://doi.org/10.3390/ijms222212554
APA StyleYu, Y., Xing, Y., Liu, F., Zhang, X., Li, X., Zhang, J., & Sun, X. (2021). The Laccase Gene Family Mediate Multi-Perspective Trade-Offs during Tea Plant (Camellia sinensis) Development and Defense Processes. International Journal of Molecular Sciences, 22(22), 12554. https://doi.org/10.3390/ijms222212554






