Cannabis sativa L. as a Natural Drug Meeting the Criteria of a Multitarget Approach to Treatment
Abstract
1. Introduction
2. Endocannabinoid System
3. Chemical Composition
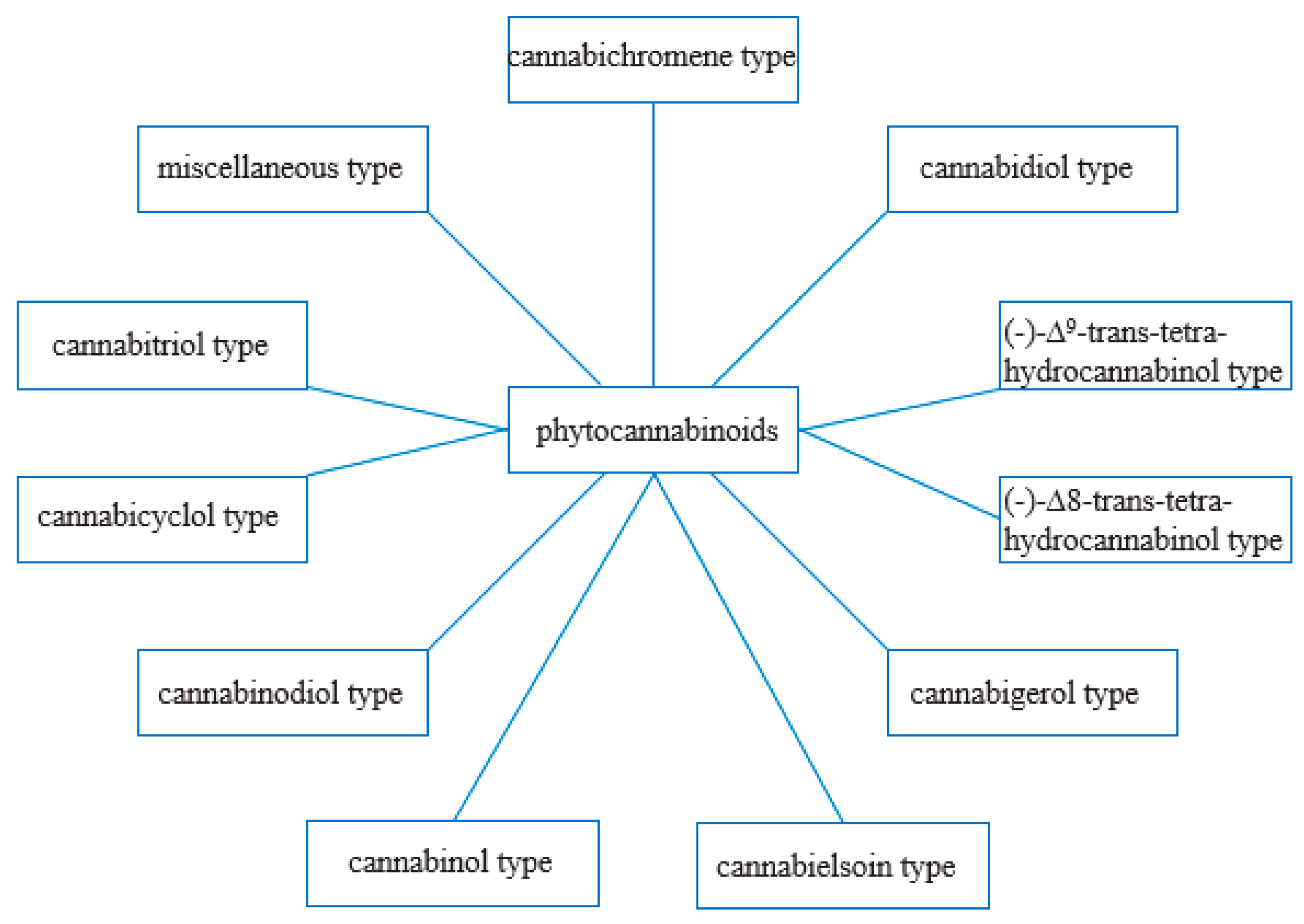
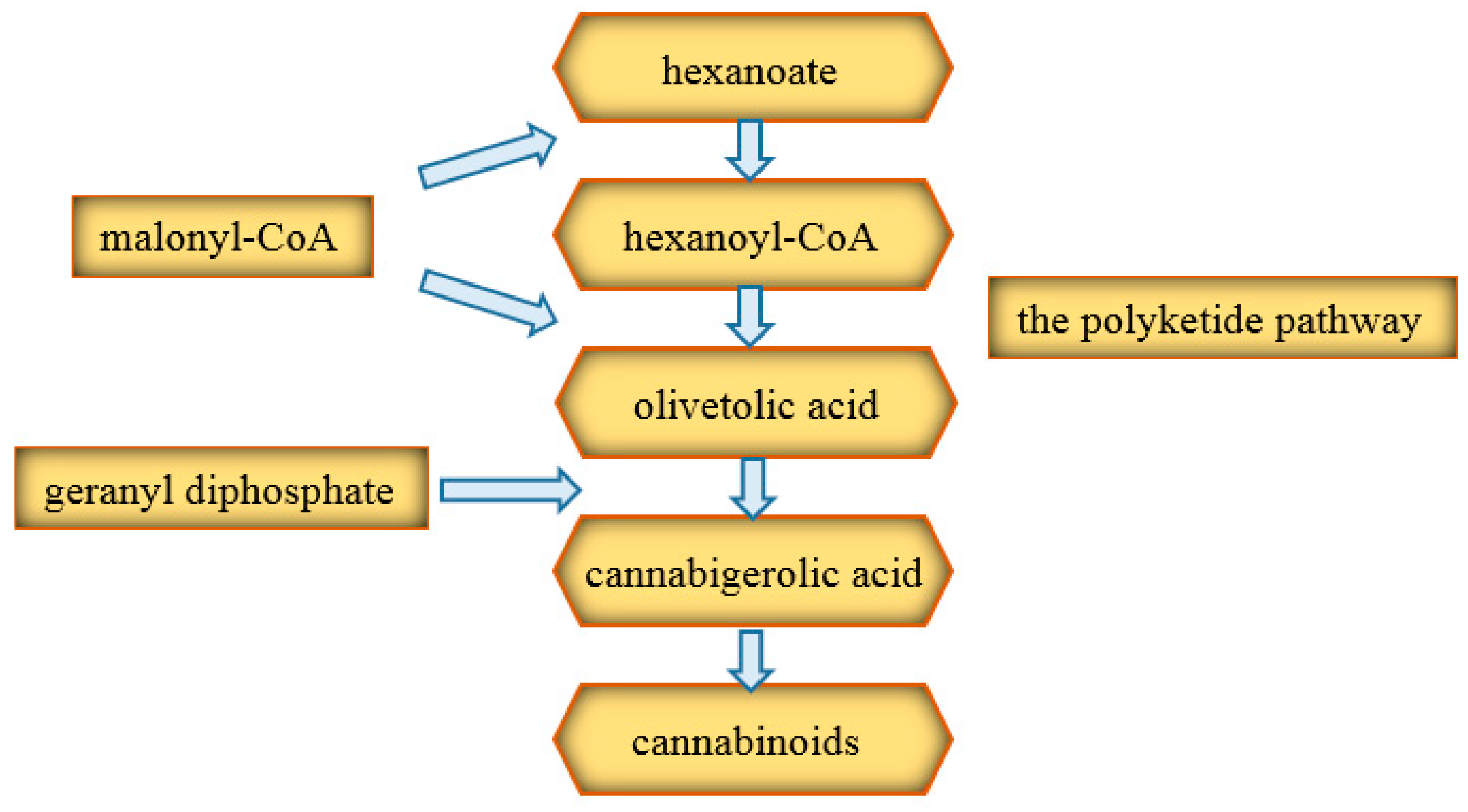
3.1. Phytocannabinoids
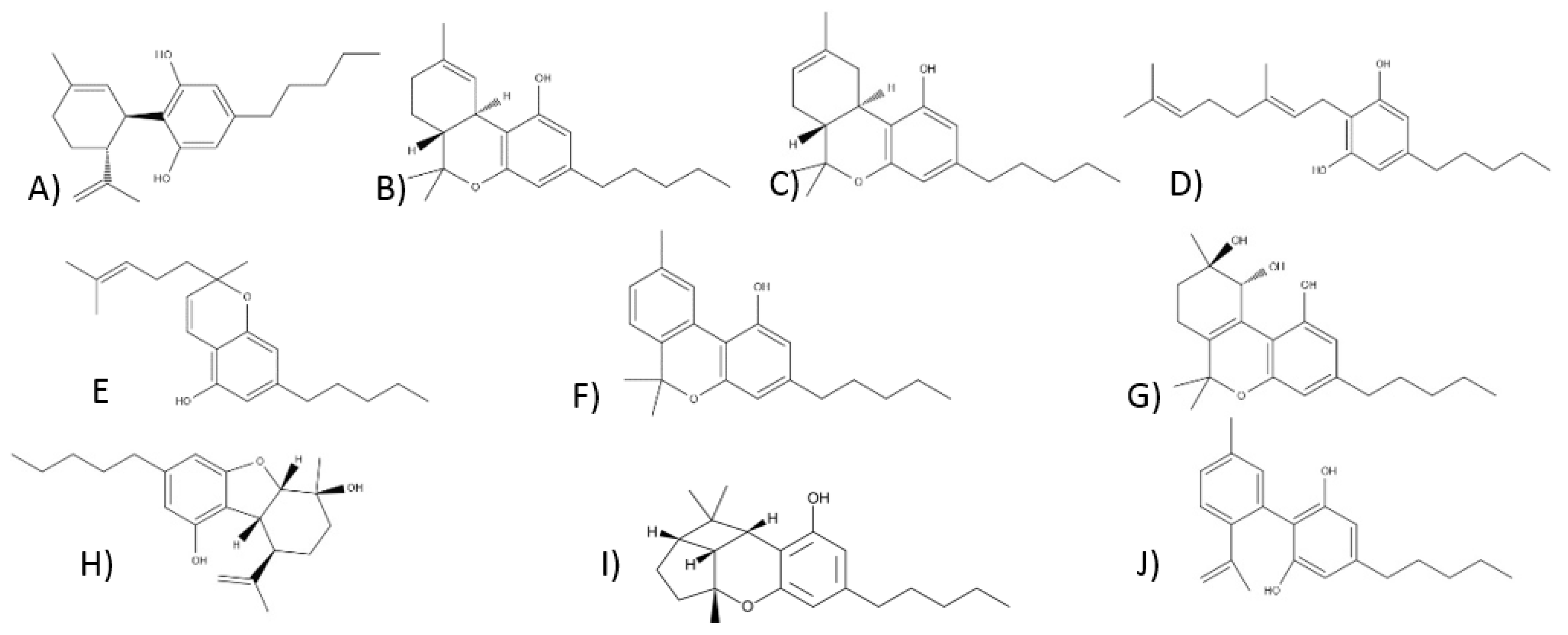
3.1.1. Cannabidiol-Type Compounds
3.1.2. Compounds of the Δ9-Tetrahydrocannabinol Type
3.1.3. Compounds of the Δ8-Tetrahydrocannabinol Type
3.1.4. Compounds of the Cannabigerol Type
3.1.5. Compounds of the Cannabichrome Type
3.1.6. Compounds of the Cannabinol Type
3.1.7. Compounds of the of Cannabitriol Type
3.1.8. Compounds of the Cannabielsoin Type
3.1.9. Compounds of the Cannabicyclol Type
3.1.10. Compounds of the Cannabinodiol Type
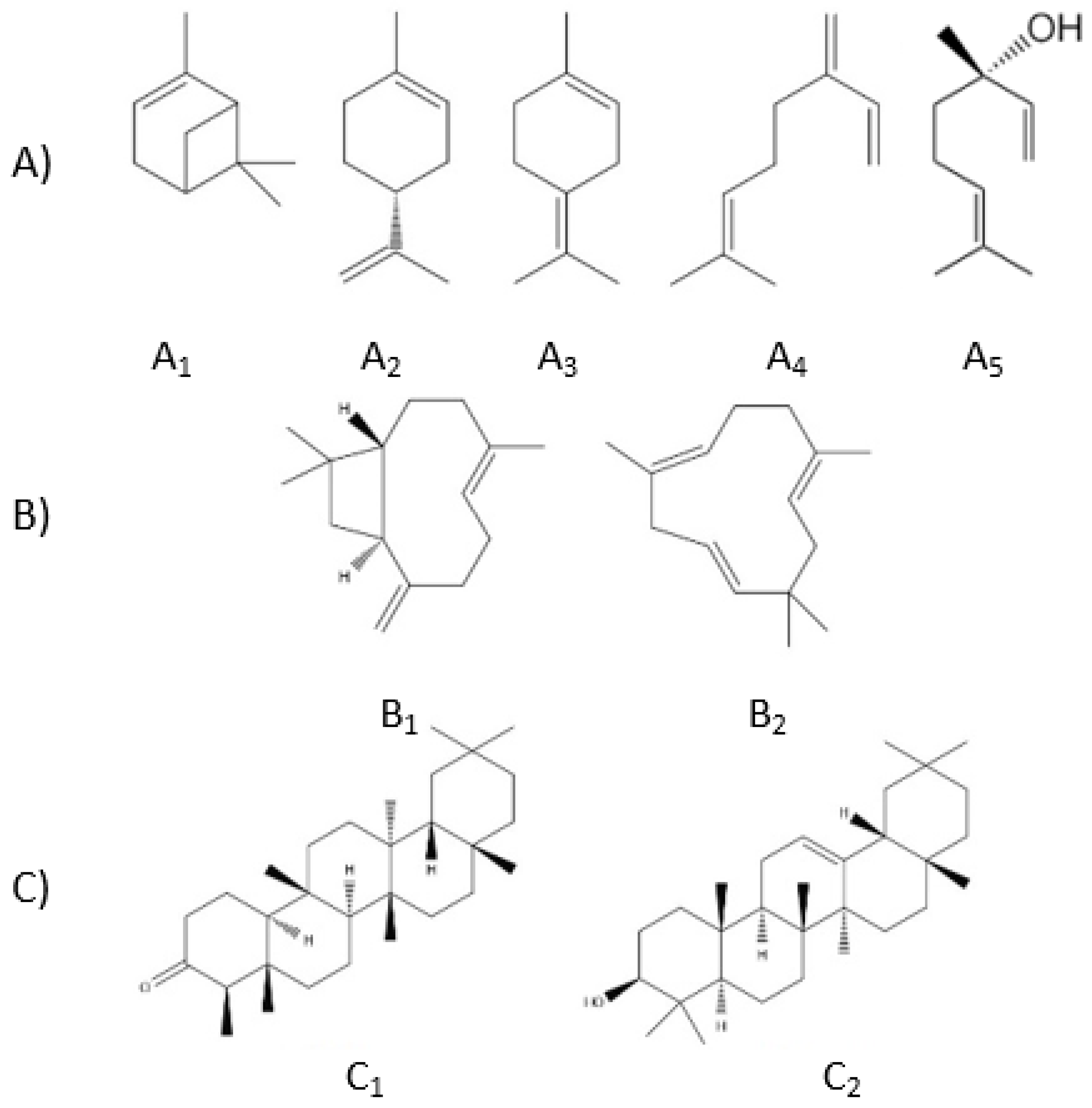
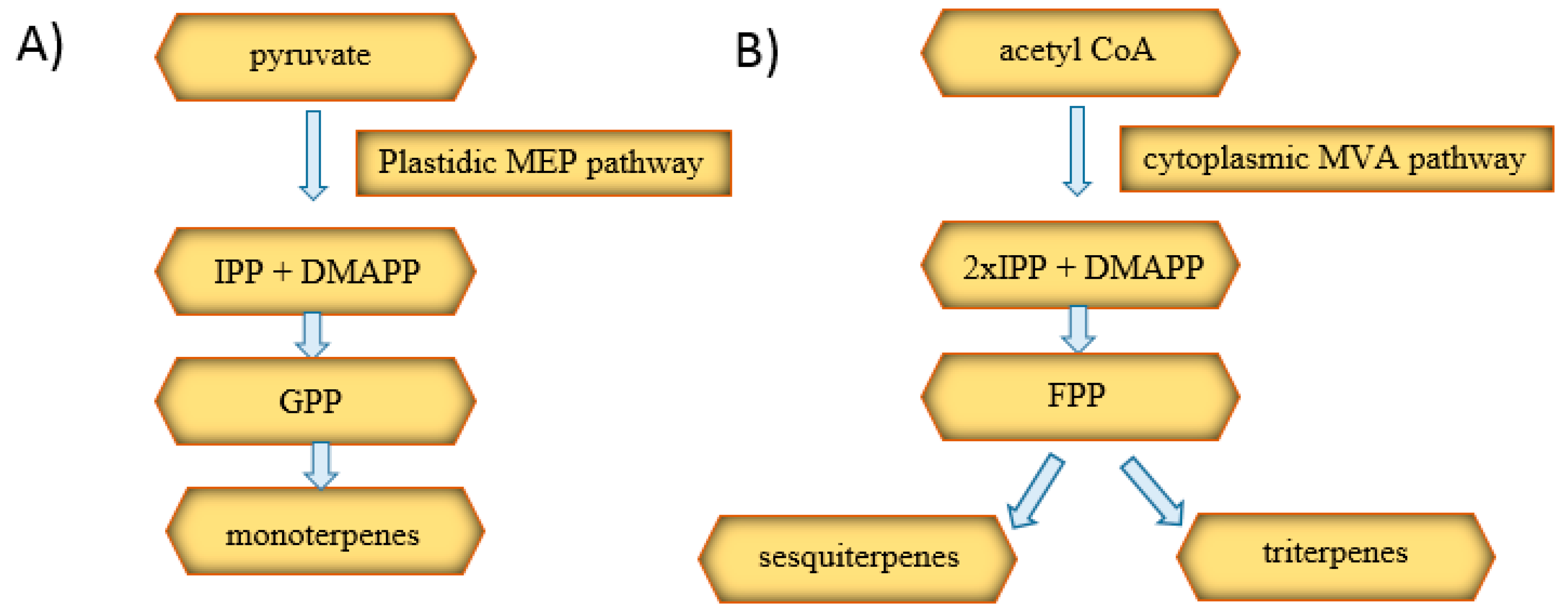
3.2. Terpenes
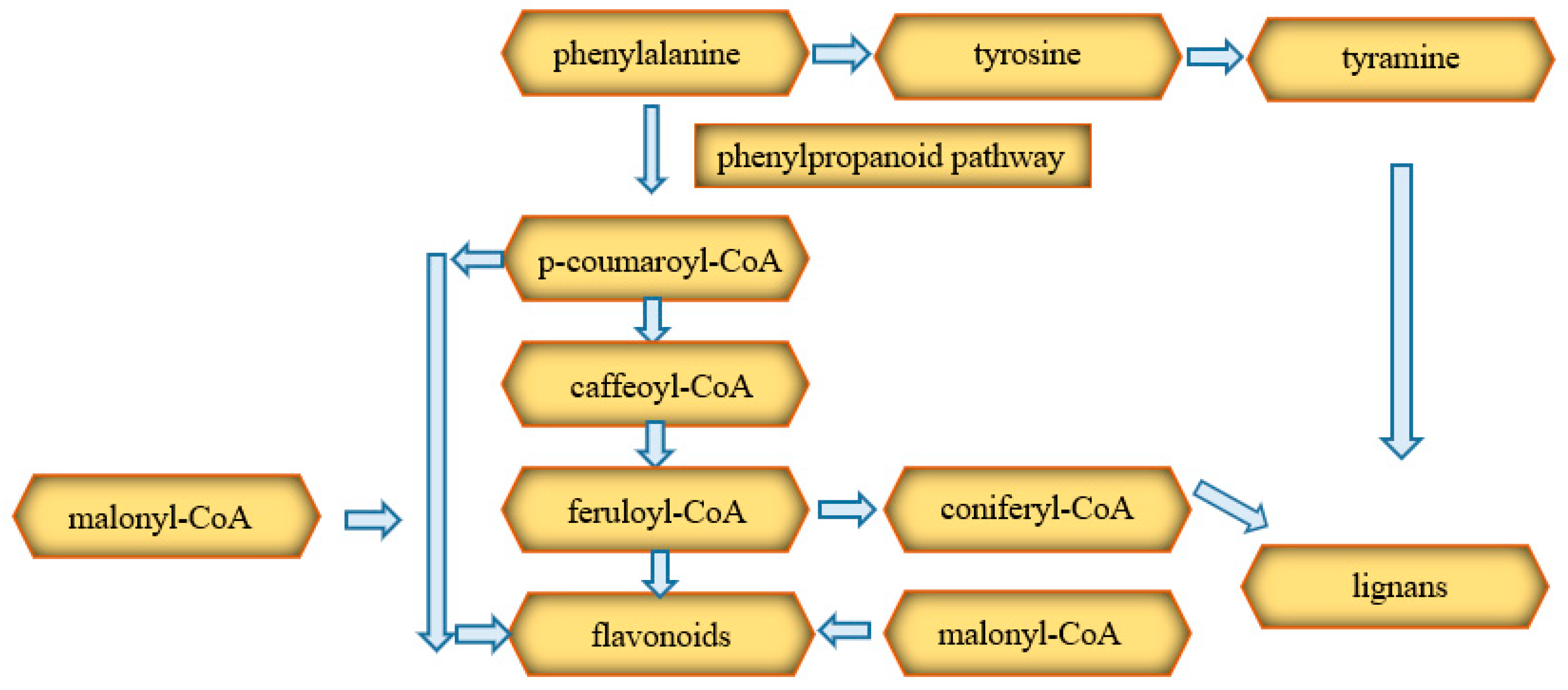
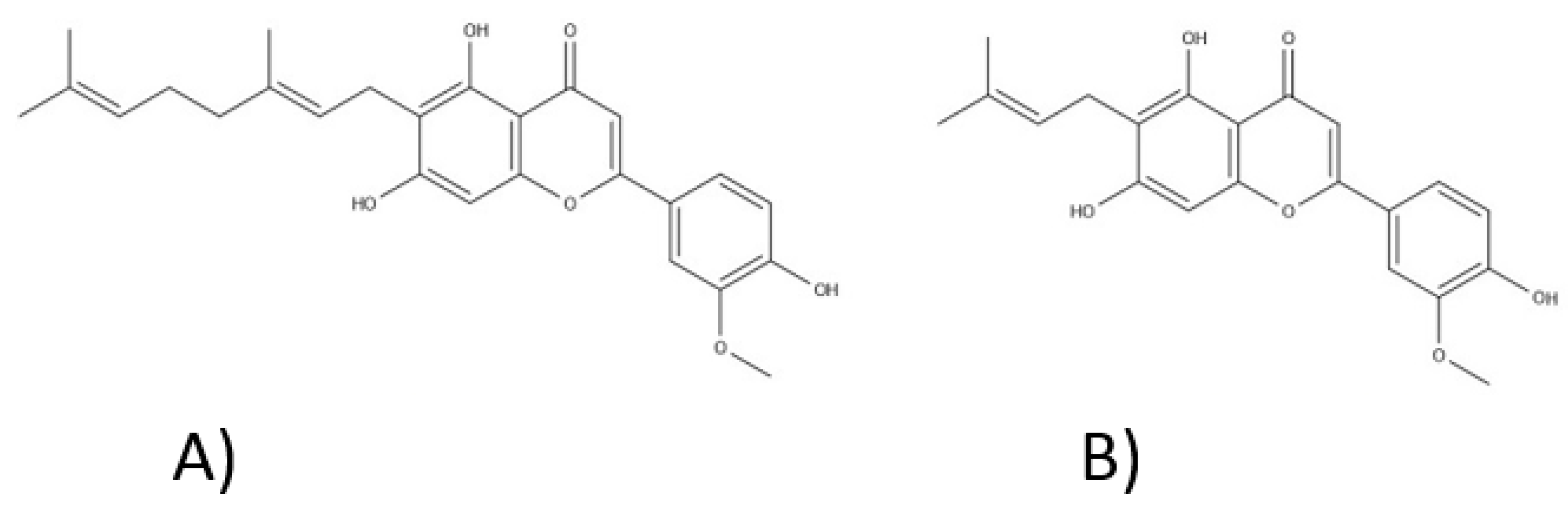
3.3. Phenolic Compounds
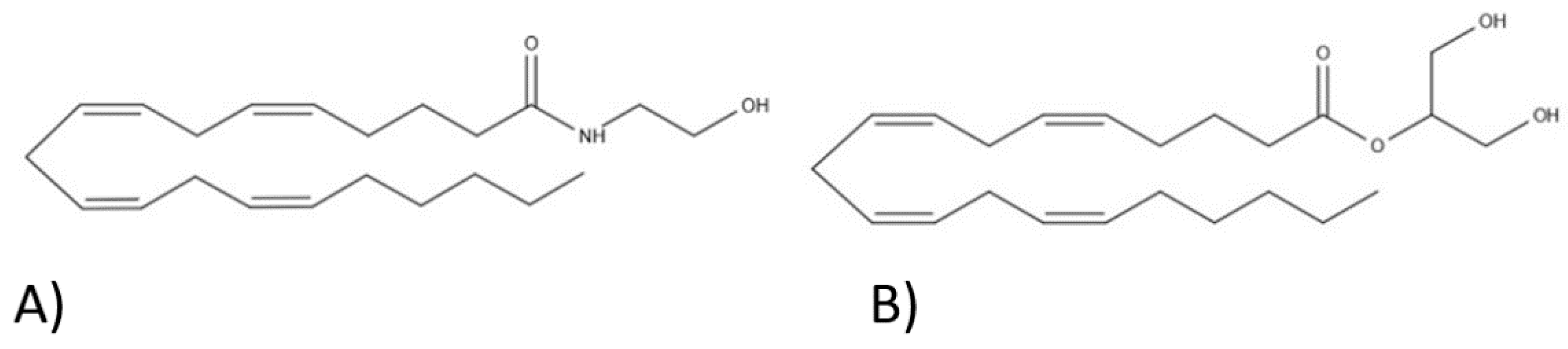
3.4. Fatty Aids
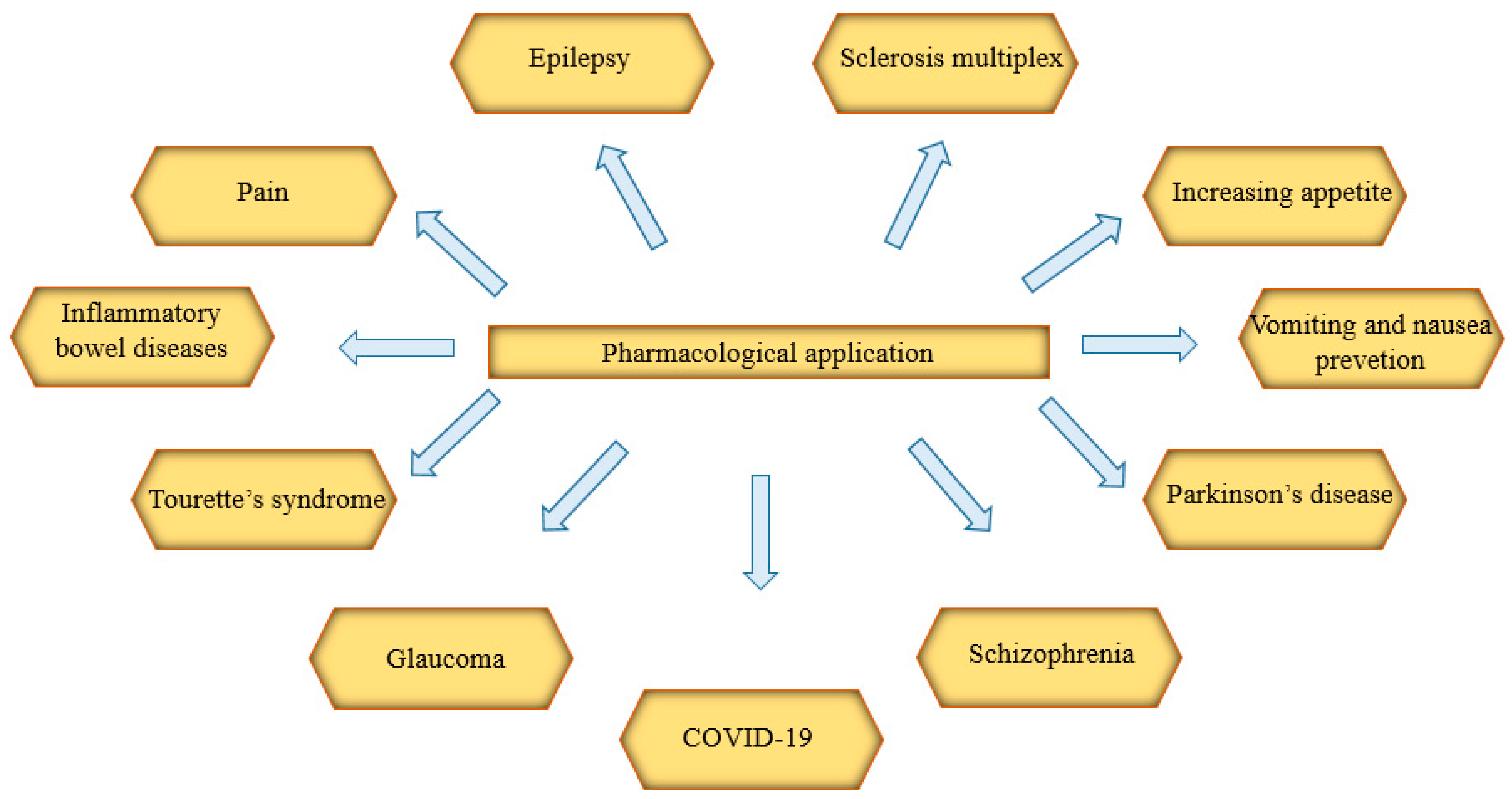
4. Pharmacological Application
4.1. Epilepsy
4.2. Sclerosis Multiplex
4.3. Vomiting and Nausea Prevention
4.4. Pain
4.5. Increasing Appetite
4.6. Inflammatory Bowel Diseases
4.7. Parkinson’s Disease
4.8. Tourette’s Syndrome
4.9. Schizophrenia
4.10. Glaucoma
4.11. COVID-19
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ∆8-THC | ∆8-tetrahydrocannabinol |
| ∆9-THC | ∆9-tetrahydrocannabinol |
| 2-AG | 2-arachidonylglycerol |
| 5-HT | serotonin receptor |
| ACE2 | angiotensin-converting enzyme 2 |
| AChE | acetylcholinesterase |
| AEA | anandamide |
| AIDS | acquired immune deficiency syndrome |
| cAMP | cyclic adenosine monophosphate |
| CB1 | cannabinoid receptor type 1 |
| CB2 | cannabinoid receptor type 2 |
| CBC | cannabichromene |
| CBD | cannabidiol |
| CBDA | cannabidiolic acid |
| CBDV | cannabidivarin |
| CBE | cannabielsoin |
| CBG | cannabigerol |
| CBGA | cannabigerolic acid |
| CBGV | cannabigerovarin |
| CBL | cannabicycline |
| CBN | cannabinol |
| CBNA | cannabinoic acid |
| CBND | cannabinodiol |
| CBT | cannabitriol |
| CDAI | Crohn’s disease activity index |
| CINV | chemotherapy-induced nausea and vomiting |
| CoA | coenzyme A |
| COVID-19 | coronavirus disease 2019 |
| CRP | C-reactive protein |
| CSG | Coronavirus Study Group |
| CXCL10/IP10 CYP | CXC-chemokine ligand cytochrome P450 |
| DMAPP | dimethylallyl diphosphate |
| DS | Dravet syndrome |
| FAAH | fatty acid amide hydrolase |
| FiO2 | fraction of inspired oxygen |
| FPP | farnesyl diphosphate |
| GABA | γ-aminobutyric acid |
| GPP | geranyl diphosphate |
| GPR | G protein-coupled receptor |
| G-SCF | granulocyte colony-stimulating factor |
| IBD | inflammatory bowel disease |
| IL | interleukin |
| IPP | isopentenyl diphosphate |
| LD50 LGS | median lethal dose Lennox-Gastaut syndrome |
| MCP1/CCL2 | monocyte chemoattractant protein-1 |
| MEP | plastidic methylerythritol phosphate |
| MIP1-α/CCL3 | macrophage inflammatory protein 1 alpha |
| MVA | mevalonic acid |
| PPAR | peroxisome proliferator-activated receptor |
| ROS | reactive oxygen species |
| SARS-CoV-2 | severe acute respiratory syndrome coronavirus 2 |
| SM | sclerosis multiplex |
| SpO2 Th1 | blood oxygen saturation T-helper type 1 |
| THC | tetrahydrocannabinol |
| THCA | tetrahydrocannabinolic acid |
| TMPRSS2 | transmembrane serine protease 2 |
| TNF-α | tumor necrosis factor |
| TRP TRPM | transient receptor potential channels transient receptor potential melastatin |
| TRPV1 | transient potential type 1 vanilloid receptor |
| TSC | tuberous sclerosis syndrome |
| USP | Pharmacopoeia of the United States |
| WHO | World Health Organization |
References
- Thatoi, H.; Patra, J.K. Biotechnology and pharmacological evaluation of medicinal plants: An overview. J. Herbs. Spices Med. Plants 2011, 17, 214–248. [Google Scholar] [CrossRef]
- Reed, J.W.; Hudlicky, T. The quest for a practical synthesis of morphine alkaloids and their derivatives by chemoenzymatic methods. Acc. Chem. Res. 2015, 48, 674–687. [Google Scholar] [CrossRef]
- Krishnamurti, C.; Rao, S.S.C.C. The isolation of morphine by Serturner. Indian J. Anaesth. 2016, 60, 861. [Google Scholar] [CrossRef]
- Chida, N. Recent advances in the synthesis of morphine and related alkaloids. In Chemistry of Opioids; Springer: Berlin/Heidelberg, Germany, 2010; pp. 1–28. [Google Scholar]
- Gulland, J.M.R.; Mem, R. Constitution of codeine and thebaine. Proc. Manch. Lit. Phil. Soc. 1925, 69, 79–86. [Google Scholar]
- Gates, M.; Tschudi, G. The Synthesis of Morphine. J. Am. Chem. Soc. 1952, 74, 1109–1110. [Google Scholar] [CrossRef]
- Bulduk, I.; Gezer, B.; Cengiz, M. Optimization of ultrasound-assisted extraction of morphine from capsules of Papaver somniferum by response surface methodology. Int. J. Anal. Chem. 2015, 2015, 1–8. [Google Scholar] [CrossRef]
- Fossati, E.; Narcross, L.; Ekins, A.; Falgueyret, J.-P.; Martin, V.J.J. Synthesis of morphinan alkaloids in Saccharomyces cerevisiae. PLoS ONE 2015, 10, e0124459. [Google Scholar] [CrossRef]
- Page, S.W.; Maddison, J.E. Principles of clinical pharmacology. Small Animal Clinical Pharmacology; Saunders Elsevier: Beijing, China, 2008; pp. 1–26. [Google Scholar]
- Maehle, A.-H. A binding question: The evolution of the receptor concept. Endeavour 2009, 33, 135–140. [Google Scholar] [CrossRef]
- Silverstein, A.M. Paul Ehrlich’s Receptor Immunology: The Magnificent Obsession; Elsevier: Amsterdam, The Netherlands, 2001; ISBN 0080538517. [Google Scholar]
- Wang, T.; Liu, X.; Guan, J.; Ge, S.; Wu, M.-B.; Lin, J.; Yang, L. Advancement of multi-target drug discoveries and promising applications in the field of Alzheimer’s disease. Eur. J. Med. Chem. 2019, 169, 200–223. [Google Scholar] [CrossRef]
- Guerrero-García, C.; Rubio-Guerra, A.F. Combination therapy in the treatment of hypertension. Drugs Context 2018, 7, 212531. [Google Scholar] [CrossRef]
- Efferth, T.; Koch, E. Complex interactions between phytochemicals. The multi-target therapeutic concept of phytotherapy. Curr. Drug Targets 2011, 12, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, H.; Kobayashi, M.; Momohara, A.; Eguchi, S.; Okamoto, T.; Yanagisawa, S.; Okubo, S.; Kiyonaga, J. Early Holocene coastal environment change inferred from deposits at Okinoshima archeological site, Boso Peninsula, central Japan. Quat. Int. 2011, 230, 87–94. [Google Scholar] [CrossRef]
- Jiang, H.-E.; Li, X.; Zhao, Y.-X.; Ferguson, D.K.; Hueber, F.; Bera, S.; Wang, Y.-F.; Zhao, L.-C.; Liu, C.-J.; Li, C.-S. A new insight into Cannabis sativa (Cannabaceae) utilization from 2500-year-old Yanghai Tombs, Xinjiang, China. J. Ethnopharmacol. 2006, 108, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Pisanti, S.; Bifulco, M. Medical Cannabis: A plurimillennial history of an evergreen. J. Cell. Physiol. 2019, 234, 8342–8351. [Google Scholar] [CrossRef] [PubMed]
- MacGillivray, N. Sir William Brooke O’Shaughnessy (1808–1889), MD, FRS, LRCS Ed: Chemical pathologist, pharmacologist and pioneer in electric telegraphy. J. Med. Biogr. 2017, 25, 186–196. [Google Scholar] [CrossRef] [PubMed]
- Zuardi, A.W. History of cannabis as a medicine: A review. Braz. J. Psychiatry 2006, 28, 153–157. [Google Scholar] [CrossRef]
- Russo, E.B. History of cannabis and its preparations in saga, science, and sobriquet. Chem. Biodivers. 2007, 4, 1614–1648. [Google Scholar] [CrossRef]
- Gaoni, Y.; Mechoulam, R. Isolation, structure, and partial synthesis of an active constituent of hashish. J. Am. Chem. Soc. 1964, 86, 1646–1647. [Google Scholar] [CrossRef]
- Ferber, S.G.; Namdar, D.; Hen-Shoval, D.; Eger, G.; Koltai, H.; Shoval, G.; Shbiro, L.; Weller, A. The “entourage effect”: Terpenes coupled with cannabinoids for the treatment of mood disorders and anxiety disorders. Curr. Neuropharmacol. 2020, 18, 87–96. [Google Scholar] [CrossRef]
- Namdar, D.; Voet, H.; Ajjampura, V.; Nadarajan, S.; Mayzlish-Gati, E.; Mazuz, M.; Shalev, N.; Koltai, H. Terpenoids and phytocannabinoids co-produced in Cannabis sativa strains show specific interaction for cell cytotoxic activity. Molecules 2019, 24, 3031. [Google Scholar] [CrossRef]
- Booth, J.K.; Bohlmann, J. Terpenes in Cannabis sativa–From plant genome to humans. Plant Sci. 2019, 284, 67–72. [Google Scholar] [CrossRef] [PubMed]
- ElSohly, M.A.; Mehmedic, Z.; Foster, S.; Gon, C.; Chandra, S.; Church, J.C. Changes in cannabis potency over the last 2 decades (1995–2014): Analysis of current data in the United States. Biol. Psychiatry 2016, 79, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Di Forti, M.; Marconi, A.; Carra, E.; Fraietta, S.; Trotta, A.; Bonomo, M.; Bianconi, F.; Gardner-Sood, P.; O’Connor, J.; Russo, M. Proportion of patients in south London with first-episode psychosis attributable to use of high potency cannabis: A case-control study. Lancet Psychiatry 2015, 2, 233–238. [Google Scholar] [CrossRef]
- Niesink, R.J.M.; van Laar, M.W. Does cannabidiol protect against adverse psychological effects of THC? Front. Psychiatry 2013, 4, 130. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Morrison, P.D.; Fusar-Poli, P.; Martin-Santos, R.; Borgwardt, S.; Winton-Brown, T.; Nosarti, C.; O’Carroll, C.M.; Seal, M.; Allen, P. Opposite effects of Δ-9-tetrahydrocannabinol and cannabidiol on human brain function and psychopathology. Neuropsychopharmacology 2010, 35, 764–774. [Google Scholar] [CrossRef] [PubMed]
- Papaseit, E.; Pérez-Mañá, C.; Pérez-Acevedo, A.P.; Hladun, O.; Torres-Moreno, M.C.; Muga, R.; Torrens, M.; Farre, M. Cannabinoids: From pot to lab. Int. J. Med. Sci. 2018, 15, 1286. [Google Scholar] [CrossRef]
- Landa, L.; Jurica, J.; Sliva, J.; Pechackova, M.; Demlova, R. Medical cannabis in the treatment of cancer pain and spastic conditions and options of drug delivery in clinical practice. Biomed. Pap. Med. Fac. Palacky Univ. Olomouc 2018, 162, 18–25. [Google Scholar] [CrossRef]
- Abrams, D.I.; Guzman, M. Cannabis in cancer care. Clin. Pharmacol. Ther. 2015, 97, 575–586. [Google Scholar] [CrossRef]
- Maida, V.; Daeninck, P.J. A user’s guide to cannabinoid therapies in oncology. Curr. Oncol. 2016, 23, 398. [Google Scholar] [CrossRef]
- Vučković, S.; Srebro, D.; Vujović, K.S.; Vučetić, Č.; Prostran, M. Cannabinoids and pain: New insights from old molecules. Front. Pharmacol. 2018, 9, 1259. [Google Scholar] [CrossRef]
- Weiss, S.R.B.; Howlett, K.D.; Baler, R.D. Building smart cannabis policy from the science up. Int. J. Drug Policy 2017, 42, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Leinwand, K.L.; Gerich, M.E.; Hoffenberg, E.J.; Collins, C.B. Manipulation of the endocannabinoid system in colitis: A comprehensive review. Inflamm. Bowel Dis. 2017, 23, 192–199. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Maccarrone, M.; Bab, I.; Bíró, T.; Cabral, G.A.; Dey, S.K.; Di Marzo, V.; Konje, J.C.; Kunos, G.; Mechoulam, R.; Pacher, P. Endocannabinoid signaling at the periphery: 50 years after THC. Trends Pharmacol. Sci. 2015, 36, 277–296. [Google Scholar] [CrossRef] [PubMed]
- Campos, A.C.; Fogaça, M.V.; Sonego, A.B.; Guimarães, F.S. Cannabidiol, neuroprotection and neuropsychiatric disorders. Pharmacol. Res. 2016, 112, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Filloux, F.M. Cannabinoids for pediatric epilepsy? Up in smoke or real science? Transl. Pediatr. 2015, 4, 271. [Google Scholar]
- Basavarajappa, B.S.; Subbanna, S. Potential mechanisms underlying the deleterious effects of synthetic cannabinoids found in Spice/K2 products. Brain Sci. 2019, 9, 14. [Google Scholar] [CrossRef]
- Maida, V. Medical cannabis in the palliation of malignant wounds—A case report. J. Pain Symptom Manage. 2017, 53, e4–e6. [Google Scholar] [CrossRef]
- Solymosi, K.; Kofalvi, A. Cannabis: A Treasure Trove or Pandora’s Box? Mini Rev. Med. Chem. 2017, 17, 1223–1291. [Google Scholar] [CrossRef]
- Andre, C.M.; Hausman, J.-F.; Guerriero, G. Cannabis sativa: The Plant of the Thousand and One Molecules. Front. Plant Sci. 2016, 7, 1–17. [Google Scholar] [CrossRef]
- Prandi, C.; Blangetti, M.; Namdar, D.; Koltai, H. Structure-activity relationship of cannabis derived compounds for the treatment of neuronal activity-related diseases. Molecules 2018, 23, 1526. [Google Scholar] [CrossRef]
- ElSohly, M.A.; Slade, D. Chemical constituents of marijuana: The complex mixture of natural cannabinoids. Life Sci. 2005, 78, 539–548. [Google Scholar] [CrossRef]
- Richins, R.D.; Rodriguez-Uribe, L.; Lowe, K.; Ferral, R.; O’Connell, M.A. Accumulation of bioactive metabolites in cultivated medical Cannabis. PLoS ONE 2018, 13, e0201119. [Google Scholar] [CrossRef]
- Hanuš, L.O.; Meyer, S.M.; Muñoz, E.; Taglialatela-Scafati, O.; Appendino, G. Phytocannabinoids: A unified critical inventory. Nat. Prod. Rep. 2016, 33, 1357–1392. [Google Scholar] [CrossRef] [PubMed]
- Abuhasira, R.; Shbiro, L.; Landschaft, Y. Medical use of cannabis and cannabinoids containing products–Regulations in Europe and North America. Eur. J. Intern. Med. 2018, 49, 2–6. [Google Scholar] [CrossRef] [PubMed]
- McPartland, J.M. Cannabis systematics at the levels of family, genus, and species. Cannabis Cannabinoid Res. 2018, 3, 203–212. [Google Scholar] [CrossRef]
- Gloss, D. An overview of products and bias in research. Neurotherapeutics 2015, 12, 731–734. [Google Scholar] [CrossRef] [PubMed]
- Furqan, T.; Batool, S.; Habib, R.; Shah, M.; Kalasz, H.; Darvas, F.; Kuca, K.; Nepovimova, E.; Batool, S.; Nurulain, S.M. Cannabis Constituents and Acetylcholinesterase Interaction: Molecular Docking, In Vitro Studies and Association with CNR1 rs806368 and ACHE rs17228602. Biomolecules 2020, 10, 758. [Google Scholar] [CrossRef] [PubMed]
- Boggs, D.L.; Nguyen, J.D.; Morgenson, D.; Taffe, M.A.; Ranganathan, M. Clinical and preclinical evidence for functional interactions of cannabidiol and Δ 9-tetrahydrocannabinol. Neuropsychopharmacology 2018, 43, 142–154. [Google Scholar] [CrossRef]
- Morales, P.; Hurst, D.P.; Reggio, P.H. Molecular targets of the phytocannabinoids: A complex picture. In Phytocannabinoids; Springer: Berlin/Heidelberg, Germany, 2017; pp. 103–131. [Google Scholar]
- Pisanti, S.; Malfitano, A.M.; Ciaglia, E.; Lamberti, A.; Ranieri, R.; Cuomo, G.; Abate, M.; Faggiana, G.; Proto, M.C.; Fiore, D. Cannabidiol: State of the art and new challenges for therapeutic applications. Pharmacol. Ther. 2017, 175, 133–150. [Google Scholar] [CrossRef]
- White, C.M. A review of human studies assessing cannabidiol’s (CBD) therapeutic actions and potential. J. Clin. Pharmacol. 2019, 59, 923–934. [Google Scholar] [CrossRef]
- Rong, C.; Lee, Y.; Carmona, N.E.; Cha, D.S.; Ragguett, R.-M.; Rosenblat, J.D.; Mansur, R.B.; Ho, R.C.; McIntyre, R.S. Cannabidiol in medical marijuana: Research vistas and potential opportunities. Pharmacol. Res. 2017, 121, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Mouhamed, Y.; Vishnyakov, A.; Qorri, B.; Sambi, M.; Frank, S.M.S.; Nowierski, C.; Lamba, A.; Bhatti, U.; Szewczuk, M.R. Therapeutic potential of medicinal marijuana: An educational primer for health care professionals. Drug. Healthc. Patient Saf. 2018, 10, 45. [Google Scholar] [CrossRef] [PubMed]
- Wollner, H.J.; Matchett, J.R.; Levine, J.; Loewe, S. Isolation of a physiologically active tetrahydrocannabinol from Cannabis sativa resin. J. Am. Chem. Soc. 1942, 64, 26–29. [Google Scholar] [CrossRef]
- Krejci, Z.; Šantavý, F. Isolace dalsˇích látek z listí indického konopí Cannabis sativa L. Acta Univ. Palacki. Olomuc. 1955, 6, 59–66. [Google Scholar]
- Santavý, F. Notes on the structure of cannabidiol compounds. Acta Univ. Palacki. Olomuc. Fac. Med. 1964, 35, 5–9. [Google Scholar]
- Fischedick, J.T. Identification of terpenoid chemotypes among high (−)-trans-Δ9-tetrahydrocannabinol-producing Cannabis sativa L. cultivars. Cannabis Cannabinoid Res. 2017, 2, 34–47. [Google Scholar] [CrossRef]
- Ujváry, I.; Hanuš, L. Human metabolites of cannabidiol: A review on their formation, biological activity, and relevance in therapy. Cannabis Cannabinoid Res. 2016, 1, 90–101. [Google Scholar] [CrossRef]
- ElSohly, M.A.; Radwan, M.M.; Gul, W.; Chandra, S.; Galal, A. Phytochemistry of Cannabis sativa L. In Phytocannabinoids; Springer: Berlin/Heidelberg, Germany, 2017; pp. 1–36. [Google Scholar]
- Valdeolivas, S.; Navarrete, C.; Cantarero, I.; Bellido, M.L.; Muñoz, E.; Sagredo, O. Neuroprotective properties of cannabigerol in Huntington’s disease: Studies in R6/2 mice and 3-nitropropionate-lesioned mice. Neurotherapeutics 2015, 12, 185–199. [Google Scholar] [CrossRef]
- Aizpurua-Olaizola, O.; Soydaner, U.; Öztürk, E.; Schibano, D.; Simsir, Y.; Navarro, P.; Etxebarria, N.; Usobiaga, A. Evolution of the cannabinoid and terpene content during the growth of Cannabis sativa plants from different chemotypes. J. Nat. Prod. 2016, 79, 324–331. [Google Scholar] [CrossRef]
- Drugs, U.N.O. Crime Recommended Methods for the Identification and Analysis of Cannabis and Cannabis Products: Manual for Use by National Drug Testing Laboratories; United Nations Publications: New York, NY, USA, 2009; ISBN 9211482429. [Google Scholar]
- Gugliandolo, A.; Pollastro, F.; Grassi, G.; Bramanti, P.; Mazzon, E. In Vitro Model of Neuroinflammation: Efficacy of Cannabigerol, a Non-Psychoactive Cannabinoid. Int. J. Mol. Sci. 2018, 19, 1992. [Google Scholar] [CrossRef]
- Claussen, U.; Von Spulak, F.; Korte, F. Chemical classification of plants. XXXI. Hashish. 10. Cannabichromene, a new hashish component. Tetrahedron 1966, 22, 1477–1479. [Google Scholar] [CrossRef]
- Gaoni, Y.; Mechoulam, R. Cannabichromene, a new active principle in hashish. Chem. Commun. 1966, 1, 20–21. [Google Scholar] [CrossRef]
- Udoh, M.; Santiago, M.; Devenish, S.; McGregor, I.S.; Connor, M. Cannabichromene is a cannabinoid CB2 receptor agonist. Br. J. Pharmacol. 2019, 176, 4537–4547. [Google Scholar] [CrossRef]
- Wood, T.B.; Spivey, W.T.N.; Easterfield, T.H. III.—Cannabinol. Part I. J. Chem. Soc. Trans. 1899, 75, 20–36. [Google Scholar] [CrossRef]
- Adams, R.; Hunt, M.; Clark, J.H. Structure of cannabidiol, a product isolated from the marihuana extract of Minnesota wild hemp. I. J. Am. Chem. Soc. 1940, 62, 196–200. [Google Scholar] [CrossRef]
- Obata, Y.; Ishikawa, Y. Constituents of hemp plant (Cannabis sativa). III. Isolation of a Gibbs-positive compound from Japanese hemp. Agric. Biol. Chem. 1966, 30, 619–620. [Google Scholar]
- Shani, A.; Mechoulam, R. A new type of cannabinoid. Synthesis of cannabielsoic acid A by a novel photo-oxidative cyclisation. J. Chem. Soc. D Chem. Commun. 1970, 273–274. [Google Scholar] [CrossRef]
- Yamaori, S.; Okushima, Y.; Masuda, K.; Kushihara, M.; Katsu, T.; Narimatsu, S.; Yamamoto, I.; Watanabe, K. Structural requirements for potent direct inhibition of human cytochrome P450 1A1 by cannabidiol: Role of pentylresorcinol moiety. Biol. Pharm. Bull. 2013, 36, 1197–1203. [Google Scholar] [CrossRef][Green Version]
- Ch, L.R.J.J.; Bercht, C.A.L.; van Ooyen, R.; Spronck, H.J.W. Cannabinodiol: Conclusive identification and synthesis of a new cannabinoid from Cannabis sativa. Phytochemistry 1977, 16, 595–597. [Google Scholar]
- Hazekamp, A.; Tejkalová, K.; Papadimitriou, S. Cannabis: From cultivar to chemovar II—A metabolomics approach to Cannabis classification. Cannabis Cannabinoid Res. 2016, 1, 202–215. [Google Scholar] [CrossRef]
- Elzinga, S.; Fischedick, J.; Podkolinski, R.; Raber, J.C. Cannabinoids and terpenes as chemotaxonomic markers in cannabis. Nat. Prod. Chem. Res. 2015, 3, 10–4172. [Google Scholar]
- Grof, C.P.L. Cannabis, from plant to pill. Br. J. Clin. Pharmacol. 2018, 84, 2463–2467. [Google Scholar] [CrossRef] [PubMed]
- Rogowska-Szadkowska, D.; Strumiło, J.; Chlabicz, S. Is medical marijuana legalisation possible in Poland? Cent. Eur. J. Public Health 2018, 26, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Pellati, F.; Brighenti, V.; Sperlea, J.; Marchetti, L.; Bertelli, D.; Benvenuti, S. New methods for the comprehensive analysis of bioactive compounds in Cannabis sativa L.(hemp). Molecules 2018, 23, 2639. [Google Scholar] [CrossRef]
- Pollastro, F.; Minassi, A.; Fresu, L.G. Cannabis phenolics and their bioactivities. Curr. Med. Chem. 2018, 25, 1160–1185. [Google Scholar] [CrossRef]
- Crombie, L.; Crombie, W.M.L.; Jamieson, S.V. Extractives of Thailand cannabis: Synthesis of canniprene and isolation of new geranylated and prenylated chrysoeriols. Tetrahedron Lett. 1980, 21, 3607–3610. [Google Scholar] [CrossRef]
- Choi, Y.H.; Hazekamp, A.; Peltenburg-Looman, A.M.G.; Frédérich, M.; Erkelens, C.; Lefeber, A.W.M.; Verpoorte, R. NMR assignments of the major cannabinoids and cannabiflavonoids isolated from flowers of Cannabis sativa. Phytochem. Anal. Int. J. Plant Chem. Biochem. Tech. 2004, 15, 345–354. [Google Scholar] [CrossRef]
- Barrett, M.L.; Gordon, D.; Evans, F.J. Isolation from Cannabis sativa L. of cannflavin—A novel inhibitor of prostaglandin production. Biochem. Pharmacol. 1985, 34, 2019–2024. [Google Scholar] [CrossRef]
- Ibrahim, R.K. A forty-year journey in plant research: Original contributions to flavonoid biochemistry. Can. J. Bot. 2005, 83, 433–450. [Google Scholar] [CrossRef]
- Sakakibara, I.; Katsuhara, T.; Ikeya, Y.; Hayashi, K.; Mitsuhashi, H. Cannabisin A, an arylnaphthalene lignanamide from fruits of Cannabis sativa. Phytochemistry 1991, 30, 3013–3016. [Google Scholar] [CrossRef]
- Yan, X.; Tang, J.; dos Santos Passos, C.; Nurisso, A.; Simoes-Pires, C.A.; Ji, M.; Lou, H.; Fan, P. Characterization of lignanamides from hemp (Cannabis sativa L.) seed and their antioxidant and acetylcholinesterase inhibitory activities. J. Agric. Food Chem. 2015, 63, 10611–10619. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Luo, Q.; Fan, P. Cannabisin F from hemp (Cannabis sativa) seed suppresses lipopolysaccharide-induced inflammatory responses in BV2 microglia as SIRT1 modulator. Int. J. Mol. Sci. 2019, 20, 507. [Google Scholar] [CrossRef] [PubMed]
- Smeds, A.I.; Eklund, P.C.; Willför, S.M. Content, composition, and stereochemical characterisation of lignans in berries and seeds. Food Chem. 2012, 134, 1991–1998. [Google Scholar] [CrossRef] [PubMed]
- Saleem, M.; Kim, H.J.; Ali, M.S.; Lee, Y.S. An update on bioactive plant lignans. Nat. Prod. Rep. 2005, 22, 696–716. [Google Scholar] [CrossRef]
- Guo, T.; Liu, Q.; Hou, P.; Li, F.; Guo, S.; Song, W.; Zhang, H.; Liu, X.; Zhang, S.; Zhang, J. Stilbenoids and cannabinoids from the leaves of Cannabis sativa f. sativa with potential reverse cholesterol transport activity. Food Funct. 2018, 9, 6608–6617. [Google Scholar] [CrossRef]
- Deferne, J.-L.; Pate, D.W. Hemp seed oil: A source of valuable essential fatty acids. J. Int. Hemp Assoc. 1996, 3, 4–7. [Google Scholar]
- Pate, D.W. Hemp seed: A valuable food source. Adv. Hemp Res. 1999, 243–255. [Google Scholar]
- Ross, S.A.; ElSohly, H.N.; ElKashoury, E.A.; ElSohly, M.A. Fatty acids of cannabis seeds. Phytochem. Anal. 1996, 7, 279–283. [Google Scholar] [CrossRef]
- De Carvalho, C.C.C.R.; Caramujo, M.J. The various roles of fatty acids. Molecules 2018, 23, 2583. [Google Scholar] [CrossRef]
- Gammone, M.A.; Riccioni, G.; Parrinello, G.; D’Orazio, N. Omega-3 polyunsaturated fatty acids: Benefits and endpoints in sport. Nutrients 2019, 11, 46. [Google Scholar] [CrossRef]
- Kiralan, M.; Gül, V.; Kara, S.M. Fatty acid composition of hempseed oils from different locatins in Turkey. Spanish J. Agric. Res. 2010, 385–390. [Google Scholar] [CrossRef]
- Caramia, G. The essential fatty acids omega-6 and omega-3: From their discovery to their use in therapy. Minerva Pediatr. 2008, 60, 219. [Google Scholar] [PubMed]
- Sills, G.J.; Rogawski, M.A. Mechanisms of action of currently used antiseizure drugs. Neuropharmacology 2020, 168, 107966. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Brodie, M.J.; Kwan, P. What has been the impact of new drug treatments on epilepsy? Curr. Opin. Neurol. 2020, 33, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Elliott, J.; DeJean, D.; Clifford, T.; Coyle, D.; Potter, B.; Skidmore, B.; Alexander, C.; Repetski, A.E.; McCoy, B.; Wells, G.A. Cannabis for pediatric epilepsy: Protocol for a living systematic review. Syst. Rev. 2018, 7, 95. [Google Scholar] [CrossRef]
- O’Connell, B.K.; Gloss, D.; Devinsky, O. Cannabinoids in treatment-resistant epilepsy: A review. Epilepsy Behav. 2017, 70, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Zaheer, S.; Kumar, D.; Khan, M.T.; Giyanwani, P.R.; Kiran, F.N.U. Epilepsy and cannabis: A literature review. Cureus 2018, 1–7. [Google Scholar] [CrossRef]
- Sekar, K.; Pack, A. Epidiolex as adjunct therapy for treatment of refractory epilepsy: A comprehensive review with a focus on adverse effects. F1000Research 2019, 8, F1000 Faculty Rev-234. [Google Scholar] [CrossRef]
- Devinsky, O.; Cross, J.H.; Laux, L.; Marsh, E.; Miller, I.; Nabbout, R.; Scheffer, I.E.; Thiele, E.A.; Wright, S. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N. Engl. J. Med. 2017, 376, 2011–2020. [Google Scholar] [CrossRef]
- Devinsky, O.; Patel, A.D.; Cross, J.H.; Villanueva, V.; Wirrell, E.C.; Privitera, M.; Greenwood, S.M.; Roberts, C.; Checketts, D.; VanLandingham, K.E. Effect of cannabidiol on drop seizures in the Lennox–Gastaut syndrome. N. Engl. J. Med. 2018, 378, 1888–1897. [Google Scholar] [CrossRef]
- Thiele, E.A.; Marsh, E.D.; French, J.A.; Mazurkiewicz-Beldzinska, M.; Benbadis, S.R.; Joshi, C.; Lyons, P.D.; Taylor, A.; Roberts, C.; Sommerville, K. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2018, 391, 1085–1096. [Google Scholar] [CrossRef]
- Chiurchiù, V.; van der Stelt, M.; Centonze, D.; Maccarrone, M. The endocannabinoid system and its therapeutic exploitation in multiple sclerosis: Clues for other neuroinflammatory diseases. Prog. Neurobiol. 2018, 160, 82–100. [Google Scholar] [CrossRef] [PubMed]
- Suryadevara, U.; Bruijnzeel, D.M.; Nuthi, M.; Jagnarine, D.A.; Tandon, R.; Bruijnzeel, A.W. Pros and cons of medical cannabis use by people with chronic brain disorders. Curr. Neuropharmacol. 2017, 15, 800–814. [Google Scholar] [CrossRef] [PubMed]
- Belendiuk, K.A.; Baldini, L.L.; Bonn-Miller, M.O. Narrative review of the safety and efficacy of marijuana for the treatment of commonly state-approved medical and psychiatric disorders. Addict. Sci. Clin. Pract. 2015, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Tintore, M.; Vidal-Jordana, A.; Sastre-Garriga, J. Treatment of multiple sclerosis—Success from bench to bedside. Nat. Rev. Neurol. 2019, 15, 53–58. [Google Scholar] [CrossRef]
- Beauchemin, M.; Sung, L.; Hershman, D.L.; Weng, C.; Dupuis, L.L.; Schnall, R. Guideline concordant care for prevention of acute chemotherapy-induced nausea and vomiting in children, adolescents, and young adults. Support. Care Cancer 2020, 28, 1–9. [Google Scholar] [CrossRef]
- Badowski, M.E. A review of oral cannabinoids and medical marijuana for the treatment of chemotherapy-induced nausea and vomiting: A focus on pharmacokinetic variability and pharmacodynamics. Cancer Chemother. Pharmacol. 2017, 80, 441–449. [Google Scholar] [CrossRef]
- May, M.B.; Glode, A.E. Dronabinol for chemotherapy-induced nausea and vomiting unresponsive to antiemetics. Cancer Manag. Res. 2016, 8, 49. [Google Scholar]
- Narain, T.; Farrah, K. Nabilone for Non-Chemotherapy Associated Nausea and Vomiting and Weight Loss Due to Medical Conditions: A Review of Clinical Effectiveness and Guidelines; Canadian Agency for Drugs and Technologies in Health: Ottawa, ON, Canada, 2017. [Google Scholar]
- Hall, W. Focus: Addiction: US Policy Responses to Calls for the Medical Use of Cannabis. Yale J. Biol. Med. 2015, 88, 257. [Google Scholar]
- Cavalli, E.; Mammana, S.; Nicoletti, F.; Bramanti, P.; Mazzon, E. The neuropathic pain: An overview of the current treatment and future therapeutic approaches. Int. J. Immunopathol. Pharmacol. 2019, 33, 1–10. [Google Scholar] [CrossRef]
- Casey, S.L.; Vaughan, C.W. Plant-based cannabinoids for the treatment of chronic neuropathic pain. Medicines 2018, 5, 67. [Google Scholar] [CrossRef] [PubMed]
- Childs, D.S.; Jatoi, A. A hunger for hunger: A review of palliative therapies for cancer-associated anorexia. Ann. Palliat. Med. 2019, 8, 50. [Google Scholar] [CrossRef] [PubMed]
- Zutt, M.; Hänßle, H.; Emmert, S.; Neumann, C.; Kretschmer, L. Dronabinol for supportive therapy in patients with malignant melanoma and liver metastases. Hautarzt 2006, 57, 423. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.Y.; Ng, S.C. The state of the art on treatment of Crohn’s disease. J. Gastroenterol. 2018, 53, 989–998. [Google Scholar] [CrossRef] [PubMed]
- Hasenoehrl, C.; Storr, M.; Schicho, R. Cannabinoids for treating inflammatory bowel diseases: Where are we and where do we go? Expert Rev. Gastroenterol. Hepatol. 2017, 11, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Storr, M.; Devlin, S.; Kaplan, G.G.; Panaccione, R.; Andrews, C.N. Cannabis use provides symptom relief in patients with inflammatory bowel disease but is associated with worse disease prognosis in patients with Crohn’s disease. Inflamm. Bowel Dis. 2014, 20, 472–480. [Google Scholar] [CrossRef]
- Naftali, T.; Schleider, L.B.-L.; Dotan, I.; Lansky, E.P.; Benjaminov, F.S.; Konikoff, F.M. Cannabis induces a clinical response in patients with Crohn’s disease: A prospective placebo-controlled study. Clin. Gastroenterol. Hepatol. 2013, 11, 1276–1280. [Google Scholar] [CrossRef]
- Armstrong, M.J.; Okun, M.S. Diagnosis and treatment of Parkinson disease: A review. JAMA 2020, 323, 548–560. [Google Scholar] [CrossRef]
- Babayeva, M.; Assefa, H.; Basu, P.; Chumki, S.; Loewy, Z. Marijuana compounds: A nonconventional approach to Parkinson’s disease therapy. Park. Dis. 2016, 2016, 1279042. [Google Scholar] [CrossRef]
- Lotan, I.; Treves, T.A.; Roditi, Y.; Djaldetti, R. Cannabis (medical marijuana) treatment for motor and non–motor symptoms of Parkinson disease: An open-label observational study. Clin. Neuropharmacol. 2014, 37, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Novotny, M.; Valis, M.; Klimova, B. Tourette syndrome: A mini-review. Front. Neurol. 2018, 9, 139. [Google Scholar] [CrossRef] [PubMed]
- Kanaan, A.S.; Jakubovski, E.; Müller-Vahl, K. Significant tic reduction in an otherwise treatment-resistant patient with Gilles de la Tourette syndrome following treatment with nabiximols. Brain Sci. 2017, 7, 47. [Google Scholar] [CrossRef] [PubMed]
- Abramovici, H. Information for Health Care Professionals: Cannabis (Marihuana, Marijuana) and the Cannabinoids; Health Canada: Ottawa, ON, Canada, 2018. [Google Scholar]
- Stępnicki, P.; Kondej, M.; Kaczor, A.A. Current concepts and treatments of schizophrenia. Molecules 2018, 23, 2087. [Google Scholar] [CrossRef]
- Weinreb, R.N.; Aung, T.; Medeiros, F.A. The pathophysiology and treatment of glaucoma: A review. JAMA 2014, 311, 1901–1911. [Google Scholar] [CrossRef]
- Sun, X.; Xu, C.S.; Chadha, N.; Chen, A.; Liu, J. Focus: Addiction: Marijuana for Glaucoma: A Recipe for Disaster or Treatment? Yale J. Biol. Med. 2015, 88, 265. [Google Scholar]
- Adelli, G.R.; Bhagav, P.; Taskar, P.; Hingorani, T.; Pettaway, S.; Gul, W.; ElSohly, M.A.; Repka, M.A.; Majumdar, S. Development of a Δ9-tetrahydrocannabinol amino acid-dicarboxylate prodrug with improved ocular bioavailability. Investig. Ophthalmol. Vis. Sci. 2017, 58, 2167–2179. [Google Scholar] [CrossRef]
- Bhartiya, S.; Ichhpujani, P. Complementary and alternate management of glaucoma: The verdict so far. J. Curr. Glaucoma Pract. 2014, 8, 54. [Google Scholar]
- Wang, C.; Horby, P.W.; Hayden, F.G.; Gao, G.F. A novel coronavirus outbreak of global health concern. Lancet 2020, 395, 470–473. [Google Scholar] [CrossRef]
- Guo, Y.-R.; Cao, Q.-D.; Hong, Z.-S.; Tan, Y.-Y.; Chen, S.-D.; Jin, H.-J.; Tan, K.-S.; Wang, D.-Y.; Yan, Y. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak–an update on the status. Mil. Med. Res. 2020, 7, 1–10. [Google Scholar] [CrossRef]
- Vannabouathong, C.; Devji, T.; Ekhtiari, S.; Chang, Y.; Phillips, S.A.; Zhu, M.; Chagla, Z.; Main, C.; Bhandari, M. Novel coronavirus COVID-19: Current evidence and evolving strategies. J. Bone Joint Surg. Am. 2020, 102, 734. [Google Scholar] [CrossRef] [PubMed]
- Onaivi, E.S.; Sharma, V. Cannabis for COVID-19: Can cannabinoids quell the cytokine storm? Future Sci OA 2020, 1–4. [Google Scholar] [CrossRef]
- Cascella, M.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; Di Napoli, R. Features, evaluation and treatment coronavirus (COVID-19). In Statpearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Tahamtan, A.; Tavakoli-Yaraki, M.; Salimi, V. Opioids/cannabinoids as a potential therapeutic approach in COVID-19 patients. Expert Rev. Respir. Med. 2020, 14, 965–967. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Liu, J.; Zhang, D.; Xu, Z.; Ji, J.; Wen, C. Cytokine storm in COVID-19: The current evidence and treatment strategies. Front. Immunol. 2020, 11, 1708. [Google Scholar] [CrossRef] [PubMed]
- Hojyo, S.; Uchida, M.; Tanaka, K.; Hasebe, R.; Tanaka, Y.; Murakami, M.; Hirano, T. How COVID-19 induces cytokine storm with high mortality. Inflamm. Regen. 2020, 40, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zaim, S.; Chong, J.H.; Sankaranarayanan, V.; Harky, A. COVID-19 and multi-organ response. Curr. Probl. Cardiol. 2020, 100618. [Google Scholar] [CrossRef]
- Rossi, F.; Tortora, C.; Argenziano, M.; Di Paola, A.; Punzo, F. Cannabinoid Receptor Type 2: A Possible Target in SARS-CoV-2 (CoV-19) Infection? Int. J. Mol. Sci. 2020, 21, 3809. [Google Scholar] [CrossRef]
- Ragia, G.; Manolopoulos, V.G. Inhibition of SARS-CoV-2 entry through the ACE2/TMPRSS2 pathway: A promising approach for uncovering early COVID-19 drug therapies. Eur. J. Clin. Pharmacol. 2020, 76, 1–8. [Google Scholar] [CrossRef]
- Esposito, G.; Pesce, M.; Seguella, L.; Sanseverino, W.; Lu, J.; Corpetti, C.; Sarnelli, G. The potential of cannabidiol in the COVID-19 pandemic: A hypothesis letter. Br. J. Pharmacol. 2020, 177, 4967–4970. [Google Scholar] [CrossRef]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and anti-inflammatory properties of cannabidiol. Antioxidants 2020, 9, 21. [Google Scholar] [CrossRef]
- Byrareddy, S.N.; Mohan, M. SARS-CoV2 induced respiratory distress: Can Cannabinoids be added to anti-viral therapies to reduce lung inflammation? Brain. Behav. Immun. 2020, 87, 120–121. [Google Scholar] [CrossRef] [PubMed]
- Khodadadi, H.; Salles, É.L.; Jarrahi, A.; Chibane, F.; Costigliola, V.; Yu, J.C.; Vaibhav, K.; Hess, D.C.; Dhandapani, K.M.; Baban, B. Cannabidiol modulates cytokine storm in acute respiratory distress syndrome induced by simulated viral infection using synthetic RNA. Cannabis Cannabinoid Res. 2020, 5, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, B.L.F.; Springs, A.E.B.; Kaminski, N.E. The profile of immune modulation by cannabidiol (CBD) involves deregulation of nuclear factor of activated T cells (NFAT). Biochem. Pharmacol. 2008, 76, 726–737. [Google Scholar] [CrossRef]
- Mamber, S.W.; Krakowka, S.; Osborn, J.; Saberski, L.; Rhodes, R.G.; Dahlberg, A.E.; Pond-Tor, S.; Fitzgerald, K.; Wright, N.; Beseme, S. Can Unconventional Immunomodulatory Agents Help Alleviate COVID-19 Symptoms and Severity? Msphere 2020, 5, e00288-20. [Google Scholar] [CrossRef] [PubMed]
- Sexton, M. Cannabis in the Time of Coronavirus Disease 2019: The Yin and Yang of the Endocannabinoid System in Immunocompetence. J. Altern. Complement. Med. 2020, 26, 444–448. [Google Scholar] [CrossRef]
| Receptor | CBD Activity | Receptor | CBD Activity |
|---|---|---|---|
| CB1 | Noncompetitive antagonist, negative allosteric modulator | 5-HT1A | Agonist |
| CB2 | Antagonist, inverse agonist | 5-HT2A | Partial agonist |
| GPR55 | Antagonist | 5-HT3A | Antagonist |
| GPR18 | Antagonist | A1A | Agonist |
| PPAR-γ | Agonist | channel TRPV1, TRPV2 | Agonist |
| TRPM8 | Antagonist | TRPA1 | Antagonist |
| α1, α3 glycine receptors | Agonist |
| Receptor | Δ9-THC Activity | Receptor | Δ9-THC Activity |
|---|---|---|---|
| CB1 | Partial agonist | opioid receptors | Allosteric modulator |
| CB2 | Partial agonist | PPAR-γ | Agonist |
| GPR55 | Agonist | TRPV2, TRPV3, TRPV4 | Agonist |
| GPR18 | Agonist | TRPA1 | Agonist |
| 5-HT3A | Antagonist | TRPM8 | Antagonist |
| Cannabinoids | Terpenes | Use |
|---|---|---|
| CBD | limonene, linalool, pinene | acne treatments |
| CBG | pinene | antiseptics |
| CBD | limonene, linalool | anxiety disorders |
| Extract CBD/THC 1:1 | caryophyllene, linalool, myrcene | sleep disorders |
| Test Group | Control Group | |
|---|---|---|
| Number of patients | 61 | 59 |
| Applied treatment | 20 mg/kg body weight of cannabidiol daily | Placebo |
| Number of seizures per month after four weeks of standard treatment | 12.4 | 14.9 |
| Number of seizures after 14 weeks of study treatment | 5.9 | 14.1 |
| The proportion of patients in whom the number of seizures decreased by at least 50% | 43% | 27% |
| Percentage of patients who were completely seizure-free | 5% | 0% |
| The Form | Effects on Symptoms |
|---|---|
| smoked marijuana | spasticity ↓, pain ↓ |
| nabiximols | spasticity ↓, pain ↓ |
| oral Cannabis extract | muscle spasticity ↓ |
| dronabinol | spasticity ↓, pain ↓ |
| nabilone (+gabapentin) | pain ↓ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stasiłowicz, A.; Tomala, A.; Podolak, I.; Cielecka-Piontek, J. Cannabis sativa L. as a Natural Drug Meeting the Criteria of a Multitarget Approach to Treatment. Int. J. Mol. Sci. 2021, 22, 778. https://doi.org/10.3390/ijms22020778
Stasiłowicz A, Tomala A, Podolak I, Cielecka-Piontek J. Cannabis sativa L. as a Natural Drug Meeting the Criteria of a Multitarget Approach to Treatment. International Journal of Molecular Sciences. 2021; 22(2):778. https://doi.org/10.3390/ijms22020778
Chicago/Turabian StyleStasiłowicz, Anna, Anna Tomala, Irma Podolak, and Judyta Cielecka-Piontek. 2021. "Cannabis sativa L. as a Natural Drug Meeting the Criteria of a Multitarget Approach to Treatment" International Journal of Molecular Sciences 22, no. 2: 778. https://doi.org/10.3390/ijms22020778
APA StyleStasiłowicz, A., Tomala, A., Podolak, I., & Cielecka-Piontek, J. (2021). Cannabis sativa L. as a Natural Drug Meeting the Criteria of a Multitarget Approach to Treatment. International Journal of Molecular Sciences, 22(2), 778. https://doi.org/10.3390/ijms22020778







