Experimental Models, Induction Protocols, and Measured Parameters in Dry Eye Disease: Focusing on Practical Implications for Experimental Research
Abstract
:1. Introduction
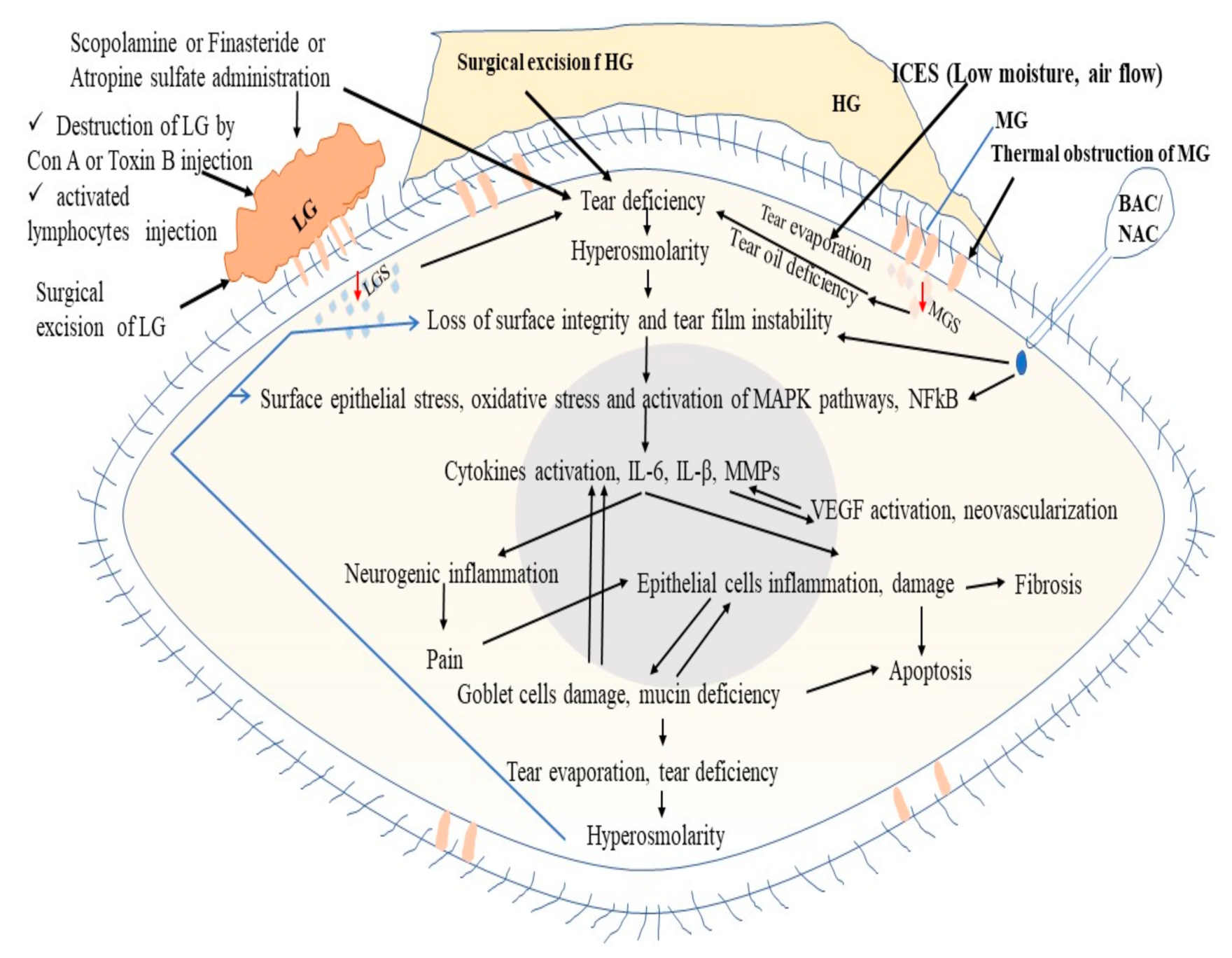
2. Pathophysiology of Dry Eye Disease (DED)
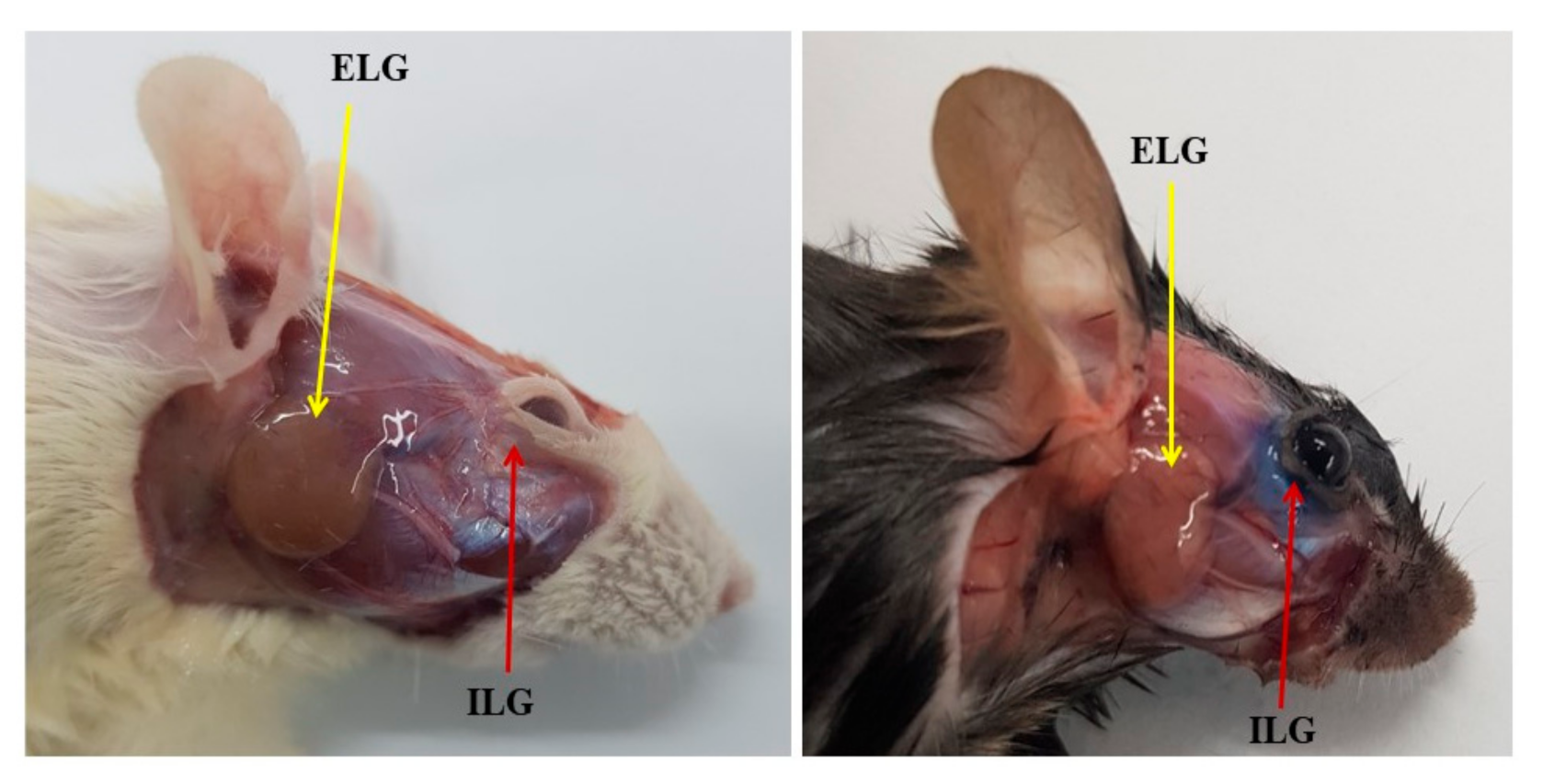
2.1. Lacrimal Glands
2.2. Meibomian Glands (MBGs)
2.3. Conjunctival and Corneal Epithelium, Goblet Cells, and Mucins
2.4. Tear Film
2.5. Harderian Gland and Nictitating Membrane (NM)
3. Classification of Dry Eye
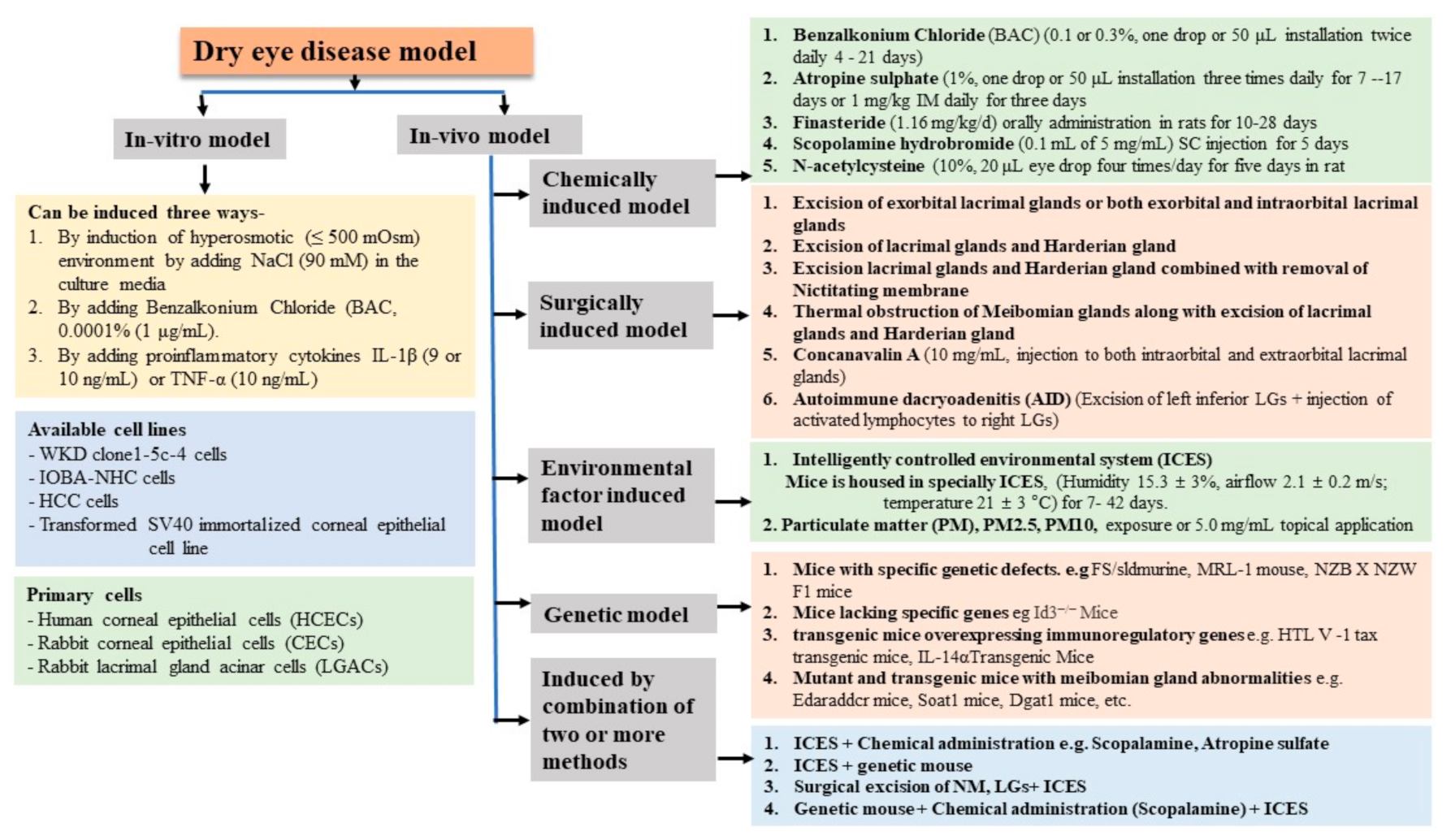
3.1. In Vitro Dry Eye Model
3.2. In Vivo Dry Eye Models
3.2.1. Chemically Induced Dry Eye Model
Benzalkonium Chloride (BAC)
3.2.2. Surgically Induced Dry Eye Model
3.2.3. Environmental Factors-Induced DED Model
3.2.4. Genetically Engineered Dry Eye Model
3.2.5. Combining Methods
4. Evaluation of DED Severity and Therapeutic Efficacy of Candidate Drugs in an Experimental Model
4.1. Inflammatory Index and Clinical Scoring
4.2. Tear Deficiency-Related Tests
4.2.1. Schirmer’s Test and Phenol Red Thread (PRT) Test
4.2.2. Tear Breakup Time
4.3. Surface Structural Damage
4.4. Analysis of Molecular Changes and Histopathological Changes
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baudouin, C.; Aragona, P.; Messmer, E.M.; Tomlinson, A.; Calonge, M.; Boboridis, K.G.; Akova, Y.A.; Geerling, G.; Labetoulle, M.; Rolando, M. Role of hyperosmolarity in the pathogenesis and management of dry eye disease: Proceedings of the OCEAN group meeting. Ocul. Surf. 2013, 11, 246–258. [Google Scholar] [CrossRef] [Green Version]
- Johnson, M.E.; Murphy, P.J. Changes in the tear film and ocular surface from dry eye syndrome. Prog. Retin. Eye Res. 2004, 23, 449–474. [Google Scholar] [CrossRef] [PubMed]
- Schaumberg, D.A.; Dana, R.; Buring, J.E.; Sullivan, D.A. Prevalence of dry eye disease among US men: Estimates from the Physicians’ Health Studies. Arch. Ophthalmol. 2009, 127, 763–768. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drew, V.J.; Tseng, C.L.; Seghatchian, J.; Burnouf, T. Reflections on Dry Eye Syndrome Treatment: Therapeutic Role of Blood Products. Front. Med. 2018, 5, 33. [Google Scholar] [CrossRef] [Green Version]
- Schaumberg, D.A.; Uchino, M.; Christen, W.G.; Semba, R.D.; Buring, J.E.; Li, J.Z. Patient reported differences in dry eye disease between men and women: Impact, management, and patient satisfaction. PLoS ONE 2013, 8, e76121. [Google Scholar] [CrossRef]
- McDonald, M.; Patel, D.A.; Keith, M.S.; Snedecor, S.J. Economic and Humanistic Burden of Dry Eye Disease in Europe, North America, and Asia: A Systematic Literature Review. Ocul. Surf. 2016, 14, 144–167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mizuno, Y.; Yamada, M.; Shigeyasu, C. Annual direct cost of dry eye in Japan. Clin. Ophthalmol. 2012, 6, 755–760. [Google Scholar] [CrossRef] [Green Version]
- Zerhouni, E.A. US biomedical research: Basic, translational, and clinical sciences. JAMA 2005, 294, 1352–1358. [Google Scholar] [CrossRef] [PubMed]
- McArthur, R.A.; Borsini, F. What Do You Mean by “Translational Research”? An Enquiry through Animal and Translational Models for CNS Drug Discovery: Psychiatric Disorders. In Animal and Translational Models for CNS Drug Discovery. Vol. 1: Psychiatric Disorders; Academic Press: New York, NY, USA, 2008. [Google Scholar]
- Mateo Orobia, A.J.; Saa, J.; Ollero Lorenzo, A.; Herreras, J.M. Combination of hyaluronic acid, carmellose, and osmoprotectants for the treatment of dry eye disease. Clin. Ophthalmol. 2018, 12, 453–461. [Google Scholar] [CrossRef] [Green Version]
- Coursey, T.G.; de Paiva, C.S. Managing Sjogren’s Syndrome and non-Sjogren Syndrome dry eye with anti-inflammatory therapy. Clin. Ophthalmol. 2014, 8, 1447–1458. [Google Scholar] [PubMed] [Green Version]
- Stern, M.E.; Gao, J.; Siemasko, K.F.; Beuerman, R.W.; Pflugfelder, S.C. The role of the lacrimal functional unit in the pathophysiology of dry eye. Exp. Eye Res. 2004, 78, 409–416. [Google Scholar] [CrossRef]
- Zhang, X.; Jeyalatha, V.M.; Qu, Y.; He, X.; Ou, S.; Bu, J.; Jia, C.; Wang, J.; Wu, H.; Liu, Z.; et al. Dry Eye Management: Targeting the Ocular Surface Microenvironment. Int. J. Mol. Sci. 2017, 18, 1398. [Google Scholar] [CrossRef] [Green Version]
- Shinomiya, K.; Ueta, M.; Kinoshita, S. A new dry eye mouse model produced by exorbital and intraorbital lacrimal gland excision. Sci. Rep. 2018, 8, 1483. [Google Scholar] [CrossRef] [PubMed]
- Conrady, C.D.; Joos, Z.P.; Patel, B.C. Review: The Lacrimal Gland and Its Role in Dry Eye. J. Ophthalmol. 2016, 2016, 7542929. [Google Scholar] [CrossRef] [Green Version]
- Hall, N.P.; Peden, R. FRCOphth Part 1: 400 SBAs and CRQs; JP Medical Ltd.: London, UK, 2016; p. 121. ISBN 9781909836365. [Google Scholar]
- Presutti, L.M.; Mattioli, F. Endoscopic Surgery of the Lacrimal Drainage System; Springer: Berlin/Heidelberg, Germany, 2015; p. 4. ISBN 978-3319206332. [Google Scholar]
- Friedman, N.J.K.; Peter, K. Essentials of Ophthalmology; Elsevier Health Sciences: Philadelphia, PA, USA, 2007; p. 7. ISBN 1416029079. [Google Scholar]
- Lemp, M.A.M.; Marquardt, R. The Dry Eye: A Comprehensive Guide; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013; p. 58. ISBN 9783642581304. [Google Scholar]
- Gipson, I.K.; Argueso, P. Role of mucins in the function of the corneal and conjunctival epithelia. Int. Rev. Cytol. 2003, 231, 1–49. [Google Scholar] [PubMed]
- Bron, A.J.; Benjamin, L.; Snibson, G.R. Meibomian gland disease. Classification and grading of lid changes. Eye 1991, 5 Pt 4, 395–411. [Google Scholar] [CrossRef] [PubMed]
- Opitz, D.; Harthan, J.; Fromstein, S.; Hauswirth, S. Diagnosis and management of meibomian gland dysfunction: Optometrists’ perspective. Clin. Optom. 2015, 7, 59–69. [Google Scholar] [CrossRef] [Green Version]
- Asano, N.; Wiseman, J.M.; Tsuji, F.; Kawazu, K. Limited Azithromycin Localization to Rabbit Meibomian Glands Revealed by LC-MS-Based Bioanalysis and DESI Imaging. Biol. Pharm. Bull. 2017, 40, 1586–1589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheriyan, T.; Schmid, T.M.; Spector, M. Presence and distribution of the lubricating protein, lubricin, in the meibomian gland in rabbits. Mol. Vis. 2011, 17, 3055–3061. [Google Scholar]
- Tong, L.; Gupta, P.K. Need for Animal Models of Meibomian Gland Dysfunction. Ophthalmol. Ther. 2016, 5, 129–134. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Posadas, L.; Contreras-Ruiz, L.; Soriano-Romani, L.; Dartt, D.A.; Diebold, Y. Conjunctival Goblet Cell Function: Effect of Contact Lens Wear and Cytokines. Eye Contact Lens 2016, 42, 83–90. [Google Scholar] [CrossRef] [Green Version]
- Dartt, D. The Conjunctiva—Structure and Function. In Duane’s Foundations of Clinical Ophthalmology 2; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2006. [Google Scholar]
- Mantelli, F.; Argueso, P. Functions of ocular surface mucins in health and disease. Curr. Opin. Allergy Clin. Immunol. 2008, 8, 477–483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bron, A.J. Non-Sjogren Dry Eye: Pathogenesis Diagnosis and Animal Models. In Lacrimal Gland, Tear Film, and Dry Eye Syndromes; Springer: Boston, MA, USA, 1994; pp. 471–488. [Google Scholar]
- Dartt, D.A.; Willcox, M.D. Complexity of the tear film: Importance in homeostasis and dysfunction during disease. Exp. Eye Res. 2013, 117, 1–3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, H.B.; Yamabayashi, S.; Ou, B.; Tanaka, Y.; Ohno, S.; Tsukahara, S. Structure and composition of rat precorneal tear film. A study by an in vivo cryofixation. Investig. Ophthalmol. Vis. Sci. 1997, 38, 381–387. [Google Scholar]
- Tran, C.H.; Routledge, C.; Miller, J.; Miller, F.; Hodson, S.A. Examination of murine tear film. Investig. Ophthalmol. Vis. Sci. 2003, 44, 3520–3525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prydal, J.I.; Muir, M.G.; Dilly, P.N. Comparison of tear film thickness in three species determined by the glass fibre method and confocal microscopy. Eye 1993, 7 Pt 3, 472–475. [Google Scholar] [CrossRef]
- Prydal, J.I.; Campbell, F.W. Study of precorneal tear film thickness and structure by interferometry and confocal microscopy. Investig. Ophthalmol. Vis. Sci. 1992, 33, 1996–2005. [Google Scholar]
- Mishima, S. Some physiological aspects of the precorneal tear film. Arch. Ophthalmol. 1965, 73, 233–241. [Google Scholar] [CrossRef]
- Payne, A.P. The harderian gland: A tercentennial review. J. Anat. 1994, 185 Pt 1, 1–49. [Google Scholar]
- Dos Reis, E.R.; Danielli Nicola, E.M.; Humberto Nicola, J. Harderian gland of wistar rats revised as a protoporphyrin IX producer. Braz. J. Morphol. Sci. 2005, 22, 43–51. [Google Scholar]
- Djeridane, Y. The harderian gland and its excretory duct in the Wistar rat. A histological and ultrastructural study. J. Anat. 1994, 184 Pt 3, 553–566. [Google Scholar]
- Umeda, Y.; Nakamura, S.; Fujiki, K.; Toshida, H.; Saito, A.; Murakami, A. Distribution of goblet cells and MUC5AC mRNA in the canine nictitating membrane. Exp. Eye Res. 2010, 91, 721–726. [Google Scholar] [CrossRef]
- Chae, J.J.; Shin, Y.J.; Lee, J.D.; Seo, K.; Elisseeff, J.H. Nictitating membrane fixation improves stability of the contact lens on the animal corneal surface. PLoS ONE 2018, 13, e0194795. [Google Scholar] [CrossRef] [Green Version]
- Gelatt, K.M. (Ed.) Canine Conjuntiva and Nictitating Membrane, 4th ed.; Wiley-Blackwell: Ames, IA, USA, 2007; p. 675. [Google Scholar]
- Ning, Y.; Bhattacharya, D.; Jones, R.E.; Zhao, F.; Chen, R.; Zhang, J.; Wang, M. Evaluating the Functionality of Conjunctiva Using a Rabbit Dry Eye Model. J. Ophthalmol. 2016, 2016, 3964642. [Google Scholar] [CrossRef] [Green Version]
- Bhattacharya, D.; Ning, Y.; Zhao, F.; Stevenson, W.; Chen, R.; Zhang, J.; Wang, M. Tear Production after Bilateral Main Lacrimal Gland Resection in Rabbits. Investig. Ophthalmol. Vis. Sci. 2015, 56, 7774–7783. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Z.Y.; Liang, Q.F.; Yu, G.Y. Establishment of a rabbit model for keratoconjunctivitis sicca. Cornea 2011, 30, 1024–1029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, N.; Deng, X.; Gao, Y.; Zhang, S.; He, M.; Zhao, D. Establishment of the mild, moderate and severe dry eye models using three methods in rabbits. BMC Ophthalmol. 2013, 13, 50. [Google Scholar] [CrossRef] [Green Version]
- Craig, J.P.; Nichols, K.K.; Akpek, E.K.; Caffery, B.; Dua, H.S.; Joo, C.K.; Liu, Z.; Nelson, J.D.; Nichols, J.J.; Tsubota, K.; et al. TFOS DEWS II Definition and Classification Report. Ocul. Surf. 2017, 15, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Brasnu, E.; Brignole-Baudouin, F.; Riancho, L.; Guenoun, J.M.; Warnet, J.M.; Baudouin, C. In vitro effects of preservative-free tafluprost and preserved latanoprost, travoprost, and bimatoprost in a conjunctival epithelial cell line. Curr. Eye Res. 2008, 33, 303–312. [Google Scholar] [CrossRef]
- Clouzeau, C.; Godefroy, D.; Riancho, L.; Rostene, W.; Baudouin, C.; Brignole-Baudouin, F. Hyperosmolarity potentiates toxic effects of benzalkonium chloride on conjunctival epithelial cells in vitro. Mol. Vis. 2012, 18, 851–863. [Google Scholar]
- Debbasch, C.; Brignole, F.; Pisella, P.J.; Warnet, J.M.; Rat, P.; Baudouin, C. Quaternary ammoniums and other preservatives’ contribution in oxidative stress and apoptosis on Chang conjunctival cells. Investig. Ophthalmol. Vis. Sci. 2001, 42, 642–652. [Google Scholar]
- Debbasch, C.; Pisella, P.J.; De Saint Jean, M.; Rat, P.; Warnet, J.M.; Baudouin, C. Mitochondrial activity and glutathione injury in apoptosis induced by unpreserved and preserved beta-blockers on Chang conjunctival cells. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2525–2533. [Google Scholar]
- De Saint Jean, M.; Brignole, F.; Bringuier, A.F.; Bauchet, A.; Feldmann, G.; Baudouin, C. Effects of benzalkonium chloride on growth and survival of Chang conjunctival cells. Investig. Ophthalmol. Vis. Sci. 1999, 40, 619–630. [Google Scholar]
- Diebold, Y.; Calonge, M.; Enriquez de Salamanca, A.; Callejo, S.; Corrales, R.M.; Saez, V.; Siemasko, K.F.; Stern, M.E. Characterization of a spontaneously immortalized cell line (IOBA-NHC) from normal human conjunctiva. Investig. Ophthalmol. Vis. Sci. 2003, 44, 4263–4274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schicht, M.; Garreis, F.; Hartjen, N.; Beileke, S.; Jacobi, C.; Sahin, A.; Holland, D.; Schroder, H.; Hammer, C.M.; Paulsen, F.; et al. SFTA3—A novel surfactant protein of the ocular surface and its role in corneal wound healing and tear film surface tension. Sci. Rep. 2018, 8, 9791. [Google Scholar] [CrossRef]
- Li, H.; Li, J.F.; Hou, C.T.; Li, J.J.; Peng, H.; Wang, Q. The effect of astaxanthin on inflammation in hyperosmolarity of experimental dry eye model in vitro and in vivo. Exp. Eye Res. 2020, 197, 108113. [Google Scholar] [CrossRef]
- Ma, B.K.; Zhou, Y.F.; Liu, R.J.; Zhang, K.; Yang, T.T.; Hu, C.X.; Gao, Y.F.; Lan, Q.Q.; Liu, Y.Y.; Yang, X.L.; et al. Pigment epithelium-derived factor (PEDF) plays anti-inflammatory roles in the pathogenesis of dry eye disease. Ocul. Surf. 2021, 20, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Park, B.; Jo, K.; Lee, T.G.; Hyun, S.W.; Kim, J.S.; Kim, C.S. Polydatin Inhibits NLRP3 Inflammasome in Dry Eye Disease by Attenuating Oxidative Stress and Inhibiting the NF-kappa B Pathway. Nutrients 2019, 11, 2792. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Z.; Chen, D.; Chen, X.; Bian, F.; Gao, N.; Li, J.M.; Pflugfelder, S.C.; Li, D.Q. Autophagy Activation Protects Ocular Surface from Inflammation in a Dry Eye Model In Vitro. Int. J. Mol. Sci. 2020, 21, 8966. [Google Scholar] [CrossRef]
- Lu, Q.Z.; Yin, H.B.; Grant, M.P.; Elisseeff, J.H. An In Vitro Model for the Ocular Surface and Tear Film System. Sci. Rep. 2017, 7, 6163. [Google Scholar] [CrossRef]
- Villani, E.; Marelli, L.; Dellavalle, A.; Serafino, M.; Nucci, P. Latest evidences on meibomian gland dysfunction diagnosis and management. Ocul. Surf. 2020, 18, 871–892. [Google Scholar] [CrossRef] [PubMed]
- Lackner, M.; Guggenbichler, J.P. “Antimicrobial Surfaces” Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2013. [Google Scholar] [CrossRef]
- Pisella, P.J.; Fillacier, K.; Elena, P.P.; Debbasch, C.; Baudouin, C. Comparison of the effects of preserved and unpreserved formulations of timolol on the ocular surface of albino rabbits. Ophthalmic Res. 2000, 32, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Baudouin, C.; Labbe, A.; Liang, H.; Pauly, A.; Brignole-Baudouin, F. Preservatives in eyedrops: The good, the bad and the ugly. Prog. Retin. Eye Res. 2010, 29, 312–334. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Baudouin, C.; Pauly, A.; Brignole-Baudouin, F. Conjunctival and corneal reactions in rabbits following short- and repeated exposure to preservative-free tafluprost, commercially available latanoprost and 0.02% benzalkonium chloride. Br. J. Ophthalmol. 2008, 92, 1275–1282. [Google Scholar] [CrossRef]
- Chen, W.; Li, Z.; Hu, J.; Zhang, Z.; Chen, L.; Chen, Y.; Liu, Z. Corneal alternations induced by topical application of benzalkonium chloride in rabbit. PLoS ONE 2011, 6, e26103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pauly, A.; Brignole-Baudouin, F.; Labbe, A.; Liang, H.; Warnet, J.M.; Baudouin, C. New tools for the evaluation of toxic ocular surface changes in the rat. Investig. Ophthalmol. Vis. Sci. 2007, 48, 5473–5483. [Google Scholar] [CrossRef] [Green Version]
- Li, C.Y.; Song, Y.Y.; Luan, S.H.; Wan, P.X.; Li, N.Y.; Tang, J.; Han, Y.; Xiong, C.J.; Wang, Z.C. Research on the Stability of a Rabbit Dry Eye Model Induced by Topical Application of the Preservative Benzalkonium Chloride. PLoS ONE 2012, 7, 33688. [Google Scholar]
- Xiong, C.; Chen, D.; Liu, J.B.; Liu, B.Q.; Li, N.Y.; Zhou, Y.; Liang, X.W.; Ma, P.; Ye, C.T.; Ge, J.; et al. A rabbit dry eye model induced by topical medication of a preservative benzalkonium chloride. Investig. Ophthalmol. Vis. Sci. 2008, 49, 1850–1856. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, K.A.; Hyun, L.C.; Jung, S.H.; Yang, S.J. The leaves of Diospyros kaki exert beneficial effects on a benzalkonium chloride-induced murine dry eye model. Mol. Vis. 2016, 22, 284–293. [Google Scholar]
- Serrander, A.M.; Peek, K.E. Changes in contact lens comfort related to the menstrual cycle and menopause. A review of articles. J. Am. Optom. Assoc. 1993, 64, 162–166. [Google Scholar]
- Singh, S.; Moksha, L.; Sharma, N.; Titiyal, J.S.; Biswas, N.R.; Velpandian, T. Development and evaluation of animal models for sex steroid deficient dry eye. J. Pharmacol. Toxicol. Methods 2014, 70, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Li, K.; Yang, Z.; Wang, Y.; Si, H. The Effect of the Aqueous Extract of Bidens Pilosa, L. on Androgen Deficiency Dry Eye in Rats. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2016, 39, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Burgalassi, S.; Panichi, L.; Chetoni, P.; Saettone, M.F.; Boldrini, E. Development of a simple dry eye model in the albino rabbit and evaluation of some tear substitutes. Ophthalmic Res. 1999, 31, 229–235. [Google Scholar] [CrossRef]
- Shafaa, M.W.; El Shazly, L.H.; El Shazly, A.H.; El Gohary, A.A.; El Hossary, G.G. Efficacy of topically applied liposome-bound tetracycline in the treatment of dry eye model. Vet. Ophthalmol. 2011, 14, 18–25. [Google Scholar] [CrossRef]
- Silva, D.A.; Nai, G.A.; Giuffrida, R.; Barbero, R.C.; Kuhn, J.M.P.; da Silva, A.C.; Pereira, R.H.Z.; Abbade, M.F.; Zulim, L.; Pereira, C.S.G.; et al. Comparison between fish and linseed oils administered orally for the treatment of experimentally induced keratoconjunctivitis sicca in rabbits. Open Vet. J. 2017, 7, 277–285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.; Kim, C.E.; Ahn, B.N.; Yang, J. Anti-inflammatory effect of hydroxyproline-GQDGLAGPK in desiccation stress-induced experimental dry eye mouse. Sci. Rep. 2017, 7, 7413. [Google Scholar] [CrossRef]
- Kim, C.E.; Oh, H.N.; Lee, J.H.; Yang, J.W. Effects of chondrocyte-derived extracellular matrix in a dry eye mouse model. Mol. Vis. 2015, 21, 1210–1223. [Google Scholar]
- Thermes, F.; Molon-Noblot, S.; Grove, J. Effects of acetylcysteine on rabbit conjunctival and corneal surfaces. A scanning electron microscopy study. Investig. Ophthalmol. Vis. Sci. 1991, 32, 2958–2963. [Google Scholar]
- Urashima, H.; Okamoto, T.; Takeji, Y.; Shinohara, H.; Fujisawa, S. Rebamipide increases the amount of mucin-like substances on the conjunctiva and cornea in the N-acetylcysteine-treated in vivo model. Cornea 2004, 23, 613–619. [Google Scholar] [CrossRef]
- Luo, L.J.; Lai, J.Y. Epigallocatechin Gallate-Loaded Gelatin-g-Poly(N-Isopropylacrylamide) as a New Ophthalmic Pharmaceutical Formulation for Topical Use in the Treatment of Dry Eye Syndrome. Sci. Rep. 2017, 7, 9380. [Google Scholar] [CrossRef] [Green Version]
- Ji, H.; Zhu, Y.; Zhang, Y.; Jia, Y.; Li, Y.; Ge, J.; Zhuo, Y. The Effect of Dry Eye Disease on Scar Formation in Rabbit Glaucoma Filtration Surgery. Int. J. Mol. Sci. 2017, 18, 1150. [Google Scholar] [CrossRef] [Green Version]
- Tseng, C.L.; Hung, Y.J.; Chen, Z.Y.; Fang, H.W.; Chen, K.H. Synergistic Effect of Artificial Tears Containing Epigallocatechin Gallate and Hyaluronic Acid for the Treatment of Rabbits with Dry Eye Syndrome. PLoS ONE 2016, 11, e0157982. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, H.C.; Chen, Z.Y.; Wang, T.J.; Drew, V.J.; Tseng, C.L.; Fang, H.W.; Lin, F.H. Herbal Supplement in a Buffer for Dry Eye Syndrome Treatment. Int. J. Mol. Sci. 2017, 18, 1697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, Z.; Liu, X.; Zhou, T.; Wang, Y.; Bai, L.; He, H.; Liu, Z. A mouse dry eye model induced by topical administration of benzalkonium chloride. Mol. Vis. 2011, 17, 257–264. [Google Scholar] [PubMed]
- Xiao, X.; Luo, P.; Zhao, H.; Chen, J.; He, H.; Xu, Y.; Lin, Z.; Zhou, Y.; Xu, J.; Liu, Z. Amniotic membrane extract ameliorates benzalkonium chloride-induced dry eye in a murine model. Exp. Eye Res. 2013, 115, 31–40. [Google Scholar] [CrossRef]
- Xiao, X.; He, H.; Lin, Z.; Luo, P.; He, H.; Zhou, T.; Zhou, Y.; Liu, Z. Therapeutic effects of epidermal growth factor on benzalkonium chloride-induced dry eye in a mouse model. Investig. Ophthalmol. Vis. Sci. 2012, 53, 191–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beyazyildiz, E.; Pinarli, F.A.; Beyazyildiz, O.; Hekimoglu, E.R.; Acar, U.; Demir, M.N.; Albayrak, A.; Kaymaz, F.; Sobaci, G.; Delibasi, T. Efficacy of topical mesenchymal stem cell therapy in the treatment of experimental dry eye syndrome model. Stem Cells Int. 2014, 2014, 250230. [Google Scholar] [CrossRef]
- Marques, D.L.; Alves, M.; Modulo, C.M.; da Silva, L.E.C.M.; Reinach, P. Lacrimal osmolarity and ocular surface in experimental model of dry eye caused by toxicity. Rev. Bras. Oftalmol. 2014, 74, 68–72. [Google Scholar] [CrossRef] [Green Version]
- Na, Y.J.; Choi, K.J.; Park, S.B.; Sung, H.R.; Jung, W.H.; Kim, H.Y.; Rhee, S.D.; Kim, K.Y. Protective effects of carbenoxolone, an 11beta-HSD1 inhibitor, against chemical induced dry eye syndrome. Int. J. Program. Cell Death 2017, 22, 1441–1453. [Google Scholar] [CrossRef] [PubMed]
- El-Shazly, A.H.; El-Gohary, A.A.; El-Shazly, L.H.; El-Hossary, G.G. Comparison between two cyclooxygenase inhibitors in an experimental dry eye model in albino rabbits. Acta Pharm. 2008, 58, 163–173. [Google Scholar] [CrossRef] [Green Version]
- Altinors, D.D.; Bozbeyoglu, S.; Karabay, G.; Akova, Y.A. Evaluation of ocular surface changes in a rabbit dry eye model using a modified impression cytology technique. Curr. Eye Res. 2007, 32, 301–307. [Google Scholar] [CrossRef]
- Li, K.; Zhang, C.; Yang, Z.; Wang, Y.; Si, H. Evaluation of a novel dry eye model induced by oral administration of finasteride. Mol. Med. Rep. 2017, 16, 8763–8770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Kang, B.; Woo, I.H.; Eom, Y.; Lee, H.K.; Kim, H.M.; Song, J.S. Effects of Topical Mucolytic Agents on the Tears and Ocular Surface: A Plausible Animal Model of Mucin-Deficient Dry Eye. Investig. Ophthalmol. Vis. Sci. 2018, 59, 3104–3114. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.; Wang, Y.; Reinach, P.S.; Ren, Y.; Li, J.; Hua, S.; Lu, H.; Chen, W. Dynamic ocular surface and lacrimal gland changes induced in experimental murine dry eye. PLoS ONE 2015, 10, e0115333. [Google Scholar]
- Viau, S.; Maire, M.A.; Pasquis, B.; Gregoire, S.; Fourgeux, C.; Acar, N.; Bretillon, L.; Creuzot-Garcher, C.P.; Joffre, C. Time course of ocular surface and lacrimal gland changes in a new scopolamine-induced dry eye model. Albrecht Graefes Arch. Klin. Exp. Ophthalmol. 2008, 246, 857–867. [Google Scholar] [CrossRef]
- Maitchouk, D.Y.; Beuerman, R.W.; Ohta, T.; Stern, M.; Varnell, R.J. Tear production after unilateral removal of the main lacrimal gland in squirrel monkeys. Arch. Ophthalmol. 2000, 118, 246–252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Polans, A.S.; McAnulty, J.F.; Phelps, P.O.; Darjatmoko, S.R.; Burris, C.K.; Albert, D.M. Pilot Study of a “Large-Eye”, Surgically Induced Dry Eye Rabbit Model by Selective Removal of the Harderian, Lacrimal, and Meibomian Glands. Ocul. Oncol. Pathol. 2017, 4, 52–56. [Google Scholar] [CrossRef]
- Guo, Z.; Song, D.; Azzarolo, A.M.; Schechter, J.E.; Warren, D.W.; Wood, R.L.; Mircheff, A.K.; Kaslow, H.R. Autologous lacrimal-lymphoid mixed-cell reactions induce dacryoadenitis in rabbits. Exp. Eye Res. 2000, 71, 23–31. [Google Scholar] [CrossRef]
- Lin, H.; Liu, Y.; Kambhampati, S.P.; Hsu, C.C.; Kannan, R.M.; Yiu, S.C. Subconjunctival dendrimer-drug therapy for the treatment of dry eye in a rabbit model of induced autoimmune dacryoadenitis. Ocul. Surf. 2018, 16, 415–423. [Google Scholar] [CrossRef]
- Tiegs, G.; Hentschel, J.; Wendel, A. A T cell-dependent experimental liver injury in mice inducible by concanavalin A. J. Clin. Investig. 1992, 90, 196–203. [Google Scholar] [CrossRef]
- Ratay, M.L.; Balmert, S.C.; Acharya, A.P.; Greene, A.C.; Meyyappan, T.; Little, S.R. TRI Microspheres prevent key signs of dry eye disease in a murine, inflammatory model. Sci. Rep. 2017, 7, 17527. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.J.; Ko, A.Y.; Ko, J.H.; Lee, H.J.; Kim, M.K.; Wee, W.R.; Khwarg, S.I.; Oh, J.Y. Mesenchymal stem/stromal cells protect the ocular surface by suppressing inflammation in an experimental dry eye. J. Am. Soc. Gene Ther. 2015, 23, 139–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, M.J.; Kim, D.H.; Ryu, J.S.; Ko, A.Y.; Ko, J.H.; Kim, M.K.; Wee, W.R.; Khwarg, S.I.; Oh, J.Y. Topical TSG-6 Administration Protects the Ocular Surface in Two Mouse Models of Inflammation-Related Dry Eye. Investig. Ophthalmol. Vis. Sci. 2015, 56, 5175–5181. [Google Scholar] [CrossRef]
- Seo, M.J.; Kim, J.M.; Lee, M.J.; Sohn, Y.S.; Kang, K.K.; Yoo, M. The therapeutic effect of DA-6034 on ocular inflammation via suppression of MMP-9 and inflammatory cytokines and activation of the MAPK signaling pathway in an experimental dry eye model. Curr. Eye Res. 2010, 35, 165–175. [Google Scholar] [CrossRef]
- Zheng, W.; Ma, M.; Du, E.; Zhang, Z.; Jiang, K.; Gu, Q.; Ke, B. Therapeutic efficacy of fibroblast growth factor 10 in a rabbit model of dry eye. Mol. Med. Rep. 2015, 12, 7344–7350. [Google Scholar] [CrossRef] [Green Version]
- Lekhanont, K.; Park, C.Y.; Combs, J.C.; Suwan-Apichon, O.; Rangsin, R.; Chuck, R.S. Effect of topical olopatadine and epinastine in the botulinum toxin B-induced mouse model of dry eye. Off. J. Assoc. Ocul. Pharmacol. Ther. 2007, 23, 83–88. [Google Scholar] [CrossRef]
- Kang, W.S.; Jung, E.; Kim, J. Aucuba japonica Extract and Aucubin Prevent Desiccating Stress-Induced Corneal Epithelial Cell Injury and Improve Tear Secretion in a Mouse Model of Dry Eye Disease. Molecules 2018, 23, 2599. [Google Scholar] [CrossRef] [Green Version]
- Kim, C.S.; Jo, K.; Lee, I.S.; Kim, J. Topical Application of Apricot Kernel Extract Improves Dry Eye Symptoms in a Unilateral Exorbital Lacrimal Gland Excision Mouse. Nutrients 2016, 8, 750. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, B.; Lee, I.S.; Hyun, S.W.; Jo, K.; Lee, T.G.; Kim, J.S.; Kim, C.S. The Protective Effect of Polygonum cuspidatum (PCE) Aqueous Extract in a Dry Eye Model. Nutrients 2018, 10, 1550. [Google Scholar] [CrossRef] [Green Version]
- She, Y.; Li, J.; Xiao, B.; Lu, H.; Liu, H.; Simmons, P.A.; Vehige, J.G.; Chen, W. Evaluation of a Novel Artificial Tear in the Prevention and Treatment of Dry Eye in an Animal Model. Off. J. Assoc. Ocul. Pharmacol. Ther. 2015, 31, 525–530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, W.; Zhang, X.; Zhang, J.; Chen, J.; Wang, S.; Wang, Q.; Qu, J. A murine model of dry eye induced by an intelligently controlled environmental system. Investig. Ophthalmol. Vis. Sci. 2008, 49, 1386–1391. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mimura, T.; Ichinose, T.; Yamagami, S.; Fujishima, H.; Kamei, Y.; Goto, M.; Takada, S.; Matsubara, M. Airborne particulate matter (PM2.5) and the prevalence of allergic conjunctivitis in Japan. Sci. Total Environ. 2014, 487, 493–499. [Google Scholar] [CrossRef]
- Song, S.J.; Hyun, S.W.; Lee, T.G.; Park, B.; Jo, K.; Kim, C.S. New application for assessment of dry eye syndrome induced by particulate matter exposure. Ecotoxicol. Environ. Saf. 2020, 205, 111125. [Google Scholar] [CrossRef]
- Tan, G.; Li, J.; Yang, Q.; Wu, A.; Qu, D.Y.; Wang, Y.; Ye, L.; Bao, J.; Shao, Y. Air pollutant particulate matter 2.5 induces dry eye syndrome in mice. Sci. Rep. 2018, 8, 17828. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Tan, G.; Ding, X.; Wang, Y.; Wu, A.; Yang, Q.; Ye, L.; Shao, Y. A mouse dry eye model induced by topical administration of the air pollutant particulate matter 10. Biomed. Pharmacother. 2017, 96, 524–534. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zhang, X.; Li, J.; Wang, Y.; Chen, Q.; Hou, C.; Garrett, Q. Efficacy of osmoprotectants on prevention and treatment of murine dry eye. Investig. Ophthalmol. Vis. Sci. 2013, 54, 6287–6297. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Zhang, X.; Liu, M.; Zhang, J.; Ye, Y.; Lin, Y.; Luyckx, J.; Qu, J. Trehalose protects against ocular surface disorders in experimental murine dry eye through suppression of apoptosis. Exp. Eye Res. 2009, 89, 311–318. [Google Scholar] [CrossRef]
- Li, J.; Roubeix, C.; Wang, Y.; Shi, S.; Liu, G.; Baudouin, C.; Chen, W. Therapeutic efficacy of trehalose eye drops for treatment of murine dry eye induced by an intelligently controlled environmental system. Mol. Vis. 2012, 18, 317–329. [Google Scholar]
- Park, Y.S.; Gauna, A.E.; Cha, S. Mouse Models of Primary Sjogren’s Syndrome. Curr. Pharm. Des. 2015, 21, 2350–2364. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.A.; Wu, Y.Y.; Lin, C.T.; Kawasumi, M.; Wu, C.H.; Kao, S.Y.; Yang, Y.P.; Hsu, C.C.; Hung, K.F.; Sun, Y.C. Animal models of dry eye: Their strengths and limitations for studying human dry eye disease. J. Chin. Med. Assoc. 2021, 84, 459–464. [Google Scholar] [CrossRef]
- Masli, S.; Dartt, D.A. Mouse Models of Sjogren’s Syndrome with Ocular Surface Disease. Int. J. Mol. Sci. 2020, 21, 9112. [Google Scholar] [CrossRef]
- Cha, S.; Peck, A.B.; Humphreys-Beher, M.G. Progress in understanding autoimmune exocrinopathy using the non-obese diabetic mouse: An update. Crit. Rev. Oral Biol. Med. Off. Publ. Am. Assoc. Oral Biol. 2002, 13, 5–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, C.E.; Lee, J.H.; Yeon, Y.K.; Park, C.H.; Yang, J. Effects of silk fibroin in murine dry eye. Sci. Rep. 2017, 7, 44364. [Google Scholar] [CrossRef] [Green Version]
- McCartney Francis, N.L.; Mizel, D.E.; Frazier Jessen, M.; Kulkarni, A.B.; McCarthy, J.B.; Wahl, S.M. Lacrimal gland inflammation is responsible for ocular pathology in TGF-beta 1 null mice. Am. J. Pathol. 1997, 151, 1281–1288. [Google Scholar]
- Vijmasi, T.; Chen, F.L.Y.T.; Chen, Y.T.; Gallup, M.; McNamara, N. Topical administration of interleukin-1 receptor antagonist as a therapy for aqueous-deficient dry eye in autoimmune disease. Mol. Vis. 2013, 19, 1957–1965. [Google Scholar]
- Nguyen, C.; Singson, E.; Kim, J.Y.; Cornelius, J.G.; Attia, R.; Doyle, M.E.; Bulosan, M.; Cha, S.; Peck, A.B. Sjogren’s syndrome-like disease of C57BL/6.NOD-Aec1Aec2 mice: Gender differences in keratoconjunctivitis sicca defined by a cross-over in the chromosome 3 Aec1 locus. Scand. J. Immunol. 2006, 64, 295–307. [Google Scholar] [CrossRef]
- Arakaki, R.; Eguchi, H.; Yamada, A.; Kudo, Y.; Iwasa, A.; Enkhmaa, T.; Hotta, F.; Mitamura-Aizawa, S.; Mitamura, Y.; Hayashi, Y.; et al. Anti-Inflammatory Effects of Rebamipide Eyedrop Administration on Ocular Lesions in a Murine Model of Primary Sjogren’s Syndrome. PLoS ONE 2014, 9, 98390. [Google Scholar] [CrossRef]
- Chen, Y.T.; Li, S.M.; Nikulina, K.; Porco, T.; Gallup, M.; McNamara, N. Immune profile of squamous metaplasia development in autoimmune regulator-deficient dry eye. Mol. Vis. 2009, 15, 563–576. [Google Scholar] [PubMed]
- Lee, H.S.; Hattori, T.; Park, E.Y.; Stevenson, W.; Chauhan, S.K.; Dana, R. Expression of Toll-Like Receptor 4 Contributes to Corneal Inflammation in Experimental Dry Eye Disease. Investig. Ophthalmol. Vis. Sci. 2012, 53, 5632–5640. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.S.; Chauhan, S.K.; Okanobo, A.; Nallasamy, N.; Dana, R. Therapeutic Efficacy of Topical Epigallocatechin Gallate in Murine Dry Eye. Cornea 2011, 30, 1465–1472. [Google Scholar] [CrossRef] [Green Version]
- Goyal, S.; Chauhan, S.K.; Zhang, Q.; Dana, R. Amelioration of Murine Dry Eye Disease by Topical Antagonist to Chemokine Receptor 2. Arch. Ophthalmol. 2009, 127, 882–887. [Google Scholar] [CrossRef] [Green Version]
- Rashid, S.; Jin, Y.P.; Ecoiffier, T.; Barabino, S.; Schaumberg, D.A.; Dana, R. Topical omega-3 and omega-6 fatty acids for treatment of dry eye. Arch. Ophthalmol. 2008, 126, 219–225. [Google Scholar] [CrossRef] [Green Version]
- Niederkorn, J.Y.; Stern, M.E.; Pflugfelder, S.C.; De Paiva, C.S.; Corrales, R.M.; Gao, J.P.; Siemasko, K. Desiccating stress induces T cell-mediated Sjogren’s syndrome-like lacrimal keratoconjunctivitis. J. Immunol. 2006, 176, 3950–3957. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.M.; Kuang, J.B.; Yu, H.Y.; Wu, Z.N.; Wang, S.Y.; Zhou, S.Y. A Novel Rabbit Dry Eye Model Induced by a Controlled Drying System. Transl. Vis. Sci. Technol. 2021, 10, 32. [Google Scholar] [CrossRef]
- Yeh, S.; Song, X.J.; Farley, W.; Li, D.Q.; Stern, M.E.; Pflugfelder, S.C. Apoptosis of ocular surface cells in experimentally induced dry eye. Investig. Ophthalmol. Vis. Sci. 2003, 44, 124–129. [Google Scholar] [CrossRef] [Green Version]
- De Paiva, C.S.; Chotikavanich, S.; Pangelinan, S.B.; Pitcher, J.D.; Fang, B.; Zheng, X.; Ma, P.; Farley, W.J.; Siemasko, K.F.; Niederkorn, J.Y.; et al. IL-17 disrupts corneal barrier following desiccating stress. Mucosal Immunol. 2009, 2, 243–253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laria, C.; Alio, J.L.; Ruiz-Moreno, J.M. Combined non-steroidal therapy in experimental corneal injury. Ophthalmic Res. 1997, 29, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Eaton, J.S.; Miller, P.E.; Bentley, E.; Thomasy, S.M.; Murphy, C.J. The SPOTS System: An Ocular Scoring System Optimized for Use in Modern Preclinical Drug Development and Toxicology. Off. J. Assoc. Ocul. Pharmacol. Ther. 2017, 33, 718–734. [Google Scholar] [CrossRef]
- Han, J.Y.; Kang, B.; Eom, Y.; Kim, H.M.; Song, J.S. Comparing the Effects of Particulate Matter on the Ocular Surfaces of Normal Eyes and a Dry Eye Rat Model. Cornea 2017, 36, 605–610. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.W.; Choi, J.A.; Shin, E.Y.; La, T.Y.; Jee, D.H.; Chung, Y.W.; Cho, Y.K. Effect of trapping vascular endothelial growth factor-A in a murine model of dry eye with inflammatory neovascularization. Int. J. Ophthalmol. 2016, 9, 1541–1548. [Google Scholar]
- De Paiva, C.S.; Corrales, R.M.; Villarreal, A.L.; Farley, W.J.; Li, D.Q.; Stern, M.E.; Pflugfelder, S.C. Corticosteroid and doxycycline suppress MMP-9 and inflammatory cytokine expression, MAPK activation in the corneal epithelium in experimental dry eye. Exp. Eye Res. 2006, 83, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Wei, D.; Xu, H.; Liu, C. Apigenin ameliorates ocular surface lesions in a rat model of dry eye disease. Eur. J. Inflamm. 2019, 17, 1–6. [Google Scholar] [CrossRef]
- Lin, T.; Lu, Y.; Zhang, X.; Gong, L.; Wei, C. Treatment of dry eye by intracanalicular injection of a thermosensitive chitosan-based hydrogel: Evaluation of biosafety and availability. Biomater. Sci. 2018, 6, 3160–3169. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, J.; Ott, L.; Roth, M.; Witt, J.; Geerling, G.; Mertsch, S.; Schrader, S. MSC Transplantation Improves Lacrimal Gland Regeneration after Surgically Induced Dry Eye Disease in Mice. Sci. Rep. 2019, 9, 18299. [Google Scholar] [CrossRef]
- Shinzawa, M.; Dogru, M.; Miyasaka, K.; Kojima, T.; Tsubota, K. The Application of Strip Meniscometry to the Evaluation of Tear Volume in Mice. Investig. Ophthalmol. Vis. Sci. 2019, 60, 2088–2091. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wei, X.E.; Markoulli, M.; Zhao, Z.; Willcox, M.D. Tear film break-up time in rabbits. Clin. Exp. Optom. 2013, 96, 70–75. [Google Scholar] [CrossRef]
- Chen, F.Y.; Lee, A.; Ge, S.; Nathan, S.; Knox, S.M.; McNamara, N.A. Aire-deficient mice provide a model of corneal and lacrimal gland neuropathy in Sjogren’s syndrome. PLoS ONE 2017, 12, e0184916. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diego, J.L.; Bidikov, L.; Pedler, M.G.; Kennedy, J.B.; Quiroz-Mercado, H.; Gregory, D.G.; Petrash, J.M.; McCourt, E.A. Effect of human milk as a treatment for dry eye syndrome in a mouse model. Mol. Vis. 2016, 22, 1095–1102. [Google Scholar]
- Jiang, D.; Liu, X.; Hu, J. Topical administration of Esculetin as a potential therapy for experimental dry eye syndrome. Eye 2017, 31, 1724–1732. [Google Scholar] [CrossRef]
- Zhao, M.; Liu, L.; Zheng, Y.; Liu, G.; Che, B.; Li, P.; Chen, H.; Dong, C.; Lin, L.; Du, Z. Anti-inflammatory effects of paeoniflorin from Paeonia lactiflora Pall. on human corneal epithelial cells and a mouse model of dry eye disease. RSC Adv. 2019, 9, 12998. [Google Scholar] [CrossRef] [Green Version]
- Yao, X.L.; Peng, Q.H.; Peng, J.; Tan, H.Y.; Wu, Q.L.; Wu, D.L.; Chen, M.; Li, C.K.; Li, D.; Zhu, H.A. Effects of extract of Buddleja officinalis on partial inflammation of lacrimal gland in castrated rabbits with dry eye. Int. J. Ophthalmol. 2010, 3, 114–119. [Google Scholar] [PubMed]
- Hamano, H.; Hoary, M.; Mitsunaga, S.; Kojima, S.; Maeshima, J. Tear test (preliminary report). J. Jpn. CL Soc. 1982, 24, 103–107. [Google Scholar]
- Saleh, T.A.; McDermott, B.; Bates, A.K.; Ewings, P. Phenol red thread test vs Schirmer’s test: A comparative study. Eye 2006, 20, 913–915. [Google Scholar] [CrossRef]
- Menzies, K.L.; Srinivasan, S.; Prokopich, C.L.; Jones, L. Infrared imaging of meibomian glands and evaluation of the lipid layer in Sjogren’s syndrome patients and nondry eye controls. Investig. Ophthalmol. Vis. Sci. 2015, 56, 836–841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Begley, C.; Caffery, B.; Chalmers, R.; Situ, P.; Simpson, T.; Nelson, J.D. Review and analysis of grading scales for ocular surface staining. Ocul. Surf. 2019, 17, 208–220. [Google Scholar] [CrossRef]
- Bron, A.J.; Evans, V.E.; Smith, J.A. Grading of corneal and conjunctival staining in the context of other dry eye tests. Cornea 2003, 22, 64. [Google Scholar] [CrossRef] [PubMed]
| Animal | Thickness | References |
|---|---|---|
| Mice | 7 μm | Tran et al. [32] 2003, Johnson et al. [2] 2004 |
| Gerbils | 15 μm | Prydal et al. [33] 1993 |
| 14 μm (interferometry) 16 μm (confocal microscopy) | Prydal et al. [34] 1992 | |
| Rat | 2–6 μm | Chen et al. [31] 1997, Johnson et al. [2] 1992 |
| 11 μm (interferometry) 11 μm (confocal microscopy) | Prydal et al. [34] 1992 | |
| 7 μm (glass fiber) 13 μm (confocal microscopy) | Prydal et al. [33] 1993 | |
| Rabbit | 4–7 μm | Mishima [35] 1965, Prydal et al. [34] 1992 |
| 12 μm (interferometry) 10 μm (confocal microscopy) | Prydal et al. [34] 1992 | |
| 7 μm (glass fiber) 10 μm (confocal microscopy) | Prydal et al. [33] 1993 |
| Name of Cells Line | Culture | Induction Methods | Starting of Treatment | End of Experiment | References |
|---|---|---|---|---|---|
| Wong Kilbourne derivative of Chang conjunctival epithelial cell line (WKD; clone 1–5c-4) | Dulbecco’s minimum essential medium supplemented with 10% fetal bovine serum, 1% glutamine, 50 UI/mL penicillin, and 50 UI/mL streptomycin | Cells were grown for 24 h. Then, benzalkonium chloride was dissolved in phosphate-buffered saline (PBS). Different concentrations of BAC (10−2%, 10−3%, were analyzed. | – | 15 min of treatment or 15 min of treatment followed by 24 h of cell recovery in complete medium | Brasnu et al. [47] 2008 |
| Dulbecco minimum essential medium supplemented with 10% fetal bovine serum (FBS), 1% glutamine (200 mM stock solution), and 1% penicillin (10,000 units/mL) and streptomycin (10,000 μg/mL) | Cells were grown for 24 h using hyperosmotic media 500 mOsM, achieved by adding 90 mM sodium chloride or media containing benzalkonium chloride at 10–4%, 3.10–4%, or 5.10–4%. | – | Cell was analyzed at 24 h and 48 h | Clouzeau et al. [48] 2012 | |
| Dulbecco’s minimum essential medium (DMEM) supplemented with 10% fetal bovine serum, 1% glutamine, 0.1% ampicillin, and 2% kanamycin | A 15 min BAC 0.001% treatment and after 15 min culture media was removed, and normal culture medium was added and allowed for 24 h | Candidate drug was mixed 1 h before BAC treatment | After 24 h | Debbasch et al. [49] 2001, Debbasch et al. [50] 2001 | |
| Eagle’s minimal essential medium supplemented with 5% fetal calf serum, 2 mM L-glutamine, 50 mg/mL streptomycin, and 50 IU/mL penicillin | 0.0001% (1 μg/mL). Cells were treated for 10 min. After this time, the BAC-containing medium was removed, cells were rinsed twice with culture medium, and normal cell culture conditions were restored. | Examined before treatment and 3, 24, 48, and 72 h later | De Saint Jean et al. [51] 1999 | ||
| IOBA-NHC cells | DMEM/F12 supplemented with 1 μg/mL bovine pancreas insulin, 2 ng/mL mouse epidermal growth factor, 0.1 μg/mL cholera toxin, 5 μg/mL hydrocortisone, 10% fetal bovine serum (FBS), 50 UI/mL penicillin, and 50 UI/mL streptomycin | Cells were grown for 24 h. Benzalkonium chloride was dissolved in PBS. Different concentrations of BAC (10–2%, 10–3%, were analyzed. | – | Two incubation times were applied to the cells: 15 min of treatment and 15 min of treatment followed by 24 h of cell recovery in complete medium | Diebold et al. [52] 2003; Brasnu et al. [47] 2008 |
| Dulbecco’s Modified Eagle Medium (DMEM)/HAM’s F12 (1:1) supplemented with 10% fetal calf serum (FCS, Biochrom AG, Berlin, Germany) in a humidified incubator containing 5% CO2 at 37 °C | For stimulation, cells (1 × 106) were seeded in Petri dishes and cultured until confluence was reached. Cells were washed PBS and changed to serum-free medium for 3 h. Afterward, cells were either treated with recombinant proinflammatory cytokine interleukin (IL)-1β (10 ng/mL) or tumor necrosis factor (TNF) α (10 ng/mL) for 6 h, 12 h, 24 h, or 48 h | – | 6 h, 12 h, 24 h, or 48 h each | Schiicht et al. [53] 2018 | |
| Human corneal epithelial cells (HCECs), a human transformed SV40 immortalized corneal epithelial cell line | Were cultured in Dulbecco’s modified Eagle’s medium/F12 with 10% fetal bovine serum and 10 ng/mL human epidermal growth factor and the medium replaced every other day. | Cells were grown for 24 h. Then, they were treated with a different osmolarity, ranging from 312 to 550 mOsm/L, which was achieved by adding 0, 70, 90, or 120 mM sodium chloride (NaCl) with or without candidate drugs. | Candidate drugs were added 2 h before adding NaCl. | Samples were after 24 h treatment | Li et al. [54] 2020 |
| Were cultured in Dulbecco’s modified Eagle medium (DMEM)/HAM’s F12 supplemented with 10% fetal bovine serum, 50 U/mL penicillin, 50 μg/mL streptomycin mixture, 1% insulin-transferrin–selenium mixture, and 10 ng/mL human epidermal growth factor | Hyperosmotic stress (500 mOsm) was achieved by adding 90 mM sodium chloride (NaCl, Sigma-Aldrich) to isosmotic medium (310 mOsm). | Candidate drugs were added 2 h before adding NaCl. | Supernatants of conditioned medium were collected at 24 h after stimulation | Ma et al. [55] 2021 | |
| Human conjunctival cell line HCC | Cells were cultured according to the manufacturer’s instruction in RPMI medium supplemented with 100 IU/mL penicillin, 100 mg/mL streptomycin, and 10% heat-inactivated FBS | Hyperosmotic media (528 mOsM) | Candidate drugs were added 2 h before adding NaCl. | 24 h after treatment | Park et al. [56] 2019 |
| Primary Culture | |||||
| Primary HCECs (human corneal epithelial cells) were cultured from donors within 72 h after death | Supplemented hormonal epidermal medium (SHEM) containing 5% FBS | The addition of 44, 69, and 94 mM of sodium chloride (NaCl) can achieve hyperosmolarity (400, 450, and 500 mOsM) from the isosmolar (312 mOsM) medium. | - | The HCECs co-incubated for 12 h, 24 h, or 48 h were used for immunostaining | Liu et al. [57] 2020 |
| Primary culture of rabbit corneal epithelial cells (CECs) or Primary rabbit LG acinar cells (LGACs) | After isolation, cultured with DMEM/F12 (Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12) with 1% antibiotic–antimycotic solution | After 24 h culture, DED-like symptom was induced by the addition of IL-1β (10 ng/mL) with the medium. | Dexamethasone (10 μM) was used combined with IL-1β for treatment | Culture was maintained for 1 week and analyzed | Lu et al. [58] 2017 |
| Animal | Dose | Frequency | Days | Starting of Treatment | End of Experiment | References |
|---|---|---|---|---|---|---|
| Rabbits | 0.01%, 0.05%, or 0.1% was applied | Twice daily to one eye | For 4 days | Chen et al. [44] 2011 | ||
| Rabbits | 0.1% BAC drops | Twice daily | For on days 5, 7, and 14 | On days 5, 7, and 14 | Xiong et al. [67] 2008 | |
| Rabbits | 0.1% BAC | Twice daily | For 14 days | After 14 weeks of BAC treatment | 3 days after treatment | Lu et al. [79] 2017 |
| Rabbits | 0.1% benzalkonium chloride (BAC) drops | Twice daily topical administration | For 3 weeks | Ji et al. [80] 2017 | ||
| Rabbits | 0.1% BAC (20 μL) | Thrice daily (10:00 a.m., 2:00 p.m., and 6:00 p.m.) | For 4 weeks | After 4 weeks of BAC treatment twice daily (10:00 a.m. and 6:00 p.m.) for 3 weeks | After 3 weeks of treatment | Tseng et al. [81] 2016 |
| Rabbits | 0.1% (wt/v) BAC (20 µL) | Thrice daily | For 4 weeks | After 4 weeks of BAC treatment | After 3 weeks of treatment | Chen et al. [82] 2017 |
| Rabbits | 0.1% BAC drops | Twice daily | For 2, 3, 4, or 5 weeks | Li et al. [66] 2012 | ||
| Mouse | 5 μL of 0.2% BAC | Twice daily (9:00 a.m., 9:00 p.m.) | For 7 days | On day 7 | Lin et al. [83] 2011 | |
| Mice | 5 μL of 0.1% BAC | Twice daily (9:00 a.m., 9:00 p.m.) | For 10 days | 5 μL, three times per day (9:00 a.m., 3:00 p.m., 9:00 p.m.) | On day 6 of treatment | Xiao et al. [84] 2012 |
| Mice | 5 μL of 0.2% BAC | Twice daily (10:00 a.m. and 10:00 p.m.) | For 14 days | After 14 days BAC instillation | On the 14th days | Kim et al. [68] 2016 |
| Mice | 5 μL of 0.2% BAC | Twice daily (9:00 a.m., 9:00 p.m.) | For 14 days | After 14 days BAC treatment 5 μL, three times per day (8:00 a.m., 3:00 p.m., 10:00 p.m.) | On 6 days after treatment | Xiao et al. [85] 2012 |
| Wister Rat | 0.2% BAC | Twice a day | For 7 days | After 7 days of BAC treatment once daily for 1 week | After 7 days of treatment | Beyazyildiz et al. [86] 2014 |
| SD rat | 5 μL 0.2% BAC | Twice daily (at 7:00 a.m. and 7:00 p.m.) | For 7 days | – | On 7th day | Marques et al. [87] 2014 |
| SD rat | 0.2% BAC | Daily, at 9:30:00 a.m. and 5:30:00 p.m. | For 10 days | Immediately after BAC treatment | After 10 days | Na et al. [88] 2017 |
| Other Chemicals | ||||||
| Atropine Sulfate | ||||||
| Animal | Dose | Frequency | Days | Starting of Treatment | End of Experiment | References |
| New Zealand adult female rabbits | One drop of 1% atropine | Three times daily 6:00 a.m., 2:00 p.m., and 10:00 p.m. | For 7 days | Oral administration of oils occurred at 8:00 a.m. from the first day with atropine sulfate to the 12 weeks | 12 weeks | Silva et al. [74] 2017 |
| White albino Rabbits | 1% atropine sulfate | Three times daily | For 1 week | After 7 days atropine sulfate treatment for 7 days | On 7th day | Shafaa et al. [73] 2017 |
| Rabbits | 1% atropine sulfate | Three times daily | For 17 days | three times daily for seven days starting on day 10 | On 17 days | El-Shazly et al. [89] 2008 |
| Rabbits | 50 μL of 1.0% atropine sulfate | Three times daily at 8:00 a.m., 1:00 p.m., and 6:00 p.m. | For 5 days. | – | – | Burgalassi et al. [72] 1999 |
| New Zealand white rabbits | 1 mg/kg atropine sulfate (1.0%). | Injected intramuscularly every day | For 3 days | – | After 3 days | Altinors et al. [90] 2007 |
| Finasteride | ||||||
| Wistar rats | Finasteride (1.16 mg/kg/d) was orally administered to all the rats | Once a daily | For 4 weeks | Same time once a day | 4 weeks | Zhang et al. [71] 2016 |
| Wistar rats | Finasteride (1.16 mg/kg/day) was orally administered to the rats | Once a day | For 4 weeks | – | – | Li et al. [91] 2018 |
| Wistar female and male rats | Finasteride (1.16 mg/kg) was orally administered to all the female and male rats | Once a day | For 10 days | – | At the end of 10 days | Singh et al. [70] 2014 |
| N-Acetylcysteine (NAC) | ||||||
| SD rats | 20 μL of 10% (wt/vol) NAC by topically | Four times (10:00 a.m., 12:00 p.m., 2:00 p.m., 4:00 p.m.) a day into the right eye of each rat | For 5 days | Li et al. [92] 2018 | ||
| rabbit | a 10% (wt/vol) NAC solutions | instilled 6 times at 2 h intervals for 1 day (9:00 a.m. to 7:00 p.m.) into the eyes | For 1 day | Next day after NAC installation | 2, 4, or 6 times a day for 3, 7, or 14 days | Urashima et al. [78] 2004 |
| Scopolamine | ||||||
| C57BL/6 mice | Subcutaneous injections of 0.1 mL of 5 mg/mL scopolamine hydrobromide | Three times daily | For 5 days | After 5 days | Xiao et al. [93] 2015 | |
| Female Lewis rats | Scopolamine was continuously and systemically delivered to the animals via an osmotic pump filled with scopolamine and implanted subcutaneously. In the first experiment, three doses of Scopolamine was delivered for 28 days, 12.5 mg/day. | At 28 days | Viau et al. [94] 2008 | |||
| Trichloroacetic Acid | ||||||
| New Zealand white rabbits | A cotton swab soaked with freshly prepared 50% trichloroacetic acid was applied to the conjunctivas of the left eyes 2–3 mm lateral to the corneal limbus | For 5 s (when blanching of the conjunctiva was observed). The conjunctival sacs were immediately washed with 100 mL 0.9% sterile saline | Li et al. [45] 2013 | |||
| Animal | Name of Parts | Starting of Treatment | End of Experiment | References |
|---|---|---|---|---|
| SD rats | The left exorbital lacrimal gland was surgically excised | At three days after surgery, orally administered for 7 days | After 7 days of treatment | Kang et al. [106] |
| C57BL/6 mice | Surgical excision of the left exorbital lacrimal gland | At three days after surgery 20 µL twice daily for 5 days | 5 days after treatment | Kim et al. [107] 2016 |
| Male Wistar rats | Surgical excision of the left exorbital lacrimal gland | At three days after surgery, 20 µL twice daily for 4 days | 7 days after operation | Park et al. [108] 2018 |
| C57BL/6 mice | Dry eye based on severe aqueous fluid deficiency, by excising both the exorbital and intraorbital lacrimal glands of mice. | – | 8 weeks | Shinomiya et al. [14] 2018 |
| Squirrel Monkey | Unilateral removal of main lacrimal gland. | 20 weeks | Maitchouk et al. [95] 2000 | |
| BALB/c mice | After anesthesia, 10 or 20 μL concanavalin A (ConA) that was diluted in phosphate-buffered solution (PBS) at concentrations of 10 mg/mL was injected into the intraorbital gland through a transconjunctival approach using a Hamilton syringe with a 33-gauge needle under an operating microscope | Immediately after ConA injection, hMSCs (1 × 103 or 1 × 105 cells/20 μL BSS), mMSCs (1 × 105 cells/20 μL BSS), or the same volume of BSS were injected into the periorbital space using a 30-gauge needle syringe | After 7 days | Lee et al. [101] 2015 |
| BALB/c mice | 20 µL ConA that was diluted in PBS at the concentration of 10 mg/mL was injected into both intraorbital and extraorbital lacrimal glands of BALB/c mice using a Hamilton syringe with a 33-gauge needle | Recombinant human (rh) TSG-6 (1 µg/10 µL was topically instilled four times a day (QID) to the ocular surface of the mice for 7 days | After 7 days of treatment | Lee et al. [102] 2015 |
| Balb/c mice | Dry eye disease was induced using 10 mg/mL of ConA (20 µL) in PBS, which was injected into the lacrimal glands with a 28.5 gauge needle using a dissecting microscope | Individually combined together and injected with ConA | After 7 days of treatment | Ratay et al. [100] 2017 |
| NZW rabbit | After anesthesia, rabbits were injected with 500 μg of Con A in 50 μL of saline in the lacrimal glands bilaterally using a 26-gauge needle | 24 h after Con A injection, ophthalmic solution four times a day for 6 days | 48 h after last Con A injection | Seo et al. [103] 2010 |
| NZW rabbit | A single 30 μL volume 300 μg Con A was injected into the lacrimal gland using a 30-gauge needle and a Hamilton syringe | Injected combined with ConA | 7 days after treatment | Zheng et al. [104] 2015 |
| New Zealand albino rabbits | Lacrimal glands and Meibomian glands. The Harderian | After 10 weeks | Polans et al. [96] 2017 | |
| Japanese albino rabbits | The lacrimal and harderian glands and nictitating membrane were removed surgically | 4 months after surgery | Chen et al. [44] 2011 | |
| New Zealand white rabbits weighing | The lacrimal gland, Harderian gland, and nictitating membrane of the left eyes were surgically removed | On day 56 | Li et al. [45] 2013 | |
| New Zealand white rabbits | Nictitating membrane (NM), Harderian gland (HG), and main LG | 4 months after excision | Bhattacharya et al. [43] 2015 | |
| New Zealand white rabbits | Resection of main LG, HG, and NM | Ning et al. [42] 2016 | ||
| CBA/J mice | Injection of 0.05 mL of 20-mU BTX-B solutions into the left lacrimal gland | 3 days after BTX injection | 4 weeks after BTX injection | Lekhanont et al. [105] 2007 |
| Animal | Humidity | Airflow | Temperature | Time of Induction | Starting of Treatment | End of Experiment | References |
|---|---|---|---|---|---|---|---|
| C57BL/6 mice | 13.1% ± 3.5% | 2.2 ± 0.2 m/s | 22 ± 2 °C | 21 days | Beginning of housing in the ICES or on day 22 after DED confirmation. 10 µL topically, four times daily until 35 days. | 35 days | Chen et al. [115] 2013 |
| BALB/c mice | 13.1% ± 3.5% | 2.2 ± 0.2 m/s | 22 ± 2 °C | 21 days | After 21 days housed in the ICES, the mice were administered 10 μ of eye drops, four times daily (every 6 h) for 14 days during which the mice remained housed in the ICES. | 35 days | Chen et al. [116] 2009 |
| C57BL/6 mice | 13.1% ± 3.5% | 2.2 ± 0.2 m/s | 22 ± 2 °C | 21 days | 10 μL/eye bilaterally four times a day for 3 weeks. | After 3 weeks | Li et al. [117] 2012 |
| C57BL/6 mice | 15.3% ± 3.0% | 2.1 ± 0.2 m/s | 21 °C ± 2 °C | 14 days | Beginning of housing in the ICES or on day 14 after DED confirmation. 10 µL topically, 4 times daily until 28 days | 28 days | She et al. [109] 2015 |
| BALB/c mice | 15.3% ± 3% | 2.1 ± 0.2 m/s | 21 ± 3 °C | 42 days | – | 42 days | Chen et al. [110] 2008 |
| C57BL/6 mice | 13.1 ± 3.5%, airflow and temperature | 2.2 ± 0.2 m/s | 22 ± 2 °C | An alternating 12 h light–dark cycle (8:00 a.m. to 8:00 p.m.) was employed for 1, 2, 4, and 6 weeks | 1, 2, 4, and 6 weeks | Xiao et al. [93] 2015 | |
| Mice | Topical application of:00 PM2.5. 5.0 mg/mL, 4 times daily | 4, 7, 10, and 14 days | Tan et al. [113] 2018 | ||||
| Mice | Topical application of PM10 5.0 mg/mL to right eyes, 4 times daily. | At 14 days | Li et al. [114] 2017 | ||||
| Rats | Rats were exposed to approximately 500 μg/m3 UPM in the exposure chamber for 5 h per day over 5 days. | 5 days after exposure of UPM | Song et al. [112] 2020 |
| Animal | Time of Induction | Detected Symptoms Related to DED | References |
|---|---|---|---|
| TGF-β1 knockout mouse | 2 and 4 weeks of age | Significant inflammatory cell infiltrates in the lacrimal gland between the ages of 2 and 4 weeks | McCartney Francis et al. [123] 1997 |
| NOD. Aire KO mice | 6 weeks of age | Severe corneal pathology observed | Vijmasi et al. [124] 2013 |
| NRTN−/− mice | 6 weeks of age | Tear volume and mucin production are decreased, and ocular surface inflammation are increased | Cha et al. [121] 2002 |
| C57BL/6.NOD-Aec1R1Aec2, | 19–22 weeks of age | Male mice displayed a high level of dacryoadenitis | Nguyen et al. [125] 2006 |
| NOD.B10.H2b | 12 weeks of age | Severe corneal pathology observed | Lee et al. [75] 2017, Kim et al. [76] 2015, Lee et al. [102] 2015 |
| TSP-1−/− mice | 6–12 weeks of age | 6–8 weeks, changes of LG epithelial cells and their functional loss are observed At 12 weeks, the loss of corneal surface integrity and corneal nerve morphology, conjunctival infiltrates, and loss of conjunctival goblet cells | Masli et al. [120] 2020 |
| IL-2Rα (CD25) knockout mice | 8 weeks of age | CD4+ cells are detected in the conjunctiva beginning at 8 weeks of age and disrupted only from 12 weeks onwards. | Masli et al. [120] 2020 |
| NFS/sld mice | 8 weeks of age | At 8 weeks, inflammatory lesions of lacrimal glands and Harderian glands, loss of ocular surface integrity, and conjunctival goblet cells observed. | Arakaki et al. [126] 2014 |
| IQI/Jic mouse | 9 months of age | After 21 days housed in the ICES, the mice were administered 10 μ of eye drops, four times daily (every 6 h) for 14 days, during which the mice remained housed in the ICES. | Chen et al. [127] 2009 |
| Animal | Name of Chemical/Surgery | Environmental Condition/Other | Starting of Treatment | End of Experiment | References |
|---|---|---|---|---|---|
| C57BL/6 mice | Scopolamine hydrobromide (0.5 mg/0.2 mL) was injected subcutaneously in the dorsal skin of mice three times per day. | Exposed to a relative humidity < 25%, temperature of 20–22 °C, and airflow of 15 L/min, 24 h per day | - | On day 7 or 9 | Lee et al. [75] 2017, Lee et al. [128] 2012 |
| C57BL/6 mice | In brief, 0.5 mg/0.2 mL scopolamine hydrobromide was injected subcutaneously in the dorsal skin of mice three times daily. | The mice placed in the CEC were continuously exposed to a relative humidity < 30%, a constant temperature of 21–23 °C, and airflow of 15 L/min, 24 h a day | From day 3 to day 9, 2 µL of the topical agent was applied to both eyes of each mouse, twice a day (9:00 a.m. and 5:00 p.m.). | On day 9 | Lee et al. [129] 2011 |
| C57BL/6 mice | Topical application of atropine sulfate, 1%, twice daily for the first 48 h. In addition, the mice also received subcutaneous 0.1-mL injections of 5 mg/mL scopolamine hydrobromide three times a day (9:00 a.m., 1:00 p.m., and 5:00 p.m.) on their dorsal surface for the duration of the experiment. | Regulation of relative humidity <30%, <30%, a constant temperature of 21–23 °C, and airflow of 15 L/min, 24 h a day | 48 h after the induction of dry eye, 3 µL of the topical formulatio nwas applied to both the eyes of the unanesthetized mice, twice a day (9:00 a.m. and 5:00 p.m.) from days 3 to 9. | 9 days of treatment | Goyal et al. [130] 2009 |
| C57BL/6 mice | Scopolamine was injected into the dorsal skin of mice (0.5 mg/0.2 mL at 9:00 a.m., 12:00 p.m., and 3:00 p.m.; 0.75 mg/0.3 mL at 6:00 p.m.). | Mice were placed in the controlled environmental chamber (relative humidity <30%, airflow 15 L/min, temperature 21–23 °C) | 1 µL of eye drop was applied topically to the eye of an unanesthetized mouse once daily from 48 h to day 4 (total three doses) or day 9 (total eight doses). | On day 5 or 10 after treatment | Rashid et al. [131] 2008 |
| C57BL/6 mice, and BALB/c | DS was induced by subcutaneous injection of scopolamine hydrobromide (0.5 mg/0.2 mL; four times a day (at 08:00 a.m., 12:00 p.m., 2:00 p.m., and 5:00 p.m.), alternating between the left and right flanks of 4–6-week-old mice | Mice were placed in a cage with a perforated plastic screen on one side to allow airflow from a fan (Cafrano) placed 6 inches in front of it for 16 h/day for 5 or 12 consecutive days; chamber (relative humidity 30–35%, airflow 15 L/min, temperature 80 °F) | – | On day 12 | Niederkom et al. [132] 2006 |
| 129SvEv/CD-1 white mice | Subcutaneous injection of scopolamine (1 mg in 0.2 mL) three times daily in the flanks of 4–6-week-old 129SvEv/CD-1 white mice. Dry eye was induced in mice with a modification of a previously described technique. | Mice were exposed to a continuous air draft from a fan placed 15.24 cm in front of the cage in an environmentally controlled room (50% humidity, 18 °C) for 10 h a day for 12 consecutive days | – | On day 12 | Yeh et al. [134] 2003 |
| C57BL/6 and IFN- γ knockout mice | Subcutaneous injection of scopolamine hydrobromide (0.5 mg per 0.2 mL four times a day (8:00 a.m. 12:00 p.m., 2:00 p.m., and 5:00 p.m.), alternating flanks of mice | Humidity was maintained at 30–35% 16 h per day. DS was induced for either 5 or 10 consecutive days | De Paiva et al. [135] 2009 | ||
| Male NOD.B10.H2b mice | 0.5 mg/0.2 mL hypodermic injection of scopolamine hydrobromide into both hindquarters (one after the other) four times (9:00 a.m., 12:00 p.m., 3:00 p.m., and 6:00 p.m.) per day for 10 days | Desiccation stress was created by low ambient humidity (30–40%) using an air draft from a fan for 18 h per day | After the desiccation stress for 10 days, the scopolamine hydrobromide injections were discontinued, and the mice were placed in an environment of normal humidity and temperature. The eye drops, PBS, and 1 mg/mL or 5 mg/mL silk fibroin were administered five times (9:00 a.m., 11:00 a.m., 1:00 p.m., 3:00 p.m., and 5:00 p.m.) per day for 10 days | After 10 days of treatment | Kim et al. [122] 2017 |
| New Zealand rabbits | Surgical dissection of nictitating membrane | After 1 week of surgery, animal were housed in controlled environment humidity, 22% ± 4%, air flow 3 to 4 m/s, and temperature 23 to 25 °C | – | After 3, 7, and 14 days | Chen et al. [133] 2021 |
| Types of Tests | Items | Animal | Methods | References |
|---|---|---|---|---|
| Clinical sings | Rabbit | Conjunctivitis (0–3), ocular discharge (0–3), corneal opacity (0–3) | Baudouin et al. [l] 2013, Silva et al. [74] 2017 | |
| Modified severity score from Eaton J.S. et al. [137]; Silva D.A. et al. [74] | The scored varies from 0 to 16 and was defined as the sum of the individual scores graded from conjunctival hyperemia (0–3), conjunctival swelling (chemosis) (0–4), conjunctival discharge (0–3), corneal opacity (area) (0–4), and corneal vascularization (0–2). | Silva et al. [74] 2017, Eaton et al. [137] | ||
| Inflammatory index/scoring and markers | Macroscopic (visually) inflammatory scoring/index | Mice | Total score of 0–9; ciliary hyperemia (0–3); central corneal edema (0–3); peripheral corneal edema (0–3) | Lin et al. [83] 2011, Xiao et al. [84] 2013, De Paiva et al. [135] 2009, Laria et al. [136] 1997 |
| Microscopically evaluated by infiltration of inflammatory cells | Rats | Evaluated by counting PMN cells in the cornea and limbus by HE stains | Han et al. [138] 2017 | |
| Inflammatory cytokines | Mice, Rat | TNF-α, IL-6, TNF- α, IL-1 a, IL-1β, and MMP-9 in corneas, CD11b+ (IHC) | Xiao et al. [84] 2012, Han et al. [138] 2017, Kwon et al. [139] 2016, De Paiva et al. [140] 2006 | |
| Rabbit | TNF-α, IL-1 β, IL-6, IL-8 in corneas | Tseng et al. [81] 2016 | ||
| Rabbit | IL-1β, TNF-a, and MMP-9 in conjunctiva epithelium | Bhattacharya et al. [43] 2015 | ||
| Test for detection of tear abnormality | Tear film osmolarity | Rats | Using Tearlab Osmolarity System® osmometer. Osmolarity increase in DED. | Marques et al. [87] 2014 |
| Schirmer’s I Test | Rabbit | Tear volume (mm) | Luo et al. [79] 2017, Tseng et al. [81] 2016 | |
| Rats | Liu et al. [141] 2019 | |||
| Phenol red thread tests | Rabbit | Lin et al. [142] 2018 | ||
| Rat | Han et al. [138] 2017 | |||
| Mice | Dietrich et al. [143] 2019 | |||
| Tear breakup time (BUT) (Specific for tear film instability) | Mice, rats, rabbit | Usually, TBUT is ≤10 s for in DED | Liu et al. [141] 2019, Shinzawa et al. [144] 2019, Wei et al. [145] 2013 | |
| Surface structural damage | Corneal Fluorescein Staining Test | Rabbit | Luo et al. [79] 2017, Tseng et al. [81] 2016 | |
| Rat | Liu et al. [141] 2019 | |||
| Mice | Oxford grading | Dietrich et al. [143] 2019 | ||
| Rose Bengal Staining Test | Rabbit | Luo et al. [79] 2017, Na et al. [88] 2017 | ||
| Rat | Beyazyildiz et al. [86] 2014 | |||
| Mice | Lin et al. [83] 2011 | |||
| Lisamine green staining | Rabbit | Chen et al. [133] 2021 | ||
| Rat | Song et al. [112] 2020 | |||
| Mice | Chen et al. [146] 2017 | |||
| Changes of epithelial layers of cornea or conjunctiva | Corneal epithelial thickness | Rabbit | Luo et al. [79] 2017, Tseng et al. [81] 2016, Luo et al. [79] 2017, Tseng et al. [81] 2016 | |
| Mice | Xiao et al. [84] 2013, Na et al. [88], Diego et al. [147] 2016 | |||
| Rat | Marques et al. [87] 2014 | |||
| Goblet cells count | Mice, Rabbit, Rats | Counting the goblet cells number by staining MUC5AC in the conjunctiva | Luo et al. [79] 2017, Lin et al. [83] 2011, Han et al. [138] 2017 | |
| Rabbit | Conjunctival impression cytology (CIC) by hematoxylin and periodic acid-Schiff (PAS) reagent | Luo et al. [79] 2017, Jiang et al. [148] 2017 | ||
| Rabbit, mice | Counting goblet cells in conjunctiva by periodic acid-Schiff (PAS) staining | Chen et al. [44] 2011, Xiao et al. [84] 2013 | ||
| Molecular analysis | MAP kinase pathways | Rabbit | p-ERK1/2 protein expression | Jiang et al. [148] 2017 |
| Mice | phospho-JNK/total JNK, phospho-ERK/total ERK, phospho-p38 (p-p38)/total p38 (p-38) in the corneal epithelia | De Paiva et al. [140] 2006 | ||
| NFkB | Mice, HCE cells | Western blot analysis of NFkB | Tan et al. [113] 2018, Zhao et al. [149] 2019 | |
| Apoptosis | Rabbit, mice, rat | TUNEL assay (apoptotic cells count) | Tseng et al. [81] 2016, Luo et al. [79] 2017, Xiao et al. [84] 2013, Han et al. [138] 2017 | |
| Mice | Bax, BCL2, Bax/BCL2 | Na et al. [88] 2017 | ||
| Vascular dysfunction | Mice | VEGF-A in corneas; cell vascular endothelial cells on corneal flat mounts | Kwon et al. [139] 2016 | |
| Rabbit | endothelial cell damage with dislocation of ZO-1, and disruption of PAMR | Liang et al. [63] 2008 | ||
| Fibrosis | Rabbit | TGF-β1 | Yao et al. [150] 2010 |
| Image Panel | Verbal Description | Dot Count | Grade and Criteria |
|---|---|---|---|
 | Absent | 0 or 1 | 0 (=Panel A or < Panel B) |
 | Minimal | 10 | 1 (≤Panel B or < Panel C) |
 | Mild | 32 | 2 (≤Panel C or < Panel D) |
 | Moderate | 100 | 3 (≤Panel D or < Panel E) |
 | Marked | 316 | 4 (≤Panel E or < Panel F) |
 | Severe | >316 | 5 (< Panel E) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahman, M.M.; Kim, D.H.; Park, C.-K.; Kim, Y.H. Experimental Models, Induction Protocols, and Measured Parameters in Dry Eye Disease: Focusing on Practical Implications for Experimental Research. Int. J. Mol. Sci. 2021, 22, 12102. https://doi.org/10.3390/ijms222212102
Rahman MM, Kim DH, Park C-K, Kim YH. Experimental Models, Induction Protocols, and Measured Parameters in Dry Eye Disease: Focusing on Practical Implications for Experimental Research. International Journal of Molecular Sciences. 2021; 22(22):12102. https://doi.org/10.3390/ijms222212102
Chicago/Turabian StyleRahman, Md. Mahbubur, Dong Hyun Kim, Chul-Kyu Park, and Yong Ho Kim. 2021. "Experimental Models, Induction Protocols, and Measured Parameters in Dry Eye Disease: Focusing on Practical Implications for Experimental Research" International Journal of Molecular Sciences 22, no. 22: 12102. https://doi.org/10.3390/ijms222212102
APA StyleRahman, M. M., Kim, D. H., Park, C.-K., & Kim, Y. H. (2021). Experimental Models, Induction Protocols, and Measured Parameters in Dry Eye Disease: Focusing on Practical Implications for Experimental Research. International Journal of Molecular Sciences, 22(22), 12102. https://doi.org/10.3390/ijms222212102







