Biphasic α2β1 Integrin Expression in Breast Cancer Metastasis to Bone
Abstract
1. Introduction
2. Results
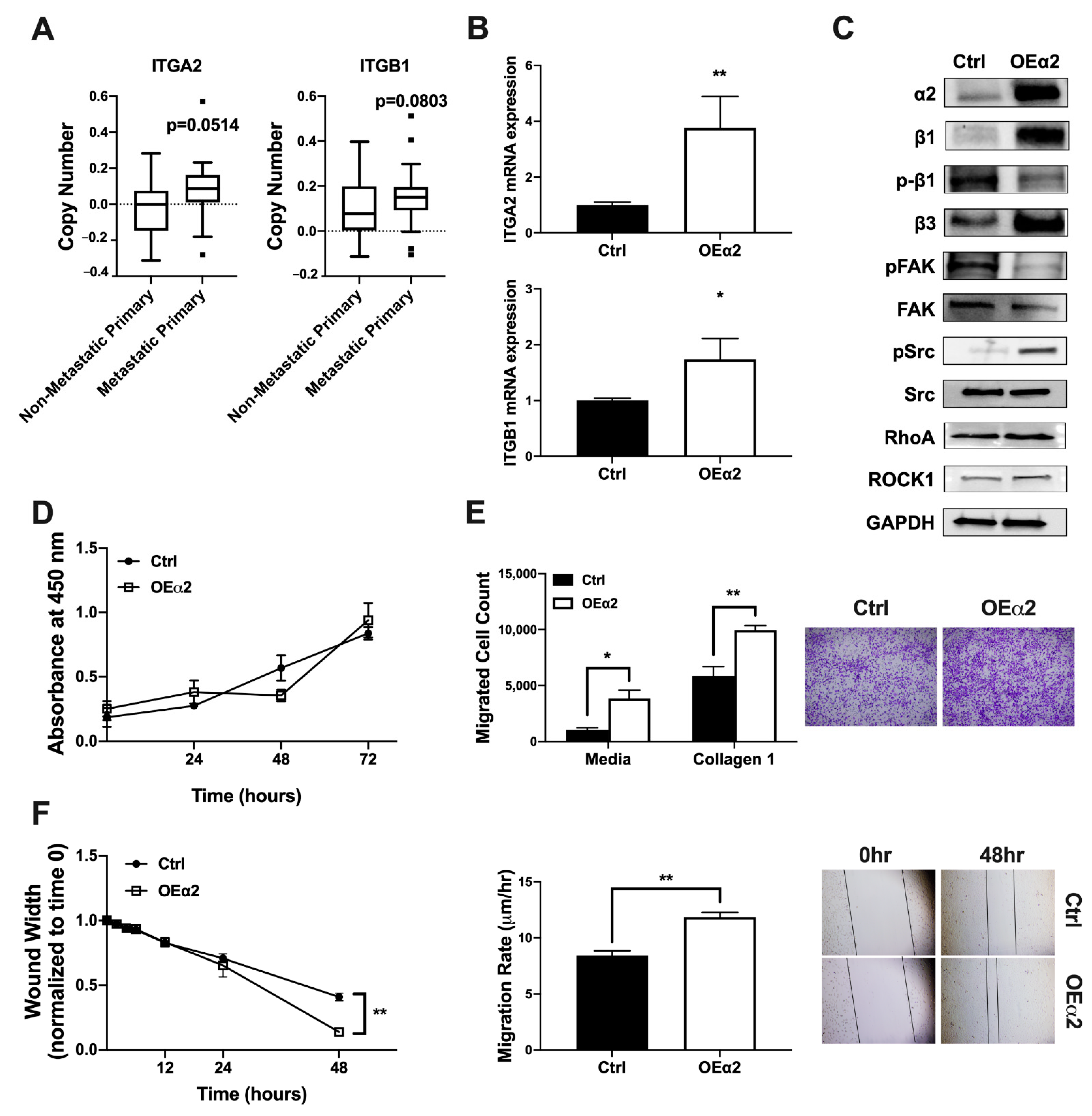
2.1. α2β1 Expression Correlates with an Invasive and Migratory Phenotype
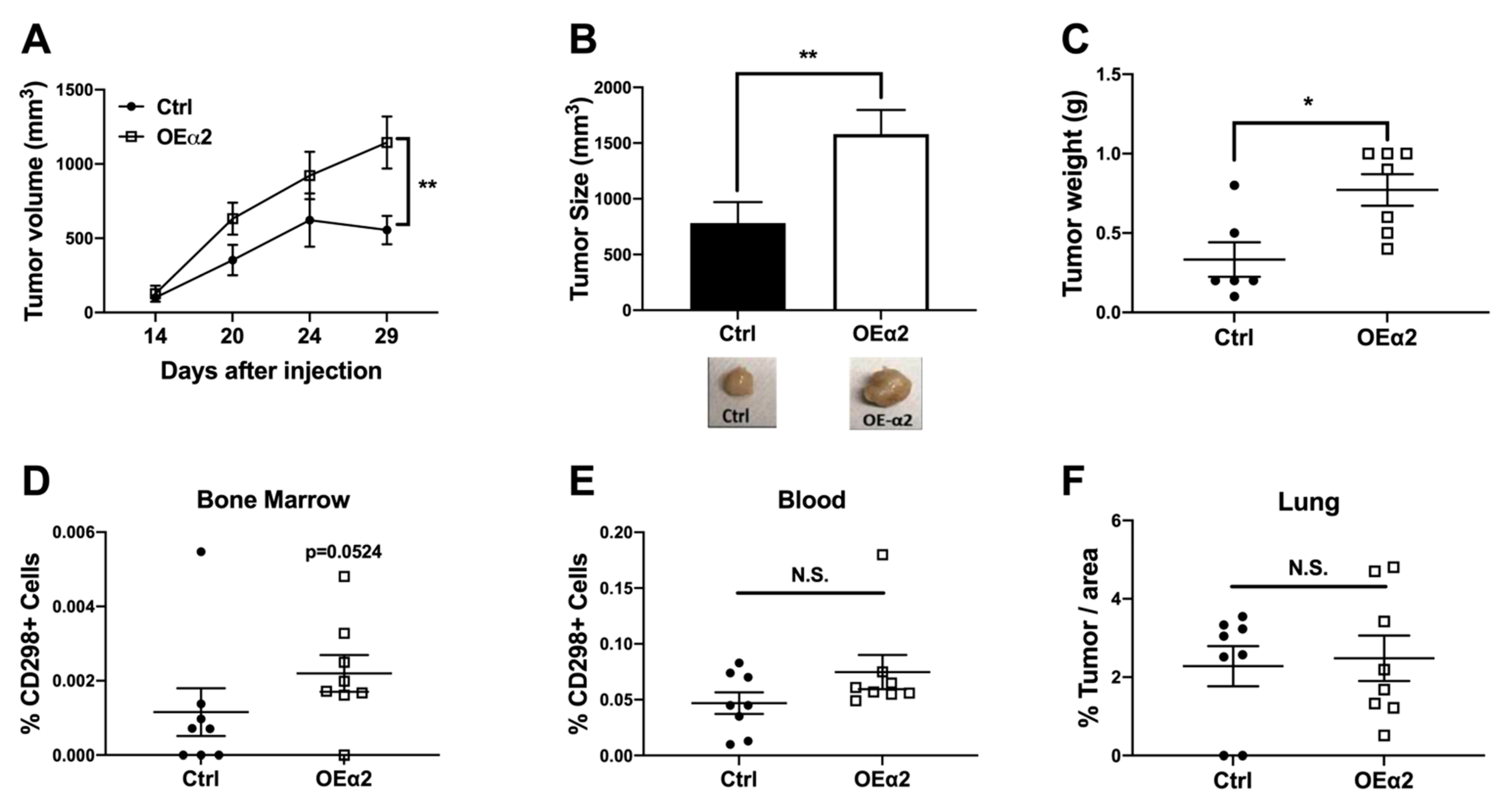
2.2. α2β1 Integrin Promotes Primary Tumor Growth and Dissemination to Bone
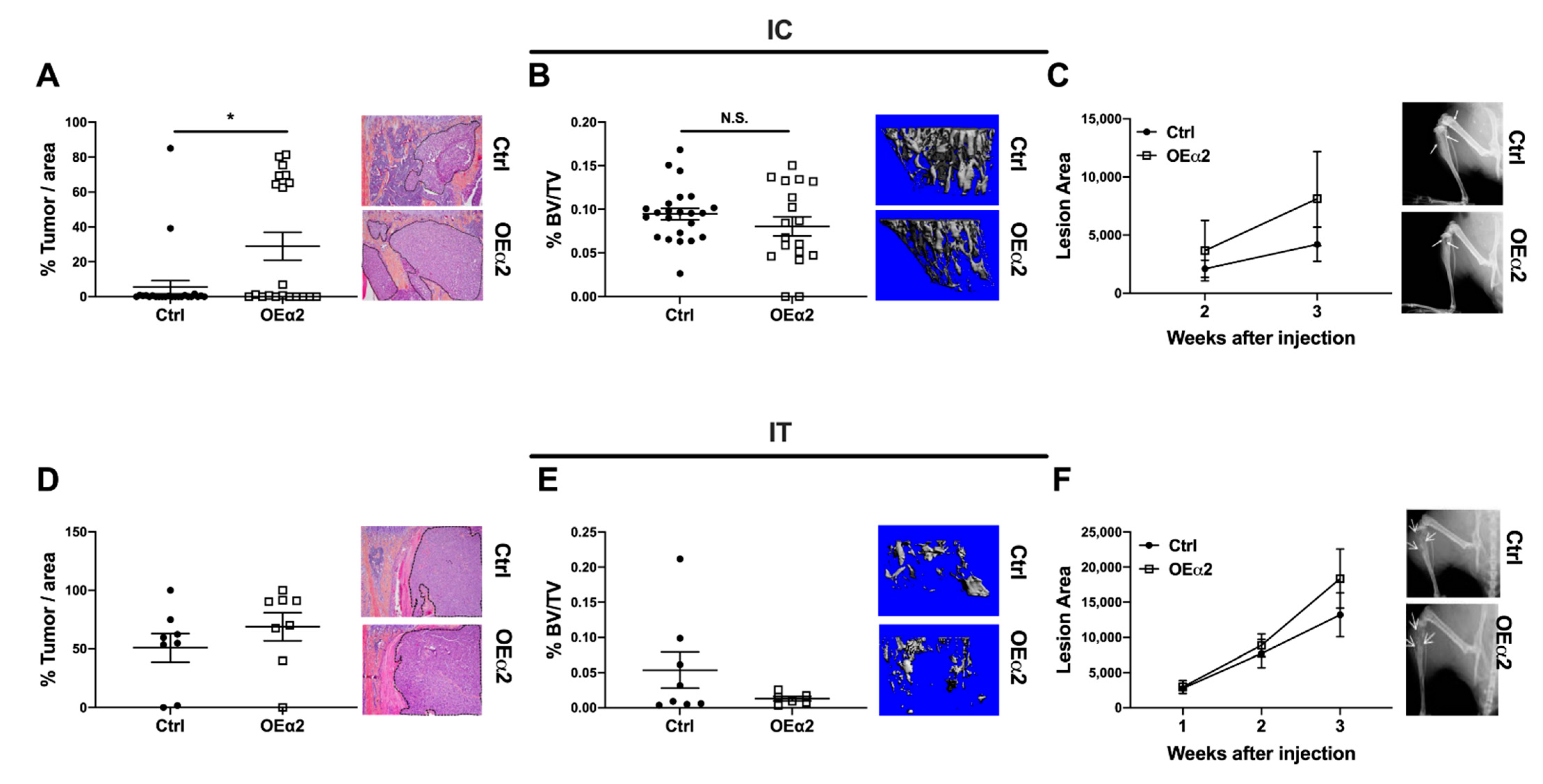
2.3. Breast Cancer Cells with High α2β1 Expression Have Increased Colonization in the Bone, but Have No Effect on Bone Destruction
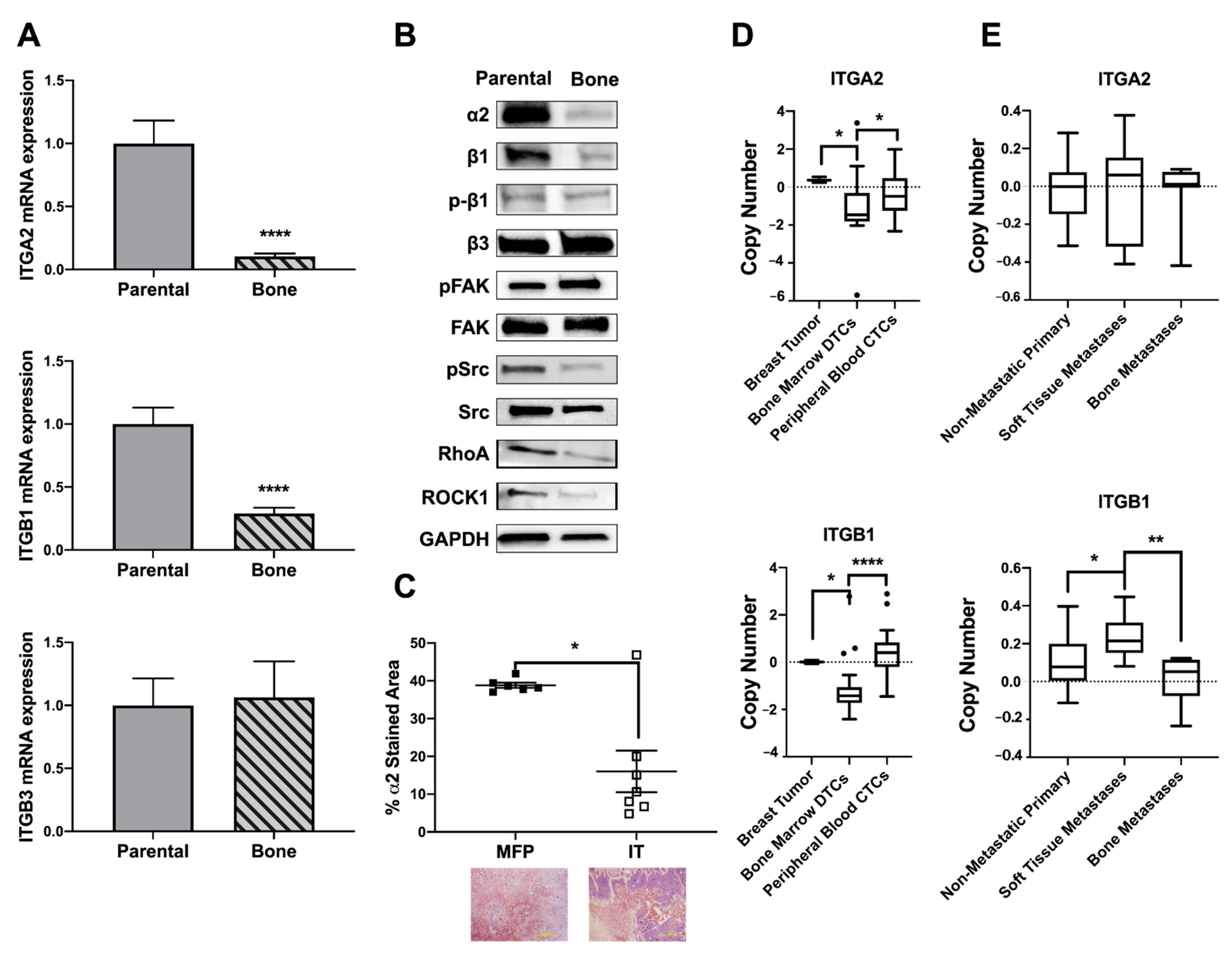
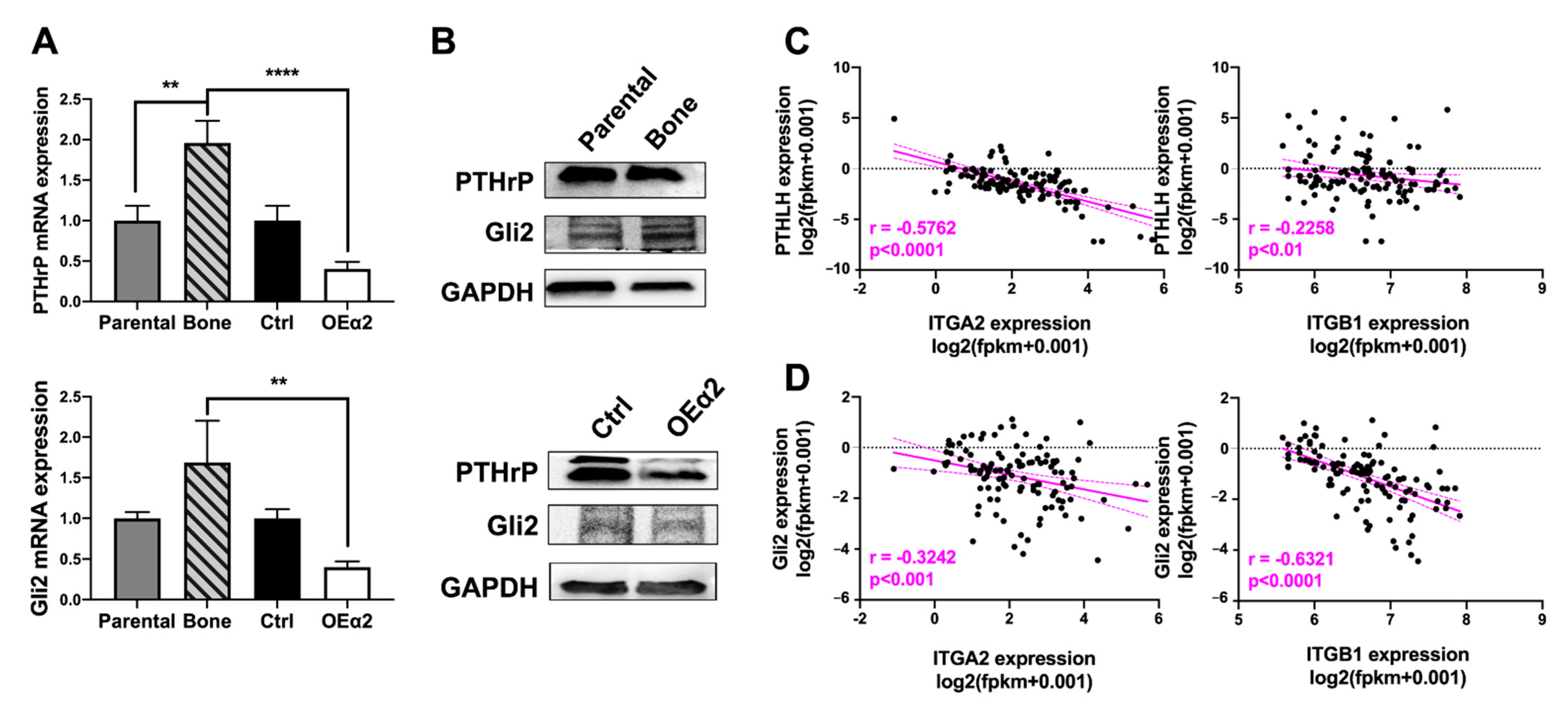
2.4. Osteolytic Breast Tumor Cells Have Decreased α2β1 Expression
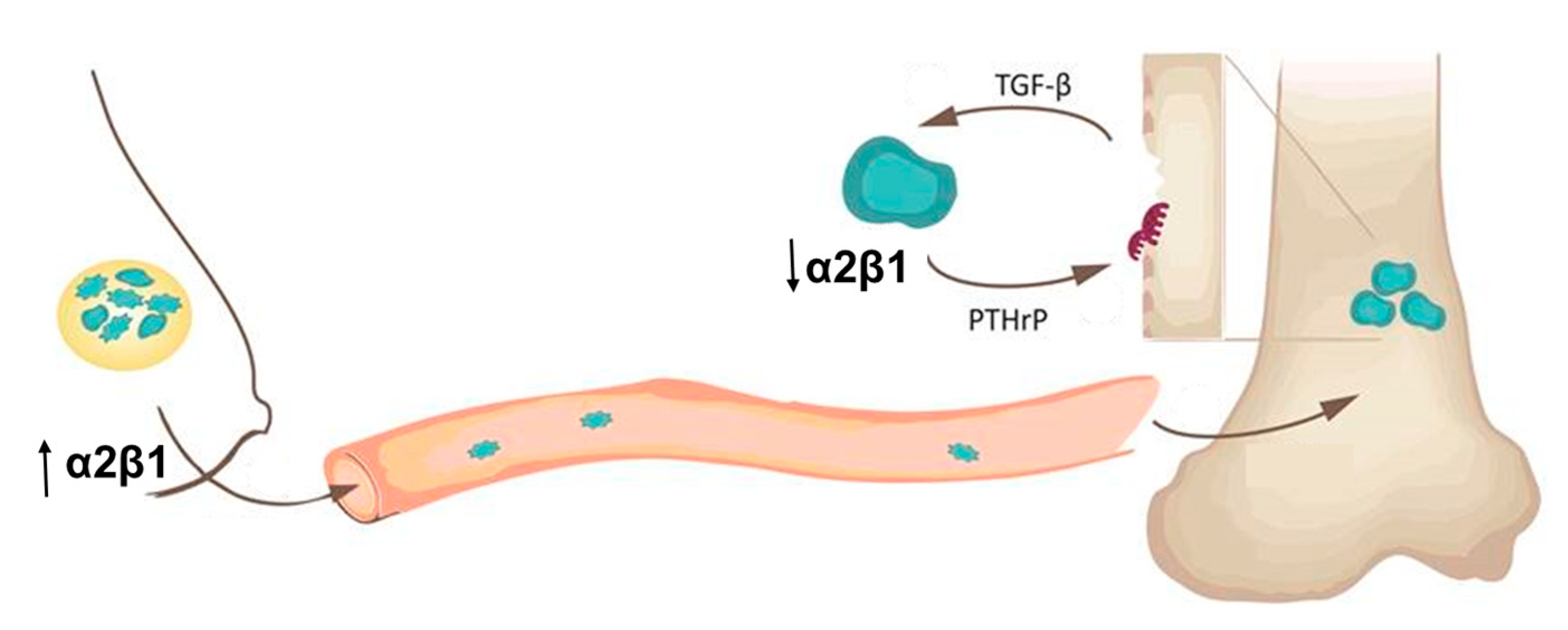
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Transfection
4.3. Quantitative Real-Time PCR
4.4. Western Blot Analysis
4.5. Proliferation Assay
4.6. Invasion Assay
4.7. Scratch Assay
4.8. In Vivo Studies
4.9. Quantification of Tumor Growth
4.10. X-ray Analysis
4.11. Micro Computed Tomography
4.12. Histomorphometry
4.13. Flow Cytometry
4.14. Patient Data Analysis
4.15. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- American Cancer Society. Cancer Facts & Figures; American Cancer Society: Atlanta, GA, USA, 2020. [Google Scholar]
- Pulido, C.; Vendrell, I.; Ferreira, A.R.; Casimiro, S.; Mansinho, A.; Alho, I.; Costa, L. Bone metastasis risk factors in breast cancer. Ecancermedicalscience 2017, 11, 715. [Google Scholar] [CrossRef] [PubMed]
- Buijs, J.T.; van der Pluijm, G. Osteotropic cancers: From primary tumor to bone. Cancer Lett. 2009, 273, 177–193. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.E. Clinical Features of Metastatic Bone Disease and Risk of Skeletal Morbidity. Clin. Cancer Res. 2006, 12, 6243s–6249s. [Google Scholar] [CrossRef] [PubMed]
- Van’T Veer, L.J.; Dai, H.; Van De Vijver, M.J.; He, Y.D.; Hart, A.A.M.; Mao, M.; Peterse, H.L.; Van Der Kooy, K.; Marton, M.J.; Witteveen, A.T.; et al. Gene expression profiling predicts clinical outcome of breast cancer. Nature 2002, 415, 530–536. [Google Scholar] [CrossRef]
- Chiang, A.C.; Massague, J. Molecular Basis of Metastasis. N. Engl. J. Med. 2008, 359, 2814–2823. [Google Scholar] [CrossRef] [PubMed]
- Weigelt, B.; Glas, A.; Wessels, L.F.A.; Witteveen, A.T.; Peterse, J.L.; Veer, L.J.V. Gene expression profiles of primary breast tumors maintained in distant metastases. Proc. Natl. Acad. Sci. USA 2003, 100, 15901–15905. [Google Scholar] [CrossRef]
- Naci, D.; Vuori, K.; Aoudjit, F. Alpha2beta1 integrin in cancer development and chemoresistance. Semin. Cancer Biol. 2015, 35, 145–153. [Google Scholar] [CrossRef]
- Plantefaber, L.C.; Hynes, R.O. Changes in integrin receptors on oncogenically transformed cells. Cell 1989, 56, 281–290. [Google Scholar] [CrossRef]
- Hynes, R.O. Integrins: Bidirectional, Allosteric Signaling Machines. Cell 2002, 110, 673–687. [Google Scholar] [CrossRef]
- Schwartz, M.A.; Schaller, M.D.; Ginsberg, M.H. Integrins: Emerging Paradigms of Signal Transduction. Annu. Rev. Cell Dev. Biol. 1995, 11, 549–599. [Google Scholar] [CrossRef]
- Shattil, S.J.; Kim, C.; Ginsberg, M.H. The final steps of integrin activation: The end game. Nat. Rev. Mol. Cell Biol. 2010, 11, 288–300. [Google Scholar] [CrossRef]
- Hamidi, H.; Ivaska, J. Every step of the way: Integrins in cancer progression and metastasis. Nat. Rev. Cancer 2018, 18, 533–548. [Google Scholar] [CrossRef]
- Albelda, S.M. Role of integrins and other cell adhesion molecules in tumor progression and metastasis—PubMed. Lab. Investig. 1993, 68, 4–17. Available online: https://pubmed.ncbi.nlm.nih.gov/8423675/ (accessed on 7 August 2020).
- Adorno-Cruz, V.; Liu, H. Regulation and functions of integrin α2 in cell adhesion and disease. Genes Dis. 2019, 6, 16–24. [Google Scholar] [CrossRef]
- Hall, C.L.; Dai, J.; Van Golen, K.L.; Keller, E.T.; Long, M.W. Type I Collagen Receptor (α2β1) Signaling Promotes the Growth of Human Prostate Cancer Cells within the Bone. Cancer Res. 2006, 66, 8648–8654. [Google Scholar] [CrossRef]
- Hall, C.L.; Dubyk, C.W.; Riesenberger, T.A.; Shein, D.; Keller, E.T.; van Golen, K.L. Type I Collagen Receptor (α2β1) Signaling Promotes Prostate Cancer Invasion through RhoC GTPase. Neoplasia 2008, 10, 797–803. [Google Scholar] [CrossRef]
- Sottnik, J.L.; Daignault-Newton, S.; Zhang, X.; Morrissey, C.; Hussain, M.H.; Keller, E.T.; Hall, C.L. Integrin alpha2beta1 (α2β1) promotes prostate cancer skeletal metastasis. Clin. Exp. Metastasis 2013, 30, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Tabaries, S.; Dong, Z.; Annis, M.G.; Omeroglu, A.; Pepin, F.; Ouellet, V.; Russo, C.; Hassanain, M.; Metrakos, P.; Diaz, Z.H.; et al. Claudin-2 is selectively enriched in and promotes the formation of breast cancer liver metastases through engagement of integrin complexes. Oncogene 2010, 30, 1318–1328. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, K.; Meckel, K.F.; Laird, L.S.; Chia, C.Y.; Park, J.-J.; Olino, K.; Tsunedomi, R.; Harada, T.; Iizuka, N.; Hazama, S.; et al. Integrin α2 Mediates Selective Metastasis to the Liver. Cancer Res. 2009, 69, 7320–7328. [Google Scholar] [CrossRef] [PubMed]
- Si, J.; Li, A.; Zhou, T.; Guo, L. Si Collagen type I regulates β-catenin tyrosine phosphorylation and nuclear translocation to promote migration and proliferation of gastric carcinoma cells. Oncol. Rep. 2010, 23, 1247–1255. [Google Scholar] [CrossRef][Green Version]
- Matsuoka, T.; Yashiro, M.; Nishimura, S.; Inoue, T.; Fujihara, T.; Sawada, T.; Kato, Y.; Seki, S.; Chung, K.H.-Y. Increased expression of alpha2beta1-integrin in the peritoneal dissemination of human gastric carcinoma. Int. J. Mol. Med. 2000, 5, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.-T.; Chang, C.-C.; Lin, B.-R.; Yang, H.-Y.; Chu, C.-Y.; Wu, M.-H.; Kuo, M.-L. Elevated Expression of Cyr61 Enhances Peritoneal Dissemination of Gastric Cancer Cells through Integrin α2β1. J. Biol. Chem. 2007, 282, 34594–34604. [Google Scholar] [CrossRef] [PubMed]
- Moretti, S.; Martini, L.; Berti, E.; Pinzi, C.; Giannotti, B. Adhesion molecule profile and malignancy of melanocytic lesions. Melanoma Res. 1993, 3, 235–239. Available online: https://pubmed.ncbi.nlm.nih.gov/8219755/ (accessed on 7 August 2020).
- Keely, P.J.; Fong, A.M.; Zutter, M.M.; Santoro, S.A. Alteration of collagen-dependent adhesion, motility, and morphogenesis by the expression of antisense alpha 2 integrin mRNA in mammary cells. J. Cell Sci. 1995, 108 (Pt 2), 595–607. [Google Scholar] [CrossRef]
- Ramirez, N.E.; Zhang, Z.; Madamanchi, A.; Boyd, K.L.; O’Rear, L.D.; Nashabi, A.; Li, Z.; Dupont, W.D.; Zijlstra, A.; Zutter, M.M. The α2β1 integrin is a metastasis suppressor in mouse models and human cancer. J. Clin. Investig. 2011, 121, 226–237. [Google Scholar] [CrossRef]
- Dudley, D.T.; Li, X.-Y.; Hu, C.Y.; Kleer, C.G.; Willis, A.L.; Weiss, S.J. A 3D matrix platform for the rapid generation of therapeutic anti-human carcinoma monoclonal antibodies. Proc. Natl. Acad. Sci. USA 2014, 111, 14882–14887. [Google Scholar] [CrossRef]
- Ibaragi, S.; Shimo, T.; Hassan, N.M.M.; Isowa, S.; Kurio, N.; Mandai, H.; Kodama, S.; Sasaki, A. Induction of MMP-13 expression in bone-metastasizing cancer cells by type I collagen through integrin α1β1 and α2β1-p38 MAPK signaling. Anticancer Res. 2011, 31, 1307–1313. [Google Scholar]
- Schneider, J.G.; Amend, S.R.; Weilbaecher, K.N. Integrins and bone metastasis: Integrating tumor cell and stromal cell interactions. Bone 2011, 48, 54–65. [Google Scholar] [CrossRef]
- Bonkhoff, H.; Stein, U.; Remberger, K. Differential expression of α6 and α2 very late antigen integrins in the normal, hyperplastic, and neoplastic prostate: Simultaneous demonstration of cell surface receptors and their extracellular ligands☆. Hum. Pathol. 1993, 24, 243–248. [Google Scholar] [CrossRef]
- Hall, C.L.; Keller, E.T. Analysis of Integrin Alpha2Beta1 (α2β1) Expression as a Biomarker of Skeletal Metastasis. Biomark. Bone Dis. 2017, 487–506. [Google Scholar]
- Pan, B.; Guo, J.; Liao, Q.; Zhao, Y. β1 and β3 integrins in breast, prostate and pancreatic cancer: A novel implication (Review). Oncol. Lett. 2018, 15, 5412–5416. [Google Scholar] [CrossRef] [PubMed]
- Zutter, M.M.; Santoro, S.A.; Staatz, W.D.; Tsung, Y.L. Re-expression of the alpha 2 beta 1 integrin abrogates the malignant phenotype of breast carcinoma cells. Proc. Natl. Acad. Sci. USA 1995, 92, 7411–7415. [Google Scholar] [CrossRef] [PubMed]
- Zutter, M.M.; Mazoujian, G.; Santoro, S.A. Decreased expression of integrin adhesive protein receptors in adenocarci-noma of the breast. Am. J. Pathol. 1990, 137, 863–870. Available online: http://www.ncbi.nlm.nih.gov/pubmed/2221016 (accessed on 7 August 2020).
- Sowder, M.E.; Johnson, R.W. Enrichment and detection of bone disseminated tumor cells in models of low tumor burden. Sci. Rep. 2018, 8, 14299. [Google Scholar] [CrossRef]
- Chen, M.B.; Lamar, J.; Li, R.; Hynes, R.O.; Kamm, R.D. Elucidation of the Roles of Tumor Integrin β1 in the Extravasation Stage of the Metastasis Cascade. Cancer Res. 2016, 76, 2513–2524. [Google Scholar] [CrossRef]
- Kwakwa, K.A.; Sterling, J.A. Integrin αvβ3 Signaling in Tumor-Induced Bone Disease. Cancers 2017, 9, 84. [Google Scholar] [CrossRef]
- Mathiesen, R.R.; Fjelldal, R.; Liestøl, K.; Due, E.U.; Geigl, J.B.; Riethdorf, S.; Borgen, E.; Rye, I.H.; Schneider, I.J.; Obenauf, A.C.; et al. High-resolution analyses of copy number changes in disseminated tumor cells of patients with breast cancer. Int. J. Cancer 2011, 131, E405–E415. [Google Scholar] [CrossRef]
- Wagle, N.; Painter, C.; Anastasio, E.; Dunphy, M.; McGillicuddy, M.; Kim, D.; Jain, E.; Buendia-Buendia, J.; Cohen, O.; Knelson, E.; et al. The Metastatic Breast Cancer (MBC) project: Accelerating translational research through direct patient engagement. J. Clin. Oncol. 2017, 35, 1076. [Google Scholar] [CrossRef]
- The Metastatic Breast Cancer Project. Available online: https://mbcproject.org/data-release (accessed on 7 August 2020).
- Buenrostro, D.; Mulcrone, P.L.; Owens, P.; Sterling, J.A. The Bone Microenvironment: A Fertile Soil for Tumor Growth. Curr. Osteoporos. Rep. 2016, 14, 151–158. [Google Scholar] [CrossRef]
- Sterling, J.A.; Edwards, J.R.; Martin, T.J.; Mundy, G.R. Advances in the biology of bone metastasis: How the skeleton affects tumor behavior. Bone 2011, 48, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Powell, G.J.; Southby, J.; Danks, J.A.; Stillwell, R.G.; Hayman, J.A.; Henderson, M.A.; Bennett, R.C.; Martin, T.J. Localization of parathyroid hormone-related protein in breast cancer metastases: Increased incidence in bone compared with other sites. Cancer Res. 1991, 51, 3059–3061. [Google Scholar] [PubMed]
- Southby, J.; Kissin, M.W.; Danks, J.A.; Hayman, J.A.; Moseley, J.M.; Henderson, M.A.; Bennett, R.C.; Martin, T.J. Immunohistochemical localization of parathyroid hormone-related protein in human breast cancer. Cancer Res. 1990, 50, 7710–7716. [Google Scholar]
- Boyle, W.J.; Simonet, W.S.; Lacey, D.L. Osteoclast differentiation and activation. Nat. Cell Biol. 2003, 423, 337–342. [Google Scholar] [CrossRef]
- Yin, J.J.; Selander, K.; Chirgwin, J.M.; Dallas, M.; Grubbs, B.G.; Wieser, R.; Massague, J.; Mundy, G.R.; Guise, T.A. TGF-β signaling blockade inhibits PTHrP secretion by breast cancer cells and bone metastases development. J. Clin. Investig. 1999, 103, 197–206. [Google Scholar] [CrossRef]
- Biswas, S.; Nyman, J.S.; Alvarez, J.; Chakrabarti, A.; Ayres, A.; Sterling, J.; Edwards, J.; Rana, T.; Johnson, R.; Perrien, D.S.; et al. Anti-Transforming Growth Factor ß Antibody Treatment Rescues Bone Loss and Prevents Breast Cancer Metastasis to Bone. PLoS ONE 2011, 6, e27090. [Google Scholar] [CrossRef]
- Johnson, R.W.; Nguyen, M.P.; Padalecki, S.S.; Grubbs, B.G.; Merkel, A.; Oyajobi, B.O.; Matrisian, L.M.; Mundy, G.R.; Sterling, J.A. TGF-β Promotion of Gli2-Induced Expression of Parathyroid Hormone-Related Protein, an Important Osteolytic Factor in Bone Metastasis, Is Independent of Canonical Hedgehog Signaling. Cancer Res. 2011, 71, 822–831. [Google Scholar] [CrossRef] [PubMed]
- Sterling, J.A.; Oyajobi, B.O.; Grubbs, B.; Padalecki, S.S.; Munoz, S.A.; Gupta, A.; Story, B.; Zhao, M.; Mundy, G.R. The Hedgehog Signaling Molecule Gli2 Induces Parathyroid Hormone-Related Peptide Expression and Osteolysis in Metastatic Human Breast Cancer Cells. Cancer Res. 2006, 66, 7548–7553. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.R.; Wu, Y.-M.; Lonigro, R.J.; Vats, P.; Cobain, E.; Everett, J.; Cao, X.; Rabban, E.; Kumar, C.; Raymond, V.; et al. Integrative clinical genomics of metastatic cancer. Nat. Cell Biol. 2017, 548, 297–303. [Google Scholar] [CrossRef]
- Adorno-Cruz, V.; Hoffmann, A.D.; Liu, X.; Dashzeveg, N.K.; Taftaf, R.; Wray, B.; Keri, R.A.; Liu, H. ITGA2 promotes expression of ACLY and CCND1 in enhancing breast cancer stemness and metastasis. Genes Dis. 2021, 8, 493–508. [Google Scholar] [CrossRef]
- Howe, G.A.; Addison, C.L. β1 integrin. Cell Adhes. Migr. 2012, 6, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P. Epithelial–mesenchymal transitions in tumour progression. Nat. Rev. Cancer 2002, 2, 442–454. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, A.; Mourtzoukou, D.; Kosmidou, V.; Avlonitis, S.; Kontogeorgos, G.; Zografos, G.; Pintzas, A. EZH2 is regulated by ERK/AKT and targets integrin alpha2 gene to control Epithelial–Mesenchymal Transition and anoikis in colon cancer cells. Int. J. Biochem. Cell Biol. 2013, 45, 243–254. [Google Scholar] [CrossRef]
- Wafai, R.; Williams, E.D.; De Souza, E.; Simpson, P.T.; Reed, A.E.M.; Kutasovic, J.R.; Waltham, M.; Snell, C.E.; Blick, T.; Thompson, E.W.; et al. Integrin alpha-2 and beta-1 expression increases through multiple generations of the EDW01 patient-derived xenograft model of breast cancer—insight into their role in epithelial mesenchymal transition in vivo gained from an in vitro model system. Breast Cancer Res. 2020, 22, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Avizienyte, E.; Frame, M.C. Src and FAK signalling controls adhesion fate and the epithelial-to-mesenchymal transition. Curr. Opin. Cell Biol. 2005, 17, 542–547. [Google Scholar] [CrossRef]
- Haage, A.; Wagner, K.; Deng, W.; Venkatesh, B.; Mitchell, C.; Goodwin, K.; Bogutz, A.; Lefebvre, L.; Van Raamsdonk, C.; Tanentzapf, G. Precise coordination of cell-ECM adhesion is essential for efficient melanoblast migration during development. Development 2020, 147. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.; Barlow, B.; O’Rear, L.; Jarvis, B.; Li, Z.; Dickeson, K.; Dupont, W.; Zutter, M. Loss of the α2β1 Integrin Alters Human Papilloma Virus-Induced Squamous Carcinoma Progression In Vivo and In Vitro. PLoS ONE 2011, 6, e26858. [Google Scholar] [CrossRef]
- Armstrong, A.J.; Marengo, M.S.; Oltean, S.; Kemeny, G.; Bitting, R.; Turnbull, J.D.; Herold, C.I.; Marcom, P.K.; George, D.J.; Garcia-Blanco, M.A. Circulating Tumor Cells from Patients with Advanced Prostate and Breast Cancer Display Both Epithelial and Mesenchymal Markers. Mol. Cancer Res. 2011, 9, 997–1007. [Google Scholar] [CrossRef]
- Liu, S. The ROCK signaling and breast cancer metastasis. Mol. Biol. Rep. 2010, 38, 1363–1366. [Google Scholar] [CrossRef]
- Sterling, J.A.; Guelcher, S.A. Bone Structural Components Regulating Sites of Tumor Metastasis. Curr. Osteoporos. Rep. 2011, 9, 89–95. [Google Scholar] [CrossRef]
- Guelcher, S.A.; Sterling, J.A. Contribution of Bone Tissue Modulus to Breast Cancer Metastasis to Bone. Cancer Microenviron. 2011, 4, 247–259. [Google Scholar] [CrossRef]
- Parvani, J.G.; Galliher-Beckley, A.J.; Schiemann, B.J.; Schiemann, W.P. Targeted inactivation of β1 integrin induces β3 integrin switching, which drives breast cancer metastasis by TGF-β. Mol. Biol. Cell 2013, 24, 3449–3459. [Google Scholar] [CrossRef]
- Zhao, Y.; Bachelier, R.; Treilleux, I.; Pujuguet, P.; Peyruchaud, O.; Baron, R.; Clément-Lacroix, P.; Clézardin, P. Tumor αvβ3 Integrin Is a Therapeutic Target for Breast Cancer Bone Metastases. Cancer Res. 2007, 67, 5821–5830. [Google Scholar] [CrossRef]
- Page, J.M.; Merkel, A.R.; Ruppender, N.S.; Guo, R.; Dadwal, U.C.; Cannonier, S.A.; Basu, S.; Guelcher, S.A.; Sterling, J.A. Matrix rigidity regulates the transition of tumor cells to a bone-destructive phenotype through integrin β3 and TGF-β receptor type II. Biomaterials 2015, 64, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Munger, J.S.; Sheppard, D. Cross Talk among TGF- Signaling Pathways, Integrins, and the Extracellular Matrix. Cold Spring Harb. Perspect. Biol. 2011, 3, a005017. [Google Scholar] [CrossRef] [PubMed]
- Yoneda, T.; Williams, P.J.; Hiraga, T.; Niewolna, M.; Nishimura, R. A Bone-Seeking Clone Exhibits Different Biological Properties from the MDA-MB-231 Parental Human Breast Cancer Cells and a Brain-Seeking Clone In Vivo and In Vitro. J. Bone Miner. Res. 2001, 16, 1486–1495. [Google Scholar] [CrossRef] [PubMed]
- Das, D.K.; Naidoo, M.K.; Ilboudo, A.; Dubois, P.; Durojaiye, V.; Liu, C.; Ogunwobi, O.O. Isolation and Propagation of Circulating Tumor Cells from a Mouse Cancer Model. J. Vis. Exp. 2015, 2015, e52861. [Google Scholar] [CrossRef]
- Edgar, R.; Domrachev, M.; Lash, A.E. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res. 2002, 30, 207–210. [Google Scholar] [CrossRef]
- Gao, J.; Aksoy, B.A.; Dogrusoz, U.; Dresdner, G.; Gross, B.; Sumer, S.O.; Sun, Y.; Jacobsen, A.; Sinha, R.; Larsson, E.; et al. Integrative Analysis of Complex Cancer Genomics and Clinical Profiles Using the cBioPortal. Sci. Signal. 2013, 6, pl1. [Google Scholar] [CrossRef] [PubMed]
- Cerami, E.; Gao, J.; Dogrusoz, U.; Gross, B.E.; Sumer, S.O.; Aksoy, B.A.; Jacobsen, A.; Byrne, C.J.; Heuer, M.L.; Larsson, E.; et al. The cBio Cancer Genomics Portal: An Open Platform for Exploring Multidimensional Cancer Genomics Data. Cancer Discov. 2012, 2, 401–404. [Google Scholar] [CrossRef]
- Correction: The cBio Cancer Genomics Portal: An Open Platform for Exploring Multidimensional Cancer Genomics Data. Cancer Discov. 2012, 2, 960. [CrossRef]
- Goldman, M.J.; Craft, B.; Hastie, M.; Repečka, K.; McDade, F.; Kamath, A.; Banerjee, A.; Luo, Y.; Rogers, D.; Brooks, A.N.; et al. Visualizing and interpreting cancer genomics data via the Xena platform. Nat. Biotechnol. 2020, 38, 675–678. [Google Scholar] [CrossRef] [PubMed]
| Target | Dilution | Catalog # | |
|---|---|---|---|
| Integrin Receptors | α2 | 1:5000 | ab133557 |
| β1 | 1:5000 | sc-8978 | |
| phospho-β1 | 1:1000 | ab5189 | |
| β3 | 1:1000 | sc13579 | |
| Integrin Signaling | FAK | 1:1000 | ab40794 |
| phospho-FAK | 1:500 | ab81298 | |
| Src | 1:1000 | 2108 1 | |
| phospho-Src | 1:1000 | 2105 1 | |
| RhoA | 1:1000 | 2117 1 | |
| ROCK-1 | 1:500 | sc-5560 | |
| Osteolytic Signaling | Gli2 | 1:500 | NB600-874 |
| PTHrP | 1:1000 | NBP1-59322 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moritz, M.N.O.; Merkel, A.R.; Feldman, E.G.; Selistre-de-Araujo, H.S.; Rhoades, J.A. Biphasic α2β1 Integrin Expression in Breast Cancer Metastasis to Bone. Int. J. Mol. Sci. 2021, 22, 6906. https://doi.org/10.3390/ijms22136906
Moritz MNO, Merkel AR, Feldman EG, Selistre-de-Araujo HS, Rhoades JA. Biphasic α2β1 Integrin Expression in Breast Cancer Metastasis to Bone. International Journal of Molecular Sciences. 2021; 22(13):6906. https://doi.org/10.3390/ijms22136906
Chicago/Turabian StyleMoritz, Milene N.O., Alyssa R. Merkel, Ean G. Feldman, Heloisa S. Selistre-de-Araujo, and Julie A. Rhoades (Sterling). 2021. "Biphasic α2β1 Integrin Expression in Breast Cancer Metastasis to Bone" International Journal of Molecular Sciences 22, no. 13: 6906. https://doi.org/10.3390/ijms22136906
APA StyleMoritz, M. N. O., Merkel, A. R., Feldman, E. G., Selistre-de-Araujo, H. S., & Rhoades, J. A. (2021). Biphasic α2β1 Integrin Expression in Breast Cancer Metastasis to Bone. International Journal of Molecular Sciences, 22(13), 6906. https://doi.org/10.3390/ijms22136906







