Angptl2 is a Marker of Cellular Senescence: The Physiological and Pathophysiological Impact of Angptl2-Related Senescence
Abstract
1. Introduction
2. Angptl2 Is a Marker of Cellular Senescence
2.1. Identification of Angptl2 in Senescent Cells
2.2. Transcriptomic and Proteomic Analyses That Identified Angptl2 as a SASP Factor
2.3. In Vivo Experimental Evidence That Angptl2 Is a Senescence Marker
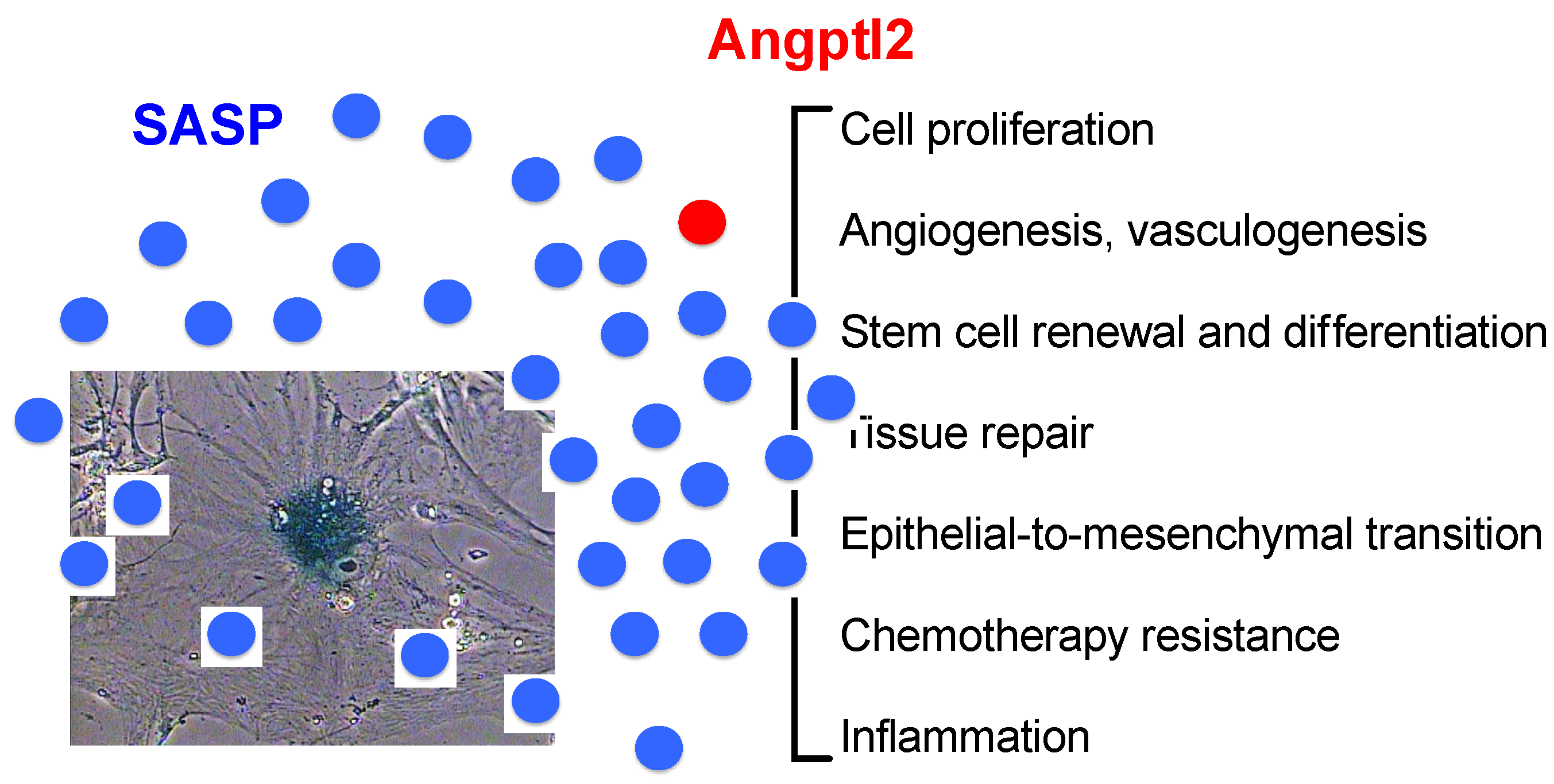
2.4. Similar Functions of the SASP and of Angptl2
3. Physiological Roles of Angptl2-Related Senescence
3.1. Potential Involvement of Angptl2-Related Senescence in Embryogenesis and Development
3.2. Physiological Impact of Angptl2-Induced Senescence in Tissue Regeneration and Repair
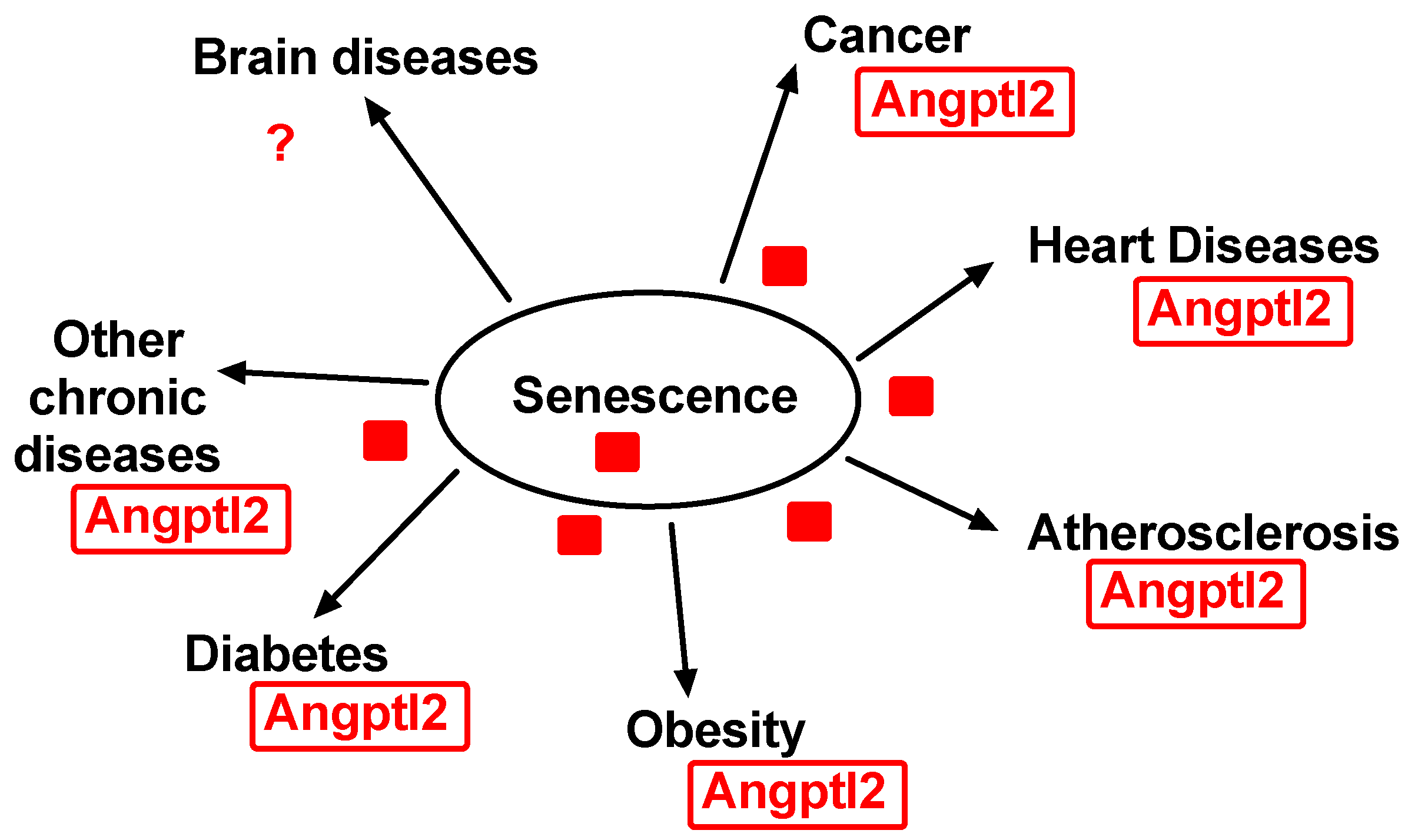
4. Pathological Impact of Angptl2-Related Senescence
5. Clinical Utility of Angptl2, a Potential Senescent Biomarker of Age-Related Diseases
6. Limitations
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Childs, B.G.; Durik, M.; Baker, D.J.; van Deursen, J.M. Cellular senescence in aging and age-related disease: From mechanisms to therapy. Nat. Med. 2015, 21, 1424–1435. [Google Scholar] [CrossRef]
- Campisi, J. Aging, cellular senescence, and cancer. Annu. Rev. Physiol. 2013, 75, 685–705. [Google Scholar] [CrossRef] [PubMed]
- Rodier, F.; Coppe, J.P.; Patil, C.K.; Hoeijmakers, W.A.; Munoz, D.P.; Raza, S.R.; Freund, A.; Campeau, E.; Davalos, A.R.; Campisi, J. Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nat. Cell Biol. 2009, 11, 973–979. [Google Scholar] [CrossRef]
- Rodier, F.; Munoz, D.P.; Teachenor, R.; Chu, V.; Le, O.; Bhaumik, D.; Coppe, J.P.; Campeau, E.; Beausejour, C.M.; Kim, S.H.; et al. DNA-SCARS: Distinct nuclear structures that sustain damage-induced senescence growth arrest and inflammatory cytokine secretion. J. Cell Sci. 2011, 124, 68–81. [Google Scholar] [CrossRef]
- Beausejour, C.M.; Krtolica, A.; Galimi, F.; Narita, M.; Lowe, S.W.; Yaswen, P.; Campisi, J. Reversal of human cellular senescence: Roles of the p53 and p16 pathways. EMBO J. 2003, 22, 4212–4222. [Google Scholar] [CrossRef] [PubMed]
- Passos, J.F.; Nelson, G.; Wang, C.; Richter, T.; Simillion, C.; Proctor, C.J.; Miwa, S.; Olijslagers, S.; Hallinan, J.; Wipat, A.; et al. Feedback between p21 and reactive oxygen production is necessary for cell senescence. Mol. Syst. Biol. 2010, 6, 347. [Google Scholar] [CrossRef] [PubMed]
- Freund, A.; Orjalo, A.V.; Desprez, P.Y.; Campisi, J. Inflammatory networks during cellular senescence: Causes and consequences. Trends Mol. Med. 2010, 16, 238–246. [Google Scholar] [CrossRef]
- Ressler, S.; Bartkova, J.; Niederegger, H.; Bartek, J.; Scharffetter-Kochanek, K.; Jansen-Durr, P.; Wlaschek, M. p16INK4A is a robust in vivo biomarker of cellular aging in human skin. Aging Cell 2006, 5, 379–389. [Google Scholar] [CrossRef]
- Dimri, G.P.; Lee, X.; Basile, G.; Acosta, M.; Scott, G.; Roskelley, C.; Medrano, E.E.; Linskens, M.; Rubelj, I.; Pereira-Smith, O.; et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc. Natl. Acad. Sci. USA 1995, 92, 9363–9367. [Google Scholar] [CrossRef]
- Ovadya, Y.; Krizhanovsky, V. Senescent cells: SASPected drivers of age-related pathologies. Biogerontology 2014, 15, 627–642. [Google Scholar] [CrossRef]
- Freund, A.; Laberge, R.M.; Demaria, M.; Campisi, J. Lamin B1 loss is a senescence-associated biomarker. Mol. Biol. Cell 2012, 23, 2066–2075. [Google Scholar] [CrossRef] [PubMed]
- Coppe, J.P.; Patil, C.K.; Rodier, F.; Sun, Y.; Munoz, D.P.; Goldstein, J.; Nelson, P.S.; Desprez, P.Y.; Campisi, J. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008, 6, e301. [Google Scholar] [CrossRef] [PubMed]
- Kuilman, T.; Peeper, D.S. Senescence-messaging secretome: SMS-ing cellular stress. Nat. Rev. Cancer 2009, 9, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Faget, D.V.; Ren, Q.; Stewart, S.A. Unmasking senescence: Context-dependent effects of SASP in cancer. Nat. Rev. Cancer 2019, 19, 439–453. [Google Scholar] [CrossRef] [PubMed]
- Malaquin, N.; Martinez, A.; Rodier, F. Keeping the senescence secretome under control: Molecular reins on the senescence-associated secretory phenotype. Exp. Gerontol. 2016, 82, 39–49. [Google Scholar] [CrossRef]
- Ito, Y.; Hoare, M.; Narita, M. Spatial and Temporal Control of Senescence. Trends Cell Biol. 2017, 27, 820–832. [Google Scholar] [CrossRef]
- Orjalo, A.V.; Bhaumik, D.; Gengler, B.K.; Scott, G.K.; Campisi, J. Cell surface-bound IL-1alpha is an upstream regulator of the senescence-associated IL-6/IL-8 cytokine network. Proc. Natl. Acad. Sci. USA 2009, 106, 17031–17036. [Google Scholar] [CrossRef]
- Tominaga, K. The emerging role of senescent cells in tissue homeostasis and pathophysiology. Pathobiol. Aging Age Relat. Dis. 2015, 5, 27743. [Google Scholar] [CrossRef]
- Nelson, G.; Wordsworth, J.; Wang, C.; Jurk, D.; Lawless, C.; Martin-Ruiz, C.; von Zglinicki, T. A senescent cell bystander effect: Senescence-induced senescence. Aging Cell 2012, 11, 345–349. [Google Scholar] [CrossRef]
- Braig, M.; Schmitt, C.A. Oncogene-induced senescence: Putting the brakes on tumor development. Cancer Res. 2006, 66, 2881–2884. [Google Scholar] [CrossRef]
- Soto-Gamez, A.; Demaria, M. Therapeutic interventions for aging: The case of cellular senescence. Drug Discov. Today 2017, 22, 786–795. [Google Scholar] [CrossRef]
- Kadomatsu, T.; Endo, M.; Miyata, K.; Oike, Y. Diverse roles of ANGPTL2 in physiology and pathophysiology. Trends Endocrinol. Metab. 2014, 25, 245–254. [Google Scholar] [CrossRef]
- Thorin-Trescases, N.; Thorin, E. Angiopoietin-like-2: A multifaceted protein with physiological and pathophysiological properties. Expert Rev. Mol. Med. 2014, 16, e17. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.; Moon, S.O.; Koh, K.N.; Kim, H.; Uhm, C.S.; Kwak, H.J.; Kim, N.G.; Koh, G.Y. Molecular cloning, expression, and characterization of angiopoietin-related protein. angiopoietin-related protein induces endothelial cell sprouting. J. Biol. Chem. 1999, 274, 26523–26528. [Google Scholar] [CrossRef] [PubMed]
- Kadomatsu, T.; Tabata, M.; Oike, Y. Angiopoietin-like proteins: Emerging targets for treatment of obesity and related metabolic diseases. FEBS J. 2011, 278, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Tabata, M.; Kadomatsu, T.; Fukuhara, S.; Miyata, K.; Ito, Y.; Endo, M.; Urano, T.; Zhu, H.J.; Tsukano, H.; Tazume, H.; et al. Angiopoietin-like protein 2 promotes chronic adipose tissue inflammation and obesity-related systemic insulin resistance. Cell Metab. 2009, 10, 178–188. [Google Scholar] [CrossRef]
- Broxmeyer, H.E.; Srour, E.F.; Cooper, S.; Wallace, C.T.; Hangoc, G.; Youn, B.S. Angiopoietin-like-2 and -3 act through their coiled-coil domains to enhance survival and replating capacity of human cord blood hematopoietic progenitors. Blood Cells Mol. Dis. 2012, 48, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Kubota, Y.; Oike, Y.; Satoh, S.; Tabata, Y.; Niikura, Y.; Morisada, T.; Akao, M.; Urano, T.; Ito, Y.; Miyamoto, T.; et al. Cooperative interaction of Angiopoietin-like proteins 1 and 2 in zebrafish vascular development. Proc. Natl. Acad. Sci. USA 2005, 102, 13502–13507. [Google Scholar] [CrossRef]
- Horio, E.; Kadomatsu, T.; Miyata, K.; Arai, Y.; Hosokawa, K.; Doi, Y.; Ninomiya, T.; Horiguchi, H.; Endo, M.; Tabata, M.; et al. Role of endothelial cell-derived angptl2 in vascular inflammation leading to endothelial dysfunction and atherosclerosis progression. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 790–800. [Google Scholar] [CrossRef]
- Zheng, J.; Umikawa, M.; Cui, C.; Li, J.; Chen, X.; Zhang, C.; Huynh, H.; Kang, X.; Silvany, R.; Wan, X.; et al. Inhibitory receptors bind ANGPTLs and support blood stem cells and leukaemia development. Nature 2012, 485, 656–660. [Google Scholar] [CrossRef]
- Fan, X.; Shi, P.; Dai, J.; Lu, Y.; Chen, X.; Liu, X.; Zhang, K.; Wu, X.; Sun, Y.; Wang, K.; et al. Paired immunoglobulin-like receptor B regulates platelet activation. Blood 2014, 124, 2421–2430. [Google Scholar] [CrossRef] [PubMed]
- Carbone, C.; Piro, G.; Fassan, M.; Tamburrino, A.; Mina, M.M.; Zanotto, M.; Chiao, P.J.; Bassi, C.; Scarpa, A.; Tortora, G.; et al. An angiopoietin-like protein 2 autocrine signaling promotes EMT during pancreatic ductal carcinogenesis. Oncotarget 2015, 6, 13822–13834. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Liu, J.; Chen, G.; Du, J.; Cai, H.; Chen, X.; Ye, G.; Luo, Y.; Luo, Y.; Zhang, L.; et al. CD146 is a Novel ANGPTL2 Receptor that Promotes Obesity by Manipulating Lipid Metabolism and Energy Expenditure. Adv. Sci. 2021, 8, 2004032. [Google Scholar] [CrossRef]
- Guo, D.F.; Chenier, I.; Tardif, V.; Orlov, S.N.; Inagami, T. Type 1 angiotensin II receptor-associated protein ARAP1 binds and recycles the receptor to the plasma membrane. Biochem. Biophys. Res. Commun. 2003, 310, 1254–1265. [Google Scholar] [CrossRef]
- Guo, D.F.; Sun, Y.L.; Hamet, P.; Inagami, T. The angiotensin II type 1 receptor and receptor-associated proteins. Cell Res. 2001, 11, 165–180. [Google Scholar] [CrossRef]
- Thorin-Trescases, N.; Thorin, E. High Circulating Levels of ANGPTL2: Beyond a Clinical Marker of Systemic Inflammation. Oxid Med. Cell. Longev. 2017, 2017, 1096385. [Google Scholar] [CrossRef]
- Shimomura, M.; Oyama, J.; Takeuchi, M.; Shibata, Y.; Yamamoto, Y.; Kawasaki, T.; Komoda, H.; Kodama, K.; Sakuma, M.; Toyoda, S.; et al. Acute effects of statin on reduction of angiopoietin-like 2 and glyceraldehyde-derived advanced glycation end-products levels in patients with acute myocardial infarction: A message from SAMIT (Statin for Acute Myocardial Infarction Trial). Heart Vessels 2016, 31, 1583–1589. [Google Scholar] [CrossRef]
- Muramoto, A.; Tsushita, K.; Kato, A.; Ozaki, N.; Tabata, M.; Endo, M.; Oike, Y.; Oiso, Y. Angiopoietin-like protein 2 sensitively responds to weight reduction induced by lifestyle intervention on overweight Japanese men. Nutr. Diabetes 2011, 1, e20. [Google Scholar] [CrossRef] [PubMed]
- Thorin-Trescases, N.; Hayami, D.; Yu, C.; Luo, X.; Nguyen, A.; Larouche, J.F.; Lalonge, J.; Henri, C.; Arsenault, A.; Gayda, M.; et al. Exercise Lowers Plasma Angiopoietin-Like 2 in Men with Post-Acute Coronary Syndrome. PLoS ONE 2016, 11, e0164598. [Google Scholar] [CrossRef]
- Park, J.; Choi, Y.; Mizushima, R.; Yoshikawa, T.; Myoenzono, K.; Tagawa, K.; Matsui, M.; Tanaka, K.; Maeda, S. Dietary modification reduces serum angiopoietin-like protein 2 levels and arterial stiffness in overweight and obese men. J. Exerc. Nutr. Biochem. 2019, 23, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Hato, T.; Tabata, M.; Oike, Y. The role of angiopoietin-like proteins in angiogenesis and metabolism. Trends Cardiovasc. Med. 2008, 18, 6–14. [Google Scholar] [CrossRef]
- Oike, Y.; Yasunaga, K.; Suda, T. Angiopoietin-related/angiopoietin-like proteins regulate angiogenesis. Int. J. Hematol. 2004, 80, 21–28. [Google Scholar] [CrossRef]
- Buga, A.M.; Margaritescu, C.; Scholz, C.J.; Radu, E.; Zelenak, C.; Popa-Wagner, A. Transcriptomics of post-stroke angiogenesis in the aged brain. Front. Aging Neurosci. 2014, 6, 44. [Google Scholar] [CrossRef] [PubMed]
- Richardson, M.R.; Robbins, E.P.; Vemula, S.; Critser, P.J.; Whittington, C.; Voytik-Harbin, S.L.; Yoder, M.C. Angiopoietin-like protein 2 regulates endothelial colony forming cell vasculogenesis. Angiogenesis 2014, 17, 675–683. [Google Scholar] [CrossRef] [PubMed]
- Akhter, S.; Rahman, M.M.; Lee, H.S.; Kim, H.J.; Hong, S.T. Dynamic roles of angiopoietin-like proteins 1, 2, 3, 4, 6 and 7 in the survival and enhancement of ex vivo expansion of bone-marrow hematopoietic stem cells. Protein Cell 2013, 4, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.I.; Price, E.N.; Boatman, S.; Hagedorn, E.J.; Trompouki, E.; Satishchandran, S.; Carspecken, C.W.; Uong, A.; DiBiase, A.; Yang, S.; et al. Angiopoietin-like proteins stimulate HSPC development through interaction with notch receptor signaling. Elife 2015, 4, e05544. [Google Scholar] [CrossRef]
- Zhang, C.C.; Kaba, M.; Ge, G.; Xie, K.; Tong, W.; Hug, C.; Lodish, H.F. Angiopoietin-like proteins stimulate ex vivo expansion of hematopoietic stem cells. Nat. Med. 2006, 12, 240–245. [Google Scholar] [CrossRef]
- Kadomatsu, T.; Oike, Y. Roles of angiopoietin-like proteins in regulation of stem cell activity. J. Biochem. 2019, 165, 309–315. [Google Scholar] [CrossRef]
- Horiguchi, H.; Endo, M.; Kawane, K.; Kadomatsu, T.; Terada, K.; Morinaga, J.; Araki, K.; Miyata, K.; Oike, Y. ANGPTL2 expression in the intestinal stem cell niche controls epithelial regeneration and homeostasis. EMBO J. 2017, 36, 409–424. [Google Scholar] [CrossRef]
- Motokawa, I.; Endo, M.; Terada, K.; Horiguchi, H.; Miyata, K.; Kadomatsu, T.; Morinaga, J.; Sugizaki, T.; Ito, T.; Araki, K.; et al. Interstitial pneumonia induced by bleomycin treatment is exacerbated in Angptl2-deficient mice. Am. J. Physiol. Lung Cell Mol. Physiol. 2016, 311, L704–L713. [Google Scholar] [CrossRef]
- Yugami, M.; Odagiri, H.; Endo, M.; Tsutsuki, H.; Fujii, S.; Kadomatsu, T.; Masuda, T.; Miyata, K.; Terada, K.; Tanoue, H.; et al. Mice Deficient in Angiopoietin-like Protein 2 (Angptl2) Gene Show Increased Susceptibility to Bacterial Infection Due to Attenuated Macrophage Activity. J. Biol. Chem. 2016, 291, 18843–18852. [Google Scholar] [CrossRef]
- Takano, A.; Fukuda, T.; Shinjo, T.; Iwashita, M.; Matsuzaki, E.; Yamamichi, K.; Takeshita, M.; Sanui, T.; Nishimura, F. Angiopoietin-like protein 2 is a positive regulator of osteoblast differentiation. Metabolism 2017, 69, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Farhat, N.; Thorin-Trescases, N.; Mamarbachi, M.; Villeneuve, L.; Yu, C.; Martel, C.; Duquette, N.; Gayda, M.; Nigam, A.; Juneau, M.; et al. Angiopoietin-like 2 promotes atherogenesis in mice. J. Am. Heart Assoc. 2013, 2, e000201. [Google Scholar] [CrossRef]
- Morinaga, J.; Kadomatsu, T.; Miyata, K.; Endo, M.; Terada, K.; Tian, Z.; Sugizaki, T.; Tanigawa, H.; Zhao, J.; Zhu, S.; et al. Angiopoietin-like protein 2 increases renal fibrosis by accelerating transforming growth factor-beta signaling in chronic kidney disease. Kidney Int. 2016, 89, 327–341. [Google Scholar] [CrossRef] [PubMed]
- Aoi, J.; Endo, M.; Kadomatsu, T.; Miyata, K.; Nakano, M.; Horiguchi, H.; Ogata, A.; Odagiri, H.; Yano, M.; Araki, K.; et al. Angiopoietin-like protein 2 is an important facilitator of inflammatory carcinogenesis and metastasis. Cancer Res. 2011, 71, 7502–7512. [Google Scholar] [CrossRef]
- Aoi, J.; Endo, M.; Kadomatsu, T.; Miyata, K.; Ogata, A.; Horiguchi, H.; Odagiri, H.; Masuda, T.; Fukushima, S.; Jinnin, M.; et al. Angiopoietin-like protein 2 accelerates carcinogenesis by activating chronic inflammation and oxidative stress. Mol. Cancer Res. 2014, 12, 239–249. [Google Scholar] [CrossRef]
- Tazume, H.; Miyata, K.; Tian, Z.; Endo, M.; Horiguchi, H.; Takahashi, O.; Horio, E.; Tsukano, H.; Kadomatsu, T.; Nakashima, Y.; et al. Macrophage-derived angiopoietin-like protein 2 accelerates development of abdominal aortic aneurysm. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 1400–1409. [Google Scholar] [CrossRef]
- Zhang, H.; Pan, K.H.; Cohen, S.N. Senescence-specific gene expression fingerprints reveal cell-type-dependent physical clustering of up-regulated chromosomal loci. Proc. Natl. Acad. Sci. USA 2003, 100, 3251–3256. [Google Scholar] [CrossRef] [PubMed]
- Farhat, N.; Thorin-Trescases, N.; Voghel, G.; Villeneuve, L.; Mamarbachi, M.; Perrault, L.P.; Carrier, M.; Thorin, E. Stress-induced senescence predominates in endothelial cells isolated from atherosclerotic chronic smokers. Can. J. Physiol. Pharmacol. 2008, 86, 761–769. [Google Scholar] [CrossRef]
- Voghel, G.; Thorin-Trescases, N.; Farhat, N.; Mamarbachi, A.M.; Villeneuve, L.; Fortier, A.; Perrault, L.P.; Carrier, M.; Thorin, E. Chronic treatment with N-acetyl-cystein delays cellular senescence in endothelial cells isolated from a subgroup of atherosclerotic patients. Mech. Ageing Dev. 2008, 129, 261–270. [Google Scholar] [CrossRef]
- Voghel, G.; Thorin-Trescases, N.; Farhat, N.; Nguyen, A.; Villeneuve, L.; Mamarbachi, A.M.; Fortier, A.; Perrault, L.P.; Carrier, M.; Thorin, E. Cellular senescence in endothelial cells from atherosclerotic patients is accelerated by oxidative stress associated with cardiovascular risk factors. Mech. Ageing Dev. 2007, 128, 662–671. [Google Scholar] [CrossRef]
- Voghel, G.; Thorin-Trescases, N.; Mamarbachi, A.M.; Villeneuve, L.; Mallette, F.A.; Ferbeyre, G.; Farhat, N.; Perrault, L.P.; Carrier, M.; Thorin, E. Endogenous oxidative stress prevents telomerase-dependent immortalization of human endothelial cells. Mech. Ageing Dev. 2010, 131, 354–363. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Acosta, J.C.; Banito, A.; Wuestefeld, T.; Georgilis, A.; Janich, P.; Morton, J.P.; Athineos, D.; Kang, T.W.; Lasitschka, F.; Andrulis, M.; et al. A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nat. Cell Biol. 2013, 15, 978–990. [Google Scholar] [CrossRef]
- Perrigue, P.M.; Rakoczy, M.; Pawlicka, K.P.; Belter, A.; Giel-Pietraszuk, M.; Naskret-Barciszewska, M.; Barciszewski, J.; Figlerowicz, M. Cancer Stem Cell-Inducing Media Activates Senescence Reprogramming in Fibroblasts. Cancers 2020, 12, 1745. [Google Scholar] [CrossRef] [PubMed]
- Casella, G.; Munk, R.; Kim, K.M.; Piao, Y.; De, S.; Abdelmohsen, K.; Gorospe, M. Transcriptome signature of cellular senescence. Nucleic Acids Res. 2019, 47, 7294–7305. [Google Scholar] [CrossRef]
- Sole-Boldo, L.; Raddatz, G.; Schutz, S.; Mallm, J.P.; Rippe, K.; Lonsdorf, A.S.; Rodriguez-Paredes, M.; Lyko, F. Single-cell transcriptomes of the human skin reveal age-related loss of fibroblast priming. Commun. Biol. 2020, 3, 188. [Google Scholar] [CrossRef] [PubMed]
- Shimamoto, A.; Kagawa, H.; Zensho, K.; Sera, Y.; Kazuki, Y.; Osaki, M.; Oshimura, M.; Ishigaki, Y.; Hamasaki, K.; Kodama, Y.; et al. Reprogramming suppresses premature senescence phenotypes of Werner syndrome cells and maintains chromosomal stability over long-term culture. PLoS ONE 2014, 9, e112900. [Google Scholar] [CrossRef]
- Marthandan, S.; Priebe, S.; Baumgart, M.; Groth, M.; Cellerino, A.; Guthke, R.; Hemmerich, P.; Diekmann, S. Similarities in Gene Expression Profiles during In Vitro Aging of Primary Human Embryonic Lung and Foreskin Fibroblasts. Biomed. Res. Int. 2015, 2015, 731938. [Google Scholar] [CrossRef]
- Marthandan, S.; Baumgart, M.; Priebe, S.; Groth, M.; Schaer, J.; Kaether, C.; Guthke, R.; Cellerino, A.; Platzer, M.; Diekmann, S.; et al. Conserved Senescence Associated Genes and Pathways in Primary Human Fibroblasts Detected by RNA-Seq. PLoS ONE 2016, 11, e0154531. [Google Scholar] [CrossRef]
- Teo, Y.V.; Rattanavirotkul, N.; Olova, N.; Salzano, A.; Quintanilla, A.; Tarrats, N.; Kiourtis, C.; Muller, M.; Green, A.R.; Adams, P.D.; et al. Notch Signaling Mediates Secondary Senescence. Cell Rep. 2019, 27, 997–1007 e1005. [Google Scholar] [CrossRef]
- Yildiz, G.; Arslan-Ergul, A.; Bagislar, S.; Konu, O.; Yuzugullu, H.; Gursoy-Yuzugullu, O.; Ozturk, N.; Ozen, C.; Ozdag, H.; Erdal, E.; et al. Genome-wide transcriptional reorganization associated with senescence-to-immortality switch during human hepatocellular carcinogenesis. PLoS ONE 2013, 8, e64016. [Google Scholar] [CrossRef]
- Nagano, T.; Nakano, M.; Nakashima, A.; Onishi, K.; Yamao, S.; Enari, M.; Kikkawa, U.; Kamada, S. Identification of cellular senescence-specific genes by comparative transcriptomics. Sci. Rep. 2016, 6, 31758. [Google Scholar] [CrossRef] [PubMed]
- Eggert, T.; Wolter, K.; Ji, J.; Ma, C.; Yevsa, T.; Klotz, S.; Medina-Echeverz, J.; Longerich, T.; Forgues, M.; Reisinger, F.; et al. Distinct Functions of Senescence-Associated Immune Responses in Liver Tumor Surveillance and Tumor Progression. Cancer Cell 2016, 30, 533–547. [Google Scholar] [CrossRef] [PubMed]
- Crowe, E.P.; Tuzer, F.; Gregory, B.D.; Donahue, G.; Gosai, S.J.; Cohen, J.; Leung, Y.Y.; Yetkin, E.; Nativio, R.; Wang, L.S.; et al. Changes in the Transcriptome of Human Astrocytes Accompanying Oxidative Stress-Induced Senescence. Front. Aging Neurosci. 2016, 8, 208. [Google Scholar] [CrossRef]
- Limbad, C.; Oron, T.R.; Alimirah, F.; Davalos, A.R.; Tracy, T.E.; Gan, L.; Desprez, P.Y.; Campisi, J. Astrocyte senescence promotes glutamate toxicity in cortical neurons. PLoS ONE 2020, 15, e0227887. [Google Scholar] [CrossRef]
- Burton, D.G.; Giles, P.J.; Sheerin, A.N.; Smith, S.K.; Lawton, J.J.; Ostler, E.L.; Rhys-Williams, W.; Kipling, D.; Faragher, R.G. Microarray analysis of senescent vascular smooth muscle cells: A link to atherosclerosis and vascular calcification. Exp. Gerontol. 2009, 44, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Kiss, T.; Nyul-Toth, A.; Balasubramanian, P.; Tarantini, S.; Ahire, C.; DelFavero, J.; Yabluchanskiy, A.; Csipo, T.; Farkas, E.; Wiley, G.; et al. Single-cell RNA sequencing identifies senescent cerebromicrovascular endothelial cells in the aged mouse brain. Geroscience 2020, 42, 429–444. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, S.; Yan, P.; Ren, J.; Song, M.; Li, J.; Lei, J.; Pan, H.; Wang, S.; Ma, X.; et al. A single-cell transcriptomic landscape of primate arterial aging. Nat. Commun. 2020, 11, 2202. [Google Scholar] [CrossRef]
- Chatsirisupachai, K.; Palmer, D.; Ferreira, S.; de Magalhaes, J.P. A human tissue-specific transcriptomic analysis reveals a complex relationship between aging, cancer, and cellular senescence. Aging Cell 2019, 18, e13041. [Google Scholar] [CrossRef] [PubMed]
- Pienimaeki-Roemer, A.; Konovalova, T.; Musri, M.M.; Sigruener, A.; Boettcher, A.; Meister, G.; Schmitz, G. Transcriptomic profiling of platelet senescence and platelet extracellular vesicles. Transfusion 2017, 57, 144–156. [Google Scholar] [CrossRef]
- Linskens, M.H.; Feng, J.; Andrews, W.H.; Enlow, B.E.; Saati, S.M.; Tonkin, L.A.; Funk, W.D.; Villeponteau, B. Cataloging altered gene expression in young and senescent cells using enhanced differential display. Nucleic Acids Res. 1995, 23, 3244–3251. [Google Scholar] [CrossRef]
- Shelton, D.N.; Chang, E.; Whittier, P.S.; Choi, D.; Funk, W.D. Microarray analysis of replicative senescence. Curr. Biol. 1999, 9, 939–945. [Google Scholar] [CrossRef]
- Krtolica, A.; Parrinello, S.; Lockett, S.; Desprez, P.Y.; Campisi, J. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: A link between cancer and aging. Proc. Natl. Acad. Sci. USA 2001, 98, 12072–12077. [Google Scholar] [CrossRef] [PubMed]
- Acosta, J.C.; O’Loghlen, A.; Banito, A.; Guijarro, M.V.; Augert, A.; Raguz, S.; Fumagalli, M.; Da Costa, M.; Brown, C.; Popov, N.; et al. Chemokine signaling via the CXCR2 receptor reinforces senescence. Cell 2008, 133, 1006–1018. [Google Scholar] [CrossRef]
- Kuilman, T.; Michaloglou, C.; Vredeveld, L.C.; Douma, S.; van Doorn, R.; Desmet, C.J.; Aarden, L.A.; Mooi, W.J.; Peeper, D.S. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell 2008, 133, 1019–1031. [Google Scholar] [CrossRef]
- Basisty, N.; Kale, A.; Jeon, O.H.; Kuehnemann, C.; Payne, T.; Rao, C.; Holtz, A.; Shah, S.; Sharma, V.; Ferrucci, L.; et al. A proteomic atlas of senescence-associated secretomes for aging biomarker development. PLoS Biol. 2020, 18, e3000599. [Google Scholar] [CrossRef] [PubMed]
- Ozcan, S.; Alessio, N.; Acar, M.B.; Mert, E.; Omerli, F.; Peluso, G.; Galderisi, U. Unbiased analysis of senescence associated secretory phenotype (SASP) to identify common components following different genotoxic stresses. Aging 2016, 8, 1316–1329. [Google Scholar] [CrossRef]
- Uyar, B.; Palmer, D.; Kowald, A.; Murua Escobar, H.; Barrantes, I.; Moller, S.; Akalin, A.; Fuellen, G. Single-cell analyses of aging, inflammation and senescence. Ageing Res. Rev. 2020, 64, 101156. [Google Scholar] [CrossRef]
- Shimamoto, A.; Yokote, K.; Tahara, H. Werner Syndrome-specific induced pluripotent stem cells: Recovery of telomere function by reprogramming. Front. Genet. 2015, 6, 10. [Google Scholar] [CrossRef]
- Hernandez-Segura, A.; de Jong, T.V.; Melov, S.; Guryev, V.; Campisi, J.; Demaria, M. Unmasking Transcriptional Heterogeneity in Senescent Cells. Curr. Biol. 2017, 27, 2652–2660.e4. [Google Scholar] [CrossRef]
- Rai, T.S.; Cole, J.J.; Nelson, D.M.; Dikovskaya, D.; Faller, W.J.; Vizioli, M.G.; Hewitt, R.N.; Anannya, O.; McBryan, T.; Manoharan, I.; et al. HIRA orchestrates a dynamic chromatin landscape in senescence and is required for suppression of neoplasia. Genes Dev. 2014, 28, 2712–2725. [Google Scholar] [CrossRef] [PubMed]
- Wiley, C.D.; Liu, S.; Limbad, C.; Zawadzka, A.M.; Beck, J.; Demaria, M.; Artwood, R.; Alimirah, F.; Lopez-Dominguez, J.A.; Kuehnemann, C.; et al. SILAC Analysis Reveals Increased Secretion of Hemostasis-Related Factors by Senescent Cells. Cell Rep. 2019, 28, 3329–3337.e5. [Google Scholar] [CrossRef] [PubMed]
- Menni, C.; Kiddle, S.J.; Mangino, M.; Vinuela, A.; Psatha, M.; Steves, C.; Sattlecker, M.; Buil, A.; Newhouse, S.; Nelson, S.; et al. Circulating Proteomic Signatures of Chronological Age. J. Gerontol. A Biol. Sci. Med. Sci. 2015, 70, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Avelar, R.A.; Ortega, J.G.; Tacutu, R.; Tyler, E.J.; Bennett, D.; Binetti, P.; Budovsky, A.; Chatsirisupachai, K.; Johnson, E.; Murray, A.; et al. A multidimensional systems biology analysis of cellular senescence in aging and disease. Genome Biol. 2020, 21, 91. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Tian, Z.; Kadomatsu, T.; Xie, P.; Miyata, K.; Sugizaki, T.; Endo, M.; Zhu, S.; Fan, H.; Horiguchi, H.; et al. Age-dependent increase in angiopoietin-like protein 2 accelerates skeletal muscle loss in mice. J. Biol. Chem. 2018, 293, 1596–1609. [Google Scholar] [CrossRef] [PubMed]
- Theodoris, C.V.; Mourkioti, F.; Huang, Y.; Ranade, S.S.; Liu, L.; Blau, H.M.; Srivastava, D. Long telomeres protect against age-dependent cardiac disease caused by NOTCH1 haploinsufficiency. J. Clin. Investig. 2017, 127, 1683–1688. [Google Scholar] [CrossRef] [PubMed]
- Caland, L.; Labbe, P.; Mamarbachi, M.; Villeneuve, L.; Ferbeyre, G.; Noly, P.E.; Carrier, M.; Thorin-Trescases, N.; Thorin, E. Knockdown of angiopoietin-like 2 induces clearance of vascular endothelial senescent cells by apoptosis, promotes endothelial repair and slows atherogenesis in mice. Aging 2019, 11, 3832–3850. [Google Scholar] [CrossRef]
- Noly, P.E.; Labbe, P.; Thorin-Trescases, N.; Fortier, A.; Nguyen, A.; Thorin, E.; Carrier, M. Reduction of plasma angiopoietin-like 2 after cardiac surgery is related to tissue inflammation and senescence status of patients. J. Thorac. Cardiovasc. Surg. 2019, 158, 792–802.e5. [Google Scholar] [CrossRef]
- Salminen, A.; Kauppinen, A.; Kaarniranta, K. Emerging role of NF-kappaB signaling in the induction of senescence-associated secretory phenotype (SASP). Cell Signal 2012, 24, 835–845. [Google Scholar] [CrossRef]
- Chien, Y.; Scuoppo, C.; Wang, X.; Fang, X.; Balgley, B.; Bolden, J.E.; Premsrirut, P.; Luo, W.; Chicas, A.; Lee, C.S.; et al. Control of the senescence-associated secretory phenotype by NF-kappaB promotes senescence and enhances chemosensitivity. Genes Dev. 2011, 25, 2125–2136. [Google Scholar] [CrossRef]
- Endo, M.; Nakano, M.; Kadomatsu, T.; Fukuhara, S.; Kuroda, H.; Mikami, S.; Hato, T.; Aoi, J.; Horiguchi, H.; Miyata, K.; et al. Tumor cell-derived angiopoietin-like protein ANGPTL2 is a critical driver of metastasis. Cancer Res. 2012, 72, 1784–1794. [Google Scholar] [CrossRef] [PubMed]
- Oike, Y.; Tian, Z.; Miyata, K.; Morinaga, J.; Endo, M.; Kadomatsu, T. ANGPTL2- A New Causal Player in Accelerating Heart Disease Development in the Aging. Circ. J. 2017, 81, 1379–1385. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, K.; Suzuki, H.I. TGF-beta Signaling in Cellular Senescence and Aging-Related Pathology. Int. J. Mol. Sci. 2019, 20, 5002. [Google Scholar] [CrossRef]
- Nakamura, T.; Okada, T.; Endo, M.; Kadomatsu, T.; Taniwaki, T.; Sei, A.; Odagiri, H.; Masuda, T.; Fujimoto, T.; Nakamura, T.; et al. Angiopoietin-like protein 2 induced by mechanical stress accelerates degeneration and hypertrophy of the ligamentum flavum in lumbar spinal canal stenosis. PLoS ONE 2014, 9, e85542. [Google Scholar] [CrossRef]
- Lee, H.J.; Kim, J.H.; Kim, J.H.; Martinus, R.D.; Park, S.H. Angiopoietin-like protein 2, a chronic inflammatory mediator, is a new target induced by TGF-beta1 through a Smad3-dependent mechanism. Biochem. Biophys. Res. Commun. 2013, 430, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Pickering, J.G. Cellular Senescence and Vascular Disease: Novel Routes to Better Understanding and Therapy. Can. J. Cardiol. 2016, 32, 612–623. [Google Scholar] [CrossRef]
- Coppe, J.P.; Kauser, K.; Campisi, J.; Beausejour, C.M. Secretion of vascular endothelial growth factor by primary human fibroblasts at senescence. J. Biol. Chem. 2006, 281, 29568–29574. [Google Scholar] [CrossRef]
- Ding, H.; Chen, C.; Shaffer, J.R.; Liu, L.; Xu, Y.; Wang, X.; Hui, R.; Wang, D.W. Telomere length and risk of stroke in Chinese. Stroke 2012, 43, 658–663. [Google Scholar] [CrossRef]
- Tian, Y.; Wang, S.; Jiao, F.; Kong, Q.; Liu, C.; Wu, Y. Telomere Length: A Potential Biomarker for the Risk and Prognosis of Stroke. Front. Neurol. 2019, 10, 624. [Google Scholar] [CrossRef]
- Yamazaki, Y.; Baker, D.J.; Tachibana, M.; Liu, C.C.; van Deursen, J.M.; Brott, T.G.; Bu, G.; Kanekiyo, T. Vascular Cell Senescence Contributes to Blood-Brain Barrier Breakdown. Stroke 2016, 47, 1068–1077. [Google Scholar] [CrossRef]
- Henze, L.; Walter, U.; Murua Escobar, H.; Junghanss, C.; Jaster, R.; Kohling, R.; Lange, F.; Salehzadeh-Yazdi, A.; Wolkenhauer, O.; Hamed, M.; et al. Towards biomarkers for outcomes after pancreatic ductal adenocarcinoma and ischaemic stroke, with focus on (co)-morbidity and ageing/cellular senescence (SASKit): Protocol for a prospective cohort study. BMJ Open 2020, 10, e039560. [Google Scholar] [CrossRef] [PubMed]
- Deng, M.; Lu, Z.; Zheng, J.; Wan, X.; Chen, X.; Hirayasu, K.; Sun, H.; Lam, Y.; Chen, L.; Wang, Q.; et al. A motif in LILRB2 critical for Angptl2 binding and activation. Blood 2014, 124, 924–935. [Google Scholar] [CrossRef] [PubMed]
- Kubota, Y. Unveiling Angptl2, a rising HSC expander. Blood 2014, 124, 833–834. [Google Scholar] [CrossRef][Green Version]
- Costa, R.A.; Cardoso, J.C.; Power, D.M. Evolution of the angiopoietin-like gene family in teleosts and their role in skin regeneration. BMC Evol. Biol. 2017, 17, 14. [Google Scholar] [CrossRef] [PubMed]
- Okumura, A.; Hayashi, T.; Ebisawa, M.; Yoshimura, M.; Sasagawa, Y.; Nikaido, I.; Umesono, Y.; Mochii, M. Cell type-specific transcriptome analysis unveils secreted signaling molecule genes expressed in apical epithelial cap during appendage regeneration. Dev. Growth Differ. 2019, 61, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Tanoue, H.; Morinaga, J.; Yoshizawa, T.; Yugami, M.; Itoh, H.; Nakamura, T.; Uehara, Y.; Masuda, T.; Odagiri, H.; Sugizaki, T.; et al. Angiopoietin-like protein 2 promotes chondrogenic differentiation during bone growth as a cartilage matrix factor. Osteoarthr. Cartil. 2018, 26, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Campisi, J.; Andersen, J.K.; Kapahi, P.; Melov, S. Cellular senescence: A link between cancer and age-related degenerative disease? Semin. Cancer Biol. 2011, 21, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Coppe, J.P.; Desprez, P.Y.; Krtolica, A.; Campisi, J. The senescence-associated secretory phenotype: The dark side of tumor suppression. Annu. Rev. Pathol. 2010, 5, 99–118. [Google Scholar] [CrossRef]
- Rodier, F.; Campisi, J. Four faces of cellular senescence. J. Cell Biol. 2011, 192, 547–556. [Google Scholar] [CrossRef]
- Sun, Y.; Coppe, J.P.; Lam, E.W. Cellular Senescence: The Sought or the Unwanted? Trends Mol. Med. 2018, 24, 871–885. [Google Scholar] [CrossRef]
- Endo, M.; Yamamoto, Y.; Nakano, M.; Masuda, T.; Odagiri, H.; Horiguchi, H.; Miyata, K.; Kadomatsu, T.; Motokawa, I.; Okada, S.; et al. Serum ANGPTL2 levels reflect clinical features of breast cancer patients: Implications for the pathogenesis of breast cancer metastasis. Int. J. Biol. Markers 2014, 29, e239–e245. [Google Scholar] [CrossRef]
- Gao, L.; Ge, C.; Fang, T.; Zhao, F.; Chen, T.; Yao, M.; Li, J.; Li, H. ANGPTL2 promotes tumor metastasis in hepatocellular carcinoma. J. Gastroenterol. Hepatol. 2015, 30, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T.; Endo, M.; Yamamoto, Y.; Odagiri, H.; Kadomatsu, T.; Nakamura, T.; Tanoue, H.; Ito, H.; Yugami, M.; Miyata, K.; et al. ANGPTL2 increases bone metastasis of breast cancer cells through enhancing CXCR4 signaling. Sci. Rep. 2015, 5, 9170. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, H.; Suzuki, A.; Shitara, M.; Hikosaka, Y.; Okuda, K.; Moriyama, S.; Yano, M.; Fujii, Y. Angiopoietin-like protein ANGPTL2 gene expression is correlated with lymph node metastasis in lung cancer. Oncol. Lett. 2012, 4, 1325–1328. [Google Scholar] [CrossRef]
- Sato, R.; Yamasaki, M.; Hirai, K.; Matsubara, T.; Nomura, T.; Sato, F.; Mimata, H. Angiopoietin-like protein 2 induces androgen-independent and malignant behavior in human prostate cancer cells. Oncol. Rep. 2015, 33, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, R.; Tsuda, H.; Kozaki, K.; Kanai, Y.; Kasamatsu, T.; Sengoku, K.; Hirohashi, S.; Inazawa, J.; Imoto, I. Frequent inactivation of a putative tumor suppressor, angiopoietin-like protein 2, in ovarian cancer. Cancer Res. 2008, 68, 5067–5075. [Google Scholar] [CrossRef]
- Horiguchi, H.; Kadomatsu, T.; Kurahashi, R.; Hara, C.; Miyata, K.; Baba, M.; Osumi, H.; Terada, K.; Araki, K.; Takai, T.; et al. Dual functions of angiopoietin-like protein 2 signaling in tumor progression and anti-tumor immunity. Genes Dev. 2019, 33, 1641–1656. [Google Scholar] [CrossRef]
- Horiguchi, H.; Kadomatsu, T.; Miyata, K.; Terada, K.; Sato, M.; Torigoe, D.; Morinaga, J.; Moroishi, T.; Oike, Y. Stroma-derived ANGPTL2 establishes an anti-tumor microenvironment during intestinal tumorigenesis. Oncogene 2021, 40, 55–67. [Google Scholar] [CrossRef]
- Horiguchi, H.; Endo, M.; Miyamoto, Y.; Sakamoto, Y.; Odagiri, H.; Masuda, T.; Kadomatsu, T.; Tanoue, H.; Motokawa, I.; Terada, K.; et al. Angiopoietin-like protein 2 renders colorectal cancer cells resistant to chemotherapy by activating spleen tyrosine kinase-phosphoinositide 3-kinase-dependent anti-apoptotic signaling. Cancer Sci. 2014, 105, 1550–1559. [Google Scholar] [CrossRef]
- Tchkonia, T.; Zhu, Y.; van Deursen, J.; Campisi, J.; Kirkland, J.L. Cellular senescence and the senescent secretory phenotype: Therapeutic opportunities. J. Clin. Investig. 2013, 123, 966–972. [Google Scholar] [CrossRef]
- Rhinn, M.; Ritschka, B.; Keyes, W.M. Cellular senescence in development, regeneration and disease. Development 2019, 146. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Espin, D.; Canamero, M.; Maraver, A.; Gomez-Lopez, G.; Contreras, J.; Murillo-Cuesta, S.; Rodriguez-Baeza, A.; Varela-Nieto, I.; Ruberte, J.; Collado, M.; et al. Programmed cell senescence during mammalian embryonic development. Cell 2013, 155, 1104–1118. [Google Scholar] [CrossRef] [PubMed]
- Storer, M.; Mas, A.; Robert-Moreno, A.; Pecoraro, M.; Ortells, M.C.; Di Giacomo, V.; Yosef, R.; Pilpel, N.; Krizhanovsky, V.; Sharpe, J.; et al. Senescence is a developmental mechanism that contributes to embryonic growth and patterning. Cell 2013, 155, 1119–1130. [Google Scholar] [CrossRef] [PubMed]
- Perez-Garijo, A.; Steller, H. The benefits of aging: Cellular senescence in normal development. EMBO J. 2014, 33, 99–100. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhao, H.; Huang, X.; Tang, J.; Zhang, S.; Li, Y.; Liu, X.; He, L.; Ju, Z.; Lui, K.O.; et al. Embryonic senescent cells re-enter cell cycle and contribute to tissues after birth. Cell Res. 2018, 28, 775–778. [Google Scholar] [CrossRef]
- Kubota, Y.; Oike, Y.; Satoh, S.; Tabata, Y.; Niikura, Y.; Morisada, T.; Akao, M.; Urano, T.; Ito, Y.; Miyamoto, T.; et al. Isolation and expression patterns of genes for three angiopoietin-like proteins, Angptl1, 2 and 6 in zebrafish. Gene Expr. Patterns 2005, 5, 679–685. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, B.; Pache, L.; Chang, M.; Khodabakhshi, A.H.; Tanaseichuk, O.; Benner, C.; Chanda, S.K. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 2019, 10, 1523. [Google Scholar] [CrossRef]
- Tang, C.; Chen, E.; Peng, K.; Wang, H.; Cheng, X.; Wang, Y.; Yu, S.; Yu, Y.; Cui, Y.; Liu, T. Mining the role of angiopoietin-like protein family in gastric cancer and seeking potential therapeutic targets by integrative bioinformatics analysis. Cancer Med. 2020, 9, 4850–4863. [Google Scholar] [CrossRef]
- Duan, X.; An, B.; Du, L.; Chang, T.; Liang, M.; Yang, B.G.; Xu, L.; Zhang, L.; Li, J.; E, G.; et al. Genome-Wide Association Analysis of Growth Curve Parameters in Chinese Simmental Beef Cattle. Animals 2021, 11, 192. [Google Scholar] [CrossRef]
- Lorda-Diez, C.I.; Solis-Mancilla, M.E.; Sanchez-Fernandez, C.; Garcia-Porrero, J.A.; Hurle, J.M.; Montero, J.A. Cell senescence, apoptosis and DNA damage cooperate in the remodeling processes accounting for heart morphogenesis. J. Anat. 2019, 234, 815–829. [Google Scholar] [CrossRef]
- Niki, D.; Katsu, K.; Yokouchi, Y. Ontogeny of angiopoietin-like protein 1, 2, 3, 4, 5, and 7 genes during chick embryonic development. Dev. Growth Differ. 2009, 51, 821–832. [Google Scholar] [CrossRef] [PubMed]
- Labbe, P.; Munoz Goyette, V.; Villeneuve, L.; Desanlis, I.; Alikashani, A.; Delwarde, C.; Thorin-Trescases, N.; Kmita, M.; Thorin, E. ANGPTL2 is essential for aortic valve development in mice. Circulation 2021, in press. [Google Scholar]
- Li, C.; Chai, Y.; Wang, L.; Gao, B.; Chen, H.; Gao, P.; Zhou, F.Q.; Luo, X.; Crane, J.L.; Yu, B.; et al. Programmed cell senescence in skeleton during late puberty. Nat. Commun. 2017, 8, 1312. [Google Scholar] [CrossRef]
- Yun, M.H. Cellular senescence in tissue repair: Every cloud has a silver lining. Int. J. Dev. Biol. 2018, 62, 591–604. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, H.N.; Hardman, M.J. Senescence in Wound Repair: Emerging Strategies to Target Chronic Healing Wounds. Front. Cell Dev. Biol. 2020, 8, 773. [Google Scholar] [CrossRef]
- Monaghan, J.R.; Walker, J.A.; Page, R.B.; Putta, S.; Beachy, C.K.; Voss, S.R. Early gene expression during natural spinal cord regeneration in the salamander Ambystoma mexicanum. J. Neurochem. 2007, 101, 27–40. [Google Scholar] [CrossRef]
- Yun, M.H.; Davaapil, H.; Brockes, J.P. Recurrent turnover of senescent cells during regeneration of a complex structure. Elife 2015, 4, e05505. [Google Scholar] [CrossRef]
- Da Silva-Alvarez, S.; Guerra-Varela, J.; Sobrido-Camean, D.; Quelle, A.; Barreiro-Iglesias, A.; Sanchez, L.; Collado, M. Cell senescence contributes to tissue regeneration in zebrafish. Aging Cell 2020, 19, e13052. [Google Scholar] [CrossRef]
- Smit, M.A.; Peeper, D.S. Epithelial-mesenchymal transition and senescence: Two cancer-related processes are crossing paths. Aging 2010, 2, 735–741. [Google Scholar] [CrossRef]
- Mavrogonatou, E.; Pratsinis, H.; Papadopoulou, A.; Karamanos, N.K.; Kletsas, D. Extracellular matrix alterations in senescent cells and their significance in tissue homeostasis. Matrix Biol. 2019, 75–76, 27–42. [Google Scholar] [CrossRef]
- Demaria, M.; Ohtani, N.; Youssef, S.A.; Rodier, F.; Toussaint, W.; Mitchell, J.R.; Laberge, R.M.; Vijg, J.; Van Steeg, H.; Dolle, M.E.; et al. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev. Cell 2014, 31, 722–733. [Google Scholar] [CrossRef] [PubMed]
- Ritschka, B.; Storer, M.; Mas, A.; Heinzmann, F.; Ortells, M.C.; Morton, J.P.; Sansom, O.J.; Zender, L.; Keyes, W.M. The senescence-associated secretory phenotype induces cellular plasticity and tissue regeneration. Genes Dev. 2017, 31, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Saul, D.; Monroe, D.G.; Rowsey, J.L.; Kosinsky, R.L.; Vos, S.J.; Doolittle, M.L.; Farr, J.N.; Khosla, S. Modulation of fracture healing by the transient accumulation of senescent cells. eLife 2021, 10, e69958. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.J.; Wijshake, T.; Tchkonia, T.; LeBrasseur, N.K.; Childs, B.G.; van de Sluis, B.; Kirkland, J.L.; van Deursen, J.M. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature 2011, 479, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.J.; Childs, B.G.; Durik, M.; Wijers, M.E.; Sieben, C.J.; Zhong, J.; Saltness, R.A.; Jeganathan, K.B.; Verzosa, G.C.; Pezeshki, A.; et al. Naturally occurring p16(Ink4a)-positive cells shorten healthy lifespan. Nature 2016, 530, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Gil, J.; Withers, D.J. Ageing: Out with the old. Nature 2016, 530, 164–165. [Google Scholar] [CrossRef]
- McHugh, D.; Gil, J. Senescence and aging: Causes, consequences, and therapeutic avenues. J. Cell Biol. 2018, 217, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Hall, B.M.; Balan, V.; Gleiberman, A.S.; Strom, E.; Krasnov, P.; Virtuoso, L.P.; Rydkina, E.; Vujcic, S.; Balan, K.; Gitlin, I.; et al. Aging of mice is associated with p16(Ink4a)- and beta-galactosidase-positive macrophage accumulation that can be induced in young mice by senescent cells. Aging 2016, 8, 1294–1315. [Google Scholar] [CrossRef]
- Xu, M.; Bradley, E.W.; Weivoda, M.M.; Hwang, S.M.; Pirtskhalava, T.; Decklever, T.; Curran, G.L.; Ogrodnik, M.; Jurk, D.; Johnson, K.O.; et al. Transplanted Senescent Cells Induce an Osteoarthritis-Like Condition in Mice. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 780–785. [Google Scholar] [CrossRef]
- Xu, M.; Pirtskhalava, T.; Farr, J.N.; Weigand, B.M.; Palmer, A.K.; Weivoda, M.M.; Inman, C.L.; Ogrodnik, M.B.; Hachfeld, C.M.; Fraser, D.G.; et al. Senolytics improve physical function and increase lifespan in old age. Nat. Med. 2018, 24, 1246–1256. [Google Scholar] [CrossRef]
- Ogrodnik, M. Cellular aging beyond cellular senescence: Markers of senescence prior to cell cycle arrest in vitro and in vivo. Aging Cell 2021, 20, e13338. [Google Scholar] [CrossRef] [PubMed]
- Straub, R.H.; Schradin, C. Chronic inflammatory systemic diseases: An evolutionary trade-off between acutely beneficial but chronically harmful programs. Evol. Med. Public Health 2016, 2016, 37–51. [Google Scholar] [CrossRef] [PubMed]
- van Deursen, J.M. Senolytic therapies for healthy longevity. Science 2019, 364, 636–637. [Google Scholar] [CrossRef]
- Velarde, M.C.; Demaria, M.; Campisi, J. Senescent cells and their secretory phenotype as targets for cancer therapy. Interdiscip. Top. Gerontol. 2013, 38, 17–27. [Google Scholar] [CrossRef]
- Shimizu, I.; Minamino, T. Cellular senescence in cardiac diseases. J. Cardiol. 2019, 74, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Mehdizadeh, M.; Aguilar, M.; Thorin, E.; Ferbeyre, G.; Nattel, S. The role of cellular senescence in cardiac disease: Basic biology and clinical relevance. Nat. Rev. 2021. [Google Scholar] [CrossRef]
- Childs, B.G.; Li, H.; van Deursen, J.M. Senescent cells: A therapeutic target for cardiovascular disease. J. Clin. Investig. 2018, 128, 1217–1228. [Google Scholar] [CrossRef]
- Katsuumi, G.; Shimizu, I.; Yoshida, Y.; Minamino, T. Vascular Senescence in Cardiovascular and Metabolic Diseases. Front. Cardiovasc. Med. 2018, 5, 18. [Google Scholar] [CrossRef]
- Minamino, T.; Komuro, I. Vascular cell senescence: Contribution to atherosclerosis. Circ. Res. 2007, 100, 15–26. [Google Scholar] [CrossRef]
- Newsholme, P.; de Bittencourt, P.I., Jr. The fat cell senescence hypothesis: A mechanism responsible for abrogating the resolution of inflammation in chronic disease. Curr. Opin. Clin. Nutr. Metab. Care 2014, 17, 295–305. [Google Scholar] [CrossRef]
- Palmer, A.K.; Tchkonia, T.; LeBrasseur, N.K.; Chini, E.N.; Xu, M.; Kirkland, J.L. Cellular Senescence in Type 2 Diabetes: A Therapeutic Opportunity. Diabetes 2015, 64, 2289–2298. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.J.; Petersen, R.C. Cellular senescence in brain aging and neurodegenerative diseases: Evidence and perspectives. J. Clin. Investig. 2018, 128, 1208–1216. [Google Scholar] [CrossRef]
- Kumar, M.; Seeger, W.; Voswinckel, R. Senescence-associated secretory phenotype and its possible role in chronic obstructive pulmonary disease. Am. J. Respir. Cell Mol. Biol. 2014, 51, 323–333. [Google Scholar] [CrossRef] [PubMed]
- Hata, J.; Mukai, N.; Nagata, M.; Ohara, T.; Yoshida, D.; Kishimoto, H.; Shibata, M.; Hirakawa, Y.; Endo, M.; Ago, T.; et al. Serum Angiopoietin-Like Protein 2 Is a Novel Risk Factor for Cardiovascular Disease in the Community: The Hisayama Study. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 1686–1691. [Google Scholar] [CrossRef]
- Doi, Y.; Ninomiya, T.; Hirakawa, Y.; Takahashi, O.; Mukai, N.; Hata, J.; Iwase, M.; Kitazono, T.; Oike, Y.; Kiyohara, Y. Angiopoietin-like protein 2 and risk of type 2 diabetes in a general Japanese population: The Hisayama study. Diabetes Care 2013, 36, 98–100. [Google Scholar] [CrossRef] [PubMed]
- Usui, T.; Ninomiya, T.; Nagata, M.; Takahashi, O.; Doi, Y.; Hata, J.; Fukuhara, M.; Kitazono, T.; Oike, Y.; Kiyohara, Y. Angiopoietin-like protein 2 is associated with chronic kidney disease in a general Japanese population: The Hisayama Study. Circ. J. 2013, 77, 2311–2317. [Google Scholar] [CrossRef] [PubMed]
- Hirata, T.; Arai, Y.; Yuasa, S.; Abe, Y.; Takayama, M.; Sasaki, T.; Kunitomi, A.; Inagaki, H.; Endo, M.; Morinaga, J.; et al. Associations of cardiovascular biomarkers and plasma albumin with exceptional survival to the highest ages. Nat. Commun. 2020, 11, 3820. [Google Scholar] [CrossRef] [PubMed]
- Gellen, B.; Thorin-Trescases, N.; Sosner, P.; Gand, E.; Saulnier, P.J.; Ragot, S.; Fraty, M.; Laugier, S.; Ducrocq, G.; Montaigne, D.; et al. ANGPTL2 is associated with an increased risk of cardiovascular events and death in diabetic patients. Diabetologia 2016, 59, 2321–2330. [Google Scholar] [CrossRef]
- Piche, M.E.; Thorin-Trescases, N.; Auclair, A.; Marceau, S.; Martin, J.; Fortier, A.; Thorin, E.; Poirier, P. Bariatric Surgery-Induced Lower Angiopoietin-Like 2 Protein Is Associated With Improved Cardiometabolic Profile. Can. J. Cardiol. 2017, 33, 1044–1051. [Google Scholar] [CrossRef]
- Okada, T.; Tsukano, H.; Endo, M.; Tabata, M.; Miyata, K.; Kadomatsu, T.; Miyashita, K.; Semba, K.; Nakamura, E.; Tsukano, M.; et al. Synoviocyte-derived angiopoietin-like protein 2 contributes to synovial chronic inflammation in rheumatoid arthritis. Am. J. Pathol. 2010, 176, 2309–2319. [Google Scholar] [CrossRef]
- Nishiyama, S.; Hirose, N.; Yanoshita, M.; Takano, M.; Kubo, N.; Yamauchi, Y.; Onishi, A.; Ito, S.; Sakata, S.; Kita, D.; et al. ANGPTL2 Induces Synovial Inflammation via LILRB2. Inflammation 2021, 44, 1108–1118. [Google Scholar] [CrossRef]
- Shan, W.; Cheng, C.; Huang, W.; Ding, Z.; Luo, S.; Cui, G.; Lu, W.; Liu, F.; Xu, J.; He, W.; et al. Angiopoietin-like 2 upregulation promotes human chondrocyte injury via NF-kappaB and p38/MAPK signaling pathway. J. Bone Miner. Metab. 2019, 37, 976–986. [Google Scholar] [CrossRef]
- Takano, M.; Hirose, N.; Sumi, C.; Yanoshita, M.; Nishiyama, S.; Onishi, A.; Asakawa, Y.; Tanimoto, K. ANGPTL2 Promotes Inflammation via Integrin alpha5beta1 in Chondrocytes. Cartilage 2019, 1947603519878242. [Google Scholar] [CrossRef]
- Tseng, C.C.; Wong, M.C.; Liao, W.T.; Chen, C.J.; Lee, S.C.; Yen, J.H.; Chang, S.J. Systemic Investigation of Promoter-wide Methylome and Genome Variations in Gout. Int. J. Mol. Sci. 2020, 21, 4702. [Google Scholar] [CrossRef] [PubMed]
- Hirasawa, M.; Takubo, K.; Osada, H.; Miyake, S.; Toda, E.; Endo, M.; Umezawa, K.; Tsubota, K.; Oike, Y.; Ozawa, Y. Angiopoietin-like Protein 2 Is a Multistep Regulator of Inflammatory Neovascularization in a Murine Model of Age-related Macular Degeneration. J. Biol. Chem. 2016, 291, 7373–7385. [Google Scholar] [CrossRef]
- Yin, R.; Zhang, N.; Zhang, D.; Zhao, W.; Ke, J.; Zhao, D. Higher levels of circulating ANGPTL2 are associated with macular edema in patients with type 2 diabetes. Medicine 2021, 100, e24638. [Google Scholar] [CrossRef]
- Ashokachakkaravarthy, K.; Rajappa, M.; Parameswaran, S.; Satheesh, S.; Priyadarshini, G.; Mohan Raj, P.S.; Revathy, G.; Priyadarssini, M. Asymmetric dimethylarginine and angiopoietin-like protein-2 are independent predictors of cardiovascular risk in pre-dialysis non-diabetic chronic kidney disease patients. Int. Urol. Nephrol. 2020, 52, 1321–1328. [Google Scholar] [CrossRef] [PubMed]
- Tian, Z.; Miyata, K.; Kadomatsu, T.; Horiguchi, H.; Fukushima, H.; Tohyama, S.; Ujihara, Y.; Okumura, T.; Yamaguchi, S.; Zhao, J.; et al. ANGPTL2 activity in cardiac pathologies accelerates heart failure by perturbing cardiac function and energy metabolism. Nat. Commun. 2016, 7, 13016. [Google Scholar] [CrossRef]
- Tian, Z.; Miyata, K.; Morinaga, J.; Horiguchi, H.; Kadomatsu, T.; Endo, M.; Zhao, J.; Zhu, S.; Sugizaki, T.; Sato, M.; et al. Circulating ANGPTL2 Levels Increase in Humans and Mice Exhibiting Cardiac Dysfunction. Circ. J. 2018, 82, 437–447. [Google Scholar] [CrossRef]
- Ishii, T.; Furuya, F.; Takahashi, K.; Shikata, M.; Takamura, T.; Kobayashi, H.; Miyazaki, A.; Morinaga, J.; Terada, K.; Oike, Y.; et al. Angiopoietin-Like Protein 2 Promotes the Progression of Diabetic Kidney Disease. J. Clin. Endocrinol. Metab. 2019, 104, 172–180. [Google Scholar] [CrossRef]
- Fukami, H.; Morinaga, J.; Okadome, Y.; Nishiguchi, Y.; Iwata, Y.; Kanki, T.; Nakagawa, T.; Izumi, Y.; Kakizoe, Y.; Kuwabara, T.; et al. Circulating angiopoietin-like protein 2 levels and arterial stiffness in patients receiving maintenance hemodialysis: A cross-sectional study. Atherosclerosis 2020, 315, 18–23. [Google Scholar] [CrossRef]
- Carbone, C.; Piro, G.; Merz, V.; Simionato, F.; Santoro, R.; Zecchetto, C.; Tortora, G.; Melisi, D. Angiopoietin-Like Proteins in Angiogenesis, Inflammation and Cancer. Int. J. Mol. Sci. 2018, 19, 431. [Google Scholar] [CrossRef] [PubMed]
- Endo, M. The Roles of ANGPTL Families in Cancer Progression. J. UOEH 2019, 41, 317–325. [Google Scholar] [CrossRef]
- Amadatsu, T.; Morinaga, J.; Kawano, T.; Terada, K.; Kadomatsu, T.; Miyata, K.; Endo, M.; Kasamo, D.; Kuratsu, J.I.; Oike, Y. Macrophage-Derived Angiopoietin-Like Protein 2 Exacerbates Brain Damage by Accelerating Acute Inflammation after Ischemia-Reperfusion. PLoS ONE 2016, 11, e0166285. [Google Scholar] [CrossRef] [PubMed]
- Hirayasu, K.; Arase, H. Functional and genetic diversity of leukocyte immunoglobulin-like receptor and implication for disease associations. J. Hum. Genet. 2015, 60, 703–708. [Google Scholar] [CrossRef]
- Tanaka, T.; Biancotto, A.; Moaddel, R.; Moore, A.Z.; Gonzalez-Freire, M.; Aon, M.A.; Candia, J.; Zhang, P.; Cheung, F.; Fantoni, G.; et al. Plasma proteomic signature of age in healthy humans. Aging Cell 2018, 17, e12799. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, C. Transcriptomic Analysis of Cellular Senescence: One Step Closer to Senescence Atlas. Mol. Cells 2021, 44, 136–145. [Google Scholar] [CrossRef]
- Basisty, N.; Kale, A.; Patel, S.; Campisi, J.; Schilling, B. The power of proteomics to monitor senescence-associated secretory phenotypes and beyond: Toward clinical applications. Expert Rev. Proteomics 2020, 17, 297–308. [Google Scholar] [CrossRef]
- Desjardins, M.P.; Thorin-Trescases, N.; Sidibe, A.; Fortier, C.; De Serres, S.A.; Lariviere, R.; Thorin, E.; Agharazii, M. Levels of Angiopoietin-Like-2 Are Positively Associated With Aortic Stiffness and Mortality After Kidney Transplantation. Am. J. Hypertens. 2017, 30, 409–416. [Google Scholar] [CrossRef]
- Morinaga, J.; Kakuma, T.; Fukami, H.; Hayata, M.; Uchimura, K.; Mizumoto, T.; Kakizoe, Y.; Miyoshi, T.; Shiraishi, N.; Adachi, M.; et al. Circulating angiopoietin-like protein 2 levels and mortality risk in patients receiving maintenance hemodialysis: A prospective cohort study. Nephrol. Dial. Transplant. 2020, 35, 854–860. [Google Scholar] [CrossRef]
- Chen, Y.; Jiang, H.; Zhu, L.; Wang, P.; Liu, S.; Xiao, X.; Yu, H.; Dong, W. Diagnostic and Prognostic Value of Serum Angiopoietin-Like Protein 2 in Patients with Non-Small Cell Lung Cancer. Clin. Lab. 2017, 63, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Ide, S.; Toiyama, Y.; Shimura, T.; Kawamura, M.; Yasuda, H.; Saigusa, S.; Ohi, M.; Tanaka, K.; Mohri, Y.; Kusunoki, M. Angiopoietin-Like Protein 2 Acts as a Novel Biomarker for Diagnosis and Prognosis in Patients with Esophageal Cancer. Ann. Surg. Oncol. 2015, 22, 2585–2592. [Google Scholar] [CrossRef] [PubMed]
- Toiyama, Y.; Tanaka, K.; Kitajima, T.; Shimura, T.; Imaoka, H.; Mori, K.; Okigami, M.; Yasuda, H.; Okugawa, Y.; Saigusa, S.; et al. Serum angiopoietin-like protein 2 as a potential biomarker for diagnosis, early recurrence and prognosis in gastric cancer patients. Carcinogenesis 2015, 36, 1474–1483. [Google Scholar] [CrossRef] [PubMed]
- Toiyama, Y.; Tanaka, K.; Kitajima, T.; Shimura, T.; Kawamura, M.; Kawamoto, A.; Okugawa, Y.; Saigusa, S.; Hiro, J.; Inoue, Y.; et al. Elevated serum angiopoietin-like protein 2 correlates with the metastatic properties of colorectal cancer: A serum biomarker for early diagnosis and recurrence. Clin. Cancer Res. 2014, 20, 6175–6186. [Google Scholar] [CrossRef] [PubMed]
- Young, A.R.; Narita, M. SASP reflects senescence. EMBO Rep. 2009, 10, 228–230. [Google Scholar] [CrossRef] [PubMed]
- Byun, H.O.; Lee, Y.K.; Kim, J.M.; Yoon, G. From cell senescence to age-related diseases: Differential mechanisms of action of senescence-associated secretory phenotypes. BMB Rep. 2015, 48, 549–558. [Google Scholar] [CrossRef]
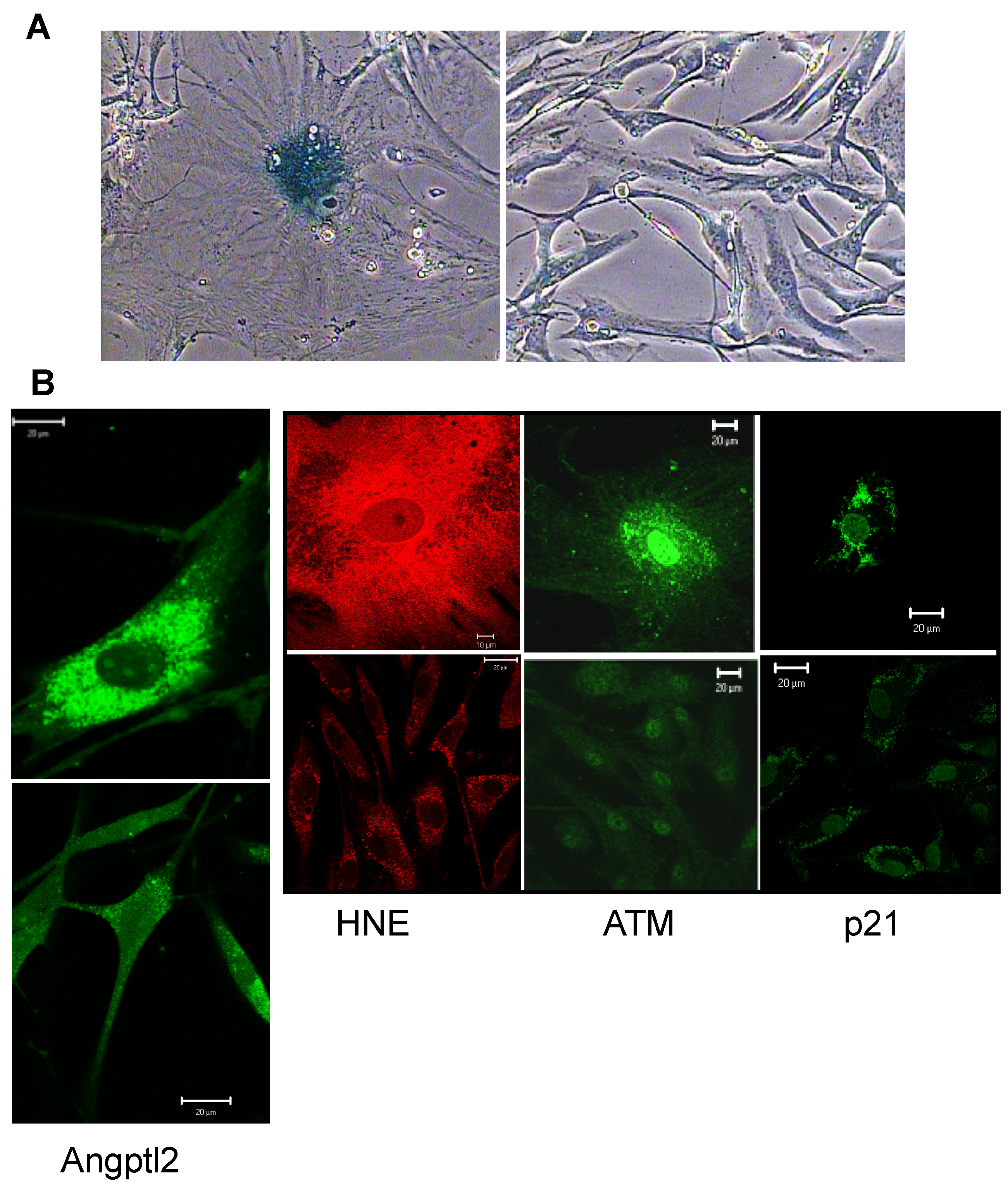
| Cell/Tissue | Species (Model Used) | Treatment/Senescence Inducer | Changes in Angptl2 Gene Expression | Fold Change | Methods | GEO Number | Reference | |
|---|---|---|---|---|---|---|---|---|
| Fibroblasts | Primary fibroblast cell lines (WS1, WI-38 & BJ) | Homo Sapiens (in vitro) | Replicative senescence (serial passages) | ↓ in senescent and quiescent fibroblasts | −0.86 range (−2.84; 1.13) | cDNA microarray | NA | [58] |
| Primary fibroblast cell line (IMR90) | Homo Sapiens (in vitro) | Oncogene-induced senescence (100 nM 4-hydroxytamoxifen treatment for 0–7 days) | ↓ in senescent fibroblasts | −1.63 | cDNA microarray | GSE41318 | [63] | |
| Embryonic fibroblast cell line (MRC-5) | Homo Sapiens (in vitro) | SCM-induced senescence (time course over 5 days) | ↓ at three time points: day 1, day 3 & day 5 vs. day 0 | D1 vs. D0 = −2.39 D3 vs. D0 = −1.93 D5 vs. D0 = −2.49 | RNA-seq | NA | [64] | |
| Primary fibroblast cell line (IMR-90) | Homo Sapiens (in vitro) | Replicative senescence (exhaustion PDL 50–59) IR-induced senescence (PDL 25 exposed to 10 Gy) | ↓ in senescent fibroblasts | RS = −1.59 IR = −1.28 | RNA-seq | GSE130727 | [65] | |
| Fibroblasts from healthy skin samples | Homo Sapiens (in vivo) | Aging (young (25–27 years) and old subjects (53–70 years) | ↓ in mesenchymal fibroblasts | −1.31 | single-cell RNA-seq | GSE130973 | [66] | |
| Fibroblasts from Werner Syndrome patients Premature aging disorder | Homo Sapiens (in vivo) | Patients# A0031: 37 years old TIG-114: 36 years old WSCU01: 63 years old | ↑ in WS fibroblasts (vs. healthy fibroblasts) | A0031 = +10.4 TIG-114 = +5.9 WSCU01 = +7.3 | cDNA microarray | NA | [67] | |
| Primary fibroblast cell lines (HFF) | Homo Sapiens (in vitro) | Replicative senescence (serial passages up to PDL 74) | ↑ in HFF senescent fibroblasts | PDL 16–26 = +1.38 PDL 16–74 = +2.68 | RNA-seq | GSE63577 | [68] | |
| Primary fibroblast cell lines (MRC-5, WI-38, BJ, IMR-90 & HFF) | Homo Sapiens (in vitro) | Replicative senescence (serial passages up to PDL 74) | ↑ in WI-38 senescent fibroblasts (vs. early PDL) | ND angptl2 is the 63rd most up-regulated gene | RNA-seq | GSE63577 | [69] | |
| Primary fibroblast cell line (WI-38) | Homo Sapiens (in vitro) | IR-induced senescence (PDL 25 exposed to 10 Gy) | ↑ in WI-38 senescent fibroblasts | +1.15 | RNA-seq | GSE130727 | [65] | |
| Primary fibroblast cell line (IMR-90) | Homo Sapiens (in vitro) | Paracrine- and oncogene-induced senescence (4-hydroxytamoxifen treatment) | ↑ in senescent fibroblasts | ND | single-cell RNA-seq | GSE115301 | [70] | |
| Hepatocytes | Hepatocellular carcinoma cell line (Huh7 clones) | Homo Sapiens (in vitro) | Reprogramming replicative senescence (clone C3: PDL80; clone G12: PDL 90) | ↓ in senescent Huh7 clones C3 and G12 | ND | cDNA microarray | GSE17546 | [71] |
| Hepatocellular carcinoma cell line (HepG2) | Homo Sapiens (in vitro) | Oncogene-induced senescence (10 µM and 100 µM etoposide treatment for 30 h) | ↑ in hepatocytes exposed to etoposide | 10 µM vs. ctrl = +3.63 100 µM vs. ctrl = +1.76 10 µM vs. 100 µM = +2.06 | cDNA microarray | GSE61110 | [72] | |
| Hepatocellular carcinoma cells from patients | Homo Sapiens (in vivo) | Oncogene-induced senescence (liver cancer) | ↑ between predicted high and low risk HCC cases | high vs. low risk = +1.16 | cDNA microarray | GSE14520 | [73] | |
| Astrocytes | Fetal astrocytes | Homo Sapiens (in vitro) | Oxidative stress-induced senescence (200 µM H2O2 treatment for 2 h) | ↑ between pre-senescent and senescent astrocytes | pre vs. senescent = +2.05 | RNA-seq | GSE58910 | [74] |
| Primary astrocyte cell line | Homo Sapiens (in vitro) | IR-induced senescence (exposed to 10 Gy) | ↑ between non-senescent and senescent astrocytes | +1.21 | RNA-seq | NA | [75] | |
| Vasculature | Aortic smooth muscle cells | Homo Sapiens (in vitro) | Replicative senescence (serial passages growing population = PDL 14 senescent population = PDL 39–42) | ↓ in VSMC from late PDL vs. early PDL | −2.8 | cDNA microarray | NA | [76] |
| Endothelial cell lines (HUVEC & HAEC) | Homo Sapiens (in vitro) | IR-induced senescence (exposed to 4 Gy) | ↓ in senescent ECs | HAECs = −0.21 | RNA-seq | GSE130727 | [65] | |
| Pericytes from brain of young and old mice (C57Bl/6) | Mus Musculus (in vivo) | Aging (young mice: 3 months old aged mice: 28 months old) | ↓ in pericytes from young to old mice brain | ND % expression: 20% (young) vs. 10% (aged) | single-cell RNA-seq | NA | [77] | |
| Arterial endothelial cells from aortic artery | Macaca Fascicularis (in vivo) | Replicative senescence (young monkey: 4–6 years old old monkey: 18–21 years old) | ↓ in senescent aortic ECs | −0.38 | single-cell RNA-seq | GSE117715 | [78] | |
| Endothelial cells derived from embryonic stem cells | Homo Sapiens (in vitro) | Replicative senescence (serial passages in FOXO3A−/− ECs) | ↓ in senescent FOXO3A−/− ECs | −0.87 | single-cell RNA-seq | GSE117715 | [78] | |
| Others | Prostate and uterus (healthy samples) | Homo Sapiens (in vivo) | Aging (1-year follow-up in healthy individuals 20–79 years old) | ↓ in healthy tissues upon aging (1 year) | Prostate = −0.01 Uterus = −0.02 | RNA-seq | NA | [79] |
| Breast and uterus tumor samples | Homo Sapiens (in vivo) | Aging (1-year follow-up in breast and uterus cancer patients) | ↓ in cancerous tissues upon aging (1 year) | Breast = −1.94 Uterus = −1.77 | RNA-seq | NA | [79] | |
| Platelets from healthy donors | Homo Sapiens (in vitro) | Replicative senescence (healthy platelets stored under standard blood banking conditions for 5 days) | ↑ between day 0 and day 5 | +3.00 | microarray | NA | [80] | |
| Brain tumor samples | Homo Sapiens (in vivo) | Aging (1-year follow-up in brain cancer patients) | ↑ in cancerous tissue upon aging (1 year) | +1.75 | RNA-seq | NA | [79] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thorin-Trescases, N.; Labbé, P.; Mury, P.; Lambert, M.; Thorin, E. Angptl2 is a Marker of Cellular Senescence: The Physiological and Pathophysiological Impact of Angptl2-Related Senescence. Int. J. Mol. Sci. 2021, 22, 12232. https://doi.org/10.3390/ijms222212232
Thorin-Trescases N, Labbé P, Mury P, Lambert M, Thorin E. Angptl2 is a Marker of Cellular Senescence: The Physiological and Pathophysiological Impact of Angptl2-Related Senescence. International Journal of Molecular Sciences. 2021; 22(22):12232. https://doi.org/10.3390/ijms222212232
Chicago/Turabian StyleThorin-Trescases, Nathalie, Pauline Labbé, Pauline Mury, Mélanie Lambert, and Eric Thorin. 2021. "Angptl2 is a Marker of Cellular Senescence: The Physiological and Pathophysiological Impact of Angptl2-Related Senescence" International Journal of Molecular Sciences 22, no. 22: 12232. https://doi.org/10.3390/ijms222212232
APA StyleThorin-Trescases, N., Labbé, P., Mury, P., Lambert, M., & Thorin, E. (2021). Angptl2 is a Marker of Cellular Senescence: The Physiological and Pathophysiological Impact of Angptl2-Related Senescence. International Journal of Molecular Sciences, 22(22), 12232. https://doi.org/10.3390/ijms222212232






