Genome-Wide Identification and Expression Profiling Analysis of WOX Family Protein-Encoded Genes in Triticeae Species
Abstract
:1. Introduction
2. Results
2.1. Identification of WOX Genes in Triticeae Species
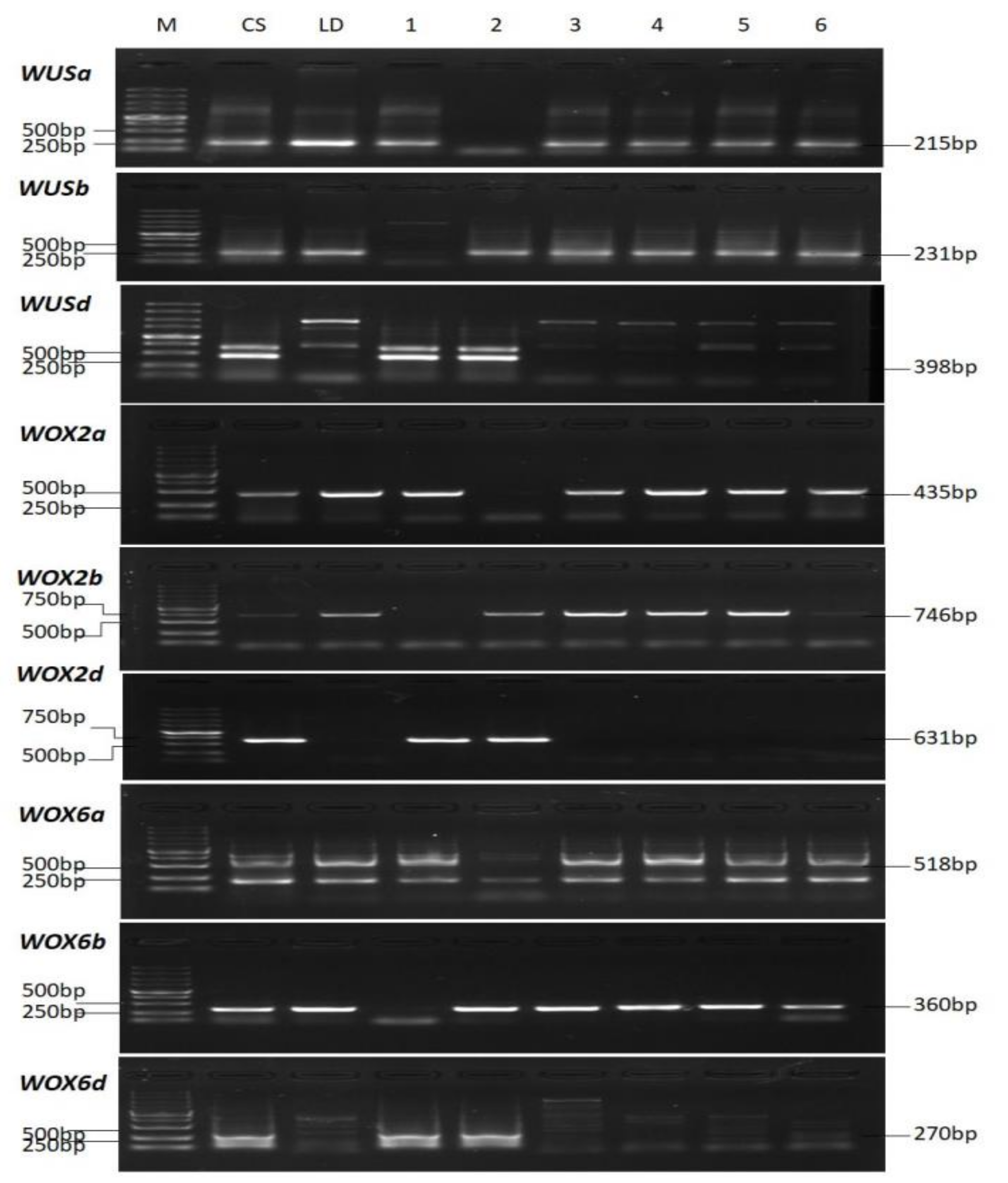
2.2. Identification of WUS Homoeologous Genes in Triticeae Species
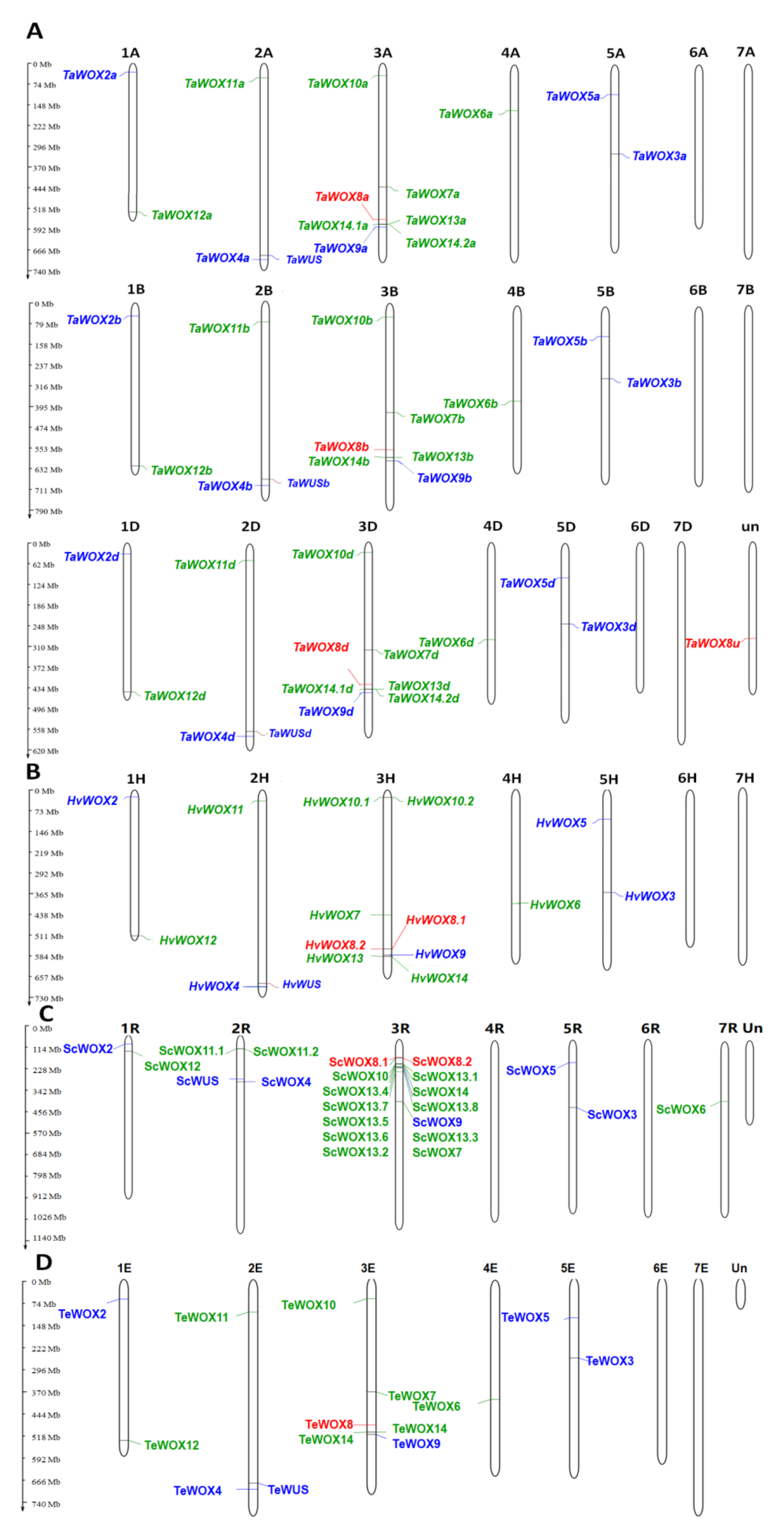
2.3. Chromosomal Location of WOX Genes in Triticeae Species
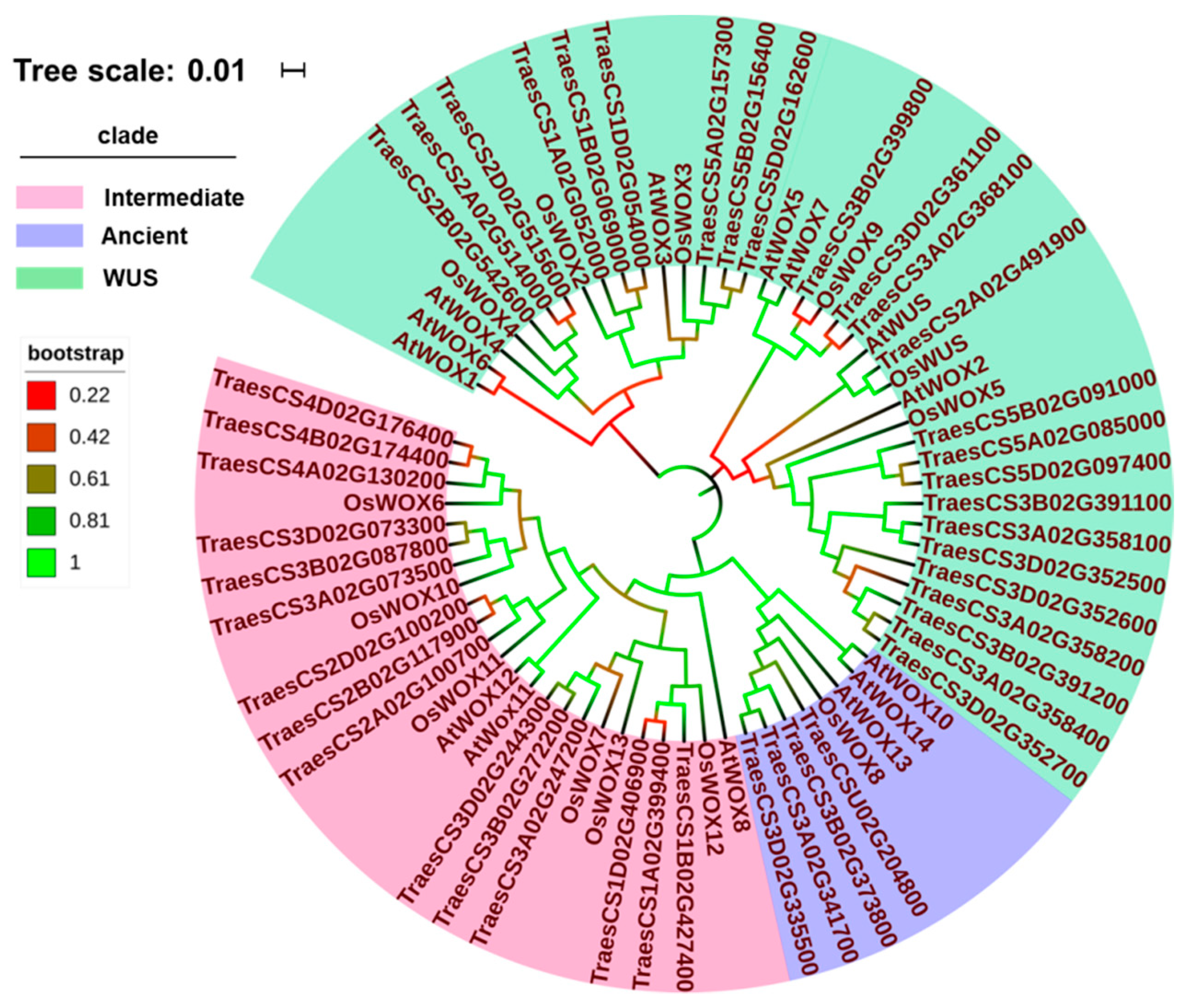
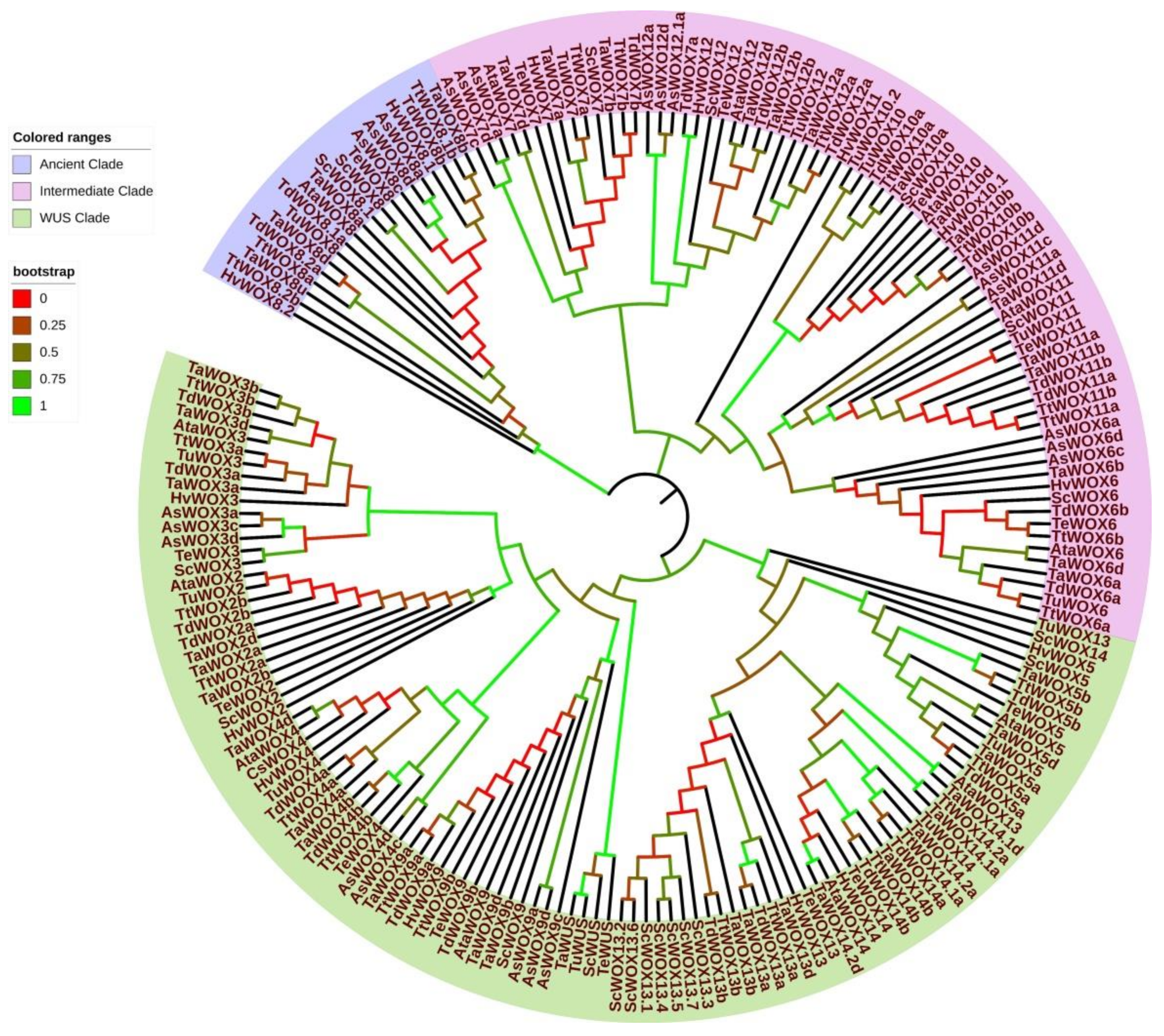
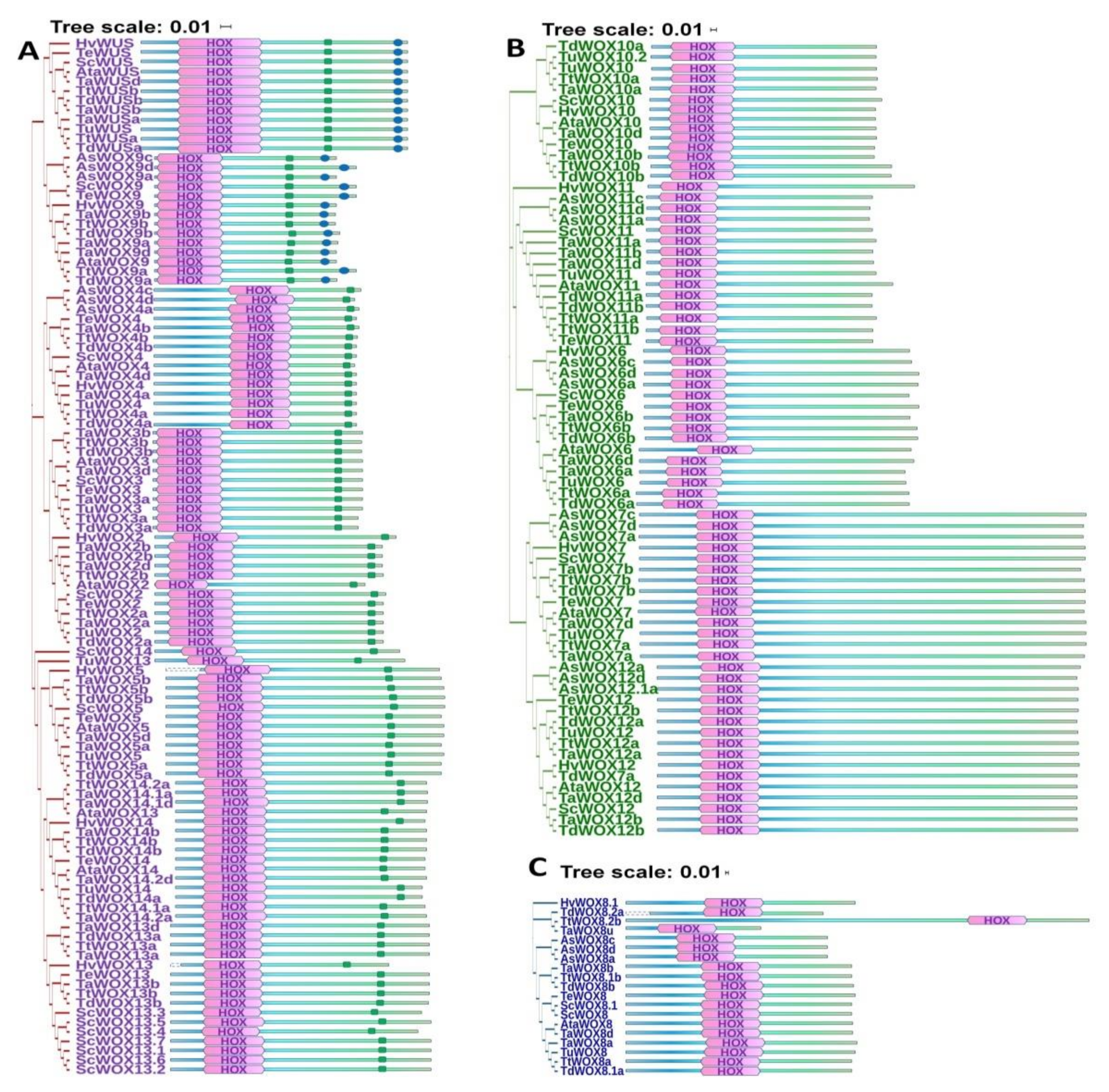
2.4. Evolution of WOX Family Proteins in Triticeae Species
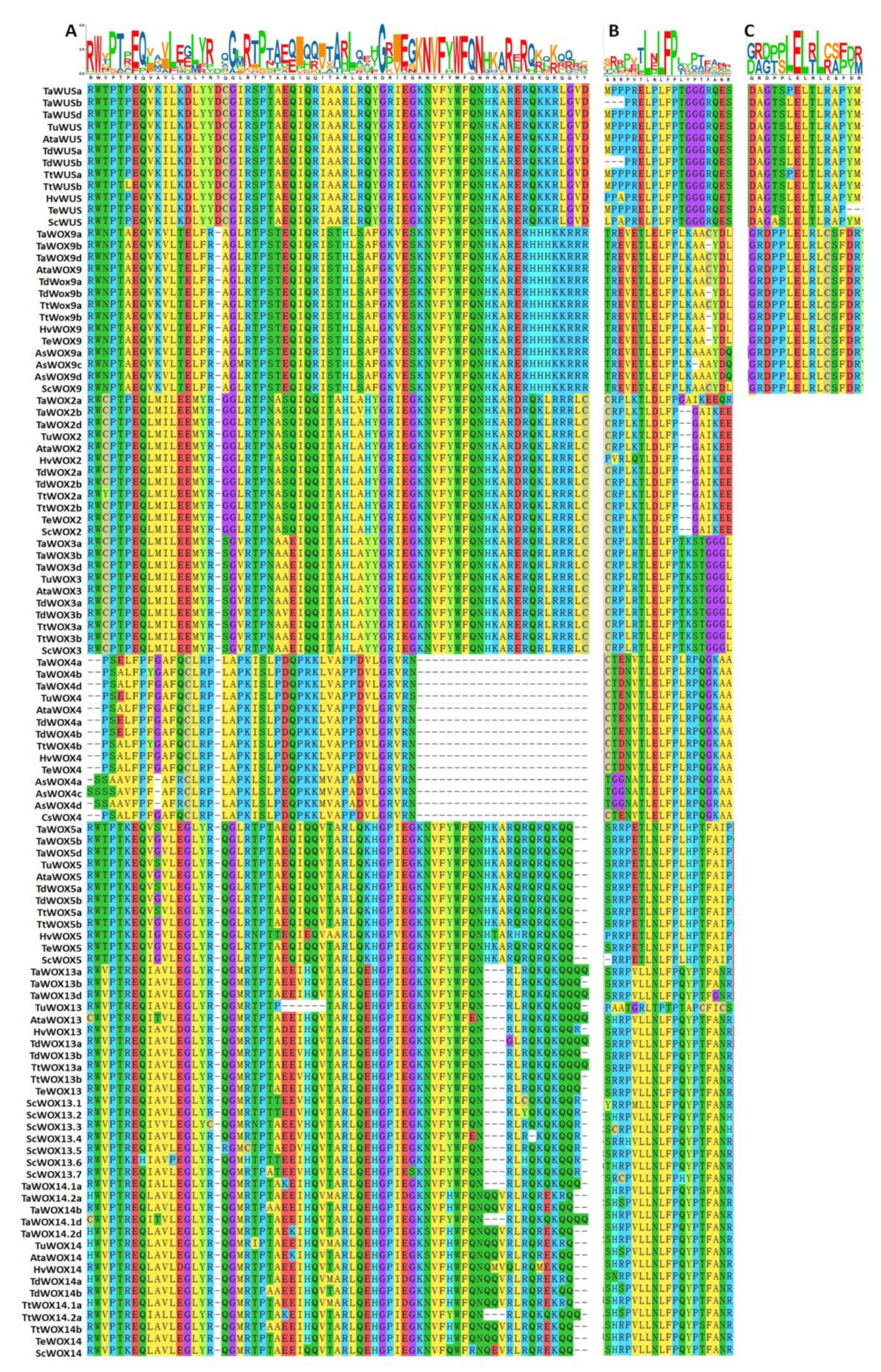
2.5. Analysis of the Conserved Motifs of WOX Proteins in Triticeae Species
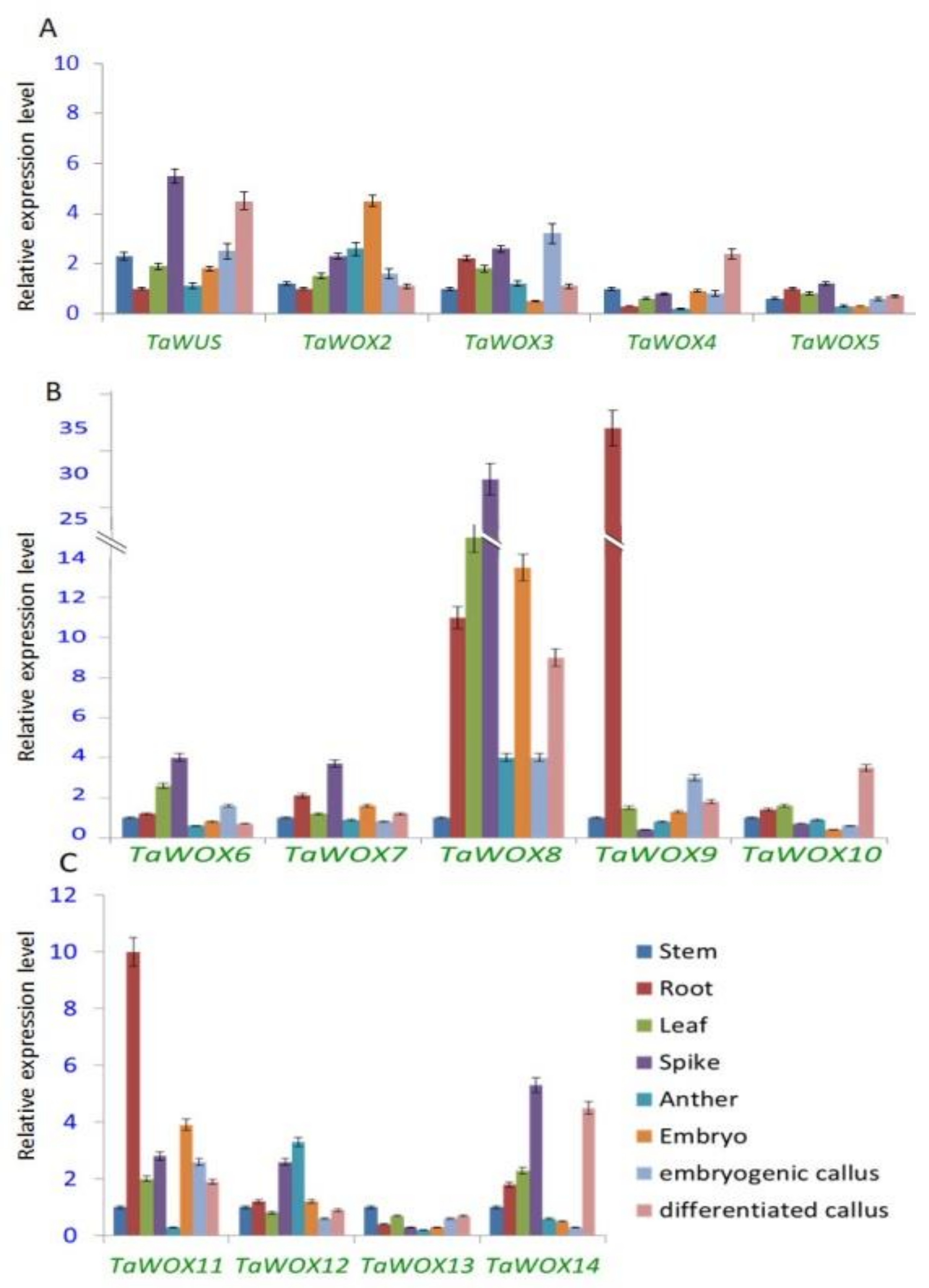
2.6. Expression Patterns of TaWOX Genes in Various Wheat Tissues
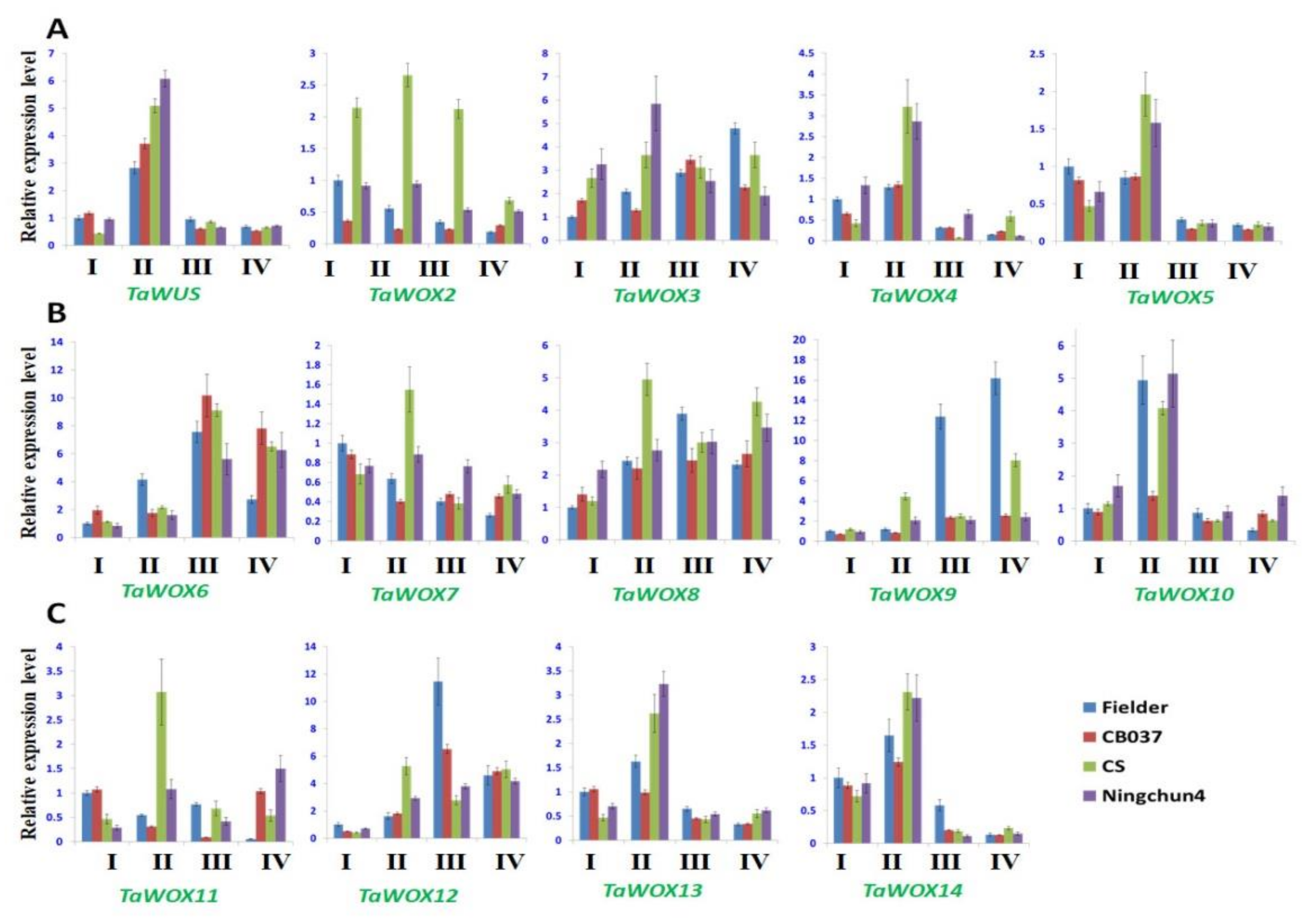
2.7. Expression Patterns of TaWOX Genes during Wheat Callus Proliferation
3. Discussion
4. Materials and Methods
4.1. Materials and Cultivation Conditions
4.2. Database Used for Searching WOX Family Genes in Triticeae Plants
4.3. Phylogenetic Trees Construction
4.4. Conserved Protein Motif Analysis
4.5. DNA Isolation and PCR Analysis
4.6. Callus Induction from Wheat Immature Embryos
4.7. RNA Isolation and qPCR Analysis
4.8. Expression Analysis of TaWOX Genes Using RNA-seq Data
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Feuillet, C.; Muehlbauer, G.J. Genetics and Genomics of the Triticeae; Springer: New York, NY, USA, 2009; p. 31. [Google Scholar]
- FAOSTAT. Available online: http://www.fao.org/faostat/en/ (accessed on 10 May 2021).
- IWGSC. Shifting the limits in wheat research and breeding using a fully annotated reference genome. Science 2018, 361, eaar7191. [Google Scholar] [CrossRef] [Green Version]
- Singh, R.K.; Prasad, M. Advances in Agrobacterium tumefaciens-mediated genetic transformation of graminaceous crops. Protoplasma 2016, 253, 691–707. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Gong, Q.; Ye, X. Recent developments and applications of genetic transformation and genome editing technologies in wheat. Theor. Appl. Genet. 2020, 133, 1603–1622. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Shirley, S.; Xu, H.; Du, L.; Clement, T. Regular production of transgenic wheat mediated by Agrobacterium tumefaciens. Agric. Sci. China 2002, 1, 239–244. [Google Scholar]
- Wang, K.; Liu, H.; Du, L.; Ye, X. Generation of marker-free transgenic hexaploid wheat via an Agrobacterium-mediated cotransformation strategy in commercial Chinese wheat varieties. Plant Biotechnol. J. 2017, 15, 614–623. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Mamrutha, H.M.; Kaur, A.; Venkatesh, K.; Grewal, A.; Kumar, R.; Tiwari, V. Development of an efficient and reproducible regeneration system in wheat (Triticum aestivum L.). Physiol. Mol. Biol. Plants 2017, 23, 945–954. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Li, X.; Qiao, M.; Li, B.; Guo, D.; Zhang, X.; Min, D. TaTCP-1, a novel regeneration-related gene involved in the molecular regulation of somatic embryogenesis in wheat (Triticum aestivum L.). Front. Plant Sci. 2020, 11, 1004. [Google Scholar] [CrossRef]
- Kareem, A.; Durgaprasad, K.; Sugimoto, K.; Du, Y.; Pulianmackal, A.J.; Trivedi, Z.B.; Abhayadev, P.V.; Pinon, V.; Meyerowitz, E.M.; Scheres, B.; et al. PLETHORA Genes Control Regeneration by a Two-Step Mechanism. Curr. Biol. 2015, 25, 1017–1030. [Google Scholar] [CrossRef] [Green Version]
- Kumar, V.; Jha, P.; van Staden, J. LEAFY COTYLEDONs (LECs): Master regulators in plant embryo development. Plant Cell Tissue Org. 2020, 140, 475–487. [Google Scholar] [CrossRef]
- Pan, J.; Zhao, F.; Zhang, G.; Pan, Y.; Sun, L.; Bao, N.; Qin, P.; Chen, L.; Yu, J.; Zhang, Y.; et al. Control of de novo root regeneration efficiency by developmental status of Arabidopsis leaf explants. J. Genet. Genomics 2019, 46, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Hofhuis, H.; Heidstra, R.; Sauer, M.; Friml, J.; Scheres, B. A molecular framework for plant regeneration. Science 2006, 311, 386–388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, L. De novo root regeneration from leaf explants: Wounding, auxin, and cell fate transition. Curr. Opin. Plant Biol. 2018, 41, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Debernardi, J.M.; Tricoli, D.M.; Ercoli, M.F.; Hayta, S.; Ronald, P.; Palatnik, J.F.; Dubcovsky, J. A GRF–GIF chimeric protein improves the regeneration efficiency of transgenic plants. Nat. Biotechnol. 2020, 38, 1274–1279. [Google Scholar] [CrossRef] [PubMed]
- Lowe, K.; Wu, E.; Wang, N.; Hoerster, G.; Hastings, C.; Cho, M.J.; Scelonge, C.; Lenderts, B.; Chamberlin, M.; Cushatt, J.; et al. Morphogenic regulators Baby boom and Wuschel improve monocot transformation. Plant Cell 2016, 28, 1998–2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lowe, K.; La Rota, M.; Hoerster, G.; Hastings, C.; Wang, N.; Chamberlin, M.; Wu, E.; Jones, T.; Gordon-Kamm, W. Rapid genotype “independent” Zea mays L. (maize) transformation via direct somatic embryogenesis. In Vitro Cell. Dev. Biol. Plant 2018, 54, 240–252. [Google Scholar] [CrossRef] [Green Version]
- Van der Graaff, E.; Laux, T.; Rensing, S.A. The WUS homeobox-containing (WOX) protein family. Genome Biol. 2009, 10, 248. [Google Scholar] [CrossRef] [PubMed]
- Gehring, W.J.; Muller, M.; Affolter, M.; Percival-Smith, A.; Billeter, M.; Qian, Y.Q.; Otting, G.; Wüthrich, K. The structure of the homeodomain and its functional implications. Trends Genet. 1990, 6, 323–329. [Google Scholar] [CrossRef]
- Gehring, W.J.; Qian, Y.Q.; Billeter, M.; Furukubo-Tokunaga, K.; Schier, A.F.; Resendez-Perez, D.; Affolter, M.; Otting, G.; Wüthrich, K. Homeodomain-DNA recognition. Cell 1994, 78, 211–223. [Google Scholar] [CrossRef]
- Breuninger, H.; Rikirsch, E.; Hermann, M.; Ueda, M.; Laux, T. Differential expression of WOX genes mediates apical-basal axis formation in the Arabidopsis embryo. Dev. Cell 2008, 14, 867–876. [Google Scholar] [CrossRef] [Green Version]
- Hao, Q.; Zhang, L.; Yang, Y.; Shan, Z.; Zhou, X.A. Genome-wide analysis of the WOX gene gamily and gunction exploration of GmWOX18 in soybean. Plants 2019, 8, 215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costanzo, E.; Trehin, C.; Vandenbussche, M. The role of WOX genes in flower development. Ann. Bot. 2014, 114, 1545–1553. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dolzblasz, A.; Nardmann, J.; Clerici, E.; Causier, B.; van der Graaff, E.; Chen, J.; Davies, B.; Werr, W.; Laux, T. Stem cell regulation by Arabidopsis WOX genes. Mol. Plant 2016, 9, 1028–1039. [Google Scholar] [CrossRef] [PubMed]
- Honda, E.; Yew, C.L.; Yoshikawa, T.; Sato, Y.; Hibara, K.; Itoh, J.I. LEAF LATERAL SYMMETRY1, a member of the WUSCHEL-RELATED HOMEOBOX3 gene family, regulates lateral organ development differentially from other paralogs, NARROW LEAF2 and NARROW LEAF3 in rice. Plant Cell Physiol. 2018, 59, 376–391. [Google Scholar] [CrossRef]
- Haecker, A.; Gross-Hardt, R.; Geiges, B.; Sarkar, A.; Breuninger, H.; Herrmann, M.; Laux, T. Expression dynamics of WOX genes mark cell fate decisions during early embryonic patterning in Arabidopsis thaliana. Development 2004, 131, 657–668. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Zong, J.; Liu, J.; Yin, J.; Zhang, D. Genome-wide analysis of WOX gene family in rice, sorghum, maize, Arabidopsis and poplar. J. Integr. Plant Biol. 2010, 52, 1016–1026. [Google Scholar] [CrossRef]
- Ma, Y.; Miotk, A.; Šutiković, Z.; Ermakova, O.; Wenzl, C.; Medzihradszky, A.; Gaillochet, C.; Forner, J.; Utan, G.; Brackmann, K.; et al. WUSCHEL acts as an auxin response rheostat to maintain apical stem cells in Arabidopsis. Nat. Commun. 2019, 10, 5093. [Google Scholar] [CrossRef] [PubMed]
- Jha, P.; Ochatt, S.J.; Kumar, V. WUSCHEL: A master regulator in plant growth signaling. Plant Cell Rep. 2020, 39, 431–444. [Google Scholar] [CrossRef]
- Zuo, J.; Niu, Q.W.; Frugis, G.; Chua, N.H. The WUSCHEL gene promotes vegetative-to-embryonic transition in Arabidopsis. Plant J. 2002, 30, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Gallois, J.L.; Nora, F.R.; Mizukami, Y.; Sablowski, R. WUSCHEL induces shoot stem cell activity and developmental plasticity in the root meristem. Gene Dev. 2004, 18, 375–380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, H.; Qu, X.; Dong, Z.; Luo, L.; Shao, C.; Forner, J.; Lohmann, J.U.; Su, M.; Xu, M.; Liu, X.; et al. WUSCHEL triggers innate antiviral immunity in plant stem cells. Science 2020, 370, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wu, R.; Qin, G.; Chen, Z.; Gu, H.; Qu, L. Over-expression of WOX1 leads to defects in meristem development and polyamine homeostasis in Arabidopsis. J. Integr. Plant Biol. 2011, 53, 493–506. [Google Scholar] [CrossRef]
- Shimizu, R.; Ji, J.; Kelsey, E.; Ohtsu, K.; Schnable, P.S.; Scanlon, M.J. Tissue specificity and evolution of meristematic WOX3 function. Plant Physiol. 2009, 149, 841–850. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.; Shang, G.; Wu, L.; Xu, Z.; Zhao, X.; Wang, J. Chromatin accessibility dynamics and a hierarchical transcriptional regulatory network structure for plant somatic embryogenesis. Dev. Cell 2020, 54, 742–757. [Google Scholar] [CrossRef] [PubMed]
- Suer, S.; Agusti, J.; Sanchez, P.; Schwarz, M.; Greb, T. WOX4 imparts auxin responsiveness to cambium cells in Arabidopsis. Plant Cell 2011, 23, 3247–3259. [Google Scholar] [CrossRef] [Green Version]
- Kong, X.; Lu, S.; Tian, H.; Ding, Z. WOX5 is shining in the root stem cell niche. Trends Plant Sci. 2015, 20, 601–603. [Google Scholar] [CrossRef] [PubMed]
- Park, S.O.; Zheng, Z.; Oppenheimer, D.G.; Hauser, B.A. The PRETTY FEW SEEDS2 gene encodes an Arabidopsis homeodomain protein that regulates ovule development. Development 2005, 132, 841–849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kong, D.; Hao, Y.; Cui, H. The WUSCHEL related Homeobox protein WOX7 regulates the sugar response of lateral root development in Arabidopsis thaliana. Mol. Plant 2016, 9, 261–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, X.; Dabi, T.; Weigel, D. Requirement of homeobox gene STIMPY/WOX9 for arabidopsis meristem growth and maintenance. Curr. Biol. 2005, 15, 436–440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, X.; Chory, J.; Weigel, D. Combinations of WOX activities regulate tissue proliferation during Arabidopsis embryonic development. Dev. Biol. 2007, 309, 306–316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ueda, M.; Zhang, Z.; Laux, T. Transcriptional activation of Arabidopsis axis patterning genes WOX8/9 links zygote polarity to embryo development. Dev. Cell 2011, 20, 264–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baesso, B.; Chiatante, D.; Terzaghi, M.; Zenga, D.; Nieminen, K.; Mahonen, A.P.; Siligato, R.; Helariutta, Y.; Scippa, G.S.; Montagnoli, A. Transcription factors PRE3 and WOX11 are involved in the formation of new lateral roots from secondary growth taproot in A. thaliana. Plant Biol. 2018, 20, 426–432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.; Sheng, L.; Xu, Y.; Li, J.; Yang, Z.; Huang, H.; Xu, L. WOX11 and 12 are involved in the first-step cell fate transition during de novo root organogenesis in Arabidopsis. Plant Cell 2014, 26, 1081–1093. [Google Scholar] [CrossRef] [Green Version]
- Hu, X.; Xu, L. Transcription factors WOX11/12 directly activate WOX5/7 to promote root primordia initiation and organogenesis. Plant Physiol. 2016, 172, 2363–2373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Romera-Branchat, M.; Ripoll, J.J.; Yanofsky, M.F.; Pelaz, S. The WOX13 homeobox gene promotes replum formation in the Arabidopsis thaliana fruit. Plant J. 2013, 73, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Denis, E.; Kbiri, N.; Mary, V.; Claisse, G.; Conde, E.S.N.; Kreis, M.; Deveaux, Y.; Silva, N.C.E. WOX14 promotes bioactive gibberellin synthesis and vascular cell differentiation in Arabidopsis. Plant J. 2017, 90, 560–572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Etchells, J.P.; Provost, C.M.; Mishra, L.; Turner, S.R. WOX4 and WOX14 act downstream of the PXY receptor kinase to regulate plant vascular proliferation independently of any role in vascular organisation. Development 2013, 140, 2224–2234. [Google Scholar] [CrossRef] [Green Version]
- Lian, G.; Ding, Z.; Wang, Q.; Zhang, D.; Xu, J. Origins and evolution of WUSCHEL-related homeobox protein family in plant kingdom. Sci. World J. 2014, 2014, 534140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, C.C.; Li, F.W.; Kramer, E.M. Large-scale phylogenomic analysis suggests three ancient superclades of the WUSCHEL-related homeobox transcription factor family in plants. PLoS ONE 2019, 14, e0223521. [Google Scholar] [CrossRef]
- Kim, D.Y.; Min, J.H.; Yong, W.S. Genome-wide transcript analysis of inflorescence development in wheat. Genome 2019, 62, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Jighly, A.; Joukhadar, R.; Sehgal, D.; Singh, S.; Ogbonnaya, F.C.; Daetwyler, H.D. Population-dependent reproducible deviation from natural bread wheat genome in synthetic hexaploid wheat. Plant J. 2019, 100, 801–812. [Google Scholar] [CrossRef]
- Jiang, W.; Yin, J.; Zhang, H.; He, Y.; Shuai, S.; Chen, S.; Cao, S.; Li, W.; Ma, D.; Chen, H. Genome-wide identification, characterization analysis and expression profiling of auxin-responsive GH3 family genes in wheat (Triticum aestivum L.). Mol. Biol. Rep. 2020, 47, 3885–3907. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Li, J.; Zhao, R.; Xu, K.; Xiao, Y.; Zhang, S.; Tian, J.; Yang, X. Genome-wide association study reveals the genetic basis of cold tolerance in wheat. Mol. Breed. 2020, 40, 36. [Google Scholar] [CrossRef]
- Brenchley, R.; Spannagl, M.; Pfeifer, M.; Barker, G.L.A.; D’Amore, R.; Allen, A.M.; McKenzie, N.; Kramer, M.; Kerhornou, A.; Bolser, D.; et al. Analysis of the bread wheat genome using whole-genome shotgun sequencing. Nature 2012, 491, 705–710. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.; Liu, D.; Xia, Y.; Li, Z.; Jing, D.; Du, J.; Niu, N.; Ma, S.; Wang, J.; Song, Y.; et al. Identification of the WUSCHEL-Related Homeobox (WOX) gene family, and interaction and functional analysis of TaWOX9 and TaWUS in wheat. Int. J. Mol. Sci. 2020, 21, 1581. [Google Scholar] [CrossRef] [Green Version]
- Zhao, S.; Jiang, Q.; Ma, J.; Zhang, X.; Zhao, Q.; Wang, X.; Wang, C.-S.; Cao, X.; Lu, Z.-X.; Zheng, Y.-L.; et al. Characterization and expression analysis of WOX5 genes from wheat and its relatives. Gene 2014, 537, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Ikeuchi, M.; Favero, D.S.; Sakamoto, Y.; Iwase, A.; Coleman, D.; Rymen, B.; Sugimoto, K. Molecular mechanisms of plant regeneration. Annu. Rev. Plant Biol. 2019, 70, 377–406. [Google Scholar] [CrossRef] [PubMed]
- Hellmann, E.; Helariutta, Y. Plant Genetics: Advances in Regeneration Pathways. Curr. Biol. 2019, 29, R702–R704. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, N. Getting to the root of regeneration: Adventitious rooting and callus formation. Plant Cell 2014, 26, 845. [Google Scholar] [CrossRef] [Green Version]
- Ishida, Y.; Tsunashima, M.; Hiei, Y.; Komari, Y. Wheat (Triticum aestivum L.) transformation using immature embryos. Methods Mol. Biol. 2015, 1223, 189–198. [Google Scholar] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Jones, D.T.; Taylor, W.R.; Thornton, J.M. The rapid generation of mutation data matrices from protein sequences. Comput. Appl. Biosci. 1992, 8, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Beitz, E. TeXshade: Shading and labeling of multiple sequence alignments using LaTeX2e. Bioinformatics 2000, 16, 135–139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.; Chen, H.; Zhang, Y.; Thomas, H.R.; Frank, M.H.; He, Y.; Xia, R. TBtools: An integrative toolkit developed for interactive analyses of big biological data. Mol. Plant 2020, 13, 1194–1202. [Google Scholar] [CrossRef] [PubMed]

| Gene | Gene Locus | Chromosome | Gene Stretch Region | mRNA Length (bp) | Protein Sequence Length (aa) | UniProt ID |
|---|---|---|---|---|---|---|
| TaWOX2a | TraesCS1A02G052000 | 1A | 33,397,501-33,398,955:−1 | 1314 | 263 | A0A1D5S1T3 |
| TaWOX12a | TraesCS1A02G399400 | 1A | 563,818,671-563,823,103:1 | 1854 | 486 | A0A1D5RPD4 |
| TaWOX2b | TraesCS1B02G069000 | 1B | 53,364,615-53,365,864:−1 | 1119 | 264 | A0A1B1XWM5 |
| TaWOX12b | TraesCS1B02G427400 | 1B | 652,781,930-652,786,496:1 | 1983 | 485 | A0A1D5SDQ8 |
| TaWOX2d | TraesCS1D02G054000 | 1D | 35,059,826-35,061,088:−1 | 1138 | 267 | W5ANF9 |
| TaWOX12d | TraesCS1D02G406900 | 1D | 470,219,711-470,224,514:1 | 2028 | 486 | A0A1D5SWV6 |
| TaWUS | TraesCS2A02G491900 | 2A | 724,513,458-724,514,647:1 | 927 | 308 | A0A1D5TC72 |
| TaWOX4a | TraesCS2A02G514000 | 2A | 738,371,677-738,372,966:1 | 1061 | 234 | A0A1D5TF70 |
| TaWOX11a | TraesCS2A02G100700 | 2A | 53,782,606-53,785,288:1 | 1380 | 265 | A0A1D5TJV0 |
| TaWOX11b | TraesCS2B02G117900 | 2B | 81,755,546-81,758,516:1 | 1366 | 261 | A0A1D5U6K9 |
| TaWOX4b | TraesCS2B02G542600 | 2B | 740,320,190-740,321,561:−1 | 1002 | 237 | W5BBK8 |
| TaWOX11d | TraesCS2D02G100200 | 2D | 52,227,203-52,229,885:1 | 1379 | 264 | A0A1D5V0E6 |
| TaWOX4d | TraesCS2D02G515600 | 2D | 606,709,221-606,710,431:1 | 979 | 237 | A0A1D5UH04 |
| TaWOX10a | TraesCS3A02G073500 | 3A | 45,776,166-45,777,448:1 | 992 | 260 | A0A1D5VKG7 |
| TaWOX7a | TraesCS3A02G247200 | 3A | 465,225,214-465,228,773:1 | 1968 | 515 | A0A1D5V4S9 |
| TaWOX8a | TraesCS3A02G341700 | 3A | 588,932,808-588,937,056:1 | 2230 | 265 | A0A077RTA5 |
| TaWOX14.1a | TraesCS3A02G358200 | 3A | 606,515,981-606,519,197:−1 | 1162 | 288 | A0A1D5VFV1 |
| TaWOX13a | TraesCS3A02G358100 | 3A | 606,444,775-606,446,830:−1 | 1138 | 301 | A0A1D5VA42 |
| TaWOX14.2a | TraesCS3A02G358400 | 3A | 606,573,438-606,576,220:−1 | 1133 | 290 | A0A1D6RQ92 |
| TaWOX9a | TraesCS3A02G368100 | 3A | 617,060,395-617,061,453:−1 | 949 | 212 | T1WFN3 |
| TaWOX10b | TraesCS3B02G087800 | 3B | 56,055,903-56,057,760:−1 | 1196 | 261 | A0A1D5VWS6 |
| TaWOX7b | TraesCS3B02G272200 | 3B | 438,378,936-438,382,259:−1 | 1776 | 515 | A0A077RSZ6 |
| TaWOX8b | TraesCS3B02G373800 | 3B | 586,694,870-586,698,391:1 | 1216 | 261 | A0A077S168 |
| TaWOX13b | TraesCS3B02G391100 | 3B | 616,425,121-616,426,978:−1 | 900 | 299 | A0A1D5VST7 |
| TaWOX14b | TraesCS3B02G391200 | 3B | 616,645,332-616,647,892:−1 | 1216 | 290 | A0A1D5WB93 |
| TaWOX9b | TraesCS3B02G399800 | 3B | 631,036,656-631,037,718:−1 | 948 | 209 | D8L9N7 |
| TaWOX10d | TraesCS3D02G073300 | 3D | 33,294,918-33,295,992:1 | 786 | 261 | A0A341T564 |
| TaWOX7d | TraesCS3D02G244300 | 3D | 339,473,290-339,476,679:−1 | 1834 | 513 | A0A1D5WHW6 |
| TaWOX8d | TraesCS3D02G335500 | 3D | 447,560,283-447,562,999:1 | 792 | 263 | A0A341TAX4 |
| TaWOX13d | TraesCS3D02G352500 | 3D | 463,197,196-463,199,275:−1 | 1112 | 298 | A0A1D5WMN9 |
| TaWOX14.1d | TraesCS3D02G352600 | 3D | 463,227,796-463,230,501:−1 | 895 | 285 | A0A1D5WPP9 |
| TaWOX14.2d | TraesCS3D02G352700 | 3D | 463,378,560-463,381,808:−1 | 942 | 291 | A0A1D5WNX7 |
| TaWOX9d | TraesCS3D02G361100 | 3D | 474,614,857-474,615,873:−1 | 901 | 210 | T1WGQ3 |
| TaWOX6a | TraesCS4A02G130200 | 4A | 170,708,103-170,711,065:−1 | 1350 | 307 | A0A1D5XNI6 |
| TaWOX6b | TraesCS4B02G174400 | 4B | 382,691,977-382,694,806:1 | 1254 | 309 | A0A1D5Y4Y9 |
| TaWOX6d | TraesCS4D02G176400 | 4D | 306,795,298-306,798,208:1 | 1262 | 306 | A0A341UK30 |
| TaWOX5a | TraesCS5A02G085000 | 5A | 111,588,730-111,590,895:1 | 1220 | 318 | A0A341UT17 |
| TaWOX3a | TraesCS5A02G157300 | 5A | 336,949,988-336,951,183:1 | 1060 | 241 | A0A1D5YD57 |
| TaWOX5b | TraesCS5B02G091000 | 5B | 118,451,983-118,454,221:1 | 1302 | 321 | A0A1D5ZG91 |
| TaWOX3b | TraesCS5B02G156400 | 5B | 288,891,901-288,893,003:−1 | 968 | 241 | W5F9A2 |
| TaWOX5d | TraesCS5D02G097400 | 5D | 108,103,399-108,105,722:1 | 1381 | 322 | W0Z680 |
| TaWOX3d | TraesCS5D02G162600 | 5D | 254,023,305-254,024,410:1 | 1006 | 242 | W5FQU4 |
| TaWOX8u | TraesCSU02G204800 | Un | 304,503,012-304,503,827:1 | 617 | 156 | A0A077RQB3 |
| TaWUSb | 2B | 714,777,526-714,778,733:1 | 921 | 306 | ||
| TaWUSd | 2D | 590,146,287-590,147,498:1 | 927 | 308 | ||
| 1D | 6,219,571-6,220,231:1 | |||||
| 3A | 64,319,914-64,325,218:−1 | |||||
| 3B | 83,465,544-83,470,232:−1 | |||||
| 3B | 83,471,253-83,471,941:−1 | |||||
| 3D | 52,801,752-52,812,298:−1 | |||||
| 3D | 463,261,309-463,261,744:−1 |
| Gene | Gene Locus | Chromosome | Gene Stretch Region | mRNA Length (bp) | Protein Sequence Length (aa) | Uniprot ID |
|---|---|---|---|---|---|---|
| HvWOX2 | HORVU1Hr1G010580 | 1H | 24,444,001-24,445,742:1 | 1742 | 279 | A0A287ELV0 |
| HvWOX12 | HORVU1Hr1G087940/50 | 1H | 540,693,806-540,698,431:−1 | 1470 | 489 | A0A287GM87 A0A287GM65 |
| HvWOX11 | HORVU2Hr1G017270 | 2H | 40,107,707-40,111,565:1 | 927 | 308 | A0A287H773 |
| HvWOX4 | HORVU2Hr1G113820 | 2H | 729,806,496-729,808,073:1 | 1151 | 228 | A0A287JHP1 |
| HvWOX10.1 | HORVU3Hr1G013290 | 3H | 28,673,837-28,674,948:−1 | 786 | 261 | M0Y8G7 |
| HvWOX10.2 | HORVU3Hr1G013330 | 3H | 28,785,048-28,786,156:−1 | 815 | 261 | A0A287K575 |
| HvWOX7 | HORVU3Hr1G060950 | 3H | 464,417,446-464,421,050:1 | 2027 | 516 | A0A287L9L2 |
| HvWOX8.1 | HORVU3Hr1G080660 | 3H | 589,829,423-589,834,968:−1 | 3229 | 267 | M0X0X0 |
| HvWOX8.2 | HORVU3Hr1G080690 | 3H | 590,115,430-590,116,290:1 | 584 | 130 | A0A287LWD8 |
| HvWOX9 | HORVU3Hr1G085050 | 3H | 610,834,437-610,835,788:−1 | 1165 | 209 | F2E473 |
| HvWOX14 | HORVU3Hr1G086430 | 3H | 616,993,938-616,996,482:−1 | 1216 | 283 | M0XTJ6 |
| HvWOX13 | HORVU3Hr1G086450 | 3H | 617,085,484-617,087,698:1 | 824 | 274 | A0A287M365 |
| HvWOX6 | HORVU4Hr1G051530 | 4H | 423,508,136-423,511,456:−1 | 1710 | 306 | M0Y4Z0 |
| HvWOX5 | HORVU5Hr1G022120 | 5H | 111,001,136-111,003,388:1 | 1046 | 276 | A0A287QMF0 |
| HvWOX3 | HORVU5Hr1G049190 | 5H | 381,765,625-381,766,908:1 | 1126 | 186 | A0A287R4V3 |
| HvWUS | 2H | 717,822,805-717,905,740:−1 | 942 | 313 |
| Gene | Chromosome | Gene Stretch Region | mRNA Length (bp) | Protein Sequence Length (aa) |
|---|---|---|---|---|
| ScWUS | 2R | 252,345,136-252,346,331:−1 | 930 | 309 |
| ScWOX2 | 1R | 48,768,047-48,768,972:−1 | 789 | 262 |
| ScWOX3 | 5R | 389,070,077-389,070,939:1 | 726 | 241 |
| ScWOX4 | 2R | 267,063,777-267,064,860:1 | 705 | 224 |
| ScWOX5 | 5R | 130,892,024-130,893,989:1 | 927 | 308 |
| ScWOX6 | 7R | 341,262,761-341,264,920:−1 | 921 | 306 |
| ScWOX7 | 3R | 358,251,618-358,254,632:−1 | 1545 | 514 |
| ScWOX8.1 | 3R | 102,984,340-102,987,559:1 | 792 | 263 |
| ScWOX8.2 | 3R | 104,728,944-104,732,163:1 | 792 | 263 |
| ScWOX9 | 3R | 154,267,694-154,268,435:−1 | 636 | 211 |
| ScWOX10 | 3R | 107,456,405-107,457,718:−1 | 786 | 261 |
| ScWOX11.1 | 2R | 77,444,261-77,446,289:1 | 795 | 264 |
| ScWOX11.2 | 2R | 77,562,117-77,564,149:1 | 795 | 264 |
| ScWOX12 | 1R | 91,075,823-91,079,684:1 | 1449 | 482 |
| ScWOX13.1 | 3R | 139,781,793-139,779,696:1 | 900 | 299 |
| ScWOX13.2 | 3R | 187,095,786-187,097,863:−1 | 897 | 298 |
| ScWOX13.3 | 3R | 164,219,576-164,221,728:−1 | 897 | 290 |
| ScWOX13.4 | 3R | 140,032,819-140,034,973:−1 | 897 | 286 |
| ScWOX13.5 | 3R | 140,194,495-140,196,649:1 | 897 | 298 |
| ScWOX13.6 | 3R | 157,852,945-157,855,106:1 | 897 | 298 |
| ScWOX13.7 | 3R | 140,118,810-140,120,961:−1 | 894 | 267 |
| ScWOX13.8 | 3R | 140138607-140140760:−1 | 366 | 121 |
| ScWOX14 | 3R | 140,085,774-140,087,928:1 | 856 | 283 |
| Gene | Chromosome | Gene Stretch Region | mRNA Length (bp) | Protein Sequence Length (aa) |
|---|---|---|---|---|
| TeWUS | 2E | 636,539,269-636,540,438:1 | 924 | 329 |
| TeWOX2 | 1E | 62,834,291-62,835,214:−1 | 789 | 262 |
| TeWOX3 | 5E | 249,414,904-249,415,769:1 | 732 | 243 |
| TeWOX4 | 2E | 657,284,040-657,284,981:−1 | 708 | 235 |
| TeWOX5 | 5E | 123,902,262-123,904,308:1 | 960 | 319 |
| TeWOX6 | 4E | 373,518,174-373,520,519:1 | 933 | 310 |
| TeWOX7 | 3E | 355,150,216-355,153,543:−1 | 1512 | 503 |
| TeWOX8 | 3E | 460,009,235-460,013,048:1 | 789 | 262 |
| TeWOX9 | 3E | 489,665,059-489,665,812:−1 | 633 | 210 |
| TeWOX10 | 3E | 63,048,238-63,049,186:1 | 786 | 261 |
| TeWOX11 | 2E | 101,851,313-101,853,580:1 | 780 | 259 |
| TeWOX12 | 1E | 503,347,861-503,351,437:1 | 1458 | 485 |
| TeWOX13 | 3E | 480,682,493-480,684,518:−1 | 903 | 300 |
| TeWOX14 | 3E | 480,874,716-480,877,294:−1 | 873 | 290 |
| Gene | Chromosome | Gene Stretch Region | mRNA Length (bp) | Protein Sequence Length (aa) |
|---|---|---|---|---|
| AsWUSa | 2A | Fragments | ||
| AsWUSc | 2C | Fragments | ||
| AsWUSd | 2D | Fragments | ||
| AsWOX2a | 1A | Fragments | ||
| AsWOX2c | 1C | Fragments | ||
| AsWOX2d | 1D | Fragments | ||
| AsWOX3a | 5A | 356,256,138-356,257,025:−1 | 741 | 246 |
| AsWOX3c | 5C | 395,717,807-395,718,695:−1 | 738 | 245 |
| AsWOX3d | 5D | 322,024,845-322,025,729:−1 | 735 | 244 |
| AsWOX4a | 2A | 396,219,690-396,220,495:−1 | 720 | 239 |
| AsWOX4c | 2C | 549,486,552-549,487,356:−1 | 717 | 238 |
| AsWOX4d | 2D | 186,662,767-186,663,482:−1 | 708 | 235 |
| AsWOX5a | 4A | Fragments | ||
| AsWOX5c | 3C | Fragments | ||
| AsWOX5d | 3D | Fragments | ||
| AsWOX6a | 5A | 53,538,777-53,540,999:−1 | 969 | 322 |
| AsWOX6c | 4C | 594,589,370-594,591,649:1 | 987 | 328 |
| AsWOX6d | 5D | 21,782,750-21,784,999:−1 | 978 | 325 |
| AsWOX7a | 3A | 376,179,787-376,182,862:−1 | 1485 | 494 |
| AsWOX7c | 3C | 514,164,874-514,167,899:−1 | 1509 | 502 |
| AsWOX7d | 3D | 327,595,075-327,598,159:−1 | 1485 | 494 |
| AsWOX8a | 4A | 369,829,463-369,832,224:−1 | 741 | 246 |
| AsWOX8c | 3C | 593,558,934-593,561,981:−1 | 738 | 245 |
| AsWOX8d | 3D | 377,117,878-377,121,109:−1 | 735 | 244 |
| AsWOX9a | 4A | 401,138,523-401,139,247:−1 | 633 | 210 |
| AsWOX9c | 3C | 621024950-621025679:−1 | 636 | 211 |
| AsWOX9d | 4D | 357331495-357332237:−1 | 651 | 216 |
| AsWOX10a | 6A | Fragments | ||
| AsWOX10.1c | 4C | Fragments | ||
| AsWOX10.2c | 7C | Fragments | ||
| AsWOX11a | 6A | 410997915-410999746:−1 | 777 | 258 |
| AsWOX11c | 4C | 24,506,573-24,508,358:1 | 786 | 261 |
| AsWOX11d | 5D | 454,073,395-454,075,249:−1 | 777 | 258 |
| AsWOX12a | 1A | 373,498,261-373,501,827:1 | 1416 | 471 |
| AsWOX12.1a | 1A | 522,889,272-522,892,564:1 | 1404 | 467 |
| AsWOX12d | 1D | 357,683,263-357,687,060:1 | 1413 | 470 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, L.; Wang, K.; Du, L.; Song, Y.; Li, H.; Ye, X. Genome-Wide Identification and Expression Profiling Analysis of WOX Family Protein-Encoded Genes in Triticeae Species. Int. J. Mol. Sci. 2021, 22, 9325. https://doi.org/10.3390/ijms22179325
Shi L, Wang K, Du L, Song Y, Li H, Ye X. Genome-Wide Identification and Expression Profiling Analysis of WOX Family Protein-Encoded Genes in Triticeae Species. International Journal of Molecular Sciences. 2021; 22(17):9325. https://doi.org/10.3390/ijms22179325
Chicago/Turabian StyleShi, Lei, Ke Wang, Lipu Du, Yuxia Song, Huihui Li, and Xingguo Ye. 2021. "Genome-Wide Identification and Expression Profiling Analysis of WOX Family Protein-Encoded Genes in Triticeae Species" International Journal of Molecular Sciences 22, no. 17: 9325. https://doi.org/10.3390/ijms22179325
APA StyleShi, L., Wang, K., Du, L., Song, Y., Li, H., & Ye, X. (2021). Genome-Wide Identification and Expression Profiling Analysis of WOX Family Protein-Encoded Genes in Triticeae Species. International Journal of Molecular Sciences, 22(17), 9325. https://doi.org/10.3390/ijms22179325







