HATMSC Secreted Factors in the Hydrogel as a Potential Treatment for Chronic Wounds—In Vitro Study
Abstract
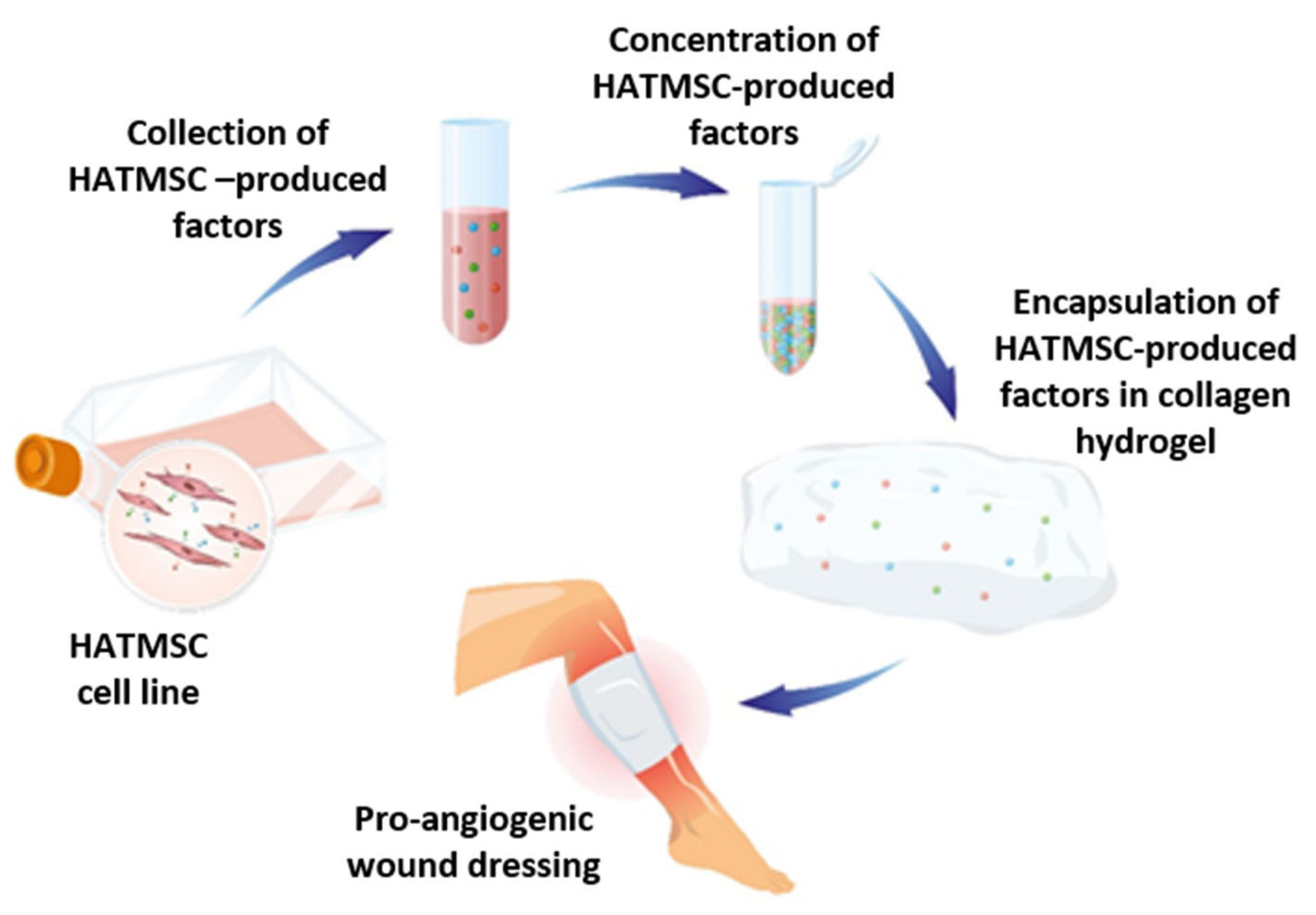
:1. Introduction
2. Results
2.1. Hydrogels Are Not Toxic to Human Skin Origin Cells
2.2. Degradation of Hydrogels
2.3. Bioactivity of Hydrogel-Released HATMSC2-Originated Trophic Factors
2.4. Release of HATMSC2 Supernatant-Present Proteins from Hydrogel
2.5. Pro-Angiogenic Activity of Hydrogel-Released HATMSC2-Originated Trophic Factors
2.6. Antimicrobial Activity of HATMSC Supernatant
3. Discussion
4. Materials and Methods
4.1. HATMSC Supernatant Preparation
4.2. Hydrogel Formation
4.3. Cytotoxicity of Hydrogel
4.4. Hydrogel Degradation
4.5. Hydroxyproline Assay
4.6. Bioactivity of the Released Trophic Factors from Hydrogel Loaded with Supernatant
4.7. Supernatant Release Study
4.8. In Vitro Angiogenesis in Matrigel Matrix
4.9. Examination of MicroRNAs Present in HATMSC Supernatant
4.10. Bacterial Growth and Antimicrobial Activity of HATMSC Supernatants
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Huang, Y.Z.; Gou, M.; Da, L.C.; Zhang, W.Q.; Xie, H.Q. Mesenchymal Stem Cells for Chronic Wound Healing: Current Status of Preclinical and Clinical Studies. Tissue Eng. Part B Rev. 2020, 26, 555–570. [Google Scholar] [CrossRef] [PubMed]
- Otero-Vinas, M.; Falanga, V. Mesenchymal Stem Cells in Chronic Wounds: The Spectrum from Basic to Advanced Therapy. Adv. Wound Care 2016, 5, 149–163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahangar, P.; Mills, S.J.; Cowin, A.J. Mesenchymal Stem Cell Secretome as an Emerging Cell-Free Alternative for Improving Wound Repair. Int. J. Mol. Sci. 2020, 21, 7038. [Google Scholar] [CrossRef] [PubMed]
- Joseph, A.; Baiju, I.; Bhat, I.A.; Pandey, S.; Bharti, M.; Verma, M.; Pratap Singh, A.; Ansari, M.M.; Chandra, V.; Saikumar, G.; et al. Mesenchymal stem cell-conditioned media: A novel alternative of stem cell therapy for quality wound healing. J. Cell Physiol. 2020, 235, 5555–5569. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Zhang, Y.; Song, X.; Zhu, J.; Zhu, Q. The Healing Effects of Conditioned Medium Derived from Mesenchymal Stem Cells on Radiation-Induced Skin Wounds in Rats. Cell Transplant. 2019, 28, 105–115. [Google Scholar] [CrossRef] [Green Version]
- Lobov, A.A.; Yudintceva, N.M.; Mittenberg, A.G.; Shabelnikov, S.V.; Mikhailova, N.A.; Malashicheva, A.B.; Khotin, M.G. Proteomic Profiling of the Human Fetal Multipotent Mesenchymal Stromal Cells Secretome. Molecules 2020, 25, 5283. [Google Scholar] [CrossRef] [PubMed]
- Madonna, R.; Angelucci, S.; Di Giuseppe, F.; Doria, V.; Giricz, Z.; Gorbe, A.; Ferdinandy, P.; De Caterina, R. Proteomic analysis of the secretome of adipose tissue-derived murine mesenchymal cells overexpressing telomerase and myocardin. J. Mol. Cell Cardiol. 2019, 131, 171–186. [Google Scholar] [CrossRef] [PubMed]
- Riis, S.; Stensballe, A.; Emmersen, J.; Pennisi, C.P.; Birkelund, S.; Zachar, V.; Fink, T. Mass spectrometry analysis of adipose-derived stem cells reveals a significant effect of hypoxia on pathways regulating extracellular matrix. Stem Cell Res. Ther. 2016, 7, 52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, S.W.; Kim, K.E.; Choi, J.W.; Lee, C.Y.; Lee, J.; Seo, H.H.; Lim, K.H.; Lim, S.; Lee, S.; Kim, S.W.; et al. Proteomic Analysis and Identification of Paracrine Factors in Mesenchymal Stem Cell-Conditioned Media under Hypoxia. Cell Physiol. Biochem. 2016, 40, 400–410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kraskiewicz, H.; Paprocka, M.; Bielawska-Pohl, A.; Krawczenko, A.; Panek, K.; Kaczynska, J.; Szyposzynska, A.; Psurski, M.; Kuropka, P.; Klimczak, A. Can supernatant from immortalized adipose tissue MSC replace cell therapy? An in vitro study in chronic wounds model. Stem Cell Res. Ther. 2020, 11, 29. [Google Scholar] [CrossRef] [PubMed]
- Alcayaga-Miranda, F.; Cuenca, J.; Khoury, M. Antimicrobial Activity of Mesenchymal Stem Cells: Current Status and New Perspectives of Antimicrobial Peptide-Based Therapies. Front. Immunol. 2017, 8, 339. [Google Scholar] [CrossRef] [PubMed]
- Harman, R.M.; Yang, S.; He, M.K.; Van de Walle, G.R. Antimicrobial peptides secreted by equine mesenchymal stromal cells inhibit the growth of bacteria commonly found in skin wounds. Stem Cell Res. Ther. 2017, 8, 157. [Google Scholar] [CrossRef] [PubMed]
- Pitocco, D.; Spanu, T.; Di Leo, M.; Vitiello, R.; Rizzi, A.; Tartaglione, L.; Fiori, B.; Caputo, S.; Tinelli, G.; Zaccardi, F.; et al. Diabetic foot infections: A comprehensive overview. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Sen, P.; Demirdal, T.; Emir, B. Meta-analysis of risk factors for amputation in diabetic foot infections. Diabetes Metab. Res. Rev. 2019, 35, e3165. [Google Scholar] [CrossRef] [PubMed]
- Serra, R.; Grande, R.; Butrico, L.; Rossi, A.; Settimio, U.F.; Caroleo, B.; Amato, B.; Gallelli, L.; de Franciscis, S. Chronic wound infections: The role of Pseudomonas aeruginosa and Staphylococcus aureus. Expert Rev. Anti Infect. Ther. 2015, 13, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Tong, S.Y.; Davis, J.S.; Eichenberger, E.; Holland, T.L.; Fowler, V.G., Jr. Staphylococcus aureus infections: Epidemiology, pathophysiology, clinical manifestations, and management. Clin. Microbiol. Rev. 2015, 28, 603–661. [Google Scholar] [CrossRef] [Green Version]
- Dong, Y.; Yang, Q.; Sun, X. Comprehensive Analysis of Cell Therapy on Chronic Skin Wound Healing: A Meta-Analysis. Hum. Gene Ther. 2021, 32, 15–16. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Shi, J.; Zhang, M.; Chen, Y.; Wang, X.; Zhang, L.; Tian, Z.; Yan, Y.; Li, Q.; Zhong, W.; et al. Mesenchymal stem cell-laden anti-inflammatory hydrogel enhances diabetic wound healing. Sci. Rep. 2015, 5, 18104. [Google Scholar] [CrossRef]
- Yang, M.; He, S.; Su, Z.; Yang, Z.; Liang, X.; Wu, Y. Thermosensitive Injectable Chitosan/Collagen/beta-Glycerophosphate Composite Hydrogels for Enhancing Wound Healing by Encapsulating Mesenchymal Stem Cell Spheroids. ACS Omega 2020, 5, 21015–21023. [Google Scholar] [CrossRef]
- Hu, P.; Yang, Q.; Wang, Q.; Shi, C.; Wang, D.; Armato, U.; Pra, I.D.; Chiarini, A. Mesenchymal stromal cells-exosomes: A promising cell-free therapeutic tool for wound healing and cutaneous regeneration. Burn. Trauma 2019, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Trzyna, A.; Banas-Zabczyk, A. Adipose-Derived Stem Cells Secretome and Its Potential Application in “Stem Cell-Free Therapy”. Biomolecules 2021, 11, 878. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, S.; Raines, R.T. Review collagen-based biomaterials for wound healing. Biopolymers 2014, 101, 821–833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fleck, C.A.; Simman, R. Modern collagen wound dressings: Function and purpose. J. Am. Col. Certif. Wound Spec. 2010, 2, 50–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gianino, E.; Miller, C.; Gilmore, J. Smart Wound Dressings for Diabetic Chronic Wounds. Bioengineering 2018, 5, 51. [Google Scholar] [CrossRef] [Green Version]
- Sarrigiannidis, S.O.; Rey, J.M.; Dobre, O.; Gonzalez-Garcia, C.; Dalby, M.J.; Salmeron-Sanchez, M. A tough act to follow: Collagen hydrogel modifications to improve mechanical and growth factor loading capabilities. Mater. Today Bio. 2021, 10, 100098. [Google Scholar] [CrossRef] [PubMed]
- Giliomee, J.; du Toit, L.C.; Kumar, P.; Klumperman, B.; Choonara, Y.E. Evaluation of Composition Effects on the Physicochemical and Biological Properties of Polypeptide-Based Hydrogels for Potential Application in Wound Healing. Polymers 2021, 13, 1828. [Google Scholar] [CrossRef] [PubMed]
- Sallent, I.; Capella-Monsonis, H.; Zeugolis, D.I. Production and Characterization of Chemically Cross-Linked Collagen Scaffolds. Methods Mol. Biol. 2019, 1944, 23–38. [Google Scholar] [CrossRef] [PubMed]
- Sargeant, T.D.; Desai, A.P.; Banerjee, S.; Agawu, A.; Stopek, J.B. An in situ forming collagen-PEG hydrogel for tissue regeneration. Acta Biomater. 2012, 8, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Ng, H.W.; Zhang, Y.; Naffa, R.; Prabakar, S. Monitoring the Degradation of Collagen Hydrogels by Collagenase Clostridium histolyticum. Gels 2020, 6, 46. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, K.M.; Han, S.H.; Ko, E.A.; Yoon, D.S.; Park, I.K.; Shin, H.C.; Park, K.H.; Lee, J.W. Development of stabilized dual growth factor-loaded hyaluronate collagen dressing matrix. J. Tissue Eng. 2021, 12, 2041731421999750. [Google Scholar] [CrossRef] [PubMed]
- Lao, G.; Yan, L.; Yang, C.; Zhang, L.; Zhang, S.; Zhou, Y. Controlled release of epidermal growth factor from hydrogels accelerates wound healing in diabetic rats. J. Am. Podiatr. Med. Assoc. 2012, 102, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.N.; Wright, K.T.; Fuller, H.R.; MacNeil, S.; Johnson, W.E. Mesenchymal stem cell-conditioned medium accelerates skin wound healing: An in vitro study of fibroblast and keratinocyte scratch assays. Exp. Cell Res. 2010, 316, 1271–1281. [Google Scholar] [CrossRef] [PubMed]
- Rabbany, S.Y.; Pastore, J.; Yamamoto, M.; Miller, T.; Rafii, S.; Aras, R.; Penn, M. Continuous delivery of stromal cell-derived factor-1 from alginate scaffolds accelerates wound healing. Cell Transplant. 2010, 19, 399–408. [Google Scholar] [CrossRef] [Green Version]
- Lin, Y.J.; Lee, G.H.; Chou, C.W.; Chen, Y.P.; Wu, T.H.; Lin, H.R. Stimulation of wound healing by PU/hydrogel composites containing fibroblast growth factor-2. J. Mater. Chem. B 2015, 3, 1931–1941. [Google Scholar] [CrossRef] [PubMed]
- Bhansali, A.; Venkatesh, S.; Dutta, P.; Dhillon, M.S.; Das, S.; Agrawal, A. Which is the better option: Recombinant human PDGF-BB 0.01% gel or standard wound care, in diabetic neuropathic large plantar ulcers off-loaded by a customized contact cast? Diabetes Res. Clin. Pract. 2009, 83, e13–e16. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Shim, S.; Jang, H.; Myung, H.; Lee, J.; Bae, C.H.; Myung, J.K.; Kim, M.J.; Lee, S.B.; Jang, W.S.; et al. Human umbilical cord blood-derived mesenchymal stromal cells and small intestinal submucosa hydrogel composite promotes combined radiation-wound healing of mice. Cytotherapy 2017, 19, 1048–1059. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Li, X.; Zhang, B.; Shi, Q.; Li, D.; Ju, X. A Human Umbilical Cord Mesenchymal Stem Cell-Conditioned Medium/Chitosan/Collagen/beta-Glycerophosphate Thermosensitive Hydrogel Promotes Burn Injury Healing in Mice. Biomed. Res. Int. 2019, 2019, 5768285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krawczenko, A.; Bielawska-Pohl, A.; Paprocka, M.; Kraskiewicz, H.; Szyposzynska, A.; Wojdat, E.; Klimczak, A. Microvesicles from Human Immortalized Cell Lines of Endothelial Progenitor Cells and Mesenchymal Stem/Stromal Cells of Adipose Tissue Origin as Carriers of Bioactive Factors Facilitating Angiogenesis. Stem Cells Int. 2020, 2020, 1289380. [Google Scholar] [CrossRef]
- Liang, X.; Lin, F.; Ding, Y.; Zhang, Y.; Li, M.; Zhou, X.; Meng, Q.; Ma, X.; Wei, L.; Fan, H.; et al. Conditioned medium from induced pluripotent stem cell-derived mesenchymal stem cells accelerates cutaneous wound healing through enhanced angiogenesis. Stem Cell Res. Ther. 2021, 12, 295. [Google Scholar] [CrossRef] [PubMed]
- Cortes-Araya, Y.; Amilon, K.; Rink, B.E.; Black, G.; Lisowski, Z.; Donadeu, F.X.; Esteves, C.L. Comparison of Antibacterial and Immunological Properties of Mesenchymal Stem/Stromal Cells from Equine Bone Marrow, Endometrium, and Adipose Tissue. Stem Cells Dev. 2018, 27, 1518–1525. [Google Scholar] [CrossRef] [PubMed]
- Krasnodembskaya, A.; Song, Y.; Fang, X.; Gupta, N.; Serikov, V.; Lee, J.W.; Matthay, M.A. Antibacterial effect of human mesenchymal stem cells is mediated in part from secretion of the antimicrobial peptide LL-37. Stem Cells 2010, 28, 2229–2238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kouhkheil, R.; Fridoni, M.; Piryaei, A.; Taheri, S.; Chirani, A.S.; Anarkooli, I.J.; Nejatbakhsh, R.; Shafikhani, S.; Schuger, L.A.; Reddy, V.B.; et al. The effect of combined pulsed wave low-level laser therapy and mesenchymal stem cell-conditioned medium on the healing of an infected wound with methicillin-resistant Staphylococcal aureus in diabetic rats. J. Cell Biochem. 2018, 119, 5788–5797. [Google Scholar] [CrossRef] [PubMed]
- Brogden, K.A. Antimicrobial peptides: Pore formers or metabolic inhibitors in bacteria? Nat. Rev. Microbiol. 2005, 3, 238–250. [Google Scholar] [CrossRef] [PubMed]
- Hurlin, P.J.; Maher, V.M.; McCormick, J.J. Malignant transformation of human fibroblasts caused by expression of a transfected T24 HRAS oncogene. Proc. Natl. Acad. Sci. USA 1989, 86, 187–191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kieda, C.; Paprocka, M.; Krawczenko, A.; Zalecki, P.; Dupuis, P.; Monsigny, M.; Radzikowski, C.; Dus, D. New human microvascular endothelial cell lines with specific adhesion molecules phenotypes. Endothelium 2002, 9, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Glynn, C.L.; Khan, S.; Kerin, M.J.; Dwyer, R.M. Isolation of secreted microRNAs (miRNAs) from cell-conditioned media. Microrna 2013, 2, 14–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cao, Z.; Zhang, J.; Niu, Y.D.; Cui, N.; Ma, Y.; Cao, F.; Jin, L.; Li, Z.; Xu, Y. Isolation and characterization of a “phiKMV-like” bacteriophage and its therapeutic effect on mink hemorrhagic pneumonia. PLoS ONE 2015, 10, e0116571. [Google Scholar] [CrossRef] [PubMed]
- Hyman, P. Phages for Phage Therapy: Isolation, Characterization, and Host Range Breadth. Pharmaceuticals 2019, 12, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]







| Bacterial Strains | HATMSC1 | HATMSC2 | HATMSC2D10 | HATMSC2F10 | Control | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 h * | 3 h * | 0 h | 3 h | 0 h | 3 h | 0 h | 3 h | 0 h | 3 h | ||
| Gram-positive bacteria | Staphylococcus aureus MRSA PCM 3144 | +++ | +++ | +++ | +++ | +++ | +++ | +++ | +++ | - | - |
| Staphylococcus aureus PCM 519 | ++ | +++ | +++ | +++ | +++ | +++ | +++ | +++ | - | - | |
| Staphylococcus aureus Covan PCM 2101 | +++ | +++ | ++ | +++ | ++ | +++ | ++ | +++ | - | - | |
| Staphylococcus epidermidis PCM 2651 | +++ | +++ | +++ | +++ | +++ | +++ | +++ | +++ | - | - | |
| Gram-negative bacteria | Escherichia coli O104 PCM 270 | +++ | +++ | +++ | +++ | +++ | +++ | +++ | +++ | - | - |
| Escherichia coli PCM 1144 | +++ | +++ | +++ | +++ | +++ | +++ | +++ | +++ | - | - | |
| Pseudomonas aeruginosa PCM 2270 | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | - | - | |
| Pseudomonas aeruginosa PCM 2058 | + | + | + | + | + | + | + | + | - | - | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kraskiewicz, H.; Hinc, P.; Krawczenko, A.; Bielawska-Pohl, A.; Paprocka, M.; Witkowska, D.; Mohd Isa, I.L.; Pandit, A.; Klimczak, A. HATMSC Secreted Factors in the Hydrogel as a Potential Treatment for Chronic Wounds—In Vitro Study. Int. J. Mol. Sci. 2021, 22, 12241. https://doi.org/10.3390/ijms222212241
Kraskiewicz H, Hinc P, Krawczenko A, Bielawska-Pohl A, Paprocka M, Witkowska D, Mohd Isa IL, Pandit A, Klimczak A. HATMSC Secreted Factors in the Hydrogel as a Potential Treatment for Chronic Wounds—In Vitro Study. International Journal of Molecular Sciences. 2021; 22(22):12241. https://doi.org/10.3390/ijms222212241
Chicago/Turabian StyleKraskiewicz, Honorata, Piotr Hinc, Agnieszka Krawczenko, Aleksandra Bielawska-Pohl, Maria Paprocka, Danuta Witkowska, Isma Liza Mohd Isa, Abhay Pandit, and Aleksandra Klimczak. 2021. "HATMSC Secreted Factors in the Hydrogel as a Potential Treatment for Chronic Wounds—In Vitro Study" International Journal of Molecular Sciences 22, no. 22: 12241. https://doi.org/10.3390/ijms222212241






