Recent Progress Regarding the Molecular Aspects of Insect Gall Formation
Abstract
:1. Introduction
2. Significance of Morphology and Function of Galls for Insects
3. Benefits of Gall Formation for Plants
4. Molecular Biology in Host Plants in Insect Gall Formation
4.1. Changes in Plant Hormonal Regulation during Gall Development
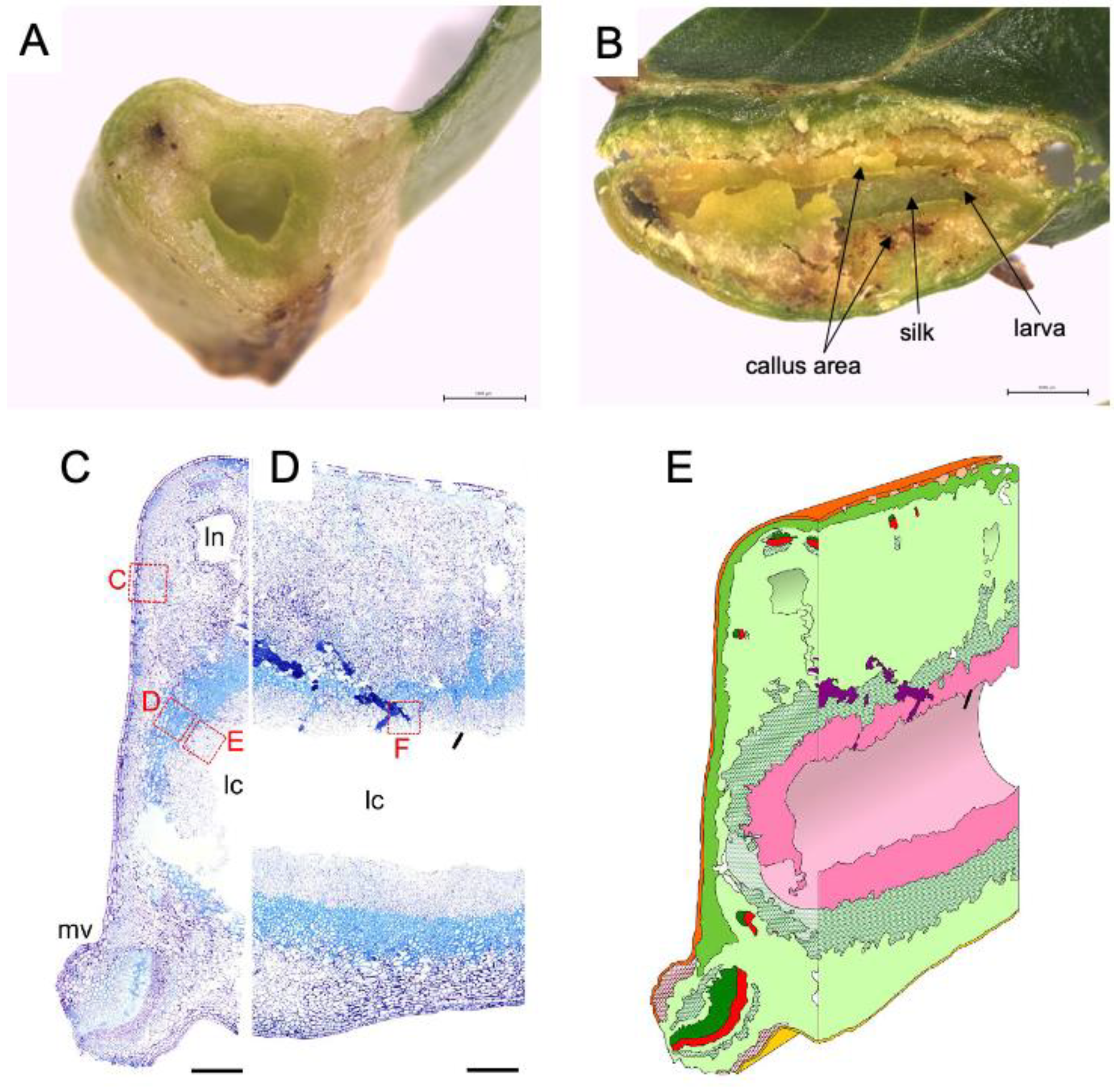
4.2. Attempts to Identification of Effector Molecules Involved in the Gall Formation
4.3. Changes in the Expression Patterns of Genes Involved in the Biosynthesis of the Metabolic Process during Gall Formation
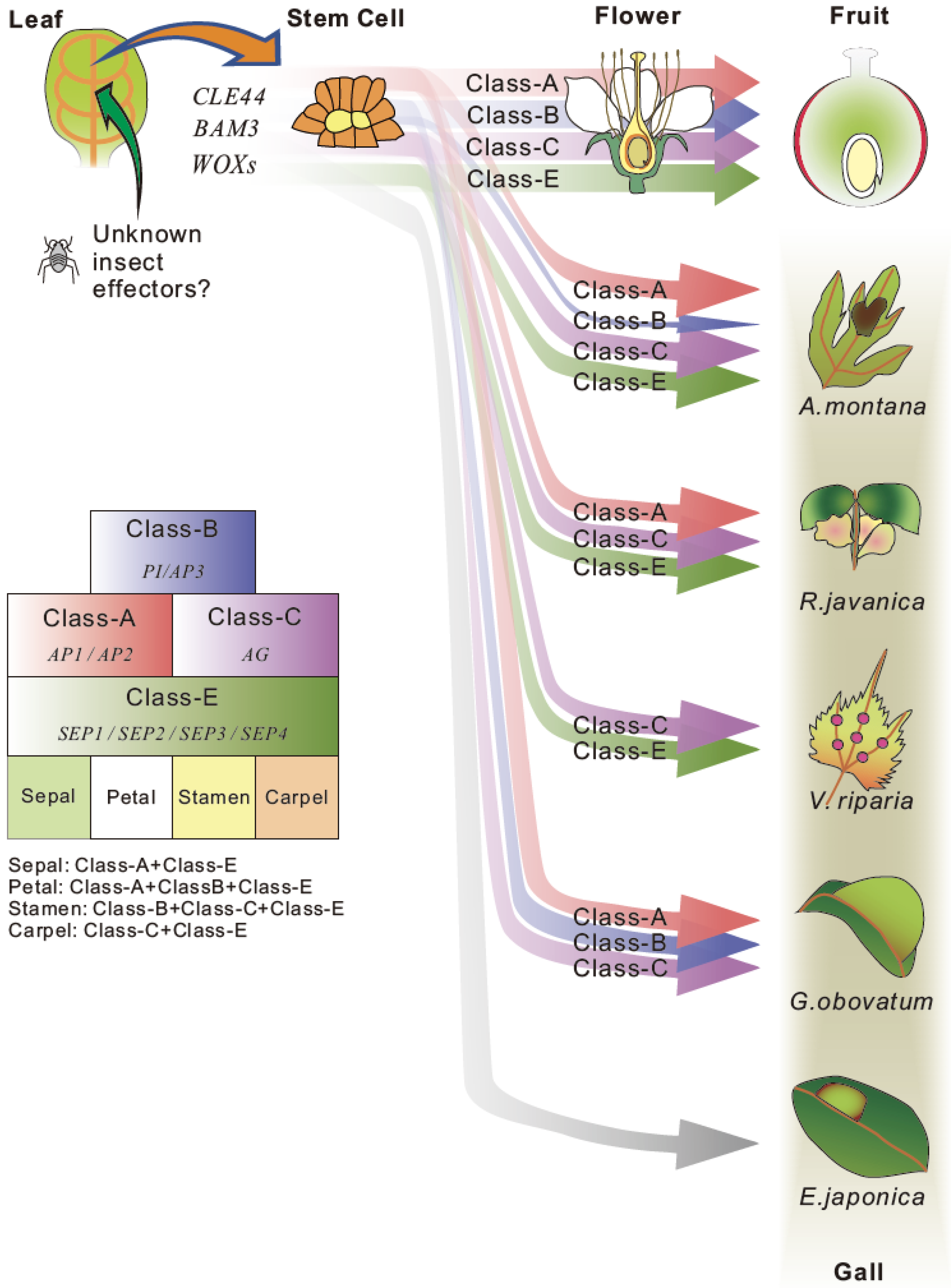
4.4. Regulation of Transcriptional Factors for Reproductive Organ Development
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mani, M.S. Ecology of Plant Galls; Junk, W., Ed.; Spring: The Hague, The Netherland, 1964. [Google Scholar]
- Felt, E.P. Gall insects and their relations to plants. Sci. Mon. 1918, 6, 509–525. [Google Scholar]
- Price, P.W.; Fernandes, G.W.; Waring, G.L. Adaptive nature of insect galls. Environ. Entomol. 1987, 16, 15–24. [Google Scholar] [CrossRef] [Green Version]
- Espirito-Santo, M.M.; Fernandes, G.W. How many species of gall-inducing insects are there on earth, and where are they? Ann. Entomol. Soc. Am. 2007, 100, 95–99. [Google Scholar]
- Giron, D.; Huguet, E.; Stone, G.N.; Body, M. Insect-induced effects on plants and possible effectors used by galling and leaf-mining insects to manipulate their host-plant. J. Insect Physiol. 2016, 84, 70–89. [Google Scholar] [CrossRef]
- Stone, G.N.; Schönrogge, K. The adaptive significance of insect gall morphology. Trends Ecol. Evol. 2003, 18, 512–522. [Google Scholar] [CrossRef]
- Takeda, S.; Yoza, M.; Amano, T.; Ohshima, I.; Hirano, T.; Sato, M.H.; Sakamoto, T.; Kimura, S. Comparative transcriptome analysis of galls from four different host plants suggests the molecular mechanism of gall development. PLoS ONE 2019, 14, e0223686. [Google Scholar]
- Yamaguchi, H.; Tanaka, H.; Hasegawa, M.; Tokuda, M.; Asami, T.; Suzuki, Y. Phytohormones and willow gall induction by a gall-inducing sawfly. New Phytol. 2012, 196, 586–595. [Google Scholar] [CrossRef] [PubMed]
- Tooker, J.F.; De Moraes, C.M. Feeding by a gall-inducing caterpillar species alters levels of indole-3-acetic and abscisic acid in Solidago altissima (Asteraceae) stems. Arthropod-Plant Inte. 2011, 5, 115–124. [Google Scholar] [CrossRef]
- Andreas, P.; Kisiala, A.; Neil Emery, R.J.; De Clerck-Floate, R.; Tooker, J.F.; Price, P.W.; Miller, D.G.; Chen, M.S.; Connor, E.F. Cytokinins are abundant and widespread among insect species. Plants 2020, 9, 208. [Google Scholar] [CrossRef] [Green Version]
- Ponce, G.E.; Fuse, M.; Chan, A.; Connor, E.F. The localization of phytohormones within the gall-inducing insect Eurosta solidaginis (Diptera: Tephritidae). Arthropod Plant Interact. 2021, 15, 375–385. [Google Scholar] [CrossRef]
- Tanaka, Y.; Okada, K.; Asami, T.; Suzuki, Y. Phytohormones in japanese mugwort gall induction by a gall-inducing gall midge. Biosci. Biotechnol. Biochem. 2013, 77, 1942–1948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barnewall, E.C.; De Clerck-Floate, R.A. A preliminary histological investigation of gall induction in an unconventional galling system. Arthropod-Plant Interact. 2012, 6, 449–459. [Google Scholar] [CrossRef]
- Koyama, Y.; Yao, I.; Akimoto, S.I. Aphid galls accumulate high concentrations of amino acids: A support for the nutrition hypothesis for gall formation. Entomol. Exp. Appl. 2004, 113, 35–44. [Google Scholar] [CrossRef]
- Suzuki, D.K.; Fukushi, Y.; Akimoto, S.I. Do aphid galls provide good nutrients for the aphids?: Comparisons of amino acid concentrations in galls among Tetraneura species (Aphididae: Eriosomatinae). Arthropod. Plant. Interact. 2009, 3, 241–247. [Google Scholar] [CrossRef]
- Larson, K.C.; Whitham, T.G. Manipulation of food resources by a gall-forming aphid: The physiology of sink-source interactions. Oecologia 1991, 88, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Larson, K.C.; Whitham, T.G. Competition between gall aphids and natural plant sinks: Plant architecture affects resistance to galling. Oecologia 1997, 109, 575–582. [Google Scholar] [CrossRef]
- Guiguet, A.; Ohshima, I.; Takeda, S.; Laurans, F.; Lopez-Vaamonde, C.; Giron, D. Origin of gall-inducing from leaf- mining in Caloptilia micromoths. Sci. Rep. 2019, 9, 6794. [Google Scholar] [CrossRef] [Green Version]
- Guiguet, A.; Hamatani, A.; Amano, T.; Takeda, S.; Lopez-vaamonde, C.; Giron, D.; Ohshima, I. Inside the horn of plenty: Leaf-mining micromoth manipulates its host plant to obtain unending food provisioning. PLoS ONE 2018, 13, e0209485. [Google Scholar] [CrossRef]
- Kawahara, A.Y.; Plotkin, D.; Ohshima, I.; Lopez-Vaamonde, C.; Houlihan, P.R.; Breinholt, J.W.; Kawakita, A.; Xiao, L.; Regier, J.C.; Davis, D.R.; et al. A molecular phylogeny and revised higher-level classification for the leaf-mining moth family Gracillariidae and its implications for larval host-use evolution. Syst. Entomol. 2017, 42, 60–81. [Google Scholar] [CrossRef]
- Li, X.; Ryan, S.L.; Chandra, E.; Camiel, D.; van Erik, N.; Don, D.; Chris, J.; Atsushi, K.; Shigeki, K.; Andreas, Z.; et al. Phylogeny of gracillariid leaf-mining moths: Evolution of larval behaviour inferred from integrated phylogenomic and Sanger data. Cladistics 2021, in press. [Google Scholar]
- Hawkins, B.A.; Cornell, H.V.; Hochberg, M.E. Predators, parasitoids, and pathogens as mortality agents in phytophagous insect populations. Ecology 1997, 78, 2145–2152. [Google Scholar] [CrossRef]
- Inbar, M.; Izhaki, I.; Koplovich, A.; Lupo, I.; Silanikove, N.; Glasser, T.; Gerchman, Y.; Perevolotsky, A.; Lev-Yadun, S. Why do many galls have conspicuous colors? A new hypothesis. Arthropod. Plant. Interact. 2010, 4, 1–6. [Google Scholar] [CrossRef]
- Price, P.W.; Pschorn-Walcher, H. Are gall-inducing insects better protected against parasitoids than exposed feeders? A test using tenthredinid sawflies. Ecol. Entomol. 1988, 13, 195–205. [Google Scholar] [CrossRef]
- Cornell, H.V. The secondary chemistry and complex morphology of galls formed by the Cynipiae (Hymenoptera): Why and How? Am. Midl. Nat. 1983, 110, 225–234. [Google Scholar] [CrossRef]
- Blanche, K.R. Diversity of insect-induced galls along a temperature-rainfall gradient in the tropical savannah region of the Northern Territory, Australia. Austral Ecol. 2000, 25, 311–318. [Google Scholar] [CrossRef]
- Kutsukake, M.; Shibao, H.; Uematsu, K.; Fukatsu, T. Scab formation and wound healing of plant tissue by soldier aphid. Proc. R. Soc. B Biol. Sci. 2009, 276, 1555–1563. [Google Scholar] [CrossRef] [Green Version]
- Fujii, T.; Matsuo, K.; Abe, Y.; Yukawa, J.; Tokuda, M. An endoparasitoid avoids hyperparasitism by manipulating immobile host herbivore to modify host plant morphology. PLoS ONE 2014, 9, e102508. [Google Scholar] [CrossRef] [Green Version]
- Connor, E.F.; Bartlett, L.; O’Toole, S.; Byrd, S.; Biskar, K.; Orozco, J. The mechanism of gall induction makes galls red. Arthropod. Plant. Interact. 2012, 6, 489–495. [Google Scholar] [CrossRef]
- White, T.C.R. Why do many galls have conspicuous colours? An alternative hypothesis revisited. Arthropod. Plant. Interact. 2010, 4, 149–150. [Google Scholar] [CrossRef]
- Kutsukake, M.; Meng, X.Y.; Katayama, N.; Nikoh, N.; Shibao, H.; Fukatsu, T. An insect-induced novel plant phenotype for sustaining social life in a closed system. Nat. Commun. 2012, 3, 1187. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Yang, Z.; Chen, H.; Qi, Q.; Liu, J.; Wang, C.; Shao, S.; Lu, Q.; Li, Y.; Wu, H.; et al. A Complex nutrient exchange between a gall-forming aphid and its plant host. Front. Plant Sci. 2020, 11, 811. [Google Scholar] [CrossRef]
- Murakami, R.; Ushima, R.; Sugimoto, R.; Tamaoki, D.; Karahara, I.; Hanba, Y.; Wakasugi, T.; Tsuchida, T. A new galling insect model enhances photosynthetic activity in an obligate holoparasitic plant. Sci. Rep. 2021, 11, 13013. [Google Scholar] [CrossRef]
- Fay, P.A.; Hartnett, D.C.; Knapp, A.K. Increased photosynthesis and water potentials in Silphium integrifolium galled by cynipid wasps. Oecologia 1993, 93, 114–120. [Google Scholar] [CrossRef]
- Rocha, S.; Branco, M.; Boas, L.V.; Almeida, M.H.; Protasov, A.; Mendel, Z. Gall induction may benefit host plant: A case of a gall wasp and eucalyptus tree. Tree Physiol. 2013, 33, 388–397. [Google Scholar] [CrossRef] [Green Version]
- Matsukura, K.; Matsumura, M.; Tokuda, M. Host manipulation by the orange leafhopper Cicadulina bipunctata: Gall induction on distant leaves by dose-dependent stimulation. Naturwissenschaften 2009, 96, 1059–1066. [Google Scholar] [CrossRef]
- Stuart, J.J.; Chen, M.-S.; Shukle, R.; Harris, M.O. Gall midges (Hessian Flies) as plant pathogens. Annu. Rev. Phytopathol. 2012, 50, 339–357. [Google Scholar] [CrossRef]
- Dorchin, N.; Hoffmann, J.H.; Stirk, W.A.; Novák, O.; Strnad, M.; Van Staden, J.; Ond, R. Sexually dimorphic gall structures correspond to differential phytohormone contents in male and. Physiol. Entomol. 2009, 34, 359–369. [Google Scholar] [CrossRef]
- Bailey, S.; Percy, D.M.; Hefer, C.A.; Cronk, Q.C.B. The transcriptional landscape of insect galls: Psyllid (Hemiptera) gall formation in Hawaiian Metrosideros polymorpha (Myrtaceae). BMC Genom. 2015, 16, 943. [Google Scholar] [CrossRef] [Green Version]
- Hirano, T.; Kimura, S.; Sakamoto, T.; Okamoto, A.; Nakayama, T.; Matsuura, T.; Ikeda, Y.; Takeda, S.; Suzuki, Y.; Ohshima, I.; et al. Reprogramming of the developmental program of Rhus javanica during initial stage of gall induction by Schlechtendalia chinensis. Front. Plant Sci. 2020, 11, 471. [Google Scholar] [CrossRef]
- Wang, H.; Cui, K.; Shao, S.; Liu, J.; Chen, H.; Wang, C.; Yang, Z.; Lu, Q.; King-jones, K.; Chen, X.; et al. Molecular response of gall induction by aphid Schlechtendalia chinensis (Bell) attack on Rhus chinensis Mill. J. Plant Interact. 2017, 12, 465–479. [Google Scholar] [CrossRef] [Green Version]
- Shin, R.; Burch, A.Y.; Huppert, K.A.; Tiwari, S.B.; Murphy, A.S.; Guilfoyle, T.J.; Schachtman, D.P. The Arabidopsis transcription factor MYB77 modulates auxin signal transduction. Plant Cell 2007, 19, 2440–2453. [Google Scholar] [CrossRef] [Green Version]
- Sugimoto, K.; Jiao, Y.; Meyerowitz, E.M. Arabidopsis regeneration from multiple tissues occurs via a root development pathway. Dev. Cell 2010, 18, 463–471. [Google Scholar] [CrossRef] [Green Version]
- Rani, D.D.; Pedada, S.; Singh, R. Hessian fly: New insights to develop a gall nutritive tissue. J. Entomol. Zool. Stud. 2018, 6, 720–728. [Google Scholar]
- Chen, M.S.; Zhao, H.X.; Zhu, Y.C.; Scheffler, B.; Liu, X.; Liu, X.; Hulbert, S.; Stuart, J.J. Analysis of transcripts and proteins expressed in the salivary glands of Hessian fly (Mayetiola destructor) larvae. J. Insect Physiol. 2008, 54, 1–16. [Google Scholar] [CrossRef]
- Chen, M.S.; Liu, X.; Yang, Z.; Zhao, H.; Shukle, R.H.; Stuart, J.J.; Hulbert, S. Unusual conservation among genes encoding small secreted salivary gland proteins from a gall midge. BMC Evol. Biol. 2010, 10, 296. [Google Scholar] [CrossRef] [Green Version]
- Zhao, C.; Escalante, L.N.; Chen, H.; Benatti, T.R.; Qu, J.; Chellapilla, S.; Waterhouse, R.M.; Wheeler, D.; Andersson, M.N.; Bao, R.; et al. A massive expansion of effector genes underlies gall-formation in the wheat pest Mayetiola destructor. Curr. Biol. 2015, 25, 613–620. [Google Scholar] [CrossRef]
- Aggarwal, R.; Subramanyam, S.; Zhao, C.; Chen, M.; Harris, M.O.; Stuart, J.J. Avirulence effector discovery in a plant galling and plant parasitic arthropod, the Hessian fly (Mayetiola destructor). PLoS ONE 2014, 9, e100958. [Google Scholar] [CrossRef] [Green Version]
- Zhao, C.; Shukle, R.; Navarro-Escalante, L.; Chen, M.; Richards, S.; Stuart, J.J. Avirulence gene mapping in the Hessian fly (Mayetiola destructor) reveals a protein phosphatase 2C effector gene family. J. Insect Physiol. 2016, 84, 22–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cambier, S.; Ginis, O.; Moreau, S.J.M.; Gayral, P.; Hearn, J.; Stone, G.N.; Giron, D.; Huguet, E.; Drezen, J.M. Gall wasp transcriptomes unravel potential effectors involved in molecular dialogues with oak and rose. Front. Physiol. 2019, 10, 926. [Google Scholar] [CrossRef] [PubMed]
- Eitle, M.W.; Carolan, J.C.; Griesser, M.; Forneck, A. The salivary gland proteome of root-galling grape phylloxera (Daktulosphaira vitifoliae Fitch) feeding on Vitis spp. PLoS ONE 2019, 14, e0225881. [Google Scholar] [CrossRef]
- Korgaonkar, A.; Han, C.; Lemire, A.L.; Siwanowicz, I.; Bennouna, D.; Kopec, R.E.; Andolfatto, P.; Shigenobu, S.; Stern, D.L. A novel family of secreted insect proteins linked to plant gall development. Curr. Biol. 2021, 31, 1836–1849.e12. [Google Scholar] [CrossRef]
- Doss, R.P.; Oliver, J.E.; Proebsting, W.M.; Potter, S.W.; Kuy, S.; Clement, S.L.; Williamson, R.T.; Carney, J.R.; DeVilbiss, E.D. Bruchins: Insect-derived plant regulators that stimulate neoplasm formation. Proc. Natl. Acad. Sci. USA 2000, 97, 6218–6223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, H.; Liu, J.; Cui, K.; Lu, Q.; Wang, C.; Wu, H.; Yang, Z.; Ding, W.; Shao, S.; Wang, H.; et al. Molecular mechanisms of tannin accumulation in Rhus galls and genes involved in plant-insect interactions. Sci. Rep. 2018, 8, 9841. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Shi, F.; Chen, Y.; Wang, M.; Zhao, Y.; Geng, G. Transcriptome analysis of Chinese chestnut (Castanea mollissima Blume) in response to Dryocosmus kuriphilus Yasumatsu infestation. Int. J. Mol. Sci. 2019, 20, 855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, H.B.; Jin, L.P.; Wei, D.; Huang, Z.H. Study on the differential gene expression of elm leaves fed on by Tetraneura akinire Sasaki. Genes Genom. 2019, 41, 1505–1516. [Google Scholar] [CrossRef] [PubMed]
- Darwin, C.R. The Variation of Animals and Plants under Domestication Volumen I; John Murray: London, UK, 1875. [Google Scholar]
- Rohfritsch, O.; Shorthouse, J.D. Insect galls. In Molecular Biology of Plant Tumors; Kahl, G., Schell, J., Eds.; Academic Press: New York, NY, USA, 1982; pp. 121–152. [Google Scholar]
- Kurosu, U.; Aoki, S. Formation of a “Cat’s-paw” gall by the aphid Ceratovacuna nekoashi (Homoptera). Jpn. J. Entomol. 1990, 58, 155–166. [Google Scholar]
- Schultz, J.C.; Edger, P.P.; Body, M.J.A.; Appel, H.M. A galling insect activates plant reproductive programs during gall development. Sci. Rep. 2019, 9, 1833. [Google Scholar] [CrossRef] [Green Version]
- Lincoln, C.; Long, J.; Yamaguchi, J.; Serikawa, K.; Hakeaibi, S. A knotted1-like homeobox gene in Arabidopsis is expressed in the vegetative meristem and dramatically alters leaf morphology when overexpressed in transgenic plants. Plant Cell 1994, 6, 1859–1876. [Google Scholar]
- Jun, J.H.; Fiume, E.; Fletcher, J.C. The CLE family of plant polypeptide signaling molecules. Cell. Mol. Life Sci. 2008, 65, 743–755. [Google Scholar] [CrossRef]
- Yamaguchi, Y.L.; Ishida, T.; Sawa, S. CLE peptides and their signaling pathways in plant development. J. Exp. Bot. 2016, 67, 4813–4826. [Google Scholar] [CrossRef] [Green Version]
- Ó’Maoiléidigh, D.S.; Graciet, E.; Wellmer, F. Gene networks controlling Arabidopsis thaliana flower development. New Phytol. 2014, 201, 16–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takeda, S.; Hirano, T.; Ohshima, I.; Sato, M.H. Recent Progress Regarding the Molecular Aspects of Insect Gall Formation. Int. J. Mol. Sci. 2021, 22, 9424. https://doi.org/10.3390/ijms22179424
Takeda S, Hirano T, Ohshima I, Sato MH. Recent Progress Regarding the Molecular Aspects of Insect Gall Formation. International Journal of Molecular Sciences. 2021; 22(17):9424. https://doi.org/10.3390/ijms22179424
Chicago/Turabian StyleTakeda, Seiji, Tomoko Hirano, Issei Ohshima, and Masa H. Sato. 2021. "Recent Progress Regarding the Molecular Aspects of Insect Gall Formation" International Journal of Molecular Sciences 22, no. 17: 9424. https://doi.org/10.3390/ijms22179424
APA StyleTakeda, S., Hirano, T., Ohshima, I., & Sato, M. H. (2021). Recent Progress Regarding the Molecular Aspects of Insect Gall Formation. International Journal of Molecular Sciences, 22(17), 9424. https://doi.org/10.3390/ijms22179424





