Why Have the Benefits of DHA Not Been Borne Out in the Treatment and Prevention of Alzheimer’s Disease? A Narrative Review Focused on DHA Metabolism and Adipose Tissue
Abstract
1. Introduction
2. DHA Benefits Alzheimer’s Disease Neuropathological Mechanisms
2.1. Amyloid, Tau, Neuroinflammation and Cell Signaling
2.2. DHA Improves Vascular Risk Factors for Dementia
3. DHA Status of Peripheral Biomarkers Correlates with AD Risk and Progression
Supplemental DHA Does Not Effectively Prevent or Treat AD
4. The Metabolic Fates of DHA Govern Uptake into Specific Tissues
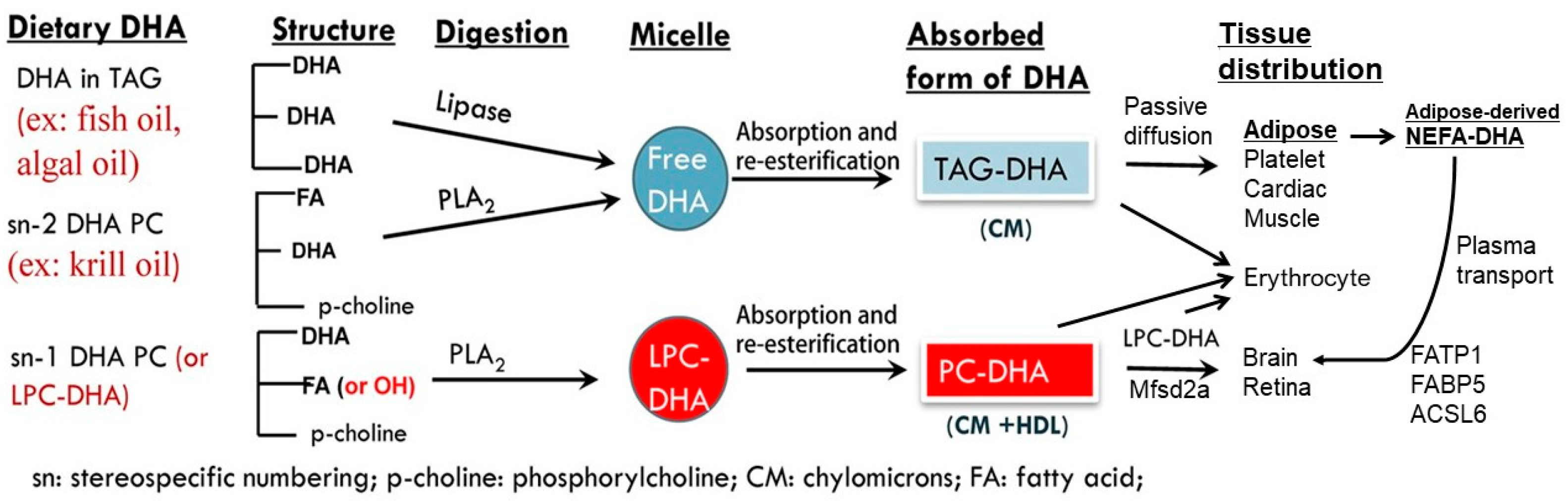
4.1. Digestion and Metabolism of Dietary DHA
4.2. Metabolic Fates of TG- and PL-DHA
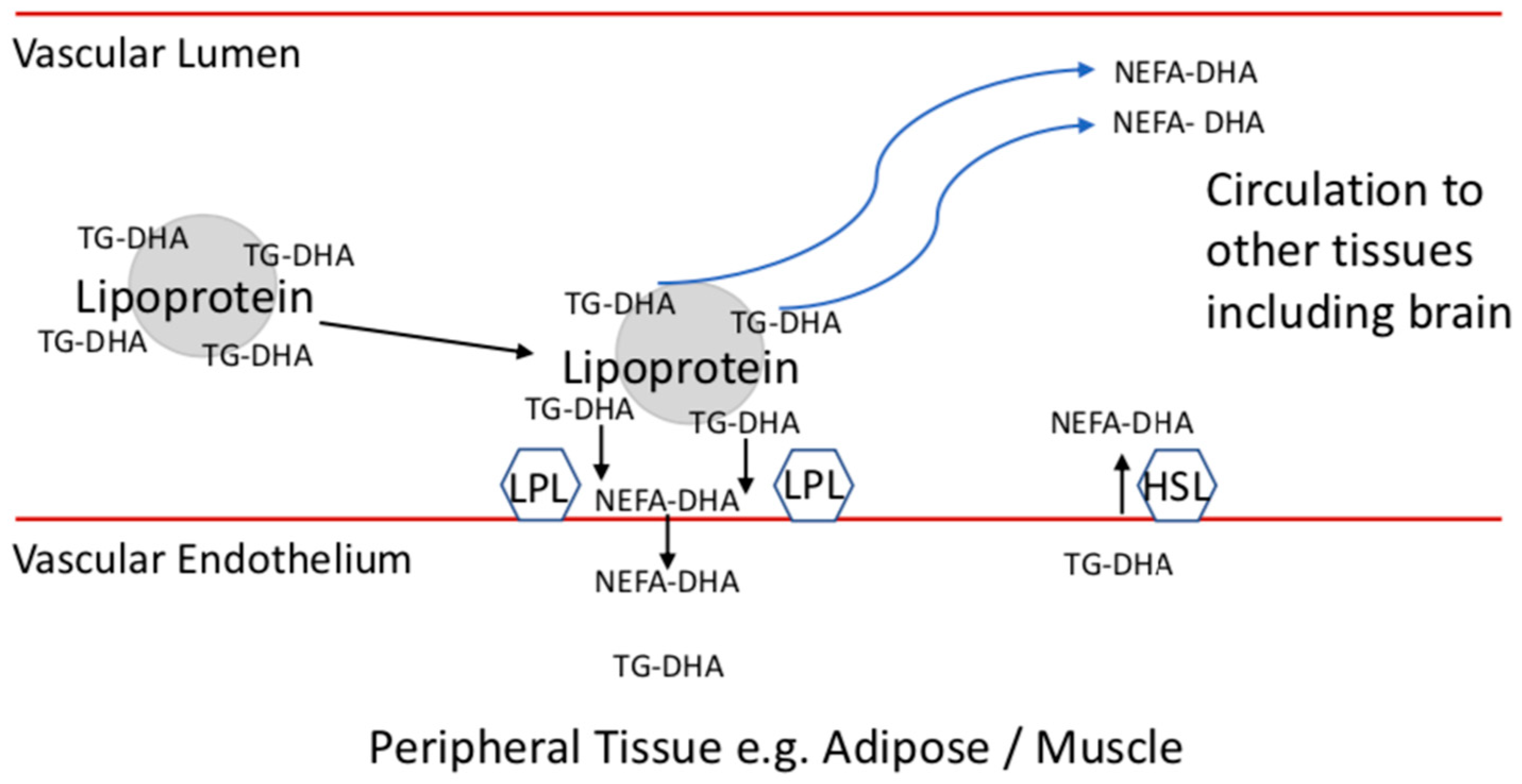
5. Plasma DHA Forms Supply Specific Tissues including Brain
6. Adipose Stores and Supplies NEFA-DHA to Tissues including Brain
7. Adipose Tissue Is an Unknown Entity in Alzheimer’s Disease
8. AD Risk Factors Alter DHA Homeostasis
8.1. Age and Metabolic Disease
8.2. APOE4 and the BBB
9. Is There an Optimal Level of Brain DHA?
Where to from Here?
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sperling, R.A.; Aisen, P.S.; Beckett, L.A.; Bennett, D.A.; Craft, S.; Fagan, A.M.; Iwatsubo, T.; Jack, C.R.; Kaye, J.; Montine, T.J.; et al. Toward defining the preclinical stages of Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement. 2011, 7, 280–292. [Google Scholar] [CrossRef]
- 2020 Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2020, 16, 391–460. [CrossRef] [PubMed]
- Kivipelto, M.; Mangialasche, F.; Ngandu, T. Lifestyle interventions to prevent cognitive impairment, dementia and Alzheimer disease. Nat. Rev. Neurol. 2018, 14, 653–666. [Google Scholar] [CrossRef] [PubMed]
- Solomon, A.; Mangialasche, F.; Richard, E.; Andrieu, S.; Bennett, D.A.; Breteler, M.; Fratiglioni, L.; Hooshmand, B.; Khachaturian, A.S.; Schneider, L.S.; et al. Advances in the prevention of Alzheimer’s disease and dementia. J. Intern. Med. 2014, 275, 229–250. [Google Scholar] [CrossRef] [PubMed]
- Horrocks, L.A.; Yeo, Y.K. Health benefits of docosahexaenoic acid (DHA). Pharmacol. Res. 1999, 40, 211–225. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Innis, S.M. Genetic Variants of the FADS1 FADS2 Gene Cluster Are Associated with Altered (n-6) and (n-3) Essential Fatty Acids in Plasma and Erythrocyte Phospholipids in Women during Pregnancy and in Breast Milk during Lactation. J. Nutr. 2008, 138, 2222–2228. [Google Scholar] [CrossRef]
- Nita, R.; Kawabata, T.; Kagawa, Y.; Nakayama, K.; Yanagisawa, Y.; Iwamoto, S.; Kimura, F.; Miyazawa, T.; Tatsuta, N.; Arima, T.; et al. Associations of Erythrocyte Fatty Acid Compositions with FADS1 Gene Polymorphism in Japanese Mothers and Infants. Prostaglandins Leukot. Essent. Fatty Acids 2020, 152, 102031. [Google Scholar] [CrossRef] [PubMed]
- Hammouda, S.; Ghzaiel, I.; Khamlaoui, W.; Hammami, S.; Mhenni, S.Y.; Samet, S.; Hammami, M.; Zarrouk, A. Genetic Variants in FADS1 and ELOVL2 Increase Level of Arachidonic Acid and the Risk of Alzheimer’s Disease in the Tunisian Population. Prostaglandins Leukot. Essent. Fat. Acids 2020, 160, 102159. [Google Scholar] [CrossRef]
- Rapoport, S.I.; Rao, J.S.; Igarashi, M. Brain metabolism of nutritionally essential polyunsaturated fatty acids depends on both the diet and the liver. Prostaglandins Leukot. Essent. Fat. Acids 2007, 77, 251–261. [Google Scholar] [CrossRef]
- Muskiet, F.A.J.; Fokkema, M.R.; Schaafsma, A.; Boersma, E.R.; Crawford, M.A. Is Docosahexaenoic Acid (DHA) Essential? Lessons from DHA Status Regulation, Our Ancient Diet, Epidemiology and Randomized Controlled Trials. J. Nutr. 2004, 134, 183–186. [Google Scholar] [CrossRef]
- Domenichiello, A.F.; Kitson, A.P.; Bazinet, R.P. Is docosahexaenoic acid synthesis from α-linolenic acid sufficient to supply the adult brain? Prog. Lipid Res. 2015, 59, 54–66. [Google Scholar] [CrossRef] [PubMed]
- Neuringer, M.; Connor, W.E.; Lin, D.S.; Barstad, L.; Luck, S. Biochemical and functional effects of prenatal and postnatal ω3 fatty acid deficiency on retina and brain in rhesus monkeys. Proc. Natl. Acad. Sci. USA 1986, 83, 4021–4025. [Google Scholar] [CrossRef]
- Colombo, J.; Kannass, K.N.; Shaddy, D.J.; Kundurthi, S.; Maikranz, J.M.; Anderson, C.J.; Blaga, O.M.; Carlson, S.E. Maternal DHA and the Development of Attention in Infancy and Toddlerhood. Child Dev. 2004, 75, 1254–1267. [Google Scholar] [CrossRef] [PubMed]
- Lucas, A.; Morley, R.; Cole, T.J.; Lister, G.; Leeson-Payne, C. Breast milk and subsequent intelligence quotient in. Lancet 1992, 339, 261–264. [Google Scholar] [CrossRef]
- Fedorova, I.; Salem, N. Omega-3 fatty acids and rodent behavior. Prostaglandins Leukot. Essent. Fat. Acids 2006, 75, 271–289. [Google Scholar] [CrossRef] [PubMed]
- DeMar, J.C.; Ma, K.; Bell, J.M.; Igarashi, M.; Greenstein, D.; Rapoport, S.I. One generation of n-3 polyunsaturated fatty acid deprivation increases depression and aggression test scores in rats. J. Lipid Res. 2006, 47, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Barberger-Gateau, P.; Raffaitin, C.; Letenneur, L.; Berr, C.; Tzourio, C.; Dartigues, J.F.; Alpérovitch, A. Dietary Patterns and Risk of Dementia: The Three-City Cohort Study. Neurology 2007, 69, 1921–1930. [Google Scholar] [CrossRef]
- Morris, M.C.; Evans, D.A.; Bienias, J.L.; Tangney, C.C.; Bennett, D.A.; Wilson, R.S.; Aggarwal, N.; Schneider, J. Consumption of Fish and N-3 Fatty Acids and Risk of Incident Alzheimer Disease. Arch. Neurol. 2003, 60, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Kalmijn, S.; Launer, L.J.; Ott, A.; Witteman, J.C.M.; Hofman, A.; Breteler, M.M.B. Dietary fat intake and the risk of incident dementia in the Rotterdam study. Ann. Neurol. 1997, 42, 776–782. [Google Scholar] [CrossRef]
- Barberger-Gateau, P.; Letenneur, L.; Deschamps, V.; Pérès, K.; Dartigues, J.F.; Renaud, S. Fish, meat, and risk of dementia: Cohort study. Br. Med. J. 2002, 325, 932–933. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, J.; Qiu, J.; Li, Y.; Wang, J.; Jiao, J. Intakes of fish and polyunsaturated fatty acids and mild-to-severe cognitive impairment risks: A dose-response meta-analysis of 21 cohort studies. Am. J. Clin. Nutr. 2016, 103, 330–340. [Google Scholar] [CrossRef]
- Hashimoto, M.; Shahdat, H.M.; Katakura, M.; Tanabe, Y.; Gamoh, S.; Miwa, K.; Shimada, T.; Shido, O. Effects of Docosahexaenoic Acid on in Vitro Amyloid Beta Peptide 25-35 Fibrillation. Biochim. Biophys. Acta—Mol. Cell Biol. Lipids 2009, 1791, 289–296. [Google Scholar] [CrossRef]
- Tatsumi, Y.; Kato, A.; Sango, K.; Himeno, T.; Kondo, M.; Kato, Y.; Kamiya, H.; Nakamura, J.; Kato, K. Omega-3 Polyunsaturated Fatty Acids Exert Anti-Oxidant Effects through the Nuclear Factor (Erythroid-Derived 2)-Related Factor 2 Pathway in Immortalized Mouse Schwann Cells. J. Diabetes Investig. 2019, 10, 602–612. [Google Scholar] [CrossRef]
- Hashimoto, M.; Hossain, S.; Al Mamun, A.; Matsuzaki, K.; Arai, H. Docosahexaenoic acid: One molecule diverse functions. Crit. Rev. Biotechnol. 2017, 37, 579–597. [Google Scholar] [CrossRef] [PubMed]
- Green, K.N.; Martinez-Coria, H.; Khashwji, H.; Hall, E.B.; Yurko-Mauro, K.A.; Ellis, L.; LaFerla, F.M. Dietary Docosahexaenoic Acid and Docosapentaenoic Acid Ameliorate Amyloid-β and Tau Pathology via a Mechanism Involving Presenilin 1 Levels. J. Neurosci. 2007, 27, 4385–4395. [Google Scholar] [CrossRef]
- Akbar, M.; Calderon, F.; Wen, Z.; Kim, H.-Y. Docosahexaenoic acid: A positive modulator of Akt signaling in neuronal survival. Proc. Natl. Acad. Sci. USA 2005, 102, 10858–10863. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Chiang, N.; van Dyke, T.E. Resolving inflammation: Dual anti-inflammatory and pro-resolution lipid mediators. Nat. Rev. Immunol. 2008, 8, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Devassy, J.G.; Leng, S.; Gabbs, M.; Monirujjaman, M.; Aukema, H.M. Omega-3 polyunsaturated fatty acids and oxylipins in neuroinflammation and management of Alzheimer disease. Adv. Nutr. 2016, 7, 905–916. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhu, M.; Hjorth, E.; Cortés-Toro, V.; Eyjolfsdottir, H.; Graff, C.; Nennesmo, I.; Palmblad, J.; Eriksdotter, M.; Sambamurti, K.; et al. Resolution of Inflammation Is Altered in Alzheimer’s Disease. Alzheimer’s Dement. 2015, 11, 40–50. [Google Scholar] [CrossRef]
- Cunnane, S.C.; Chouinard-Watkins, R.; Castellano, C.A.; Barberger-Gateau, P. Docosahexaenoic acid homeostasis, brain aging and Alzheimer’s disease: Can we reconcile the evidence? Prostaglandins Leukot. Essent. Fat. Acids 2013, 88, 61–70. [Google Scholar] [CrossRef]
- Ahmmed, M.K.; Ahmmed, F.; Tian, H.; Carne, A.; Bekhit, A.E.D. Marine omega-3 (n-3) phospholipids: A comprehensive review of their properties, sources, bioavailability, and relation to brain health. Compr. Rev. Food Sci. Food Saf. 2020, 19, 64–123. [Google Scholar] [CrossRef]
- Sugasini, D.; Yalagala, P.C.R.; Goggin, A.; Tai, L.M.; Subbaiah, P.V. Enrichment of brain docosahexaenoic acid (DHA) is highly dependent upon the molecular carrier of dietary DHA: Lysophosphatidylcholine is more efficient than either phosphatidylcholine or triacylglycerol. J. Nutr. Biochem. 2019, 74, 108231. [Google Scholar] [CrossRef]
- Yalagala, P.C.R.; Sugasini, D.; Zaldua, S.B.; Tai, L.M.; Subbaiah, P.V. Lipase Treatment of Dietary Krill Oil, but Not Fish Oil, Enables Enrichment of Brain Eicosapentaenoic Acid and Docosahexaenoic Acid. Mol. Nutr. Food Res. 2020, 64, 1–13. [Google Scholar] [CrossRef]
- Bazinet, R.P.; Bernoud-Hubac, N.; Lagarde, M. How the plasma lysophospholipid and unesterified fatty acid pools supply the brain with docosahexaenoic acid. Prostaglandins Leukot. Essent. Fat. Acids 2019, 142, 1–3. [Google Scholar] [CrossRef]
- Eser Ocak, P.; Ocak, U.; Sherchan, P.; Zhang, J.H.; Tang, J. Insights into major facilitator superfamily domain-containing protein-2a (Mfsd2a) in physiology and pathophysiology. What do we know so far? J. Neurosci. Res. 2020, 98, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Hachem, M.; Belkouch, M.; Lo Van, A.; Picq, M.; Bernoud-Hubac, N.; Lagarde, M. Brain targeting with docosahexaenoic acid as a prospective therapy for neurodegenerative diseases and its passage across blood brain barrier. Biochimie 2020, 170, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Harrington, C.R. The Molecular Pathology of Alzheimer’s Disease. Neuroimaging Clin. N. Am. 2012, 22, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Lim, G.P.; Calon, F.; Morihara, T.; Yang, F.; Teter, B.; Ubeda, O.; Salem, N.; Frautschy, S.A.; Cole, G.M. A Diet Enriched with the Omega-3 Fatty Acid Docosahexaenoic Acid Reduces Amyloid Burden in an Aged Alzheimer Mouse Model. J. Neurosci. 2005, 25, 3032–3040. [Google Scholar] [CrossRef] [PubMed]
- Oksman, M.; Iivonen, H.; Hogyes, E.; Amtul, Z.; Penke, B.; Leenders, I.; Broersen, L.; Lütjohann, D.; Hartmann, T.; Tanila, H. Impact of Different Saturated Fatty Acid, Polyunsaturated Fatty Acid and Cholesterol Containing Diets on Beta-Amyloid Accumulation in APP/PS1 Transgenic Mice. Neurobiol. Dis. 2006, 23, 563–572. [Google Scholar] [CrossRef]
- Hashimoto, M.; Hossain, S.; Shimada, T.; Shido, O. Docosahexaenoic acid-induced protective effect against impaired learning in amyloid β-infused rats is associated with increased synaptosomal membrane fluidity. Clin. Exp. Pharmacol. Physiol. 2006, 33, 934–939. [Google Scholar] [CrossRef]
- Clandinin, M.T.; Jumpsen, J. Fatty Acid Metabolism in Brain in Relation to Development, Membrane Structure, and Signaling. In Handbook of Essential Fatty Acid Biology: Biochemistry, Physiology, and Behavioral Neurobiology; Yehuda, S., Mostofsky, D.I., Eds.; Humana Press: Totowa, NJ, USA, 1997; pp. 15–65. [Google Scholar] [CrossRef]
- Onuki, Y.; Morishita, M.; Chiba, Y.; Tokiwa, S.; Takayama, K. Docosahexaenoic acid and eicosapentaenoic acid induce changes in the physical properties of a lipid bilayer model membrane. Chem. Pharm. Bull. 2006, 54, 68–71. [Google Scholar] [CrossRef]
- Kawakita, E.; Hashimoto, M.; Shido, O. Docosahexaenoic acid promotes neurogenesis in vitro and in vivo. Neuroscience 2006, 139, 991–997. [Google Scholar] [CrossRef]
- Moreno-Jiménez, E.P.; Flor-García, M.; Terreros-Roncal, J.; Rábano, A.; Cafini, F.; Pallas-Bazarra, N.; Ávila, J.; Llorens-Martín, M. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer’s disease. Nat. Med. 2019, 25, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zhang, L.; Wen, M.; Du, L.; Gao, X.; Xue, C.; Xu, J.; Wang, Y. Enhanced Neuroprotective Effect of DHA and EPA-Enriched Phospholipids against 1-Methyl-4-Phenyl-1,2,3,6-Tetrahydropyridine (MPTP) Induced Oxidative Stress in Mice Brain. J. Funct. Foods 2016, 25, 385–396. [Google Scholar] [CrossRef]
- Liu, Q.; Wu, D.; Ni, N.; Ren, H.; Luo, C.; He, C.; Kang, J.X.; Wan, J.B.; Su, H. Omega-3 polyunsaturated fatty acids protect neural progenitor cells against oxidative injury. Mar. Drugs 2014, 12, 2341–2356. [Google Scholar] [CrossRef]
- Belayev, L.; Khoutorova, L.; Atkins, K.D.; Bazan, N.G. Robust docosahexaenoic acid-mediated neuroprotection in a rat model of transient, focal cerebral ischemia. Stroke. J. Cereb. Circ. 2009, 40, 3121–3126. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, H.; Zhang, H.; Leak, R.K.; Shi, Y.; Hu, X.; Gao, Y.; Chen, J. Dietary supplementation with omega-3 polyunsaturated fatty acids robustly promotes neurovascular restorative dynamics and improves neurological functions after stroke. Exp. Neurol. 2015, 272, 170–180. [Google Scholar] [CrossRef] [PubMed]
- Sastre, M.; Klockgether, T.; Heneka, M.T. Contribution of inflammatory processes to Alzheimer’s disease: Molecular mechanisms. Int. J. Dev. Neurosci. 2006, 24, 167–176. [Google Scholar] [CrossRef]
- Lukiw, W.J.; Cui, J.-G.; Marcheselli, V.L.; Bodker, M.; Botkjaer, A.; Gotlinger, K.; Serhan, C.N.; Bazan, N.G. A Role for Docosahexaenoic Acid-Derived Neuroprotectin D1 in Neural Cell Survival and Alzheimer Disease. J. Clin. Investig. 2005, 115, 2774–2783. [Google Scholar] [CrossRef]
- Miyazawa, K.; Fukunaga, H.; Tatewaki, Y.; Takano, Y.; Yamamoto, S.; Mutoh, T.; Taki, Y. Alzheimer’s Disease and Specialized pro-Resolving Lipid Mediators: Do MAR1, RVD1, and NPD1 Show Promise for Prevention and Treatment? Int. J. Mol. Sci. 2020, 21, 5783. [Google Scholar] [CrossRef]
- Bazan, N.G. Omega-3 fatty acids, pro-inflammatory signaling and neuroprotection. Curr. Opin. Clin. Nutr. Metab. Care 2007, 10, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Marcheselli, V.L.; Hong, S.; Lukiw, W.J.; Tian, X.H.; Gronert, K.; Musto, A.; Hardy, M.; Gimenez, J.M.; Chiang, N.; Serhan, C.N.; et al. Novel Docosanoids Inhibit Brain Ischemia-Reperfusion-Mediated Leukocyte Infiltration and Pro-Inflammatory Gene Expression. J. Biol. Chem. 2003, 278, 43807–43817. [Google Scholar] [CrossRef]
- Bazan, N.G.; Calandria, J.M.; Gordon, W.C. Docosahexaenoic acid and its derivative neuroprotectin d1 display neuroprotective properties in the retina, brain and central nervous system. Nestle Nutr. Inst. Workshop Ser. 2013, 77, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Beydoun, M.A.; Kaufman, J.S.; Sloane, P.D.; Heiss, G.; Ibrahim, J. N-3 Fatty Acids, Hypertension and Risk of Cognitive Decline among Older Adults in the Atherosclerosis Risk in Communities (ARIC) Study. Public Health Nutr. 2008, 11, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Dunn, H.C.; Ager, R.R.; Baglietto-Vargas, D.; Cheng, D.; Kitazawa, M.; Cribbs, D.H.; Medeiros, R. Restoration of Lipoxin A4 Signaling Reduces Alzheimer’s Disease-Like Pathology in the 3xTg-AD Mouse Model. J. Alzheimer’s Dis. 2014, 43, 893–903. [Google Scholar] [CrossRef]
- Kuszewski, J.C.; Wong, R.H.X.; Wood, L.G.; Howe, P.R.C. Effects of Fish Oil and Curcumin Supplementation on Cerebrovascular Function in Older Adults: A Randomized Controlled Trial. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.C.; Brockman, J.; Schneider, J.A.; Wang, Y.; Bennett, D.A.; Tangney, C.C.; van de Rest, O. Association of Seafood Consumption, Brain Mercury Level, and APOE Ε4 Status With Brain Neuropathology in Older Adults. JAMA 2016, 315, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Pettegrew, J.W.; Panchalingam, K.; Hamilton, R.L.; Mcclure, R.J. Brain Membrane Phospholipid Alterations in Alzheimer’s Disease. Neurochem. Res. 2001, 26, 771–782. [Google Scholar] [CrossRef]
- Yassine, H.N.; Feng, Q.; Azizkhanian, I.; Rawat, V.; Castor, K.; Fonteh, A.N.; Harrington, M.G.; Zheng, L.; Reed, B.R.; DeCarli, C.; et al. Association of Serum Docosahexaenoic Acid with Cerebral Amyloidosis. JAMA Neurol. 2016, 73, 1208–1216. [Google Scholar] [CrossRef]
- Gottfries, C.G.; Karlsson, I.; Svennerholm, L. Membrane Components Separate Early-Onset Alzheimer’s Disease from Senile Dementia of the Alzheimer Type. Int. Psychogeriatr. 1996, 8, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Söderberg, M.; Edlund, C.; Kristensson, K.; Dallner, G. Fatty acid composition of brain phospholipids in aging and in Alzheimer’s disease. Lipids 1991, 26, 421–425. [Google Scholar] [CrossRef]
- Soderberg, M.; Edlund, C.; Alafuzoff, I.; Kristensson, K.; Dallner, G. Lipid Composition in Different Regions of the Brain in Alzheimer’s Disease/Senile Dementia of Alzheimer’s Type. J. Neurochem. 1992, 59, 1646–1653. [Google Scholar] [CrossRef]
- Stokes, C.E.; Hawthorne, J.N. Reduced Phosphoinositide Concentrations in Anterior Temporal Cortex of Alzheimer-Diseased Brains. J. Neurochem. 1987, 48, 1018–1021. [Google Scholar] [CrossRef] [PubMed]
- Nitsch, R.M.; Blusztajn, J.K.; Pittas, A.G.; Slack, B.E.; Growdon, J.H.; Wurtman, R.J. Evidence for a membrane defect in Alzheimer disease brain. Proc. Natl. Acad. Sci. USA 1992, 89, 1671–1675. [Google Scholar] [CrossRef]
- Wells, K.; Farooqui, A.A.; Liss, L.; Horrocks, L.A. Neural membrane phospholipids in alzheimer disease. Neurochem. Res. 1995, 20, 1329–1333. [Google Scholar] [CrossRef] [PubMed]
- Farooqui, A.A.; Rapoport, S.I.; Horrocks, L.A. Membrane phospholipid alterations in Alzheimer’s disease: Deficiency of ethanolamine plasmalogens. Neurochem. Res. 1997, 22, 523–527. [Google Scholar] [CrossRef]
- Prasad, M.R.; Lovell, M.A.; Yatin, M.; Dhillon, H.; Markesbery, W.R. Regional membrane phospholipid alterations in Alzheimer’s disease. Neurochem. Res. 1998, 23, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Guan, Z.Z.; Söderberg, M.; Sindelar, P.; Edlund, C. Content and fatty acid composition of cardiolipin in the brain of patients with alzheimer’s disease. Neurochem. Int. 1994, 25, 295–300. [Google Scholar] [CrossRef]
- Nakada, T.; Kwee, I.L.; Ellis, W.G. Membrane Fatty Acid Composition shows. NeuroReport 1990, 1, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Skinner, E.R.; Watt, C.; Besson, J.A.O.; Best, P.V. Differences in the fatty acid composition of the grey and white matter of different regions of the brains of patients with alzheimer’s disease and control subjects. Brain 1993, 116, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, M.; Ma, K.; Gao, F.; Kim, H.; Rapoport, S.I.; Rao, J.S. concentrations in Alzheimer disease prefrontal cortex. Brain 2011, 24, 507–517. [Google Scholar] [CrossRef]
- Fraser, T.; Tayler, H.; Love, S. Fatty acid composition of frontal, temporal and parietal neocortex in the normal human brain and in Alzheimer’s disease. Neurochem. Res. 2010, 35, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Tully, A.M.; Roche, H.M.; Doyle, R.; Fallon, C.; Bruce, I.; Lawlor, B.; Coakley, D.; Gibney, M.J. Low Serum Cholesteryl Ester-Docosahexaenoic Acid Levels in Alzheimer’s Disease: A Case–Control Study. Br. J. Nutr. 2003, 89, 483–489. [Google Scholar] [CrossRef]
- Conquer, J.A.; Tierney, M.C.; Zecevic, J.; Bettger, W.J.; Fisher, R.H. Fatty acid analysis of blood plasma of patients with Alzheimer’s disease, other types of dementia, and cognitive impairment. Lipids 2000, 35, 1305–1312. [Google Scholar] [CrossRef] [PubMed]
- Kyle, D.J.; Schaefer, E.; Patton, G.; Beiser, A. Low serum docosahexaenoic acid is a significant risk factor for Alzheimer’s dementia. Lipids 1999, 34 (Suppl. 6), S245. [Google Scholar] [CrossRef]
- Heude, B.; Ducimetière, P.; Berr, C. Cognitive decline and fatty acid composition of erythrocyte membranes—The EVA Study. Am. J. Clin. Nutr. 2003, 77, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, M.; Poljak, A.; Braidy, N.; Crawford, J.; Sachdev, P. Blood fatty acids in Alzheimer’s disease and mild cognitive impairment: A meta-analysis and systematic review. Ageing Res. Rev. 2020, 60, 101043. [Google Scholar] [CrossRef] [PubMed]
- Hodson, L.; Skeaff, C.M.; Fielding, B.A. Fatty acid composition of adipose tissue and blood in humans and its use as a biomarker of dietary intake. Prog. Lipid Res. 2008, 47, 348–380. [Google Scholar] [CrossRef]
- Arab, L. Biomarkers of Fat and Fatty Acid Intake. J. Nutr. 2003, 133, 925S–932S. [Google Scholar] [CrossRef]
- Jackson, K.H.; Polreis, J.M.; Tintle, N.L.; Kris-Etherton, P.M.; Harris, W.S. Association of reported fish intake and supplementation status with the omega-3 index. Prostaglandins Leukot. Essent. Fat. Acids 2019, 142, 4–10. [Google Scholar] [CrossRef]
- Laidlaw, M.; Holub, B.J. Effects of supplementation with fish oil-derived n-3 fatty acids and γ-linolenic acid on circulating plasma lipids and fatty acid profiles in women. Am. J. Clin. Nutr. 2003, 77, 37–42. [Google Scholar] [CrossRef]
- Conklin, S.M.; Gianaros, P.J.; Brown, S.M.; Yao, J.K.; Hariri, A.R.; Manuck, S.B.; Muldoon, M.F. Long-Chain Omega-3 Fatty Acid Intake Is Associated Positively with Corticolimbic Gray Matter Volume in Healthy Adults. Neurosci. Lett. 2007, 421, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Connor, W.E.; Neuringer, M.; Lin, D.S. Dietary effects on brain fatty acid composition: The reversibility of n-3 fatty acid deficiency and turnover of docosahexaenoic acid in the brain, erythrocytes, and plasma of rhesus monkeys. J. Lipid Res. 1990, 31, 237–247. [Google Scholar] [CrossRef]
- Arellanes, I.C.; Choe, N.; Solomon, V.; He, X.; Kavin, B.; Martinez, A.E.; Kono, N.; Buennagel, D.P.; Hazra, N.; Kim, G.; et al. Brain Delivery of Supplemental Docosahexaenoic Acid (DHA): A Randomized Placebo-Controlled Clinical Trial. EBioMedicine 2020, 59, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Cunnane, S.C.; Schneider, J.A.; Tangney, C.; Tremblay-Mercier, J.; Fortier, M.; Bennett, D.A.; Morris, M.C. Plasma and Brain Fatty Acid Profiles in Mild Cognitive Impairment and Alzheimer’s Disease. J. Alzheimer’s Dis. 2012, 29, 691–697. [Google Scholar] [CrossRef]
- Faxen-Irving, G.; Falahati, F.; Basun, H.; Eriksdotter, M.; Vedin, I.; Wahlund, L.O.; Schultzberg, M.; Hjorth, E.; Palmblad, J.; Cederholm, T.; et al. Does Fatty Acid Composition in Subcutaneous Adipose Tissue Differ between Patients with Alzheimer’s Disease and Cohabiting Proxies? J. Alzheimer’s Dis. 2017, 61, 515–519. [Google Scholar] [CrossRef] [PubMed]
- Eriksdotter, M.; Vedin, I.; Falahati, F.; Freund-Levi, Y.; Hjorth, E.; Faxen-Irving, G.; Wahlund, L.O.; Schultzberg, M.; Basun, H.; Cederholm, T.; et al. Plasma Fatty Acid Profiles in Relation to Cognition and Gender in Alzheimer’s Disease Patients during Oral Omega-3 Fatty Acid Supplementation: The OmegAD Study. J. Alzheimer’s Dis. 2015, 48, 805–812. [Google Scholar] [CrossRef]
- Sydenham, E.; Dangour, A.D.; Lim, W.S. Omega 3 fatty acid for the prevention of cognitive decline and dementia. Sao Paulo Med. J. 2012, 130, 419. [Google Scholar] [CrossRef]
- Fotuhi, M.; Mohassel, P.; Yaffe, K. Fish consumption, long-chain omega-3 fatty acids and risk of cognitive decline or Alzheimer disease: A complex association. Nat. Clin. Pract. Neurol. 2009, 5, 140–152. [Google Scholar] [CrossRef]
- Cunnane, S.; Plourde, M.; Pifferi, F.; Bégin, M.; Féart, C.; Barberger-Gateau, P. Fish, docosahexaenoic acid and Alzheimer’s disease. Prog. Lipid Res. 2009, 48, 239–256. [Google Scholar] [CrossRef]
- Alex, A.; Abbott, K.A.; McEvoy, M.; Schofield, P.W.; Garg, M.L. Long-chain omega-3 polyunsaturated fatty acids and cognitive decline in non-demented adults: A systematic review and meta-analysis. Nutr. Rev. 2020, 78, 563–578. [Google Scholar] [CrossRef]
- Zhang, X.; Han, H.; Ge, X.; Liu, L.; Wang, T.; Yu, H. Effect of n-3 long-chain polyunsaturated fatty acids on mild cognitive impairment: A meta-analysis of randomized clinical trials. Eur. J. Clin. Nutr. 2020, 74, 548–554. [Google Scholar] [CrossRef] [PubMed]
- Balachandar, R.; Soundararajan, S.; Bagepally, B.S. Docosahexaenoic acid supplementation in age-related cognitive decline: A systematic review and meta-analysis. Eur. J. Clin. Pharmacol. 2020, 76, 639–648. [Google Scholar] [CrossRef]
- Stough, C.; Downey, L.; Silber, B.; Lloyd, J.; Kure, C.; Wesnes, K.; Camfield, D. The effects of 90-day supplementation with the Omega-3 essential fatty acid docosahexaenoic acid (DHA) on cognitive function and visual acuity in a healthy aging population. Neurobiol. Aging 2012, 33, e1–e824. [Google Scholar] [CrossRef] [PubMed]
- Van De Rest, O.; Geleijnse, J.M.; Kok, F.J.; Van Staveren, W.A.; Dullemeijer, C.; OldeRikkert, M.G.M.; Beekman, A.T.F.; De Groot, C.P.G.M. Effect of fish oil on cognitive performance in older subjects: A randomized, controlled trial. Neurology 2008, 71, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M.A.; Childs, C.E.; Calder, P.C.; Rogers, P.J. No effect of omega-3 fatty acid supplementation on cognition and mood in individuals with cognitive impairment and probable Alzheimer’s disease: A randomised controlled trial. Int. J. Mol. Sci. 2015, 16, 24600–24613. [Google Scholar] [CrossRef] [PubMed]
- Barbaresko, J.; Lellmann, A.W.; Schmidt, A.; Lehmann, A.; Amini, A.M.; Egert, S.; Schlesinger, S.; Nöthlings, U. Dietary Factors and Neurodegenerative Disorders: An Umbrella Review of Meta-Analyses of Prospective Studies. Adv. Nutr. 2020, 11, 1161–1173. [Google Scholar] [CrossRef] [PubMed]
- Quinn, J.F.; Raman, R.; Thomas, R.G.; Yurko-Mauro, K.; Nelson, E.B.; Van Dyck, C.; Galvin, J.E.; Emond, J.; Jack, C.R.; Weiner, M.; et al. Docosahexaenoic Acid Supplementation and Cognitive Decline in Alzheimer Disease. JAMA 2010, 304, 1903. [Google Scholar] [CrossRef]
- Dyerberg, J.; Madsen, P.; Møller, J.M.; Aardestrup, I.; Schmidt, E.B. Bioavailability of marine n-3 fatty acid formulations. Prostaglandins Leukot. Essent. Fat. Acids 2010, 83, 137–141. [Google Scholar] [CrossRef]
- Patrick, R.P. Role of phosphatidylcholine-DHA in preventing APOE4-associated Alzheimer’s disease. FASEB J. 2019, 33, 1554–1564. [Google Scholar] [CrossRef]
- Tou, J.C.; Jaczynski, J.; Chen, Y.C. Krill for human consumption: Nutritional value and potential health benefits. Nutr. Rev. 2007, 65, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Subbaiah, P.V.; Sowa, J.M.; Davidson, M.H. Evidence for altered positional specificity of LCAT in vivo: Studies with docosahexaenoic acid feeding in humans. J. Lipid Res. 2004, 45, 2245–2251. [Google Scholar] [CrossRef]
- Chen, S.; Subbaiah, P.V. Regioisomers of phosphatidylcholine containing DHA and their potential to deliver dha to the brain: Role of phospholipase specificities. Lipids 2013, 48, 675–686. [Google Scholar] [CrossRef] [PubMed]
- Subbaiah, P.V.; Liu, M.; Bolan, P.J.; Paltauf, F. Altered positional specificity of human plasma lecithin-cholesterol acyltransferase in the presence of sn-2 arachidonoyl phosphatidyl cholines. Mechanism of formation of saturated cholesteryl esters. Biochim. Biophys. Acta (BBA)/Lipids Lipid Metab. 1992, 1128, 83–92. [Google Scholar] [CrossRef]
- Sugasini, D.; Thomas, R.; Yalagala, P.C.R.; Tai, L.M.; Subbaiah, P.V. Dietary docosahexaenoic acid (DHA) as lysophosphatidylcholine, but not as free acid, enriches brain DHA and improves memory in adult mice. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Le Kim, D.; Betzing, H. Intestinal Absorption of Polyunsaturated Phosphatidylcholine in the Rat. Hoppe-Seyler’S Z. Physiol. Chem. 1976, 357, 1321–1332. [Google Scholar] [CrossRef]
- Lemaitre-Delaunay, D.; Pachiaudi, C.; Laville, M.; Pousin, J.; Armstrong, M.; Lagarde, M. Blood compartmental metabolism of docosahexaenoic acid (DHA) in humans after ingestion of a single dose of [13C]DHA in phosphatidylcholine. J. Lipid Res. 1999, 40, 1867–1874. [Google Scholar] [CrossRef]
- Subbaiah, P.V.; Dammanahalli, K.J.; Yang, P.; Bi, J.; O’Donnell, J.M. Enhanced incorporation of dietary DHA into lymph phospholipids by altering its molecular carrier. Biochim. Biophys. Acta (BBA)—Mol. Cell Biol. Lipids 2016, 1861, 723–729. [Google Scholar] [CrossRef] [PubMed]
- Yalagala, P.C.R.; Sugasini, D.; Dasarathi, S.; Pahan, K.; Subbaiah, P.V. Dietary lysophosphatidylcholine-EPA enriches both EPA and DHA in the brain: Potential treatment for depression. J. Lipid Res. 2019, 60, 566–578. [Google Scholar] [CrossRef]
- Zierenberg, O.; Grundy, S.M. Intestinal absorption of polyenephosphatidylcholine in man. J. Lipid Res. 1982, 23, 1136–1142. [Google Scholar] [CrossRef]
- Tall, A.R.; Green, P.H.R.; Glickman, R.M.; Riley, J.W. Metabolic fate of chylomicron phospholipids and apoproteins in the rat. J. Clin. Investig. 1979, 64, 977–989. [Google Scholar] [CrossRef] [PubMed]
- Amate, L.; Gil, A.; Ramírez, M. Feeding infant piglets formula with long-chain polyunsaturated fatty acids as triacylglycerols or phospholipids influences the distribution of these fatty acids in plasma lipoprotein fractions. J. Nutr. 2001, 131, 1250–1255. [Google Scholar] [CrossRef] [PubMed]
- Croset, M.; Brossard, N.; Pachiaudi, C.; Normand, S.; Lecerf, J.; Chirouze, V.; Riou, J.P.; Tayot, J.L.; Lagarde, M. In vivo compartmental metabolism of13C docosahexaenoic acid, studied by gas chromatography-combustion isotope ratio mass spectrometry. Lipids 1996, 31, S109–S115. [Google Scholar] [CrossRef] [PubMed]
- Brossard, N.; Croset, M.; Lecerf, J.; Pachiaudi, C.; Normand, S.; Chirouze, V.; Macovschi, O.; Riou, J.P.; Tayot, J.L.; Lagarde, M. Metabolic fate of an oral tracer dose of [13C]docosahexaenoic acid triglycerides in the rat. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 1996, 270, R846–R854. [Google Scholar] [CrossRef]
- Graf, B.A.; Duchateau, G.S.M.J.E.; Patterson, A.B.; Mitchell, E.S.; van Bruggen, P.; Koek, J.H.; Melville, S.; Verkade, H.J. Age dependent incorporation of 14C-DHA into rat brain and body tissues after dosing various 14C-DHA-esters. Prostaglandins Leukot. Essent. Fat. Acids 2010, 83, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Bickerton, A.S.T.; Roberts, R.; Fielding, B.A.; Hodson, L.; Blaak, E.E.; Wagenmakers, A.J.M.; Gilbert, M.; Karpe, F.; Frayn, K.N. Preferential uptake of dietary fatty acids in adipose tissue and muscle in the postprandial period. Diabetes 2007, 56, 168–176. [Google Scholar] [CrossRef]
- Chouinard-Watkins, R.; Lacombe, R.J.S.; Metherel, A.H.; Masoodi, M.; Bazinet, R.P. DHA Esterified to Phosphatidylserine or Phosphatidylcholine is More Efficient at Targeting the Brain than DHA Esterified to Triacylglycerol. Mol. Nutr. Food Res. 2019, 63, 1801224. [Google Scholar] [CrossRef]
- Umhau, J.C.; Zhou, W.; Carson, R.E.; Rapoport, S.I.; Polozova, A.; Demar, J.; Hussein, N.; Bhattacharjee, A.K.; Ma, K.; Esposito, G.; et al. Imaging incorporation of circulating docosahexaenoic acid into the human brain using positron emission tomography. J. Lipid Res. 2009, 50, 1259–1268. [Google Scholar] [CrossRef]
- Rahman, T.; Taha, A.Y.; Jun Song, B.; Orr, S.K.; Liu, Z.; Chen, C.T.; Bazinet, R.P. The very low density lipoprotein receptor is not necessary for maintaining brain polyunsaturated fatty acid concentrations. Prostaglandins Leukot. Essent. Fat. Acids 2010, 82, 141–145. [Google Scholar] [CrossRef]
- Chen, C.T.; Ma, D.W.L.; Kim, J.H.; Mount, H.T.J.; Bazinet, R.P. The low density lipoprotein receptor is not necessary for maintaining mouse brain polyunsaturated fatty acid concentrations. J. Lipid Res. 2008, 49, 147–152. [Google Scholar] [CrossRef]
- Spector, A.A. Plasma free fatty acid and lipoproteins as sources of polyunsaturated fatty acid for the brain. J. Mol. Neurosci. 2001, 16, 159–165. [Google Scholar] [CrossRef]
- Frayn, K.N.; Summers, L.K.M.; Fielding, B.A. Regulation of the plasma non-esterified fatty acid concentration in the postprandial state. Proc. Nutr. Soc. 1997, 56, 713–721. [Google Scholar] [CrossRef]
- Griffiths, A.J.; Humphreys, S.M.; Clark, M.L.; Fielding, B.A.; Frayn, K.N. Immediate metabolic availability of dietary fat in combination with carbohydrate. Am. J. Clin. Nutr. 1994, 59, 53–59. [Google Scholar] [CrossRef]
- Purdon, D.; Arai, T.; Rapoport, S. No evidence for direct incorporation of esterified palmitic acid from plasma into brain lipids of awake adult rat. J. Lipid Res. 1997, 38, 526–530. [Google Scholar] [CrossRef]
- Ochiai, Y.; Uchida, Y.; Ohtsuki, S.; Tachikawa, M.; Aizawa, S.; Terasaki, T. The blood-brain barrier fatty acid transport protein 1 (FATP1/SLC27A1) supplies docosahexaenoic acid to the brain, and insulin facilitates transport. J. Neurochem. 2017, 141, 400–412. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Short, J.L.; Choy, K.H.C.; Zeng, A.X.; Marriott, P.J.; Owad, Y.; Scanlon, M.J.; Porte, C.J.H.; Nicolazzo, J.A. Fatty acid-binding protein 5 at the blood–brain barrier regulates endogenous brain docosahexaenoic acid levels and cognitive function. J. Neurosci. 2016, 36, 11755–11767. [Google Scholar] [CrossRef]
- Hamilton, J.A.; Brunaldi, K. A model for fatty acid transport into the brain. J. Mol. Neurosci. 2007, 33, 12–17. [Google Scholar] [CrossRef]
- Chouinard-Watkins, R.; Bazinet, R.P. ACSL6 is critical for maintaining brain DHA levels. Proc. Natl. Acad. Sci. USA 2018, 115, 12343–12345. [Google Scholar] [CrossRef]
- Coe, N.R.; Smith, A.J.; Frohnert, B.I.; Watkins, P.A.; Bernlohr, D.A. The fatty acid transport protein (FATP1) is a very long chain acyl-CoA synthetase. J. Biol. Chem. 1999, 274, 36300–36304. [Google Scholar] [CrossRef]
- Fernandez, R.F.; Kim, S.Q.; Zhao, Y.; Foguth, R.M.; Weera, M.M.; Counihan, J.L.; Nomura, D.K.; Chester, J.A.; Cannon, J.R.; Ellis, J.M. Acyl-CoA synthetase 6 enriches the neuroprotective omega-3 fatty acid DHA in the brain. Proc. Natl. Acad. Sci. USA 2018, 115, 12525–12530. [Google Scholar] [CrossRef]
- Gauster, M.; Rechberger, G.; Sovic, A.; Hörl, G.; Steyrer, E.; Sattler, W.; Frank, S. Endothelial lipase releases saturated and unsaturated fatty acids of high density lipoprotein phosphatidylcholine. J. Lipid Res. 2005, 46, 1517–1525. [Google Scholar] [CrossRef]
- Duong, M.N.; Psaltis, M.; Rader, D.J.; Marchadier, D.; Barter, P.J.; Rye, K.A. Evidence That Hepatic Lipase and Endothelial Lipase Have Different Substrate Specificities for High-Density Lipoprotein Phospholipids. Biochemistry 2003, 42, 13778–13785. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Subbaiah, P.V. Phospholipid and fatty acid specificity of endothelial lipase: Potential role of the enzyme in the delivery of docosahexaenoic acid (DHA) to tissues. Biochim. Biophys. Acta—Mol. Cell Biol. Lipids 2007, 1771, 1319–1328. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, J.; Miyashita, K.; Nakajima, K.; Mabuchi, H. Hepatic lipase: A comprehensive view of its role on plasma lipid and lipoprotein metabolism. J. Atheroscler. Thromb. 2015, 22, 1001–1011. [Google Scholar] [CrossRef]
- Brindley, D. Hepatic secretion of lysophosphatidylcholine: A novel transport system for polyunsaturated fatty acids and choline. J. Nutr. Biochem. 1993, 4, 442–449. [Google Scholar] [CrossRef]
- Nguyen, L.N.; Ma, D.; Shui, G.; Wong, P.; Cazenave-Gassiot, A.; Zhang, X.; Wenk, M.R.; Goh, E.L.K.; Silver, D.L. Mfsd2a Is a Transporter for the Essential Omega-3 Fatty Acid Docosahexaenoic Acid. Nature 2014, 509, 503–506. [Google Scholar] [CrossRef]
- Rapoport, S.I.; Ramadan, E.; Basselin, M. Docosahexaenoic acid (DHA) incorporation into the brain from plasma, as an in vivo biomarker of brain DHA metabolism and neurotransmission. Prostaglandins Other Lipid Mediat. 2011, 96, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Lagarde, M.; Bernoud, N.; Thiès, F.; Brossard, N.; Lemaitre-Delaunay, D.; Croset, M.; Lecerf, J. Lysophosphatidylcholine as a carrier of docosahexaenoic acid to target tissues. World Rev. Nutr. Diet. 2001, 88, 173–177. [Google Scholar] [CrossRef]
- Bernoud, N.; Fenart, L.; Molière, P.; Dehouck, M.P.; Lagarde, M.; Cecchelli, R.; Lecerf, J. Preferential transfer of 2-docosahexaenoyl-1-lysophosphatidylcholine through an in vitro blood-brain barrier over unesterified docosahexaenoic acid. J. Neurochem. 1999, 72, 338–345. [Google Scholar] [CrossRef]
- Hachem, M.; Géloën, A.; Van, A.L.; Foumaux, B.; Fenart, L.; Gosselet, F.; Da Silva, P.; Breton, G.; Lagarde, M.; Picq, M.; et al. Efficient Docosahexaenoic Acid Uptake by the Brain from a Structured Phospholipid. Mol. Neurobiol. 2016, 53, 3205–3215. [Google Scholar] [CrossRef]
- Thies, F.; Pillon, C.; Moliere, P.; Lagarde, M.; Lecerf, J. Preferential incorporation of sn-2 lysoPC DHA over unesterified DHA in the young rat brain. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 1994, 267, R1273–R1279. [Google Scholar] [CrossRef] [PubMed]
- Croset, M.; Brossard, N.; Polette, A.; Lagarde, M. Characterization of plasma unsaturated lysophosphatidylcholines in human and rat. Biochem. J. 2000, 345, 61. [Google Scholar] [CrossRef]
- Köhler, A.; Sarkkinen, E.; Tapola, N.; Niskanen, T.; Bruheim, I. Bioavailability of fatty acids from krill oil, krill meal and fish oil in healthy subjects-a randomized, single-dose, cross-over trial. Lipids Health Dis. 2015, 14, 1–10. [Google Scholar] [CrossRef]
- Schuchardt, J.; Schneider, I.; Meyer, H.; Neubronner, J.; von Schacky, C.; Hahn, A. Incorporation of EPA and DHA into plasma phospholipids in response to different omega-3 fatty acid formulations—A comparative bioavailability study of fish oil vs. krill oil. Lipids Health Dis. 2011, 10, 145. [Google Scholar] [CrossRef]
- Liu, L.; Bartke, N.; van Daele, H.; Lawrence, P.; Qin, X.; Park, H.G.; Kothapalli, K.; Windust, A.; Bindels, J.; Wang, Z.; et al. Higher efficacy of dietary DHA provided as a phospholipid than as a triglyceride for brain DHA accretion in neonatal piglets. J. Lipid Res. 2014, 55, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Yurko-Mauro, K.; Kralovec, J.; Bailey-Hall, E.; Smeberg, V.; Stark, J.G.; Salem, N. Similar Eicosapentaenoic Acid and Docosahexaenoic Acid Plasma Levels Achieved with Fish Oil or Krill Oil in a Randomized Double-Blind Four-Week Bioavailability Study. Lipids in Health and Disease 2015, 14, 1–9. [Google Scholar] [CrossRef]
- Adkins, Y.; Laugero, K.D.; Mackey, B.; Kelley, D.S. Accretion of Dietary Docosahexaenoic Acid in Mouse Tissues Did Not Differ between Its Purified Phospholipid and Triacylglycerol Forms. Lipids 2019, 54, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Hiratsuka, S.; Koizumi, K.; Ooba, T.; Yokogoshi, H. Effects of Dietary Docosahexaenoic Acid Connecting Phospholipids on the Learning Ability and Fatty Acid Composition of the Brain. J. Nutr. Sci. Vitaminol. 2009, 55, 374–380. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ghasemifard, S.; Hermon, K.; Turchini, G.M.; Sinclair, A.J. Metabolic fate (absorption, β -oxidation and deposition) of long-chain n -3 fatty acids is affected by sex and by the oil source (krill oil or fish oil) in the rat. Br. J. Nutr. 2015, 114, 684–692. [Google Scholar] [CrossRef] [PubMed]
- Vaisman, N.; Kaysar, N.; Zaruk-Adasha, Y.; Pelled, D.; Brichon, G.; Zwingelstein, G.; Bodennec, J. Correlation between Changes in Blood Fatty Acid Composition and Visual Sustained Attention Performance in Children with Inattention: Effect of Dietary N−3 Fatty Acids Containing Phospholipids. Am. J. Clin. Nutr. 2008, 87, 1170–1180. [Google Scholar] [CrossRef] [PubMed]
- Cook, C.M.; Hallaråker, H.; Sæbø, P.C.; Innis, S.M.; Kelley, K.M.; Sanoshy, K.D.; Berger, A.; Maki, K.C. Bioavailability of Long Chain Omega-3 Polyunsaturated Fatty Acids from Phospholipid-Rich Herring Roe Oil in Men and Women with Mildly Elevated Triacylglycerols. Prostaglandins Leukot. Essent. Fat. Acids 2016, 111, 17–24. [Google Scholar] [CrossRef]
- Ahn, S.H.; Lim, S.J.; Ryu, Y.M.; Park, H.-R.; Suh, H.J.; Han, S.H. Absorption Rate of Krill Oil and Fish Oil in Blood and Brain of Rats. Lipids Health Dis. 2018, 17, 162. [Google Scholar] [CrossRef]
- Kitson, A.P.; Metherel, A.H.; Chen, C.T.; Domenichiello, A.F.; Trépanier, M.O.; Berger, A.; Bazinet, R.P. Effect of Dietary Docosahexaenoic Acid (DHA) in Phospholipids or Triglycerides on Brain DHA Uptake and Accretion. J. Nutr. Biochem. 2016, 33, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Brossard, N.; Croset, M.; Normand, S.; Pousin, J.; Lecerf, J.; Laville, M.; Tayot, J.L.; Lagarde, M. Human Plasma Albumin Transports [13C]Docosahexaenoic Acid in Two Lipid Forms to Blood Cells. J. Lipid Res. 1997, 38, 1571–1582. [Google Scholar] [CrossRef]
- Chen, C.T.; Kitson, A.P.; Hopperton, K.E.; Domenichiello, A.F.; Trépanier, M.O.; Lin, L.E.; Ermini, L.; Post, M.; Thies, F.; Bazinet, R.P. Plasma Non-Esterified Docosahexaenoic Acid Is the Major Pool Supplying the Brain. Sci. Rep. 2015, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- DeMar, J.C.; Ma, K.; Bell, J.M.; Rapoport, S.I. Half-lives of docosahexaenoic acid in rat brain phospholipids are prolonged by 15 weeks of nutritional deprivation of n-3 polyunsaturated fatty acids. J. Neurochem. 2004, 91, 1125–1137. [Google Scholar] [CrossRef] [PubMed]
- Dayton, S.; Hashimoto, S.; Dixon, W.; Pearce, M.L. Composition of lipids in human serum and adipose tissue during prolonged feeding of a diet high in unsaturated fat. J. Lipid Res. 1966, 7, 103–111. [Google Scholar] [CrossRef]
- Ogura, T.; Takada, H.; Okuno, M.; Kitade, H.; Matsuura, T.; Kwon, M.; Arita, S.; Hamazaki, K.; Itomura, M.; Hamazaki, T. Fatty Acid Composition of Plasma, Erythrocytes and Adipose: Their Correlations and Effects of Age and Sex. Lipids 2010, 45, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Browning, L.M.; Walker, C.G.; Mander, A.P.; West, A.L.; Madden, J.; Gambell, J.M.; Young, S.; Wang, L.; Jebb, S.A.; Calder, P.C. Incorporation of Eicosapentaenoic and Docosahexaenoic Acids into Lipid Pools When given as Supplements Providing Doses Equivalent to Typical Intakes of Oily Fish. Am. J. Clin. Nutr. 2012, 96, 748–758. [Google Scholar] [CrossRef]
- Luxwolda, M.F.; Kuipers, R.S.; Koops, J.H.; Muller, S.; de Graaf, D.; Dijck-Brouwer, D.A.J.; Muskiet, F.A.J. Interrelationships between Maternal DHA in Erythrocytes, Milk and Adipose Tissue. Is 1 Wt% DHA the Optimal Human Milk Content? Data from Four Tanzanian Tribes Differing in Lifetime Stable Intakes of Fish. Br. J. Nutr. 2014, 111, 854–866. [Google Scholar] [CrossRef]
- Böckmann, K.A.; von Stumpff, A.; Bernhard, W.; Shunova, A.; Minarski, M.; Frische, B.; Warmann, S.; Schleicher, E.; Poets, C.F.; Franz, A.R. Fatty Acid Composition of Adipose Tissue at Term Indicates Deficiency of Arachidonic and Docosahexaenoic Acid and Excessive Linoleic Acid Supply in Preterm Infants. Eur. J. Nutr. 2021, 60, 861–872. [Google Scholar] [CrossRef] [PubMed]
- Clandinin, M.T.; Chappell, J.E.; Leong, S.; Heim, T.; Swyer, P.R.; Chance, G.W. Extrauterine fatty acid accretion in infant brain: Implications for fatty acid requirements. Early Hum. Dev. 1980, 4, 131–138. [Google Scholar] [CrossRef]
- Haggarty, P. Fatty acid supply to the human fetus. Annu. Rev. Nutr. 2010, 30, 237–255. [Google Scholar] [CrossRef]
- Clandinin, M.T.; Chappell, J.E.; Heim, T.; Swyer, P.R.; Chance, G.W. Fatty acid utilization in perinatal de novo synthesis of tissues. Early Hum. Dev. 1981, 5, 355–366. [Google Scholar] [CrossRef]
- Cunnane, S.C.; Francescutti, V.; Brenna, J.T.; Crawford, M.A. Breast-fed infants achieve a higher rate of brain and whole body docosahexaenoate accumulation than formula-fed infants not consuming dietary docosahexaenoate. Lipids 2000, 35, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Kuipers, R.S.; Luxwolda, M.F.; Offringa, P.J.; Rudi Boersma, E.; Dijck-Brouwer, D.A.J.; Muskiet, F.A.J. Fetal intrauterine whole body linoleic, arachidonic and docosahexaenoic acid contents and accretion rates. Prostaglandins Leukot. Essent. Fat. Acids 2012, 86, 13–20. [Google Scholar] [CrossRef]
- Crawford, M.A.; Williams, G.; Hassam, A.G.; Whitehouse, W.L. Essential Fatty Acids and Fetal Brain Growth. Lancet 1976, 307, 452–453. [Google Scholar] [CrossRef]
- Bourre, J.M.; Piciotti, M. Delta-6 desaturation of alpha-linolenic acid in brain and liver during development and aging in the mouse. Neurosci. Lett. 1992, 141, 65–68. [Google Scholar] [CrossRef]
- Chappus-McCendie, H.; Chevalier, L.; Roberge, C.; Plourde, M. Omega-3 PUFA metabolism and brain modifications during aging. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2019, 94, 109662. [Google Scholar] [CrossRef] [PubMed]
- Fink, R.I.; Kolterman, O.G.; Griffin, J.; Olefsky, J.M. Mechanisms of insulin resistance in aging. J. Clin. Investig. 1983, 71, 1523–1535. [Google Scholar] [CrossRef]
- Ott, A.; Stolk, R.P.; van Harskamp, F.; Pols, H.A.; Hofman, A.; Breteler, M.M. Diabetes mellitus and the risk of dementia: The Rotterdam Study. Neurology 1999, 53, 1937–1942. [Google Scholar] [CrossRef]
- Arvanitakis, Z.; Wilson, R.S.; Bienias, J.L.; Evans, D.A.; Bennett, D.A. Diabetes mellitus and risk of Alzheimer disease and decline in cognitive function. Arch. Neurol. 2004, 61, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Peters, S.A.E.; Woodward, M.; Mejia Arango, S.; Batty, G.D.; Beckett, N.; Beiser, A.; Borenstein, A.R.; Crane, P.K.; Haan, M.; et al. Type 2 Diabetes as a Risk Factor for Dementia in Women Compared With Men: A Pooled Analysis of 2.3 Million People Comprising More Than 100,000 Cases of Dementia. Diabetes Care 2016, 39, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Mosconi, L.; Sorbi, S.; De Leon, M.J.; Li, Y.; Nacmias, B.; Myoung, P.S.; Tsui, W.; Ginestroni, A.; Bessi, V.; Fayyazz, M.; et al. Hypometabolism exceeds atrophy in presymptomatic early-onset familial Alzheimer’s disease. J. Nucl. Med. 2006, 47, 1778–1786. [Google Scholar] [PubMed]
- Silverman, D.H.S.; Small, G.W.; Chang, C.Y.; Lu, C.S.; de Aburto, M.A.K.; Chen, W.; Czernin, J.; Rapoport, S.I.; Pietrini, P.; Alexander, G.E.; et al. Positron Emission Tomography in Evaluation of Dementia. JAMA 2001, 286, 2120. [Google Scholar] [CrossRef] [PubMed]
- Ekblad, L.L.; Johansson, J.; Helin, S.; Viitanen, M.; Laine, H.; Puukka, P.; Jula, A.; Rinne, J.O. Midlife insulin resistance, APOE genotype, and late-life brain amyloid accumulation. Neurology 2018, 90, e1150–e1157. [Google Scholar] [CrossRef]
- Craft, S.; Newcomer, J.; Kanne, S.; Dagogo-Jack, S.; Cryer, P.; Sheline, Y.; Luby, J.; Dagogo-Jack, A.; Alderson, A. Memory improvement following induced hyperinsulinemia in Alzheimer’s disease. Neurobiol. Aging 1996, 17, 123–130. [Google Scholar] [CrossRef]
- Craft, S.; Peskind, E.; Schwartz, M.W.; Schellenberg, G.D.; Raskind, M.; Porte, D. Cerebrospinal fluid and plasma insulin levels in Alzheimer’s disease: Relationship to severity of dementia and apolipoprotein E genotype. Neurology 1998, 50, 164–168. [Google Scholar] [CrossRef]
- Frölich, L.; Blum-Degen, D.; Bernstein, H.G.; Engelsberger, S.; Humrich, J.; Laufer, S.; Muschner, D.; Thalheimer, A.; Türk, A.; Hoyer, S.; et al. Brain insulin and insulin receptors in aging and sporadic Alzheimer’s disease. J. Neural Transm. 1998, 105, 423–438. [Google Scholar] [CrossRef] [PubMed]
- Kellar, D.; Craft, S. Brain insulin resistance in Alzheimer’s disease and related disorders: Mechanisms and therapeutic approaches. Lancet Neurol. 2020, 19, 758–766. [Google Scholar] [CrossRef]
- Digel, M.; Staffer, S.; Ehehalt, F.; Stremmel, W.; Ehehalt, R.; Füllekrug, J. FATP4 contributes as an enzyme to the basal and insulin-mediated fatty acid uptake of C2C12 muscle cells. Am. J. Physiol.-Endocrinol. Metab. 2011, 301, E785–E796. [Google Scholar] [CrossRef]
- Vandal, M.; Freemantle, E.; Tremblay-Mercier, J.; Plourde, M.; Fortier, M.; Bruneau, J.; Gagnon, J.; Bégin, M.; Cunnane, S.C. Plasma omega-3 fatty acid response to a fish oil supplement in the healthy elderly. Lipids 2008, 43, 1085–1089. [Google Scholar] [CrossRef]
- Plourde, M.; Chouinard-Watkins, R.; Vandal, M.; Zhang, Y.; Lawrence, P.; Brenna, J.T.; Cunnane, S.C. Plasma incorporation, apparent retroconversion and β-oxidation of 13C-docosahexaenoic acid in the elderly. Nutr. Metab. 2011, 8, 1–9. [Google Scholar] [CrossRef]
- Corder, E.H.; Saunders, A.M.; Strittmatter, W.J.; Schmechel, D.E.; Gaskell, P.C.; Small, G.W.; Roses, A.D.; Haines, J.L.; Pericak-Vance, M.A. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science 1993, 261, 921–923. [Google Scholar] [CrossRef] [PubMed]
- Riedel, B.C.; Thompson, P.M.; Brinton, R.D. Age, APOE and sex: Triad of risk of Alzheimer’s disease. J. Steroid Biochem. Mol. Biol. 2016, 160, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Cardona, F.; Morcillo, S.; Gonzalo-Marin, M.; Tinahones, F.J. The apolipoprotein E genotype predicts postprandial hypertriglyceridemia in patients with the metabolic syndrome. J. Clin. Endocrinol. Metab. 2005, 90, 2972–2975. [Google Scholar] [CrossRef][Green Version]
- Shin, M.H.; Hee, N.K.; Cui, L.H.; Kweon, S.S.; Park, K.S.; Heo, H.; Nam, H.S.; Jeong, S.K.; Chung, E.K.; Choi, J.S. The Effect of Apolipoprotein E Polymorphism on Lipid Levels in Korean Adults. J. Korean Med Sci. 2005, 20, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Reznik, Y.; Morello, R.; Pousse, P.; Mahoudeau, J.; Fradin, S. The Effect of Age, Body Mass Index, and Fasting Triglyceride Level on Postprandial Lipemia Is Dependent on Apolipoprotein E Polymorphism in Subjects with Non-Insulin-Dependent Diabetes Mellitus. Metab. Clin. Exp. 2002, 51, 1088–1092. [Google Scholar] [CrossRef]
- Plourde, M.; Vohl, M.C.; Vandal, M.; Couture, P.; Lemieux, S.; Cunnane, S.C. Plasma n-3 fatty acid response to an n-3 fatty acid supplement is modulated by apoE 4 but not by the common PPAR- L162V polymorphism in men. Br. J. Nutr. 2009, 102, 1121–1124. [Google Scholar] [CrossRef]
- Chouinard-Watkins, R.; Rioux-Perreault, C.; Fortier, M.; Tremblay-Mercier, J.; Zhang, Y.; Lawrence, P.; Vohl, M.C.; Perron, P.; Lorrain, D.; Brenna, J.T.; et al. Disturbance in uniformly 13C-labelled DHA metabolism in elderly human subjects carrying the apoE Î4 allele. Br. J. Nutr. 2013, 110, 1751–1759. [Google Scholar] [CrossRef]
- Cole, G.M.; Frautschy, S.A. DHA may prevent age-related dementia. J. Nutr. 2010, 140, 869–874. [Google Scholar] [CrossRef] [PubMed]
- Rhea, E.M.; Banks, W.A. Role of the Blood-Brain Barrier in Central Nervous System Insulin Resistance. Front. Neurosci. 2019, 13, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.T.; Bazinet, R.P. Β-Oxidation and Rapid Metabolism, But Not Uptake Regulate Brain Eicosapentaenoic Acid Levels. Prostaglandins Leukot. Essent. Fat. Acids 2015, 92, 33–40. [Google Scholar] [CrossRef]
- Su, H.M.; Bernardo, L.; Mirmiran, M.; Ma, X.H.; Nathanielsz, P.W.; Brenna, J.T. Dietary 18:3n-3 and 22:6n-3 as sources of 22:6n-3 accretion in neonatal baboon brain and associated organs. Lipids 1999, 34, 347–350. [Google Scholar] [CrossRef]
- Abate, G.; Vezzoli, M.; Sandri, M.; Rungratanawanich, W.; Memo, M.; Uberti, D. Mitochondria and cellular redox state on the route from ageing to Alzheimer’s disease. Mech. Ageing Dev. 2020, 192, 111385. [Google Scholar] [CrossRef]
- Swerdlow, R.H.; Burns, J.M.; Khan, S.M. The Alzheimer’s disease mitochondrial cascade hypothesis. J. Alzheimer’s Dis. 2010, 20 (Suppl. 2), S265–S279. [Google Scholar] [CrossRef] [PubMed]
- Calingasan, N.Y.; Uchida, K.; Gibson, G.E. Protein-bound acrolein: A novel marker of oxidative stress in Alzheimer’s disease. J. Neurochem. 1999, 72, 751–756. [Google Scholar] [CrossRef] [PubMed]
- Bayir, H. Reactive oxygen species. Crit. Care Med. 2005, 33 (Suppl. 12), S498–S501. [Google Scholar] [CrossRef]
- Bazinet, R.P.; Layé, S. Polyunsaturated fatty acids and their metabolites in brain function and disease. Nat. Rev. Neurosci. 2014, 15, 771–785. [Google Scholar] [CrossRef]
- Cameron-Smith, D.; Albert, B.B.; Cutfield, W.S. Fishing for answers: Is oxidation of fish oil supplements a problem? J. Nutr. Sci. 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Albert, B.B.; Cameron-Smith, D.; Hofman, P.L.; Cutfield, W.S. Oxidation of marine omega-3 supplements and human health. BioMed Res. Int. 2013, 2013, 464921. [Google Scholar] [CrossRef] [PubMed]
- Lagarde, M. Docosahexaenoic acid: Nutrient and precursor of bioactive lipids. Eur. J. Lipid Sci. Technol. 2008, 110, 673–678. [Google Scholar] [CrossRef]
- Tanito, M.; Brush, R.S.; Elliott, M.H.; Wicker, L.D.; Henry, K.R.; Anderson, R.E. High levels of retinal membrane docosahexaenoic acid increase susceptibility to stress-induced degeneration. J. Lipid Res. 2009, 50, 807–819. [Google Scholar] [CrossRef] [PubMed]
- Grimm, M.O.W.; Haupenthal, V.J.; Mett, J.; Stahlmann, C.P.; Blumel, T.; Mylonas, N.T.; Endres, K.; Grimm, H.S.; Hartmann, T. Oxidized Docosahexaenoic Acid Species and Lipid Peroxidation Products Increase Amyloidogenic Amyloid Precursor Protein Processing. Neurodegener. Dis. 2016, 16, 44–54. [Google Scholar] [CrossRef]
- Serra-Majem, L.; Nissensohn, M.; Øverby, N.C.; Fekete, K. Dietary methods and biomarkers of omega 3 fatty acids: A systematic review. Br. J. Nutr. 2012, 107 (Suppl. 2), S64–S76. [Google Scholar] [CrossRef]
- Kuratko, C.N.; Salem, N. Biomarkers of DHA status. Prostaglandins Leukot. Essent. Fat. Acids 2009, 81, 111–118. [Google Scholar] [CrossRef]
- Sparkes, C.; Sinclair, A.J.; Gibson, R.A.; Else, P.L.; Meyer, B.J. High variability in erythrocyte, plasma and whole blood EPA and DHA levels in response to supplementation. Nutrients 2020, 12, 1017. [Google Scholar] [CrossRef]
- Harris, W.S. Omega-3 fatty acids and cardiovascular disease: A case for omega-3 index as a new risk factor. Pharmacol. Res. 2007, 55, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Shearer, G.C.; Pottala, J.V.; Spertus, J.A.; Harris, W.S. Red Blood Cell Fatty Acid Patterns and Acute Coronary Syndrome. PLoS ONE 2009, 4, e5444. [Google Scholar] [CrossRef]
- Harris, W.S.; Assaad, B.; Poston, W.C. Tissue Omega-6/Omega-3 Fatty Acid Ratio and Risk for Coronary Artery Disease. Am. J. Cardiol. 2006, 98 (Suppl. 1), 19–26. [Google Scholar] [CrossRef]
- Martínez-Fernández, L.; Laiglesia, L.M.; Huerta, A.E.; Martínez, J.A.; Moreno-Aliaga, M.J. Omega-3 fatty acids and adipose tissue function in obesity and metabolic syndrome. Prostaglandins Other Lipid Mediat. 2015, 121 Pt A, 24–41. [Google Scholar] [CrossRef]
- Kalupahana, N.S.; Claycombe, K.J.; Moustaid-Moussa, N. (n-3) Fatty Acids Alleviate Adipose Tissue Inflammation and Insulin Resistance: Mechanistic Insights. Adv. Nutr. 2011, 2, 304–316. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.R.; Midgette, Y.; Shah, R. Fish Oil Derived Omega 3 Fatty Acids Suppress Adipose NLRP3 Inflammasome Signaling in Human Obesity. J. Endocr. Soc. 2019, 3, 504–515. [Google Scholar] [CrossRef] [PubMed]
- Todorčević, M.; Hodson, L. The Effect of Marine Derived n-3 Fatty Acids on Adipose Tissue Metabolism and Function. J. Clin. Med. 2016, 5, 3. [Google Scholar] [CrossRef] [PubMed]
- Hames, K.C.; Morgan-Bathke, M.; Harteneck, D.A.; Zhou, L.; Port, J.D.; Lanza, I.R.; Jensen, M.D. Very-Long-Chain ω-3 Fatty Acid Supplements and Adipose Tissue Functions: A Randomized Controlled Trial. Am. J. Clin. Nutr. 2017, 105, 1552–1558. [Google Scholar] [CrossRef] [PubMed]
- Hrboticky, N.; MacKinnon, M.J.; Innis, S.M. Effect of a vegetable oil formula rich in linoleic acid on tissue fatty acid accretion in the brain, liver, plasma, and erythrocytes of infant piglets. Am. J. Clin. Nutr. 1990, 51, 173–182. [Google Scholar] [CrossRef]
- Hrboticky, N.; MacKinnon, M.J. Retina Fatty Acid Composition of Piglets Fed from Birth with a Linoleic Acid-Rich Vegetable-Oil Formula for Infants13. 1991, Volume 53. Available online: https://academic.oup.com/ajcn/article/53/2/483/4694259 (accessed on 3 March 2021).
- Ramsden, C.E.; Hennebelle, M.; Schuster, S.; Keyes, G.S.; Johnson, C.D.; Kirpich, I.A.; Dahlen, J.E.; Horowitz, M.S.; Zamora, D.; Feldstein, A.E.; et al. Effects of diets enriched in linoleic acid and its peroxidation products on brain fatty acids, oxylipins, and aldehydes in mice. Biochim. Biophys. Acta-Mol. Cell Biol. Lipids 2018, 1863, 1206–1213. [Google Scholar] [CrossRef] [PubMed]
- Ramsden, C.E.; Ringel, A.; Feldstein, A.E.; Taha, A.Y.; MacIntosh, B.A.; Hibbeln, J.R.; Majchrzak-Hong, S.F.; Faurot, K.R.; Rapoport, S.I.; Cheon, Y.; et al. Lowering dietary linoleic acid reduces bioactive oxidized linoleic acid metabolites in humans. Prostaglandins Leukot. Essent. Fat. Acids 2012, 87, 135–141. [Google Scholar] [CrossRef]
- Serhan, C.N.; Clish, C.B.; Brannon, J.; Colgan, S.P.; Chiang, N.; Gronert, K. Novel functional sets of lipid-derived mediators with antiinflammatory actions generated from omega-3 fatty acids via cyclooxygenase 2-nonsteroidal antiinflammatory drugs and transcellular processing. J. Exp. Med. 2000, 192, 1197–1204. [Google Scholar] [CrossRef]
- Cayer, L.G.J.; Mendonça, A.M.; Pauls, S.D.; Winter, T.; Leng, S.; Taylor, C.G.; Zahradka, P.; Aukema, H.M. Adipose tissue oxylipin profiles vary by anatomical site and are altered by dietary linoleic acid in rats. Prostaglandins Leukot. Essent. Fat. Acids 2019, 141, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.; Kevala, K.; Kim, H.Y. Depletion of brain docosahexaenoic acid impairs recovery from traumatic brain injury. PLoS ONE 2014, 9, 86472. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y. Neuroprotection by docosahexaenoic acid in brain injury. Mil. Med. 2014, 179, 106–111. [Google Scholar] [CrossRef] [PubMed]
| Author | Model/Population | Tissue | Study Duration | Comparison | Results | Comment |
|---|---|---|---|---|---|---|
| Chouinard-Watkins et al., 2019 [118] | Rats | Brain cortex and serum lipid classes | 6 h | DHA esterified to phosphatidylcholine (DHA-PtdCho), phosphatidylethanolamine (DHA-PtdEtn), phosphatidylserine (DHA-PtdSer) or as triglyceride (TG-DHA) | Brain DHA levels 5-7 fold higher in DHA-PtdCho and DHA-PtdSer groups than in TG-DHA group. | |
| Graf et al., 2010 [116] | Rats | Brain | 24 h | PL- vs. TG-DHA | In 10-week-old rats, tissues such as liver, brain, kidney and anterior uveal tract (retina) accumulated 2–3 fold more PL-DHA-derived radioactivity than compared with TG-DHA. 14C-DHA derived radioactivity after 14C-DHA-PC dosing compared with 14C-DHA-TG dosing. | DHA-ester type did not influence tissue uptake in rats aged <10 weeks old, while age influenced tissue uptake regardless of DHA-ester type. |
| Köhler et al., 2015 [144] | Adult humans | Plasma phospholipids | 72 h | Two krill products (krill oil, krill meal) vs. FO | A larger AUC of plasma DHA was detected for krill oil in comparison to krill meal or fish oil. Bioavailability of EPA+DHA was not different between krill meal and fish oil. | A large inter-individual variability in response was observed. |
| Schuchdart et al., 2011 [145] | Adult humans | Plasma phospholipids | 72 h | Two FO products containing DHA as either ethyl-ester or re-esterified TG compared against KO | Nonsignificant differences in AUC and maximum plasma phospholipid concentration of DHA between all groups. | High standard deviation values. |
| Liu et al., 2014 [146] | Piglets | Brain | 6 days | PL- vs. TG-DHA. Results normalised as %Dose of radiolabelled DHA found in the grey matter of the cerebral cortex for each PL-DHA and TG-DHA. | The %dose of PL-DHA was 1.9× more efficacious for grey matter DHA accretion than TG-DHA. | Less retro-conversion to N3 DPA in the TG-DHA group (PC > TG 2.8 fold). TG-DHA provided as 4.8 mg/500 mL feed vs. PL-DHA 1.8 mg/500 mL feed. Data regarding brain updake from total dietary TG-DHA/PL-DHA was not displayed. |
| Yurko-Mauro et al., 2015 [147] | Adult humans | Plasma and Erythrocyte | 28 days | FO (containing DHA as either ethyl-ester or re-esterified TG) and KO. | No significant differences in plasma or erythrocyte EPA + DHA at 28 days between groups. | |
| Adkins et al., 2019 [148] | Mice | Liver, Adipose, Heart, Eye, Brain. | 38 days | PL- vs. TG-DHA | No difference in brain DHA concentration. | |
| Hiratsuka et al., 2009 [149] | Mice | Liver and Brain | 5 weeks | PL- vs. TG-DHA | No significant differences in brain or liver fatty acid contents or of DHA content. | |
| Ghasemifard et al., 2015 [150] | Rats | Whole body, Liver, heart, white gastrocnemius muscle and perirenal adipose tissue | 6 weeks | FO vs. KO | No significant effect of diet on net accumulation of DHA. | |
| Vaisman et al., 2008 [151] | Children aged 8-13 years | Blood lipid profile | 3 months | PL-DHA vs. FO | No significant change to blood lipids after three months. | |
| Studies with acute and delayed measurement phases | ||||||
| Author | Model/Population | Tissue | Study Duration | Comparison | First Timepoint Results | Second Timepoint Results |
| Cook et al., 2016 [152] | Adult humans | Plasma phospholipids | 12 h and 14 days | PL-rich herring roe oil or TG-rich FO | After 12 h, the ability of PL-DHA to increase the AUC of EPA, DHA and EPA +DHA was 2-fold that of TG-DHA. | After 2 weeks, there was no significant difference in the abilities of each supplement to increase plasma EPA+DHA. |
| Ahn et al., 2018 [153] | Rats | Blood and Brain | 48 h and 14 days | FO and two forms of KO | TG-DHA increased brain DHA the most at 2 h, but PL-DHA in KO achieved greatest brain DHA at 48 h. The statistical significance of these findings was not described in the paper. | No significant difference in DHA content between FO, KO, and CKO groups in brain or blood. |
| Kitson et al., 2016 [154] | Rats | Brain | 6 h and 4 weeks | PL- vs. TG-DHA | PL-DHA achieved 78%, 140% and 69% greater labelling in cerebellum, hippocampus and remainder of brain than the TG-DHA group. | No difference in brain DHA concentration between groups fed PC-DHA, TG-DHA or a combination of both PC- and TG-DHA. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heath, R.J.; Wood, T.R. Why Have the Benefits of DHA Not Been Borne Out in the Treatment and Prevention of Alzheimer’s Disease? A Narrative Review Focused on DHA Metabolism and Adipose Tissue. Int. J. Mol. Sci. 2021, 22, 11826. https://doi.org/10.3390/ijms222111826
Heath RJ, Wood TR. Why Have the Benefits of DHA Not Been Borne Out in the Treatment and Prevention of Alzheimer’s Disease? A Narrative Review Focused on DHA Metabolism and Adipose Tissue. International Journal of Molecular Sciences. 2021; 22(21):11826. https://doi.org/10.3390/ijms222111826
Chicago/Turabian StyleHeath, Rory J., and Thomas R. Wood. 2021. "Why Have the Benefits of DHA Not Been Borne Out in the Treatment and Prevention of Alzheimer’s Disease? A Narrative Review Focused on DHA Metabolism and Adipose Tissue" International Journal of Molecular Sciences 22, no. 21: 11826. https://doi.org/10.3390/ijms222111826
APA StyleHeath, R. J., & Wood, T. R. (2021). Why Have the Benefits of DHA Not Been Borne Out in the Treatment and Prevention of Alzheimer’s Disease? A Narrative Review Focused on DHA Metabolism and Adipose Tissue. International Journal of Molecular Sciences, 22(21), 11826. https://doi.org/10.3390/ijms222111826






