Molecular Hydrogen as a Novel Antitumor Agent: Possible Mechanisms Underlying Gene Expression
Abstract
:1. Introduction
2. Molecular Biology of Cancer
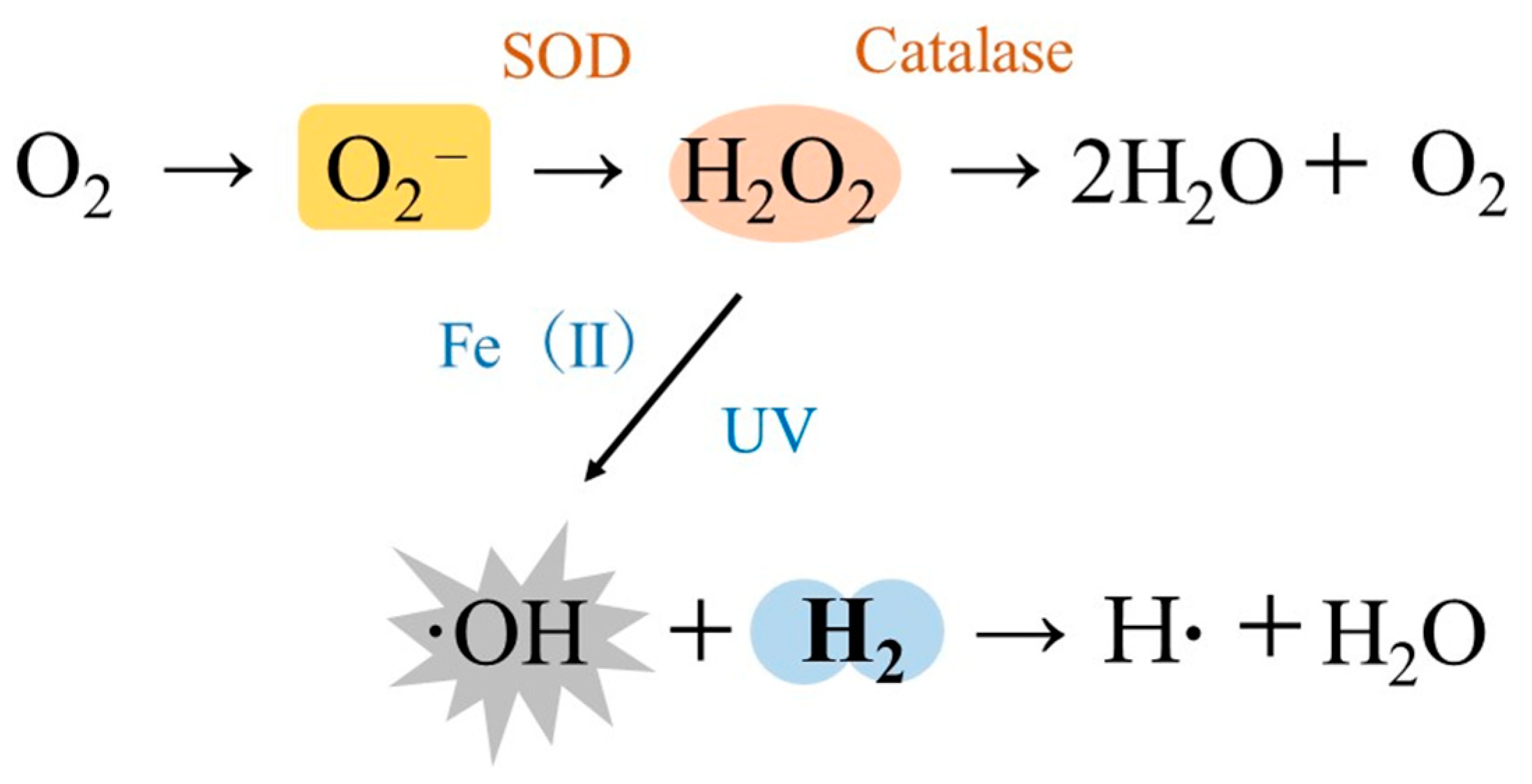
3. Redox Control in Cancer
4. The Relationship between Cancer, Chronic Inflammation and Ageing
5. Antitumor Effects of H2
5.1. Antitumor Effects in Cellular or Animal Models
5.1.1. Antitumor Effects in Cellular Models
5.1.2. Antitumor Effects in Animal Models
5.1.3. Antitumor Effects in UV or Ionizing Radiation Models
5.1.4. Inhibition of Carcinogenesis in the NASH Model
5.1.5. Inhibitory Effects on Angiogenesis
5.2. Antitumor Effects in Human Clinical Trials
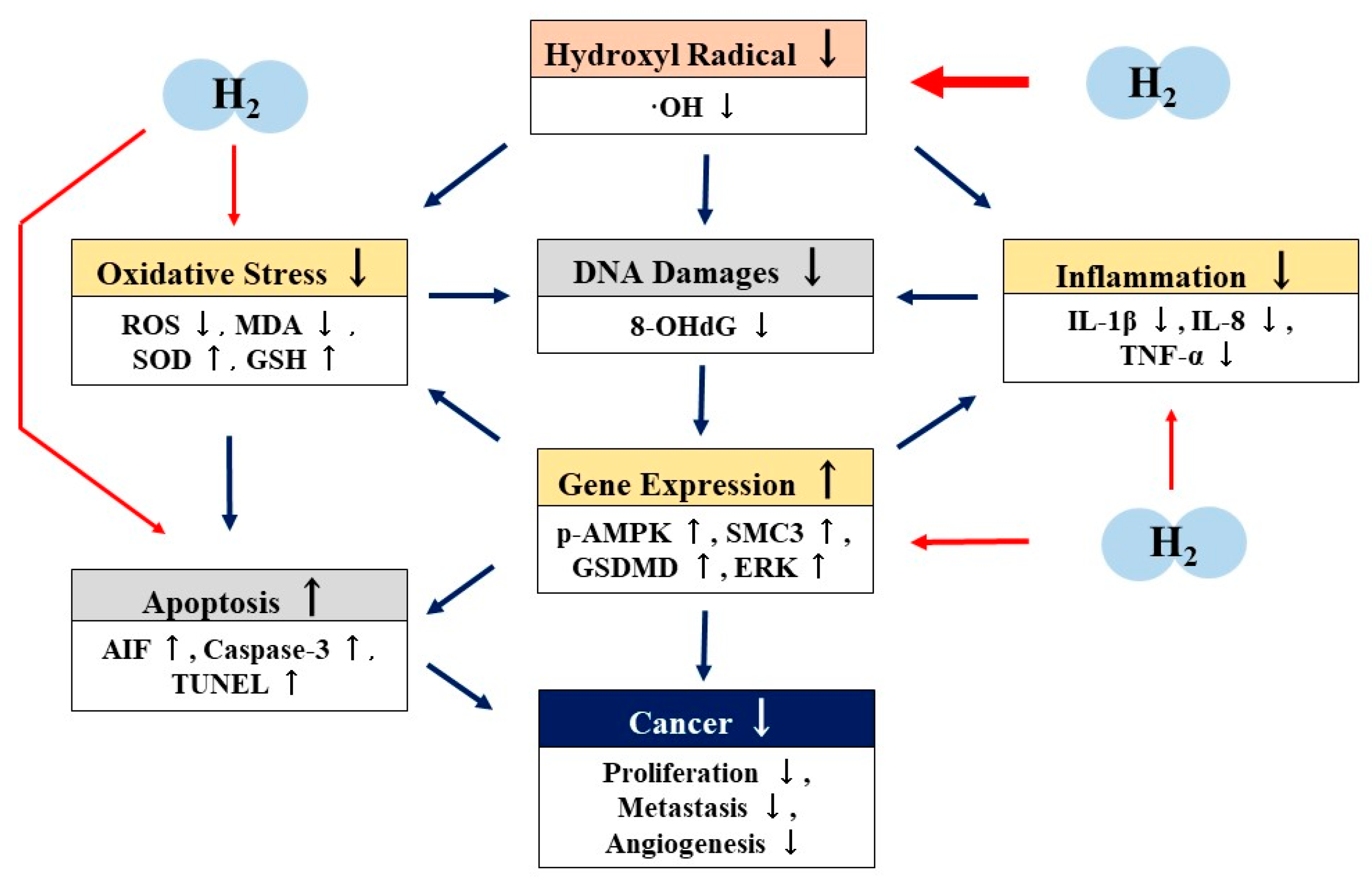
6. Possible Mechanisms of the Antitumor Effects of H2
7. Prospects of H2 as an Antitumor Agent
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- National Cancer Institute. What Is Cancer? Available online: https://www.cancer.gov/about-cancer/understanding/what-is-cancer (accessed on 2 July 2021).
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Kindler, H.L. Malignant pleural mesothelioma. Curr. Treat. Options Oncol. 2000, 1, 313–326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, Y.T.; Tan, Y.J.; Oon, C.E. Molecular targeted therapy: Treating cancer with specificity. Eur. J. Pharmacol. 2018, 834, 188–196. [Google Scholar] [CrossRef]
- Opalinska, J.B.; Gewirtz, A.M. Nucleic-acid therapeutics: Basic principals and recent applications. Nat. Rev. Drug Discov. 2002, 1, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Chen, Y.P.; Du, X.J.; Liu, J.Q.; Huang, C.L.; Chen, L.; Zhou, G.Q.; Li, W.F.; Mao, Y.P.; Hsu, C. Comparative safety of immune checkpoint inhibitors in cancer: Systematic review and network meta-analysis. BMJ 2018, 363, k4226. [Google Scholar] [CrossRef]
- Vasan, N.; Baselga, J.; Hyman, D.M. A view on drug resistance in cancer. Nature 2019, 575, 299–309. [Google Scholar] [CrossRef] [Green Version]
- Dole, M.; Wilson, F.R.; Fife, W.P. Hyperbaric hydrogen therapy: A possible treatment for cancer. Science 1975, 190, 152–154. [Google Scholar] [CrossRef]
- Ohsawa, I.; Ishikawa, M.; Takahashi, K.; Watanabe, M.; Nishimaki, K.; Yamagata, K.; Katsura, K.I.; Katayama, Y.; Asoh, S.; Ohta, S. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nat. Med. 2007, 13, 688–694. [Google Scholar] [CrossRef]
- Yanagihara, T.; Arai, K.; Miyamae, K.; Sato, B.; Shudo, T.; Yamada, M.; Aoyama, M. Electrolyzed hydrogen-saturated water for drinking use elicits an antioxidative effect; a feeding test with rats. Biosci. Biotrechnol. Biochem. 2005, 69, 1985–1987. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ohta, S. Molecular hydrogen as a preventive and therapeutic medical gas: Initiation, development and potential of hydrogen medicine. Pharmacol. Ther. 2014, 144, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Ohta, S. Molecular hydrogen as a novel antioxidant: Overview of the advantages of hydrogen for medical applications. Methods Enzymol. 2015, 555, 289–317. [Google Scholar]
- Hirano, S.-i.; Ichikawa, Y.; Sato, B.; Satoh, F.; Takefuji, Y. Hydrogen is promising for medical applications. Clean Technol. 2020, 2, 529–541. [Google Scholar] [CrossRef]
- Hirano, S.-i.; Ichikawa, Y.; Kurokawa, R.; Takefuji, Y.; Satoh, F. A “philosophical molecule,” hydrogen may overcome senescence and intractable diseases. Med. Gas Res. 2020, 10, 47–49. [Google Scholar] [CrossRef]
- Ono, H.; Nishijima, Y.; Adachi, N.; Tachibana, S.; Chitoku, S.; Mukaihara, S.; Sakamoto, M.; Kudo, Y.; Nakazawa, J.; Kaneko, K.; et al. Improved brain MRI indices in the acute brain stem infarct sites treated with hydroxyl radical scavengers, Edaravone and hydrogen, as compared to Edaravone alone. A non-controlled study. Med. Gas Res. 2011, 1, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoritaka, A.; Takanashi, M.; Hirayama, M.; Nakahara, T.; Ohta, S.; Hattori, N. Pilot study of H2 therapy in Parkinson’s disease. A randomized double-blind placebo-controlled trial. Mov. Disord. 2013, 28, 836–839. [Google Scholar] [CrossRef] [PubMed]
- Ono, H.; Nishijima, Y.; Ohta, S.; Sakamoto, M.; Kinone, K.; Horikoshi, T.; Tamaki, M.; Takeshita, H.; Futatuki, T.; Ohishi, W.; et al. Hydrogen gas inhalation treatment in acute cerebral infarction: A randomized controlled clinical study on safety and neuroprotection. J. Stroke Cerebrovasc. 2017, 26, 2587–2594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Katsumata, Y.; Sano, F.; Abe, T.; Tamura, T.; Fujisawa, T.; Shiraishi, Y.; Khosaka, S.; Ueda, I.; Honmma, K.; Suzuki, M.; et al. The effects of hydrogen gas inhalation on adverse left ventricular remodeling after percutaneous coronary intervention for ST-elevated myocardial infraction. First pilot study in humans. Circ. J. 2017, 81, 940–947. [Google Scholar] [CrossRef] [Green Version]
- Tamura, T.; Hayashida, K.; Sano, M.; Suzuki, M.; Shibusawa, T.; Yoshizawa, J.; Kabayashi, Y.; Suzuki, T.; Ohta, S.; Morisaki, H.; et al. Feasibility and safety of hydrogen gas inhalation for post-cardiac arrest syndrome. Circ. J. 2016, 80, 1870–1873. [Google Scholar] [CrossRef] [Green Version]
- Guan, W.J.; Wei, C.H.; Chen, A.L.; Sun, X.C.; Guo, G.Y.; Zou, X.; Shi, J.D.; Lai, P.Z.; Zheng, Z.G.; Zhong, N.S. Hydrogen/oxygen mixed gas inhalation improves disease severity and dyspnea in patients with Coronavirus disease 2019 in a recent multicenter, open-label clinical trial. J. Thorac. Dis. 2020, 12, 3448–3452. [Google Scholar] [CrossRef]
- Gong, Z.; Guan, J.; Ren, X.; Meng, D.; Zhang, H.; Wang, B.; Yan, X. Protective effect of hydrogen on the lung of sanitation workers exposed to haze. Chin. J. Tuberc. Respir. Dis. 2016, 39, 916–923. [Google Scholar]
- Kajiyama, S.; Hasegawa, G.; Asano, M.; Hosoda, H.; Fukui, M.; Nakamura, N.; Kitawaki, J.; Imai, S.; Nakano, K.; Ohta, M.; et al. Supplementation of hydrogen-rich water improves lipid and glucose metabolism in patients with type 2 diabetes or impaired glucose tolerance. Nutr. Res. 2008, 28, 137–143. [Google Scholar] [CrossRef]
- Xia, C.; Liu, W.; Zeng, D.; Zhu, L.; Sun, X.; Sun, X. Effect of hydrogen-rich water on oxidative stress, liver function, and viral load in patients with chronic hepatitis B. Clin. Trans. Sci. 2013, 6, 372–375. [Google Scholar] [CrossRef] [Green Version]
- Song, G.; Li, M.; Sang, H.; Zhang, L.; Li, X.; Yao, S.; Yu, Y.; Zong, C.; Xue, Y.; Qin, S. Hydrogen-rich water decreases serum low-density lipoprotein cholesterol levels and improves high-density lipoprotein function in patients with potential metabolic syndrome. J. Lipid Res. 2013, 54, 1884–1893. [Google Scholar] [CrossRef] [Green Version]
- Saitoh, Y.; Okayasu, H.; Xiao, L.; Harata, Y.; Miwa, N. Neutral pH hydrogen-enriched electrolyzed water achieves tumor-preferential clonal growth inhibition over normal cells and tumor invasion inhibition concurrently with intracellular oxidant repression. Oncol. Res. 2008, 17, 247–255. [Google Scholar] [CrossRef]
- Saitoh, Y.; Yoshimura, Y.; Nakano, K.; Miwa, N. Platinum nanocolloid-supplemented hydrogen-dissolved water inhibits growth of human tongue carcinoma cells preferentially over normal cells. Exp. Oncol. 2009, 31, 156–162. [Google Scholar] [PubMed]
- Asada, R.; Kageyama, K.; Tanaka, H.; Matsui, H.; Kimura, M.; Saitoh, Y.; Miwa, N. Antitumor effects of nano-bubble hydrogen-dissolved water are enhanced by coexistent platinum colloid and the combined hyperthermia with apoptosis-like cell death. Oncol. Rep. 2010, 24, 1463–1470. [Google Scholar] [PubMed] [Green Version]
- Saitoh, Y.; Ikeshima, M.; Kawasaki, N.; Masumoto, A.; Miwa, N. Transient generation of hydrogen peroxide is responsible for carcinostatic effects of hydrogen combined with platinum nanocolloid, together with increase intracellular ROS, DNA cleavages, and proportion of G2/M-phase. Free Radic. Res. 2016, 50, 385–395. [Google Scholar] [CrossRef]
- Kato, S.; Saitoh, Y.; Miwa, N. Carcinostatic effects of alkanoyl ascorbate plus platinum nano-colloid and stabilization of the esterolytically resultant ascorbate by hydrogen. Hum. Cell 2021, 34, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Kagawa, A.; Katsura, K.; Mizumoto, M.; Tagawa, Y.; Mashiko, Y. Influence of hydrogen discharged from palladium base hydrogen storage alloys on cancer cells. Mater. Sci. Forum 2012, 706–709, 520–525. [Google Scholar] [CrossRef]
- Runtuwene, J.; Amitani, H.; Amitani, M.; Asakawa, A.; Cheng, K.C.; Inui, K. Hydrogen-water enhances 5-fluorouracil-induced inhibition of colon cancer. PeerJ 2015, 3, e859. [Google Scholar] [CrossRef] [Green Version]
- Wang, D.; Wang, L.; Zhang, Y.; Zhao, Y.; Chen, G. Hydrogen gas inhibits lung cancer progression through targeting SMC3. Biomed. Pharmacol. 2018, 104, 788–797. [Google Scholar] [CrossRef]
- Zhao, P.; Jin, Z.; Chen, Q.; Yang, T.; Chen, D.; Meng, J.; Lu, X.; Gu, Z.; He, Q. Local generation of hydrogen for enhanced photothermal therapy. Nat. Commun. 2018, 9, 4241. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.U.; Xie, F.; Zhang, Y.; Wang, T.T.; Ma, S.N.; Zhao, P.X.; Zhang, X.; Lebaron, T.W.; Yan, X.L.; Ma, X.M. Molecular hydrogen suppresses glioblastoma growth via inducing the glioma stem-like cell differentiation. Stem Cell Res. Ther. 2019, 10, 145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Y.; Liu, P.Y.; Bao, W.; Chen, S.J.; Wu, F.S.; Zhu, P.Y. Hydrogen inhibits endometrial cancer growth via a ROS/NLRP3/Caspase-1/GSDMD-mediated pyroptotic pathway. BMC Cancer 2020, 20, 28. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhou, C.; Zhang, J.; Gao, F.; Li, B.; Chuai, Y.; Liu, C.; Cai, J. Hydrogen protects mice from radiation induced thymic lymphoma in BALB/c mice. Int. J. Biol. Sci. 2011, 7, 297–300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawai, D.; Takaki, A.; Nakatsuka, A.; Wada, J.; Tamaki, N.; Yasunaka, T.; Koike, K.; Tsuzaki, R.; Matsumoto, K.; Miyake, Y.; et al. Hydrogen-rich water prevents progression of nonalcoholic steatohepatitis and accompanying hepatocarcinogenesis in mice. Hepatology 2012, 56, 912–921. [Google Scholar] [CrossRef]
- Ye, J.; Li, Y.; Hamasaki, T.; Nakamichi, N.; Komatsu, T.; Kashiwagi, T.; Teruya, K.; Nishikawa, R.; Osada, K.; Toh, K.; et al. Inhibitory effect of electrolyzed reduced water on tumor angiogenesis. Biol. Pharm. Bull. 2008, 31, 19–26. [Google Scholar] [CrossRef] [Green Version]
- Akagi, J.; Baba, H. Hydrogen gas restores exhausted CD8+ T cells in patients with advanced colorectal cancer to improve prognosis. Oncol. Rep. 2018, 41, 301–311. [Google Scholar] [CrossRef] [Green Version]
- Akagi, J.; Baba, H. Hydrogen gas activates coenzyme Q10 to restore exhausted CD8+ T cells, especially PD-1+Timterminal CD8+ T cells, leading to better nivolumab outcomes in patients with lung cancer. Oncol. Lett. 2020, 20, 258. [Google Scholar] [CrossRef]
- Chen, J.B.; Kong, X.F.; Lv, Y.Y.; Qin, S.C.; Sun, X.J.; Mu, F.; Lu, T.Y.; Xu, K.C. “Real world survey” of hydrogen-controlled cancer: A follow-up report of 82 advanced cancer patients. Med. Gas Res. 2019, 9, 115–121. [Google Scholar]
- Chen, J.B.; Pan, Z.B.; Du, D.M.; Qian, W.; Ma, Y.Y.; Mu, F.; Xu, K.C. Hydrogen gas therapy shrinkage of metastatic gallbladder cancer: A case report. World J. Clin. Cases 2019, 7, 2065–2074. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Mu, F.; Lu, T.; Ma, Y.; Du, D.; Xu, K. A gallbladder carcinoma patient with pseudo-progressive remission after hydrogen inhalation. Onco Targets Ther. 2019, 12, 8645–8651. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Mu, F.; Lu, T.; Du, D.; Xu, K. Brain metastases completely disappear in non-small cell lung cancer using hydrogen gas inhalation: A case report. Onco Target Ther. 2019, 12, 11145–11151. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.B.; Kong, X.F.; Mu, F.; Lu, T.Y.; Lu, Y.Y.; Xu, K.C. Hydrogen therapy can be used to control tumor progression and alleviate the adverse events of medications in patients with advanced non-small cell lung cancer. Med. Gas Res. 2020, 10, 75–80. [Google Scholar] [PubMed]
- Chen, J.B.; Kong, X.F.; Qian, W.; Mu, F.; Lu, T.Y.; Lu, Y.Y.; Xu, K.C. Two weeks of hydrogen inhalation can significantly reverse adaptive and innate immune system senescence patients with advanced non-small cell lung cancer: A self-controlled study. Med. Gas Res. 2020, 10, 149–154. [Google Scholar]
- Compagni, A.; Christofori, G. Recent advances in research on multistage tumorigenesis. Br. J. Cancer 2000, 83, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.J.; Oren, M. The first 30 years of p53: Growing ever more complex. Nat. Rev. Cancer 2009, 9, 749–758. [Google Scholar] [CrossRef] [Green Version]
- Sigal, A.; Rotter, V. Oncogenic mutations of the p53 tumor suppressor: The demons of the guardian of the genome. Cancer Res. 2000, 60, 6788–6793. [Google Scholar] [PubMed]
- Vousden, K.H.; Lu, X. Live or let die: The cell’s response to p53. Nat. Rev. Cancer 2002, 2, 594–604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kasai, H. Analysis of a form of oxidative DNA damage, 8-hydroxy-2′-deoxyguanosine, as a marker of cellular oxidative stress during carcinogenesis. Mutat. Res. 1997, 387, 147–163. [Google Scholar] [CrossRef]
- Toyokuni, S.; Mori, T.; Dizdaroglu, M. DNA base modifications in renal chromatin of Wister rats treated with a renal carcinogen, ferric nitrilotriacetate. Int. J. Cancer 1994, 57, 123–128. [Google Scholar] [CrossRef]
- Tsukahara, H.; Hiraoka, M.; Kobata, R.; Hata, I.; Ohshima, Y.; Jiang, M.Z.; Noiri, E.; Mayumi, M. Increased oxidative stress in rats with chronic nitric oxide depletion: Measurement of urinary 8-hydroxy-2′-deoxyguanosine excretion. Redox Rep. 2000, 5, 23–28. [Google Scholar] [CrossRef]
- Floyd, R.A. The role of 8-hydroxyguanine in carcinogenesis. Carcinogenesis 1990, 11, 1447–1450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirano, S.-i.; Ichikawa, Y.; Sato, B.; Yamamoto, H.; Takefuji, Y.; Satoh, F. Molecular hydrogen as a potential clinically applicable radioprotective agent. Int. J. Mol. Sci. 2021, 22, 4566. [Google Scholar] [CrossRef]
- Harris, S.L.; Levine, A.J. The p53 pathway: Positive and negative feedback loops. Oncogene 2005, 24, 2899–2908. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hasty, P.; Christy, B.A. p53 as an intervention target for cancer and aging. Pathobiol. Aging Age Relat. Dis. 2013, 3, 22702. [Google Scholar] [CrossRef]
- Leonhardt, H.; Cardoso, M.C. DNA methylation, nuclear structure, gene expression and cancer. J. Cell Biochem. Suppl. 2000, 79 (Suppl. 35), 78–83. [Google Scholar] [CrossRef] [Green Version]
- U.S. National Library of Medicine. Available online: https://clinicaltrials.gov/ct2/home (accessed on 2 July 2021).
- Hirano, S.-i.; Ichikawa, Y.; Sato, B.; Yamamoto, H.; Takefuji, Y.; Satoh, F. Potential therapeutic applications of hydrogen in chronic inflammatory disease: Possible inhibiting role on mitochondrial stress. Int. J. Mol. Sci. 2021, 22, 2549. [Google Scholar] [CrossRef] [PubMed]
- World Cancer Research Fund International. The Third Expert Report. 2018. Available online: https://www.wcrf.org/sites/default/files/Summary-of-Third-Expert-Report-2018.pdf (accessed on 2 July 2021).
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-related inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef]
- Greider, C.W. Cellular responses to telomere shortening: Cellular senescence as a tumor suppressor mechanism. Harvey Lect. 2000, 96, 33–50. [Google Scholar] [PubMed]
- Lundberg, A.S.; Hahn, W.C.; Gupta, P.; Weinberg, R.A. Genes involved in senescence and immortalization. Curr. Opin. Cell Biol. 2000, 12, 705–709. [Google Scholar] [CrossRef]
- Chambers, C.R.; Ritchie, S.; Pereira, B.A.; Timpson, P. Overcoming the senescence-associated secretory phenotype (SASP): A complex mechanism of resistance in the treatment of cancer. Mol. Oncol. 2021. online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Kamimura, N.; Ichimiya, H.; Iuchi, K.; Ohta, S. Molecular hydrogen stimulates the gene expression of transcriptional coactivator PGC-1α to enhance fatty acid metabolism. NPJ Aging Mech. Dis. 2016, 2, 16008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radiation Chemistry Data Center; Notre Dame Radiation Laboratory (n.d.). NERL Data. 2011. Available online: http://kinetics.nist.gov/solution/ (accessed on 5 August 2021).
- Wood, K.C.; Gladwin, M.T. The hydrogen highway to reperfusion therapy. Nat. Med. 2007, 13, 673–674. [Google Scholar] [CrossRef] [PubMed]
- Hoovis, M.L. Response of endometrial stromal sarcoma to cyclophosphamide. Am. J. Obstet. Gynecol. 1970, 108, 1117–1119. [Google Scholar] [CrossRef]
- Kim, R.; Nishimoto, N.; Inoue, H.; Yoshida, K.; Toge, T. An analysis of the therapeutic efficacy of protracted infusion of low-dose 5-fluorouracil and cisplatin in advanced gastric cancer. J. Infect. Chemother. 2000, 6, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Levine, M. Epirubicin in breast cancer: Present and future. Clin. Breast Cancer 2000, S62–S67. [Google Scholar] [CrossRef]
- Sekine, I.; Saijo, N. Novel combination chemotherapy in the treatment of non-small cancer lung cancer. Expert Opin. Pharmacother. 2000, 1, 1131–1161. [Google Scholar] [CrossRef]
- Hirano, S.-i.; Agata, N.; Hara, Y.; Iguchi, H.; Shirai, M.; Tone, H.; Urakawa, N. Effects of pirarubicin, an antitumor antibiotic, on the cardiovascular system. Cancer Chemother. Pharmacol. 1991, 28, 266–272. [Google Scholar]
- Hirano, S.-i.; Agata, N.; Hara, Y.; Iguchi, H.; Shirai, M.; Tone, H.; Urakawa, N. Pirarubicin-induced endotherium-dependent relaxation in the rat isolated aorta. J. Pharm. Pharmacol. 1991, 43, 848–854. [Google Scholar] [CrossRef]
- Hirano, S.-i.; Agata, N.; Hara, Y.; Iguchi, H.; Shirai, M.; Tone, H.; Urakawa, N. A possible mechanism of endothelium-dependent relaxation induced by pirarubicin and carbachol in rat isolated aorta. J. Pharm. Pharmacol. 1992, 44, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Hirano, S.-i.; Agata, N.; Iguchi, H.; Tone, H. Effects of pirarubicin in comparison with epirubicin and doxorubicin on the contractile function in rat isolated cardiac muscles. Gen. Pharmacol. 1995, 26, 1339–1347. [Google Scholar] [CrossRef]
- Rochette, L.; Zeller, M.; Cottin, Y.; Vergely, C. Antitumor activity of protons and molecular hydrogen underlying mechanism. Cancer 2021, 13, 893. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhu, Y.; Xi, X. Anti-inflammatory and antitumor action of hydrogen via reactive oxygen species. Oncol. Lett. 2018, 16, 2771–2776. [Google Scholar] [CrossRef] [Green Version]
- Klein, E.A.; Thompson, I.M.; Tangen, C.M.; Growley, J.J.; Lucia, M.S.; Goodman, P.J.; Minasian, L.M.; Ford, L.G.; Parnes, H.L.; Gaziano, J.M.; et al. Vitamin E and the risk of prostate cancer. The selenium and vitamin E cancer prevention trial (Select). J. Am. Med. Assoc. 2011, 306, 1549–1556. [Google Scholar] [CrossRef]
- Chandel, N.S.; Tuveson, D.A. The promise and perils of antioxidants for cancer patients. N. Engl. J. Med. 2014, 371, 177–178. [Google Scholar] [CrossRef]
- Sayin, V.I.; Ibrahim, M.X.; Larsson, E.; Nilsson, J.A.; Lindahl, P.; Bergo, M.O. Antioxidants accelerate lung progression in mice. Sci. Transl. Med. 2014, 6, 221ra15. [Google Scholar] [CrossRef]
- DeNicola, G.M.; Karreth, F.A.; Humpton, T.J.; Gopinathan, A.; Wei, C.; Frese, K.; Mangal, D.; Yu, K.H.; Yeo, C.J.; Calhoun, E.S.; et al. Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature 2011, 475, 106–109. [Google Scholar] [CrossRef]
- Schafer, Z.T.; Grassian, A.R.; Song, L.; Jiang, Z.; Gerhart-Hines, Z.; Irie, H.Y.; Gao, S.; Puigserver, P.; Brugge, J.S. Antioxidant and oncogene rescue of metabolic defects caused by loss of matrix attachment. Nature 2009, 461, 109–113. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Li, B.; Liu, C.; Chuai, Y.; Lei, J.; Gao, F.; Cui, J.; Sun, D.; Cheng, Y.; Zhou, C.; et al. Hydrogen-rich saline protects immunocytes from radiation-induced apoptosis. Med. Sci. Monit. 2012, 18, BR144–BR148. [Google Scholar] [CrossRef] [Green Version]
- Zhao, S.; Yang, Y.; Liu, W.; Xuan, Z.; Wu, S.; Yu, S.; Mei, K.; Huang, Y.; Zhang, P.; Cai, J.; et al. Protective effect of hydrogen-rich saline against radiation-induced immune dysfunction. J. Cell Mol. Med. 2014, 18, 938–946. [Google Scholar] [CrossRef]
- Ozeki, N.; Yamawaki-Ogata, A.; Narita, Y.; Mii, S.; Ushida, K.; Ito, M.; Hirano, S.-i.; Kurokawa, R.; Ohno, K.; Usui, A. Hydrogen water alleviates obliterative airway disease in mice. Gen. Thorac. Cardiovasc. Surg. 2019, 68, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Kurokawa, R.; Fujino, M.; Hirano, S.-I.; Sato, B.; Li, X.K. Estimation of the hydrogen concentration in rat tissue using an airtight tube following the administration of hydrogen via various routes. Sci. Rep. 2014, 4, 5485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamamoto, R.; Homma, K.; Suzuki, S.; Sano, M.; Sasaki, J. Hydrogen gas distribution in organs after inhalation: Real-time monitoring of tissue hydrogen concentration in rat. Sci. Rep. 2019, 9, 1255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Experimental System | Cancer Types | Effects of Molecular Hydrogen (H2) | Ref. No. |
|---|---|---|---|
| Cultured cells | Human tongue cancer and Human fibrosarcoma | H2-rich electrolyzed water showed inhibitory effects on ROS production and cell proliferation. | [25] |
| Human tongue cancer and Human fibrosarcoma | H2-dissolved water, in combination with platinum nano-colloid, showed enhanced suppression of ROS production and cell proliferation. | [26] | |
| Mouse Ehrlich’s ascites tumor | Nanobubble H2 water with platinum nano-colloid demonstrated inhibitory effects on the production of ROS and cell proliferation. | [27] | |
| Mouse Ehrlich’s ascites tumor | The combination of H2-dissolved water and platinum nano-colloid showed an inhibitory effect on cell proliferation, and the effect involved the transient generation of hydrogen peroxide. | [28] | |
| Human esophageal cancer | The combination of electrolytic H2 water and PVP-Pt colloid demonstrated enhanced inhibition of cell proliferation. | [29] | |
| Human cervical cancer, Human soft tissue tumor, etc. | Hydrogen radicals and H2 produced from Pd-Ni hydrogen storage alloys showed a killing effect on four types of tumor cells. | [30] | |
| Experimental animals (Transplant models) | Mouse colon cancer | The combination of H2 water and 5-fluorouracil showed antitumor effects via activation of the apoptotic pathway. | [31] |
| Human lung cancer | H2 gas showed antitumor effects through down-regulation of the expression of SMC3, a regulator of chromosome condensation. | [32] | |
| Mouse breast cancer and Human melanoma | The combination of palladium hydride and laser light irradiation was effective in suppressing tumor volume and tumor weight. | [33] | |
| Rat glioma and Human glioma | H2 gas inhibited the growth of glioblastoma through differentiation of glioma stem-like cells. | [34] | |
| Human endometrial cancer | H2-rich water showed antitumor effects by activating the ROS/NLRP3/caspase-1/GSDMD-mediated pyrotrophic pathway. | [35] | |
| Experimental animals (Induced models) | UV-induced skin cancer | Hyperbaric treatment with H2 gas inhibited the growth and regression of skin cancer in mice induced by UV. | [8] |
| Ionizing radiation-induced thymic lymphoma | H2-rich saline suppressed the development of thymic lymphoma in mice induced by ionizing radiation. | [36] | |
| High-fat diet-induced liver cancer | H2-rich water showed carcinogenic effects in an experimental system that progressed from NASH to fibrosis and liver cancer. | [37] | |
| Angiogenesis | Co-culture experiment of 2 cell lines | Electrolyzed reduced H2 water inhibited lumen formation via suppression of VEGF expression in cultured cells. | [38] |
| Cancer Types | No. of Patients | Administration Periods | Effects of Molecular Hydrogen (H2) | Ref. No. |
|---|---|---|---|---|
| Colorectal cancer | 55 | 3-months | Inhalation of H2 gas prolonged progression-free survival and overall survival in patients with stage IV, and the mechanism involved the recovery of CD8+ T cells. | [39] |
| Lung cancer | 42 | 60-months | The combination of H2 gas and Nivolumab showed prolonged overall survival, and the mechanism involved increased coenzyme Q10. | [40] |
| Lung cancer, liver cancer, pancreatic cancer, etc. | 82 | At least 3-months | Inhalation of H2 gas in patients with stage III or IV resulted in complete (CR) and partial remission (PR), with a disease control rate of 57.5%. | [41] |
| Gallbladder cancer | 1 | 10-months | A case study of a patient with gallbladder cancer whose primary tumor was in the liver showed that inhalation of H2 gas had a tumor-reducing effect. | [42,43] |
| Brain tumor | 1 | 4-months | A case study of a patient with a brain tumor whose primary tumor was lung cancer showed that inhalation of H2 gas had a tumor-reducing effect. | [44] |
| Lung cancer | 48 | 5-months | Treatment with H2 gas inhalation alone (10 patients) or in combination with anticancer drugs (38 patients) prolonged progression-free survival in lung cancer patients. | [45] |
| Lung cancer | 20 | 2-weeks | Inhalation of H2 gas improved adaptation and innate immune senescence in lung cancer patients. | [46] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hirano, S.-i.; Yamamoto, H.; Ichikawa, Y.; Sato, B.; Takefuji, Y.; Satoh, F. Molecular Hydrogen as a Novel Antitumor Agent: Possible Mechanisms Underlying Gene Expression. Int. J. Mol. Sci. 2021, 22, 8724. https://doi.org/10.3390/ijms22168724
Hirano S-i, Yamamoto H, Ichikawa Y, Sato B, Takefuji Y, Satoh F. Molecular Hydrogen as a Novel Antitumor Agent: Possible Mechanisms Underlying Gene Expression. International Journal of Molecular Sciences. 2021; 22(16):8724. https://doi.org/10.3390/ijms22168724
Chicago/Turabian StyleHirano, Shin-ichi, Haru Yamamoto, Yusuke Ichikawa, Bunpei Sato, Yoshiyasu Takefuji, and Fumitake Satoh. 2021. "Molecular Hydrogen as a Novel Antitumor Agent: Possible Mechanisms Underlying Gene Expression" International Journal of Molecular Sciences 22, no. 16: 8724. https://doi.org/10.3390/ijms22168724
APA StyleHirano, S.-i., Yamamoto, H., Ichikawa, Y., Sato, B., Takefuji, Y., & Satoh, F. (2021). Molecular Hydrogen as a Novel Antitumor Agent: Possible Mechanisms Underlying Gene Expression. International Journal of Molecular Sciences, 22(16), 8724. https://doi.org/10.3390/ijms22168724








