Shared Biological Pathways between Antipsychotics and Omega-3 Fatty Acids: A Key Feature for Schizophrenia Preventive Treatment?
Abstract
1. Introduction
2. The Role of Omega-3 in the Plasma Membrane Composition and Dynamics
3. Effects of Omega-3 on the Endocannabinoid System
4. Effects of Omega-3 on the Dopaminergic and Serotonergic Systems
5. Effects of Omega-3 on the Glutamatergic System
6. The Role of Omega-3 in Myelin Constitution and Function
7. The Effect of Omega-3 in Anti-Inflammatory and Antioxidant Pathways
8. Effect of Omega-3 on the One-Carbon Metabolism
9. Omega-3 Overall Actions and Shared Biological Pathways with Antipsychotics
10. Omega-3 Supplementation at Different Stages of Schizophrenia
11. Conclusions
Funding
Conflicts of Interest
References
- Charlson, F.J.; Ferrari, A.J.; Santomauro, D.F.; Diminic, S.; Stockings, E.; Scott, J.G.; McGrath, J.J.; Whiteford, H.A. Global Epidemiology and Burden of Schizophrenia: Findings from the Global Burden of Disease Study 2016. Schizophr. Bull. 2018. [Google Scholar] [CrossRef]
- Fusar-Poli, P.; Bonoldi, I.; Yung, A.R.; Borgwardt, S.; Kempton, M.J.; Valmaggia, L.; Barale, F.; Caverzasi, E.; McGuire, P. Predicting Psychosis: Meta-Analysis of Transition Outcomes in Individuals at High Clinical Risk. Arch. Gen. Psychiatry 2012, 69, 220–229. [Google Scholar] [CrossRef]
- Pillinger, T.; D’Ambrosio, E.; McCutcheon, R.; Howes, O.D. Is Psychosis a Multisystem Disorder? A Meta-Review of Central Nervous System, Immune, Cardiometabolic, and Endocrine Alterations in First-Episode Psychosis and Perspective on Potential Models. Mol. Psychiatry 2019, 24, 776–794. [Google Scholar] [CrossRef] [PubMed]
- Howes, O.; McCutcheon, R.; Stone, J. Glutamate and Dopamine in Schizophrenia: An Update for the 21st Century. J. Psychopharmacol. 2015, 29, 97–115. [Google Scholar] [CrossRef]
- Koga, M.; Serritella, A.V.; Sawa, A.; Sedlak, T.W. Implications for Reactive Oxygen Species in Schizophrenia Pathogenesis. Schizophr. Res. 2016, 176, 52–71. [Google Scholar] [CrossRef] [PubMed]
- Pillinger, T.; Osimo, E.F.; Brugger, S.; Mondelli, V.; McCutcheon, R.A.; Howes, O.D. A Meta-Analysis of Immune Parameters, Variability, and Assessment of Modal Distribution in Psychosis and Test of the Immune Subgroup Hypothesis. Schizophr. Bull. 2018. [Google Scholar] [CrossRef] [PubMed]
- Frajerman, A.; Kebir, O.; Chaumette, B.; Tessier, C.; Lamazière, A.; Nuss, P.; Krebs, M.-O. Membrane lipids in schizophrenia and early phases of psychosis: Potential biomarkers and therapeutic targets? Encephale 2020. [Google Scholar] [CrossRef]
- Krebs, M.O.; Bellon, A.; Mainguy, G.; Jay, T.M.; Frieling, H. One-Carbon Metabolism and Schizophrenia: Current Challenges and Future Directions. Trends Mol. Med. 2009, 15, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Chokhawala, K.; Stevens, L. Antipsychotic Medications. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Sumiyoshi, T.; Matsui, M.; Itoh, H.; Higuchi, Y.; Arai, H.; Takamiya, C.; Kurachi, M. Essential Polyunsaturated Fatty Acids and Social Cognition in Schizophrenia. Psychiatry Res. 2008, 157, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Bowling, H.; Santini, E. Unlocking the Molecular Mechanisms of Antipsychotics—A New Frontier for Discovery. Swiss. Med. Wkly. 2016, 146, w14314. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sadowska-Bartosz, I.; Galiniak, S.; Bartosz, G.; Zuberek, M.; Grzelak, A.; Dietrich-Muszalska, A. Antioxidant Properties of Atypical Antipsychotic Drugs Used in the Treatment of Schizophrenia. Schizophr. Res. 2016, 176, 245–251. [Google Scholar] [CrossRef]
- Spertus, J.; Horvitz-Lennon, M.; Abing, H.; Normand, S.-L. Risk of Weight Gain for Specific Antipsychotic Drugs: A Meta-Analysis. NPJ Schizophr. 2018, 4, 12. [Google Scholar] [CrossRef]
- Lally, J.; Ajnakina, O.; Stubbs, B.; Williams, H.R.; Colizzi, M.; Carra, E.; Fraietta, S.; Gardner-Sood, P.; Greenwood, K.E.; Atakan, Z.; et al. Hyperprolactinaemia in First Episode Psychosis—A Longitudinal Assessment. Schizophr. Res. 2017, 189, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Monteleone, P.; Cascino, G.; Monteleone, A.M.; Rocca, P.; Rossi, A.; Bertolino, A.; Aguglia, E.; Amore, M.; Collantoni, E.; Corrivetti, G.; et al. Prevalence of Antipsychotic-Induced Extrapyramidal Symptoms and Their Association with Neurocognition and Social Cognition in Outpatients with Schizophrenia in the “Real-Life”. Prog. Neuropsychopharmacol. Biol. Psychiatry 2021, 109, 110250. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Xu, L.; Tang, X.; Wei, Y.; Hu, Q.; Hu, Y.; Cui, H.; Tang, Y.; Hui, L.; Li, C.; et al. Real-World Effectiveness of Antipsychotic Treatment in Psychosis Prevention in a 3-Year Cohort of 517 Individuals at Clinical High Risk from the SHARP (ShangHai at Risk for Psychosis). Aust. N. Z. J. Psychiatry 2020, 54, 696–706. [Google Scholar] [CrossRef]
- Fahy, E.; Subramaniam, S.; Murphy, R.C.; Nishijima, M.; Raetz, C.R.H.; Shimizu, T.; Spener, F.; van Meer, G.; Wakelam, M.J.O.; Dennis, E.A. Update of the LIPID MAPS Comprehensive Classification System for Lipids. J. Lipid Res. 2009, 50, S9–S14. [Google Scholar] [CrossRef] [PubMed]
- Liebisch, G.; Fahy, E.; Aoki, J.; Dennis, E.A.; Durand, T.; Ejsing, C.S.; Fedorova, M.; Feussner, I.; Griffiths, W.J.; Köfeler, H.; et al. Update on LIPID MAPS Classification, Nomenclature, and Shorthand Notation for MS-Derived Lipid Structures. J. Lipid Res. 2020, 61, 1539–1555. [Google Scholar] [CrossRef] [PubMed]
- Rizos, E.C.; Markozannes, G.; Tsapas, A.; Mantzoros, C.S.; Ntzani, E.E. Omega-3 Supplementation and Cardiovascular Disease: Formulation-Based Systematic Review and Meta-Analysis with Trial Sequential Analysis. Heart 2021, 107, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Perica, M.M.; Delas, I. Essential Fatty Acids and Psychiatric Disorders. Nutr. Clin. Pract. 2011, 26, 409–425. [Google Scholar] [CrossRef]
- Bentsen, H.; Solberg, D.K.; Refsum, H.; Bøhmer, T. Clinical and Biochemical Validation of Two Endophenotypes of Schizophrenia Defined by Levels of Polyunsaturated Fatty Acids in Red Blood Cells. Prostaglandins Leukot. Essent. Fatty Acids 2012, 87, 35–41. [Google Scholar] [CrossRef]
- Solberg, D.K.; Bentsen, H.; Refsum, H.; Andreassen, O.A. Association between Serum Lipids and Membrane Fatty Acids and Clinical Characteristics in Patients with Schizophrenia. Acta Psychiatr. Scand. 2015. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.; Nelson, B.; Markulev, C.; Yuen, H.P.; Schäfer, M.R.; Mossaheb, N.; Schlögelhofer, M.; Smesny, S.; Hickie, I.B.; Berger, G.E.; et al. Relationship Between Polyunsaturated Fatty Acids and Psychopathology in the NEURAPRO Clinical Trial. Front. Psychiatry 2019, 10, 393. [Google Scholar] [CrossRef]
- Kim, S.-W.; Schäfer, M.R.; Klier, C.M.; Berk, M.; Rice, S.; Allott, K.; Bartholomeusz, C.F.; Whittle, S.L.; Pilioussis, E.; Pantelis, C.; et al. Relationship between Membrane Fatty Acids and Cognitive Symptoms and Information Processing in Individuals at Ultra-High Risk for Psychosis. Schizophr. Res. 2014, 158, 39–44. [Google Scholar] [CrossRef]
- Kuharic, D.B.; Kekin, I.; Hew, J.; Kuzman, M.R.; Puljak, L. Interventions for Prodromal Stage of Psychosis. Cochrane Database Syst. Rev. 2019. [Google Scholar] [CrossRef]
- Chang, C.-H.; Tseng, P.-T.; Chen, N.-Y.; Lin, P.-C.; Lin, P.-Y.; Chang, J.P.-C.; Kuo, F.-Y.; Lin, J.; Wu, M.-C.; Su, K.-P. Safety and Tolerability of Prescription Omega-3 Fatty Acids: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Prostaglandins Leukot. Essent. Fatty Acids 2018, 129, 1–12. [Google Scholar] [CrossRef]
- Simons, M.; Krämer, E.-M.; Thiele, C.; Stoffel, W.; Trotter, J. Assembly of Myelin by Association of Proteolipid Protein with Cholesterol-and Galactosylceramide-Rich Membrane Domains. J. Cell. Biol. 2000, 151, 143–154. [Google Scholar] [CrossRef]
- Jacobson, K.; Mouritsen, O.G.; Anderson, R.G.W. Lipid Rafts: At a Crossroad between Cell Biology and Physics. Nat. Cell Biol. 2007, 9, 7–14. [Google Scholar] [CrossRef]
- Kasahara, K.; Sanai, Y. Functional Roles of Glycosphingolipids in Signal Transduction via Lipid Rafts. Glycoconj J. 2000, 17, 153–162. [Google Scholar] [CrossRef]
- Brown, D.A.; London, E. Structure and Function of Sphingolipid- and Cholesterol-Rich Membrane Rafts. J. Biol. Chem. 2000, 275, 17221–17224. [Google Scholar] [CrossRef] [PubMed]
- Borroni, M.V.; Vallés, A.S.; Barrantes, F.J. The Lipid Habitats of Neurotransmitter Receptors in Brain. Biochim. Biophys. Acta 2016, 1858, 2662–2670. [Google Scholar] [CrossRef]
- Fantini, J.; Barrantes, F.J. Sphingolipid/Cholesterol Regulation of Neurotransmitter Receptor Conformation and Function. Biochim. Biophys. Acta 2009, 1788, 2345–2361. [Google Scholar] [CrossRef] [PubMed]
- Bazinet, R.P.; Layé, S. Polyunsaturated Fatty Acids and Their Metabolites in Brain Function and Disease. Nature Rev. Neurosci. 2014, 15, 771–785. [Google Scholar] [CrossRef]
- Horrobin, D.F.; Glen, A.I.M.; Vaddadi, K. The Membrane Hypothesis of Schizophrenia. Schizophr. Res. 1994, 13, 195–207. [Google Scholar] [CrossRef]
- Guixà-González, R.; Javanainen, M.; Gómez-Soler, M.; Cordobilla, B.; Domingo, J.C.; Sanz, F.; Pastor, M.; Ciruela, F.; Martinez-Seara, H.; Selent, J. Membrane Omega-3 Fatty Acids Modulate the Oligomerisation Kinetics of Adenosine A2A and Dopamine D2 Receptors. Sci. Rep. 2016, 6, 19839. [Google Scholar] [CrossRef]
- Pérez-Isidoro, R.; Costas, M. The Effect of Neuroleptic Drugs on DPPC/Sphingomyelin/Cholesterol Membranes. Chem. Phys. Lipids 2020, 229, 104913. [Google Scholar] [CrossRef] [PubMed]
- Sethom, M.M.; Fares, S.; Bouaziz, N.; Melki, W.; Jemaa, R.; Feki, M.; Hechmi, Z.; Kaabachi, N. Polyunsaturated Fatty Acids Deficits Are Associated with Psychotic State and Negative Symptoms in Patients with Schizophrenia. Prostaglandins Leukot. Essent. Fatty Acids 2010, 83, 131–136. [Google Scholar] [CrossRef]
- Morgan, K.; Martucci, N.; Kozlowska, A.; Gamal, W.; Brzeszczyński, F.; Treskes, P.; Samuel, K.; Hayes, P.; Nelson, L.; Bagnaninchi, P.; et al. Chlorpromazine Toxicity Is Associated with Disruption of Cell Membrane Integrity and Initiation of a Pro-Inflammatory Response in the HepaRG Hepatic Cell Line. Biomed. Pharmacother. 2019, 111, 1408–1416. [Google Scholar] [CrossRef]
- Weston-Green, K.; Babic, I.; de Santis, M.; Pan, B.; Montgomery, M.K.; Mitchell, T.; Huang, X.-F.; Nealon, J. Disrupted Sphingolipid Metabolism Following Acute Clozapine and Olanzapine Administration. J. Biomed. Sci. 2018, 25, 40. [Google Scholar] [CrossRef]
- McNamara, R.K.; Jandacek, R.; Rider, T.; Tso, P.; Cole-Strauss, A.; Lipton, J.W. Differential Effects of Antipsychotic Medications on Polyunsaturated Fatty Acid Biosynthesis in Rats: Relationship with Liver Delta6-Desaturase Expression. Schizophr. Res. 2011, 129, 57–65. [Google Scholar] [CrossRef]
- Yao, J.K.; van Kammen, D.P.; Gurklis, J.A. Abnormal Incorporation of Arachidonic Acid into Platelets of Drug-Free Patients with Schizophrenia. Psychiatry Res. 1996, 60, 11–21. [Google Scholar] [CrossRef]
- Hou, T.Y.; McMurray, D.N.; Chapkin, R.S. Omega-3 Fatty Acids, Lipid Rafts, and T Cell Signaling. Eur. J. Pharmacol. 2016, 785, 2–9. [Google Scholar] [CrossRef]
- Georgieva, R.; Mircheva, K.; Vitkova, V.; Balashev, K.; Ivanova, T.; Tessier, C.; Koumanov, K.; Nuss, P.; Momchilova, A.; Staneva, G. Phospholipase A 2 -Induced Remodeling Processes on Liquid-Ordered/Liquid-Disordered Membranes Containing Docosahexaenoic or Oleic Acid: A Comparison Study. Langmuir 2016, 32, 1756–1770. [Google Scholar] [CrossRef]
- Rogero, M.M.; Leão, M.D.C.; Santana, T.M.; Pimentel, M.V.D.M.; Carlini, G.C.; da Silveira, T.F.; Gonçalves, R.C.; Castro, I.A. Potential Benefits and Risks of Omega-3 Fatty Acids Supplementation to Patients with COVID-19. Available online: https://pubmed.ncbi.nlm.nih.gov/32653511/ (accessed on 17 July 2020).
- Dennis, E.A.; Cao, J.; Hsu, Y.-H.; Magrioti, V.; Kokotos, G. Phospholipase A2 Enzymes: Physical Structure, Biological Function, Disease Implication, Chemical Inhibition, and Therapeutic Intervention. Chem Rev. 2011, 111, 6130–6185. [Google Scholar] [CrossRef] [PubMed]
- Mouchlis, V.D.; Dennis, E.A. Membrane and Inhibitor Interactions of Intracellular Phospholipases A2. Adv. Biol. Regul. 2016, 61, 17–24. [Google Scholar] [CrossRef]
- Smesny, S.; Milleit, B.; Nenadic, I.; Preul, C.; Kinder, D.; Lasch, J.; Willhardt, I.; Sauer, H.; Gaser, C. Phospholipase A2 Activity Is Associated with Structural Brain Changes in Schizophrenia. Neuroimage 2010, 52, 1314–1327. [Google Scholar] [CrossRef]
- Gattaz, W.F.; Köllisch, M.; Thuren, T.; Virtanen, J.A.; Kinnunen, P.K.J. Increased Plasma Phospholipase-A2 Activity in Schizophrenic Patients: Reduction after Neuroleptic Therapy. Biol. Psychiatry 1987, 22, 421–426. [Google Scholar] [CrossRef]
- Smesny, S.; Milleit, B.; Hipler, U.-C.; Milleit, C.; Schäfer, M.R.; Klier, C.M.; Holub, M.; Holzer, I.; Berger, G.E.; Otto, M.; et al. Omega-3 Fatty Acid Supplementation Changes Intracellular Phospholipase A2 Activity and Membrane Fatty Acid Profiles in Individuals at Ultra-High Risk for Psychosis. Mol. Psychiatry 2014, 19, 317–324. [Google Scholar] [CrossRef]
- van Rensburg, S.J.; Smuts, C.M.; Hon, D.; Kidd, M.; van der Merwe, S.; Myburgh, C.; Oosthuizen, P.; Emsley, R. Changes in Erythrocyte Membrane Fatty Acids during a Clinical Trial of Eicosapentaenoic Acid (EPA) Supplementation in Schizophrenia. Metab. Brain Dis. 2009, 24, 659–672. [Google Scholar] [CrossRef] [PubMed]
- Alqarni, A.; Mitchell, T.W.; McGorry, P.D.; Nelson, B.; Markulev, C.; Yuen, H.P.; Schäfer, M.R.; Berger, M.; Mossaheb, N.; Schlögelhofer, M.; et al. Supplementation with the Omega-3 Long Chain Polyunsaturated Fatty Acids: Changes in the Concentrations of Omega-3 Index, Fatty Acids and Molecular Phospholipids of People at Ultra High Risk of Developing Psychosis. Schizophr. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Dainese, E.; Sabatucci, A.; Angelucci, C.B.; Barsacchi, D.; Chiarini, M.; Maccarrone, M. Impact of Embedded Endocannabinoids and Their Oxygenation by Lipoxygenase on Membrane Properties. ACS Chem. Neurosci. 2012, 3, 386–392. [Google Scholar] [CrossRef] [PubMed]
- Gallelli, C.A.; Calcagnini, S.; Romano, A.; Koczwara, J.B.; de Ceglia, M.; Dante, D.; Villani, R.; Giudetti, A.M.; Cassano, T.; Gaetani, S. Modulation of the Oxidative Stress and Lipid Peroxidation by Endocannabinoids and Their Lipid Analogues. Antioxidants 2018, 7, 93. [Google Scholar] [CrossRef]
- Heilman, P.; Hill, M.N.; Coussons-Read, M.; Brundin, L.; Coccaro, E.F. Role of the Kynurenine Pathway and the Endocannabinoid System as Modulators of Inflammation and Personality Traits. Psychoneuroendocrinology 2019, 110, 104434. [Google Scholar] [CrossRef] [PubMed]
- Minichino, A.; Senior, M.; Brondino, N.; Zhang, S.H.; Godwlewska, B.R.; Burnet, P.W.J.; Cipriani, A.; Lennox, B.R. Measuring Disturbance of the Endocannabinoid System in Psychosis: A Systematic Review and Meta-Analysis. JAMA Psychiatry 2019. [Google Scholar] [CrossRef] [PubMed]
- Leweke, F.M.; Giuffrida, A.; Wurster, U.; Emrich, H.M.; Piomelli, D. Elevated Endogenous Cannabinoids in Schizophrenia. Neuroreport 1999, 10, 1665–1669. [Google Scholar] [CrossRef] [PubMed]
- Leweke, F.M.; Giuffrida, A.; Koethe, D.; Schreiber, D.; Nolden, B.M.; Kranaster, L.; Neatby, M.A.; Schneider, M.; Gerth, C.W.; Hellmich, M.; et al. Anandamide Levels in Cerebrospinal Fluid of First-Episode Schizophrenic Patients: Impact of Cannabis Use. Schizophr. Res. 2007, 94, 29–36. [Google Scholar] [CrossRef]
- Muguruza, C.; Lehtonen, M.; Aaltonen, N.; Morentin, B.; Meana, J.J.; Callado, L.F. Quantification of Endocannabinoids in Postmortem Brain of Schizophrenic Subjects. Schizophr. Res. 2013, 148, 145–150. [Google Scholar] [CrossRef]
- Rodríguez-Muñoz, M.; Sánchez-Blázquez, P.; Merlos, M.; Garzón-Niño, J. Endocannabinoid Control of Glutamate NMDA Receptors: The Therapeutic Potential and Consequences of Dysfunction. Oncotarget 2016, 7, 55840–55862. [Google Scholar] [CrossRef]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and Anti-Inflammatory Properties of Cannabidiol. Antioxidants 2019, 9, 21. [Google Scholar] [CrossRef]
- Booz, G.W. Cannabidiol as an Emergent Therapeutic Strategy for Lessening the Impact of Inflammation on Oxidative Stress. Free Radic. Biol. Med. 2011, 51, 1054–1061. [Google Scholar] [CrossRef]
- Kathmann, M.; Flau, K.; Redmer, A.; Tränkle, C.; Schlicker, E. Cannabidiol Is an Allosteric Modulator at Mu- and Delta-Opioid Receptors. Naunyn Schmiedebergs Arch Pharmacol. 2006, 372, 354–361. [Google Scholar] [CrossRef]
- Martínez-Aguirre, C.; Carmona-Cruz, F.; Velasco, A.L.; Velasco, F.; Aguado-Carrillo, G.; Cuéllar-Herrera, M.; Rocha, L. Cannabidiol Acts at 5-HT1A Receptors in the Human Brain: Relevance for Treating Temporal Lobe Epilepsy. Front. Behav. Neurosci. 2020, 14, 611278. [Google Scholar] [CrossRef] [PubMed]
- Pertwee, R.G.; Ross, R.A.; Craib, S.J.; Thomas, A. (-)-Cannabidiol Antagonizes Cannabinoid Receptor Agonists and Noradrenaline in the Mouse Vas Deferens. Eur. J. Pharmacol. 2002, 456, 99–106. [Google Scholar] [CrossRef]
- Pertwee, R.G. The Diverse CB1 and CB2 Receptor Pharmacology of Three Plant Cannabinoids: Delta9-Tetrahydrocannabinol, Cannabidiol and Delta9-Tetrahydrocannabivarin. Br. J. Pharmacol. 2008, 153, 199–215. [Google Scholar] [CrossRef]
- Tham, M.; Yilmaz, O.; Alaverdashvili, M.; Kelly, M.E.M.; Denovan-Wright, E.M.; Laprairie, R.B. Allosteric and Orthosteric Pharmacology of Cannabidiol and Cannabidiol-Dimethylheptyl at the Type 1 and Type 2 Cannabinoid Receptors. Br. J. Pharmacol. 2019, 176, 1455–1469. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Pinilla, E.; Varani, K.; Reyes-Resina, I.; Angelats, E.; Vincenzi, F.; Ferreiro-Vera, C.; Oyarzabal, J.; Canela, E.I.; Lanciego, J.L.; Nadal, X.; et al. Binding and Signaling Studies Disclose a Potential Allosteric Site for Cannabidiol in Cannabinoid CB2 Receptors. Front. Pharmacol. 2017, 8. [Google Scholar] [CrossRef]
- Laprairie, R.B.; Bagher, A.M.; Kelly, M.E.M.; Denovan-Wright, E.M. Cannabidiol Is a Negative Allosteric Modulator of the Cannabinoid CB1 Receptor. Br. J. Pharmacol. 2015, 172, 4790–4805. [Google Scholar] [CrossRef]
- Kopelli, E.; Samara, M.; Siargkas, A.; Goulas, A.; Papazisis, G.; Chourdakis, M. The Role of Cannabidiol Oil in Schizophrenia Treatment. a Systematic Review and Meta-Analysis. Psychiatry Res. 2020, 291, 113246. [Google Scholar] [CrossRef]
- Jung, T.; Hudson, R.; Rushlow, W.; Laviolette, S.R. Functional Interactions between Cannabinoids, Omega-3 Fatty Acids and Peroxisome Proliferator-Activated Receptors: Implications for Mental Health Pharmacotherapies. Eur. J. Neurosci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Lafourcade, M.; Larrieu, T.; Mato, S.; Duffaud, A.; Sepers, M.; Matias, I.; De Smedt-Peyrusse, V.; Labrousse, V.F.; Bretillon, L.; Matute, C.; et al. Nutritional Omega-3 Deficiency Abolishes Endocannabinoid-Mediated Neuronal Functions. Nat. Neurosci. 2011, 14, 345–350. [Google Scholar] [CrossRef]
- Larrieu, T.; Madore, C.; Joffre, C.; Layé, S. Nutritional N-3 Polyunsaturated Fatty Acids Deficiency Alters Cannabinoid Receptor Signaling Pathway in the Brain and Associated Anxiety-like Behavior in Mice. J. Physiol. Biochem. 2012, 68, 671–681. [Google Scholar] [CrossRef]
- Watanabe, S.; Doshi, M.; Hamazaki, T. N-3 Polyunsaturated Fatty Acid (PUFA) Deficiency Elevates and n-3 PUFA Enrichment Reduces Brain 2-Arachidonoylglycerol Level in Mice. Prostaglandins Leukot. Essent. Fatty Acids 2003, 69, 51–59. [Google Scholar] [CrossRef]
- Healy-Stoffel, M.; Levant, B. N-3 (Omega-3) Fatty Acids: Effects on Brain Dopamine Systems and Potential Role in the Etiology and Treatment of Neuropsychiatric Disorders. CNS Neurol. Disord. Drug Targets 2018, 17, 216–232. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Snyder, G.L.; Vanover, K.E. Dopamine Targeting Drugs for the Treatment of Schizophrenia: Past, Present and Future. Curr. Top. Med. Chem. 2016, 16, 3385–3403. [Google Scholar] [CrossRef]
- Ohara, K. The N-3 Polyunsaturated Fatty Acid/Dopamine Hypothesis of Schizophrenia. Prog. Neuropsychopharmacol. Biol. Psychiatry 2007, 31, 469–474. [Google Scholar] [CrossRef]
- Hibbeln, J.R.; Linnoila, M.; Umhau, J.C.; Rawlings, R.; George, D.T.; Salem, N. Essential Fatty Acids Predict Metabolites of Serotonin and Dopamine in Cerebrospinal Fluid among Healthy Control Subjects, and Early- and Late-Onset Alcoholics. Biol Psychiatry 1998, 44, 235–242. [Google Scholar] [CrossRef]
- Sublette, M.E.; Galfalvy, H.C.; Hibbeln, J.R.; Keilp, J.G.; Malone, K.M.; Oquendo, M.A.; Mann, J.J. Polyunsaturated Fatty Acid Associations with Dopaminergic Indices in Major Depressive Disorder. Int. J. Neuropsychopharmacol. 2014, 17, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Bondi, C.O.; Taha, A.Y.; Tock, J.L.; Totah, N.K.; Cheon, Y.; Torres, G.E.; Rapoport, S.I.; Moghaddam, B. Adolescent Behavior and Dopamine Availability are Uniquely Sensitive to Dietary Omega-3 Fatty Acid Deficiency. Biol. Psychiatry 2014, 75. [Google Scholar] [CrossRef] [PubMed]
- Chung, W.-L.; Chen, J.-J.; Su, H.-M. Fish Oil Supplementation of Control and (n-3) Fatty Acid-Deficient Male Rats Enhances Reference and Working Memory Performance and Increases Brain Regional Docosahexaenoic Acid Levels. J. Nutr. 2008, 138, 1165–1171. [Google Scholar] [CrossRef]
- Kuperstein, F.; Eilam, R.; Yavin, E. Altered Expression of Key Dopaminergic Regulatory Proteins in the Postnatal Brain Following Perinatal N-3 Fatty Acid Dietary Deficiency. J. Neurochem. 2008, 106, 662–671. [Google Scholar] [CrossRef]
- Zimmer, L.; Hembert, S.; Durand, G.; Breton, P.; Guilloteau, D.; Besnard, J.-C.; Chalon, S. Chronic N-3 Polyunsaturated Fatty Acid Diet-Deficiency Acts on Dopamine Metabolism in the Rat Frontal Cortex: A Microdialysis Study. Neurosci. Lett. 1998, 240, 177–181. [Google Scholar] [CrossRef]
- McNamara, R.K.; Able, J.; Liu, Y.; Jandacek, R.; Rider, T.; Tso, P.; Lipton, J.W. Omega-3 Fatty Acid Deficiency during Perinatal Development Increases Serotonin Turnover in the Prefrontal Cortex and Decreases Midbrain Tryptophan Hydroxylase-2 Expression in Adult Female Rats: Dissociation from Estrogenic Effects. J. Psychiatr. Res. 2009, 43, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, L.; Vancassel, S.; Cantagrel, S.; Breton, P.; Delamanche, S.; Guilloteau, D.; Durand, G.; Chalon, S. The Dopamine Mesocorticolimbic Pathway Is Affected by Deficiency in N-3 Polyunsaturated Fatty Acids. Am. J. Clin. Nutr. 2002, 75, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Chalon, S.; Delion-Vancassel, S.; Belzung, C.; Guilloteau, D.; Leguisquet, A.M.; Besnard, J.C.; Durand, G. Dietary Fish Oil Affects Monoaminergic Neurotransmission and Behavior in Rats. J. Nutr. 1998, 128, 2512–2519. [Google Scholar] [CrossRef]
- Metz, V.G.; Segat, H.J.; Dias, V.T.; Barcelos, R.C.S.; Maurer, L.H.; Stiebe, J.; Emanuelli, T.; Burger, M.E.; Pase, C.S. Omega-3 Decreases D1 and D2 Receptors Expression in the Prefrontal Cortex and Prevents Amphetamine-Induced Conditioned Place Preference in Rats. J. Nutr. Biochem. 2019, 67, 182–189. [Google Scholar] [CrossRef]
- Naneix, F.; Tantot, F.; Glangetas, C.; Kaufling, J.; Janthakhin, Y.; Boitard, C.; De Smedt-Peyrusse, V.; Pape, J.R.; Vancassel, S.; Trifilieff, P.; et al. Impact of Early Consumption of High-Fat Diet on the Mesolimbic Dopaminergic System. eNeuro 2017, 4. [Google Scholar] [CrossRef] [PubMed]
- Chitre, N.M.; Wood, B.J.; Ray, A.; Moniri, N.H.; Murnane, K.S. Docosahexaenoic Acid Protects Motor Function and Increases Dopamine Synthesis in a Rat Model of Parkinson’s Disease via Mechanisms Associated with Increased Protein Kinase Activity in the Striatum. Neuropharmacology 2020, 167, 107976. [Google Scholar] [CrossRef] [PubMed]
- Martel, J.C.; Gatti McArthur, S. Dopamine Receptor Subtypes, Physiology and Pharmacology: New Ligands and Concepts in Schizophrenia. Front. Pharmacol. 2020, 11, 1003. [Google Scholar] [CrossRef] [PubMed]
- Sibarov, D.A.; Poguzhelskaya, E.E.; Antonov, S.M. Downregulation of Calcium-Dependent NMDA Receptor Desensitization by Sodium-Calcium Exchangers: A Role of Membrane Cholesterol. BMC Neurosci. 2018, 19, 73. [Google Scholar] [CrossRef]
- Schrattenholz, A.; Soskic, V. NMDA Receptors Are Not Alone: Dynamic Regulation of NMDA Receptor Structure and Function by Neuregulins and Transient Cholesterol-Rich Membrane Domains Leads to Disease-Specific Nuances of Glutamate-Signalling. Curr. Top. Med. Chem. 2006, 6, 663–686. [Google Scholar] [CrossRef]
- Isu, U.; Tabari, S.H.; Kumar, V.G.; Moradi, M. Effect of Cholesterol on the Structural Dynamics of Metabotropic Glutamate Receptor (MGluR1): A Molecular Dynamics Study. Biophys. J. 2020, 118, 525a. [Google Scholar] [CrossRef]
- Sodero, A.O.; Vriens, J.; Ghosh, D.; Stegner, D.; Brachet, A.; Pallotto, M.; Sassoè-Pognetto, M.; Brouwers, J.F.; Helms, J.B.; Nieswandt, B.; et al. Cholesterol Loss during Glutamate-Mediated Excitotoxicity. EMBO J. 2012, 31, 1764–1773. [Google Scholar] [CrossRef]
- Egerton, A.; Grace, A.A.; Stone, J.; Bossong, M.G.; Sand, M.; McGuire, P. Glutamate in Schizophrenia: Neurodevelopmental Perspectives and Drug Development. Schizophr. Res. 2020. [Google Scholar] [CrossRef]
- Scoriels, L.; Salek, R.M.; Goodby, E.; Grainger, D.; Dean, A.M.; West, J.A.; Griffin, J.L.; Suckling, J.; Nathan, P.J.; Lennox, B.R.; et al. Behavioural and Molecular Endophenotypes in Psychotic Disorders Reveal Heritable Abnormalities in Glutamatergic Neurotransmission. Transl. Psychiatry 2015, 5, e540. [Google Scholar] [CrossRef]
- Egerton, A.; Bhachu, A.; Merritt, K.; McQueen, G.; Szulc, A.; McGuire, P. Effects of Antipsychotic Administration on Brain Glutamate in Schizophrenia: A Systematic Review of Longitudinal 1H-MRS Studies. Front. Psychiatry 2017, 8, 66. [Google Scholar] [CrossRef]
- Miller, B.; Sarantis, M.; Traynelis, S.F.; Attwell, D. Potentiation of NMDA Receptor Currents by Arachidonic Acid. Nature 1992, 355, 722–725. [Google Scholar] [CrossRef]
- Moreira, J.D.; Knorr, L.; Ganzella, M.; Thomazi, A.P.; de Souza, C.G.; de Souza, D.G.; Pitta, C.F.; Mello e Souza, T.; Wofchuk, S.; Elisabetsky, E.; et al. Omega-3 Fatty Acids Deprivation Affects Ontogeny of Glutamatergic Synapses in Rats: Relevance for Behavior Alterations. Neurochem. Int. 2010, 56, 753–759. [Google Scholar] [CrossRef]
- Latour, A.; Grintal, B.; Champeil-Potokar, G.; Hennebelle, M.; Lavialle, M.; Dutar, P.; Potier, B.; Billard, J.-M.; Vancassel, S.; Denis, I. Omega-3 Fatty Acids Deficiency Aggravates Glutamatergic Synapse and Astroglial Aging in the Rat Hippocampal CA1. Aging Cell 2013, 12, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Keleshian, V.L.; Kellom, M.; Kim, H.-W.; Taha, A.Y.; Cheon, Y.; Igarashi, M.; Rapoport, S.I.; Rao, J.S. Neuropathological Responses to Chronic NMDA in Rats Are Worsened by Dietary N-3 PUFA Deprivation but Are Not Ameliorated by Fish Oil Supplementation. PLoS ONE 2014, 9, e95318. [Google Scholar] [CrossRef]
- Bach, S.A.; de Siqueira, L.V.; Müller, A.P.; Oses, J.P.; Quatrim, A.; Emanuelli, T.; Vinadé, L.; Souza, D.O.; Moreira, J.D. Dietary Omega-3 Deficiency Reduces BDNF Content and Activation NMDA Receptor and Fyn in Dorsal Hippocampus: Implications on Persistence of Long-Term Memory in Rats. Nutr. Neurosci. 2014, 17, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Calon, F.; Lim, G.P.; Morihara, T.; Yang, F.; Ubeda, O.; Salem, N.; Frautschy, S.A.; Cole, G.M. Dietary N-3 Polyunsaturated Fatty Acid Depletion Activates Caspases and Decreases NMDA Receptors in the Brain of a Transgenic Mouse Model of Alzheimer’s Disease. Eur. J. Neurosci. 2005, 22, 617–626. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Kevala, K.; Kim, J.; Moon, H.-S.; Jun, S.B.; Lovinger, D.; Kim, H.-Y. Docosahexaenoic Acid Promotes Hippocampal Neuronal Development and Synaptic Function. J. Neurochem. 2009, 111, 510–521. [Google Scholar] [CrossRef] [PubMed]
- Aryal, S.; Hussain, S.; Drevon, C.A.; Nagelhus, E.; Hvalby, Ø.; Jensen, V.; Walaas, S.I.; Davanger, S. Omega-3 Fatty Acids Regulate Plasticity in Distinct Hippocampal Glutamatergic Synapses. Eur. J. Neurosci. 2019, 49, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Cadinu, D.; Grayson, B.; Podda, G.; Harte, M.K.; Doostdar, N.; Neill, J.C. NMDA Receptor Antagonist Rodent Models for Cognition in Schizophrenia and Identification of Novel Drug Treatments, an Update. Neuropharmacology 2018, 142, 41–62. [Google Scholar] [CrossRef]
- Neill, J.C.; Barnes, S.; Cook, S.; Grayson, B.; Idris, N.F.; McLean, S.L.; Snigdha, S.; Rajagopal, L.; Harte, M.K. Animal Models of Cognitive Dysfunction and Negative Symptoms of Schizophrenia: Focus on NMDA Receptor Antagonism. Pharmacol. Ther. 2010, 128, 419–432. [Google Scholar] [CrossRef] [PubMed]
- Islam, R.; Trépanier, M.-O.; Milenkovic, M.; Horsfall, W.; Salahpour, A.; Bazinet, R.P.; Ramsey, A.J. Vulnerability to Omega-3 Deprivation in a Mouse Model of NMDA Receptor Hypofunction. NPJ Schizophr. 2017, 3, 12. [Google Scholar] [CrossRef] [PubMed]
- Manduca, A.; Bara, A.; Larrieu, T.; Lassalle, O.; Joffre, C.; Lay, S.; Manzoni, O.J. Amplification of MGlu 5 -Endocannabinoid Signaling Rescues Behavioral and Synaptic Deficits in a Mouse Model of Adolescent and Adult Dietary Polyunsaturated Fatty Acids Imbalance. J. Neurosci. 2017, 37, 6851–6868. [Google Scholar] [CrossRef]
- Ménard, C.; Patenaude, C.; Gagné, A.-M.; Massicotte, G. AMPA Receptor-Mediated Cell Death Is Reduced by Docosahexaenoic Acid but Not by Eicosapentaenoic Acid in Area CA1 of Hippocampal Slice Cultures. J. Neurosci. Res. 2009, 87, 876–886. [Google Scholar] [CrossRef] [PubMed]
- Grintal, B.; Champeil-Potokar, G.; Lavialle, M.; Vancassel, S.; Breton, S.; Denis, I. Inhibition of Astroglial Glutamate Transport by Polyunsaturated Fatty Acids: Evidence for a Signalling Role of Docosahexaenoic Acid. Neurochem. Int. 2009, 54, 535–543. [Google Scholar] [CrossRef]
- Relton, J.K.; Strijbos, P.J.; Cooper, A.L.; Rothwell, N.J. Dietary N-3 Fatty Acids Inhibit Ischaemic and Excitotoxic Brain Damage in the Rat. Brain Res. Bull. 1993, 32, 223–226. [Google Scholar] [CrossRef]
- Högyes, E.; Nyakas, C.; Kiliaan, A.; Farkas, T.; Penke, B.; Luiten, P.G.M. Neuroprotective Effect of Developmental Docosahexaenoic Acid Supplement against Excitotoxic Brain Damage in Infant Rats. Neuroscience 2003, 119, 999–1012. [Google Scholar] [CrossRef]
- Torres, M.; Price, S.L.; Fiol-Deroque, M.A.; Marcilla-Etxenike, A.; Ahyayauch, H.; Barceló-Coblijn, G.; Terés, S.; Katsouri, L.; Ordinas, M.; López, D.J.; et al. Membrane Lipid Modifications and Therapeutic Effects Mediated by Hydroxydocosahexaenoic Acid on Alzheimer’s Disease. Biochim. Biophys. Acta 2014, 1838, 1680–1692. [Google Scholar] [CrossRef]
- Gama, C.S.; Canever, L.; Panizzutti, B.; Gubert, C.; Stertz, L.; Massuda, R.; Pedrini, M.; de Lucena, D.F.; Luca, R.D.; Fraga, D.B.; et al. Effects of Omega-3 Dietary Supplement in Prevention of Positive, Negative and Cognitive Symptoms: A Study in Adolescent Rats with Ketamine-Induced Model of Schizophrenia. Schizophr. Res. 2012, 141, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Zugno, A.I.; Chipindo, H.L.; Volpato, A.M.; Budni, J.; Steckert, A.V.; de Oliveira, M.B.; Heylmann, A.S.; da Silveira, F.R.; Mastella, G.A.; Maravai, S.G.; et al. Omega-3 Prevents Behavior Response and Brain Oxidative Damage in the Ketamine Model of Schizophrenia. Neuroscience 2014, 259, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Du, F.; Cooper, A.J.; Thida, T.; Shinn, A.K.; Cohen, B.M.; Öngür, D. Myelin and Axon Abnormalities in Schizophrenia Measured with Magnetic Resonance Imaging Techniques. Biol. Psychiatry 2013, 74, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Wood, P.L.; Holderman, N.R. Dysfunctional Glycosynapses in Schizophrenia: Disease and Regional Specificity. Schizophr. Res. 2015. [Google Scholar] [CrossRef] [PubMed]
- Peters, B.D.; Machielsen, M.W.J.; Hoen, W.P.; Caan, M.W.A.; Malhotra, A.K.; Szeszko, P.R.; Duran, M.; Olabarriaga, S.D.; de Haan, L. Polyunsaturated Fatty Acid Concentration Predicts Myelin Integrity in Early-Phase Psychosis. Schizophr. Bull. 2013, 39, 830–838. [Google Scholar] [CrossRef] [PubMed]
- Castillo, R.I.; Rojo, L.E.; Henriquez-Henriquez, M.; Silva, H.; Maturana, A.; Villar, M.J.; Fuentes, M.; Gaspar, P.A. From Molecules to the Clinic: Linking Schizophrenia and Metabolic Syndrome through Sphingolipids Metabolism. Front. Neurosci. 2016, 10. [Google Scholar] [CrossRef]
- Gouvêa-Junqueira, D.; Falvella, A.C.B.; Antunes, A.S.L.M.; Seabra, G.; Brandão-Teles, C.; Martins-de-Souza, D.; Crunfli, F. Novel Treatment Strategies Targeting Myelin and Oligodendrocyte Dysfunction in Schizophrenia. Front. Psychiatry 2020, 11, 379. [Google Scholar] [CrossRef]
- Maas, D.A.; Eijsink, V.D.; Spoelder, M.; Van Hulten, J.A.; De Weerd, P.; Homberg, J.R.; Vallès, A.; Nait-Oumesmar, B.; Martens, G.J.M. Interneuron Hypomyelination Is Associated with Cognitive Inflexibility in a Rat Model of Schizophrenia. Nat. Commun. 2020, 11, 2329. [Google Scholar] [CrossRef]
- Makinodan, M.; Yamauchi, T.; Tatsumi, K.; Okuda, H.; Takeda, T.; Kiuchi, K.; Sadamatsu, M.; Wanaka, A.; Kishimoto, T. Demyelination in the Juvenile Period, but Not in Adulthood, Leads to Long-Lasting Cognitive Impairment and Deficient Social Interaction in Mice. Prog. Neuropsychopharmacol. Biol. Psychiatry 2009, 33, 978–985. [Google Scholar] [CrossRef]
- Zhou, C.; Xue, S.; Xue, F.; Liu, L.; Liu, J.; Ma, Q.; Qin, J.; Tan, Q.; Wang, H.; Peng, Z. The Impact of Quetiapine on the Brain Lipidome in a Cuprizone-Induced Mouse Model of Schizophrenia. Biomed. Pharmacother. 2020, 131, 110707. [Google Scholar] [CrossRef]
- Narayan, S.; Head, S.R.; Gilmartin, T.J.; Dean, B.; Thomas, E.A. Evidence for Disruption of Sphingolipid Metabolism in Schizophrenia. J. Neurosci. Res. 2009, 87, 278–288. [Google Scholar] [CrossRef]
- Bartzokis, G.; Lu, P.H.; Stewart, S.B.; Oluwadara, B.; Lucas, A.J.; Pantages, J.; Pratt, E.; Sherin, J.E.; Altshuler, L.L.; Mintz, J.; et al. In Vivo Evidence of Differential Impact of Typical and Atypical Antipsychotics on Intracortical Myelin in Adults with Schizophrenia. Schizophr. Res. 2009, 113, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Bartzokis, G.; Lu, P.H.; Nuechterlein, K.H.; Gitlin, M.; Doi, C.; Edwards, N.; Lieu, C.; Altshuler, L.L.; Mintz, J. Differential Effects of Typical and Atypical Antipsychotics on Brain Myelination in Schizophrenia. Schizoph. Res. 2007, 93, 13–22. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Saher, G.; Brügger, B.; Lappe-Siefke, C.; Möbius, W.; Tozawa, R.; Wehr, M.C.; Wieland, F.; Ishibashi, S.; Nave, K.-A. High Cholesterol Level Is Essential for Myelin Membrane Growth. Nat. Neurosci. 2005, 8, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Pajevic, S.; Basser, P.J.; Fields, R.D. Role of Myelin Plasticity in Oscillations and Synchrony of Neuronal Activity. Neuroscience 2014, 276, 135–147. [Google Scholar] [CrossRef]
- Di Biase, A.; Salvati, S.; Di Benedetto, R.; Attorri, L.; Martinelli, A.; Malchiodi, F. Eicosapentaenoic Acid Pre-Treatment Reduces Biochemical Changes Induced in Total Brain and Myelin of Weanling Wistar Rats by Cuprizone Feeding. Prostaglandins Leukot. Essent. Fatty Acids 2014, 90, 99–104. [Google Scholar] [CrossRef]
- Farooqui, A.A. Lipid Mediators in the Neural Cell Nucleus: Their Metabolism, Signaling, and Association with Neurological Disorders. Neuroscientist 2009. [Google Scholar] [CrossRef]
- Calder, P.C. Eicosapentaenoic and Docosahexaenoic Acid Derived Specialised Pro-Resolving Mediators: Concentrations in Humans and the Effects of Age, Sex, Disease and Increased Omega-3 Fatty Acid Intake. Biochimie 2020. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Marine Omega-3 Fatty Acids and Inflammatory Processes: Effects, Mechanisms and Clinical Relevance. Biochim. Biophys. Acta 2015, 1851, 469–484. [Google Scholar] [CrossRef]
- Zuo, L.; Prather, E.R.; Stetskiv, M.; Garrison, D.E.; Meade, J.R.; Peace, T.I.; Zhou, T. Inflammaging and Oxidative Stress in Human Diseases: From Molecular Mechanisms to Novel Treatments. Int. J. Mol. Sci. 2019, 20, 4472. [Google Scholar] [CrossRef]
- Dickerson, F.; Stallings, C.; Origoni, A.; Vaughan, C.; Khushalani, S.; Yang, S.; Yolken, R. C-Reactive Protein Is Elevated in Schizophrenia. Schizophr. Res. 2013, 143, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Flatow, J.; Buckley, P.; Miller, B.J. Meta-Analysis of Oxidative Stress in Schizophrenia. Biol. Psychiatry 2013, 74, 400–409. [Google Scholar] [CrossRef]
- Fraguas, D.; Díaz-Caneja, C.M.; Rodríguez-Quiroga, A.; Arango, C. Oxidative Stress and Inflammation in Early Onset First Episode Psychosis: A Systematic Review and Meta-Analysis. Int. J. Neuropsychopharmacol. 2017, 20, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Li, Q.; Luo, X.; Tian, L.; Wang, Z.; Tan, S.; Chen, S.; Yang, G.; An, H.; Yang, F.; et al. Plasma Total Antioxidant Status and Cognitive Impairments in First-Episode Drug-Naïve Patients with Schizophrenia. Cogn. Neurodyn. 2019, 13, 357–365. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Chen, D.C.; Xiu, M.H.; Tang, W.; Zhang, F.; Liu, L.; Chen, Y.; Liu, J.; Yao, J.K.; Kosten, T.A.; et al. Plasma Total Antioxidant Status and Cognitive Impairments in Schizophrenia. Schizophr. Res. 2012, 139, 66–72. [Google Scholar] [CrossRef]
- Prestwood, T.R.; Asgariroozbehani, R.; Wu, S.; Agarwal, S.M.; Logan, R.W.; Ballon, J.S.; Hahn, M.K.; Freyberg, Z. Roles of Inflammation in Intrinsic Pathophysiology and Antipsychotic Drug-Induced Metabolic Disturbances of Schizophrenia. Behav. Brain Res. 2021, 402, 113101. [Google Scholar] [CrossRef] [PubMed]
- Caruso, G.; Grasso, M.; Fidilio, A.; Tascedda, F.; Drago, F.; Caraci, F. Antioxidant Properties of Second-Generation Antipsychotics: Focus on Microglia. Pharmaceuticals 2020, 13, 457. [Google Scholar] [CrossRef]
- Delpech, J.-C.; Madore, C.; Joffre, C.; Aubert, A.; Kang, J.X.; Nadjar, A.; Layé, S. Transgenic Increase in N-3/n-6 Fatty Acid Ratio Protects against Cognitive Deficits Induced by an Immune Challenge through Decrease of Neuroinflammation. Neuropsychopharmacology 2015, 40, 525–536. [Google Scholar] [CrossRef]
- Delpech, J.-C.; Thomazeau, A.; Madore, C.; Bosch-Bouju, C.; Larrieu, T.; Lacabanne, C.; Remus-Borel, J.; Aubert, A.; Joffre, C.; Nadjar, A.; et al. Dietary N-3 PUFAs Deficiency Increases Vulnerability to Inflammation-Induced Spatial Memory Impairment. Neuropsychopharmacology 2015, 40, 2774–2787. [Google Scholar] [CrossRef] [PubMed]
- Mingam, R.; Moranis, A.; Bluthé, R.-M.; De Smedt-Peyrusse, V.; Kelley, K.W.; Guesnet, P.; Lavialle, M.; Dantzer, R.; Layé, S. Uncoupling of Interleukin-6 from Its Signalling Pathway by Dietary n-3-Polyunsaturated Fatty Acid Deprivation Alters Sickness Behaviour in Mice. Eur. J. Neurosci. 2008, 28, 1877–1886. [Google Scholar] [CrossRef]
- Shi, J.; Wang, W.; Sang, G.; Xi, H.; Sun, Y.; Lu, C.; Ye, H.; Huang, L. Short Term Usage of Omega-3 Polyunsaturated Fatty Acids Ameliorate Lipopolysaccharide-Induced Inflammatory Response and Oxidative Stress in the Neonatal Rat Hippocampal Tissue. Front. Nutr. 2020, 7, 572363. [Google Scholar] [CrossRef]
- Zugno, A.I.; Chipindo, H.; Canever, L.; Budni, J.; Alves de Castro, A.; Bittencourt de Oliveira, M.; Heylmann, A.S.; Gomes Wessler, P.; da Rosa Silveira, F.; Damázio, L.S.; et al. Omega-3 Fatty Acids Prevent the Ketamine-Induced Increase in Acetylcholinesterase Activity in an Animal Model of Schizophrenia. Life Sci. 2015, 121, 65–69. [Google Scholar] [CrossRef]
- Buonocore, D.; Verri, M.; Giolitto, A.; Doria, E.; Ghitti, M.; Dossena, M. Effect of 8-Week n-3 Fatty-Acid Supplementation on Oxidative Stress and Inflammation in Middle- and Long-Distance Running Athletes: A Pilot Study. J. Int. Soc. Sports Nutr. 2020, 17, 55. [Google Scholar] [CrossRef] [PubMed]
- Heshmati, J.; Morvaridzadeh, M.; Maroufizadeh, S.; Akbari, A.; Yavari, M.; Amirinejhad, A.; Maleki-Hajiagha, A.; Sepidarkish, M. Omega-3 Fatty Acids Supplementation and Oxidative Stress Parameters: A Systematic Review and Meta-Analysis of Clinical Trials. Pharmacol. Res. 2019, 104462. [Google Scholar] [CrossRef]
- Sepidarkish, M.; Akbari-Fakhrabadi, M.; Daneshzad, E.; Yavari, M.; Rezaeinejad, M.; Morvaridzadeh, M.; Heshmati, J. Effect of Omega-3 Fatty Acid plus Vitamin E Co-Supplementation on Oxidative Stress Parameters: A Systematic Review and Meta-Analysis. Clin. Nutr. 2020, 39, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Koga, N.; Ogura, J.; Yoshida, F.; Hattori, K.; Hori, H.; Aizawa, E.; Ishida, I.; Kunugi, H. Altered Polyunsaturated Fatty Acid Levels in Relation to Proinflammatory Cytokines, Fatty Acid Desaturase Genotype, and Diet in Bipolar Disorder. Transl. Psychiatry 2019, 9, 208. [Google Scholar] [CrossRef]
- Rapaport, M.H.; Nierenberg, A.A.; Schettler, P.J.; Kinkead, B.; Cardoos, A.; Walker, R.; Mischoulon, D. Inflammation as a Predictive Biomarker for Response to Omega-3 Fatty Acids in Major Depressive Disorder: A Proof-of-Concept Study. Mol. Psychiatry 2016, 21, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Fond, G.; Lançon, C.; Korchia, T.; Auquier, P.; Boyer, L. The Role of Inflammation in the Treatment of Schizophrenia. Front. Psychiatry 2020, 11, 160. [Google Scholar] [CrossRef]
- Cho, M.; Lee, T.Y.; Kwak, Y.B.; Yoon, Y.B.; Kim, M.; Kwon, J.S. Adjunctive Use of Anti-Inflammatory Drugs for Schizophrenia: A Meta-Analytic Investigation of Randomized Controlled Trials. Aust. N. Z. J. Psychiatry 2019, 53, 742–759. [Google Scholar] [CrossRef]
- Pawe?czyk, T.; Grancow-Grabka, M.; Trafalska, E.; Szemraj, J.; Pawe?czyk, A. Oxidative Stress Reduction Related to the Efficacy of N-3 Polyunsaturated Fatty Acids in First Episode Schizophrenia: Secondary Outcome Analysis of the OFFER Randomized Trial. Prostaglandins Leukot. Essent. Fatty Acids 2017, 121, 7–13. [Google Scholar] [CrossRef]
- Smesny, S.; Milleit, B.; Schaefer, M.R.; Hesse, J.; Schlögelhofer, M.; Langbein, K.; Hipler, U.-C.; Berger, M.; Cotter, D.R.; Sauer, H.; et al. Effects of Omega-3 PUFA on Immune Markers in Adolescent Individuals at Ultra-High Risk for Psychosis—Results of the Randomized Controlled Vienna Omega-3 Study. Schizophr. Res. 2017. [Google Scholar] [CrossRef]
- Smesny, S.; Milleit, B.; Schaefer, M.R.; Hipler, U.-C.; Milleit, C.; Wiegand, C.; Hesse, J.; Klier, C.M.; Holub, M.; Holzer, I.; et al. Effects of Omega-3 PUFA on the Vitamin E and Glutathione Antioxidant Defense System in Individuals at Ultra-High Risk of Psychosis. Prostaglandins Leukot. Essent. Fatty Acids 2015, 101, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Kurniawan, H.; Kobayashi, T.; Brenner, D. The Emerging Role of One-Carbon Metabolism in T Cells. Curr. Opin. Biotechnol. 2021, 68, 193–201. [Google Scholar] [CrossRef]
- Coppedè, F. One-Carbon Epigenetics and Redox Biology of Neurodegeneration. Free Radic. Biol. Med. 2020. [Google Scholar] [CrossRef]
- Bagnyukova, T.V.; Powell, C.L.; Pavliv, O.; Tryndyak, V.P.; Pogribny, I.P. Induction of Oxidative Stress and DNA Damage in Rat Brain by a Folate/Methyl-Deficient Diet. Brain Res. 2008, 1237, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Peifer, J.J.; Lewis, R.D. Effects of Vitamin B-12 Deprivation on Phospholipid Fatty Acid Patterns in Liver and Brain of Rats Fed High and Low Levels of Linoleate in Low Methionine Diets. J. Nutr. 1979, 109, 2160–2172. [Google Scholar] [CrossRef]
- Nguyen, L.N.; Ma, D.; Shui, G.; Wong, P.; Cazenave-Gassiot, A.; Zhang, X.; Wenk, M.R.; Goh, E.L.K.; Silver, D.L. Mfsd2a Is a Transporter for the Essential Omega-3 Fatty Acid Docosahexaenoic Acid. Nature 2014, 509, 503–506. [Google Scholar] [CrossRef]
- Miller, R.R.; Leanza, C.M.; Phillips, E.E.; Blacquire, K.D. Homocysteine-Induced Changes in Brain Membrane Composition Correlate with Increased Brain Caspase-3 Activities and Reduced Chick Embryo Viability. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2003, 136, 521–532. [Google Scholar] [CrossRef]
- Li, D.; Mann, N.J.; Sinclair, A.J. A Significant Inverse Relationship between Concentrations of Plasma Homocysteine and Phospholipid Docosahexaenoic Acid in Healthy Male Subjects. Lipids 2006, 41, 85–89. [Google Scholar] [CrossRef]
- Kale, A.; Naphade, N.; Sapkale, S.; Kamaraju, M.; Pillai, A.; Joshi, S.; Mahadik, S. Reduced Folic Acid, Vitamin B12 and Docosahexaenoic Acid and Increased Homocysteine and Cortisol in Never-Medicated Schizophrenia Patients: Implications for Altered One-Carbon Metabolism. Psychiatry Res. 2010, 175, 47–53. [Google Scholar] [CrossRef]
- Fan, N.; Tan, Y.; Yang, F.; Tian, L.; Chen, S.; Li, J.; Wang, Z.; Zhang, X. Effect of Risperidone on Serum Homocysteine Levels in First-Episode, Drug-Naïve Patients with Schizophrenia. Neurosci. Lett. 2017, 650, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Assies, J.; Mocking, R.J.T.; Lok, A.; Ruhé, H.G.; Pouwer, F.; Schene, A.H. Effects of Oxidative Stress on Fatty Acid- and One-Carbon-Metabolism in Psychiatric and Cardiovascular Disease Comorbidity. Acta Psychiatr. Scand. 2014, 130, 163–180. [Google Scholar] [CrossRef]
- Réus, G.Z.; Maciel, A.L.; Abelaira, H.M.; de Moura, A.B.; de Souza, T.G.; Dos Santos, T.R.; Darabas, A.C.; Parzianello, M.; Matos, D.; Abatti, M.; et al. ω-3 and Folic Acid Act against Depressive-like Behavior and Oxidative Damage in the Brain of Rats Subjected to Early- or Late-Life Stress. Nutrition 2018, 53, 120–133. [Google Scholar] [CrossRef]
- Roy, S.; Kale, A.; Dangat, K.; Sable, P.; Kulkarni, A.; Joshi, S. Maternal Micronutrients (Folic Acid and Vitamin B12) and Omega 3 Fatty Acids: Implications for Neurodevelopmental Risk in the Rat Offspring. Brain Dev. 2012, 34, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Dawson, S.L.; Bowe, S.J.; Crowe, T.C. A Combination of Omega-3 Fatty Acids, Folic Acid and B-Group Vitamins Is Superior at Lowering Homocysteine than Omega-3 Alone: A Meta-Analysis. Nutr. Res. 2016, 36, 499–508. [Google Scholar] [CrossRef]
- Oulhaj, A.; Jernerén, F.; Refsum, H.; Smith, A.D.; de Jager, C.A. Omega-3 Fatty Acid Status Enhances the Prevention of Cognitive Decline by B Vitamins in Mild Cognitive Impairment. J. Alzheimer’s Dis. 2016, 50, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Jernerén, F.; Cederholm, T.; Refsum, H.; Smith, A.D.; Turner, C.; Palmblad, J.; Eriksdotter, M.; Hjorth, E.; Faxen-Irving, G.; Wahlund, L.-O.; et al. Homocysteine Status Modifies the Treatment Effect of Omega-3 Fatty Acids on Cognition in a Randomized Clinical Trial in Mild to Moderate Alzheimer’s Disease: The OmegAD Study. J. Alzheimer’s Dis. 2019, 69, 189–197. [Google Scholar] [CrossRef]
- Millan, M.J.; Andrieux, A.; Bartzokis, G.; Cadenhead, K.; Dazzan, P.; Fusar-Poli, P.; Gallinat, J.; Giedd, J.; Grayson, D.R.; Heinrichs, M.; et al. Altering the Course of Schizophrenia: Progress and Perspectives. Nat. Rev. Drug Discov. 2016, 15, 485–515. [Google Scholar] [CrossRef] [PubMed]
- Glen, A.I.; Glen, E.M.; Horrobin, D.F.; Vaddadi, K.S.; Spellman, M.; Morse-Fisher, N.; Ellis, K.; Skinner, F.S. A Red Cell Membrane Abnormality in a Subgroup of Schizophrenic Patients: Evidence for Two Diseases. Schizophr. Res. 1994, 12, 53–61. [Google Scholar] [CrossRef]
- Tessier, C.; Sweers, K.; Frajerman, A.; Bergaoui, H.; Ferreri, F.; Delva, C.; Lapidus, N.; Lamaziere, A.; Roiser, J.P.; De Hert, M.; et al. Membrane Lipidomics in Schizophrenia Patients: A Correlational Study with Clinical and Cognitive Manifestations. Transl. Psychiatry 2016, 6, e906. [Google Scholar] [CrossRef] [PubMed]
- Amminger, G.P.; Mechelli, A.; Rice, S.; Kim, S.-W.; Klier, C.M.; McNamara, R.K.; Berk, M.; McGorry, P.D.; Schäfer, M.R. Predictors of Treatment Response in Young People at Ultra-High Risk for Psychosis Who Received Long-Chain Omega-3 Fatty Acids. Transl. Psychiatry 2015, 5, e495. [Google Scholar] [CrossRef] [PubMed]
- Sen, P.; Lamichhane, S.; Mathema, V.B.; McGlinchey, A.; Dickens, A.M.; Khoomrung, S.; Orešič, M. Deep Learning Meets Metabolomics: A Methodological Perspective. Brief Bioinform. 2021, 22, 1531–1542. [Google Scholar] [CrossRef] [PubMed]
- Stahl, S.M. Drugs for Psychosis and Mood: Unique Actions at D3, D2, and D1 Dopamine Receptor Subtypes. CNS Spectr. 2017, 22, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Moosavian, S.P.; Arab, A.; Mehrabani, S.; Moradi, S.; Nasirian, M. The Effect of Omega-3 and Vitamin E on Oxidative Stress and Inflammation: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Int. J. Vitam. Nutr. Res. 2020, 90, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Dyall, S.C. Interplay Between N-3 and n-6 Long-Chain Polyunsaturated Fatty Acids and the Endocannabinoid System in Brain Protection and Repair. Lipids 2017, 52, 885–900. [Google Scholar] [CrossRef]
- Goh, K.K.; Chen, C.Y.-A.; Chen, C.-H.; Lu, M.-L. Effects of Omega-3 Polyunsaturated Fatty Acids Supplements on Psychopathology and Metabolic Parameters in Schizophrenia: A Meta-Analysis of Randomized Controlled Trials. J. Psychopharmacol. 2021, 269881120981392. [Google Scholar] [CrossRef]
- Chen, A.T.; Chibnall, J.T.; Nasrallah, H.A. A Meta-Analysis of Placebo-Controlled Trials of Omega-3 Fatty Acid Augmentation in Schizophrenia: Possible Stage-Specific Effects. Ann. Clin. Psychiatry 2015, 27, 289–296. [Google Scholar]
- Sivrioglu, E.Y.; Kirli, S.; Sipahioglu, D.; Gursoy, B.; Sarandöl, E. The Impact of Omega-3 Fatty Acids, Vitamins E and C Supplementation on Treatment Outcome and Side Effects in Schizophrenia Patients Treated with Haloperidol: An Open-Label Pilot Study. Prog. Neuropsychopharmacol. Biol. Psychiatry 2007, 31, 1493–1499. [Google Scholar] [CrossRef]
- Jamilian, H.; Solhi, H.; Jamilian, M. Randomized, Placebo-Controlled Clinical Trial of Omega-3 as Supplemental Treatment in Schizophrenia. Glob. J. Health Sci. 2014, 6, 103–108. [Google Scholar] [CrossRef]
- Fenton, W.S.; Dickerson, F.; Boronow, J.; Hibbeln, J.R.; Knable, M. A Placebo-Controlled Trial of Omega-3 Fatty Acid (Ethyl Eicosapentaenoic Acid) Supplementation for Residual Symptoms and Cognitive Impairment in Schizophrenia. Am. J. Psychiatry 2001, 158, 2071–2074. [Google Scholar] [CrossRef]
- Bentsen, H.; Osnes, K.; Refsum, H.; Solberg, D.K.; Bøhmer, T. A Randomized Placebo-Controlled Trial of an Omega-3 Fatty Acid and Vitamins E+C in Schizophrenia. Transl. Psychiatry 2013, 3, e335. [Google Scholar] [CrossRef] [PubMed]
- Berger, G.E.; Proffitt, T.-M.; McConchie, M.; Yuen, H.; Wood, S.J.; Amminger, G.P.; Brewer, W.; McGorry, P.D. Ethyl-Eicosapentaenoic Acid in First-Episode Psychosis: A Randomized, Placebo-Controlled Trial. J. Clin. Psychiatry 2007, 68, 1867–1875. [Google Scholar] [CrossRef]
- Pawełczyk, T.; Grancow-Grabka, M.; Kotlicka-Antczak, M.; Trafalska, E.; Pawełczyk, A. A Randomized Controlled Study of the Efficacy of Six-Month Supplementation with Concentrated Fish Oil Rich in Omega-3 Polyunsaturated Fatty Acids in First Episode Schizophrenia. J. Psychiatr. Res. 2016, 73, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Amminger, G.P.; Schäfer, M.R.; Papageorgiou, K.; Klier, C.M.; Cotton, S.M.; Harrigan, S.M.; Mackinnon, A.; McGorry, P.D.; Berger, G.E. Long-Chain Omega-3 Fatty Acids for Indicated Prevention of Psychotic Disorders: A Randomized, Placebo-Controlled Trial. Arch. Gen. Psychiatry 2010, 67, 146–154. [Google Scholar] [CrossRef]
- Amminger, G.P.; Schäfer, M.R.; Schlögelhofer, M.; Klier, C.M.; McGorry, P.D. Longer-Term Outcome in the Prevention of Psychotic Disorders by the Vienna Omega-3 Study. Nat. Commun. 2015, 6, 7934. [Google Scholar] [CrossRef]
- McGorry, P.D.; Nelson, B.; Markulev, C.; Yuen, H.P.; Schäfer, M.R.; Mossaheb, N.; Schlögelhofer, M.; Smesny, S.; Hickie, I.B.; Berger, G.E.; et al. Effect of ω-3 Polyunsaturated Fatty Acids in Young People at Ultrahigh Risk for Psychotic Disorders: The NEURAPRO Randomized Clinical Trial. JAMA Psychiatry 2017, 74, 19–27. [Google Scholar] [CrossRef]
- Messamore, E.; McNamara, R.K. Detection and Treatment of Omega-3 Fatty Acid Deficiency in Psychiatric Practice: Rationale and Implementation. Lipids Health Dis. 2016, 15, 25. [Google Scholar] [CrossRef] [PubMed]
- Schlögelhofer, M.; McGorry, P.D.; Nelson, B.; Markulev, C.; Yuen, H.P.; Schäfer, M.; Mossaheb, N.; Smesny, S.; Hickie, I.B.; Berger, G.; et al. T49. The Neurapro Study: Adherence To Study Medication. Schizophr. Bull. 2018, 44, S132–S133. [Google Scholar] [CrossRef]
- Deckelbaum, R.J.; Calder, P.C. Editorial: Is It Time to Separate EPA from DHA When Using Omega-3 Fatty Acids to Protect Heart and Brain? Curr. Opin. Clin. Nutr. Metab. Care 2020, 23, 65–67. [Google Scholar] [CrossRef] [PubMed]
- Bentsen, H.; Solberg, D.K.; Refsum, H.; Gran, J.M.; Bøhmer, T.; Torjesen, P.A.; Halvorsen, O.; Lingjærde, O. Bimodal Distribution of Polyunsaturated Fatty Acids in Schizophrenia Suggests Two Endophenotypes of the Disorder. Biol. Psychiatry 2011, 70, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Yang, X.; Jiang, J.; Hu, X.; Qing, Y.; Wang, D.; Yang, T.; Yang, C.; Zhang, J.; Yang, P.; et al. Identification of the Niacin-Blunted Subgroup of Schizophrenia Patients from Mood Disorders and Healthy Individuals in Chinese Population. Schizophr. Bull. 2017, 44, 896–907. [Google Scholar] [CrossRef] [PubMed]
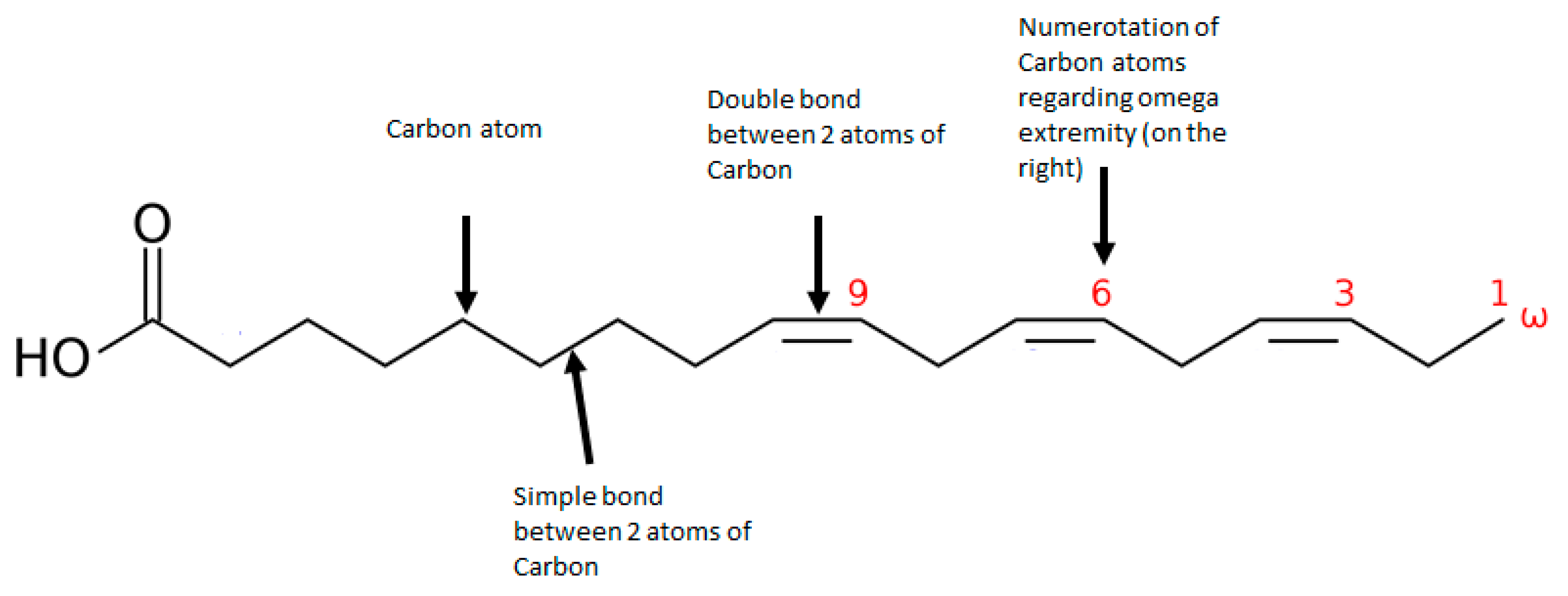
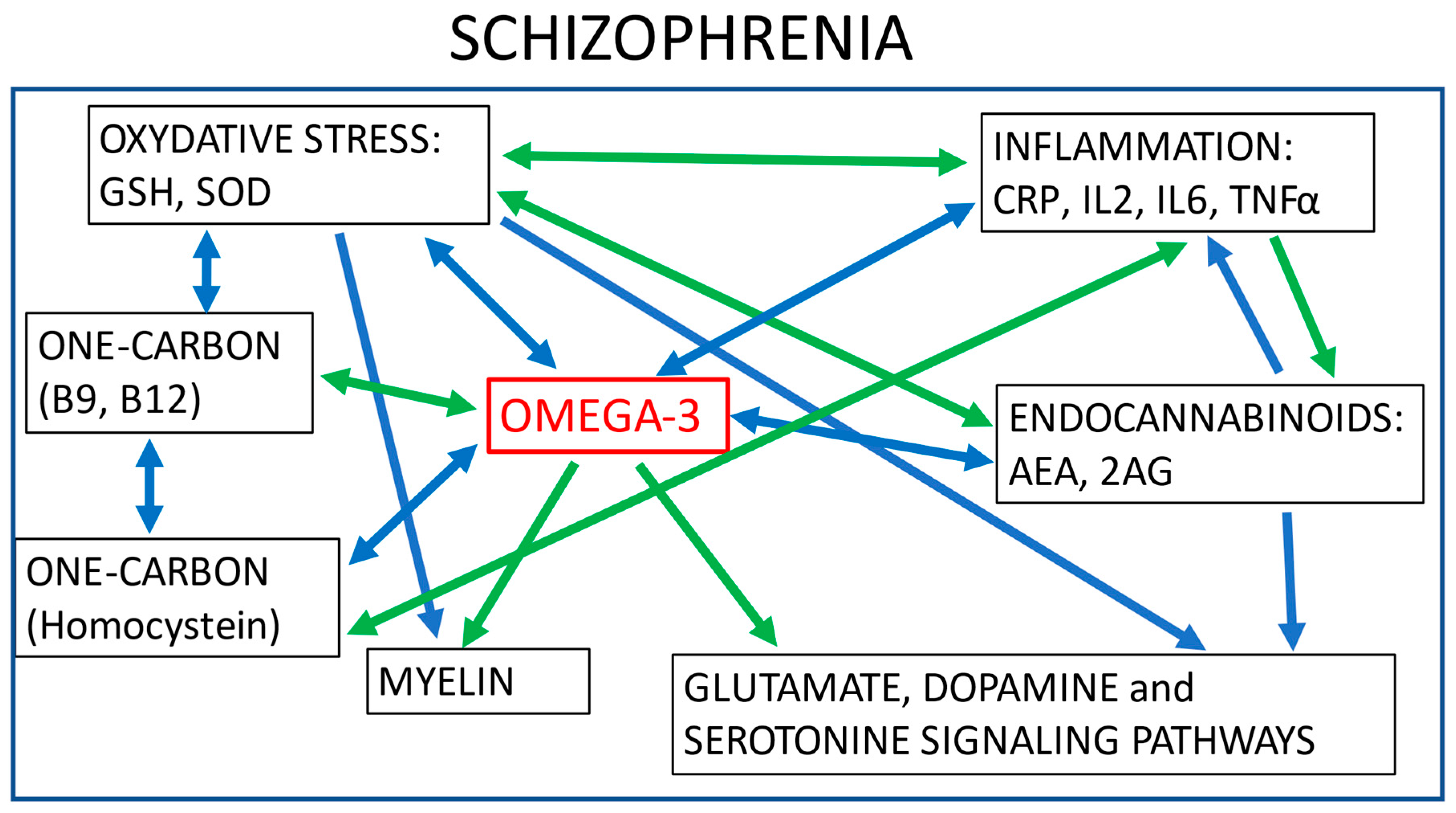
| Schizophrenia Patients with No Treatment | Effect of Antipsychotics | Effect of Omega-3 Supplementation | |
|---|---|---|---|
| Biological systems | |||
| Dopamine and glutamate neurotransmission pathways | Altered [4] | Rebalanced [96,176] | Rebalanced [88] |
| Myelination | ↓ [116] | ↑ [125] | ↑ [129] |
| Inflammation | ↑ [134] | ↓ [139] | ↓ [12] |
| Oxidative stress | ↑ [135] | ↓ [140] | ↓ [177] |
| Molecules | |||
| Homocysteine | ↑ [163] | ↓ [164] | ↓ [168] |
| B9, B12 | ↓ [163] | - | Positive correlation [162] |
| Phospholipase A2 | ↑ [47] | ↓ [48] | ↓ [49] |
| Endocannabinoids (anandamide and 2AG) | ↑ [62] | - | ↓ [178] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frajerman, A.; Scoriels, L.; Kebir, O.; Chaumette, B. Shared Biological Pathways between Antipsychotics and Omega-3 Fatty Acids: A Key Feature for Schizophrenia Preventive Treatment? Int. J. Mol. Sci. 2021, 22, 6881. https://doi.org/10.3390/ijms22136881
Frajerman A, Scoriels L, Kebir O, Chaumette B. Shared Biological Pathways between Antipsychotics and Omega-3 Fatty Acids: A Key Feature for Schizophrenia Preventive Treatment? International Journal of Molecular Sciences. 2021; 22(13):6881. https://doi.org/10.3390/ijms22136881
Chicago/Turabian StyleFrajerman, Ariel, Linda Scoriels, Oussama Kebir, and Boris Chaumette. 2021. "Shared Biological Pathways between Antipsychotics and Omega-3 Fatty Acids: A Key Feature for Schizophrenia Preventive Treatment?" International Journal of Molecular Sciences 22, no. 13: 6881. https://doi.org/10.3390/ijms22136881
APA StyleFrajerman, A., Scoriels, L., Kebir, O., & Chaumette, B. (2021). Shared Biological Pathways between Antipsychotics and Omega-3 Fatty Acids: A Key Feature for Schizophrenia Preventive Treatment? International Journal of Molecular Sciences, 22(13), 6881. https://doi.org/10.3390/ijms22136881






