Perinatal Gram-Positive Bacteria Exposure Elicits Distinct Cytokine Responses In Vitro
Abstract
1. Introduction
2. Results
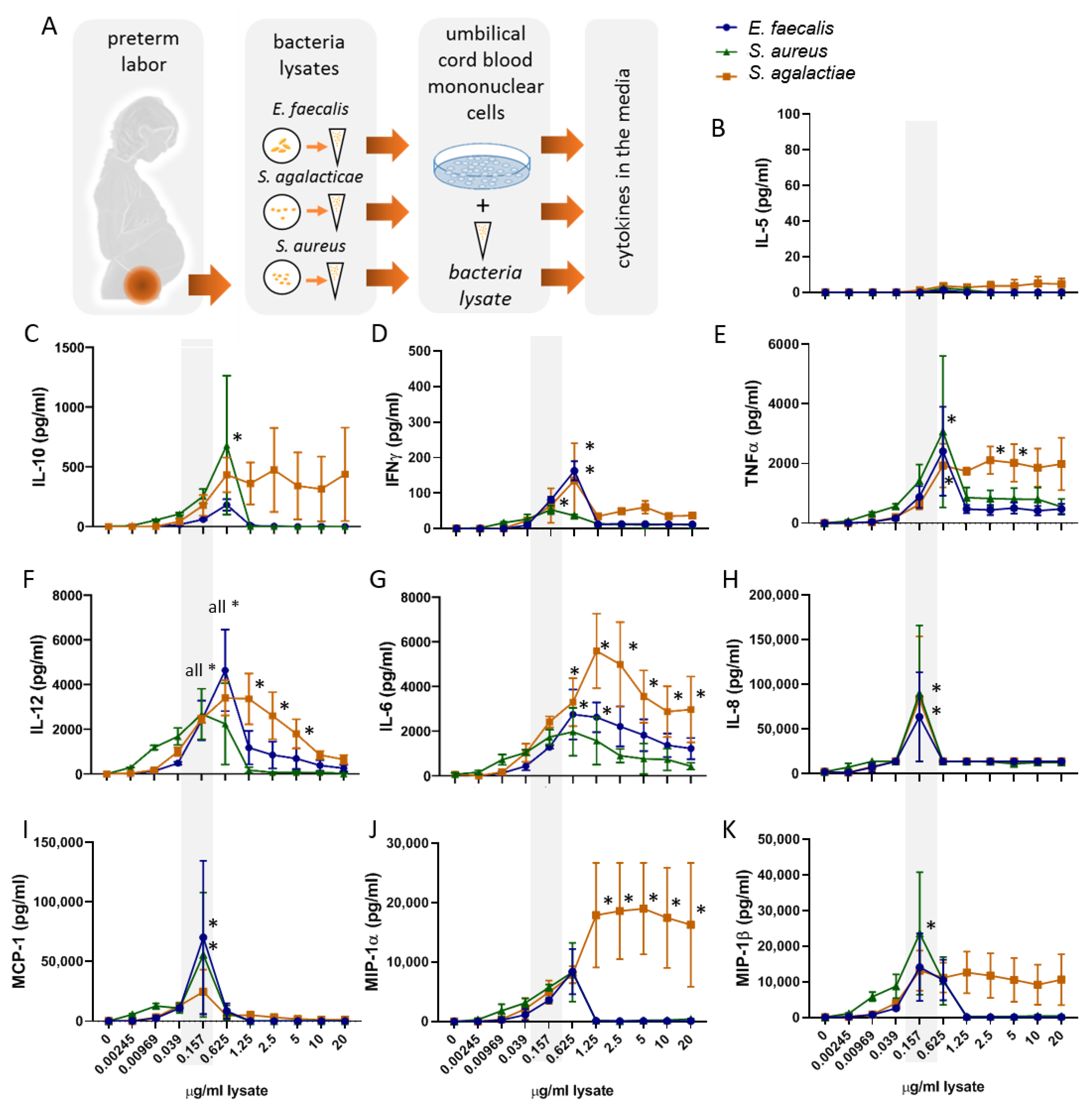
2.1. Bacterial Lysates Trigger the Secretion of Cytokines and Chemokines by Umbilical Cord Blood Mononuclear Cells
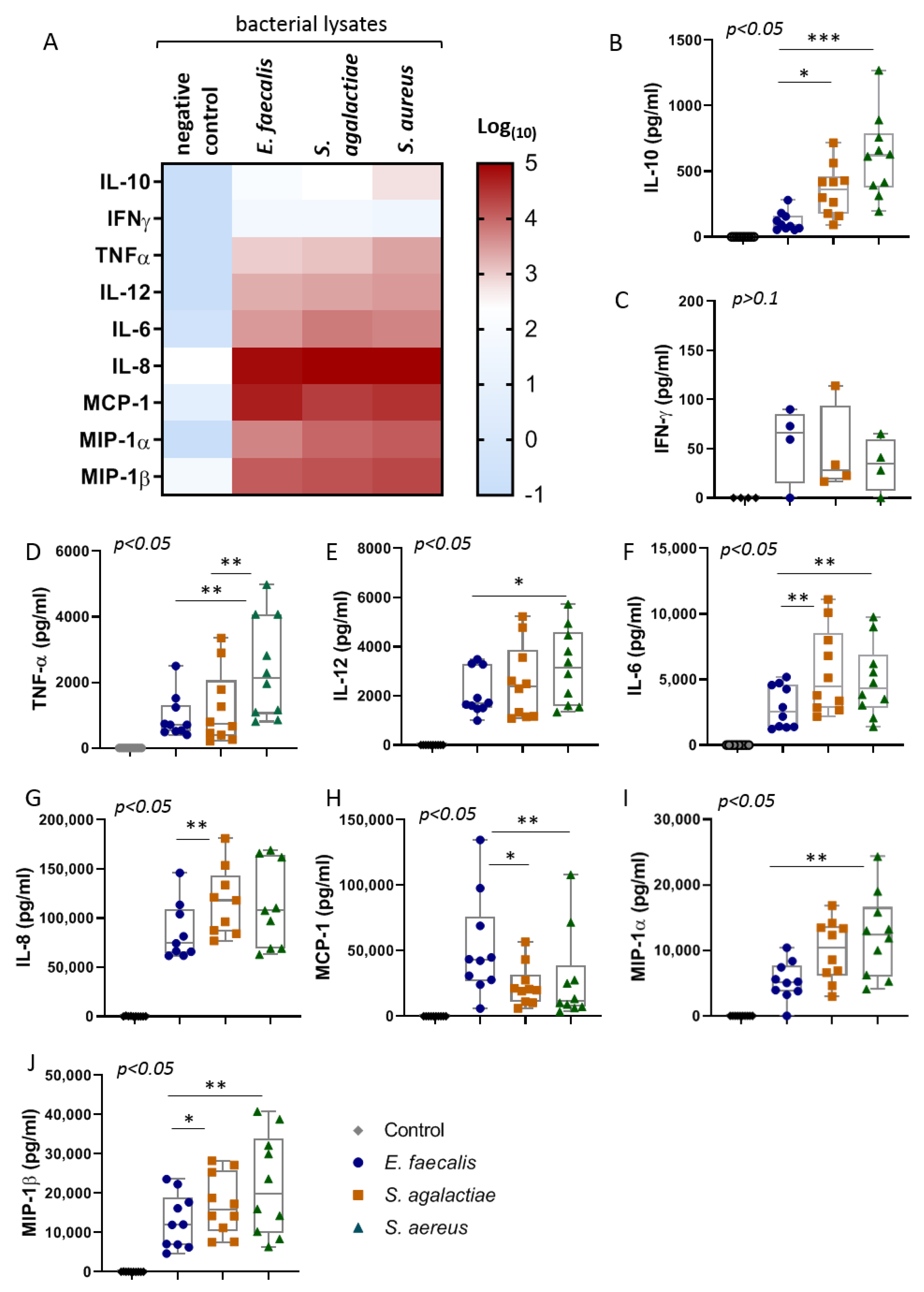
2.2. The Bacterial Lysates Elicit Distinct Patterns of Cytokines Responses
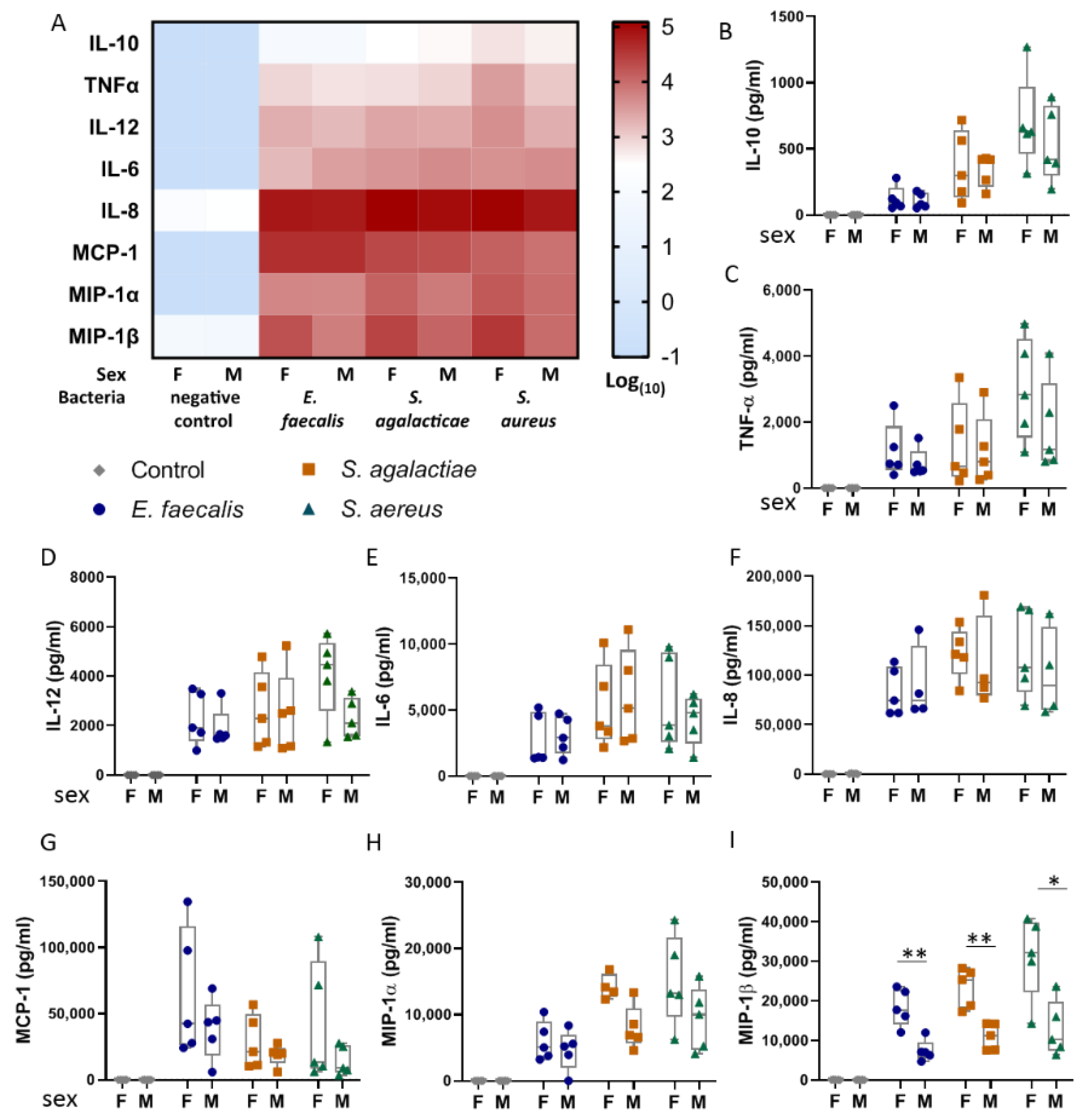
2.3. Umbilical Blood Mononuclear Cells from Female Infants Respond More Pronouncedly to Bacterial Lysates than Those from Male Infants
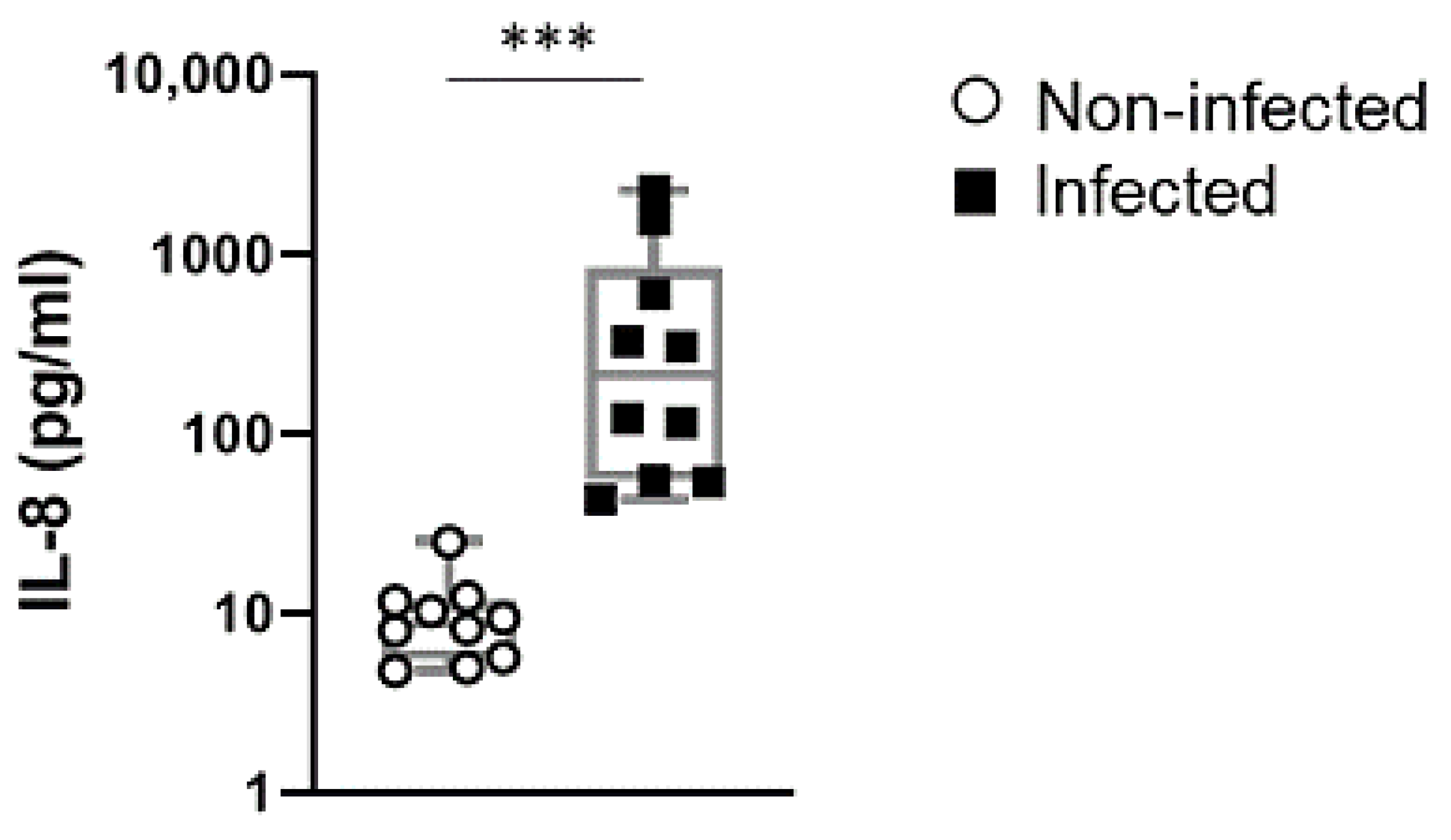
2.4. IL-8 Levels in Cord Blood of Preterm Newborn Infants is a Stable Indicator of Perinatal Infections
3. Discussion
4. Materials and Methods
4.1. Umbilical Cord Blood Source
4.2. Isolation of Umbilical Cord Blood Mononuclear Cells (UBMC)
4.3. Preparation of Bacterial Lysates
4.4. Stimulation of UBMC with Gram-Positive Bacterial Lysates
4.5. Determination of Cytokine and Chemokine Concentrations in UBMC Supernatants
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goldenberg, R.L. The management of preterm labor. Obstet. Gynecol. 2002, 100, 1020–1037. [Google Scholar] [PubMed]
- Chawanpaiboon, S.; Vogel, J.P.; Moller, A.-B.; Lumbiganon, P.; Petzold, M.; Hogan, D.; Landoulsi, S.; Jampathong, N.; Kongwattanakul, K.; Laopaiboon, M.; et al. Global, regional, and national estimates of levels of preterm birth in 2014: A systematic review and modelling analysis. Lancet Glob. Health 2019, 7, e37–e46. [Google Scholar] [CrossRef]
- Martin, J.A.; Hamilton, B.E.; Osterman, M.J.K.; Driscoll, A.K.; Drake, P. Births: Final Data for 2016. Natl. Vital. Stat. Rep. 2018, 67, 1–55. [Google Scholar] [PubMed]
- Romero, R.; Espinoza, J.; Gonçalves, L.F.; Kusanovic, J.P.; Friel, L.; Hassan, S. The Role of Inflammation and Infection in Preterm Birth. Semin. Reprod. Med. 2007, 25, 21–39. [Google Scholar] [CrossRef] [PubMed]
- Romero, R.; Dey, S.K.; Fisher, S.J. Preterm labor: One syndrome, many causes. Science 2014, 345, 760–765. [Google Scholar] [CrossRef]
- Frascoli, M.; Coniglio, L.; Witt, R.; Jeanty, C.; Fleck-Derderian, S.; Myers, D.E.; Lee, T.-H.; Keating, S.M.; Busch, M.P.; Norris, P.J.; et al. Alloreactive fetal T cells promote uterine contractility in preterm labor via IFN-γ and TNF-α. Sci. Transl. Med. 2018, 10, eaan2263. [Google Scholar] [CrossRef]
- Montalbano, A.P.; Hawgood, S.; Mendelson, C.R. Mice Deficient in Surfactant Protein A (SP-A) and SP-D or in TLR2 Manifest Delayed Parturition and Decreased Expression of Inflammatory and Contractile Genes. Endocrinology 2013, 154, 483–498. [Google Scholar] [CrossRef]
- Muhammad, Z.; Ahmed, A.; Hayat, U.; Wazir, M.S.; Rafiyatullah; Waqas, H. Neonatal sepsis: Causative bacteria and their resistance to antibiotics. J. Ayub. Med. Coll. Abbottabad JAMC 2012, 22, 33–36. [Google Scholar]
- Weinstein, R.A.; Gaynes, R.; Edwards, J.R.; System, N.N.I.S. Overview of Nosocomial Infections Caused by Gram-Negative Bacilli. Clin. Infect. Dis. 2005, 41, 848–854. [Google Scholar] [CrossRef]
- Chen, C.; Song, X.; Chunwei, Z.; Zhong, H.; Dai, J.; Lan, Z.; Li, F.; Yu, X.; Feng, Q.; Wang, Z.; et al. The microbiota continuum along the female reproductive tract and its relation to uterine-related diseases. Nat. Commun. 2017, 8, 1–11. [Google Scholar] [CrossRef]
- De Gier, B.; Van Kassel, M.N.; Sanders, E.A.M.; Van De Beek, D.; Hahné, S.J.M.; Van Der Ende, A.; Bijlsma, M.W. Disease burden of neonatal invasive Group B Streptococcus infection in the Netherlands. PLoS ONE 2019, 14, e0216749. [Google Scholar] [CrossRef] [PubMed]
- Christie, C.; Hammond, J.; Reising, S.; Evans-Patterson, J. Clinical and molecular epidemiology of enterococcal bacteremia in a pediatric teaching hospital. J. Pediatr. 1994, 125, 392–399. [Google Scholar] [CrossRef]
- Kaufman, D.A.; Fairchild, K.D. Clinical Microbiology of Bacterial and Fungal Sepsis in Very-Low-Birth-Weight Infants. Clin. Microbiol. Rev. 2004, 17, 638–680. [Google Scholar] [CrossRef] [PubMed]
- Bianconi, S.; Gudavalli, M.; Sutija, V.G.; Lopez, A.L.; Barillas-Arias, L.; Ron, N. Ranitidine and late-onset sepsis in the neonatal intensive care unit. J. Perinat. Med. 2007, 35, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Stoll, B.J.; Hansen, N.I.; Sánchez, P.J.; Faix, R.G.; Poindexter, B.B.; Van Meurs, K.P.; Bizzarro, M.J.; Goldberg, R.N.; Frantz, I.D.; Hale, E.C.; et al. Early Onset Neonatal Sepsis: The Burden of Group B Streptococcal and E. coli Disease Continues. Pediatry 2011, 127, 817–826. [Google Scholar] [CrossRef] [PubMed]
- Maisey, H.C.; Doran, K.S.; Nizet, V. Recent advances in understanding the molecular basis of group BStreptococcusvirulence. Expert Rev. Mol. Med. 2008, 10, e27. [Google Scholar] [CrossRef]
- Doster, R.S.; Kirk, L.A.; Tetz, L.M.; Rogers, L.M.; Aronoff, D.M.; Gaddy, J.A. Staphylococcus aureus Infection of Human Gestational Membranes Induces Bacterial Biofilm Formation and Host Production of Cytokines. J. Infect. Dis. 2017, 215, 653–657. [Google Scholar] [CrossRef]
- Jimenez-Truque, N.; Tedeschi, S.; Saye, E.J.; McKenna, B.D.; Langdon, W.; Wright, J.P.; Alsentzer, A.; Arnold, S.; Saville, B.R.; Wang, W.; et al. Relationship Between Maternal and Neonatal Staphylococcus aureus Colonization. Pediatry 2012, 129, e1252–e1259. [Google Scholar] [CrossRef]
- Geng, W.; Qi, Y.; Li, W.; McConville, T.H.; Hill-Ricciuti, A.; Grohs, E.C.; Saiman, L.; Uhlemann, A.-C. Epidemiology of Staphylococcus aureus in neonates on admission to a Chinese neonatal intensive care unit. PLoS ONE 2020, 15, e0211845. [Google Scholar] [CrossRef]
- Romero, R.; Chaiworapongsa, T.; Espinoza, J. Micronutrients and Intrauterine Infection, Preterm Birth and the Fetal Inflammatory Response Syndrome. J. Nutr. 2003, 133, 1668S–1673S. [Google Scholar] [CrossRef]
- Agrawal, V.; Hirsch, E. Intrauterine infection and preterm labor. Semin. Fetal Neonatal Med. 2012, 17, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Shane, A.L.; Sánchez, P.J.; Stoll, B.J. Neonatal sepsis. Lancet 2017, 390, 1770–1780. [Google Scholar] [CrossRef]
- Stefanovic, I.M. Neonatal sepsis. Biochem. Med. 2011, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Dey, P.; Fabri-Faja, N.; Calvo-Lozano, O.; Terborg, R.A.; Belushkin, A.; Yesilkoy, F.; Fàbrega, A.; Ruiz-Rodriguez, J.C.; Ferrer, R.; González-López, J.J.; et al. Label-free Bacteria Quantification in Blood Plasma by a Bioprinted Microarray Based Interferometric Point-of-Care Device. ACS Sens. 2019, 4, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Popovich, J.; Zhang, W.; Ganser, C.; Haydel, S.E.; Seo, D.-K. Superior ion release properties and antibacterial efficacy of nanostructured zeolites ion-exchanged with zinc, copper, and iron. RSC Adv. 2018, 8, 37949–37957. [Google Scholar] [CrossRef]
- Reuschel, E.; Toelge, M.; Entleutner, K.; Deml, L.; Seelbach-Goebel, B. Cytokine profiles of umbilical cord blood mononuclear cells upon in vitro stimulation with lipopolysaccharides of different vaginal gram-negative bacteria. PLoS ONE 2019, 14, e0222465. [Google Scholar] [CrossRef]
- Kim, C.J.; Romero, R.; Chaemsaithong, P.; Chaiyasit, N.; Yoon, B.H.; Kim, Y.M. Acute chorioamnionitis and funisitis: Definition, pathologic features, and clinical significance. Am. J. Obstet. Gynecol. 2015, 213, S29–S52. [Google Scholar] [CrossRef]
- Zeitlin, J.; Saurel-Cubizolles, M.-J.; De Mouzon, J.; Rivera, L.; Ancel, P.; Blondel, B.; Kaminski, M. Fetal sex and preterm birth: Are males at greater risk? Hum. Reprod. 2002, 17, 2762–2768. [Google Scholar] [CrossRef]
- Cortese, F.; Scicchitano, P.; Gesualdo, M.; Filaninno, A.; De Giorgi, E.; Schettini, F.; Laforgia, N.; Ciccone, M.M. Early and Late Infections in Newborns: Where Do We Stand? A Review. Pediatr. Neonatol. 2016, 57, 265–273. [Google Scholar] [CrossRef]
- Scotland, R.S.; Stables, M.J.; Madalli, S.; Watson, P.; Gilroy, D.W. Sex differences in resident immune cell phenotype underlie more efficient acute inflammatory responses in female mice. Blood 2011, 118, 5918–5927. [Google Scholar] [CrossRef]
- Le Rouzic, V.; Corona, J.; Zhou, H. Postnatal Development of Hepatic Innate Immune Response. Inflammation 2010, 34, 576–584. [Google Scholar] [CrossRef][Green Version]
- Madalli, S.; Beyrau, M.; Whiteford, J.R.; Duchêne, J.; Nandhra, I.S.A.; Patel, N.S.; Motwani, M.P.; Gilroy, D.W.; Thiemermann, C.; Nourshargh, S.; et al. Sex-specific regulation of chemokine Cxcl5/6 controls neutrophil recruitment and tissue injury in acute inflammatory states. Biol. Sex Differ. 2015, 6, 27. [Google Scholar] [CrossRef] [PubMed]
- Marriott, I.; Huet-Hudson, Y.M. Sexual Dimorphism in Innate Immune Responses to Infectious Organisms. Immunol. Res. 2006, 34, 177–192. [Google Scholar] [CrossRef]
- Speirs, I.C.; Tronson, N.C. Sex differences in hippocampal cytokines after systemic immune challenge. Neuroscience 2018. [Google Scholar] [CrossRef]
- Erickson, M.E.; Liang, W.S.; Fernandez, E.G.; Bullock, K.M.; Thysell, J.A.; Banks, W.A. Genetics and sex influence peripheral and central innate immune responses and blood-brain barrier integrity. PLoS ONE 2018, 13, e0205769. [Google Scholar] [CrossRef] [PubMed]
- Bystry, R.S.; Aluvihare, V.R.; Welch, K.A.; Kallikourdis, M.; Betz, A.G. B cells and professional APCs recruit regulatory T cells via CCL4. Nat. Immunol. 2001, 2, 1126–1132. [Google Scholar] [CrossRef] [PubMed]
- O’Driscoll, D.N.; McGovern, M.; Greene, C.M.; Molloy, E.J. Gender disparities in preterm neonatal outcomes. Acta Paediatr. 2018, 107, 1494–1499. [Google Scholar] [CrossRef]
- Zhou, M.; Cheng, S.; Yu, J.; Lu, Q. Interleukin-8 for Diagnosis of Neonatal Sepsis: A Meta-Analysis. PLoS ONE 2015, 10, e0127170. [Google Scholar] [CrossRef]
- Boskabadi, H.; Zakerihamidi, M. Evaluate the diagnosis of neonatal sepsis by measuring interleukins: A systematic review. Pediatr. Neonatol. 2018, 59, 329–338. [Google Scholar] [CrossRef]
- Sacks, G.P.; Redman, C.W.G.; Sargent, I.L. Monocytes are primed to produce the Th1 type cytokine IL-12 in normal human pregnancy: An intracellular flow cytometric analysis of peripheral blood mononuclear cells. Clin. Exp. Immunol. 2003, 131, 490–497. [Google Scholar] [CrossRef]
- Dudley, N.J.; Edwin, S.S.; Van Wagoner, J.; Augustine, N.H.; Hill, H.R.; Mitchell, M.D.; Van Wagoner, J. Regulation of decidual cell chemokine production by group B streptococci and purified bacterial cell wall components. Am. J. Obstet. Gynecol. 1997, 177, 666–672. [Google Scholar] [CrossRef]
- Gibbons, D.; Fleming, P.; Virasami, A.; Michel, M.-L.; Sebire, N.J.; Costeloe, K.; Carr, R.; Klein, N.; Hayday, A. Interleukin-8 (CXCL8) production is a signatory T cell effector function of human newborn infants. Nat. Med. 2014, 20, 1206–1210. [Google Scholar] [CrossRef] [PubMed]
- Hansen-Pupp, I.; Harling, S.; Berg, A.-C.; Cilio, C.; Hellström-Westas, L.; Ley, D.; Hellstr, L. Circulating Interferon-gamma and White Matter Brain Damage in Preterm Infants. Pediatr. Res. 2005, 58, 946–952. [Google Scholar] [CrossRef]
- Mancuso, G.; Cusumano, V.; Genovese, F.; Gambuzza, M.; Beninati, C.; Teti, G. Role of interleukin 12 in experimental neonatal sepsis caused by group B streptococci. Infect. Immun. 1997, 65, 3731–3735. [Google Scholar] [CrossRef]
- Fournier, B.; Philpott, D.J. Recognition of Staphylococcus aureus by the Innate Immune System. Clin. Microbiol. Rev. 2005, 18, 521–540. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, P.; Basu, S. Enterococcus fecalis Sepsis and Leukemoid Reaction. J. Pediatr. Hematol. 2015, 37, e419–e420. [Google Scholar] [CrossRef]
- Araújo, T.F.; Ferreira, C.L.D.L.F. The genus Enterococcus as probiotic: Safety concerns. Braz. Arch. Biol. Technol. 2013, 56, 457–466. [Google Scholar] [CrossRef]
- Bhagwat, A.; Annapure, U.S. Maternal-neonatal transmission of Enterococcus strains during delivery. Beni-Suef Univ. J. Basic Appl. Sci. 2019, 8, 1–9. [Google Scholar] [CrossRef]
- Shigematsu, K.; Asai, A.; Kobayashi, M.; Herndon, D.N.; Suzuki, F. Enterococcus faecalis translocation in mice with severe burn injury: A pathogenic role of CCL2 and alternatively activated macrophages (M2aMϕ and M2cMϕ). J. Leukoc. Biol. 2009, 86, 999–1005. [Google Scholar] [CrossRef]
- Xu, Z.; Tong, Z.; Neelakantan, P.; Cai, Y.; Wei, X. Enterococcus faecalis immunoregulates osteoclastogenesis of macrophages. Exp. Cell Res. 2018, 362, 152–158. [Google Scholar] [CrossRef]
- Wolpe, S.D.; Davatelis, G.; Sherry, B.; Beutler, B.; Hesse, D.G.; Nguyen, H.T.; Moldawer, L.L.; Nathan, C.F.; Lowry, S.F.; Cerami, A. Macrophages secrete a novel heparin-binding protein with inflammatory and neutrophil chemokinetic properties. J. Exp. Med. 1988, 167, 570–581. [Google Scholar] [CrossRef] [PubMed]
- Hieshima, K.; Imai, T.; Baba, M.; Shoudai, K.; Ishizuka, K.; Nakagawa, T.; Tsuruta, J.; Takeya, M.; Sakaki, Y.; Takatsuki, K.; et al. A novel human CC chemokine PARC that is most homologous to macrophage-inflammatory protein-1 alpha/LD78 alpha and chemotactic for T lymphocytes, but not for monocytes. J. Immunol. 1997, 159, 1140–1149. [Google Scholar] [PubMed]
- Peltier, M.R. Immunology of term and preterm labor. Reprod. Biol. Endocrinol. 2003, 1, 122. [Google Scholar] [CrossRef]
- Cusumano, V.; Genovese, F.; Mancuso, G.; Carbone, M.; Fera, M.T.; Teti, G. Interleukin-10 protects neonatal mice from lethal group B streptococcal infection. Infect. Immun. 1996, 64, 2850–2852. [Google Scholar] [CrossRef]
- Ouyang, W.; Rutz, S.; Crellin, N.K.; Valdez, P.; Hymowitz, S.G. Regulation and Functions of the IL-10 Family of Cytokines in Inflammation and Disease. Annu. Rev. Immunol. 2011, 29, 71–109. [Google Scholar] [CrossRef]
- Flynn, J.L.; Goldstein, M.M.; Chan, J.; Triebold, K.J.; Pfeffer, K.; Lowenstein, C.J.; Schrelber, R.; Mak, T.W.; Bloom, B.R. Tumor necrosis factor-α is required in the protective immune response against mycobacterium tuberculosis in mice. Immunity 1995, 2, 561–572. [Google Scholar] [CrossRef]
- Echtenacher, B. Treatment of experimental sepsis-induced immunoparalysis with TNF. Immunobiology 2003, 208, 381–389. [Google Scholar] [CrossRef]
- Mohammad, M.; Nguyen, M.; Engdahl, C.; Na, M.; Jarneborn, A.; Hu, Z.; Karlsson-Bengtsson, A.; Pullerits, R.; Ali, A.; Götz, F.; et al. The YIN and YANG of lipoproteins in developing and preventing infectious arthritis by Staphylococcus aureus. PLOS Pathog. 2019, 15, e1007877. [Google Scholar] [CrossRef]
- Ali, A.; Zhu, X.; Kwiecinski, J.; Gjertsson, I.; Lindholm, C.; Iwakura, Y.; Wang, X.; Lycke, N.; Josefsson, E.; Pullerits, R.; et al. Antibiotic-KilledStaphylococcus aureusInduces Destructive Arthritis in Mice. Arthritis Rheumatol. 2014, 67, 107–116. [Google Scholar] [CrossRef]
- Herbert, A.M.; Beveridge, C.J.; Saunders, N.J. Bacterial virulence factors in neonatal sepsis: Group B streptococcus. Curr. Opin. Infect. Dis. 2004, 17, 225–229. [Google Scholar] [CrossRef]
- Bray, B.A.; Sutcliffe, I.C.; Harrington, D.J. Impact of lgt mutation on lipoprotein biosynthesis and in vitro phenotypes of Streptococcus agalactiae. Microbiology 2009, 155, 1451–1458. [Google Scholar] [CrossRef] [PubMed]
- Ernst, W.; Kusi, E.; Malfertheiner, S.F.; Reuschel, E.; Deml, L.; Seelbach-Göbel, B. The effect of Indomethacin and Betamethasone on the cytokine response of human neonatal mononuclear cells to gram-positive bacteria. Cytokine 2015, 73, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, A.R.; Dorrington, M.G.; Dutta, B.; Krauss, K.S.; Martins, A.J.; Uderhardt, S.; Chan, W.; Tsang, J.S.; Torabi-Parizi, P.; Fraser, I.D.; et al. IFN-mediated negative feedback supports bacteria class-specific macrophage inflammatory responses. eLife 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Helwig, U.; Lammers, K.M.; Rizzello, F.; Brigidi, P.; Rohleder, V.; Caramelli, E.; Gionchetti, P.; Schrezenmeir, J.; Foelsch, U.R.; Schreiber, S.; et al. Lactobacilli, bifidobacteriaandE. colinissle induce pro- and anti-inflammatory cytokines in peripheral blood mononuclear cells. World J. Gastroenterol. 2006, 12, 5978–5986. [Google Scholar] [CrossRef] [PubMed]
- Memedovski, Z.; Czerwonka, E.; Han, J.; Mayer, J.; Luce, M.; Klemm, L.C.; Hall, M.L.; Mayer, A.M. Classical and Alternative Activation of Rat Microglia Treated with Ultrapure Porphyromonas gingivalis Lipopolysaccharide In Vitro. Toxins 2020, 12, 333. [Google Scholar] [CrossRef]
- Mayer, A.M.; Murphy, J.; Macadam, D.; Osterbauer, C.; Baseer, I.; Hall, M.L.; Feher, D.; Williams, P. Classical and Alternative Activation of CyanobacteriumOscillatoriasp. Lipopolysaccharide-Treated Rat Microgliain vitro. Toxicol. Sci. 2015, 149, 484–495. [Google Scholar] [CrossRef]
- Meyer, F.; Wilson, K.T.; James, S.P. Modulation of Innate Cytokine Responses by Products of Helicobacter pylori. Infect. Immun. 2000, 68, 6265–6272. [Google Scholar] [CrossRef]
- Koch, L.; Frommhold, D.; Buschmann, K.; Kuss, N.; Poeschl, J.; Ruef, P. LPS- and LTA-Induced Expression of IL-6 and TNF-αin Neonatal and Adult Blood: Role of MAPKs and NF-κB. Mediat. Inflamm. 2014, 2014, 1–8. [Google Scholar] [CrossRef]
- Kranzer, K.; Eckhardt, A.; Aigner, M.; Knoll, G.; Deml, L.; Speth, C.; Lehn, N.; Rehli, M.; Schneider-Brachert, W. Induction of Maturation and Cytokine Release of Human Dendritic Cells by Helicobacter pylori. Infect. Immun. 2004, 72, 4552–4560. [Google Scholar] [CrossRef]
- Deml, L.; Aigner, M.; Decker, J.; Eckhardt, A.; Schütz, C.; Mittl, P.R.E.; Barabas, S.; Denk, S.; Knoll, G.; Lehn, N.; et al. Characterization of the Helicobacter pylori Cysteine-Rich Protein A as a T-Helper Cell Type 1 Polarizing Agent. Infect. Immun. 2005, 73, 4732–4742. [Google Scholar] [CrossRef]
- Simpson, S.; Kaislasuo, J.; Guller, S.; Pal, L. Thermal stability of cytokines: A review. Cytokine 2020, 125, 154829. [Google Scholar] [CrossRef] [PubMed]
| Subject Number | Maternal Age (Years) | Parity | Gestational Age (Weeks + Days) | Maternal BMI Before Pregnancy | Neonatal Birth Weight (g) | Neonatalsex |
|---|---|---|---|---|---|---|
| 1 | 31 | 6 | 40 + 3 | 25.9 | 3810 | Male |
| 2 | 29 | 2 | 41 + 0 | 24.1 | 3540 | Female |
| 3 | 33 | 1 | 39 + 4 | 31.9 | 4150 | Female |
| 4 | 29 | 2 | 40 + 6 | 30.8 | 3740 | Male |
| 5 | 27 | 1 | 40 + 1 | 22.1 | 3150 | Female |
| 6 | 34 | 1 | 40 + 5 | 35.0 | 3300 | Male |
| 7 | 25 | 2 | 38 + 5 | 32.4 | 3800 | Male |
| 8 | 27 | 1 | 39 + 4 | 22.5 | 3620 | Male |
| 9 | 30 | 2 | 37 + 1 | 21.5 | 2830 | Female |
| 10 | 27 | 1 | 37 + 0 | 22.9 | 2570 | Female |
| Median | 29 | 1.5 | 39 + 3 | 25.0 | 3580 | |
| Range | 25–31 | 1–6 | 37–41 | 21.5–35.0 | 2570–4150 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reuschel, E.; Toelge, M.; Haeusler, S.; Deml, L.; Seelbach-Goebel, B.; Solano, M.E. Perinatal Gram-Positive Bacteria Exposure Elicits Distinct Cytokine Responses In Vitro. Int. J. Mol. Sci. 2021, 22, 332. https://doi.org/10.3390/ijms22010332
Reuschel E, Toelge M, Haeusler S, Deml L, Seelbach-Goebel B, Solano ME. Perinatal Gram-Positive Bacteria Exposure Elicits Distinct Cytokine Responses In Vitro. International Journal of Molecular Sciences. 2021; 22(1):332. https://doi.org/10.3390/ijms22010332
Chicago/Turabian StyleReuschel, Edith, Martina Toelge, Sebastian Haeusler, Ludwig Deml, Birgit Seelbach-Goebel, and Maria Emilia Solano. 2021. "Perinatal Gram-Positive Bacteria Exposure Elicits Distinct Cytokine Responses In Vitro" International Journal of Molecular Sciences 22, no. 1: 332. https://doi.org/10.3390/ijms22010332
APA StyleReuschel, E., Toelge, M., Haeusler, S., Deml, L., Seelbach-Goebel, B., & Solano, M. E. (2021). Perinatal Gram-Positive Bacteria Exposure Elicits Distinct Cytokine Responses In Vitro. International Journal of Molecular Sciences, 22(1), 332. https://doi.org/10.3390/ijms22010332





