Hygiene Hypothesis as the Etiology of Kawasaki Disease: Dysregulation of Early B Cell Development
Abstract
1. Clinical Characteristics
2. Etiology of KD
2.1. KD Cannot Be Explained by the Infection Model
2.2. KD Cannot Be Explained by the Autoimmunity Model
2.3. KD Can Be Explained by the Hygiene Hypothesis
3. Epidemiological Evidence for the Hygiene Hypothesis as the Pathogenesis of KD
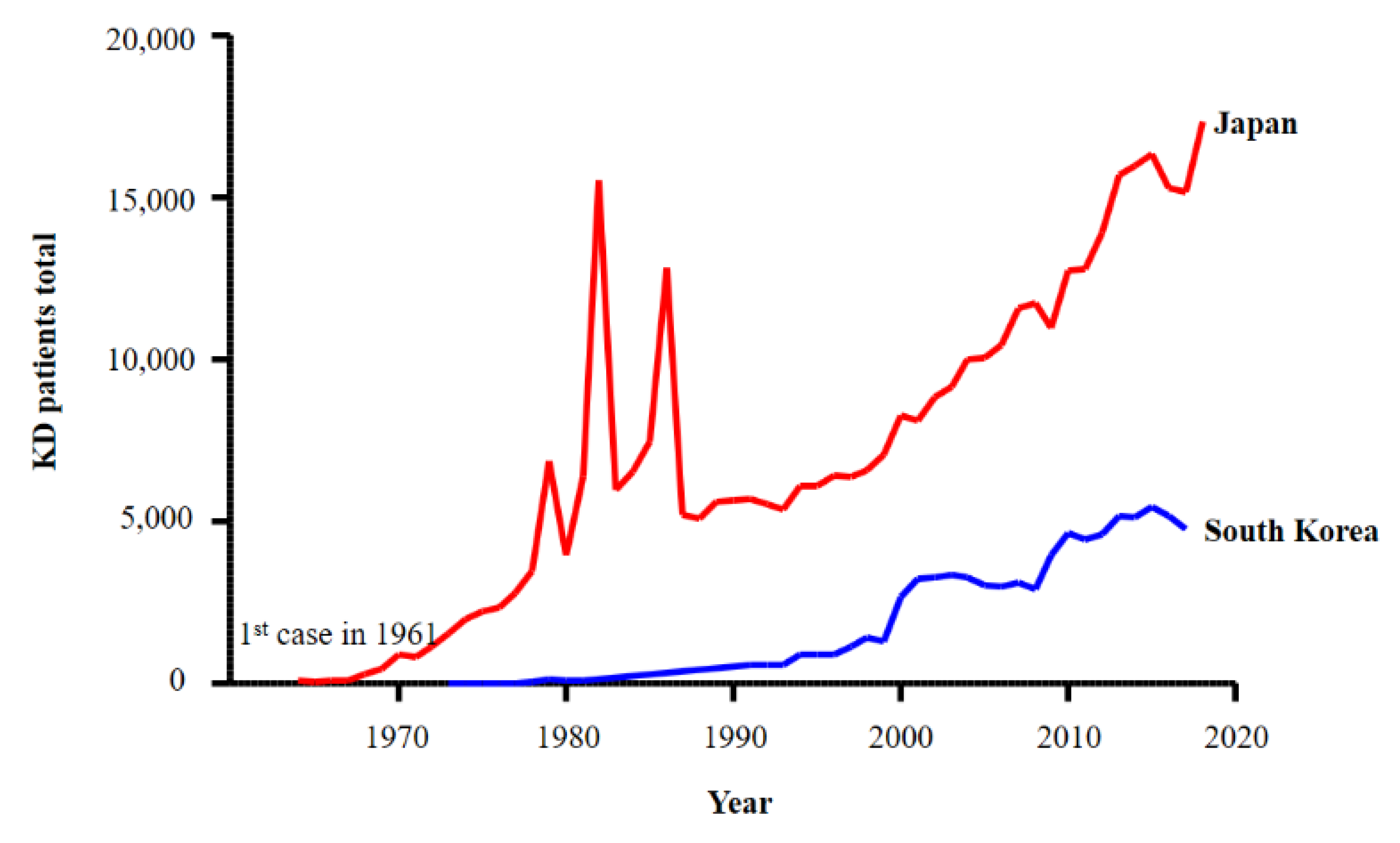
3.1. The Incidence of KD in East Asia Has Been Increasing Since the First Case in 1961
3.2. Improved Socioeconomic Environments Increase the Incidence of KD
3.3. Vaccination Protects against KD
| Factor | Effect on KD | References |
|---|---|---|
| Risk | [44] |
| Risk | [42,44,45,46] |
| Risk | [32] |
| Protective | [49] |
| Protective | [52,53,54] |
3.4. Breastfeeding Protects against KD
4. Insufficient B Cell Immunity Is Crucial for the Development of KD
4.1. The Peak Incidence of KD Overlaps with the Lowest Level of B Cell Immunity
4.2. KD Occurs in Children and Adults with Primary and Secondary Immunodeficiencies
4.3. Patients with KD Have a High Risk for Allergic Diseases and Vice Versa
5. Immunological Evidence from Patients with KD Demonstrates that B Cell Immunity Is Crucial for the Etiopathogenesis of KD
5.1. Severe Inflammatory Responses in Patients with KD
5.2. Low IgG Levels in Patients with KD
5.3. High IgE and Eosinophil Levels in Patients with KD
6. Efficacy of IVIG Treatment Demonstrates That B Cell Development and Activation Are Crucial for the Pathogenesis of KD
6.1. Lower IgG Levels in Patients with KD Correlate with Worse Clinical Outcomes, and IVIG Is the Standard Therapy for KD
6.2. The Therapeutic Effect of IVIG Is Not Likely Due to Passive Protection or Anti-Inflammatory Effects
6.3. The Therapeutic Effect of IVIG Is Likely Due to the Activation of Endogenous B Cell Development and Function
7. Genetic Evidence Indicates That Defects in Early B Cell Development May Be Critical for the Etiopathogenesis of KD
7.1. Genetic Susceptibility Affects the Incidence of KD
7.2. KD Susceptibility Genes Are Involved in Early B Cell Development and Function
7.3. Reduced Expression of Risk Alleles of BLK and BCL2L11 Genes in KD Suggests That the Development of KD Is Due to Defects or Dysregulation of Early B Cell Development
7.4. Male-Dominant Incidence of KD May Be Due to Male-Specific Susceptibility through the FCGR2A Gene
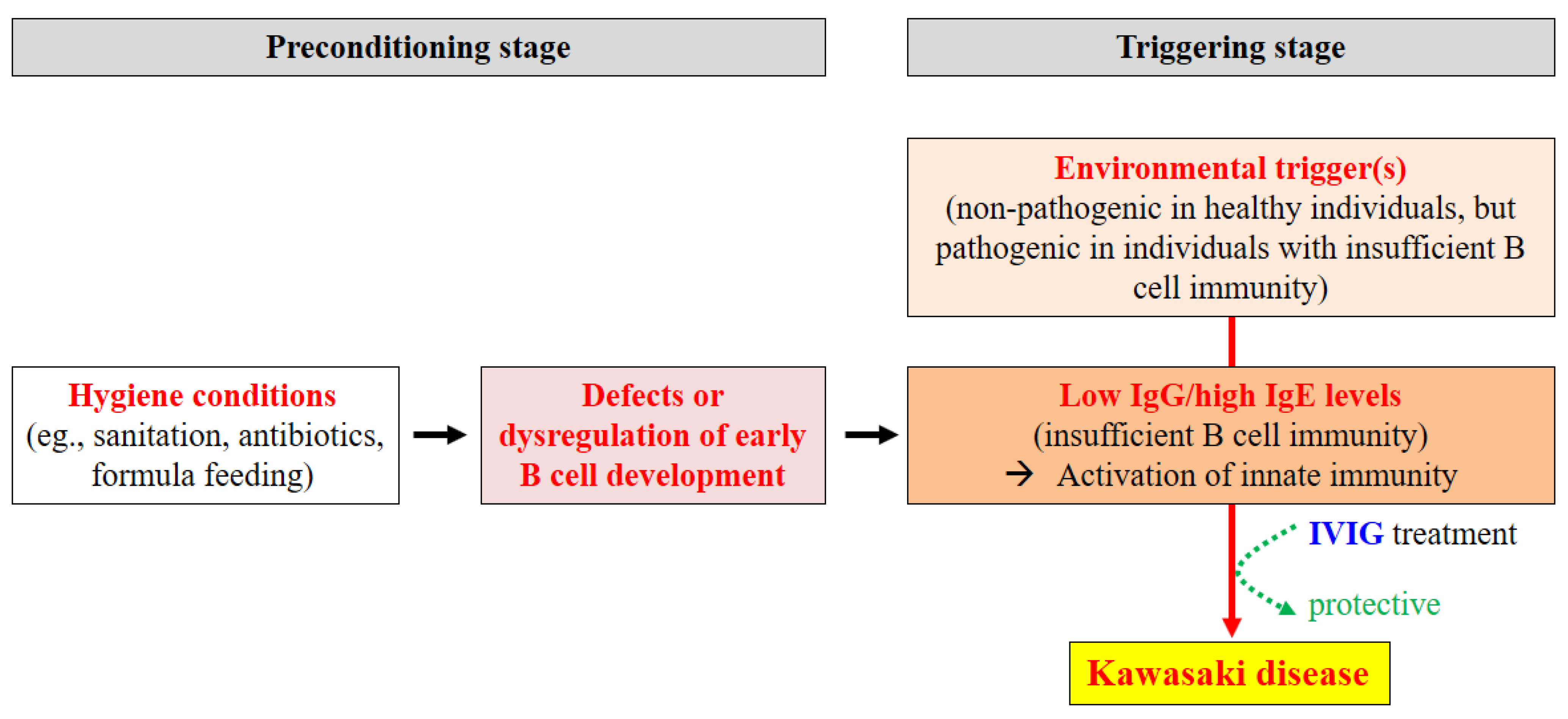
8. Conclusions and Proposed Mechanism Underlying the Etiopathogenesis of KD
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Makino, N.; Nakamura, Y.; Yashiro, M.; Sano, T.; Ae, R.; Kosami, K.; Kojo, T.; Aoyama, Y.; Kotani, K.; Yanagawa, H. Epidemiological observations of Kawasaki disease in Japan, 2013–2014. Pediatr. Int. 2018, 60, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, T. Acute febrile mucocutaneous syndrome with lymphoid involvement with specific desquamation of the fingers and toes in children. Arerugi 1967, 16, 178–222, Note: An English translation was published in Pediat. Infect. Dis. J. 2002, 21, 1–38.. [Google Scholar]
- Kato, H.; Koike, S.; Yamamoto, M.; Ito, Y.; Yano, E. Coronary aneurysms in infants and young children with acute febrile mucocutaneous lymph node syndrome. J. Pediatr. 1975, 86, 892–898. [Google Scholar] [CrossRef]
- Newburger, J.W.; Takahashi, M.; Burns, J.C.; Beiser, A.S.; Chung, K.J.; Duffy, C.E.; Glode, M.P.; Mason, W.H.; Reddy, V.; Sanders, S.P.; et al. The treatment of Kawasaki syndrome with intravenous gamma globulin. N. Engl. J. Med. 1986, 315, 341–347. [Google Scholar] [CrossRef]
- Durongpisitkul, K.; Gururaj, V.J.; Park, J.M.; Martin, C.F. The prevention of coronary artery aneurysm in Kawasaki disease: A meta-analysis on the efficacy of aspirin and immunoglobulin treatment. Pediatrics 1995, 96, 1057–1061. [Google Scholar]
- Manlhiot, C.; Mueller, B.; O’Shea, S.; Majeed, H.; Bernknopf, B.; Labelle, M.; Westcott, K.V.; Bai, H.; Chahal, N.; Birken, C.S.; et al. Environmental epidemiology of Kawasaki disease: Linking disease etiology, pathogenesis and global distribution. PLoS ONE 2018, 13, e0191087. [Google Scholar] [CrossRef]
- Rowley, A.H. Kawasaki disease: Novel insights into etiology and genetic susceptibility. Annu. Rev. Med. 2011, 62, 69–77. [Google Scholar] [CrossRef]
- Satou, G.M.; Giamelli, J.; Gewitz, M.H. Kawasaki disease: Diagnosis, management, and long-term implications. Cardiol. Rev. 2007, 15, 163–169. [Google Scholar] [CrossRef]
- Rowley, A.H.; Baker, S.C.; Orenstein, J.M.; Shulman, S.T. Searching for the cause of Kawasaki disease--cytoplasmic inclusion bodies provide new insight. Nat. Rev. Microbiol. 2008, 6, 394–401. [Google Scholar] [CrossRef]
- Kawasaki, T.; Kosaki, F.; Okawa, S.; Shigematsu, I.; Yanagawa, H. A new infantile acute febrile mucocutaneous lymph node syndrome (MLNS) prevailing in Japan. Pediatrics 1974, 54, 271–276. [Google Scholar] [PubMed]
- Hara, T.; Nakashima, Y.; Sakai, Y.; Nishio, H.; Motomura, Y.; Yamasaki, S. Kawasaki disease: A matter of innate immunity. Clin. Exp. Immunol. 2016, 186, 134–143. [Google Scholar] [CrossRef]
- Marrani, E.; Burns, J.C.; Cimaz, R. How Should We Classify Kawasaki Disease? Front. Immunol. 2018, 9, 2974. [Google Scholar] [CrossRef]
- Strachan, D.P. Hay fever, hygiene, and household size. BMJ 1989, 299, 1259–1260. [Google Scholar] [CrossRef]
- Burgner, D.; Carter, K.; Webster, R.; Kuijpers, T.W. Kawasaki disease, childhood allergy and the hygiene hypothesis. Pediatr. Allergy Immunol. 2011, 22, 751. [Google Scholar] [CrossRef]
- Lee, K.Y.; Han, J.W.; Lee, J.S. Kawasaki disease may be a hyperimmune reaction of genetically susceptible children to variants of normal environmental flora. Med. Hypotheses 2007, 69, 642–651. [Google Scholar] [CrossRef]
- Park, J.S.; Seo, C.J.; Cho, S.H.; Lee, D.B. Clinical observation of mucocutaneous lymph node syndrome: 5 cases. J. Korean Pediatr. Soc. 1973, 16, 61–67. [Google Scholar]
- Melish, M.E.; Hicks, R.M.; Larson, E.J. Mucocutaneous lymph node syndrome in the United States. Am. J. Dis. Child. 1976, 130, 599–607. [Google Scholar] [CrossRef]
- Yang, H.Y.; Lin, G.J.; Lee, C.Y.; Lue, H.C. Clinical observation of mucocutaneous lymph node syndrome. Acta Paediatr. Sin. 1985, 26, 213–222. [Google Scholar]
- Singh, S.; Vignesh, P.; Burgner, D. The epidemiology of Kawasaki disease: A global update. Arch. Dis. Child. 2015, 100, 1084–1088. [Google Scholar] [CrossRef]
- Nakamura, Y. Kawasaki disease: Epidemiology and the lessons from it. Int. J. Rheum. Dis. 2018, 21, 16–19. [Google Scholar] [CrossRef]
- Kim, G.B. Reality of Kawasaki disease epidemiology. Korean J. Pediatr. 2019, 62, 292–296. [Google Scholar] [CrossRef]
- Yanagawa, H.; Kawasaki, T.; Shigematsu, I. Nationwide survey on Kawasaki disease in Japan. Pediatrics 1987, 80, 58–62. [Google Scholar]
- Uehara, R.; Nakamura, Y.; Yanagawa, H. Epidemiology of Kawasaki disease in Japan. JMAJ 2005, 48, 183–193. [Google Scholar]
- Yanagawa, H.; Uashiro, M.; Nakamura, Y.; Kawasaki, T.; Kato, H. Epidemiologic pictures of Kawasaki disease in Japan: From the nationwide incidence survey in 1991 and 1992. Pediatrics 1995, 95, 475–479. [Google Scholar]
- Yanagawa, H.; Nakamura, Y.; Yashiro, M.; Ojima, T.; Koyanagi, H.; Kawasaki, T. Update of the epidemiology of Kawasaki disease in Japan—from the results of 1993-94 nationwide survey. J. Epidemiol. 1996, 6, 148–157. [Google Scholar] [CrossRef]
- Yanagawa, H.; Nakamura, Y.; Yashiro, M.; Ojima, T.; Tanihara, S.; Oki, I.; Zhang, T. Results of the nationwide epidemiologic survey of Kawasaki disease in 1995 and 1996 in Japan. Pediatrics 1998, 102, E65. [Google Scholar] [CrossRef]
- Yanagawa, H.; Nakamura, Y.; Yashiro, M.; Oki, I.; Hirata, S.; Zhang, T.; Kawasaki, T. Incidence survey of Kawasaki disease in 1997 and 1998 in Japan. Pediatrics 2001, 107, E33. [Google Scholar] [CrossRef]
- Yanagawa, H.; Nakamura, Y.; Yashiro, M.; Uehara, R.; Oki, I.; Kayaba, K. Incidence of Kawasaki disease in Japan: The nationwide surveys in 1999–2002. Pediatr. Int. 2006, 48, 356–361. [Google Scholar] [CrossRef]
- Nakamura, Y.; Yashiro, M.; Uehara, R.; Oki, I.; Kayaba, K.; Yanagawa, H. Increasing incidence of Kawasaki disease in Japan: Nationwide survey. Pediatr. Int. 2008, 50, 287–290. [Google Scholar] [CrossRef]
- Nakamura, Y.; Yashiro, M.; Uehara, R.; Oki, I.; Watanabe, M.; Yanagawa, H. Epidemiologic features of Kawasaki disease in Japan: Results from the nationwide survey in 2005–2006. J. Epidemiol. 2008, 18, 167–172. [Google Scholar] [CrossRef]
- Nakamura, Y.; Yashiro, M.; Uehara, R.; Sasakane, A.; Chihara, I.; Aoyama, Y.; Kotani, K.; Yanagawa, H. Epidemiologic features of Kawasaki disease in Japan: Results of the 2007–2008 nationwide survey. J. Epidemiol. 2010, 20, 302–307. [Google Scholar] [CrossRef]
- Nakamura, Y.; Yashiro, M.; Uehara, R.; Sadakane, A.; Tsuboi, S.; Aoyama, Y.; Kotani, K.; Tsogzolbaatar, E.O.; Yanagawa, H. Epidemiologic features of Kawasaki disease in Japan: Results of the 2009–2010 nationwide survey. J. Epidemiol. 2012, 22, 216–221. [Google Scholar] [CrossRef]
- Makino, N.; Nakamura, Y.; Yashiro, M.; Ae, R.; Tsuboi, S.; Aoyama, Y.; Kojo, T.; Uehara, R.; Kotani, K.; Yanagawa, H. Descriptive epidemiology of Kawasaki disease in Japan, 2011–2012: From the results of the 22nd nationwide survey. J. Epidemiol. 2015, 25, 239–245. [Google Scholar] [CrossRef]
- Makino, N.; Nakamura, Y.; Yashiro, M.; Kosami, K.; Matsubara, Y.; Ae, R.; Aoyama, Y.; Yanagawa, H. Nationwide epidemiologic survey of Kawasaki disease in Japan, 2015–2016. Pediatr. Int. 2019, 61, 397–403. [Google Scholar] [CrossRef]
- Ae, R.; Makino, N.; Kosami, K.; Kuwabara, M.; Matsubara, Y.; Nakamura, Y. Epidemiology, treatments, and cardiac complications in patients with Kawasaki disease: The nationwide survey in Japan, 2017–2018. J. Pediatr. 2020, 225, 23–29. [Google Scholar] [CrossRef]
- Lee, D.B.; Lee, K.S.; Lee, B.C.; Lee, I.J. Epidemiologic and clinical study of mucocutaneous lymph node syndrome in Korea. J. Korean Pediatr. Soc. 1982, 25, 977–993. [Google Scholar]
- Park, Y.W.; Park, I.S.; Kim, C.H.; Ma, J.S.; Lee, S.B.; Kim, C.H.; Yun, Y.S.; Lee, H.J.; Lee, S.K.; Tockgo, Y.C. Epidemiologic study of Kawasaki disease in Korea, 1997–1999: Comparison with previous studies during 1991–1996. J. Korean Med. Sci. 2002, 17, 453–456. [Google Scholar] [CrossRef]
- Park, Y.W.; Han, J.W.; Park, I.S.; Kim, C.H.; Yun, Y.S.; Cha, S.H.; Ma, J.S.; Lee, S.B.; Kim, C.H.; Lee, H.J.; et al. Epidemiologic picture of Kawasaki disease in Korea, 2000–2002. Pediatr. Int. 2005, 47, 382–387. [Google Scholar] [CrossRef]
- Park, Y.W.; Han, J.W.; Park, I.S.; Kim, C.H.; Cha, S.H.; Ma, J.S.; Lee, J.S.; Kwon, T.C.; Lee, S.B.; Kim, C.H.; et al. Kawasaki disease in Korea, 2003–2005. Pediatr. Infect. Dis. J. 2007, 26, 821–823. [Google Scholar] [CrossRef]
- Park, Y.W.; Han, J.W.; Hong, Y.M.; Ma, J.S.; Cha, S.H.; Kwon, T.C.; Lee, S.B.; Kim, C.H.; Lee, J.S.; Kim, C.H. Epidemiological features of Kawasaki disease in Korea, 2006–2008. Pediatr. Int. 2011, 53, 36–39. [Google Scholar] [CrossRef]
- Kim, G.B.; Han, J.W.; Park, Y.W.; Song, M.S.; Hong, Y.M.; Cha, S.H.; Kim, D.S.; Park, S. Epidemiologic features of Kawasaki disease in South Korea: Data from nationwide survey, 2009–2011. Pediatr. Infect. Dis. J. 2014, 33, 24–27. [Google Scholar] [CrossRef]
- Kim, G.B.; Park, S.; Eun, L.Y.; Han, J.W.; Lee, S.Y.; Yoon, K.L.; Yu, J.J.; Choi, J.W.; Lee, K.Y. Epidemiology and Clinical Features of Kawasaki Disease in South Korea, 2012–2014. Pediatr. Infect. Dis. J. 2017, 36, 482–485. [Google Scholar] [CrossRef]
- Kim, G.B.; Eun, L.Y.; Han, J.W.; Kim, S.H.; Yoon, K.L.; Han, M.Y.; Yu, J.J.; Choi, J.W.; Rhim, J.W. Epidemiology of Kawasaki Disease in South Korea: A Nationwide Survey 2015–2017. Pediatr. Infect. Dis. J. 2020, 39, 1012–1016. [Google Scholar] [CrossRef]
- Fujiwara, T.; Shobugawa, Y.; Matsumoto, K.; Kawachi, I. Association of early social environment with the onset of pediatric Kawasaki disease. Ann. Epidemiol. 2019, 29, 74–80. [Google Scholar] [CrossRef]
- Nakamura, Y.; Yanagawa, I.; Kawasaki, T. Temporal and geographical clustering of Kawasaki disease in Japan. Prog. Clin. Biol. Res. 1987, 250, 19–32. [Google Scholar]
- Chang, Y.T.; Wang, H.S.; Liu, J.R.; Tseng, C.N.; Chou, I.J.; Luo, S.F.; Kuo, C.F.; See, L.C. Association of maternal ethnicity and urbanicity on severe pediatric disease: A nationwide cohort study. BMC Pediatr. 2019, 19, 514. [Google Scholar] [CrossRef]
- Bonetto, C.; Trotta, F.; Felicetti, P.; Alarcón, G.S.; Santuccio, C.; Bachtiar, N.S.; Brauchli Pernus, Y.; Chandler, R.; Girolomoni, G.; Hadden, R.D.; et al. Vasculitis as an adverse event following immunization—Systematic literature review. Vaccine 2016, 34, 6641–6651. [Google Scholar] [CrossRef]
- Hua, W.; Izurieta, H.S.; Slade, B.; Belay, E.D.; Haber, P.; Tiernan, R.; Woo, E.J.; Iskander, J.; Braun, M.M.; Ball, R. Kawasaki disease after vaccination: Reports to the vaccine adverse event reporting system 1990–2007. Pediatr. Infect. Dis. J. 2009, 28, 943–947. [Google Scholar] [CrossRef]
- Abrams, J.Y.; Weintraub, E.S.; Baggs, J.M.; McCarthy, N.L.; Schonberger, L.B.; Lee, G.M.; Klein, N.P.; Belongia, E.A.; Jackson, M.L.; Naleway, A.L.; et al. Childhood vaccines and Kawasaki disease, Vaccine Safety Datalink, 1996–2006. Vaccine 2015, 33, 382–387. [Google Scholar] [CrossRef]
- Center, K.J.; Hansen, J.R.; Lewis, E.; Fireman, B.H.; Hilton, B. Lack of association of Kawasaki disease after immunization in a cohort of infants followed for multiple autoimmune diagnoses in a large, phase-4 observational database safety study of 7-valent pneumococcal conjugate vaccine: Lack of association between Kawasaki disease and seven-valent pneumococcal conjugate vaccine. Pediatr. Infect. Dis. J. 2009, 28, 438–440. [Google Scholar]
- Esposito, S.; Bianchini, S.; Dellepiane, R.M.; Principi, N. Vaccines and Kawasaki disease. Expert Rev. Vaccines 2016, 15, 417–424. [Google Scholar]
- Yorifuji, T.; Tsukahara, H.; Doi, H. Breastfeeding and Risk of Kawasaki Disease: A Nationwide Longitudinal Survey in Japan. Pediatrics 2016, 137, e20153919. [Google Scholar] [CrossRef]
- Meyer, K.; Volkmann, A.; Hufnagel, M.; Schachinger, E.; Klau, S.; Horstmann, J.; Berner, R.; Fischer, M.; Lehner, A.; Haas, N.; et al. Breastfeeding and vitamin D supplementation reduce the risk of Kawasaki disease in a German population-based case-control study. BMC Pediatr. 2019, 19, 66. [Google Scholar] [CrossRef]
- Wang, S.; Xiang, D.; Fang, C.; Yao, B. Association between breastfeeding and Kawasaki disease: A case-control study. Eur. J. Pediatr. 2020, 179, 447–453. [Google Scholar] [CrossRef]
- Iyengar, S.R.; Walker, W.A. Immune factors in breast milk and the development of atopic disease. J. Pediatr. Gastroenterol. Nutr. 2012, 55, 641–647. [Google Scholar] [CrossRef]
- M’Rabet, L.; Vos, A.P.; Boehm, G.; Garssen, J. Breast-feeding and its role in early development of the immune system in infants: Consequences for health later in life. J. Nutr. 2008, 138, 1782S–1790S. [Google Scholar] [CrossRef]
- McCrindle, B.W.; Rowley, A.H.; Newburger, J.W.; Burns, J.C.; Bolger, A.F.; Gewitz, M.; Baker, A.L.; Jackson, M.A.; Takahashi, M.; Shah, P.B.; et al. Diagnosis, Treatment, and Long-Term Management of Kawasaki Disease: A Scientific Statement for Health Professionals From the American Heart Association. Circulation 2017, 135, e927–e999. [Google Scholar] [CrossRef]
- Janeway, C.A.; Travers, P.; Walport, M.; Shlomchik, M.J. Immunobiology: The Immune System in Health & Disease, 6th ed.; Garland Science Publishing: New York, NY, USA, 2005. [Google Scholar]
- Rivas-Larrauri, F.; Aguilar-Zanela, L.; Castro-Oteo, P.; Rosales-Hernandez, L.A.; Otero-Mendoza, F.; López-Herrera, G.; Ordoñez-Ortega, J.; Garrido-García, M.; Yamazaki-Nakashimada, M.A. Kawasaki disease and immunodeficiencies in children: Case reports and literature review. Rheumatol. Int. 2019, 39, 1829–1838. [Google Scholar] [CrossRef]
- Matsuoka, S.; Tatara, K.; Nakagawa, R.; Mori, K.; Kuroda, Y. Tendency toward atopy in Kawasaki disease. Eur. J. Pediatr. 1997, 156, 30–32. [Google Scholar] [CrossRef]
- Woon, P.Y.; Chang, W.C.; Liang, C.C.; Hsu, C.H.; Klahan, S.; Huang, Y.H.; Chang, W.P.; Kuo, H.C. Increased risk of atopic dermatitis in preschool children with kawasaki disease: A population-based study in Taiwan. Evid. Based Complement Alternat. Med. 2013, 2013, 605123. [Google Scholar] [CrossRef]
- Tsai, Y.J.; Lin, C.H.; Fu, L.S.; Fu, Y.C.; Lin, M.C.; Jan, S.L. The association between Kawasaki disease and allergic diseases, from infancy to school age. Allergy Asthma Proc. 2013, 34, 467–472. [Google Scholar] [CrossRef]
- Kuo, H.C.; Chang, W.C.; Yang, K.D.; Yu, H.R.; Wang, C.L.; Ho, S.C.; Yang, C.Y. Kawasaki disease and subsequent risk of allergic diseases: A population-based matched cohort study. BMC Pediatr. 2013, 13, 38. [Google Scholar] [CrossRef]
- Liew, W.K.; Lim, C.W.; Tan, T.H.; Wong, K.Y.; Tai, B.C.; Quek, S.C.; Bever, H.v. The effect of Kawasaki disease on childhood allergies-A sibling control study. Pediatr. Allergy Immunol. 2011, 22, 488–493. [Google Scholar] [CrossRef]
- Webster, R.J.; Carter, K.W.; Warrington, N.M.; Loh, A.M.; Zaloumis, S.; Kuijpers, T.W.; Palmer, L.J.; Burgner, D.P. Hospitalisation with infection, asthma and allergy in Kawasaki disease patients and their families: Genealogical analysis using linked population data. PLoS ONE 2011, 6, e28004. [Google Scholar] [CrossRef]
- Hassidim, A.; Merdler, I.; Chorin, O.; Merdler-Rabinowicz, R.; Dallal, I.; Perlman, M.; Chorin, E. Atopic Predilection among Kawasaki Disease Patients: A Cross-Sectional Study of 1,187,757 Teenagers. Int. Arch. Allergy Immunol. 2016, 170, 92–96. [Google Scholar] [CrossRef]
- Wei, C.C.; Lin, C.L.; Kao, C.H.; Liao, Y.H.; Shen, T.C.; Tsai, J.D.; Chang, Y.J.; Li, T.C. Increased risk of Kawasaki disease in children with common allergic diseases. Ann. Epidemiol. 2014, 24, 340–343. [Google Scholar] [CrossRef]
- Furukawa, S.; Matsubara, T.; Yabuta, K. Mononuclear cell subsets and coronary artery lesions in Kawasaki disease. Arch. Dis. Child. 1992, 67, 706–708. [Google Scholar] [CrossRef]
- Tsujimoto, H.; Takeshita, S.; Nakatani, K.; Kawamura, Y.; Tokutomi, T.; Sekine, I. Intravenous immunoglobulin therapy induces neutrophil apoptosis in Kawasaki disease. Clin. Immunol. 2002, 103, 161–168. [Google Scholar] [CrossRef]
- Lee, H.H.; Park, I.H.; Shin, J.S.; Kim, D.S. Immunoglobulin V(H) chain gene analysis of peripheral blood IgM-producing B cells in patients with Kawasaki disease. Yonsei Med. J. 2009, 50, 493–504. [Google Scholar] [CrossRef][Green Version]
- Ha, K.S.; Jang, G.Y.; Lee, J.; Lee, K.C.; Son, C.S. Laboratory Markers in Incomplete Kawasaki Disease according to Coronary Artery Outcome. Korean Circ. J. 2018, 48, 287–295. [Google Scholar] [CrossRef]
- Ling, X.B.; Lau, K.; Kanegaye, J.T.; Pan, Z.; Peng, S.; Ji, J.; Liu, G.; Sato, Y.; Yu, T.T.; Whitin, J.C.; et al. A diagnostic algorithm combining clinical and molecular data distinguishes Kawasaki disease from other febrile illnesses. BMC Med. 2011, 9, 130. [Google Scholar] [CrossRef]
- Newburger, J.W.; Takahashi, M.; Beiser, A.S.; Burns, J.C.; Bastian, J.; Chung, K.J.; Colan, S.D.; Duffy, C.E.; Fulton, D.R.; Glode, M.P. A single intravenous infusion of gamma globulin as compared with four infusions in the treatment of acute Kawasaki syndrome. N. Engl. J. Med. 1991, 324, 1633–1639. [Google Scholar] [CrossRef]
- Ding, Y.; Li, G.; Xiong, L.J.; Yin, W.; Liu, J.; Liu, F.; Wang, R.G.; Xia, K.; Zhang, S.L.; Zhao, L. Profiles of responses of immunological factors to different subtypes of Kawasaki disease. BMC Musculoskelet. Disord. 2015, 16, 315. [Google Scholar] [CrossRef]
- Kuo, H.C.; Huang, Y.H.; Chung, F.H.; Chen, P.C.; Sung, T.C.; Chen, Y.W.; Hsieh, K.S.; Chen, C.S.; Syu, G.D. Antibody Profiling of Kawasaki Disease Using Escherichia coli Proteome Microarrays. Mol. Cell. Proteomics 2018, 17, 472–481. [Google Scholar] [CrossRef]
- Yamazaki-Nakashimada, M.A.; Gámez-González, L.B.; Murata, C.; Honda, T.; Yasukawa, K.; Hamada, H. IgG levels in Kawasaki disease and its association with clinical outcomes. Clin. Rheumatol. 2019, 38, 749–754. [Google Scholar] [CrossRef]
- Kawamori, J.; Miyake, T.; Yoshida, T. B-cell function in Kawasaki disease and the effect of high-dose gamma-globulin therapy. Acta Paediatr. Jpn. 1989, 31, 537–543. [Google Scholar] [CrossRef]
- Han, J.W.; Oh, J.H.; Rhim, J.W.; Lee, K.Y. Correlation between elevated platelet count and immunoglobulin levels in the early convalescent stage of Kawasaki disease. Medicine 2017, 96, e7583. [Google Scholar] [CrossRef]
- Sawaji, Y.; Haneda, N.; Yamaguchi, S.; Kajino, Y.; Kishida, K.; Seto, S.; Konishi, N.; Waki, K.; Baba, K.; Arisawa, K.; et al. Coronary risk factors in acute Kawasaki disease: Correlation of serum immunoglobulin levels with coronary complications. Acta Paediatr. Jpn. 1998, 40, 218–225. [Google Scholar] [CrossRef]
- Morikawa, Y.; Ohashi, Y.; Harada, K.; Asai, T.; Okawa, S.; Nagashima, M.; Katoh, T.; Baba, K.; Frusho, K.; Okuni, M.; et al. Coronary risks after high-dose gamma-globulin in children with Kawasaki disease. Pediatr. Int. 2000, 42, 464–469. [Google Scholar] [CrossRef]
- Kim, J.J.; Kim, H.J.; Yu, J.J.; Yun, S.W.; Lee, K.Y.; Yoon, K.L.; Kil, H.R.; Kim, G.B.; Han, M.K.; Song, M.S.; et al. IgA Levels Are Associated with Coronary Artery Lesions in Kawasaki Disease. Korean Circ. J. 2021, 51, 267–278. [Google Scholar] [CrossRef]
- Kuo, H.C.; Pan, C.T.; Huang, Y.H.; Huang, F.C.; Lin, Y.S.; Li, S.C.; Huang, L.H. Global Investigation of Immune Repertoire Suggests Kawasaki Disease Has Infectious Cause. Circ. J. 2019, 83, 2070–2078. [Google Scholar] [CrossRef]
- Ko, T.M.; Kiyotani, K.; Chang, J.S.; Park, J.H.; Yin Yew, P.; Chen, Y.T.; Wu, J.Y.; Nakamura, Y. Immunoglobulin profiling identifies unique signatures in patients with Kawasaki disease during intravenous immunoglobulin treatment. Hum. Mol. Genet. 2018, 27, 2671–2677. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.; Wrotniak, B.H.; Hicar, M. Suppressed plasmablast responses in febrile infants, including children with Kawasaki disease. PLoS ONE 2018, 13, e0193539. [Google Scholar] [CrossRef]
- Furukawa, S.; Matsubara, T.; Motohashi, T.; Sasai, K.; Nakachi, S.; Umezawa, Y.; Yabuta, K. Increased expression of Fc epsilon R2/CD23 on peripheral blood B lymphocytes and serum IgE levels in Kawasaki disease. Int. Arch. Allergy Appl. Immunol. 1991, 95, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Kusakawa, S.; Heiner, D.C. Elevated levels of immunoglobulin E in the acute febrile mucocutaneous lymph node syndrome. Pediatr. Res. 1976, 10, 108–111. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Hwang, B. Serial immunologic studies in patients with mucocutaneous lymph node syndrome (Kawasaki disease). Ann. Allergy 1987, 59, 291–297. [Google Scholar] [PubMed]
- Koo, C.M.; Choi, S.Y.; Kim, D.S.; Kim, K.W. Relation between Kawasaki Disease and Immunoglobulin E. J. Rheum. Dis. 2013, 20, 4–8. [Google Scholar] [CrossRef]
- Cahenzli, J.; Köller, Y.; Wyss, M.; Geuking, M.B.; McCoy, K.D. Intestinal microbial diversity during early-life colonization shapes long-term IgE levels. Cell Host Microbe 2013, 14, 559–570. [Google Scholar] [CrossRef] [PubMed]
- Hill, D.A.; Siracusa, M.C.; Abt, M.C.; Kim, B.S.; Kobuley, D.; Kubo, M.; Kambayashi, T.; Larosa, D.F.; Renner, E.D.; Orange, J.S.; et al. Commensal bacteria-derived signals regulate basophil hematopoiesis and allergic inflammation. Nat. Med. 2012, 18, 538–546. [Google Scholar] [CrossRef]
- Wesemann, D.R.; Magee, J.M.; Boboila, C.; Calado, D.P.; Gallagher, M.P.; Portuguese, A.J.; Manis, J.P.; Zhou, X.; Recher, M.; Rajewsky, K.; et al. Immature B cells preferentially switch to IgE with increased direct Sμ to Sε recombination. J. Exp. Med. 2011, 208, 2733–2746. [Google Scholar] [CrossRef]
- Brosius, C.L.; Newburger, J.W.; Burns, J.C.; Hojnowski-Diaz, P.; Zierler, S.; Leung, D.Y. Increased prevalence of atopic dermatitis in Kawasaki disease. Pediatr. Infect. Dis. J. 1988, 7, 863–866. [Google Scholar] [CrossRef]
- Park, K.S.; Lee, K.S.; Kim, C.W. Study on Serum IgE Levels in Healthy Korean. Korean J. Clin. Pathol. 1982, 2, 65–71. [Google Scholar]
- Holman, R.C.; Belay, E.D.; Christensen, K.Y.; Folkema, A.M.; Steiner, C.A.; Schonberger, L.B. Hospitalizations for Kawasaki syndrome among children in the United States, 1997–2007. Pediatr. Infect. Dis. J. 2010, 29, 483–488. [Google Scholar] [CrossRef]
- Kuo, H.C.; Yang, K.D.; Liang, C.D.; Bong, C.N.; Yu, H.R.; Wang, L.; Wang, C.L. The relationship of eosinophilia to intravenous immunoglobulin treatment failure in Kawasaki disease. Pediatr. Allergy Immunol. 2007, 18, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Terai, M.; Yasukawa, K.; Honda, T.; Jibiki, T.; Hirano, K.; Sato, J.; Ishiwada, N.; Seguchi, M.; Ueda, S.; Kohno, Y. Peripheral blood eosinophilia and eosinophil accumulation in coronary microvessels in acute Kawasaki disease. Pediatr. Infect. Dis. J. 2002, 21, 777–781. [Google Scholar] [CrossRef] [PubMed]
- Öner, T.; Yılmazer, M.M.; Güven, B.; Devrim, I.; Cilengiroğlu, Ö.V.; Demirpençe, S.; Meşe, T.; Tavlı, V.; Vitrinel, A. An observational study on peripheral blood eosinophilia in incomplete Kawasaki disease. Anadolu Kardiyol Derg 2012, 12, 160–164. [Google Scholar] [PubMed]
- Hwang, J.Y.; Lee, K.Y.; Rhim, J.W.; Youn, Y.S.; Oh, J.H.; Han, J.W.; Lee, J.S.; Burgner, D. Assessment of intravenous immunoglobulin non-responders in Kawasaki disease. Arch. Dis. Child. 2011, 96, 1088–1090. [Google Scholar] [CrossRef]
- Seo, Y.M.; Kang, H.M.; Lee, S.C.; Yu, J.W.; Kil, H.R.; Rhim, J.W.; Han, J.W.; Lee, K.Y. Clinical implications in laboratory parameter values in acute Kawasaki disease for early diagnosis and proper treatment. Korean J. Pediatr. 2018, 61, 160–166. [Google Scholar] [CrossRef]
- Kazatchkine, M.D.; Kaveri, S.V. Immunomodulation of autoimmune and inflammatory diseases with intravenous immune globulin. N. Engl. J. Med. 2001, 345, 747–755. [Google Scholar] [CrossRef]
- Kato, H.; Koike, S.; Yokoyama, T. Kawasaki disease: Effect of treatment on coronary artery involvement. Pediatrics 1979, 63, 175–179. [Google Scholar]
- Newburger, J.W.; Sleeper, L.A.; McCrindle, B.W.; Minich, L.L.; Gersony, W.; Vetter, V.L.; Atz, A.M.; Li, J.S.; Takahashi, M.; Baker, A.L.; et al. Randomized trial of pulsed corticosteroid therapy for primary treatment of Kawasaki disease. N. Engl. J. Med. 2007, 356, 663–675. [Google Scholar] [CrossRef]
- Okubo, Y.; Michihata, N.; Morisaki, N.; Sundel, R.P.; Matsui, H.; Fushimi, K.; Yasunaga, H. Association Between Dose of Glucocorticoids and Coronary Artery Lesions in Kawasaki Disease. Arthritis Care Res. 2018, 70, 1052–1057. [Google Scholar] [CrossRef]
- Miura, M. Role of glucocorticoids in Kawasaki disease. Int. J. Rheum. Dis. 2018, 21, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Anthony, R.M.; Ravetch, J.V. A novel role for the IgG Fc glycan: The anti-inflammatory activity of sialylated IgG Fcs. J. Clin. Immunol. 2010, 30, S9–S14. [Google Scholar] [CrossRef]
- Kuwabara, M.; Yashiro, M.; Ae, R.; Yanagawa, H.; Nakamura, Y. The effects of early intravenous immunoglobulin therapy for Kawasaki disease: The 22nd nationwide survey in Japan. Int. J. Cardiol. 2018, 269, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Sugimura, T.; Akagi, T.; Sato, N.; Hashino, K.; Maeno, Y.; Kazue, T.; Eto, G.; Yamakawa, R. Long-term consequences of Kawasaki disease. A 10- to 21-year follow-up study of 594 patients. Circulation 1996, 94, 1379–1385. [Google Scholar] [CrossRef] [PubMed]
- Ogata, S.; Shimizu, C.; Franco, A.; Touma, R.; Kanegaye, J.T.; Choudhury, B.P.; Naidu, N.N.; Kanda, Y.; Hoang, L.T.; Hibberd, M.L.; et al. Treatment response in Kawasaki disease is associated with sialylation levels of endogenous but not therapeutic intravenous immunoglobulin G. PLoS ONE 2013, 8, e81448. [Google Scholar] [CrossRef]
- Maddox, R.A.; Holman, R.C.; Uehara, R.; Callinan, L.S.; Guest, J.L.; Schonberger, L.B.; Nakamura, Y.; Yashiro, M.; Belay, E.D. Recurrent Kawasaki disease: USA and Japan. Pediatr. Int. 2015, 57, 1116–1120. [Google Scholar] [CrossRef]
- Balasubramanian, S.; Ganesh, R. Recurrent Kawasaki disease. Indian J. Pediatr. 2009, 76, 848–849. [Google Scholar] [CrossRef]
- Chen, C.Y.; Wu, J.R. Relapse of Kawasaki disease: A case report. Gaoxiong Yi Xue Ke Xue Za Zhi 1989, 5, 189–193. [Google Scholar]
- Cook, D.H.; Antia, A.; Attie, F.; Gersony, W.M.; Kamiya, T.; Kato, H.; Kusakawa, S.; Rowe, R.D.; Takao, A.; Van der Hauwaert, L.; et al. Results from an international survey of Kawasaki disease in 1979–1982. Can. J. Cardiol. 1989, 5, 389–394. [Google Scholar]
- Uehara, R.; Belay, E.D. Epidemiology of Kawasaki disease in Asia, Europe, and the United States. J. Epidemiol. 2012, 22, 79–85. [Google Scholar] [CrossRef]
- Holman, R.C.; Shahriari, A.; Effler, P.V.; Belay, E.D.; Schonberger, L.B. Kawasaki syndrome hospitalizations among children in Hawaii and Connecticut. Arch. Pediatr. Adolesc. Med. 2000, 154, 804–808. [Google Scholar] [CrossRef]
- Chang, R.K. Epidemiologic characteristics of children hospitalized for Kawasaki disease in California. Pediatr. Infect. Dis. J. 2002, 21, 1150–1155. [Google Scholar] [CrossRef]
- Fujita, Y.; Nakamura, Y.; Sakata, K.; Hara, N.; Kobayashi, M.; Nagai, M.; Yanagawa, H.; Kawasaki, T. Kawasaki disease in families. Pediatrics 1989, 84, 666–669. [Google Scholar]
- Uehara, R.; Yashiro, M.; Nakamura, Y.; Yanagawa, H. Kawasaki disease in parents and children. Acta Paediatr. 2003, 92, 694–697. [Google Scholar] [CrossRef]
- Onouchi, Y. Genetics of Kawasaki disease: What we know and don’t know. Circ. J. 2012, 76, 1581–1586. [Google Scholar] [CrossRef]
- Onouchi, Y.; Ozaki, K.; Burns, J.C.; Shimizu, C.; Terai, M.; Hamada, H.; Honda, T.; Suzuki, H.; Suenaga, T.; Takeuchi, T.; et al. A genome-wide association study identifies three new risk loci for Kawasaki disease. Nat. Genet. 2012, 44, 517–521. [Google Scholar] [CrossRef]
- Lee, Y.C.; Kuo, H.C.; Chang, J.S.; Chang, L.Y.; Huang, L.M.; Chen, M.R.; Liang, C.D.; Chi, H.; Huang, F.Y.; Lee, M.L.; et al. Two new susceptibility loci for Kawasaki disease identified through genome-wide association analysis. Nat. Genet. 2012, 44, 522–525. [Google Scholar] [CrossRef]
- Kim, J.J.; Yun, S.W.; Yu, J.J.; Yoon, K.L.; Lee, K.Y.; Kil, H.R.; Kim, G.B.; Han, M.K.; Song, M.S.; Lee, H.D.; et al. A genome-wide association analysis identifies NMNAT2 and HCP5 as susceptibility loci for Kawasaki disease. J. Hum. Genet. 2017, 62, 1023–1029. [Google Scholar] [CrossRef]
- Kwon, Y.C.; Kim, J.J.; Yun, S.W.; Yu, J.J.; Yoon, K.L.; Lee, K.Y.; Kil, H.R.; Kim, G.B.; Han, M.K.; Song, M.S.; et al. BCL2L11 Is Associated With Kawasaki Disease in Intravenous Immunoglobulin Responder Patients. Circ. Genom. Precis. Med. 2018, 11, e002020. [Google Scholar] [CrossRef]
- Khor, C.C.; Davila, S.; Breunis, W.B.; Lee, Y.C.; Shimizu, C.; Wright, V.J.; Yeung, R.S.; Tan, D.E.; Sim, K.S.; Wang, J.J.; et al. Genome-wide association study identifies FCGR2A as a susceptibility locus for Kawasaki disease. Nat. Genet. 2011, 43, 1241–1246. [Google Scholar] [CrossRef]
- Johnson, T.A.; Mashimo, Y.; Wu, J.Y.; Yoon, D.; Hata, A.; Kubo, M.; Takahashi, A.; Tsunoda, T.; Ozaki, K.; Tanaka, T.; et al. Association of an IGHV3-66 gene variant with Kawasaki disease. J. Hum. Genet. 2021, 66, 475–489. [Google Scholar] [CrossRef]
- Chang, C.J.; Kuo, H.C.; Chang, J.S.; Lee, J.K.; Tsai, F.J.; Khor, C.C.; Chang, L.C.; Chen, S.P.; Ko, T.M.; Liu, Y.M.; et al. Replication and meta-analysis of GWAS identified susceptibility loci in Kawasaki disease confirm the importance of B lymphoid tyrosine kinase (BLK) in disease susceptibility. PLoS ONE 2013, 8, e72037. [Google Scholar] [CrossRef]
- Dymecki, S.M.; Zwollo, P.; Zeller, K.; Kuhajda, F.P.; Desiderio, S.V. Structure and developmental regulation of the B-lymphoid tyrosine kinase gene blk. J. Biol. Chem. 1992, 267, 4815–4823. [Google Scholar] [CrossRef]
- Bewarder, N.; Weinrich, V.; Budde, P.; Hartmann, D.; Flaswinkel, H.; Reth, M.; Frey, J. In vivo and in vitro specificity of protein tyrosine kinases for immunoglobulin G receptor (FcgammaRII) phosphorylation. Mol. Cell. Biol. 1996, 16, 4735–4743. [Google Scholar] [CrossRef]
- Reth, M.; Wienands, J. Initiation and processing of signals from the B cell antigen receptor. Annu. Rev. Immunol. 1997, 15, 453–479. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.R.; Scott, D.W. Antisense oligodeoxynucleotides to the blk tyrosine kinase prevent anti-mu-chain-mediated growth inhibition and apoptosis in a B-cell lymphoma. Proc. Natl. Acad. Sci. USA 1993, 90, 7946–7950. [Google Scholar] [CrossRef] [PubMed]
- Simpfendorfer, K.R.; Olsson, L.M.; Manjarrez Orduño, N.; Khalili, H.; Simeone, A.M.; Katz, M.S.; Lee, A.T.; Diamond, B.; Gregersen, P.K. The autoimmunity-associated BLK haplotype exhibits cis-regulatory effects on mRNA and protein expression that are prominently observed in B cells early in development. Hum. Mol. Genet. 2012, 21, 3918–3925. [Google Scholar] [CrossRef] [PubMed]
- Wasserman, R.; Li, Y.S.; Hardy, R.R. Differential expression of the blk and ret tyrosine kinases during B lineage development is dependent on Ig rearrangement. J. Immunol. 1995, 155, 644–651. [Google Scholar] [PubMed]
- Zeller, T.; Wild, P.; Szymczak, S.; Rotival, M.; Schillert, A.; Castagne, R.; Maouche, S.; Germain, M.; Lackner, K.; Rossmann, H.; et al. Genetics and beyond--the transcriptome of human monocytes and disease susceptibility. PLoS ONE 2010, 5, e10693. [Google Scholar] [CrossRef] [PubMed]
- Han, J.W.; Zheng, H.F.; Cui, Y.; Sun, L.D.; Ye, D.Q.; Hu, Z.; Xu, J.H.; Cai, Z.M.; Huang, W.; Zhao, G.P.; et al. Genome-wide association study in a Chinese Han population identifies nine new susceptibility loci for systemic lupus erythematosus. Nat. Genet. 2009, 41, 1234–1237. [Google Scholar] [CrossRef] [PubMed]
- Okada, Y.; Wu, D.; Trynka, G.; Raj, T.; Terao, C.; Ikari, K.; Kochi, Y.; Ohmura, K.; Suzuki, A.; Yoshida, S.; et al. Genetics of rheumatoid arthritis contributes to biology and drug discovery. Nature 2014, 506, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Herrera, M.; Kerick, M.; González-Serna, D.; Myositis Genetics Consortium; Scleroderma Genetics Consortium; Wijmenga, C.; Franke, A.; Gregersen, P.K.; Padyukov, L.; Worthington, J.; et al. Genome-wide meta-analysis reveals shared new loci in systemic seropositive rheumatic diseases. Ann. Rheum. Dis. 2019, 78, 311–319. [Google Scholar] [CrossRef]
- Hom, G.; Graham, R.R.; Modrek, B.; Taylor, K.E.; Ortmann, W.; Garnier, S.; Lee, A.T.; Chung, S.A.; Ferreira, R.C.; Pant, P.V.; et al. Association of systemic lupus erythematosus with C8orf13-BLK and ITGAM-ITGAX. N. Engl. J. Med. 2008, 358, 900–909. [Google Scholar]
- Snow, A.L.; Oliveira, J.B.; Zheng, L.; Dale, J.K.; Fleisher, T.A.; Lenardo, M.J. Critical role for BIM in T cell receptor restimulation-induced death. Biol. Direct 2008, 3, 34. [Google Scholar] [CrossRef]
- Enders, A.; Bouillet, P.; Puthalakath, H.; Xu, Y.; Tarlinton, D.M.; Strasser, A. Loss of the pro-apoptotic BH3-only Bcl-2 family member Bim inhibits BCR stimulation-induced apoptosis and deletion of autoreactive B cells. J. Exp. Med. 2003, 198, 1119–1126. [Google Scholar] [CrossRef]
- Bouillet, P.; Metcalf, D.; Huang, D.C.; Tarlinton, D.M.; Kay, T.W.; Köntgen, F.; Adams, J.M.; Strasser, A. Proapoptotic Bcl-2 relative Bim required for certain apoptotic responses, leukocyte homeostasis, and to preclude autoimmunity. Science 1999, 286, 1735–1738. [Google Scholar] [CrossRef]
- Astle, W.J.; Elding, H.; Jiang, T.; Allen, D.; Ruklisa, D.; Mann, A.L.; Mead, D.; Bouman, H.; Riveros-Mckay, F.; Kostadima, M.A.; et al. The Allelic Landscape of Human Blood Cell Trait Variation and Links to Common Complex Disease. Cell 2016, 167, 1415–1429. [Google Scholar] [CrossRef]
- Vuckovic, D.; Bao, E.L.; Akbari, P.; Lareau, C.A.; Mousas, A.; Jiang, T.; Chen, M.H.; Raffield, L.M.; Tardaguila, M.; Huffman, J.E.; et al. The Polygenic and Monogenic Basis of Blood Traits and Diseases. Cell 2020, 182, 1214–1231. [Google Scholar] [CrossRef]
- Speedy, H.E.; Di Bernardo, M.C.; Sava, G.P.; Dyer, M.J.; Holroyd, A.; Wang, Y.; Sunter, N.J.; Mansouri, L.; Juliusson, G.; Smedby, K.E.; et al. A genome-wide association study identifies multiple susceptibility loci for chronic lymphocytic leukemia. Nat. Genet. 2014, 46, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Berndt, S.I.; Camp, N.J.; Skibola, C.F.; Vijai, J.; Wang, Z.; Gu, J.; Nieters, A.; Kelly, R.S.; Smedby, K.E.; Monnereau, A.; et al. Meta-analysis of genome-wide association studies discovers multiple loci for chronic lymphocytic leukemia. Nat. Commun. 2016, 7, 10933. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Jia, Q.; Jahani, P.S.; Hurrell, B.P.; Pan, C.; Huang, P.; Gukasyan, J.; Woodward, N.C.; Eskin, E.; Gilliland, F.D.; et al. Genome-wide analysis highlights contribution of immune system pathways to the genetic architecture of asthma. Nat. Commun. 2020, 11, 1776. [Google Scholar] [CrossRef] [PubMed]
- Suhre, K.; Shin, S.Y.; Petersen, A.K.; Mohney, R.P.; Meredith, D.; Wägele, B.; Altmaier, E.; Deloukas, P.; Erdmann, J.; Grundberg, E.; et al. Human metabolic individuality in biomedical and pharmaceutical research. Nature 2011, 477, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Tian, X.; Li, Q.; Wang, G.; Li, C.; Yang, J. Expression of FcRs on monocytes among Kawasaki disease patients with coronary artery lesions. Int. Immunopharmacol. 2017, 45, 1–5. [Google Scholar] [CrossRef]
- Fridman, W.H.; Teillaud, J.L.; Bouchard, C.; Teillaud, C.; Astier, A.; Tartour, E.; Galon, J.; Mathiot, C.; Sautès, C. Soluble Fc gamma receptors. J. Leukoc. Biol. 1993, 54, 504–512. [Google Scholar] [CrossRef]
- International Genetics of Ankylosing Spondylitis Consortium (IGAS); Cortes, A.; Hadler, J.; Pointon, J.P.; Robinson, P.C.; Karaderi, T.; Leo, P.; Cremin, K.; Pryce, K.; Harris, J.; et al. Identification of multiple risk variants for ankylosing spondylitis through high-density genotyping of immune-related loci. Nat. Genet. 2013, 45, 730–738. [Google Scholar] [CrossRef]
- Bentham, J.; Morris, D.L.; Graham, D.S.C.; Pinder, C.L.; Tombleson, P.; Behrens, T.W.; Martín, J.; Fairfax, B.P.; Knight, J.C.; Chen, L.; et al. Genetic association analyses implicate aberrant regulation of innate and adaptive immunity genes in the pathogenesis of systemic lupus erythematosus. Nat. Genet. 2015, 47, 1457–1464. [Google Scholar] [CrossRef]
- Liu, J.Z.; van Sommeren, S.; Huang, H.; Ng, S.C.; Alberts, R.; Takahashi, A.; Ripke, S.; Lee, J.C.; Jostins, L.; Shah, T.; et al. Association analyses identify 38 susceptibility loci for inflammatory bowel disease and highlight shared genetic risk across populations. Nat. Genet. 2015, 47, 979–986. [Google Scholar] [CrossRef]
- Anderson, C.A.; Boucher, G.; Lees, C.W.; Franke, A.; D’Amato, M.; Taylor, K.D.; Lee, J.C.; Goyette, P.; Imielinski, M.; Latiano, A.; et al. Meta-analysis identifies 29 additional ulcerative colitis risk loci, increasing the number of confirmed associations to 47. Nat. Genet. 2011, 43, 246–252. [Google Scholar] [CrossRef]
- Jostins, L.; Ripke, S.; Weersma, R.K.; Duerr, R.H.; McGovern, D.P.; Hui, K.Y.; Lee, J.C.; Schumm, L.P.; Sharma, Y.; Anderson, C.A.; et al. Host-microbe interactions have shaped the genetic architecture of inflammatory bowel disease. Nature 2012, 491, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Warmerdam, P.A.; van de Winkel, J.G.; Vlug, A.; Westerdaal, N.A.; Capel, P.J. A single amino acid in the second Ig-like domain of the human Fc gamma receptor II is critical for human IgG2 binding. J. Immunol. 1991, 147, 1338–1343. [Google Scholar] [PubMed]
- Clark, M.R.; Stuart, S.G.; Kimberly, R.P.; Ory, P.A.; Goldstein, I.M. A single amino acid distinguishes the high-responder from the low-responder form of Fc receptor II on human monocytes. Eur. J. Immunol. 1991, 21, 1911–1916. [Google Scholar] [CrossRef]
- Parren, P.W.; Warmerdam, P.A.; Boeije, L.C.; Arts, J.; Westerdaal, N.A.; Vlug, A.; Capel, P.J.; Aarden, L.A.; van de Winkel, J.G. On the interaction of IgG subclasses with the low affinity Fc gamma RIIa (CD32) on human monocytes, neutrophils, and platelets. Analysis of a functional polymorphism to human IgG2. J. Clin. Investig. 1992, 90, 1537–1546. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.C.; Kim, J.J.; Yun, S.W.; Yu, J.J.; Yoon, K.L.; Lee, K.Y.; Kil, H.R.; Kim, G.B.; Han, M.K.; Song, M.S.; et al. Male-specific association of the FCGR2A His167Arg polymorphism with Kawasaki disease. PLoS ONE 2017, 12, e0184248. [Google Scholar] [CrossRef] [PubMed]
- Sim, B.K.; Park, H.; Kim, J.J.; Yun, S.W.; Yu, J.J.; Yoon, K.L.; Lee, K.Y.; Kil, H.R.; Kim, G.B.; Han, M.K.; et al. Assessment of the Clinical Heterogeneity of Kawasaki Disease Using Genetic Variants of BLK and FCGR2A. Korean Circ. J. 2019, 49, 99–108. [Google Scholar] [CrossRef]
- Nusbaum, J.S.; Mirza, I.; Shum, J.; Freilich, R.W.; Cohen, R.E.; Pillinger, M.H.; Izmirly, P.M.; Buyon, J.P. Sex Differences in Systemic Lupus Erythematosus: Epidemiology, Clinical Considerations, and Disease Pathogenesis. Mayo Clin. Proc. 2020, 95, 384–394. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, C.M.; Cleary, J.; Dagtas, A.S.; Moussai, D.; Diamond, B. Estrogen alters thresholds for B cell apoptosis and activation. J. Clin. Investig. 2002, 109, 1625–1633. [Google Scholar] [CrossRef]
- Kanda, N.; Tamaki, K. Estrogen enhances immunoglobulin production by human PBMCs. J. Allergy Clin. Immunol. 1999, 103, 282–288. [Google Scholar] [CrossRef]
- Ruth, K.S.; Day, F.R.; Tyrrell, J.; Thompson, D.J.; Wood, A.R.; Mahajan, A.; Beaumont, R.N.; Wittemans, L.; Martin, S.; Busch, A.S.; et al. Using human genetics to understand the disease impacts of testosterone in men and women. Nat. Med. 2020, 26, 252–258. [Google Scholar] [CrossRef]
- Sun, B.B.; Maranville, J.C.; Peters, J.E.; Stacey, D.; Staley, J.R.; Blackshaw, J.; Burgess, S.; Jiang, T.; Paige, E.; Surendran, P.; et al. Genomic atlas of the human plasma proteome. Nature 2018, 558, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Tretter, T.; Ross, A.E.; Dordai, D.I.; Desiderio, S. Mimicry of pre-B cell receptor signaling by activation of the tyrosine kinase Blk. J. Exp. Med. 2003, 198, 1863–1873. [Google Scholar] [CrossRef] [PubMed]
- Oliver, P.M.; Wang, M.; Zhu, Y.; White, J.; Kappler, J.; Marrack, P. Loss of Bim allows precursor B cell survival but not precursor B cell differentiation in the absence of interleukin 7. J. Exp. Med. 2004, 200, 1179–1187. [Google Scholar] [CrossRef] [PubMed]



| Infection Hypothesis | Autoimmunity Hypothesis | Hygiene Hypothesis | |
|---|---|---|---|
| Pros |
|
|
|
| Cons |
|
|
| Cell Type | No. of Samples (Acute KD: Convalescent KD: Healthy Controls) | -Fold Change (Compared with Healthy Controls) | p-Value | References | |
|---|---|---|---|---|---|
| Acute KD | Convalescent KD | ||||
| WBC (#) | 33:33:25 | 1.75 ** | 1.05 | <0.01 | [69] |
| 106:68:22 | 1.85 ** | 1.02 | <0.01 | [68] | |
| PBMC (#) | 106:68:22 | 0.94 | 1.08 | ns | [68] |
| Neutrophils (#) | 33:33:25 | 2.54 ** | 0.92 | <0.01 | [69] |
| Immature neutrophils (#) | 33:33:25 | 37 ** | 2.55 | <0.01 | [69] |
| Monocytes (#) | 33:33:25 | 1.58 | 1.06 | ns | [69] |
| 106:68:22 | 2.89 ** | 1.39 | <0.01 | [68] | |
| Lymphocytes (#): | 106:68:22 | 0.87 | 1.04 | ns | [68] |
| CD3+ T cells | 106:68:22 | 0.80 | 1.06 | ns | [68] |
| CD4+ T cells | 106:68:22 | 0.78 * | 0.97 | <0.05 | [68] |
| CD8+ T cells | 106:68:22 | 0.83 * | 1.21 | <0.05 | [68] |
| CD57+ NK cells | 106:68:22 | 0.69 * | 1.66 | <0.05 | [68] |
| CD19+ B cells | 106:68:22 | 1.31 * | 1.06 | <0.05 | [68] |
| IgG (mg/dL) | 33:33:25 | 0.95 | 1.50 ** | ns | [69] |
| CRP (mg/dL) | 33:33:25 | 39 ** | 1 | <0.01 | [69] |
| Gene | SNP | Population | Sample Size (Case:Control) | OR | p-Value | References |
|---|---|---|---|---|---|---|
| BLK | rs2254546 | Japan | 1182:4326 | 1.85 | 8.2 × 10−21 | [119] |
| rs2736340 | Taiwan | 883:1657 | 1.54 | 9.0 × 10−10 | [120] | |
| rs6993775 | Korea | 915:4553 | 1.52 | 2.5 × 10−11 | [121] | |
| BCL2L11 | rs3789065 | Korea | 846:4553 | 1.42 | 4.4 × 10−11 | [122] |
| CD40 | rs4813003 | Japan | 1182:4326 | 1.41 | 4.8 × 10−8 | [119] |
| rs1569723 | Taiwan | 883:1657 | 1.42 | 5.7 × 10−9 | [120] | |
| rs1883834 | Korea | 915:4553 | 1.18 | 0.003912 | unpublished data | |
| FCGR2A | rs1801274 | Multi-ethnic | 1433:7764 | 1.32 | 7.4 × 10−11 | [123] |
| Japan | 1182:4326 | na | 1.6 × 10−6 | [119] | ||
| Korea | 915:4553 | 1.30 | 5.7 × 10−5 | [121] | ||
| IGHV | rs4774175 | Japan, Taiwan, Korea | 3428:7837 | 1.20 | 6.0 × 10−9 | [124] |
| rs6423677 * | Japan | 3603:5731 | 1.25 | 6.8 × 10−10 | [124] |
| Gene | SNP | Risk Allele | Functional Effects of Risk Alleles | References |
|---|---|---|---|---|
| BLK | rs2254546 (A/G) | G | Decreased BLK mRNA expression | [125] |
| BCL2L11 | rs3789065 (C/G) | C | Reduced BCL2L11 mRNA expression | [126] |
| CD40 | rs4813003 (C/T) | C | Enhancement of CD40 function | [118] |
| FCGR2A | rs1801274 (A/G; H167R *) | A | High-affinity receptor leading to immune activation | [118,123] |
| IGHV3-66 | rs6423677 (A/C; p.Cys/Gly) | C | C (risk allele): very high mRNA expression. | [124] |
| Risk Allele of the FCGR2A Variant (rs1801274; A/G = H167R *) | Effect of Risk Allele on IgG Binding | Disease or Trait Associated with Same Risk Allele | OR or Beta | p-Value | References |
|---|---|---|---|---|---|
| A allele encoding His (H) | high affinity | AS | 1.11 | 1 × 10−9 | [148] |
| IBD | 1.13 | 9 × 10−36 | [150] | ||
| IBD | 1.12 | 2 × 10−38 | [152] | ||
| UC | 1.19 | 1 × 10−41 | [150] | ||
| UC | 1.21 | 2 × 10−20 | [151] | ||
| CD | 1.08 | 9 × 10−11 | [150] | ||
| KD | 1.32 | 7 × 10−11 | [123] | ||
| G allele encoding Arg (R) | little or no affinity | SLE | 1.16 | 1 × 10−12 | [149] |
| Basophil count | 0.017 unit increase | 3 × 10−14 | [141] | ||
| Blood FcγRIIa levels | 1.24 unit increase | 1 × 10−2102 | [162] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.-K. Hygiene Hypothesis as the Etiology of Kawasaki Disease: Dysregulation of Early B Cell Development. Int. J. Mol. Sci. 2021, 22, 12334. https://doi.org/10.3390/ijms222212334
Lee J-K. Hygiene Hypothesis as the Etiology of Kawasaki Disease: Dysregulation of Early B Cell Development. International Journal of Molecular Sciences. 2021; 22(22):12334. https://doi.org/10.3390/ijms222212334
Chicago/Turabian StyleLee, Jong-Keuk. 2021. "Hygiene Hypothesis as the Etiology of Kawasaki Disease: Dysregulation of Early B Cell Development" International Journal of Molecular Sciences 22, no. 22: 12334. https://doi.org/10.3390/ijms222212334
APA StyleLee, J.-K. (2021). Hygiene Hypothesis as the Etiology of Kawasaki Disease: Dysregulation of Early B Cell Development. International Journal of Molecular Sciences, 22(22), 12334. https://doi.org/10.3390/ijms222212334






