Insights into the Role of Tick Salivary Protease Inhibitors during Ectoparasite–Host Crosstalk
Abstract
1. Introduction
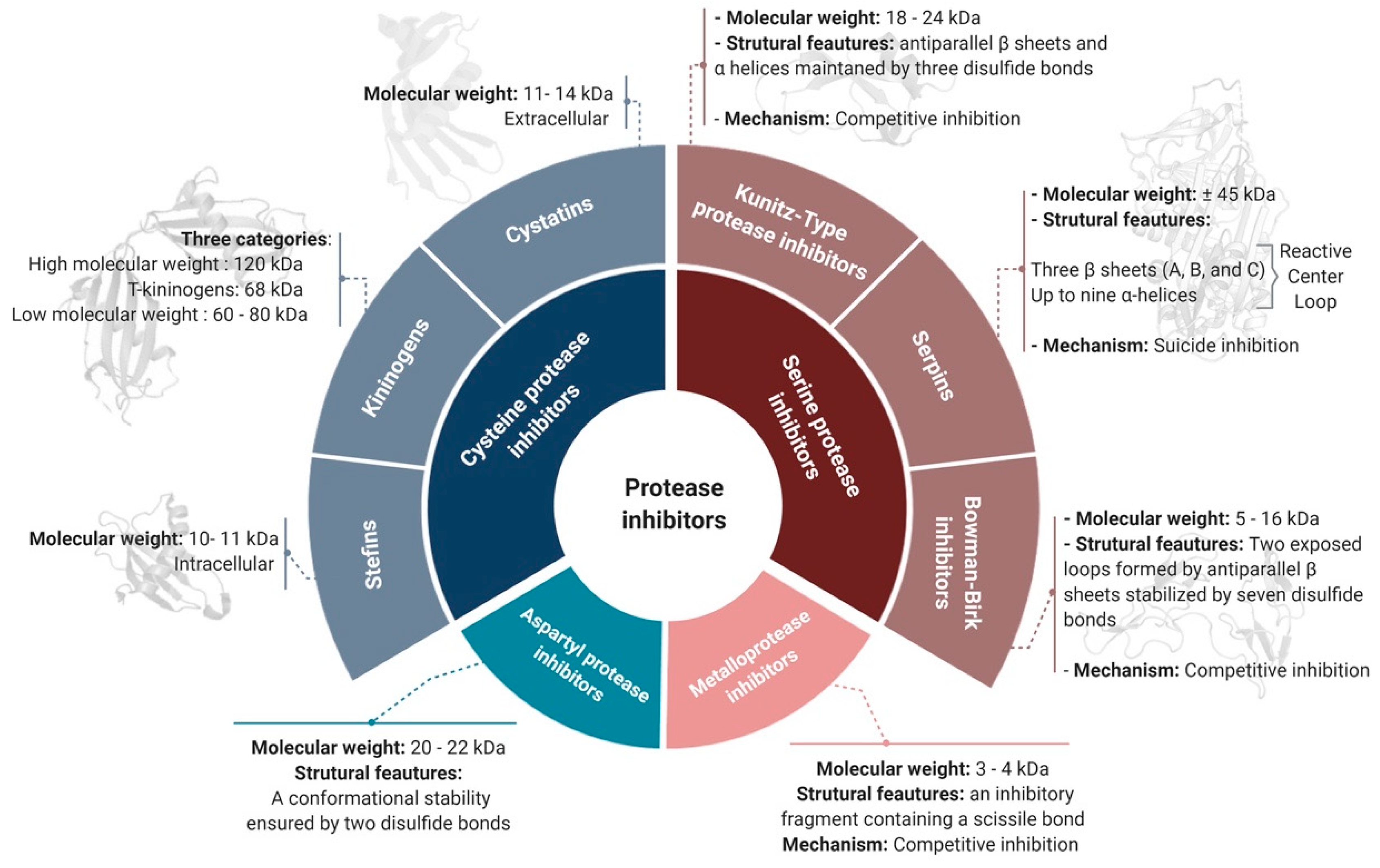
2. Classification of Protease Inhibitors
2.1. Target-Based Classification
2.1.1. Serine Protease Inhibitors
Serpins
Kunitz-Type PIs
Bowman–Birk Inhibitors (BBIs)
2.1.2. Cysteine Protease Inhibitors
2.1.3. Metalloproteases Inhibitors
2.1.4. Aspartyl Protease Inhibitors
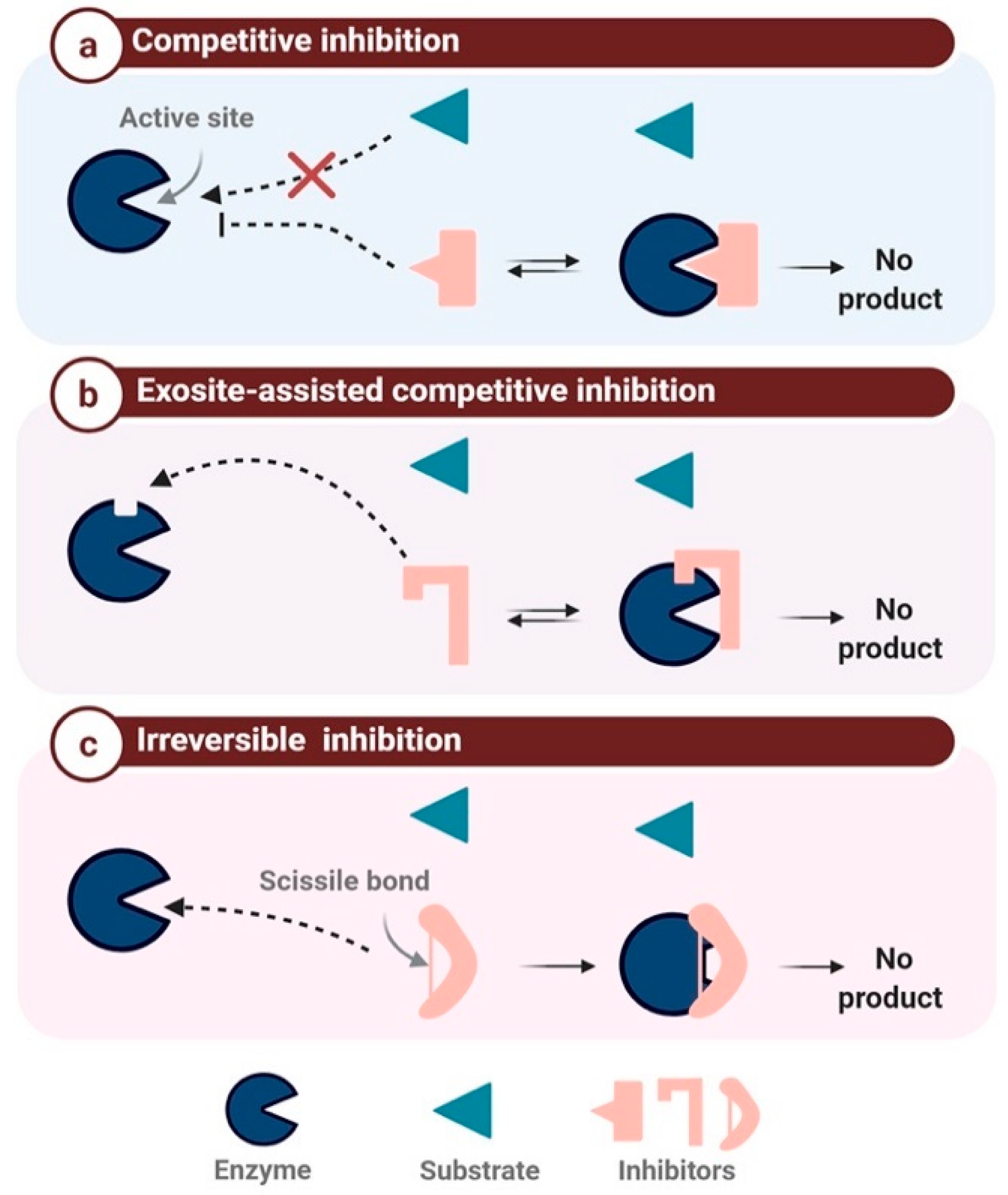
2.2. Mechanism-Based Classification
2.2.1. Competitive Protease Inhibitors
2.2.2. Exosite-Assisted Competitive Inhibitors
2.2.3. Irreversible Inhibition
3. Applications of Protease Inhibitors
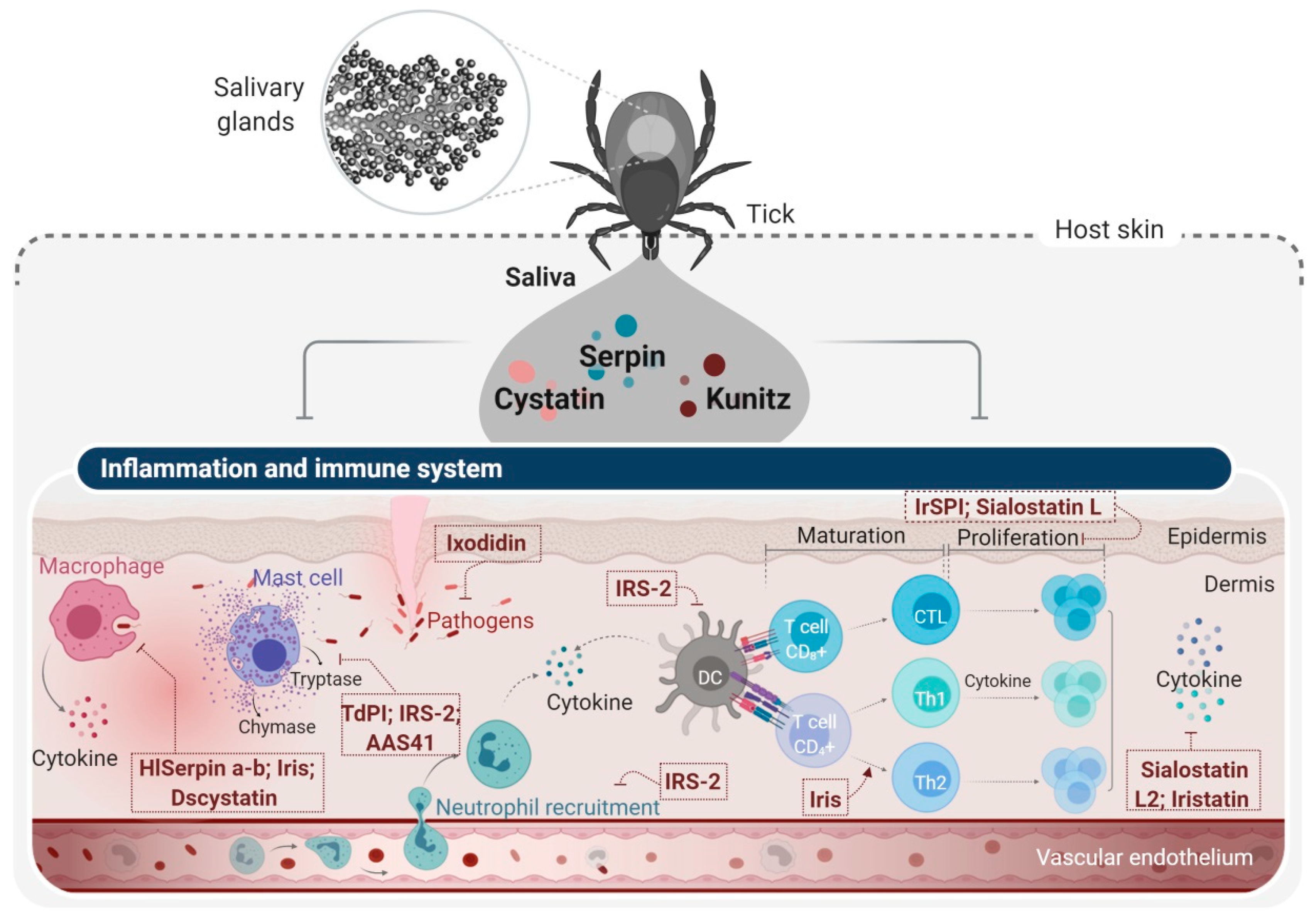
4. Tick Salivary Glands: An Attractive Source of PIs with Potential Pharmacological Applications
4.1. Tick Serine Protease Inhibitors and Their Applications
4.2. Tick Cysteine PIs and Their Applications
5. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ADP | Adenosine diphosphate |
| BBI | Bowman–Birk inhibitor |
| BMDM | Bone-marrow-derived macrophages |
| CAM | Chicken chorioallantoic membrane |
| EAE | Experimental autoimmune encephalomyelitis |
| HIV | Human immunodeficiency virus |
| IFN | Interferon |
| IL | Interleukin |
| Ir-CPI | Ixodes ricinus contact phase inhibitor |
| IRF | IFN-regulatory factor |
| IrSPI | Ixodes Ricinus serine protease inhibitor |
| IRS-2 | Ixodes ricinus serpin-2 |
| KPIs | Kazal-type proteinase inhibitors |
| MMP | Matrix metallopeptidase |
| MPI | Metalloprotease inhibitor |
| NRTI | Nucleotide/nucleoside reverse transcriptase inhibitor |
| NNRTI | Non-nucleoside reverse transcriptase inhibitor |
| OVA | Ovalbumin |
| PBMC | Peripheral blood mononuclear cells |
| PDGF | Platelet-Derived Growth Factor |
| PI | Protease inhibitor |
| RCL | Reactive center loop |
| RENCA | Murine renal adenocarcinoma |
| RGD | Arginylglycylaspartic acid |
| RHS8 | Rhipicephalus haemaphysaloides |
| ROS | Reactive oxygen species |
| STAT | Signal transducer and activator of transcription |
| TAP | Tick anticoagulant peptide |
| TdPI | Tick-derived protease inhibitor |
| TIL | Trypsin inhibitor like |
| TLR | Toll-like receptor |
| TNF | Tumor necrosis factor |
| TRAF | TNF receptor-associated factor |
| VEGF | Vascular endothelial growth factor |
References
- Hartl, M.; Giri, A.P.; Kaur, H.; Baldwin, I.T. The multiple functions of plant serine protease inhibitors: Defense against herbivores and beyond. Plant Signal. Behav. 2011, 6, 1009–1011. [Google Scholar] [CrossRef] [PubMed]
- Craik, D.J.; Fairlie, D.P.; Liras, S.; Price, D. The future of peptide-based drugs. Chem. Biol. Drug Des. 2013, 81, 136–147. [Google Scholar] [CrossRef]
- Shamsi, T.N.; Parveen, R.; Fatima, S. Characterization, biomedical and agricultural applications of protease inhibitors: A review. Int. J. Biol. Macromol. 2016, 91, 1120–1133. [Google Scholar] [CrossRef] [PubMed]
- Harish, B.S.; Uppuluri, K.B. Microbial serine protease inhibitors and their therapeutic applications. Int. J. Biol. Macromol. 2018, 107, 1373–1387. [Google Scholar] [CrossRef]
- Dunaevsky, Y.E.; Popova, V.V.; Semenova, T.A.; Beliakova, G.A.; Belozersky, M.A. Fungal inhibitors of proteolytic enzymes: Classification, properties, possible biological roles, and perspectives for practical use. Biochimie 2014, 101, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Sabotic, J.; Kos, J. Microbial and fungal protease inhibitors—Current and potential applications. Appl. Microbiol. Biotechnol. 2012, 93, 1351–1375. [Google Scholar] [CrossRef]
- Oliva, M.L.; Sampaio, M.U. Action of plant proteinase inhibitors on enzymes of physiopathological importance. An. Acad. Bras. Cienc. 2009, 81, 615–621. [Google Scholar] [CrossRef]
- Gagaoua, M.; Hafid, K.; Boudida, Y.; Becila, S.; Ouali, A.; Picard, B.; Boudjellal, A.; Sentandreu, M.A. Caspases and Thrombin Activity Regulation by Specific Serpin Inhibitors in Bovine Skeletal Muscle. Appl. Biochem. Biotechnol. 2015, 177, 279–303. [Google Scholar] [CrossRef] [PubMed]
- Doljak, B.; Cateni, F.; Anderluh, M.; Procida, G.; Zilic, J.; Zacchigna, M. Glycerolipids as selective thrombin inhibitors from the fungus Stereum hirsutum. Drug Dev. Ind. Pharm. 2006, 32, 635–643. [Google Scholar] [CrossRef]
- Overall, C.M.; Blobel, C.P. In search of partners: Linking extracellular proteases to substrates. Nat. Rev. Mol. Cell Biol. 2007, 8, 245–257. [Google Scholar] [CrossRef]
- Stibraniova, I.; Bartikova, P.; Holikova, V.; Kazimirova, M. Deciphering Biological Processes at the Tick-Host Interface Opens New Strategies for Treatment of Human Diseases. Front. Physiol. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Chmelar, J.; Kotal, J.; Langhansova, H.; Kotsyfakis, M. Protease Inhibitors in Tick Saliva: The Role of Serpins and Cystatins in Tick-host-Pathogen Interaction. Front. Cell. Infect. Microbiol. 2017, 7, 216. [Google Scholar] [CrossRef] [PubMed]
- Rawlings, N.D.; Alan, J.; Thomas, P.D.; Huang, X.D.; Bateman, A.; Finn, R.D. The MEROPS database of proteolytic enzymes, their substrates and inhibitors in 2017 and a comparison with peptidases in the PANTHER database. Nucleic Acids Res. 2018, 46, D624–D632. [Google Scholar] [CrossRef] [PubMed]
- Laskowski, M.; Kato, I. Protein Inhibitors of Proteinases. Annu. Rev. Biochem. 1980, 49, 593–626. [Google Scholar] [CrossRef] [PubMed]
- Rawlings, N.D.; Tolle, D.P.; Barrett, A.J. Evolutionary families of peptidase inhibitors. Biochem. J. 2004, 378, 705–716. [Google Scholar] [CrossRef]
- Jacobson, A.R.; Adler, M.; Silvaggi, N.R.; Allen, K.N.; Smith, G.M.; Fredenburg, R.A.; Stein, R.L.; Park, J.B.; Feng, X.C.; Shoemaker, C.B.; et al. Small molecule metalloprotease inhibitor with in vitro, ex vivo and in vivo efficacy against botulinum neurotoxin serotype A. Toxicon Off. J. Int. Soc. Toxinol. 2017, 137, 36–47. [Google Scholar] [CrossRef]
- Page, M.J.; Di Cera, E. Serine peptidases: Classification, structure and function. Cell. Mol. Life Sci. 2008, 65, 1220–1236. [Google Scholar] [CrossRef]
- Billinger, E.; Zuo, S.S.; Johansson, G. Characterization of Serine Protease Inhibitor from Solanum tuberosum Conjugated to Soluble Dextran and Particle Carriers. ACS Omega 2019, 4, 18456–18464. [Google Scholar] [CrossRef]
- Durvanger, Z.; Boros, E.; Hegedus, R.; Dobo, J.; Kocsis, A.; Fodor, K.; Gal, P.; Mezo, G.; Pal, G.; Harmat, V.; et al. Studying the Structural Basis for Selectivity in Complexes of Peptide Inhibitors and Serine-Proteases of the Complement System. Acta. Crystallogr. A 2019, 75, E120. [Google Scholar] [CrossRef]
- Shi, Y.H.; Shao, Y.N.; Lv, Z.M.; Li, C.H. Serpin-type serine protease inhibitor mediates coelomocyte apoptosis in Apostichopus japonicus. Fish Shellfish Immunol. 2020, 104, 410–418. [Google Scholar] [CrossRef]
- Wei, X.M.; Xu, J.; Yang, J.M.; Liu, X.Q.; Zhang, R.R.; Wang, W.J.; Yang, J.L. Involvement of a Serpin serine protease inhibitor (OoSerpin) from mollusc Octopus ocellatus in antibacterial response. Fish Shellfish Immunol. 2015, 42, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Gettins, P.G.W. Serpin structure, mechanism, and function. Chem. Rev. 2002, 102, 4751–4803. [Google Scholar] [CrossRef] [PubMed]
- Hellinger, R.; Gruber, C.W. Peptide-based protease inhibitors from plants. Drug Discov. Today 2019, 24, 1877–1889. [Google Scholar] [CrossRef] [PubMed]
- Huntington, J.A. Serpin structure, function and dysfunction. J. Thromb. Haemost. 2011, 9, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Turk, B.; Turk, D.; Turk, V. Protease signalling: The cutting edge. EMBO J. 2012, 31, 1630–1643. [Google Scholar] [CrossRef]
- Bendre, A.D.; Ramasamy, S.; Suresh, C.G. Analysis of Kunitz inhibitors from plants for comprehensive structural and functional insights. Int. J. Biol. Macromol. 2018, 113, 933–943. [Google Scholar] [CrossRef] [PubMed]
- Blisnick, A.A.; Foulon, T.; Bonnet, S.I. Serine Protease Inhibitors in Ticks: An Overview of Their Role in Tick Biology and Tick-Borne Pathogen Transmission. Front. Cell. Infect. Microbiol. 2017, 7, 199. [Google Scholar] [CrossRef]
- Gomes, M.T.; Oliva, M.L.; Lopes, M.T.; Salas, C.E. Plant proteinases and inhibitors: An overview of biological function and pharmacological activity. Curr. Protein Pept. Sci. 2011, 12, 417–436. [Google Scholar] [CrossRef]
- Yang, X.; van der Donk, W.A. Ribosomally synthesized and post-translationally modified peptide natural products: New insights into the role of leader and core peptides during biosynthesis. Chemistry 2013, 19, 7662–7677. [Google Scholar] [CrossRef]
- Armstrong, W.B.; Taylor, T.H.; Kennedy, A.R.; Melrose, R.J.; Messadi, D.V.; Gu, M.; Le, A.D.; Perloff, M.; Civantos, F.; Goodwin, W.J.; et al. Bowman birk inhibitor concentrate and oral leukoplakia: A randomized phase IIb trial. Cancer Prev. Res. 2013, 6, 410–418. [Google Scholar] [CrossRef]
- Dai, H.; Ciric, B.; Zhang, G.X.; Rostami, A. Bowman-Birk Inhibitor attenuates experimental autoimmune encephalomyelitis by delaying infiltration of inflammatory cells into the CNS. Immunol. Res. 2011, 51, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Safavi, F.; Rostami, A. Role of serine proteases in inflammation: Bowman-Birk protease inhibitor (BBI) as a potential therapy for autoimmune diseases. Exp. Mol. Pathol. 2012, 93, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Palavalli, M.H.; Natarajan, S.S.; Wang, T.T.; Krishnan, H.B. Imbibition of soybean seeds in warm water results in the release of copious amounts of Bowman-Birk protease inhibitor, a putative anticarcinogenic agent. J. Agric. Food Chem. 2012, 60, 3135–3143. [Google Scholar] [CrossRef] [PubMed]
- Zajc, I.; Sever, N.; Bervar, A.; Lah, T.T. Expression of cysteine peptidase cathepsin L and its inhibitors stefins A and B in relation to tumorigenicity of breast cancer cell lines. Cancer Lett. 2002, 187, 185–190. [Google Scholar] [CrossRef]
- Martins, L.A.; Kotal, J.; Bensaoud, C.; Chmelar, J.; Kotsyfakis, M. Small protease inhibitors in tick saliva and salivary glands and their role in tick-host-pathogen interactions. Biochim. Biophys. Acta Proteins Proteom. 2020, 1868, 140336. [Google Scholar] [CrossRef]
- Srikanth, S.; Chen, Z. Plant Protease Inhibitors in Therapeutics-Focus on Cancer Therapy. Front. Pharmacol. 2016, 7, 470. [Google Scholar] [CrossRef]
- Filler, G.; Bokenkamp, A.; Hofmann, W.; Le Bricon, T.; Martinez-Bru, C.; Grubb, A. Cystatin C as a marker of GFR-history, indications, and future research. Clin. Biochem. 2005, 38, 1–8. [Google Scholar] [CrossRef]
- Priyadarshini, M.; Khan, R.H.; Bano, B. Physicochemical properties of thiol proteinase inhibitor isolated from goat pancreas. Biopolymers 2010, 93, 708–717. [Google Scholar] [CrossRef]
- Benchabane, M.; Schluter, U.; Vorster, J.; Goulet, M.C.; Michaud, D. Plant cystatins. Biochimie 2010, 92, 1657–1666. [Google Scholar] [CrossRef]
- Vorster, B.J.; Goulet, M.C.; Michaud, D. Plant cystatins and insect cysteine proteases: Weapons in a molecular arms race. S. Afr. J. Bot. 2012, 79, 221–222. [Google Scholar]
- Farady, C.J.; Craik, C.S. Mechanisms of macromolecular protease inhibitors. Chembiochem A Eur. J. Chem. Biol. 2010, 11, 2341–2346. [Google Scholar] [CrossRef] [PubMed]
- Gomis-Ruth, F.X.; Maskos, K.; Betz, M.; Bergner, A.; Huber, R.; Suzuki, K.; Yoshida, N.; Nagase, H.; Brew, K.; Bourenkov, G.P.; et al. Mechanism of inhibition of the human matrix metalloproteinase stromelysin-1 by TIMP-1. Nature 1997, 389, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Bateman, K.S.; James, M.N. Plant protein proteinase inhibitors: Structure and mechanism of inhibition. Curr. Protein Pept. Sci. 2011, 12, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Turra, D.; Lorito, M. Potato type I and II proteinase inhibitors: Modulating plant physiology and host resistance. Curr. Protein Pept. Sci. 2011, 12, 374–385. [Google Scholar] [CrossRef] [PubMed]
- Li, B.J.; Gadahi, J.A.; Gao, W.X.; Zhang, Z.C.; Ehsan, M.; Xu, L.X.; Song, X.K.; Li, X.R.; Yan, R.F. Characterization of a novel aspartyl protease inhibitor from Haemonchus contortus. Parasites Vectors 2017, 10. [Google Scholar] [CrossRef]
- Mondal, M.; Radeva, N.; Koster, H.; Park, A.; Potamitis, C.; Zervou, M.; Klebe, G.; Hirsch, A.K.H. Structure-Based Design of Inhibitors of the Aspartic Protease Endothiapepsin by Exploiting Dynamic Combinatorial Chemistry. Angew. Chem. Int. Edit. 2014, 53, 3259–3263. [Google Scholar] [CrossRef]
- Motwani, H.V.; De Rosa, M.; Odell, L.R.; Hallberg, A.; Larhed, M. Aspartic protease inhibitors containing tertiary alcohol transition-state mimics. Eur. J. Med. Chem. 2015, 90, 462–490. [Google Scholar] [CrossRef]
- Jiang, L.G.; Andersen, L.M.; Andreasen, P.A.; Chen, L.Q.; Huang, M.D. Insights into the serine protease mechanism based on structural observations of the conversion of a peptidyl serine protease inhibitor to a substrate. BBA Gen. Subj. 2016, 1860, 599–606. [Google Scholar] [CrossRef]
- Zuchowski, J.; Grzywnowicz, K. Partial purification of proteinase K inhibitors from liquid-cultured mycelia of the white rot basidiomycete Trametes versicolor. Curr. Microbiol. 2006, 53, 259–264. [Google Scholar] [CrossRef]
- Laskowski, M.; Qasim, M.A. What can the structures of enzyme-inhibitor complexes tell us about the structures of enzyme substrate complexes? BBA Protein Struct. M 2000, 1477, 324–337. [Google Scholar] [CrossRef]
- Grosse-Holz, F.M.; van der Hoorn, R.A.L. Juggling jobs: Roles and mechanisms of multifunctional protease inhibitors in plants. New Phytol. 2016, 210, 794–807. [Google Scholar] [CrossRef] [PubMed]
- Clemente, M.; Corigliano, M.G.; Pariani, S.A.; Sanchez-Lopez, E.F.; Sander, V.A.; Ramos-Duarte, V.A. Plant Serine Protease Inhibitors: Biotechnology Application in Agriculture and Molecular Farming. Int. J. Mol. Sci. 2019, 20, 1345. [Google Scholar] [CrossRef] [PubMed]
- Joshi, R.S.; Mishra, M.; Suresh, C.G.; Gupta, V.S.; Giri, A.P. Complementation of intramolecular interactions for structural-functional stability of plant serine proteinase inhibitors. BBA Gen. Subj. 2013, 1830, 5087–5094. [Google Scholar] [CrossRef] [PubMed]
- Stoops, J.K.; Schroeter, J.P.; Kolodziej, S.J.; Strickland, D.K. Structure-Function-Relationships of Human Alpha(2)-Macroglobulin-3-Dimensional Structures of Native Alpha(2)-Macroglobulin and Its Methylamine and Chymotrypsin Derivatives. Biol. Alpha2 Macroglobulin Recept. Relat. Proteins 1994, 737, 212–228. [Google Scholar] [CrossRef]
- Antao, C.M.; Malcata, F.X. Plant serine proteases: Biochemical, physiological and molecular features. Plant Physiol. Bioch. 2005, 43, 637–650. [Google Scholar] [CrossRef]
- Kodoth, S.M.; Chavan, S.J.; Pahwa, S.G. Immunomodulatory effect of protease inhibitor Nelfinavir on host cells: Implications for treatment in HIV disease. J. Allergy Clin. Immun. 2001, 107, S199. [Google Scholar]
- Sierko, E.; Wojtukiewicz, M.Z.; Zimnoch, L.; Tokajuk, P.; Ostrowska-Cichocka, K.; Kisiel, W. Co-localization of Protein Z, Protein Z-Dependent protease inhibitor and coagulation factor X in human colon cancer tissue: Implications for coagulation regulation on tumor cells. Thromb. Res. 2012, 129, E112–E118. [Google Scholar] [CrossRef]
- Barragan, P.; Podzamczer, D. Lopinavir/ritonavir: A protease inhibitor for HIV-1 treatment. Expert Opin. Pharmaco. 2008, 9, 2363–2375. [Google Scholar] [CrossRef]
- Ng, T.B.; Lam, S.K.; Fong, W.P. A homodimeric sporamin-type trypsin inhibitor with antiproliferative, HIV reverse transcriptase-inhibitory and antifungal activities from wampee (Clausena lansium) seeds. Biol. Chem. 2003, 384, 289–293. [Google Scholar] [CrossRef]
- Koblinski, J.E.; Ahram, M.; Sloane, B.F. Unraveling the role of proteases in cancer. Clin. Chim. Acta 2000, 291, 113–135. [Google Scholar] [CrossRef]
- Clemente, A.; Arques, M.D. Bowman-Birk inhibitors from legumes as colorectal chemopreventive agents. World J. Gastroentero. 2014, 20, 10305–10315. [Google Scholar] [CrossRef] [PubMed]
- Mannisto, T.K.; Karvonen, K.E.; Kerola, T.V.; Ryhanen, L.J. Inhibitory effect of the angiotensin converting enzyme inhibitors captopril and enalapril on the conversion of procollagen to collagen. J. Hypertens. 2001, 19, 1835–1839. [Google Scholar] [CrossRef] [PubMed]
- Wiggins, B.S.; Spinler, S.; Wittkowsky, A.K.; Stringer, K.A. Bivalirudin: A direct thrombin inhibitor for percutaneous transluminal coronary angioplasty. Pharmacotherapy 2002, 22, 1007–1018. [Google Scholar] [CrossRef] [PubMed]
- Ussuf, K.K.; Laxmi, N.H.; Mitra, R. Proteinase inhibitors: Plant-derived genes of insecticidal protein for developing insect-resistant transgenic plants. Curr. Sci. India 2001, 80, 847–853. [Google Scholar]
- Schuler, T.H.; Poppy, G.M.; Kerry, B.R.; Denholm, I. Insect-resistant transgenic plants. Trends Biotechnol. 1998, 16, 168–175. [Google Scholar] [CrossRef]
- Ahmed, A.; Shamsi, A.; Bano, B. Deciphering the toxic effects of iprodione, a fungicide and malathion, an insecticide on thiol protease inhibitor isolated from yellow Indian mustard seeds. Environ. Toxicol. Phar. 2018, 61, 52–60. [Google Scholar] [CrossRef]
- Braga-Silva, L.A.; Santos, A.L.S. Aspartic Protease Inhibitors as Potential Anti-Candida albicans Drugs: Impacts on Fungal Biology, Virulence and Pathogenesis. Curr. Med. Chem. 2011, 18, 2401–2419. [Google Scholar] [CrossRef]
- Zhao, Y.R.; Xu, Y.H.; Jiang, H.S.; Xu, S.; Zhao, X.F.; Wang, J.X. Antibacterial activity of serine protease inhibitor 1 from kuruma shrimp Marsupenaeus japonicus. Dev. Comp. Immunol. 2014, 44, 261–269. [Google Scholar] [CrossRef]
- Jongsma, M.A.; Beekwilder, J. Co-Evolution of Insect Proteases and Plant Protease Inhibitors. Curr. Protein Pept. Sci. 2011, 12, 437–447. [Google Scholar] [CrossRef]
- Zhu, J.Y.; He, Y.X.; Yan, X.M.; Liu, L.; Guo, R.; Xia, X.B.; Cheng, D.J.; Mi, X.Z.; Samarina, L.; Liu, S.R.; et al. Duplication and transcriptional divergence of three Kunitz protease inhibitor genes that modulate insect and pathogen defenses in tea plant (Camellia sinensis). Hortic. Res. 2019, 6. [Google Scholar] [CrossRef]
- de Almeida Barros, B.; da Silva, W.G.; Moreira, M.A.; de Barros, E.G. In silico characterization and expression analysis of the multigene family encoding the Bowman-Birk protease inhibitor in soybean. Mol. Biol. Rep. 2012, 39, 327–334. [Google Scholar] [CrossRef]
- Odeny, D.A.; Stich, B.; Gebhardt, C. Physical organization of mixed protease inhibitor gene clusters, coordinated expression and association with resistance to late blight at the StKI locus on potato chromosome III. Plant Cell Environ. 2010, 33, 2149–2161. [Google Scholar] [CrossRef]
- Alvarez-Alfageme, F.; Maharramov, J.; Carrillo, L.; Vandenabeele, S.; Vercammen, D.; Van Breusegem, F.; Smagghe, G. Potential Use of a Serpin from Arabidopsis for Pest Control. PLoS ONE 2011, 6, e0020278. [Google Scholar] [CrossRef]
- Goulet, M.C.; Dallaire, C.; Vaillancourt, L.P.; Khalf, M.; Badri, A.M.; Preradov, A.; Duceppe, M.O.; Goulet, C.; Cloutier, C.; Michaud, D. Tailoring the specificity of a plant cystatin toward herbivorous insect digestive cysteine proteases by single mutations at positively selected amino acid sites. Plant Physiol. 2008, 146, 1010–1019. [Google Scholar] [CrossRef] [PubMed]
- Garcia, V.A.; Freire, M.; Novello, J.C.; Marangoni, S.; Macedo, M.L. Trypsin inhibitor from Poecilanthe parviflora seeds: Purification, characterization, and activity against pest proteases. Protein J. 2004, 23, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Manara, A.; Fasani, E.; Molesini, B.; DalCorso, G.; Pennisi, F.; Pandolfini, T.; Furini, A. The Tomato Metallocarboxypeptidase Inhibitor I, which Interacts with a Heavy Metal-Associated Isoprenylated Protein, Is Implicated in Plant Response to Cadmium. Molecules 2020, 25, 700. [Google Scholar] [CrossRef] [PubMed]
- Diez-Diaz, M.; Conejero, V.; Rodrigo, I.; Pearce, G.; Ryan, C.A. Isolation and characterization of wound-inducible carboxypeptidase inhibitor from tomato leaves. Phytochemistry 2004, 65, 1919–1924. [Google Scholar] [CrossRef]
- Guimaraes, L.C.; de Oliveira, C.F.; Marangoni, S.; de Oliveira, D.G.; Macedo, M.L. Purification and characterization of a Kunitz inhibitor from Poincianella pyramidalis with insecticide activity against the Mediterranean flour moth. Pestic. Biochem. Physiol. 2015, 118, 1–9. [Google Scholar] [CrossRef]
- Dunaevsky, Y.E.; Gladysheva, I.P.; Pavlukova, E.B.; Beliakova, G.A.; Gladyshev, D.P.; Papisova, A.I.; Larionova, N.I.; Belozersky, M.A. The anionic protease inhibitor BWI-1 from buckwheat seeds. Kinetic properties and possible biological role. Physiol. Plant. 1997, 101, 483–488. [Google Scholar] [CrossRef]
- Aguiar, J.M.; Franco, O.L.; Rigden, D.J.; Bloch, C., Jr.; Monteiro, A.C.; Flores, V.M.; Jacinto, T.; Xavier-Filho, J.; Oliveira, A.E.; Grossi-de-Sa, M.F.; et al. Molecular modeling and inhibitory activity of cowpea cystatin against bean bruchid pests. Proteins 2006, 63, 662–670. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, S.; Takano, T. Two cysteine proteinase inhibitors from Arabidopsis thaliana, AtCYSa and AtCYSb, increasing the salt, drought, oxidation and cold tolerance. Plant Mol. Biol. 2008, 68, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Maass, N.; Teffner, M.; Rosel, F.; Pawaresch, R.; Jonat, W.; Nagasaki, K.; Rudolph, P. Decline in the expression of the serine proteinase inhibitor maspin is associated with tumour progression in ductal carcinomas of the breast. J. Pathol. 2001, 195, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Cooley, J.; Takayama, T.K.; Shapiro, S.D.; Schechter, N.M.; Remold-O’Donnell, E. The serpin MNEI inhibits elastase-like and chymotrypsin-like serine proteases through efficient reactions at two active sites. Biochemistry 2001, 40, 15762–15770. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, A.; Babu, C.R. Purification and biochemical characterization of a serine proteinase inhibitor from Derris trifoliata Lour. seeds: Insight into structural and antimalarial features. Phytochemistry 2009, 70, 703–712. [Google Scholar] [CrossRef]
- Touil, T.; Ciric, B.; Ventura, E.; Shindler, K.S.; Gran, B.; Rostami, A. Bowman-Birk inhibitor suppresses autoimmune inflammation and neuronal loss in a mouse model of multiple sclerosis. J. Neurol. Sci. 2008, 271, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, A.R. The Bowman-Birk inhibitor from soybeans as an anticarcinogenic agent. Am. J. Clin. Nutr. 1998, 68, 1406s–1412s. [Google Scholar] [CrossRef] [PubMed]
- Bell-McGuinn, K.M.; Garfall, A.L.; Bogyo, M.; Hanahan, D.; Joyce, J.A. Inhibition of cysteine cathepsin protease activity enhances chemotherapy regimens by decreasing tumor growth and invasiveness in a mouse model of multistage cancer. Cancer Res. 2007, 67, 7378–7385. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.X.; Erskine, P.T.; Coker, A.R.; Wood, S.P.; Cooper, J.B. Structure of a Kunitz-type potato cathepsin D inhibitor. J. Struct. Biol. 2015, 192, 554–560. [Google Scholar] [CrossRef]
- Krishnamoorthi, R.; Gong, Y.X.; Richardson, M. A New-Protein Inhibitor of Trypsin and Activated Hageman-Factor from Pumpkin (Cucurbita-Maxima) Seeds. FEBS Lett. 1990, 273, 163–167. [Google Scholar] [CrossRef]
- Visetnan, S.; Donpudsa, S.; Supungul, P.; Tassanakajon, A.; Rimphanitchayakit, V. Domain 2 of a Kazal serine proteinase inhibitor SPIPm2 from Penaeus monodon possesses antiviral activity against WSSV. Fish Shellfish Immunol. 2014, 41, 526–530. [Google Scholar] [CrossRef]
- Lamarre, D.; Anderson, P.C.; Bailey, M.; Beaulieu, P.; Bolger, G.; Bonneau, P.; Bos, M.; Cameron, D.R.; Cartier, M.; Cordingley, M.G.; et al. An NS3 protease inhibitor with antiviral effects in humans infected with hepatitis C virus. Nature 2003, 426, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Hinrichsen, H.; Benhamou, Y.; Wedemeyer, H.; Reiser, M.; Sentjens, R.E.; Calleja, J.L.; Forns, X.; Erhardt, A.; Cronlein, J.; Chaves, R.L.; et al. Short-term antiviral efficacy of BILN 2061, a hepatitis C virus serine protease inhibitor, in hepatitis C genotype 1 patients. Gastroenterology 2004, 127, 1347–1355. [Google Scholar] [CrossRef] [PubMed]
- Okumura, Y.; Ogawa, K.; Uchiya, K.; Nikai, T. Isolation and characterization of a novel elastase inhibitor, AFLEI from Aspergillus flavus. Nihon Ishinkin Gakkai Zasshi 2006, 47, 219–224. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hibbetts, K.; Hines, B.; Williams, D. An overview of proteinase inhibitors. J. Vet. Intern. Med. 1999, 13, 302–308. [Google Scholar] [CrossRef] [PubMed]
- Abrahamson, M.; Mason, R.W.; Hansson, H.; Buttle, D.J.; Grubb, A.; Ohlsson, K. Human cystatin C. role of the N-terminal segment in the inhibition of human cysteine proteinases and in its inactivation by leucocyte elastase. Biochem. J. 1991, 273, 621–626. [Google Scholar] [CrossRef]
- Travis, J.; Bangalore, N. Biochemical mechanisms for disrupting the proteinase-proteinase inhibitor balance in tissues. Agents Actions Suppl. 1993, 42, 19–25. [Google Scholar] [CrossRef]
- Jongejan, F.; Uilenberg, G. The global importance of ticks. Parasitology 2004, 129, S3–S14. [Google Scholar] [CrossRef]
- Francischetti, I.M.B.; Sa-Nunes, A.; Mans, B.J.; Santos, I.M.; Ribeiro, J.M.C. The role of saliva in tick feeding. Front. Biosci. A J. Virtual Libr. 2009, 14, 2051–2088. [Google Scholar] [CrossRef]
- Mans, B.J. Evolution of Vertebrate Hemostatic and Inflammatory Control Mechanisms in Blood-Feeding Arthropods. J. Innate Immun. 2011, 3, 41–51. [Google Scholar] [CrossRef]
- Mans, B.J.; Neitz, A.W.H. Adaptation of ticks to a blood-feeding environment: Evolution from a functional perspective. Insect Biochem. Mol. Biol. 2004, 34, 1–17. [Google Scholar] [CrossRef]
- Zavasnik-Bergant, T.; Vidmar, R.; Sekirnik, A.; Fonovic, M.; Salat, J.; Grunclova, L.; Kopacek, P.; Turk, B. Salivary Tick Cystatin OmC2 Targets Lysosomal Cathepsins S and C in Human Dendritic Cells. Front. Cell. Infect. Microbiol. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Nuttall, P.A. Wonders of tick saliva. Ticks Tickborne Dis. 2019, 10, 470–481. [Google Scholar] [CrossRef] [PubMed]
- Parizi, L.F.; Ali, A.; Tirloni, L.; Oldiges, D.P.; Sabadin, G.A.; Coutinho, M.L.; Seixas, A.; Logullo, C.; Termignoni, C.; Vaz, I.D. Peptidase inhibitors in tick physiology. Med. Vet. Entomol. 2018, 32, 129–144. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Y.; de la Fuente, J.; Cote, M.; Galindo, R.C.; Moutailler, S.; Vayssier-Taussat, M.; Bonnet, S.I. IrSPI, a Tick Serine Protease Inhibitor Involved in Tick Feeding and Bartonella henselae Infection. PLoS Negl. Trop. Dis. 2014, 8, e2993. [Google Scholar] [CrossRef] [PubMed]
- Sojka, D.; Pytelkova, J.; Perner, J.; Horn, M.; Konvickova, J.; Schrenkova, J.; Mares, M.; Kopacek, P. Multienzyme degradation of host serum albumin in ticks. Ticks TickBorne Dis. 2016, 7, 604–613. [Google Scholar] [CrossRef] [PubMed]
- Estrela, A.; Seixas, A.; Termignoni, C. A cysteine endopeptidase from tick (Rhipicephalus (Boophilus) microplus) larvae with vitellin digestion activity. Comp. Biochem. Phys. B 2007, 148, 410–416. [Google Scholar] [CrossRef]
- Limo, M.K.; Voigt, W.P.; Tumbo-Oeri, A.G.; Njogu, R.M.; ole-MoiYoi, O.K. Purification and characterization of an anticoagulant from the salivary glands of the ixodid tick Rhipicephalus appendiculatus. Exp. Parasitol. 1991, 72, 418–429. [Google Scholar] [CrossRef]
- Katz, L.; Baltz, R.H. Natural product discovery: Past, present, and future. J. Ind. Microbiol. Biot. 2016, 43, 155–176. [Google Scholar] [CrossRef]
- Gileadi, O. Recombinant Protein Expression in E. coli: A Historical Perspective. Methods Mol. Biol. 2017, 1586, 3–10. [Google Scholar] [CrossRef]
- Baghban, R.; Farajnia, S.; Ghasemi, Y.; Mortazavi, M.; Zarghami, N.; Samadi, N. New Developments in Pichia pastoris Expression System, Review and Update. Curr. Pharm. Biotechnol. 2018, 19, 451–467. [Google Scholar] [CrossRef]
- Rozov, S.M.; Permyakova, N.V.; Deineko, E.V. Main Strategies of Plant Expression System Glycoengineering for Producing Humanized Recombinant Pharmaceutical Proteins. Biochem. Biokhimiia 2018, 83, 215–232. [Google Scholar] [CrossRef] [PubMed]
- Chmelar, J.; Kotal, J.; Karim, S.; Kopacek, P.; Francischetti, I.M.B.; Pedra, J.H.F.; Kotsyfakis, M. Sialomes and Mialomes: A Systems-Biology View of Tick Tissues and Tick-Host Interactions. Trends Parasitol. 2016, 32, 242–254. [Google Scholar] [CrossRef] [PubMed]
- Koh, C.Y.; Kini, R.M. Anticoagulants from hematophagous animals. Expert Rev. Hematol. 2008, 1, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Aounallah, H.; Bensaoud, C.; M’ghirbi, Y.; Faria, F.; Chmelar, J.; Kotsyfakis, M. Tick Salivary Compounds for Targeted Immunomodulatory Therapy. Front. Immunol. 2020, 11. [Google Scholar] [CrossRef]
- Ranasinghe, S.; McManus, D.P. Structure and function of invertebrate Kunitz serine protease inhibitors. Dev. Comp. Immunol. 2013, 39, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, J.M.C.; Alarcon-Chaidez, F.; Francischetti, I.M.B.; Mans, B.J.; Mather, T.N.; Valenzuela, J.G.; Wikel, S.K. An annotated catalog of salivary gland transcripts from Ixodes scapularis ticks. Insect Biochem. Mol. Biol. 2006, 36, 111–129. [Google Scholar] [CrossRef]
- Chmelar, J.; Calvo, E.; Pedra, J.H.F.; Francischetti, I.M.B.; Kotsyfakis, M. Tick salivary secretion as a source of antihemostatics. J. Proteom. 2012, 75, 3842–3854. [Google Scholar] [CrossRef]
- Decrem, Y.; Rath, G.; Blasioli, V.; Cauchie, P.; Robert, S.; Beaufays, J.; Frere, J.M.; Feron, O.; Dogne, J.M.; Dessy, C.; et al. Ir-CPI, a coagulation contact phase inhibitor from the tick Ixodes ricinus, inhibits thrombus formation without impairing hemostasis. J. Exp. Med. 2009, 206, 2381–2395. [Google Scholar] [CrossRef]
- Neeper, M.P.; Waxman, L.; Smith, D.E.; Schulman, C.A.; Sardana, M.; Ellis, R.W.; Schaffer, L.W.; Siegl, P.K.S.; Vlasuk, G.P. Characterization of Recombinant Tick Anticoagulant Peptide—A Highly Selective Inhibitor of Blood-Coagulation Factor-Xa. J. Biol. Chem. 1990, 265, 17746–17752. [Google Scholar] [CrossRef]
- Pireaux, V.; Tassignon, J.; Demoulin, S.; Derochette, S.; Borenstein, N.; Ente, A.; Fiette, L.; Douxfils, J.; Lancellotti, P.; Guyaux, M.; et al. Anticoagulation With an Inhibitor of Factors XIa and XIIa During Cardiopulmonary Bypass. J. Am. Coll. Cardiol. 2019, 74, 2178–2189. [Google Scholar] [CrossRef]
- Schaffer, L.W.; Davidson, J.T.; Vlasuk, G.P.; Siegl, P.K.S. Antithrombotic Efficacy of Recombinant Tick Anticoagulant Peptide—A Potent Inhibitor of Coagulation Factor-Xa in a Primate Model of Arterial Thrombosis. Circulation 1991, 84, 1741–1748. [Google Scholar] [CrossRef] [PubMed]
- Onishi, A.; St Ange, K.; Dordick, J.S.; Linhardt, R.J. Heparin and anticoagulation. Front. Biosci. Landmrk. 2016, 21, 1372–1392. [Google Scholar] [CrossRef]
- Stoll, P.; Bassler, N.; Hagemeyer, C.E.; Eisenhardt, S.U.; Chen, Y.C.; Schmidt, R.; Schwarz, M.; Ahrens, I.; Katagiri, Y.; Pannen, B.; et al. Targeting ligand-induced binding sites on GPIIb/IIIa via single-chain antibody allows effective anticoagulation without bleeding time prolongation. Arterioscl. Throm. Vas. 2007, 27, 1206–1212. [Google Scholar] [CrossRef] [PubMed]
- Karczewski, J.; Connolly, T.M. The interaction of disagregin with the platelet fibrinogen receptor, glycoprotein IIb-IIIa. Biochem. Biophys. Res. Commun. 1997, 241, 744–748. [Google Scholar] [CrossRef] [PubMed]
- Barboza, T.; Gomes, T.; da Costa Medeiros, P.; Ramos, I.P.; Francischetti, I.; Monteiro, R.Q.; Gutfilen, B.; de Souza, S.A.L. Development of 131I-ixolaris as a theranostic agent: Metastatic melanoma preclinical studies. Clin. Exp. Metastasis. 2020, 37, 489–497. [Google Scholar] [CrossRef]
- Boufleur, P.; Sciani, J.M.; Goldfeder, M.; Faria, F.; Branco, V.; Chudzinski-Tavassi, A.M. Biodistribution and Pharmacokinetics of Amblyomin-X, a Novel Antitumour Protein Drug in Healthy Mice. Eur. J. Drug Metab. Pharmacokinet. 2019, 44, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Francischetti, I.M.B.; Valenzuela, J.G.; Andersen, J.F.; Mather, T.N.; Ribeiro, J.M.C. Ixolaris, a novel recombinant tissue factor pathway inhibitor (TFPI) from the salivary gland of the tick, Ixodes scapularis: Identification of factor X and factor Xa as scaffolds for the inhibition of factor VIIa/tissue factor complex. Blood 2002, 99, 3602–3612. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, R.Q.; Rezaie, A.R.; Ribeiro, J.M.C.; Francischetti, I.M.B. Ixolaris: A factor Xa heparin-binding exosite inhibitor. Biochem. J. 2005, 387, 871–877. [Google Scholar] [CrossRef]
- Monteiro, R.Q.; Rezaie, A.R.; Bae, J.-S.; Calvo, E.; Andersen, J.F.; Francischetti, I.M.B. Ixolaris binding to factor X reveals a precursor state of factor Xa heparin-binding exosite. Protein Sci. 2008, 17, 146–153. [Google Scholar] [CrossRef]
- De Paula, V.S.; Sgourakis, N.G.; Francischetti, I.M.B.; Almeida, F.C.L.; Monteiro, R.Q.; Valente, A.P. NMR structure determination of Ixolaris and factor X(a) interaction reveals a noncanonical mechanism of Kunitz inhibition. Blood 2019, 134, 699–708. [Google Scholar] [CrossRef]
- Monteiro, R.Q. Targeting exosites on blood coagulation proteases. An Acad. Bras. Cienc. 2005, 77, 275–280. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nazareth, R.A.; Tomaz, L.S.; Ortiz-Costa, S.; Atella, G.C.; Ribeiro, J.M.C.; Francischetti, I.M.B.; Monteiro, R.Q. Antithrombotic properties of Ixolaris, a potent inhibitor of the extrinsic pathway of the coagulation cascade. Thromb. Haemost. 2006, 96, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Chudzinski-Tavassi, A.M.; Morais, K.L.P.; Pacheco, M.T.F.; Pasqualoto, K.F.M.; de Souza, J.G. Tick salivary gland as potential natural source for the discovery of promising antitumor drug candidates. Biomed. Pharmacother. 2016, 77, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Carneiro-Lobo, T.C.; Konig, S.; Machado, D.E.; Nasciutti, L.E.; Forni, M.F.; Francischetti, I.M.B.; Sogayar, M.C.; Monteiro, R.Q. Ixolaris, a tissue factor inhibitor, blocks primary tumor growth and angiogenesis in a glioblastoma model. J. Thromb. Haemost. 2009, 7, 1855–1864. [Google Scholar] [CrossRef] [PubMed]
- Carneiro-Lobo, T.C.; Schaffner, F.; Disse, J.; Ostergaard, H.; Francischetti, I.M.B.; Monteiro, R.Q.; Ruf, W. The tick-derived inhibitor Ixolaris prevents tissue factor signaling on tumor cells. J. Thromb. Haemost. 2012, 10, 1849–1858. [Google Scholar] [CrossRef]
- Batista, I.F.C.; Ramos, O.H.P.; Ventura, J.S.; Junqueira-de-Azevedo, I.L.M.; Ho, P.L.; Chudzinski-Tavassi, A.M. A new Factor Xa inhibitor from Amblyomma cajennense with a unique domain composition. Arch. Biochem. Biophys. 2010, 493, 151–156. [Google Scholar] [CrossRef]
- Branco, V.G.; Iqbal, A.; Alvarez-Flores, M.P.; Sciani, J.M.; de Andrade, S.A.; Iwai, L.K.; Serrano, S.M.T.; Chudzinski-Tavassi, A.M. Amblyomin-X having a Kunitz-type homologous domain, is a noncompetitive inhibitor of FXa and induces anticoagulation in vitro and in vivo. Biochim. Biophys. Acta 2016, 1864, 1428–1435. [Google Scholar] [CrossRef]
- Akagi, E.M.; Júnior, P.L.d.S.; Simons, S.M.; Bellini, M.H.; Barreto, S.A.; Chudzinski-Tavassi, A.M. Pro-apoptotic effects of Amblyomin-X in murine renal cell carcinoma “in vitro”. Biomed. Pharmacother. 2012, 66, 64–69. [Google Scholar] [CrossRef]
- Maria, D.A.; de Souza, J.G.; Morais, K.L.P.; Berra, C.M.; Zampolli, H.d.C.; Demasi, M.; Simons, S.M.; de Freitas Saito, R.; Chammas, R.; Chudzinski-Tavassi, A.M. A novel proteasome inhibitor acting in mitochondrial dysfunction, ER stress and ROS production. Invest New Drugs 2013, 31, 493–505. [Google Scholar] [CrossRef]
- Schmidt, M.C.B.; Morais, K.L.P.; Almeida, M.E.S.d.; Iqbal, A.; Goldfeder, M.B.; Chudzinski-Tavassi, A.M. Amblyomin-X, a recombinant Kunitz-type inhibitor, regulates cell adhesion and migration of human tumor cells. Cell. Adh. Migr. 2020, 14, 129–138. [Google Scholar] [CrossRef]
- Drewes, C.C.; Dias, R.Y.S.; Hebeda, C.B.; Simons, S.M.; Barreto, S.A.; Ferreira, J.M.; Chudzinski-Tavassi, A.M.; Farsky, S.H.P. Actions of the Kunitz-type serine protease inhibitor Amblyomin-X on VEGF-A-induced angiogenesis. Toxicon 2012, 60, 333–340. [Google Scholar] [CrossRef]
- Drewes, C.C.; Dias, R.Y.; Branco, V.G.; Cavalcante, M.F.; Souza, J.G.; Abdalla, D.S.P.; Chudzinski-Tavassi, A.M.; Farsky, S.H.P. Post-transcriptional control of Amblyomin-X on secretion of vascular endothelial growth factor and expression of adhesion molecules in endothelial cells. Toxicon 2015, 101, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Maria, D.A.; Will, S.E.A.L.; Bosch, R.V.; Souza, J.G.; Sciani, J.M.; Goldfeder, M.B.; Rondon, G.G.; Chudzinski-Tavassi, A.M. Preclinical evaluation of Amblyomin-X, a Kunitz-type protease inhibitor with antitumor activity. Toxicol. Rep. 2019, 6, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, F.; Iqbal, A.; de Lima Will, S.E.A.; Bosch, R.V.; DeOcesano-Pereira, C.; Goldfeder, M.B.; Chammas, R.; Trufen, C.E.M.; Morais, K.L.P.; de Souza, J.G.; et al. Modulation of Stress and Immune Response by AMBLYOMIN-X Results in Tumor Cell Death in a Horse Melanoma Model. Available online: https://pubmed.ncbi.nlm.nih.gov/32286411/ (accessed on 15 January 2021).
- Blisnick, A.A.; Šimo, L.; Grillon, C.; Fasani, F.; Brûlé, S.; Le Bonniec, B.; Prina, E.; Marsot, M.; Relmy, A.; Blaise-Boisseau, S.; et al. The Immunomodulatory Effect of IrSPI, a Tick Salivary Gland Serine Protease Inhibitor Involved in Ixodes ricinus Tick Feeding. Vaccines 2019, 7, 148. [Google Scholar] [CrossRef]
- Duran, A.F.A.; Neves, L.d.P.; da Silva, F.R.S.; Machado, G.C.; Ferreira, G.C.; Lourenço, J.D.; Tanaka, A.S.; Martins, M.d.A.; Lopes, F.D.T.Q.S.; Sasaki, S.D. rBmTI-6 attenuates pathophysiological and inflammatory parameters of induced emphysema in mice. Int. J. Biol. Macromol. 2018, 111, 1214–1221. [Google Scholar] [CrossRef] [PubMed]
- Paesen, G.C.; Siebold, C.; Harlos, K.; Peacey, M.F.; Nuttall, P.A.; Stuart, D.I. A tick protein with a modified Kunitz fold inhibits human tryptase. J. Mol. Biol. 2007, 368, 1172–1186. [Google Scholar] [CrossRef] [PubMed]
- Sommerhoff, C.P.; Schaschke, N. Mast cell tryptase beta as a target in allergic inflammation: An evolving story. Curr. Pharm. Des. 2007, 13, 313–332. [Google Scholar] [CrossRef] [PubMed]
- Valdés, J.J.; Schwarz, A.; Cabeza de Vaca, I.; Calvo, E.; Pedra, J.H.F.; Guallar, V.; Kotsyfakis, M. Tryptogalinin is a tick Kunitz serine protease inhibitor with a unique intrinsic disorder. PLoS ONE 2013, 8, e62562. [Google Scholar] [CrossRef]
- Castaneda, O.; Harvey, A.L. Discovery and characterization of cnidarian peptide toxins that affect neuronal potassium ion channels. Toxicon 2009, 54, 1119–1124. [Google Scholar] [CrossRef]
- Lucchesi, K.J.; Moczydlowski, E. On the Interaction of Bovine Pancreatic Trypsin-Inhibitor with Maxi Ca2+-Activated K+ Channels—A Model System for Analysis of Peptide-Induced Subconductance States. J. Gen. Physiol. 1991, 97, 1295–1319. [Google Scholar] [CrossRef]
- Paesen, G.C.; Siebold, C.; Dallas, M.L.; Peers, C.; Harlos, K.; Nuttall, P.A.; Nunn, M.A.; Stuart, D.I.; Esnouf, R.M. An Ion-channel Modulator from the Saliva of the Brown Ear Tick has a Highly Modified Kunitz/BPTI Structure. J. Mol. Biol. 2009, 389, 734–747. [Google Scholar] [CrossRef]
- Lucas, A.; Yaron, J.R.; Zhang, L.Q.; Ambadapadi, S. Overview of Serpins and Their Roles in Biological Systems. Methods Protoc. 2018, 1826, 1–7. [Google Scholar] [CrossRef]
- Kim, T.K.; Tirloni, L.; Berger, M.; Diedrich, J.K.; Yates, I.I.I.; Termignoni, C.; Vaz, I.D.; Mulenga, A. Amblyomma americanum serpin 41 (AAS41) inhibits inflammation by targeting chymase and chymotrypsin. Int. J. Biol. Macromol. 2020, 156, 1007–1021. [Google Scholar] [CrossRef] [PubMed]
- Tirloni, L.; Kim, T.K.; Berger, M.; Termignoni, C.; Vaz, I.D.; Mulenga, A. Amblyomma americanum serpin 27 (AAS27) is a tick salivary anti-inflammatory protein secreted into the host during feeding. PLoS Negl. Trop. Dis. 2019, 13, e7660. [Google Scholar] [CrossRef] [PubMed]
- Mulenga, A.; Kim, T.; Ibelli, A.M.G. Amblyomma americanum tick saliva serine protease inhibitor 6 is a cross-class inhibitor of serine proteases and papain-like cysteine proteases that delays plasma clotting and inhibits platelet aggregation. Insect Mol. Biol. 2013, 22, 306–319. [Google Scholar] [CrossRef] [PubMed]
- Ibelli, A.M.G.; Kim, T.K.; Hill, C.C.; Lewis, L.A.; Bakshi, M.; Miller, S.; Porter, L.; Mulenga, A. A blood meal-induced Ixodes scapularis tick saliva serpin inhibits trypsin and thrombin, and interferes with platelet aggregation and blood clotting. Int. J. Parasitol. 2014, 44, 369–379. [Google Scholar] [CrossRef]
- Tirloni, L.; Kim, T.K.; Coutinho, M.L.; Ali, A.; Seixas, A.; Termignoni, C.; Mulenga, A.; Vaz, I.D. The putative role of Rhipicephalus microplus salivary serpins in the tick-host relationship. Insect Biochem. Mol. Biol. 2016, 71, 12–28. [Google Scholar] [CrossRef]
- Radulovic, Z.M.; Mulenga, A. Heparan sulfate/heparin glycosaminoglycan binding alters inhibitory profile and enhances anticoagulant function of conserved Amblyomma americanum tick saliva serpin 19. Insect Biochem. Mol. Biol. 2017, 80, 1–10. [Google Scholar] [CrossRef][Green Version]
- Kim, T.K.; Tirloni, L.; Radulovic, Z.; Lewis, L.; Bakshi, M.; Hill, C.; Vaz, I.D.; Logullo, C.; Termignoni, C.; Mulenga, A. Conserved Amblyomma americanum tick Serpin19, an inhibitor of blood clotting factors Xa and XIa, trypsin and plasmin, has anti-haemostatic functions. Int. J. Parasitol. 2015, 45, 613–627. [Google Scholar] [CrossRef]
- Sanrattana, W.; Maas, C.; de Maat, S. SERPINs-From Trap to Treatment. Front. Med. 2019, 6. [Google Scholar] [CrossRef]
- Wang, F.Q.; Song, Z.Y.; Chen, J.; Wu, Q.H.; Zhou, X.; Ni, X.H.; Dai, J.F. The immunosuppressive functions of two novel tick serpins, HlSerpin-a and HlSerpin-b, from Haemaphysalis longicornis. Immunology 2020, 159, 109–120. [Google Scholar] [CrossRef]
- Xu, Z.M.; Yan, Y.J.; Zhang, H.S.; Cao, J.; Zhou, Y.Z.; Xu, Q.M.; Zhou, J.L. A serpin from the tick Rhipicephalus haemaphysaloides: Involvement in vitellogenesis. Vet. Parasitol. 2020, 279. [Google Scholar] [CrossRef] [PubMed]
- Jittapalapong, S.; Kaewhom, P.; Pumhom, P.; Canales, M.; de la Fuente, J.; Stich, R.W. Immunization of rabbits with recombinant serine protease inhibitor reduces the performance of adult female Rhipicephalus microplus. Transbound. Emerg. Dis. 2010, 57, 103–106. [Google Scholar] [CrossRef] [PubMed]
- Prevot, P.P.; Couvreur, B.; Denis, V.; Brossard, A.; Vanhamme, L.; Godfroid, E. Protective immunity against Ixodes ricinus induced by a salivary serpin. Vaccine 2007, 25, 3284–3292. [Google Scholar] [CrossRef] [PubMed]
- Lucas, A.; Yaron, J.R.; Zhang, L.Q.; Macaulay, C.; McFadden, G. Serpins: Development for Therapeutic Applications. Serpins Methods Protoc. 2018, 1826, 255–265. [Google Scholar] [CrossRef]
- Leboulle, G.; Crippa, M.; Decrem, Y.; Mejri, N.; Brossard, M.; Bollen, A.; Godfroid, E. Characterization of a novel salivary immunosuppressive protein from Ixodes ricinus ticks. J. Biol. Chem. 2002, 277, 10083–10089. [Google Scholar] [CrossRef]
- Prevot, P.P.; Adam, B.; Boudjeltia, K.Z.; Brossard, M.; Lins, L.; Cauchie, P.; Brasseur, R.; Vanhaeverbeek, M.; Vanhamme, L.; Godfroid, E. Anti-hemostatic effects of a serpin from the saliva of the tick Ixodes ricinus. J. Biol. Chem. 2006, 281, 26361–26369. [Google Scholar] [CrossRef]
- Prevot, P.P.; Beschin, A.; Lins, L.; Beaufays, J.; Grosjean, A.; Bruys, L.; Adam, B.; Brossard, M.; Brasseur, R.; Boudjeltia, K.Z.; et al. Exosites mediate the anti-inflammatory effects of a multifunctional serpin from the saliva of the tick Ixodes ricinus. Febs. J. 2009, 276, 3235–3246. [Google Scholar] [CrossRef]
- Chmelar, J.; Oliveira, C.J.; Rezacova, P.; Francischetti, I.M.B.; Kovarova, Z.; Pejler, G.; Kopacek, P.; Ribeiro, J.M.C.; Mares, M.; Kopecky, J.; et al. A tick salivary protein targets cathepsin G and chymase and inhibits host inflammation and platelet aggregation. Blood 2011, 117, 736–744. [Google Scholar] [CrossRef]
- Palenkova, J.; Lieskovska, J.; Langhansova, H.; Kotsyfakis, M.; Chmelar, J.; Kopecky, J. Ixodes ricinus Salivary Serpin IRS-2 Affects Th17 Differentiation via Inhibition of the Interleukin-6/STAT-3 Signaling Pathway. Infect. Immun. 2015, 83, 1949–1956. [Google Scholar] [CrossRef]
- Fogaca, A.C.; Almeida, I.C.; Eberlin, M.N.; Tanaka, A.S.; Bulet, P.; Daffre, S. Ixodidin, a novel antimicrobial peptide from the hemocytes of the cattle tick Boophilus microplus with inhibitory activity against serine proteinases. Peptides 2006, 27, 667–674. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, A.; Goldfeder, M.B.; Marques-Porto, R.; Asif, H.; Souza, J.G.; Faria, F.; Chudzinski-Tavassi, A.M. Revisiting antithrombotic therapeutics; sculptin, a novel specific, competitive, reversible, scissile and tight binding inhibitor of thrombin. Sci. Rep. 2017, 7, 1431. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.J.; Ansell, J.E. Direct thrombin inhibitors. Br. J. Clin. Pharmacol. 2011, 72, 581–592. [Google Scholar] [CrossRef]
- Schwarz, A.; Valdes, J.J.; Kotsyfakis, M. The role of cystatins in tick physiology and blood feeding. Ticks Tickborne Dis. 2012, 3, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Kotsyfakis, M.; Horka, H.; Salat, J.; Andersen, J.F. The crystal structures of two salivary cystatins from the tick Ixodes scapularis and the effect of these inhibitors on the establishment of Borrelia burgdorferi infection in a murine model. Mol. Microbiol. 2010, 77, 456–470. [Google Scholar] [CrossRef] [PubMed]
- Kotsyfakis, M.; Sa-Nunes, A.; Francischetti, I.M.B.; Mather, T.N.; Andersen, J.F.; Ribeiro, J.M.C. Antiinflammatory and immunosuppressive activity of sialostatin L, a salivary cystatin from the tick Ixodes scapularis. J. Biol. Chem. 2006, 281, 26298–26307. [Google Scholar] [CrossRef] [PubMed]
- Sa-Nunes, A.; Bafica, A.; Lucas, D.A.; Conrads, T.P.; Veenstra, T.D.; Andersen, J.F.; Mather, T.N.; Ribeiro, J.M.C.; Francischetti, I.M.B. Prostaglandin E-2 is a major inhibitor of dendritic cell maturation and function in Ixodes scapularis saliva. J. Immunol. 2007, 179, 1497–1505. [Google Scholar] [CrossRef]
- Horka, H.; Staudt, V.; Klein, M.; Taube, C.; Reuter, S.; Dehzad, N.; Andersen, J.F.; Kopecky, J.; Schild, H.; Kotsyfakis, M.; et al. The Tick Salivary Protein Sialostatin L Inhibits the Th9-Derived Production of the Asthma-Promoting Cytokine IL-9 and Is Effective in the Prevention of Experimental Asthma. J. Immunol. 2012, 188, 2669–2676. [Google Scholar] [CrossRef]
- Klein, M.; Bruhl, T.J.; Staudt, V.; Reuter, S.; Grebe, N.; Gerlitzki, B.; Hoffmann, M.; Bohn, T.; Ulges, A.; Stergiou, N.; et al. Tick Salivary Sialostatin L Represses the Initiation of Immune Responses by Targeting IRF4-Dependent Transcription in Murine Mast Cells. J. Immunol. 2015, 195, 621–631. [Google Scholar] [CrossRef]
- Sa-Nunes, A.; Bafica, A.; Antonelli, L.R.; Choi, E.Y.; Francischetti, I.M.B.; Andersen, J.F.; Shi, G.P.; Chavakis, T.; Ribeiro, J.M.; Kotsyfakis, M. The Immunomodulatory Action of Sialostatin L on Dendritic Cells Reveals Its Potential to Interfere with Autoimmunity. J. Immunol. 2009, 182, 7422–7429. [Google Scholar] [CrossRef]
- Chen, G.; Wang, X.W.; Severo, M.S.; Sakhon, O.S.; Sohail, M.; Brown, L.J.; Sircar, M.; Snyder, G.A.; Sundberg, E.J.; Ulland, T.K.; et al. The Tick Salivary Protein Sialostatin L2 Inhibits Caspase-1-Mediated Inflammation during Anaplasma phagocytophilum Infection. Infect. Immun. 2014, 82, 2553–2564. [Google Scholar] [CrossRef] [PubMed]
- Lieskovska, J.; Palenikova, J.; Sirmarova, J.; Elsterova, J.; Kotsyfakis, M.; Chagas, A.C.; Calvo, E.; Ruzek, D.; Kopecky, J. Tick salivary cystatin sialostatin L2 suppresses IFN responses in mouse dendritic cells. Parasite Immunol. 2015, 37, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Wang, F.Q.; Pan, W.; Wu, Q.H.; Wang, J.W.; Dai, J.F. An Immunosuppressive Tick Salivary Gland Protein DsCystatin Interferes With Toll-Like Receptor Signaling by Downregulating TRAF6. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Kotal, J.; Stergiou, N.; Busa, M.; Chlastakova, A.; Berankova, Z.; Rezacova, P.; Langhansova, H.; Schwarz, A.; Calvo, E.; Kopecky, J.; et al. The structure and function of Iristatin, a novel immunosuppressive tick salivary cystatin. Cell. Mol. Life Sci. 2019, 76, 2003–2013. [Google Scholar] [CrossRef] [PubMed]

| PI | Type | Source | Application/Role | Reference |
|---|---|---|---|---|
| SlCYS8 | Cysteine protease inhibitor | Solanum lycopersicum | Inhibition of herbivorous insects | [74] |
| PPTI | Kunitz-type inhibitor | Poecilanthe parviflora seeds | Pesticide | [75] |
| TCMP-1 | Metalloprotease inhibitor | Tomato | Protect plants from cadmium accumulation | [76] |
| CPI | Metalloprotease inhibitor | Tomato leaves | Protect plants against herbivores attacks | [77] |
| PpyTI | Kunitz-type inhibitor | Poincianella pyramidalis | Insecticide | [78] |
| BWI-1 | Serine protease inhibitor | Buckwheat seeds | Suppression of filamentous fungi growth | [79] |
| Cowpea cystatin | Cysteine protease inhibitor | Cowpea seeds | Pesticide | [80] |
| AtCYSa/AtCYSb | Cysteine protease inhibitor | Arabidopsis thaliana | Improve plants tolerance to various stresses | [81] |
| Maspin | Serine protease inhibitor | Humans | Tumor suppressor | [82] |
| MNEI | Serpin | Humans | Regulation of extravascular sites inflammation | [83] |
| DtTC | Serine protease inhibitor | Derris trifoliata | Antimalarial agent | [84] |
| BBI | Bowman–Birk inhibitor | Soybean | Suppresses autoimmune encephalomyelitis | [85] |
| BBI | Bowman–Birk inhibitor | Soybean | Anticarcinogenic agent | [86] |
| JPM-OEt | Cysteine protease inhibitor | - | Inhibition of tumor growth | [87] |
| BBI | Bowman–Birk inhibitor | Soybean, pea, lentil, and chickpea | Colorectal cancer prevention | [61] |
| PDI | Kunitz-type inhibitor | Potato | Trypsin and cathepsin D inhibitor | [88] |
| CMTI-V | Serine protease inhibitor | Pumpkin seeds | Trypsin inhibition | [89] |
| SPIPm2 | Kazal-type inhibitor | Shrimp (Penaeus monodon) | Immune response against white spot syndrome virus | [90] |
| BILN 2061 | Serine protease inhibitor | - | Antiviral effect against hepatitis C virus | [91,92] |
| AFLEI | Serine protease inhibitor | Aspergillus flavus | Elastase inhibitor | [93] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jmel, M.A.; Aounallah, H.; Bensaoud, C.; Mekki, I.; Chmelař, J.; Faria, F.; M’ghirbi, Y.; Kotsyfakis, M. Insights into the Role of Tick Salivary Protease Inhibitors during Ectoparasite–Host Crosstalk. Int. J. Mol. Sci. 2021, 22, 892. https://doi.org/10.3390/ijms22020892
Jmel MA, Aounallah H, Bensaoud C, Mekki I, Chmelař J, Faria F, M’ghirbi Y, Kotsyfakis M. Insights into the Role of Tick Salivary Protease Inhibitors during Ectoparasite–Host Crosstalk. International Journal of Molecular Sciences. 2021; 22(2):892. https://doi.org/10.3390/ijms22020892
Chicago/Turabian StyleJmel, Mohamed Amine, Hajer Aounallah, Chaima Bensaoud, Imen Mekki, Jindřich Chmelař, Fernanda Faria, Youmna M’ghirbi, and Michalis Kotsyfakis. 2021. "Insights into the Role of Tick Salivary Protease Inhibitors during Ectoparasite–Host Crosstalk" International Journal of Molecular Sciences 22, no. 2: 892. https://doi.org/10.3390/ijms22020892
APA StyleJmel, M. A., Aounallah, H., Bensaoud, C., Mekki, I., Chmelař, J., Faria, F., M’ghirbi, Y., & Kotsyfakis, M. (2021). Insights into the Role of Tick Salivary Protease Inhibitors during Ectoparasite–Host Crosstalk. International Journal of Molecular Sciences, 22(2), 892. https://doi.org/10.3390/ijms22020892







